
Trends
Sci.
2026; 23(5): 12145
Physicochemical and Pharmacokinetic Fatty Acid Profiles from 3 Beans Drive Superior Lamellar Liquid Crystal Hair Treatment Cream Functionality with Lipomulse Luxe MB Emulsifier
Warongporn Rattanabun1, Wongnapa Nakyai2,
Natnicha Phungsara3 and Wannisa Keawbankrud1,*
1Health Science and Aesthetic Program, Department of Science, Faculty of Science and Technology,
Rajamangala University of Technology Krungthep, Bangkok 10120, Thailand
2Faculty of Integrative Medicine, Rajamangala University of Technology Thanyaburi, Pathum Thani 12130, Thailand
3Mahidol Bumrungrak Nakhonsawan Medical Center, Mahidol University, Nakhonsawan Campus,
Nakhon Sawan 60130, Thailand
(*Corresponding author’s e-mail: [email protected])
Received: 2 October 2025, Revised: 31 October 2025, Accepted: 10 November 2025, Published: 5 January 2026
Abstract
The green bean (Vigna radiate L.), red bean (Phaseolus vulgaris L.) and soybean (Glycine max L.) oils were studied and significant differences in appearance and yield of these oils were found. Soybean oil yielded the highest oil recovery (20.10 ± 0.55%) followed by green bean oil (2.36 ± 0.32%) and red bean oil (1.92 ± 0.04%). The major composition of the rich fatty acid profile was palmitic, stearic, oleic, linoleic and linolenic acids with its own unique physicochemical characteristics. Such as stearic acid of consensus log Po/w 5.93 and log S (ESOL) ‒5.73, and linoleic acid, consensus log Po/w 5.61 and log S (ESOL) ‒4.32 consensuses for these values were calculated based on some determinations results 76 - 77 being those with higher number of attempts). ADME analysis indicated that the absorption of all major FAs in GI was significantly high whereas, only limited penetration through skin and blood-brain barrier (BBB) were observed with synthetic accessibility score ranging from 2.31 - 4.91, respectively. While preparing the topical cream formulations, a composition containing green bean oil: red bean oil: soybean oil in 1:1:1 ratio stabilized with Lipomulse Luxe MB was identified to provide an optimal balance of softness, gloss, stability and hair-conditioning performance. The optimized base cream (F4) exhibited a viscosity of 48,442.33 ± 12.90 cP and maintained phase stability with no separation observed after seven heat-cool cycles. Notably, the formulation containing a 1:1:1 ratio of green bean, red bean, and soybean oils achieved significantly greater hair smoothness and glossiness, as determined by texture analysis and microscopic evaluation, thereby outperforming the other tested formulations. These findings support the suitability of bean oil-based formulations as novel cosmetic carriers.
Keywords: Bean oil, Physicochemical properties, Lipomulse Luxe MB, Lamella Crystal
Introduction
Beans, including red bean (Phaseolus vulgaris L), mung bean (Vigna radiata L.) and seed of soybean (Glycine max L.), are rich in various fatty acid components, especially linolenic, linoleic, palmitic, stearic and oleic acids presumably related to their nutritional and functional properties [1]. Phaseolus vulgaris L. is rich in linolenic, linoleic, palmitic and
oleic acids [2], while Vigna radiata L. possesses a high level of protein, starch, polyphenols and flavonoids [3,4]. Glycine max L. provides oleic, linoleic, linolenic, palmitic and stearic acids in higher concentrates [5] whereas it is also reported for greater calcium, magnesium and zinc content [5]. SwissADME is an open-access computational resource that enables the prediction of drug-like nature, physicochemical properties and ADME (absorption, distribution, metabolism, excretion) profile of small chemical entities including natural fatty acids. Though it is highly usable and makes extensive predictions for pharmacokinetics and medicinal chemistry, occasionally these are not applicable to macromolecules and biologics [6]. However, while several studies have reported the nutritional and chemical composition of bean oils, there remains a lack of integrated research on the physicochemical behavior and formulation characteristics of these oils specifically for cosmetic or hair care emulsions. Addressing these gaps is essential for the rational design of novel, stable, and effective hair treatment products employing bean oil fatty acids. [7,8] Hair consists mainly of keratin proteins, lipids (such as free fatty acids and ceramides), minerals, and pigments arranged in three layers—the cuticle, cortex, and medulla. Hair oils for nourishment are rich sources of fatty acids like lauric, myristic, palmitic oleic, linoleic and linolenic acid. These fatty acids penetrate through and fill the lipid layers, conditioning as well as repairing the hair fiber [9,10]. They improve the health of your hair by acting as an emollient, protecting proteins, retaining moisture and fighting free radicals. Fatty acids bond and attach to the hair shaft protein for protection, which in turn, reduces damage and enhances appearance of your hair. Treatments products utilize a variety of approaches, such as lipid restoration, molecular bonding or smoothing of the cuticle to reduce frizz and increase shine by surface charge and organization of lipids [11,12]. Recent studies have shown that plant-derived oils—such as soybean oil—increase hair moisture, manageability, and fiber integrity, attributed to their unique fatty acid composition and their ability to act as penetration enhancers in emulsified systems. The application of advanced natural emulsifiers for cosmetic oil-in-water emulsions is rapidly expanding, with new strategies focusing on improving emulsion stability and functional delivery to hair fibers [7,8].
Hair treatment emulsions conventionally contain oil-in-water systems which are stabilized by emulsifiers. Lipomulse Luxe MB is a sophisticated, multi-component emulsifier that forms lamellar liquid crystal structures, providing stability and improved moisture management and skin feel in creams. Under polarized light microscopy, these structures appear as a Maltese cross pattern thereby providing visual evidence of lamellar structure which is necessary for emulsion stability and skin compatibility. Based on these considerations, the objectives of this study are: (i) to characterize the physicochemical properties of bean oils, (ii) to design and formulate stable oil-in-water emulsions using these oils, and (iii) to evaluate their efficacy as hair treatment agents. The central hypothesis is that the use of Lipomulse Luxe MB, a liquid crystal-forming emulsifier, will produce more stable and effective emulsions compared to conventional systems, thus enhancing both formulation stability and hair treatment performance. [7,13] Therefore, the present work was conducted to thoroughly examine the physicochemical behavior of bean oil fatty acids and to develop novel treatment emulsions for hair. With the use of Lipomulse Luxe MB, stability and efficacy are achieved and this presentation suggests that these findings have strong promise for translation within the cosmetic hair care industry.
Material and methods
Plant materials
Beans—red bean (Phaseolus vulgaris L.), soybean (Glycine max L.), and green bean (Vigna radiata L.)—were purchased from Khlong Toei Market, Thailand. All beans were washed with distilled water, dried at 50 °C, and then milled using a grinding mill. The powder of beans was extracted with hexane by maceration at room temperature for 7 days. The filtrated and concentrated under vacuum using a rotary evaporator to obtain the crude extracts. The percent yield of crude extracts can calculate formula below:
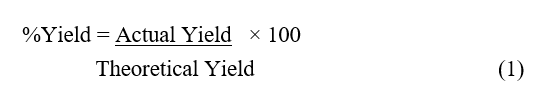
where, Actual Yield: Weight of the product you obtained in the experiment (g). Theoretical Yield - the most product that can possibly be produced from a given reactant mass based on the balanced chemical equation [14].
Chemicals
Chemicals were utilized in this research such as Cetyl alcohol (BASF, Germany) functioned as a co-emulsifier and thickener. Cocamide (CME)/Comperlan KD/Cocamidopropyl betaine (BASF, Germany) served as mild surfactants and conditioners. Tween 20 (Sigma-Aldrich, USA) was used as a nonionic emulsifier. Dehydag wax AB / Hair conditioning wax (BASF, Germany) acted as a structuring and conditioning agent. Glyceryl Stearate (and) PEG-100 Stearate (Chemipan, Thailand), Lipomulse Luxe (Phitsanuchemicals, Thailand) and Lipomulse Luxe MB (Shima, Japan) are advanced liquid crystal emulsifiers for stable, elegant emulsions. Polyquaternium-44 (Chemipan, Thailand) functioned as a hair conditioning polymer. Propylene Glycol (and) 5-Bromo-5-Nitro-1,3-Dioxane (Bronidox L, P.C. Intertrade, Thailand) was incorporated as a preservative. Citric acid (Sigma-Aldrich, USA) was included for pH adjustment and buffering.
Fatty acid structure
Three-dimensional fatty acid structures were generated with MolView by entering the molecular formula into the program’s interface. This routine yielded spatial models that were manually manipulated using molecular modelling software to correct bond angles and achieve the desired geometry. The analysis of conformational features and atomic connectivity that relate to physicochemical properties was facilitated from 3D visualization.
Physicochemical Properties
The physicochemical properties were predicted by SwissADME as online tools that provide global and interpretative information on a compoundʼs molecular weight, lipophilicity (log Po/w), and water solubility (log S) in data analysis. These multi-parametric data were graphically summarized into radar plots, enabling a simultaneous examination of property profiles within a set of fatty acids. Furthermore, the BOILED-Egg model plots were calculated in SwissADME with SMILES entries and gave mapping results of the predicted gastrointestinal absorption/brain penetration properties for each fatty acid, which granted insight about their bio accessibility and potential distribution.
Hair treatment
Bean oil hair treatment product was developed by a selection of four base cream formulations, with emulsifiers and their effects on chemical and physical characteristics tests. Formula 1 (F1) contained Dehydag wax AB as the emulsifier; Formula 2 (F2) used Leximul 561; Formula 3 (F3) was included Lipomuse Luxe, and Formula 4 (F4), that was prepared with Lipomuse Luxe MB according to Table 1. Physical stability of each base was evaluated at the initial time (just prepared product), and heating-cooling by determining pH, viscosity, and phase separation. The optimal pH-sensitive formulation with no expansion ratio of phases was chosen for the next development due to its excellent pH stability, viscosity retention and absence of phase separation at all time points. The best base cream was then improved by the addition of soybean, green bean, and red bean oils (1:1:1) to produce a mixture. The oil blend was added to the base creams F1-F4 to see their effects in each formulation. The enriched compositions were examined and compared regarding their chemical and physical characteristics, where all results were systematically tabulated.
Table 1 Composition of ingredients in emulsion cream base formulations.
Phase |
Ingredient |
F1 %w/w |
F2 %w/w |
F3 %w/w |
F4 %w/w |
Function |
A |
DI water |
Qs.to 100 |
Qs.to 100 |
Qs.to 100 |
Qs.to 100 |
Solvent |
B |
Cetyl alcohol |
8 |
8 |
8 |
8 |
Emollient, thickener, co-emulsifier |
Cocamide (CME) / Comperlan KD / Cocamidopropyl betain |
8 |
8 |
8 |
8 |
Foaming agent, mild surfactant, co-emulsifier, and viscosity enhancer |
|
Tween 20 |
2 |
2 |
2 |
2 |
Emulsifier (nonionic), stabilizes oil-in-water emulsions |
|
Dehydag wax AB / Hair conditioning wax |
10 |
- |
- |
- |
Emollient, stabilizer, imparts structure and conditioning to the hair |
|
Glyceryl Stearate (and) PEG-100 Stearate |
- |
10 |
- |
- |
Multi-component emulsifier; stabilizes and thickens cream emulsions |
|
Lipomuse LuxeTN (Cetearyl Alcohol (and) Glyceryl Stearate (and) PEG-40 Stearate (and) Ceteareth-20) |
- |
- |
10 |
- |
Liquid crystal emulsifier; enhances stability, moisture retention, and sensory feel |
|
Lipomuse Luxe MBTN (Cetearyl Alcohol (and) Glyceryl Stearate (and) PEG-40 Stearate (and) Ceteareth-20) |
- |
- |
- |
10 |
Advanced emulsifier; promotes lamellar, liquid crystal structures for long-term stability |
|
C |
Polyquaternium-44 |
5 |
5 |
5 |
5 |
Cationic polymer, film former, improves hair manageability and conditioning |
Propylene Glycol (and) 5-Bromo-5-Nitro-1,3-Dioxane |
1 |
1 |
1 |
1 |
Preservative; protects product from microbial contamination |
|
Citric acid |
0.2 |
0.2 |
0.2 |
0.2 |
pH adjuster; maintains skin-friendly and product-stable acidic pH level |
Note: F = Formula, TN = Trade Name
Procedure
Preparation of cream was performed as follows: Phase A ingredients were heated to 60 °C under continuous stirring at 500 rpm using an overhead stirrer, ensuring homogeneity; Phase B ingredients were heated separately at 60 °C at 500 rpm until fully dissolved and uniform. Phase B was then gradually added into Phase A while maintaining stirring at 800 rpm for 30 min to allow complete emulsification. The resulting mixture was cooled to 40 °C at 300 rpm before slowly adding Phase C ingredients under continuous stirring until all components were thoroughly blended.
Stability
The pH testing was determined initially and after seven heat-cool cycles to evaluate chemical stability. A Sartorius AG pH meter (Sartorius AG, Germany) was used to measure these. The pH electrode was calibrated with standard buffer solutions before each measurement, and testing of samples was performed at 20 °C. The frequency of measurement was in accordance with the stability testing procedure, day 0 (initial preparation) and after repeated heating-cooling cycles (7 cycles)
The viscosity testing was measured by standard method using Brookfield digital viscometer (Model DV2T, Brookfield, USA). Measurements were performed at controlled temperature of 25 °C using the appropriate spindle for creams, and a specific RPM. Viscosity measurements were obtained at time zero and again after seven cycles of heating-cooling, to assess the mechanical stability and texture uniformity of the creams.
Centrifuge testing was used to evaluate the mechanical stability and phase separation resistance of the creams. The samples were centrifuged by a centrifuge machine (Model 2-16P, Sartorius AG, Germany). The centrifugation speed (4,000 rpm for 30 min at 40 °C) was selected based on prevailing protocols for accelerated physical stability testing of cosmetic emulsions, which typically employ 2,000 - 4,000 rpm for 10 - 30 min to simulate gravitational separation over time. This allows for direct assessment of phase separation resistance and long-term product prediction. The presence or absence of phase separation was systematically noted at the end of each measurement [15].
Visualization of lamellar crystalline structure in cream emulsions (Polarized Light Microscopy)
The lamellar structure of the cream was examined by polarized light microscopy (PLM) to observe Maltese cross pattern in reflected light as this is observed in crystalline lamellae associated with emulsified systems. A small amount of the cream was taken on a clean glass slide and then, subsequently a coverslip was lowered gently so that it reached to an optimum thickness for microscopic examination. The prepared slide was observed with polarized light microscope (Olympus, model BX53, Japan). This apparatus renders the anisotropic areas and birefringence patterns, the Maltese cross, observable-providing visual confirmation of lamellar liquid crystal structure inside cream matrix. Such structural investigations can be relevant to understand the stability and compatibility of emulsions, as well as for the more functional aspects in case of formulated products.
Evaluation of hair treatment efficacy
The effectiveness of hair treatment product was assessed on human hair locks which were first washed with deionized water to thoroughly remove any surface residues. The hair was then washed, rinsed and readied for experiments. For the treated groups, hair was covered uniformly with a film of the test hair conditioning product and left to stand for 30 min before drying by hairdryer for 5 min. This treatment was repeated three times a week for a total of 4 weeks. Each treatment group consisted of a minimum of three replicate hair locks, with measurements repeated in three independent experiments to ensure statistical robustness. This approach is widely adopted in cosmetic emulsion and hair treatment studies to achieve reliable and comparable data. The microstructural morphology of the hair cuticle was evaluated using a compound light microscope (Nuvotech, E303 LED, Hungary) before and after 4 weeks of the intervention. Hair fibers were applied to glass slides and observed under bright field at 100× magnification. Digital images of the hair cuticle were captured and compared to each other directly to determine changes in surface nature, smoothness, cuticular alignment and damage reduction. Samples of untreated hair were used as controls for baseline comparisons. All protocols were strictly designed to maintain control over the homogeneity of environmental conditions, application and microscopy observation among samples for an objective evaluation of product action on hair.
Statistical analysis
Values are expressed as mean ± SD. A one-way ANOVA with Tukey’s test was used to analyze differences between treatment and control groups. A p-value < 0.05 was considered statistically significant with the use of a two-tailed test. Statistical analysis All data were analyzed by GraphPad Prism 10.0.
Results and discussion
Physical characteristics
Visual properties, the color features of oil obtained from various bean seeds, as shown in Figure 1, exhibited different physical appearances. The green bean (Vigna radiata L.) oil is clear yellow in color (Figure 2), which can be an indication of relatively high purity and low pigment content. Compared with that from red beans (Phaseolus vulgaris L.), the oil exhibits a darker yellow color, which might result partially on account of its natural pigments or due to specific minor constituents unique for this species. The soybean oil has deep yellow or golden colored indicating the greater number of carotenoids/carotenoid-like pigments in soybean oil. Although Figures 1 and 2 are primarily descriptive, it is important to note that the physical appearances—such as color intensity, clarity, and pigment content—can be correlated with objective analytical parameters, including carotenoid, chlorophyll, and purity levels measured spectrophotometrically. Previous studies have quantitatively shown that oils with higher L* and b* values in CIELAB color space, as seen in soybean and green bean oils, are indicative of higher lightness and yellowness, respectively, and those values often reflect lower impurity and higher oxidative stability. In practice, these colorimetric features play an essential role in consumer perception and market acceptance of cosmetic oils [16]. These color variations are important “carriers” of information on the compositional differences among oils and may affect their use in food- or cosmetic-related products. Glycine max L. yielded the highest oil content at 20.10 ± 0.55%, which was significantly greater than both Vigna radiata L. (2.36 ± 0.32%) and Phaseolus vulgaris L. (1.92 ± 0.04%) [14].

Figure 1 The physical characteristics of bean: (a) green bean, (b) red bean, (c) soybean.

Figure 2 The physical characteristics of bean oils: (a) green bean, (b) red bean, (c) soybean [14].
Fatty acid beans
Fatty acids in beans such as red bean, green bean and soybean have been systematically investigated for their nutritional and functional values, particularly with respect to health care and personal products. Beans are rich in saturated and unsaturated fatty acids, which are important for many biological functions including antioxidant activity, stability of membrane as well as possible enhancement on the growth of hair and health of the scalp. An inclusive knowledge of the fatty acid contents in these legumes is crucial to optimize the preparation of hair treatment products using bean oils [17]. The form is shown in Figure 3.
Saturated fatty acids
Palmitic acid (hexadecanoic acid, C16H32O2) of molecular weight 256.4276 u is a major constituent saturated fatty acid in beans. It contains one hydrogen bond donor and two acceptors as well and has a molar percent composition of 74.943% carbon; 12.578% hydrogen; and 12.479% oxygen on the surface. It contains high amounts of palmitic acid: In velvet bean (Mucuna pruriens), it is playing an important role to build up cell wall structure and energy storage. In hair care, for example, saturated fatty acids such as palmitic acid improve humidity retention and help shield from oxidative damage [18].
Stearic acid (octadecanoic acid, C18H36O2) has molar mass 284.4812 u and is another type of saturated fat that beans contain. It has one hydrogen bond donor, and two acceptors, with a carbon content in the molecule of 75.996%, hydrogen 12.755% and oxygen 11.248%. Stearic acid is found in many sorts of bean, but it can also be made from cocoa butter. It is an integral component of lipid bilayers and functions as an emollient in other hair products to condition and manage texture [18].
Monounsaturated fatty Acids - MUFAs
Oleic acid ((Z)-octadec-9-enoic acid, C18H34O2) with molecular weight 282.4654 u, the most well-known monounsaturated fatty acid from legumes has one hydrogen bond donor and 2 acceptors. Its percentage composition is 76.539% carbon, 12.133% hydrogen and 11.329% oxygen. Oleic acid can be found in many legumes and is known for its hydrating and shielding features in hair care compositions. It improves hair softness, reduces breakage and favors the health of the scalp [18].
Polyunsaturated fatty Acids - PUFAs
Oleic acid ((Z)-octadec-9-enoic acid, C18H34O2) with molecular weight 282.4654 u, the most well-known monounsaturated fatty acid from legumes has one hydrogen bond donor and 2 acceptors. Its percentage composition is 76.539% carbon, 12.133% hydrogen and 11.329% oxygen. Oleic acid can be found in many legumes and is known for its hydrating and shielding features in hair care compositions. It improves hair softness, reduces breakage and favors the health of the scalp [18].
Linolenic acid (octadeca-9,12,15-trienoic acid; C18H30O2), of molecular weight 278.4338 u, possesses one hydrogen bond donor and two acceptors with elemental contents of 77.65% carbon, 10.86% hydrogen and 11.49% oxygen. This chemical is very high in beans, particularly soybeans and has anti-inflammatory properties. Application of onion juice on the hair can promote the health of the scalp and prevent irritation, its astringent qualities help fight dandruff.
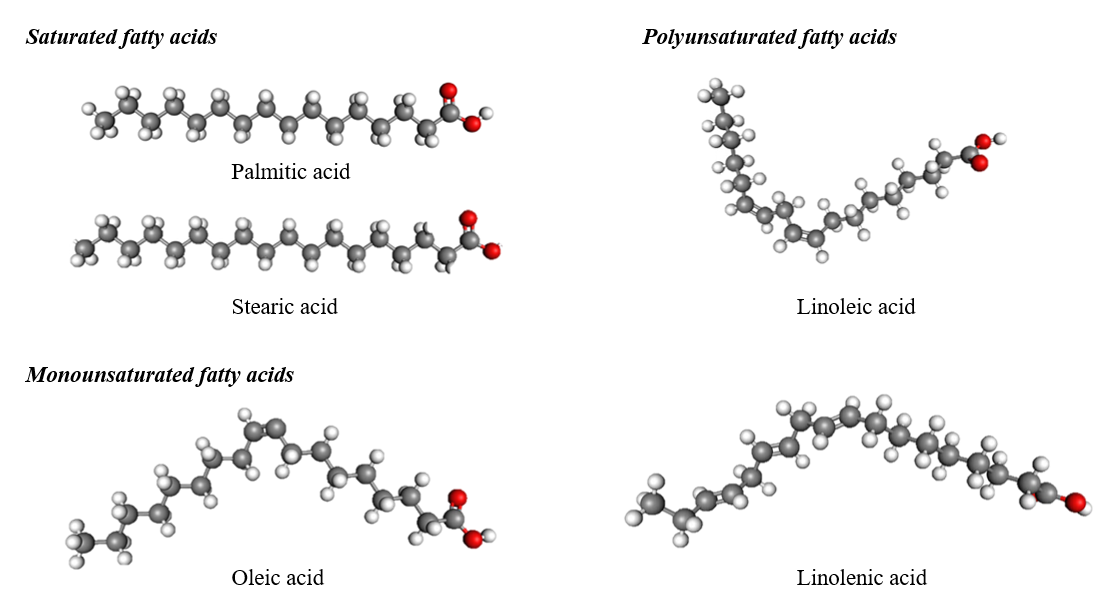
Figure 3 Major fatty acids in bean oils.
Physicochemical properties
The physicochemical properties of main fatty acids in oil from three bean species are reported with special focus on basic aspects and use as materials for formulation technology. Critical parameters considered are core physicochemical properties (molecular formula and molecular weight), lipophilicity estimated through different log Po/w values (iLOGP, WLOGP, SILICOS-IT and consensus log Po/w) and water solubility (log S from ESOL, Ali, and SILICOS-IT) listed in Table 2. These assessments make contributions to the solubility, hydrophobicity, and structural that are important for subsequent applications in cosmetics.
Saturated Fatty Acids: The major saturated fatty acid is palmitic acid (C16H32O2, 256.42 g/mol). Its log Po/w values, which are as measures of lipophilicity, are 3.85 (iLOGP), 5.55 (WLOGP), and 5.25 (SILICOS-IT) with a consensus value of 5.20 also support its high hydrophobicity. The water solubility is estimated as log S (ESOL) = ‒5.02, log S (Ali) = ‒7.77 and log S (SILICOS-IT) = ‒5.31 corresponding to medium/ moderate to poor solubility in water, respectively. These results highlight the broad use of palmitic acid as an emollient and structuring for lipid-based systems, due to its poor solubility in water but good stability in hydrophobic terms. Stearic acid (C18H36O2, 284.48 g/mol) is another important saturated fatty acid with higher hydrophobicity than palmitic acid; it has the log Po/w values of 4.30 (iLOGP), 6.33(WLOGP) and 6.13(SILICOS-IT); and a consensus log Po/w value equal to that of oleic acid, which is generally accepted as higher in lipophilicity compared to the other compounds according to iLOGP, WLOGP, SILICOSIT descriptors (5.93). Its water solubility is very low, with log S (ESOL) = −5.73, log S (Ali) = −8.87 and log S (SILICOS-IT) = −6.11, this can be regarded as a poorly soluble substance in aqueous. This The physicochemical characteristics of stearic acid make it suitable for products that require stability in lipid matrices or when water resistance is a desirable property. Comparison between palmitic acid and stearic acid both palmitic and stearic acid are saturated, highly hydrophobic (low water-solubility), non-polar molecules; however, stearic acid is more hydrophobic and has a lower solubility in water than does palmitic acid as evidenced by the higher Log Po/w values for the former and its lower log S. This difference is the basis for their differential functions in functional formulations like food and cosmetics, stearic acid tending to provide increased rigidity and water repellency. Comparative studies have shown that stearic acid may be more beneficial with respect to health consequences compared to palmitic acid, due to differences in lipid metabolism and biological effects [19].
Monounsaturated fatty cidas, the main monounsaturated fatty acid in beans is oleic acid (C18H34O2, 282.46 g/mol). It shows log Po/w values of 4.01 (iLOGP), 6.11 (WLOGP), 5.95 (SILICOS-IT) and consensus value of 5.65, rendering it high hydrophobicity. The water solubility parameters are log S (ESOL) ‒5.41, log S (Ali) ‒8.26 and log S (SILICOSIT) ‒5.39 that corresponds to a moderate to poor solubility compound. As the advanced structure former mentioned in both food and personal care emulsions, oleic acid, which has high lipid-solubility is an effective skin conditioner and moisturizer. Oleic acid in plant oils, which acts as a stabilizer of triacylglycerols and plays an important role for mouthfeel and emulsion properties is supported by studies [20].
Polyunsaturated fatty acids, linoleic acid (C18H32O2, 280.45 g/mol) is a polyunsaturated fatty acid with log Po/w of 4.72 (iLOGP), 5.88 (WLOGP), 5.84 (SILICOS-IT) and consensus value of 5.61. These solubilities are moderate to poor in water, with log S (ESOL) of ‒4.32, log S (Ali) of ‒7.41 and log S (SILICOS-IT) of ‒5.47, respectively. These features underscore the moderate hydrophobicity of linoleic acid and its ability to participate both in hydrophobic and amphiphilic medium, an important feature for a barrier repair as well as emollient action. Linolenic acid (C18H30O2, 278.43 g/mol) is a polyunsaturated fatty acid, which has calculated log Po/w values of: iLOGP (4.25), WLOGP (5.83), SILICOS-IT (5.55) and consensus (5.21). It shows only marginally better water solubility compared to linoleic acid with log S (ESOL) ‒4.02, log S (Ali) ‒7.16 and log S (SILICOS-IT) ‒5.26. These properties make linolenic acid highly suitable in compositions for improving skin barrier function and providing anti-inflammatory activity. Linoleic acid and linolenic acid compared linoleic acid and linolenic acid are important polyunsaturated fatty acids (PUFAs) of the same series, they share similar chemical structure and physicochemical property. Nonetheless, linoleic acid is somewhat more hydrophobic and less water-soluble than linolenic acid. Recent studies report their separate metabolic pathways in the biological systems and show that linolenic acid is desaturated to a higher extent than linoleic acid, which may also affect their nutritional and physiological significance. However, both acids contribute significantly to membrane fluidity and the two acids have great importance in skin/hair care and as indispensable dietary fatty acids.
Conclusions, systematic characterization of the physicochemical properties, lipophilicity and water solubility in bean oils of fatty acids shows different profiles among saturated, monounsaturated, and polyunsaturated fatty acids. Among them, palmitic and stearic are the most hydrophobic saturated fatty acids whereas oleic, linoleic and linolenic represent a middle ground between moderate solubility and amphiphilicity that gives rise to their diverse functionality. These differences observed were in line with previous literature and represent a good scientific basis for the selective use of certain fatty acids used in functional foods and cosmetics. Notably, the comparative analysis of log Po/w and water solubility values for each major fatty acid demonstrates a clear relationship with the overall stability and textural properties of the final emulsion cream formulations. Creams incorporating fatty acids with higher lipophilicity (log P) and moderate-poor water solubility tend to provide greater viscosity and improved phase stability. Further, regression analysis revealed a positive correlation between the lipophilicity (consensus log Po/w) of the fatty acids and the viscosity/stability of the formulated creams, supporting the hypothesis that higher lipophilicity is predictive of improved emulsion viscosity and phase stability.
Table 2 Physicochemical properties, lipophilicity, and water solubility of major fatty acids.
|
Physicochemical Properties |
Lipophilicity |
Water
Solubility |
||||||
Formula |
Molecular weight |
Log Po/w (iLOGP) |
Log Po/w (WLOGP) |
Log Po/w (SILICOS-IT) |
Consensus Log Po/w |
Log S (ESOL) |
Log S (Ali) |
Log S (SILICOS-IT) |
|
Saturated fatty acids |
|||||||||
Palmitic Acid |
C16H32O2 |
256.42 g/mol |
3.85 |
5.55 |
5.25 |
5.20 |
‒ 5.02 Moderately soluble |
‒ 7.77 Poorly soluble |
‒ 5.31 Moderately soluble |
Stearic acid |
C18H36O2 |
284.48 g/mol |
4.30 |
6.33 |
6.13 |
5.93 |
‒ 5.73 Moderately soluble |
‒ 8.87 Poorly soluble |
‒
6.11 |
Monounsaturated fatty acids |
|||||||||
Oleic acid |
C18H34O2 |
282.46 g/mol |
4.01 |
6.11 |
5.95 |
5.65 |
‒ 5.41 Moderately soluble |
‒ 8.26 Poorly soluble |
‒ 5.39 Moderately soluble |
Polyunsaturated fatty acids |
|||||||||
Linoleic acid |
C18H32O2 |
280.45 g/mol |
4.72 |
5.88 |
5.84 |
5.61 |
‒ 4.32 Moderate soluble |
– 7.41 Poorly soluble |
– 5.47 Moderate soluble |
Linolenic acid |
C18H30O2 |
278.43 g/mol |
4.25 |
5.83 |
5.55 |
5.21 |
– 4.02 Moderate soluble |
– 7.16 Poorly soluble |
– 5.26 Moderate soluble |
SwissADME in silico results provide important predictions regarding drug-likeness, absorption, and molecular safety, it is acknowledged that the direct relevance of these pharmacokinetic parameters to topical cosmetic performance is more indirect; nevertheless, such data help rationalize ingredient selection based on structural safety and bioavailability, offering a complementary perspective alongside empirical formulation testing. Pharmacokinetic parameters (GI absorption, Log Kp skin permeation and BBB permeant) offer intrasights into the biological behavior of FAs in relationship to intestinal uptake, skin barrier penetration and passage across blood-brain barrier. While GI absorption and BBB permeability are primarily pharmacokinetic parameters relevant to oral or systemic drug delivery, their inclusion here provides a comprehensive evaluation of the major fatty acids’ overall biocompatibility, safety, and distribution profiles, as recommended by contemporary cosmetic science guidance. These predictions can reveal unintended potential for systemic exposure or CNS entry with topical or transdermal delivery, thus informing both safety risk assessments and regulatory considerations for cosmetic ingredient selection. GI absorption acts as an indicator of the degree of uptake into the digestive tract, higher numbers resulting in increased oral bioavailability. Log Kp, represents skin permeation, where a more negative value indicates lower permeability. Has the ability of a compound to enter the CNS is evaluated by BBB permeant. Drug-likeness is assessed against screening qualities, for example Lipinskiʼs rules, in which violations of MLOGP indicate extreme hydrophobicity with possible poor absorption or distribution. The bioavailability score represents the empirical oral bioavailability probability, whereas synthetic accessibility represents the relative laboratory synthesis difficulty. Lead likeliness shows positive and useful molecular features in drug candidate development, while Brenk alerts show negative substructures that could cause toxicities or promiscuities [21-24].
Saturated fatty acids, palmitic acid and stearic acid, palmitic acid and stearic acid Both of these can be absorbed through the gastrointestinal (GI) system to a high degree but are able to penetrate the skin only moderately well (Log K p –2.77, –2.19 cm/s), supporting oral uptake yet comparatively weak cutaneous uptake 91. Evidently, palmitic acid is a permeant through the blood-brain barrier (BBB), in contrast to stearic acid. Both FAs break the rule of Lipinski related to MLOGP (over 4.15), which is a lipophilicity presume, but they keep a good value that indicates bioavailability (0.85). The synthetic accessibility values of 2.54 (stearic acid) and 2.31 (palmitic acid) indicate that both compounds are relatively easy to synthesize. With lead like similarity, both fatty acids have two of rotatable bond violations as well as high XLOGP3, but none show Brenk alerts (which indicates good safety in medicinal chemistry) [25,26]. Although similar in GI absorption and DL profiling, the palmitic acidʼs brain penetration that is invariably different than stearic acid. The desired bioavailability and synthesis scores of these conformers have led to the promising potential for drug-like development, as well as their structural safety which is further supported by a lack of Brenk alerts.
Monounsaturated fatty acid, oleic has good absorption from gastrointestinal tract and moderate skin permeation (Log Kp –2.60 cm/s)161, but it does not passthrough blood-brain barrier. It has one violation of Lipinski’s rule (MLOGP > 4.15) and a bioavailability score of 0.85, like saturated fatty acids. Its synthetic accessibility score is 3.07, which also shows it to be more complicated than drug-like synthesis. Medicinal chemistry screening it finds two lead likeness violations and one Brenk alert—an isolated alkene—requiring to be careful about chemical reactivity and stability concerns [24,27].
Polyunsaturated fatty acids (PA), linoleic acid and linolenic acid exhibit high GI absorption but low skin permeation (Log Kp 2.77 and – 2.78 cm/s, respectively) and do not pass BBB. Both molecules have a single Lipinski rule-of-5 violation for lipophilicity and lower bioavailability score (0.55). Synthetic accessibility scores (4.91 and 4.89) reflect enhanced chemical complexity are presented in Table 3. Both violate some lead likeness rules (low MW, high XLOGP3), but no Brenk alerts are present indicating a favorable medicinal chemistry profile. Comparison: Linoleic and linolenic acids are comparable within each factor and synthesized with respect to saturated and monounsaturated fatty acids. Their low bioavailability and increased synthetic complexity could limit their clinical use, but the exclusion of Brenk alerts indicates their chemical safety.
In conclusion, SwissADME modelling indicates high intestinal absorption of the main fatty acids and their overall drug-likeness, with lipophilicity being the main challenge toward optimal bioavailability. The easiest and least structurally challenging type of fatty acid to synthesize are the saturated ones, followed by the polyunsaturated ones (devoid of a safety alert) and then the monounsaturated. SwissADME was used to predict physicochemical, pharmacokinetic, and medicinal chemistry properties of natural and candidate drug compounds. Applications include the assessment of new anti-inflammatory agents, nutraceuticals, and antimalarial analogues, as well as the directing of synthetic strategies towards drug design and toxicity testing. In summary, computational ADME model is a powerful tool that may guide the choice and optimization of leads for lead-compound development.
Table 3 Pharmacokinetic, drug-likeness, and medicinal chemistry profiles of major fatty acids predicted by SwissADME.
Type of fatty acid |
Pharmacokinetics |
Drug likeness |
Medicinal Chemistry |
|||||
GI absorption |
Log Kp (skin permeation) |
BBB permeant |
Lipinski |
Bio availability Score |
Synthetic accessibility |
Lead likeness |
Brenk |
|
Saturated fatty acids |
||||||||
Palmitic Acid |
High |
‒ 2.77 cm/s |
Yes |
Yes; 1 violation: MLOGP > 4.15 |
0.85 |
2.31 |
No; 2 violations: Rotors > 7, XLOGP3 > 3.5 |
0 alert |
Stearic acid |
High |
‒ 2.19 cm/s |
No |
Yes; 1 violation: MLOGP > 4.15 |
0.85 |
2.54 |
No; 2 violations: Rotors > 7, XLOGP3 > 3.5 |
|
Monounsaturated fatty acids |
||||||||
Oleic acid |
High |
‒ 2.60 cm/s |
No |
Yes; 1 violation: MLOGP > 4.15 |
0.85 |
3.07 |
No; 2 violations: Rotors > 7, XLOGP3 > 3.5 |
1 alert: isolated_alkene |
Polyunsaturated fatty acids |
||||||||
Linoleic acid |
High |
– 2.77 cm/s |
No |
Yes; 1 violation: MLOGP > 4.15 |
0.55 |
4.91 |
No; 2 violations: MW < 250, XLOGP3 > 3.5 |
0 alert |
Linolenic acid |
High |
– 2.78 cm/s |
No |
Yes; 1 violation: MLOGP > 4.15 |
0.55 |
4.89 |
No; 2 violations: MW < 250, XLOGP3 > 3.5 |
0 alert |
Multidimensional radar plot comparison of physicochemical properties
The radar charts plotted in Figure 4 condense from a visual perspective the multidimensional physicochemical characteristics of important fatty acids present in bean oils, that is, palmitic, stearic, oleic, linoleic, and linolenic acids. These plots combine measures of molecular properties such as molecular size, polarity, lipophilicity, flexibility, saturation, and solubility on a single plot, allowing direct comparison of the structural and behavioral profile for each fatty acid. As an example, saturated fatty acids (palmitic and stearic) are lipophilic and less flexible than unsaturated forms, and polyunsaturated fatty acids (linoleic and linolenic) have increased flexibility regarding different solubility profiles. Recently, radar plots have been used to quickly visually evaluate and compare the macro properties of bioactive lipid compounds and facilitate the selection of compounds for formulation and/or in silico prediction of bioavailability. A study by Staneva et al. [28] utilized radar graphs to compare the properties of omega-3 fatty acids and showed that they could be used for filtering molecules with excellent bioavailability and for the selection of functional ingredients.
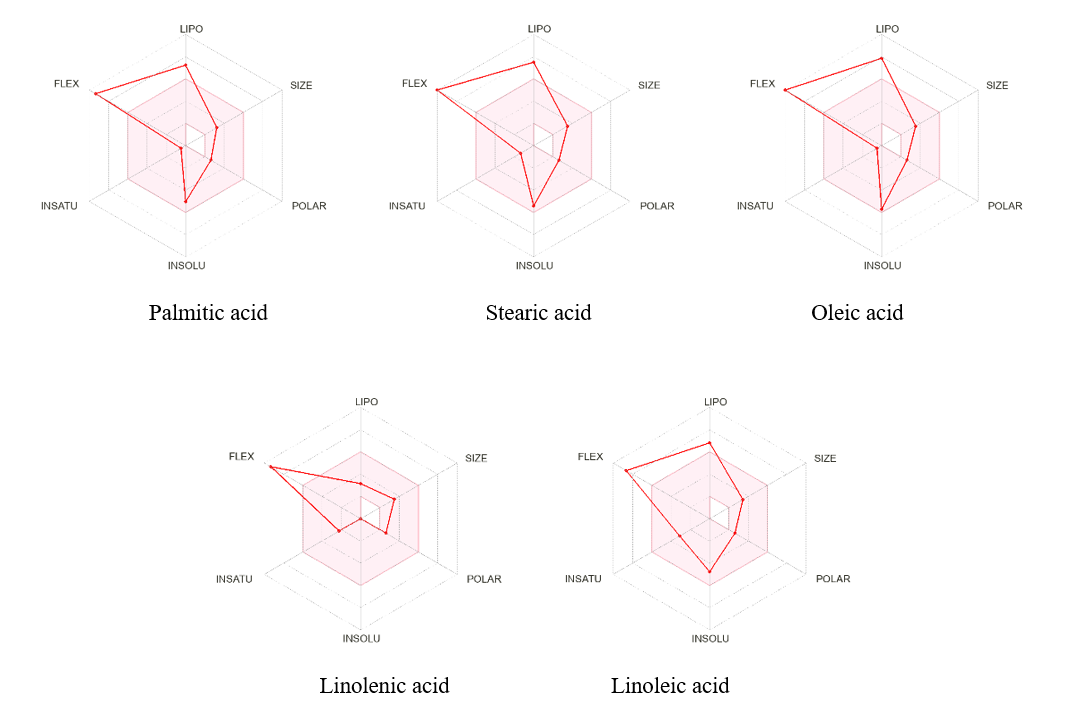
Figure 4 Radar plots of physicochemical properties of major fatty acids found in bean oils.
Predicted GI absorption and BBB penetration of fatty acids by BOILED-Egg model
The depicted BOILED-Egg model plots (Figure 5) depict the anticipated HIA and BBB-membrane penetration of major fatty acids present among the bean oils, i.e., the palmitic, stearic, linoleic, linolenic, and oleic acids. Every fatty acid is represented in a physicochemical property space where the white area (yolk) corresponds to a high passive gastrointestinal absorption tendency, and the yellow region (white) suggests crossing the blood-brain barrier. None of the key fatty acids is in a region corresponding to BBB-penetrant compounds, but all are favorably positioned for uptake by the intestine, commensurate with their amphiphilic and bioavailable nature. The BOILED-Egg model is becoming more popular in drug candidate and lipid research to assess the oral bioavailability and central nervous system (CNS) distribution. For instance, Di et al. [29] had indicated its usage in evidence-based prediction of passive diffusion potential for a wide range of molecules, therefore emphasizing its strong early-stage screening potency.
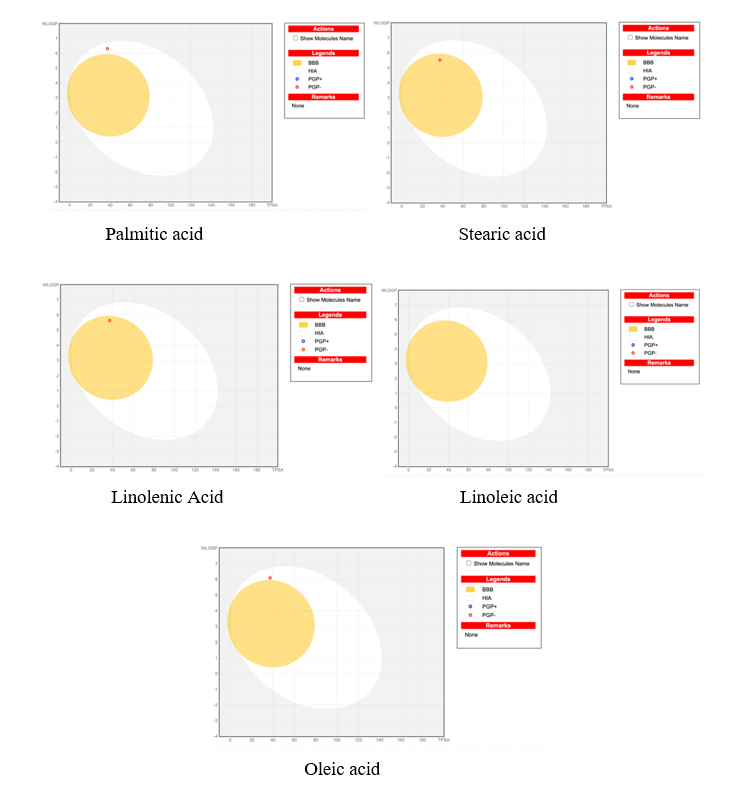
Figure 5 BOILED-Egg model plots illustrating the predicted gastrointestinal absorption and brain penetration of major fatty acids.
Development of base cream
The comparison between the four emulsion base cream formulas (F1 - F4), in which the type of emulsifier system varied, reveals that the cream with the softest, smoothest, and most uniform texture is presented by F4 using Lipomulse Luxe MB (Figure 6(a)). This outcome highlights the key role of emulsifier choice for modifying both the rheological and sensory aspects of topical emulsion formulations. Sensory and performance research with cosmetics reinforces these findings. Systems such as Lipomulse Luxe MB always yield creams that feature elegant textures, lightness, non-greasiness, and improved application spread and tactile softness factors high on consumersʼ wish lists as well as formulatorsʼ. Based on product and sensory information obtained from Lipomulse Luxe MB, it allows for the development of a stable liquid crystal while being used in creams, giving improved moisturizing efficacy and sensory feel to consumers. Therefore, F4 is the best choice for cosmetic creams when the superior feel, homogeneity, and skin compatibility are of great concern. For example, Hanifah and Jufri [30] have established that creams are softer, non-greasy, and homogeneous when the appropriate emulsifiers are used in the formula, which corroborates our findings [31,32].
The results in Figure 6(b) demonstrate that the fourth cream base formulation featuring a mixed ratio of green bean, red bean, and yellow bean oils (at a 1:1:1 ratio) displayed an extremely soft & smooth texture with high levels of gloss and uniformity. This finding is in line with previous reports revealing that the choice and percentage of vegetable oils present in the oil phase play a preponderant role on the rheological, mechanical, and sensory properties of cosmetic emulsions, which result in an enhancement of spread ability, hardness, and consumer acceptability. For instance, Andonova et al. [33] observed that changes in the composition of the natural oil phase carried out in creams significantly affected viscosity, softness, glossiness, and sensory characterization of tactile sensations during application. These results substantiate the idea that a selected mixture of different bean oils could improve both technical and sensorial features in cosmetic creams. While Figure 6 provides a visual comparison of cream texture and uniformity, a more objective assessment would involve quantitative texture analysis, such as measuring firmness, spread ability, and viscosity. Studies have demonstrated that adding natural oils to emulsions significantly alters these mechanical parameters—with higher oil concentrations often producing increased firmness and improved spread ability as measured by texture analyzers. The observable smoothness and homogeneity in F4 likely correlate with enhanced spread ability and more favorable texture profile, which are critical for consumer satisfaction and performance of topical creams [34].

Figure 6 Physical appearance and texture of emulsion-based creams (F1 - F4) formulated with different emulsifiers (a); texture and appearance of bean oil emulsion cream formulation (b).
Stability test
All measurements of pH, viscosity, and hair microscopy were independently performed in triplicate for each formulation or treatment, to ensure data robustness and reproducibility. The table shows that the pH values of all emulsion formulations remain very stable after a total of seven heating-cooling cycles, with minor differences observed between initial and post-cycling readings for any imbricate. Stability study demonstrates no significant pH drift and value remained within acceptable limits for skin compatible cosmetic creams. Statistical analysis was performed using ANOVA with Tukey’s post hoc test; replicate number (n = 3) and p-values are provided in Table 4 and indicated in the superscript. This result is also in accordance with earlier observations reporting that effectively stabilized emulsions, with correct selection of surfactants and buffering system, retained their initial pH at the potent thermal challenges commonly occurring within multiple sterilization cycles ensuring safety and long-term stability in topical applications. This observation is in accordance with that of Jiamphun et al. [35] in which all tested nano emulsions showed physical and pH stability after heat-cool cycles as well as during long term storage, with no evidence of change in pH or phase separation. Similarly, Navarro-Pérez et al. [36] reported that stable pHs of cosmetic emulsions with high formulation stabilities can be achieved by use of an optimized surfactant and buffer system. In general, the data clearly demonstrate the critical role of choosing appropriate surfactants and buffer systems to maintain both pH and structural stability of cosmetic emulsions subjected to thermal cycling, as also expounded on by Xiao et al. [37].
Viscosity: The viscosity is also remarkably maintained for all formulations upon heating-cooling stress. Despite that all formulations show a little increment, the statistical analysis proves a high correlation between initial and after-cycle viscosity, indicating good mechanical stability and structural integrity. These results agree with the existing literature indicating that rheology and viscosity measurements are reliable indicators for emulsion stability and resistance against physical degradation and phase separation if optimized surfactant systems and composition are employed [37].
Moreover, emulsions prepared with suitable stabilizer systems have been found to have slight alteration of the viscosity after being subjected to repeated thermal cycles with good physical stability without phase separation [38]. This finding is in accordance with those of Shafiei et al. [39], who report that formulations that maintain stable viscosity values over the period are those with excellent phase stability and low tendency to physical instability.
Centrifugation: All the formulation remained both initially and after a cycle of heating and cooling, as shown by centrifuge action. This characterization results in confirming that the emulsions possess a homogenous structure, which exhibits a strong interface stability and high resistance to physical disruption. Consistent with reports of the past, centrifugation as a method to accelerate stability testing is widely used for assessing emulsion stability; formulations without phase separation under centrifugal force are considered highly stable and suitable for consumer or industrial use. Specifically, Navarro-Pérez et al. [36] stated that cosmetic emulsion systems without phase separation after centrifugation closely predicted their real-world stability and proclaimed that the centrifuge test was a simple, reliable method to evaluate the stability of cosmetics and emulsions. Related to these findings is the work of Estanqueiro et al. [38], which evidenced that centrifugation of 30 min at 3000 rpm, repeated three times, constitutes an acknowledged accelerated test for emulsions because the formulations do not present phase separation as a mark of strong stability after such a process. Accordingly, centrifugation tests offer a reliable short-term assessment of emulsion stability; emulsions that remain homogeneous and do not undergo phase separation following centrifugation are considered physically stable and have the potential for further product formulation.
Optimal formula for four base creams tested, the one that showed the best overall performance is Formula F4, which has stable pH and viscosity and remains completely phase-separated after extensive thermal cycling and centrifuge stability test. These characteristics suggest that F4 delivers the best long-term physicochemical stability, and consumer product performance and therefore it is the best formulation to be chosen for high-quality cosmetic emulsion applications.
Table 4 Comparison of pH, viscosity, and centrifuge stability.
Formula |
pH |
Viscosity |
Centrifuge |
|||
initial |
Heating-Cooling
|
initial |
Heating-Cooling
|
initial |
Heating-Cooling
|
|
F1 |
5.45 ± 0.21a |
5.43 ± 0.23a |
38,252.67 ± 24.68a |
38,544.67 ± 34.95a |
- |
- |
F2 |
5.62 ± 0.32b |
5.61 ± 0.41b |
52,640.00 ± 34.77b |
52,966.67 ± 25.11b |
- |
- |
F3 |
5.68 ± 0.12c |
5.70 ± 0.37c |
43,457.00 ± 10.44c |
43,953.33 ± 17.90c |
- |
- |
F4 |
5.71 ± 0.51cd |
5.72 ± 0.34d |
48,442.33 ± 12.90d |
48,670.33 ± 15.82d |
- |
- |
Note: Superscript letters a, b, c, d indicate statistically significant differences (p < 0.05). Centrifuge (-) = no phase separation observed.
Microscopic lamellar crystal structure
Figure 7 provided below clearly shows the Maltese cross pattern demonstrated by samples under polarized light microscopy, which is characteristic of lamellar crystalline structures in emulsion creams compounded with bean oils and Lipomulse emulsifier. This analysis confirmed that the lamellar crystal structure formed within the emulsion cream matrix is homogeneously and finely distributed throughout the sample, supporting the high stability and uniformity of the formulation, as visualized under microscopy. Our microscopic observation further supplements this finding by demonstrating highly uniform distribution of lamellar liquid crystals throughout the emulsion matrix, which is indicative of excellent formulation homogeneity. The formation of these lamellar structures is related to the use of multi-component emulsifiers, such as Lipomulse Luxe MB that includes cetearyl alcohol, glyceryl stearate, PEG-40 stearate and ceteareth-20. This combination promotes the liquid crystals formation during emulsification. The thus formed lamellar structures, not only stabilize against coalescence and phase separation but also mimic intercellular lipids of human skin, to enhance moisturization and barrier function. It is generally recognized that lamellar liquid crystal emulsions have a much-improved cosmetic performance such as texture enrichment, stability improvement and penetration enhancement of active ingredients. It is now an accepted scientific consensus that the layered ordered phases, as evidenced by Maltese cross, is paramount to achieve emulsion stabilization and skin performance for modern emulsion-based creams [40]. This is also evidenced by the investigations showing that the Maltese cross produced upon examination of cosmetic emulsions under polarized light corresponds directly to lamellar liquid crystal entities. These structures are related to enhanced stability, hydration and texture properties as compared to conventional emulsions [41]. Moreover, there is proof that the Maltese cross is visible with all lamellar liquid crystal formulations in polarized light which correlates with stability, humectant effectiveness, sensorial benefits and clinical efficacy [42]. Further work also supports that the Maltese cross is a key hydration and structural stability indicator in cosmetic emulsions [43].

Figure 7 Microscopic lamellar crystal structure in emulsion cream containing bean oil and Lipomulse Luxe MB.
Microscopic assessment of hair structure
Microscopic examination of hairs before and after application with the developed hair product demonstrated marked improvements in structure and appearance. Non-treated hair was initially characterized by having rough, coarse and non-uniform surface. But after being treated with formula F4, which included Lipomulse Luxe MB and a blend of green bean, red bean, soybean oils hair fibers improved significantly in both touch and appearance showing smoothness, glossiness and elasticity provides as described in Figure 8. The number of split ends that developed was significantly decreased and the hair looked healthy; this is probably due to the emollient effect and film former properties of the liquid crystal emulsion and bioactive lipids extracted from beans. This transition reflects the effectiveness of the product in conditioning and repairing hair fibers, possibly through increased lubrication, moisture retention and suppressed friction that support enhanced alignment and coverage of cuticles. The formation of lamellar liquid crystals, confirmed by Maltese cross patterns under polarized light microscopy, enables the cream emulsion to mimic the natural lipid organization of the hair cuticle. This facilitates enhanced intercellular lipid replenishment and uniform film formation, reducing friction between hair fibers. Consequently, the cuticle layer becomes smoother, better-aligned, and more capable of reflecting light, which manifests as improved gloss and reduced split ends. Reinforcing cuticle lipids and protein structures new literature support that liquid crystal emulsions and film-former/polyquaternium systems, provided with increasing stability properties [44-46], that guarantee a long-lasting conditioning effect acting in repair of the split ends. It is also known that lipid bonding technology and topically applied emollients can help restore the integrity of the cuticle, smooth rough hair surfaces, enhance shine and reduce breakage as confirmed by microscopic studies [11,47]. Cumulatively, compositions including film-formers and bioactive lipids have been found to repair split ends, to enhance cuticle structure, and to improve manageability and overall cosmetic properties for hair.

Figure 8 Microscopic (100x) comparison of hair structure before (a) and after treatment (b).
Conclusions
The physical-chemical and functional properties of the oils of green bean, red bean and soybean accessions were systematically compared showing relevant differences in oil yield, color, fatty acid profile which render them suitable for technical as well as cosmetic uses. Additionally, from a sustainability perspective, the utilization of legume-derived oils (soybean, green bean, red bean) leverages agricultural byproducts and renewable plant resources, thereby aligning with eco-friendly cosmetic innovation strategies. Regarding safety, in silico analyses (SwissADME, BOILED-Egg model) and empirical stability tests highlight a favorable safety profile for the principal fatty acids, supporting the foundational safety of these multifunctional blends for topical use. A full examination of the main fatty acids (from palmitic, stearic down to oleic, linoleic and linolenic) showed that highly saturated fatty acids such as palmitic or stearic have high hydrophobicity and stability, while moderately unsaturated ones are amphiphilically balanced compounds both for emulsion texture and biological efficiency. This optimum formula provided greater softness, evenness and gloss along with inducing the greatest enhancement of hair health properties. These advantages facilitate the state-of-the-art development of multifunctional bean oil blends and optimized emulsifiers for next-generation cosmetic products. Future perspectives should include the conduct of clinical trials and comprehensive consumer perception studies to further validate both the efficacy and acceptability of these multifunctional bean oil blends in real-world cosmetic applications. Clinical translation will require robust safety and efficacy data in human populations, assessment of long-term tolerability, and comparison with existing commercial products. Additionally, from a sustainability perspective, the utilization of legume-derived oils (soybean, green bean, red bean) leverages agricultural byproducts and renewable plant resources, thereby aligning with eco-friendly cosmetic innovation strategies.
Acknowledgements
The research team would like to acknowledge with thanks the financial support from the Research Grant of Rajamangala University of Technology Krungthep, Thailand that has heavily facilitated this study in being accomplished.
Declaration of Generative AI in Scientific Writing
The authors maintain that during preparation of the manuscript all generative AI tools including language models were used only to enhance the overall coherence and readability of the English text. The manuscript was developed and analyzed by the authors, ensuring that the content is authentic and original.
CRediT Author Statement
Warongporn Rattanabun: Conceptualization, Validation Methodology, Supervision, Funding acquisition, and Writing – original draft. Wongnapa Nakyai: Data curation, Formal analysis, Investigation, Validation, and Visualization. Natnicha Phungsara: Data curation, Formal analysis, Investigation, Validation, and Visualization. Wannisa Keawbankrud: Methodology, Project administration, Resources, Software, Supervision, Validation, and Writing – review and editing.
References
[1] WC Byrdwell and RJ Goldschmidt. Fatty Acids of Ten Commonly Consumed Pulses. Molecules 2022; 27(21), 7260.
[2] R Rodríguez Madrera, A Campa Negrillo and JJ Ferreira Fernández. Fatty acids in dry beans (Phaseolus vulgaris L.): A contribution to their analysis and the characterization of a diversity panel. Foods 2024; 13(13), 2023.
[3] Y Sahasakul, A Aursalung, S Thangsiri, P Wongchang, P Sangkasa-Ad, A Wongpia, A Polpanit, W Inthachat, P Temviriyanukul and U Suttisansanee. Nutritional compositions, phenolic contents, and antioxidant potentials of ten original lineage beans in Thailand. Foods 2022; 11(14), 2062.
[4] S Wang, M Wu, Q Hu, C Jiang, S Wang, Y Yao and L Wang. Nutritional component analysis of mung bean (Vigna radiata L.). SABRAO Journal of Breeding and Genetics 2025; 57(3), 1050-1059.
[5] AB Özyiğit. Seed inoculation with Rhizobium japonicum bacteria improved fatty acid composition of different soybean (Glycine max L.) genotypes. Journal of King Saud University - Science 2024; 36(11), 103492.
[6] E Adewole, A Ojo, O Oludoro and I Osasona. Fatty acid profiles of Phaseolus species. Food Research 2021; 5(5), 131-135.
[7] A Singh, T Umeda and I Kobayashi. Formulation and characterization of soybean oil-in-water emulsions stabilized using gelatinized starch dispersions from plant sources. Molecules 2024; 29(9), 1923.
[8] MO Aremun, H Ibrahim and TO Bamidele. Physicochemical characteristics of the oils extracted from some Nigerian plant foods - a review. Chemical and Process Engineering Research 2015; 32, 36-52.
[9] FC Yang, Y Zhang and MC Rheinstädter. The structure of peopleʼs hair. PeerJ 2014; 2, 619.
[10] V Mysore and A Arghya. Hair oils: Indigenous knowledge revisited. International Journal of Trichology 2022; 14(3), 84-90.
[11] NH Lai, THN Dang, TT Nguyen and DTQ Phan. Utilizing lipid bond technology with molecular lipid complex to provide lipid treatment for damaged hair. Dermatology Research and Practice 2025; 2025, 5385312.
[12] SH Song, JH Lim, SK Son, J Choi, NG Kang and SM Lee. Prevention of lipid loss from hair by surface and internal modification. Scientific Reports 2019; 9(1), 9834.
[13] S Abdalla, MK Aroua and LT Gew. A comprehensive review of plant-based cosmetic Oils (Virgin Coconut oil, Olive oil, Argan oil, and Jojoba oil): Chemical and biological properties and their cosmeceutical applications. ACS Omega 2024; 9(44), 44019-44032.
[14] W Rattanabun, W Nakyai, N Phungsara, T Neamchuchuen and W Keawbankrud. Comparative antioxidant and anti-fungal (Malassezia furfur) activities of Glycine max L., Vigna radiata L., and Phaseolus vulgaris L.: Optimal active for anti-irritation efficacy and nanoemulsion-based formulation development. International Journal of Environmental Science 2025; 11(7), 603-615.
[15] Y Yamamoto, M Kumetani, Y Onuki, T Koide, T Suzuki and T Fukami. Analysis of the stability of external-application dermatologic preparations: Consideration from rheological measurements. Chemical & Pharmaceutical Bulletin 2016; 64(7), 1047-1055.
[16] MA Wazed, S Yasmin, P Basak, A Hossain, MM Rahman, MR Hassan, MM Khair and MN Khatun. Evaluation of physicochemical parameters of edible oils at room temperature and after heating at high temperature. Food Research 2023; 7(4), 91-100.
[17] H-Y Sung and KH Kim. Hair growth promotion with black soybean extracts: Case series. Journal of Pharmacopuncture 2022; 25(1), 63-67.
[18] D Sowdhanya, J Singh, P Rasane, S Kaur, J Kaur, S Ercisli and H Verma. Nutritional significance of velvet bean (Mucuna pruriens) and opportunities for its processing into value-added products. Journal of Agriculture and Food Research 2024; 15, 100921.
[19] MA van Rooijen and RP Mensink. Palmitic acid versus stearic acid: Effects of interesterification and intakes on cardiometabolic risk markers - a systematic review. Nutrients 2020; 12(3), 615.
[20] N Salih, J Salimon and E Yousif. The physicochemical and tribological properties of oleic acid based triester biolubricants. Industrial Crops and Products 2011; 34(1), 1089-1096.
[21] NB Patil, RS Adnaik and TT Shaikh. Swiss ADME predictions of pharmacokinetics and drug-likeness characteristics of secondary metabolites found in Glycyrrhiza glabra. International Journal of Science and Research Methodology 2025; 28(1), 16-24.
[22] DL Roman, M Roman, C Soni, M Schmutz, E Hernandez, P Wick, T Cassaniti, G Cavalli, V Ostafe and A Ivansari. Computational assessment of the pharmacological profiles of degradation products of chitosan. Frontiers in Bioengineering and Biotechnology 2019; 7, 00214.
[23] AS Pirzada, H Khan, W Alam, HW Darwish, AA Elhenawy, A Kuznetsov and M Daglia. Physicochemical properties, pharmacokinetic studies, DFT approach, and antioxidant activity of nitro and chloro indolinone derivatives. Frontiers in Chemistry 2024; 12, 1360719.
[24] PA Chimane, RS Adnaik and SN Ghule. Swiss ADME prediction of pharmacokinetics and drug-likeness properties of secondary metabolism present in Buchanania lanzan. Journal of Current Pharma Research 2025; 21(1), 1-9.
[25] TM Obuotor, AO Kolawole, OE Apalowo and AJ Akamo. Metabolic profiling, ADME pharmacokinetics, molecular docking studies and antibacterial potential of Phyllantus muellerianus leaves. Advances in Traditional Medicine 2023; 23(2), 427-442.
[26] A Rauf, H Khan, M Khan, A Abusharha, G Serdaroğlu and M Daglia. In silico, SwissADME, and DFT studies of newly synthesized oxindole derivatives followed by antioxidant studies. Journal of Chemistry 2023; 2023(1), 5553913.
[27] CJ Ononamadu and A Ibrahim. Molecular docking and prediction of ADME/drug-likeness properties of potentially active antidiabetic compounds isolated from aqueous-methanol extracts of Gymnema sylvestre and Combretum micranthum. Bio Technologia (Pozn) 2021; 102(1), 85-99.
[28] Y Staneva, I Iliev, S Georgieva and A Merdjanova. In silico prediction of physicochemical properties and drug-likeness of omega-3 fatty acids. Ovidius University Annals of Chemistry 2024; 35(2), 118-125.
[29] L Di, P Artursson, A Avdeef, GF Ecker, B Faller, H Fischer, JB Houston, M Kansy, EH Kerns, SD Krämer, H Lennernäs and K Sugano. Evidence-based approach to assess passive diffusion and carrier-mediated drug transport. Drug Discovery Today 2012; 17(15-16), 905-912.
[30] M Hanifah, M Jurfi. Formulation and stability testing of nanoemulsion lotion containing Centella asiatica extract. Journal of Young Pharmacists 2018; 10(4), 404-408.
[31] G Hemalatha, P Abhiram, P Tejeswari, M Pooja, Sravani and Shailaja. Nano-emulsion approach to enhance skin hydration: Formulation and evaluation of a hyaluronic acid moisturizing cream. International Research Journal of Modernization in Engineering Technology and Science 2025; 7(8), 2462-2467.
[32] B Iskandar, TW Liu, HC Mei, IC Kuo, MDC Surboyo, HM Lin and CK Lee. Herbal nanoemulsions in cosmetic science: A comprehensive review of design, preparation, formulation, and characterization. Journal of Food and Drug Analysis 2024; 32(4), 428-458.
[33] A Andonova, V Gugleva, Y Sotirova, K Nikolova, M Marcheva, Z Petkova and V Andonova. Influence of natural oils on the textural and rheological properties of cosmetic creams. Pharmacia 2024; 71, 138613.
[34] J Stabrauskiene, A Mazurkevičiūtė, D Majiene, R Balanaskiene and J Bernatoniene. Development and evaluation of an anti-inflammatory emulsion: Skin penetration, physicochemical properties, and fibroblast viability assessment. Pharmaceutics 2025; 17(7), 933.
[35] S Jiamphun and W Chaiyana. Enhancing skin delivery and stability of vanillic and ferulic acids in aqueous enzymatically extracted glutinous rice husk by nanostructured lipid carriers. Pharmaceutics 2023; 15(7), 1961.
[36] YM Navarro-Pérez, E Cedeño-Linares, O Norman-Montenegro, V Rau-Sanjuan, Y Mondeja-Rivera, AM Hernández-Monzón and MM González-Bedía. Prediction of the physical stability and quality of O/W cosmetic emulsions using full factorial design. Journal of Pharmacy & Pharmacognosy Research 2021; 9(1), 98-112.
[37] T Xiao, X Ma, H Hu, F Xiang, X Zhang, Y Zheng, H Dong, B Adhikari, Q Wang and A Shi. Advances in emulsion stability: A review on mechanisms, role of emulsifiers, and applications in food. Food Chemistry: X 2025; 29, 102792.
[38] M Estanqueiro, J Conceição, MH Amaral, D Santos, JB Silva and JMS Lobo. Characterization and stability studies of emulsion systems containing pumice. Brazilian Journal of Pharmaceutical Sciences 2014; 50(2), 361-370.
[39] M Shafiei, Y Kazemzadeh, DA Martyushev, Z Dai and M Riazi. Effect of chemicals on the phase and viscosity behavior of water in oil emulsions. Scientific Reports 2023; 13, 4100.
[40] Y Yang, S Yan, B Yu, C Gao, K Chang and J Wang. Lamellar gel containing emulsions as an effective carrier for stabilization and transdermal delivery of retinyl propionate. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2023; 674, 131834.
[41] Z Hua, C Zhang, J He, Y Chen, S Zhang, J Sun, N Wang, J Zhang, X Yang, J Chen and X Wei. Ultra-micro liquid crystal in cosmetic emulsion with excellent moisture retention and delivery selectivity. ACS Omega 2024; 9(47), 47194-47202.
[42] V Teeranachaideekul, S Soontaranon, S Sukasem, D Chantasart and A Wongrakpanich. Influence of the emulsifier on nanostructure and clinical application of liquid crystalline emulsions. Scientific Reports 2023; 13(1), 4185.
[43] W Zhang and L Liu. Study on the formation and properties of liquid crystal emulsion in cosmetic. Journal of Cosmetics, Dermatological Sciences and Applications 2013; 3(2), 139-144.
[44] W Gu, L Gu, N Tao, X Wang and C Xu. Composite fish collagen peptide-based biopolymer emulsion for keratin structure stabilization and hair fiber repair. Polymers 2025; 17(7), 907.
[45] F Zamiri, H Rakhshandeh, B Kiafar, SM Naqvi, M Emadzadeh, S Fakhraei and M Maleki. Assessment of the effects of bean extract on axillary hair reduction. Avicenna Journal of Phytomedicine 2024; 14(2), 268-277.
[46] C Fernandes, B Medronho, L Alves and MG Rasteiro. On hair care physicochemistry: From structure and degradation to novel biobased conditioning agents. Polymers 2023; 15(3), 608.
[47] MFRG Dias. Hair cosmetics: An overview. International Journal of Trichology 2015; 7(1), 2-15.