
Trends
Sci.
2026;
23(5):
11985
Innovative Polyherbal Nanoemulsion for Cardioprotection in Diabetes:
Impact on Cardiac Biomarker
Rafiastiana Capritasari1,2, Akrom1,*, Arif Budi Setianto1 and Titiek Hidayati3
1Doctoral Program of Pharmacy Sciences, Faculty of Pharmacy, Universitas Ahmad Dahlan, Yogyakarta, Indonesia
2Department of Pharmacy, Faculty of Health Sciences, Universitas Muhammadiyah Magelang, Central Java, Indonesia
3Department of Epidemiology, Family Medicine, and Community Medicine, Faculty of Medical and Health Sciences,
Universitas Muhammadiyah Yogyakarta, Yogyakarta, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 21 September 2025, Revised: 22 October 2025, Accepted: 5 November 2025, Published: 5 January 2026
Abstract
Diabetes mellitus is a chronic metabolic disease that increases the risk of cardiovascular disease, which is the leading cause of death in people with diabetes. One of the main mechanisms contributing to cardiovascular complications in diabetes is an increase in cardiac enzyme levels, such as ACE, NO, CK-MB, LDH, and liver enzymes SGOT and SGPT. This study aims to explore the cardioprotective potential of a novel polyherbal nanoemulsion formulation containing Nigella sativa oil, Centella asiatica, and Moringa oleifera in streptozotocin (STZ)-induced diabetic rats. The focus of this study was to measure the impact of nanoemulsion therapy on cardiac biomarker levels, namely ACE, NO, CK-MB, LDH, SGOT, and SGPT, which play an important role in monitoring heart function in diabetes. The results showed that administration of polyherbal nanoemulsion significantly reduced the levels of these cardiac enzymes, including ACE, NO, CK-MB, LDH, SGOT, and SGPT, compared to the positive control group that was only given STZ. Therapy with higher doses of polyherbal nanoemulsion showed better therapeutic effects, indicating that nanoemulsion technology can increase the bioavailability and therapeutic efficacy of bioactive compounds from these plants in protecting the heart. These findings suggest that polyherbal nanoemulsion may be a promising therapeutic approach for reducing the risk of cardiovascular complications in diabetic patients, particularly through the regulation of cardiac biomarkers associated with heart damage.
Keywords: Biomarker cardiotoxic, Cardiac enzyme, Nanoemulsion polyherbal, Diabetic rat, Cardioprotective, Streptozotocin-induced cardiotoxic, Nanoencapsulation
Introduction
Diabetes mellitus is a chronic metabolic disease characterized by hyperglycemia due to impaired insulin secretion, insulin action, or both [1]. Diabetes mellitus significantly increases the risk of cardiovascular disease, which is the leading cause of mortality and morbidity in diabetic patients [2]. Chronic hyperglycemia, which is a major characteristic of diabetes mellitus, not only affects glucose metabolism but also has a detrimental effect on various organs of the body, one of which is the heart. One of the most significant complications of diabetes related to heart disorders is diabetic cardiomyopathy, a condition that causes damage to the heart muscle without classic ischemia (as in coronary heart disease). In diabetic cardiomyopathy, myocardial damage is caused by oxidative stress, chronic inflammation, and fibrosis formation, which ultimately leads to heart failure [3]. When blood glucose levels remain high, the body experiences increased production of free radicals, known as oxidative stress These free radicals attack heart cells, particularly heart muscle cells (myocytes), causing damage to cell membranes, proteins, and DNA. This ongoing cellular damage leads to impaired cardiac contractility, thereby affecting the heart’s ability to pump blood efficiently [4]. In addition, chronic hyperglycemia also activates systemic inflammation. This condition causes the release of various pro-inflammatory cytokines, such as TNF-α and IL-6, which further exacerbate myocardial damage. This prolonged inflammation triggers changes in the structure of the myocardium, where healthy tissue is replaced by fibrosis, the process of scar tissue formation that replaces damaged muscle tissue. This fibrosis reduces myocardial elasticity, worsens the heart’s ability to contract, and causes ventricular dysfunction, which contributes to the development of heart failure [5].
The increase in the prevalence of diabetes is closely related to the increase in the incidence of heart failure among diabetic patients. Studies show that heart failure in diabetic patients has a much higher incidence rate than in the non-diabetic population. Based on estimates from the International Diabetes Federation (IDF) that 2.7 million people have been diagnosed with Type 2 diabetes (T2DM) and assuming a complication rate of 55% [6]. In Indonesia, data shows that 57% of the population experiences diabetes-related complications. Most of the population involved in this study was between 51 and 70 years old and lived in Java. The most common complication, found in about 24% of cases, was cardiovascular disease [7]. In 2019, Indonesia had 10.7 million people with diabetes, making it one of the highest prevalence rates in the world. It is estimated that this number will increase to 16.6 million by 2045. Diabetes was also one of the leading causes of death in Indonesia in 2021 [8]. The main factor contributing to this is diabetic cardiomyopathy, in which damage to the myocardium caused by chronic hyperglycemia leads to disturbances in the structure and function of the heart.
Diabetes, especially when poorly controlled, increases the prevalence of heart failure, and diabetic cardiomyopathy is one of the main factors contributing to this condition. Therefore, a better understanding of diabetic cardiomyopathy, including the underlying mechanisms of oxidative stress, inflammation, and myocardial fibrosis, is essential in the prevention and treatment of this disease [9].
Uncontrolled diabetes increases the risk of ischemic stroke [10]. Prolonged hyperglycemia can cause sympathetic nervous system dysregulation, which leads to increased blood pressure through increased renin-angiotensin-aldosterone secretion [11]. One of the mechanisms involved in cardiovascular complications in diabetes is an increase in cardiac enzyme levels such as angiotensin-converting enzyme (ACE), creatine kinase myocardial band (CKMB), lactate dehydrogenase (LDH), as well as liver transaminases SGOT (serum glutamic-oxaloacetic transaminase) and SGPT (serum glutamic-pyruvic transaminase) [12]. Elevated levels of these enzymes may indicate damage to heart and liver tissue associated with the progression of heart disease in diabetic patients [13].
Cardiovascular disease is one of the serious complications that often occur in patients with uncontrolled diabetes [14]. Chronically elevated blood sugar levels can damage blood vessels and increase the risk of various heart conditions, such as hypertension, stroke, and coronary heart disease [15]. Conventional treatment with antihypertensive medications like telmisartan has proven effective in controlling blood pressure in diabetic patients [16], but additional efforts are still needed to achieve comprehensive recovery. In this context, the use of herbal supplements as an adjunct to conventional therapy may offer a promising option, aimed at improving heart health, enhancing blood circulation, and reducing inflammation, a high-risk factor for cardiovascular disease in patients with uncontrolled diabetes. However, the use of plant-based therapies has great potential as a safer and more effective alternative. One promising approach is the use of herbal nanoemulsion, which can enhance the bioavailability and efficacy of bioactive compounds from medicinal plants. Nanoemulsion are nanotechnology-based delivery systems that combine the benefits of emulsions with extremely small particle sizes, enabling better penetration into body tissues and enhancing their therapeutic effects [17].
Several herbal plants containing polyphenols, flavonoids, and lycopene have potential as herbal medicines that can prevent atherosclerosis and act as cardioprotective agents due to their anti-inflammatory and antioxidant effects [18]. Several terpenoid and polyphenolic compounds, including flavonoids, hydrolysable tannins, xanthones, procyanidins, and caffeoylquinic acid derivatives, are effective natural ACE inhibitors. Flavonoids have the ability to scavenge free radicals and inhibit lipid oxidation [19]. Moringa Oleifera contains Moringin, which has antioxidant properties [20]. Centella asiatica contains triterpenoids such as asiaticosides [21], which has cardioprotective properties. In addition, Nigella sativa oil contains linoleic acid and thymoquinone, which have anti-inflammatory and immunomodulatory properties [22]. A combination of Nigella sativa, Centella asiatica, and Moringa oleifera has been previously studied for its antioxidant and hepatoprotective effects. These 3 herbs were found to reduce SGOT, SGPT, and malondialdehyde (MDA) levels in rats induced with alloxan [23].
This study aims to explore the cardioprotective potential of a mixed herbal nanoemulsion preparation containing extracts from these 3 plants in a diabetic rat model. This study will assess its impact on the levels of key cardiac enzymes, namely ACE, CKMB, LDH, SGOT, and SGPT, which play an important role in monitoring heart and liver function in diabetic conditions. The focus on using nanoemulsion in this study is to enhance the stability and bioavailability of the bioactive compounds contained in the 3 herbal plants, which can enhance their therapeutic potential and strengthen their cardioprotective effects.
Thus, this study is expected to contribute significantly to the development of more effective and safer herbal-based alternative therapies for treating cardiovascular complications in diabetic patients, as well as opening opportunities for the development of nanotechnology-based therapies in the fields of pharmacy and cardiovascular health.
Materials and methods
Ethical committee approval
Before conducting experiments with animals, ethical approval was obtained to ensure that the research complied with appropriate animal welfare standards. The animal ethics approval process was carried out by the Ahmad Dahlan University Research Ethics Committee, and the research received official approval with ethics permit number No.012407168. This approval ensures that the study complies with ethical guidelines for the use of animals in research and that the welfare of the test animals is protected throughout the research.
Animal
A total of 35 male Sprague Dawley (SD) rats, aged approximately 5 - 6 months, weighing 200 - 300 g, were adapted to standard laboratory animal care conditions and fed and watered ad libitum. These test animals were obtained from the breeding facility of Taufik Rat and Mice in Bogor, West Java, Indonesia, accompanied by animal health certificate No. 335/TRM/SK/XI/2024. After the adaptation period was completed, the rats were divided into 7 treatment groups (I, II, III, IV, V, VI and VII), each consisting of 5 rats. The mice were acclimatized for 7 days to ensure that they adapted to the new environment and minimized potential stress from environmental changes. During the acclimatization period, they received normal food and had free access to drinking water. Rats were housed in a controlled environment with regulated lighting (12/12 h light/dark cycle), temperature (22 ± 1 °C), and humidity (55 ± 5%) and provided free access to a standard pellet diet and water ad libitum. One week before to the research, the animals were acclimated [24]. Sample size calculation was based on the Degree of Freedom formula, with a minimum of 5 rats for each group.
Chemicals
Streptozotocin (sigma, St.Louis, USA) was employed as diabetic models. Elisa kit ACE (Reed Biotech with catalog number RE3201), CK MB enzyme (DiaSys, LOT 33695), LDH enzyme (DiaSys, LOT 35100), SGOT (DiaSys, LOT 10523003) and SGPT (DiaSys, LOT 10623005), Glucose reagent (Stanbio, 1071-250), NO (Griess reagent LOT SLCH9583).
Experimental design
The rats were divided into 7 groups, i.e:
Group I (Normal control): Healthy male SD rats without treatment.
Group II (Positive control): Male SD rats induced with Streptozotocin 45 mg/kg of body weight single dose intraperitoneal (i.p)
Group III (Standard): Male SD rats induced with Streptozotocin 45 mg/kg of body weight single dose and Telmisartan 10 mg/kg of body weight per oral (p.o) for 2 weeks.
Group IV (Treatment-NE100): Male SD rats induced with Streptozotocin 45 mg/kg of body weight single dose and polyherbal nanoemulsion 100 mg/kg body weight per oral (p.o) for 2 weeks.
Group V (Treatment-NE200): Male SD rats induced with Streptozotocin 45 mg/kg of body weight single dose and polyherbal nanoemulsion 200 mg/kg body weight per oral (p.o) for 2 weeks.
Group VI (Treatment-NE300): Male SD rats induced with Streptozotocin 45 mg/kg of body weight single dose and polyherbal nanoemulsion 300 mg/kg body weight per oral (p.o) for 2 weeks.
Group VII (Treatment-P200): Male SD rats induced with Streptozotocin 45 mg/kg of body weight single dose and polyherbal 200 mg/kg body weight per oral (p.o) for 2 weeks.
Male SD rats were sacrificed under anesthesia using gas CO2. Blood samples were collected for further analysis.
Blood sampling and serum isolation
Blood samples were collected from the orbital vein at room temperature. Samples were left to clot for 30 min and centrifuged g for 15 min. the serum was separated and stored at temperature −80 °C for quantitative determination of biomarker cardiac enzyme. Serum ACE levels were measured using an enzyme-linked immunosorbent assay (ELISA) kit, and plates were analyzed using an automated plate reader (ThermoFisher Scientific, Varioskan ALF). Serum NO, CK MB, LDH, SGOT and SGPT were measured using colorimetry method.
Statistical analysis
All experiments data was presented as mean± standard deviation (SD). SPSS versions 27.0 for Windows were used to conduct the statistical analysis. All experiments data count were test for normality using Shapiro-Wilk test, and homogeneity using Levene Test. Data normal and homogeny distributions were analyzed using the One-Way ANOVA. If the data shows a difference (p < 0.05), continue with the post-Hoc LSD test to determine the significant differences between groups.
Results and discussion
Formulation of polyherbal nanoemulsion
This study presents an innovative polyherbal nanoemulsion formulation developed exclusively for this research. Prior to this main test, we conducted a series of preliminary assessments of the test active ingredient compound’s characteristics and formula optimization, with the following results: Particle size: 115.463 ± 9.563 nm, PDI: 0.490 ± 0.050, zeta potential: −29.737 ± 1.665 mV, supported by Transmission Electron Microscopy (TEM) measurements [25].
Animal model cardiotoxic
The modeling of cardiotoxic test animals can be described based on the data results provided above, which focus on the effect of STZ (Streptozotocin) dosage on 2 main parameters, namely GDS (Daily Blood Glucose) and CK-MB (Creatine Kinase-Myocardial Band). These 2 parameters are used to evaluate the cardiotoxic effects of a compound on test animals, in this case mice. The results of the preliminary test animal modeling are presented in Figure 1.
The results of the One Way ANOVA with a p-value of 0.0004 (Blood Glucose) and a p-value of 0.0001 (CKMB) indicate that there is a statistically significant difference between the measurement time groups, suggesting that the timing of STZ administration consistently affects blood glucose levels in rats. This shows that STZ administration not only increases blood glucose levels but also has a significant impact on heart muscle damage, which can be detected through CK-MB levels.
This reinforces the hypothesis that STZ can cause a measurable increase in blood glucose at various times after administration. This hyperglycemic effect is a potential indicator of broader cardiotoxicity, leading to increased metabolic stress and possible impairment of heart function. Hyperglycemia can directly affect cardiovascular function through increased blood glucose levels, which can cause metabolic changes in the heart and increase the risk of heart muscle damage or other heart disorders [26].
With the variability observed between individual mice (as seen from the larger SD at certain times), this model also shows that the response to STZ can vary between individuals, which may be related to differences in metabolism or the body’s capacity to cope with toxic effects. The administration of STZ at a dose of 45 mg/kg BW aims to cause metabolic disorders, particularly in liver, kidney, or cardiovascular system function, as well as heart damage or cardiotoxic disorders. STZ is a compound known for its ability to induce diabetes in test animals, particularly in mice, by damaging the pancreatic β cells responsible for insulin production [27]. Therefore, this model is often used to study the effects of diabetes or metabolic disorders on heart function and other organs. Intraperitoneal administration of STZ allows the compound to be rapidly absorbed into the blood circulation of the test animal. A dose of 45 mg/kg body weight is commonly used in animal diabetes models, where STZ is expected to damage the pancreas within a short period of time and cause hyperglycemia (high blood glucose levels) (Figure 1), as well as other effects on the heart and related organs.
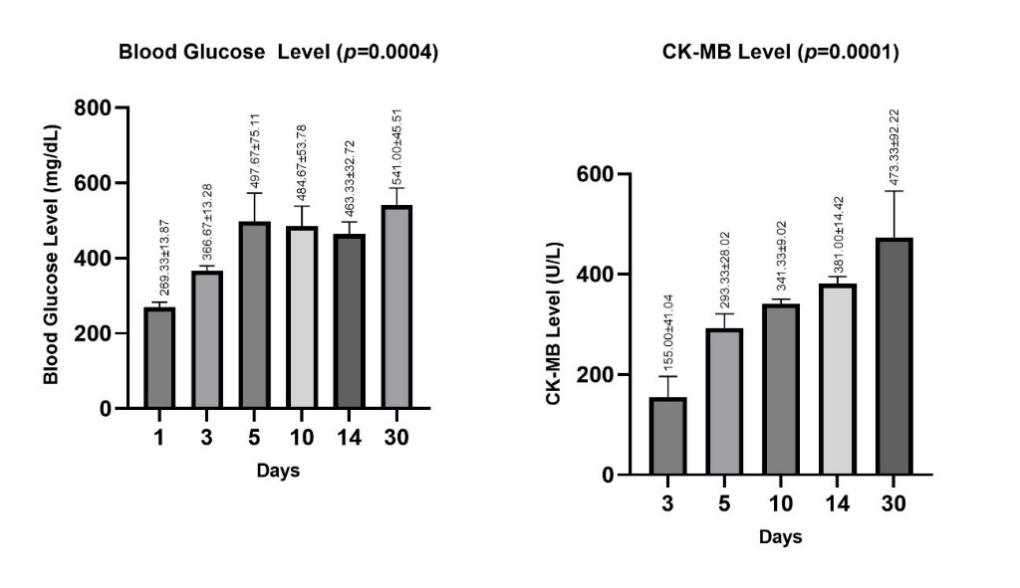
Figure 1 The results of the preliminary test animal modeling.
STZ has a fairly complex mechanism of action, which causes damage to body cells, especially pancreatic β cells, through several pathways, including: DNA damage: STZ induces the formation of free radicals that damage pancreatic β cell DNA [28]. Oxidative stress: This process causes disturbances in the body’s antioxidant balance, which in turn can cause damage to other tissues and organs, including the heart [29]. Increased glucose levels: Damage to the pancreas causes a decrease in insulin production, leading to hyperglycemia, which can affect the metabolism and function of various organs, including the heart [30].
Hyperglycemia can directly influence cardiovascular function through various critical biochemical and physiological mechanisms: 1) Oxidative Stress: Prolonged elevated glucose levels result in endothelial cells within blood vessels encountering an abundance of substrate for glucose metabolism. This results in heightened production of reactive oxygen species (ROS) free radicals within the mitochondria. Reactive oxygen species (ROS) compromise the lipid membranes, proteins, and DNA of endothelial cells, thereby impairing endothelial function and ultimately diminishing the release of nitric oxide (NO), a crucial vasodilator. The effect is an elevation in vasoconstriction, resulting in impaired blood flow and a heightened risk of hypertension [31]. 2) Endothelial dysfunction: Elevated glucose levels induce non-enzymatic glycation of endothelial proteins, leading to the formation of Advanced Glycation End Products (AGEs). AGEs interact with RAGE receptors on endothelial cells, initiating chronic inflammation and enhancing vascular permeability. Endothel loses its capacity to regulate the equilibrium between vasodilators (nitric oxide) and vasoconstrictors (endothelin-1). The consequence is that the vascular walls become rigid, the reaction to blood pressure is compromised, and the heart’s perfusion response diminishes [32]. 3) Alterations in myocardial metabolism: The heart typically utilizes fatty acids and glucose as energy substrates. Under hyperglycemic conditions, myocardial cells exhibit insulin resistance, hindering glucose utilization. The heart predominantly utilizes fatty acid oxidation, which is less efficient and generates a greater amount of reactive oxygen species (ROS). Consequently, the heart’s operational efficiency diminishes, potentially leading to myocardial contractile dysfunction (diabetic cardiomyopathy) [33,34]. 4) Activation of pro-inflammatory and coagulation pathways: Chronic hyperglycemia induces the activation of protein kinase C (PKC), resulting in enhanced expression of adhesion molecules (VCAM-1, ICAM-1) that recruit leukocytes. Inflammatory cytokines, including TNF-alpha and IL-6, induce vasodilation. The coagulation pathway elevates the risk of thrombosis and coronary artery occlusion. The consequence is an elevated risk of coronary heart disease, stroke, and microcirculatory disorders [35].
Cardiotoxic effects can be observed after STZ administration, particularly in terms of heart tissue damage, which is often characterized by an increase in cardiac enzymes such as CK-MB (Creatine Kinase Myocardial Band) [36]. Some potential cardiotoxic effects include Heart Contraction Disorders: Chronic hyperglycemia can interfere with heart function and affect heart muscle contraction [37]. Increased Cardiac Enzymes: Elevated levels of enzymes such as CK-MB in the blood may indicate damage to heart muscle cells, which is a sign of myocardial infarction or cardiac stress [38]. Changes in Heart Structure: Morphological changes in heart tissue can also be observed through histological examination, which shows inflammation, cell degeneration, or fibrosis [39].
This model provides insight into the impact of STZ on the metabolic and cardiovascular systems and demonstrates its potential as an agent capable of causing clear cardiotoxic damage, which can be used for further studies on toxic effects on the heart and metabolism.
Blood glucose levels, blood pressure levels, and CK-MB levels
Five days after STZ 45 mg/kg of body weight induction, the average blood glucose, blood pressure and CK MB levels were obtained in the normal control and positive control that had received STZ induction (Table 1). In rats, an intravenous dosage of 25 - 100 mg/kg of STZ has been shown to induce dose-dependent hyperglycemia [40]. STZ (45 mg/kg BW, i.p) injection after 4 weeks of a high-fat diet resulted in hyperglycemia with the highest success rate [41].
Intravenous administration of STZ at doses between 25 and 100 mg/kg has been empirically shown to elicit dose-dependent hyperglycemia in mice. As STZ systematically destroys pancreatic beta cells based on the dosage given, this happens, causing insulin production problems to get worse and blood glucose levels to rise with higher doses [42].
Table 1 Results of blood glucose, blood pressure and CK-MB levels after STZ Induction at 5 days.
Group |
Parameter |
|||
Blood Glucose Levels (mg/dL) |
Blood Pressure Levels (mmHg) |
CK-MB Levels (U/L) |
||
Systolic |
Diastolic |
|||
Normal Control |
137.87 ± 14.49** |
96.61 ± 5.03* |
171,69 ± 8,06* |
|
Positive Control |
380.4 ± 8.79* |
156.2 ± 18.71** |
119.76 ± 12.34* |
488,61 ± 6,98* |
*Superscripts in the same row (normal and positive control) indicate significant differences (p < 0.05), **Superscripts indicate not significant differences (p > 0.05).
In the normal group, blood glucose levels, blood pressure, and CK-MB enzyme activity were within the normal physiological range, reflecting stable metabolic and cardiovascular conditions. In contrast, the streptozotocin-induced group (positive control) showed a significant increase in blood glucose levels compared to the normal group (normal control), this is consistent with the expected results, as streptozotocin is known to be a compound that damages pancreatic β cells, causing disruption in insulin production and ultimately increasing blood glucose levels [43]. The normal blood glucose levels in this study, consistent with the report by Nazaruddin et al. [44] on male Sprague Dawley rats which was 118.4 ± 1.14 mg/dL, while hyperglycemic rats had blood glucose levels > 200 mg/dL [45].
The streptozotocin-induced group (positive control) exhibited an inclination for elevated blood pressure relative to the control group. STZ preferentially penetrates beta cells through the glucose transporter GLUT2. STZ causes DNA damage, makes free radicals, and kills beta cells (necrosis or apoptosis). Insulin production from the pancreas is greatly reduced because the beta cells do not work properly. Insulin lowers blood sugar levels by helping the body’s cells take in glucose. Lower insulin levels make it harder for the body to use glucose properly, which causes it to build up in the blood (hyperglycemia). The significant increase in blood glucose levels in the STZ-induced group is due to damage to pancreatic beta cells and a decrease in insulin production, which makes it harder to control glucose levels [42]. This finding confirms that STZ is effectively utilized to induce hyperglycemic or experimental diabetes conditions in animal models.
Chronic hyperglycemia occurring in the induction group can cause endothelial dysfunction, disturbances in vascular tone regulation, and increased peripheral resistance, which can indirectly affect blood pressure. Although streptozotocin (STZ) can cause significant metabolic disturbances, such as increased blood glucose levels, its effects on blood pressure can vary depending on the severity of diabetes, the duration of hyperglycemia, and the individual’s response to the condition. In diabetes, especially in the early stages or in mild hyperglycemia, changes in blood pressure may not be immediately apparent in systolic parameters. Systolic blood pressure is more influenced by factors such as blood vessel stiffness, blood volume, and heart function, which may not yet be significantly impaired in the early stages of diabetes induction [46]. The diastolic mechanism that causes a significant increase due to hyperglycemia in diabetes mellitus is related to several factors that affect myocardial contraction and relaxation. Hyperglycemia in diabetes mellitus leads to the accelerated accumulation of advanced glycation end products (AGEs) in the myocardium [47].
Hyperglycemia in diabetes mellitus can significantly increase the synthesis of advanced glycation end products (AGEs). AGEs are harmful chemicals that form when glucose (or other reducing sugars) reacts with proteins, lipids, or nucleic acids without the help of enzymes. Chronic hyperglycemia leads to increased glucose concentrations in the blood and tissues. Glucose interacts with the free amino groups of proteins to form Schiff bases, which then turn into Amadori products. After that, the product goes through a series of oxidative and dehydration steps that make AGEs. Higher blood sugar levels speed up the production of advanced glycation end products (AGEs). This process, which does not need enzymes and depends on the amount of glucose, is sped up a lot by hyperglycemia in the making of AGEs. Advanced glycation end products (AGEs) cause proteins to become permanently damaged, especially those with long lifespans like collagen. AGEs can bind to their receptors (RAGE) on cell surfaces, which starts pathways for inflammation and oxidative stress [48]. This causes problems with diabetes, such as nephropathy [48], retinopathy [49], and neuropathy [47].
These AGEs bind to AGE receptors (RAGE) on myocardial cells, activating inflammatory and fibrotic pathways. As a result, myocardial stiffness increases, reducing the ability of the left ventricle (LV) to perform normal filling during the diastolic phase. This leads to an increase in end-diastolic pressure (LV-PED), which indicates left ventricular stiffness [50]. These results are consistent with previous research findings showing that diabetes can increase the risk of hypertension due to disturbances in the cardiovascular system [51]. In CK-MB enzyme activity measurements, the streptozotocin-induced group (positive control) showed a more significant increase compared to the control group (normal control). This increase indicates myocardial stress or damage, which can be caused by metabolic disorders associated with diabetes. In conditions of uncontrolled hyperglycemia, oxidative stress and inflammation in the cardiovascular system can contribute to increased CK-MB activity, which is a marker of heart muscle damage [52].
Cardiotoxic prolonged hyperglycemia can lead to dysregulation of the sympathetic nervous system, causing increased blood pressure thru increased renin-angiotensin-aldosterone excretion [11]. Recent findings regarding the G-protein-coupled receptor (GPR91) activated by succinate, an intermediate product of the citric acid cycle, are associated with high glucose levels and renin release from the juxtaglomerular apparatus (JGA) in the kidneys [53]. GPR91 is a metabolic receptor expressed in the kidneys that can cause the renin-angiotensin-aldosterone system (RAAS) activation and increase systemic blood pressure. Its ligand, succinate, is an intermediate product in the citric acid cycle that causes renin release from the JGA. High glucose levels and succinate-induced GPR91 activation trigger paracrine signals from the juxtaglomerular endothelium, which produces renin to increase renin synthesis and release, a step in RAS activation. Elements of the signal transduction pathway involve succinate and increased endothelial vascular Ca2+ ions, which are GPR91-dependent, such as the synthesis and release of NO and PGE2, a mediator for renin release. The direct production of endothelial NO and prostaglandins directly causes vasodilation of the afferent arterioles, which is important in the development of glomerular hypertrophy [43]. RAAS also plays an important role in regulating the mechanical properties of arterial elasticity and increasing collagen synthesis [54]. The RAAS particularly angiotensin II, plays a crucial role in hemodynamic changes within the kidneys. Angiotensin II can increase renal sodium reabsorption by affecting the proximal tubules and stimulating aldosterone secretion. Angiotensin II interacts with 2 specific receptors: The angiotensin II type 1 receptor (AT1R) and the angiotensin II type 2 (AT2R) [55].
High blood glucose levels are associated with increased expression of Transforming Growth Factor β (TGF-β) and increased production of extracellular matrix. TGF-β is closely linked to RAAS activation [55]. TGF-β plays a crucial role in myocardial fibrosis by inducing the production and secretion of collagen mediated by angiotensin II. Excessive expression of TGF-β can contribute to the pathogenesis of fibrosis, leading to remodeling or cardiac hypertrophy, which ultimately results in heart failure [56]. RAAS antagonists such as ACE inhibitors and angiotensin receptor blockers can weaken fibrotic changes mediated by TGF-β [57].
Changing how things spread TGF-β (factor-beta) is a cytokine that has many functions, including controlling cell growth, differentiation, immunomodulation, and tissue fibrosis. In the setting of chronic hyperglycemia in diabetes mellitus, the synthesis of TGF-β, particularly TGF-β1, is increased, resulting in the development of microvascular and macrovascular complications. Hyperglycemia can directly activate intracellular signaling pathways, such as protein kinase C (PKC), mitogen-activated protein kinase (MAPK), and oxidative stress pathways. This pathway’s activation causes the TGF-β1 gene to be made in many types of cells, such as fibroblasts, renal mesangial cells, and endothelial cells. Higher levels of TGF-β will speed up the production of collagen, fibronectin, and other proteins that make up the extracellular matrix, while also slowing down their breakdown [58,59]. This process leads to tissue fibrosis, as evidenced in diabetic nephropathy [60], diabetic cardiomyopathy [61], and diabetic retinopathy [49].
Biomarker of cardio
Cardioprotective activity testing aims to evaluate the ability of a compound to protect the heart from damage caused by various factors, including oxidative stress, hypoxia, or myocardial infarction. One approach to measuring cardioprotective activity is to monitor changes in the levels of enzymes involved in heart damage. ACE, NO, CK-MB, LDH, SGOT, and SGPT enzymes are the primary biomarkers used in this testing (data measurements are presented in Figure 2).
In the ACE level measurement results, it was observed that the healthy group showed lower ACE levels compared to the other treatment groups, with a value of 36.45 ± 7.36 ng/mL. In group II, which was given 45 mg/kg of body weight STZ induction, higher ACE levels were observed (116.92 ± 14.84 ng/mL). This increase in ACE levels is caused by damage or physiological changes resulting from STZ treatment, which is intended to induce diabetic conditions in the test animals. In Group IV, which received 100 mg/kg polyherbal nanoemulsion treatment after STZ induction, ACE levels were higher than in Group III, which received Telmisartan 10 mg/kg of body weight, but lower than in Group II. This indicates that polyherbal nanoemulsion administration has an effect in reducing ACE levels in the serum of STZ-induced test animals. Group V (200 mg/kg polyherbal nanoemulsion) showed lower ACE levels compared to groups II and IV, while group VI (300 mg/kg polyherbal nanoemulsion) showed lower ACE levels compared to the other groups. This indicates that higher doses of polyherbal nanoemulsion have a therapeutic effect in lowering ACE levels, which is associated with cardiac protective effects through the renin-angiotensin system mechanism.
The increase in ACE levels observed after STZ treatment is closely associated with the physiological harm inflicted by STZ, notably the impairment of pancreatic beta cells and the systemic repercussions of hyperglycemia. STZ selectively targets pancreatic beta cells, inhibiting insulin production and leading to chronic hyperglycemia. Hyperglycemia induces oxidative stress, inflammation, and endothelial dysfunction, resulting in damage to the cardiovascular and renal systems. The RAAS turns on to make up for metabolic stress situations like diabetes. ACE, one of its parts, changes angiotensin I into angiotensin II, a peptide that makes blood vessels constrict and causes inflammation and fibrosis. Higher levels of ACE are a sign that the body is not working properly because of tissue damage and stress caused by diabetes. Hyperglycemia also activates endothelial and renal tubular cells, which makes ACE more active both in and out of the body [62].
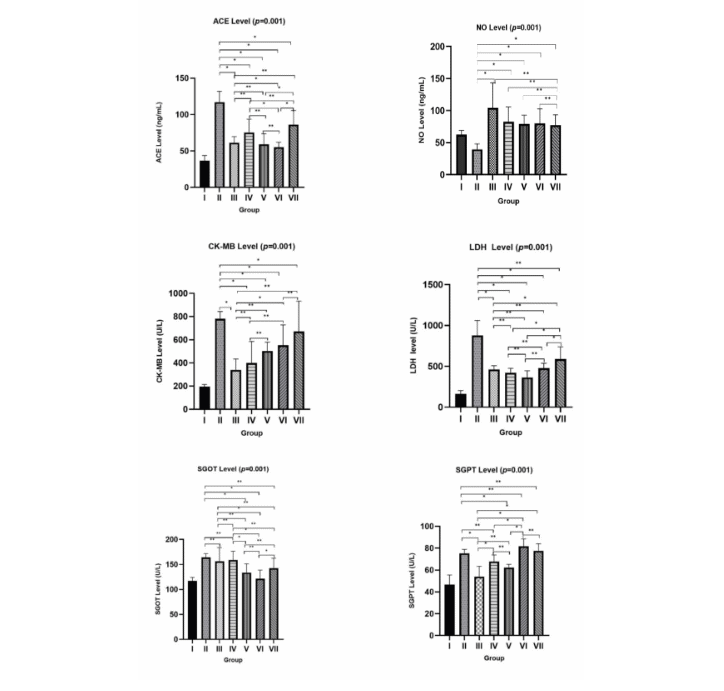
Figure 2 Cardiac marker Levels across different treatment groups. Bars represent mean ± SD of Cardiac marker count in serum from 7 experimental groups. Group I (Normal Control), II (Positive Control), III (Standard Telmisartan 10 mg/kg BW), IV (Polyherbal Nanoemulsion 100 mg/kg BW rats), V (Polyherbal Nanoemulsion 200 mg/kg BW rats), VI (Polyherbal Nanoemulsion 300 mg/kg BW rats), and VII (Polyherbal 200 mg/kg BW rats). Statistical significance was analyzed with a p-value of 0.001. Superscripts (*) denote statistically significant differences between groups, with each letter indicating distinct groupings (p < 0.05). Superscripts (**) denote statistically not significant differences between groups, with each letter indicating distinct groupings (p > 0.05).
ACE and AT1R inhibition increase basal NO release and coronary or arm blood flow response to acetylcholine. The mechanism begins with the emergence of risk factors such as age, family history, metabolic syndrome, diabetes mellitus, dyslipidemia, obesity, microalbuminuria, smoking, and stress, which lead to increased vascular resistance. This leads to a decrease in blood flow to the kidneys, causing the kidneys to stimulate the release of the enzyme renin stored in the juxtaglomerular cells of the kidneys to convert Angiotensinogen (renin substrate) into Angiotensin I (an inactive decapeptide) [63].
Several pathological mechanisms, including chronic hyperglycemia, activation of the renin-angiotensin system, and elevated ACE levels, as observed in STZ-induced diabetes mellitus models, lead to diminished renal blood flow. In situations of metabolic stress, like diabetes, the RAAS system is turned on, which raises the levels of angiotensin II. Angiotensin II is a strong vasoconstrictor, especially in the kidney’s afferent and efferent arterioles. This vasoconstriction makes the blood vessels in the kidneys more resistant, which slows down blood flow to the glomerulus. ACE changes angiotensin I into angiotensin II, which makes blood vessels constrict more. The rise in ACE caused by diabetes makes this effect stronger, making the drop in renal perfusion even worse.
Then, this angiotensin I will be converted by Angiotensin Converting Enzyme (ACE), which is a dipeptidyl carboxypeptidase that cleaves histidyl-leucine from inactive angiotensin I to form angiotensin II. It is angiotensin II that causes arteriolar vasoconstriction, leading to increased peripheral resistance and increased blood pressure. Additionally, angiotensin II can also stimulate the adrenal cortex, triggering the release of aldosterone, which in turn increases sodium ion reabsorption in the kidneys and water reabsorption, resulting in increased blood pressure [64].
ACE not only catalyzes the formation of angiotensin II but also catalyzes the degradation of bradykinin. Unlike angiotensin II, bradykinin has vasodilatory, anti-inflammatory, ROS-reducing, antifibrinolytic, and antithrombotic effects [65]. The usefulness of bradykinin is mediated by the release of bradykinin-induced NO. NO is a vasodilator factor in tissues and a major regulator of endothelial function [66].
NO production by endothelial cells is mediated by eNOS, stimulated by acetylcholine and shear stress thru receptor and non-receptor pathways, calcium-dependent and calcium-independent pathways. In the presence of endothelial cell dysfunction, the effect of acetylcholine on vascular smooth muscle cells directly causes constriction and inhibits the release of NO into the lumen. Vasoconstriction increases shear stress, the most potent physiological stimulus for NO production [67]. The imbalance between angiotensin II and bradykinin, with increased levels of angiotensin II and decreased levels of bradykinin due to ACE overexpression, ultimately leads to endothelial dysfunction. By targeting ACE, ACE inhibitors not only inhibit the formation of angiotensin II but also increase the availability and prevent the degradation of bradykinin [68].
Angiotensin II not only raises blood pressure, but it also activates different pathways that are involved in cardiac remodeling, such as the growth of fibroblast cells. These fibroblasts, in turn, make too much extracellular matrix, which causes myocardial fibrosis [69]. Fibroblasts are the main cells in connective tissue that make and keep the extracellular matrix (ECM), which is made up of structural proteins like collagen, fibronectin, and proteoglycans. Fibroblast activity is tightly controlled to keep tissues in balance when things are normal. Fibroblasts produce too much extracellular matrix when TGF-β is activated by high blood sugar, oxidative stress, or tissue damage. TGF-β helps fibroblasts turn into myofibroblasts, which are very good at making ECM parts. These cells make a lot of type I and III collagen, fibronectin, and other matrix proteins. Pathological conditions lead to an elevation in ECM formation, alongside a reduction in the activity of proteolytic enzymes, including MMPs (Matrix Metalloproteinases), and an enhancement in the expression of TIMPs (Tissue Inhibitors of Metalloproteinases). This imbalance causes too much ECM to build up in the tissue. Fibrosis happens when extracellular matrix builds up and replaces functional tissue with connective tissue.
Myocardial fibrosis is a condition in which connective tissue replaces healthy heart muscle tissue, resulting in a decrease in the heart’s ability to contract and relax effectively. In patients with DM, increased ACE is often more pronounced due to the influence of high glucose levels, which stimulate the renin-angiotensin-aldosterone system, exacerbating this process and contributing to the development of heart failure [70].
Beside measuring ACE levels, another biomarker of heart damage is CK-MB. Creatine-Kinase MB (CK-MB) is a cardiac enzyme composed of M and/or B subunits. CK plays a role in regulating the production of high-energy phosphates and their utilization for tissue contraction. Generally, CK acts as an intermediary for high-energy phosphate binding thru creatine phosphate from the mitochondria to the cytoplasm. Therefore, this enzyme is found in tissues with high energy demands, such as the renal tubules and cardiac muscle. CK-MB is found in large quantities in heart muscle, so total serum CK and CKMB concentrations increase when there is injury to the myocardium. Elevated CK-MB levels in heart failure patients are often associated with a poorer prognosis. This is because high CK-MB levels indicate ongoing myocardial damage, which has the potential to worsen heart failure. Several studies have shown that high CK-MB levels are associated with an increased risk of heart failure worsening, especially in patients who have previously experienced myocardial infarction. However, CKMB is more specific to myocardial injury than CK [71]. The results of CK-MB data measurements are presented in Figure 2.
In the healthy group, the CK-MB enzyme value was 195.36 ± 19.57 U/L. The CK-MB level in the normal group showed no significant heart disorders. These results are in line with previous studies that reported normal CK-MB values in healthy rats of 137.70 ± 20.31 U/L [72] and 205.00 ± 17.32 [73]. In group II, which was given a single dose of 45 mg/kg body weight of STZ, there was a significant increase in CK-MB levels compared to group I (healthy group), namely 780.65 ± 63.48 U/L after 14 days. The increase in CK-MB levels indicates damage to the heart muscle through oxidative stress and inflammation, which worsens heart function in diabetic rats [74]. In group III, which was given STZ at a dose of 45 mg/kg BW and Telmisartan at a dose of 10 mg/kg BW as a control group, there was a decrease in CK-MB levels of 340.11 ± 94.01 U/L compared to group II. Telmisartan, an angiotensin II receptor antagonist, has a protective effect on the heart and can reduce inflammation and oxidative stress in diabetic rats [74,75]. In groups IV, V, and VI, which were administered STZ at a dose of 45 mg/kg body weight and doses of polyherbal nanoemulsion preparations at 100, 200, and 300 mg/kg BW, CK-MB levels decreased sequentially to 399.44 ± 186.03, 503.74 ± 76.17, and 552.35 ± 176.21 U/L, respectively, compared to Group II. This occurred due to the antioxidant and anti-inflammatory effects of the active compounds in the nano-polyherbal formulation, which can help reduce damage to the heart muscle [76]. The results show that increasing the active ingredient content in polyherbal nanoemulsion preparations can provide better cardioprotective effects compared to lower concentrations, but still show higher CK-MB levels compared to group III, which was given Telmisartan treatment. In Group VII, which received a 200 mg/kg BW polyherbal formulation, there was a decrease in CK-MB levels compared to Group II, which was 672.17 ± 261.83 U/L. Although there was a decrease in CK-MB levels, this group still showed higher CK-MB levels compared to the group treated with Telmisartan and polyherbal nanoemulsion at concentration a 100 mg/kg BW.
In addition to measuring CK-MB levels, another biomarker of heart damage, Lactate Dehydrogenase (LDH), is also measured for cardioprotective activity. LDH is an enzyme involved in cellular energy metabolism, and LDH concentration in the blood can increase in response to heart tissue damage caused by physiological or pathological stress [77]. Increased LDH levels are closely related to myocardial fibrosis, which contributes to heart failure. LDH is an enzyme involved in energy metabolism, particularly in the conversion of lactate to pyruvate. LDH is found in various tissues throughout the body, including the heart, liver, muscles, kidneys, and brain, so its elevation can serve as an indicator of damage or stress in various organs, including the heart [78]. Therefore, testing cardioprotective activity with the LDH marker provides important insights into measuring the potential for heart protection.
From the results of LDH level measurements over 14 days (Figure 2), in Group II, which only received a single dose of 45 mg/kg BW STZ induction, the LDH levels showed a significant increase over 14 days compared to Group I, indicating heart damage due to STZ induction. In Group III, which received a single dose of 45 mg/kg BW STZ induction and was treated with Telmisartan at a dose of 10 mg/kg BW, LDH levels decreased, indicating that Telmisartan has a cardioprotective effect. These results are consistent with previous studies stating that there was a significant decrease after treatment with Telmisartan at a dose of 10 mg/kg BW (p < 0.001) [79]. Meanwhile, in groups IV, V, VI, and VII, there was a decrease in LDH levels, but it was not significant between the conditions before and after treatment. This result may be due to several factors, including the limited treatment time (14 days), which may not have been long enough to observe significant protective effects from both the nanopolyherbal and polyherbal preparations. Additionally, variations in the data can also lead to non-significant results. Previous research results related to measuring LDH levels induced by streptozotocin showed that serum LDL levels decreased significantly in all treated groups [80].
Elevated LDH can be an indicator of more extensive myocardial cell damage, including fibrosis. In patients with heart failure, elevated LDH indicates damage or inflammation in the heart that triggers the formation of scar tissue (fibrosis) in response to repeated damage [81]. During myocardial fibrosis, oxidative stress and inflammation play a role in damaging the structure of the heart [82]. Elevated LDH, caused by cell damage, may indicate that this process is ongoing and may worsen myocardial fibrosis.
Additionally, the comparison between the groups administered the nanoemulsion preparation (groups IV, V, and VI) and the group administered the polyherbal preparation (group VII) showed significantly different results. This means that the nanoemulsion had a greater or different effect compared to the polyherbal preparation in influencing LDH levels. Nanoemulsion are a more stable and easily absorbed form of drug preparation by the body, which can increase therapeutic effectiveness. Conversely, the polyherbal preparation, which consists of multiple plant extracts, may have a more complex or different By optimizing drug delivery thru a nanoemulsion system, the therapy provided becomes more effective because nanoemulsion allow for controlled drug release and more focused delivery to the target site mechanism of action in influencing biochemical processes in the body [83].
Then, measurements of the SGOT and SGPT enzymes were taken from several different treatment groups. The SGOT (Serum Glutamate-Oxaloacetate Transaminase) enzyme, also known as AST (Aspartate Aminotransferase), and the SGPT (Serum Glutamate-Pyruvate Transaminase) enzyme, also called ALT, are 2 types of enzymes found in various body organs, especially in the liver, heart, muscles, and kidneys. Both enzymes play a role in amino acid metabolism, which is the process of converting amino acids into energy. SGOT and SGPT enzymes are found primarily in the liver, heart, muscles, kidneys, and brain, where they are involved in the conversion of glutamate and oxaloacetic acid into alpha-ketoglutarate and aspartic acid [84]. This enzyme helps in the process of cellular energy metabolism, particularly within liver and heart cells. An increase in SGOT levels in the blood can indicate damage to the liver and heart. High SGOT levels are found in conditions such as heart disease, hepatitis, and myocardial infarction. These 2 enzymes (SGOT and SGPT) can be associated with cardiovascular disease. Elevated SGOT and SGPT enzymes in cardiovascular disease are associated with myocardial infarction and cardiomyopathy, which cause damage to the heart muscle and blood vessels. This is because SGOT, found in high amounts in heart muscle, can be released into the bloodstream when damage occurs. The results of the SGOT and SGPT enzyme measurements are presented in Figure 2.
The results obtained show that Group I (healthy) had SGOT levels of 117.00 ± 7.11 U/L and SGPT levels of 46.60 ± 8.90 U/L. These levels reflect normal physiological conditions without any organ dysfunction or damage, and an increase in these enzyme levels in the healthy group is generally considered part of the body’s normal metabolic process [85]. The SGPT and SGOT levels in this healthy group are consistent with previous research, which found SGPT levels in the healthy control group to be 116.92 ± 22.35 U/L and SGOT levels to be 58.72 ± 8.79 U/L [86]. In Group II, the SGOT level obtained was 164.20 ± 7.66 U/L and the SGPT level was 75.42 ± 3.51 U/L. This SGOT level is consistent with previous research, which was 138.16 ± 8.83 U/L [87], and for the SGPT level of 62.55 ± 0.62 U/L [88].
The use of streptozotocin in experimental animals aims to induce diabetes mellitus. Streptozotocin has properties as a diabetogenic agent that can trigger excessive production of free radicals and cause oxidative stress [27]. STZ works by targeting pancreatic β-cells, which impacts the reduction of insulin levels in the blood, leading to hyperglycemia [45]. Type 2 diabetes mellitus (T2DM) can lead to various complications, one of which is liver dysfunction. Liver dysfunction is a significant cause of death in patients with diabetes mellitus [89]. The heart or liver is the site of glucose storage and metabolism to maintain blood glucose levels in balance. The body needs insulin to help absorb glucose from the bloodstream into all body cells [90].
Group III (STZ 45 mg/kg BW and Telmisartan 10 mg/kg BW) produced SGOT levels of 156.40 ± 27.49 U/L and SGPT levels of 54.00 ± 4.33 U/L. These results are consistent with previous research stating that there was a decrease in SGOT levels in rats after Telmisartan therapy for 137.47 ± 0.85 U/L compared to the sick group, which was 230.34 ± 13.43 U/L [91]. It is evident that there is a significant difference between group I and the treatment groups (II, III, IV, and VII). Additionally, groups II, V, and VII show significantly different results, indicating that treatment with 100 and 300 mg/kg BW nanopolyherbal preparations resulted in a decrease in SGOT enzyme levels compared to group II. However, there was no significant difference in SGOT enzyme levels between groups IV, V, and VI and polyherbal VII.
SGOT and SGPT levels can also be influenced by the duration of diabetes. In hyperglycemic conditions, the liver plays an important role in glucose metabolism, as it can store glycogen and producing glucose thru glycogenolysis and gluconeogenesis [92]. Additionally, the increase in glucose followed by elevated SGOT and SGPT levels indicates organ damage. The decline in heart function in DM leads to increased SGOT levels, which is more sensitive to heart damage [84]. STZ triggers the formation of free radicals, leading to oxidative stress in the liver. This oxidative stress can damage hepatocyte cells, including SGPT and SGOT. Increased levels of both enzymes are indicators of liver cell damage [84].
Myocardial fibrosis is a condition in which connective tissue replaces healthy heart muscle tissue due to long-term damage or stress on the heart. This fibrosis process reduces the heart muscle’s ability to contract effectively, contributing to heart failure. In heart failure, an increased workload on the heart, especially due to conditions such as hypertension or ischemic heart disease, can cause damage to heart muscle fibers. This damage leads to the release of SGOT and SGPT into the blood [93].
Oxidative stress and inflammation in heart failure patients often contribute to heart damage. During this process, not only cardiac enzymes such as CK-MB increase, but also SGOT and SGPT due to more extensive damage to myocardial tissue and muscle cells involved in cardiac remodeling [94]. In chronic heart failure, increased SGOT and SGPT may reflect more extensive myocardial tissue damage, which is associated with the development of myocardial fibrosis.
Cardiotoxicity due to uncontrolled hyperglycemia is one condition that can worsen heart function, especially in patients with diabetes mellitus. This damage can affect various cardiac biochemical parameters such as ACE (angiotensin-converting enzyme), CK-MB (creatine kinase MB), LDH (lactate dehydrogenase), and liver enzymes SGOT (serum glutamic oxaloacetic transaminase) and SGPT (serum glutamic pyruvic transaminase). Given the negative impact of hyperglycemia on the heart and liver, the use of herbal-based alternative therapies is increasingly attractive to reduce such damage. A combination of Nigella sativa, which contains thymoquinone, Moringa oleifera with the active ingredient moringin, and Centella asiatica, which contains asiaticosides, was chosen because it has been proven to have the potential to improve metabolic balance and provide protection to heart tissue.
Thymoquinone, as one of the main active substances contained in Nigella sativa, has been extensively studied for its various properties, including as an antioxidant [95], anti-inflammatory [96], and immunomodulator [97]. In the context of cardiotoxicity, thymoquinone has the potential to help reduce heart damage caused by various stress factors, including uncontrolled hyperglycemia.
As an immunomodulator, thymoquinone works by balancing the body’s immune response. This can reduce excessive inflammation in heart tissue, which is often the main cause of heart damage due to hyperglycemia or other factors. In addition, thymoquinone can also reduce the formation of free radicals, which contribute to cell oxidation and heart cell damage. By regulating immune cell activity and reducing uncontrolled inflammatory responses [98], thymoquinone can help improve heart function and reduce the severity of cardiotoxicity [99], especially that caused by conditions such as uncontrolled diabetes mellitus.
Asiaticosides, an active compound contained in Centella asiatica [100], has various therapeutic benefits that have been proven in traditional and modern medicine, including as an agent that supports protection against the severity of cardiotoxicity [21]. In the context of cardiotoxicity, especially that caused by oxidative stress and inflammation, asiaticosides plays an important role in supporting recovery and protecting the heart from further damage.
One of the main properties of asiaticosides is as a powerful antioxidant asiaticosides can help reduce the number of free radicals that cause oxidative stress, one of the main causes of heart cell damage in cardiotoxicity conditions. By reducing oxidative damage to heart cells, asiaticosides serves to maintain the integrity and function of this organ [101]. In addition, asiaticosides also has significant anti-inflammatory effects. Chronic inflammation due to uncontrolled hyperglycemia can damage heart tissue and blood vessels, contributing to the development of cardiovascular disease and cardiotoxicity. Asiaticosides can reduce this inflammatory response by inhibiting pro-inflammatory pathways, thereby helping to prevent further damage to the heart. Several studies have also shown that asiaticosides can improve blood circulation and accelerate tissue healing. This is particularly important in the context of cardiotoxicity, where damage to heart muscle and blood vessels requires repair to prevent more serious heart dysfunction [102].
Moringa oleifera, commonly known as the drumstick tree, contains various bioactive compounds such as moringin, flavonoids, and polyphenols, which offer numerous health benefits [103,104]. One of the main benefits of moringin is as a powerful antioxidant [20] and antiinflammatory agent [105], which can help reduce the severity of cardiotoxicity, especially in conditions caused by oxidative stress and inflammation, such as uncontrolled hyperglycemia.
Moringin, as the main active ingredient in Moringa oleifera, has been shown to reduce cell and tissue damage through several mechanisms. As an antioxidant, moringin can reduce the number of free radicals in the body that play a role in the oxidation process, which causes damage to heart cells and blood vessels. In this case, moringin helps maintain the integrity of heart cells, reduces inflammation, and protects the heart from oxidative stress, which is often the main cause of cardiotoxicity [20,103,105].
In addition, moringin also has anti-inflammatory properties that can reduce chronic inflammation in the heart and blood vessels. Uncontrolled hyperglycemia often causes inflammation in various organs, including the heart. Moringin can inhibit this inflammatory pathway, which reduces the damage caused by long-term inflammation [105]. With its antioxidant and anti-inflammatory properties, moringa can help reduce the severity of cardiotoxicity and support heart recovery from damage caused by hyperglycemia or other stress factors. Thus, moringa plays an important role in cardiovascular protection, especially in situations that require support for heart health compromised by metabolic conditions such as diabetes.
Histological examination, a microscopic analysis of tissue structure, can identify morphological alterations in cardiac tissue, including necrosis, hypertrophy, inflammatory cell infiltration, and fibrosis. Histological examination is essential for assessing the direct pathogenic effects on the myocardium, especially in cases of myocardial infarction, myocarditis, or cardiomyopathy. Nevertheless, the focus of this study was limited to the analysis of cardiac biomarkers, including CK-MB, ACE, LDH, SGOT, and SGPT, which serve as biochemical indicators of injury or stress on cardiac tissue. While biomarkers offer prompt and precise data on potential cardiac injury, they cannot replace histological analysis for assessing structural and morphological alterations in tissue. Consequently, the study’s focus is confined to the examination of cardiac biomarkers as indicators of myocardial injury. While the results suggest an initial indication of possible myocardial injury, no histological analysis has been conducted to directly ascertain morphological changes in the tissue. Consequently, further research employing histological analysis is necessary to achieve a more comprehensive understanding of the pathogenic effects on cardiac tissue.
Conclusions
Polyherbal nanoemulsion shown protective effects against myocardial injury in a diabetic paradigm, as indicated by enhanced enzymatic injury indicators (ACE, CK-MB, LDH, SGOT, SGPT) and elevated in vivo nitric oxide levels, suggesting improved endothelial function. These findings are encouraging and necessitate a more extensive oxidative/inflammatory panel and functional testing in further trials to definitively validate cardioprotective effectiveness.
Acknowledgements
This study was supported by Directorate of Research, Technology, and Community Services, Ministry of Education, Culture, Research and Technology, Indonesia, under the Grant No. 126/C3/DT.05.00/PL/2025; 0498.12/LL5-INT/AL.04/2025; 008/PDD/LPPM.UAD/V/2025.
Declaration of generative AI in scientific writing
The authors acknowledge the use of generative artificial intelligence tools (such as OpenAI's ChatGPT) during the manuscript writing process, particularly for grammar correction and language editing. No content creation or data interpretation was performed by artificial intelligence; therefore, the author is solely responsible for the content and results of this work.
CRediT author statement
Rafiastiana Capritasari: Project Administration, Data Curation, Formal Analysis, Resources, Visualization, and Writing – original draft. Akrom: Conceptualization, Methodology, Supervision, Validation, Revision, Funding Acquisition, and Writing – original draft. Arif Budi Setianto: Data Curation, Formal Analysis, Investigation, Validation, and Visualization. Titiek Hidayati: Conceptualization, Software, Supervision, Validation, and Writing – original draft.
References
[1] BK Sarkar, J Das, A Das, P Dodak, KS Kumar, R Akter, P Modak, S Halder and SA Prosun. Diabetes mellitus: A Comprehensive review. Journal of Pharmacognosy and Phytochemistry 2019; 8(6), 2362-2371.
[2] D Glovaci, W Fan and ND Wong. Epidemiology of diabetes mellitus and cardiovascular disease. Current Cardiology Reports 2019; 21(21), 1-8.
[3] G Jia, MA Hill and JR Sowers. Diabetic cardiomyopathy: An update of mechanisms contributing to this clinical entity. Circulation Research 2019; 122(4), 624-638.
[4] RF Mapanga and MF Essop. Damaging effects of hyperglycemia on cardiovascular function: Spotlight on glucose metabolic pathways. American Journal of Physiology-Heart and Circulatory Physiology 2016; 310(2), H153-H173.
[5] G Frati, L Schirone, I Chimenti, D Yee, G Biondi-Zoccai, M Volpe and S Sciarretta. An overview of the inflammatory signalling mechanisms in the myocardium underlying the development of diabetic cardiomyopathy. Cardiovascular Research 2017; 113(4), 378-388.
[6] HTK Pham, TTM Kieu, TD Duong, KD Van Nguyen, NQ Tran, TH Tran and JYS Ng. Direct medical costs of diabetes and its complications in Vietnam: A national health insurance database study. Diabetes Research and Clinical Practice 2020; 162, 108051.
[7] B Hidayat, RV Ramadani, A Rudijanto, P Soewondo, K Suastika and JYS Ng. Direct medical cost of type 2 diabetes mellitus and its associated complications in Indonesia. Value in Health Regional Issues 2021; 28, 82-89.
[8] M Naghavi, KL Ong, A Aali, HS Ababneh and YH Abate. Global burden of 288 causes of death and life expectancy decomposition in 204 countries and territories and 811 subnational locations, 1990 - 2021: A systematic analysis for the global burden of disease study 2021. Lancet 2024; 403(10440), 2100-2132.
[9] B De Geest and M Mishra. Role of oxidative stress in diabetic cardiomyopathy. Antioxidants 2022; 11(4), 784.
[10] DM Bravata, N Kim, J Concato and LM Brass. Hyperglycaemia in patients with acute ischaemic stroke: How often do we screen for undiagnosed diabetes? An International Journal of Medicine 2003; 96(7), 491-497.
[11] WA Hsueh and K Wyne. Renin-angiotensin-aldosterone system in diabetes and hypertension. The Journal of Clinical Hypertension 2011; 13(4), 224-237.
[12] T Zhu, Q Han, X Zhang and Q Hou. Effects of xinnaoning combined with trimetazidine on the levels of CK and its isoenzymes, AST, ALT And LDH in patients with myocardial ischemia. American Journal of Translational Research 2021; 13(4), 2875-2882.
[13] MS Alam, YA Kaabi, MM Hoque, F Riyaz, MH Shah, MS Akhter, ZH Hakami and Y Mjery. Association between cardiac enzymes level in type 2 diabetes mellitus from jazan region, Saudi Arabia. South Eastern European Journal of Public Health 2025; 26, 280-286.
[14] PP Swoboda, AK McDiarmid, B Erhayiem, DP Ripley, LE Dobson, P Garg, TA Musa, KK Witte, TM Kearney, JH Barth, R Ajjan, JP Greenwood and S Plein. Diabetes mellitus, microalbuminuria, and subclinical cardiac disease: Identification and monitoring of individuals at risk of heart failure. Journal of the American Heart Association 2017; 6(7), e005539.
[15] E Dal Canto, A Ceriello, L Rydén, M Ferrini, TB Hansen, O Schnell, E Standl and JWJ Beulens. Diabetes as a cardiovascular risk factor: An overview of global trends of macro and micro vascular complications. European Journal of Preventive Cardiology 2019; 26(2), 25-32.
[16] MS Ola, MM Ahmed, HM Abuohashish, SS Al-Rejaie and AS Alhomida. Telmisartan Ameliorates neurotrophic support and oxidative stress in the retina of streptozotocin-induced diabetic rats. Neurochemical Research 2013; 38(8), 1572-1579.
[17] BV Bonifácio, PB Da Silva, MA Dos, S Ramos, KMS Negri, TM Bauab and Marluschorilli. Nanotechnology-based drug delivery systems and herbal medicines: A review. International Journal of Nanomedicine 2014; 9, 1-15.
[18] F Gutama, MI Barliana and IM Puspitasari. Factors associated with health-related quality of life in patients with coronary heart disease. Pharmacia 2022; 69(3), 771-777.
[19] SDS Banjarnahor and N Artanti. Antioxidant properties of flavonoids. Medical Journal of Indonesia 2014; 23(4), 239-244.
[20] A Gugliandolo, C Gabriella, M Claudia, A Osvaldo, R Patrick, P Daniele, I Renato, M Emanuela and C Luigi. α-cyclodextrin/moringin induces an antioxidant transcriptional response activating Nrf2 in differentiated NSC-34 motor neurons. Antioxidants 2024; 13(813), 1-16.
[21] NNM Razali, CT Ng and LY Fong. Cardiovascular protective effects of centella asiatica and its triterpenes: A review. Planta Medica 2019; 85(16), 1203-1215.
[22] T Hidayati, A Akrom and AB Setianto. Black cumin seed oil preparation consumption potentially improves adaptive cellular immune response among healthy volunteers. International Journal of Public Health 2022; 11(2), 680-686.
[23] A Akrom, T Hidayati and AB Setianto. Polyherbal preparations as antioxidant and hepatoprotector in alloxan-induced Sprague Dawley (SD) rats). AIP Conference Proceedings 2024; 3155(1), 080002.
[24] IS Muflihah, S Soetrisno, R Reviono, A Prayitno, B Wasita and EP Pamungkasari. Ovalbumin-induced chronic asthma in female wistar rats: Eosinophilic infiltration, IL-5 profiles and bronchial histopathology. Trends in Sciences 2025; 22(7), 1-14.
[25] R Capritasari, AB Setianto, IR Akrom and Y Sapto. Design and characterization of optimized polyherbal nanoencapsulation: In vitro antioxidant activity targeting Nrf2 and gst pathways. International Journal of Applied Pharmaceutics 2025; 17(3), 50-59.
[26] M Scheen, R Giraud and K Bendjelid. Stress hyperglycemia, cardiac glucotoxicity, and critically ill patient outcomes current clinical and pathophysiological evidence. Physiological Reports 2021; 9(2), 1-19.
[27] BL Furman. Streptozotocin-induced diabetic models in mice and rats. Current Protocols in Pharmacology 2015; 70(1), 5-47.
[28] PA Gerber and GA Rutter. The role of oxidative stress and hypoxia in pancreatic beta-cell dysfunction in diabetes mellitus. Antioxidants Redox Signal 2017; 26(10), 501-518.
[29] N Eguchi, ND Vaziri, DC Dafoe and H Ichii. The role of oxidative stress in pancreatic β cell dysfunction in diabetes. International Journal of Molecular 2021; 22(4), 1-18.
[30] T Szkudelski. The mechanism of alloxan and streptozotocin action in b cells of the rat pancreas. Physiological Research 2001; 50(6), 537-546.
[31] P González, P Lozano, G Ros and F Solano. Hyperglycemia and oxidative stress: An integral, updated and critical overview of their metabolic interconnections. International Journal of Molecular Sciences 2023; 24(11), 9352.
[32] DR Yang, MY Wang, CL Zhang and Y Wang. Endothelial dysfunction in vascular complications of diabetes: A comprehensive review of mechanisms and implications. Frontiers in Endocrinology 2024; 15, 1359255.
[33] N Mylonas, K Drosatos and S Mia. The role of glucose in cardiac physiology and pathophysiology. Current Opinion in Clinical Nutrition & Metabolic Care 2023; 26(4), 323-329.
[34] X Guan, C Zhou, X Zhuo, W Yang and H Wang. The heart of diabetes: Unraveling metabolic drivers of cardiomyopathy. Cardiovascular Diabetology - Endocrinology Reports 2025; 11(1), 18.
[35] M Wang, Y Liu, Y Liang, K Naruse and K Takahashi. Systematic understanding of pathophysiological mechanisms of oxidative stress-related conditions - diabetes mellitus, cardiovascular diseases, and ischemia–reperfusion injury. Frontiers in Cardiovascular Medicine 2021; 8, 649785.
[36] EAE Elbassuoni. Incretin Attenuates diabetes-induced damage in rat cardiac tissue. The Journal of Physiological Sciences 2014; 64(5), 357-364.
[37] RM Singh, T Waqar, FC Howarth, E Adeghate, K Bidasee and J Singh. Hyperglycemia-Induced cardiac contractile dysfunction in the diabetic heart. Heart Failure Reviews 2018; 23(1), 37-54.
[38] KKA Kasar, MH Al-Bahrani and AA Mohsin. Interpreting myocardial enzymatic biomarkers in the setting of acute myocardial infraction AMI. Journal of Techniques 2022; 4(4), 105-110.
[39] A Abdelatty, MS Ahmed, MA Abdel-Kareem, M Dmerdash, R Mady, AS Saad, A Albrakati, KE Kotb, A Elsawak and W Abdo. Acute and delayed doxorubicin-induced myocardiotoxicity associated with elevation of cardiac biomarkers, depletion of cellular antioxidant enzymes, and several histopathological and ultrastructural changes. Life 2021; 11(9), 880.
[40] MA Abeeleh, ZB Ismail, KR Alzaben, SA Abu-Halaweh, MK Al-Essa, J Abuabeele and MM Alsmady. Induction of diabetes mellitus in rats using intraperitoneal streptozotocin: A comparison between 2 strains of rats. European Journal of Scientific Research 2009; 32(3), 398-402.
[41] M Zhang, XY Lv, J Li, ZG Xu and L Chen. The characterization of high-fat diet and multiple low-dose streptozotocin induced type 2 diabetes rat model. Journal of Diabetes Research 2008; 2008, 704045.
[42] SN Goyal, NM Reddy, KR Patil, KT Nakhate, S Ojha, CR Patil and YO Agrawal. Challenges and issues with streptozotocin-induced diabetes - a clinically relevant animal model to understand the diabetes pathogenesis and evaluate therapeutics. Chemico-Biological Interactions 2016; 244, 49-63.
[43] T Micakovic, S Papagiannarou, E Clark, Y Kuzay, K Abramovic, J Peters, C Sticht, N Volk, T Fleming, P Nawroth, HP Hammes, N Alenina, HJ Gröne and SC Hoffmann. The angiotensin II type 2 receptors protect renal tubule mitochondria in early stages of diabetes mellitus. Kidney International 2018; 94(5), 937-950.
[44] N Nazaruddin, TN Siregar, A Sutriana, S Wahyuni and T Armansyah. The Impact of streptozotocin-induced diabetes on testosterone hormone and androgen receptor expression and correlation with sperm quality impairment in sprague dawley rats. Trends in Sciences 2025; 22(9), 10409.
[45] DC Damasceno, AO Netto, IL Iessi, FQ Gallego, SB Corvino, B Dallaqua, YK Sinzato, A Bueno, IMP Calderon, and MVC Rudge. Streptozotocin-induced diabetes models: Pathophysiological mechanisms and fetal outcomes. BioMed Research International 2014; 2014(1), 819065.
[46] H Smulyan, A Lieber and ME Safar. Hypertension, diabetes type II, and their association: Role of arterial stiffness. American Journal of Hypertension 2016; 29(1), 5-13.
[47] G Jia, MA Hill and JR Sowers. Diabetic cardiomyopathy: An update of mechanisms contributing to this clinical entity. Circulation Research 2018; 122(4), 624-638.
[48] N Hadzi-Petrushev, M Angelovski and M Mladenov. Advanced glycation end products and diabetes. Obesity, Diabetes and Inflammation: Molecular Mechanisms and Clinical Management 2023; F1468, 99-127.
[49] R Sayon, A Shruthi and R Suman. Retinal fibrosis in diabetic retinopathy. Experimental Eye Research 2016; 2(74), 71-75.
[50] SH Ihm, K Chang, HY Kim, SH Baek, HJ Youn, KB Seung and JH Kim. Peroxisome proliferator-activated receptor-γ activation attenuates cardiac fibrosis in type 2 diabetic rats: The effect of rosiglitazone on myocardial expression of receptor for advanced glycation end products and of connective tissue growth factor. Basic Research in Cardiology 2010; 105(3), 399-407.
[51] AO Abiodun, DI AlDosari, A Alghamdi, AA Al-Amri, S Ahmad and MS Ola. Diabetes-induced stimulation of the renin-angiotensin system in the rat brain cortex. Saudi Journal of Biological Sciences 2023; 30(9), 103779.
[52] W Chen, L Juan, F Yu, J Yuanyuan, Y Runan, C Jinyu, L Wenxiu, L Yue and Y Xinhua. Cardioprotection of cortistatin against isoproterenol-induced myocardial injury in rats. Annals of Translational Medicine 2020; 8(6), 309-309.
[53] J Peti-Peterdi. High glucose and renin release: The role of succinate and GPR91. Kidney international 2010; 78(12), 1214-1217.
[54] C Savoia, RM Touyz, F Amiri and EL Schiffrin. Selective mineralocorticoid receptor blocker eplerenone reduces resistance artery stiffness in hypertensive patients. Hypertension 2008; 51(2), 432-439.
[55] GA Favre, VLM Esnault and EV Obberghen. Modulation of glucose metabolism by the renin-angiotensin-aldosterone system. American Journal of Physiology-Endocrinology and Metabolism 2015; 308(6), E435-E449.
[56] C Yu and RW Jeremy. Angiotensin, transforming growth factor Β and aortic dilatation in marfan syndrome: Of mice and humans. IJC Heart & Vasculature 2018; 18, 71-80.
[57] JJL Grobe, AP Mecca, M Lingis, RC Speth, MK Raizada and MJ Katovich. Prevention of angiotensin ii-induced cardiac remodeling by angiotensin-(1-7). American Journal of Physiology-Heart and Circulatory Physiology 2007; 292(2), 736-742.
[58] Y Tie, F Tang, D Peng, Y Zhang and H Shi. TGF-beta signal transduction: Biology, function and therapy for diseases. Molecular Biomedicine 2022; 3(1), 45.
[59] Z Deng, T Fan, C Xiao, H Tian, Y Zheng, C Li and J He. TGF-β signaling in health, disease, and therapeutics. Signal Transduction and Targeted Therapy 2024; 9(1), 61.
[60] Y Zhang, D Jin, X Kang, R Zhou, Y Sun, F Lian and X Tong. Signaling pathways involved in diabetic renal fibrosis. Frontiers in Cell and Developmental Biology 2021; 9, 696542.
[61] KL Pan, YC Hsu, ST Chang, CM Chung and CL Lin. The role of cardiac fibrosis in diabetic cardiomyopathy: From pathophysiology to clinical diagnostic tools. International Journal of Molecular Sciences 2023; 24(10), 8604.
[62] JL Gamboa, M Pretorius, KC Sprinkel, NJ Brown and TA Ikizler. Angiotensin converting enzyme inhibition increases adma concentration in patients on maintenance hemodialysis - a randomized cross-over study. BMC Nephrology 2015; 16, 167.
[63] A Krüger-Genge, A Blocki, RP Franke and F Jung. Vascular endothelial cell biology: An update. International Journal of Molecular Sciences 2019; 20(18), 4411.
[64] RAS Santos, AJ Ferreira, T Verano-Braga and M Bader. Angiotensin-converting enzyme 2, angiotensin-(1-7) and mas: New players of the renin-angiotensin system. Journal of Endocrinology 2013; 216(2), R1-R17.
[65] CR Alves, GB Alves, AC Pereira, IC Trombetta, RG Dias, GFA Mota, T Fernandes, JE Krieger, CE Negrão and EM Oliveira. Vascular reactivity and ACE activity response to exercise training are modulated by the +9/-9 bradykinin B2 receptor gene functional polymorphism. Physiological Genomics 2013; 45(12), 487-492.
[66] MG Scioli, G Storti, F D’Amico, R Rodríguez Guzmán, F Centofanti, E Doldo, EMC Miranda and A Orlandi. Oxidative stress and new pathogenetic mechanisms in endothelial dysfunction: Potential diagnostic biomarkers and therapeutic targets. Journal of Clinical Medicine 2020; 9(6), 1-39.
[67] J Herrmann, L Lerman and A Lerman. Simply say yes to no? nitric oxide (NO) sensor-based assessment of coronary endothelial function. European Heart Journal 2010; 31(23), 2834-2836.
[68] A Ancion, J Tridetti, ML Nguyen Trung, C Oury and P Lancellotti. A review of the role of bradykinin and nitric oxide in the cardioprotective action of angiotensin-converting enzyme inhibitors: Focus on perindopril. Cardiology and Therapy 2019; 8(2), 179-191.
[69] O Eldimassi. Angiotensin II binds to one of its two receptors, inducing three major signaling pathways to regulate blood pressure. International Journal of High School Research 2024; 6(2), P26.
[70] G Díaz-Araya, R Vivar, C Humeres, P Boza, S Bolivar and C Muñoz. Cardiac fibroblasts as sentinel cells in cardiac tissue: Receptors, signaling pathways and cellular functions. Pharmacological Research 2015; 101, 30-40.
[71] N Gusti, A Putu, L Santika, AAW Lestari, IW Putu and S Yasa. Relationship between Troponin T (TNT) and Creatine Kinase-Myocardial Band (CK-MB) levels in Acute Myocardial Infarction (AMI) patients at Sanglah General Hospital Denpasar. E-Jurnal Medika Udayana 2018; 7(1), 43-48.
[72] MN Alam, MM Hossain, MM Rahman, N Subhan, MAA Mamun, A Ulla, HM Reza and MA Alam. Astaxanthin prevented oxidative stress in heart and kidneys of isoproterenol-administered aged rats. Journal of Dietary Supplements 2018; 15(1), 42-54.
[73] AMS Gomaa, AT Abdelhafe and HA Aamer. Garlic (Allium Sativum) exhibits a cardioprotective effect in experimental chronic renal failure rat model by reducing oxidative stress and controlling cardiac Na + /K + -atpase activity and Ca2+ levels. Cell Stress Chaperones 2018; 23(5), 913-920.
[74] C Malfitano, CA Barboza, C Mostarda, RK da Palma, CP dos Santos, B Rodrigues, SCF Freitas, A Belló-Klein, S Llesuy, MC Irigoyen and KD Angelis. Diabetic hyperglycemia attenuates sympathetic dysfunction and oxidative stress after myocardial infarction in rats. Cardiovascular Diabetology 2014; 13(1), 1-9.
[75] BR Goyal, K Parmar, RK Goyal and AA Mehta. Beneficial role of telmisartan on cardiovascular complications associated with STZ-induced type 2 diabetes in rats. Pharmacological Reports 2011; 63(4), 956-966.
[76] PM Boarescu, I Boarescu, IC Bocșan, D Gheban, AE Bulboacă, C Nicula, RM Pop, RM Râjnoveanu and SD Bolboacă. Antioxidant and anti-inflammatory effects of curcumin nanoparticles on drug-induced acute myocardial infarction in diabetic rats. Antioxidants 2019; 8(10), 504.
[77] L Brigic, M Ehlimana, K Faris, H Mirela, DN Azra, D Lejla and G Una. Role of lactate dehydrogenase as a biomarker of early cardiac remodeling: A cross-sectional study. Cureus 2024; 16(9), e68906.
[78] R Yang, Q Jia, XF Liu and SF Ma. Effect of genistein on myocardial fibrosis in diabetic rats and its mechanism. Molecular Medicine Reports 2018; 17(2), 2929-2936.
[79] S Garg, KS Irfan, MR Kumar, SM Kumar, K Manoj, K Punit, NT Chandra, R Ruma, B Jagriti and AD Singh. Cardioprotective effects of azilsartan compared with that of telmisartan on an in vivo model of myocardial ischemia-reperfusion injury. Journal of Biochemical and Molecular Toxicology 2021; 35(7), e22785.
[80] H Mollazadeh, HR Sadeghnia, A Hoseini, M Farzadnia and MT Boroushaki. Effects Of pomegranate seed oil on oxidative stress markers, serum biochemical parameters and pathological findings in kidney and heart of streptozotocin-induced diabetic rats. Renal Failure 2016; 38(8), 1256-1266.
[81] C Liu, C Ni, W Liu, X Yang, R Zhang, J Zhang, M Luo, J Xu and J Yu. Effects of long-term nonylphenol exposure on myocardial fibrosis and cardiac function in rats. Environmental Sciences Europe2021; 33(1), 96.
[82] A Aimo, V Castiglione, C Borrelli, LF Saccaro, M Franzini, S Masi, M Emdin and A Giannoni. Oxidative stress and inflammation in the evolution of heart failure: From pathophysiology to therapeutic strategies. European Journal of Preventive Cardiology 2020; 27(5), 494-510.
[83] Preeti, S Sambhakar, R Malik, S Bhatia, A Al Harrasi, C Rani, R Saharan, S Kumar, Geeta and R Sehrawat. Nanoemulsion: An emerging novel technology for improving the bioavailability of drugs. Scientifica 2023; 2023(1), 6640103.
[84] SA Husen, D Winarni, ANM Ansori, RJK Susilo and S Hayaza. Hepatoprotective effect of gamma-mangostin for amelioration of impaired liver structure and function in streptozotocin-induced diabetic mice. IOP Conference Series: Earth and Environmental Science 2019; 217(1), 012031.
[85] EM Youssef and GY Wu. Subnormal serum liver enzyme levels: A review of pathophysiology and clinical significance. Journal of Clinical and Translational Hepatology 2024; 12(4), 428-435.
[86] RD Yudhani, RN Pesik, S Azzahro, AF Anisa and R Hendriyani. Acute toxicity test of amomum cardamomum (kapulaga) seed extract on hepatic trasaminase enzyme in winstar rats. Indonesian Journal of Clinical Pharmacy Volume 2020; 9(4), 288.
[87] NR Rahayu, MS Bachri and LH Nurani. Hepatoprotective potentials of green chireta (Andrographis paniculata) and moringa leaves (Moringa oleifera) extracts against streptozotocin-induced hepatotoxicity in rats. Journal of Research in Pharmaceutical Science 2021; 7(11), 21-27.
[88] S Rahman, MAHM Jamal, A Parvin and MR Islam. Antidiabetic activity of Centella asiatica (L.) urbana in alloxan induced type 1 diabetic model rats. Journal of Bio-Science 2011; 1980, 23-27.
[89] KK Gangopadhyay and P Singh. Consensus statement on dose modifications of antidiabetic agents in patients with hepatic impairment. Indian Journal of Endocrinology and Metabolism 2017; 21(2), 341-354.
[90] EEP Oktaviona, N Qomariyah and F Khaleyla. Aktivitas hepatoprotektif ekstrak daun Phyllanthus acidus L. pada mencit diabetes mellitus tipe 2. LenteraBio: Berkala Ilmiah Biologi 2023; 12(3), 381-388.
[91] NAT Abbas, FA Fayed, RS El Sebaey and HA Hassan. Telmisartan and candesartan promote browning of white adipose tissue and reverse fatty liver changes in high fat diet fed male albino rats. Naunyn-Schmiedeberg’s Archives of Pharmacology 2024; 397(4), 2359-2378.
[92] S Rafaqat, A Sattar, A Khalid and S Rafaqat. Role of liver parameters in diabetes mellitus - a narrative review. Endocrine Regulations 2023; 57(1), 200-220.
[93] M Correale, N Tarantino, R Petrucci, L Tricarico, I Laonigro, M Di Biase and ND Brunetti. Liver disease and heart failure: Back and forth. European Journal of Internal Medicine 2018; 48, 25-34.
[94] V Bhayana and AR Henderson. Biochemical markers of myocardial damage. Clinical Biochemistry 1995; 28(1), 1-29.
[95] NK Isaev, EE Genrikhs and EV Stelmashook. Antioxidant thymoquinone and its potential in the treatment of neurological diseases. Antioxidants 2023; 12(2), 433.
[96] Z Kohandel, T Farkhondeh, M Aschner and S Samarghandian. Anti-inflammatory effects of thymoquinone and its protective effects against several diseases. Biomedicine & Pharmacotherapy 2021; 138, 111492.
[97] A Akrom, T Hidayati, N Umam and GZ Saputri. Combination of black cumin seeds with curcuma xanthorrhiza extract as an antioxidant and immune-modulator agent in the COVID-19 pandemic era. International Journal of Public Health 2022; 11(3), 1073-1081.
[98] A Parlar and SO Arslan. Thymoquinone exhibits anti-inflammatory, antioxidant, and immunomodulatory effects on allergic airway inflammation. Archives of Clinical and Experimental Medicine 2019; 4(2), 60-65.
[99] GH Danaei, B Memar, R Ataee and M Karami. Protective effect of thymoquinone, the main component of nigella sativa, against diazinon cardio-toxicity in rats. Drug and Chemical Toxicology 2019; 42(6), 585-591.
[100] S Pramono and D Ajiastuti. Standardization of gotu kola (Centella asiatica (L.) Urban) herbal extract based on asiaticoside content by TLC-densitometry. Majalah Farmasi Indonesia 2004; 15(3), 118-123.
[101] Y Luo, C Fu, Z Wang, Z Zhang, H Wang and Y Liu. Asiaticoside attenuates the effects of spinal cord injury through antioxidant and anti-inflammatory effects, and inhibition of the P38-MAPK mechanism. Molecular Medicine Reports 2015; 12(6), 8294-8300.
[102] C Xu, L Xia, D Xu, Y Liu, P Jin, M Zhai, Y Mao, Y Wang, A Wen, J Yang and L Yang. Cardioprotective effects of asiaticoside against diabetic cardiomyopathy: Activation of the AMPK/Nrf2 pathway. Journal of Cellular and Molecular Medicine 2024; 28(2), e18055.
[103] G Borgonovo, L De Petrocellis, A Schiano Moriello, S Bertoli, A Leone, A Battezzati, S Mazzini and A Bassoli. Moringin, a stable isothiocyanate from Moringa oleifera, activates the somatosensory and pain receptor TRPA1 channel in vitro. Molecules 2020; 25(4), 1-12.
[104] P. Kashyap, Shiv Riar, Charanjit Singh, Jindal Navdeep, Baniwal Poonam, Guiné Raquel P.F. Correia Paula M.R, Mehra Rahul, and Kumar Harish. Recent advances in drumstick (Moringa oleifera) leaves bioactive compounds: composition, health benefits, bioaccessibility, and dietary applications. Antioxidants 2022; 11(2), 402.
[105] NA Lopez-Rodriguez, M Gaytán-Martínez, M de la Luz Reyes-Vega and G Loarca-Piña. Glucosinolates and isothiocyanates from Moringa oleifera: Chemical and biological approaches. Plant Foods for Human Nutrition 2020; 75(4), 447-457.