Trends
Sci.
2026; 23(5): 11792
Effects of Kenikir (C. caudatus Kunth) Extract on Biomarker and Renal Function in A Preeclamptic Rat Model Study
Devi Kurniasari1,2,*, Soetrisno Soetrisno1,3, Adi Prayitno1,4, Brian Wasita1,5,
Pramasari Dirgahayu6, Risya Cilmiaty7 and Vitri Widyaningsih8
1Doctoral Program of Medical Sciences, Faculty of Medicine, Universitas Sebelas Maret, Central Java, Indonesia
2Department of Midwifery, Faculty of Health, Universitas Malahayati, Lampung, Indonesia
3Department of Obstetrics and Gynecology, Faculty of Medicine, Universitas Sebelas Maret,
Central Java, Indonesia
4Department of Oral Diseases and Doctoral Program of Medical Sciences, Faculty of Medicine,
Universitas Sebelas Maret, Central Java, Indonesia
5Department of Anatomical Pathology, Faculty of Medicine, Universitas Sebelas Maret, Central Java, Indonesia
6Faculty of Medicine, Universitas Sebelas Maret, Central Java, Indonesia
7Department of Oral Diseases and Doctoral Program of Medical Sciences, Faculty of Medicine,
Universitas Sebelas Maret, Central Java, Indonesia
8Department of Public Health, Faculty of Medicine, Universitas Sebelas Maret, Central Java, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 5 September 2025, Revised: 4 November 2025, Accepted: 11 November 2025, Published: 5 January 2026
Introduction: Preeclampsia is a systemic pregnancy syndrome characterized by oxidative stress and inflammation, leading to significant renal and placental dysfunction. The therapeutic potential of Cosmos caudatus Kunth (Kenikir) is largely derived from quercitrin, a flavonoid whose potent antioxidant and anti-inflammatory properties directly target key aspects of this pathogenesis. This study aimed to assess the therapeutic potential of C. caudatus Kunth leaf extract (CCE) by measuring its effects on key preeclampsia markers, including MDA, IL-6, sFlt-1, renal parameters, and placental histopathology. Materials and methods: An experimental laboratory study with a pre-posttest-only design for the proteinuria and blood pressure and a posttest-only control group design for other parameters. The samples comprised 30 pregnant Sprague Dawley rats, divided into 5 groups with 6 members each. Rats were then randomized into normal (N) and Negative control groups exposed to L-NAME (PE), L-NAME + Nifedipine 50 mg/kgBW (CPos), L-NAME + CCE 200 mg/kgBW (PI), and L-NAME + Nifedipine 50 mg/kgBW+ CK 200 mg/kgBW (P2). levels of MDA, sFlt-1, IL-6, and Cystatin-C, were analyzed using enzyme-linked immunosorbent assay (ELISA). Glomerular fibrosis and placental spiral artery diameter were assessed using Masson’s Trichrome and HE staining, respectively. Results and discussion: CCE monotherapy (P1) demonstrated superior efficacy, significantly reducing systolic blood pressure and proteinuria to levels comparable to the normal group (p > 0.05), outperforming Nifedipine monotherapy. CCE treatment reduced MDA levels by 60% (from 3.397 to 1.358 Nmol/mL, p < 0.001), sFlt-1 by 57% (from 9.881 to 4.238 Nmol/mL, p < 0.001), and IL-6 by 75% (from 10.718 to 2.650 Nmol/mL, p < 0.001) compared to the PE group. Histopathological analysis revealed complete absence of glomerulosclerosis in the P1 group, indicating profound renoprotection. 4) Conclusions: The combination therapy (P2) showed complex interactions, occasionally less effective than CCE alone. These findings strongly suggest that CCE is a promising multi-target therapeutic agent for preeclampsia.
Keywords: Preeclampsia, Malondialdehyde (MDA), soluble Fms-like tyrosine kinase-1 (sFlt-1), Interleukin-6 (IL-6), Cystatin-C, C. caudatus Kunth, Glomerulosclerosis
Introduction
Preeclampsia (PE) remains a formidable challenge in obstetrics, constituting a major threat to maternal and fetal health globally [1-4]. This condition is diagnostically defined as the new onset of hypertension (systolic blood pressure ≥ 140 mmHg and/or diastolic blood pressure ≥ 90 mmHg) accompanied by significant proteinuria (≥ 0.3 g/24 h) or signs of end-organ dysfunction, manifesting after the 20th week of gestation [5,6]. The clinical significance of PE is profound. As a leading cause of maternal mortality worldwide, its impact extends beyond the immediate perinatal period, being strongly associated with substantial long-term morbidity in both mothers and their offsprin [6,7]. Epidemiologically, preeclampsia complicates an estimated 2% - 8% of all pregnancies, underscoring its widespread nature and substantial public health burden [6,8,9].
A critical aspect of this multifaceted disorder is its pervasive effect on maternal organ systems. Notably, the kidneys are among the organs most severely affected [10]. Pregnancy itself induces significant functional adaptations that alter renal hemodynamics, and preeclampsia exacerbates this state, leading to renal dysfunction [10-12]. This renal impairment not only contributes to the acute diagnostic criteria of the disease but is also a key driver of the associated long-term sequelae [13,14]. Consequently, mothers with a history of PE face an elevated lifetime risk of developing chronic cardiovascular and renal diseases [15]. Understanding the pathophysiological mechanisms underlying this renal involvement is therefore paramount for improving both immediate clinical management and long-term health outcomes for affected women. The pathogenesis of preeclampsia is widely attributed to aberrant placentation, characterized by inadequate remodeling of the spiral arteries. This malfunctioning placenta becomes a source of anti-angiogenic factors, such as soluble fms-like tyrosine kinase-1 (sFlt-1), and pro-inflammatory cytokines that are released into the maternal circulation [16]. A key consequence of this anti-angiogenic state is systemic endothelial dysfunction, the central hallmark of the disorder [17,18]. Endothelial injury manifests as widespread vasoconstriction and reduced perfusion, leading to ischemia and oxidative stress in vital organs [19,20]. This is evidenced by an increase in lipid peroxidation products like Malondialdehyde (MDA), a reliable marker of oxidative damage. Concurrently, the heightened inflammatory response, marked by elevated levels of pro-inflammatory cytokines such as Interleukin-6 (IL-6), further exacerbates endothelial injury and contributes to the systemic clinical manifestations [21]. The renal system is a primary target in this pathophysiological cascade [22]. The glomerular endotheliosis results in altered glomerular morphology and a compromised ultrafiltration capacity, leading to proteinuria. Beyond traditional markers, renal dysfunction in PE is also reflected by alterations in novel biomarkers like Cystatin-C, which serves as a sensitive indicator of glomerular filtration rate (GFR) impairment [23]. These pathological changes are a direct result of the interplay between angiogenic imbalance, oxidative stress, and inflammatory processes.
Given the central role of oxidative stress, inflammation, and angiogenic imbalance in PE pathophysiology, interventions targeting these pathways hold significant therapeutic promise. Natural compounds with potent antioxidant and anti-inflammatory properties are thus compelling candidates for adjunctive investigation [24,25]. Cosmos caudatus Kunth (commonly known as Kenikir in Indonesia) is a readily available medicinal plant traditionally consumed for its health benefits [26-28]. It is particularly rich in bioactive flavonoids, including Quercetin, Quercitrin, Kaempferol, and Chlorogenic acid, which are known for their potent antioxidant and anti-inflammatory activities [25,29,30]. Emerging evidence from in vivo studies suggests that CCE and its constituent quercetin can ameliorate key pathological features of hypertensive disorders. Quercetin administration has been shown to reduce blood pressure, decrease plasma levels of endothelin-1 (ET-1), sFlt-1, TNF-α, and IL-6, while increasing vascular endothelial growth factor (VEGF) in animal models [31-34]. However, despite these promising pharmacological properties, the therapeutic potential of CCE specifically for preeclampsia, particularly its efficacy in simultaneously modulating the defining angiogenic, inflammatory, and oxidative stress markers and its protective effect on renal function, remains largely unexplored. Therefore, this study aims to comprehensively evaluate the therapeutic effects of C. caudatus leaf extract (CCE) in a preeclamptic rat model. We hypothesize that CCE administration will ameliorate the PE phenotype by reducing oxidative stress (as measured by MDA), mitigating inflammation (IL-6), counteracting anti-angiogenic activity (sFlt-1), and improving renal function (Cystatin-C), consequently leading to improved birth outcomes and histopathological findings in the placenta and kidney.
Materials and methods
Plant material and preparation
Cosmos caudatus Kunth used in this study was procured from local farmers in the East Lampung agricultural region, Indonesia. The freshly harvested leaves were meticulously sorted and washed to prevent damage. The samples were then air-dried away from direct sunlight, followed by oven-drying until a moisture content of below 10% was achieved. The dried leaves were ground into a fine powder using a mechanical grinder.
The maceration extraction was performed at the Research and Development Centre for Medicinal Plants and Traditional Medicine (B2P2TOOT) in Tawangmangu, Indonesia. A 500 g sample of the powder was macerated in a macerator using 3 different concentrations of re-distilled ethanol (50%, 70%, and 96%). The maceration process was conducted for 72 h with the solvent replaced every 24 h. The resultant crude extracts from each concentration were concentrated separately using a rotary evaporator at 40 °C to obtain viscous ethanol extracts. The extracts were then centrifuged at 1006×g for 15 min.
The extraction yields from the 3 different ethanol concentrations were calculated and compared. Based on this comparison, the 70% ethanol extract demonstrated the highest yield and was selected for further analysis. This selected extract was designated as CCE (Cosmos caudatus ethanol extract). The chemical constituents of the CCE were subsequently analysed using Quadrupole Time-of-Flight (Q-TOF) mass spectrometry at the Saraswati Indo Genetech (SIG) laboratory in Bogor, Indonesia.
Molecular docking analysis
The in-silico phase commenced with the preparation of ligand and protein structures. The ligand structures were retrieved from the PubChem database in SDF format and subsequently converted to PDB format using Discovery Studio Visualizer. The target proteins, namely MDA (PDB ID: 6VJ3), IL-6 (PDB ID: 4O9H), and sFlt-1 (PDB ID: 5BK7), were acquired from the RCSB Protein Data Bank. All structures were then loaded into PyRx software (version 0.8). The docking calculations were executed using the VinaWizard plugin, which employs the AutoDock Vina algorithm to predict the binding affinity (ΔG, kcal/mol) and the preferred binding orientation (pose) of the ligand within the protein’s active site.
Ethical approval
The animal study protocol received ethical approval from The Bioethics Commission for Medical and Health Research, Faculty of Medicine, Sultan Agung University, Semarang (Approval Number: 79/11/2025/Komisi Bioetik) prior to the commencement of the study. All experimental procedures involving animals were performed in accordance with standard international guidelines for the care and use of laboratory animals
Animal care
Pregnant female Sprague-Dawley rats, aged 10 - 12 weeks and weighing 160 - 200 g, were obtained from the Integrated Animal Laboratory of the Faculty of Medicine at Sultan Agung University. The animals were housed in a controlled environment under a 12/12-hour light/dark cycle, at a temperature of 22 ± 1 °C, and humidity of 55 ± 5%. They were provided with ad libitum access to food and water. A 1-week acclimatization period was implemented prior to the start of the experiment. To induce preeclampsia, a rat model was established following the method described by Fitriana et al. [35,36]. Briefly, pregnant rats received daily subcutaneous injections of L-NAME (Cat.GA11233-5G, GLPBIO, USA) at a dosage of 75 mg/kg body weight, dissolved in 200 µL of solvent, from gestational day 9 to day 19. Animals were randomly grouped and researchers assessing outcomes were blinded to treatments to reduce bias.
Treatment
The rats were randomly allocated into 5 groups (n = 6). The Normal group (N) received no treatment. The Negative Control group (PE) received a daily subcutaneous injection of L-NAME (75 mg/kg BW) from gestational day (GD) 9 to 19. The Positive Control group (PE + Nif) received the same L-NAME regimen plus a daily oral of nifedipine (50 mg/kg BW) from GD10 to 19. Treatment group P1 (PE + CCE) received L-NAME plus a daily oral of CCE extract (200 mg/kg BW) from GD10 to 19. Treatment group P2 (PE + Nif + CCE) received L-NAME plus a combination of nifedipine (50 mg/kg BW) and CCE extract (200 mg/kg BW) from GD10 to 19. Blood pressure and urine samples were collected for protein analysis on GD7 (pre-treatment baseline), GD13, and GD19. On GD20, all rats were euthanized via inhalation of a high dose of isoflurane (10% - 20%). Prior to euthanasia, a caesarean section was performed. The kidneys and placentas were weighed, rinsed with 0.9 %NaCl, and fixed in 4% paraformaldehyde for subsequent histopathological examination. Urinary protein concentration was quantified using BCA Protein Colourimetric Assay kit (Elabscience, Beijing, China) on an automated biochemical analyzer. Blood pressure was measured non-invasively using a CODA monitor (BP-2010A, Softron, Beijing, China), following a standardized protocol. Rats were warmed at 40 °C for 5 minutes to facilitate tail blood flow. A sensor was placed at the tail base, and measurements commenced after curve stabilization. Five readings were taken per session, and the mean value was calculated for analysis.
Measurement of biomarker
Biomarkers related to Preeclamsia were measured using the Enzyme-Linked Immunosorbent Assay (ELISA) kits (BT-LAB, Shanghai, China), strictly following the manufacturer’s protocols. Four specific biomarkers were selected based on their roles in Preeclamsia pathogenesis: MDA as an Oxidative Stress marker, IL-6 as an inflammatory marker, sFlt-1 as an Angiogenesis marker, and Cystatin-C as a Renal Function marker. All 4 biomarkers (MDA, IL-6, sFlt-1, and Cystatin-C) were measured from serum samples. On gestational day (GD) 20, rats were deeply anesthetized. Blood samples (approximately 2 mL) were collected aseptically via terminal bleeding from the inferior vena cava using a sterile syringe without anticoagulant. The blood was transferred into serum separator tubes, allowed to clot for 30 minutes at room temperature, and then centrifuged at 3,000× g for 15 min to isolate the serum. The serum aliquots were stored at −80 °C until analysis. For the ELISA, 25 μL of each serum sample was analyzed in duplicate. The manufacturer’s protocol was followed precisely: 200 μL of enzyme conjugate was added to each well and incubated for 60 min at room temperature. After incubation, the plate was washed 3 times with wash buffer. Then, 200 μL of substrate solution was added and incubated for 15 min at room temperature in the dark. The reaction was stopped by adding 100 μL of stop solution. The absorbance was immediately measured at 450 nm using a microplate reader (xMark™ Microplate Absorbance Spectrophotometer, Bio-Rad Laboratories Inc.). The concentrations of each biomarker were calculated based on a standard curve generated using Microplate Manager® 6 Software (Bio-Rad Laboratories Inc.).
Histological examination
Histopathology Hematoxylin and Eosin (H&E) staining was performed as previously described. Placental and renal tissue sections were examined under 20× magnification, and representative images were captured for analysis. Five images were taken for each section, and staining was measured using ImageJ software (version 1.53t). To evaluate the histopathology of the placenta and kidney, H&E staining of the placental sections and Masson’s trichrome staining of the kidney sections were performed. Five randomly selected fields were photographed for histological assessment of each section. The histology of the placenta and kidney was examined and evaluated by an Anatomical Pathologist. Histological changes in the placenta were measured through 4 fields of spiral artery diameter measurements and averaged. Glomerular fibrosis was assessed by Masson’s staining with a score of 0 - 3 (0 ≤ 5%, 1 = 5% - 10%, 2 = 11% - 25%, 3 ≥ 25%).
Urine analysis and arterial pressure measurement
Rats were housed in separate metabolic cages for 24 h, followed by measurement of urine output on gestational day 4, gestational day 12, and gestational day 19. The BP-2000 Blood Pressure Analysis System (Visitech Systems, Inc., Apex, NC, USA) was used to perform a non-invasive tail-cuff to measure mean arterial pressure (MAP) on gestational day 7, gestational day 13, and gestational day 19. Subsequently, all female rats were warmed to a temperature of 38 °C for further analysis.
Statistical analysis
All data are expressed as mean ± standard deviation. Data analysis was performed using SPSS version 24.0. The Shapiro-Wilk test confirmed data normality. One-way ANOVA with Tukey’s post-hoc test was used to analyze differences in MDA, sFlt-1, and IL-6 levels, as well as placental weight. For non-normally distributed data, including Cystatin-C levels and histopathological scores of kidney and placental tissues, the Kruskal-Walli’s test followed by the Mann-Whitney U test with Bonferroni correction was applied. Changes in systolic blood pressure, diastolic blood pressure, and urinary protein levels over time were analyzed using Repeated Measures ANOVA with an appropriate post-hoc test. A p-value < 0.05 was considered statistically significant.
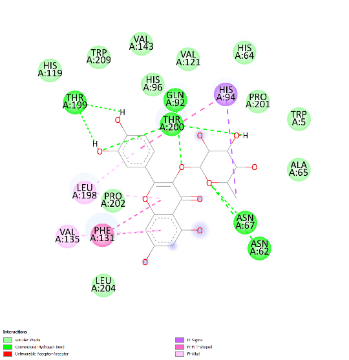
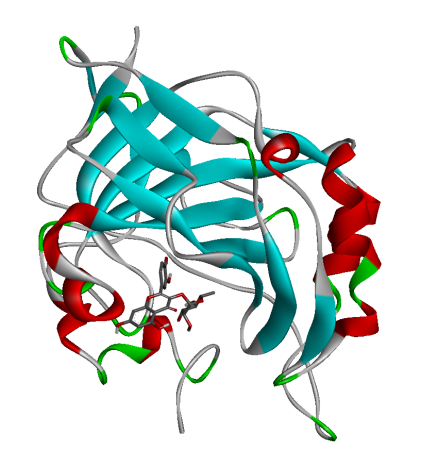
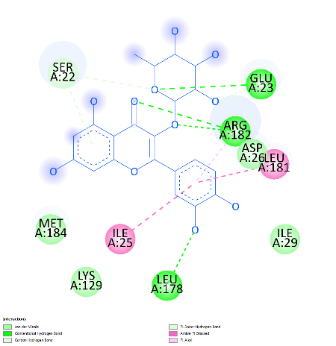
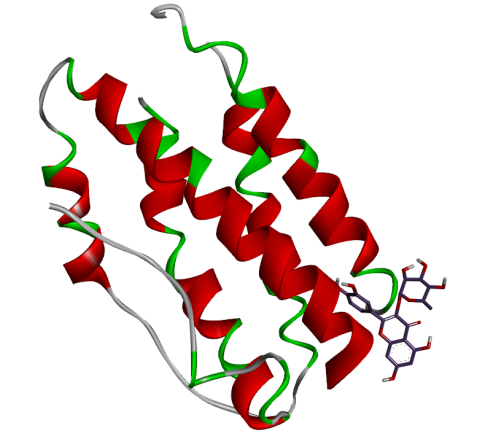
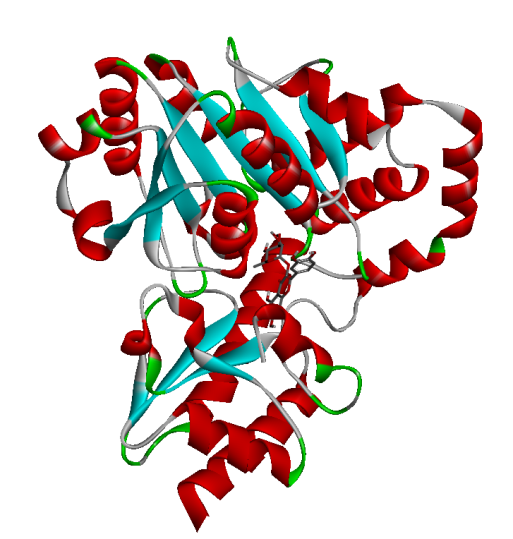
Prior to investigating the therapeutic outcomes, a phytochemical profiling of Cosmos caudatus extract (CCE) was conducted using Liquid Chromatography-Mass Spectrometry/Tandem Mass Spectrometry Quadrupole Time-of-Flight (LC-MS/MS-QTOF) to identify bioactive constituents targeting key pathways in preeclampsia pathogenesis, including oxidative stress (MDA), inflammation (IL-6), angiogenic imbalance (sFlt-1), and renal dysfunction (Cystatin-C). The analysis revealed a diverse phytochemical profile. From the identified compounds, quercitrin was selected for further investigation. This selection was based on its well-documented biological activities - particularly its potent anti-inflammatory and antioxidant properties - which strongly align with the pathological pathways of preeclampsia, as well as the established safety profile of its natural source. The 3D structures of the target proteins (MDA: 6VJ3, IL-6: 4O9H, sFlt-1: 5BK7) and the chemical structure of Quercitrin, retrieved from the Protein Data Bank and PubChem respectively, are presented in Figure 1.
Ligand-Protein |
2D Visualization |
3D Visualization |
Quercitrin – MDA (6VJ3) |
|
Quercitrin |
Quercitrin-IL-6 (4O9H) |
|
Quercitrin |
Quercitrin – sFlt-1(5BK7) |
|
Quercitrin |
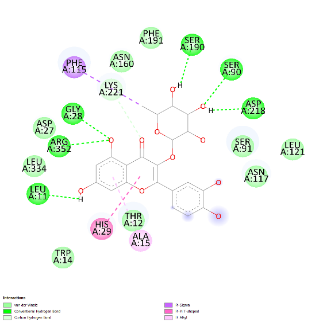
Figure 1 Docking pose of hydrogen bonding interaction of target proteins with Quercitrin.
Results and discussion
Molecular docking analysis of CCE targeting MDA, IL16 and sFlt-1 receptor
Molecular docking simulations were performed to investigate the binding interactions between quercitrin, a key bioactive constituent of Cosmos caudatus Kunth extract (CCE), and 3 preeclampsia-related protein targets: malondialdehyde (MDA; PDB: 6VJ3), interleukin-6 (IL-6; PDB: 4O9H), and soluble Fms-like tyrosine kinase-1 (sFlt-1; PDB: 5BK7). The results demonstrated that quercitrin exhibited remarkably strong binding affinity towards all 3 targets (Table 1). Notably, it showed the highest affinity for MDA with a binding energy (ΔG) of −9.62 kcal/mol, followed closely by sFlt-1 at −9.21 kcal/mol, and IL-6 at −7.02 kcal/mol. These values indicate spontaneous and highly stable binding interactions. A detailed analysis of the binding interactions revealed that quercitrin engages with each target through a complex network of molecular interactions. For the MDA target, the strong binding was stabilized by multiple Van der Waals interactions with residues including His119, Trp209, and Val143. With sFlt-1, quercitrin formed an extensive network of 7 conventional hydrogen bonds with key residues including Gly28, Arg352, and Ser190, along with additional π-π and π-alkyl interactions. Similarly, its interaction with the inflammatory marker IL-6 was characterized by strong hydrogen bonds with Glu23, Arg182, and Leu178, supplemented by various hydrophobic interactions. The consistently strong binding affinity of quercitrin across these pathologically distinct targets underscores its potential as a multi-target therapeutic agent for preeclampsia. This polypharmacological profile represents a significant advantage over other herbal therapies that typically modulate single pathways, as it enables simultaneous targeting of the oxidative stress (via MDA), inflammation (via IL-6), and angiogenic imbalance (via sFlt-1) that collectively drive disease progression. Furthermore, the exceptional binding affinity for MDA (ΔG = −9.62 kcal/mol) contextualizes its potent antioxidant capacity when compared to standard antioxidants; molecular docking studies report that ascorbic acid typically exhibits binding energies in the range of −4.0 to −6.0 kcal/mol for various targets, substantially weaker than the values observed for quercitrin in this study. This provides a robust molecular rationale for the significant reduction in oxidative stress markers observed in our in vivo experiments and highlights the superior potential of quercitrin as a natural therapeutic compound against preeclampsia.
Table 1 Molecular docking results of quercitrin with preeclampsia-related protein targets.
Target Protein (PDB ID) |
Binding Affinity (ΔG, kcal/mol) |
Interaction |
Amino Acid Residue |
MDA (6VJ3) |
-9.62 |
Van der Waals |
His119, Trp209, Val143, Val121, His64, His96, Pro201, Trp5, Ala65, Leu204, Pro202 |
Conventional H-Bond |
Thr200, Thr199, Asn67, Asn62 |
||
Pi-Pi T-shaped |
His94, Phe131 |
||
Pi-Sigma |
His94 |
||
Pi-Alkyl |
Val135, Leu198 |
||
IL-6 (4O9H) |
-7.02 |
Van der Waals |
Ser22, Asp26, Ile29, Lys129, Leu181, Met184 |
Conventional H-Bond |
Glu23, Arg182, Leu178 |
||
Carbon H- Bond |
Arg182 |
||
Pi-Donor H- Bond |
Ser22 |
||
Amide-Pi Stacked |
Ile25, Leu181 |
||
Pi-Alkyl |
Ile25 |
||
sFlt-1 (5BK7) |
-9.21 |
Van der Waals |
Phe191, Asn160, Asp27, Leu334, Ser91, Asn117, Leu121, Trp14 |
Conventional H- Bond |
Gly28, Arg352, Ser190, Ser90, Asp218, Leu11, Thr12 |
||
Carbon H-Bond |
Lys21 |
||
Pi-Sigma |
Phe115 |
||
Pi-Pi T-shaped |
His29 |
||
Pi-Alkyl |
Ala15 |
Effects of CCE on MDA, IL16, sFlt-1 and expression
The in vivo effects of Cosmos caudatus extract (CCE), both as a monotherapy and in combination with nifedipine, on preeclampsia-related biomarkers are presented in Figure 2 and detailed in Table 2. The 1-way ANOVA analysis revealed a statistically significant difference in post-therapy levels across all groups for each biomarker (p < 0.001).
Regarding the inflammatory pathway, IL-6 levels were significantly suppressed by both CCE monotherapy (P1) and the combination therapy (P2) to concentrations comparable to the normal control group. This is a crucial finding, as the standard drug nifedipine alone failed to demonstrate a significant effect (PE vs. PE + Nif, p = 0.104). The efficacy of CCE in modulating IL-6 is highly relevant to PE pathogenesis, given that the downregulation of this key inflammatory cytokine can not only mitigate systemic inflammation but also alleviate associated oxidative stress and reduce apoptosis of placental trophoblastic cells [37,38].
For the angiogenic marker sFlt-1, the most pronounced reduction was observed in the combination therapy group (P2), which achieved the lowest post-therapy level (3.267 ± 0.525 ng/mL). The magnitude of reduction in the P2 group was significantly greater than that achieved by either monotherapy. This is particularly significant because sFlt-1 is a central driver of PE; its elevated levels cause angiogenic imbalance and directly reflect placental vascular damage. The ability of CCE, especially in combination, to normalize sFlt-1 suggests a potent effect on a core pathological mechanism of the disease [39].
A similar superior effect was observed for the renal biomarker Cystatin-C, where the P2 group also showed the most dramatic decrease (3.032 ± 2.426 ng/mL). The selection of Cystatin-C is justified by its role as a sensitive early marker of renal impairment, a major complication of PE. Unlike in normotensive pregnancies, serum Cystatin-C rises with reduced glomerular filtration rate, providing a direct measure of PE-induced renal dysfunction [40]. The profound reduction seen here indicates a strong renoprotective effect of the CCE-nifedipine combination.
Post-hoc tests confirmed that all CCE treatment groups showed statistically significant improvement compared to the negative control group (PE, p < 0.001). The superior outcomes with combination therapy suggest a synergistic interaction, offering a more comprehensive approach to managing the multifactorial pathogenesis of preeclampsia.
Table 2 Post-therapy CCE on serum levels of preeclampsia biomarkers and statistical analysis.
Biomarkers |
After therapy CCE, mean±SD |
p-value after therapy |
p-value (between groups) |
MDA (Nmol/mL) |
|
|
|
Normal |
1.336 ± 0.062 |
0.001 |
0.001 |
PE |
3.397 ± 0.317 |
|
Ref. |
PE + Nif |
1.494 ± 0.033 |
|
0.001 |
PE + CCE |
1.358 ± 0.138 |
|
0.001 |
PE + Nif + CCE |
1.134 ± 0.124 |
|
0.001 |
sFlt-1 (Nmol/mL) |
|
|
|
Normal |
4.214 ± 0.392 |
0.001 |
0.001 |
PE |
9.881 ± 1.508 |
|
Ref. |
PE + Nif |
6.221 ± 0.827 |
|
0.001 |
PE + CCE |
4.238 ± 0.136 |
|
0.001 |
PE + Nif + CCE |
3.267 ± 0.525 |
|
0.001 |
IL-6 (Nmol/mL) |
|
|
|
Normal |
2.759 ± 0.532 |
0.001 |
0.001 |
PE |
10.718 ± 0.713 |
|
Ref. |
PE + Nif |
8.252 ± 0.3406 |
|
0.104 |
PE + CCE |
2.650 ± 0.962 |
|
0.001 |
PE + Nif + CCE |
1.726 ± 0.559 |
|
0.001 |
Cystatin-C (Nmol/mL) |
|
|
|
Normal |
22.178 ± 6.735 |
0.001 |
0.001 |
PE |
58.968 ± 13,639 |
|
Ref. |
PE + Nif |
18.657 ± 4.729 |
|
0.001 |
PE + CCE |
8.044 ± 1.620 |
|
0.001 |
PE + Nif + CCE |
3.032 ± 2.426 |
|
0.001 |
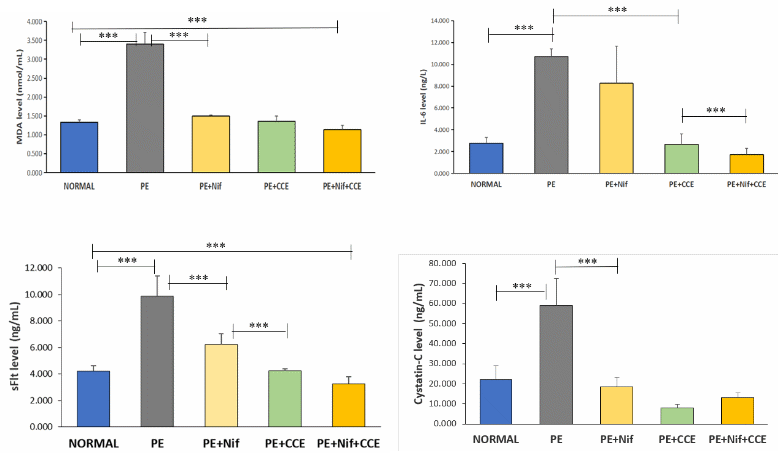
Figure 2 Post-therapy CCE on serum levels of MDA, sFlt-1, IL-6, and Cystatin-C across experimental groups.
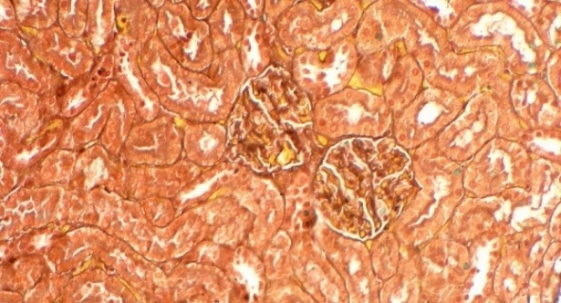
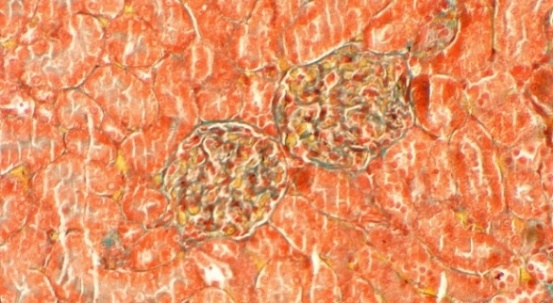
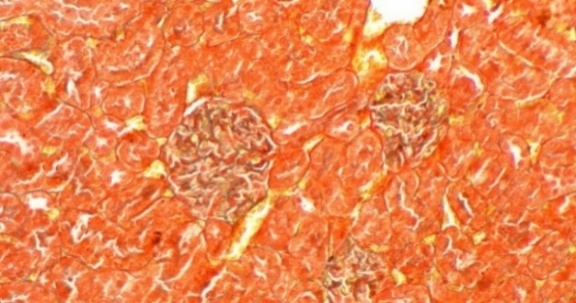
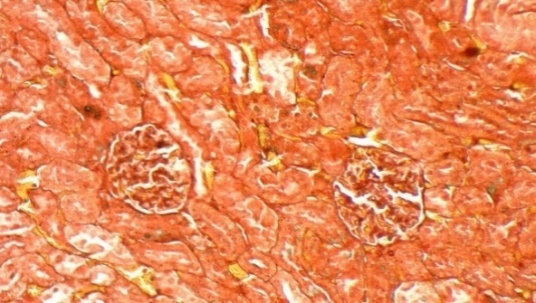
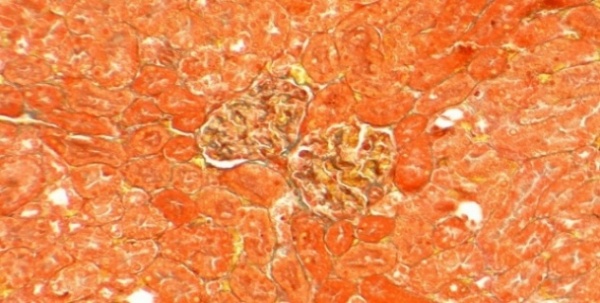
Effects of CCE on glomerulosclerosis on Renal Glomeruli
The results, presented in Figure 3, revealed pronounced differences between groups. Histological analysis of the Normal group showed intact glomerular architecture with no signs of pathology. In contrast, the negative control (PE) group exhibited significant renal damage, with glomerulosclerosis affecting approximately 5% of the glomerular area. The standard drug nifedipine alone (PE + Nif), provided a moderate protective effect, reducing the sclerotic area to 2%. Remarkably, renal morphology in the P1 group (PE + CCE) was completely preserved, with no histological evidence of glomerulosclerosis observed. Unexpectedly, P2 group (PE+Nif + CCE) showed minimal fibrous tissue formation in 1% of the glomerular area. While this represents a significant improvement over the rat model, the presence of minor damage suggests a potential interaction between the 2 compounds that merits further investigation. The histopathological results provide compelling visual evidence of the profound Reno protective effects of CCE. The complete absence of glomerulosclerosis in the CCE monotherapy group (P1) is a particularly significant finding. This result not only demonstrates a protective effect but also suggests that CCE monotherapy may be superior to nifedipine monotherapy in preventing the structural renal damage that characterizes preeclampsia. The superior outcome in the P1 group indicates that the mechanism of CCE’s action likely extends beyond blood pressure control. This potent protection is almost certainly mediated by the rich array of antioxidant and anti-inflammatory bioactive compounds in CCE, such as quercitrin. These compounds presumably mitigate the oxidative stress and inflammation that are central to the endothelial dysfunction and glomerular injury in preeclampsia [41]. The combination therapy (P2) showed minimal damage, representing an improvement over the disease model but slightly reduced efficacy compared to CCE alone. This suggests a complex interaction between nifedipine and CCE bioactives, potentially requiring optimized dosing for true synergy. Ultimately, CCE monotherapy demonstrated exceptional potential as a standalone natural treatment for preventing renal damage in preeclampsia.
Normal |
PE |
|
|
PE+Nif |
PE+CCE |
|
|
PE+Nif+CCE |
|
|
|
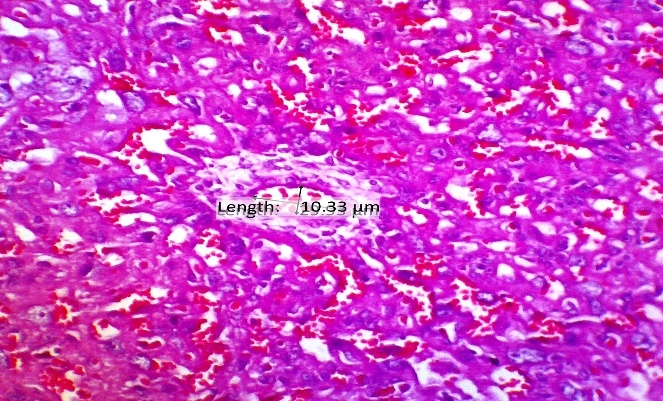
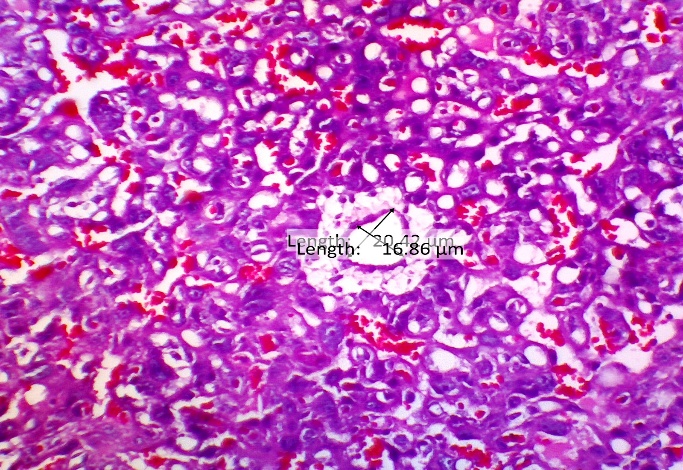
Figure 3 Masson’s Trichrome Staining of Renal Glomeruli: Percentages indicate affected glomerular area. (A) Normal, (B) PE (5%), (C) Nifedipine (2%), (D) CCE (0%), (E) Nifedipine + CCE (1%). Scale bar: 50 μm.
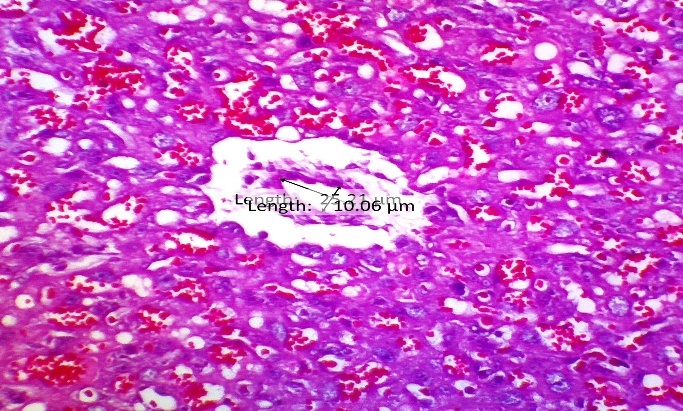
Effect of CCE on placental spiral artery diameter
Evaluation of morphological and vascular parameters revealed significant treatment effects. Placental weight was significantly reduced in the PE group compared to normal controls (Figure 5), indicating compromised placental development. Concomitantly, analysis of spiral artery diameter (Figure 4) showed marked pathological enlargement in the PE group (18.28 ± 5.60 μm) compared to the normal group (14.74 ± 3.66 μm), reflecting impaired vascular remodeling. This pathological alteration is further visually supported by histopathological observations in Figure 6, which shows aberrant spiral artery morphology in the PE group.
The positive control group treated with nifedipine (PE + Nif) also exhibited a similarly elevated mean diameter (18.66 ± 2.55 μm), suggesting limited efficacy of the standard therapy alone in fully normalizing spiral artery architecture.
The most compelling finding emerged from the treatment groups. Administration of CCE alone (PE + CCE) significantly reduced the diameter to 15.65 ± 3.01 μm, approaching the normal baseline. This pronounced reduction indicates that CCE monotherapy is effective in reversing the pathological changes in spiral arteries, likely by mitigating the oxidative stress and endothelial dysfunction that impede proper vascular remodeling in preeclampsia.
Notably, the combination therapy of CCE and nifedipine (PE + Nif + CCE) yielded the most favorable outcome, with the lowest mean diameter of all groups (13.99 ± 2.29 μm), even slightly below the normal baseline. This suggests a potential synergistic effect between the vasodilatory action of nifedipine and the multifaceted vasoprotective mechanisms of CCE's bioactive compounds, ultimately promoting the most significant restoration of vascular function, as also observed in Figure 6.
In summary, these results, supported by both statistical analysis of spiral artery diameter (Figure 4) and histopathological evidence (Figure 6), demonstrate that CCE, particularly when combined with nifedipine, facilitates significant improvement in spiral artery diameter and morphology. This critical improvement is essential for enhancing uteroplacental blood flow and addressing the fundamental placental insufficiency in preeclampsia.
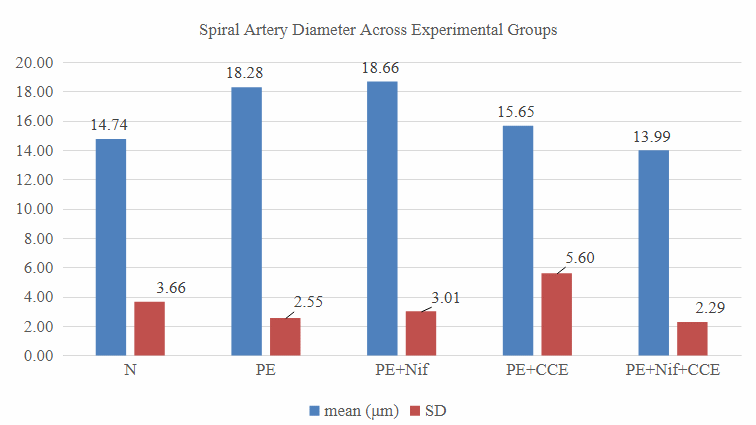
Figure 4 Spiral artery diameter distribution.
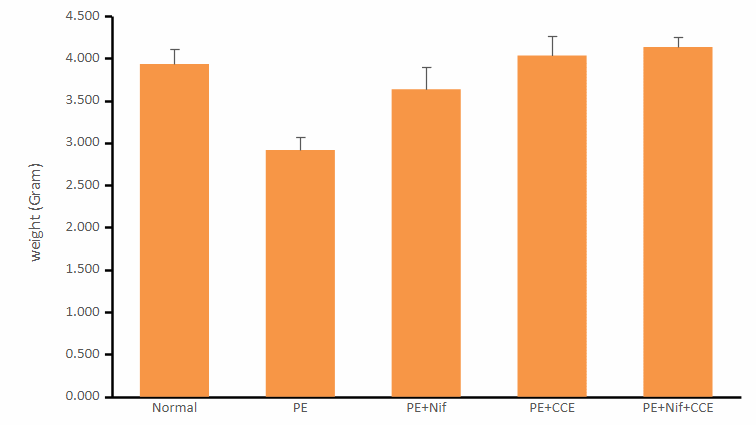
Figure 5 Placental weight in experimental groups.
Normal |
PE |
|
|
PE+Nif |
PE+CCE |
|
|
PE+Nif+CCE |
|
|
|
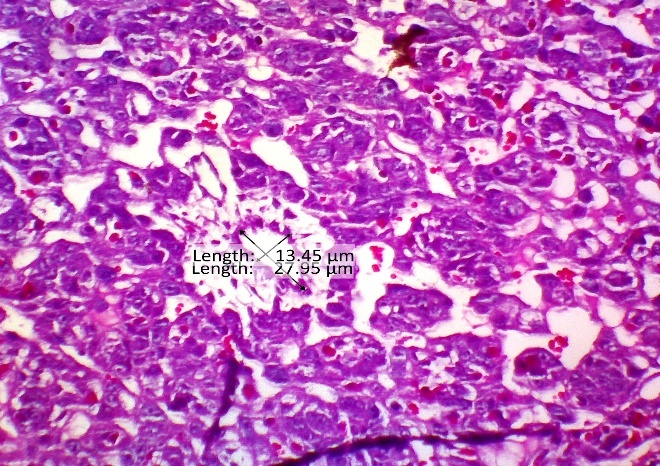
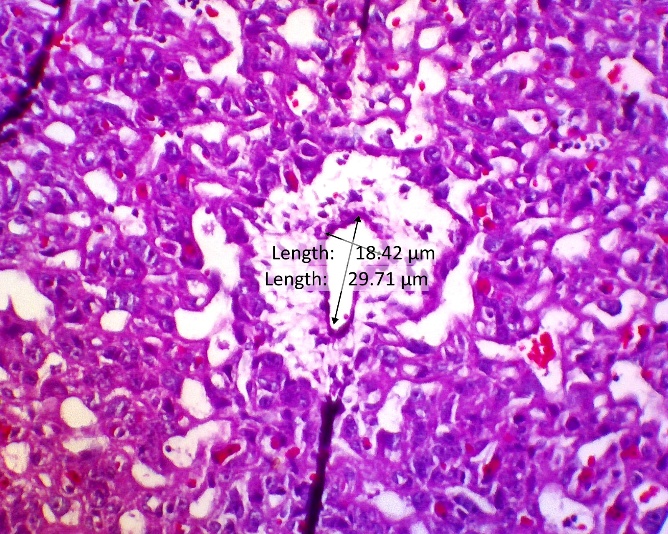
Figure 6 HE Image of the placental spiral arteries, this study proves that there is widening or vasodilation in the placental spiral arteries given CCE therapy so that there is improvement in the placenta in preeclampsia.
Effect CCE on urinary protein levels
Urinary protein levels, a clinical hallmark of preeclampsia-related renal impairment, were significantly elevated in the negative control (PE) group (p < 0.001), confirming successful disease induction (Table 3). Notably, CCE monotherapy (PE + CCE) demonstrated potent renoprotective effects, effectively maintaining urinary protein levels near physiological baseline values (11.14 ± 0.19 mg/mL) at GD-19 performance that was comparable to the normal control group (10.55 ± 0.19 mg/mL) and superior to nifedipine monotherapy (11.80 ± 0.39 mg/mL). These results suggest that the renoprotective mechanism of CCE extends beyond antihypertensive action, likely involving direct modulation of oxidative stress and inflammatory pathways that underlie glomerular damage in preeclampsia. Unexpectedly, the combination therapy group (PE + Nif + CCE) showed higher protein levels (13.34 ± 0.90 mg/mL) than either monotherapy, suggesting a potentially suboptimal interaction between CCE and nifedipine that merits further investigation into dosing regimens. In conclusion, CCE monotherapy significantly attenuated proteinuria in this preeclampsia model, highlighting its promise as a therapeutic agent for preserving renal function.
Measuring Time |
Group |
||||
Normal |
PE |
PE + Nif |
PE + CCE |
PE + Nif + CCE |
|
Protein Urin |
|||||
GD-7 |
10.53 ± 0.39 |
10.45 ± 0.19 |
11.14 ± 0.43 |
10.72 ± 0.51 |
10.86 ± 0.47 |
GD-13 |
10.43 ± 0.14 |
14.36 ± 0.27 |
13.61 ± 0.50 |
12.82 ± 0.41 |
15.05 ± 0.23 |
GD-19 |
10.55 ± 0.19 |
20.43 ± 1.05 |
11.80 ± 0.39 |
11.14 ± 0.19 |
13.34 ± 0.90 |
Effect CCE on Systolic and Siastolic blood pressure
The hemodynamic results presented in Table 4 provide compelling evidence for the potent antihypertensive properties of CCE in a rat model of preeclampsia. The key finding of this study is that CCE monotherapy (PE + CCE) was as effective as the combination of CCE and nifedipine (PE + Nif + CCE) in normalizing systolic blood pressure, and it was superior in normalizing diastolic pressure. This suggests that CCE possesses a robust mechanism of action that does not merely augment the effect of standard calcium channel blocker therapy but may act effectively on its own. The efficacy of CCE is likely mediated through its high concentration of bioactive flavonoids, such as quercitrin and quercetin, which are known for their potent antioxidant and vasodilatory effects. These compounds are postulated to ameliorate endothelial dysfunction by scavenging reactive oxygen species (ROS) and improving nitric oxide (NO) bioavailability, thereby counteracting the L-NAME-induced inhibition of NO synthase. The slightly higher, though not statistically significant, diastolic pressure in the combination therapy group (PE + Nif + CCE) warrants further investigation. It may indicate a complex pharmacodynamic interaction between nifedipine and compounds in CCE, suggesting that an optimal dosing strategy for combination therapy might differ from that of monotherapies. In conclusion, the ability of CCE monotherapy to restore blood pressure to normotensive levels underscores its significant potential as a standalone natural therapeutic agent for the management of preeclampsia-associated hypertension. Its multi-target action, targeting both oxidative stress and vascular function, presents a distinct advantage over single-mechanism approaches
Table 4 Systolic and diastolic blood pressure in preeclamptic rats across experimental groups.
Measuring Time |
Group |
||||
Normal |
PE |
PE + Nif |
PE + CCE |
PE + Nif + CCE |
|
Sistolik |
|||||
GD - 7 |
113.34 ± 6.28 |
110.84 ± 4.96 |
107.84 ± 7.42 |
105.67 ± 7.82 |
108.17 ± 6.34 |
GD-13 |
110.34 ± 13.96 |
149.50 ± 9.82 |
116.00 ± 10.87 |
109.17 ± 11.02 |
120.84 ± 2.64 |
GD-19 |
113.50 ± 2.89 |
155.50 ± 7.69 |
112.67 ± 8.90 |
107.67 ± 2.58 |
107.34 ± 0.56 |
p-value* |
0.802 |
< 0.001 |
0.364 |
0.629 |
< 0.005 |
Diastolik |
|||||
GD - 7 |
79.00 ± 2.53 |
76.00 ± 4.60 |
72.00 ± 5.59 |
70.34 ± 4.72 |
74.34 ± 8.31 |
GD-13 |
76.67 ± 5.16 |
114.34 ± 10.05 |
79.34 ± 6.75 |
76.67 ± 8.19 |
93.17 ± 9.37 |
GD-19 |
77.17 ± 7.36 |
116.84 ± 9.13 |
77.50 ± 9.42 |
75.17 ± 5.27 |
82.84 ± 7.03 |
p-value* |
0.621 |
0.001 |
0.313 |
0.293 |
0.002 |
Conclusions
This study demonstrates that Cosmos caudatus extract (CCE) is a highly effective multi-target therapeutic agent against preeclampsia. CCE monotherapy outperformed the standard drug nifedipine in normalizing blood pressure, preventing proteinuria, and providing complete histoprotection to the kidneys by abolishing glomerulosclerosis. Furthermore, CCE significantly improved placental health by restoring spiral artery remodeling and increasing placental weight. The superior efficacy of CCE is attributed to its multifaceted mechanism of action, which simultaneously addresses the core pathophysiological pathways of preeclampsia: it mitigates oxidative stress (reduced MDA), rectifies angiogenic imbalance (lowered sFlt-1), suppresses inflammation (reduced IL-6), and protects renal function (lowered Cystatin-C). This holistic approach offers a significant advantage over conventional single-mechanism antihypertensive therapy. While the combination of CCE and nifedipine showed beneficial effects, its slightly reduced efficacy in some parameters compared to CCE alone suggests a complex interaction that warrants further investigation to optimize synergistic potential. In conclusion CCE extract presents a promising, potent, and natural strategy for the prevention and treatment of preeclampsia.
Acknowledgements
The authors would like to thank the research supervisors from the Medical Sciences Study Program, Faculty of Medicine, Sebelas Maret University, Surakarta, Central Java, Indonesia, and Malahayati University, Bandar Lampung, Lampung, Indonesia. The authors also thank the Indonesian Education Scholarship Program (BPI) of the Ministry of Education and Culture of the Republic of Indonesia for their support in completing this research. The Integrated Laboratory Unit of the Faculty of Medicine, Sultan Agung Islamic University, Semarang, Central Java, Indonesia, and the Clinical Pathology and Anatomical Pathology Laboratory, Faculty of Medicine, Sebelas Maret University, Surakarta, Indonesia.
Declaration of Generative AI in Scientific Writing
In the preparation of this manuscript, the authors utilised generative artificial intelligence tools specifically DeepSeek strictly for the purpose of language enhancement, including editing and grammatical checks. We hereby explicitly affirm that these tools were not employed in any capacity for generating scholarly content, formulating ideas, analysing data, interpreting results, or drawing conclusions. The authors take full responsibility for the entire scientific content, integrity, and conclusions of this work.
CRediT Author Statement
Devi Kurniasari, Brian Wasita: Conceptualization; Methodology; Software. Devi Kurniasari, Vitri Widyaningsih: Data curation; Writing - Original draft preparation. Soetrisno: Visualization; Investigation. Adi prayitmo: Supervision. Risya Cilmiyati, Pramasari Dirgahayu: Software; Validation. Devi Kurniasari, Vitri Widyaningsih: Writing - Reviewing and Editing.
References
[1] KJ Chang, KM Seow and KH Chen. Preeclampsia: Recent advances in predicting, preventing, and managing the maternal and fetal life-threatening condition. International Journal of Environmental Research and Public Health 2023; 20(4), 2994.
[2] LC Poon, A Shennan, JA Hyett, A Kapur, E Hadar, H Divakar, F McAuliffe, F da Silva Costa, P von Dadelszen, HD McIntyre, AB Kihara, GC Di Renzo, R Romero, M D’Alton, V Berghella, KH Nicolaides and M Hod. The International Federation of Gynecology and Obstetrics (FIGO) initiative on pre-eclampsia: A pragmatic guide for first-trimester screening and prevention. International Journal of Gynecology & Obstetrics 2019; 145(S1), 1-33.
[3] S Rana, E Lemoine, JP Granger and SA Karumanchi. Preeclampsia: Pathophysiology, challenges, and perspectives. Circulation Research 2019; 124(7), 1094-1112.
[4] Pre-eclampsia, Available at: https://www.who.int/news-room/fact-sheets/detail/pre-eclampsia, accessed September 2025.
[5] RA Abbas, L Ghulmiyyah, E Hobeika, IM Usta, F Mirza and AH Nassar. Preeclampsia: A review of early predictors. Maternal-Fetal Medicine 2021; 3(3), 197-202.
[6] S Koulouraki, V Paschos, P Pervanidou, P Christopoulos, A Gerede and M Eleftheriades. Short- and long-term outcomes of preeclampsia in offspring: Review of the literature. Children 2023; 10(5), 826.
[7] M Naser, MM Nasr and LH Shehata. Prediction and management of preeclampsia: Literature review. International Journal of Progressive Sciences and Technologies 2024; 16, 48-66.
[8] Preeclampsia, Available at: https://www.ncbi.nlm.nih.gov/books/NBK570611/, accessed September 2025.
[9] E Overton, D Tobes and A Lee. Preeclampsia diagnosis and management. Best Practice & Research Clinical Anaesthesiology 2022; 36(1), 107-121.
[10] PM Barrett, FP McCarthy, M Evans, M Kublickas, IJ Perry, P Stenvinkel, AS Khashan and K Kublickiene. Hypertensive disorders of pregnancy and the risk of chronic kidney disease: A Swedish registry-based cohort study. PLoS Medicine 2020; 17(8), e1003255.
[11] V Dines, S Suvakov, A Kattah, J Vermunt, K Narang, M Jayachandran, C A. Hassan, AM Norby and VD Garovic. Preeclampsia and the kidney: Pathophysiology and clinical implications. Comprehensive Physiology 2023; 13(1), 4231-4267.
[12] X Hu and L Zhang. Uteroplacental circulation in normal pregnancy and preeclampsia: Functional adaptation and maladaptation. International Journal of Molecular Sciences 2021; 22(16), 8622.
[13] J Gameiro, F Marques and JA Lopes. Long-term consequences of acute kidney injury: A narrative review. Clinical Kidney Journal 2021; 14(3), 789-804.
[14] P Koirala, V Garovic, MI Dato and A Kattah. Role of chronic kidney disease and risk factors in preeclampsia. Pregnancy Hypertension 2024; 37, 101146.
[15] S Marek-Iannucci, W Ullah, U Patricia, E Oliveros, Y Brailovsky, B Ramu and I Rajapreyar. Association of chronic kidney dysfunction and preeclampsia: Insights of the Nationwide Inpatient Sample. American Journal of Obstetrics & Gynecology MFM 2023; 5(6), 100928.
[16] R Vogtmann, J Heupel, F Herse, M Matin, H Hagmann, I Bendix, K Kräker, R Dechend, E Winterhager, R Kimmig, A Köninger and A Gellhaus. Circulating maternal sFLT1 (Soluble fms-Like Tyrosine Kinase-1) is sufficient to impair spiral arterial remodeling in a preeclampsia mouse model. Hypertension 2021; 78(4), 1067-1079.
[17] JH Jeong, U Ojha and YM Lee. Pathological angiogenesis and inflammation in tissues. Archives of Pharmacal Research 2021; 44(1), 1-15.
[18] J Torres-Torres, S Espino-y-Sosa, R Martinez-Portilla, H Borboa-Olivares, G Estrada-Gutierrez, S Acevedo-Gallegos, E Ruiz-Ramirez, M Velasco-Espin, P Cerda-Flores, A Ramirez-Gonzalez and L Rojas-Zepeda. A narrative review on the pathophysiology of preeclampsia. International Journal of Molecular Sciences 2024; 25(14), 7569.
[19] X Wang and B He. Endothelial dysfunction: Molecular mechanisms and clinical implications. MedComm 2024; 5(8), e651.
[20] Y Higashi. Roles of oxidative stress and inflammation in vascular endothelial dysfunction-related disease. Antioxidants 2022; 11(10), 1958.
[21] K Palm, C Cluver, E Langenegger, S Tong, S Walker, H Imberg, R Hastie and L Bergman. Circulating concentrations of pro-inflammatory cytokines in preeclampsia with varying disease severity. Pregnancy Hypertension 2024; 38, 101168.
[22] L Dobrek. An outline of renal artery stenosis pathophysiology - A narrative review. Life 2021; 11(3), 208.
[23] AT Taşkömür and Ö Erten. The role of cystatin C, neutrophil-lymphocyte ratio and platelet-lymphocyte ratio in the evaluation of kidney function in women with preeclampsia. Taiwanese Journal of Obstetrics and Gynecology 2021; 60(4), 615-620.
[24] M Ahda, I Jaswir, A Khatib, QU Ahmed and SNA Syed Mohamad. A review on Cosmos caudatus as A potential medicinal plant based on pharmacognosy, phytochemistry, and pharmacological activities. International Jornal of Food Properties 2023; 26(1), 344-358.
[25] Z Abidin, Aminah, R Razak and M Pratama. Phytochemical constituents, antioxidant and anti-inflammatory potentials of dechlorophyllized extract of kenikir leaves (Cosmos caudatus Kunth). Tropical Journal of Natural Product Research 2025; 9(5), 1958.
[26] AA Senjaya, NND Supariani and NM Sirat. Phytochemicals and toxicity of the extract from Cosmos caudatus leaves. Biomedical and Pharmacology Journal 2024; 17(3), 2055-2064.
[27] NND Supariani, NM Sirat, AA Senjaya, NK Ratmini, IK Ariana and AE Yulianti. Preference of the mouthwash ingredients wild cosmos leaf extract (Cosmos caudatus) among organoleptic test panelists. Journal of Health and Medical Sciences 2024; 7(4), 72-78.
[28] N Susanti, DLestari and R Anwar. Ekstraksi dan deteksi fitokimia kenikir (Cosmos caudatus Kunth.) asal Panjang Utara, Lampung (in Indonesian). AGRORADIX: Jurnal Ilmu Pertanian 2024; 8(1), 113-120.
[29] GM Sari, I Kusumawati, YA Arifandi and JB Swannjo. Effects of cosmos caudatus (Kenikir) antioxidant properties on bone metabolism marker in rat. Current Research in Physiology 2024; 7, 100128.
[30] W Hendrarti, AH Umar, R Syahruni, M Rafi and WA Kusuma. Deciphering the mechanism of action cosmos caudatus compounds against breast neoplasm: A combination of pharmacological networking and molecular docking approach with bibliometric analysis. Indonesian Journal of Science and Technology 2024; 9(2), 527-556.
[31] M Nacka-Aleksić, A Pirković, A Vilotić, Ž Bojić-Trbojević, MJ Krivokuća, F Giampieri, M Battino and D Dekanski. The role of dietary polyphenols in pregnancy and pregnancy-related disorders. Nutrients 2022; 14(24), 5246.
[32] M Ożarowski, PŁ. Mikołajczak, R Kujawski, K Wielgus, A Klejewski, H Wolski and A Seremak-Mrozikiewicz. Pharmacological effect of quercetin in hypertension and its potential application in pregnancy‐induced hypertension: Review of in vitro, in vivo, and clinical studies. Evidence-Based Complementary and Alternative Medicine 2018; 2018(1), 7421489.
[33] X Sun, S Zhang and H Song. Quercetin attenuates reduced uterine perfusion pressure -induced hypertension in pregnant rats through regulation of endothelin-1 and endothelin-1 type A receptor. Lipids in Health Disease 2020; 19(1), 180.
[34] Y Yin, J Xu, I Ilyas and S Xu. Bioactive flavonoids in protecting against endothelial dysfunction and atherosclerosis. In: CL Wainwright and VB Schini-Kerth (Eds.). Natural products as sources of novel drugs. Springer, Cham, Switzerland, 2025.
[35] F Fitriana, S Sulistyowati, D Indarto, S Soetrisno, I Nurwati and V Widyaningsih. Effect of kopyor coconut water on early-onset preeclampsia-like impairments in rats induced by L-nitro-arginine methyl ester. Pharmacia 2024; 71, 1-11.
[36] S Zeng, Z Liu, J Yin, S Li, M Jiang, H Yang and Y Long. Improvement in clinical features of L-NAME-induced preeclampsia-like rats through reduced SERPINA5 expression. Biomolecules 2023; 13(12), 1792.
[37] Y Wang, Y Gu, JS Alexander and DF Lewis. Preeclampsia status controls Interleukin-6 and Soluble IL-6 receptor release from neutrophils and endothelial cells: Relevance to increased inflammatory responses. Pathophysiology 2021; 28(2), 202-211.
[38] GJ Wang, Z Yang, J HuaI and QQ Xiang. Pravastatin alleviates oxidative stress and decreases placental trophoblastic cell apoptosis through IL-6/STAT3 signaling pathway in preeclampsia rats. European Review for Medical and Pharmacological Sciences 2020; 24, 12955-12962.
[39] M Rybak-Krzyszkowska, J Staniczek, A Kondracka, J Bogusławska, S Kwiatkowski, T Góra, M Strus and W Górczewski. From biomarkers to the molecular mechanism of preeclampsia - A comprehensive literature review. International Journal of Molecular Sciences 2023; 24(17), 13252.
[40] HM Sani, SZ Vahed and M Ardalan. Preeclampsia: A close look at renal dysfunction. Biomedicine & Pharmacotherapy 2019; 109, 408-416.
[41] L Zhang, L Xu, F Tang, D Liu, X Zhao, J Zhang, J Xia, J Wu, Y Yang, C Peng and H Ao. New perspectives on the therapeutic potential of quercetin in non-communicable diseases: Targeting Nrf2 to counteract oxidative stress and inflammation. Journal of Pharmaceutical Analysis 2024; 14(6), 100930.
[42] C Kuo, M Shevchuk, J Opfermann, T Guo, M Santoro, JP Fisher and PCW Kim. Trophoblast-endothelium signaling involves angiogenesis and apoptosis in a dynamic bioprinted placenta model. Biotechnology and Bioengineering 2019; 116(1), 181-192.