
Trends
Sci.
2026;
23(5):
11749
Effect of Natural Dietary Formulation LekCapp on the Development of Knee Joint Pain in A Rat Model of Monoiodoacetate-Induced Osteoarthritis
Orada
Sriwatananukulkit1,2,
![]() ,
Scarlett
Desclaux1,
,
Scarlett
Desclaux1,
![]() ,
Kasem Rattanapinyopituk3,
,
Kasem Rattanapinyopituk3,
![]() ,
,
Wanwisa
Himakhun4,
![]() ,
Yupaporn Sampaopan5,
,
Yupaporn Sampaopan5,
![]() ,
Sareeya Reungpatthanaphong6,
,
Sareeya Reungpatthanaphong6,
![]() ,
,
Surapote
Wongyai6,
![]() and Ruedee Hemstapat1,*
and Ruedee Hemstapat1,*
1Department of Pharmacology, Faculty of Science, Mahidol University, Bangkok 10400, Thailand
2Centre for Integrated Preclinical Drug Development, The University of Queensland, Queensland 4072, Australia
3Department of Pathology, Faculty of Veterinary Science, Chulalongkorn University, Bangkok 10330, Thailand
4Department of Pathology, Faculty of Medicine, Thammasat University, Pathum Thani 10120, Thailand
5College of Oriental Medicine, Rangsit University, Pathum Thani 12000, Thailand
6Cosmetic Science and Natural Products, College of Health and Wellness, Dhurakij Pundit University,
Bangkok 10210, Thailand
(*Corresponding author’s email: [email protected])
Received: 1 September 2025, Revised: 14 September 2025, Accepted: 21 September 2025, Published: 1 January 2026
Abstract
In recent years, the use of herbal supplements has risen markedly, with many products readily accessible through online retailers. Despite their growing popularity, scientific evidence supporting their efficacy and safety remains limited. In this study, we investigated the therapeutic potential of a natural product, LekCapp, in alleviating knee pain in a rat model of osteoarthritis (OA). OA knee pain was induced by a unilateral intra-articular injection of 2 mg monoiodoacetate (MIA). LekCapp was administered orally once daily for 14 days following MIA injection, following a preventive treatment protocol. Pain - related behaviors were assessed using weight-bearing distribution and von Frey tests. Histological analysis of knee joints was performed to evaluate potential cartilage-protective effects. Additionally, blood chemical analysis and histological examination of digestive organs were conducted to assess potential systemic toxicity. The results showed that LekCapp significantly reduced knee joint swelling and pain in MIA-treated rats compared with vehicle-treated controls. Although its protective effect against cartilage damage was modest, LekCapp administration preserved normal serum biochemical profiles, and histological evaluation revealed no abnormalities in the livers, kidneys, or stomach. These findings suggest that LekCapp may provide a safe and effective option for relieving OA-related knee pain.
Keywords: Herbal supplement, LekCapp, Monoiodoacetate, Knee pain, Osteoarthritis
Introduction
Knee joint pain has been associated with substantial healthcare expenses and impaired productivity for several decades [1]. Current treatments primarily involve pharmacological approaches, such as non-steroidal anti-inflammatory drugs (NSAIDs) and corticosteroid injections. However, these are often accompanied by serious side effects, including gastrointestinal ulceration and bleeding [2]. As a result,
there is growing interest in developing less invasive alternatives with improved safety profiles and long-term efficacy. Herbal supplements derived from medicinal plants and natural products have emerged as promising candidates for managing knee joint conditions such as osteoarthritis (OA) [3].
Since the supplement industry is not required to conduct pre-market safety and efficacy testing, the mechanisms of action for many products remain poorly understood [4]. Therefore, rigorous studies evaluating both the effectiveness and safety of dietary supplements are essential. To date, multi-ingredient herbal supplements have been increasing popularity. This study aimed to investigate the therapeutic potential of LekCapp, a patented multi-ingredient dietary supplement developed by Herbplus co., ltd., Thailand. According to the company, the formulation comprises hydrolyzed collagen, widely known for their role in restoring damaged cartilage [5] and extracts from various fruits and herbs known for their diverse pharmacological activities that may help alleviate OA pain, including acerola cherry, goji berry, turmeric, dong quai, reishi mushroom, ginseng and jiaogulan. Previous studies have demonstrated that several natural products possess antioxidant, anti-inflammatory, and chondroprotective properties relevant to osteoarthritis (OA). Acerola cherry [6] and goji berry [7] are rich in antioxidants that reduce reactive oxygen species, key contributors to age-related OA degeneration. Turmeric [8] and dong quai [9] have been extensively investigated in both preclinical and clinical studies for their antioxidant, anti-inflammatory, and chondro-protective properties. Reishi mushroom exhibits anti-nociceptive and anti-inflammatory activities in animal models of chronic inflammation [10], while ginseng extract has been reported to improve knee arthritis symptoms in clinical settings [11]. Additionally, jiaogulan has been reported to inhibit inflammatory mediators, attenuate pain, and restore cartilage integrity in animal models of inflammation [12]. Collectively, these findings support the potential of natural compounds in modulating OA progression and provide a rationale for exploring multi-component formulations in disease management.
Although LekCapp is readily available through online retailers in Thailand as a dietary supplement for individuals with symptomatic knee pain, there is limited scientific evidence supporting its efficacy in relieving knee joint pain under controlled experimental conditions. To address this gap, we evaluated knee joint pain-related behaviour, including nociceptive pain and mechanical allodynia, common and challenging symptoms of neuropathic pain, alongside structural joint pathology, using the well-established monoiodoacetate (MIA) model of knee joint pain in rats. In addition, biochemical blood analysis and histological examination of digestive organs were performed to assess potential systemic toxicity. The findings from this study will provide evidence-based support for the use of this supplement, enabling the general public, including those without a scientific background, to better understand its potential benefits and use it appropriately.
Materials and methods
Chemicals and reagents
LekCapp (patented formulation, as shown in Table 1; manufacturer: Herb Plus Co., Ltd.; batch number: 01/02/2021) was used as provided. The detailed composition is as declared by the manufacturer (patent number: 2103000583). Only the characterization of turmeric was performed in this study. HPLC-grade acetonitrile was purchased from Honeywell Burdick & Jackson (Muskegon, MI, USA). Glacial acetic acid and methanol were purchased from Merck (Darmstadt, Germany). HPLC-grade water was obtained from RCI Labscan Limited (Bangkok, Thailand). Curcumin primary standard (Product No. ASB-00003926), demethoxycurcumin primary standard (Product No. ASB-00004230), and bisdemethoxycurcumin primary standard (Product No. ASB-00004231) were obtained from ChromaDex (Irvine, CA, USA). MIA was purchased from Sigma-Aldrich (St Louis, MO, USA). All other reagents were of analytical grade unless otherwise noted. Celecoxib was obtained from Siam Pharmaceutical Co., Ltd. (Bangkok, Thailand). Methylcellulose was purchased from S. Tong chemicals Co., Ltd. (Bangkok, Thailand). Neutral buffered formalin and ethylenediaminetetraacetic acid (EDTA) were obtained from Ajax Finechem Pty., Ltd (New South Wales, Australia).
Table 1 The composition of LekCapp as described by the product company.
Ingredients |
Scientific names |
Extracted from |
Ratio (%) |
Supplier names |
Collagen |
Hydrolyzed Collagen |
Fish |
71.54 |
CNABIOTECH Co., Ltd, Korea |
Acerola cherry |
Malpighia emarginata |
Fruit |
7.43 |
Hunan Huacheng Biotech Inc., Hunan, China |
Turmeric |
Curcuma longa |
Rhizome |
6.68 |
CAVACURMIN Curcumin complex, Adrian, MI, USA |
Dong quai |
Angelica sinensis |
Root |
3.96 |
Fortress Hilltop LTD., Kowloon, Hongkong |
Jiaogulan |
Gynostemma pentaphyllum |
Leaf |
3.71 |
Hunan Huacheng Biotech Inc., Hunan, China |
Goji berry |
Lycium barbarum |
Fruit |
3.71 |
Guilin Layn Natural Ingredients Corp., Guilin, China |
Reishi mushroom |
Ganoderma lucidum |
Mushroom |
2.23 |
Hunan Huacheng Biotech Inc., Hunan, China |
Korean ginseng |
Panax ginseng |
Root |
0.74 |
Hunan Huacheng Biotech Inc., Hunan, China |
Composition of LekCapp
LekCapp is a commercially available dietary supplement in capsule form, approved for human consumption in Thailand. The composition of LekCapp is outlined in Table 1, based on the product description provided by the manufacturer.
Animals
Forty-nine male Wistar rats (6-weeks of age, 160 - 180 g) were obtained from Nomura Siam International Co., Ltd., Bangkok, Thailand. The animals were housed in an Association for the Assessment and Accreditation of Laboratory Animal Care (AAALAC) accredited facility under a 12-hour light/dark cycle, with a temperature of 22 ± 1 °C and humidity ranging from 30% to 70%. They were provided access to standard commercial food and water ad libitum. Prior to initiation of experiments, the rats were acclimatized for a minimum of 5 days. All animal welfare and experimental procedures adhered to the ARRIVE guidelines and were approved by the Institutional Animal Care and Use Committee (IACUC) of the Faculty of Science, Mahidol University (Approval No: MUSC64-003-552).
Knee pain model establishment and animal grouping
Knee joint pain was induced under anesthesia using inhaled isoflurane (Attane™; 5% for induction; 2% - 3% for maintenance). An intra-articular (i.a.) injection of 2 mg monoiodoacetate (MIA) dissolved in 50 μL of 0.9% sterile saline (40 mg/mL) was administered into the intra-articular space of the right knee joint. The injection was performed through the intrapatellar ligament using a 26-G needle attached to a SGE syringe (250 µL). Meanwhile, control rats were administered an i.a. injection of 50 μL sterile saline, while the contralateral knee joint remained untreated. As illustrated in Figure 1, animals were randomly assigned to 6 experimental groups by an independent researcher using a simple randomization protocol to ensure unbiased allocation. The researcher responsible for group assignment was blinded to the treatment conditions, and allocation remained concealed until all behavioral data had been recorded and finalized to minimize selection and observer bias. Animals (≥ 6 animals per group) were assigned to 6 experimental groups as follows: (1) Saline + vehicle (n = 6), (2) MIA + vehicle (n = 7), (3) MIA + 10 mg/kg celecoxib (n = 8), (4) MIA + 200 mg/kg LekCapp (n = 6), (5) MIA + 400 mg/kg LekCapp (n = 12), (6) MIA + 800 mg/kg LekCapp (n = 10). Sterile distilled water was used as the vehicle for oral administration. Celecoxib served as the positive control due to its well-documented pain-relieving and anti-inflammatory properties [13]. The celecoxib dose was selected based on the study by Kockaya et al. [14]. The LekCapp doses were determined using the human equivalent dose (HED) calculation method described by Nair and Jacob [15], based on the recommended adult dose of approximately 2,500 - 3,000 mg per day.

Figure 1 Schematic diagram summarizing the experimental design. MIA or saline was administered via intra-articular injection, followed by daily oral treatment with vehicle, celecoxib, or LekCapp for 14 days. Behavioral and histological assessments were conducted at specified time points to evaluate joint swelling, pain-related behaviors, and tissue integrity. I.A.: Intra-articular.
Experimental design
Baseline measurements, including body weight, knee joint width, hind limb weight distribution, and von Frey filament tests, were conducted prior to MIA injection. One day after MIA-injection, animals were orally administered vehicle (sterile distilled water), celecoxib or LekCapp once daily for 14 consecutive days. LekCapp was prepared in sterile distilled water, while celecoxib was suspended in 0.5% methylcellulose. Rat body weights were measured every other day, and knee joint width was assessed on days 1, 3, 5, 7, and 13 post-MIA injection. The von Frey filament test was performed on days 2, 6, 10, and 14 post-MIA injection, while hind limb weight distribution was evaluated on days 3, 7, and 13 post-MIA injection. After 14 days of subacute oral dosing, rats were euthanized by intraperitoneal injection of an overdose of thiopental (100 mg/kg; Anesthal®, Scott-Edil Pharmacia Ltd., Solan, India). Terminal blood samples were collected via the posterior vena cava. The knee joints of the injured hind limb were harvested for further evaluation of the cartilage-protective effect of the test compounds. Additionally, the stomach, liver, and kidneys were collected for safety assessment of the test compounds.
Assessment of pain-like behaviors
Pain-like behaviors were assessed using the hind limb weight bearing distribution (HLWD) test and von Frey paw withdrawal threshold measurements at various time points. The HLWD test was conducted using an incapacitance meter (Columbus Instruments), as previously described [16]. Briefly, animals were allowed to acclimate in an acrylic chamber, with each paw placed on a separate sensor pad to record weight distribution. The weight (g) borne by the left (contralateral) and right (ipsilateral) hind limbs was measured in 10 consecutive readings per rat, and the average value was calculated. Mechanical allodynia was assessed using von Frey filaments (Touch Test® Sensory Evaluator), following the methodology previously described by Chaplan et al. [17]. Briefly, rats were placed individually in wire mesh cages and acclimatized for at least 15 min to minimize stress-induced variability. Calibrated von Frey filaments ranging from 2 to 16 g were applied perpendicularly to the plantar surface of the ipsilateral hind paw, starting with the lowest force and gradually increasing force until a withdrawal response (paw withdrawal, licking, or flinching) was observed. The paw withdrawal threshold (PWT) was defined as the lowest filament force (g) that elicited a response, based on 3 independent measurements taken at 5-min intervals. All testing was performed by the same experimenter, who was blinded to the treatment groups. Detailed methodology for each test can be found in Supplemental method 1.
Knee joint width measurement
Knee joint swelling (inflammation) was measured on days 0, 1, 3, 5, 7, and 13 post-MIA injection using a digital Vernier caliper (resolution: 0.01 mm, SV-03-050, E-Base Measuring Tools Co., Taiwan). This measurement served as an indicator of joint inflammation, as previously described [18]. Detailed methodology is provided in Supplemental method 2.
Histopathological examination
As described in Supplemental method 3, coronal sections of the knee joints were stained with hematoxylin and eosin (H & E) and safranin-O/fast green to assess general morphology and proteoglycan content, respectively. The severity of articular cartilage damage was blindly quantified by 2 independent observers using the Mankin histologic scoring system [19]. H & E staining of the stomach, liver and kidneys was also performed, with evaluation based on histological criteria, including gastric lesion scoring at the forestomach (non-glandular keratinized epithelium) and glandular regions (fundus and pylorus) [20] (Tables S1 and S2). For a semi-quantitative comparison of structural changes, abnormalities in liver and kidney tissue sections were graded on a scale from 0 (normal structure) to 3 (severe pathological changes), as previously described [21] (Tables S3 and S4). Images were captured using a Nikon Eclipse Ci light microscope equipped with a Nikon DS-Ri2 camera, and image acquisition was performed with NIS-Elements BR software (version 5.21.00).
Biochemical blood analysis
Following terminal blood collection, serum was obtained by centrifugation at 3,000 rpm for 10 min at 4 °C. The serum was then sent to the Vet Central Lab (Bangkok, Thailand), a commercial testing laboratory, for analysis of potential liver and kidney toxicity. Liver function parameters assessed included aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), albumin, and total protein. Kidney function was evaluated by measuring blood urea nitrogen (BUN) and creatinine levels.
Reversed-phase high-performance liquid chromatography (RP-HPLC) analysis of LekCapp
RP-HPLC was conducted to identify the presence of curcumin, and its derivatives, following methods described previously [22,23]. Detailed methodological procedures are provided in Supplemental method 4.
Statistical analysis
Data are presented as the mean ± standard error of the mean (SEM) for each measurement within each treatment group. All graphs and statistical analyses were performed using GraphPad Prism version 6.0.1 (GraphPad Software, La Jolla, CA, USA). Sample sizes were determined based on our previous studies, which indicated that at least 6 animals per group would provide sufficient statistical power. The Shapiro-Wilks test was used to assess the normality of the data. Scoring parameters and biochemical blood analysis were evaluated using non-parametric tests, including the Kruskal-Wallis test with Dunn’s post hoc analysis. Pain behavioral tests and knee joint width measurements were analyzed with a 2-way analysis of variance (2-way ANOVA), followed by post hoc Bonferroni multiple comparison tests. A p-value less than 0.05 was considered statistically significant.
Results and discussion
LekCapp prevented the development of pain-like behaviors in MIA-induced knee pain
In the weight-bearing distribution test (Figure 2(a)), a marked reduction in the mean% hindlimb weight distribution (HLWD) was observed at 3 days post-MIA injection in all MIA-treated rats, while no change occurred in the vehicle-treated saline-injected group. At this time-point, the mean ± SEM% HLWD in the 10 mg/kg celecoxib-treated group decreased from 50.37 ± 0.15 on day 0 to 33.82 ± 2.67 on day 3 (p < 0.01) compared to a decline from 50.13 ± 0.09 to 26.28 ± 1.41 in the vehicle-treated MIA group. However, this significant anti-nociceptive effect of celecoxib was transient, lasting only up to 7 days post-MIA injection. By day 13 post-MIA injection, rats treated with LekCapp at 400 mg/kg (41.50 ± 0.88; p < 0.0001) and 800 mg/kg (43.24 ± 1.15; p < 0.0001) showed significantly improved %HLWD compared to the vehicle-treated MIA-injected group (33.42 ± 1.61). This improvement was not significantly different from that observed in the celecoxib-treated group (38.10 ± 2.50; p > 0.05).
As shown in Figure 2(b), similar to the weight-bearing distribution test, mechanical allodynia of the ipsilateral hind limb was markedly reduced in all MIA-treated groups, except the vehicle-treated saline-injected group, by 2 weeks post-MIA injection. Celecoxib (10 mg/kg/day) exhibited the most significant anti-allodynic effect, which persisted for up to 10 days before diminishing. Notably, after 14 days of treatment, paw withdrawal thresholds (PWTs; mean ± SEM) were significantly improved in rats receiving LekCapp at 400 mg/kg/day (9.61 ± 0.76; p < 0.05) and 800 mg/kg/day (10.33 ± 0.77; p < 0.01), compared to the vehicle-treated MIA group (7.14 ± 0.40). However, these values were not significantly different from those observed in the celecoxib-treated MIA group (9.00 ± 1.13; p > 0.05).
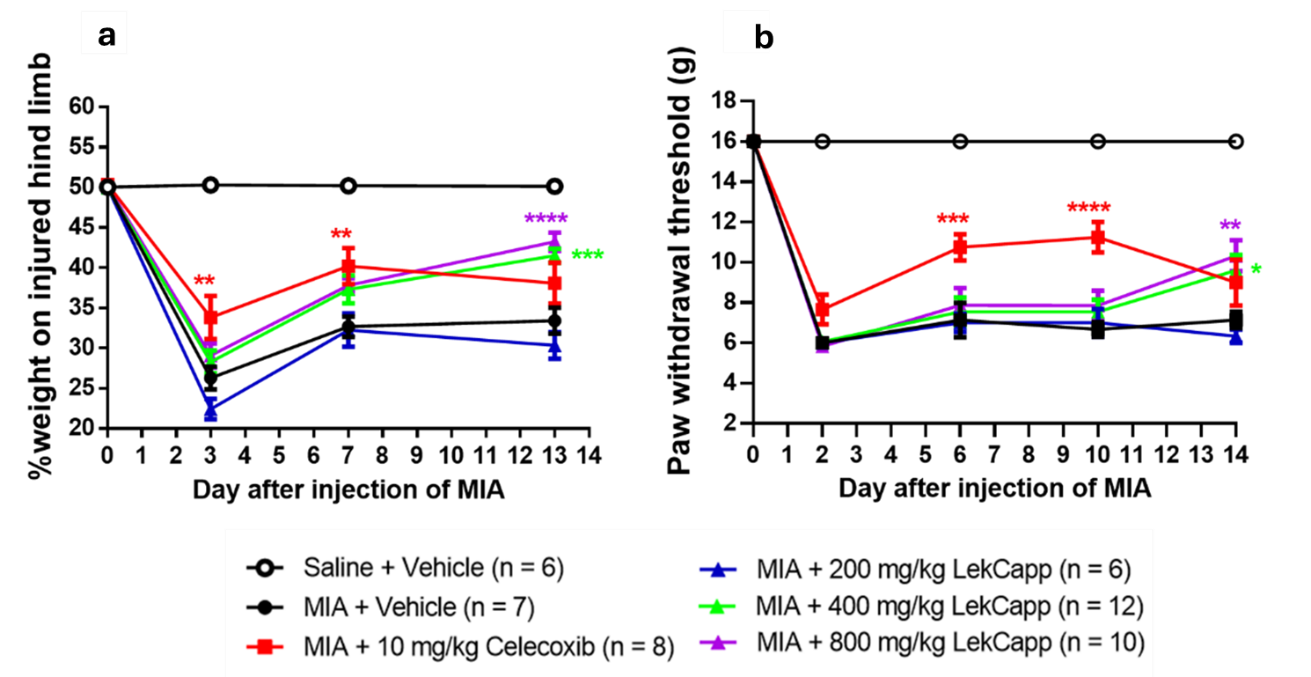
Figure 2 Orally administered LekCapp (400 and 800 mg/kg/day) according to a prevention protocol attenuated the development of pain-like behaviors in MIA-injected rats over a 14-day treatment period. (a) Mean percentage of hind limb weight-bearing distribution and (b) Mean paw withdrawal threshold values measured in the ipsilateral hind limb of MIA-injected rats. Data are expressed as mean ± SEM. Statistical significance was determined using ordinary 2-way ANOVA, followed by post hoc Bonferroni multiple comparison tests. p < 0.05 (*), p < 0.01 (**), p < 0.001 (***) and p < 0.0001 (****) were considered statistically significant.
LekCapp reduced MIA-induced knee swelling in rats.
One day post-MIA injection, all experimental groups, excepted the vehicle-treated saline-injected group, exhibited maximal knee swelling (Figure 3). By day 3 post-MIA injection, a downward trend in mean ± SEM change (Δ) in knee width was observed in all MIA-injected groups. The most pronounced reduction occurred in the celecoxib-treated group (10 mg/kg), with knee swelling decreasing from 2.76 ± 0.18 on day 1 to 0.55 ± 0.12 mm on day 3 (p < 0.0001), compared to the vehicle-treated MIA group (3.27 ± 0.31 to 2.03 ± 0.33 mm). Similarly, rats treated with daily oral LekCapp at 400 mg/kg and 800 mg/kg showed significant reductions in knee swelling from day 1 to day 3: 400 mg/kg: 2.75 ± 0.28 to 0.97 ± 0.16 mm (p < 0.0001) and 800 mg/kg: 2.67 ± 0.22 to 1.12 ± 0.14 mm (p < 0.01). By day 13 post-MIA injection, no significant differences in knee swelling were observed among the experimental groups and the vehicle-treated MIA group. The vehicle-treated saline-injected group maintained consistent Δ knee width throughout the study, showing no signs of inflammation.
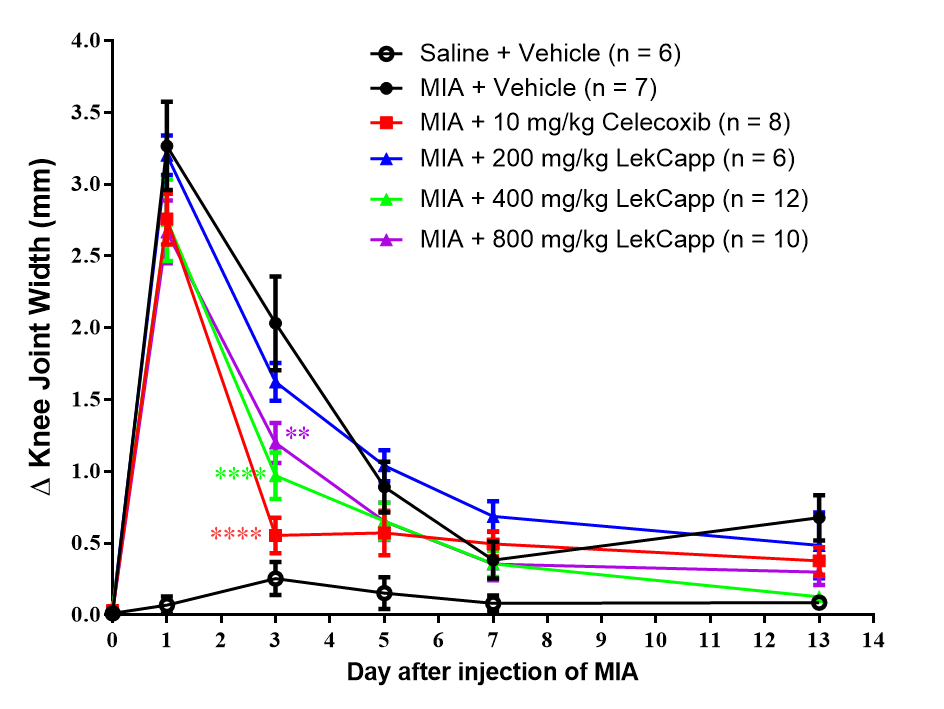
Figure 3 LekCapp (400 and 800 mg/kg/day), administered according to a prevention protocol, significantly reduced joint swelling in MIA-injected rats by day 3 post-dosing. Changes in knee joint width (Δ knee width) were measured to assess joint swelling. Data are expressed as mean ± SEM. Statistical analysis was performed using ordinary 2-way ANOVA, followed by post hoc Bonferroni multiple comparison tests. p < 0.01 (**), and p < 0.0001 (****) were considered statistically significant.
LekCapp had a minor effect on cartilage damage in MIA-induced osteoarthritis rats
At 14 days post-injection, vehicle-treated saline-injected rats displayed intact articular cartilage with normal histological architecture (Figures 4(a) and 4(b)). In contrast, vehicle-treated MIA-injected rats exhibited severe pathological changes, including cartilage surface tears, cleft extending to the transitional zone, subchondral bone destruction, loss of safranin-O staining, and chondrocyte hypocellularity (Figures 4(c) and 4(d)). Treatment with celecoxib (10 mg/kg) did not prevent the progression of cartilage degeneration (Figures 4(e) and 4(f)). Notably, rats treated with LekCapp at 400 mg/kg (Figures 4(g) and 4(h)) and 800 mg/kg (Figures 4(i) and 4(j)) showed histological improvement compared to the vehicle-treated MIA group, although the effect was modest. Quantitatively, the mean ± SEM Mankin score for the vehicle-treated MIA group (9.81 ± 0.61) was significantly higher than that of the saline-injected control group (1.58 ± 0.37; p < 0.05) (Figure 4(k)). However, no significant difference was found between the 800 mg/kg LekCapp group (10.07 ± 0.64) and the vehicle-treated MIA group (9.81 ± 0.61), indicating limited chondroprotective effects. Due to the minimal impact observed at higher doses, histological analysis was not conducted for the lower dose of LekCapp (200 mg/kg).
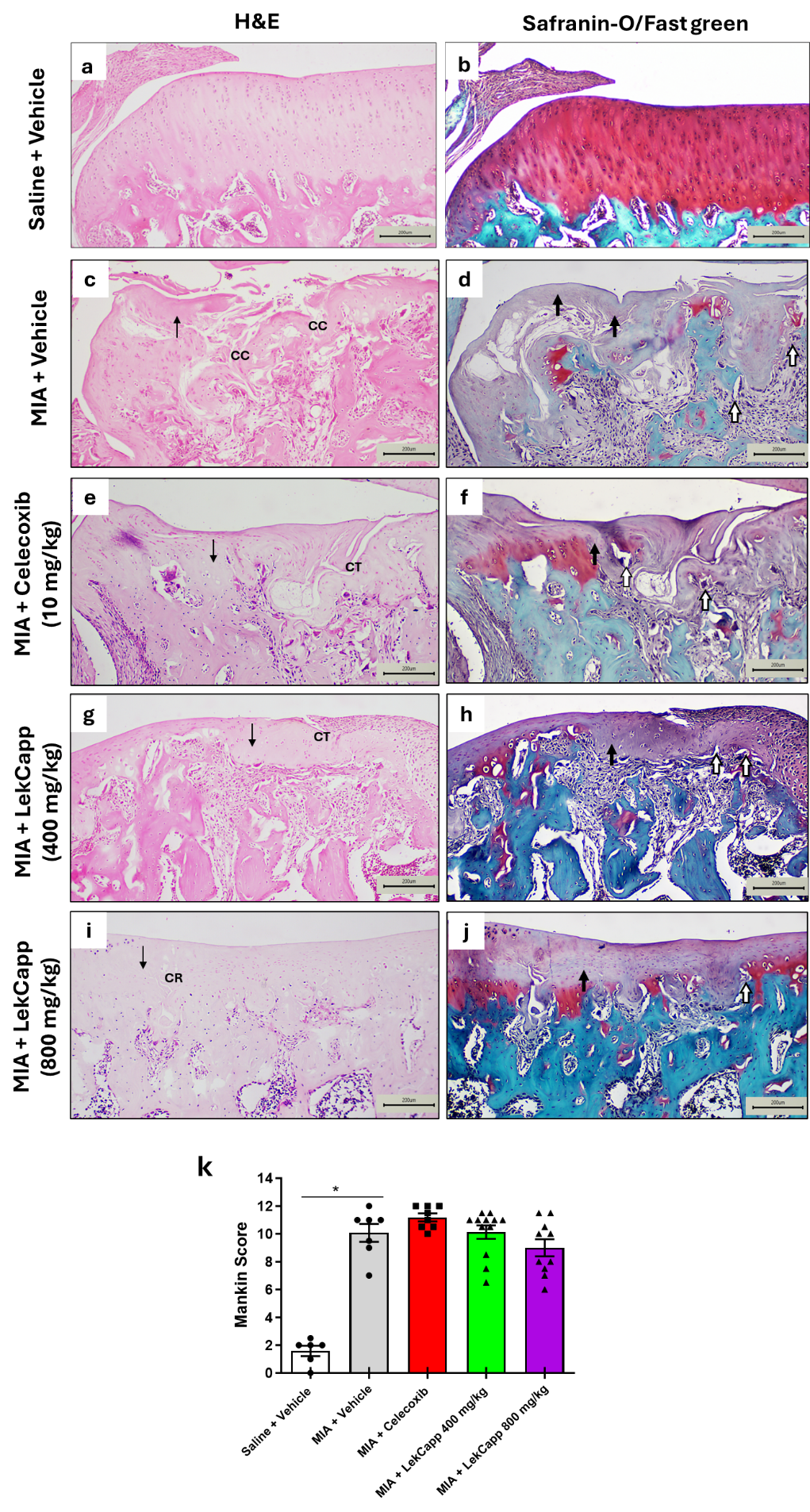
Figure 4 Oral administration of LekCapp (400 and 800 mg/kg/day), following a prevention protocol, did not significantly improve cartilage degeneration in MIA-induced OA rats. Representative histological images of articular cartilage stained with H & E and Safranin O/fast green from the following groups are shown: (a,b) Saline + vehicle (n = 6), (c,d) MIA + vehicle (n = 7), (e,f) MIA + celecoxib 10 mg/kg (n = 8), (g,h) MIA + LekCapp 400 mg/kg (n = 12) and (i,j) MIA + LekCapp 800 mg/kg (n = 10). Scale bars 200 µm. (k) Quantification of cartilage damage using the Mankin score. Thin black arrows indicate chondrocyte hypocellularity; thick black arrows indicate severe reduction of Safranin-O staining; white arrows indicate blood vessel crossing the tidemark. CT: Cleft to transitional zone; CC cleft to calcified zone; CR cleft to radial zone. Data are expressed as mean ± SEM. Statistical significance was determined using the Kruskal-Wallis test with Dunn’s post hoc comparison. p < 0.05 (*) was considered statistically significant.
Subacute oral toxicity study of LekCapp
No behavioral abnormalities, clinical signs of toxicity, or mortality were observed in any treatment groups during the 14-day administration period. As shown in Figure 5, the mean ± SEM body weight gain in all experimental groups remained comparable to that of the age-matched vehicle-treated saline-injected and MIA-injected control groups throughout the dosing period. Furthermore, there were no significant differences (p > 0.05) in biochemical markers of liver and kidney function between the vehicle-treated saline group and the MIA-injected groups treated with vehicle, 10 mg/kg celecoxib (MIA + Celecoxib), or 800 mg/kg LekCapp (MIA + 800 mg LekCapp), as presented in Table 2. These findings indicate that oral administration of LekCapp, even at the highest tested dose, was well-tolerated and did not induce systemic toxicity under the conditions of this study.
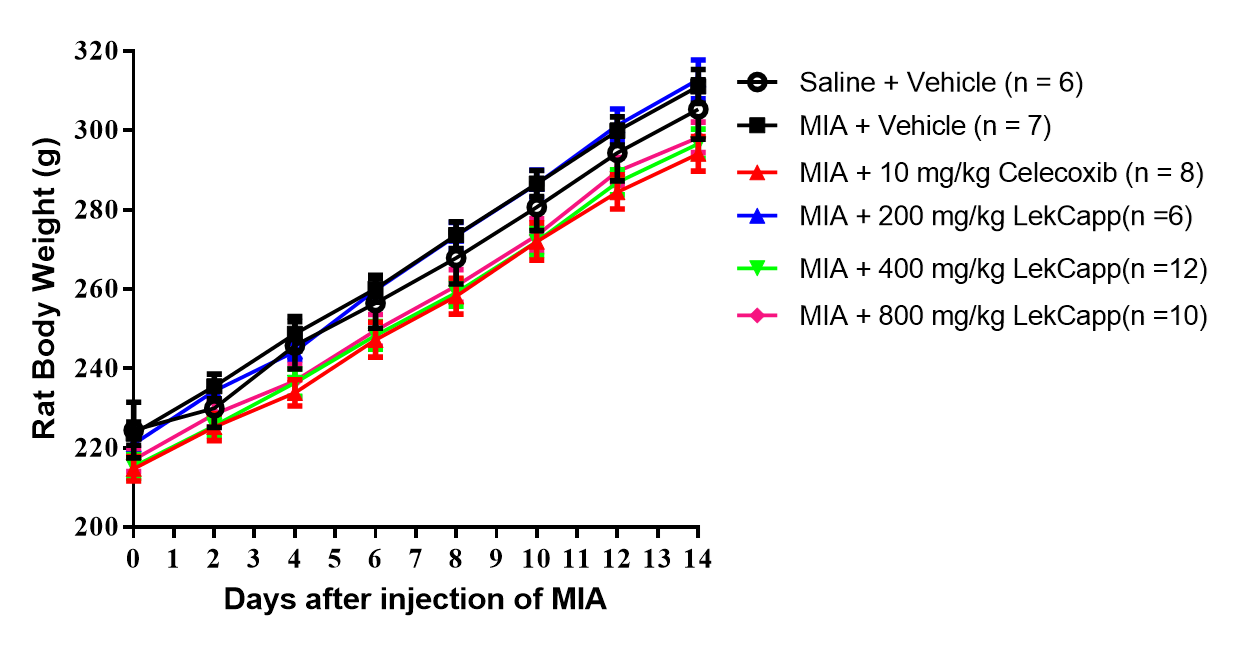
Figure 5 Oral administration of LekCapp did not affect body weight in rats. No significant changes in body weight were observed among the treatment groups throughout the 14-day dosing period. Data are expressed as mean ± SEM.
Table 2 Biochemical blood analysis.
Biochemical parameters |
Liver function test |
Kidney function test |
|||||
Groups |
ALT (U/L) |
AST (U/L) |
ALP (U/L) |
Albumin (g/dL) |
Total Protein (g/dL) |
Creatinine (mg/dL) |
BUN (mg/dL) |
Saline + Vehicle (n = 6) |
37.2 ± 3.7 |
102.5 ± 11.3 |
260.0 ± 44.9 |
2.7 ± 0.1 |
5.5 ± 0.3 |
0.7 ± 0.0 |
21.8 ± 1.3 |
MIA + Vehicle (n = 7) |
35.9 ± 3.8 |
92.7 ± 8.6 |
248.1 ± 27.2 |
2.6 ± 0.1 |
5.3 ± 0.1 |
0.6 ± 0.0 |
22.0 ± 0.7 |
MIA + Celecoxib (10 mg/kg) (n = 8) |
33.4 ± 2.2 |
84.1 ± 4.1 |
217.3 ± 13.4 |
2.6 ± 0.1 |
5.5 ± 0.2 |
0.6 ± 0.0 |
20.3 ± 1.5 |
MIA + LekCapp (800 mg/kg) (n = 10) |
34.7 ± 2.6 |
92.5 ± 6.0 |
211.0 ± 11.6 |
2.6 ± 0.1 |
5.4 ± 0.1 |
0.6 ± 0.0 |
22.7 ± 0.8 |
Histopathological evaluation of gastrointestinal tract, liver, and kidneys
As shown in Figure 6(a), no significant histological changes were observed in the forestomach, fundic, or pyloric regions of the stomach across all treatment groups. Specifically, there was no evidence of epithelial erosion, hemorrhage, or inflammation (histological score = 0; Table S5), indicating the absence of gastric mucosal toxicity. Similarly, no significant histopathological alterations were detected in the liver or kidneys of any group (score = 0), as indicated by the absence of hepatic degeneration, necrosis, and inflammatory cell infiltration in the liver (Figure 6(b); Table S6), and the lack of tubular degeneration, necrosis, or renal hemorrhage in the kidneys (Figure 6(c); Table S7). Mild findings such as fatty infiltration of hepatocytes, tubular cast, and crystals deposit were occasionally observed (score = 1) in a few animals across all experimental groups, suggesting non-specific background changes rather than treatment-related toxicity.
In this study, histopathological evaluations were initially conducted on tissue samples from animals treated with LekCapp at 400 and 800 mg/kg. The corresponding results for the 400 mg/kg group are provided in Tables S5 and S6. However, only representative histological images from the 800 mg/kg group are presented in the main figures. No abnormal morphology or histological lesions were observed in any tissues examined from animals receiving either 400 or 800 mg/kg of LekCapp. Based on these findings, histopathological assessment was not performed for the lower dose group (200 mg/kg).
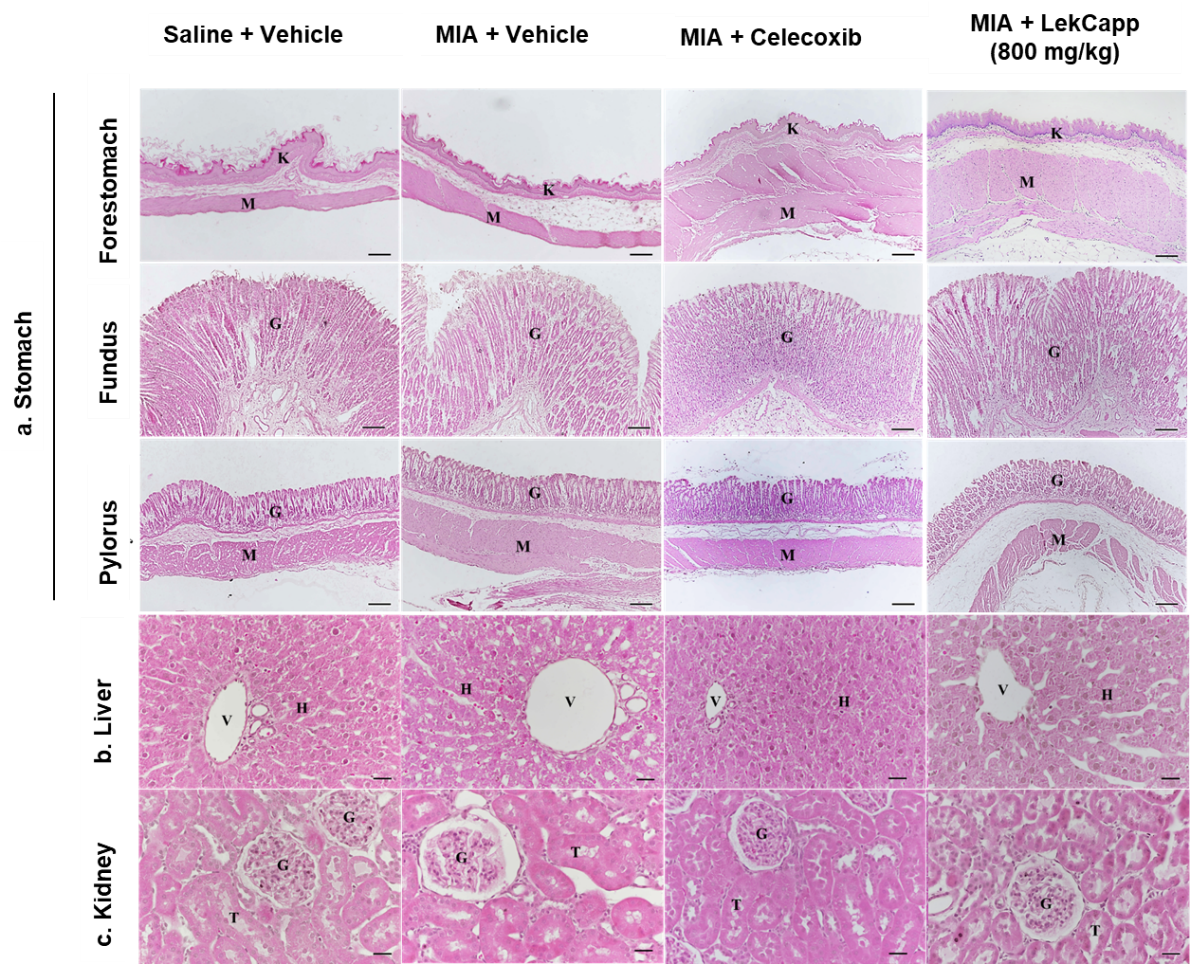
Figure 6 LekCapp did not induce significant histopathological changes in the stomach, liver or kidneys of MIA-injected rats treated with LekCapp or celecoxib, compared to vehicle-treated saline-injected controls. (a) Representative histological images of the rat stomach, showing the forestomach and glandular regions. K = Keratinized epithelium; M = Muscularis mucosae; G = Glandular epithelium. Scale bar = 50 μm. (b) Histological findings of liver tissue. H = Hepatic parenchyma; V = Hepatic vessel. and (c) Histological findings of kidney tissue. G = Renal glomerulus; T = Renal tubules. Scale bar at 20 μm.
Identification of the constituents of LekCapp
To explore the potential bioactive compounds responsible for the observed pain-relieving effects of LekCapp, we conducted a preliminary phytochemical investigation focusing on its turmeric content. This focus was based on extensive prior research demonstrating the analgesic and anti-inflammatory properties of curcumin in OA and neuropathic pain models [24-28]. Chromatographic fingerprinting and quantification of the 3 major curcuminoids, including bisdemethoxycurcumin, demethoxycurcumin and curcumin, were performed using reverse-phase high-performance liquid chromatography (RP-HPLC). A gradient elution system comprising 2% acetic acid in water and acetonitrile was employed as the mobile phase, enabling the separation of all 3 curcuminoids in a single run within an acceptable time frame. The presence of these curcuminoids was confirmed in the LekCapp formulation, with the concentration ratio of bisdemethoxycurcumin: demethoxycurcumin: curcumin measured at 1.56:0.49:1.00. The HPLC chromatogram of the LekCapp capsules is shown in Figure 7.
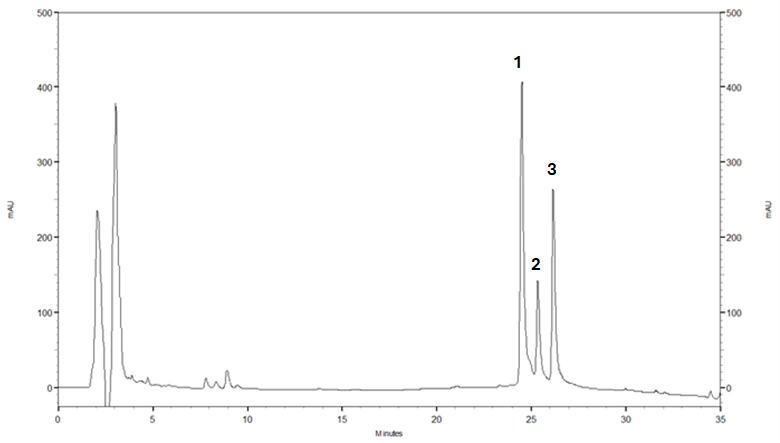
Figure 7 Representative HPLC chromatogram of LekCapp capsules. Each capsule contained 3 known marker compounds from turmeric: Bisdemethoxycurcumin (peak 1), demethoxycurcumin (peak 2), and curcumin (peak 3). The quantities of these compounds were 1.53 mg% relative standard deviation (%RSD = 1.53), 1.24 mg (%RSD = 1.62) and 2.50 mg (%RSD = 1.53), respectively (n = 6). The average total weight per capsule was 446.79 mg. HPLC conditions: Stationary phase: VDSpher PUR 100 C18-E (250×4.6 mm2 i.d., 5 µm); mobile phases: A = 2% (v/v) acetic acid in water, B = B: Acetonitrile; gradient elution: 40% B for 15 min, ramped to 100% B at 20 min; flow-rate: 1 mL/min; detection wavelength: 254 nm; injection volume: 20 µL.
Natural products are increasingly recognized as dietary supplements for managing knee joint pain conditions such as OA due to their potential to protect cartilage, reduce inflammation, and promote cartilage repair with minimal side effects [29]. In this study, we investigated whether LekCapp could alleviate knee joint pain in a well-established rat model of OA induced by MIA. We hypothesized that LekCapp would reduce knee joint swelling and pain during the acute inflammatory phase following MIA injection. Our findings demonstrated that daily oral administration of LekCapp (400 - 800 mg/kg/day) significantly reduced knee swelling and attenuated pain-related behaviors, including mechanical allodynia and impaired weight-bearing, in MIA-injected rats. These effects were most pronounced during the early phase, supporting the hypothesis that LekCapp may mitigate acute inflammatory responses associated with OA.
The MIA-induced OA model is well-established and widely used for studying joint pain and evaluating therapeutic interventions due to its ease of induction, reproducibility, and ability to replicate key features of human OA. Importantly, it captures both inflammatory and neuropathic pain components, along with progressive cartilage degradation, which are hallmarks of clinical OA [30]. Therefore, this model provides a relevant platform for assessing the anti-inflammatory and analgesic efficacy of LekCapp.
Previous studies have shown that MIA injection induces extensive infiltration of macrophages into the infrapatellar fat pad/synovium as early as day 1 post-injection, triggering a robust inflammatory response [31]. This inflammatory activity corresponds with the pronounced joint swelling observed on day 1 in our study. Between day 3 and 7 post-injection, further accumulation of inflammatory cells and extracellular matrix components contributes to the progression of joint pathology. Given this timeline, LekCapp administration was initiated on day 1 to target the acute phase of inflammation. Our results support this approach, as a significant reduction in knee joint swelling was observed in LekCapp-treated rats by day 3, compared to vehicle-treated MIA controls. These findings suggest that LekCapp may exert anti-inflammatory effects during the early phase of OA pathogenesis. This observation is consistent with previous studies reporting that several components of LekCapp possess anti-inflammatory properties. For example, Zhang and Zeng [32] reported that daily administration of curcumin, a major component of turmeric, attenuated knee joint swelling in the monosodium iodoacetate (MIA)-induced OA after 14 consecutive days of treatment, which aligns with the results of our study. In addition, ginsenosides, another ingredient in the formulation, dose-dependently reduced MIA-induced knee joint swelling [33]. Joint swelling is a clinical feature of OA, reflecting synovitis caused by synovial thickening or effusion and serving as a clinical indicator of inflammation. Mechanistically, several studies have unveiled that mechanism underlying this pathogenesis is linked to elevated levels of tumor necrosis factor-a (TNF-a), interleukin-6 (IL-6) and interleukin-1β (IL-1β) in the knee synovium and capsule [34]. Furthermore, it is well established that inflammation is closely associated with oxidative stress. Acerola cherry has been previously reported to exert strong antioxidants and anti-inflammatory effects by reducing the secretion of pro-inflammatory cytokines, including IL-1β, IL-6 and TNF-α [35]. Given these findings, further studies are warranted to confirm the underlying mechanism of LekCapp. Specifically, examining pro-inflammatory cytokines and other molecular markers at early time points (e.g., days 1 and 3 post-MIA injection) would help clarify the pathways through which LekCapp exerts its effects.
Using the static weight-bearing test to assess nociceptive pain and von Frey filaments to evaluate mechanical allodynia, our key findings demonstrate that daily oral administration of LekCapp (400 and 800 mg/kg) dose-dependently attenuated pain-related behaviors in the ipsilateral hind paw of MIA-injected rats over a 14-day period. These results suggest that prolonged administration of LekCapp is necessary to achieve sustained pain relief in the MIA-induced knee joint pain model. In contrast, the analgesic effect of celecoxib (10 mg/kg/day) declined after the first week of treatment, showing reduced efficacy beyond day 10 post-MIA injection. This observation aligns with previous reports indicating that NSAIDs are ineffective in the later stages of MIA-induced OA. For instance, Jiang et al. [36] reported that NSAIDs failed to alleviate pain-related behavior after 10 days post-MIA injection. Similarly, Fernihough et al. [37] found that diclofenac and paracetamol were only effective within the first 3 days post-injection. This decline in NSAIDs efficacy over time can be attributed to the evolving pathophysiology of the MIA model. While the early phase is dominated by acute inflammation, the later stages are characterized by progressive cartilage degradation and the development of a neuropathic pain, driven in part by central sensitization mechanisms in the spinal dorsal horn [38]. As such, conventional anti-inflammatory drugs like NSAIDs may fail to address the chronic and neuropathic dimensions of OA pain, highlighting the need for alternative or adjunct therapies, such as LekCapp that may exert broader or more sustained analgesic effects.
Previous studies have reported that collagen derivatives, the main component of LekCapp, as indicated by the manufacturer, may confer beneficial effects in OA and promote cartilage repair. Based on this, we evaluated the potential of LekCapp to prevent or reverse articular cartilage degradation in the MIA-induced OA model. However, our results revealed only a modest cartilage-protective effect. The explicitly underlying mechanism of collagen, the major component of Lekcapp, may be partly explained by previous studies showing that hydrolyzed collagens exhibit high bioavailability, enabling absorption into the bloodstream and accumulation in cartilage tissue, where they stimulate chondrocytes to promote extracellular matrix synthesis [39]. However, although histological examination showed some improvements in cartilage structure in LekCapp-treated rats compared to vehicle-treated MIA-controls, these differences did not achieve statistical significance. Specifically, persistent features of cartilage degeneration, including chondrocyte hypocellularity, proteoglycan loss, and vascular penetration through the tidemark, were still evident in the LekCapp-treated groups. These findings suggest that the dose and duration of treatment employed in this study may have been insufficient to produce measurable structural restoration. Cartilage repair is inherently a slow and complex biological process; therefore, a longer treatment period and/ or higher dosage of LekCapp may be necessary to achieve significant histological improvements. We propose that future studies incorporate extended treatment durations and investigate additional dosing regimens to more accurately assess LekCapp’s potential as a disease-modifying therapy for osteoarthritis.
To assess the gastrointestinal and systemic safety of LekCapp at therapeutic doses, we evaluated blood biochemical parameters alongside histological examination of major internal organs. Across all treatment groups, animals remained in good general health, with no signs of toxicity as indicated by stable body weight, normal biochemical markers of liver and kidney function, and the absence of significant histopathological lesions. These findings suggest that oral administration of LekCapp at doses up to 800 mg/kg/day for 14 days is well tolerated. While mild hepatic fatty infiltration, tubular casts, and crystal deposits were observed in some animals, including controls, these findings are consistent with background lesions commonly seen in healthy laboratory rats. Fatty liver changes have been documented as spontaneous occurrences in rats under normal conditions [40,41]. Similarly, the presence of renal casts (e.g., hyaline casts) and urinary crystals in small amounts is considered physiologically normal and not indicative of renal pathology [42]. Together, these results support the safety of short-term oral administration of LekCapp and warrant further investigation into its long-term safety profile.
Given the technical limitations and the complex composition of LekCapp, our phytochemical investigation primarily focused on identifying the presence of turmeric-derived constituents. Curcumin, a well-known bioactive compound in turmeric, has been extensively studied for its role in alleviating neuropathic and inflammatory pain (25 - 28). Using RP-HPLC, we confirmed the presence of 3 major curcuminoids in LekCapp, including curcumin, bisdemethoxycurcumin, and demethoxycurcumin. It is therefore plausible that these curcuminoids may partially contribute to the analgesic effects observed in our study. However, the major bioactive constituents responsible for LekCapp’s overall analgesic effects remain undefined. LekCapp contains multiple components beyond turmeric, including hydrolyzed collagen and other herbal ingredients, which were not characterized in the current study. Further research is warranted to identify and quantify the full spectrum of bioactive compounds in LekCapp and to determine the extent to which each contributes to its observed pain-relieving effects. Until such data are available, conclusion regarding the primary active constituents should be considered preliminary.
This study has several limitations. First, we evaluated the effects of a commercially formulated and patented dietary supplement (LekCapp) without conducting an independent chemical characterization of its individual components. While this reflected the real-world use of the marketed product, it limits our ability to attribute the observed effects to specific bioactive compounds. Future studies incorporating detailed phytochemical and analytical profiling are needed to identify which components, or combinations thereof, contribute to the pharmacological and histological outcomes. Second, this investigation provides preliminary evidence that oral administration of LekCapp attenuates knee joint pain-related behaviors over a 14-day period. However, the underlying cellular and molecular mechanisms driving these effects remain unclear. Further studies are necessary to elucidate the specific pathways involved in the modulation of pain and joint pathology, particularly under defined disease conditions. Third, the current study was conducted over a relatively short duration with a limited sample size. Larger-scale studies with extended treatment periods and comprehensive safety evaluations are required to confirm the reproducibility, generalizability, and translational relevance of these findings. Additionally, this study was restricted to a single animal model of knee pain and inflammation (MIA-induced OA), which may not fully represent the multifactorial nature of human OA or other joint disorders. Further preclinical investigations using diverse models, such as surgically-induced OA, along with clinical trials in human subjects, are essential to validate the therapeutic potential and real-world applicability of LekCapp.
Conclusions
This study provides the first scientific evidence that oral administration of LekCapp, following a preventive protocol, alleviates knee joint swelling and produced dose-dependent pain relief in a rat model of MIA-induced knee joint pain. These effects were observed after daily administration for at least 14 days. While the findings are promising, additional studies are necessary to further validate these results, clarify the underlying mechanisms, and assess the long-term safety and therapeutic potential of LekCapp in both preclinical and clinical setting.
Acknowledgements
We are grateful to Central Instrument Facilities (CIF), Faculty of Science, Mahidol University, Thailand for partially supporting instrument usage. We also thank Miss. Nawarat Aphaibanditkun for her technical assistance with the histological techniques. Additionally, a special thank you to Dr. Jennifer Edl Ayala and Asst. Prof. Alita Kongchanagul for proofreading this manuscript and for valuable suggestions.
Declaration of Generative AI in Scientific Writing
The authors acknowledge the use of generative AI tools (e.g., ChatGPT by OpenAI) in the preparation of this manuscript, specifically for language editing and grammar correction. No content generation or data interpretation was performed by AI. The authors take full responsibility for the content and conclusions of this work.
CRediT Author Statement
Orada Sriwatananukulkit: Conceptualization; Contributions; Methodology; Investigation; Visualization; Formal analysis; Writing - original draft preparation; Writing - review and editing. Scarlett Desclaux: Conceptualization; Contributions; Methodology; Investigation; Visualization; Formal analysis; Writing - original draft preparation; Writing - review and editing. Kasem Rattanapinyopituk: Methodology; Investigation; Visualization; Formal analysis; Writing - review and editing. Wanwisa Himakhun: Methodology; Investigation; Visualization; Formal analysis; Writing - review and editing. Yupaporn Sampaopan: Methodology; Investigation; Writing - review and editing. Sareeya Reungpatthanaphong: Methodology; Formal analysis; Validation; Resources; Writing - review and editing. Surapote Wongyai: Methodology; Visualization; Formal analysis; Writing – review and editing; Validation; Resources; Supervision. Ruedee Hemstapat: Conceptualization; Contributions; Methodology; Investigation; Writing - original draft preparation; Writing - review and editing; Validation; Resources; Supervision; Project administration; Funding acquisition.
References
[1] VP Leifer, JN Katz and E Losina. The burden of OA-health services and economics. Osteoarthritis Cartilage 2022; 30(1), 10-16.
[2] GL Nielsen, HT Sorensen, L Mellemkjoer, WJ Blot, JK McLaughlin, U Tage-Jensen and JH Olsen. Risk of hospitalization resulting from upper gastrointestinal bleeding among patients taking corticosteroids: A register-based cohort study. The American Journal of Medicine 2001; 111(7), 541-545.
[3] Z Zegota, J Gozdzik and J Glogowska-Szelag. Efficacy of herbal and naturally-derived dietary supplements for the management of knee osteoarthritis: A mini-review. Wiadomości Lekarskie 2021; 74(8), 1975-1983.
[4] JM Benson, AK Miller, N Cooper, DN Muanza, JR Smith and DM Shepherd. Anti-inflammatory effects of natural product formulations on murine macrophages. Journal of Dietary Supplements 2010; 7(3), 227-239.
[5] G Honvo, L Lengele, A Charles, JY Reginster and O Bruyere. Role of collagen derivatives in osteoarthritis and cartilage repair: A systematic scoping review with evidence mapping. Rheumatology and Therapy 2020; 7(4), 703-740.
[6] A Prakash and R Baskaran. Acerola, an untapped functional superfruit: A review on latest frontiers. Journal of Food Science and Technology 2018; 55(9), 3373-3384.
[7] B Kulczyński and A Gramza-Michałowska. Goji Berry (Lycium barbarum): Composition and health effects - a Review. Polish Journal of Food and Nutrition Sciences 2016; 66(2), 67-75.
[8] KV Peddada, KV Peddada, SK Shukla, A Mishra and V Verma. Role of curcumin in common musculoskeletal disorders: A review of current laboratory, translational, and clinical data. Orthopaedic Surgery 2015; 7(3), 222-231.
[9] C Feng, M Zhao, L Jiang, Z Hu and X Fan. Mechanism of modified Danggui Sini decoction for knee osteoarthritis based on network pharmacology and molecular docking. Evidence‐Based Complementary and Alternative Medicine 2021; 2021(1), 6680637.
[10] N Sheena, TA Ajith and KK Janardhanan. Anti-inflammatory and anti-nociceptive activities of Ganoderma lucidum occurring in South India. Pharmaceutical Biology 2008; 41(4), 301-304.
[11] SJ Jung, MR Oh, DY Lee, YS Lee, GS Kim, SH Park, SK Han, YO Kim, SJ Yoon and SW Chae. Effect of Ginseng extracts on the improvement of osteopathic and arthritis symptoms in women with osteopenia: A randomized, double-blind, placebo-controlled clinical trial. Nutrients 2021; 13(10), 3352.
[12] HG Jo, CY Baek, Y Hwang, E Baek, C Park, HS Song and D Lee. Investigating the anti-inflammatory, analgesic, and chondroprotective effects of Gynostemma pentaphyllum (Thunb.) Makino in osteoarthritis: An in vitro and in vivo study. International Journal of Molecular Sciences 2024; 25(17), 9594.
[13] D Clemett and KL Goa. Celecoxib: A review of its use in osteoarthritis, rheumatoid arthritis and acute pain. Drugs 2000; 59(4), 957-980.
[14] EA Kockaya, G Selmanoglu, K Kismet and MT Akay. Pathological and biochemical effects of therapeutic and supratherapeutic doses of celecoxib in Wistar albino male rats. Drug and Chemical Toxicology 2010; 33(4), 410-414.
[15] AB Nair and S Jacob. A simple practice guide for dose conversion between animals and human. Journal of Basic and Clinical Pharmacy 2016; 7(2), 27-31.
[16] SE Bove, SL Calcaterra, RM Brooker, CM Huber, RE Guzman, PL Juneau, DJ Schrier and KS Kilgore. Weight bearing as a measure of disease progression and efficacy of anti-inflammatory compounds in a model of monosodium iodoacetate-induced osteoarthritis. Osteoarthritis and Cartilage 2003; 11(11), 821-830.
[17] SR Chaplan, FW Bach, JW Pogrel, JM Chung and TL Yaksh. Quantitative assessment of tactile allodynia in the rat paw. Journal of Neuroscience Methods 1994; 53(1), 55-63.
[18] IJ Chen, SH Lin and CS Wong. Oral shea nut oil triterpene concentrate supplement ameliorates pain and histological assessment of articular cartilage deterioration in an ACLT injured rat knee osteoarthritis model. PLoS One 2019; 14(4), e0215812.
[19] HJ Mankin, H Dorfman, L Lippiello and A Zarins. Biochemical and metabolic abnormalities in articular cartilage from osteo-arthritic human hips. II. Correlation of morphology with biochemical and metabolic data. The Journal of Bone & Joint Surgery 1971; 53(3), 523-537.
[20] S Simoes, R Lopes, MCD Campos, MJ Marruz, MEM da Cruz and L Corvo. Animal models of acute gastric mucosal injury: Macroscopic and microscopic evaluation. Animal Models and Experimental Medicine 2019; 2(2), 121-126.
[21] KE Ibrahim, MG Al-Mutary, AO Bakhiet and HA Khan. Histopathology of the liver, kidney, and spleen of mice exposed to gold nanoparticles. Molecules 2018; 23(8), 1848.
[22] R Wulandari, Sudjadi, S Martono and A Rohman. Liquid chromatography and fourier transform infrared spectroscopy for quantitative analysis of individual and total curcuminoid in Curcuma longa extract. Journal of Applied Pharmaceutical Science 2018; 8(9), 107-113.
[23] NK Prabaningdyah, S Riyanto, A Rohman and C Siregar. Application of HPLC and response surface methodology for simultaneous determination of curcumin and desmethoxy curcumin in Curcuma syrup formulation. Journal of Applied Pharmaceutical Science 2017; 7(12), 058-064.
[24] CT Huang, PH Chen, SH Chen, JH Lue and YJ Tsai. Curcumin promotes microglial M2 polarization and suppresses chronic constriction: Injury-induced neuropathic pain in a rat model of peripheral neuropathy. Nutrition 2023; 109, 112004.
[25] X Zhang, Z Guan, X Wang, D Sun, D Wang, Y Li, B Pei, M Ye, J Xu and X Yue. Curcumin alleviates oxaliplatin-induced peripheral neuropathic pain through inhibiting oxidative stress-mediated activation of NF-kappaB and mitigating inflammation. Biological and Pharmaceutical Bulletin 2020; 43(2), 348-355.
[26] S Liu, Q Li, MT Zhang, QL Mao-Ying, LY Hu, GC Wu, WL Mi and YQ Wang. Curcumin ameliorates neuropathic pain by down-regulating spinal IL-1beta via suppressing astroglial NALP1 inflammasome and JAK2-STAT3 signalling. Scientific Reports 2016; 6(1), 28956.
[27] Y Li, Y Zhang, DB Liu, HY Liu, WG Hou and YS Dong. Curcumin attenuates diabetic neuropathic pain by downregulating TNF-alpha in a rat model. International Journal of Medical Sciences 2013; 10(4), 377-381.
[28] Y Jeon, CE Kim, D Jung, K Kwak, S Park, D Lim, S Kim and W Baek. Curcumin could prevent the development of chronic neuropathic pain in rats with peripheral nerve injury. Current Therapeutic Research 2013; 74, 1-4.
[29] YH Kang, HJ Lee, CJ Lee and JS Park. Natural products as sources of novel drug candidates for the pharmacological management of osteoarthritis: A narrative review. Biomolecules & Therapeutics 2019; 27(6), 503-513.
[30] M Thakur, W Rahman, C Hobbs, AH Dickenson and DL Bennett. Characterisation of a peripheral neuropathic component of the rat monoiodoacetate model of osteoarthritis. PLoS One 2012; 7(3), e33730.
[31] K Inomata, K Tsuji, H Onuma, T Hoshino, M Udo, M Akiyama, Y Nakagawa, H Katagiri, K Miyatake, I Sekiya, T Muneta and H Koga. Time course analyses of structural changes in the infrapatellar fat pad and synovial membrane during inflammation-induced persistent pain development in rat knee joint. BMC Musculoskeletal Disorders 2019; 20(1), 8.
[32] Y Zhang and Y Zeng. Curcumin reduces inflammation in knee osteoarthritis rats through blocking TLR4 /MyD88/NF-kappaB signal pathway. Drug Development Research 2019; 80, 353-359.
[33] A Aravinthan, MA Hossain, B Kim, CW Kang, NS Kim, KC Hwang, NS Kim, KC Hwang and JH Kim. Ginsenoside Rb(1) inhibits monoiodoacetate-induced osteoarthritis in postmenopausal rats through prevention of cartilage degradation. Journal of Ginseng Research 2021; 45, 287-294.
[34] RX Zhang, K Ren and R Dubner. Osteoarthritis pain mechanisms: Basic studies in animal models. Osteoarthritis Cartilage 2013; 21(9), 1308-1315.
[35] R Oledzki and J Harasym. Acerola (Malpighia emarginata) anti-inflammatory activity - a review. International Journal of Molecular Science 2024; 25(4), 2089.
[36] H Jiang, L Xu, W Liu, M Xiao, J Ke and X Long. Chronic pain causes peripheral and central responses in MIA-induced TMJOA rats. Cellular and Molecular Neurobiology 2022; 42(5), 1441-1451.
[37] J Fernihough, C Gentry, M Malcangio, A Fox, J Rediske, T Pellas, B Kidd, S Bevan and J Winter. Pain related behaviour in two models of osteoarthritis in the rat knee. Pain 2004; 112(1-2), 83-93.
[38] SM Lockwood, DM Lopes, SB McMahon and AH Dickenson. Characterisation of peripheral and central components of the rat monoiodoacetate model of Osteoarthritis. Osteoarthritis Cartilage 2019; 27(4), 712-722.
[39] D Martinez-Puig, E Costa-Larrion, N Rubio-Rodriguez and P Galvez-Martin. Collagen supplementation for joint health: The link between composition and scientific knowledge. Nutrients 2023; 15(6), 1332.
[40] RL Maynard and N Downes. Liver. In: RL Maynard and N Downes (Eds.). Anatomy and histology of the laboratory rat in toxicology and biomedical research. 1st ed. Academic Press, London, 2019, p. 159-168.
[41] SW Barthhold, SM Griffy and DH Percy. Pathology of laboratory rodents and rabbits. 4th ed. Wiley-Blackwell, New Jersey, United States, 2016. p. 81-106.
[42] JC Seely, GC Hard and B Blankenship. Kidney. In: AW Suttie (Ed.). Boorman’s pathology of the rat. 2nd ed. Academic Press, London, 2018, p.125-166.