
Trends
Sci.
2026;
23(5):
12105
ZnO/Chitosan Nanoconjugates from Chlorella vulgaris as Nanostimulants Against Viral Nervous Necrosis in Grouper
Uun
Yanuhar1,*,
Heru Suryanto2,
![]() ,
Herly Evanuarini3,
,
Herly Evanuarini3,
![]() ,
Nico Rahman Caesar1,
,
Nico Rahman Caesar1,
![]() ,
,
Defa
Rizqi Machfuda4,
![]() ,
Nezya Pramudya Wardani4
and Aisha Surya Ananda5
,
Nezya Pramudya Wardani4
and Aisha Surya Ananda5
1Study Program of Fisheries Resources Management, Faculty of Fisheries and Marine Sciences, Brawijaya University,
East Java, Indonesia
2Department of Mechanical and Industrial Engineering, Faculty of Engineering, Universitas Negeri Malang,
Malang 65145, Indonesia
3Department of Animal Product Technology, Faculty of Animal Science, Brawijaya University,
Malang 65141, Indonesia
4Master of Aquaculture Study Program, Faculty of Fisheries and Marine Sciences, Brawijaya University,
East Java, Indonesia
5Faculty of Veterinary Medicine, Brawijaya University, East Java, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 29 September 2025, Revised: 17 October 2025, Accepted: 24 October 2025, Published: 10 January 2026
Abstract
Grouper farming underpins Indonesia’s coastal livelihoods and supplies both domestic and export markets, yet disease-especially Viral Nervous Necrosis (VNN) in juveniles-remains a major constraint. Conventional tools (antibiotics, standard vaccines) have delivered limited protection, motivating alternative approaches. Leveraging nanotechnology, we evaluated a ZnO-chitosan nanoconjugate carrying a recombinant Chlorella vulgaris antigen for antiviral protection in cantang grouper (E. fuscoguttatus×E. lanceolatus). In a 56-day randomized block trial, 160 fish were allocated to 5 treatments (T1 - T5) and two controls. Endpoints included growth (length, weight, survival rate, SR; relative percent survival, RPS), hematology (erythrocytes, leukocytes, hematocrit, hemoglobin), gill histopathology, and flow cytometry of CD4⁺/CD8⁺ T-cell markers. The 33 µL regimen (T3) was consistently superior: length rose from 9.94 → 12.73 cm and weight from 13.57 → 18.00 g, with SR = 95% and RPS = 92%. Hematological profiles improved across all indices; gill lesions were minimal (mild edema, hyperplasia, lamellar fusion); and immunophenotyping showed moderated CD4⁺ activation alongside a marked increase in CD8⁺ expression, indicating effective cellular immunity. While T1 and T4 performed well and T2/T5 yielded moderate responses, T3 delivered the most balanced protection and significantly lowered viral load. These results highlight the promise of recombinant Chlorella antigens formulated in ZnO-chitosan nanoconjugates to strengthen grouper health and bolster aquaculture sustainability.
Keywords: Grouper, Viral Nervous Necrosis, Chlorella vulgaris, ZnO/Chitosan nanoconjugates, Nanostimulants
Introduction
The cultivation of groupers represents a vital aspect of Indonesia’s marine fisheries industry, playing a crucial role in the livelihoods of local communities while also catering to global market demands. In 2020, Indonesia’s seafood sector witnessed a substantial production of around 60,000 tons of grouper, reflecting
a growing appetite among consumers both on home shores and abroad, largely due to the speciesʼ appealing taste and high economic value [1] However, this thriving industry is not without significant challenges. Specifically, grouper aquaculture grapples with severe threats from infectious diseases, with one of the most alarming being Viral Nervous Necrosis (VNN). This virus, belonging to the Betanodavirus genus, primarily affects young groupers, larvae, and juveniles, which are particularly vulnerable. When outbreaks occur, they can devastate populations, with mortality rates soaring to devastatingly high levels, often reaching 100% [2] Such losses not only disrupt the ecosystem but also inflict significant economic hardships on fish farmers, with estimated damages running into the millions of dollars each year [3].
Traditional methods targeting the management of these diseases, such as the use of vaccines and antibiotics, have not yielded the desired results. Although vaccines can offer some level of protection, their effectiveness can be unpredictable and varies with different grouper species and farming conditions [4]. Moreover, the heavy reliance on antibiotics raises alarm bells regarding the emergence of antimicrobial resistance and potential harm to the surrounding environment. These challenges underscore an urgent need for innovative and sustainable solutions that protect the future of grouper farming.
Exciting advancements in fields like nanotechnology and biotechnology are beginning to shine light on potential remedies to these pressing issues. Particular focus has been placed on nanoparticles, such as ZnO/Chitosan nanoconjugates, which show significant promise due to their antibacterial and immunostimulatory properties [5]. These nanoparticles are not only biocompatible, making them safe for use in aquaculture, but they also enhance the effectiveness of vaccines. By allowing for controlled and sustained release of antigens, they can invoke stronger and more lasting immune responses in fish, potentially reducing viral loads and activating key immune cells that are vital for combating diseases like VNN [6]. However, hurdles still exist; ensuring that these treatments are consistently effective in varying aquaculture environments remains a significant concern, as stress factors in these settings can adversely affect immune function. Additionally, the sustainability and long-term effectiveness of nanoparticle-based vaccines necessitate more thorough investigations to confirm their safety for both fish populations and the wider environment [7]. In light of these ongoing challenges, our study introduces an innovative approach: The development of a recombinant protein-based nanostimulant derived from Chlorella vulgaris, intertwined with ZnO/Chitosan nanoconjugates. This combination of ZnO-chitosan and recombinant Chlorella vulgaris antigen aims to effectively enhance the immune responses of groupers, thereby reducing the risks posed by Viral Nervous Necrosis (VNN). Through this approach, we not only aim to improve the resilience of Indonesia’s grouper farming but also contribute positively to the growth of the industry, ensuring it continues to provide vital resources and economic opportunities for coastal communities in the long term. The specific objectives of this research are: (1) to evaluate the antiviral efficacy of ZnO/Chitosan nanoconjugates formulated with recombinant Chlorella vulgaris in enhancing grouper growth performance, including measurable parameters such as length, weight, survival rate (SR), and relative percent survival (RPS); (2) to assess the effects of the treatment on the immune response by measuring CD8+ T-cell expression and other hematological indices (erythrocyte and leukocyte counts, hematocrit, hemoglobin levels); and (3) to analyze tissue integrity through histopathological analysis. These measurable parameters will allow us to determine the potential of this approach in improving grouper aquaculture sustainability and enhancing the resistance of groupers to viral infections.
Materials and methods
Materials
This study focused on using a few key materials that were essential to our research. We collected Chlorella vulgaris, a type of green microalgae, from the coastal waters of Situbondo, where the sediment conditions were just right for our experiments. To complement the algae, we sourced zinc oxide (ZnO) through Sigma-Aldrich, a well-known supplier in the field, and chitosan from PT. Bioteknologi Indonesia, ensuring that we used high-quality materials for our work. For the experimental trials, we set up aquarium tanks to simulate natural conditions in which we could conduct our in vivo tests. When it came to analyzing our molecular data, we utilized specialized kits from Qiagen for isolating RNA and cDNA. Our laboratory was equipped with several essential tools, including micropipettes from Eppendorf for precise measurement, as well as mortars and pestles for preparing our samples before further analysis. To separate and analyze our samples, we employed centrifuges from Thermo Fisher Scientific and used agarose gel electrophoresis systems from Bio-Rad to visualize our results. In addition, we conducted polymerase chain reaction (PCR) with reliable machines from Applied Biosystems, and utilized 1.5 mL microtubes from Eppendorf for handling our samples with care. For the preservation of our samples, we relied on liquid nitrogen, which allowed us to maintain their integrity during storage. Throughout our various laboratory procedures, we utilized both deionized water (ddH₂O) and distilled water to ensure that our experiments were conducted with the utmost precision and purity.
Research design
To investigate the antiviral capabilities of ZnO/Chitosan nanoconjugates combined with recombinant Chlorella vulgaris, we adopted an experimental approach focusing on the cantang grouper, a hybrid of E. fuscoguttatus and E. lanceolatus. A total of 160 cantang grouper fish were allocated into six groups: Five treatment groups (T1 - T5) and one control group, with 20 fish in each treatment group. The sample size was determined through statistical calculations to ensure sufficient analytical power, considering the desired significance level and the ability to detect meaningful differences between treatments. The fish selected for the study were 6-month-old cantang groupers, each measuring approximately 10 - 12 cm, to minimize size-related variability. They were thoroughly screened by an ichthyologist to confirm they were free from diseases or infections that could influence the studyʼs outcomes. The selected fish exhibited normal activity levels and were representative of the broader population. Random selection was conducted using a block randomization approach to ensure that both the treatment and control groups included fish of similar size and health, minimizing bias in group assignment and ensuring a fair representation of the overall population. Our study unfolded over 56 days, during which we collected samples at two critical points: At the start (day 0) and after the treatment concluded (day 56). Throughout this research, we closely monitored several key parameters. First, we evaluated the growth performance of the grouper, looking particularly at their survival rate (SR) and relative percent survival (RPS). We also assessed various hematological parameters, including the counts of erythrocytes (red blood cells) and leukocytes (white blood cells), along with measurements of hematocrit, hemoglobin levels, and the differentiation of leukocytes. These indicators are crucial for understanding the overall health and physiological responses of the fish. Additionally, we took a closer look at the condition of the gill tissues, as these are vital for the fishʼs respiration and overall well-being. To gain deeper insights into how the treatments influenced the immune system, we employed flow cytometry. This method allowed us to assess the responses of immune cells, providing us with a comprehensive understanding of the immunomodulatory effects of our treatment approach.
Production of ZnO/Chitosan nanoconjugates
To create the ZnO/Chitosan nanoconjugates, we followed a series of well-established methods that ensured high quality and effectiveness. The process began by dissolving chitosan in a 1% acetic acid solution at room temperature, aiming for a final concentration between 1% and 3% weight/volume. Meanwhile, we turned our attention to the zinc oxide (ZnO) nanoparticles. This involved dissolving a zinc precursor, either zinc acetate or zinc nitrate, in distilled water at a concentration of 0.1 M. We then carefully added sodium hydroxide gradually, which initiated the formation of the nanoparticles. As this reaction proceeded, we noticed a cloudy suspension forming, signaling that the nanoparticles were coming together. Once we reached this stage, we collected the newly formed ZnO nanoparticles by centrifuging the mixture. To ensure purity, we washed them with distilled water to remove any leftover impurities and then dried them at a temperature between 40 and 50 °C. Next, we brought the two components together for conjugation. This involved mixing the ZnO suspension with our prepared chitosan solution in a 1:1 ratio. We stirred this mixture for 4 to 6 h at room temperature, promoting the necessary interactions, such as electrostatic forces or hydrogen bonds, between the chitosan and the zinc oxide particles. After this process, we separated the ZnO/Chitosan nanoconjugates from the solution, filtered them, and allowed them to dry at a lower temperature, preparing them for future applications [8].
Production of recombinant Chlorella vulgaris combined with ZnO/Chitosan nanoconjugates
The recombinant protein was produced from C. vulgaris sourced from Situbondo waters, following protocols adapted from [9]. Microalgal biomass was washed by sedimentation, rinsed with freshwater 2 - 3 times, harvested by centrifugation at 4,000 rpm for 10 min, freeze-dried, and stored at – 20 °C until further use. Recombinant protein expression was performed in Escherichia coli BL21 (DE3) harboring a plasmid encoding the target C. vulgaris protein. Induction was carried out with 0.1 mM IPTG at 37 °C for 4 h, after which recombinant proteins were purified by Ni-NTA affinity chromatography (Qiagen) and dialyzed against PBS. For nanostimulant formulation, purified recombinant protein was combined with ZnO/Chitosan nanoconjugates according to the optimal ratio of 1: 0.25 (protein:ZnO), as previously described [10]. This ratio was selected for its efficacy in nanoparticle delivery and immunostimulation against viral infections such as VNN. The mixture was stored at 4 °C for 24 h to ensure encapsulation of the recombinant protein within the ZnO/Chitosan matrix, then frozen at – 80 °C for subsequent antiviral application in grouper.
In vivo evaluation of recombinant Chlorella vulgaris combined with ZnO/Chitosan nanoconjugates
This study used grouper fish sourced from Teluk Kode, Malaka, Selamat, North Lombok, and West Nusa Tenggara, with individual fish weighing between 10 and 12 g. A total of 20 fish were placed in each treatment tank. Before beginning the in vivo experiment, the fish were acclimatized for seven days to ensure they adapted properly to their new environment. The formulated Recombinant Chlorella vulgaris-based ZnO/Chitosan Nanoconjugates were administered orally to the fish. The nanostimulant was given in two doses: on day 0 and day 14. To assess the vaccineʼs effectiveness, a Viral Nervous Necrosis (VNN) challenge test was performed on day 28. The virus was prepared by homogenizing the organs of fish that had been previously infected with VNN, then mixing the homogenate with distilled water and centrifuging the mixture. The supernatant, containing the virus, was then administered orally to the experimental fish. This study followed a completely randomized factorial design (CRD) with eight treatments, each replicated three times, as detailed in Table 1.
Table 1 Group experiment in research design.
Growth rate analysis
The growth performance of the cantang grouper was evaluated by measuring both survival rate (SR) and relative percentage survival (RPS). Fish length and body weight were recorded before and after administration of the nanostimulant, as well as following the VNN challenge test. SR was calculated as the percentage of fish that survived during the experimental period, while RPS was used to assess treatment effectiveness against infection. This rate can be calculated using the formula in Eq. (1).

Description:
SR: Survival Rate (%)
No: Number of fish alive at the beginning of the study (individuals)
Nt: Number of fish alive at the end of the study (individuals)
Relative Percentage Survival (RPS) is widely applied as a critical parameter for evaluating the protective efficacy of vaccines or immunostimulants, reflecting their ability to enhance disease resistance and improve survival following pathogen exposure [10]. This measure helps to gauge the overall success of vaccination in improving fish survival rates. RPS can be calculated using the formula in Eq. (2) [10].
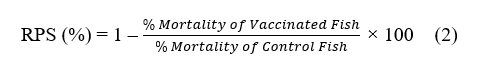
Blood cell observation
We collected blood samples using a 1 mL syringe from Terumo (Japan) that we had prepped by rinsing it with a 3% sodium citrate solution from Merck (Germany). This step was essential to prevent the blood from clotting during the collection process. For this collection, we drew blood from the muscle tissue located near the anal fin of the fish, carefully transferring these samples into 1.5 mL microtubes from Eppendorf (Germany) for further analysis. In addition to this, we also took separate blood samples specifically for hematological analysis using a dry syringe, which did not contain any anticoagulant. These samples were stored in vacuum tubes lined with EDTA from OneMed (Indonesia) to preserve their integrity. To measure hematocrit levels, we employed the microhematocrit method, placing the blood samples into specialized hematocrit tubes. These tubes were then centrifuged at a high speed of 10,000×g for 10 min, allowing the different components of the blood to separate. We measured the separation using a hematocrit reader, or sometimes did it manually, calculating the percentage of the red blood cell (RBC) volume. While this method is standard, it is important to note that hematocrit readings can show some variability due to factors such as sampling technique, individual variations among the fish, and accuracy in centrifugation and reading. Additionally, factors like the fish’s physical condition (e.g., stress or infection) may also influence RBC volume and hematocrit levels. For the leukocyte differential analysis, we prepared blood smears from the samples we collected. These smears were then stained using a Giemsa solution, following standard protocols to ensure accurate results [11,12]. Under a microscope, the differentiation of leukocytes was evaluated by counting the different types of white blood cells—monocytes, lymphocytes, basophils, eosinophils, and neutrophils—based on their characteristic morphology and structure. Red blood cell (RBC) and white blood cell (WBC) counts were determined using a hemocytometer, while hemoglobin (Hb) concentration was measured with a Sahli hemometer.
Histopathological examination
Gill tissues of cantang grouper (E. fuscoguttatus×E. lanceolatus) were collected and processed for histopathological analysis following the protocol described by [13]. Samples were fixed in 10% neutral-buffered formalin, sectioned, and stained with hematoxylin and eosin (H&E) (Sigma-Aldrich, USA). Microscopic examinations were performed using an Optilab camera under magnifications of 100x to 400x. Tissue alterations, including edema, hyperplasia, and lamellar fusion, were assessed in 5 random fields of view per sample. The extent of damage was quantified as the percentage of affected cells, and severity was graded on a scale of 1 to 4, as described by [14]. This approach enabled a systematic evaluation of gill tissue health.
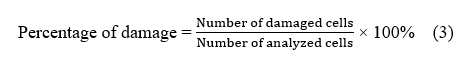
The obtained percentage damage has scores ranging from 0 to 3 (Aminin et al. 2024), with criteria as follows:
-Score 0: No damage at all.
-Score 1: Mild damage with a percentage of < 15%.
-Score 2: Moderate damage with a percentage of 15% - 30%.
-Score 3: Severe damage with a percentage of > 30%.
Flowcytometry analysis
Cells were isolated from immune-related organs, particularly the gills, of cantang grouper treated with recombinant Chlorella vulgaris in combination with ZnO/Chitosan nanoconjugates. The isolated cells were treated with trypsin, resuspended in phosphate-buffered saline (PBS), and subsequently analyzed using a FACSCalibur flow cytometer (BD Biosciences, Germany). Immune responses were characterized with monoclonal antibodies (BioLegend, Inc., San Diego, USA) specific for CD4 and CD8 markers, which enabled identification of lymphocyte subsets based on size and granularity. A total of 10,000 events were acquired per sample to determine the immunological effect of the nanostimulant [15].
Analysis data
All data were analyzed using one-way analysis of variance (ANOVA) to determine statistically significant differences among treatment groups, with significance set at p < 0.05. Data processing, statistical analyses, and graphical presentations were carried out using Microsoft Excel and OriginPro 2019b (OriginLab Corporation, USA). Results of growth performance, hematological indices, histopathological scores, and flow cytometry analyses were expressed as mean ± standard deviation (SD). Graphical outputs were generated to aid interpretation, including visualization of ANOVA comparisons.
Results and discussion
Results of grouper fish growth rate analysis
Growth in organisms refers to the increase in size, both in length and weight, which occurs due to the enlargement and multiplication of cells. Several factors play a role in determining growth, including internal aspects such as heredity, gender, and age, as well as external influences like water quality and feeding conditions. For fish, measuring growth in terms of length and weight is a key indicator of how successful various cultivation treatments are [16-19]. In our study, we found that the treatment significantly affected the growth of the fish, enhancing both their length and weight after the administration of the nanostimulant and following the virus challenge. For those interested in the specific measurements of the cantang grouperʼs length and weight, these details are provided in Table 2.
Nanostimulant treatment has a noticeable effect on the growth of organisms, both in terms of length and weight, following nanostimulant administration and the VNN virus challenge test. In the control group without nanostimulants (K‒), the average length of the organisms after nanostimulant administration was 9.19 cm, and after the virus challenge, it increased to 10.61 cm (Table 2). On the other hand, in the control group with nanostimulants (K+), the organisms had a slightly higher initial length of 9.23 cm after nanostimulant administration, and their length increased to 11.20 cm after the virus challenge. In treatments T1 - T5, a significant increase in length was observed after the viral challenge compared with the control group. Notably, treatment T3, which involved the administration of recombinant Chlorella vulgaris combined with ZnO/Chitosan nanoconjugates (33 µL), resulted in the greatest improvement, with fish length increasing from 9.94 to 12.73 cm. This finding indicates that the T3 nanostimulant treatment provided the most favorable response to viral exposure, thereby promoting optimal growth.
Table 2 Average growth in length and weight of grouper fish.
Treatment |
Average final length (cm) |
Average final weight (g) |
||
Post-Nanostimulant administration |
Post-VNN virus challenge test |
Post-Nanostimulant administration |
Post-VNN virus challenge test |
|
K‒ |
9.19 ± 0.58 |
10.61 ± 0.37 |
12.92 ± 0.66 |
15.27 ± 0.84 |
K+ |
9.23 ± 0.60 |
11.20 ± 0.50 |
13.20 ± 0.92 |
15.82 ± 0.82 |
T1 |
9.34 ± 0.60 |
11.20 ± 0.37 |
13.31 ± 0.66 |
18.00 ± 0.82 |
T2 |
9.38 ± 0.58 |
11.10 ± 0.50 |
13.34 ± 0.92 |
17.90 ± 0.84 |
T3 |
9.94 ± 0.89 |
12.73 ± 0.87 |
13.57 ± 1.24 |
18.00 ± 1.50 |
T4 |
9.48 ± 0.66 |
11.52 ± 0.60 |
13.21 ± 0.91 |
16.17 ± 1.22 |
T5 |
9.74 ± 0.80 |
11.96 ± 0.49 |
13.51 ± 0.93 |
16.48 ± 1.13 |
The trend in weight gain mirrored the improvements seen in length. In the control group (K-group), the average weight of the fish rose from 12.92 g after the nanostimulant was given to 15.27 g following the viral challenge. Meanwhile, the K+ group showed a similar rise, increasing from 13.20 to 15.82 g. In contrast, the fish in the T3 group experienced a remarkable boost, with weights climbing from 13.57 to 18.00 g after the challenge. This clearly indicates that the T3 treatment not only resulted in the largest increase in length but also achieved the highest weight gain in response to the viral infection.
The primary aim of using nanostimulants in grouper is to bolster their immune responses against viral threats, such as Viral Nervous Necrosis (VNN) [10,20]. The increases in both length and weight point to a positive effect of the nanostimulants on immune performance, allowing the fish to effectively build a stronger defense against viral infections [21]. Among all the treatments tested, T3 emerged as the standout, showing the most significant enhancement in growth, which hints at its superior ability to strengthen both immunity and growth performance. The improvements in growth metrics are likely linked to enhanced immune competence, which equips the fish to better tackle VNN infections. When fish have stronger immune systems, they generally enjoy better survival rates and growth outcomes, even when faced with viral stress [22].
Moreover, nanostimulants are recognized for their role in boosting metabolic activity, which helps fish utilize nutrients more effectively, promoting growth in both length and weight [23,24]. Fish that manage to keep their viral loads low can redirect more energy toward growth, thereby emphasizing the dual benefits of nanostimulants in both immune support and metabolism. After receiving the nanostimulants and then being challenged with the virus, the group of fish displayed improved growth rates. This suggests that although they were infected, the nanostimulants helped mitigate the virusʼs adverse effects on their physical development [25,26]. However, treatments in groups T4 and T5 did not show optimal results like those in T3. This could be due to the nanostimulant dose being too high in these groups, potentially reducing its effectiveness in enhancing the immune response of the grouper fish. Excessively high doses may disrupt the metabolism or balance of the fishʼs body, diminishing the ability of the nanostimulants to support optimal growth [27].
Survival Rate (SR) and Relative Percentage Survival (RPS)
Survival Rate (SR), or the survival rate of grouper fish, is influenced by nanostimulant treatment and the VNN virus challenge. In the control group without a nanostimulant (K‒), the survival rate was very low, at around 40%. Meanwhile, in the control group exposed to the VNN virus (K+), the survival rate dropped even further to just 20% (Figure 1(A)). This indicates that the VNN virus, when administered without nanostimulant treatment, significantly impacts the survival of grouper fish by weakening their immune response and causing damage to organ functions. Fish that were treated with nanostimulants experienced notably higher survival rates compared to those that weren’t given these treatments. For instance, in Treatment T1, where healthy hybrid grouper-the species Epinephelus fuscoguttatus crossed with Epinephelus lanceolatus-were combined with ZnO, we observed a survival rate of 75%. This was a marked improvement over the control group.
In Treatment T2, where hybrid grouper were exposed to both ZnO and the VNN virus, the survival rate was 55%. This lower figure suggests that ZnO alone isn’t sufficient to effectively inhibit the replication of the virus or provoke a strong immune response against VNN [28].
Figure 1 Measurement results: (A) Survival Rate (SR); (B) Relative Percentage Survival (RPS).
In stark contrast, Treatment T3 yielded the most impressive results. By combining recombinant Chlorella vulgaris with ZnO/Chitosan nanoconjugates (33 µL), this group achieved the highest survival rate of 95%. This highlights the powerful protective effect of the nanostimulant, enhancing the fishʼs resilience against VNN infection. Treatments T4 (66 µL) and T5 (112 µL) also showed strong survival rates of 90% and 85%, respectively. Although these rates were slightly lower than T3, they still reflect positive outcomes. The decreased effectiveness observed in T4 and T5 might be due to the higher doses, which could hinder an optimal immune response. The overall goal of using nanostimulants in grouper aquaculture is to strengthen the immune system against VNN infection. Notably, survival rates in all groups treated with nanostimulants (T1 - T5) were significantly higher than those in the K+ group, which faced the virus challenge without treatment. These findings reinforce the importance of nanostimulants in boosting disease resistance and enhancing survival when fish are exposed to viral pathogens [29,30].
While Treatments T1 and T2 exhibited lower survival rates compared to T3 - T5, this likely stemmed from their reliance on ZnO nanoparticles alone, without a full nanovaccine formulation. Although ZnO is known for its antioxidant properties, its standalone administration did not seem capable of triggering a strong enough immune response for effective protection [31]. In contrast, the superior survival rate in T3 underscores the effectiveness of a well-balanced nanovaccine dosage, which optimally elicits immune protection. On the other hand, the higher doses in T4 and T5 might have led to reduced outcomes due to potential overstimulation or toxicity, compromising the immune response [28,32].
Additionally, administering nanostimulants significantly improved the Relative Percent Survival (RPS) when compared with the control group. In the uninfected control group (K−), the RPS reached 50%, showcasing the grouperʼs natural resilience against VNN infection without immunization (Figure 1(B)). These results highlight the critical role of proper nanostimulant formulation and dosage to maximize survival and ensure effective antiviral defense in grouper aquaculture. Conversely, the control group exposed to the VNN virus (K+) had a much lower RPS of 30%, illustrating that exposure to the virus without any nanovaccine treatment can lead to high mortality. This underscores the importance of activating the immune system through vaccination to combat viral infections [33].
For Treatments T1 and T2, where only ZnO nanoparticles were introduced, the RPS values stood at 58% and 44%, respectively. While these figures were better than those recorded for the VNN control group, they remained lower than those of the groups that received nanostimulants. This suggests that while ZnOʼs antioxidant properties may provide some level of protection against oxidative stress from viral infections, it doesn’t directly enhance the adaptive immune response. Therefore, relying solely on ZnO nanoparticles is not enough to fully safeguard fish from VNN infection [34].
In contrast, the groups treated with the nanovaccine combination showed a much greater increase in RPS. Treatment T3, which involved Recombinant Chlorella vulgaris combined with ZnO/Chitosan Nanoconjugates (33 µL), yielded the best result, with an RPS of 92%. This indicates that the nanovaccine dose in T3 was optimal for stimulating the fishʼs immune system and providing antiviral protection against the VNN virus. Treatments T4 (recombinant Chlorella vulgaris combined with ZnO/Chitosan nanoconjugates, 66 µL) and T5 (112 µL) demonstrated relatively high survival rates of 88% and 75%, respectively, although both were lower than that observed in T3. These findings indicate a dose-dependent relationship between nanovaccine administration and immune response. While vaccine efficacy appears to increase up to an optimal threshold, excessively high doses, as in T4 and T5, resulted in reduced effectiveness. Such outcomes may be attributed to adverse immunological effects, including overstimulation or the induction of immune tolerance, which ultimately diminishes the protective capacity of the treatment [35].
Results of grouper fish blood cell analysis
The red blood cell (RBC) count in grouper fish remained relatively stable across all treatments, ranging from 2.50 to 2.75×10⁶ cells/mm³. In grouper fish, the normal RBC value ranges from 2.5 to 3.5×10⁶ cells/mm³ (for example, in other teleost fish) [36]. In the control group with VNN infection (K+), the RBC count was at its lowest, 2.50×10⁶ cells/mm³ (Figure 2(A)). The presence of the VNN virus appears to cause significant physiological disruptions when fish do not receive nanostimulant treatment, particularly evident in the decrease of red blood cell (RBC) counts. This decline may be linked to oxidative stress and damage to hematopoietic tissues caused by the infection, which inhibits the normal production of red blood cells. In contrast, the control group of fish that were not exposed to the virus (K−) exhibited a slightly higher RBC count of 2.57×10⁶ cells/mm³, suggesting that these fish maintained a more stable physiological state without the added stress of a viral infection [35].
Figure 2 Measurement results: (A) Erythrocytes; (B) Leukocytes; (C) Hemoglobin; (D) Hematocrit.
In the groups that were treated with ZnO nanoparticles (T1 and T2), the RBC counts were similar to those in the uninfected control group, measuring at 2.55 and 2.56×10⁶ cells/mm³, respectively. This similarity indicates that while ZnO helps maintain physiological stability through its antioxidant effects, it does not seem to encourage increased production of red blood cells [37]. However, the fish receiving nanostimulants (T3 and T4), which combined recombinant Chlorella vulgaris with ZnO/Chitosan nanoconjugates, showed a notable increase in their RBC counts, rising to 2.75×10⁶ cells/mm³. This enhancement suggests that the nanostimulant formulation is more effective at optimizing physiological functions, thereby boosting adaptive immune responses and improving overall blood health [38]. Interestingly, Treatment T5 produced a slightly lower RBC count of 2.60×10⁶ cells/mm³. This might indicate that while the higher doses of the nanostimulant initially seem beneficial, they could eventually lead to diminishing returns once an optimal threshold is surpassed.
Leukocyte counts also varied among treatments, ranging from 1.26 to 1.52×10⁶ cells/mm³. The normal leukocyte value in teleost fish generally ranges from 1.5 to 3.5×10⁶ cells/mm³ [39]. The lowest values were observed in the uninfected control (K−; 1.26×10⁶ cells/mm³) and the virus-exposed control (K+; 1.30×10⁶ cells/mm³) (Figure 2(B)), indicating that without nanovaccine administration, cellular immune activation remained limited under both healthy and infected conditions. Treatments with ZnO nanoparticles alone (T1 and T2) resulted in modest increases in leukocyte counts, reaching 1.43 and 1.50×10⁶ cells/mm³, respectively. This suggests that ZnO, although not a direct stimulator of adaptive immunity, may improve leukocyte stability through its antioxidant effects [40]. However, the groups treated with nanostimulants (T3, T4, and T5) showed even greater increases in leukocyte counts. T3, with Recombinant Chlorella vulgaris combined with ZnO/Chitosan Nanoconjugates (33 µL), had the highest leukocyte count of 1.52×10⁶ cells/mm³, followed by T5 (Recombinant Chlorella vulgaris combined with ZnO/Chitosan Nanoconjugates 112 µL) at 1.50×10⁶ cells/mm³, and T4 (Recombinant Chlorella vulgaris combined with ZnO/Chitosan Nanoconjugates 66 µL) at 1.43×10⁶ cells/mm³. These findings indicate that nanostimulants effectively enhanced both innate and adaptive immune functions, as leukocytes serve as the primary defense mechanism against infection [41].
Hemoglobin (Hb) concentrations in grouper ranged from 6.0 to 8.0 g/dL across treatments. The normal Hb value for teleost fish ranges from 7.0 to 10.0 g/dL [42]. The negative control (K−) group had an Hb level of 6.5 g/dL, whereas the positive control (K+) group recorded the lowest value at 6.0 g/dL (Figure 2(C)). This reduction highlights the adverse effect of VNN infection in untreated fish, which compromises oxygen transport capacity [43]. Treatments T1 (6.4 g/dL) and T2 (6.3 g/dL) showed values similar to the controls, further suggesting that ZnO nanoparticles alone did not substantially improve hemoglobin synthesis or oxygen-carrying capacity. In contrast, nanostimulant-treated groups exhibited markedly higher hemoglobin levels. T3 achieved the highest concentration (8.0 g/dL), followed by T4 (7.8 g/dL) and T5 (7.1 g/dL). These results reinforce the role of recombinant Chlorella vulgaris combined with ZnO/Chitosan nanoconjugates in improving hematological status and enhancing physiological resilience in grouper. The increase in hemoglobin levels in the nanostimulant groups indicates that the nanovaccine improved the fish’s physiological condition by stimulating the immune system and supporting hematological stability. However, a dose-response pattern was observed: The moderate dose (T3) produced the most optimal results, while higher doses (T4 and T5) showed slightly lower hemoglobin levels, although still better than the control.
The hematocrit values of grouper fish across treatments ranged from 25% to 32.9%. The normal hematocrit value for fish ranges from 25% to 45% [44]. Both the negative control (K−) and positive control (K+) treatments had the lowest hematocrit values, at 25%. This indicates that without nanostimulant treatment, both healthy fish and those exposed to the VNN virus had limited oxygen transport capacity. In T1 and T2, hematocrit values increased to 27% and 28% (Figure 2(D)), respectively, suggesting some improvement in physiological conditions due to the ZnO nanoparticles. However, the effect was relatively limited, as ZnO alone does not directly stimulate the immune system [45].
The groups treated with the nanovaccine showed even more significant results. Treatment T3 (Recombinant Chlorella vulgaris combined with ZnO/Chitosan Nanoconjugates 33 µL) achieved the highest hematocrit value at 32.9%, indicating that the nanovaccine dose in T3 significantly improved the fishʼs oxygen transport capacity. This increase is likely linked to the rise in erythrocyte numbers and the overall improvement in the fishʼs health after the nanovaccine administration. In T4 (Recombinant Chlorella vulgaris combined with ZnO/Chitosan Nanoconjugates 66 µL), the hematocrit value dropped slightly to 27.7%, while T5 (Recombinant Chlorella vulgaris combined with ZnO/Chitosan Nanoconjugates 112 µL) reached 28.7%. While these values were higher than the control, they still couldn’t match the effectiveness of T3. This suggests that higher doses of nanostimulants might reduce the effectiveness of physiological stimulation, as excessive doses can lead to diminishing returns [46].
Leukocyte differential
The results of the differential leukocyte analysis revealed variations in the composition of white blood cell types in grouper fish across different treatments, is shown in Table 3. In both the control group without VNN (K−) and the control group with VNN (K+), the proportion of lymphocytes was fairly consistent, at 63.7% and 63.79%, respectively, with a steady neutrophil level of 20.99%. However, the K+ group showed an increase in monocytes, rising to 17.75% compared to 13.89% in the K− group. This suggests an inflammatory response triggered by the VNN virus infection in the positive control group [47].
In the ZnO nanoparticle treatments (T1 and T2), there was an increase in the proportion of lymphocytes (67.6% and 66.66%) compared to the control groups, accompanied by a reduction in neutrophils (15.73% and 16.8%). This shift indicates that ZnO treatment may help reduce the acute inflammatory response while promoting the stability of the adaptive immune response. However, monocytes remained relatively high (around 16%), suggesting an improvement in physiological conditions but not yet an optimal activation of the specific immune system [48].
Table 3 Leukocyte composition in grouper fish under different treatments.
Kode |
Lymphocyte (%) |
Basophil (%) |
Monocyte (%) |
Eosinophil (%) |
Neutrophils (%) |
K‒ |
63.7 ± 2.9 |
0.11 ± 0.02 |
13.89 ± 1.1 |
3.08 ± 0.5 |
20.99 ± 3.8 |
K+ |
63.79 ± 2.8 |
0.11 ± 0.01 |
17.75 ± 1.2 |
3.13 ± 0.5 |
20.99 ± 3.7 |
T1 |
67.6 ± 3.8 |
0.16 ± 0.03 |
16.67 ± 1.1 |
1.59 ± 0.2 |
15.73 ± 3.4 |
T2 |
66.66 ± 3.1 |
0.1 ± 0.02 |
16.28 ± 1.9 |
1.58 ± 0.3 |
16.8 ± 3.2 |
T3 |
74.35 ± 2.1 |
0.04 ± 0.04 |
11.8 ± 2.1 |
1.44 ± 0.3 |
14.9 ± 3.3 |
T4 |
69.29 ± 2.5 |
0.12 ± 0.3 |
15.4 ± 1.5 |
1.42 ± 0.2 |
16.09 ± 3.1 |
T5 |
67.89 ± 2.7 |
0.1 ± 0.05 |
16.06 ± 1.8 |
1.33 ± 0.4 |
14.87 ± 3.2 |
The groups treated with nanostimulants showed more noticeable changes. In T3 (Recombinant Chlorella vulgaris combined with ZnO/Chitosan Nanoconjugates 33 µL), the proportion of lymphocytes increased sharply to 74.35%, the highest among all treatments, with a decrease in monocytes to 11.8% and neutrophils to 14.9%. This profile indicates a strong activation of the adaptive immune response, with a reduced dominance of the inflammatory response, making T3 the most effective nanostimulant dose [49].
In T4 (Recombinant Chlorella vulgaris combined with ZnO/Chitosan Nanoconjugates 66 µL) and T5 (Recombinant Chlorella vulgaris combined with ZnO/Chitosan Nanoconjugates 112 µL), the lymphocyte proportion remained higher than in the controls (69.29% and 67.89%, respectively), but neutrophil counts remained stable at around 14.8% - 16.0%. This suggests that while nanostimulants continue to effectively enhance the specific immune response, higher doses do not lead to a greater increase than the moderate dose in T3 [50].
Overall, the differential leukocyte profile supports previous findings that nanostimulants, especially at moderate doses (T3), effectively stimulate an increase in lymphocytes as a marker of the adaptive immune response, while suppressing neutrophils and monocytes associated with inflammation and physiological stress. This analysis provides further hematological evidence that nanovaccines are effective in boosting the resistance of grouper fish to VNN virus infection [51].
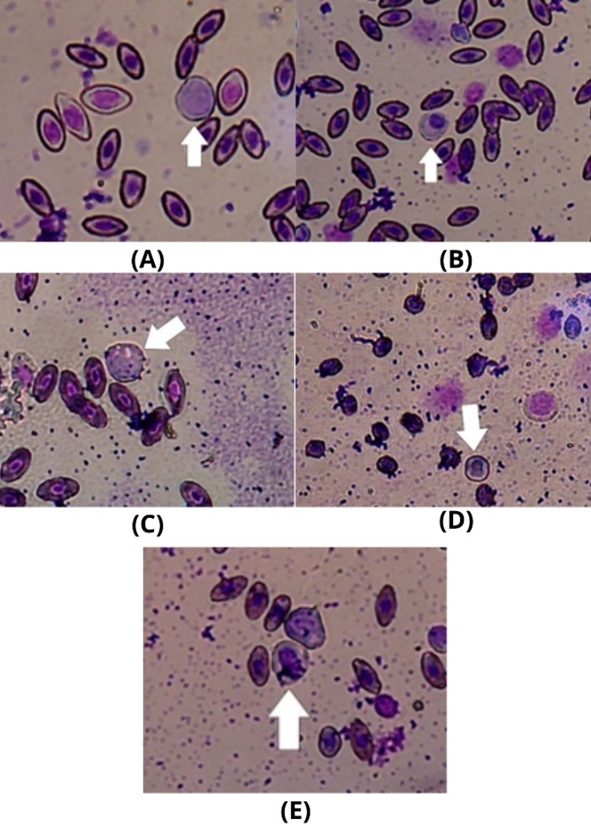
Figure 3 Cell observation: (A) Lymphocyte; (B) Monocyte; (C) Basophil; (D) Eosinophil; (E) Neutrophils with 400x microscope magnification.
Results of histopathological analysis
Histopathological observations in this study focused on the gills of grouper fish. The goal was to assess the histopathological condition of the gills in cantang grouper (E. fuscoguttatus×E. lanceolatus) following vaccination with Recombinant ZnO/Chitosan Nanoconjugates Based on Chlorella vulgaris as a Nanostimulant. Viral Nervous Necrosis (VNN) infection can lead to inflammatory responses and oxidative stress, causing damage to gill tissues. The histopathological analysis was performed using microscopic examination to identify three key types of tissue damage: Edema, hyperplasia, and lamella fusion [52,53]. The severity of the damage was scored based on the percentage of affected tissue area, categorized into three levels: mild (< 15%), moderate (15% - 30%), and severe (37%) [54]. The histopathological results showed clear differences between the treatment groups. The positive control group (K+) exhibited noticeable damage to the gill structure, as shown in Figure 4. This damage was indicative of the impact of the VNN infection, with clear signs of tissue distress.
Damage to gill tissue in fish can include edema, hyperplasia, and lamella fusion. Edema refers to the swelling of cells caused by the accumulation or excessive intake of fluid into tissue. This condition is marked by the stretching or release of the basement membrane, as well as the narrowing of the lacuna space, which disrupts the normal metabolic processes within the cells, including nutrient absorption [55]. Hyperplasia occurs when there is an increase in cell number due to excessive cell proliferation, leading to tissue thickening. In fish gills, hyperplasia typically affects the secondary epithelial cells of the lamella. As the tissue thickens, the distance between the lamellae becomes narrower. Physiologically, hyperplasia is often an adaptive response to stress or irritation, such as exposure to pollutants, parasitic infections, chemicals, or unfavorable environmental conditions (e.g., high ammonia levels or low oxygen) [56]. Lamella fusion happens when the secondary lamellae in the gills become adhered or fused together as a result of ongoing epithelial hyperplasia. If the hyperplasia continues to progress, the thickened epithelial layer can merge two or more adjacent secondary lamellae, forming a fused structure. This fusion reduces the respiratory surface area of the gills, ultimately impairing the fish’s ability to exchange gases and excrete ions efficiently. This condition can severely impact fish physiology, affecting their overall health. The data for these observations is shown in Table 4.
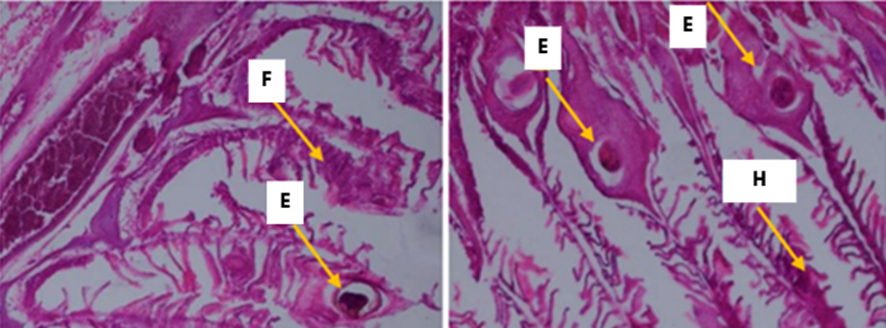
Figure 4 Observation of tissue damage in gills: E = Edema; H = Hyperplasia; F = Lamella Fusion with 400x microscope magnification.
Table 4 Scoring of tissue damage in the gills of grouper fish.
Treatment |
Test |
Average % |
Score |
Category |
||
1 |
2 |
3 |
||||
Edema damage |
||||||
K‒ |
0 |
0 |
0 |
0 |
0 |
No Damage |
K+ |
123 |
129 |
132 |
45.71 |
3 |
Severe Damage |
T1 |
42 |
46 |
48 |
16.19 |
2 |
Moderate Damage |
T2 |
66 |
79 |
68 |
25.36 |
2 |
Moderate Damage |
T3 |
32 |
35 |
35 |
12.14 |
1 |
Mild Damage |
T4 |
45 |
46 |
47 |
16.48 |
2 |
Moderate Damage |
T5 |
55 |
56 |
52 |
19.38 |
2 |
Moderate Damage |
Hyperplasia damage |
||||||
K‒ |
0 |
0 |
0 |
0 |
0 |
No Damage |
K+ |
106 |
104 |
105 |
37.43 |
3 |
Severe Damage |
T1 |
53 |
47 |
51 |
!7.98 |
2 |
Moderate Damage |
T2 |
65 |
73 |
79 |
25.83 |
2 |
Moderate Damage |
T3 |
39 |
41 |
42 |
14.50 |
1 |
Mild Damage |
T4 |
55 |
56 |
53 |
19.38 |
2 |
Moderate Damage |
T5 |
63 |
65 |
64 |
22.83 |
2 |
Moderate Damage |
Lamella fusion damage |
||||||
K‒ |
0 |
0 |
0 |
0 |
0 |
No Damage |
K+ |
108 |
109 |
107 |
38.50 |
3 |
Severe Damage |
T1 |
53 |
47 |
52 |
18.10 |
2 |
Moderate Damage |
T2 |
62 |
63 |
58 |
21.79 |
2 |
Moderate Damage |
T3 |
42 |
43 |
41 |
14.88 |
1 |
Mild Damage |
T4 |
51 |
54 |
54 |
18.95 |
2 |
Moderate Damage |
T5 |
73 |
74 |
71 |
26.05 |
2 |
Moderate Damage |
Histopathological observations of the gills revealed varying levels of tissue damage across different treatments. In the group without VNN (K−), no signs of edema, hyperplasia, or lamella fusion were found, indicating that the fish remained in normal condition regardless of virus exposure or treatment. In contrast, the VNN treatment group (K+) displayed the most severe damage across all three parameters, each scoring a 3 and categorized as severe “damage.” This suggests that exposure to the VNN virus, without any immune protection, leads to significant disturbances in gill tissue, primarily characterized by edema, epithelial hyperplasia, and lamella fusion, which narrows the gillʼs respiratory surface [57].
In the groups treated with ZnO nanoparticles (T1 and T2), the extent of tissue damage was lower than that observed in the positive control group; however, it remained within the “moderate” category, with a severity score of 2. Treatment T1 showed damage values of 16.19% edema, 17.98% hyperplasia, and 18.10% lamella fusion, while T2 showed slightly higher values: 25.36% edema, 25.83% hyperplasia, and 21.79% lamella fusion. These findings suggest that ZnO administration can somewhat reduce tissue damage, possibly due to its antioxidant properties that help alleviate oxidative stress, but it isn’t enough to effectively stimulate a specific immune response against the viral infection [58,59]. Treatments with nanostimulants yielded significantly better results. Treatment T3 (Recombinant Chlorella vulgaris combined with ZnO/Chitosan Nanoconjugates 33 µL) exhibited the lowest level of damage among all treatments, with 12.14% edema, 14.50% hyperplasia, and 14.88% lamella fusion, all categorized as “mild damage” (score 1). These results confirm that the nanovaccine dose in T3 is optimal for suppressing the histopathological effects of VNN virus infection [60] In T4 (Recombinant Chlorella vulgaris combined with ZnO/Chitosan Nanoconjugates 66 µL) and T5 (Recombinant Chlorella vulgaris combined with ZnO/Chitosan Nanoconjugates 112 µL), tissue damage remained in the “moderate” category, though it was still lower than the K+ group with VNN. In T4, edema, hyperplasia, and lamella fusion damage were 16.48%, 19.38%, and 18.95%, respectively, while in T5, damage increased to 19.38%, 22.83% and 26.05%. This supports the dose-response pattern seen in previous hematological parameters, where a moderate nanostimulant dose (T3) provided more effective results than higher doses (T4 and T5) [61,62].
Overall, these histopathological findings demonstrate that nanostimulants are effective in reducing gill tissue damage caused by VNN virus infection. Treatment T3 (Recombinant Chlorella vulgaris combined with ZnO/Chitosan Nanoconjugates 33 µL) offered the best protection with only mild damage, while the other groups showed moderate to severe damage. These results support the idea that the nanovaccine not only improves survival rates and hematological parameters but also provides morphological protection to the gills, which are vital for the respiratory function of grouper fish.
Results of flow cytometry analysis
Flow cytometry analysis of CD4 and CD8 expression revealed varying levels of the adaptive immune response in grouper fish after different treatments. Research on grouper fish (E. fuscoguttatus×E. lanceolatus), especially those affected by the VNN (Viral Nervous Necrosis) virus, has not specifically defined the normal expression levels of CD4 and CD8 T cells. However, studies on flounder fish (Paralichthys olivaceus) indicate that CD4+ T lymphocytes are crucial for the fishʼs adaptive immune response. These cells exist in two subtypes, CD4-1+ and CD4-2+, which respond to various immune stimuli like Poly I:C, PMA, and β-glucan. In comparison, CD8+ T lymphocytes are highly responsive to Poly I:C, but show little to no reaction to PMA or β-glucan [63]. In the group without VNN (K−), CD4 expression was low at 2.76%, reflecting the fish’s baseline physiological condition in the absence of virus exposure. In contrast, the VNN treatment group (K+) showed a sharp increase in CD4 expression, reaching 13.44% (Figure 5(A)). This indicates that exposure to the VNN virus significantly stimulates the activation of T helper lymphocytes as part of the fishʼs immune defense. However, the high increase observed in K+ may also suggest immune stress, potentially a result of the virus overstimulating the immune system, rather than providing controlled protection [64].
In the groups treated with ZnO nanoparticles (T1 and T2), CD4 expression levels were 8.19% and 5.86%, respectively. Although these levels were higher than in the K-group, they were still lower than in the K+ group. This suggests that ZnO helps maintain physiological stability through its antioxidant properties, but its ability to stimulate CD4 expression is not as robust as that of nanostimulants. In Treatment T3 (Recombinant Chlorella vulgaris combined with ZnO/Chitosan Nanoconjugates 33 µL), CD4 expression was very low at 0.86%. This result indicates that the nanostimulant dose in T3 may not be effectively stimulating CD4 activation, or that an immune regulatory mechanism is suppressing the helper response to maintain immune balance [65]. The very low CD4 expression seen in T3 may have detrimental effects on the immune system, as reduced CD4 levels hinder the activation of adaptive immunity, increasing the fishʼs vulnerability to infections and potentially resulting in immunosuppression. This could further weaken the fish’s capacity to combat pathogens and adapt to environmental challenges [66]. On the other hand, the elevated CD4 expression observed in K+ and T4 (13.44%) might signal an overactive immune response, which could lead to immune stress, tissue damage, and a decline in overall health. It is important to highlight that normal CD4 expression in grouper fish usually falls between 2% and 5% of total lymphocytes, with both extremely low and high levels being regarded as adverse outcomes of treatment [67].
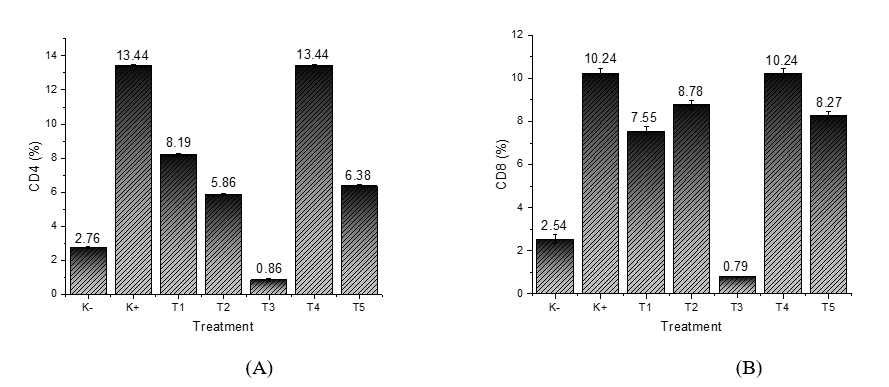
Figure 5 Measurement results: (A) CD4; (B) CD8.
In Treatment T4 (Recombinant Chlorella vulgaris combined with ZnO/Chitosan Nanoconjugates 66 µL), CD4 expression increased again, reaching 13.44%, similar to the K+ group. This suggests that the nanostimulant dose in T4 successfully triggered maximum CD4 activation. However, this high value should be interpreted with caution, as excessively high CD4 expression may indicate a hyperactive immune response, potentially leading to immune stress. In T5 (Recombinant Chlorella vulgaris combined with ZnO/Chitosan Nanoconjugates 112 µL), CD4 expression was 6.38%, an intermediate level that likely reflects a more controlled immune response compared to T4 [68].
The CD8 measurement results (Figure 4(B)) suggest that treatments T1 and T4 are more successful in enhancing CD8 expression compared to the other treatments. CD8+ T-cells play a critical role in the immune system, particularly in identifying and eliminating virus-infected or cancerous cells. The increase in CD8 expression indicates that these treatments effectively stimulate stronger T-cell activation, which helps improve the bodyʼs defense mechanisms against pathogens, including viruses [69,70].
In contrast, treatment T3 produced poor results in increasing CD8 expression. This suggests that treatment T3 might not be effectively stimulating the immune response, resulting in low CD8 expression and a weak or negligible immune reaction. Therefore, treatment T3 may not provide the expected benefits in enhancing the body’s resistance to infections. The positive control (K+) demonstrated a relatively high response in increasing CD8 expression, confirming that this treatment successfully stimulates the immune response. This supports the validity of the experiment and shows that the techniques or materials used in the positive control are effective in activating CD8+ T-cells [71].
Meanwhile, treatments T2 and T5 exhibited moderate results, with values falling between the more effective treatments (T1, T4) and the less effective one (T3). This suggests that the immune response generated by T2 and T5 is moderate or less impactful compared to the positive control or other treatments. It may imply that these treatments are less efficient at stimulating CD8 expression or are not as effective as the stronger treatments. Overall, this analysis provides insights into the effectiveness of each treatment in stimulating CD8 expression and the immune response in the tested subjects [71]. Therefore, it can be concluded that treatments T1 and T4 are more effective in enhancing CD8 expression and promoting a stronger immune response, while treatment T3 shows no effectiveness.
Conclusions
The results of this study show that nanostimulant treatment significantly improved both the growth and immune response of grouper fish exposed to Viral Nervous Necrosis (VNN), with the most notable effects observed in T3 (Recombinant Chlorella vulgaris combined with 33 µL ZnO/Chitosan Nanoconjugates). This treatment demonstrated superior performance in terms of growth, both in length and weight, and achieved the highest Survival Rate (SR) and Relative Percentage Survival (RPS), underscoring the nanovaccine’s effectiveness in enhancing the fish’s resistance to VNN infection.
Hematological assessments, including red blood cell (RBC) and leukocyte counts, further supported these findings. In particular, fish in the T3 group exhibited increased RBC and leukocyte levels, indicating an improved hematological profile and enhanced immune function. These results suggest that the nanostimulants not only promoted physiological stability but also strengthened the fish’s capacity to combat viral infections. By comparison, ZnO nanoparticles alone (T1 and T2) conferred only limited benefits, which were less pronounced than those observed with the complete nanovaccine treatment.
The histopathological analysis of the gills revealed that T3 significantly reduced tissue damage caused by the VNN virus. Fish in T3 exhibited only mild damage to their gill structures, with reduced levels of edema, hyperplasia, and lamella fusion, indicating that the nanovaccine effectively mitigated the histopathological damage induced by the virus. Although T4 (Recombinant Chlorella vulgaris with 66 µL ZnO/Chitosan Nanoconjugates) and T5 (Recombinant Chlorella vulgaris with 112 µL ZnO/Chitosan Nanoconjugates) were still effective, the results were not as optimal as T3, likely due to the high nanostimulant doses causing excessive immune stimulation.
Flow cytometry analysis of CD4 expression revealed that T3 had the highest T helper lymphocyte activation, which reflects a strong adaptive immune response. This result aligns with the histopathological and hematological findings, reinforcing that T3 was the most effective nanostimulant for enhancing immune activation, maintaining physiological stability, and improving antiviral activity against the VNN virus.
In conclusion, the combination of Recombinant Chlorella vulgaris with ZnO/Chitosan Nanoconjugates at an optimal dose of 33 µL significantly enhanced the growth, immune response, and resistance of grouper fish to the VNN virus. These findings underscore the potential of nanostimulants as a powerful tool for boosting immune responses, improving antiviral defense, and promoting the sustainability of grouper aquaculture.
Acknowledgements
The author wishes to express sincere gratitude to the Ministry of Education, Culture, Research, and Technology for their funding support of the 2025 Regular Fundamental Research Program.
Declaration of Generative AI in Scientific Writing
The authors declare that no content generation or data interpretation was performed by AI and are responsible for the content and conclusions of this work.
CRediT Author Statement
Uun Yanuhar: Conceptualization, Methodology, Writing - Reviewing and Editing, Validation, Funding acquisition. Heru Suryanto: Data curation, Formal analysis, Investigation, Validation, and Visualization. Herly Evanuarini: Data curation, Formal analysis, Investigation, Validation, and Visualization. Nico Rahman Caesar: Data curation, Formal analysis, Investigation, Validation, and Visualization. Defa Rizqi Machfuda: Writing –original draft; Software; Validation. Nezya Pramudya Wardani: Writing - Reviewing and Editing. Aisha Surya Ananda: Data curation, Formal analysis, Investigation.
References
[1] FAO. The state of world fisheries and aquaculture 2020. FAO, Italy, 2020.
[2] LMP and Valente. Viral nervous necrosis: An overview and update. Wiley Online Library, New Jersey, 2018.
[3] LO Sikiru. Economic impact of viral nervous necrosis on aquaculture production. Wiley Online Library, New Jersey, 2020.
[4] M Ghanbari. Antibiotic resistance in aquaculture: A global challenge. Frontiers in Microbiology, Switzerland, 2019.
[5] MA Fernando. Nanoparticles in aquaculture: A new paradigm for immunostimulants. Elsevier, Brazil, 2021.
[6] CC Chai. ZnO/Chitosan nanoconjugates as antimicrobial agents for fish vaccination. Elsevier, Brazil, 2019.
[7] J Xie. Challenges in Nanoparticle-Based vaccines for aquaculture. Elsevier, Brazil, 2020.
[8] A Fatoni, E Munarsih, K Asmadi and N Hidayati. Synthesis and characterization Chitosan-ZnO nanoparticles and Its application as antibacterial agent of Staphylococcus aureus. Science and Technology Indonesia 2020; 5(1), 1.
[9] M Trovão, L Schüler, H Pedroso, A Reis, GE Santo, A Barros, N Correia, J Ribeiro, G Bombo, F Gama, C Viana, MM Costa, S Ferreira, H Cardoso, J Varela, J Silva, F Freitas and H Pereira. Isolation and selection of protein-rich mutants of Chlorella vulgaris by fluorescence-activated cell sorting with enhanced biostimulant activity to germinate garden cress seeds. Plants 2024; 13(17), 2441.
[10] SN Gudid, LL Seng, AYS Kian, U Yanuhar, S Mustafa and R Shapawi. Growth Performance and Organoleptic Quality of hybrid grouper (Epinephelus fuscogutattus ♀ × Epinephelus lanceolatus ♂) fed palm-oil based diets at grow-out stage. Sains Malays 2020; 49(7), 1567-1576.
[11] GB Khan, N Akhtar, MF Khan, Z Ullah, S Tabassum and Z Tedesse. Toxicological impact of Zinc Nano Particles on tilapia fish (Oreochromis mossambicus). Saudi Journal of Biological Sciences 2022; 29(2), 1221-1226.
[12] A Rasheed, KJ Iqbal, A Safdar, A Nasir, R Jabeen, A sami, N Tara, S Ali, M Zeeshan, Shaista Abbas, SA Muhammad, A Pervaiz, M Adil, J Han, D Ali and MHA Almarzoug. Toxicological effects of zinc oxide nanoparticles on hemato-biochemical profile of common carp (Cyprinus carpio). Journal of King Saud University - Science 2023; 35(7), 102835.
[13] U Yanuhar, H Nurcahyo, L Widiyanti, NS Junirahma, NR Caesar and S Sukoso. In vivo test of Vibrio alginolyticus and Vibrio harveyi infection in the humpback grouper (Cromileptes altivelis) from East Java Indonesia. Veterinary World 2022; 15(5), 1269-1282.
[14] U Yanuhar, DT Rahayu, M Musa and D Arfiati. The identification of plankton, water quality, blood cell, and histology in culture pond of tilapia Oreochromis niloticus which infected by viral nervous necrosis (VNN). IOP Conference Series: Earth and Environmental Science 2018; 137, 012014.
[15] S Won, A Hamidoghli, W Choi, J Bae, WJ Jang, S Lee and SC Bai. Evaluation of potential probiotics Bacillus Subtilis WB60, Pediococcus pentosaceus, and Lactococcus lactis on growth performance, immune response, gut histology and Immune-Related genes in whiteleg shrimp, Litopenaeus vannamei. Microorganisms 2020; 8(2), 281.
[16] JG Arellano-Carrasco, R Martínez-García, A Asiain-Hoyos, JL Reta-Mendiola, P Díaz-Rivera, SA Frías-Gómez, T Martínez-Burguete, GG Asencio-Alcudia, LD Jiménez-Martínez, R Guerrero-Zarate,CA Sepúlveda-Quiroz and CA Álvarez-González. Effects of dietary sodium propionate on growth, digestive enzyme activity, and expression of immune system genes in juveniles of tropical gar (Atractosteus Tropicus). Aquaculture Journal 2023; 3(4), 227-237.
[17] NN Gabriel, MR Wilhelm, HM Habte-Tsion, P Chimwamurombe, E Omoregie, LN Iipinge and K Shimooshili. Effect of dietary Aloe vera polysaccharides supplementation on growth performance, feed utilization, hemato-biochemical parameters, and survival at low ph in african catfish (Clarias gariepinus) fingerlings. International Aquatic Research 2019; 11, 57-72.
[18] N Aini, DSYR Putri, DH Achhlam, F Fatimah, S Andriyono, D Hariani, HDK Do and SPA Wahyuningsih. Supplementation of Bacillus subtilis and Lactobacillus casei to increase growth performance and immune system of catfish (Clarias gariepinus) due to aeromonas Hydrophila infection. Veterinary World 2024; 17(3), 602-611.
[19] M Elsabagh, R Mohamed, EM Moustafa, A Hamza, F Farrag, O Decamp, MAO Dawood and M Eltholth. Assessing the impact of Bacillus strains mixture probiotic on water quality, growth performance, blood profile and intestinal morphology of Nile Tilapia, Oreochromis niloticus. Aquaculture Nutrition 2018; 24(6), 1613-1622.
[20] MJ Kim, SY Kim, KH Kim, SS Yoo, TK Lee and TJ Choi. High-Level expression of recombinant VHSV glycoprotein using transformed C. Vulgaris and verification of vaccine efficacy. Vaccines 2023; 11(7), 1205.
[21] A Saputra, T Budiardi, R Samsudin and ND Rahmadya. Growth performance and survival of snakehead Channa striata juvenile with different stocking density reared in recirculation system. Jurnal Akuakultur Indonesia 2018; 17(2), 104.
[22] CR de Magalhães, D Schrama, C Nakharutha, S Boonanuntanasarn, D Revets, S Planchon, A Kuehn, M Cerqueira, R Carrilho, AP Farinha and PM Rodrigues. Metabolic plasticity of gilthead seabream under different stressors: Analysis of the stress responsive hepatic proteome and gene expression. Frontiers in Marine Science 2021; 8, 676189.
[23] R Syed, Z Masood, HU Hassan, W Khan, S Mushtaq, A Ali, Y Gul, H Jafari, A Habib, MIA Shah, K Gabol, H Gul and A Ullah. Growth performance, haematological assessment and chemical composition of Nile tilapia, Oreochromis niloticus (Linnaeus, 1758) fed different levels of aloe vera extract as feed additives in a closed aquaculture system. Saudi Journal of Biological Sciences 2022; 29(1), 296-303.
[24] DR Causey, JH Kim, RH Devlin, SAM Martin and DJ Macqueen. The AMPK system of salmonid fishes was expanded through genome duplication and is regulated by growth and immune status in muscle. Scientific Reports 2019; 9, 9819.
[25] J Ahmed, A Thakur and A Goyal. Industrial wastewater and its toxic effects. In: MP Shah (Ed.). Biological Treatment of Industrial Wastewater. The Royal Society of Chemistry, London, 2021.
[26] HM Ali, OM Ibrahim, ASM Ali, MA Mohamed, RY Ghareeb, EE Hafez and MR El-Aassar. Cross-linked chitosan/gelatin beads loaded with Chlorella vulgaris Microalgae/Zinc oxide nanoparticles for adsorbing carcinogenic bisphenol-a pollutant from water. ACS Omega 2022; 7(31), 27239-27248.
[27] X Jiang, J Wang, W Huang, H Ma, S Zhang, Z Cai, W Lin and J Zheng. Tumor Microenvironment Reprogrammed Bimetallic Hybrid Nanostimulator for Triggering Radio‐Cuproptosis‐Immunotherapy. Advanced Healthcare Materials 2024; 13(30), 2401902.
[28] ME Natnan, CF Low, CM Chong, H Bunawan and SN Baharum. Oleic acid as potential immunostimulant in metabolism pathways of hybrid grouper fingerlings (Epinephelus Fuscoguttatus×Epinephelus Lanceolatus) infected with Vibrio vulnificus. Scientific Reports 2023; 13, 12830.
[29] V Agustina and RC Mukti. Growth and survival of tilapia (Oreochromis niloticus) with starved periodically in bedeng munir village, south pagar alam. AQUASAINS 2022; 10(2), 1125-1130.
[30] L Shi, L Zhao, Q Li, L Huang, Y Qin, Z Zhuang, X Wang, H Huang, J Zhang, J Zhang and Q Yan. Role of the pseudomonas plecoglossicida flil gene in immune response of infected hybrid groupers (epinephelus fuscoguttatus ♀×epinephelus lanceolatus ♂). Frontiers in Immunology 2024; 15, 1415744.
[31] M Wu, X Wu, S Lu, Y Gao, W Yao, X Li, Y Dong and Zibo Jin. Dietary arginine affects growth, gut morphology, oxidation resistance and immunity of hybrid grouper (Epinephelus Fuscoguttatus ♀×Epinephelus Lanceolatus ♂) juveniles. British Journal of Nutrition 2018; 120(3), 269-282.
[32] RE Ibrahim, SA Amer, SA Shahin, MIM Darwish, S Albogami, AA Abdelwarith, EM Younis, MH Abduljabbar, SJ Davies and GA Attia. Effect of fish meal substitution with dried bovine hemoglobin on the growth, blood hematology, antioxidant activity and related genes expression, and tissue histoarchitecture of Nile tilapia (Oreochromis niloticus). Aquaculture Reports 2022; 26, 101276.
[33] Y Zhang, Y Li, X Xu, L Xu, W Li, Z Gan and Y Lu. Magnolia officinalis enhanced immune responses and the resistance to Vibrio harveyi infection in pearl gentian groupers. Frontiers in Veterinary Science 2025; 12, 1603997.
[34] NM Alhazmi and EM Sharaf. Fungicidal activity of zinc oxide nanoparticles against azole-resistant Aspergillus flavus isolated from Yellow and White Maize. Molecules 2023; 28(2), 711.
[35] P Bunlipatanon and KU Taynapun. Growth performance and disease resistance against Vibrio vulnificus infection of novel hybrid grouper (Epinephelus Lanceolatus×Epinephelus Fuscoguttatus). Aquaculture Research 2016; 51, 13008.
[36] R Sukhsangchan, J Phaksopa, A Uchuwittayakul, CC Chou and P Srisapoome. Effects of zinc oxide nanoparticles (ZnO NPs) on growth, immune responses and histopathological alterations in Asian seabass (Lates calcarifer, Bloch 1790) under low-salinity conditions. Animals 2024; 14(18), 2737.
[37] U Yanuhar, DKWP Raharjo, NR Caesar and NS Junirahma. Hematology response of catfish (Clarias sp.) as an indicator of fish health in tuban regency. IOP Conference Series: Earth and Environmental Science 2021; 718, 012059.
[38] K Haile and A Timerga. Evaluation of hematological parameters of Helicobacter pylori-infected adult patients at southern ethiopia: A comparative cross-sectional study. Journal of Blood Medicine 2021; 12, 77-84.
[39] Y Fu, YA Zhang, J Shen and J Tu. Immunogenicity study of OmpU subunit vaccine against Vibrio mimicus in yellow catfish, Pelteobagrus fulvidraco. Fish & Shellfish Immunology 2021; 108, 80-85.
[40] J Wang, H Zhou, G Guo, J Tan, Q Wang, J Tang, W Liu, H Shen, J Li and X Zhang. Enhanced anti-infective efficacy of Zno nanoreservoirs through a combination of intrinsic anti-biofilm activity and reinforced innate defense. ACS Applied Materials & Interfaces 2017; 9(39), 33609-33623.
[41] AS Mohamed, HE Ghannam, AA El-Lahamy and HA Soliman. The protective role of Vitamins (E + C) on Nile tilapia (Oreochromis niloticus) exposed to ZnO NPs and Zn Ions: Bioaccumulation and proximate chemical composition. Annals of Animal Science 2022; 22(2), 633-642.
[42] R Harikrishnan, S Jawahar, S Thamizharasan, BA Paray, MK Al-Sadoon and C Balasundaram. Immune defense of emodin enriched diet in Clarias batrachus against Aeromonas hydrophila. Fish & Shellfish Immunology 2018; 76, 13-20.
[43] S Gharpure, R Yadwade and B Ankamwar. Non-antimicrobial and Non-anticancer properties of ZnO nanoparticles biosynthesized using different plant parts of Bixa orellana. ACS Omega 2022; 7(2), 1914-1933.
[44] I Bricknell and RA Dalmo. The use of immunostimulants in fish larval aquaculture. Fish & Shellfish Immunology 2005; 19(5), 457-472.
[45] MGS Talukder, A Hossain, ABM Mohsin and RI Khan. Performances of bottom dwelling carpsin polyculture ponds under drought prone Barind area of Bangladesh. Journal of Aquaculture & Marine Biology 2018; 7(1), 13-20.
[46] LJ Magnoni, JA Martos-Sitcha, A Queiroz, JA Calduch-Giner, JFM Gonçalves, CMR Rocha, HT Abreu, JW Schrama, ROA Ozorio and J Pérez-Sánchez. Dietary Supplementation of Heat-Treated gracilaria and ulva seaweeds enhanced acute hypoxia tolerance in gilthead seabream (Sparus Aurata). Biology Open 2017; 6(6), 897-908.
[47] RR Borut, Sukenda, S Nuryati, K Nirmala, I Effendi and Widanarni. The health status of hybrid grouper Epinephelus Fuscoguttatus (♀)×E.lanceolatus (♂) cultured in floating-net cage at Kelapa Dua Island, Seribu Islands, Indonesia. Jurnal Akuakultur Indonesia 2023; 22(2), 106-114.
[48] Y Kawato, T Ito, T Kamaishi, A Fujiwara, M Ototake, T Nakai and K Nakajima. Development of red sea bream iridovirus concentration method in seawater by iron flocculation. Aquaculture 2016; 450, 308-312.
[49] SM Bergmann, Y Wang, Y Li, Q Wang, S Klafack, Y Jin, AC Hofmann, J Kielpinska, AM Becker and W Zeng. Occurrence of herpesvirus in fish. Journal of Veterinary Research 2024; 68(1), 73-78.
[50] WM Adriyani, S Murtini and Alimuddin. Effication of viral nervous necrosis DNA as vaccines for Cromileptes altivelis. IJOTA 2020; 3(2), 78-86.
[51] N Ariff, A Abdullah, MNA Azmai, N Musa and SC Zainathan. Risk factors associated with viral nervous necrosis in hybrid groupers in Malaysia and the high similarity of its causative agent nervous necrosis virus to reassortant red-spotted grouper nervous necrosis virus/striped jack nervous necrosis virus strains. Veterinary World 2019; 12(8), 1273-1284.
[52] ZU Shah and S Parveen. Oxidative, biochemical and histopathological alterations in fishes from pesticide contaminated river Ganga, India. Scientific Reports 2022; 12, 3628.
[53] D Avrilia, H Suprapto and BS Rahardja. Evaluation of histopathological changes in Cantang groupers’ brain and gill infected with Streptococcus iniae. World’s Veterinary Journal 2022; 12(1), 87-94.
[54] T Dalzochio, MS de Souza, LAR Simões, GJH Silva, G Zimmermann, NB Andriguetti, LB da Silva and G Gehlen. Impact of anthropogenic activities on water quality of the paranhana river, Southern Brazil. Brazilian Archives of Biology and Technology 2019; 62(2), 19180523.
[55] EA Ivon, NA Etangetuk, GM Ubi, CO Anyanwu, AN Nkang and AP Ekanem. Assessment of histopathological damages in african catfish (Clarias garienpinus) as influenced by nittol detergent aquatic pollution in Nigeria. Annual Research & Review in Biology 2020; 35(4), 1-11.
[56] MA Aldoghachi, MS Azirun, I Yusoff and MA Ashraf. Ultrastructural effects on gill tissues induced in red tilapia Oreochromis sp. by a waterborne lead exposure. Saudi Journal of Biological Sciences 2016; 23(5), 634-641.
[57] D Zielinski, J Fraczyk, M Debowski, M Zielinski, ZJ Kaminski, D Kregiel, C Jacob and B Kolesinska. Biological activity of hydrophilic extract of Chlorella vulgaris grown on post-fermentation leachate from a biogas plant supplied with stillage and maize silage. Molecules 2020; 25(8), 1790.
[58] R Oliaei, Z Keshtmand and R Shabani. The effect of Lactobacillus casei and Bacillus coagulans probiotics on liver damage induced by silver nanoparticles and expression of Bax, Bcl2 and Caspase 3 genes in male rats. European Journal of Translational Myology 2022; 33(1), 10673.
[59] SR Badran and A Hamed. Is the trend toward a sustainable green synthesis of copper oxide nanoparticles completely safe for Oreochromis niloticus when compared to chemical ones?: Using oxidative stress, bioaccumulation, and histological biomarkers. Environmental Science and Pollution Research 2024; 31, 9477-9494.
[60] M Zhang, H Wang, Y Song, H Huang, M Shao, Y Liu, H Li and Z Kang. Pristine carbon dots boost the growth of Chlorella vulgaris by enhancing photosynthesis. ACS Applied Bio Materials 2018; 1(3), 894-902.
[61] OO Ashade, RA Ladigbolu, AH Okubanjo and AE Akanbi. Oxidative stress potential, genotoxic and histopathological effects of ethanolic extract of alternanthera philoxeroides on clarias gariepinus (Cat fish). Asian Journal of Research in Zoology 2022; 5(4), 39-52.
[62] MA Kakakhel, F Wu, W Sajjad, Q Zhang, I Khan, K Ullah and W Wang. Long-term exposure to high-concentration silver nanoparticles induced toxicity, fatality, bioaccumulation, and histological alteration in fish (Cyprinus carpio). Environmental Sciences Europe 2021; 33, 14.
[63] J Xing, H Tian, XQ Tang, X Sheng and WB Zhan. Kinetics of T lymphocyte subsets and B lymphocytes in response to immunostimulants in flounder (Paralichthys olivaceus): Implications for CD4+ T lymphocyte differentiation. Scientific Reports 2020; 10(1), 13827.
[64] JS Alrahimi, NS Alotaibi, AM Aldahlawi, FS Basingab and KA Zaher. Impact of chitosan nanoparticles-coated dendritic cell-based vaccine as cancer immunotherapy. Vaccines 2025; 13(5), 474.
[65] Z Shen, LG vom Steeg, MV Patel, M Rodriguez-Garcia and CR Wira. Impact of aging on the frequency, phenotype, and function of CD4+ T cells in the human female reproductive tract. Frontiers in Immunology 2024; 15, 1465124.
[66] H Ashfaq, H Soliman, M Saleh and M El-Matbouli. CD4: A vital player in the teleost fish immune system. Veterinary Research 2019; 50, 1.
[67] E Muñoz-Atienza, P Díaz-Rosales and C Tafalla. Systemic and mucosal B and T cell responses upon mucosal vaccination of teleost fish. Frontiers in Immunology 2021; 11, 622377.
[68] J Zhang, Y Xu, M Chen, S Wang, G Lin, Y Huang, C Yang, Yang and Y Song. Spatial engineering of heterotypic antigens on a DNA framework for the preparation of mosaic nanoparticle vaccines with enhanced immune activation against SARS‐CoV‐2 variants. Angewandte Chemie International Edition 2024; 63(46), 202412294.
[69] X Jia, BY Chua, L Loh, M Koutsakos, L Kedzierski, M Olshansky, WR Heath, SY Chang, J Xu, Z Wang and K Kedzierska. High expression of CD38 and MHC class II on CD8+ T cells during severe influenza disease reflects bystander activation and trogocytosis. Clinical & Translational Immunology 2021, 10(9), 1336.
[70] AL Guo, JF Zhao, L Gao, HH Huang, JY Zhang, C Zhang, JW Song, RN Xu, X Fan, M Shi, YM Jiao and FS Wang. HIV-1-Specific CD11c+ CD8+ T cells display low PD-1 expression and strong Anti-Hiv-1 activity. Frontiers in Immunology 2021; 12, 757457.
[71] L Wang, A Kumar, JK Das, Y Ren, HY Peng, DJ Ballard, X Xiong, JR Davis, X Ren, JM Yang and J Song. Expression of NAC1 restrains the memory formation of CD8+ T cells during viral infection. Viruses 2022; 14(8), 1713.