
Trends
Sci.
2026;
23(5):
12030
In Vivo Evaluation of the Immunomodulatory and Anti-Inflammatory Effects of Crude Red Fruit (Pandanus Conoideus Lamk.) Oil
Zita
Letviany Sarungallo1,
![]() ,
Budi Santoso1,*,
Mathelda Kurniaty Roreng1,
,
Budi Santoso1,*,
Mathelda Kurniaty Roreng1,
Indah
Epriliati2,
![]() and Mardiah3
and Mardiah3
1Study Program of Agricultural Product Technology, Faculty of Agricultural Technology, Papua University,
West Papua 98314, Indonesia
2Study Program of Food Technology, Faculty of Agricultural Technology, Catholic Widya Mandala University,
East Java 60265, Indonesia
3Study Program of Food Technology, Postgraduate School, Djuanda University, West Java 16720, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 24 September 2025, Revised: 21 October 2025, Accepted: 28 October 2025, Published: 1 January 2026
Abstract
This study evaluates the effectiveness of crude red fruit oil (CRFO) as an immunomodulator and anti-inflammatory agent through in vivo testing on Sprague Dawley (SD) rats. The red fruit oil was extracted using a dry extraction method and administered to male rats, which were divided into six treatment groups (n=5) in a completely randomized design. Acute inflammation was induced using carrageenan injection, while immunomodulatory effects were tested through the cotton pellet granuloma method. The treatment groups consisted of normal control, negative (Na-CMC 0.5%), positive (imboost) and CRFO at doses of 0.081, 0.243 and 0.813 mL/kg body weight, except normal control, all treatment groups were involving phagocytosis and acute-chronic inflammation. Data were analyzed using Analysis of Variance (ANOVA) followed by Duncan’s Multiple Range Test for statistical significance (p-value < 0.05). The results showed that CRFO contained 8,135 ± 152 mg/kg carotenoids and 15,482 ± 122 mg/kg tocopherols, contributing to its antioxidant and anti-inflammatory activity. CRFO significantly inhibited edema by up to 62.60%, enhanced peritoneal macrophage phagocytosis capacity (p-value < 0.05), reduced granuloma formation (p-value < 0.05) and suppressed inflammatory exudates (p-value < 0.05). The CRFO doses of 0.243 and 0.813 mL/kg body weight provided anti-inflammatory effects comparable to the positive control. Moreover, these CRFO levels demonstrated good safety profiles without significantly affecting the rats’ physiological parameters. This study provides scientific evidence that CRFO has strong potential as a natural immunomodulatory and anti-inflammatory agent. These findings support the development of CRFO as an adjunctive therapy for managing inflammatory conditions and as a functional supplement to enhance immune system function. This discovery offers a safer, nature-based alternative to existing commercial supplements.
Keywords: Anti-inflammatory, Immunomodulator, Oil, Pandanus conoideus
Introduction
Immunomodulators and anti-inflammatory agents are essential components in the biomedical field, particularly in the development of safe, natural, and locally sourced alternative therapeutic agents. Immunomodulators function to regulate and maintain the balance of the body's immune responses against pathogenic onset, while anti-inflammatory agents suppress excessive inflammation occurrences that may
cause tissue damages. Both roles are crucial for disease prevention, accelerating recovery, and reducing the risk of chronic illnesses such as autoimmune, degenerative, and cardiovascular diseases [1]. In this regard, Indonesia's biodiversity, particularly in Papua, contains many natural resources with significant potential to be developed as raw materials for biopharmaceuticals and functional foods. One of such resources is the red fruit (Pandanus conoideus Lamk.), an endemic plant that the local community has long used either as food or traditional medicines.
Red fruit is well-known for its richness in bioactive compounds, particularly in its oil extract known as red fruit oil (RFO), which contains various nutraceutical components such as carotenoids (β-carotene, cryptoxanthin, phytoene, capsanthin, and violaxanthin) [2,3], tocopherols (α- and γ-tocopherol), and unsaturated fatty acids, mainly oleic and linoleic acids [4]. In addition, RFO contains considerable amounts of flavonoids and phenolic compounds [4,5], which contribute to its potent antioxidant properties. This antioxidant activity enables RFO to neutralize reactive oxygen species (ROS) and protect cells from oxidative damage, which is a significant trigger of inflammation and immune dysfunction. Several in vivo studies have demonstrated that RFO exhibits various biological activities, including anticancer effects through apoptosis induction [6], anti-inflammatory effects via inhibition of the NF-κB signaling pathway [7], and immunomodulatory potential through T-cell activation and enhancement of immune function [8]. Furthermore, RFO has been shown to reduce oxidative stress in endothelial cells by modulating nitric oxide production and scavenging ROS [9], and it also plays a role in regulating immune cells such as T lymphocytes, NK cells, and macrophages. By suppressing the expression of pro-inflammatory mediators such as TNF-α and IL-6 through NF-κB inhibition [10], RFO demonstrates intense anti-inflammatory actions. The combination of these bioactive compounds makes RFO a promising candidate for development of natural therapeutic agent with antioxidant, immunomodulatory, and anti-inflammatory activities.
Traditionally, Papuans extract RFO using a wet method, which involves boiling the drupes (fruit granules) in water for several hours, grinding them to separate the seeds from the pulp, and then reheating the pulp paste until oil appears on the surface, after which it is manually collected. However, this traditional method has several limitations, including prolonged heating that can degrade active compounds, low oil yield, and unstable chemical quality. As an alternative, Sarungallo et al. [11] developed a more modern dry extraction method consisting of three stages: Pressurized steaming, hydraulic pressing, and centrifugation to separate the oil from the pulp. This method has proven more efficient in improving both the chemical quality and yield of the oil compared to traditional and wet rendering extraction methods [11]. Furthermore, Sarungallo et al. [12] reported that heating red fruit drupes at 120 °C for 20 min (autoclaving) before hydraulic pressing produced the highest oil yield (11.5 ± 0.3%) with total carotenoid and tocopherol contents of 6,148 ± 65 ppm and 1,515 ± 24 ppm, respectively. The free fatty acid content (0.76 ± 0.1%) showed no significant difference (p-value < 0.05) compared to the unheated control (0.71 ± 0.2%). Therefore, the modern dry extraction method represents an improved approach for obtaining crude red fruit oil (CRFO) with superior bioactive quality and stability, making it suitable for further biological evaluation.
Although the chemical composition and antioxidant activity of red fruit oil have been widely reported, scientific evidence explicitly describing the potential of crude red fruit oil (CRFO) obtained through dry extraction as an immunomodulatory and anti-inflammatory agent remains limited. This study is the first in vivo investigation explicitly designed to evaluate the effects of CRFO on modulating immune and inflammatory responses. The objectives of this study were to investigate the potential of CRFO in modulating the immune system of Sprague-Dawley rats through three approaches: (1) to evaluate the effectiveness of CRFO in inhibiting carrageenan-induced acute inflammation by measuring paw edema, (2) to assess phagocytic capacity as an indicator of enhanced immune activity, and (3) to analyze the effect of CRFO on granuloma formation as a model of chronic inflammation. The findings of this study are expected to provide new insights into the mechanisms of CRFO in regulating immune and inflammatory responses and to support its potential application as a functional food ingredient and a complementary natural therapeutic candidate. This study is the first in vivo investigation explicitly designed to evaluate the effects of CRFO on modulating immune and inflammatory responses, i.e., a preliminary research which will be confirmed further on molecular mechanism in the next research program.
Materials and methods
Source and extraction of red fruit oil
Red fruit (with local name “Memeri”) was cultivated in SP 5 Kali Merah Village, Masni District, Manokwari, West Papua Province, Indonesia. The RFO extract was obtained using a method described by Sarungallo et al. [11], specifically a portable pressure steam sterilizer (Faithful, model FSF-18ML, China) at a temperature of 120 °C and 14.9 Psi, which was held for 20 min on manually chopped red fruit hordes. Then, the red fruit grains were detached from the hordes, wrapped in cheesecloth, and pressed using a hydraulic press (Jack hydraulic, 5 tons capacity, Indonesia). The resulting oil was then separated from water and residues by centrifugation (Hettich, EBA, Germany) with an operating speed of 2,000 rpm for 10 min to obtain CRFO. The CRFO produced was stored in a dark glass bottle in a refrigerator (temperature ranged 4 - 10 °C) until the time for conducting in vivo experiments, determination of total carotenoids [13], and tocopherol [14].
Experimental animals
The experiment employed thirty (30) Sprague Dawley (SD) white rats, aged 6 - 8 weeks, with body weights of 120 - 130 g, obtained from the iRatco laboratory in Bogor, Indonesia. The CRFO dose was determined based on the average daily consumption of the red fruits by Papuans, i.e., 15 mL (1 tablespoon per day), which was then converted to 0.081 mL/kg BW in rats (dose 1). Dose 2 and 3 were three and ten times folds, respectively. The rats were divided into six treatment groups, i.e., (I) normal control (Na-CMC suspension 0.5%), (II) negative control (phagocytosis + acute-chronic inflammation, and Na-CMC 0.5%), (III) positive control (phagocytosis + acute-chronic inflammation and immune booster 78.4 mg/day), (IV) Dose 1 (phagocytosis and CRFO 0.081 mL/kg BW), (V) Dose 2 (Phagocytosis + acute-chronic inflammation and CRFO 0.243 mL/kg BW), and (VI) Dose 3 (Phagocytosis and CRFO 0.813 mL/kg BW). Codes of CRFO dosages were BM 1 dosages (rats with CRFO dosage 1), BM 2 dosages (rats with CRFO dosage 2), and BM 3 dosages (rats with CRFO dosage 3) as listed in Table 1. The subjected rats were grouped through a random sampling technique.
Table 1 Rat groups treatments for CRFO in anti-inflammatory and immunomodulatory experiments.
Rat’s Groups |
Treatments |
Acute inflammation (1% Carrageenan intraplantar on day 7) |
Chronic inflammation (cotton pouch granuloma on day 8) |
Fagocytosis (peritoneal 10% indian ink on day 15) |
Normal Control |
14 days Na-CMC 0.5% |
- |
- |
- |
Negative Control |
14 days Na-CMC 0.5% |
+ |
+ + + + + |
+ + + + + |
Positive Control |
14 days Imboost 78.4 mg/day |
+ |
||
CRFO Dose 1 |
14 days CRFO 0.081 mL/kg BW |
+ |
||
CRFO Dose 2 |
14 days CRFO 0.243 mL/kg BW |
+ |
||
CRFO Dose 3 |
14 days CRFO 0.813 mL/kg BW |
+ |
*CRFO: Crude red fruit oil; BW = body weights.
The acclimatization was done for 7 days in a room with a temperature of 25 ± 2 °C at Iratco Veterinary Laboratory Services, Bogor, Indonesia. They were given standardized foods and room temperature drinking water that had been boiled. This was aimed at helping the rats adapt to their new environment to comply with the proper requisites of an in vivo experiment. During the acclimatization, the rat’s weights were monitored periodically once a week.
The intragastric commercial Imboost (PT Soho Industri Pharmacy, Bandung, Indonesia) or CRFO dosages were given once a day for 14 days (H1-H14). The acute inflammation was induced by intraplanar injection of 0,1 mL carrageenan 1% (Sigma Aldrich, St. Louis, MO, USA) suspended in 0.5% Na-CMC solution at day 7, and the chronic inflammation was induced using a Cotton Pouch granuloma method at day 8 for observation at day 15. The immunomodulator capacity of the CRFO was carried out by aseptically inducing the rats with a 10% Indian ink through an intraperitoneal route at day 15. The rats were sacrificed after phagocytosis tests. The in vivo experiments fulfilled the ethical code of Helsinki (Ethical approval letter no. 4.2.003/KEHI/II/2021) by Animal Ethics Committee of iRATco Veterinary Laboratory Services, Bogor Jawa Barat, Indonesia.
Paw oedema (acute inflammation) test
Paw oedema was used for an acute anti-inflammation test, which was carried out by measuring the degree of swelling of the rats’ paws (feet) and its reduction or its inhibition using a plethysmometer. The paw swelling or its reduction/inhibition indicated anti-inflammation activity of the treatments. All groups obtained 1% carrageenan solution intraplantar (0.1 mL) on day 7. The swelling volumes were measured after 1, 2, 3, 4, 5, and 24 h after injections. Data were expressed as a percentage of inhibition of edema compared to the normal control group, calculated according to Eq. (1) [15].

Immunomodulator test
The experiments were carried out by intraperitoneal induced using one drop of 10% Indian ink, and then the intraperitoneal liquid was collected, swabbed onto an object glass, and stained with Giemsa for observation of actively and non-actively macrophages acting as phagocytosis towards foreign materials entering the body (indian ink). The observation of peritoneal macrophages was carried out by injection of 5mL of saline, and then it was taken back. The blood was centrifuged (Tomy Portable Refrigerated Centrifuge MX-201) at a speed of 3,000 rpm for 5 min, the supernatant was removed and then added with another 100 µL of saline. The amounts of blood cells were microscopically counted using a hemocytometer glass at a sample volume of 100 µL. Another 100 µL was used for methylene blue staining to determine the number of macrophages that phagocytized the Indian ink. The amount of phagocytosis of macrophages frequently represents immunological parameters that can be used to evaluate the health status/function of the immune system. Justification of phagocytosis capacity or activity can be determined as phagocytosis capacity [16], as shown in Eq. (2).
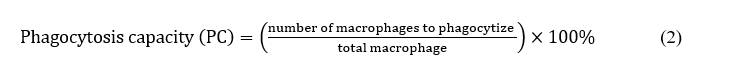
Cotton pellet-induced granuloma test
Chronic inflammation was induced with a cotton pouch that had the same weights and sizes on day 8, and it was left until day 15. A sterile cotton (autoclaved) was implanted subcutaneously into the rats. The observations of granuloma formations were taken after 7 days of implant. The rats were sacrificed after phagocytosis observation, and the size, thickness, area, and weight of granulomas (macromorphology) on their skins were measured; stored in 10% buffered neutral formalin for histopathological observation using Hematoxylin-Eosin (H&E) staining. Tissue observation using a microscope to examine the density of inflammatory cells (lymphocytes, macrophages), and fibroblast proliferation, using Image software version 1.5.
Exudate and granuloma formation were determined by measuring the weight of the cotton after implantation. The cotton was weighed wet after removal, then dried at 60 °C for 24 h to a constant weight, and the net dry weight, that is, after subtracting the weight of the cotton pellets, was calculated.
Measure of exudate formation = Immediate wet weight of pellet – Constant dry weight of pellet.
Measure of granuloma tissue formation = Constant dry weight – Initial weight of cotton pellet.
Furthermore, the percentage of inhibition of the formation of exudate and granuloma was determined using Eqs. (3) - (4) [17].


Data analysis
The quantitative data of anti-inflammatory activity were statistically analyzed using analysis of variance (ANOVA) at the confidence level of 95% (p-value < 0.05). When it was detected to have significant effects of the treatments, then it was continued for Duncan’s Multiple Range Test (DMRT) to compare among different treatment groups. The statistical analysis was performed by using SPSS software version 16.0.
Results and discussion
Carotenoid and tocopherol content of CRFO
The total carotenoid and tocopherol contents of crude red fruit oil (CRFO) obtained in this study were 8,135 ± 152 mg/kg and 15,482±122 mg/kg, respectively (Table 2). These concentrations are significantly higher than those reported by Sarungallo et al. [18], who found 6,784 ± 206 mg/kg of carotenoids and 1,765 ± 57 mg/kg of tocopherols using a wet extraction method. The concentration of bioactive compounds in crude red fruit oil (CRFO) is significantly influenced by the extraction method used. The dry extraction technique applied in this study is considered more effective in preserving heat-sensitive compounds, such as carotenoids and tocopherols, by minimizing thermal degradation and oxidative loss during the isolation process. Red fruit extract (Memeri clone) obtained using a chloroform-methanol (2:1) solvent system, has been reported to contain α-cryptoxanthin (10.38 ± 1.2 mg/kg), β-cryptoxanthin (9.8 ± 0.9 mg/kg), α-carotene (10.5 ± 1.6 mg/kg), and β-carotene (55.8 ± 2.7 mg/kg) [3]. It also contains significant levels of α-tocopherol (272 ± 27 mg/kg) and γ-tocopherol (67 ± 3.2 mg/kg) [4]. In addition, CRFO includes other carotenoids such as phytoene, capsanthin, and violaxanthin [2], which contribute synergistically to its antioxidant potential. These compounds play a critical role in neutralizing reactive oxygen species (ROS) and prevent oxidative cellular damage, thereby reducing the risk of oxidative stress-related diseases such as cardiovascular and neurodegenerative disorders [19].
Table 2 Content of total carotenoid and tocopherol of crude red fruit (Pandanus conoideus Lamk.) oil.
Sample |
Total carotenoid (mg/kg) (Meanstandard deviation) |
Total tocopherol (mg/kg) (Meanstandard deviation) |
CFRO |
8,135 152 |
15,482 122 |
Carotenoids and tocopherols in crude red fruit oil (CRFO) act synergistically as antioxidants, anticarcinogens, and radioprotective agents. Their high concentrations in CRFO contribute to its potential in preventing degenerative diseases, including colorectal and breast cancers [20]. β-carotene functions as a potent antioxidant and modulates immune responses by enhancing T-cell activity and inducing apoptosis via the Bcl-2/Bax pathway [21]. α-Tocopherol plays a crucial role in cardiovascular protection by neutralizing nitrogen oxides and inhibiting LDL oxidation, a key factor in preventing atherosclerosis [22]. These properties position CRFO as a promising functional food and complementary therapy for managing oxidative stress-related conditions.
Effect of CRFO treatment on body weight change of rats
During the research period, all treatment groups exhibited consistent weight gain, with no significant differences observed. Minor variations appeared from days 8 to 15, as shown in Figure 1. Initial weight gains reflected the study’s preventive design, with CRFO administered before inflammation induction, indicating baseline stability. After inflammation induction on day eight, minor weight gain differences emerged, potentially due to metabolic influences. However, ANOVA analysis (α = 0.01) showed no significant differences in body weight between treatment groups (p-value > 0.01), suggesting that CRFO did not negatively affect rat growth, which supports its safety for long-term use. Consistent with this, Fitri and Pudjirahayu [23] reported no statistical significance of weight gain in RFO-supplemented rats on an atherogenic diet. These findings indicate that CRFO, across varying doses, does not negatively impact weight gain, underscoring its potential as a safe therapeutic agent for chronic inflammatory conditions without disrupting normal growth.
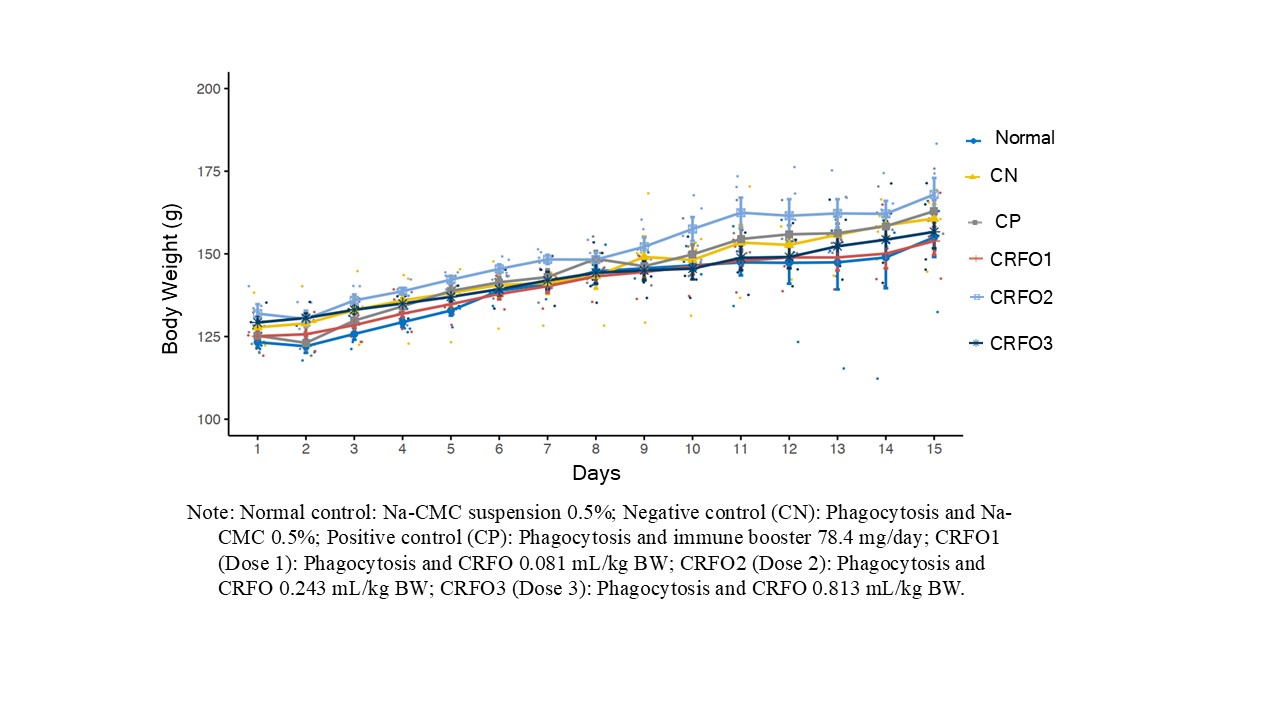
Figure 1 Changes in body weight of rats during anti-inflammatory test of crude red fruit (Pandanus conoideus Lamk.) oil (CRFO) during 15 days.
Effects of CRFO treatment on carrageenan-induced rat paw edema
The paw oedema test assessed CRFO's acute anti-inflammatory effect by measuring carrageenan-induced swelling in mice using a plethysmometer. The test used Imboost (78.4 mg) as a positive control, a supplement containing Echinacea purpurea and zinc picolinate, which is known for its anti-inflammatory effects [23]. Paw oedema was assessed at hourly intervals over a 24-hour period following intraplantar injection of carrageenan. As shown in Figure 2(a), the administration of CRFO at doses of 0.081, 0.243, and 0.813 mL/kg BW resulted in a dose-dependent reduction in paw swelling. Generally, all doses of CRFO treatments were lower than the positive control treatment. These findings suggest that CRFO mitigates acute inflammation, possibly through the downregulation of proinflammatory mediators such as prostaglandins and cytokines (e.g., TNF-α, IL-6), potentially via inhibition of the NF-κB signaling pathway. The comparable efficacy of CRFO to a well-established supplement like Imboost highlights its potential as a natural anti-inflammatory agent in therapeutic applications.
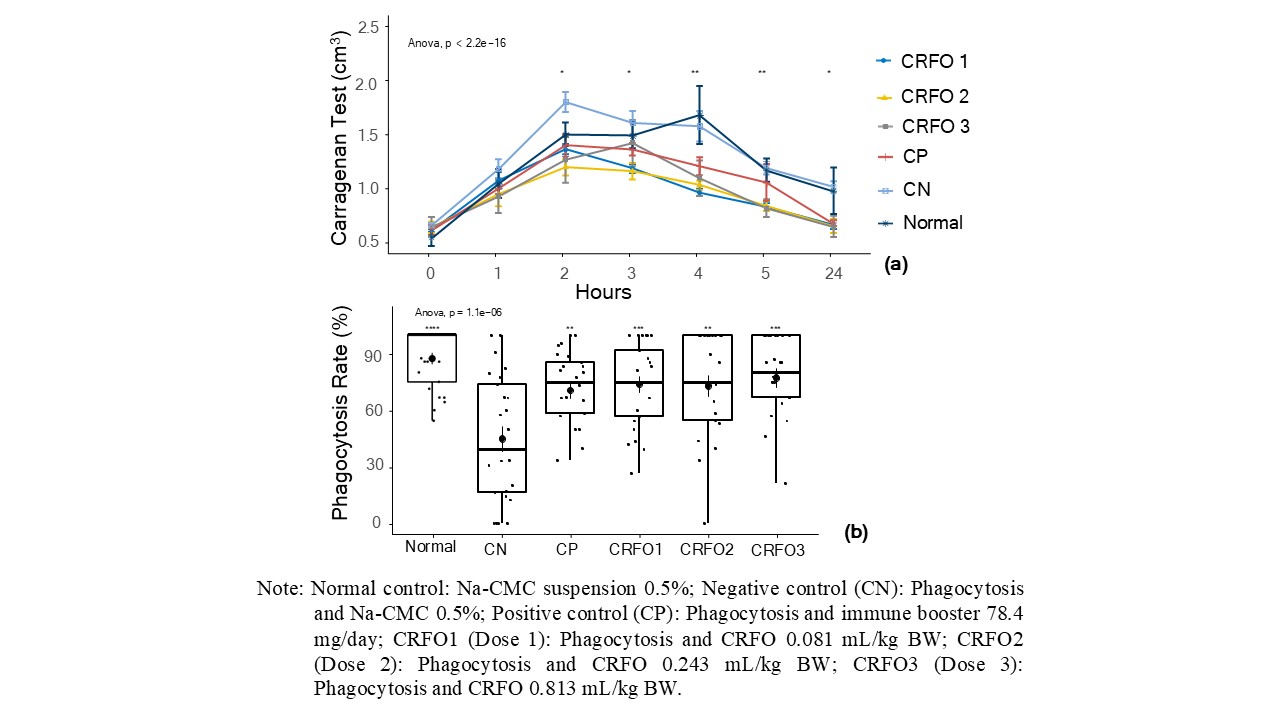
Figure 2 Changes in rat paw swelling after carrageenan induction, as observed over 15 h in an anti-inflammatory test of crude red fruit (Pandanus conoideus Lamk.) oil (CRFO) (a); Rates of macrophage phagocytosis obtained during testing of the anti-inflammatory effects of CRFO in rats (b).
Figure 2(a) shows a significant difference (p-value < 0.05) in paw oedema between the negative control and treatment groups (Imboost and CRFO). The negative control group had greater swelling due to the lack of anti-inflammatory treatment after carrageenan induction. Oedema began decreasing from the third hour in all treatment groups, with the most notable reduction in the Imboost and CRFO groups, continuing through the 24th hour. These results indicate that both Imboost and CRFO effectively and sustainably suppressed inflammation. Karim et al. [24] stated that the carrageenan-induced paw oedema model, featuring a biphasic inflammatory response, is commonly used to evaluate natural anti-inflammatory agents. The early phase involves histamine and serotonin, whereas the late phase is driven by cytokines such as TNF-α and IL-6. As shown in Figure 2(a), inflammation peaks in the second hour, then significantly decreases in both Imboost- and CRFO-treated groups. This indicates that CRFO may reduce inflammation by inhibiting proinflammatory mediators, similar to Imboost, which contain Echinacea purpurea and zinc picolinate. Prior to investigation on immune biomarkers, which is the limitation of this study, the current study plays an important step to set a further in depth research in the near future using cell culture epigenetic experimental base studies comprehensively while other related functional physiological investigation on osteoporosis effects is ongoing in our laboratory.
The percentage of inflammation inhibition was determined by comparing paw volume differences between treated and negative control groups after carrageenan-induced inflammation. Table 3 shows that CRFO at the highest dose 3 (0.813 mL/kg BW) inhibited inflammation by 62.60%, comparable to the positive control (Imboost) with 60.0% inhibition. ANOVA analysis indicated a significant reduction (p-value < 0.05) in paw oedema in CRFO-treated groups compared to the negative control 22.90%. These results suggest CRFO effectively reduces inflammation, presumably follows the well-known mechanisms, potentially by modulating proinflammatory mediators and inhibiting the NF-κB pathway, indicating its potential as a natural anti-inflammatory agent. These anti-inflammatory activities are therefore considered to be generated by CRFO bioactive compounds such as carotenoids, tocopherols, and flavonoids as general knowledge currently obtained from literatures. Several studies have shown that these compounds can inhibit the activation of the NF-κB pathway, a key regulator in the inflammatory response, by suppressing the expression of pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6 which also actively involved in other functional physiological biomarkers including osteoporosis as a holistic healthy state nowadays. Hence, the inhibition of paw inflammation observed in the CRFO treatment group indicates a potential mechanism involving a reduction in NF-κB signaling, consistent with the natural antioxidant effects reported in previous in vivo studies [8,25].
Table 3 also shows that CRFO administration at doses 1 (0.081 mL/kg BW), dose 2 (0.243 mL/kg BW), and dose 3 (0.813 mL/kg BW) effectively inhibited paw swelling in carrageenan-induced rats, with dose 3 showing the highest inhibition (62.6%), comparable to dose 2 (62.19%). A substance is considered anti-inflammatory if it achieves 50% or greater inhibition in carrageenan-induced inflammation models. Pakale et al. [26] reported that the aqueous and methanolic extracts of fresh ginger rhizomes demonstrated significant anti-inflammatory effects, with inhibition rates of 62.03 - 65.7% and 73.14% at doses of 100 mg/kg and 200 mg/kg, respectively, showing their potential as natural therapeutic agents comparable to Aspirin, which showed 85.5% inhibition at a 3-hour interval. While CRFO did not surpass the standard anti-inflammatory drug, its performance as a natural product is noteworthy.
Table 3 Increased swelling and percentage of inflammation inhibition were obtained during experiments testing the anti-inflammatory effects of crude red fruit (Pandanus conoideus Lamk.) oil (CRFO) in rats.
Treatment Groups* |
Increased swelling of the rat's paws (mL)** |
Inhibition of Inflammation (%) |
Control Normal |
0.150 ± 0.60ab |
- |
Control Negative |
0.161 ± 0.32a |
22.90 |
Control Positive |
0.136 ±0.18b |
60.00 |
CRFO Dose 1 |
0.119 ±0.08c |
61.03 |
CRFO Dose 2 |
0.116 ±0.09c |
62.19 |
CRFO Dose 3 |
0.113 ± 0.37c |
62.60 |
*: Normal control: Na-CMC suspension 0.5%; Negative control: Phagocytosis and Na-CMC 0.5%; Positive control: Phagocytosis and immune booster 78.4 mg/day; Dose 1: Phagocytosis and CRFO 0.081 mL/kg BW; Dose 2: Phagocytosis and CRFO 0.243 mL/kg BW; Dose 3: Phagocytosis and CRFO 0.813 mL/kg BW.
**: Different letters in the same column indicate significant differences (p-value < 0.05).
Effects of CRFO treatment on characteristics of peritoneal macrophages
Peritoneal macrophages are key to immune defense, clearing infections and foreign bodies through phagocytosis as part of the mononuclear phagocyte system alongside monocytes and lymphocytes [27]. To assess their activity, an Indian ink assay was conducted by injecting ink into the peritoneal cavity and then staining the collected cells with methylene blue. Active macrophages appear dark from ink uptake, allowing calculation of the phagocytic index, a key marker of immune function and CRFO’s potential anti-inflammatory effects.
Administration of CRFO (at doses of 0.081, 0.243, and 0.813 mL/kg BW) significantly increased the phagocytic capacity of peritoneal mononuclear macrophages compared to the negative control (p-value < 0.01), as demonstrated in Figure 2(b). The highest dose of CRFO (0.813 mL/kg BW) enhanced the phagocytic rate up to 80%, comparable to the positive control group Imboost (75%) and substantially higher than the negative control (40%). The α-tocopherol and carotenoid content in CRFO acts as an antioxidant, neutralizing ROS and promoting macrophage phagocytosis. The anti-inflammatory and immunomodulatory effects of CRFO are likely attributed to its bioactive compounds (tocopherols, carotenoids, phenolic compounds, and unsaturated fatty acids) that modulate immune signaling pathways such as NF-κB and MAPK [3,4,8] generally understood. The comparable efficacy of CRFO to Imboost suggests its potential as a natural alternative for enhancing immune function and managing inflammatory conditions.
Effects of CRFO treatment on histopathological characteristics
Macroscopic granuloma observations were performed using the cotton pouch method, a well-established model for evaluating chronic inflammation and tissue response to persistent irritation. This method provides valuable insights into the body’s response to stimuli and enhances understanding of chronic inflammation, including the structure of granuloma and tissue composition. The staining results of chronic inflammation granulomas in CRFO-treated rats are shown in Figure 3(a), while the morphological assessment of granuloma area is presented in Figure 3(b) as a preliminary observation prior to set a more comprehensive functionality of physiological biomarkers orchestra.
Figure 3(a) illustrates changes in the inflammatory marker hyperaemia across treatment groups. The negative control showed pronounced hyperaemia, while the positive control (Imboost) and CRFO-treated groups exhibited a marked reduction, indicating the qualitative CRFO’s anti-inflammatory effects. These histopathological findings suggest CRFO may exert its anti-inflammatory effects by modulating the immune response, potentially by inhibiting proinflammatory cytokines (TNF-α, IL-6) and suppressing capillary permeability, which paves a building block for further research how the networking of general biomarkes orchestrated in regulating health biomarkers. Arguably, the dose-dependent reduction in granuloma size and thickness further supports CRFO's potential as a natural anti-inflammatory agent for managing chronic inflammatory conditions.
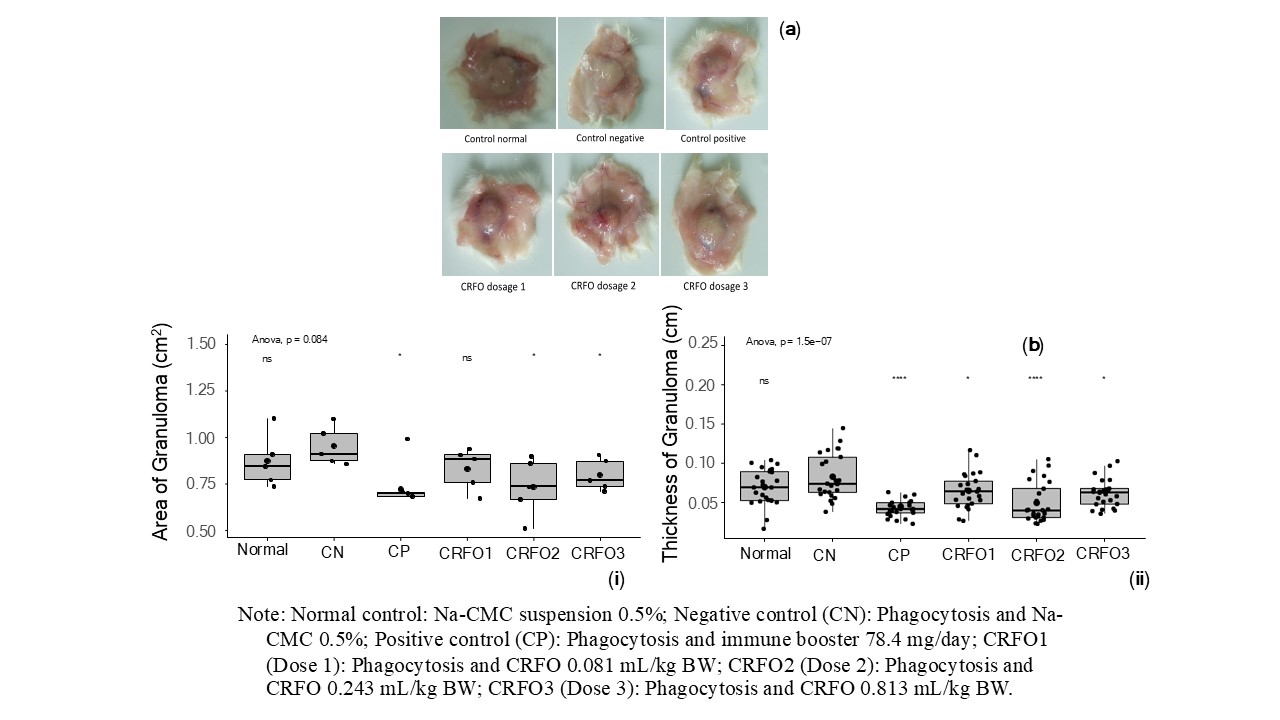
Figure 3 Macroscopic changes (hyperemia) in granulomas during anti-inflammatory testing of crude red fruit (Pandanus conoideus Lamk.) oil (CRFO) in rats (a); Quantitative area of granulomas (i) and thickness of granuloma (ii) obtained during experiments testing of the anti-inflammatory effects of CRFO in rats (b).
Statistical analysis of the granuloma area (Figures 3(b) - 3(i)) revealed no significant difference (p-value > 0.05) between the normal (0.87 ± 0.14 cm2) and negative control groups (0.95 ± 0.10 cm2). Nevertheless, the inflammation state definitely signals that epigenetically unhealthy body. It can be seen that Imboost and CRFO at dose 2 (0.73 ± 0.16 cm2) and dose 3 (0.80 ± 0.09 cm2) has a capability of qualitatively controlling (p-value < 0.05) the granuloma expanding area compared to the negative control. Therefore, dose 2 of CRFO was the best treatment for controlling the granuloma expansion. This anti-inflammatory effect is likely mediated by CRFO’s bioactive components (carotenoids and tocopherols), which may modulate immune responses by enhancing macrophage activity, promoting phagocytosis, and inhibiting proinflammatory cytokines (TNF-α and IL-6) based on the current understanding. The comparable efficacy to Imboost (positive control with known mechanism) highlights CRFO’s potential as a natural therapeutic agent for enhancing immune function and managing chronic inflammatory conditions through similar molecular pathways such as NF-κB and MAPK signaling.
Figures 3(b) - 3(i) shows that Imboost exhibited the lowest granuloma thickness (0.043 ± 0.015 cm), which was insignificantly different (p-value > 0.05) from CRFO at doses 2 (0.049 ± 0.025 cm) and dose 3 (0.064 ± 0.024 cm). However, all treatment groups showed significant (p-value < 0.05) reductions in granuloma thickness compared to the negative control (0.082 ± 0.029 cm). CRFO’s anti-inflammatory effect is likely due to its high antioxidant capacity from carotenoids and tocopherols [5], by which their current understandings can modulate immune responses, inhibit fibroblast activation, reduce collagen deposition, and suppress proinflammatory cytokines. The comparable efficacy of CRFO to Imboost obviously show the possible similarity of its potential as a natural therapeutic agent for managing chronic inflammation by reducing granuloma thickness and promoting tissue homeostasis. Further research is needed to understand the specific molecular mechanisms and clinical applications of CRFO in chronic inflammatory disorders.
Effects of CRFO treatment on microscopic observation of granuloma
Granulomas, which are aggregates of immune cells formed in response to persistent stimuli, were microscopically observed after cotton pouch implantation by quantifying the number of macrophages, lymphocytes, and fibroblasts to evaluate the immune response and inflammatory process. Cellular histopathology assesses the microscopic architecture of granulomas, including chronic inflammation-associated cells. Figure 4 shows the histopathological observation of granuloma cells using Hematoxylin-Eosin (H&E) staining, where the tissue from the normal control group exhibited healthy morphology, with immune cells (lymphocytes) and fibroblasts present at normal levels. In contrast, the negative control group showed disrupted tissue structure and an increased number of inflammatory cells, indicating an active inflammatory response. The positive control group displayed denser tissue structure, suggesting a more controlled inflammatory response or ongoing tissue repair process. The CRFO treatment groups (Doses 1, 2, and 3) exhibited varying cellular responses, particularly in the number and distribution of lymphocytes, macrophages, and fibroblasts, indicating dose-dependent biological effects. Dose 1 showed relatively high signs of inflammation, although still better than the negative control. Dose 2 appeared to be the most effective, showing more organized tissue structure and reduced inflammation, while Dose 3 remained effective but slightly less optimal than Dose 2, possibly due to a saturation effect.
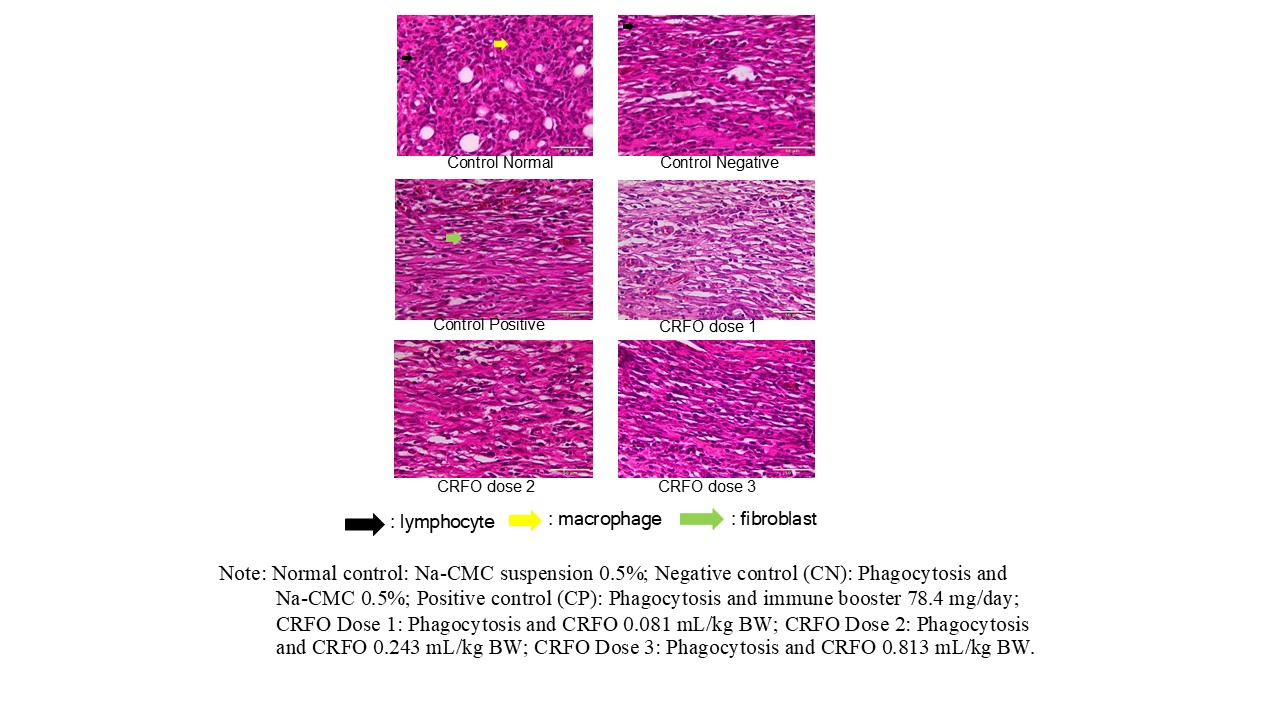
Figure 4 Cellular histopathological features of granuloma tissue in testing the anti-inflammatory effect of crude red fruit (Pandanus conoideus Lamk.) oil (CRFO) in rat.
The significant numbers of macrophages, lymphocytes, and fibroblasts formed because of CRFO treatment (doses 1, 2, and 3) and the positive control (Imboost) is presented in Figure 5. The number of macrophages and lymphocytes showed a significant decrease compared to the negative control, as shown in Figures 5(a) and 5(b). The greatest reduction in the number of macrophages was observed with CRFO dose 2 (172.44 ± 36.99), followed by dose 1 (195 ± 39.22). Meanwhile, dose 3 (208.28 ± 37.02) showed a similar effect to the positive control (207.28 ± 38.81). This decrease in the number of macrophages indicates the potential immunomodulatory activity of CRFO in regulating inflammatory responses, as macrophages are the main phagocytic cells involved in the early phase of inflammation. The greatest reduction in the number of lymphocytes was also found with CRFO dose 2 (101.76 ± 21.58), followed by dose 3 (109.32 ± 20.85). The effects of CRFO dose 1 and the positive control (Imboost) were relatively similar in reducing lymphocyte levels. The lymphocytes are key components of adaptive immunity, and a decrease in their number may indicate suppression of excessive immune activity, which is beneficial in managing chronic inflammatory conditions.
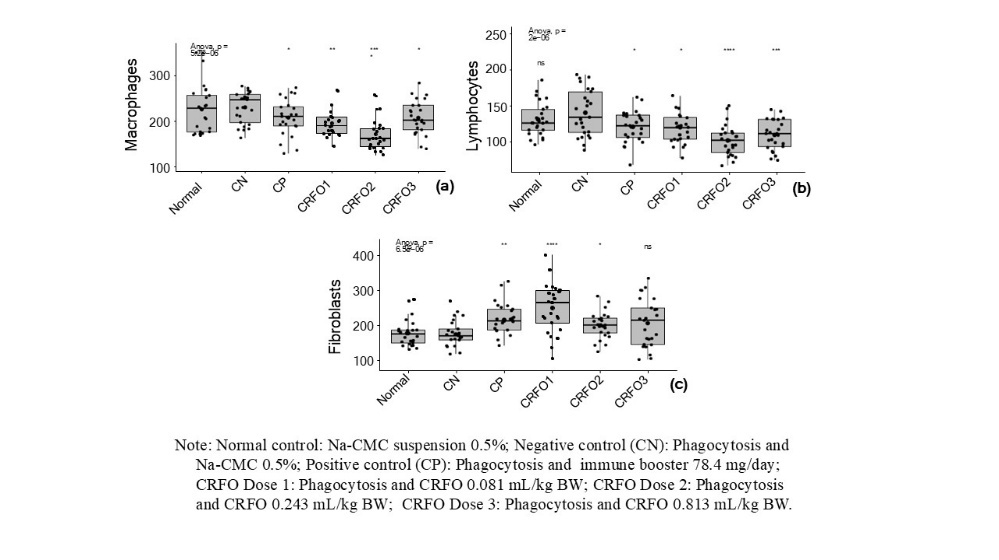
Figure 5 The number of macrophages (a), the number of lymphocytes (b), and the number of fibroblasts (c) in the tissue around the granuloma obtained during testing of the anti-inflammatory effects of crude red fruit (Pandanus conoideus Lamk.) oil (CRFO) in rats.
Meanwhile, all CRFO and Imboost treatment increased the number of fibroblasts compared to the negative control (Figure 5(c)). The highest increase was observed with CRFO dose 1 (250.44 ± 70.54), while doses 2 and 3 showed results comparable to the positive control. Fibroblasts play an essential role in wound healing through the production of collagen and other extracellular matrix components. These findings indicate that CRFO not only has immunomodulatory properties but also promotes tissue regeneration. Notably, CRFO dose 2 demonstrates a balanced effect by suppressing inflammation while supporting healing process.
It is consistently observed in the present study that bioactive components such as carotenoids and tocopherols in CRFO phenomenally influence the NF-κB and TGF-β signaling pathways, leading to reduced cell infiltration and fibrosis. Rhee et al. [8] demonstrated in LPS-stimulated murine RAW 264.7 macrophages that CRFO, containing 14.05 ± 0.8 mg/g of β-cryptoxanthin and 7.4 ± 0.7 mg/g of β-carotene, significantly reduced NO, cyclooxygenase-2, prostaglandin E2 production, and related protein and mRNA levels without affecting cell viability. CRFO also regulated ROS and inhibited NF-κB p65 subunit translocation, I-κB phosphorylation, and p38, ERK, and JNK activation dose-dependently [8]. These findings suggest CRFO’s anti-inflammatory effects are mediated by regulating ROS and the NF-κB pathway via MAPK, likely due to its antioxidant components [2-8]. Furthermore, Lin et al. [28] noted that carotenoids in CRFO can enhance immune responses by increasing INF-γ and IL-2 production without cytotoxicity, with INF-γ activating macrophages and stimulating other immune cells. The dual anti-inflammatory and immunomodulatory properties of CRFO reveal its potential as a natural therapeutic agent for chronic inflammation and immune modulation through specific molecular mechanisms.
Inflammation also involves fluid exudation, peaking within 24 h [29]. Figure 6(a) shows that exudate inhibition was highest in the CRFO dose 2 group (0.243 mL/kg BW), with an inhibition rate of 60.64%, which was insignificantly different from the positive control (51.67%) but was substantially higher (p-value < 0.01) than the negative control (29.34%). Similarly, granuloma inhibition (Figure 6(b)) was most effective with CRFO dose 2 (73.42%), comparable to dose 3 (65.18%) and Imboost (67.67%), and significantly higher (p-value < 0.01) than the negative control (48.86%). These results indicate that CRFO has a strong ability to inhibit exudate and granuloma formation, with dose 2 showing superior performance. This anti-inflammatory effect is likely due to its rich content of bioactive compounds, including carotenoids, tocopherols, and various phytochemicals [3,4]. Flavonoids in CRFO can also regulate the expression of proinflammatory genes (NF-κB, AP-1, ICAM, VCAM, and E-selectin) and inhibit proinflammatory enzymes such as iNOS, COX-2, and LOX [25]. These findings highlight CRFO’s potential as a natural anti-inflammatory agent for managing chronic inflammation by modulating immune responses and reducing tissue damage.
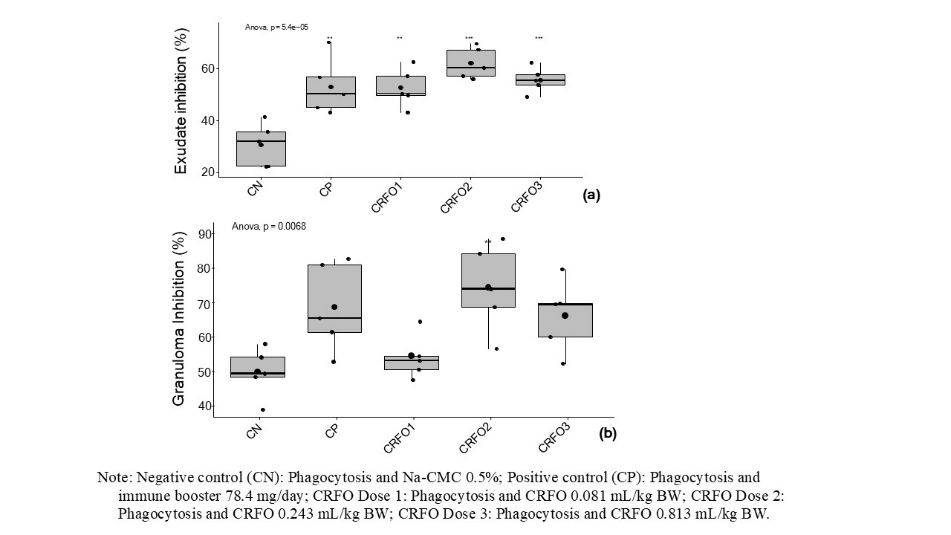
Figure 6 Percentage of exudate inhibition (a), and granuloma inhibition (b) obtained during testing of the anti-inflammatory effects of crude red fruit (Pandanus conoideus Lamk.) oil (CRFO) in rats.
Conclusions
Based on the results of this study, the in vivo administration of CRFO in Sprague-Dawley (SD) rats demonstrated significant anti-inflammatory and immunomodulatory effects referring to its phenomenal observed comparable to Imboost similarity. The CRFO effectively inhibited inflammation-induced oedema by up to 62.6%, enhanced the phagocytic capacity of peritoneal mononuclear macrophages in clearing carbon particles, and reduced chronic inflammation by decreasing macrophage and lymphocyte infiltration in granulomatous tissues. Additionally, CRFO treatment significantly inhibited exudate formation at doses 1, 2, and 3 by 51.30%, 60.64%, and 54.26%, respectively, compared to the negative control (29.34%). Granuloma inhibition was also notable, with doses 1, 2, and 3 showing inhibition rates of 53.45%, 73.42%, and 65.18%, demonstrating comparably highest efficacy, markedly prominent at dose 2. These findings reveal that CRFO is a promising and safe natural therapeutic agent for boosting the immune system and managing chronic inflammation. More importantly, no adverse effects were observed in CRFO-treated rats, indicating a favorable safety profile. Further studies, including clinical trials, are needed to elaborate further deeply these results and explore the clinical potential of CRFO as a viable alternative to conventional immunomodulatory and anti-inflammatory treatments.
Acknowledgements
The authors would like to thank the Directorate of Research and Community Service, Directorate General of Higher Education, Ministry of Education, Culture, Research, and Technology of the Republic of Indonesia for the funding provided through the National Strategic Research Grant, as well as Iratco Laboratory for facilitating the implementation of this research.
Declaration of Generative AI in Scientific Writing
In this paper, the authors acknowledge the use of a generative AI tool (ChatGPT) for language editing purposes. However, the AI was not used in the creation of content or the interpretation of data. Consequently, the authors bear full responsibility for the content of this paper.
CRediT Author Statement
Zita Letviany Sarungallo: Conceptualization, Methodology, Writing - original draft, Writing review & editing, Project administration. Budi Santoso: Methodology, Formal analysis, Writing - original draft, Writing review & editing, Visualization. Mathelda Kurniaty Roreng: Validation, Visualization, Writing - original draft. Indah Epriliati: Validation, Formal analysis, Writing review & editing, Project administration. Mardiah: Conceptualization, Methodology, Validation, Formal analysis, Writing - original draft, Visualization, Project administration.
References
[1] W Feleszko, GA Rossi, R Krenke, GW Canonica, LV Gerven and O Kalyuzhin. Immunoactive preparations and regulatory responses in the respiratory tract: Potential for clinical application in chronic inflammatory airway diseases. Expert Review of Respiratory Medicine 2020; 14(6), 603-619.
[2] E Fitria, N Wulandari, P Hariyadi and H Wijaya. Identification and fractionation of carotenoids in red fruit oil (Pandanus conoideus) (in Indonesian). Warta IHP 2020; 37(1), 7-19.
[3] ZL Sarungallo, P Hariyadi, N Andarwulan and EH Purnomo. Analysis of α-cryptoxanthin, β-cryptoxanthin, α-carotene, and β-carotene of Pandanus conoideus oil by high-performance liquid chromatography (HPLC). Procedia Food Science 2015; 3, 231-243.
[4] ZL Sarungallo, P Hariyadi, N Andarwulan and EH Purnomo. Characterization of chemical properties, lipid profile, total phenol and tocopherol content of oils extracted from nine clones of red fruit (Pandanus conoideus). Agriculture and Natural Resources 2015; 49(2), 237-250.
[5] A Rohman, S Riyanto, N Yuniarti, WR Saputra, R Utami and W Mulatsih. Antioxidant activity, total phenolic, total flavanoid of extracts and fractions of red fruit (Pandanus conoideus Lam). International Food Research Journal 2010; 17, 97-106.
[6] Achadiyani, L Septiani, A Faried, HMS Bolly and D Kurnia. Role of the red fruit (Pandanus conoideus Lamk) ethyl acetate fraction on the induction of apoptosis vs. downregulation of survival signaling pathways in cervical cancer cells. Europe Journal of Medicinals Plants 2016; 13(2), 1-9.
[7] T Tambaip, MB Karo, M Hatta, R Dwiyanti, R Natzir, MN Massi, AA Islam and K Djawad. Immunomodulatory effect of orally red fruit (Pandanus conoideus) extract on the expression of CC chemokine receptor 5-mRNA in HIV patients with antiretroviral therapy. Journal of Immunology Research 2018; 11(1), 15-21.
[8] YH Rhee, YK Park and JS Kim. Pandanus conoideus Lamk oil protects against inflammation through regulating reactive oxygen species in LPS-induced murine macrophages. Natural Product Communications 2020; 15(9), 1-8.
[9] N Xia, C Schirra, S Hasselwander, U Förstermann and H Li. Red fruit (Pandanus conoideus Lam) oil stimulates nitric oxide production and reduces oxidative stress in endothelial cells. Journal of Functional Foods 2018; 51, 65-74.
[10] P Annamalai and EB Thangam. Local and systemic profiles of inflammatory cytokines in carrageenan-induced paw inflammation in rats. Immunological Investigations 2016; 46(3), 274-283.
[11] ZL Sarungallo, P Hariyadi, N Andarwulan and EH Purnomo. The effect of extraction method on the chemical quality and fatty acid composition of red fruit (Pandanus conoideus) oil (in Indonesian). Journal of Agroindustry Technology 2014; 24(3), 209-217.
[12] ZL Sarungallo, P Hariyadi, N Andarwulan and EH Purnomo. Effect of heat treatment prior to extraction on the yield and quality of red fruit (Pandanus conoideus) oil. Food Research 2020; 4(3), 659-665.
[13] G Knockaert, L Lemmens, S Van-Buggenhout, M Hendrickx and A Van-Loey. Changes in β-carotene bioaccessibility and concentration during processing of carrot puree. Food Chemistry 2012; 133(1), 60-67.
[14] ML Wong, RE Timms and EM Goh. Colorimetric determination of total tocopherols in palm olein and stearin. Journal of the American Oil Chemists’ Society 1988; 65(2), 258-261.
[15] S Sarkhel. Evaluation of the anti-inflammatory activities of Quillaja saponaria Mol. saponin extract in mice. Toxicology Report 2015; 3, 1-3.
[16] A Fountain, M Mansat, T Lackraj, MC Gimenez, S Moussaoui, M Ezzo, S Soffiaturo, E Urdaneta, MB Verdawala, K Fung, C Lancaster, E Somerville, B Hinz, MR Terebiznik and RJ Botelho. Depletion of endomembrane reservoirs drives phagocytic appetite exhaustion in macrophages. Journal of Cell Science 2025; 138(16), 263539.
[17] H Tazeze, S Mequanente, D Nigussie, B Legesse, E Makonnen and T Mengie. Investigation of wound healing and anti-inflammatory activities of leaf gel of Aloe trigonantha L.C. leach in rats. Journal of Inflammation Research 2021; 14, 5567-5580.
[18] ZL Sarungallo, B Santoso, RU Situngkir, MK Roreng and MM Lisangan. Determination of chemical properties, composition of fatty acid, carotenoids and tocopherols of degummed and neutralized red fruit (Pandanus conoideus) oil. Jurnal Teknologi 2020; 82(6), 71-78.
[19] M Rodriguez-Concepcion, J Avalos, ML Bonet, A Boronat, L Gomez-Gomez and D Hornero- Mendez, MC Limon, AJ Meléndez-Martínez, B Olmedilla-Alonso, A Palou, J Ribot, MJ Rodrigo, L Zacarias and C Zhu. A global perspective on carotenoids: Metabolism, biotechnology, and benefits for nutrition and health. Progress in Lipid Research 2018; 70, 62-93.
[20] O Merhan. The biochemistry and antioxidant properties of carotenoids. In: J Dragan and GS Nikolic (Eds.). Carotenoids. IntechOpen, London, 2017, p. 51-66.
[21] R Li, Y Yang, P Hong, Z Zhang, L Li, J Hui, X Zheng. β-carotene attenuates weaning-induced apoptosis via inhibition of PERK-CHOP and IRE1-JNK/p38 MAPK signalling pathways in piglet jejunum. Journal of Animal Physiology and Animal Nutrition 2020; 104(1), 280-290.
[22] HKM Alboaklah and DS Leake. Effect of vitamin E on low density lipoprotein oxidation at lysosomal pH. Free Radical Research 2020; 54(8-9), 574-584.
[23] S Syarkiah, LE Fitri and A Pudjirahaju. The effect of red fruit (Pandanus conoideus) oil toward the formation of foam cells in aorta of wistar strain rat (Rattus norvegicus) with atherogenic diet (in bahasa Indonesia). Jurnal Kedokteran Brawijaya 2008. https://doi.org/10.21776/ub.jkb.2008.024.01.1
[24] N Karim, I Khan, W Khan, I Khan, A Khan, SA Halim, H Khan, J Hussain and A Al-Haarasi. Anti-nociceptive and anti-inflammatory activities of asparacosin a involve selective cyclooxygenase 2 and inflammatory cytokines inhibition: An in-vitro, in-vivo, and in-silico Approach. Frontier Immunollogy 2019; 10, 00581.
[25] JM Al-Khayri, GR Sahana, P Nagella, BV Joseph, FM Alessa and MQ Al-Mssallem. Flavonoids as potential anti-inflammatory molecules: A Review. Molecules 2022; 27(9), 2901.
[26] PV Pakale, CC Khanwelkar and SA Jadhav. Study of anti-inflammatory activity of aqueous and methanolic extracts of fresh rhizome of Zingiber officinale in Wistar rats. International Journal of Health Sciences 2022; 6(S3), 2209-2217.
[27] AA Cassado, MRD Lima and KR Bortoluci. Revisiting mouse peritoneal macrophages: Heterogeneity, development, and function. Frontiers in Immunology 2015; 6, 225.
[28] KH Lin, KC Lin, WJ Lu, PA Thomas, T Jayakumar and JR Sheu. Astaxanthin, a carotenoid, stimulates immune responses by enhancing IFN-γ and IL-2 secretion in primary cultured lymphocytes in vitro and ex vivo. International Journal of Molecular Science 2016; 17(1), 44.
[29] HJ Justil-Guerrero, JL Arroyo-Acevedo, JP Rojas-Armas, CO García-Bustamante, M Palomino-Pacheco, RD Almonacid-Román and JWC Torres. Evaluation of bioactive compounds, antioxidant capacity, and anti-inflammatory effects of lipophilic and hydrophilic extracts of the pericarp of Passiflora tripartita var. mollissima at two stages of ripening. Molecules 2024; 29(20), 4964.