Trends
Sci.
2026;
23(5):
12241
Pomegranate Extract as A Natural Therapy for Preeclampsia: Enhancing Endothelial Function, Modulating Inflammation, and Boosting Antioxidant Activity
Endang
Sri Wahyuni1,2,*,
Soetrisno
Soetrisno1,3,
![]() ,
Bambang Purwanto1,4,
,
Bambang Purwanto1,4,
![]() ,
Brian Wasita1,5,
,
Brian Wasita1,5,
![]() ,
Vitri Widyaningsih1,6,
,
Vitri Widyaningsih1,6,
![]() ,
Risya Cilmiaty1,7,
,
Risya Cilmiaty1,7,
![]() and Paramasari Dirgahayu1,8,
and Paramasari Dirgahayu1,8,
![]()
1Doctoral Program of Medical Sciences, Faculty of Medicine, Universitas Sebelas Maret, Surakarta 57126, Indonesia
2Departement of Midwifery, Faculty of Health Sciences, Universitas Aisyiyah Surakarta, Surakarta 57126, Indonesia
3Department of Obstetrics and Gynaecology, Faculty of Medicine, Universitas Sebelas Maret,
Surakarta 57126, Indonesia
4Department of Internal Medicine, Faculty of Medicine, Universitas Sebelas Maret, Surakarta 57126, Indonesia 5Department of Anatomical Pathology, Faculty of Medicine, Universitas Sebelas Maret, Surakarta 57126, Indonesia
6Department of Public Health, Faculty of Medicine, Universitas Sebelas Maret, Surakarta 57126, Indonesia
7Department of Oral Disease, Faculty of Medicine, UNS Hospital, Universitas Sebelas Maret,
Surakarta 57126, Indonesia
8Departement of Parasitology, Faculty of Medicine, Universitas Sebelas Maret, Surakarta 57126, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 11 October 2025, Revised: 14 November 2025, Accepted: 21 November 2025, Published: 10 January 2026
Abstract
Preeclampsia is a pregnancy complication characterised by hypertension in which blood flow to the mother and baby is reduced. It significantly affects the health of the mother and foetus and can lead to organ damage. Pomegranates are rich in bioactive compounds, especially polyphenols, which have antioxidant and anti-inflammatory properties, making them a potential therapeutic strategy for preeclampsia. Inflammation, oxidative stress, and autoimmune responses are significant in the pathogenesis of preeclampsia. This study aimed to explore the bioactive compounds in pomegranate (Punica granatum) fruit extract through phytochemical analysis; 2,2-diphenyl-1-picrylhydrazyl (DPPH) antioxidant assays; absorption, distribution, metabolism, excretion, and toxicity (ADMET) prediction; molecular docking; and in silico simulations using molecular targets involved in the pathophysiology of preeclampsia. Pomegranate fruit extract was obtained using a maceration technique. The extract was found to contain high levels of polyphenols, flavonoids, and tannins. It also exhibited strong antioxidant activity, with a half maximal inhibitory concentration of 20.967 (± 0.149) µg/mL. Molecular docking indicated strong binding affinity between luteolin and aschantin and the key targets carbonic anhydrase II, endothelial nitric oxide synthase (eNOS) and nuclear factor kappa-light-chain enhancer of activated B cells (NF-kB), demonstrating their potential reduce oxidative stress and inflammation and improve endothelial function. Our ADMET predictions demonstrated favourable pharmacokinetic properties and low toxicity, supporting the safety of luteolin and aschantin. These findings demonstrate the potential of pomegranate extract as a natural therapeutic agent for the management of preeclampsia and provide a basis for future clinical studies.
Keywords: Antioxidants, Bioactive compounds, Endothelial function, Inflammation, Molecular docking, Pomegranate, Preeclampsia
Introduction
Preeclampsia is a severe pregnancy complication characterized by hypertension and proteinuria, leading to potential damage in various maternal organs, including the kidneys and liver. It poses a significant threat to maternal and foetal health, contributing to increased maternal morbidity and mortality. Globally, preeclampsia accounts for approximately 9% - 26% of maternal deaths, highlighting the urgent need for effective therapeutic interventions [1-3]. Recent research has identified various factors involved in the pathophysiology of preeclampsia, including endothelial dysfunction, oxidative stress and inflammation [4]. The complex interplay between these factors underscores the need for innovative approaches in the management of preeclampsia.
Numerous
therapeutic strategies have been explored, including the use of
antihypertensive agents and corticosteroids. However, these
treatments have various limitations and side effects, especially in
pregnant women and when used over extended periods. The limitations
of antihypertensive and corticosteroid therapy in preeclampsia are
[5,6] primarily due to their inability to address the underlying
cause of the disease, the potential risks to fetal development, and
the lack of strong clinical evidence supporting their efficacy in
altering disease progression [7]. Consequently, there has been
growing interest in exploring natural compounds as alternative or
complementary therapies for preeclampsia. Among these, pomegranate
(Punica
granatum)
has emerged as a promising candidate due to its rich phytochemical
profile, including bioactive compounds such as polyphenols,
flavonoids and tannins, which are known for their antioxidant,
anti-inflammatory and vasculoprotective [8-10]. Pomegranate offers
several unique advantages compared to other plants that are also
rich in polyphenols, namely a synergistic effect on the vascular
endothelium, a balanced increase in immunity, thereby improving
endothelial function by modifying the inflammatory profile and
oxidative stress [5,6]. The use of pomegranate extract, particularly
in the management of oxidative stress and inflammation, may offer an
alternative or complement to conventional therapies.
Pomegranate extract can reduce the levels of biomarkers of oxidative stress, such as carbonic anhydrase II, and increase the activity of antioxidant enzymes, making it a viable candidate for use in the reduction of oxidative damage during preeclampsia [11]. Furthermore, the anti-inflammatory effects of pomegranate, as evidenced by its ability to decrease levels of pro-inflammatory cytokines such as TNF-α and IL-6, may help to improve endothelial function and reduce hypertension in preeclampsia [1]. Despite these promising findings, there have been few comprehensive studies into the molecular mechanisms through which pomegranate compounds, particularly their individual bioactive components, target the key pathways involved in preeclampsia.
This study aims to explore the bioactive compounds in pomegranate extract and able to interact beneficially with molecular targets such as carbonic anhydrase II, endothelial nitric oxide synthase (eNOS) and nuclear factor kappa-light-chain enhancer of activated B cells (NF-kB), which are crucial in the pathophysiology of preeclampsia. Using an integrated approach that includes phytochemical analysis, antioxidant assays, molecular docking, and in silico simulations, we sought to identify key compounds in pomegranate able to interact beneficially with molecular targets such as carbonic anhydrase II, endothelial nitric oxide synthase (eNOS) and nuclear factor kappa-light-chain enhancer of activated B cells (NF-kB) in women with preeclampsia. This study offers a novel and profound contribution to the understanding of the therapeutic potential of pomegranate extract in preeclampsia, paving the way for its future clinical application, and distinguishing our study from previous studies.
Materials and methods
Materials and sample collection
The primary material used in this study was pomegranate (Punica granatum) fruit, which was selected based on optimal ripeness and freshness criteria. The criteria for a ripe pomegranate are that it has a bright red and even skin color, and has a weight that matches its size [12]. The freshness of the pomegranate can be seen from the condition of the skin, which must be free from damage, spots, or dents [13]. The fruit samples were collected from a pomegranate farm in Dusun Merak, Desa Sumberwaru, Kecamatan Banyuputih, Kabupaten Situbondo, Jawa Timur, Indonesia, on November 2, 2024. The specimens were identified and verified by the Unit Pelaksana Fungsional Pelayanan Kesehatan Tradisional Tawangmangu RSUP Dr Sardjito (certification no. TL.02.04/D.XI.6/24934.1145/2024).
Sample preparation and extraction procedure
The pomegranate fruit were washed thoroughly with aquades to remove any contaminants, then dried in a forced-air oven at 50 °C for 72 h, and ground into a fine powder into a fine powder using a mechanical grinder and sieved through a 60-mesh screen. Extraction was performed using the maceration method. The pomegranate powder was mixed with 70 %(v/v) ethanol and stirred intermittently using a magnetic stirrer to ensure homogeneity. The mixture was left to macerate for 120 h at room temperature (at 24 ± 2 °C) in a closed container. This was protected from light to prevent the degradation of sensitive compounds. The mixture was then separated by vacuum filtration using Whatman No. 1, and the ethanol solvent was evaporated using a rotary evaporator, resulting in a concentrated extract. The extract was then dried in an oven at 40 °C until a constant weight was achieved, yielding a thick paste ready for further analysis.
Phytochemical analysis methods
Phytochemical screening
The total content of flavonoids, phenols, tannins, alkaloids, and saponins in the pomegranate extract was determined using Ultraviolet-visible (UV-vis) spectroscopy with a Shimadzu UV-1800 Spectrophotometer (Shimadzu Corporation, Japan). The instrument was calibrated according to internal procedures using Holmium oxide, Didymium, K₂Cr₂O₇, and toluene/hexane standards. Sample testing followed the laboratory’s internal procedures at LPPT UGM. The reagents and conditions used for each assay were:
Total phenols: Measured with Folin-Ciocalteu reagent, incubated for 30 min at room temperature, absorbance read at 760 nm, results expressed as gallic acid equivalents.
Total flavonoids: Measured using the aluminum chloride method, incubated for 30 min at room temperature, absorbance read at 510 nm, expressed as quercetin equivalents.
Total tannins: Also measured with Folin-Ciocalteu reagent, absorbance at 760 nm, expressed as tannic acid equivalents.
Total alkaloids: Colorimetric method, absorbance at 470 nm, expressed as quinine equivalents.
Total saponins: Assay based on Quillaja bark, absorbance at 435 nm.
Calibration curves for each assay were constructed using standard compounds: Gallic acid (phenols), quercetin (flavonoids), tannic acid (tannins), quinine (alkaloids), and saponins from Quillaja bark. Screening was performed at the LPPT of Universitas Gadjah Mada (certificate no. 1797/UN1/LPPT/TR2024), in Yogyakarta city, Indonesia.
Gas chromatography-mass spectrometry (GC-MS)
Gas chromatography-mass spectrometry (GC-MS) analysis was performed to identify volatile and semi-volatile compounds in the Punica granatum (pomegranate) extract. The extract (50 mg) was dissolved in 1 mL of ethanol to achieve a final concentration of A mg/mL. The solution was homogenized, centrifuged, and the supernatant was transferred into a vial for injection. The analysis was conducted at LPPT UGM (certificate no. 1798/UN1/LPPT/TR2024), in Yogyakarta city, Indonesia, using a Thermo Scientific TRACE1310 GC coupled with a Thermo Scientific ISQ LT MS system. The system was equipped with an HP-5MS UI capillary column (30 m×0.25 mm×0.25 µm film thickness). Mass spectrometric data were obtained over a range of 40 to 500 m/z. Chromatographic separation was performed with the following temperature program: The oven was initially set to 110 °C, held for 2 min, then increased to 200 °C at 10 °C/min and held for 5 min, followed by an increase to 280 at 5 °C/min with a final hold for 10 min. Helium was used as the carrier gas at a flow rate of 1 mL/min. Electron Ionization (EI) was used as the ionization mode, with the ion source temperature set to 230 °C and the interface temperature at 250 °C. Data were processed by comparing the mass spectra to public and internal metabolite databases for bioactive compound identification, with criteria including mass error ≤ 5 ppm, isotope matching, and signal intensity matching.
Liquid chromatography-mass spectrometry (LC-MS/MS) with quadrupole time-of-flight (QTOF)
The metabolite profile of the pomegranate extract was analyzed using LC-MS/MS with QTOF technology. The analysis was performed using a Waters Xevo G2-XS QTOF mass spectrometer (Waters Corporation, USA) coupled with an Agilent 1290 Infinity II HPLC system (Agilent Technologies, USA). The extract (50 mg) was dissolved in 1 mL of ethanol to achieve a final concentration of 50 mg/mL. The prepared sample was injected into the HPLC system equipped with a Kinetex C18 column (100×2.1 mm2, 1.7 µm particle size; Phenomenex, USA). The mobile phase consisted of: Solvent A: Water with 0.1% formic acid; and Solvent B: Acetonitrile. The gradient elution program was as follows: 0 to 2 min: 5% Solvent B; 2 to 10 min: 5% to 95% Solvent B; 10 to 12 min: 95% Solvent B; 12 to 12.1 min: 95% to 5% Solvent B; 12.1 to 15 min: 5% Solvent B. Mass spectrometric analysis was conducted using the QTOF-MS system to acquire high-resolution mass spectra. The data obtained were compared to both public and internal metabolite databases to identify the bioactive compounds in the extract. Identification was based on strict criteria, including mass error less than or equal to 5 ppm, isotope matching, and signal intensity matching. LC-MS/MS analyses were performed at PT Saraswanti Indo Genetech (Certificate No. SIG.LHP.I.2025.141511551) in Bogor city, Indonesia.
Antioxidant activity assay
The antioxidant activity within the extract was evaluated using a 2,2-diphenyl-1-picrylhydrazyl (DPPH) assay. The extract was prepared at various concentrations (2.52 - 37.80 µg/mL), and each was mixed with a 0.4 mM DPPH solution. The mixture was incubated in the dark for 30 min, and the absorbance was measured at 516.5 nm using a spectrophotometer. The percentage of radical scavenging activity was calculated. The half maximal inhibitory concentration (IC50) value was determined from the regression curve of concentration versus inhibition. This represents the concentration of the extract required to inhibit 50 % of the DPPH radicals. Measurements were performed in triplicate, and the results were described as mean (± standard error of the mean [SEM]). DPPH assay were conducted at the LPPT of Universitas Gadjah Mada (certificate no. 52/UN1/LPPT/TR2025), in Yogyakarta city, Indonesia.
Molecular docking and ADMET predictions
Molecular docking simulations were performed to investigate potential interactions between pomegranate bioactive compounds and key molecular targets involved in the pathophysiology of preeclampsia. These targets were carbonic anhydrase II, eNOS, and NF-kB. The 3D structures of the phytochemical compounds and receptors were retrieved from PubChem (in SDF format), while the structures of eNOS (PDB ID: 3NOS), carbonic anhydrase II (PDB ID: 6VJ3), and NF-kB (PDB ID: 4DN5) were obtained from the Protein Data Bank (PDB) [14-16]. The bioactive compounds analyzed were luteolin, dencichine, aschantin, kaempferol, quercetin, ellagic acid, and punicalagin, selected based on their known biological activities.
Prior to docking, the ligands underwent energy minimization using the MMFF94 force field. Hydrogens were added, Gasteiger charges were assigned, and the protonation states were adjusted based on the physiological pH (7.4) where applicable. The ligands were then converted into the appropriate format (PDBQT) for docking with AutoDock and PyRx software. Docking simulations were carried out using AutoDock and PyRx (version 1.0.0), with the binding affinity of each compound evaluated by calculating the docking energy (ΔG) and the inhibition constant (Ki). The results provided insights into the potential modulation of target proteins by these compounds.
To assess the pharmacokinetic properties, bioavailability, and potential toxicity of the selected bioactive compounds, ADMET predictions were performed using SwissADME and pkCSM. The software versions used were SwissADME (version 1.0.0) and pkCSM (version 3.0), with the predictions conducted on [May 3, 2025]. The following ADMET properties were predicted: Lipinski’s Rule of Five to assess drug-likeness; Gastrointestinal absorption to predict bioavailability; Blood-brain barrier (BBB) penetration to evaluate CNS availability; Cytochrome P450 (CYP) inhibition potential; Acute oral toxicity (LD50); Hepatotoxicity; Carcinogenicity.
Statistical analysis
The results were presented as mean (SEM). The IC50 values were calculated by plotting the concentration of the extract against the percentage of inhibition using linear regression analysis. Differences between groups were assessed using one-way ANOVA, and p-value < 0.05 were considered statistically significant.
Ethical considerations
The research involving plant material was approved by the Ethics Committee of Universitas Sebelas Maret, Surakarta, Indonesia, (certification no. 254/UN27.06.11/KEP/EC/2024).
Results and discussion
Phytochemical analysis of pomegranate extract
The bioactive compounds present in pomegranate extract were identified and quantified using phytochemical analysis. Our analysis found various key compounds known for their antioxidant and anti-inflammatory properties, including phenols, polyphenols, flavonoids, tannins, and alkaloids. The phytochemical analysis of the pomegranate extract revealed the presence and quantification of several bioactive compound classes. As detailed in Tables 1 and 2, the extract contained significant concentrations of total phenols, flavonoids, tannins, alkaloids, and saponins. These findings are consistent with the established rich phytochemical profile of Punica granatum, underscoring the extract’s potential therapeutic value attributed to the well-documented antioxidant and anti-inflammatory properties of these compounds.
Qualitative and quantitative phytochemical screening
Quantitative analysis was conducted to determine the concentrations of key phytochemicals in the extract. Per 100 g of pomegranate extract, we found 0.31 g of flavonoids, 12.73 g of phenolics, 3.31 g of saponins, and 19.02 g of tannins. Each gram of pomegranate extract contained 467.74 μg of alkaloids. Flavonoids are antioxidant compounds that help protect cells from free radical damage. They exert their antioxidants effects through the reduction of inflammation and the protection of cells from oxidative damage. The higher the flavonoid concentration of a given substance, the greater its antioxidant potential. Phenolic compounds are also known for their antioxidant properties, as well as their protective effects against heart disease and cancer. High phenol content imbues significant potential health benefits. Saponins have anti-inflammatory properties and can influence lipid metabolism. Thus, the saponin content found in our extract likely offers health benefits worthy of further exploration. Tannins are known for their astringent (tissue-shrinking) and antioxidant properties. High tannin content can provide health benefits, such as protection against certain diseases. Alkaloids have various pharmacological effects. Although these were present in the pomegranate extract in relatively small amounts compared to the other compounds, their presence may still contribute some therapeutic effects [6,7,18].
Gas chromatography-mass spectrometry (GC-MS)
Our
examination of pomegranate extract using GC-MS identified 31
phytochemical compounds
(Table
1 and
Figure 1).
Among these, several, including 5-hydroxymethylfurfural (HMF),
melezitose, desulphosinigrin, and ethyl iso-allocholate (EIA), have
great potential as natural therapies for preeclampsia [17,19,20].
Their antioxidant, anti-inflammatory and blood pressure-regulating
properties make these compounds highly relevant to the management of
preeclampsia [18,21].
Table 1 Compounds identified in pomegranate extract using gas chromatography-mass spectrometry.
Peak |
Retention time, min |
Compound |
Molecular weight (Da) |
Relative area % |
1 |
4.25 |
1-nitro-2-acetamido-1,2-dideoxy-d-mannitol |
252 |
1.14 |
2 |
4.68 |
5-hydroxymethylfurfural |
126 |
6.26 |
3 |
4.86 |
6-acetyl-ß-d-mannose |
222 |
3.08 |
4 |
5.11 |
6-acetyl-ß-d-mannose |
222 |
0.36 |
5 |
5.54 |
melezitose |
504 |
0.38 |
6 |
5.75 |
2-myristynoyl pantetheine |
484 |
1.01 |
7 |
6.54 |
2-myristynoyl pantetheine |
484 |
0.93 |
8 |
7.06 |
2-myristynoyl pantetheine |
484 |
1.92 |
9 |
7.97 |
melezitose |
504 |
28.01 |
10 |
8.76 |
d- mannose |
180 |
7.16 |
11 |
9.12 |
d- mannose |
180 |
4.45 |
12 |
9.49 |
Desulphosinigrin |
279 |
3.33 |
13 |
10.15 |
Desulphosinigrin |
279 |
20.45 |
14 |
10.48 |
Desulphosinigrin |
279 |
8.77 |
15 |
10.92 |
[1,1’-bicyclopropyl]-2-octanoic acid, 2’-hexyl-, methyl ester |
322 |
0.35 |
16 |
11.32 |
2-myristynoyl pantetheine |
484 |
0.23 |
17 |
12.50 |
Cyclopropanebutanoic acid, 2-[[2-[[2-[(2-pentylcyclopropyl)methyl]cyclopropyl]methyl]cyclopropyl]methyl]-, methyl ester |
374 |
0.36 |
18 |
12.91 |
Ethyl iso-allocholate |
436 |
0.10 |
19 |
13.11 |
Estra-1,3,5(10)-trien-17ß-ol |
256 |
2.01 |
20 |
13.30 |
a-D-glucopyranoside, methyl 2-(acetylamino)-2-deoxy-3-O-(trimethylsilyl)-, cyclic methylboronate |
331 |
0.76 |
21 |
13.50 |
a-D-glucopyranoside, methyl 2-(acetylamino)-2-deoxy-3-O-(trimethylsilyl)-, cyclic methylboronate |
331 |
2.96 |
22 |
13.99 |
a-D-glucopyranoside, methyl 2-(acetylamino)-2-deoxy-3-O-(trimethylsilyl)-, cyclic methylboronate |
331 |
0.85 |
23 |
14.09 |
a-D-glucopyranoside, methyl 2-(acetylamino)-2-deoxy-3-O-(trimethylsilyl)-, cyclic methylboronate |
331 |
0.02 |
24 |
14.67 |
Ethyl iso-allocholate |
436 |
0.33 |
25 |
15.34 |
trans-13-octadecenoic acid |
282 |
3.26 |
26 |
15.65 |
Ethyl iso-allocholate |
436 |
0.32 |
27 |
16.57 |
Ethyl iso-allocholate |
436 |
0.13 |
28 |
22.72 |
Ethyl iso-allocholate |
436 |
0.14 |
29 |
22.99 |
Ethyl iso-allocholate |
436 |
0.62 |
30 |
23.35 |
Ethyl iso-allocholate |
436 |
0.01 |
31 |
32.18 |
Ethyl iso-allocholate |
436 |
0.32 |
The compounds melezitose, d-mannose, and desulphosinigrin were detected in high concentrations. These are known to have anti-inflammatory, antidiabetic, and antitumor properties and could therefore potentially address vascular inflammation and endothelial dysfunction in preeclampsia [22]. The 2-myristynoyl pantetheine compounds detected have the potential to increase cellular energy metabolism, reduce inflammation and repair the membrane structure of damaged endothelial cells [23,24]. Ethyl iso-allocholate and trans-13-octadecenoic acid have previously been found to regulate lipid metabolism and excert anti-inflammatory effecs. As such, they may play a role in preeclampsia treatment through the management of hypertension and vascular health [1,22]. Estra-1,3,5(10)-trien-17ß-ol can stabilise hormonal balance and improve endothelial function. This is particularly relevant to preeclampsia, as hormonal fluctuations can exacerbate endothelial cell impairment [25].
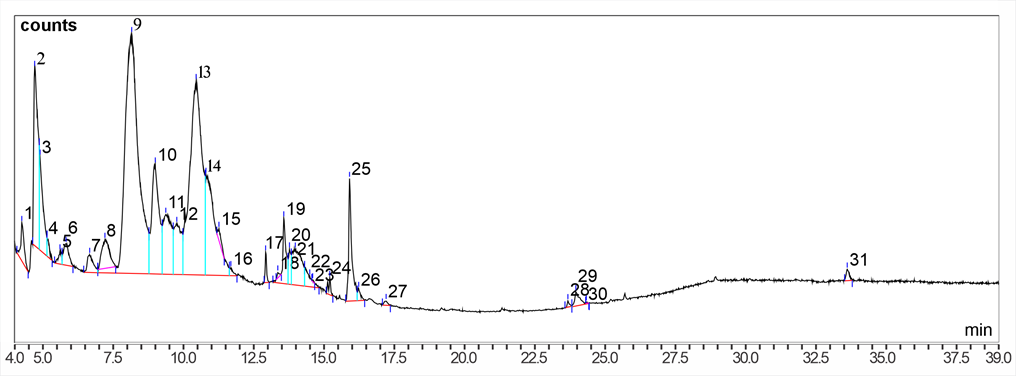
Figure 1 Chromatogram of gas chromatography-mass spectrometry analysis of the bioactive content of pomegranate fruit extract. The graph shows the distribution of the compounds in the extract based on their retention times. Large peaks indicate the dominant compounds in the sample.
Liquid chromatography-mass spectrometry (LC-MS/MS) with quadrupole time-of-flight (QTOF)
A number of the compounds detected by LC-MS/MS QTOF (Table 2) have therapeutic potential for preeclampsia, including dencichine, aschantin, brevifolin, and kaempferol [26]. These are known for their antioxidant and anti-inflammatory properties and may reduce inflammation and improve vascular health. High levels of kaempferol-3-glucuronide and quercetin-3-O-xyloside were detected, indicating that these flavonoid compounds could exert significant effects in the management of preeclampsia. These compounds can improve endothelial function, reduce inflammation, and reduce the oxidative stress associated with hypertension during pregnancy [1,11]. In addition, several compounds in the extract also have the potential to lower blood pressure and improve vascular health [21,27,28].
Table 2 Bioactive compounds identified in pomegranate extract using liquid chromatography-mass spectrometry with quadrupole time-of-flight.
No |
ESI |
Compound |
Compound groups |
1 |
(+) |
Dencichine |
Amino acids |
2 |
(+) |
(-)-olivil-4’’-O-β-D-glucopyranoside |
Coumarins & lignanoids |
3 |
(+) |
Aschantin |
Coumarins & lignanoids |
4 |
(+) |
Brevifolin |
Coumarins & lignanoids |
5 |
(+) |
Imperanene |
Coumarins & lignanoids |
6 |
(+) |
Kushecarpins B |
Coumarins & lignanoids |
7 |
(+) |
Myristicanol B |
Coumarins & lignanoids |
8 |
(+) |
Urolignoside |
Coumarins & lignanoids |
9 |
(−) |
(-)-olivil-4’-O-β-D-glucopyranoside |
Coumarins & lignanoids |
10 |
(−) |
(-)-secoisolariciresinol-4-O-β-D-glucopyranoside |
Coumarins & lignanoids |
11 |
(−) |
8-(1,1-dimethylallyl)-5-hydroxypsorolen |
Coumarins & lignanoids |
12 |
(−) |
Bavacoumestan B |
Coumarins & lignanoids |
13 |
(−) |
Brevifolin |
Coumarins & lignanoids |
14 |
(−) |
Citrusin B |
Coumarins & lignanoids |
15 |
(−) |
Interiotherin C |
Coumarins & lignanoids |
16 |
(−) |
Kushecarpins B |
Coumarins & lignanoids |
17 |
(−) |
Macrophylloside B |
Coumarins & lignanoids |
18 |
(−) |
Phellopterin |
Coumarins & lignanoids |
19 |
(−) |
Piperenone |
Coumarins & lignanoids |
20 |
(−) |
Tamariscinoside B |
Coumarins & lignanoids |
21 |
(−) |
Tellimagrandin I |
Coumarins & lignanoids |
22 |
(−) |
Urolignoside |
Coumarins & lignanoids |
23 |
(+) |
(2R,3R)-taxifolin-3′-O-β-D-glucopyranside |
Flavonoids & glycosides |
24 |
(+) |
1,8-dihydrxy-3,5-dimethoxyxanthone |
Flavonoids & glycosides |
25 |
(+) |
5,7,8,2′-tetrahydroxy-flavone-7-O-β-D-glucoside |
Flavonoids & glycosides |
26 |
(+) |
7-hydroxy-1-methoxy-2-methoxyxanthone |
Flavonoids & glycosides |
27 |
(+) |
Apigenol |
Flavonoids & glycosides |
28 |
(+) |
Genistein_1 |
Flavonoids & glycosides |
29 |
(+) |
Isoetin |
Flavonoids & glycosides |
30 |
(+) |
Isohyperoside |
Flavonoids & glycosides |
31 |
(+) |
Juglanin |
Flavonoids & glycosides |
32 |
(+) |
Kaempferol-3-gentiobioside |
Flavonoids & glycosides |
33 |
(+) |
Kaempferol-3-glucuronide |
Flavonoids & glycosides |
34 |
(+) |
Kaempferol-7-O-α-L-arabinofuranoside |
Flavonoids & glycosides |
35 |
(+) |
Luteolin |
Flavonoids & glycosides |
36 |
(+) |
Naringenin |
Flavonoids & glycosides |
37 |
(+) |
Nobiletin |
Flavonoids & glycosides |
38 |
(+) |
Quercetin-3-O-xyloside |
Flavonoids & glycosides |
39 |
(+) |
Quercetin-3-O-α-L-rhamnoside |
Flavonoids & glycosides |
40 |
(+) |
Viscidulin I |
Flavonoids & glycosides |
41 |
(−) |
(-)-epiafzelechin-3-O-(6"-O-acetyl)-β-D-allosepyranoside |
Flavonoids & glycosides |
42 |
(−) |
(-)-Epiafzelechin-3-O-β-D-allopyranoside |
Flavonoids & glycosides |
43 |
(−) |
3-O-β-D-Galacopyanosyl quercetin |
Flavonoids & glycosides |
44 |
(−) |
5-Hydroxy-6,4’-dimethoxy-flavone-7-O-β-D-glucopyranoside |
Flavonoids & glycosides |
45 |
(−) |
6-Hydroxykaempferol-3-O-glucoside |
Flavonoids & glycosides |
46 |
(−) |
Aloeresin |
Flavonoids & glycosides |
47 |
(−) |
Aloeresin C |
Flavonoids & glycosides |
48 |
(−) |
Cyclomorusin |
Flavonoids & glycosides |
49 |
(−) |
Epicatechin 5-O-β-D-glucopyranoside |
Flavonoids & glycosides |
50 |
(−) |
Hibiscetin-3-O-glucoside |
Flavonoids & glycosides |
51 |
(−) |
Icariin |
Flavonoids & glycosides |
52 |
(−) |
Juglanin |
Flavonoids & glycosides |
53 |
(−) |
Kaempferol 3-O-β-D-glucuronopyranosyl methyl ester |
Flavonoids & glycosides |
54 |
(−) |
Ledebouriellol |
Flavonoids & glycosides |
55 |
(−) |
Methyl ophiopogonone B |
Flavonoids & glycosides |
56 |
(−) |
Morusin hydroperoxide |
Flavonoids & glycosides |
57 |
(−) |
Myricetin |
Flavonoids & glycosides |
58 |
(−) |
Ombuine |
Flavonoids & glycosides |
59 |
(−) |
Quercetin 3-O-neohesperidoside |
Flavonoids & glycosides |
60 |
(−) |
Quercetin-3-O-xyloside |
Flavonoids & glycosides |
61 |
(−) |
Quercetin-3-O-α-L-rhamnoside |
Flavonoids & glycosides |
62 |
(−) |
Quercetin-6-O-glucoside |
Flavonoids & glycosides |
63 |
(−) |
Scutellarein |
Flavonoids & glycosides |
64 |
(−) |
Sibiricaxanthone B |
Flavonoids & glycosides |
65 |
(+) |
3,7-Dihydroxy-2,4-dimethoxyphenanthrene-3-O-glucoside |
Phenols |
66 |
(+) |
Collinin |
Phenols |
67 |
(+) |
Demethoxycurcumin |
Phenols |
68 |
(+) |
Didemethoxylcurcumin |
Phenols |
69 |
(+) |
Gemin D |
Phenols |
70 |
(+) |
Mallotinic acid |
Phenols |
71 |
(+) |
Pedunculagin |
Phenols |
72 |
(+) |
Stilbostemin B |
Phenols |
73 |
(−) |
1,2,3,6-Tetra-O-galloyl-β-D-glucopyranoside |
Phenols |
74 |
(−) |
1-Galloyl-β-D-glucose |
Phenols |
75 |
(−) |
2,3,5,4’-Tetrahydroxystilbene-2-O-(6’’-O-α-D-glucopyranosyl)-β-D-glucopyranoside |
Phenols |
76 |
(−) |
Castalagin |
Phenols |
77 |
(−) |
Cyclocurcumin |
Phenols |
78 |
(−) |
Demethoxycurcumin |
Phenols |
79 |
(−) |
Digupigan A |
Phenols |
80 |
(−) |
Eckol |
Phenols |
81 |
(−) |
Furosin |
Phenols |
82 |
(−) |
Gemin D |
Phenols |
83 |
(−) |
Geraniin |
Phenols |
84 |
(−) |
Meliadanoside B |
Phenols |
85 |
(−) |
Polygoacetophenoside |
Phenols |
86 |
(−) |
Rugosin C |
Phenols |
87 |
(−) |
Tubuloside E |
Phenols |
88 |
(+) |
Casuarinin |
Polyphenols (tannin) |
89 |
(+) |
Corilagin |
Polyphenols (tannin) |
90 |
(+) |
Ellagic acid |
Polyphenols (tannin) |
91 |
(+) |
Geraniin |
Polyphenols (tannin) |
92 |
(+) |
Icaritin |
Polyphenols (tannin) |
93 |
(+) |
Nilocitin |
Polyphenols (tannin) |
94 |
(+) |
Terchebulin |
Polyphenols (tannin) |
95 |
(+) |
Terflavin A |
Polyphenols (tannin) |
96 |
(−) |
1,2,3,4,6-penta-O-galloyl-β-D-glucopyranoside |
Polyphenols (tannin) |
97 |
(−) |
2,3-(S)-hexahydroxydiphenoyl-D-glucose |
Polyphenols (tannin) |
98 |
(−) |
2,4,6-tri-O-galloyl-β-D-glucose |
Polyphenols (tannin) |
99 |
(−) |
6′-O-galloyl-homoarbutin |
Polyphenols (tannin) |
100 |
(−) |
Casuarinin |
Polyphenols (tannin) |
101 |
(−) |
Corilagin |
Polyphenols (tannin) |
102 |
(−) |
Laevigatin A |
Polyphenols (tannin) |
103 |
(−) |
Nilocitin |
Polyphenols (tannin) |
104 |
(−) |
Potentillin |
Polyphenols (tannin) |
105 |
(−) |
Terchebin |
Polyphenols (tannin) |
106 |
(−) |
Terchebulin |
Polyphenols (tannin) |
107 |
(−) |
Terflavin A |
Polyphenols (tannin) |
The compound validation criteria were: Mass error ≤ 5 PPM, isotope match with MZ RMS PPM ≤ 6 ppm and isotope match with MZ RMS % ≤ 10 %, intensity/response ≥ 300 and fragment match ≥ 1 mass fragment.
ESI, electrospray ionization; MZ, mass-to-charge ratio of an ion; PPM, parts per million; RMS, root mean square.
Antioxidant activity of pomegranate extract
In our assessment of its antioxidant activity, pomegranate extract demonstrated radical scavenging activity, with an IC50 value of 20.97 µg/mL (Figure 2). This value indicates that the extract has the capacity to effectively neutralise free radicals at relatively low concentrations, suggesting potent antioxidant properties.
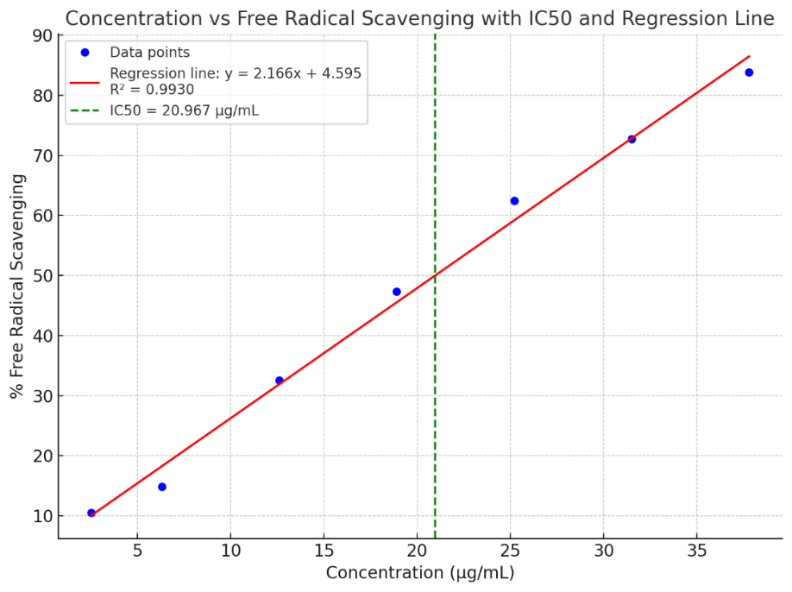
Figure 2 Antioxidant activity of pomegranate ethanol extract.
The graph shows the relationship between the concentration of pomegranate ethanol extract (µg/mL) and its free radical scavenging activity (%). The blue data points represent experimental values, while the red line is a best-fit linear regression curve (y = 2.166x+4.5849, R² = 0.9931), modelling the trend in antioxidant activity with increasing concentrations. The green dashed vertical line represents the IC50 value, which is the concentration required to inhibit 50% of free radicals. Thus the IC50 value of 20.967 (± 0.149) µg/mL, in this case is the concentration at which the extract exhibits 50% scavenging activity.
Molecular docking results
Docking with carbonic anhydrase II
In our molecular docking studies of pomegranate extract compounds and preeclampsia-related carbonic anhydrase II targets, luteolin and aschantin exhibited strong binding affinity with carbonic anhydrase II. Luteolin exhibited a binding energy of –5.9 kcal/mol and an inhibition constant of approximately 47 µM, This is close to the values for the standard drug preeclampsia treatment [29,30], have (–5.87 kcal/mol, 49.65 µM). Aschantin exhibited the strongest affinity, with a binding energy of –6.96 kcal/mol and an inhibition constant of 7.97 µM. These findings suggest that these compounds may be effective carbonic anhydrase II inhibitors in the treatment of preeclampsia.
Docking with endothelial nitric oxide synthase
The molecular docking results of pomegranate extract compounds and endothelial nitric oxide synthase (eNOS) revealed several important findings. Aschantin displayed the greatest binding potential, with a binding energy of –7.21 kcal/mol and a low inhibition constant of 5.21 µM, indicating strong increase of eNOS. Luteolin also showed strong binding 6.45 kcal/mol with an inhibition constant around 19 µM, suggesting its potential as an eNOS modulate. Ellagic acid had a promising binding energy of –7.01 kcal/mol, with inhibition constants around 7-11 µM, also indicating strong modulation potential. Despite its high binding energy (126.84 kcal/mol), puncalagin showed a very weak inhibition constant, suggesting less potent effect on eNOS modulate others compounds. These findings indicate that aschantin, luteolin, and ellagic acid have significant enhancer potential against eNOS and are promising therapeutic candidates for preeclampsia treatment.
Docking with nuclear factor kappa-light-chain enhancer of activated B cells
Our molecular docking study on pomegranate extract compounds and NF-kB revealed aschantin to have the most potent binding energy of –9.41 kcal/mol, with an extremely low inhibition constant (0.126 µM), demonstrating the ability to strongly inhibit NF-kB. Luteolin also exhibited a good binding energy of –7.75 kcal/mol, with an inhibition constant of 2.07 µM, indicating good inhibition. Dencichin, demonstrated weak binding energy between (–6.61 and –6.68 kcal/mol) with a very high inhibition constants (12.76 - 3,910 µM), suggesting limited potential in the inhibition of NF-kB. Punicalagin showed little binding affinity, and an inhibition constant exceeding, limiting its therapeutic potential. We found aschantin to be the compound in pomegranate extract with the most promise for use in the treatment of NF-kB-related diseases like preeclampsia.
Luteolin and aschantin was found to interacts with carbonic anhydrase II. Carbonic anhydrase II contributes to the regulation of endothelial function by modifying local pH. In the context of preeclampsia, where endothelial dysfunction plays a central role, increased expression of carbonic anhydrase II in endothelial cells may influence vascular reactivity and improve placental blood flow, which is important for maternal and fetal health [16,31]. Luteolin exhibited good interaction with eNOS, and an ability to bind to active sites similar to that of the native ligand. As such, it has the potential to modulate eNOS, thereby influencing nitric oxide (NO) production and helping to stabilise vascular function. Aschantin exhibited very strong binding to eNOS, superior to that of luteolin. Its anti-inflammatory abilities mean that it may reduced the risk of vascular complications in preeclampsia. Luteolin was found to interacts with NF-kB a key inflammation pathway. It showed the potential to inhibit NF-kB activation, which would reduce the production of pro-inflammatory cytokines. By inhibiting NF-kB, luteolin may reduce inflammation and control the immune response, which is crucial for the prevention of vascular inflammation in preeclampsia. Aschantin also demonstrated this capacity, but with stronger inhibitory potency than luteolin. Thus, aschantin may more effectively reduce the inflammation and oxidative stress that contributes to vascular dysfunction and hypertension in preeclampsia. Luteolin and aschantin both exhibited potent biological activity against eNOS, carbonic anhydrase II, and NF-kB, with aschantin demonstrating greater potency in the inhibitione of carbonic anhydrase II and NF-kB, as well as the reduction of oxidative stress, making it a more promising therapy for preeclampsia.
Quercetin exhibited similar interactions with the native ligand, but with significant differences at several residues. Quercetin bound more strongly to different polar and hydrophobic residues at the protein’s active site. Hydrogen bonds formed between quercetin and polar residues (such as Thr199, Gln92, and His94) indicate strong interactions and strengthen the binding affinity of quercetin to the target protein. Quercetin exhibited greater binding stability than the native ligand. This suggests greater anti-inflammatory, antioxidant, and antihypertensive therapeutic potential.
We found ellagic acid to interacts with the active site of the carbonic anhydrase II protein through both hydrogen bonds and non-hydrogen bonds. It formed hydrogen bonds with the polar residues Thr199, Gln92, and His94 indicating strong interactions. Its non-hydrogen hydrophobic interactions helped to increase the stability of the complex. While ellagic acid also exhibited weaker interactions than the native ligand with several hydrophobic residues, it still showed stable binding affinity with the target protein. Therefore ellagic acid has therapeutic potential in the management of preeclampsia due to its ability to reduce oxidative stress, inflammation, stabilise endothelial function, and reduce hypertension.
Table 3 The root mean square deviations obtained using molecular docking for preeclampsia-related ligands and their replication on the target proteins.
Repetition |
RMSD (Ǻ) |
Overlay |
Overlay replications 1-3 |
A. Carbonic Anhydrase II |
|||
Replication 1 |
1,409 |
|
Mean RMSD (± SD) 1,191 (± 0.190)
|
Replication 2 |
1,107 |
|
|
Replication 3 |
1,058 |
|
|
B. Endothelial nitric oxide synthase (eNOS) |
|||
Replication 1 |
1,798 |
|
Mean RMSD (± SD) 1,663 (± 279.49)
|
Replication 2 |
1,342 |
|
|
Replication 3 |
1,850 |
|
|
C. Nuclear factor kappa B (NF-kB) |
|||
Replication 1 |
2,168 |
|
Mean RMSD (± SD) 1,857 (± 566.30)
|
Replication 2 |
2,199 |
|
|
Replication 3 |
1,203 |
|
|
Description: Molecular docking was performed using AutoDock 1.5.6 open-access software. We compared the binding values of the reference ligand-ligand binding site receptor and the copy ligand-ligand binding site receptor. The crystallographic structure results were expressed as RMSDs. The docking method is considered valid when the RMSD value is ≤2 - 5 Å. RMSD, root mean square deviation; SD, standard deviation
We found that each ligand and replication on the carbonic anhydrase II target protein in Table 3(A) showed that the molecular docking was valid because the RMSD value was ≤2 Ǻ. Similarly, the ligand and replication test on the eNOS target protein in (B) showed that the molecular docking was valid because the RMSD value was ≤2 Ǻ. (C) the molecular docking was valid with an RMSD value of ≤2 Ǻ on the 3rd replication. However, replications 1 and 2 obtained RMSD values >2 Ǻ.
When we attempted to bind luteolin, dencichin, aschantin, kaempferol, quercetin, ellagic acid, and punicalagin to carbonic anhydrase II, eNOS, and NF-kB, only two of the compounds luteolin and aschantin, consistently boud well to these molecules (Figure 3).
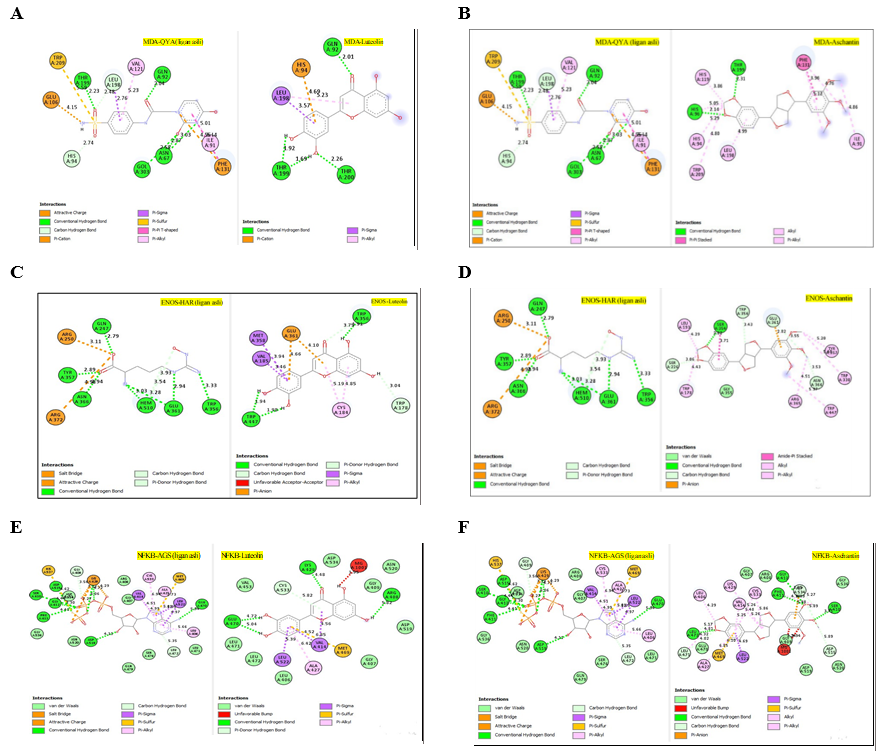
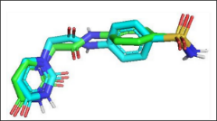
Figure 3 Comparisons of the interactions of luteolin and aschantin, and native ligands with target protein residues. (A) 2D diagram of native ligand vs luteolin interactions with carbonic anhydrase II residues. (B) 2D diagram of native ligand vs aschantin interactions with carbonic anhydrase II residues. (C) 2D diagram of native ligand vs luteolin interactions with eNOS residues. (D) 2D diagram of native ligand vs aschantin interactions with eNOS residues. (E) 2D diagram of native ligand vs luteolin interactions with NF-kB residues. (F) 2D diagram of native ligand vs aschantin interactions with NF-kB residues. The interaction were examined using AutoDock and Docking Server software eNOS, endothelial nitric oxide synthase; NF-kB, nuclear factor kappa-light-chain enhancer of activated B cells.
The hydrogen and non-hydrogen interactions of luteolin with carbonic anhydrase II protein residues were similar to those of the native ligand, but with additional interactions (Figure 3(a)). This may contribute to greater stability and therapeutic effects of this compound against the protein target. Hydrogen bonds between polar residues such as Thr199, Gln92, and Thr200 indicate stronger interactions, while non-hydrogen bonds are stabilising and hydrophobic. Aschantin showed stronger interactions with hydrophobic carbonic anhydrase II residues than the native ligand, suggesting a more stable binding affinity (Figure 3(b)). Luteolin exhibited stable binding to eNOS, with lower binding energy than the native ligand in its interactions with Thr199, Thr200, Gln92, Leu198 and, His94, which are involved in hydrogen and non-hydrogen bonds. The low inhibition constant (approximately 19 µM) indicated luteolin’s potential ability to modulate eNOS (Figure 3(c)). Aschantin exhibited a stronger affinity energy –7.21 kcal/mol, indicating that it binds more strongly to eNOS than luteolin. The inhibition constant of aschantin was 5.21 µM, which was lower than that of luteolin, indicating a greater eNOS modulation potential. Aschantin was found to interacts with amino acid residues Ser354, Leu193, Trp178, and Trp447, demonstrating more hydrogen and non-hydrogen interactions than luteolin, so greater influence on eNOS activity. Aschantin shows very strong binding results (with more negative binding) and very low inhibition constants, making it one of the best candidates for preeclampsia therapy (Figure 3(d)). Luteolin showed weaker binding energy than the native ligand for NF-kB. This was seen in its, interactions with the residues Glu470, Arg408, Lys429, Val414, Leu522, Met469, Ala427, and Cys533, for which the interaction distances range between 3.71 and 7.75 Å. However, luteolin still has therapeutic potential, especially as an anti-inflammatory (Figures 3(e) - 3(f)) Aschantin demonstrated much greater inhibitory effects on NF-kB than both luteolin and the native ligand. Aschantin was found to interacts with more amino acid residues, including Leu472, Phe411, Gly412, Cys533, and Lys429, with tighter interaction distances, (ranging from 3.71 to 7.73 Å), indicating stronger, more stable bonds with NF-kB.
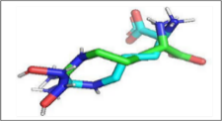
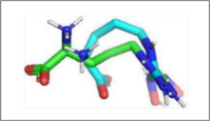
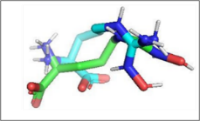
Figure 4 Visualisations of luteolin and aschantin’s interactions with target preeclampsia molecules. (A) carbonic anhydrase II – Luteolin interaction, (B) eNOS – Luteolin interaction, (C) NF-kB – Luteolin interaction, (D) carbonic anhydrase II – Aschantin interaction, (E) eNOS – Aschantin interaction, (F) NF-kB – Aschantin interaction.
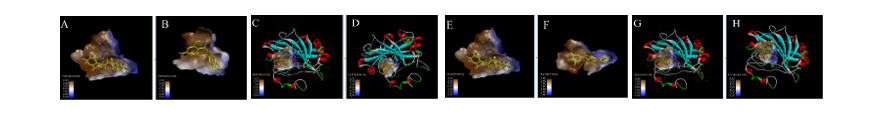
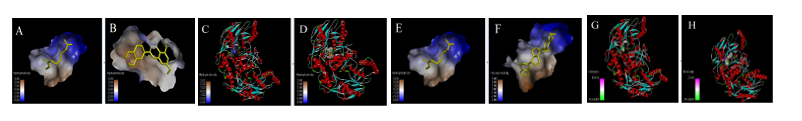
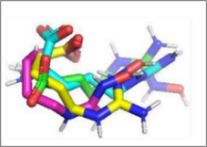
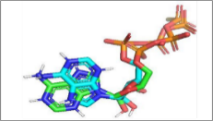
Figure 6 Visualisations of the hydrophobic and hydrogen surface areas of luteolin, aschantin and native ligands in their molecular interactions with endothelial nitric oxide synthase (eNOS) receptors., (A) Native ligand, (B) Luteolin, (C) Native ligand, (D) Molecular interactions of luteolin with eNOS receptor. (E) Native ligand, (F) Aschantin, (G) Native ligand, (H) Molecular interactions of aschantin with eNOS receptors.
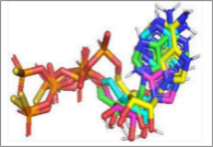
Figure 7 Visualisations of the hydrophobic and hydrogen surface areas of luteolin, aschantin and native ligands in their molecular interactions with, nuclear factor kappa-light-chain enhancer of activated B cells (NF-kB) receptors., (A) Native ligand, (B) Luteolin, (C) Native ligand, (D) Molecular interactions of luteolin with NF-kB receptor. (E) Native ligand, (F) Aschantin, (G) Native ligand, (H) Molecular interactions of aschantin with NF-kB receptors.
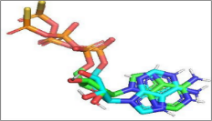
Visualization of molecular Interactions of luteolin and aschantin with carbonic anhydrase II, eNOS, and NF-kB (Figure 4) shows panels A - C (luteolin): The image of luteolin interactions with carbonic anhydrase II, eNOS, and NF-kB can be seen how luteolin interacts with amino acid residues on all three targets, with many hydrogen and non-hydrogen interactions. Panels D - F (aschantin): Shows the interaction of aschantin with targets carbonic anhydrase II, eNOS, and NF-kB showing stronger binding to the targets compared to luteolin. Figures 5 - 7 show stronger and more interactions of aschantin with all molecular targets carbonic anhydrase II, eNOS, and NF-kB compared to luteolin and native ligand. Aschantin has a larger hydrophobic surface area and hydrogen interactions on each target, indicating a higher potential in binding and inhibiting the activity of these targets. On carbonic anhydrase II , aschantin forms tighter bonds with hydrophobic residues, while luteolin has weaker bonds. At eNOS, aschantin also exhibits higher affinity by forming more hydrogen bonds than luteolin. At NF-kB, aschantin has stronger binding and more hydrophobic surface area than luteolin. Overall, aschantin exhibits greater potency in inhibiting all three targets than luteolin, thanks to its stronger and more extensive interactions with hydrophobic surface area and hydrogen bonds.
ADMET predictions
Absorption, distribution, metabolism, excretion, and toxicity (ADMET) predictions for seven of the pomegranate extract compounds. Our assessment of their toxicity and pharmacokinetic potential (is summarised in Table 4). Kaempferol (median lethal dose [LD50] 3,919 mg/kg, class V) demonstrated good bioavailability, but did not meet the necessary saturation parameters. Dencichin (LD50 5,000 mg/kg, class V). Was found not to penetrate the blood-brain barrier (BBB) and exhibited limited cell penetration. Ellagic acid (LD50 2991 mg/kg, class V), failed to meet several of the bioavailability parameters, including polarity and saturation. Luteolin (LD50 3,919 mg/kg, class V). Had good bioavailability, but did not meet the required saturation parameters. Punicalagin (LD50 3,700 mg/kg, class V), had low bioavailability and poor solubilisation. Aschantin (LD50 2,391 mg/kg, class V), showed good bioavailability and met most ADMET parameters. Despite its therapeutic potential, the bioavailability of quercetin (LD50 159 mg/kg, class III), was limited by its polarity and saturation. Overall, aschantin and kaempferol exhibited better pharmacokinetic profiles with lower toxicity, than quercetin.
Table 4 ADMET analysis of compounds from pomegranate fruit extract.
Compounds |
Boiled egg method |
Radar bioavailability |
LD50 (mg/kg) |
Toxicity risk |
Therapeutic potential |
Aschantin |
The compound penetrates the blood-brain barrier. |
Optimal lipophilicity and oral bioavailability, good saturation |
2,391 |
Low toxicity. Class V (may be harmful if swallowed): (2,000 < LD50 ≤ 5,000 mg/kg) |
Requires optimisation of its interactions with P-glycoprotein. Potential for development in an oral dosage form.
|
Luteolin |
Does not penetrate the blood-brain barrier |
Insufficient saturation |
3,919 |
Low toxicity. Class V (may be harmful if swallowed): (2,000 < LD5 ≤ 5,000 mg/kg)
|
Requires further evaluation of bioavailability and absorption. |
Kaempferol |
Does not penetrate the blood-brain barrier |
Insufficient saturation |
3,919 |
Low toxicity. Class V (may be harmful if swallowed): (2,000 < LD50 ≤ 5,000 mg/kg)
|
Requires further evaluation of bioavailability and absorption. |
Punicalagin |
Does not penetrate the blood-brain barrier |
Does not meet polarity and molecular weight requirements |
3,700 |
Low toxicity. Class V (may be harmful if swallowed): (2,000 < LD50 ≤ 5,000 mg/kg)
|
Further research is needed to improve oral bioavailability. Special formulations are needed to improve oral absorption.
|
Dencichin |
Does not penetrate the blood-brain barrier |
Insufficient lipofilicity |
5,000 |
Low toxicity. Class V (may be harmful if swallowed): (2,000 < LD50 ≤ 5,000 mg/kg)
|
Lipophilicity improvement is needed. |
Ellagic acid |
Does not penetrate the blood-brain barrier |
Does not meet saturation and polarity requirements |
2,991 |
Low toxicity. Class V (may be harmful if swallowed): (2,000 < LD50 ≤ 5,000 mg/kg)
|
Does not meet the criteria for oral administration. |
Quercetin |
Does not penetrate the blood-brain barrier |
Does not meet saturation and polarity requirements |
159 |
Moderate toxicity. Class III (toxic if swallowed): (50 < LD50 ≤ 300 mg/kg) |
Requires further research, with attention to appropriate dosing to minimise the risk of toxicity. |
LD50, median lethal dose.
Table 5 Results of biological activity predictions of the compounds of interest.
Compound |
Pa aryl hydrocarbon receptor |
Pa estrogen receptor alpha |
Pa aromatase |
Related biological activities |
|
Luteolin |
1 |
1 |
1 |
0.96 |
Anti-inflammatory, Antioxidant, Vasoprotective, Antihypertensive |
Kaempferol |
1 |
1 |
1 |
0.91 |
Anti-inflammatory, Antioxidant, Vasoprotective, Antihypertensive |
Aschantin |
0.96 |
0.87 |
0.57 |
0.96 |
Anti-inflammatory, Antioxidant, Vasoprotective |
Punicalagin |
0.96 |
0.88 |
0.96 |
1 |
Anti-inflammatory, Antioxidant, Vasoprotective |
Quercetin |
0.91 |
0.95 |
1 |
0.95 |
Anti-inflammatory, Antioxidant, Vasoprotective |
Dencichin |
0.91 |
0.98 |
1 |
0.57 |
Antioxidant, Vasoprotective |
Ellagic acid |
0.77 |
0.87 |
0.95 |
0.95 |
Anti-inflammatory, Antioxidant, Vasoprotective |
ADMET, absorption, distribution, metabolism, excretion and toxicity; Pa, probability ativity.
Our prediction results (Table 5) indicated that the main compounds in pomegranate extract demonstrate broad biological activity, especially anti-inflammatory and antioxidant [18], and vascular modulatory properties. Their high prediction values predicted activity (Pa) > 0.9 provide scientific justification for their further development as phytopharmaceutical candidates or health supplements. As anti-inflammatory and antioxidant agent [1]: They show high consistency, with high Pa mitochondrial membrane potential (MMP) and Pa against against aryl hydrocarbon receptor (AhR/) and oestrogen receptor alpha (ERα). Flavonoids and phenolics tend to neutralise free radicals, protect mitochondria, and reduce pro-inflammatory gene expression. They demonstrate vasoprotective and antihypertensive effects. Their interaction with ERα modulate endothelial function, and their antioxidant effects modulate NO scavenging. Moreover, they modulate of vascular enzymes and metabolites (For example, their aromatase modulation effects the balance of vasoregulatory steroids). We found some differences between the compounds: In particular punicalagin showed Pa aromatase of 1 and Pa MMP of 0.96 indicating very strong potential as an aromatase inhibitor and mitochondrial protector [32]. Awhereasa aschantin had a relatively low Pa MMP of 0.57, suggesting either a weaker mitochondrial protective effect or a different mechanism.
The therapeutic potential of pomegranate extract for the management of preeclampsia has garnered attention due to its bioactive compounds contents, which includes flavonoids, polyphenols and tannins, known for their antioxidant, anti-inflammatory, and endothelial function-enhancing properties. This study aimed to investigate the could influence of pomegranate extract through molecular docking, antioxidant activity assays, and ADMET predictions to explore its possible role in preeclampsia treatment. Our results suggest that pomegranate extract could serve as a promising natural remedy, highlighting the abilities of key bioactive compounds like luteolin and aschantin to target oxidative stress, inflammation, and endothelial dysfunction.
One of the main findings of this study was the significant antioxidant activity of pomegranate extract, which has an IC50 of 20.97 µg/mL. This value is indicative of a potent ability to neutralise the free radicals, central to the development of oxidative stress-induced damage in preeclampsia [11]. By reducing oxidative damage, the extract may mitigate endothelial dysfunction, a key contributor to preeclampsia hypertension. This is in concordance with the findings of previous studies indicating that polyphenolic compounds in pomegranate, particularly flavonoids like luteolin, possess strong antioxidant and anti-inflammatory properties [9,10,27,33-35].
Our molecular docking results further validated the ability of pomegranate extract to target specific molecular pathways in preeclampsia. Luteolin and aschantin exhibited particularly strong binding affinities to carbonic anhydrase II, eNOS and NF-kB. Carbonic anhydrase II contributes to the regulation of endothelial function by modifying local pH. In the context of preeclampsia, where endothelial dysfunction plays a central role, increased Carbonic anhydrase II expression in endothelial cells may influence vascular reactivity and improve placental blood flow, which is important for maternal and fetal health [31]. eNOS plays a crucial role in endothelial function by facilitating the production of NO, which regulates vascular tone. Impairment of eNOS activity is a hallmark of endothelial dysfunction in preeclampsia, which leads to reduced NO availability and contributes to hypertension [1,9]. Luteolin and aschantin showed more favorable binding energies, particularly aschantin, which demonstrated the strongest interaction with carbonic anhydrase II , eNOS, and NF-kB. NF-kB activation is known to increase the expression of pro-inflammatory cytokines, such as TNF-α and IL-6, which contribute to vascular inflammation and endothelial damage. The strong binding of aschantin to NF-kB suggests that it could effectively suppress the inflammatory cascade, thereby reducing the inflammatory environment that exacerbates preeclampsia symptoms [2]. These findings underscore the potential of these compounds to mitigate oxidative stress, inflammation, and endothelial dysfunction in preeclampsia.
Kaempferol, another key bioactive compound, exhibited notable antioxidant activity, though its molecular docking results were less impressive than those of luteolin and aschantin. It showed binding affinity to carbonic anhydrase II and eNOS, but a higher inhibition constant than luteolin and aschantin, suggesting that its modulation of the target molecules may be less potent. Nonetheless, endothelial-protective and anti-inflammatory properties have previously been identified in kaempferol, and it could still provide complementary effects in the management of preeclampsia [21,26].
Despite the promising molecular docking results, it is important to note that punicalagin, a major compound in pomegranate, showed weak binding affinity with both carbonic anhydrase II and NF-kB, indicating that its potential therapeutic role in preeclampsia treatment may be limited. Similarly, despite showing some binding affinity, dencichin exhibited high inhibition constants, suggesting limited modulation of oxidative stress and inflammation compared to other compounds like luteolin and aschantin. These findings align with those of a previous study, which also demonstrated variability in the efficacy of pomegranate extract compounds in modulating oxidative stress and inflammation [18].
The ADMET predictions for kaempferol, aschantin, luteolin, and ellagic acid collectively indicated favorable pharmacokinetic profiles, characterized by low toxicity and good bioavailability, suggest their potential as promising candidates for safe therapeutic agents for preeclampsia treatment. Aschantin displayed the most favourable pharmacokinetic profile, with good bioavailability and an LD50 of 2,391 mg/kg, placing it in class V. Conversely quercetin, was predicted to have relatively high toxicity, with an LD50 of 159 mg/kg, which placed it in class III (Table 4). Therefore, despite its therapeutic potential, the predicted toxicity of quercetin and low bioavailability reduce its clinical utility [1,36]
Pomegranate extract has emerged as a promising candidate as a therapeutic agent for preeclampsia, primarily due to its rich bioactive content of compounds like luteolin and aschantin. These compounds are predicted to possess strong antioxidant, anti-inflammatory, and endothelial-protective properties [37], which are essential for managing preeclampsia. Compared to conventional therapies such as antihypertensive agents (example methyldopa) and magnesium sulphate, pomegranate extract offers several advantages. First, it targets multiple pathophysiological mechanisms including oxidative stress, inflammation, and endothelial dysfunction suggesting it could offer a more holistic treatment option. Second, the natural origin of pomegranate extract is often associated with a perception of potentially reduced long-term side effects compared to synthetic drugs, although this requires rigorous clinical validation. Our study’s integrated approach, combining antioxidant assays, molecular docking, and ADMET predictions, strengthens the case for pomegranate as a natural therapeutic agent. However, further in vivo studies and clinical trials are essential to validate our findings and further explore the therapeutic potential of luteolin, aschantin and other bioactive compounds within pomegranate extract. Future research should also aim to optimise the formulation of pomegranate-based therapies to maximise their efficacy and minimise any adverse effects on pregnant women and their foetuses.
Limitations of this study include the lack of in vivo testing to confirm the molecular findings and the limited variety of preeclampsia models tested. Furthermore, potential interactions of the compound with other drugs and optimal dosages require further exploration through clinical trials.
Conclusions
Pomegranate extract, particularly its bioactive compounds luteolin and aschantin, potential significant therapeutic potential for managing preeclampsia. Through molecular docking, antioxidant activity assays, and ADMET predictions, these compounds showed strong predicted interactions with key biological targets involved in oxidative stress, inflammation, and endothelial dysfunction. Our findings suggest that pomegranate extract could serve as a natural, could potentially, and promising alternative to conventional therapies for preeclampsia treatment. Further clinical studies are needed to validate these results and optimise pomegranate-based therapies for use in pregnant women.
This study makes an important contribution to the understanding and development of pomegranate extract-based therapies for the management of preeclampsia. The potential use of pomegranate extract as an antioxidant and anti-inflammatory agent lays the groundwork for safer and more effective natural therapies for pregnant women with preeclampsia. However, these results need to be validated through in vivo studies and clinical trials to assess the safety, efficacy, and pharmacokinetics of pomegranate extract in humans. Future research should focus on comprehensive clinical investigations to establish standardized dosing regimens, bioavailability, and long-term outcomes associated with pomegranate extract, as well as its potential interactions with other medications commonly used in preeclampsia management. These studies are crucial for confirming the therapeutic potential of pomegranate extract and developing evidence-based guidelines for its use in clinical practice for preeclampsia.
Acknowledgements
The authors would like to thank the research supervisors from the Medical Sciences Study Program, Faculty of Medicine, Sebelas Maret University, Surakarta, Indonesia, and ‘Aisyiyah University of Surakarta, Surakarta, Central Java, Indonesia. The authors also express their gratitude to the Indonesian Education Scholarship Program (BPI), the Center for Funding and Assessment of Higher Education (PPAPT), the Center for Funding and Assessment of Higher Education, Ministry of Higher Education, Science, and Technology of the Republic of Indonesia, and the Education Fund Management Institute (LPDP).
Declaration of Generative AI in Scientific Writing
During the preparation of this work, generative artificial intelligence tools, primarily OpenAI’s ChatGPT, were used to enhance the language quality, correct grammatical errors, and improve the clarity of the text. This contributed to the overall readability of the manuscript. However, all scientific aspects of the study, including the formulation of the research design and procedures, data analysis and interpretation of the results, as well as the discussion points, and conclusions reached, were fully conceptualised and rigorously reviewed by the authors. The authors take full responsibility for the integrity, originality, and accuracy of the study and the content of this manuscript.
CRediT Author Statement
Endang Sri Wahyuni: Conceptualization, Methodology, Investigation, Formal analysis, Data curation, Writing – Original Draft. Soetrisno Soetrisno: Supervision, Conceptualization, Expertise in preeclampsia, Writing – Review and Editing. Bambang Purwanto: Supervision, Conceptualization, Validation, Writing – Review and Editing. Brian Wasita: Supervision, Methodology, Research design, Data analysis, Tissue examination preparation, Interpretation of results, Writing – Review and Editing. Vitri Widyaningsih: Data analysis, Methodology, Research design, Writing – Review & Editing. Risya Cilmiaty: Validation, Resources, Interpretation of result, Writing – Review and Editing. Paramasari Dirgahayu: Project administration, Research preparation, Methodology, Writing – Review & Editing.
References
[1] MY Ali, S Jannat and MS Chang. Discovery of potent angiotensin-converting enzyme inhibitors in pomegranate as a treatment for hypertension. Journal of Agricultural and Food Chemistry 2023; 71(30), 11476-11490.
[2] M Ożarowski, TM Karpiński, M Szulc, K Wielgus, R Kujawski, H Wolski and A Seremak-Mrozikiewicz. Plant phenolics and extracts in animal models of preeclampsia and clinical trials—review of perspectives for novel therapies. Pharmaceuticals 2021; 14(3), 269.
[3] S Gu, C Zhou, J Pei, Y Wu, S Wan, X Zhao, J Hu and X Hua. Esomeprazole inhibits hypoxia/endothelial dysfunction-induced autophagy in preeclampsia. Cell and Tissue Research 2022; 388(1), 181-194.
[4] J Jin, L Gao, X Zou, Y Zhang, Z Zheng, X Zhang, J Li, Z Tian, X Wang, J Gu, Z Wang and Q Zhang. Gut dysbiosis promotes preeclampsia by regulating macrophages and trophoblasts. Circulation Research 2022; 131(6), 492-506.
[5] ES Wahyuni, L Firrahmawati, IN Mahmudah and JD Handayani. Potensi buah delima (punica granatum) sebagai terapi adjuvan preeklampsia: A scoping review. Holistik Jurnal Kesehatan 2025; 19(5), 987-995.
[6] S Kerdsuknirund, A Thaeomor, P Kupittayanant, P Khunkaewla, S Chanlun, R Srisawat, P Tongdee, P Nimkuntod and S Kupittayanant. Pomegranate juice alleviates preeclampsia symptoms in an L-NAME-Induced Rat Model: A dose-dependent study. Nutrients 2025; 17(7), 1143.
[7] DD Sharma, NR Chandresh, A Javed, P Girgis, M Zeeshan, SS Fatima, TT Arab, S Gopidasan, VC Daddala, KV Vaghasiya, A Soofia and M Mylavarapu. The management of preeclampsia: A comprehensive review of current practices and future directions. Cureus 2024; 16(1), e51512.
[8] S Khan, T Khan and AJ Shah. Total phenolic and flavonoid contents and antihypertensive effect of the crude extract and fractions of Calamintha vulgaris. Phytomedicine 2018; 47, 174-183.
[9] M Trapali and V Lagouri. Vasculoprotective and neuroprotective effects of various parts of pomegranate: In vitro, in vivo, and preclinical studies. IntechOpen, London, 2021.
[10] D Wang, C Özen, IM Abu-Reidah, S Chigurupati, JK Patra, JO Horbanczuk, A Jóźwik, NT Tzvetkov, P Uhrin and AG Atanasov. Vasculoprotective effects of pomegranate (Punica granatum L.). Frontiers in Pharmacology 2018; 9, 00544.
[11] I Nasifah, S Soeharto, Nooryanto and Mukhamad. Effects of anti-lipid peroxidation of Punica granatum fruit extract in endothelial cells induced by plasma of severe pre-eclamptic patients. Journal of Ayurveda and Integrative Medicine 2017; 8(4), 215-217.
[12] I Tinebra, D Scuderi, G Sortino, A Mazzaglia and V Farina. Pomegranate cultivation in mediterranean climate: Plant adaptation and fruit quality of “Mollar de elche” and “Wonderful” cultivars. Agronomy 2021; 11(1), 156.
[13] BD Millo, V Martínez-Blay, MB Pérez-Gago M, Argente-Sanchis, A Grimal, E Baraldi and L Palou. Antifungal hydroxypropyl methylcellulose (HPMC)-lipid composite edible coatings and modified atmosphere packaging (MAP) to reduce postharvest decay and improve storability of “Mollar De Elche” pomegranates. Coatings 2021; 11(3), 308.
[14] TO Fischmann and PC Weber. Human endhothelial nitric oxide synthase with arginine substrate. Nature Structural & Molecular Biology 1999; 6, 233-242.
[15] X Min, J Liu, A Sudom, NP Walker and Z Wang. Crystal structure of NF-kB-inducing Kinase (NIK). Worldwide Protein Data Bank 2012. https://doi.org/10.2210/pdb4dn5/pdb
[16] CL Lomelino and R McKenna. Carbonic anhydrase II in complex with pyrimidine-based inhibitor. European Journal of Medicinal Chemistry 2020. https://www.wwpdb.org/pdb?id=pdb_00006vj3
[17] JY Jang, D Kim, E Im and ND Kim. Therapeutic potential of pomegranate extract for women’s reproductive health and breast cancer. Life 2024; 14(10), 1264.
[18] J Du, H Wang, L Zhong, S Wei, X Min, H Deng, X Zhang, M Zhong and Y Huang. Bioactivity and biomedical applications of pomegranate peel extract: A comprehensive review. Frontiers in Pharmacology 2025; 16, 1569141.
[19] E Palabıyık, H Uğuz, B Avcı, AN Sulumer, B Yılmaz and H Aşkın. Bioactive component analysis of seed coat hexane extract of Ardahan (Türkiye) walnut. Frontiers in Life Sciences and Related Technologies 2024; 5, 89-94.
[20] B Mészáros, Z Kukor and S Valent. Recent advances in the prevention and screening of preeclampsia. Journal of Clinical Medicine 2023; 12(18), 6020.
[21] SA Siddiqui, S Singh and GA Nayik. Bioactive compounds from pomegranate peels - Biological properties, structure–function relationships, health benefits and food applications – A comprehensive review. Journal of Functional Foods 2024; 116, 106132.
[22] M Yousaf, V Razmovski-Naumovski, M Zubair, D Chang and X Zhou. Synergistic effects of natural product combinations in protecting the endothelium against cardiovascular risk factors. Journal of Evidence-based Integrative Medicine 2022; 27, 2515690X221113327.
[23] G Li, M Chen, J Chen, Y Shang, X Lian, P Wang, H Lei and Q Ma. Chemical composition analysis of pomegranate seeds based on ultra-high-performance liquid chromatography coupled with quadrupole-Orbitrap high-resolution mass spectrometry. Journal of Pharmaceutical and Biomedical Analysis 2020; 187, 113357.
[24] L Jia, L Shi, J Li, Y Zeng, S Tang, W Liu, X Mo and X Liu. Total flavonoids from celery suppresses RANKL-induced osteoclast differentiation and bone resorption function via attenuating NF-κB and p38 pathways in RAW264.7 cells. Journal of Functional Foods 2020; 69, 103949.
[25] YY Shin, SM An, JS Jeong, SY Yang, GS Lee, EJ Hong, EB Jeung, SC Kim and BS An. Comparison of steroid hormones in three different preeclamptic models. Molecular Medicine Reports 2021; 23(4), 252.
[26] B Singh, JP Singh, A Kaur and N Singh. Phenolic compounds as beneficial phytochemicals in pomegranate (Punica granatum L.) peel: A review. Food Chemistry 2018; 261, 75-86.
[27] L Ciccone, S Nencetti, A Rossello and E Orlandini. Pomegranate: A source of multifunctional bioactive compounds potentially beneficial in Alzheimer’s disease. Pharmaceuticals 2023; 16(7), 1036.
[28] M Fourati, S Smaoui, HB Hlima, K Elhadef, OB Braïek, K Ennouri, AC Mtibaa and L Mellouli. Bioactive compounds and pharmacological potential of pomegranate (Punica granatum) seeds - A review. Plant Foods for Human Nutrition 2020; 75(4), 477-486.
[29] R Nirupama, S Divyashree, P Janhavi, SP Muthukumar and PV Ravindra. Preeclampsia: Pathophysiology and management. Journal of Gynecology Obstetrics and Human Reproduction 2021; 50, 101975.
[30] RA Sari, S Sulistiawati and E Ernawati. Maternal and perinatal outcomes of pre-referral magnesium sulfate treatment in severe preeclampsia patients. Majalah Obstetri & Ginekologi 2022; 30(1), 17-23.
[31] R Aliyazicioglu, S Guven, A Mentese, S Kolayli, S Cengiz, O Deger and A Alver. Serum anti-carbonic anhydrase II antibodies and oxidant-antioxidant balance in pre-eclampsia. American Journal of Reproductive Immunology 2011; 66(4), 297-303.
[32] Y Wang, M Huang, X Yang, Z Yang, L Li and J Mei. Supplementing punicalagin reduces oxidative stress markers and restores angiogenic balance in a rat model of pregnancy-induced hypertension. The Korean Journal of Physiology & Pharmacology 2018; 22(4), 409-417.
[33] MM Rahman, MR Islam, S Akash, ME Hossain, AA Tumpa, GMA Ishtiaque, L Ahmed, A Rauf, AA Khalil, WA Abdulmonem and J Simal-Gandara. Pomegranate-specific natural compounds as onco-preventive and onco-therapeutic compounds: Comparison with conventional drugs acting on the same molecular mechanisms. Heliyon 2023; 9, e18090.
[34] National Library of Medicine. Pomegranate. National Library of Medicine, Bethesda, 2023.
[35] AM Mahdavi and Z Javadivala. Systematic review of the effects of pomegranate (Punica granatum) on osteoarthritis. Health Promotion Perspectives 2021; 11(4), 411-425.
[36] S Zeng, Z Liu, J Yin, S Li, M Jiang, H Yang and Y Long. Improvement in clinical features of L-NAME-Induced preeclampsia-like rats through reduced SERPINA5 expression. Biomolecules 2023; 13(12), 1792.
[37] DK Patel. Biological potential and therapeutic effectiveness of aschantin and epiaschantin: Natural lignans abundantly found in magnolia flos. Coronaviruses 2023; 4(3), e220823220193.