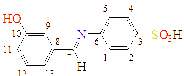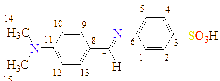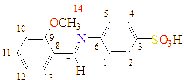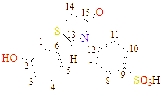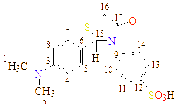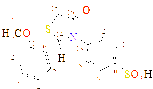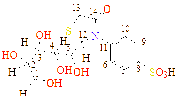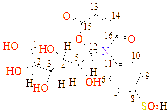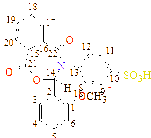![]() Trends
Sci. 2026; 23(5): 11951
Trends
Sci. 2026; 23(5): 11951
Synthesis and Characterization of Some Heterocyclic Compounds Derived from
Schiff Base and Evaluation of Their Biological Activities
Zainab Faiyq Saeed1, Ghufran Thanoon Sadeek2 and Rafid Kamal Jameel3
1Department of Physiology, Biochemistry, and Pharmacology, College of Veterinary Medicine, University of Mosul, Nineveh, Iraq
2Department of Chemistry, College of Education for Pure Science, University of Mosul, Nineveh, Iraq
3College of Dentistry, Al-Noor University, Nineveh, Iraq
(*Corresponding author’s e-mail: [email protected])
Received: 18 September 2025, Revised: 8 October 2025, Accepted: 18 October 2025, Published: 1 January 2026
Abstract
Heterocyclic compounds represent one of the most important classes in organic chemistry due to their wide range of biological and pharmacological activities. In this study, new Schiff bases were synthesized through the condensation of benzaldehyde derivatives with para-aminosulfonic acid. These intermediates were subsequently cyclized to obtain four-, five-, and seven-membered heterocyclic systems, including thiazolidine, oxazepine, and benzooxazepine derivatives. The structures of the synthesized compounds were confirmed using elemental analyses and spectral techniques (IR, ¹H-NMR, and ¹³C-NMR). The antimicrobial activity of the prepared compounds was evaluated against Gram-positive and Gram-negative bacterial strains, as well as yeast species. Results revealed that compounds 8, 10, 11, and 13 displayed notable inhibitory effects, particularly at higher concentrations, with compound 13 showing the strongest biological activity. Furthermore, molecular docking simulations demonstrated favorable binding interactions of selected compounds with the active sites of target proteins, indicating their potential as promising leads for drug development.
Keywords: P-aminosulfonic, Biological activities, Schiff base, Docking study, Heterocyclic, Antibacterial
Introduction
The heterogeneous ring systems containing nitrogen, sulfur, and oxygen atoms are of the largest classes, where many of them showed biological and medical efficacy and acted as anti-bacterial [1,2] and anti-fungal [3,4]. One of the most important methods of synthesis of heterocyclic compounds is by using Schiffʼs bases as intermediate to synthesis a new compounds thiazole and oxazipine where these compounds showed many biological activities and activities of a wide range as anti-inflammatory [5,6], analgesic [7], antimicrobial [8,9] and anti-cancer factors [10,11], antioxidant [12,13] and antidepressant [14,15]. In addition to other industrial uses as a nucleus for dyes [16,17] and surface corrosion inhibitors [18,19].
Sulfanilic acid is one of the compounds containing a sulfur atom in addition to the amine group (NH2) that was used in the preparation of types of dyes [20,21] as well as in the estimation of some pharmaceutical compounds [22,23]. Sulfanlic acid was used as a catalyst for many reactions [24,25] and some derivatives were prepared from it that gave biological activity as antibacterials [26,27].
Materials and methods
Synthesis of Schiff base (1 - 4) [28]
mixed (0.01 mol) of one of the benzaldehyde substitutes with (20 mL) of absolute ethanol, then (0.01 mol) para-sulfanilic acid and some drops of glacial acetic acid are added to it, the mixture reflux for 2 - 4 h, then it is cooled for sedimentation and crushed ice is added. The precipitate was filtered, then recrystallized with ethanol. Some physical constants and spectral properties are shown in Tables 1 - 4.
Table 1 The physical properties of Schiff base compounds (1 - 4).
Comp. No. |
R |
Molecular formula |
M.P. °C |
Yield % |
Color |
1 |
-OH |
C13H11NO4S |
183 - 185 |
55 |
Brawn |
2 |
-N(CH3) |
C15H16N2O3S |
113 - 114 |
65 |
Yellow |
3 |
-OCH3 |
C14H13NO4S |
137 - 139 |
60 |
Yellow |
4 |
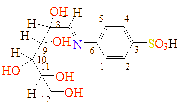
-C4H9O4 |
C13H19NO8S |
188 - 189 |
30 |
Synthesis of 4-(2-subsituted-4-thiazolidin-3-yl)benzenesulfonic acid (2 - 5) [29]
A mixture of equivalent moles of one of Schiffʼs bases (1 - 4) (mole 0.002) with (mole 0.002) (0.18 g) of thioglycolic acid was dissolved in (25 mL) of absolute ethanol, then (0.01 mole) (1.36) was added to it g) of anhydrous zinc chloride. The reaction mixture was reflux for (8 h), after which the mixture was cooled, then the formed precipitate was filtered and washed with a solution of (3%) sodium bicarbonate, then recrystallized from (dioxane - water). physical characteristics and spectral properties are shown in Tables 2.
Table 2 The physical properties of thiazolidine compounds (5 - 8).
Comp. No. |
R |
Molecular formula |
M.P. °C |
Yield % |
Color |
5 |
-OH |
C17H13NO7S |
161 - 162 |
45 |
Red |
6 |
-N(CH3) |
C19H18N2O6S |
139 - 140 |
55 |
Pall Yellow |
7 |
-OCH3 |
C18H15NO7S |
169 - 171 |
62 |
Orang |
8 |
-C4H9O4 |
C16H19NO11S |
166 - 167 |
57 |
Brawn |
Synthesis of 4-(2-subsituted-4,7-dioxo-4,7-dihydro-1,3-oxazepin-3(2H)-yl)benzenesulfonic acid (6 - 9) [30]
Equivalent moles consisting of (0.001 mole) of one of the prepared Schiff bases (1 - 4) are mixed with (0.001 mole) of maleic anhydride in (20 mL) of dry benzene. The reaction mixture ascends for (4 h). After that, the solution is concentrated under pressure. Sieve,
then filter or put the solution in a beaker and leave in the hood for a few hours until the precipitate forms alone. The precipitate is taken and recrystallized in ethanol. The physical constants and spectral properties are fixed in Tables 3, 11-13.
Table 3 The physical properties of oxazepine compounds (9 - 12).
Comp. No. |
R |
Molecular formula |
M.P. °C |
Yield % |
Color |
9 |
-OH |
C15H13NO5S2 |
225 - 226 |
44 |
Brawn |
10 |
-N(CH3) |
C17H18N2O4S2 |
198 - 199 |
39 |
Dark Brawn |
11 |
-OCH3 |
C16H15NO5S2 |
206 - 208 |
37 |
Brawen |
12 |
-C4H9O4 |
C14H19NO9S2 |
266 - 267 |
51 |
Pall Brawn |
Synthesis of 4-(3-subsituted-1,5-dioxo-1,5-dihydrobenzo[e][1,3]oxazepin-4(3H)-yl)benzenesulfonic acid (1013) [31]
Equivalent moles consisting of (0.001 mole) of one of the prepared Schiff bases (1 - 4) are mixed with 0.001 mole of phthalic acid anhydride in (20 mL) of dry benzene. The reaction mixture is ascended for (4 h). After that, the solution is concentrated under A sieve pressure, then filtered or the solution is placed in a beaker and left in the hood for a few hours until the precipitate forms alone. The precipitate is taken and recrystallized in ethanol. The physical constants and spectral properties are fixed in (4, 14, 15 and 16).
Table 4 The physical properties of benzooxazepine compounds (13 - 16).
Comp. No. |
R |
Molecular formula |
M.P. °C |
Yield % |
Color |
13 |
-OH |
C21H15NO7S |
222-224 |
55 |
Dark Brawn |
14 |
-N(CH3) |
C23H20N2O6S |
188-190 |
60 |
Orang |
15 |
-OCH3 |
C22H17NO7S |
212-213 |
54 |
Brawn |
16 |
-C4H9O4 |
C20H21NO11S |
287 Dec. |
59 |
Black |
Antibacterial activity [26,27,32]
The novel synthesized heterocyclic compounds were screened for their in-vitro antimicrobial activity using cup plate Agar disc-diffusion method against two Gram-positive bacterial strains, streptococcus pneumoniae and Staphylococcus aureus and two Gram-negative strains, Escherichia coli and klebsiella spp against .and strains of yeast (candidan albicans).the bacterial strains were obtained from clinical cases-Al-Salam hospital-Mosul .all bacterial strains were subcultured on Brain heart infusion agar at 37 °C for 24 h, before performing antimicrobial susceptibility test. General procedure for the determination of zone of inhibition by Agar disc-diffusion method Test solutions were prepared with known weight of compound in dimethyl sulfoxide (DMSO) and diluted suitably to give the resultant concentration of 25, 50, 75 and 100 mg/mL Concentrations (25, 50, 75 and 100) mg were dissolved in 1 mL of DMSO and mixed until dissolved, then used in tablets and added to the culture medium. sterile filter paper discs (6 mm) were impregnated with solution and allowed to dry at room temperature. In-vitro antibacterial activity was determined using Mueller Hinton Agar 24 h old culture of selected bacterial strain was mixed with physiological saline and the turbidity was corrected by adding sterile physiological saline and sub cultured on Sabouraud Dextrose and suspended in sterile distilled water to an Petri plates were prepared by pouring 10 mL of Mueller Hinton Agar for bacteria containing microbial culture was allowed to solidify. The discs were then applied, and the plates were incubated at 37 °C for 24 h (bacteria) A graduated cylinder in mm was used to measure the diameter of the formed circle (slide cage). and the inhibition zone was measured as diameter, Zone of inhibition in mm are given in the Table 9.
Results and discussion
Schiff base were prepared from the condensation of benzaldehyde substitutes with sulfanalic acid in the presence of absolute ethanol as a solvent and in the presence of glacial acetic acid as a catalyst. The interaction occurs through the mechanism of addition and deletion, as shown in Scheme 1 following [33]:
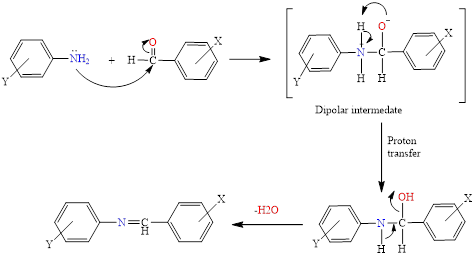
Scheme 1 Mechanism of Schiff base reaction.
Schiff base (1 - 4) were diagnosed using the infrared spectrum, as the spectrum showed an absorption band at the range (1,661 - 1,629) cm-1, which refers to the (C=N) bond, while the (C=C) bond stretch returns For the structural vibrations of the aromatic ring, it was shown at the frequency (1,601 - 1,458) cm-1. The (IR) spectral shows in Table 5
Table 5 The (IR) spectral properties of the Schiff base compounds (1 - 4).
Comp. No. |
Y |
C=C Aromatic cm-1 |
C−H Aromatic cm-1 |
C−H Aliphatic cm-1 |
C=N cm-1 |
S=O, S-O |
Others cm-1 |
1 |
-OH |
1,570 - 1,458 |
3063 |
2,836, 2,739 |
1,661 |
1,630 1,319 |
OH(3199), C-O(1237) |
2 |
-N(CH3) |
1,595 - 1,498 |
3045 |
2,911 |
1,654 |
1,608 1,281 |
- |
3 |
-OCH3 |
1,591 - 1,471 |
3050 |
2,964 |
1,644 |
1,605 1,276 |
C-O-C(1064,1198) |
4 |
-C4H9O4 |
1,601 - 1,498 |
3,061 |
2,862 |
1,629 |
1,614 1,240 |
OH(3440), C-O(1033,1153) |
The proton nuclear magnetic resonance (1H-NMR) spectrum of compound (1 - 4) was studied, as it showed a single signal at (6.998 ppm) for phenol ring at, single signal at (7.403 - 7.884 ppm) for sulfanilic acid ring and a single signal at (7.903 ppm) referring to H–C=N group, single signaling at (9.915 ppm) for proton of the sulfone group. the (1HNMR) and (13C-NMR) show in Tables 6 and 7.
Table 6 The (1H-NMR) spectral properties of Schiff base compounds (1 - 4).
No. Comp. |
1HNMR (δ, ppm) |
1 |
6.998 (t,H,C2 phenol ring);7.403-7.884 (d:d,4H,sulfanilic acid ring); 7.296,7.332,7.375 (m,3H,(C3,4,6)phenol ring); 7.903 (s,H, CH=N); 8.675 (s,H, OH phenol); 9.915 (s,H,OH sulfonic acid).
|
2 |
3.83(s,6H, di CH3); 6.781-7.076 (d,4H,N,N-dimethyl aniline ring); 7.49-7.51 (d:d,4H,sulfanilic acid ring); 8.105 (s,H,CH=N); 9.667 (s,H,OH sulfonic acid).
|
3 |
3.952(s,3H,OCH3); 7.095-7.21 (d,2H,(C3,4)anisole ring); 7.62 (d,H,C5 anisole ring); 7.287 (t,H,C2 anisole ring); 7.239-7.771 (d:,4H,sulfanilic acid ring); 9.031 (s,H,CH=N); 10.362 (s,H,OH sulfonic acid).
|
4 |
3.168,4.130 (d,2H,C1(H+OH)); 3.421,4.175 (q,1H,C2(H+OH)); 3.506,4.221 (t,1H,C3(H+OH)); 3.567,4.257(t,1H,C4(H+OH)); 4.081,4.227 (t,1H,C5(H+OH)); 7.169-7.653 (d:d,4H,sulfanilic acid ring); 8.134 (d,H,CH=N, C6); 9.434 (s,H,OH sulfonic acid).
|
Table 7 The (C-13 NMR) spectral properties of Schiff base compounds (1 - 4).
No. Comp. |
Structure compounds |
13C-NMR (δ, ppm) |
1 |
|
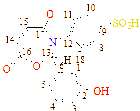
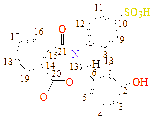
C1(115), C2(121), C3(148), C4(121), C5(115), C6(120), C7(193), C8(132), C9(127), C10(158), C11(127), C12(130), C13(130)
|
2 |
|
C1(111), C2(119), C3(147), C4(119), C5(109), C6(120), C7(186), C8(132), C9(127), C10(151), C11(127), C12(129), C13(129), C14(39), C15(39)
|
3 |
|
C1(119), C2(123), C3(142), C4(123), C5(119), C6(120), C7(178), C8(134), C9(146), C10(149), C11(126), C12(131), C13(131), C14(44)
|
4 |
|
C1(121), C2(127), C3(133), C4(127), C5(121), C6(138), C7(181), C8(54), C9(44), C10(44), C11(41), C12(39)
|
Scheme 2 shows the reaction of amine with substituted aldehyde to give Schiff base:
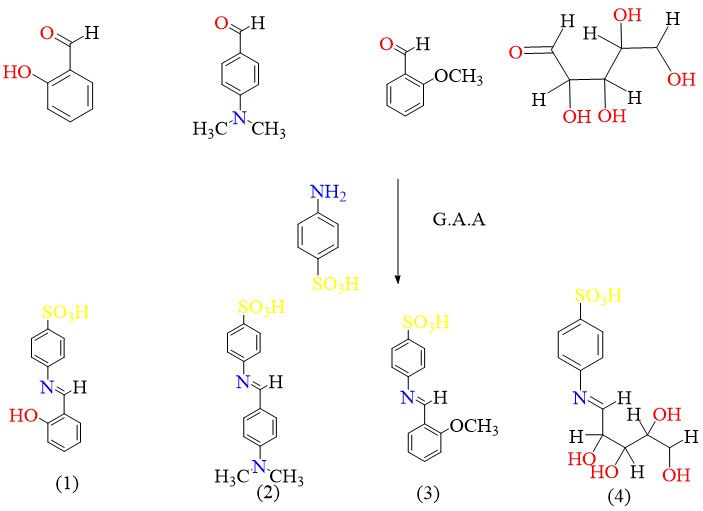
Scheme 2 Synthesis of Schiff base derivative.
Thiazolidinenes were prepared from the condensation of Schiff bases with thiocyclic acid using anhydrous zinc chloride as a catalyst in absolute ethanol and according to the following mechanics in Scheme 3 [34]:
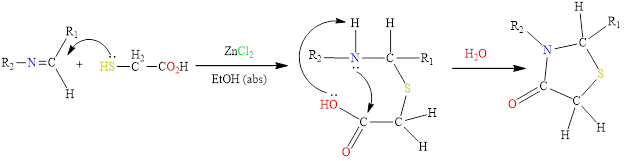
Scheme 3 Mechanism of cyclization of thiazolidine.
The mechanics, we note that the nucleophile attack of the nitrogen atom on the carboxyl carbonyl group occurs with the contribution of the cofactor ZnCl2, which works to hold the (OH) group in the carboxylate and then the loss of a water molecule to give the final product.
The thiozolidine-4-one compounds (5-8) were identified using the infrared spectrum, as the spectrum showed bands at 1,719 - 1,674 cm-1 that belong to the carbonyl group (C=O) in the thiozolidin-4-one, and the spectrum showed bands at (1,187 - 1,005).) cm-1 belongs to the stretch of (C-S-C) group. As for the absorption spectrum of the aromatic double bond (C=C), it appeared at the range (1,600 - 1,437) cm-1, as shown in the Table 8.
Table 8 The (IR) spectral properties of thiazolidine compounds (5 - 8).
Comp. No. |
Y |
C=C Aromatic cm-1 |
C−H Aromatic cm-1 |
C−H Aliphatic cm-1 |
Amide C=O cm-1 |
C−S−C cm-1 |
S=O, S−O |
Others cm-1 |
5 |
-OH |
1,600 - 1,498 |
3,062 |
2,876 |
1,719 |
1,111, 1,006 |
1,630 1,296 |
OH(3178) |
6 |
-N(CH3) |
1,595 - 1,437 |
3,036 |
2,895 |
1,709 |
1,163, 1,067 |
1,622 1,284 |
- |
7 |
-OCH3 |
1,600 -1,474 |
3,034 |
2,880 |
1,690 |
1,134, 1,076 |
1,646 1,274 |
C-O-C (1205,1034) |
8 |
-C4H9O4 |
1,600 - 1,443 |
3,063 |
2,917, 2,865 |
1,674 |
1,187, 1,005 |
1,634 1,280 |
OH(3340) C-O(1054,1155) |
Table 9 The (1H-NMR) spectral properties of thiazolidine compounds (5 - 8).
No. Comp. |
1HNMR (δ, ppm) |
9 |
2.527(s,H,CH-N); 3.922(s,2H,CH2-thiazol); 7.581 (t,H,C2 phenol ring);7.597-7.700 (d:d,4H,sulfanilic acid ring); 7.922,8.035,8.035(m,3H,(C3,4,6)phenol ring); 8.306 (s,H, OH phenol); 10.449 (s,H,OH sulfonic acid). |
10 |
2.201(s,H,CH-N); 3.105(s,2H,CH2-thiazol); 3.212(s,6H, di CH3); 7.097-7.314 (d,4H,N,N-dimethyl aniline ring); 7.635-7.720 (d:d,4H,sulfanilic acid ring); 9.112 (s,H,OH sulfonic acid). |
11 |
2.090(s,H,CH-N); 2.507(s,2H,CH2-thiazol); 3.570(s,3H,OCH3); 7.156-7.161 (d,2H,(C3,4)anisole ring); 7.176 (d,H,C5 anisole ring); 7.627 (t,H,C2 anisole ring); 7.647-7.669 (d:,4H,sulfanilic acid ring); 9.168 (s,H,OH sulfonic acid). |
12 |
2.505(s,H,CH-N); 3.333(s,2H,CH2-thiazol); 3.441,4.012 (d,2H,C1(H+OH)); 3.587,4.019 (q,1H,C2(H+OH)); 3.685,4.107 (t,1H,C3(H+OH)); 3.999,4.125(t,1H,C4(H+OH)); 4.143,5.604 (t,1H,C5(H+OH)); 6.945-7.693 (d:d,4H,sulfanilic acid ring); 8.440 (s,H,OH sulfonic acid). |
Table 10 The (C-13 NMR) spectral properties of thiazolidine compounds (5 - 8).
No. Comp. |
Structure compounds |
13C-NMR (δ, ppm) |
5 |
|
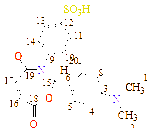
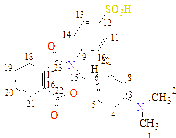
C1(118), C2(148), C3(121), C4(121), C5(123), C6(126), C7(127), C8(128), C9(140), C10(128), C11(127), C12(148), C13(169), C14(162), C15(189)
|
6 |
|
C1(56), C2(56), C3(113), C4(118), C5(121), C6(123), C7(121), C8(118), C9(132), C10(127), C11(128), C12(148), C13(128), C14(127), C15(166), C16(158), C17(187) |
7 |
|
C1(111), C2(119), C3(118), C4(119), C5(121), C6(124), C7(63), C8(127), C9(129), C10(149), C11(129), C12(127), C13(133), C14(169), C15(149), C16(190) |
8 |
|
C1(32), C2(34), C3(46), C4(56), C5(61), C6(111), C7(120), C8(133)), C9(122), C10(127), C11(147), C12(170), C13(156), C14(195) |
These compounds were prepared through the reaction of stoichiometric moles of Schiff bases with maleic anhydride and thermal sublimation for 4 h in the presence of dry benzene as a solvent, and the reaction takes place according to the following mechanism in Scheme 4 [35]:
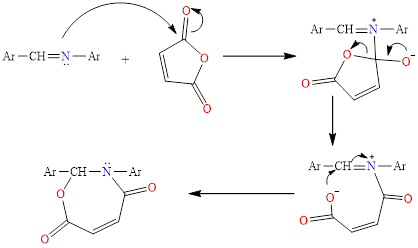
Scheme 4 Mechanism of cyclization of oxazepane.
Compounds (9 - 12) were identified by infrared spectrum, where it was found that the (C=O) group in the lactone appears at the range (1,719 - 1,651) cm-1, while the carbonyl group (C=O) in the lactam showed firmness within the range (1,683 - 1,641) cm-1, while bands appeared within the range (1,609 - 1,467) cm-1 due to the stretching of the double bond (C=C) of the benzene ring, as shown in Table 11.
Table 11 The (IR) spectral properties of oxazepine compounds (9 - 12).
Comp. No. |
Y |
C=C Aromatic cm-1 |
C−H Aromatic cm-1 |
C−H Aliphatic cm-1 |
C=O cm-1 Ester, Amid |
C=C cm-1 |
S=O, S-O |
Others cm-1 |
9 |
-OH |
1,588 - 1,467 |
3,051 |
2,990, 2,851 |
1,651, 1,644 |
1,633 |
1,622 1,274 |
OH(3397) |
10 |
-N(CH3) |
1,593 -1,490 |
3,057 |
2,960, 2,850 |
1,719, 1,641 |
1,622 |
1,606 |
- |
11 |
-OCH3 |
1,609 - 1,485 |
3,098 |
2,981, 2,880 |
1,716, 1,683 |
1,653 |
1,637 |
C-O-C (1263,1032) |
12 |
-C4H9O4 |
1,600 - 1,474 |
3,063 |
2,965, 2,874 |
1,705, 1,666 |
1,647 |
1,628 |
OH(3410) C-O(1060,1156) |
Table 12 shows the (1H-NMR) spectral properties of oxazepine compounds (9 - 12).
No. Comp. |
1HNMR (δ, ppm) |
5 |
2.510(s,H,CH-N); 3.972(d, H,=CH,);4.095(d,H,=CH); 7.061 (t,H,C2 phenol ring);7.221-7.323 (d:d,4H,sulfanilic acid ring); 7.554,7.583,7.607(m,3H,(C3,4,6)phenol ring); 9.088 (s,H, OH phenol); 10.368 (s,H,OH sulfonic acid). |
6 |
2.509(s,H,CH-N); 3.229(d, H,=CH,);3.507(d,H,=CH); 3.16(s,6H, di CH3); 6.782-7.000 (d,4H,N,Ndimethyl aniline ring); 7.533-7.620 (d:d,4H,sulfanilic acid ring); 9.667 (s,H,OH sulfonic acid). |
7 |
2.507(s,H,CH-N); 2.672(d, H,=CH,); 3.047(d,H,=CH); 3.699(s,3H,OCH3); 6.781-7.021 (d,2H,(C3,4)anisole ring); 7.574 (d,H,C5 anisole ring); 7.596 (t,H,C2 anisole ring); 7.627-7.762 (d:,4H,sulfanilic acid ring); 9.668 (s,H,OH sulfonic acid). |
8 |
2.505(s,H,CH-N); 3.315(d, H,=CH,);3.428(d,H,=CH); 3.441,4.012 (d,2H,C1(H+OH)); 3.587,4.019 (q,1H,C2(H+OH)); 3.685,4.107 (t,1H,C3(H+OH)); 3.999,4.125(t,1H,C4(H+OH)); 4.143,5.604 (t,1H,C5(H+OH)); 6.945-7.693 (d:d,4H,sulfanilic acid ring); 8.440 (s,H,OH sulfonic acid). |
Table 13 The (C-13 NMR) spectral properties of oxazepine compounds (9 - 12).
No. Comp. |
Structure compounds |
13C-NMR (δ, ppm) |
9 |
|
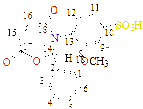
C1(113), C2(133), C3(118), C4(121), C5(121), C6(125), C7(121), C8(125), C9(131), C10(126), C11(128), C12(131), C13(56), C14(163), C15(167), C16(173), C17(189)
|
10 |
|
C1(34), C2(34), C3(117), C4(123), C5(126), C6(128), C7(123), C8(126), C9(131), C10(126), C11(123), C12(134), C13(123), C14(126), C15(52), C16(163), C17(169), C18(179), C19(187) |
11 |
|
C1(137), C2(131), C3(117), C4(124), C5(124), C6(124), C7(45), C8(126), C9(126), C10(132), C11(127), C12(127), C13(133), C14(54), C15(163), C16(165), C17(179), C18(183) |
12 |
|
C1(37), C2(33), C3(34), C4(33), C5(42), C6(121), C7(121), C8(133), C9(123), C10(121), C11(131), C12(55), C13(161), C14(163), C15(171), C16(179) |
These compounds were prepared through the reaction of stoichiometric moles of Schiff bases with phthalic anhydride and thermal sublimation for 4 h in the presence of dry benzene as a solvent. The reaction takes place with the same previous mechanism . Compounds (13 - 16) were identified by infrared spectrum, where it was found that the (C=O) group in the lactone appears at the range (1,735 - 1,690) cm-1, while the carbonyl group (C=O) in the lactam showed firmness within the range (1,650 - 1,644) cm-1, while bands appeared within the range (1,600 - 1,440) cm-1 due to the stretching of the double bond (C=C) of the benzene ring, the IR spectra show in the Table 14.
Table 14 The (IR) spectral properties of benzoxazepine compounds (13 - 16).
Comp. No. |
Y |
C=C Aromatic cm-1 |
C−H Aromatic cm-1 |
C−H Aliphatic cm-1 |
C=O ester, amid cm-1 |
S=O, S-O |
Others cm-1 |
13 |
-OH |
1,578 - 1,467 |
3,061 |
2,981, 2,938 |
1,735, 1,648 |
1,611 1,264 |
OH(3211) |
14 |
-N(CH3) |
1,593 - 1,492 |
3,050 |
2,933 |
1,722, 1,650 |
1,605 1,279 |
- |
15 |
-OCH3 |
1,591 - 1,470 |
3,063 |
2,962 |
1,700, 1,644 |
1,604 1,276 |
C-O-C (1199,1034) |
16 |
-C4H9O4 |
1,600 - 1,440 |
3,054 |
2,980, 2,870 |
1,690, 1,648 |
1,612 1,266 |
OH(3401) C-O(1054,1150) |
Table 15 The (1H-NMR) spectral properties of benzoxazepine compounds (13 - 16).
No. Comp. |
1HNMR (δ, ppm) |
13 |
2.504(s,H,CH-N); 7.389 (t,H,C2 phenol ring);6.281(s,4H,benzene ring); 7.690-7.979 (d:d,4H,sulfanilic acid ring); 8.072,8.286,8.879(m,3H,(C3,4,6)phenol ring); 9.485 (s,H, OH phenol); 10.221 (s,H,OH sulfonic acid). |
14 |
2.501(s,H,CH-N); 3.016(s,6H, di CH3); 7.332-7.445(m,4H, benzene ring); 7.720-7.890 (d,4H,N,Ndimethyl aniline ring); 8.133-8.620 (d:d,4H,sulfanilic acid ring); 9.067 (s,H,OH sulfonic acid). |
15 |
2.809(s,H,CH-N); 3.909(s,3H,OCH3); 7.433(s,4H, benzene ring); 7.120-7.612 (d.d,2H,(C3,4)anisole ring); 7.799 (d,H,C5 anisole ring); 7.960 (t,H,C2 anisole ring); 7.670-8.162 (d:,4H,sulfanilic acid ring); 9.968 (s,H,OH sulfonic acid). |
16 |
2.506(s,H,CH-N); 3.516,3.952 (d,2H,C1(H+OH)); 3.726,4.089 (q,1H,C2(H+OH)); 3.743,4.132 (t,1H,C3(H+OH)); 3.768,4.132(t,1H,C4(H+OH)); 3.910,5.104 (t,1H,C5(H+OH)); 7.078- 7.307(m,4H, benzene ring); 7.481-7.068 (d:d,4H,sulfanilic acid ring); 10.368 (s,H,OH sulfonic acid). |
Table 16 The (C-13 NMR) spectral properties of benzoxazepine compounds (13 - 16).
No. Comp. |
Structure compounds |
13C-NMR (δ, ppm) |
13 |
|
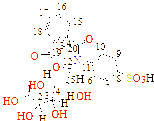
C1(122), C2(131), C3(122), C4(121), C5(126), C6(129), C7(132), C8(122), C9(122), C10(137), C11(122), C12(121), C13(40), C14(122), C15(125), C16(122), C17(123), C18(122), C19(126), C20(167), C21(178)
|
14 |
|
C1(27), C2(27), C3(127), C4(122), C5(127), C6(129), C7(127), C8(122), C9(131), C10(122), C11(126), C12(133), C13(126), C14(122), C15(55), C16(129), C17(129), C18(122), C19(122), C20(126), C21(128), C22(178), C23(187) |
15 |
|
C1(137), C2(131), C3(124), C4(118), C5(119), C6(124), C7(58), C8(123), C9(122), C10(135), C11(123), C12(122), C13(129), C14(47), C15(122), C16(127), C17(120), C18(121), C19(121), C20(120), C21(171), C22(179) |
16 |
|
C1(56), C2(40), C3(41), C4(40), C5(56), C6(112), C7(120), C8(136), C9(113), C10(121), C11(129), C12(56), C13(124), C14(126), C15(128), C16(127), C17(131), C18(132), C19(167), C20(189) |
Scheme 5 shows the reaction of Schiff base with thioglycolic acid, malic acid & phthalic acid to give
thiazolidine, oxazepine & benzoxazepine, respectively.
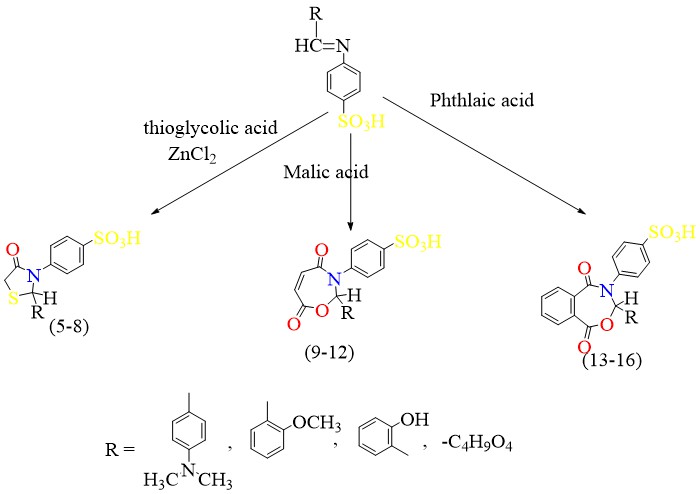
Scheme 5 Synthesis of thiazolidine compounds (5-8) oxazepine (9-12) benzooxazepine(13-16).
Docking results and conclusions
The molecular docking simulations of the five tested compounds revealed distinct interaction profiles with the target protein.
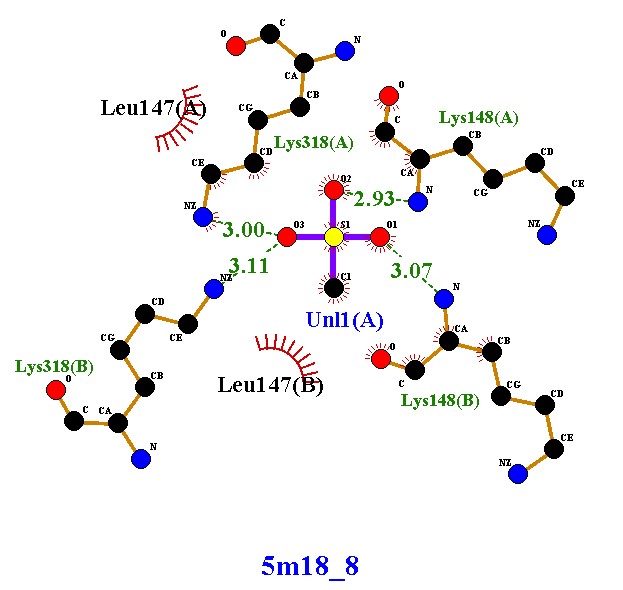
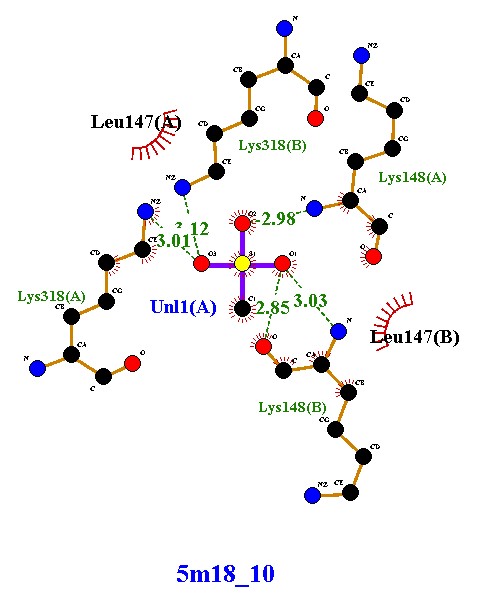
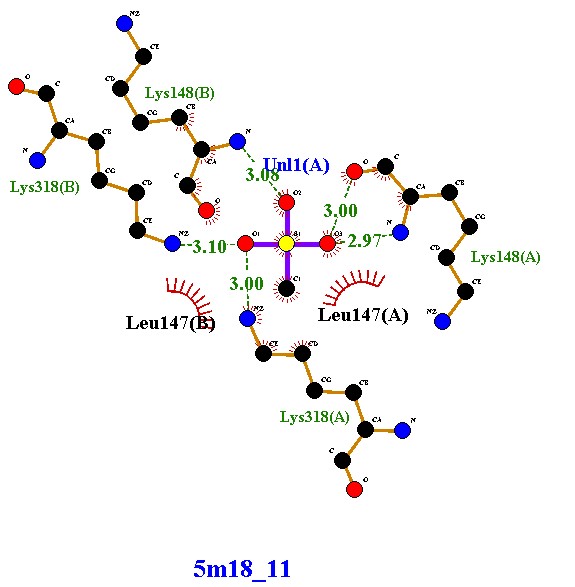
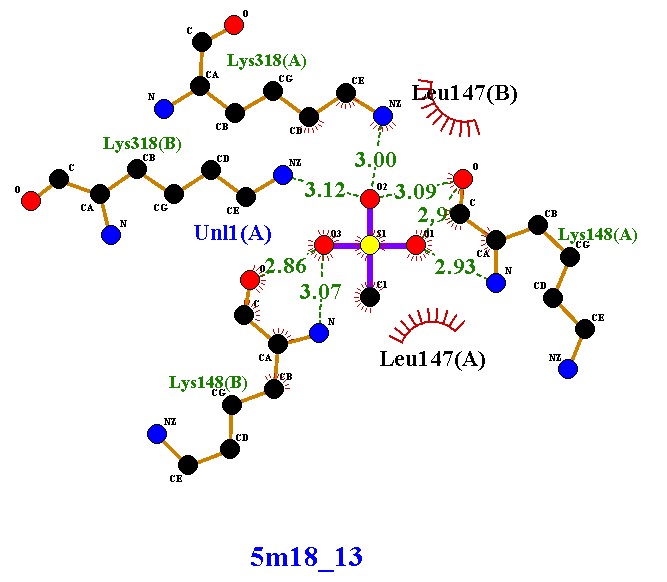
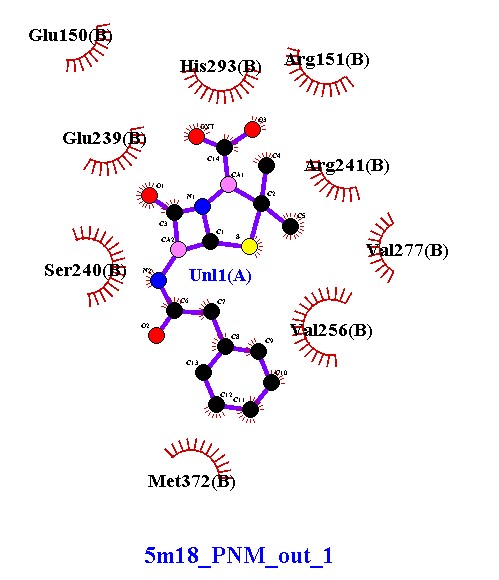
Compound 5m18_8 established four hydrogen bonds with Lys318 and Lys148 (chains A and B), along with hydrophobic contacts with Leu147. These interactions indicate moderate stability, with dual-chain engagement supporting its inhibitory potential. Compound 5m18_10 demonstrated an improved binding profile, forming five hydrogen bonds with lysine residues and Uni1(A), in addition to hydrophobic stabilization by Leu147. The extra hydrogen bond, compared to 5m18_8, suggests enhanced affinity and tighter binding. Compound 5m18_11 also formed five hydrogen bonds with a symmetrical distribution across both chains, coupled with hydrophobic interactions. This balanced binding pattern indicates robust and consistent stabilization, positioning it as a strong inhibitory candidate. Compound 5m18_13 displayed the highest binding stability, with six hydrogen bonds, several at optimal short distances (2.86–2.93 Å), and additional hydrophobic contacts. This ligand exhibited the strongest affinity among the tested molecules, making it the most promising inhibitor in this series. Compound 5m18_PNM_out_1, in contrast, did not form classical hydrogen bonds. Instead, it relied on extensive hydrophobic and electrostatic interactions with residues such as Glu150, Glu239, Arg241, Val277, Ile256, Met372, and His293. While this indicates a weaker hydrogen-bonding profile, the compound remains stabilized through non-polar contacts, suggesting a complementary binding mode. In summary, compounds 5m18_10, 5m18_11, and 5m18_13 demonstrated strong hydrogen bonding and hydrophobic stabilization, with 5m18_13 emerging as the lead candidate due to its superior binding affinity and interaction profile. Compound 5m18_PNM_out_1 may still offer value through an alternative hydrophobic binding mechanism, while 5m18_8 exhibited moderate inhibitory potential. Collectively, these findings highlight 5m18_13 as the most promising scaffold for further optimization, followed by 5m18_11 and 5m18_10
Table 17 The results and discussion of molecular docking of compounds (8-16).
Comp. No. |
Docking structure |
Results |
Dissociation |
8 |
|
Forms four hydrogen bonds with Lys318(A), Lys318(B), Lys148(A), and Lys148(B), at distances of 2.93–3.11 Å (optimal range for stability). Establishes hydrophobic interactions with Leu147(A) and Leu147(B). The binding involves both protein chains (A and B), suggesting stronger stabilization |
Compound 5m18_8 shows strong and stable binding within the active site. The multiple hydrogen bonds with lysine residues, combined with hydrophobic contacts, indicate good affinity and stability. Its ability to interact with both chains highlights its potential inhibitory effect on the protein by stabilizing the dimer structure. |
10 |
|
Forms five hydrogen bonds with Lys318(A), Lys318(B), Lys148(A), Lys148(B), and Uni1(A), with bond lengths ranging 2.85 - 3.12 Å (within the optimal range). Shows hydrophobic interactions with Leu147(A) and Leu147(B), contributing to additional stability. Interactions involve both protein chains (A and B), similar to compound 5m18_8, but here the ligand forms |
Compound 5m18_10 demonstrates enhanced binding affinity compared to 5m18_8, due to the presence of an extra hydrogen bond. The shorter bond distances (~2.85 - 2.98 Å) further suggest tighter binding. Combined with hydrophobic interactions, these features indicate that 5m18_10 could be a more potent inhibitor, effectively stabilizing the dimer interface and strengthening its therapeutic potential. |
|
|
more H-bonds, indicating stronger stabilization. |
|
11 |
|
Forms five hydrogen bonds with Lys318(A), Lys318(B), Lys148(A), and Uni1(A), with bond distances ranging 2.97–3.10 Å (stable and within optimal range). Exhibits hydrophobic interactions with Leu147(A) and Leu147(B), reinforcing the complex stability. Notably, this compound shows a balanced hydrogen bonding pattern across both chains, ensuring symmetrical stabilization of the protein interface. |
Compound 5m18_11 presents a robust binding profile, comparable to compound 5m18_10. The hydrogen bonds, coupled with hydrophobic contacts, indicate strong and consistent stabilization within the binding pocket. Its symmetrical engagement with residues from both chains suggests a reliable inhibitory effect, potentially preventing conformational flexibility of the protein. This makes 5m18_11 a competitive candidate with strong therapeutic promise.
|
13 |
|
Forms six hydrogen bonds with Lys318(A), Lys318(B), Lys148(A), Lys148(B), and Uni1(A), with bond distances ranging 2.86–3.12 Å. Exhibits hydrophobic interactions with Leu147(A) and Leu147(B). This compound shows the highest number of hydrogen bonds among the tested ligands, with several at short distances (2.86–2.93 Å), suggesting very strong stabilization. |
Compound 5m18_13 demonstrates the strongest binding profile compared to the other ligands (5m18_8, 5m18_10, and 5m18_11). The presence of six hydrogen bonds, particularly with optimal bond lengths (<3.0 Å), indicates high affinity and strong stability within the active site. Together with hydrophobic contacts, these interactions highlight 5m18_13 as the most promising candidate, potentially offering superior inhibitory activity through robust multi-residue and dualchain engagement. |
16 |
|
Unlike the previous ligands, this compound shows no classical hydrogen bonds with active site residues. The main stabilization is achieved through hydrophobic interactions with multiple residues: Glu150(B), Glu239(B), Ser240(B), Arg241(B), |
Compound 5m18_PNM_out_1 exhibits a different binding mode compared to the other ligands. While it lacks hydrogen bonding, it establishes extensive hydrophobic and electrostatic interactions with several residues, forming a stable non-polar pocket. This suggests that its binding affinity may be weaker than compounds 5m18_10, 5m18_11, and |
|
|
Val277(B), Ile256(B), Met372(B), and His293(B). Binding is mainly governed by hydrophobic and electrostatic contacts, not by strong hydrogen bonding. |
5m18_13, which rely on multiple hydrogen bonds. However, such a binding pattern may still provide stability through van der Waals and hydrophobic packing, indicating a potential complementary inhibitory effect. |
|
|||
Biological activity
We note from the table regarding the values of the biological activity results for compounds 8, 10, 11 and 13 that they give good results. At a concentration of 25 mg, they gave weak results, but at a higher concentration of 100 mg, the results were excellent for the four compounds that were tested with four types of bacteria except for Staphylococcus bacteria. Which was not affected at all by any type of prepared materials.
The test results in the Table 18 showed that the prepared compounds are of great biological importance, as if toxicity tests are conducted on the compounds, the work can be completed to obtain good pharmaceutical compounds, as the sulfonyl group as well as the last active groups in the compounds make them easily soluble in water, which facilitates their absorption by the body. And converting it to blood as a therapeutic material.
Table 18 The results of the biological effective of the compounds (8,10,11,13).
Microorganisms |
8 |
10 |
11 |
13 |
||||||||||||
25 mg |
50 mg |
75 mg |
100 mg |
25 mg |
50 mg |
75 mg |
100 mg |
25 mg |
50 mg |
75 mg |
100 mg |
25 mg |
50 mg |
75 mg |
100 mg |
|
Staphlococcus aurens |
13.1 |
12.2 |
14.8 |
15 |
20.5 |
20.7 |
29 |
30.3 |
9.1 |
14 |
14.6 |
18.2 |
17.4 |
25.4 |
27.4 |
30.7 |
Streptococcus pneumoniae |
- |
- |
- |
- |
- |
- |
- |
- |
- |
- |
- |
- |
- |
- |
- |
- |
E. coli |
- |
- |
- |
13.6 |
- |
- |
- |
- |
- |
- |
- |
- |
- |
- |
10.6 |
11.2 |
Klebsiella spp. |
- |
- |
- |
- |
- |
- |
12.5 |
13.5 |
- |
- |
- |
11.3 |
- |
12.1 |
13.1 |
14.8 |
Canolida albicans |
12.7 |
13.4 |
13.5 |
17.3 |
R |
15.7 |
18.0 |
18.2 |
- |
- |
- |
- |
- |
- |
- |
- |
(-) = NO activity
Conclusions
In this work, new organic compounds were synthesized by converting the amine group in the paraaminosulfanilic compound into Schiff bases, and then cyclizing the result using several factors to obtain penta- and heptacyclic heterocyclic compounds. Compounds containing a sulfone group with heterogeneous rings are effective against Gram-positive and Gramnegative bacteria, as sulfur compounds are among the compounds that are used as excellent therapeutic agents in many medical drugs. The biological effectiveness of compounds 8, 10, 11, and 13 has been shown to be superior to many types of antibacterials at four different concentrations, and they were effective in eliminating bacterial colonies in them.
Declaration of Generative AI in Scientific Writing
The authors acknowledge the use of generative AI tools (ChatGPT by OpenAI) in the preparation of this manuscript, specifically for language editing and grammar correction. No content generation or data interpretation was performed by AI. The authors take full responsibility for the content and conclusions of this work.
CRediT Author Statement
Zainab Faiyq Saeed: Data curation, Formal analysis, Investigation, Validation, and Visualization. Ghufran Thanoon. Sadeek: Methodology, Project administration, Resources, Supervision, Validation, and Writing –original draft. Rafid Kamal Jameel: Methodology, Supervision, Validation, Funding acquisition, and Writing –original draft.
Acknowledgments
The authors express their gratitude to Al-Noor University for its financial support of this manuscript under project number (ANUI/2025/SCI20). The researchers also thank the University of Mosul for its support in providing raw materials and laboratories for the practical component.
References
[1] KTA Al-Sultani. Synthesis, identification and evaluation the biological activity for some new heterocyclic compounds derived from schiff bases. IOSR Journal of Pharmacy and Biological Sciences 2017; 12(2), 39-47.
[2] AM Asiri, SA Khan, HM Marwani and K Sharma. Synthesis, spectroscopic and physicochemical investigations of environmentally benign heterocyclic Schiff base derivatives as antibacterial agents on the bases of in vitro and density functional theory. Journal of Photochemistry and Photobiology B: Biology 2013; 120, 82-89.
[3] MA Ibrahim and KM El-Mahdy. Synthesis and Antimicrobial activity of some new heterocyclic Schiff bases derived from 2-amino-3-formylchromone. Phosphorus, Sulfur, and Silicon and the Related Elements 2009; 184(11), 2945-2958.
[4] GT Sadeek, ZF Saeed and MY Saleh. Synthesis and pharmacological profile of hydrazide compounds. Research Journal of Pharmacy and Technology 2023; 16(2), 975-982.
[5] AM Alafeefy, MA Bakht, MA Ganaie, MN Ansarie, NN El-Sayed and AS Awaad. Synthesis, analgesic, anti-inflammatory and anti-ulcerogenic activities of certain novel Schiff’s bases as fenamate isosteres. Bioorganic & medicinal chemistry letters 2015; 25(2), 179-183.
[6] HS Abood, U Ramadhan and H Hamza. Synthesis and anti-inflammatory activity study of Schiff bases complexes. Biochemical and Cellular Archives 2020; 20(2), 5627-5631.
[7] M Jesmin, MK Islam and SMM Ali. Analgesic and anti-inflammatory activities of some transition metal schiff base complexes. International Letters of Chemistry, Physics and Astronomy 2014; 27, 64-72.
[8] HMY Al-Labban, HM Sadiq and AAJ Aljanaby. Synthesis, Characterization and study biological activity of some Schiff bases derivatives from 4-amino antipyrine as a starting material. In: Proceedings of the 249th ECS Meeting Seattle Convention Center – Arch and Sheraton Grand Seattle, Seattle, WA. 2025, 1294(5), p. 052007
[9] OY AL-Abbasy, WI Ali, AI Rashan and SA AL-Bajari. Purification, characterization, and inhibition of tyrosinase from jerusalem artichoke (Helianthus tuberosus L.) tuber. Reports of Biochemistry & Molecular Biology 2021; 10(3), 495-505.
[10] AI Rashan, RT Altaee, FS Salh, OY Al-abbasy and N Al-Lehebe. The role of polyamines in plants: A review. Plant Science Today 2023; 10(2), 164-171.
[11] AA Sultan, MY Saleh and EFA Alkhalidi. Nano-Hydroxyapatite remineralization of in-situ induced enamel caries. Journal of Nanostructures 2022; 12(4), 1067-1074.
[12] HMS Al-Jubori, TSF Al-Mathkuri, ZR Banoon and MY Saleh. Design, synthesis and molecular docking study of novel quinolinethioacetohydrazides bearing α-aminophosphonate and their antimicrobial evaluation. Results in Chemistry 2024; 8, 101586.
[13] MY Saleh, AKO Aldulaimi, SM Saeed and AH Adhab. TiFe2O4@ SiO2–SO3H: A novel and effective catalyst for esterification reaction. Heliyon 2024; 10(4), 26286.
[14] NT Al-Thakafy, MA Abdelzaher, HG Abdelzaher, MY Saleh, MS Al-Enizzi, SM Saied, M Elbagory, S El-Nahrawy, A El-Dein Omara, M Al-Shalawi and FS Moghanm. A novel chalcone compound as a reagent for the validation of pharmaceutical cefotaxime sodium preparations. Results in Chemistry 2024; 7, 101387.
[15] MY Saleh, GT Sadeek and SM Saied. Preparation and characterization of a dual acidic ionic liquid functionalized graphene oxide nanosheets as a heterogeneous catalyst for the synthesis of pyrimido [4, 5-b] quinolines in water. Iranian Journal of Catalysis 2023; 13(4), 499-516.
[16] MY Saleh, MA Abdelzaher, AH Ali and KA Owaid. Synthesis and characterization of some new heterocyclic polymer compounds from benzo [1, 2-d: 4, 5-d] bis (thiazole)-2, 6-diamine and [6, 6'-Bibenzo [d] thiazole]-2, 2'-diamine. Rafidain Journal of Science 2023; 32(4), 57-69.
[17] A Ayoob, OM Yahya and MY Saleh. Synthesis and biological activity of 2-chloro-3-formyl-1, 8naphthyridine chalcone derivative. Egyptian Journal of Chemistry 2022; 65(13), 549-555.
[18] RMH Al-Sultan, A Ammar, Al-Sultan, MA Hayawi, JM Bilal, B Aldahham, MY Saleh, H Mohammed. The effect of subclinical thyroid dysfunction on B-type natriuretic peptide level. Revista Bionatura 2022; 7(2), 99-108.
[19] SM Saied, SJ Mohammed, BT Khaleel and MY Saleh. Comparative studies between conventional techniques and green chemistry to synthesis of novel piperidinium salts ionic liquids (PBSILs). Journal of Chemical Health Risks 2021; 11(4), 451-456.
[20] AA Majhool, MY Saleh, AKO Aldulaimi, SM Saeed, SM Hassan, MF El-Shehry, SM Awad and SSSA Azziz. Synthesis of new azo dyes of uracil via ecofriendly method and evaluation for the breast, liver and lung cancer cells in vitro. Chemical Review and Letters 2023; 6(4), 442-448.
[21] MY Saleh, AKO Aldulaimi, SM Saeed and AH Adhab. Palladium fabricated on Fe3O4 as an organic-inorganic hybrid nanocatalyst for the Suzuki and Stille coupling reactions. Journal of Molecular Structure 2025; 1321, 139597.
[22] AH Ali, MY Saleh and KA Owaid. Mild synthesis, characterization, and application of some polythioester polymers catalyzed by cetrimide ionic liquid as a green and eco-friendly phase-transfer catalyst. Iranian Journal of Catalysis 2023; 13(1), 73-83.
[23] ER Mohammed, SM Saied and MY Saleh. Synthesis, characterization and biological evaluation study of cephalexin (Ceph) and Isatin Schiff base and its complexation with some divalent metal ions. Egyptian Journal of Chemistry 2022; 65(7), 595-603.
[24] SM Saied, MY Saleh and AM Hamdoon. Multicomponent synthesis of tetrahydrobenzo [a] xanthene and tetrahydrobenzo [a] acridine derivatives using sulfonated multi-walled carbon nanotubes as heterogeneous nanocatalysts. Iranian Journal of Catalysis 2022; 12(2), 189-205.
[25] SA Raoof, FJ Ahmed, ASMO Al-barwarib and MY Saleh. Synthesis, characterization, and biological activity of chromium complexes as efficient and novel catalysts for direct synthesis of carbonyl compounds from benzyl/cycloalkyl bromides in water under aerobic oxidation. Iranian Journal of Catalysis 2022; 12(1), 55-68.
[26] V Kuete, KO Eyong, GN Folefoc, VP Beng, H Hussain, K Krohn and AE Nkengfack. Antimicrobial activity of the methanolic extract and of the chemical constituents isolated from Newbouldia laevis. Die Pharmazie-An International Journal of Pharmaceutical Sciences 2007; 62(7), 552-556.
[27] N Pfoze, Y Kumar, B Myrboh, DRK Bhagobaty and SR Joshi. In vitro antibacterial activity of alkaloid extract from stem bark of Mahonia manipurensis Takeda. Journal of Medicinal Plants Research 2011; 5(5), 859-861.
[28] ZF Saeed, MY Saleh and GT Sadeek. Synthesis and biological evolution of novel substituted 1, 2, 4-triazine from sulfanilic acid. Egyptian Journal of Chemistry 2023; 66(1), 555-561.
[29] A Saleh and MY Saleh. Synthesis of heterocyclic compounds by cyclization of Schiff bases prepared from capric acid hydrazide and study of biological activity. Egyptian Journal of Chemistry 2022; 65(12), 783-792.
[30] AM Hamdoon, MY Saleh and SM Saied. Synthesis & biological evaluation of novel series of benzo [f] indazole derivatives. Egyptian Journal of Chemistry 2022; 65(11), 305-312.
[31] MY Saleh, ASMO Al-barwari and AI Ayoob. Synthesis of some Novel 1, 8-Naphthyridine chalcones as antibacterial agents. Journal of Nanostructures 2022; 12(3), 598-606.
[32] M Faisal, RF Al-Smaism and ZM Abd Al-Khaliq. Synthesis, characterization and antibacterial activity of new series of sulfamethoxazole derivatives. World Journal of Pharmacy and Pharmaceutical Sciences 2015; 4(10), 284-293.
[33] NK Chaudhary. Synthesis and medicinal use of Metal complexes of Schiff Bases. Bibechana 2013; 9, 75-80.
[34] ZY Kadhim, HGJ Alqaraghuli and MT Abd. Synthesis, characterization, molecular docking, in vitro biological evaluation and in vitro cytotoxicity study of novel Thiazolidine-4-One derivatives as anti-breast cancer agents. Anti-Cancer Agents in Medicinal Chemistry-Anti-Cancer Agents) 2021; 21(17), 2379-2406.
[35] WK Jassim. Preparation and characterization of some new heterocyclic compounds with evaluating of its biological activity. Karbala Journal of Pharmaceutical Sciences 2011; 2, 241-256.