
Trends
Sci.
2026;
23(5):
12196
Comparative Efficacy of Endogenous Versus Exogenous Antioxidants in Small Ruminants Sperm Cryopreservation
Yudit
Oktanella1,2,*,
Imam Mustofa3,
![]() ,
Suherni Susilowati3,
,
Suherni Susilowati3,
![]() ,
Widjiati Widjiati4,
,
Widjiati Widjiati4,
![]() ,
,
Bambang
Purwantara5,
![]() ,
Adeyinka Oye Akintunde6,
,
Adeyinka Oye Akintunde6,
![]() ,
Tatik Hernawati3,
,
Tatik Hernawati3,
![]() ,
,
Tri
Wahyu Suprayogi3,
![]() ,
Suzanita Utama3,
,
Suzanita Utama3,
![]() and Eka Pramyrtha Hestianah7,
and Eka Pramyrtha Hestianah7,
![]()
1Doctorate Program of Sains Veterinary, Faculty of Veterinary Medicine, Universitas Airlangga,
East Java 60115, Indonesia
2Department of Veterinary Reproduction, Faculty of Veterinary Medicine, Universitas Brawijaya,
East Java 65151, Indonesia
3Division of Veterinary Reproduction, Faculty of Veterinary Medicine, Universitas Airlangga,
East Java 60115, Indonesia
4Division of Veterinary Embriology, Faculty of Veterinary Medicine, Universitas Airlangga,
East Java 60115, Indonesia
5Division of Reproduction and Obstetrics, School of Veterinary Medicine and Biomedical Sciences,
IPB University, West Java 16680, Indonesia
6Department of Agriculture and Industrial Technology, Babcock University, Ogun State Postcode 121003, Nigeria
7Department of Basic Veterinary Medicine, Faculty of Veterinary Medicine, Universitas Airlangga,
East Java 60115, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 7 October 2025, Revised: 24 October 2025, Accepted: 5 November 2025, Published: 5 January 2026
Abstract
Small ruminant semen is highly susceptible to freeze-thaw oxidative damage due to PUFA-rich membranes, making antioxidant defense central to cryopreservation outcomes. Following PRISMA guidance, we searched Scopus, PubMed, and Google Scholar (2015 - 2025). Of 250 records screened, 25 studies met inclusion criteria. Effects were pooled as Hedges’ g under random-effects models (primary analyses in OpenMEE, DerSimonian-Laird; sensitivity verified with REML). Percentage outcomes (motility, viability) were arcsine-square-root transformed before SMD calculation. Subgroups contrasted endogenous vs exogenous antioxidants; heterogeneity (I²) and 95% CIs are reported. For motility, endogenous antioxidants showed a small decrease versus controls (SMD = –0.182; 95% CI –0.359 to –0.004; p = 0.045; I² = 12%), while exogenous antioxidants showed no clear effect (SMD = 0.025; 95% CI –0.144 to 0.194; p = 0.773; I² = 0%). The overall effect on motility was non-significant (SMD = –0.080; 95% CI –0.199 to 0.039; p = 0.186). For viability, both subgroups improved outcomes versus controls, with a larger pooled effect for exogenous (SMD = 5.150; 95% CI 2.810 to 7.490; I² = 59.94%) than endogenous (SMD = 4.105; 95% CI 0.100 to 8.109; I² = 92.14%); the overall pooled estimate was positive (SMD = 4.621; 95% CI 2.070 to 7.172). Sensitivity analyses (excluding higher-risk studies and using MD on the original percentage scale) did not materially change interpretations. The data suggest that endogenous antioxidants improve post-thaw viability but may not reliably sustain motility, whereas exogenous antioxidants may offer superior advantages to viability under particular circumstances. These results should be approached with caution due to significant variability in reporting and substantial differences in viability. Standardized methods, fixed dosages, and consistent outcome metrics are crucial to clarify the timing and recipients of external supplementation that improves beyond innate defenses.
Keywords: Antioxidant, Cryopreservation, Oxidative stress, Goat sperm, Zero hunger
Introduction
The development of innovative semen cryopreservation techniques for goats has become increasingly important in Indonesia, especially in line with the country’s commitment to achieving the United Nations Sustainable Development Goals (SDGs), notably SDG 2 (Zero Hunger) and SDG 1 (No Poverty). As an essential livestock species, goats contribute significantly to rural livelihoods and food security, providing meat, milk, and income to millions of smallholder farmers - an economic sector that supports over 30 million Indonesians [1]. However, the low productivity of goats, mainly due to poor genetic quality and reproductive inefficiencies, poses a significant challenge to improving national food security. Recent advances, such as the incorporation of antioxidants into cryopreservation protocols, offer new opportunities to improve post-thaw sperm viability and fertility rates. These improvements are in line with the objectives of Indonesia’s National Livestock Revitalization Program, which targets a 20% increase in livestock productivity by 2025 [2]. Better breeding outcomes through cryopreservation can help smallholder farmers to improve herd quality, increase income, and contribute directly to poverty reduction (SDG 1) while securing protein sources to fight malnutrition (SDG 2).
Cryopreservation of goat semen plays a crucial role in genetic improvement, species conservation, and the efficient application of artificial insemination (AI) programs. The cryopreservation or freezing processes exert severe oxidative stress on spermatozoa, mitochondria, and cell membranes, which can the integrity and functionality of these components. The key challenge is the lipid-rich membrane of small-ruminant spermatozoa, which is enriched in polyunsaturated fatty acids (PUFAs) [3,11]. Lipidomic studies show breed-dependent profiles: SFA ~40% - 50% and the remainder largely MUFA + PUFA (~50% - 60%). This higher unsaturation - especially PUFA enrichment - makes small-ruminant sperm highly prone to lipid peroxidation and oxidative injury during freeze-thaw [3]. Consequently, antioxidant supplementation must be carefully tailored for small-ruminant semen preservation. Notably, while these PUFAs support membrane structure and function, they also heighten membrane vulnerability during freezing and thawing [4]. Oxidative damage can weaken the membranes, impair motility, or cause fragmentation of DNA, all of which can reduce fertility.
To mitigate the effects of oxidative stress during the cryopreservation of goat semen, the use of extenders containing antioxidants has been explored. Some antioxidants like coenzyme Q10 [5,6], glutathione [7], and melatonin [8,9,10] are considered to be endogenous and have the alluring characteristic of being naturally synthesized within the sperm cell. The intrinsic biocompatibility of these antioxidants is advantageous because the likelihood of being harmful is minimized. Their main role is to bolster the sperm’s built-in antioxidant defenses, neutralize harmful reactive oxygen species (ROS), and protect mitochondrial and membrane integrity. At the molecular level, melatonin also regulates apoptotic signaling by upregulating anti-apoptotic proteins (e.g., BCL-2) and downregulating pro-apoptotic factors (e.g., BAX), thereby safeguarding spermatozoa from oxidative damage. That said, even these natural defenders have their limits, especially when faced with extreme oxidative stress [11,12].
On the other hand, external antioxidants, including some plant-based substances like Turraea fischeri extract [13], resveratrol [12], and proline [3], as well as novel nanoparticles such as ZnO or selenium [14,15], are more effective and flexible in protecting against oxidative damage. These factors can be tailored in concentration and composition to suit different qualities of semen and freezing methods. Their applicability, however, has disadvantages. Inevitably, high doses may prove to be cytotoxic, and lack of preparation or purity can lead to inconsistencies with unpredictable outcomes. These external antioxidants may be more effective in treating lipid peroxidation and ROS buildup, but the balance of these antioxidants to protect against oxidative damage while preserving sperm cell viability is still a critical challenge [16,13,17]. With the choice of endogenous versus exogenous antioxidants, considerations of the protective capability alongside endurance of spermatozoa and compatibility with extenders for recovery post-thaw should be prioritized.
Despite all the advances in technology, scientists are still debating which antioxidant works best. Butylated hydroxytoluene (BHT), although synthetic, has been shown to substantially reduce lipid peroxidation at the cellular level [18,19]. However, the potential for cellular toxicity remains to be evaluated. On the natural side, oils such as Nigella sativa [20], green tea [21], and whey protein via Nrf2 activation [22] have shown clear advantages, especially in the recovery of post-thaw motility and cryo-survival in ovine and caprine sperm. Also, the recent introduction of selenium-loaded berberine nanoparticles [23], cysteine supplementation [24], mitochondria-targeted antioxidants such as Mito-TEMPO [25], and metabolic regulators such as sodium salicylate [4] demonstrate the promising potential of cryopreservation without cryoinjury risk.
This review looks at specific challenges involving small ruminant semen preservation: (a) preserving the structural integrity of the spermatozoa during freezing, (b) achieving restoration of function after thawing, and (c) reduction of oxidative stress damage. It assesses the active and passive cryoprotectants: endogenous, antioxidant, synthetic, and phytochemical and their effects on the quality of frozen-thawed goat sperm. It also aims at the successful use of the more fertile males of the species, and at the same time points out the problems that need to be studied more deeply.
Materials and methods
This literature review systematically explores experimental studies investigating the use of antioxidants in goat sperm cryopreservation to promote better sperm quality post-thawing. Several major scientific databases were systematically explored, including Scopus, PubMed, and Google Scholar, for experimental studies on antioxidant use in goat and/or sheep semen cryopreservation, covering 1 January 2015 to 30 July 2025. The final search was run on 30 July 2025 (Asia/Jakarta). Reference lists of relevant reviews and included studies were also screened to identify additional records. To streamline the search process, a specific set of keywords was applied, combining terms related to species, preservation methods, and antioxidant interventions. The primary keywords included:
Species-specific terms: “goat” “sheep” OR “Caprine”
Preservation techniques: “cryopreservation”
Antioxidant compounds: “antioxidants”.
The search identified 250 records (databases, n = 240; registers, n = 10). Before screening, 40 duplicate records and 10 records flagged as ineligible by automation tools were removed (other reasons, n = 0), leaving 200 records for title/abstract screening. At screening, 30 records were excluded with reasons: species other than goats or sheep (n = 10), no antioxidant intervention (n = 8), and no post-thaw outcomes (n = 12). We sought 170 full-text reports; 20 were not retrieved. Thus, 150 reports were assessed for eligibility, and 105 were excluded with reasons: study design (n = 38), irrelevant parameter (n = 45), and not enough information (n = 22). After full-text screening, 25 articles were included in the qualitative synthesis. The PRISMA flow diagram summarizing selection is shown in Figure 1.
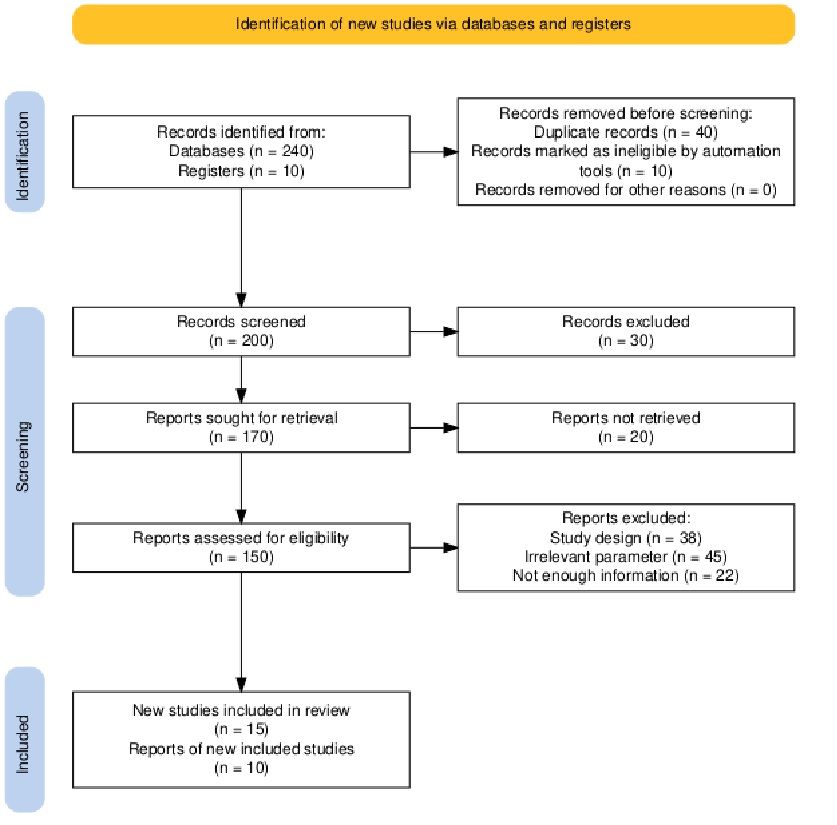
Figure 1 The PRISMA flow chart visually outlines how studies were selected for this systematic review of antioxidant use in goat sperm freezing techniques, covering research published between 2015 and 2025.
Key study details were systematically extracted, covering the type and concentration of antioxidants, extender composition, freezing protocol, and post-thaw outcomes such as motility and viability. Operational definition used in this review: “endogenous” denotes antioxidants that are naturally present in fresh semen, whereas “exogenous” denotes antioxidants that are not naturally present in fresh semen and are introduced from outside but can act as antioxidants in the cryopreservation system.
Data were organized in Table 1 to provide an overview of study characteristics. Outcomes expressed as percentages (motility, viability) underwent arcsine-square-root transformation before effect-size computation to stabilize variances. Subsequently, standardized mean differences were calculated as Hedges’ g (REML) using the converted means and standard deviations. To ensure robustness, sensitivity analyses were conducted utilizing the raw mean difference (MD) on the untransformed % scale and juxtaposing the findings with the SMD results.
Risk of bias was assessed with the Cochrane RoB 2 tool across 5 domains: D1 randomization process, D2 deviations from intended interventions, D3 missing outcome data, D4 measurement of outcomes, and D5 selection of the reported result. Two reviewers independently completed RoB 2 signaling questions and assigned domain-level and overall judgments (low risk, some concerns, high risk); disagreements were resolved by consensus. Results are summarized in a traffic-light plot and summary figure (Figures 3 and 4). Sensitivity analyses excluded studies rated high risk overall and, in exploratory meta-regression, included the overall RoB judgment as a moderator to examine its impact on pooled effects and heterogeneity (Hedges’ g, τ² and I²).
Quantitative synthesis was then performed using OpenMEE software (build 2016-07-26), applying a continuous random-effects model to calculate standardized mean differences (SMD) with 95% confidence intervals. Subgroup analyses were conducted to directly compare the effects of endogenous and exogenous antioxidants.
The characteristics of the studies (Tables 2 and 4), the outcomes of these meta-analyses (Tables 3 and .5) and illustrated in forest plots (Figures 5 and 6), providing pooled estimates for motility and viability. Statistical heterogeneity was assessed using the I² statistic, with values of 25%, 50%, and 75% interpreted as low, moderate, and high heterogeneity, respectively. In this analysis, motility outcomes showed low heterogeneity across both subgroups (I² = 12% for endogenous, I² = 0% for exogenous), while viability outcomes exhibited substantial heterogeneity (I² = 92.14% for endogenous, I² = 59.94% for exogenous). These metrics were reported alongside pooled estimates to provide a clearer interpretation of effect consistency across studies. By combining systematic evidence extraction with quantitative synthesis, this approach not only contrasts with the relative effectiveness of endogenous versus exogenous antioxidants but also identifies which supplementation strategies offer more reliable protection for goat sperm during cryopreservation.
The critical need for optimal antioxidants in goat sperm cryopreservation why are goat and sheep sperm particularly vulnerable to freezing damage?
Sperm cryopreservation doesn't work equally well across all species, and unfortunately, goat sperm ranks among the most vulnerable. Unlike cattle, pigs, or even horses, goats and sheep suffer much more from freezing damage, often showing the poorest quality after thawing. The main culprit behind this vulnerability is their cell membrane structure. The primary factor underlying this vulnerability is the membrane composition of goat spermatozoa. Their membranes are enriched with polyunsaturated fatty acids (PUFAs), which contribute to membrane fluidity but simultaneously increase susceptibility to oxidative damage. During freezing, these PUFAs break down through a process called lipid peroxidation, which weakens the membrane, slashes sperm motility, and can even destroy the acrosome - a vital structure the sperm needs to penetrate and fertilize an egg [27]. In contrast, cattle sperm have fewer PUFAs in their membranes, giving them better cold resistance [28], while pig sperm benefit from higher cholesterol, which stabilizes their membranes [29].
Another issue lies in the mitochondria, the energy-producing powerhouses of sperm. In goats and sheep, these mitochondria have fewer inner folds (cristae), making them less efficient and more likely to leak harmful reactive oxygen species (ROS) during freezing and thawing. This not only depletes energy but can also trigger cell death [30,31]. Stallion sperm, in contrast, has sturdier mitochondria, allowing them to recover better post-thaw [32]. Making matters worse, goat semen naturally contains lower levels of protective antioxidants like glutathione peroxidase (GPx) and superoxide dismutase (SOD), which normally help neutralize ROS and prevent cell damage [11]. Bulls, however, have higher concentrations of these enzymes, giving their sperm a stronger natural defense against freezing injury [33].
The takeaway is clear: standard cryopreservation protocols may not be enough for goats’ sperm. While species like cattle and pigs do reasonably well with basic cryoprotectants, goat sperm needs specialized antioxidant support - both from within and added externally - to survive freezing and stay functional for successful fertilization.
Mechanisms of oxidative stress in cryopreserved goat sperm
Cryopreserving goat sperm for artificial insemination is far more complicated than it looks. Unlike bull or pig sperm, which handle freezing relatively well, goat sperm are naturally fragile, largely due to their unique cellular structure. The first major hurdle lies in their cell membranes. Goat sperm are rich in polyunsaturated fatty acids (PUFAs), which normally help maintain membrane flexibility. But when it froze, these fats become a liability [34]. Ice crystals and oxidative stress tear through the membranes, destroying critical structures like the acrosome and severely reducing the sperm’s ability to swim.
But it’s not just about the membranes. The mitochondria - tiny structures that power the sperm - also play a role in this fragility. Goat sperm mitochondria have fewer inner folds (called cristae), which makes them less efficient at producing energy and more prone to leaking harmful reactive oxygen species. On top of that, goat semen naturally has lower levels of protective antioxidants, so these sperm enter the cryopreservation process without much in the way of defense [35]. Add all this together and you get a perfect storm: damaged membranes, weak energy production, and DNA that’s vulnerable to breakage. The illustration and its effect display in (Figure 2).
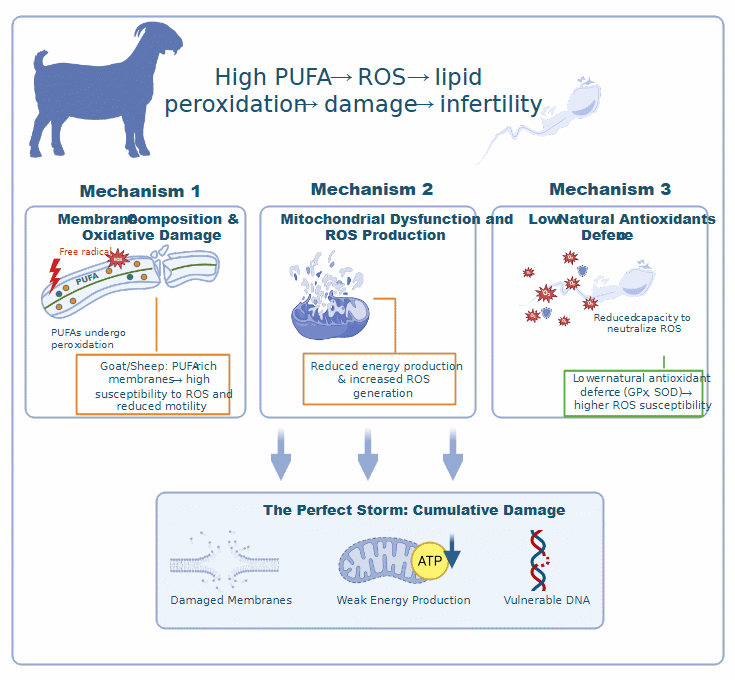
Figure 2 Mechanisms of PUFA-driven oxidative damage in goat sperm.
Classification of antioxidants in sperm cryopreservation
When freezing goat sperm, antioxidants act as cellular bodyguards against the damaging effects of oxidative stress. The freezing process generates harmful reactive oxygen species (ROS) that can wreak havoc through lipid peroxidation, but antioxidants step in to neutralize these threats. Researchers typically divide these protective compounds into 2 main teams:
1) Enzymatic defenders - including superoxide dismutase, catalase, and glutathione peroxidase
2) Non-enzymatic protectors - such as melatonin, vitamin E, plant polyphenols, and synthetic options like sodium salicylate
Each type works differently to shield sperm cells. To compare their effectiveness, we've organized key data - including how they work and their impact on thawed sperm quality - in Table 1. This comprehensive analysis helps identify which antioxidant approaches work best for maintaining goat sperm viability after freezing and thawing.
Table 1 Antioxidants in sperm cryopreservation - mechanisms and post-thaw effects.
Antioxidant |
Type |
Category |
Effect on Post-Thaw Sperm Quality |
Key References |
Superoxide Dismutase (SOD) |
Endogenous |
Enzymatic |
↑ motility, viability, and membrane integrity; ↓ ROS; ↑ cleavage rate after IVF |
[36] |
Glutathione Peroxidase (GPx) |
Endogenous |
Enzymatic |
↓ plasma membrane damage and lipid peroxidation; no significant on DNA methylation |
[37] |
Catalase (CAT) |
Endogenous |
Enzymatic |
↑ Motility, ↑ viability, ↑ plasma membrane intact, ↑ intact acrosome |
[38] |
Coenzyme Q10
|
Endogenous |
Non-enzymatic
|
↑ Motility, ↑ viability, ↑ intact acrosome |
[39] |
Maintain plasma membrane integrity and membrane stability, ↑ pregnancy rates |
[40] |
|||
Glutathione (GSH) |
Endogenous |
Non-enzymatic |
↑ Motility, viability, plasma membrane intact; ↓ ROS and MDA; ↑SOD and GSH-PX |
[7] |
L-Carnitine |
Endogenous |
Non-enzymatic |
↑ motility, maintain membrane integrity, ↓ lipid peroxidation |
[41] |
↑ motility, maintain membrane integrity, ↓ sperm early capacitation |
[42] |
|||
Melatonin |
Endogenous/ exogenous |
Non-enzymatic |
↑ progressive motility |
[43] |
Vitamin E (α-Tocopherol) |
Exogenous |
Non-enzymatic |
↑ motility and viability, ↓ MDA, maintain membrane integrity |
[44] |
Glycine |
Exogenous |
Non-enzymatic |
↑ motility, ↑ viability, ↑ plasma membrane and acrosome integrity, ↑ mitochondrial membrane potential, and ↑ DNA integrity |
[45] |
Quercetin |
Exogenous |
Non-enzymatic |
↑ motility, ↑total antioxidant capacity, ↓ MDA, ↓total oxidant capacity |
[46] |
Isoglycyrrhizin |
Exogenous |
Non-enzymatic |
↑ motility, ↑antioxidant enzyme activities, ↓cleavage and blastocyst formation rates in vitro |
[17] |
Resveratrol |
Exogenous |
Non-enzymatic |
↑motility, ↑ viability, and maintain plasma membrane integrity, ↓MDA, ↑total antioxidant capacity |
[47] |
Green Tea Extract (EGCG) |
Exogenous |
Non-enzymatic |
↑ viability, ↑ intact plasma membrane, ↓DNA fragmentation, ↓amino acids mutation |
[21] |
↑motility, ↑ viability, and maintain plasma membrane integrity, ↓DNA fragmentation |
[48] |
|||
↑motility, ↑ viability, and maintain plasma membrane integrity, ↓MDA level, ↓DNA fragmentation |
[49] |
|||
Butylated Hydroxytoluene (BHT) |
Exogenous |
Non-enzymatic |
↑ Post-thaw motility, ↑ Membrane and acrosome integrity, ↓ MDA, ↓ ROS, but high doses may impair sperm function |
[18] |
Sodium Salicylate |
Exogenous |
Non-enzymatic |
↑motility, ↑ Membrane and acrosome integrity, ↓ ROS, ↓ MDA, ↑catalase, ↑SOD, ↑mitochondrial membrane potential |
[4] |
Proline |
Exogenous |
Non-enzymatic |
↑motility, ↑viability, and ↑acrosome integrity, ↓ sperm apoptosis, ↓ oxidative stress via Proline Dehydrogenase |
[3] |
Nano-Berberine |
Exogenous |
Non-enzymatic |
↑motility, ↑viability, ↑DNA and membrane integrity, ↓ROS, ↓lipid peroxidation, ↑ in vitro fertilization |
[23] |
Selenium Nanoparticles |
Exogenous |
Non-enzymatic |
↑motility, ↑viability, maintain membrane integrity, ↓lipid peroxidation |
[50] |
Fumaric Acid |
Exogenous |
Non-enzymatic |
↑motility, ↑viability, and ↑acrosome integrity, maintain membrane integrity, ↑ mitochondrial function |
[51] |
Folic Acid |
Exogenous |
Non-enzymatic |
↑motility, ↑viability, maintain membrane integrity, ↓lipid peroxidation |
[52] |
Risk of bias
Risk of bias (RoB 2) is a significant concern in research. Most studies showed low risk in D1 (randomization process) and D4 (measurement of outcomes), while D2 (deviations from intended interventions) frequently carried some concerns due to limited reporting of blinding or protocol adherence. D3 (missing outcome data) was flagged as high risk in 2 studies (visible as the red markers), whereas the remainder were low/some concerns. For D4 - D5 (measurement and selection of reported results), several studies provided insufficient information (blue markers), typically because assessor blinding or a prespecified analysis plan was not stated. Overall, most studies were judged to have “some concerns,” with a subset rated overall low risk (e.g., Carrico et al. [43]; Akhter et al. [43]; Nazif et al. [43]); no study was rated overall high risk. Excluding the high-risk (D3) studies in a sensitivity analysis did not materially change the pooled effects, and heterogeneity decreased slightly, indicating that the main conclusions are robust to study quality.
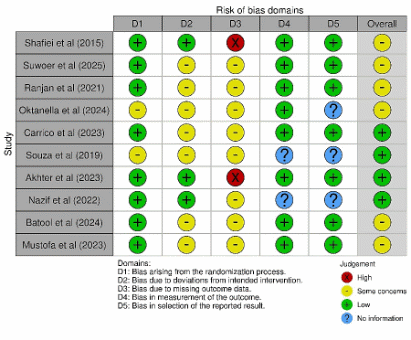
Figure 3 Traffic-light risk-of-bias plot (RoB 2.0). Each row represents a study and columns D1–D5 show domain-level judgments: D1 randomization process, D2 deviations from intended interventions, D3 missing outcome data, D4 measurement of the outcome, and D5 selection of the reported result. Symbols/colors indicate the rating (green “+” = low risk; yellow “○” = some concerns; red “×” = high risk; blue “?” = no information). The Overall column summarizes the study-level risk of bias across domains.
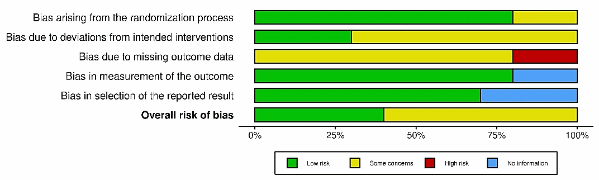
Figure 4 Summary risk-of-bias across domains (RoB 2.0). Stacked bars show the percentage of the 10 included studies judged in each category - low risk (green), some concerns (yellow), high risk (red), and no information (blue) - for the 5 RoB 2.0 domains and the overall judgment (bars sum to 100%).
Meta analysis
Additional antioxidants' effect on sperm’s post-thawed motility
The subgroup meta-analysis demonstrated that endogenous antioxidants were significantly associated with a reduction in sperm motility (SMD = –0.182; 95% CI = –0.359 to –0.004; p = 0.045), whereas exogenous antioxidants showed no significant effect (SMD = 0.025; 95% CI = –0.144 to 0.194; p = 0.773). The overall pooled estimate across studies indicated a non-significant decrease in motility (SMD = –0.080; 95% CI = –0.199 to 0.039; p = 0.186). Heterogeneity was low in both subgroups (I² = 12% for endogenous; I² = 0% for exogenous), suggesting consistent results within groups. The corresponding forest plot (Figure 5) illustrates these findings, where the diamond shapes for endogenous and overall estimates lie to the left of the null line, indicating a trend toward reduced motility, while exogenous antioxidants cluster around the line of no effect.
Table 2 Characteristics of the studies included in the meta-analysis (motility).
Study References |
Type of antioxidants |
Samples (n) |
% motility (control) |
% motility (supplementation) |
Type |
Group |
Shafiei et al. [36] |
Superoxide Dismutase (SOD) |
10 |
40.7 |
55.0 |
Enzymatic |
Endogenous |
Suwor et al. [37] |
Glutathione Peroxidase (GPx) |
5 |
66.3 |
53.5 |
Enzymatic |
Endogenous |
Ranjan et al. [38] |
Catalase (CAT) |
6 |
52.5 |
65.8 |
Enzymatic |
Endogenous |
Oktanella et al. [5] |
Coenzyme Q10 |
5 |
51.2 |
61.4 |
Non-enzymatic |
Endogenous |
Carrico et al. [43] |
Glutathione (GSH) |
5 |
52.6 |
64.6 |
Non-enzymatic |
Endogenous |
Souza et al. [41] |
L-Carnitine |
8 |
43.8 |
56.3 |
Non-enzymatic |
Exogenous |
Akhter et al. [44] |
Vitamin E (α-Tocopherol) |
5 |
37.6 |
55.2 |
Non-enzymatic |
Exogenous |
Nazif et al. [45] |
Glycine |
5 |
52.4 |
68.5 |
Non-enzymatic |
Exogenous |
Batool et al. [46] |
Quercetin |
6 |
53.3 |
68.2 |
Non-enzymatic |
Exogenous |
Mustofa et al. [48] |
Green Tea Extract |
5 |
46.7 |
66.7 |
Non-enzymatic |
Table 3 Subgroup meta-analysis of antioxidant effects on sperm motility.
Subgroup |
Estimate (SMD) |
95% CI (Lower - Upper) |
Std. Error |
p-Value |
Endogenous antioxidants |
–0.182 |
–0.359 to –0.004 |
0.091 |
0.045 |
Exogenous antioxidants |
0.025 |
–0.144 to 0.194 |
0.086 |
0.773 |
Overall |
–0.080 |
–0.199 to 0.039 |
0.061 |
0.186 |
Note: SMD = Standardized Mean Difference; CI = Confidence Interval. Negative SMD values indicate a decrease in sperm motility, whereas positive values indicate an increase. A p-value < 0.05 was considered statistically significant.
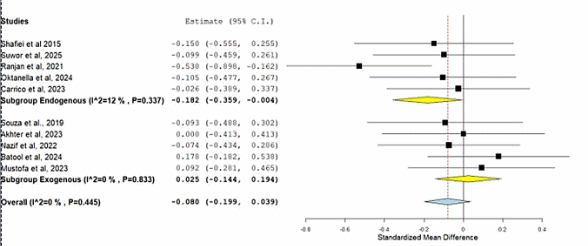
Figure 5 Forest plot of subgroup meta-analysis of endogenous and exogenous antioxidant supplementation on sperm motility.
Additional antioxidants effect on sperm’s post-thawed viability
The subgroup meta-analysis demonstrated that both endogenous and exogenous antioxidants significantly improved sperm viability compared with controls. Endogenous antioxidants yielded a moderate but significant effect (SMD = 4.105; 95% CI = 0.100 - 8.109; p = 0.045), whereas exogenous antioxidants showed a stronger and highly significant effect (SMD = 5.150; 95% CI = 2.810 - 7.490; p < 0.001). The overall pooled estimate confirmed a robust positive impact of antioxidant supplementation on sperm viability (SMD = 4.621; 95% CI = 2.070 - 7.172; p < 0.001). Heterogeneity was substantial in both subgroups (I² = 92.14% for endogenous; I² = 59.94% for exogenous), indicating variability across studies. The forest plot (Figure 6) illustrates these findings, with both subgroup diamonds positioned clearly to the right of the null line, highlighting the consistent enhancement of viability following antioxidant supplementation.
A high level of heterogeneity appears in both endogenous and exogenous subgroups, likely stemming from a mix of methodological and biological differences. Doses vary widely, and several antioxidants show dose-dependent - sometimes U-shaped - responses that can blur pooled effects. Extender formulations and handling steps (cooling/thawing rates, cryoprotectant ratios) differ across laboratories and can modify antioxidant action, especially in lipid-rich sperm membranes. Biological factors - species, breed, season, and baseline semen quality - add further variability. Measurement choices (CASA settings, viability assays, timepoints) and differences in study quality can also widen between-study dispersion. These factors explain much, though not all, of the variance, consistent with wide prediction intervals. Reducing heterogeneity will require standardized extenders and protocols, pre-specified and biologically justified doses, and fully reported methods.
Table 4 Characteristics of the studies included in the meta-analysis (viability).
Study References |
Type of antioxidants |
Samples (n) |
% viability (control) |
%viability (supplementation) |
Type |
Group |
Shafiei et al. [36] |
Superoxide Dismutase (SOD) |
5 |
32.22 |
45 |
Enzimatic |
Endogenous |
Oktanella et al. [5] |
Coenzyme-Q10 |
5 |
56.5 |
67.5 |
Non-enzymatic |
Endogenous |
Carrico et al. [43] |
Glutathione (GSH) |
5 |
40.1 |
40.9 |
Non-enzymatic |
Endogenous |
Nazif et al. [45] |
Glycine |
6 |
62.96 |
72.41 |
Non-enzymatic |
Exogenous |
Batool et al. [46] |
Quercetin |
5 |
44.22 |
51.08 |
Non-enzymatic |
Exogenous |
Mustofa et al. [48] |
Green Tea Extract |
5 |
30.5 |
34.83 |
Non-enzymatic |
Exogenous |
Table 5 Subgroup meta-analysis of antioxidant effects on sperm viability.
Subgroup |
Estimate (SMD) |
95% CI (Lower - Upper) |
Std. Error |
p-value |
Endogenous antioxidants |
4.105 |
0.100 - 8.109 |
2.043 |
0.045 |
Exogenous antioxidants |
5.150 |
2.810 - 7.490 |
1.194 |
< 0.001 |
Overall |
4.621 |
2.070 - 7.172 |
1.302 |
< 0.001 |
Note: SMD = Standardized Mean Difference; CI = Confidence Interval. Negative SMD values indicate a decrease in sperm motility, whereas positive values indicate an increase. A p-value < 0,05 was considered statistically significant.
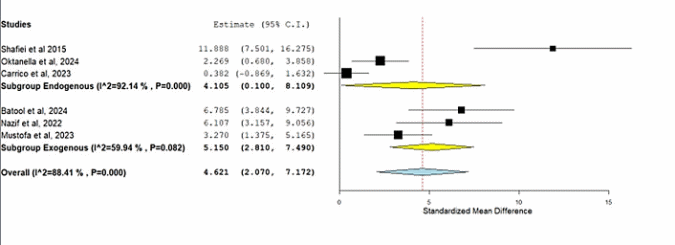
Figure 6 Forest plot of subgroup meta-analysis showing the effect of endogenous and exogenous antioxidant supplementation on sperm viability.
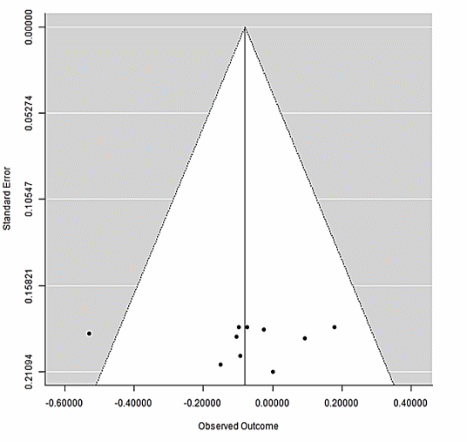
Figure 7 Funnel plot of small-study effects/publication bias. Points are individual comparisons plotted as SMD (x-axis) against standard error (y-axis; higher precision at the top). The vertical line is the pooled effect; dotted diagonals are pseudo-95% confidence limits forming the triangle. Shaded areas lie outside these limits. This display complements the forest and risk-of-bias results by visualizing potential small-study or selective-reporting patterns without relying on formal asymmetry tests.
Figure 7 (k ≈ 9 comparisons) shows a largely symmetric scatter around the pooled effect (near 0 SMD), with one small study exhibiting a relatively large negative effect size; there is no obvious “missing” tail on either side of the triangle. Because the number of studies is < 10, formal small-study tests (Egger’s regression) and trim-and-fill are underpowered and can be unstable; therefore, we treat this evaluation as exploratory. Overall, the visual pattern does not suggest pronounced small-study or publication bias, and our inference for this outcome remains unchanged.
Inside every sperm cell, there is a built-in defence system constantly working to counter oxidative damage - a challenge that becomes especially critical during cryopreservation. This system relies on both enzymatic and non-enzymatic antioxidants, each with specific roles. Enzymatic antioxidants such as glutathione peroxidase (GPx), superoxide dismutase (SOD), and catalase (CAT) are frontline protectors. GPx reduces hydrogen peroxide (H₂O₂) into water, limiting lipid peroxidation and preserving DNA integrity [11,53]. SOD neutralises superoxide radicals (O₂⁻), improving mitochondrial function and lowering apoptosis from reactive oxygen species (ROS) buildup [54]. In turn, CAT eliminates remaining H₂O₂, protecting crucial structures like the acrosome that are essential for fertilisation [29]. Alongside these enzymes, natural non-enzyme antioxidants - glutathione (GSH), Coenzyme Q10, L-carnitine, and melatonin - further reinforce the sperm’s defence. GSH scavenges ROS and strengthens membrane stability [55], while Coenzyme Q10 safeguards mitochondrial integrity and reduces DNA fragmentation [5]. L-carnitine boosts energy metabolism, enhancing motility and viability while reducing cryocapacitation [40]. Melatonin is unique because it works both internally and externally: it enhances the activity of SOD and GPx, maintains membrane resilience, and prevents sperm from triggering apoptotic pathways [8].
Building on this natural defence, researchers have explored supplementing semen extenders with additional antioxidants to better withstand oxidative stress during cryopreservation. Endogenous compounds like Coenzyme Q10 [5,56], glutathione [7,37], and melatonin [8,57] are particularly attractive since they are naturally synthesised within sperm, reducing the risk of toxicity while strengthening the cell’s own antioxidant shield. These agents help neutralise ROS, protect mitochondria and membranes, and regulate apoptotic signalling by upregulating anti-apoptotic proteins (e.g., BCL-2) and downregulating pro-apoptotic factors (e.g., BAX) [11]. However, even these natural defenders have limitations under extreme oxidative conditions, highlighting why optimising antioxidant supplementation remains a critical strategy in improving sperm cryosurvival.
Exogenous antioxidants are externally supplemented compounds designed to reinforce the body's natural antioxidant defences, especially critical during sperm freezing and thawing. External antioxidants, including some plant-based substances like Turraea fischeri extract [13], proline [3,58,59], and resveratrol [12,60,61], are more effective and flexible in protecting against oxidative damage. These factors can be tailored in concentration and composition to suit different qualities of semen and freezing methods. Their applicability, however, has disadvantages. Inevitably, high doses may prove to be cytotoxic, and lack of preparation or purity can lead to inconsistencies with unpredictable outcomes. These external antioxidants may be more effective in treating lipid peroxidation and ROS buildup, but the balance of these antioxidants to protect against oxidative damage while preserving sperm cell viability is still a critical challenge [16,13,62]. With the choice of endogenous versus exogenous antioxidants, considerations of the protective capability alongside endurance of spermatozoa and compatibility with extenders for recovery post-thaw should be prioritized.
Despite all the advances in technology, scientists are still debating which antioxidant works best. BHT is synthetic, yet study suggests it prevents cell fat breakdown [18]. However, the potential for cellular toxicity remains to be evaluated. On the natural side, oils such as Nigella sativa [20], green tea [21,63], and coconut milk or pyridoxine supplementation [64,65] have shown clear advantages, especially in the recovery of post-thaw motility and cryo-survival in ovine and caprine sperm. Also, the recent introduction of selenium-loaded berberine nanoparticles [23], isoglycyrrhizin [62], platelet-rich plasma [66,67], sericin [68], quercetin [69], N-acetylcysteine [27], IGF-1 [70], MitoQ [71,72], and metabolic regulators such as sodium salicylate [4] demonstrate the promising potential of cryopreservation without the cryoinjury risk.
Comparative advantages and limitations of endogenous vs exogenous antioxidants
Across outcomes, the patterns were not uniform. For motility, the endogenous subgroup showed a small but statistically significant decrease (SMD = –0.182; 95% CI –0.359 to –0.004), while the exogenous subgroup showed no clear effect (SMD = 0.025; 95% CI –0.144 to 0.194). The overall pooled estimate was non-significant (SMD = –0.080; 95% CI –0.199 to 0.039), with low within-subgroup heterogeneity (I² = 12% endogenous; 0% exogenous), indicating fairly consistent findings for motility.
For viability, both subgroups showed improvement compared to controls. The pooled impact was bigger for exogenous antioxidants (SMD = 5.150; 95% CI 2.810 to 7.490) than for endogenous antioxidants (SMD = 4.105; 95% CI 0.100 to 8.109). However, these estimates were accompanied by strong heterogeneity (I² = 92.14% endogenous; 59.94% exogenous), indicating that effects differ significantly among studies and settings.
Mechanistically, these patterns are plausible: endogenous defences sustain baseline redox balance but may be insufficient against the abrupt oxidative challenge of freeze–thaw, whereas exogenous supplementation can, under some conditions, provide additional membrane and mitochondrial protection. Still, the magnitude of viability effects appears to depend on dose, formulation, extender, and handling steps, and may be attenuated when methodological variability is accounted for.
Sensitivity checks supported the robustness of the main conclusions. Excluding studies with high risk in missing-data (D3) did not materially change pooled effects, and heterogeneity decreased slightly. A funnel-plot screen (k ≈ 9) did not suggest pronounced small-study or publication bias, though formal tests are underpowered below 10 studies; we therefore treat the bias assessment as exploratory. Despite inconsistent motility increases, endogenous antioxidants remain a key defense during cryopreservation, supporting post-thaw viability. Their physiological compatibility and baseline redox support make them a good first-line method when dose, extender composition, and handling can be standardized. Selecting relevant endogenous substances (enzymatic and non-enzymatic), calibrating dosages, and aligning them with extender chemistry and cooling/thawing procedures may produce dependable benefits with decreased formulation-related toxicity. Exogenous supplements can be used carefully but future research should focus on endogenous regimens and their optimal performance across breeds, seasons, and baseline semen quality.
Summary of current findings
Small ruminant sperm are susceptible to freeze–thaw injury, hence antioxidant defence is crucial to cryopreservation. Endogenous antioxidants—parts of the intrinsic redox system - supported post-thaw viability in our meta-analysis, even though motility did not increase and, in pooled estimates, reduced slightly. The results indicate that endogenous defences provide a physiologically compatible baseline buffer against oxidative stress but do not always retain movement-related function.
Exogenous antioxidants had bigger pooled effects on viability than endogenous agents, although motility effects were only minor. Heterogeneity (dose, formulation, extender, handling procedures) reduces assurance. Thus, their advantages appear condition-dependent, with dose and toxicity trade-offs that must be clarified before routine usage is advised.
The evidence suggests that endogenous mechanisms can maintain viability following freezing, but neither endogenous nor exogenous techniques improve motility. Future work should standardize extenders and protocols, pre-specify biologically justified doses, and report methods in full to determine when adjunct exogenous supplementation adds value to the endogenous baseline and its net effect on fertility-relevant outcomes.
Conclusions
Based on the evidence, endogenous antioxidants, which are part of the intrinsic redox system, consistently support post-thaw viability but do not reliably maintain motility, suggesting functional limitations under freeze-thaw stress. Exogenous antioxidants can provide greater improvements in viability; however, their effects seem to be contingent upon specific conditions (dose, formulation, extender, handling) and are influenced by significant variability among studies and uncertainties regarding study quality. The data collectively endorse endogenous strategies as a physiologically compatible foundation, with selective, cautiously optimized exogenous supplementation regarded as a potential complementary rather than an entirely superior treatment.
Acknowledgements
We extend our appreciation to our respective universities and research groups for providing the resources necessary to complete this work.
Declaration of generative AI in scientific writing
The authors acknowledge the use of generative AI tools in the preparation of this manuscript. ChatGPT (OpenAI) was employed for language editing and grammar refinement, while BioRender was used to generate illustrative figures. No content generation, data interpretation, or scientific conclusions were produced by AI. The authors take full responsibility for the accuracy, integrity, and conclusions of this work.
CRediT author statement
Yudit Oktanella: Conceptualization, Methodology, Writing – Original Draft. Imam Mustofa: Data Curation, Funding Acquisition. Suherni Susilowati: Screening, Data Curation. Widjiati Widjiati: Supervision, Validation, Resources. Bambang Purwantara: Supervision, Writing - Review & Editing. Adeyinka Oye Akintunde: Software, Visualization. Tatik Hernawati: Screening, Validation. Tri Wahyu Suprayogi: Formal Analysis, Methodology. Suzanita Utama: Supervision, Writing - Review & Editing. Eka Pramyrtha Hestianah: Writing - Review & Editing.
References
Ministry of Agriculture. Annual report on livestock and animal health sector in Indonesia. Ministry of Agriculture, Jakarta, Indonesia, 2023.
Directorate General of Livestock and Animal Health. National livestock revitalization program 2020 - 2025. Ministry of Agriculture, Jakarta, Indonesia, 2022.
W Zhang, L Min, Y Li, Y Lang, SAM Hoque, AO Adetunji and Z Zhu. Beneficial effect of proline supplementation on goat spermatozoa quality during cryopreservation. Animals 2022; 12(19), 2626.
W Shen, Y Fu, H Bai, Z Zhang, Z Cao, Z Liu, C Yang, S Sun, L Wang, C Ren, Y Ling, Z Zhang and H Cao. Antioxidant activity and metabolic regulation of sodium salicylate on goat sperm at low temperature. Animal Bioscience 2024; 37(4), 640-654.
Y Oktanella, I Mustofa, FAR An-Haru, DDM Putri, VF Hendrawan, S Susilowati, NY Degu and T Hernawati. Conserving goat sperm post-thawed gene expression and cellular characteristics using the antioxidant coenzyme Q10 supplementation. Veterinary world 2024; 17(7), 1637-1647.
B Khazravi, M Khodaei-Motlagh, R Masoudi and M Yahyaei. Coenzyme Q10 preserves buck’s sperm quality during cryopreservation process in plant-based extender. Animal Reproduction Science 2024; 265, 107487.
J Zou, L Wei, D Li, Y Zhang, G Wang, L Zhang, P Cao, S Yang and G Li. Effect of glutathione on sperm quality in Guanzhong dairy goat sperm during cryopreservation. Frontiers in Veterinary Science 2021; 8, 771440.
MG Al-Mutary, MQ Al-Ghadi, AA Ammari, AR Al-Himadi, AH Al-Jolimeed, MW Arafah, RA Amran, MS Aleissa and AA-A Swelum. Effect of different concentrations of resveratrol on the quality and in vitro fertilizing ability of ram semen stored at 5 °C for up to 168 h. Theriogenology 2020; 152, 139-146.
IŽ Žaja, V Berta, H Valpotić, M Samardžija, S Milinković-Tur, M Vilić and S Vince. The influence of exogenous melatonin on antioxidative status in seminal plasma and spermatozoa in French Alpine bucks during the nonbreeding season. Domestic Animal Endocrinology 2020; 71, 106400.
M Monteiro, M Silva, RAJA Arruda, LCP Oliveira, AS Mergulhão, FCC Monteiro Júnior, PLJ Batista, AM and MMP Guerra. Effect of melatonin in different extenders on the quality of frozen semen of goats. Emerging Animal Species 2022; 5(1-4), 100015.
M Alvarez-Rodriguez, M Alvarez, L Anel-Lopez, F Martinez-Pastor, S Borragan, L Anel and P de Pazz. Sperm membrane fatty acid composition in the cryopreservation of goat semen: a pilot study. Andrologia 2020; 52(2), e13484.
L Falchi, S Pau, I Pivato, L Bogliolo and MT Zedda. Resveratrol supplementation and cryopreservation of buck semen. Cryobiology 2020; 95, 60-67.
SA Hassan, WA Khalil, MAE Hassan, AI Yousif, OM Sabry, M Wink and M Sobeh. Antioxidant and antiapoptotic effects of a Turraea fischeri leaf extract on cryopreserved goat sperm. Animals 2021; 11(10), 2840.
SN Abedin, A Baruah, KK Baruah, A Bora, DJ Dutta, G Kadirvel, R Katiyar, S Doley, S Das, G Khargharia, B Sarkar, S Sinha, A Phookan, RK Dewry, MK Kalita, H Chakravarty and S Deori. Zinc oxide and selenium nanoparticles can improve semen quality and heat shock protein expression in cryopreserved goat (Capra hircus) spermatozoa. Journal of Trace Elements in Medicine and Biology 2023; 80, 127296.
MA Khalique, H Rehman, SMH Andrabi, KA Majeed, N Ahmad, MH Fayyaz, M Haider, S Naz, SS Qureshi and S Sulaiman. Antioxidant effects of zinc-oxide nanoparticles on post-thaw quality and in vivo fertility of Beetal buck spermatozoa. Small Ruminant Research 2023; 225, 107012.
C Li, Y Zhang, J Wang, H Liu, X Zhou and W Li. Effects of quercetin supplementation on cryopreserved goat sperm quality: Focus on oxidative stress mitigation and mitochondrial protection. PeerJ 2024a; 12, e17580.
F Liu, J Dai, J Gao, M He, J Xu, C Wu, S Zhang, X Zhu and L Sun. BGP-15 improves quality of goat sperm by mitigating oxidative stress during cryopreservation. Cryobiology 2025a; 119, 105232.
C Li, L Allai, J Liang, C Lv, X Zhao, X Ni, G Wu, W Deng, B Badaoui and G Quan. The antioxidant effects of butylated hydroxytoluene on cryopreserved goat sperm from a proteomic perspective. PeerJ 2024b; 12, e17580.
A Memon, HW Wahid, Y Rosnina, YM Goh, M Ebrahimi, FM Nadia and G Audrey. Effect of butylated hydroxytoluene on cryopreservation of Boer goat semen in Tris egg yolk extender. Animal Reproduction Science 2011; 129(1-2), 44-49.
AG Miah, R Bathgate, KI Hamano and U Salma. Effects of pre-freeze Nigella sativa oil supplementation on cryosurvival of ovine spermatozoa. Reproduction in Domestic Animals 2018; 53(6), 1424-1433.
SS Susilowati, I Mustofa, W Wurlina, T Hernawati, Y Oktanella, S Soeharsono and DA Purwanto. Green tea extract in the extender improved the post-thawed semen quality and decreased amino acid mutation of Kacang buck sperm. Veterinary Sciences 2022; 9(8), 403.
E Kerasioti, D Stagos, A Tzimi and D Kouretas. Increase in antioxidant activity by sheep/goat whey protein through nuclear factor-like 2 (Nrf2) is cell type dependent. Food and Chemical Toxicology 2016; 97, 47-56.
M Piri, AH Mahdavi, M Hajian, A Shahverdi, A Asadi, V Asgari, MH NasrEsfahani and H Gourabi. Effects of nano-berberine and berberine loaded on green synthesized selenium nanoparticles on cryopreservation and in vitro fertilization of goat sperm. Scientific Reports 2024; 14, 24171.
A Kafi, M Khalid, T Herath and C Kershaw. Cysteine supplementation pre-freeze and post-thaw improves integrity and reduces oxidative stress in cryopreserved ram spermatozoa. Cryobiology 2024; 114, 104854.
N Asadzadeh, Z Abdollahi, S Esmaeilkhanian and R Masoudi. Fertility and flow cytometry evaluations of ram frozen semen in plant-based extender supplemented with Mito-TEMPO. Animal Reproduction Science 2021; 233, 106836.
MJ Page, D Moher, PM Bossuyt, I Boutron, TC Hoffmann, CD Mulrow, L Shamseer, JM Tetzlaff, EA Akl, SE Brennan, R Chou, J Glanville, JM Grimshaw, A Hróbjartsson, MM Lalu, T Li, EW Loder, E Mayo-Wilson, S McDonald, LA McGuinness, LA Stewart, J Thomas, AC Tricco, VA Welch, P Whiting and JE McKenzie. PRISMA 2020 explanation and elaboration: Updated guidance and exemplars for reporting systematic reviews. BMJ 2021; 372, n160.
Y Wang, X Fu and H Li. Mechanisms of oxidative stress-induced sperm dysfunction. Frontiers in Endocrinology 2025; 16, 1520835.
I Yánez-Ortiz, J Catalán, JE Rodríguez-Gil, J Miró and M Yeste. Advances in sperm cryopreservation in farm animals: Cattle, horse, pig and sheep. Animal Reproduction Science 2022; 246, 106904.
MJ Shepherd and LA Herickhoff. A novel experimental design for boar sperm cryopreservation. Journal of Animal Science 2022; 100(7), skac169.
B Kushawaha, RS Yadav, DK Swain, P Kumari, A Kumar, B Yadav, M Anand, S Yadav, D Singh and SK Garg. Collapsed mitochondrial cristae in goat spermatozoa due to mercury result in lethality and compromised motility along with altered kinematic patterns. Scientific Reports 2021; 11(1), 646.
S Zang, S Zou, X Chen, B Pan, A Ning, J Qin, Y Wei, K Du, J Ye, Q Liang, Y Fang, Q Cirenlamu, T Song and G Zhou. Abnormalities in mitochondrial energy metabolism induced by cryopreservation negatively affect goat sperm motility. Frontiers in Veterinary Science 2025; 11, 1514362.
Z Al-Kass and JM Morrell. Freezing stallion semen - What do we need to focus on for the future? Veterinary Sciences 2024; 11(2), 65.
A Kowalczyk. The role of the natural antioxidant mechanism in sperm cells. Reproductive Sciences 2022; 29, 1387-1394.
F Ren, T Feng, G Dai, Y Wang, H Zhu and J Hu. Lycopene and alpha-lipoic acid improve semen antioxidant enzymes activity and cashmere goat sperm function after cryopreservation. Cryobiology 2018; 84, 27-32.
Y Oktanella, I Mustofa, S Susilowati, VV Hendrawan and T Hernawati. Effect of cryopreservation duration time on post-thawing sperms’ characteristics of goat semen. Jurnal Veteriner 2024b; 25(2), 175-185.
M Shafiei, M Forouzanfar, SM Hosseini and MH Esfahani. The effect of superoxide dismutase mimetic and catalase on the quality of post-thawed goat semen. Theriogenology 2015; 83(8), 1321-1327.
F Suwor, S Kubota, S Nawong, A Thuangsanthia, M Toyra, P Paengkoum and S Ponchunchoovong. Effects of cryoprotectant combinations on post-thawed sperm quality, biomolecular changes, DNA methylation, and pregnancy rates in Boer goat semen. Veterinary Sciences 2025; 12(2), 178.
R Ranjan, P Singh, C Gangwar, SP Singh, DK Swain and SD Kharche. Fortification of catalase improves post-thaw fertility of goat semen. Iranian Journal of Applied Animal Science 2021; 11(3), 587-593.
A Sarangi, P Singh, M Virmani, AS Yadav, S Sahu, HM Ajithakumar, A Kumari and AP Rath. Effect of antioxidants supplementation on the quality of Beetal buck semen stored at 4°C. Veterinary World 2017; 10(10), 1184-1188.
SMT Tironi, L Sitó-Silva, BL de Camillo, R Denadai, ALA Silva, CP Freitas-Dell'Aqua, JAD Junior, RA de Oliveira, MIL Souza and E Oba. Use of coenzyme Q-10 to improve the pregnancy rate in sheep. Animal Reproduction Science 2024, 266, 107498.
CV Souza, FZ Brandão, JDR Santos, VAP Alfradique, VMB Santos, MCC Morais, PSC Rangel, AA Silva and JMG Souza-Fabjan. Effect of different concentrations of L-carnitine in extender for semen cryopreservation in sheep. Cryobiology 2019; 89, 104-108.
A Najafi, MH Najafi, Z Zanganeh, M Sharafi, F Martínez-Pastor and H Adeldust. Effect of different concentrations of L-carnitine in extender for semen cryopreservation in sheep. Cryobiology 2019; 89, 6-10.
C Carriço, JP Barbas, J Pimenta and J Simões. Effect of in vitro addition of melatonin and glutathione on seminal parameters of rams in diluted semen and after thawing. Veterinary Sciences 2023; 10(7), 446.
S Akhter, M Zubair, M Mahmood, SMH Andrabi, N Hameed, E Ahmad and MK Saleemi. Effects of vitamins C and E in tris citric acid glucose extender on chilled semen quality of Kail ram during different storage times. Scientific Reports 2023; 13(1), 18123.
MS Nazif, ZU Rehman, H Khan, FA Khan, T Hussain, A Ahmad, Farmanullah, A Husnain, S Muhammad, G Murtaza and L Gang. Glycine improved cryopreserved spermatozoa quality in Achai bull. BioMed Research International 2022; 2022(1), 8282387.
I Batool, MH Fayyaz, A Hameed, SMH Andrabi, R Kausar, M Shahzad, Y Mubashir, AD Omur, G Murtaza, A Ditta and T Hussain. Quercetin in semen extender improves frozen-thawed spermatozoa quality and in-vivo fertility in crossbred Kamori goats. Frontiers in Veterinary Science 2024; 11, 1385642.
Z Zhu, H Zhao, H Cui, AO Adetunji and L Min. Resveratrol improves the frozen-thawed ram sperm quality. Animals 2023; 13(24), 3887.
I Mustofa, S Susilowati, TW Suprayogi, Y Oktanella, DA Purwanto and AO Akintunde. Combination of nanoparticle green tea extract in tris-egg yolk extender and 39 °C thawing temperatures improve the sperm quality of post-thawed Kacang goat semen. Animal Reproduction 2023; 19(4), e20220025.
T Suprayogi, W Wurlina, T Hernawati, S Susilowati, Y Oktanella and DA Purwanto. Improved quality of Kambing Kacang sexing frozen semen with the addition of green tea extract. Journal of Advanced Veterinary and Animal Research 2022; 9(3), 412.
S Nateq, G Moghaddam, S Alijani and M Behnam. The effects of different levels of nano selenium on the quality of frozen-thawed sperm in ram. Journal of Applied Animal Research 2020; 48(1), 434-439.
A Saratsi, F Samartzi, I Panagiotidis, A Basioura, D Tsiokos, C Ligda and CA Rekkas. Post-thaw parameters of buck semen quality after soy lecithin extender supplementation with fumaric acid. Veterinary Sciences 2023; 10(9), 569.
Z Abdollahi, S Zeinoaldini, M Zhandi, A Towhidi and H Baghshahi. Supplementation of plant-based cryopreservation medium with folic acid conserves the quality of bulk post-thawed spermatozoa. Archives of Razi Institute 2021; 76(3), 553-559.
DI Berean, LM Bogdan and R Cimpean. Advancements in understanding and enhancing antioxidant-mediated sperm cryopreservation in small ruminants: Challenges and perspectives. Antioxidants 2024; 13(6), 624.
H Younus. Therapeutic potentials of superoxide dismutase. International Journal of Health Sciences 2018; 12(3), 88-93.
J Zou, Y Chen, J Yang, H Shi, Z Wang and Y Xu. Melatonin protects goat spermatozoa against oxidative damage during cryopreservation by improving antioxidant capacity and mitochondrial function. Frontiers in Veterinary Science 2021; 8, 771440.
I Yousefian, M Emamverdi, A Karamzadeh-Dehaghani, R Sabzian-Melei, M Zhandi and A Zare-Shahneh. Attenuation of cryopreservation-induced oxidative stress by antioxidant: Impact of Coenzyme Q10 on the quality of post-thawed buck spermatozoa. Cryobiology 2018; 81, 88-93.
L Gallego-Calvo, MC Gatica, J Santiago-Moreno, JL Guzmán and LA Zarazaga. Exogenous melatonin does not improve the freezability of Blanca Andaluza goat semen over exposure to 2 months of short days. Animal Reproduction Science 2015; 157, 24-32.
A Sharon Sangeeta, S Arangasamy, S Kulkarni and S Selvaraju. Role of amino acids as additives on sperm motility, plasma membrane integrity and lipid peroxidation levels at pre-freeze and post-thawed ram semen. Animal Reproduction Science 2015; 161, 82-88.
J Zhao, P Meng, M Jin, X Ma, H Ma, H Yang and J Liu. Combined addition of L-carnitine and L-proline improves cryopreservation of dairy goat semen. Animal Reproduction Science 2023; 257, 107325.
C Lv, A Larbi, G Wu, Q Hong and G Quan. Improving the quality of cryopreserved goat semen with a commercial bull extender supplemented with resveratrol. Animal Reproduction Science 2019; 208, 106127.
T Hussain, MH Fayyaz, A Hameed, SMH Andrabi, R Kausar, Y Mubashir and AD Omur. Effect of resveratrol on post-thaw motility, kinematics, structural parameters and antioxidant/oxidant status of Kamori buck spermatozoa. Cryobiology 2025; 118, 105202.
F Liu, L Sun, J Xu, M He, C Wu, H Shen, H Zhu, F Luo, J Gao and J Dai. Isoglycyrrhizin supplementation of frozen goat semen-extender improves post-thaw sperm quality and in vitro fertilization rates. Theriogenology 2025; 234, 133-142.
I Mustofa, S Susilowati, W Wurlina, T Hernawati and Y Oktanella. Green tea extract increases the quality and reduced DNA mutation of post-thawed Kacang buck sperm. Heliyon 2021; 7(3), e06372.
JO Daramola, EO Adekunle, OE Oke, OM Onagbesan, OS Iyasere, TJ Williams and JA Oyewusi. Effects of pyridoxine supplementation or in combination with other antioxidants on motility, in vitro capacitation and acrosome reaction of goat buck spermatozoa during cryopreservation. Small Ruminant Research 2015; 131, 113-117.
JO Daramola, EO Adekunle, OS Iyasere, OE Oke, TA Sorongbe, OA Iyanda, AR Kehinde, SP Aluko, IO Olaoye, OE Gbadebo, LI Falolu, EO Olukayode, RA Ajayi, OJ Enikannaye and ED Osunjaiye. Effects of coconut milk alone or supplementation with pyridoxine in tris-extenders on viability of buck spermatozoa during vitrification. Small Ruminant Research 2016; 136, 208-213.
S Alcay, A Aktar, D Koca, MA Kilic, M Akkasoglu and H Sagirkaya. Positive effect of autologous platelet rich plasma on Saanen buck semen cryopreservation in non-breeding season. Cryobiology 2021; 103, 45-48.
MS Salama, AM Shehabeldin, MA Ashour, MQ Al-Ghadi, BH Marghani, I El-kon and M Shukry. Effect of the addition of platelet-rich plasma to Boer buck semen on sperm quality and antioxidant activity before and after cryopreservation and in vivo fertility. Small Ruminant Research 2024; 230, 107167.
VS Reddy, B Yadav, CL Yadav, M Anand, DK Swain, D Kumar and S Yadav. Effect of sericin supplementation on heat shock protein 70 (HSP70) expression, redox status and post thaw semen quality in goat. Cryobiology 2018; 84, 33-39.
A Kumar, JK Prasad, N Kumar, M Anand, S Verma, R Dhariya, A Kumar and A Gattani. Quercetin in semen extender curtails reactive oxygen and nitrogen species and improves functional attributes of cryopreserved buck semen. Cryobiology 2024; 116, 104931.
WL Singh, S Sinha, K Ahmed, L Buragohain, M Das Gupta, D Bhuyan, S Tamuly, SN Abedin, S Dhara and M Nath. IGF-1 outperforms vitamin E and crocin in improving semen quality, antioxidant profile and fertility gene expression in cryopreserved goat (Capra hircus) semen. Animal Reproduction Science 2025; 274, 107791.
A Rezaei, HR Bahmani, S Mafakheri, A Farshad, P Nazari and R Masoudi. Protective effects of different doses of MitoQ separately and combined with trehalose on oxidative stress and sperm function of cryopreserved Markhoz goat semen. Cryobiology 2023; 110, 36-43.
M Hatami, R Masoudi, A Hatefi, P Alipour-Jenaghard and V Esmaeili. The effects of MitoQ as a mitochondrial-targeted antioxidant in a plant-based extender on buck sperm quality parameters during cryopreservation. Animal Reproduction Science 2024; 266, 107517.