
Trends
Sci.
2026;
23(5):
12221
Liposomal Vitamin D3 as A Dual Modulator of Bone Density and Estrogen Homeostasis in Postmenopausal Osteoporosis Model
Ari
Budi Suryawinata1,
![]() ,
Sri Puji Astuti Wahyuningsih1,2,*,
Listyani Suhargo1,2,
,
Sri Puji Astuti Wahyuningsih1,2,*,
Listyani Suhargo1,2,
![]() ,
,
Dwi
Roudlotul Firda1,
Alvin Oktaviana Puspitasari1
and Lionel In Lian Aun3,
![]()
1Departmen of Biology, Faculty of Science and Technology, Universitas Airlangga, Surabaya 60115, Indonesia
2RG Developmental Biology & Biomedical Science, Universitas Airlangga, Surabaya 60115, Indonesia
3Department of Biotechnology, Faculty of Applied Sciences, UCSI University, Kuala Lumpur 56000, Malaysia
(*Corresponding author’s e-mail: [email protected])
Received: 9 October 2025, Revised: 31 October 2025, Accepted: 7 November 2025, Published: 10 January 2026
Abstract
Postmenopausal osteoporosis, driven by estrogen decline, leads to significant bone loss and increased fracture risk. This study evaluated if a liposomal vitamin D3 (VD3) formulation offers superior therapeutic benefits over conventional VD3 in an ovariectomized (OVX) mouse model of postmenopause, based on the rationale that enhanced liposomal bioavailability may amplify VD3’s mechanistic role in regulating steroidogenesis and estrogen homeostasis. Thirty-six female BALB/c mice were ovariectomized and allocated into 6 groups (n = 6/group): Control groups or treatment groups receiving conventional VD3 (2,000 IU) or liposomal VD3 (1,000, 2,000, or 4,000 IU) daily for 2 months. Serum estrogen, calcium, phosphorus, and femoral bone histomorphometry (specifically trabecular area and osteoblast, osteoclast, and osteocyte counts) were analyzed. Liposomal VD3 at 2,000 IU and 4,000 IU doses significantly restored serum estrogen levels, an effect not seen with the conventional form. The 2000 IU liposomal dose was most effective, restoring the trabecular area to near-normal levels and was unique among the interventions in significantly increasing the osteoblast population. All VD3 treatments reduced osteoclast numbers; however, the liposomal formulation showed no superiority in elevating serum calcium compared to the standard form. In conclusion, liposomal VD3 (2000 IU) is significantly more effective than its conventional counterpart in reversing bone loss and restoring estrogen by beneficially modulating bone cell populations. Its primary advantage lies in hormonal regulation and bone remodeling rather than mineral homeostasis, highlighting it as a promising strategy for managing postmenopausal osteoporosis.
Keywords: Postmenopausal osteoporosis model, Liposomal vitamin D3, Estrogen homeostasis, Bone remodeling, Ovariectomized mice
Introduction
Menopause, marked by the permanent cessation of ovarian function, leads to a significant decline in estrogen production. This hormonal shift is a primary etiological factor in the development of postmenopausal osteoporosis, a systemic skeletal disorder characterized by low bone mass and micro-architectural deterioration of bone tissue [1,2]. The resulting increase in bone fragility elevates the risk of fractures, posing a considerable public health challenge and diminishing the quality of life for aging women worldwide. The
underlying pathophysiology involves an imbalance in bone remodeling, where the rate of bone resorption by osteoclasts surpasses the rate of bone formation by osteoblasts, a process heavily influenced by estrogen deficiency [2].
Vitamin D (VD) is a cornerstone in maintaining mineral metabolism and skeletal integrity. Its active metabolite, 1,25(OH)2D3 (calcitriol), is crucial for calcium and phosphorus homeostasis. It enhances the intestinal absorption of these minerals and modulates the secretion of parathyroid hormone (PTH), both of which are vital for proper bone mineralization [3,4]. Consequently, ensuring adequate VD levels is a fundamental strategy in the prevention and therapeutic management of osteoporosis.
Pre-clinical studies utilizing the ovariectomized (OVX) mouse, a well-established animal model that mimics the estrogen-deficient state of post-menopause, have substantiated the beneficial effects of VD3 supplementation. Research in these models demonstrates that VD3 administration can effectively increase serum calcium levels and improve bone mineral density (BMD) [5]. At the cellular level, VD directly influences bone cell populations; it regulates osteoclast activity via the RANKL pathway, promotes the differentiation of osteoblasts, and has been shown to decrease osteocyte apoptosis [6]. These cellular effects translate to tangible improvements in bone structure, including an increase in trabecular bone volume [5]. However, the therapeutic window for VD is critical, as supra-physiological or high doses have been observed to paradoxically inhibit osteoblast mineralization in vitro, highlighting the need for optimized delivery and dosing strategies [7].
Despite its importance, the clinical efficacy of conventional VD3 can be constrained by its lipophilic nature, which may affect its oral bio-availability. To address this challenge, advanced drug delivery systems have been explored. Liposome encapsulation, a technology that entraps active compounds within phospholipid vesicles, offers a promising approach to enhance the solubility, stability, and systemic bio-availability of lipophilic molecules like VD3. This improved delivery may allow for greater therapeutic effects at potentially lower and safer concentrations.
Furthermore, the rationale for investigating liposomal VD3 extends beyond its established role in mineral metabolism to its potential influence on estrogen homeostasis itself. While VD3 is primarily known for bone integrity, emerging evidence indicates it also functions as an endocrine regulator. Research has demonstrated that VD3 can mechanistically influence steroidogenesis; for example, it has been shown to increase the expression of genes involved in estrogen biosynthesis within ovarian granulosa cells [9,10]. This process may be modulated through the activation of specific signaling pathways, such as the AMPK pathway, which plays a role in regulating steroid production [10]. Moreover, VD3 has been reported to possess direct estrogenic activity, capable of influencing the estrous cycle and uterine growth in animal models, further indicating its role in hormonal balance [11]. Even its classical function in calcium homeostasis is relevant, as proper calcium levels are essential for normal granulosa cell function and subsequent hormone biosynthesis [9,12].
Therefore, it is hypothesized that the superior bioavailability afforded by liposomal encapsulation [8], which enhances systemic absorption and stability would not only optimize VD3’s effects on bone remodeling but also amplify its less-appreciated effects on these estrogenic pathways. This enhanced delivery could lead to a more significant restoration of serum estrogen levels compared to conventional VD3, providing a dual-pronged therapeutic strategy against postmenopausal osteoporosis by addressing both mineral deficiency and the underlying hormonal imbalance.
Given the critical role of VD in bone health and the potential advantages of liposome formulations, this study was designed to comprehensively evaluate the impact of liposomal VD3 in a postmenopausal osteoporosis model. Therefore, the present study aims to investigate the effects of liposomal VD3 administration on serum estrogen, calcium, and phosphorus levels, as well as its influence on bone histomorphometry specifically trabecular area, and the number of osteocytes, osteoblasts, and osteoclasts in OVX mice.
Materials and methods
Ethical approval
All experimental protocols involving animal subjects were reviewed and approved by the Health Research Ethics Committee (KEPK) of the Faculty of Dental Medicine, Universitas Airlangga (Ethical Clearance No. 0923/HRECC.FODM/VIII/2024). All procedures were conducted in accordance with the applicable ethical principles and guidelines for animal research.
Animals and housing
Thirty-six female BALB/c mice, initially weighing 25 g, were procured from the Center for Veterinary Farm (PUSVETMA), Surabaya, Indonesia. The animals were housed in eight standard laboratory cages (47×33×15 cm3), with 6 to 7 mice per cage. Each cage was equipped with a food and water dispenser. The animals were maintained in the Animal Laboratory of Universitas Airlangga under controlled environmental conditions with adequate lighting. A 3-week acclimatization period was observed to allow the mice to adapt to the new environment and reach a target body weight of approximately 28 g prior to the surgical procedures.
Ovariectomy
Mice in the ovariectomy groups were anesthetized with ketamine (10 mg/kg body weight). Following anesthetization, the abdominal area was shaved and disinfected with alcohol. A mid-line incision was made to expose the ovaries, which were then ligated with catgut sutures and excised. The peritoneum was sutured with catgut, and the skin was closed with silk sutures. Post-surgery, Nebacetin antibiotic powder was applied to the surgical wound to prevent infection. The animals were allowed a 2-week recovery period. The success of the ovariectomy was confirmed by vaginal smear analysis, ensuring the mice were in the diestrus phase.
Experimental design
A completely randomized design was employed for this study. The mice were randomly allocated into 6 groups, with 6 animals (n = 6) in each group: Control (K): Sham-operated mice receiving the vehicle (coconut oil); Negative Control (P-): OVX mice receiving the vehicle (coconut oil); Positive Control (P+): OVX mice receiving 2000 IU of regular VD3 (NOW Foods, USA); Treatment 1 (P1): OVX mice receiving 1000 IU of Liposomal VD3 (Upnourish, USA); Treatment 2 (P2): OVX mice receiving 2000 IU of Liposomal VD3; Treatment 3 (P3): OVX mice receiving 4000 IU of Liposomal VD3.
A completely randomized design was employed for this study. The mice were randomly allocated into 6 groups, with 6 animals (n = 6) in each group. The groups were specifically defined as follows: Control (K): Sham-operated (non-OVX) mice receiving the vehicle (coconut oil), Negative Control (P-): OVX mice receiving the vehicle (coconut oil), Positive Control (P+): OVX mice receiving 2000 IU of regular VD3 (NOW Foods, USA) Treatment (P1): OVX mice receiving 1,000 IU of Liposomal VD3 (Upnourish, USA), Treatment (P2): OVX mice receiving 2,000 IU of Liposomal VD3, Treatment (P3): OVX mice receiving 4,000 IU of Liposomal VD3. The experimental dosages were derived from standard human daily supplementation regimens of 1,000, 2,000, and 4,000 IU. These human reference doses were normalized to body weight and pharmacologically scaled to determine the equivalent doses for mice (in IU/kg). This established conversion process was employed to ensure that the dosages administered per-orally were analogous to the intended human intakes. After the recovery and validation period, the 2-month treatment regimen was initiated. Formulations were administered daily via oral gavage (using a feeding needle). The superior bioavailability of liposomal formulations [8,33]. After the recovery and validation period, a 2-month treatment regimen was initiated. VD3 was prepared in various International Unit (IU) concentrations by dissolving it in coconut oil. This preparation was administered to the mice daily via oral gavage (using a feeding needle).
Serum collection and estrogen analysis
At the end of the treatment period, the mice were euthanized by chloroform inhalation. Blood was collected via cardiac puncture into micro-tubes and allowed to clot for 2 h at room temperature. The samples were then centrifuged at 3,000 rpm for 15 min at 37 °C. The resulting serum was collected and stored at −20 °C until analysis. Serum estrogen levels were quantified using a competitive Enzyme-Linked Immunosorbent Assay (ELISA) kit (BT-Lab, Cat. No. EA0012Mo) according to the manufacturer’s protocol.
Femur collection and histological analysis
The right femur from each mouse was dissected, cleaned of soft tissue, and fixed in a Neutral Buffer Formalin solution. The bones were then decalcified in 10% EDTA for 5 days. Following decalsification, the femurs underwent dehydration through a graded series of ethanol (70%, 80%, 90% and absolute), cleared with xylene, and embedded in paraffin blocks. The paraffin-embedded tissues were sectioned at a 5 µm thickness using a micro-tome. The sections were stained with Hematoxylin and Eosin (H&E) for histological examination.
Quantification of calcium and phosphorus levels
For the quantification of bone minerals, the excised femur bones were first cleaned of adherent soft tissue, dried in an oven at 100 °C to a constant weight, and then ashed in a muffle furnace at 700 °C for 6 h. A known mass of the resulting bone ash was prepared for analysis by dissolving it in a solution containing 20 mL of deionized water, 0.5 mL of 65% concentrated nitric acid (HNO3), and 3 mL of 32% concentrated hydrochloric acid (HCl), followed by heating until the solution became clear. After cooling, the digestate was quantitatively transferred to a 100 mL volumetric flask and diluted to the mark with deionized water. Calcium content was then determined using an Atomic Absorption Spectrophotometer (AAS) at a wavelength of 422.7 nm, with the final concentration (ppm) calculated by comparing sample absorbance against a standard curve prepared from calcium chloride (CaCl2) solutions. For phosphorus analysis, a 1 mL aliquot from the same acid-digested solution was treated with 10 mL of deionized water, 1 mL of 5% ammonium molybdate reagent, and 50 mg of ascorbic acid. This mixture was heated to boiling, cooled, diluted in a 100 mL volumetric flask, and incubated for 10 min to allow for the development of a blue color complex. Phosphorus concentration (ppm) was measured using a visible spectrophotometer at a wavelength of 830 nm and determined by comparison to a standard curve prepared from potassium dihydrogen phosphate (KH2PO4)
Histomorphometry analysis
Histological images of the epiphyseal region of the femur were captured using a microscope equipped with an Optilab camera at 400× magnification. The images were analyzed using ImageJ software (National Institutes of Health, USA). To prevent operator bias, all histological slides were coded, and the image analysis was performed by an investigator blinded to the treatment group allocations. The following parameters were quantified:
Trabecular Area: The percentage of the total area occupied by trabecular bone was measured.
Cell Counts: The number of osteoblasts, osteoclasts, and osteocytes were counted in 4 different fields of view for each sample.
Statistical analysis
Data were analyzed using GraphPad Prism software. Descriptive statistics were used to calculate the mean and standard deviation (SD). The normality of the data was assessed using the Shapiro-Wilk test, and homogeneity of variances was tested with Levene’s test. For normally distributed and homogeneous data, a one-way ANOVA followed by Tukey’s post-hoc test was used. For data that were normally distributed but not homogeneous, Welch’s ANOVA with a Dunnett T3 post-hoc test was performed. For non-normally distributed data, the Kruskal-Wallis test followed by Dunn’s multiple comparison test was used. A p-value of < 0.05 was considered statistically significant.
Results and discussion
Liposomal VD3 restores serum estrogen levels in ovariectomized mice
Based on the graph in Figure 1, the ovariectomy (OVX) procedure resulted in a significant decrease of 78.5% in serum estrogen concentration in the negative control group (P-), with a mean level of 123.2 ± 44.52 ng/L compared to 572.1 ± 211.8 ng/L in the normal control group (K) (p < 0.0001). Supplementation with liposomal VD3 showed a dose-dependent restorative effect. Doses of 2000 IU (P2) and 4000 IU (P3) significantly increased serum estrogen concentrations to 222.5 ± 89.22 ng/L (an 80.6% increase vs P-) and 271.8 ± 78.69 ng/L (a 120.6% increase vs P-), respectively (both p < 0.001 vs P-). This efficacy was superior to the conventional VD3 treatment (P+), which failed to significantly restore estrogen levels (114.9 ± 39.60 ng/L), a value that was not significantly different from the P- group (p > 0.05).
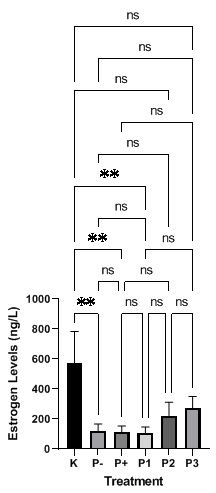
Figure 1 The effect of liposomal VD3 on estrogen levels in ovariectomized mice. The bar charts illustrate estrogen concentrations (ng/L) across different treatment groups. The experimental groups are defined as follows: K (sham-operated control), P- (ovariectomized negative control), P+ (ovariectomized mice treated with standard VD3), P1 (ovariectomized mice treated with 1,000 IU liposomal VD3), P2 (ovariectomized mice treated with 2,000 IU liposomal VD3), and P3 (ovariectomized mice treated with 4,000 IU liposomal VD3). Data are presented as mean ± standard deviation (SD). Data were analyzed using the Kruskal-Wallis test followed by Dunn’s multiple comparison test, as normality was not assumed (Shapiro-Wilk test, p < 0.05). Statistical significance between groups is denoted by asterisks (p < 0.05; **p < 0.001), while ‘ns’ indicates a non-significant difference.
Serum calcium and phosphorus levels
Based on the graph in Figure 2, all VD3 treatment groups (P+, P1, P2 and P3) significantly increased both calcium (p < 0.001) and phosphorus levels (p < 0.0001 for liposome; p < 0.001 for standard) compared to the negative control group (P-). A primary finding is the superiority of the liposome formulation in phosphorus modulation; all groups receiving liposomal VD3 (P1, P2 and P3) exhibited statistically significantly higher phosphorus levels than the group receiving the standard formulation (P+) (p < 0.05, p < 0.01, and p < 0.001, respectively). However, this advantage was not observed in calcium metabolism, where the efficacy of liposomal VD3 was statistically equivalent to the standard formulation (p > 0.05).
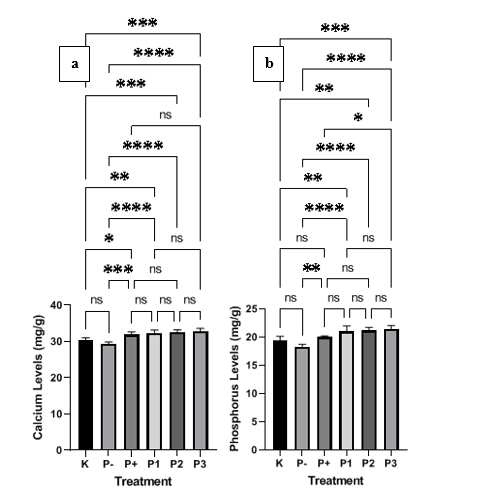
Figure 2 Effect of liposomal VD3 on bone mineral content in ovariectomized mice. The bar charts display the concentration (mg/g) of (a) Calcium and (b) Phosphorus across different treatment groups. The experimental groups are defined as: K (sham-operated control), P- (ovariectomized negative control), P+ (ovariectomized mice treated with standard VD3), P1 (ovariectomized mice treated with 1,000 IU liposomal VD3), P2 (ovariectomized mice treated with 2,000 IU liposomal VD3), and P3 (ovariectomized mice treated with 4,000 IU liposomal VD3). Data are presented as mean ± standard deviation (SD). Statistical significance between groups is denoted by asterisks: p < 0.05, *p < 0.01, **p < 0.001, and ***p < 0.0001. Data for calcium and phosphorus levels were analyzed using a 1-way ANOVA followed by Tukey’s post-hoc test, as data were normally distributed and variances were homogeneous. ‘ns’ indicates a non-significant difference.
Effect of liposomal VD3 on trabecular bone area
Quantitative analysis of the femoral trabecular area (Figure 3) revealed a statistically highly significant difference among treatment groups (Welch’s ANOVA, p < 0.0001). The OVX procedure was confirmed to cause a significant bone mass reduction of 35.8% in the negative control group (P-) to 2,776 ± 226.0 μm², which was significantly lower than the normal control group (K) (4,322 ± 911.8 μm²) (p < 0.0001).
Administration of conventional VD3 (P+) was able to significantly increase the trabecular area by 26.2% to 3,503 ± 168.1 μm², compared to the P- group (p < 0.01). However, its efficacy was sub-optimal compared to the liposome treatments. The liposomal VD3 2000 IU (P2) treatment group showed the most optimal results. The trabecular area in the P2 group reached 4,221 ± 447.6 μm², representing a 52.0% increase from the P- group (p < 0.001). This level successfully restored the bone condition to 97.7% of the normal control level (K), and there was no statistically significant difference (p > 0.05) between P2 and K. The 4,000 IU dose (P3) also demonstrated a significant increase of 41.9% (3,941 ± 472.1 μm²) compared to P- (p < 0.001).
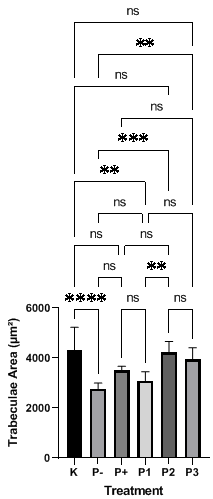
Figure 3 Histomorphometry analysis of trabecular area in ovariectomized mice femur following liposomal VD3 treatment. The bar charts illustrate the trabecular bone area (µm²) measured. The treatment groups are defined as: K (sham-operated control), P- (ovariectomized negative control), P+ (ovariectomized mice treated with standard VD3), P1 (ovariectomized mice treated with 1,000 IU liposomal VD3), P2 (ovariectomized mice treated with 2,000 IU liposomal VD3), and P3 (ovariectomized mice treated with 4,000 IU liposomal VD3). All values are presented as mean ± standard deviation (SD). Statistical significance between groups is denoted by asterisks: p < 0.05, *p < 0.01, **p < 0.001, and ***p < 0.0001. ‘ns’ indicates a non-significant difference.
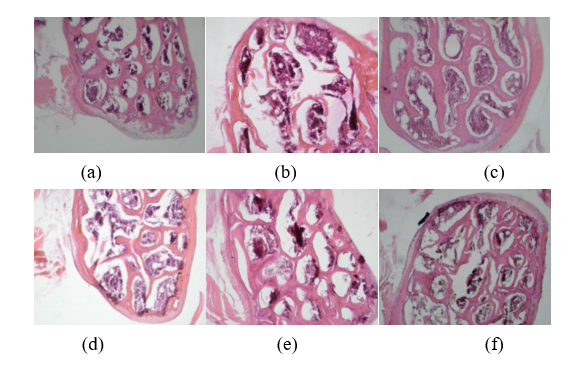
Figure 4 Histological Analysis of Trabecular Bone Femur Ovariectomized Mice Microarchitecture with Hematoxylin and Eosin (H&E) Staining. Representative micrographs of the bone tissue from each experimental group. (a) The sham-operated control group (K) showing a dense, thick, and well-connected trabecular network. (b) The ovariectomized negative control group (P-) exhibiting significant bone loss, characterized by thin, disconnected trabeculae and enlarged bone marrow spaces. (c) The positive control group (P+) treated with standard VD3 shows slight improvement compared to P-. (d) - (f) Groups treated with 1,000 IU (P1), 2,000 IU (P2), and 4,000 IU (P3) of liposomal VD3, respectively, demonstrate a progressive, dose-dependent restoration of trabecular bone structure and density. All images were taken at 400× magnification.
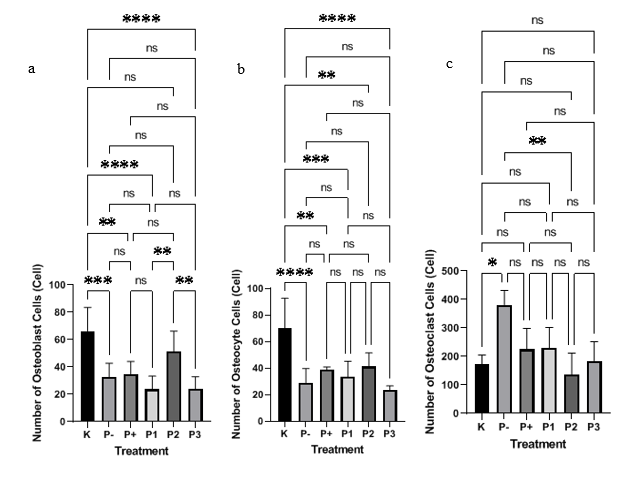 Figure
5
Quantitative analysis of bone cell populations in ovariectomized
mice treated with liposomal VD3. The bar charts represent the cell
counts for (A) Osteoblasts (bone-forming cells), (B) Osteoclasts
(bone-resorbing cells), and (C) Osteocytes (mature bone cells)
across the different experimental groups. The groups are defined as:
K (sham-operated control), P- (ovariectomized negative control), P+
(ovariectomized mice treated with standard VD3), P1 (ovariectomized
mice treated with 1,000 IU liposomal VD3), P2 (ovariectomized mice
treated with 2,000 IU liposomal VD3), and P3 (ovariectomized mice
treated with 4,000 IU liposomal VD3). All data are expressed as mean
cell number ± standard deviation (SD). Statistical significance
between groups is denoted by asterisks: p
< 0.05, *p
< 0.01, **p
< 0.001, and ***p
< 0.0001. ‘ns’ indicates a non-significant difference.
Figure
5
Quantitative analysis of bone cell populations in ovariectomized
mice treated with liposomal VD3. The bar charts represent the cell
counts for (A) Osteoblasts (bone-forming cells), (B) Osteoclasts
(bone-resorbing cells), and (C) Osteocytes (mature bone cells)
across the different experimental groups. The groups are defined as:
K (sham-operated control), P- (ovariectomized negative control), P+
(ovariectomized mice treated with standard VD3), P1 (ovariectomized
mice treated with 1,000 IU liposomal VD3), P2 (ovariectomized mice
treated with 2,000 IU liposomal VD3), and P3 (ovariectomized mice
treated with 4,000 IU liposomal VD3). All data are expressed as mean
cell number ± standard deviation (SD). Statistical significance
between groups is denoted by asterisks: p
< 0.05, *p
< 0.01, **p
< 0.001, and ***p
< 0.0001. ‘ns’ indicates a non-significant difference.
Histomorphometry analysis (Figure 5) revealed that OVX significantly altered bone cell populations. Osteoclasts (Figure 5(c)): The number of osteoclasts (bone-resorbing cells) in the P- group was significantly increased (p < 0.05) compared to the normal control (K). All VD3 interventions showed a suppressive trend against this increase. However, only the liposomal VD3 2000 IU (P2) intervention succeeded in statistically significantly (p < 0.01 vs P-) suppressing the osteoclast count, reducing it to the lowest level (136.6 ± 75.09 cells), which was equivalent to the normal control level (p > 0.05 vs K).
Osteoblasts (Figure 5(b)): Conversely, the number of osteoblasts (bone-forming cells) markedly decreased in the P- group (p < 0.001 vs K). Only the liposomal VD3 2000 IU (P2) treatment succeeded in significantly increasing the osteoblast population (51.20 ± 15.07 cells; p < 0.01 vs P-), reaching a level comparable to the normal control group (K) (p > 0.05 vs K). Other interventions, including conventional VD3 (P+), did not show significant improvement in osteoblast numbers (p > 0.05 vs P-).
Osteocytes (Figure 5(a)): The osteocyte count also significantly decreased in the P- group (p < 0.0001 vs K). Treatment with conventional VD3 (P+), as well as liposomal doses of 1,000 IU (P1) and 2,000 IU (P2), successfully restored a portion of the osteocyte count, but the count was still significantly lower than the normal control (K) (p < 0.01, p < 0.001, and p < 0.01, respectively). The highest dose (P3) resulted in the lowest osteocyte count, which was also significantly lower than the normal control (p < 0.0001).
The present study successfully validated the use of an OVX mouse model to simulate menopausal conditions, as confirmed by the drastic reduction in estrogen levels observed in the negative control group (P-). Against this established baseline, the primary finding demonstrates that VD3 supplementation via a liposomal delivery system is significantly superior in increasing estrogen levels compared to conventional VD3. This advantage, particularly evident at higher doses of 2,000 IU (P2) and 4,000 IU (P3), is attributable to the fundamental mechanism of liposomal encapsulation, which is proven to enhance the bioavailability and efficiency of VD3 absorption into the circulatory system [8]. This enhanced bioavailability underpins the series of physiological effects observed.
The superiority of the liposomal formulation in restoring estrogen is supported by several mechanisms. Firstly, VD3 possesses intrinsic estrogenic activity, which has been shown to regulate the estrous cycle and increase uterine weight, an indicator of estrogenic effects [11]. With higher bioavailability, this effect becomes more pronounced. Secondly, VD3 plays a crucial role in estrogen metabolism, where its active metabolite, 1,25-dihydroxyVD3, can interact with estrogen receptors and potentially stimulate endogenous estrogen production [13]. The superior absorption of the liposomal formulation ensures higher levels of this active metabolite. Lastly, VD3’s antioxidant and anti-inflammatory properties [13,14] can improve the microenvironment within adrenal tissues, making it more conducive to steroidogenesis, including estrogen synthesis, to compensate for the loss of ovarian function.
The benefits of this enhanced bioavailability also extend significantly to bone health parameters. The results indicate that the liposomal VD3 formulation was superior in improving bone trabecular area compared to conventional VD3. The increased availability of VD3 [5,15] directly optimizes its anabolic effects on bone, such as stimulating an increase in trabecular number and thickness [16]. Furthermore, a more bioavailable VD3 can more effectively suppress the activity of osteoclasts the cells responsible for bone resorption thereby preserving trabecular structure [17,18]. This process is supported by the more efficient absorption of calcium and phosphate required for robust bone mineralization [16,19].
These findings are further corroborated at the cellular level. The administration of liposomal VD3 was shown to ameliorate bone remodeling parameters, evidenced by a significant decrease in osteoclast count alongside a tendency towards an increase in osteoblast and osteocyte numbers. The superior delivery efficiency of the liposomal system [20,121] allows VD3 to more effectively stimulate the differentiation and activity of osteoblasts, the bone-forming cells [22,23], and to suppress bone resorption by regulating the expression of RANKL, a key cytokine for osteoclast activation [24,25]. Collectively, the liposomal technology fosters a more favorable balance between bone formation and resorption, which is crucial for preventing bone degradation in postmenopausal conditions [6,26].
In contrast to its demonstrated superiority in hormonal and bone architectural parameters, a divergent finding emerged concerning serum mineral homeostasis. The analysis revealed that while all forms of VD3 supplementation successfully increased calcium and phosphorus levels compared to the untreated negative control, there was no statistically significant difference in efficacy between liposomal and conventional VD3. Even at the highest dose (P3), the liposomal formulation conferred no additional advantage in elevating serum mineral levels.
This phenomenon suggests the influence of limiting factors within the biological system. A multifactorial explanation can be proposed. The tightly regulated nature of calcium homeostasis may have reached a response ceiling or saturation point at the tested dosages, where further increases in bioavailability no longer translate into proportional increases in serum mineral levels [27]. Moreover, the efficacy of supplementation may be constrained by the rate-limiting enzymatic conversion of VD3 to its active metabolite, a step unaffected by the delivery system [28].
Other factors, such as the challenging physiological milieu of the OVX animal model [29] and the primary objective of liposomal encapsulation, which may prioritize stability and toxicity reduction over maximizing physiological effects [30], likely contribute to this observation. Thus, this study underscores that the superiority of the liposomal formulation is most pronounced in its long-term impacts on hormonal regulation and bone architectural remodeling rather than on the acute modulation of serum mineral levels.
A primary finding of this study highlights a non-linear, dose-dependent response to VD3 supplementation on bone remodeling in the OVX animal model. Our data consistently demonstrate that the 2000 IU dose, particularly when liposomally formulated, provides superior efficacy in achieving balanced bone remodeling. This dosage appears to achieve an optimal threshold for VD Receptor (VDR) activation, which is essential for promoting osteoblast differentiation while concurrently suppressing osteoclast activity. This mechanism is likely mediated by the upregulation of osteoprotegerin (OPG) and suppression of RANKL, which shifts the remodeling balance toward bone formation [24,26,34,35].
Significantly, and contrary to the hypothesis that a higher dose would confer greater benefit, the 4,000 IU dose failed to demonstrate additional superiority. Conversely, our data align with studies indicating a plateau or even a decline in efficacy [26,36]. This phenomenon may be explained by the activation of negative feedback mechanisms at supraphysiological doses. Excessive VDR activation at the 4000 IU dose has been shown to trigger suppression of the primary osteogenic transcription factor, Runx2, which directly inhibits osteoblast maturation [37,38]. Furthermore, this “overactivation” may disrupt the sensitive OPG/RANKL signaling axis, potentially reducing the antiresorptive effect or even paradoxically promoting osteoclastogenesis [39].
Another critical aspect of our study is the role of the delivery vehicle. The liposomal formulation demonstrated enhanced bioavailability, yet this advantage was most pronounced at the 2,000 IU dose [40-42]. Liposomal encapsulation is known to improve intestinal absorption and protect VD3 from degradation [33,43-45]. The fact that this superiority diminished at the 4,000 IU dose [46] suggests a possible saturation of the intestinal absorption mechanisms. This implies that for therapeutic efficacy in postmenopausal conditions (as mimicked by the OVX model), a more effective strategy is to optimize bioavailability (via liposomes) at a moderate dose (2000 IU), rather than merely increasing the total dosage (4,000 IU).
These findings must be interpreted within the context of the study’s limitations. Reliance on the OVX rodent model, while a gold standard [47], necessitates caution in direct extrapolation to human physiology. There is also variability in site-specific bone responses (e.g., femur vs. maxilla) that cannot be overlooked [5,48], as well as limited pharmacokinetic data directly comparing the liposomal and regular formulations at both dosages [49, 50]. Furthermore, the risk of high-dose toxicity, such as altered metabolite profiles [51,52] and potential vitamin K depletion a critical cofactor for matrix protein carboxylation remains a significant concern [53].
Conclusions
This study concludes that the liposomal formulation, particularly at a dose of 2,000 IU, is notably more effective in ameliorating bone loss, as evidenced by the significant restoration of femoral trabecular area to near-normal levels. Furthermore, the liposomal preparation showed a superior capacity to restore serum estrogen concentrations, an effect not observed with conventional VD3. This superior hormonal effect is likely attributable to the liposomal delivery significantly enhancing VD3 bioavailability. This increased bioavailability is hypothesized to potentiate a dual mechanism: enabling the intrinsic estrogenic activity of VD3 to exert a more pronounced effect and stimulating endogenous steroidogenesis, possibly in adrenal tissues, to compensate for the loss of ovarian production. These structural and hormonal improvements are underpinned by a more effective modulation of bone cell populations, including a significant reduction in osteoclast numbers and a unique ability to restore the osteoblast population at the 2000 IU dosage.
Acknowledgements
This study was carried out in collaboration with a Postgraduate Research-Master's Thesis Research 2024 (No. 2440/B/UN3.LPPM/PT.01.03/2025). We are sincerely grateful to Pusvetma Surabaya for providing the experimental animals, and to the Laboratory of the Faculty of Science and Technology, Universitas Airlangga, for their invaluable support throughout the research process. Their contributions were instrumental to the successful completion of this study contributions
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Generative AI in Scientific Writing
The authors acknowledge the use of generative AI tools (e.g., Gemini Ai) in the preparation of this manuscript, specifically for grammar correction. No content generation or data interpretation was performed by AI. The authors take full responsibility for the content and conclusions of this work.
CRediT Author Statement
Sri Puji Astuti Wahyuningsih: Conceptualization; Methodology; Data curation; Validation; Investigation; Supervision; Project administration. Ari Budi Suryawinata: Formal analysis; Investigation; Visualization; Writing - Original draft preparation Writing - Review & Editing. Listyani Suhargo: Conceptualization; Methodology; Data curation; Validation; Supervision; Visualization; Investigation; Project administration. Lionel In Lian Aun: Supervision. Dwi Roudlotul Firda: Formal analysis; Investigation; Visualization; Writing - Original draft preparation Writing - Review & Editing. Alvin Oktaviana Puspitasari: Formal analysis; Investigation; Visualization; Writing - Original draft preparation Writing - Review & Editing.
References
[1] S Kavosian, G Amirbozorgi, T Shomali, H Hashempoor, M Khorshidi, AA Ahmadi, H Haghi-Aminjan, M Asouri, A Alizadeh, R Ataee, SMB Hashemi and H Akhavan Niaki. Association between vitamin D receptor gene Fok1 polymorphism and bone mineral density in a population of postmenopausal women with osteoporosis. Journal of Mazandaran University of Medical Sciences 2014; 24(118), 73-82.
[2] EF Hamzah, MF Smaism and AH Al-Algawy. Investigation of the role of VDR C>A rs7975232 SNP in the incidence of osteoporosis in Iraqi women. International Journal of Drug Delivery Technology 2021; 11(4), 1190-1194.
[3] GA Reinhart. Vitamin D analogs: Novel therapeutic agents for cardiovascular disease?. Current Opinion in Investigational Drugs 2004; 5(9), 947-951.
[4] BE Kream and AC Lichtler. Vitamin D regulation of type I collagen expression in bone. In: D Feldman, JW Pike and JS Adams (Eds.). Academic Press, San Diego, 2011, p. 403-409.
[5] S Körmendi, B Vecsei, S Ambrus, K Orhan and C Dobó-Nagy. Evaluation of the effect of vitamin D3 on mandibular condyles in an ovariectomized mouse model: A micro-CT study. BMC Oral Health 2021; 21(1), 627.
[6] A Kaspiris, ID Iliopoulos, E Chronopoulos, OD Savvidou, E Vasiliadis and S Kotsela. Implication of vitamin D deficiency in altered osseous remodeling and development of bone marrow edema syndrome. In: J Andělová (Ed.). The essential guide to vitamin D. Nova Science Publishers, New York, 2019, p. 109-122.
[7] M Yamaguchi and MN Weitzmann. High dose 1,25(OH)2D3 inhibits osteoblast mineralization in vitro. International Journal of Molecular Medicine 2012; 29(5), 934-938.
[8] V Yanachkova, R Staynova, S Stoev and E Naseva. Benefits of using a microencapsulated vitamin D delivery system in women with polycystic ovary syndrome. European Journal of Hospital Pharmacy 2023; 30(5), 284-287.
[9] SH Hong, JE Lee, SM An, YY Shin, DY Hwang, SY Yang, SK Cho and BS An. Effect of vitamin D3 on biosynthesis of estrogen in porcine granulosa cells via modulation of steroidogenic enzymes.Toxicological Research 2017; 33(1), 49-54.
[10] S Bakhshalizadeh, F Amidi, R Shirazi and MS Nashtaei. Vitamin D3 regulates steroidogenesis in granulosa cells through AMP-activated protein kinase (AMPK) activation in a mouse model of polycystic ovary syndrome. Cell Biochemistry and Function 2018; 36(4), 183-193.
[11] EC Mbegbu, CT Mbah, IR Obidike and LO Aka. Oral supplementation of VD3 regulates estrous cycle due to its estrogenic effects in rats. Pakistan Veterinary Journal 2020; 40(4), 585-590.
[12] J Gattineni, P Alphonse, Q Zhang, N Mathews, CM Bates and M Baum. Regulation of renal phosphate transport by FGF23 is mediated by FGFR1 and FGFR4. American Journal of Physiology. Renal Physiology 2014; 306(3), F351-F358.
[13] NA Mohammed, NF El-Malkey, AA Ibrahim and DM Abdullah. VD3 supplementation ameliorates ovariectomy-induced cardiac apoptotic and structural changes in adult albino rats. Canadian Journal of Physiology and Pharmacology 2019; 97(7), 647-654.
[14] T Vulcan, TS Suciu, LM Lenghel, VA Toma, N Decea and R Moldovan. The impact of VD3 administration and of high fat diet on oxidative stress and inflammation in experimentally induced polycystic ovary syndrome. Medical and Pharmacy Reports 2024; 97(4), 516-627.
[15] CY Lai, JY Yang, S Rayalam, MA Della-Fera, S Ambati and RD Lewis. Preventing bone loss and weight gain with combinations of vitamin D and phytochemicals. Journal of Medicinal Food 2011; 14(11), 1352-1362.
[16] M Li, DR Healy, HA Simmons, HZ Ke and DD Thompson. Alfacalcidol restores cancellous bone in ovariectomized rats. Journal of Musculoskeletal & Neuronal Interactions 2003; 3(1), 39-46.
[17] H Hongo, M Sasaki, T Hasegawa and N Amizuka. Clinical application of active VD3 and its analogues for bone diseases. Clinical Calcium 2011; 21(11), 63-70.
[18] M Noa, S Mendoza, R Mas, N Mendoza and E Goicochea. Long-term effects of D-003, a mixture of high molecular weight acids from sugarcane wax, on bones of ovariectomized rats: A one year study. Pharmazie 2008; 63(6), 486-488.
[19] C Galeşanu, C Ciubotariu, G Melnic and MR Galeşanu. Postmenopausal osteoporosis. Digital Rx radiogrammetry in the diagnosis and follow-up of treatment with alfacalcidol. Revista Medico-Chirurgicala a Societatii de Medici si Naturalisti din Iasi 2006; 110(4), 833-841.
[20] M Demirbilek, N Laçin Türkoglu, S Aktürk and C Akça. VitD3-loaded solid lipid nanoparticles: stability, cytotoxicity and cytokine levels. Journal of Microencapsulation 2017; 34(5), 454-462.
[21] N Ignjatović, V Uskoković, Z Ajduković and D Uskoković. Multifunctional hydroxyapatite and poly(D,L-lactide-co-glycolide) nanoparticles for the local delivery of cholecalciferol. Materials Science and Engineering: C 2013; 33(2), 943-950.
[22] RC Pereira, IB Salusky, RE Bowen, EG Freymiller and K Wesseling-Perry. Vitamin D sterols increase FGF23 expression by stimulating osteoblast and osteocyte maturation in CKD bone. Bone 2019; 127, 626-634.
[23] T Rolvien, M Krause, A Jeschke, T Yorgan, K Püschel and T Schinke. Vitamin D regulates osteocyte survival and perilacunar remodeling in human and murine bone. Bone 2017; 103, 78-87.
[24] Y Nakamichi, N Udagawa, T Suda and N Takahashi. The Journal of Steroid Biochemistry and Molecular Biology 2018; 177, 70-76.
[25] Y Nakamichi, N Takahashi, T Suda and N Udagawa. Osteoclastogenesis and vitamin D. In: D Feldman and JW Pike (Eds.). Feldman and Pike’s vitamin D. Academic Press, Massachusetts, 2024, p. 395-408.
[26] PR Ebeling. Vitamin D and osteoporosis. In: M Hewison, R Bouillon, E Giovannucci, D Goltzman, M Meyer and J Welsh (Eds.). Feldman and Pike’s vitamin D: Volume two: Disease and therapeutics. Mosby, Missouri, 2024, p. 411-433.
[27] JF Aloia, R Dhaliwal, A Shieh, M Mikhail, M Fazzari and L Ragolia. Vitamin D supplementation increases calcium absorption without a threshold effect. American Journal of Clinical Nutrition 2014; 99(3), 624-631.
[28] K Uenishi, M Tokiwa, S Kato and M Shiraki. Stimulation of intestinal calcium absorption by orally administrated VD3 compounds: A prospective open-label randomized trial in osteoporosis. Osteoporosis International 2018; 29(3), 723-732.
[29] RHM Salama, SS Ali, THM Salama, MA Almged, TA Alsanory and AA Alsanory. Dietary effects of nanopowder eggshells on mineral contents, bone turnover biomarkers, and regulators of bone resorption in healthy rats and ovariectomy-induced osteoporosis rat model. Applied Biochemistry and Biotechnology 2023; 195(8), 5034-5052.
[30] M Ezcurra-Hualde, S Zalba, Á Bella, L Arrizabalaga, A Risson, R García-Fuentes, C Gomar, N Ardaiz, V Belsue, D Ruiz-Guillamon, A Serrano-Alcaide, A Sargado, F Aranda, MJ Garrido and P Berraondo. Liposomal encapsulation of cholecalciferol mitigates in vivo toxicity and delays tumor growth. Frontiers in Immunology 2025; 16, 1529007.
[31] Y Tayem, R Alotaibi, R Hozayen and A Hassan. Therapeutic regimens for vitamin D deficiency in postmenopausal women: A systematic review. Przeglad Menopauzalny 2019; 18(1), 57-62.
[32] IY Torshin, OA Gromova and NK Tetruashvili. Concerning effective and safe dosages of vitamin D3: Mega-analysis of clinically effective studies as a basis for actual evidence. Meditsinskiy Sovet Medical Council 2021; 13, 112-120.
[33] P Dałek, D Drabik, H Wołczańska, A Foryś, M Jagas, N Jędruchniewicz, M Przybyło, W Witkiewiczand and M Langner. Bioavailability by design - vitamin D3 liposomal delivery vehicles. Nanomedicine 2022; 43, 102552.
[34] Y Nakamichi and N Takahashi. Current topics on vitamin D. The role of active forms of vitamin D in regulation of bone remodeling. Clinical Calcium 2015; 25, 395-402.
[35] Y Nakamichi, Z Liu, T Mori, Z He, H Yasuda, N Takahashi and N Udagawa. The vitamin D receptor in osteoblastic cells but not secreted parathyroid hormone is crucial for soft tissue calcification induced by the proresorptive activity of 1,25(OH)2D3. The Journal of Steroid Biochemistry and Molecular Biology 2023; 232, 106351.
[36] T Hasegawa, T Yamamoto, S Sakai, Y Miyamoto, H Hongo, Z Qiu, M Abe, S Takeda, K Oda, PHL de Freitas, M Li, K Endo and N Amizuka. Histological effects of the combined administration of Eldecalcitol and a parathyroid hormone in the metaphyseal trabeculae of ovariectomized rats. The Journal of Histochemistry and Cytochemistry: Official Journal of the Histochemistry Society 2019; 67(3), 169-184.
[37] H Drissi, A Pouliot, C Koolloos, JL Stein, JB Lian, GS Stein and AJ van Wijnen. 1,25-(OH)2-vitamin D3 suppresses the bone-related Runx2/Cbfa1 gene promoter. Experimental Cell Research 2002; 274(2), 323-333.
[38] D Yang, PH Anderson, AR Wijenayaka, KR Barratt, R Triliana, CJM Stapledon, H Zhou, DM Findlay, HA Morris and GJ Atkins. Both ligand and VDR expression levels critically determine the effect of 1α,25-dihydroxyvitamin-D3 on osteoblast differentiation. The Journal of Steroid Biochemistry and Molecular Biology 2018; 177, 83-90.
[39] PA Baldock, GP Thomas, JM Hodge, SU Baker, U Dressel, PD O’Loughlin, GC Nicholson, KH Briffa, JA Eisman and EM Gardiner. Vitamin D action and regulation of bone remodeling: Suppression of osteoclastogenesis by the mature osteoblast. Journal of Bone and Mineral Research: The Official Journal of the American Society for Bone and Mineral Research 2006; 21(10), 1618-1626.
[40] A Fratter and M Pellizzato. Novel micellar system for vitamin D3 oral delivery: Assessment of enteric absorption through a digestion-like in vitro model. Journal of Drug Delivery Science and Technology 2020; 59, 101840.
[41] RK Marwaha, T Dev, A Mittal, K Mani, A Narang, P Arora, A Singh, A Chadha, N Dang, M Goel, VK Sharma and G Sethuraman. A randomised controlled trial comparing the efficacy of micellised and fat-soluble vitamin D3 supplementation in healthy adults. The British Journal of Nutrition 2019; 121(8), 859-865.
[42] J Solnier, C Chang, Y Zhang, YC Kuo, M Du, YS Roh, J See, J Brix, RJ Gahler, T Green and S Wood. A comparison and safety evaluation of micellar versus standard vitamin D3 oral supplementation in a randomized, double-blind human pilot study. Nutrients 2024; 16, 1573.
[43] G Anjani, R Achmad Maulana, SR Putri, F Fulyani, A Syauqy, DN Afifah, F Ayustaningwarno, RA Kusuma and Z Masruroh. Ameliorative effect of liprotide-encapsulated vitamin D3 on blood glucose, calcium homeostasis, and vitamin D level in a vitamin D and calcium deficient rat model. Frontiers in Nutrition 2025; 12, 1514179.
[44] H Annappa, A Tamatam, I Nallamuthu and K Ranganathan. Formulation of pH-responsive nanoparticles using zein/sodium alginate polymers for enhanced bioavailability of the vitamin D3. International Journal of Biological Macromolecules 2025; 313, 144140.
[45] SJ Park, CV Garcia, GH Shin and JT Kim. Development of nanostructured lipid carriers for the encapsulation and controlled release of vitamin D3. Food Chemistry 2017; 225, 213-219.
[46] J Chen, L Dehabadi, YC Ma and LD Wilson. Development of novel lipid-based formulations for water-soluble vitamin C versus fat-soluble vitamin D3. Bioengineering 2022; 9(12), 819.
[47] YP Kharode, MC Sharp and PV Bodine. Utility of the ovariectomized rat as a model for human osteoporosis in drug discovery. Methods in Molecular Biology 2008; 455, 111-124.
[48] CM Esteves, RM Moraes, FC Gomes, MS Marcondes, GM Lima and AL Anbinder. Ovariectomy-associated changes in interradicular septum and in tibia metaphysis in different observation periods in rats. Pathology, Research and Practice 2015; 211, 125-129.
[49] CH Chang, SJ Yang, TH Young and WC Yao. Effect of co-loaded vitamin D3 on intravenous injectable raloxifene delivery system. Colloids and Surfaces B: Biointerfaces 2025; 246, 114379.
[50] P Sharifan, A Ziaee, S Darroudi, M Rezaie, M Safarian, S Eslami, M Khadem-Rezaiyan, M Tayefi, M Bajgiran, H Ghazizadeh, Z Khorasanchi, M Bagherniya, MA Sardar, G Ferns, H Vatanparast and MG Mobarhan. Effect of low-fat dairy products fortified with 1500IU nano encapsulated vitamin D3 on cardiometabolic indicators in adults with abdominal obesity: A total blinded randomized controlled trial. Current Medical Research and Opinion 2021; 37(4), 579-588.
[51] S Qiu, G Divine and SD Rao. Effect of vitamin D metabolites on bone histomorphometry in healthy black and white women: An attempt to unravel the so-called vitamin D paradox in blacks. Bone Reports 2022; 18, 101650.
[52] A Zittermann, JB Ernst, S Prokop, U Fuchs, J Dreier, J Kuhn, HK Berthold, S Pilz, I Gouni-Berthold and JF Gummert. Vitamin D supplementation and bone turnover in advanced heart failure: The EVITA trial. Osteoporosis International 2018; 29(3), 579-586.
[53] GN Varlıhan, ÖB Ağralı, HS Güngörmek, SD Delipınar, L Kuru and HÖ Özener. Vitamins K2 and D3 enhance guided bone regeneration in rat calvarial bone defects. Journal of Periodontology 2025. https://doi.org/10.1002/jper.11372