
Trends
Sci.
2026;
23(5):
12054
Enhancing the Lipid-Lowering Activity of Apigenin through Nanostructured Lipid Carrier (NLC) Modification
Fajar
Setiawan1,
![]() ,
Nur
Rahayuningsih1,
Maya Nurul Rahma1,2,
,
Nur
Rahayuningsih1,
Maya Nurul Rahma1,2,
Wulan Aprilia Susilawati1, Laras Rizkia Widyastuti1 and Lusi Nurdianti1,*
1Department of Pharmaceutics, Faculty of Pharmacy, Bakti Tunas Husada University, Tasikmalaya 46115, Indonesia
2Department of Pharmaceutics and Pharmaceutical Technology, Padjadjaran University, West Java 45363, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 25 September 2025, Revised: 7 October 2025, Accepted: 14 October 2025, Published: 10 January 2026
Abstract
A total of 2.6 million deaths and 29.7 million Disability-Adjusted Life Years (DALYs) worldwide are caused by hyperlipidemia. Apigenin is known to play a role in cholesterol regulation, but it is limited in terms of solubility and stability. Modification of Nanostructured Lipid Carriers (NLC) is known to increase the bioavailability and effectiveness of hyperlipidemia drugs. This study aims to analyze the potential of NLC drug delivery systems containing apigenin to increase antihyperlipidemia efficacy. Apigenin NLC was formulated using emulsification and melting methods. The formulation was characterized based on particle size, PDI, zeta potential, %EE, and morphology. Subsequently, in vitro release and antihyperlipidemic activity tests were conducted using the photometric method. The apigenin NLC formulation was prepared with a ratio of solid lipid to liquid lipid of 30:70 (F1), 40:60 (F2), and 50:50 (F3). The characterization results showed F3 as the optimal formula with a particle size of 172.30 ± 35.66 nm, polydispersity index of 0.57 ± 0.16, zeta potential of −26.48 ± 0.28 mV, and %EE of 77.8870 ± 0.05%. The NLC modification successfully increased apigenin release by 3-fold compared to unmodified apigenin and the activity of lowering cholesterol levels increased with a decrease in the IC50 value of apigenin’s antihyperlipidemic activity from 132.56 to 101.09 ppm in NLC-Apigenin. This study demonstrates that NLC Apigenin holds promising potential in the treatment of hyperlipidemia.
Keywords: Hyperlipidemia, Apigenin, Nanocarrier, NLC, Release, Cholesterol, Photometric method
Introduction
Hyperlipidemia is a condition that occurs due to an increase in blood lipid levels, including cholesterol, triglycerides, LDL, and HDL. Hyperlipidemia is estimated to cause 2.6 million deaths (4.5% of total deaths) [1] or about 2% of the total disease burden worldwide [2]. Hyperlipidemia can cause atherosclerosis, which leads to cardiovascular diseases such as coronary heart disease, hypertension, and stroke. According to WHO data, these 3 diseases are among the top 10 causes of death globally [3]. Long-term use of medications, such as statins, has the potential to cause side effects such as myalgia and myopathy. In more severe cases, statins can increase the risk of hepatotoxicity, type 2 diabetes, and peripheral
neuropathy [4]. As an alternative, the use of herbal remedies is gaining attention due to their potential to lower lipid levels with significantly lower risks of side effects.
Apigenin is a flavonoid found in celery (Apium graveolens) [5] and has many pharmacological activities [6]. In terms of cholesterol regulation, previous studies have shown that apigenin plays a role in regulating blood lipids. At a dose of 3×10−7 mol/L, apigenin can significantly reduce total cholesterol (TC), triglycerides (TG), and low-density lipoprotein cholesterol (LDL-C) levels in the serum of high-fat model rats [7]. Despite apigenin’s therapeutic potential as an antihyperlipidemic agent, it has limitations because it belongs to BCS (Biopharmaceutical Classification System) Class II. Its low water solubility and slow dissolution rate cause apigenin to have bioavailability issues [8].
Nanostructured Lipid Carriers (NLC) are a type of lipid-based drug delivery system. NLC is a potential drug carrier for increasing the solubility of drugs that are difficult to dissolve in water or lipophilic drugs and increasing their bioavailability [9]. In previous studies, several antihyperlipidemic drugs such as simvastatin. Modification of NLC for simvastatin showed a more significant reduction in cholesterol and LDL-C levels compared to drug suspensions in test animals [10]. In addition to simvastatin, NLC modification was also performed on fenofibrate, and the AUC value of unmodified simvastatin was successfully increased from 68.9 ± 2.9 to 227.6 ± 7.2 h·μ g/mL [11]. Ezetimibe, another antihyperlipidemic drug model, also successfully increased its bioavailability with NLC modification [12]. In this study, the synergistic combination of apigenin with NLC technology is expected to enhance efficacy in lowering cholesterol levels in hyperlipidemia conditions
Materials and methods
Apigenin (Hefei Dielegance Biotechnology Co., Ltd), soy lecithin, stearic acid (Sigma-Aldrich), oleic acid (Sigma-Aldrich), potassium dihydrogen phosphate (KH2PO4) (Sigma-Aldrich), chloroform (Sigma-Aldrich), ethanol (Sigma- Aldrich), Tween 80 (Sigma-Aldrich), cholesterol (Sigma-Aldrich), Lieberman-Burchard reagent.
Preparation of nanostructured lipid carriers apigenin (NLC-apigenin)
The formula for making NLC apigenin is shown in Table 1. Apigenin is dissolved in 95% ethanol, then mixed with stearic acid melted together with oleic acid. This mixture is slowly added drop by drop to the aqueous phase, which consists of a phosphate buffer solution and Tween 80 with a pH of 7.4 at a constant temperature of 85 °C (3 h). Sonication for 20 min, using a cycle of 0.5 s−1 and an amplitude power of 75% [13].
Table 1 Formula of NLC-apigenin.
Composition |
|
Formula |
|
|
F1 |
F2 |
F3 |
||
Apigenin (mg) |
API |
10 |
10 |
10 |
Stearic acid (%) |
Solid Lipid |
30 |
40 |
50 |
Oleic Acid (%) |
Liquid Lipids |
70 |
60 |
50 |
Tween 80 (g) |
Water Phase |
2 |
2 |
2 |
Phosphate Buffer (mL) |
10 |
10 |
10 |
|
Characterization of NLC-apigenin
Particle size was analyzed using a Particle Size Analyzer. NLC-Apigenin was dissolved in distilled water at a ratio of 1:10, then the sample was poured into a flow cell. Zeta potential was analyzed by pouring the sample into a cuvette and storing it in a zetasizer probe [14]. Morphological characteristics were observed using transmission electron microscopy (TEM) set at 200 KV [13]. The % entrapment efficiency (%EE) was determined by placing the sample in a viva spin tube, then centrifuging it using an ultracentrifugator set at a temperature of 4 °C for 60 min at a speed of 12,000 rpm. The supernatant was taken to measure the amount of Apigenin that was not entrapped in NLC using a UV Vis spectrophotometer [15]. The %EE calculation was performed using the formula:
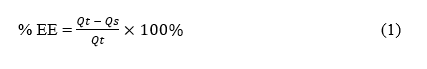
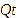 is the theoretical amount of Apigenin added.
is the theoretical amount of Apigenin added.
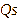 is the amount of Apigenin detected in the supernatant.
is the amount of Apigenin detected in the supernatant.
In vitro drug release
A total of 5 mL of sample and apigenin powder as a reference were each placed in a dialysis bag and then placed in 100 mL phosphate buffer pH 7.4 as the dissolution medium in beaker glass. Stirring was performed at a speed of 50 rpm and the temperature was maintained at 37 ± 0.5 °C. Sample extracts were taken at 5 mL at 0, 1, 2, 4, 6, and 8 h. [16]
In vitro anti-hyperlipidemic studies of NLC-apigenin
The anti-cholesterol activity of NLC-Apigenin was tested using a photometric method. In this test, cholesterol was reacted with Liebermann-Burchard reagent. Samples of apigenin and NLC-Apigenin were tested at various concentrations (15.625, 31.25, 62.5, 125 and 250 mg/L). A total of 4 mL of sample was taken and placed in a covered test tube, then 1 mL of 1,000 ppm cholesterol stock solution was added. The mixture was shaken for 2 min, then 2 mL of Liebermann-Burchard reagent was added. The mixture was incubated for 15 min in dark conditions. Color changes were measured using a UV-Vis spectrophotometer at a wavelength of 620 nm. [17]
Results and discussion
Preparation of nanostructured lipid carriers apigenin (NLC-Apigenin)
The NLC system is a form of nanocarrier because several studies have shown the superiority of NLC compared to other types of nanocarrier drug delivery systems. As a nano lipid carrier, NLC is capable of carrying lipophilic active compounds [18]. In addition, NLC has the advantage of higher drug loading capacity when compared to previous generation lipid carriers, namely solid lipid nanoparticles (SLN) [19].
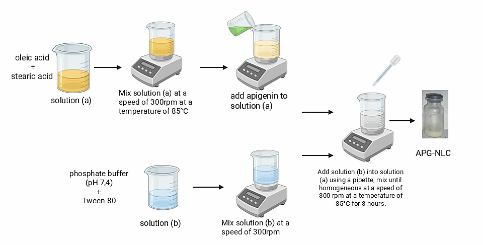
Figure 1 NLC-Apigenin preparation using the emulsification-melting method.
Figure 1 illustrates the procedure for preparing NLC-Apigenin. NLC apigenin was formulated using the emulsification and melting method and followed by sonication. Stearic acid was used as the solid lipid and oleic acid was used as the liquid lipid. The effect of the difference in the ratio between solid lipids and liquid lipids was evaluated during formulation, because the amount of oleic acid plays a role in the viscosity value in the NLC system. The apigenin NLC formulation was carried out with solid lipid and liquid lipid ratios of 30:70 (F1); 40:60 (F2); and 50:50 (F3), while the Tween 80 component was kept constant. Tween 80 was chosen as a surfactant due to its high emulsifying properties for lipid components in the NLC system and its biocompatibility, especially for topical applications [20]. In addition, the melting emulsification technique combined with ultrasonication was chosen because it is easier to perform and eliminates the need for organic solvents [21]. Sonication was then performed to form NLC on a nano scale [22]
Characterization of NLC-apigenin
The formulated NLC-Apigenin formulation was then analyzed, including colloidal properties such as polydispersity index (PDI), particle size, zeta potential, surface and morphology analysis using TEM, adsorption efficiency, and dissolution ability. Each evaluation was performed to ensure the suitability of the formulated product with the predetermined nanocarrier requirements.
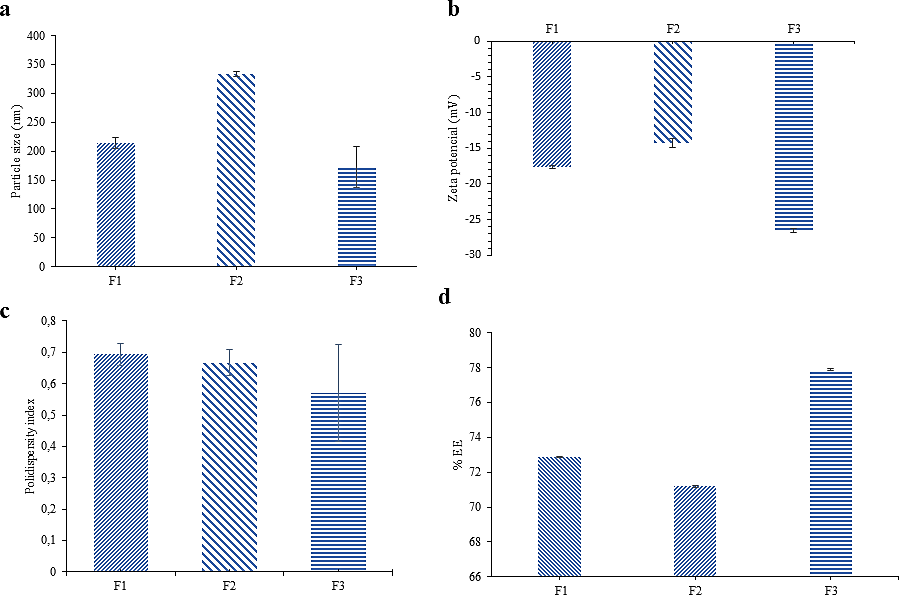
Figure 2 (a) Particle Size (b) Zeta Potential (c) Polydispersity Index (d) %EE of various NLC-Apigenin F1, F2, and F3. Particle size, distribution, and zeta potential were obtained via analysis with a particle size analyzer (PSA). Mean ± standard deviation with n = 3.
Figure 2 shows the results of NLC-Apigenin characterization. The characterization results indicate that NLC- Apigenin particles in all formulations fall within the nano formulation requirements for PDI, particle size, and zeta potential. F1 has a size of 213.90 ± 9.21 nm, F2 has a size of 331 ± 1.015 nm, and F3 has a size of 172.30 ± 35.66 nm (Figure 2(a)). These results meet the size limits for nanocarrier-based delivery systems, which are between 1 - 1,000 nm [23]. The NLC particle sizes obtained are similar to those reported in other studies. In a study conducted by Bonilla et al. [24], NLC-Apigenin obtained by the hot high-pressure homogenization method had a particle size of 146.8 ± 0.8 nm. This slight difference may be due to the different methods used, particularly the use of sonication, which can help break down molecules into the nano range [22].
The zeta potential of formulas F1, F2, and F3 had values of −17.61 ± 0.24, −14.25 ± 0.61 and −26.48 ± 0.28 mV, respectively (Figure 2(b)). The zeta potential values obtained are in line with the ionization of carboxylate groups (−COO−) in stearic acid and oleic acid, which dominate the negative charge on the particle surface [25,26], while Tween 80 is a nonionic stabilizer that has a relatively neutral charge. Zeta potential values in the range below ± 10 mV indicate an unstable formula, ± 10 - 20 mV indicates a relatively stable formula, ± 20 - 30 mV indicates a fairly stable formula, and above ± 30 mV indicates a very stable formula [27].
The lowest polydispersity index value was observed in formula F3 at 0.57 ± 0.16 (Figure 2(c)). These results indicate that the particles have a homogeneous size distribution based on the criteria defining nanoformulations with a polydispersity index below 0.7, suggesting that the preparation has a homogeneous particle size distribution [28]. Formula F3 also showed the best %EE value among all formulas (Figure 2(d)). The %EE values for F1, F2, and F3 were 72.8851 ± 0.02%, 71.2023 ± 0.05%, and 77.8870 ± 0.05%, respectively. This indicates that the F3 formulation of NLC-Apigenin performs better than the F1 and F2 formulations, which was then subjected to morphological analysis, release testing, and antihyperlipidemic activity testing.
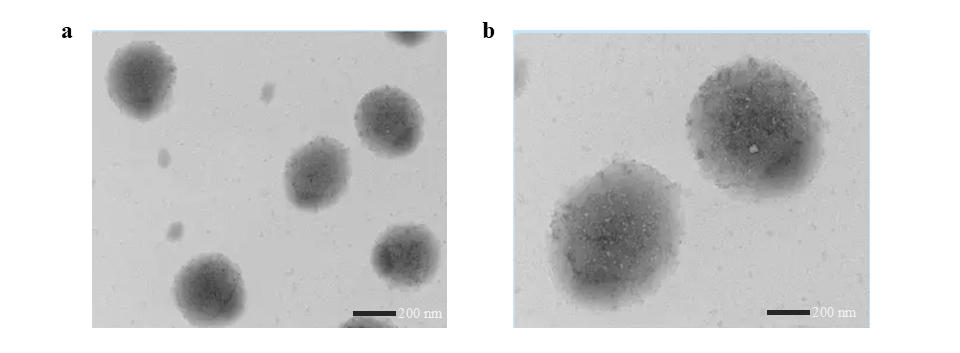
Figure 3 Results of morphological analysis (a) Magnification = ×10.0 K (b) Magnification = ×20.0 K calculated through transmission electron microscopy (TEM) imaging.
Figure 3 shows the results of morphological analysis of NLC-Apigenin. NLC-Apigenin particles exhibit a spherical morphology. The imaging reveals the presence of lipid components inside the NLC particles, surrounded by surfactants on their surface. This finding is consistent with the characteristics of NLC, which usually has a round or spherical shape with lipids located inside the particle [29].
In vitro drug release
The dissolution profile of NLC-Apigenin compared to apigenin powder is illustrated in Figure 4. Apigenin released from the NLC system showed a high release rate since the first sampling time at of 78.78 23 ± 0.82%. The amount of apigenin contained in the medium from NLC-Apigenin was significantly higher than that of unmodified apigenin. NLC-Apigenin showed a dissolution percentage of 84.92 ± 1.37% at the 2nd h, while apigenin powder only reached a dissolution percentage of 25.77 ± 1.10%. At hour 8, NLC-Apigenin dissolved by 92.99 ± 0.25%, while apigenin powder dissolved by 30.78 ± 0.05%. These results conclude that there is an approximately 3-fold increase in the dissolution capacity of apigenin in the NLC system. Thus, the modified NLC system successfully overcomes the shortcomings of apigenin in terms of its low solubility in aqueous media. This increase may be due to the solubilization process influenced by the lipid components in the NLC system [30].
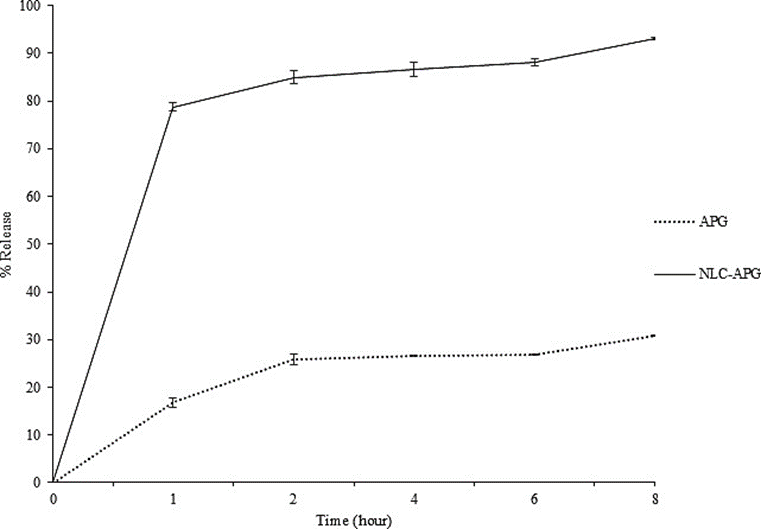
Figure 4 In vitro release of NLC-apigenin (NLC-APG) and unloaded Apigenin (APG) at phosphate buffer saline (pH 7.4). Data are presented as mean ± standard deviation (n = 3).
In vitro anti-hyperlipidemic studies of NLC-apigenin
Based on the IC50 value analysis (Figure 5) obtained from the inhibition percentage data, an overview of the antihyperlipidemic activity effectiveness of each formulation can be seen. The IC50 value represents the concentration required to achieve 50% inhibition, where the lower the value, the higher the level of effectiveness. When compared to apigenin, the NLC-Apigenin formulation showed the most significant antihyperlipidemic activity. This is related to the nature of apigenin, which naturally has low solubility [31], thus limiting its ability to interact with cholesterol [32] Encapsulation in NLC increases the solubility and stability of the active substance [33]. In addition, the very small particle size and homogeneous distribution in NLC also increase the surface area [34], which implies an increase in interaction with cholesterol during the reaction with the Liebermann-Burchard reagent. These conditions result in a higher inhibition percentage, reflected in a lower IC₅₀ value for the NLC formulation. Thus, the use of NLC plays an important role in maximizing the effectiveness of apigenin as a candidate antihyperlipidemic agent
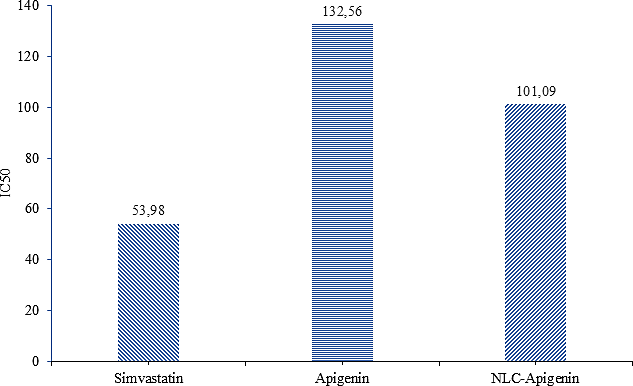
Figure 5 IC50 value of antihyperlipidemic activity.
Conclusions
The application of NLC technology to apigenin as an active ingredient resulted in a significant increase in dissolution capacity and cholesterol-lowering activity compared to unmodified apigenin. Among the various formulas tested, formula F3 with a stearic acid:oleic acid ratio (50:50) performs better than the F1 and F2 formulations, showing superior results in all colloidal characterization processes evaluated. These results highlight the potential of NLC as an innovative drug delivery system to enhance antihyperlipidemic activity. Further research is expected to prioritize in vivo testing to strengthen the efficacy and safety data of the new formulation. This study has implications for the advancement of NLC systems for antihyperlipidemic therapy, paving the way for the development of natural materials as therapeutic agents.
Acknowledgements
This research was funded by the Ministry of Education, Culture, Science, and Technology, Indonesia; Year of 2025 (Grant Number: 0070/C3/AL.04/2025).
CRediT Author Statement
Fajar Setiawan: Conceptualization, Methodology, Software. Nur Rahayuningsih: Project administration. Maya Nurul Rahma: Data curation, Writing Original draft preparation. Wulan Aprilia Susilawati: Visualization, Investigation. Lusi Nurdianti: Supervision. Ardianes Firmansya: Software, Validation. Laras Rizkia Widyastuti: Writing - Reviewing and Editing.
References
N Kullawong, T Apidechkul, P Upala, R Tamornpark, V Keawdounglek, C Wongfu, F Yeemard, S Khunthason and C Chomchoei. Factors associated with elevated low-density lipoprotein cholesterol levels among hill tribe people aged 30 years and over in Thailand: A cross-sectional study. BMC Public Health 2021; 21(1), 498.
HN Pham, R Ibrahim, E Sainbayar, A Olson, A Singh, MY Khanji, J Lee, VK Somers, C Wenger, AA Chahal and MA Mamas. Burden of hyperlipidemia, cardiovascular mortality, and COVID‐19: A retrospective‐cohort analysis of US data. Journal of the American Heart Association 2025; 14(5), e037381.
D Meidayanti. The benefits of lycopene in tomatoes for the prevention of atherosclerosis. Jurnal Medika Hutama 2021; 02(03), 2-6.
S Ramkumar, A Raghunath and S Raghunath. Statin therapy: Review of safety and potential side effects. Acta Cardiologica Sinica 2016; 32(6), 631-639.
MSS Chagas, MD Behrens, CJ Moragas-Tellis, GXM Penedo, AR Silva and CF Gonçalves-de-Albuquerque. Flavonols and flavones as potential anti‐inflammatory, antioxidant, and antibacterial compounds. Oxidative Medicine and Cellular Longevity 2022; 2022(1), 9966750.
D Tang, K Chen, L Huang and J Li. Pharmacokinetic properties and drug interactions of apigenin, a natural flavone. Expert Opinion on Drug Metabolism & Toxicology 2017; 13(3), 323-330.
KK Zhang, W Song, D Li and X Jin. Apigenin in the regulation of cholesterol metabolism and protection of blood vessels. Experimental and Therapeutic Medicine 2017; 13(5), 1719-1724.
J Zhang, D Liu, Y Huang, Y Gao and S Qian. Biopharmaceutics classification and intestinal absorption study of apigenin. International Journal of Pharmaceutics 2012; 436(1-2), 311-317.
O Elkhateeb, MEI Badawy, HG Tohamy, H Abou-Ahmed, M El-Kammar and H Elkhenany. Curcumin-infused nanostructured lipid carriers: A promising strategy for enhancing skin regeneration and combating microbial infection. BMC Veterinary Research 2023; 19(1), 206.
HA Fathi, A Allam, M Elsabahy, G Fetih and M El-Badry. Nanostructured lipid carriers for improved oral delivery and prolonged antihyperlipidemic effect of simvastatin. Colloids Surfaces B Biointerfaces 2018; 162, 236-245.
YC Pyo, P Tran, DH Kim and JS Park. Chitosan-coated nanostructured lipid carriers of fenofibrate with enhanced oral bioavailability and efficacy. Colloids Surfaces B Biointerfaces 2019; 196, 111331.
YO Agrawal, UB Mahajan, VV Agnihotri, MS Nilange, HS Mahajan, C Sharma, S Ojha, CR Patil and SN Goyal. Ezetimibe-loaded nanostructured lipid carrier based formulation ameliorates hyperlipidaemia in an experimental model of high fat diet. Molecules 2021; 26(5), 1485.
C Suhandi, G Wilar, R Lesmana, F Zulhendri, I Suharyani, N Hasan and N Wathoni. Propolis-based nanostructured lipid carriers for α-mangostin delivery: Formulation, characterization, and in vitro antioxidant activity evaluation. Molecules 2023; 28(16), 6057.
X Wang, J Liu, Y Ma, X Cui, C Chen, G Zhu, Y Sun and L Tong. Development of a nanostructured lipid carrier-based drug delivery strategy for apigenin: experimental design based on CCD-RSM and evaluation against NSCLC in vitro. Molecules 2023; 28(18), 6668.
A Firmansya, F Setiawan, L Nurdianti and A Yuliana. Formulation and characterization of buccal film nanoemulsion apigenin as antidiabetic. Indonesian Journal of Pharmaceutical Science and Technology 2022; 1(1), 22-32.
AC Ortiz, O Yañez, E Salas-Huenuleo and JO Morales. Development of an lipid carrier (NLC) by a low-energy method, comparison of release kinetics and molecular dynamics simulation. Pharmaceutics 2021; 13(4), 531.
MME Aldeeb, G Wilar, C Suhandi, AFA Mohammed, A El-Rayyes, KM Elamin and N Wathoni. Formulation and characterization of Sonchus arvensis L. nanosuspension for enhanced antioxidant and lipid-lowering activities. International Journal of Nanomedicine 2025; 20, 5457-5473.
HM Alatawi, SS Alhwiti, KA Alsharif, SS Albalawi, SM Abusaleh, GK Sror and M Qushawy. Nanostructured lipid carriers (NLCs) as effective drug delivery systems: Methods of preparation and their therapeutic applications. Recent Patents on Nanotechnology 2024; 18(2), 179-189.
A Khosa, S Reddi and RN Saha. Nanostructured lipid carriers for site-specific drug delivery. Biomedicine & Pharmacotherapy 2018; 103, 598-613.
D Rahmasari, N Rosita and W Soeratri. Physicochemical characteristics, stability, and irritability of nanostructured lipid carrier system stabilized with different surfactant ratios. Jurnal Farmasi Dan Ilmu Kefarmasian Indonesia 2022; 9(1), 8-16.
S Feng, Z Wang, J Zhao, Z Luo, P Shao, N Xiang and P Sun. Fabrication and characterization of water-soluble phytosterol ester nanodispersion by emulsification-evaporation combined ultrasonic method. Journal of Food Engineering 2020; 276, 109895.
G Jafar, M Abdassah, T Rusdiana and R Khairunisa. Development and characterization of precirol ato 88 base in nanostructured lipid carriers (Nlc) formulation with the probe sonication method. International Journal of Applied Pharmaceutics 2021; 13(3), 43-46.
D Dipahayu and GG Kusumo. Formulation and evaluation of ethanol extract nanoparticles from purple sweet potato leaves (Ipomoea batatas L.) Antin-3 variety. Jurnal Sains Dan Kesehatan 2021; 3(6), 781-785.
L Bonilla-Vidal, M Espina, ML García, L Baldomá, J Badía, JA González, LM Delgado, A Gliszczyńska, EB Souto and E Sánchez-López. Novel nanostructured lipid carriers loading Apigenin for anterior segment ocular pathologies. International Journal of Pharmaceutics 2024; 658, 124222.
RM Shah, DS Eldridge, EA Palombo and IH Harding. Stability mechanisms for microwave- produced solid lipid nanoparticles. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2022; 643, 128774.
CW Lai, FW Low, MF Tai and SB Abdul Hamid. Iron oxide nanoparticles decorated oleic acid for high colloidal stability. Advances in Polymer Technology 2018; 37(6), 1712-1721.
S Bhattacharjee. In relation to the following article ‘DLS and zeta potential - What they are and what they are not?’ Journal of Controlled Release 2016; 235, 337-351.
M Danaei, M Dehghankhold, S Ataei, F Hasanzadeh Davarani, R Javanmard, A Dokhani, S Khorasani and MR Mozafari. Impact of particle size and polydispersity index on the clinical applications of lipidic nanocarrier systems. Pharmaceutics 2018; 10(2), 57.
OM Elkhateeb, MEI Badawy, AE Noreldin, HM Abou-Ahmed, MH El-Kammar and HA Elkhenany. Comparative evaluation of propolis nanostructured lipid carriers and its crude extract for antioxidants, antimicrobial activity, and skin regeneration potential. BMC Complementary Medicine and Therapies 2022; 22(1), 256.
L Bonilla-Vidal, M Świtalska, M Espina, J Wietrzyk, ML García, EB Souto, A Gliszczyńska and ES López. Dually active apigenin-loaded nanostructured lipid carriers for cancer treatment. International Journal of Nanomedicine 2023; 18, 6979.
SN Stiani, T Rusdiana and A Subarnas. Improving solubility and dissolution of a natural product apigenin via preparation of solid dispersion by hot melt extrusion. International Journal of Applied Pharmaceutics 2021; 13(3), 47-52.
G Kumar, P Jain, T Virmani, A Sharma, MS Akhtar, SA Aldosari, MF Khan, SOD Duarte and P Fonte. Enhancing therapy with nano-based delivery systems: Exploring the bioactive properties and effects of apigenin. Therapeutic Delivery 2024; 15(9), 717-735.
JE Hyun, HY Yi, GP Hong and JY Chun. Digestion stability of curcumin-loaded nanostructured lipid carrier. LWT 2022; 162, 113474.
P Riangjanapatee and S Okonogi. Effect of surfactant on lycopene-loaded nanostructured lipid carriers. Drug Discoveries & Therapeutics 2012; 6(3), 163-168.