
Trends
Sci.
2026;
23(5):
11170
Hyperglycemia-Induced Apoptosis and Phagocytosis Suppression in Drosophila
Anggun Nurhidayah1, Habibie Habibie2, Filmaharani Filmaharani1, Alfreds Roosevelt1,3, Muhammad Rayza Azmin1, Ratnawati Ratnawati1, Jumriani Jumriani1, Widya Hardiyanti4,5, Asbah Asbah4, Nadila Pratiwi Latada4, Mukarram Mudjahid2,4 and Firzan Nainu2,4,*
1Postgraduate Program in Pharmacy, Faculty of Pharmacy, Hasanuddin University, Makassar 90245, Indonesia
2Department of Pharmacy, Faculty of Pharmacy, Hasanuddin University, Makassar 90245, Indonesia
3Departement of Pharmacy, Sandi Karsa Polytechnic, Sulawesi Selatan 90245, Indonesia
4Unhas Fly Research Group, Faculty of Pharmacy, Hasanuddin University, Makassar 90245, Indonesia
5Study Program of Pharmacy, Faculty of Medicine and Health Sciences, Universitas Muhammadiyah Makassar, Makassar 90221, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 3 July 2025, Revised: 6 October 2025, Accepted: 13 October 2025, Published: 1 January 2026
Abstract
Hyperglycemia, characterized by elevated blood glucose levels, is a major risk factor for the development of type 2 diabetes mellitus and its associated complications, including organ dysfunction and immune system impairment. Oxidative stress induced by hyperglycemia is known to disrupt cellular immune mechanisms, including phagocytosis, which is crucial for eliminating pathogens and damaged cells. This study aimed to assess the effects of a high-sugar diet (HSD) on phenotypic traits, phagocytosis, and expression of immune-related genes in Drosophila melanogaster wild-type w1118 and mutant itgbn2, which serves as a model organism for hyperglycemia. A phenotypic assay was conducted by observing glucose levels, development, body weight, crawling, and activity, as well as molecular analysis using the RT-qPCR method. The results showed that HSD treatment significantly increased hemolymph glucose levels and reduced larvae body weight, crawling ability, and developmental progression to the pupae and adult stages. Molecular analysis revealed a significant upregulation of pro-apoptotic gene expression in w1118 larvae, rpr expression increased by approximately 9% (p < 0.05), while in the itgbn² mutant, the increase was more substantial, reaching about 21% (p < 0.01). The grim expression was also significantly elevated in itgbn² larvae by around 25% (p < 0.001). In contrast, the phagocytic receptor gene drpr was markedly downregulated in w1118 larvae, showing a 50% reduction under HSD (p < 0.0001). In contrast, no significant changes were observed in the itgbn² mutant, a phagocytosis receptor involved in the elimination of apoptotic cells, which may reflect limited activation of integrin associated signaling pathways necessary for transcriptional responsiveness under hyperglycemic stress. Overall, these findings suggest that hyperglycemia induced by an HSD is associated with increased apoptosis and reduced phagocytic activity, possibly through disrupted integrin-mediated signaling in the itgbn² mutant, which may contribute to cellular immune dysfunction under hyperglycemic conditions. This study offers new insights into the molecular mechanisms underlying immune dysregulation in hyperglycemic conditions, highlighting the therapeutic potential of targeting phagocytic pathways to prevent diabetes-related complications in the future.
Keywords: Hyperglycemia, Apoptosis, Phagocytosis receptors, Inflammation, Drosophila melanogaster
Introduction
Hyperglycemia, characterized by blood glucose levels exceeding normal physiological limits, is a major contributor to mortality in individuals aged < 70 years
[1]. In 2021, over 500 million people worldwide were living with diabetes, a number expected to rise to 1.3 billion by 2050 [2]. Indonesia currently ranks fifth globally in diabetes prevalence [3]. Uncontrolled hyperglycemia significantly increases the risk of organ damage especially in the eyes, kidneys, heart, and nerves primarily through inflammation and oxidative stress [4, 5].
Hyperglycemia-induced oxidative stress triggers the release of pro-inflammatory mediators, such as IL-6 and TNF-α, while activating biological pathways that contribute to cellular damage, including glucose autoxidation, the polyol pathway, advanced glycation end product (AGE) formation, and Protein Kinase C activation [6, 7]. These processes refer to chronic hyperglycemia-induced cellular stress, including oxidative stress and inflammation, which contribute to β-cell apoptosis [8, 9]. Apoptosis is a programmed cell death essential for maintaining cellular homeostasis and facilitating tissue regeneration [10]. Chronic hyperglycemia disrupts various physiological functions, particularly in the cellular immune system. One of the most affected mechanisms is phagocytosis, the process by which immune cells remove apoptotic cells and harmful foreign substances [9]. Impaired phagocytic activity exacerbates inflammation, diminishes the efficacy of the immune system, and increases susceptibility to chronic diseases [11].
Phagocytic cells clear apoptotic cells to prevent further damage to the surrounding tissues [12]. An enhanced phagocytic response can reduce necrosis and accelerate tissue repair [13, 14]. Previous studies in mammalian models have shown that HSD can downregulate the expression of phagocytic receptors, potentially exacerbating tissue damage [15]. Although mammalian models have provided important insights into immune-metabolic interactions, their use is often limited by ethical concerns, high experimental costs, and complex handling requirements. Thus, alternative models are needed to explore conserved mechanisms in a simpler and more genetically tractable organism. Drosophila melanogaster serves as an ideal model in this regard, offering low maintenance cost, ease of handling, extensive genetic tools, and evolutionary conservation of major immune pathways [16].
Drosophila has been extensively used as a research model to investigate various biological mechanisms at the cellular and molecular levels, including apoptosis, necrosis and phagocytosis [17]. A key advantage of this model is the remarkable similarity between its cellular immune system and that of mammals, particularly the role of hemocytes in phagocytosis and immune responses [18, 19]. With approximately 75% genetic homology to humans, Drosophila serves as a powerful model for studying the pathways and receptors involved in the recognition and elimination of apoptotic and necrotic cells [20]. In Drosophila, phagocytic receptors such as Integrin βν and Draper play crucial roles in recognizing and eliminating apoptotic and necrotic cells [21, 22]. Integrin βν shares structural and functional similarities with mammalian integrins, whereas Draper is homologous to mammalian MEGF10 (Multiple EGF-like Domains 10) [23]. Given these characteristics, Drosophila represents a highly advantageous model for investigating immune and metabolic homeostasis in vivo. In the context of drug discovery, biological models such as Drosophila contribute to the early preclinical phase of research. Drug development typically progresses stepwise, beginning with in vitro and in vivo preclinical studies to identify molecular targets and mechanisms, which are then validated through clinical trials in humans. Therefore, this model plays a crucial role in exploring the molecular mechanisms underlying disease pathology and in identifying potential therapeutic targets for future intervention.
This study utilized the Drosophila itgbn2 mutant, which lacks Integrin βν receptors, in conjunction with the w1118 strain as a control, to analyze phenotypic characteristics and gene expression. In addition, Draper gene expression was assessed as a marker of phagocytic activity. This study aimed to evaluate the role of the cellular immune system, particularly phagocytic mechanisms, under hyperglycemic conditions. This investigation focused on the function of phagocytic receptors, such as Integrin βν and Draper, in the recognition and elimination of apoptotic and necrotic cells. The findings from this study are expected to provide novel insights into immune dysfunction associated with hyperglycemia and support the development of innovative therapeutic strategies targeting phagocytic receptors to enhance immune responses in hyperglycemia related complications. Particularly, they will leverage Drosophila as a genetically tractable model to explore conserved mechanisms that remain underexplored in current literature.
Materials and methods
Sucrose (CAS No.: 57-50-1, Smart Lab, Indonesia) and Glucosexidase-Peroxidase Aminoantipyrine (GOD-PAP) reagent (Glory Diagnostics, Barcelona, Spain) were used in this study. Drosophila w1118 and itgbn2 lines (Laboratory of Host Defence and Responses, Kanazawa University, Japan) were used as model organisms in all experiments. The flies were maintained at a constant temperature of 25°C. In the current study, flies were subjected to a normal diet (ND) or a high-sugar diet (HSD). The diet compositions are listed in Table 1.
Table 1 Composition of the experimental diets.
Ingredients |
Normal Diet (ND) |
High Sugar Diet (HSD) |
Corn meal (g) |
7.5 |
7.5 |
Yeast (g) |
2.5 |
2.5 |
Agar (g) |
0.9 |
0.9 |
Sucrose (g) |
4.5 |
30 [24] |
Propionic Acid (µL) |
400 |
400 |
Methyl Paraben (µL) |
450 |
450 |
Water (mL) |
100 |
100 |
Hyperglycemia model establishment
Drosophila used in this study were 3 - 5 days old. Twenty flies (10 males and 10 females) were mated on either ND or HSD diet. The mating period lasted 5 - 7 days to produce third-instar larvae, which were identified by their movement along the walls of the vial. The collected larvae were used for further analysis.
Figure 1 The experimental groups consisted of Drosophila larvae from 2 genotypes (w1118 and itgbn²) fed either ND or HSD, resulting in 4 treatment combinations: w1118 ND, w1118 HSD, itgbn² ND, and itgbn² HSD. ND, normal diet. HSD, high sugar diet.
Hemolymph glucose levels
Approximately 70 third instar larvae were collected to obtain hemolymph for glucose concentration measurements [25]. Larvae were collected at different time points between the control and HSD groups because of the observed developmental delays in the HSD group. The larvae were placed in microtubes and homogenized using a micropestle. Following homogenization, the samples were centrifuged at 4 °C and 1,600 rpm to extract the hemolymph. Hemolymph (10 μL) was pipetted and transferred to microtubes containing 1 mL of GOD-PAP reagent. The resulting mixture was incubated for 10 min at room temperature, after which the absorbance was measured using a UV-Vis spectrophotometer (Shimadzu UV-1800, Shimadzu Corp., Kyoto, Japan) at a wavelength of 500 nm.
Survival assay
A survival assay was conducted to assess the lifespan of the treatment group, starting from the second-instar larvae. A total of 20 larvae from each group were placed in vials containing fresh feed according to group division. The number of larvae that developed into pupae and the number of pupae that emerged as adult flies were recorded.
Determination of body weight and size of larvae
Body weight measurements were conducted on third-instar larvae that were washed with NaCl using an analytical balance (Sartorius®) [26]. Body size was measured using a digital caliper (Taffware, Indonesia).
Crawling and locomotor assay
The crawling assay was conducted as previously described [27]. The objective of this study was to evaluate the impact of HSD on the motor activity of Drosophila larvae. Third-instar larvae from each treatment group were placed in Petri dishes containing 2% agar. Their crawling activity was observed for 1 min, during which the number of squares traversed by the larvae was recorded. Each square on the grid paper measured 1×1 mm2.
Real-time qPCR analysis
Total RNA was extracted from Drosophila larvae using the PureLink™ RNA Mini Kit (Invitrogen, Thermo Fisher Scientific Inc., MA, US). following the manufacturer’s protocol. Briefly, ten third-instar larvae of each w1118 and itgbn2 were homogenized in Treff tubes containing lysis buffer supplemented with 1% 2-mercaptoethanol and subjected to the remaining total RNA isolation procedure. Gene expression analysis was performed on the isolated total RNA using the reverse transcriptase quantitative PCR (RT-qPCR) method, with the Universal One-Step RT-qPCR Kit (Luna®, New England Biolabs, Inc., MA., US). Following the manufacturer’s protocol. Each RT-qPCR reaction was performed in a total volume of 10 μL. Reactions were performed on the Rotor-Gene Q (Qiagen, Germany), starting with a reverse transcription step at 50 °C for 10 min, followed by 40 cycles of PCR amplification. The PCR protocol consisted of denaturation at 95 °C for 10 s, followed by annealing and elongation at 60 °C for 30 s. After the PCR cycle was completed, melting curve analysis was performed at 40 °C for 1 min to assess the specificity of the amplified products. The primer sequences used for each target gene are shown in Table 2.
Table 2 Primer sequences (Drosophila) used for real-time qPCR.
Genes |
Forward Primers (5′–3′) |
Reverse Primers (5′–3′) |
dilp3 |
5′-ATCCTTATGATCGGCGGTGT-3′ |
5′-GTTCACGGGGTCCAAAGTTC-3′ |
rpr |
5′-ACTGGATCCCAATGGCAGTGGCATTCT-3′ |
5′-AAAGGATCCTCATTGCGATGGCTTGC-3′ |
hid |
5′-TGCGAAATACACGGGTTCA-3′ |
5′-CCAATATCACCCAGTCCCG-3′ |
grim |
5′-TCGGAGTTTGGATGCTGGGATCTT-3′ |
5′-AGTCACGTCGTCCTCATCGTTGTT-3′ |
atg5 |
5′-GCACTACATGTCCTGCCTGA-3′ |
AGATTCGCAGGGGAATGTTT-3′ |
drpr |
5′-CGGAATTCTCTGCCGCACGGGTTACATAG-3′ |
5′-CCGCTCGAGCCGGCTCGAATTTTCGCTT-3′ |
rp49 |
5′-CGCTTCAAGGGACAGTATCTG-3′ |
5′-AAACGCGGTTCTGCATGAG-3′ |
Statistical analyses of hemolymph glucose levels, body size, body weight, larval crawling ability, and negative geotaxis were performed using unpaired 2-tailed t-tests, as only 2 dietary groups (ND and HSD) were compared. For the survival assay, the percentage of larvae reaching the pupae or adult stage was recorded at regular intervals (every 12 h or daily) until all individuals either completed metamorphosis or failed to develop. RT-qPCR data (dilp3, rpr, hid, grim, atg5 and drpr) were analyzed using Qgene software (Qiagen, Germany), followed by statistical evaluation using unpaired t-test for comparisons between ND and HSD with in each genotype. All analyses were conducted using Prism® 9 (GraphPad Software, Boston, MA, U.S.).
Results and discussion
Elevation of hemolymph glucose levels in Drosophila melanogaster larvae by a high-sugar diet
In this study, we established a hyperglycemia model in Drosophila by administering HSD containing 30% sucrose, following a previously established protocol [24]. Although this approach effectively induces a hyperglycemic phenotype in flies, it should be noted that such a high sucrose concentration represents an artificial condition and may not fully replicate physiological hyperglycemia in mammals. This model aims to evaluate the metabolic effects of excessive nutrient intake, which has been associated with disorders such as insulin resistance and diabetes mellitus [28]. This study utilized both w1118 and itgbn2 Drosophila strains. This model was used to investigate alterations in hemolymph glucose levels and the expression of genes associated with the insulin signaling pathway, specifically dilp3. Drosophila was selected based on its conserved insulin signaling pathway, which closely resembles that of humans, and its hemolymph, which functions analogously to mammalian blood [29, 30]. Furthermore, insulin-like peptides (DILPs), produced by insulin-producing cells (IPCs) in the brain, serve a role analogous to that of the mammalian pancreas in regulating metabolism and maintaining energy homeostasis [25,31]. Third-instar larvae were selected for testing because of their high feeding activity during this developmental stage, which amplifies their sensitivity to dietary changes [32]. The results, as depicted in Figure 2, demonstrated that HSD significantly elevated hemolymph glucose levels (Figure 2(A)). Concurrently, a marked decrease in the expression of dilp3 was observed in both genotypes (Figure 2(B)).
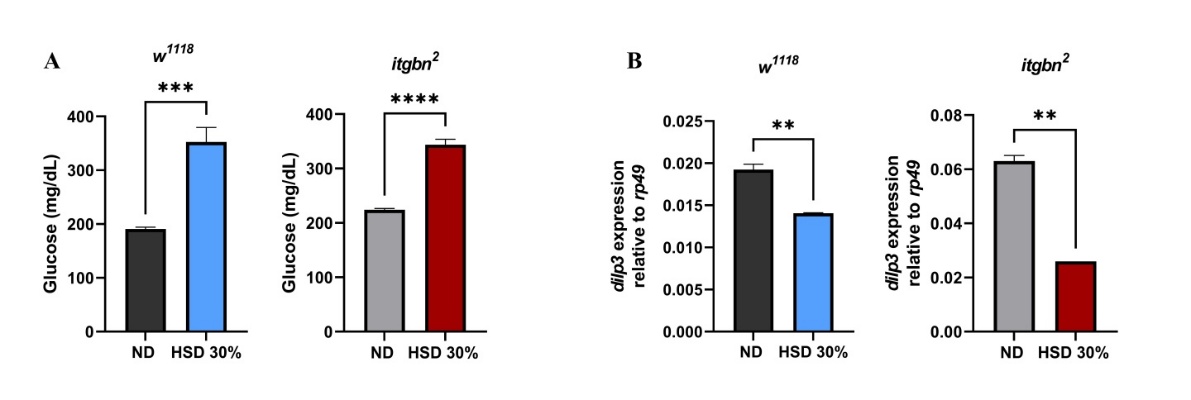
Figure 2 Effect of HSD on glucose levels and insulin-like peptide gene expression in Drosophila larvae. (A) HSD induced hyperglycemia in Drosophila w1118 and itgbn2 larvae, as indicated by a significant increase in hemolymph glucose levels. (B) A significant reduction in dilp3 expression was observed in both genotypes. Absolute values and effect sizes are provided in the discussion. ND, normal diet. HSD, high sugar diet; **, p < 0.01; ***, p < 0.001, **** p < 0.0001.
The results indicated that HSD markedly increased hemolymph glucose concentrations in the larvae (Figure 2(A)). In w1118 larvae, hemolymph glucose levels increased from 190.7 mg/dL under ND to 352.3 mg/dL following HSD, indicating an 85% elevation (p < 0.0001). In the itgbn² mutant, glucose levels rose from 224.0 to 343.7 mg/dL, representing a 53% increase (p < 0.0001). In parallel, dilp3 expression was significantly reduced under HSD. In w1118 larvae, expression decreased by 26%, while in the itgbn² mutant, the reduction reached 59% compared with ND (p < 0.0001). These results indicate that both genotypes exhibit hyperglycemia under HSD, accompanied by decreased dilp3 expression, which was more pronounced in the itgbn² mutant. This observation aligns with previous studies that demonstrated that HSD can induce hyperglycemic conditions [33].
In addition, dilp3 gene expression (Figure 2(B)) was significantly reduced in the HSD treated groups of both w1118 and itgbn2 larvae. This altered expression pattern may reflect a chronic hyperglycemic state in both genotypes following prolonged HSD exposure. The dilp3 gene plays a crucial role in regulating metabolism and glucose levels to maintain energy homeostasis, support growth, and facilitate physiological adaptation to nutritional changes induced by stress [34]. Integrin βν has been implicated in phagocytic activity in Drosophila, particularly through glial cells and hemocytes, which play essential roles in clearing apoptotic cells and maintaining tissue homeostasis [35]. These results suggest that metabolic stress and impaired immune homeostasis in itgbn2 mutants may exacerbate hyperglycemia.
Reduction in survival of Drosophila under hyperglycemic conditions
Survival assays were conducted using a hyperglycemia model of Drosophila to evaluate the systemic effects of metabolic disturbances induced by HSD. Survival is a key indicator of an organism’s resilience to metabolic stress [36, 37]. This study assessed the effects of HSD on the developmental stages of both w1118 and itgbn2, as shown in Figure 3. The results demonstrated that HSD significantly reduced the survival rate from the larval to pupal stages (Figure 3(A)), and from pupae to adult flies (Figure 3(B)).
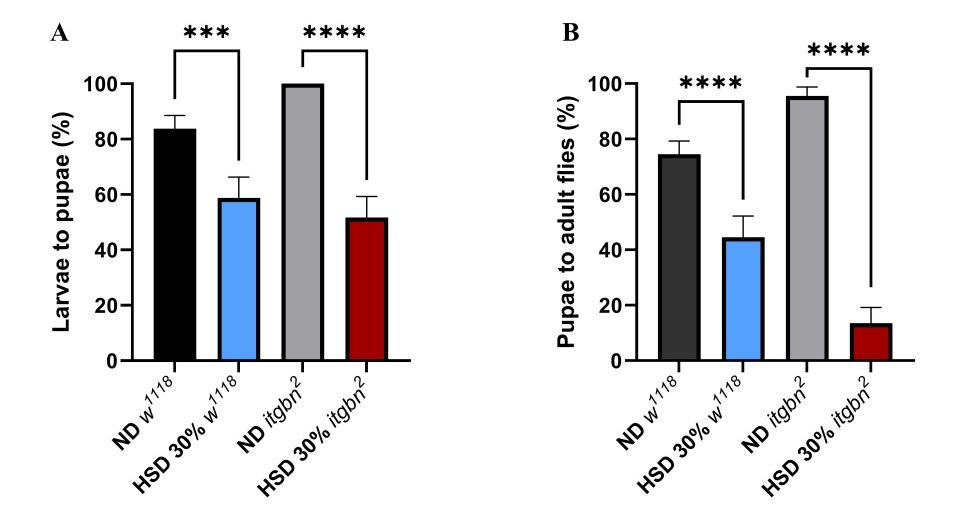
Figure 3 HSD exposure affects the developmental progression of Drosophila at multiple stages. (A) The effect of HSD on survival from the larvae to pupae stage was significantly reduced. (B) Survival from pupae to adult flies also showed a significant decrease. ND, normal diet. HSD, high sugar diet; ns (non-significant); * p < 0.05; ** p < 0.01; **** p < 0.0001.
These findings suggest that HSD impairs developmental processes (Figures 3(A) - 3(B)). The HSD caused an apparent developmental delay compared to the control group on an ND. In w1118 larvae, the transition from larvae to pupae was extended from 3 to 5 days, and from pupae to adult from 3 to 4 days. The delay was more pronounced in the itgbn² mutant, where the larvae to pupae transition increased from 5 to 10 days and the pupae to adult stage from 6 to 7 days under HSD. Consistently, HSD markedly reduced survival at both developmental stages. During the larvae to pupae transition, survival in w1118 decreased by 28%, while in the itgbn² mutant, survival dropped by 36%. At the pupae-to-adult stage, survival declined by 40% in w1118 and by more than 90% in the itgbn² mutant (p < 0.0001). These findings indicate that excessive dietary sugar impairs survival and disrupts expected developmental timing, with more severe effects observed in the itgbn² mutant.
Mechanistically, the observed developmental delay under HSD is likely associated with the disruption of dilp signalling, which plays a critical role in regulating growth and metabolism. The reduced expression of dilp3 in larvae exposed to HSD may reflect systemic metabolic dysregulation. This finding aligns with Kim and Neufeld [38], who showed that dietary sugar can modulate dilp expression and thereby influence systemic metabolic homeostasis. Under metabolic stress conditions such as hyperglycemia, IPCs are hypothesised to undergo functional alter ations that result in decreased dilp expression. Although the current data do not directly demonstrate impairment of the entire insulin signalling cascade, the downregulation of dilp3 can be considered an early indicator of potential metabolic dysfunction affecting growth related pathways.
In addition, the more pronounced developmental delay observed in itgbn² mutants may imply that itgbn², characterized by the absence of Integrin βν receptors, may be more vulnerable to development, especially following exposure to an HSD. This increased sensitivity is potentially linked to compromised phagocytic activity, as integrin βν receptors are essential components in mediating effective cellular immune responses. Consequently, the metabolic stress induced by HSD may not be effectively counteracted in the itgbn² mutant due to the absence of integrin-mediated cellular functions. These findings further suggest that Integrin βν is not only essential for phagocytic activity but also plays a critical role in supporting organismal viability and developmental progression under hyperglycemic stress. These findings are supported by evidence that HSD-induced hyperglycemia can impair developmental processes in Drosophila [32,39].
The reductions in larval body size, weight and motor activity resulting from hyperglycemic conditions
Dietary interventions significantly influence organismal phenotypes, including those of Drosophila. HSD has been shown to alter phenotypic traits, such as body size, weight, and motor activity [40]. The results shown in Figure 4 indicate that exposure of Drosophila larvae to HSD significantly reduced larvae body weight (Figure 4(A)), and width (Figure 4(B)), and length (Figure 4(C)) and diminished crawling activity in third instar larvae (Figure 4(D)). These phenotypic alterations were observed in both w1118 and itgbn2 mutant strains.
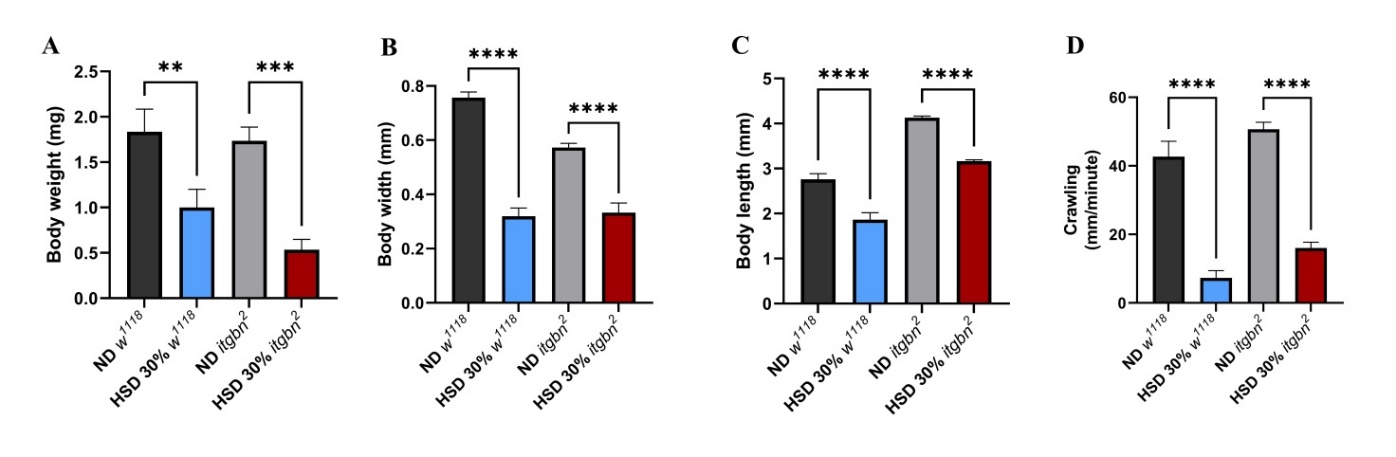
Figure 4 HSD exposure adversely affected the physical development and motor function of Drosophila larvae. (A) HSD significantly decreased larvae body weight, (B) length, (C) width, and (D) crawling ability of Drosophila larvae. ND, normal diet. HSD, high sugar diet; ** p < 0.01; *** p < 0.001; **** p < 0.0001.
The results shown in Figures 4(A) - 4(C) revealed a significant decrease in body weight in w1118 larvae, with a substantially greater reduction observed in itgbn2 larvae. Under HSD, both w1118 and itgbn² larvae exhibited marked reductions in body size and motility. In w1118, body weight, width, and length decreased by approximately 45%, 58%, and 32%, respectively. The itgbn² mutant showed an even stronger response, with reductions of about 69% in body weight, 42% in body width, and 23% in body length. Crawling activity was also severely impaired, declining by more than 80% in w1118 and by nearly 70% in the itgbn² mutant (p < 0.0001). These results indicate that excessive dietary sugar profoundly suppresses growth and locomotor performance, with greater sensitivity observed in the itgbn² mutant background.
Our findings demonstrated a significant reduction in body weight, body size, and motor activity (Figure 4). The decrease in larval body weight is associated with hyperglycemic conditions, which may exacerbate metabolic homeostasis, reduce energy storage efficiency, and disrupt lipid synthesis and storage [36]. Consequently, metabolic efficiency decreases, leading to a reduction in both body weight and size (Figures 4(A) - 4(C)). These findings are consistent with those of mammalian studies, in which HSD has been associated with reduced body weight in Sprague-Dawley rats [41].
Similarly, analysis of crawling ability revealed a significant decrease in itgbn2 compared to w1118 larvae (Figure 4(D)). This result is supported by previous research demonstrating that a HSD can lead to reduced physical activity in Wistar rats [42]. This condition may impair metabolic function, particularly in itgbn2 larvae, which are more vulnerable to the effects on their phagocytic immune system. The increased susceptibility of itgbn2 became more evident based on the phenotypic data following HSD exposure. These findings show that impairment of integrin function under hyperglycemic conditions increases cellular vulnerability to metabolic stress.
Induction of apoptosis and suppression of phagocytosis by hyperglycemia
Gene expression analysis indicated that HSD induced physiological alterations in Drosophila that resembled type 2 diabetes, which consequently affected cellular immune functions, such as phagocytosis and apoptosis (Figure 5).
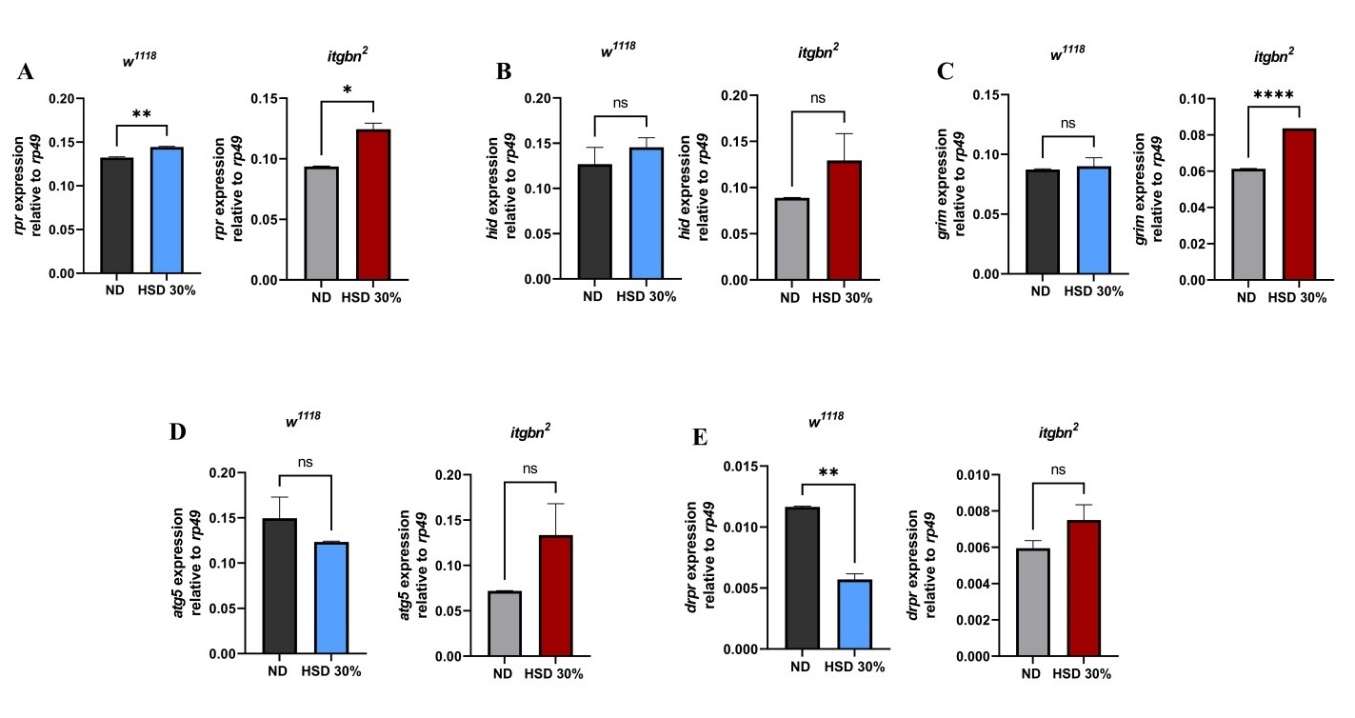
Figure 5 HSD exposure modulates the expression of genes involved in apoptosis, autophagy, and phagocytosis in Drosophila larvae. (A) rpr was significantly upregulated in both w1118 and itgbn². (B) hid expression remained unchanged in both strains. (C) grim was significantly upregulated only in itgbn². (D) atg5 expression did not differ across strains or treatments. (E) drpr was downregulated in w1118 but remained unchanged in itgbn². ND, normal diet. HSD, high sugar diet; ns (non-significant); * p < 0.05; ** p < 0.01; **** p < 0.0001.
As shown in Figures 5(A) - 5(E), the expression of genes exhibited genotype-specific responses to the high-sugar diet (HSD). In w1118 larvae, rpr expression increased by approximately 9% under HSD compared to ND (p < 0.001), while in the itgbn² mutant, the increase was more substantial, reaching about 21% (p < 0.01). Expression of hid and grim showed only modest and variable changes in w1118 (less than 10%, p > 0.05), but in itgbn² larvae, both genes tended to be upregulated, showing increases of about 20% - 25%, though not all changes reached statistical significance (p = 0.06 - 0.09). For the autophagy-related gene atg5, expression decreased slightly in w1118 (around 10%, p > 0.05) but increased by approximately 35% in the itgbn² mutant (p < 0.05), suggesting differential autophagic regulation between genotypes. Notably, drpr expression was significantly reduced by 50% in w1118 larvae (p < 0.0001), whereas it remained essentially unchanged in itgbn² mutants (p > 0.05).
The administration of HSD in Drosophila potentially disrupts metabolic homeostasis, likely due to oxidative stress induced by elevated glucose levels. As shown in Figure 5(A), we observed an increase in the expression of pro-apoptotic genes, suggesting that HSD may trigger apoptosis possibly mediated by ROS accumulation. This condition is known to activate the intrinsic apoptotic pathway, involving pro-apoptotic proteins such as reaper (rpr), Head Involution Defective (hid), and grim, which inhibit Diap1 and promote apoptosome formation (Dark), followed by activation of Dronc and effector caspases Drice and Dcp-1, ultimately leading to programmed cell death [43].
The increased rpr expression suggests a potential disruption of tissue homeostasis caused by oxidative stress (Figure 5(A)), which may damage the cellular structures and accelerate apoptotic processes. In contrast, the expression of another pro-apoptotic gene, hid, did not show any significant change (Figure 5(B)). Interestingly, the expression of grim did not exhibit significant changes in w1118; however, it was significantly upregulated in the itgbn2 mutant exposed to HSD (Figure 5(C)). These findings suggest that Drosophila itgbn2 experiences enhanced apoptotic activity, as indicated by the increased expression of rpr and grim. The rpr gene plays a crucial role in regulating apoptosis during both embryonic and adult stages of Drosophila. The expression of rpr can be induced by the activation of the p53 pathway or DNA damage caused by oxidative stress, specifically contributing to the activation of the caspase cascade. The hid gene functions through a mechanism similar to rpr and grim, although it is expressed at lower levels. Meanwhile, grim primarily inhibits the activity of Diap1, an antiapoptotic protein essential for regulating cell survival [44]. Collectively, these data suggest that HSD-induced apoptosis plays a more prominent role in driving cell death in the itgbn2 mutant than in the w1118 strain, as evidenced by the heightened expression of key pro-apoptotic genes, such as rpr and grim. In contrast, the expression of atg5, a gene involved in regulating autophagy and immune responses [45], did not differ significantly between the ND and HSD groups (Figure 5(D)). These findings suggest that HSD primarily affects apoptotic regulatory pathways associated with oxidative stress, rather than autophagic pathways.
This study utilized Integrin βν as phagocytic receptor models and analyzed the expression of the drpr gene (Figure 5(E)), which encodes the Draper protein. The results demonstrated that drpr expression in the itgbn² mutant did not differ significantly between the ND and HSD groups. This lack of drpr expression response is likely attributable to impaired activation of apoptotic cell recognition signaling resulting from the loss of Integrin βν function as a phagocytic receptor. These findings indicate a disruption of phagocytic regulatory mechanisms caused by the itgbn² mutation. This supports the hypothesis that this gene plays a crucial role in cellular immune responses to metabolic stress induced by an HSD, consistent with previous reports highlighting the involvement of Integrin βν in metabolic signaling and insulin sensitivity [46]. Furthermore, a sustained increase in apoptotic activity may exacerbate tissue damage, increasing the risk of necrosis and the development of other pathologies. As reported by Dey et al. [47]; LaRocca et al. [48], chronic hyperglycemia is known to exacerbate inflammation through the activation of toll-like receptors (TLRs), which subsequently elevates the release of pro-inflammatory cytokines.
These findings indicate a disruption in the phagocytic regulatory mechanisms caused by the itgbn² mutation. This supports the hypothesis that this gene plays a role in immune responses to metabolic stress induced by an HSD. While previous studies have explored metabolic and inflammatory pathways as potential therapeutic targets for diabetes [49, 50], our results may offer an additional perspective by suggesting the possible involvement of phagocytosis receptors in maintaining cellular homeostasis under hyperglycemic conditions.
Limitation
Although the mechanisms of phagocytosis and apoptosis are largely conserved between Drosophila and mammals, the translational relevance of our findings remains to be fully established. Key signalling components, including integrins, Draper (the homolog of mammalian MGF10), and apoptotic pathways, exhibit conserved roles; however, the present study is exploratory and did not include validation in mammalian models or clinical settings. Additionally, rescue experiments to directly confirm the involvement of dilp signalling or Integrin βν function were not performed, which limits the mechanistic conclusions. Future studies addressing these gaps will be necessary to strengthen the evidence and better assess the applicability of this Drosophila model for understanding immune dysfunction under hyperglycemic conditions.
Conclusions
Notably, the itgbn² mutant exhibited impaired phagocytic signaling and distinct transcriptional responses under hyperglycemic conditions. These findings suggest a possible involvement of Integrin βν in coordinating immune and metabolic responses to dietary stress. While this model enhances our understanding of immune metabolic interactions under hyperglycemia, further studies using mammalian models will be essential to determine whether these mechanisms are conserved and may inform potential therapeutic strategies in the future. This cautious interpretation aligns the conclusions with the current data while highlighting clear directions for future research.
Acknowledgements
The authors extend their gratitude to Prof. Takayuki Kuraishi from Kanazawa University, Japan for generously providing the Drosophila lines used in this study. We would also like to appreciate the support provided by Prof. Elly Wahyudin from the Faculty of Pharmacy, Hasanuddin University, Indonesia, for facilitating our molecular experiments at Biofarmaka Laboratory. This study was funded by the Ministry of Education, Culture, Research, and Technology of the Republic of Indonesia (KEMDIKBUDRISTEK) as part of a 2024 Master’s Thesis Research Grant (050/E5/PG.02.00.PL/2024).
Declaration of Generative AI in Scientific Writing
The authors declare that AI tools (ChatGPT and Grammarly) were used to assist in language editing and translation during the preparation of this manuscript. The content, ideas, interpretations, and conclusions remain entirely the authors’ original work and responsibility.
CRediT Author Statement
Anggun Nurhidayah: Conceptualization; Methodology; Software; Data curation; Writing - Original draft preparation; Visualization. Habibie Habibie: Software; Validation; Supervision; Funding acquisition. Filmaharani Filmaharani, Alfreds Roosevelt, Muhammad Rayza Azmin, Ratnawati Ratnawati, Jumriani Jumriani: Investigation; Writing - Review & Editing. Widya Hardiyanti, Asbah Asbah, Nadila Pratiwi Latada, Mukarram Mudjahid: Investigation; Resources; Writing - Review & Editing. Firzan Nainu: Conceptualization; Methodology; Resources; Writing - Review & Editing; Supervision; Project administration; Funding acquisition.
References
M Islam, MA Hoshen, FI Ayshasiddeka and T Yeasmin. Antimicrobial, membrane stabilizing and thrombolytic activities of ethanolic extract of Curcuma zedoaria Rosc. Rhizome. Journal of Pharmacognosy and Phytochemistry 2017; 6(5), 38-41.
KL Ong, LK Stafford, SA McLaughlin, EJ Boyko, SE Vollset, AE Smith, BE Dalton, J Duprey, JA Cruz, H Hagins, PA Lindstedt, A Aali, YH Abate, MD Abate, M Abbasian, Z Abbasi-Kangevari, M Abbasi-Kangevari, SA ElHafeez, R Abd-Rabu, …, T Vos. Global, regional, and national burden of diabetes from 1990 to 2021, with projections of prevalence to 2050: A systematic analysis for the Global Burden of Disease Study 2021. The Lancet 2023; 402(10397), 203-234.
ID Federation. Diabetes around the world 2021. International Diabetes Federation, Brussels, Belgium, 2025.
MZ Banday, AS Sameer and S Nissar. Pathophysiology of diabetes: An overview. Avicenna Journal of Medicine 2020; 10(04), 174-188.
D Reyes-Umpierrez, G Davis, S Cardona, FJ Pasquel, L Peng, S Jacobs, P Vellanki, M Fayfman, S Haw, M Halkos, RA Guyton, VH Thourani and GE Umpierrez. Inflammation and oxidative stress in cardiac surgery patients treated to intensive versus conservative glucose targets. The Journal of Clinical Endocrinology & Metabolism 2017; 102(1), 309-315.
Y Li, Y Liu, S Liu, M Gao, W Wang, K Chen, L Huang and Y Liu. Diabetic vascular diseases: Molecular mechanisms and therapeutic strategies. Signal Transduction and Targeted Therapy 2023; 8(1), 152.
S Tangvarasittichai. Oxidative stress, insulin resistance, dyslipidemia and type 2 diabetes mellitus. World Journal of Diabetes 2015; 6(3), 456.
B Zhang, Y Chen, Q Shen, G Liu, J Ye, G Sun and X Sun. Myricitrin attenuates high glucose-induced apoptosis through activating Akt-Nrf2 signaling in H9c2 cardiomyocytes. Molecules 2016; 21(7), 880.
B Giri, S Dey, T Das, M Sarkar, J Banerjee and SK Dash. Chronic hyperglycemia mediated physiological alteration and metabolic distortion leads to organ dysfunction, infection, cancer progression and other pathophysiological consequences: An update on glucose toxicity. Biomedicine & Pharmacotherapy 2018; 107, 306-328.
S Riwaldt, TJ Corydon, D Pantalone, J Sahana, P Wise, P Wise, M Wehland, M Krüger, D Melnik, Daniela, S Kopp, M Infanger and D Grimm. Role of apoptosis in wound healing and apoptosis alterations in microgravity. Frontiers in Bioengineering and Biotechnology 2021; 9, 679650.
SA Antar, NA Ashour, M Sharaky, M Khattab, NA Ashour, RT Zaid, EJ Roh, A Elkamhawy and AA Al-Karmalawy. Diabetes mellitus: Classification, mediators, and complications; A gate to identify potential targets for the development of new effective treatments. Biomedicine & Pharmacotherapy 2023; 168, 115734.
S Arandjelovic and KS Ravichandran. Phagocytosis of apoptotic cells in homeostasis. Nature Immunology 2015; 16(9), 907-917.
IKH Poon, CD Lucas, AG Rossi and KS Ravichandran. Apoptotic cell clearance: basic biology and therapeutic potential. Nature Reviews Immunology 2014; 14(3), 166-180.
S Nagata. Autoimmune diseases caused by defects in clearing dead cells and nuclei expelled from erythroid precursors. Immunological Reviews 2007; 220(1), 237-250.
S Pavlou, J Lindsay, R Ingram, H Xu and M Chen. Sustained high glucose exposure sensitizes macrophage responses to cytokine stimuli but reduces their phagocytic activity. BMC Immunology 2018; 19(1), 24.
TS Salminen and PF Vale. Drosophila as a model system to investigate the effects of mitochondrial variation on innate immunity. Frontiers in Immunology 2020; 11, 521.
F Nainu, Y Tanaka, A Shiratsuchi and Y Nakanishi. Protection of insects against viral infection by apoptosis-dependent phagocytosis. The Journal of Immunology 2015; 195(12), 5696-5706.
N Buchon, N Silverman and S Cherry. Immunity in Drosophila melanogaster--from microbial recognition to whole-organism physiology. Nature Reviews Immunology 2014; 14(12), 796-810.
S Yu, F Luo, Y Xu, Y Zhang and LH Jin. Drosophila innate immunity involves multiple signaling pathways and coordinated communication between different tissues. Frontiers in Immunology 2022; 13, 905370.
KA Hope and LT Reiter. Understanding human genetic disease with the fly. In: K Walz and JI Young (Eds.). Cellular and animal models in human genomics research. Academic Press, New York, 2019, p. 69-87.
J Manaka, T Kuraishi, A Shiratsuchi, Y Nakai, H Higashida, P Henson and Y Nakanishi. Draper-mediated and phosphatidylserine-independent phagocytosis of apoptotic cells by Drosophila hemocytes/macrophages. Journal of Biological Chemistry 2004; 279(46), 48466-48476.
K Nagaosa, R Okada, S Nonaka, K Takeuchi, Y Fujita, T Miyasaka, J Manaka, I Ando and Y Nakanishi. Integrin βν-mediated phagocytosis of apoptotic cells in Drosophila embryos. Journal of Biological Chemistry 2011; 286(29), 25770-25777.
F Nainu, A Shiratsuchi and Y Nakanishi. Induction of apoptosis and subsequent phagocytosis of virus-infected cells as an antiviral mechanism. Frontiers in Immunology 2017; 8, 1220.
N Baenas and AE Wagner. Drosophila melanogaster as a model organism for obesity and type-2 diabetes mellitus by applying high-sugar and high-fat diets. Biomolecules 2022; 12(2), 307.
F Liguori, E Mascolo and F Vernì. The genetics of diabetes: What we can learn from Drosophila. International Journal of Molecular Sciences 2021; 22(20), 11295.
F Lourido, D Quenti, D Salgado-Canales and N Tobar. Domeless receptor loss in fat body tissue reverts insulin resistance induced by a high-sugar diet in Drosophila melanogaster. Scientific Reports 2021; 11(1), 3263.
CD Nichols, J Becnel and UB Pandey. Methods to assay Drosophila behavior. Journal of Visualized Experiments 2012; 61, e3795.
YH Alam, R Kim and C Jang. Metabolism and health impacts of dietary sugars. Journal of Lipid and Atherosclerosis 2022; 11(1), 20.
C Guney and F Akar. The possible mechanisms of high-fructose diet-induced pancreatic disturbances. Journal of Research in Pharmacy 2023; 27(2), 753-761.
K Jones and I Eleftherianos. A simple protocol for isolating hemolymph from single Drosophila melanogaster adult flies. Methods and Protocols 2023; 6(5), 100.
K Kannan and YW Fridell. Functional implications of Drosophila insulin-like peptides in metabolism, aging, and dietary restriction. Frontiers in Physiology 2013; 4, 288.
LP Musselman, JL Fink, K Narzinski, PV Ramachandran, SS Hathiramani, RL Cagan and TJ Baranski. A high-sugar diet produces obesity and insulin resistance in wild-type Drosophila. Disease Models & Mechanisms 2011; 4(6), 842-849.
D Chen, J Yang, Z Xiao, S Zhou and L Wang. A diet-induced type 2 diabetes model in Drosophila. Science China Life Sciences 2021; 64, 326-329.
DR Nässel and JV Broeck. Insulin/IGF signaling in Drosophila and other insects: Factors that regulate production, release and post-release action of the insulin-like peptides. Cellular and Molecular Life Sciences 2016; 73, 271-290.
C Melcarne, B Lemaitre and E Kurant. Phagocytosis in Drosophila: From molecules and cellular machinery to physiology. Insect Biochemistry and Molecular Biology 2019; 109, 1-12.
SNS Morris, C Coogan, K Chamseddin, SO Fernandez-Kim, S Kolli, JN Keller and JH Bauer. Development of diet-induced insulin resistance in adult Drosophila melanogaster. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease 2012; 1822(8), 1230-1237.
BM Rovenko, OI Kubrak, DV Gospodaryov, NV Perkhulyn, IS Yurkevych, A Sanz, OV Lushchak and VI Lushchak. High sucrose consumption promotes obesity whereas its low consumption induces oxidative stress in Drosophila melanogaster. Journal of Insect Physiology 2015; 79, 42-54.
J Kim and T Neufeld. Dietary sugar promotes systemic TOR activation in Drosophila through AKH-dependent selective secretion of Dilp3. Nature Communications 2015; 6(1), 6846.
MR Azmin, H Habibie, F Filmaharani, A Roosevelt, A Nurhidayah, MR Pratama, W Hardiyanti, NP Latada, M Mudjahid, D Yuliana and F Nainu. Aspirin-mediated reduction of glucose level and inflammation in Drosophila melanogaster. ACS Omega 2025; 10(18), 18622-18628.
V Eickelberg, K Lüersen, S Staats and G Rimbach. Phenotyping of Drosophila melanogaster - a nutritional perspective. Biomolecules 2022; 12(2), 221.
L Cao, X Liu, H Cao, Q Lv and N Tong. Modified high‐sucrose diet‐induced abdominally obese and normal‐weight rats developed high plasma free fatty acid and insulin resistance. Oxidative Medicine and Cellular Longevity 2012; 2012(1), 374346.
SV Bădescu, CP Tătaru, L Kobylinska, EL Georgescu, DM Zahiu, AM Zăgrean and L Zăgrean. Effects of caffeine on locomotor activity in streptozotocin-induced diabetic rats. Journal of Medicine and Life 2016; 9(3), 275.
R Umargamwala, J Manning, L Dorstyn, D Denton and S Kumar. Understanding developmental cell death using Drosophila as a model system. Cells 2024; 13(4), 347.
C Peterson, GE Carney, BJ Taylor and K White. Reaper is required for neuroblast apoptosis during Drosophila development. Development 2002; 129(6), 1467-1476.
X Ye, XJ Zhou and H Zhang. Exploring the role of autophagy-related gene 5 (ATG5) yields important insights into autophagy in autoimmune/autoinflammatory diseases. Frontiers in Immunology 2018; 9, 2334.
J Kim, D Bilder and TP Neufeld. Mechanical stress regulates insulin sensitivity through integrin-dependent control of insulin receptor localization. Genes & Development 2018; 32(2), 156-164.
S Dey, N Murmu, T Mondal, I Saha, S Chatterjee, R Manna, S Haldar, SK Dash, TR Sarkar and B Giri. Multifaceted entrancing role of glucose and its analogue, 2-deoxy-D-glucose in cancer cell proliferation, inflammation, and virus infection. Biomedicine & Pharmacotherapy 2022; 156, 113801.
TJ LaRocca, SA Sosunov, NL Shakerley, VS Ten and AJ Ratner. Hyperglycemic conditions prime cells for RIP1-dependent necroptosis. Journal of Biological Chemistry 2016; 291(26), 13753-13761.
TM Belete. A recent achievement in the discovery and development of novel targets for the treatment of type-2 diabetes mellitus. Journal of Experimental Pharmacology 2020; 12, 1-15.
S Dhankhar, S Chauhan, DK Mehta, Nitika, K Saini, M Saini, R Das, S Gupta and V Gautam. Novel targets for potential therapeutic use in Diabetes mellitus. Diabetology & Metabolic Syndrome 2023; 15(1), 17