
Trends
Sci.
2026;
23(5):
11649
Optimization of Polysaccharides Matrix for Enhancing Physicochemical Properties and Stability of Synbiotic Encapsulation
Lintang Dion Pangestu1, I Made Dedi Mahariawan 2, Muhammad Fakhri2,
Abdul Rahem Faqih2, Ating Yuniarti2 and Anik Martinah Hariati2*
1Postgraduate Program in Fisheries and Marine Science, Faculty of Fisheries and Marine Science, Universitas Brawijaya, East Java 65145, Indonesia
2Department of Aquaculture, Faculty of Fisheries and Marine Science, Universitas Brawijaya,
East Java 65145, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 20 August 2025, Revised: 17 September 2025, Accepted: 10 October 2025, Published: 5 January 2026
Abstract
Encapsulation techniques can prevent the decrease in viability and maintain stability in the digestive tract. To support the success of encapsulation application, the matrix material must be selected with an appropriate composition to support the protection of the core material in the capsule. This study evaluates the matrix composition of the synbiotics’ encapsulation characteristics and viability during the processing period. An experimental method was used in this study with a complete randomized design with 2 factors: The first factor is the type of matrix, including Sodium Alginate (SA), Carrageenan (CG) and a combination of both (SC), and the second factor is the dose used, including 1.25%, 1.75% and 2.25%. The results showed that SA with a dose of 2.25% was the best treatment with a viscosity value of 3,260 cp, gel strength of 14.6 N, viability of 1.45 Log CFU mL−1, and microcapsule efficiency of 97.61%. The quality of capsules is strongly influenced by viscosity and gel strength parameters. The higher the viscosity, the faster the gel formation process, and the stronger the resulting gel, the better it maintains the stability of bacteria in the capsule. Functional group analysis showed similar results, with carboxyl and hydroxyl groups in sodium alginate and sulphate ester groups in carrageenan detected in the IR spectrum absorption. The main groups in each structure play a role in the cross-linking process. The surface characteristics of the capsules also showed drier, rougher, and stronger conditions in alginate. Based on the data obtained, using SA at a dose of 2.25% is the appropriate composition that can be used.
Keywords: Encapsulation, Synbiotic, Probiotic, Prebiotic, Viability, Efficiency of encapsulation, Viscosity, Gel strength, Alginate, Carrageenan
The use of probiotics in aquaculture is a breakthrough has helped to increase aquaculture production. The use of probiotics in feed can help increase the level of feed digestibility and provide good health impacts for the host [1]. Probiotics are highly recommended in aquaculture as an environmentally friendly disease prevention method [2]. Probiotics can benefit the host if they are given in sufficient quantities [3]. The use of probiotics is good at a minimum dose of 106 CFU mL−1 and is recommended at a dose of 107 - 109 CFU mL−1 [4,5]. Appropriate probiotics will make it easier for probiotic bacteria to form colonies and multiply in the intestines. Lactobacillus casei, Bacillus subtilis and Saccharomyces cerevisiae are probiotics that are often used and have a role in increasing digestive enzyme activity [6-8]. in addition, nitrifying bacteria are also often used in feed to maximize feed utilization and decompose organic matter from feed residues in water. The application of Nitrosomonas and Nitrobacter bacteria has been reported to reduce ammonia and Total Suspended Solid (TSS) levels in water [9].
Synbiotics are the latest method to encourage the development of good microbiota in the intestine. The application of synergy by providing probiotics and prebiotics as nutrients is an alternative to maintaining bacterial stability in the intestine. The use of synbiotics has also been shown to be more effective when compared to giving probiotics alone [10]. Synbiotic powder has excellent advantages in maintaining stability in the digestive environment by forming probiotic bacterial sporulation. The formation of bacillus bacterial spores has been reported to support bacterial survival in less supportive environmental conditions [11]. However, this application is still limited to bacteria that can form spores only, the digestive tract has extreme conditions and still threatens the stability and viability of several other probiotic bacteria to reach the target location [12]. The diversity of temperature, pH, oxygen and water content conditions in the digestive system is still variable, which affects bacteria’s survival and viability [13]. This will be worse if it occurs together with other inhibiting factors such as diet, age, host genetics and environment [14]
Encapsulation techniques are a measure that can be used to protect functional ingredients, such as synbiotic powder, in the digestive environment. Encapsulation techniques can prevent a decrease in viability and maintain stability in the digestive tract [15]. Additionally, it has been reported that synbiotic encapsulation can enhance bacterial viability [16]. However, environmental conditions during encapsulation often pose challenges that can reduce probiotic viability. Previous studies have reported that during the drying process of encapsulation, there was a significant decrease in the viability of the probiotic bacteria used [17]. Another study also reported that temperature during the encapsulation process significantly affects the survival of probiotic cells [18]. The composition of the matrix material in encapsulation needs to be optimized to enhance the protection of the core material from environmental influences (temperature, humidity, and other interfering substances) during the encapsulation processing period. Additionally, resistance to the digestive tract must be considered when selecting matrix materials. The chosen material must withstand the physical and chemical properties of the digestive tract and facilitate controlled release at the target area [19].
Polysaccharides are the most commonly used materials as a matrix for probiotic encapsulation. Polysaccharides have characteristics that are good enough to be used as microcapsule walls, such as gelation properties, structural strengthening agents, and enteric dissolution, which allows polysaccharides to dissolve only in the intestinal environment and are prebiotic [20]. Maltodextrin is a material often used as a microcapsule matrix; this modified starch material is still imported for food and medicine needs [21]. Therefore, it is necessary to use other alternatives that can meet the availability of capsules. Alginate and carrageenan are types of polysaccharides contained in seaweed, both of which have abundant sources of materials. Alginate and carrageenan have properties suitable as gel-forming compounds, with sufficiently high viscosity and good gel strength, making them potentially useful as encapsulation matrix materials [22-24]. Therefore, it is necessary to optimize the composition of the matrix and the appropriate processing techniques to maintain the stability of the synbiotic powder within the capsules, thereby enhancing its effectiveness in cultivation.
Materials and methods
Materials
Sodium Alginate was extracted directly from E. cottoni from the Balai Perikanan Budidaya Air Payau (BPBAP) Situbondo, and Carrageenan from Sargassum sp. from the Balai Besar Perikanan Budidaya Air Payau (BBPBAP) Jepara. Probiotics used in this study were Bacillus subtilis, Lactobacillus casei, Nitrosomonas, Nitrobacter and Yeast Saccharomyces cerevisiae obtained from the Faculty of Medicine, Universitas Brawijaya. The probiotic fermentation ingredients used were molasses, brown sugar, rice bran and other additives such as ginger, turmeric, javanese tumeric pineapple, and milk. The synbiotic ingredients used were rice flour, tapioca flour, coconut water and garlic as a source of prebiotics. Encapsulation process materials included 0.9% NaCl, 0.2 M CaCl2 and 0.3 M KCL. The ingredients for calculating total bacteria were agar (PCA), 0.9% NaCl and distilled water.
Seaweed extraction
The carrageenan extraction method refers to previous research methods [25], where seaweed is washed to remove salt content and cut into 2 - 4 cm lengths. Extraction is carried out using 100 grams of dried seaweed, then extracted using 4% hot KOH solution (1L) for 2 h at a temperature of 80 °C. Then the seaweed is washed to remove KOH, and the pH of the seaweed becomes 8 - 9. The seaweed is soaked in water with a ratio of 1:20 for 30 min at a temperature of 80 °C and then filtered with a calico cloth. The resulting filtrate is then added to 2% KCl solution (1:2) at a temperature of 30 °C. The obtained carrageenan fibres were then dried at 60 °C and ground into a fine powder until they passed a sieve of 60 mesh.
The SA extraction method used refers to previous research methods [26,27], Extraction was done using 25 g of dried seaweed, then soaked in 800 mL of 2% formaldehyde for 24 h at room temperature. The sample was washed with water, then 0.2 M HCl (800 mL) was added and left again for 24 h. After that, the sample was washed with deionized water and extracted using 2% sodium carbonate for 3 h at 100 °C. The extracted solution was filtered and centrifuged (4,000 rpm) for 30 min. After that, the filtrate was added with 2.5% sodium hypochlorite to be bleached until yellowish white. Then, the products were precipitated with 95% ethanol, three times the sample volume. The collected sodium alginate was washed twice with 100 mL of acetone and dried at 60 °C.
Probiotic culture
Cultures were carried out using the streak plate method on the prepared TSA and NA media. After that, incubation was carried out at 37 °C for 24 h. After the re-culture process, a scale-up was carried out on liquid media. The bacterial culture process on liquid media was carried out using TSB media for Nitrosomonas and Nitrobacter bacteria, NB media for B. subtilis and L. casei bacteria, and 0.9% NaCl for S. cerevisiae yeast. After planting, the culture process was carried out using a shaker incubator at 37° C for 24 h at 175 rpm.
The preparation of synbiotics begins with making a probiotic fermentation solution. The fermentation preparation process begins with mixing red ginger, turmeric, and javanese turmeric (1 kg) and boiling in 11 L of water. Next, 1 kg of brown sugar and 400 g of rice bran are added and reheated until boiling and well mixed. After that, wait for the temperature to drop to 60 - 70 °C to add 600 g of pineapple, 1 L of milk, and 1 L of molasses, and reheat until boiling at medium temperature. After boiling, the solution is then allowed to cool to room temperature. Next, probiotics were mixed with 100 mL each of Nitrosomonas, Nitrobacter, B. subtilis, and L. casei and 24 mL each of S. cerevisiae with a density of 108 CFU mL−1 [5]. The solution was then fermented for 2 weeks at room temperature and in dark conditions [28].
Synbiotics were prepared by mixing (spray method) 100 mL of fermented liquid probiotics into 100 g of sterilised tapioca and rice flour. After that, 50 mL of coconut water and 50 g of garlic were added. After the ingredients were mixed, they were dried in a dehydrator at 35 °C for 48 h. The manufacturing process is also presented in Figure 1.
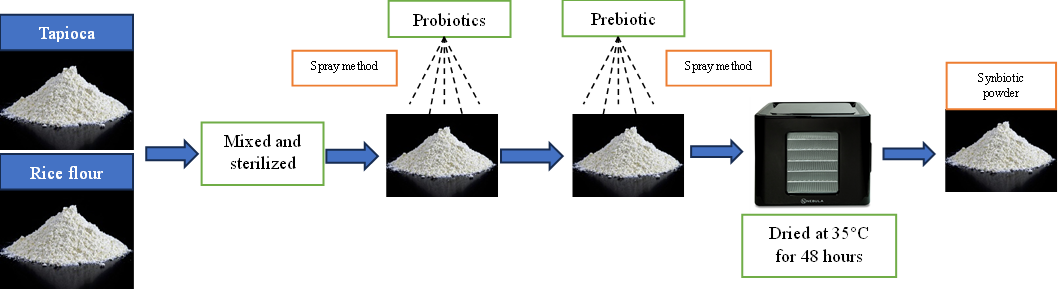
Figure 1 Synbiotic preparation.
The synbiotic encapsulation process was carried out using the extrusion method [29]. The encapsulation process was carried out by dissolving the matrix (SA, CG, and SC) as much as 1.25%, 1.75% and 2.25% into 0.9% NaCl and heating until the material dissolved. Then, let the matrix solution stand at 40 °C and mix the synbiotic powder into it. The suspension obtained was then put into a syringe, and microcapsule granules were formed by pouring it into 0.3 M KCl or 0.2 M CaCl2. The formed capsules were then filtered and rinsed again with 0.9% sterile NaCl and dried in a dehydrator at 40 °C for 2 h (Figure 2).
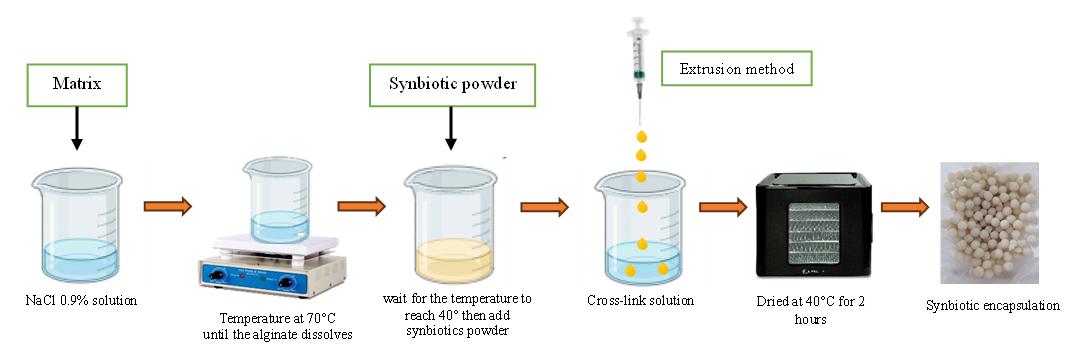
Figure 2 Encapsulation precessing.
Analysis parameters
Microcapsule structure and diameter
Morphological capsule analysis used the Scanning Electron Microscope (SEM) method. Testing was carried out at the Integrated Research Laboratory, Universitas Brawijaya. SEM testing was carried out to determine capsule characteristics, the structure and shape of the particles, and the surface condition, specifically to determine differences in the analysis of the use of different matrix materials and capsule characteristics in the storage process.
Viability of probiotic
Bacterial viability was determined to determine the number of probiotic bacteria in the sample to be taken and to identify changes in viability that occurred before and after treatment. This bacterial cell calculation was done by taking 1 g of the capsules used as a sample and suspending them in 9 mL of 0.9% NaCl [30]. Furthermore, the sample was stirred for 25 min until the microcapsule polymer broke (the mixture was cloudy) and vortexed until homogeneous. After homogeneity, the next step is to dilute and count bacterial cells by taking 100 microliters of the dilution results into agar media, then incubating for 48 h at 37 °C [31].
Encapsulation efficiency
Encapsulation efficiency was calculated by comparing the efficiency of capsule use in protecting bacterial viability before and after encapsulation. This parameter was used to determine how efficient the material was in protecting the core material (bacteria) from other factors. Microcapsule efficiency can be calculated using the following formula [32].
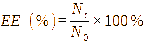
where, EE: Encapsulation Efficiency; Nt: Bacterial viability after encapsulation (Log CFU mL−1); N0: Bacterial viability before encapsulation (Log CFU mL−1)
Viscosity of capsules
Viscosity measures a liquid's ability to not flow. Viscosity testing is carried out using a Brookfield brand Vicometer at the Food Safety and Quality Testing Laboratory, Faculty of Agricultural Technology, Universitas Brawijaya, Malang. The encapsulation matrix formulation must have optimal viscosity; this allows the core material to remain stable and facilitates optimal release transitions.
Functional group analysis
This test was conducted using a Fourier Transform Infrared Spectrometer (FT-IR) at the Integrated Laboratory, Faculty of Agricultural Technology, Universitas Brawijaya. This test aimed to determine the functional groups contained in the capsules before and after the encapsulation process and shelf life testing.
Results and discussion
Viscosity of encapsulation
The use of different types and doses of matrix, based on Table 1, shows a significantly different effect on the viscosity value of the capsules. The viscosity results for each matrix type increased along with the matrix dose used. SA and CG experienced a significant increase in viscosity values at a dose of 2.25%. However, they had the same increase pattern; the viscosity value of SA at that dose was much higher than that of CG and all treatments. While SC had a different increase pattern compared to SA and CG. The viscosity value of SC also increased at a dose of 2.25% but was not higher than the increase that occurred at a dose of 1.75%. The combination of alginate and carrageenan can form hydrogen bonds through molecular interactions through the carboxyl and hydroxyl ether groups in it [33], so that the microstructure formed from this polymer combination can experience changes in physical and chemical properties, such as viscosity and tensile strength, from the use of a single polymer. In addition, it has been reported that each coating material has a different maximum limit for increasing viscosity until there is no further increase [34].
Table 1 The effect of different types of matrix and doses.
Parameters |
SA |
CG |
SC |
||||||
1.25% |
1.75% |
2.25% |
1.25% |
1.75% |
2.25% |
1.25% |
1.75% |
2.25% |
|
Viscosity (cp) |
540.00f |
623.33f |
3260.00a |
783.33ef |
996.67e |
2633.33b |
523.33f |
1616.67d |
1943.33c |
Gel strength (N) |
7.03d |
7.90cd |
14.60a |
4.10e |
7.43d |
11.13b |
5.10e |
8.60c |
7.90cd |
Viability (Log CFU mL−1) |
9.23cd |
9.29bc |
9.43a |
9.13e |
9.25bc |
9.32b |
9.18de |
9.27bc |
9.27bc |
EE (%) |
97.47cd |
98.11bc |
99.55a |
96.42e |
97.68bcd |
98.40b |
96.95de |
97.92bc |
97.86bc |
Gel strength of encapsulation
Gel strength has a result that is directly proportional to the dose used; the higher the dose used, the greater the gel strength will be (Table 1). This increase is also the same as the viscosity value, which increases with the addition of the dose given. SA has a higher gel strength value than other types of matrix. The gel strength of CG has a relatively similar increase in each dose addition, while in the SA matrix, with a low dose, there is a low increase, different from the dose of 2.25% which experienced a significant increase of more than 80% from the dose of 1.75%. The gel strength of SA with a dose of 2.25% is the highest result compared to other treatments; this result is also comparable to the viscosity value in this treatment, which is also the highest result. Previous studies have reported alginate has a higher tensile strength than carrageenan [35]. Linear copolymer blocks of guluronic acid (G) and mannuronic acid (M) are polymers that play a vital role in the physical properties of alginate, including gel strength [36]. The proximity of the number of G-block bonds and the length of G-block damage influences the gel strength value of sodium alginate.
Viability of synbiotic encapsulation
The viability value in each treatment is directly proportional to the gel strength value of the capsules (Table 1). Increasing the gel strength of the capsules also increases the viability value of bacteria in the capsules. SA has the highest viability value among other treatments; the higher the dose used, the higher the bacterial viability value. Gel strength and viscosity are the main parameters to determine the quality of entrapment in capsules, based on previous studies with different matrix materials showing that materials with high viscosity and strength values also have high viability and
encapsulation efficiency [37]. These results indicate that the higher the viscosity and gel strength values, the faster the gel formation process and the stronger the gel. The higher the viscosity and gel strength values, the higher the chance of the core material being protected in the capsules. Environmental parameters are factors that affect the viability of capsules. The drying process in capsules can cause damage and evaporation of the core material in the capsules if the matrix material used has low gel strength and viscosity. Other studies have also explained that high viability values can be caused by calcium cross-linking with alginate, which increases the strength of the gel to form a protective matrix [38].
The encapsulation efficiency value compares the viability of bacteria in the powder and the viability of bacteria after the encapsulation process. The viability value obtained in the synbiotic powder before encapsulation was 9.47 Log CFU mL−1. The efficiency value from Table 1 shows that the type of matrix and different doses greatly affect the encapsulation efficiency value. The highest efficiency value in this study was 99.55% in SA with a dose of 2.25%, which was higher than several other materials that have been used, such as starch, gellan, and gum arabic, which were 85% - 95% [39-41]. The efficiency value is directly proportional to the viability value of bacteria in the capsules. The increase in viability in capsules also indicates that the capsules made have high efficiency. This efficiency value is influenced by high gel strength and viscosity; high viscosity helps accelerate gel formation and increase gel strength, so that the core material can be better protected [42]. Higher doses in each matrix showed an average increase in the resulting efficiency value, which is also directly proportional to the gel strength value obtained. This result can be explained by the fact that increasing biopolymer molecules in the microcapsule volume, which can also increase the number of calcium ion binding sites [43]. The high content of -COOH and -OH in alginate and carrageenan allows the formation of a dense network to occur because the cross-linking of biopolymers through hydrogen bonds and ionic interactions causes a decrease in the distance between alginate and carrageenan molecules, so that it can encourage the formation of a denser network structure [44].
IR spectral characterization
Based on the spectral results of the IR functional group analysis, the capsules in the SA and CG matrix have similar spectra. The functional groups in the capsules show the presence of C=O groups in each treatment with absorption wave numbers of 1,632.36 - 1,635.73 cm−1 (Figure 3(a)). In addition, absorption at a wavelength of 1,233.62 - 1,236.49 cm−1 in SA indicates the presence of a C-O group (Figure 3SA(b)). The C-O group that appears at these wave numbers has low intensity, indicating that many C-O groups in alginate are bound to Ca2+ as a cross-linking agent for the -COO group [45]. Carboxyl groups (COOH) in alginate and carrageenan were detected through absorption at wavelengths of 3,235.55 - 3,239.86 cm−1 (Figure 3(c)). β-D-mannuronic acid (M) and α-L-guluronic acid (G) are the structural components of alginate that have free carboxyl and hydroxyl groups in their polymer chains as the main functional groups [46]. These groups play a role in facilitating the gelation process with divalent cations in the form of calcium ions through a cross-linking process [47]. The presence of this absorption indicates that both functional groups are active in the process of gel structure formation in the cross-linking process. The carboxyl group of alginate, the carboxyl group, and the sulphate group of carrageenan can interact with calcium ions through ionic cross-linking with divalent calcium ions [48]. These bonds play a role in gel formation in capsules with a combination of SA and CG matrix.
Absorption at wavelengths of 1,232.18 - 1,233.62 cm−1 in the CG matrix indicates the presence of sulphate ester groups that play a role in gel formation in the CG matrix (Figure 3CG(b)). In general, carrageenan has sulphate ester groups (-OSO₃⁻) as the main anionic groups. Carrageenan contains about 15% - 40% sulphate esters, except in the β-carrageenan structure, which does not contain sulphate [49]. Sulphate ester groups in carrageenan play a role in the cross-linking process for gel structure formation. Ca2+ ions form intra- or intermolecular cross-bridges between sulphate ester groups in the carrageenan chain [50].
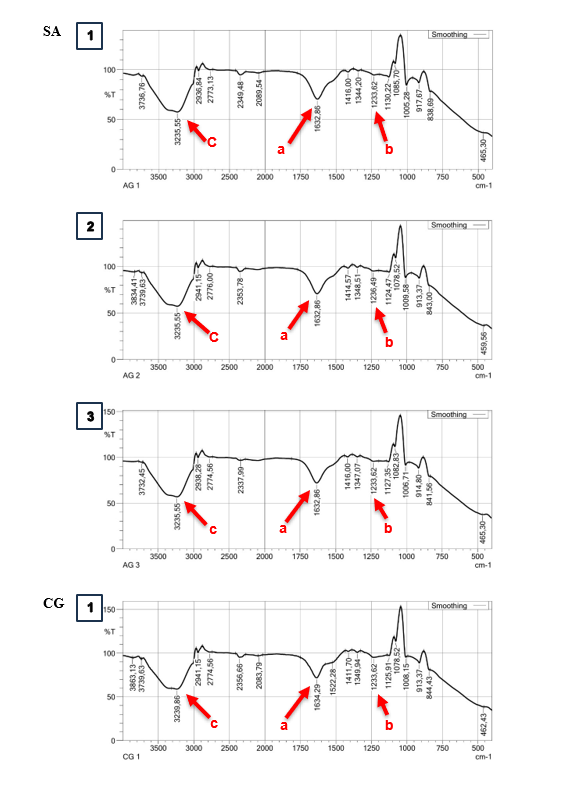
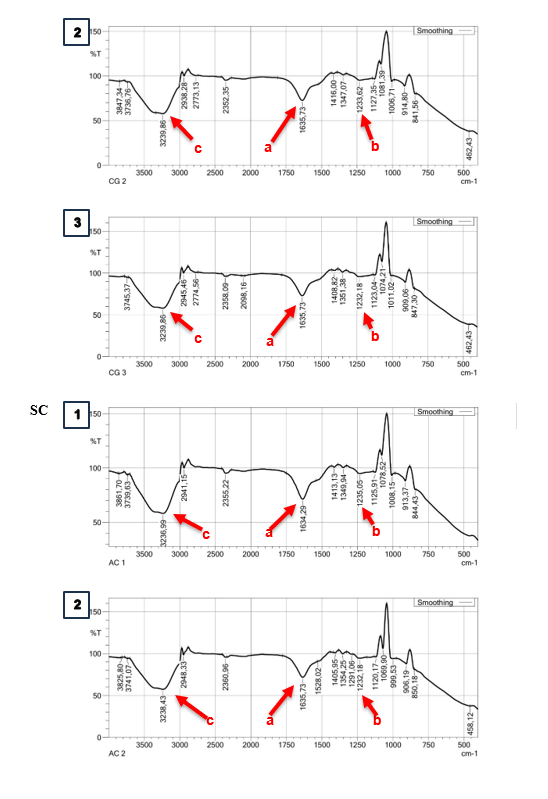
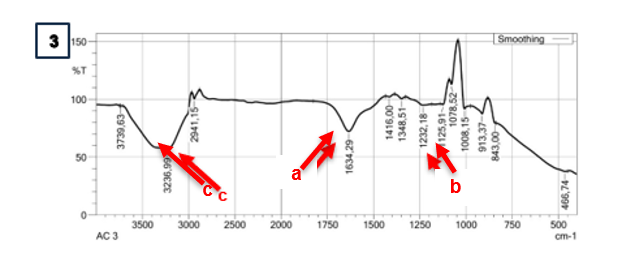
Figure 3 IR spectra analysis on different types of matrix and doses. Sodium Alginate (SA); Carrageenan (CG); Alginate + Carrageenan (SC); 1,25% (1); 1,75% (2); 2,25% (3).
Analysis of encapsulation surface structure
A scanning electron microscope (SEM) was used in this study to evaluate the surface structure produced on various types of matrix in this study. The SA 2.25% matrix type was observed to be the best treatment compared to other treatments in this study (Table 1). Maltodextrin and dextrin were also observed as comparison matrix in combination with 1% SA (Figure 4). Based on the results obtained, it can be observed that the particle structure formed on the surface of each material has an irregular agglomeration shape, but with different conditions. The matrix type and prebiotic material used have hygroscopic properties, causing the material to clump, resulting in an irregular structure [51]. The surface structure of the maltodextrin and dextrin matrix capsules is smoother and moister (Figure 4, DX; MD). Maltodextrin and dextrin matrix have lower viscosity than alginate, so during the mixing process, the core material dissolves more easily, resulting in a smoother and moister surface structure. Based on research [52], it also shows that alginate and maltodextrin microparticles have a smoother surface structure. However, the moist condition of the capsule can cause a decrease in shelf life quality because it can cause biological damage [53].
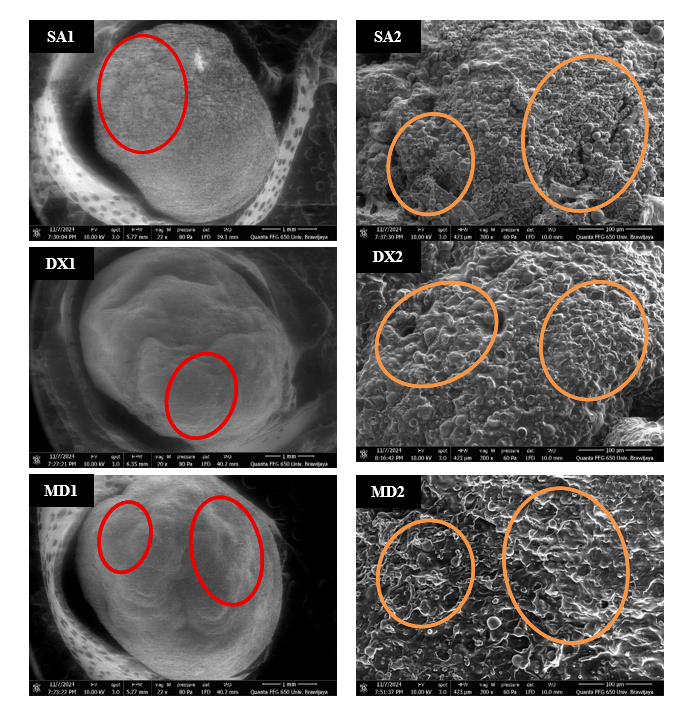
Figure 4 Scanning Electron Microscopy (SEM) analysis; Sodium Alginate 2.25% (SA1 (Magnification: 22×) and SA2 (Magnification: 300×)); Alginate 1% + dextrin (DX1 (Magnification: 22×) and DX2 (Magnification: 300×)); Alginate 1% + Maltodextrin (MD1 (Magnification: 22×) and MD2 (Magnification: 300×)). The red circles indicate the rough and smooth characteristics of the capsule surface. The orange circles indicate the dry and moist characteristics of the capsule surface.
In addition, capsules with the SA matrix showed rougher, wrinkled, and drier surface structures than other matrix structures (Figure 4SA). Another study explained that CaCl₂ as a binding solution plays a role in changing the surface properties of microparticles [54]. Calcium ions in solution can form cross-links with alginate gel, which can cause the outer layer to become denser [55]. A denser capsule surface indicates good strength in protecting the capsule core material; surface density can prevent leakage of the core material and protect it from external disturbances. Previous studies have also shown that capsules with alginate material exhibit a stronger gel structure than other materials. The absence of gaps or cracks on the capsule surface is significant in ensuring lower gas permeability and improving the protection of encapsulated probiotic cells, thereby reducing the adverse effects of external factors on the cells [52,56]. Additionally, using FOS in crude garlic extract also plays a role in narrowing the encapsulation pores. FOS can produce a more cohesive structure, smaller pores, and higher viscosity, significantly affecting the gel surface structure [57].
Conclusions
Based on the results obtained, conclusions were drawn to determine the optimal process of making synbiotic powder encapsulation. SA at a dose of 2.25% is the best treatment application in this study to maintain viability and increase the Encapsulation Efficiency value. This treatment obtained the highest viscosity and gel strength values at 3,260 cp and 14.6 N, respectively. High viscosity and gel strength values can improve the function of the capsule wall in protecting the core material from environmental influences. The surface structure of the capsule also showed a dense and dry condition, making it very suitable for protecting the core material from external factors.
Acknowledgements
This work was supported by Universitas Brawijaya, Indonesia. Professor Grant Research Scheme, Universitas Brawijaya, with contract number: 3709/UN10.F06/KS/2024.
Declaration of generative AI in scientific writing
The authors acknowledge the use of generative AI tools such as DeepL and ChatGPT from OpenAI, which were used specifically to assist with language editing and grammar correction in the preparation of this manuscript. The authors did not use AI tools to create content or interpret data. The authors take full responsibility for the content and conclusions written in this manuscript.
CRediT author statement
Lintang Dion Pangestu: Conceptualization, Methodology, Data curation, Software, Formal analysis, Investigation, Validation, Visualization, and Writing - Drafting of original manuscript. I Made Dedi Mahariawan: Conceptualization, Methodology, Supervision, Data curation, Formal analysis, Investigation, Validation, Project administration, and Writing - Review & Editing manuscript. Muhammad Fakhri: Conceptualization, Methodology, Supervision, Validation, and Writing - Review & Editing manuscript. Abdul Rahem Faqih: Conceptualization, Methodology, Supervision, Validation, and Writing - Review & Editing manuscript. Ating Yuniarti: Conceptualization, Methodology, Supervision, Validation, and Writing - Review & Editing manuscript. Anik Martinah Hariati: Conceptualization, Methodology, Supervision, Validation, Fundraising, Project Administration and Writing - Review & Editing manuscript.
[1] AM Tangko, A Mansyur and R Reski. Use of probiotics in milkfish cultivation feed in floating net cages in the sea. Jurnal Riset Akuakultur 2016; 2(1), 33-40.
[2] YB Wang, JR Li and J Lin. Probiotics in aquaculture: Challenges and outlook. Aquaculture 2008; 281(1-4), 1-4.
[3] G De Marco, T Cappello and M Maisano. Histomorphological changes in fish gut in response to prebiotics and probiotics treatment to improve their health status: A Review. Animals 2023; 13(18), 2860.
[4] M Mohamadzadeh, A Fazeli and SA Shojaosadati. Polysaccharides and proteins-based bionanocomposites for microencapsulation of probiotics to improve stability and viability in the gastrointestinal tract: A review. International Journal of Biological Macromolecules 2024; 259, 129287.
[5] MK Tripathi and SK Giri. Probiotic functional foods: Survival of probiotics during processing and storage. Journal of Functional Foods 2014; 9, 225-241.
[6] N Aini, DSYR Putri, DH Achhlam, F Fatimah, S Andriyono, D Hariani, HDK Do and SPA Wahyuningsih. Supplementation of Bacillus subtilis and Lactobacillus casei to increase growth performance and immune system of catfish (Clarias gariepinus) due to Aeromonas hydrophila infection. Veterinary World 2024; 17(3), 602.
[7] MN Monier, H Kabary, A Elfeky, S Saadony, NNB Abd El‑Hamed, MEH Eissa and ESH Eissa. The effects of Bacillus species probiotics (Bacillus subtilis and B. licheniformis) on the water quality, immune responses, and resistance of whiteleg shrimp (Litopenaeus vannamei) against Fusarium solani infection. Aquaculture International 2023; 31(6), 3437-3455.
[8] N Sutthi and W Thaimuangphol. Effects of yeast (Saccharomyces cerevisiae) on growth performances, body composition and blood chemistry of Nile tilapia (Oreochromis niloticus Linnaeus, 1758) under different salinity conditions. Iranian journal of Fisheries Sciences 2020; 19(3), 1428-1446.
[9] KH Dwiardani, Prayogo and BS Rahardja. Utilization of Nitrosomonas sp and Nitrobacter sp probiotic towards total suspended solid and ammonia level in nile tilapia culturing using aquaponic system. IOP Conference Series: Earth and Environmental Science 2021; 679(1), 012067.
[10] DL Merrifield, A Dimitroglou, A Foey, SJ Davies, RTM Baker, J Bøgwald, M Castex and E Ringø. The current status and future focus of probiotic and prebiotic applications for salmonids. Aquaculture 2010; 302(1-2), 1-18.
[11] IMD Mahariawan, NB Ariffin, WE Kusuma, A Yuniarti, MAG Beltran and AM Hariati. Effect of different carbon doses of tapioca (Manihot esculenta) flour on vegetative cells and spore production of Bacillus megaterium. IOP Conference Series: Earth and Environmental Science 2020; 441(1), 012106.
[12] WY Koh, XX Lim, TC Tan, R Kobun and B Rasti. Encapsulated probiotics: Potential techniques and coating materials for non-dairy food applications. Applied Sciences 2022; 12(19), 10005.
[13] S Razavi, S Janfaza, N Tasnim, DL Gibson and M Hoorfar. Microencapsulating polymers for probiotics delivery systems: Preparation, characterization, and applications. Food Hydrocolloids 2020; 120, 106882.
[14] AD Diwan, SN Harke and AN Panche. Host-microbiome interaction in fish and shellfish: An overview. Fish and Shellfish Immunology Reports 2023; 4, 100091.
[15] S Iravani, H Korbekandi and SV Mirmohammadi. Technology and potential applications of probiotic encapsulation in fermented milk products. Journal of Food Science and Technology 2015; 52(8), 4679-4696.
[16] AP do Carmo Alves, A do Carmo Alves, RA Ferreira Rodrigues, B da Silva Cerozi and JE Possebon Cyrino. Microencapsulation of Bacillus subtilis and oat β-glucan and their application as a synbiotic in fish feed. Journal of Microencapsulation 2023; 40(7), 491-501.
[17] MP Silva, FL Tulini, E Martins, M Penning, CS Fávaro-Trindade and D Poncelet. Comparison of extrusion and co-extrusion encapsulation techniques to protect Lactobacillus acidophilus LA3 in simulated gastrointestinal fluids. LWT 2018; 89, 392-399.
[18] M Yin, L Chen, M Chen, Y Yuan, F Liu and F Zhong. Encapsulation of Lactobacillus rhamnosus GG in double emulsions: Role of prebiotics in improving probiotics survival during spray drying and storage. Food Hydrocolloids 2024; 151, 109792.
[19] A Al-Hamayda, B Abu-Jdayil, M Ayash and J Tannous. Advances in microencapsulation techniques using Arabic gum: A comprehensive review. Industrial Crops and Products 2023; 205, 117556.
[20] H Liu, M Xie and S Nie. Recent trends and applications of polysaccharides for microencapsulation of probiotics. Food Frontiers 2020; 1(1), 45-59.
[21] V Derosya and A Kasim. Optimization of maltodextrin production based on sago starch using α-amylase and spray drying method. Jurnal Teknologi Pertanian Andalas 2017; 21(1), 28-32.
[22] LP Ta, E Bujna, O Antal, M Ladányi, R Juhász, A Szécsi, S Kun, S Sudheer, VK Gupta and QD Nguyen. Effects of various polysaccharides (alginate, carrageenan, gums, chitosan) and their combination with prebiotic saccharides (resistant starch, lactosucrose, lactulose) on the encapsulation of probiotic bacteria Lactobacillus casei 01 strain. International Journal of Biological Macromolecules 2021; 183, 1136-1144.
[23] J Lai, AK Azad, WMAW Sulaiman, V Kumarasamy, V Subramaniyan and SA Alshehade. Alginate-Based Encapsulation fabrication technique for drug delivery: an updated review of particle type, formulation technique, pharmaceutical ingredient, and targeted delivery system. multidisciplinary digital publishing institute (MDPI). Pharmaceutics 2024; 16(3), 370.
[24] Z Cheng, B Zhang, D Qiao, X Yan, S Zhao, C Jia, M Niu and Y Xu. Addition of κ-carrageenan increases the strength and chewiness of gelatin-based composite gel. Food Hydrocolloids 2022; 128, 107565.
[25] A Diharmi, Rusnawati and N Irasari. Characteristic of carrageenan Eucheuma cottonii collected from the coast of Tanjung Medang Village and Jaga Island, Riau. IOP Conference Series: Earth and Environmental Science 2019; 404(1), 012049.
[26] TA Fenoradosoa, G Ali, C Delattre, C Laroche, E Petit, A Wadouachi and P Michaud. Extraction and characterization of an alginate from the brown seaweed Sargassum turbinarioides Grunow. Journal of Applied Phycology 2010; 22(2), 131-137.
[27] SH Rashedy, MSM Abd El Hafez, MA Dar, J Cotas and L Pereira. Evaluation and characterization of alginate extracted from brown seaweed collected in the red sea. Applied Sciences 2021; 11(14), 6290.
[28] DA Fernanda and D Hariani. The effect of administration of synbiotic and enzyme with various concentrations on feed to the growth of GIFT tilapia seeds (Oreochromis sp.). LenteraBio: Berkala Ilmiah Biologi 2021; 23(2), 113-118.
[29] S Moumita, K Goderska, EM Johnson, B Das, D Indira, R Yadav, S Kumar and R Jayabalan. Evaluation of the viability of free and encapsulated lactic acid bacteria using in-vitro gastro intestinal model and survivability studies of synbiotic microcapsules in dry food matrix during storage. LWT 2017; 77, 460-467.
[30] DC Campos, F Acevedo, E Morales, J Aravena, V Amiard, MA Jorquera, NG Inostroza and M Rubilar. Microencapsulation by spray drying of nitrogen-fixing bacteria associated with lupin nodules. World Journal of Microbiology and Biotechnology 2014; 30(9), 2371-2378.
[31] P Pupa, P Apiwatsiri, W Sirichokchatchawan, N Pirarat, N Muangsin, AA Shah and N Prapasarakul. The efficacy of three double-microencapsulation methods for preservation of probiotic bacteria. Scientific Reports 2021; 11(1), 13753.
[32] W Sun, QD Nguyen, G Sipiczki, SR Ziane, K Hristovski, L Friedrich, A Visy, G Hitka, A Gere and E Bujna. Microencapsulation of Lactobacillus plantarum 299v strain with whey proteins by lyophilization and its application in production of probiotic apple juices. Applied Sciences 2023; 13(1), 318.
[33] RA Rather, MA Bhat and AH Shalla. Multicomponent interpenetrating metal based Alginate-Carrageenan biopolymer hydrogel beads substantiated by graphene oxide for efficient removal of methylene blue from waste water. Chemical Engineering Research and Design 2022; 182, 604-615.
[34] DM Sumanti, I Lanti, II Hanidah, E Sukarminah and A Giovanni. The effect ofskim milk and maltodextrin concentration as coating agent towards viability and characteristics of Lactobacillus plantarum bacteria microencapsulated suspension using freeze drying method. Jurnal Penelitian Pangan 2016; 1, 7-13.
[35] G Kavoosi, M Derakhshan, M Salehi and L Rahmati. Microencapsulation of zataria essential oil in agar, alginate and carrageenan. Innovative Food Science and Emerging Technologies 2018; 45, 418-425.
[36] MP Silva, IJ Badruddin, T Tonon, S Rahatekar and LD Gomez. Environmentally benign alginate extraction and fibres spinning from different European Brown algae species. International Journal of Biological Macromolecules 2023; 226, 434-442.
[37] M Khorshidi, A Heshmati, M Taheri, M Karami and R Mahjub. Effect of whey protein- and xanthan-based coating on the viability of microencapsulated Lactobacillus acidophilus and physiochemical, textural, and sensorial properties of yogurt. Food Science & Nutrition 2021; 9(7), 3942-3953.
[38] NMY Jacobsen, I Caglayan, A Caglayan, D Bar-Shalom and A Müllertz. Achieving delayed release of freeze-dried probiotic strains by extrusion, spheronization and fluid bed coating - evaluated using a three-step in vitro model. International Journal of Pharmaceutics 2020; 591, 120022.
[39] L Shoukat, S Javed, M Afzaal, N Akhter and YA Shah. Starch-based encapsulation to enhance probiotic viability in simulated digestion conditions. International Journal of Biological Macromolecules 2024; 283, 137606.
[40] R González-Cuello, J Hernández-Fernández and R Ortega-Toro. Optimization of the viability of microencapsulated Lactobacillus reuteri in gellan gum-based composites using a box-behnken design. Journal of Composites Science 2025; 9(8), 419.
[41] R Kamwa, B Khurajog, N Muangsin, P Pupa, DJ Hampson and N Prapasarakul. Water-soluble microencapsulation using gum Arabic and skim milk enhances viability and efficacy of Pediococcus acidilactici probiotic strains for application in broiler chickens. Animal Bioscience 2024; 37(8), 1440.
[42] MM Saputra, Y Wuryandari and N Ramadhini. Analysis of encapsulation efficiency Bacillus sp. based variations in sodium alginate concentration in the beads. NST Proceeding 2024; 2024, 7-14.
[43] Y Zhou, D Xu, H Yu, J Han, W Liu and D Qu. Encapsulation of Salmonella phage SL01 in alginate/carrageenan microcapsules as a delivery system and its application in vitro. Frontiers in Microbiology 2022; 13, 906103.
[44] F Yu, T Cui, C Yang, X Dai and J Ma. Κ-Carrageenan/sodium alginate double-network hydrogel with enhanced mechanical properties, anti-swelling, and adsorption capacity. Chemosphere 2019; 237, 124417.
[45] TN Hidayah, D Djaenudin and N Lubis. Encapsulation of probiotic Lactobacillus sp. using alginate and chitosan biopolymers with one-step method. Jurnal Serambi Engineering 2021; 6(2), 1814-1825.
[46] S Akshaya and AJ Nathanael. A review on hydrophobically associated alginates: Approaches and applications. American Chemical Society 2024; 9(4), 4246-4262.
[47] SM Shahrbabak, SM Jalali, MF Fathabadi, V Tayebi-Khorrami, M Amirinejad, S Forootan, M Saberifar, MR Fadaei, Z Najafi and VR Askari. Modified alginates for precision drug delivery: Advances in controlled-release and targeting systems. International Journal of Pharmaceutics: X 2025; 10, 100381.
[48] MH Kim, YW Lee, WK Jung, J Oh and SY Nam. Enhanced rheological behaviors of alginate hydrogels with carrageenan for extrusion-based bioprinting. Journal of the Mechanical Behavior of Biomedical Materials 2019; 98, 187-194.
[49] L Cunha and A Grenha. Sulfated seaweed polysaccharides as multifunctional materials in drug delivery applications. Marine drugs 2016; 14(3), 42.
[50] M Ciancia, MC Matulewicz and R Tuvikene. Structural diversity in galactans from red seaweeds and its influence on rheological properties. Frontier in Plant Science 2020; 11, 559986.
[51] R Rajam, SB Kumar, P Prabhasankar and C Anandharamakrishnan. Microencapsulation of Lactobacillus plantarum MTCC 5422 in fructooligosaccharide and whey protein wall systems and its impact on noodle quality. Journal of Food Science and Technology 2015; 52(7), 4029-4041.
[52] S Pandey, Y Premjit, A Khuntia, A Kadival and J Mitra. Development of probiotic-loaded calcium alginate-maltodextrin microparticles based on electrohydrodynamic technique. Powder Technology 2023; 428, 118808.
[53] LD Pangestu, IMD Mahariawan, A Yuniarti and AM Hariati. Optimization of temperature and drying time of encapsulated synbiotic powder on the characteristics and viability of microcapsules. Jurnal Penelitian Pendidikan 2025; 11(1), 1049-1056.
[54] ES Chan, SL Wong, PP Lee, JS Lee, TB Ti, Z Zhang, D Poncelet, P Ravindra, SH Phan and ZH Yim. Effects of starch filler on the physical properties of lyophilized calcium-alginate beads and the viability of encapsulated cells. Carbohydrate Polymers 2011; 83(1), 225-232.
[55] B Németh, A Ujhidy, J Tóth, L Trif, T Feczkó and R Rauch. Gelation elimination in eco-friendly preparation of double-layered calcium alginate-coconut oil latent heat energy storing microcapsules. Materials Chemistry and Physics 2023; 293, 126889.
[56] Y Premjit and J Mitra. Optimization of electrospray-assisted microencapsulation of probiotics (Leuconostoc lactis) in soy protein isolate-oil particles using box-behnken experimental design. Food and Bioprocess Technology 2021; 14(9), 1712-1729.
[57] KCG Silva and ACK Sato. Biopolymer gels containing fructooligosaccharides. Food Research International 2017; 101, 88-95.