
Trends
Sci.
2026;
23(5):
12139
Characteristics of Intelligent Film based on Glucomannan Porang (Amorphophallus oncophyllus) and Gelatin with Addition of Copigmented Anthocyanins from Rambutan Peel for Shrimp Freshness Monitoring
Ida Bagus Gede Mahatmananda1, Arum Widyastuti Perdani1,2,
Bambang Purwono3, Andriati Ningrum1,* and Supriyadi Supriyadi1,4
1Department of Food and Agricultural Product Technology, Faculty of Agricultural Technology,
Universitas Gadjah Mada, Yogyakarta 55281, Indonesia
2Applied Culinary Arts Study Program, Faculty of Vocational Studies, Universitas Negeri Yogyakarta,
Special Region of Yogyakarta, Gunungkidul Campus, Yogyakarta 55893, Indonesia
3Department of Chemistry, Faculty of Mathematics and Natural Sciences, Universitas Gadjah Mada,
Yogyakarta 55281, Indonesia
4Centre of Excellent for Indonesian Gastronomy, Universitas Gadjah Mada, Yogykarta 55281, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 1 October 2025, Revised: 22 October 2025, Accepted: 10 November 2025, Published: 5 January 2026
Abstract
This study develops intelligent films from porang-derived glucomannan and gelatin enriched with RPAE (native and gallic-acid-copigmented) for shrimp freshness monitoring. The films showed improved performance, TS up to 9.37 MPa, EAB up to 31.90%, and WVP of 3.10-4.72×10⁻7 g·m⁻1·Pa⁻1·h⁻1 together with enhanced radical-scavenging activity. FTIR indicated hydrogen-bonding interactions among gelatin, glucomannan, and anthocyanins, while SEM revealed smoother, more homogeneous morphologies for copigmented systems. The films exhibited a clear pH-dependent optical response with visually perceptible shifts (ΔE > 5). During shrimp storage, TVB-N increased from 28.11 to 71.03 mg N/100 g and TVC from 5.24 to 6.27 log CFU/g; the film color transitioned from green to yellow in parallel with quality deterioration, enabling non-invasive, real-time indication of spoilage. Leveraging locally abundant biopolymers and colorants, these films advance sustainable, intelligent packaging for seafood quality and safety.
Keywords: Anthocyanins, Rambutan, Intelligent films, Glucomannan, Gelatin
Abbreviation
RPAE: Rambutan Peel Anthocyanin Extract
UAE: Ultrasound Assisted Extraction
GA: Gallic Acid
TS: Tensile Strength
EAB: Elongation at Break
∆E: Color Changes
L: Lightness
a: Redness
b: Yellowness
ABTS: 2,2′-Azino-bis-(3-ethylbenzothiazoline-6-sulfonic acid)
DPPH: 2,2-diphenyl-1-picrylhydrazyl
FTIR: Fourier Transform Infra-Red
RSA: Radical Scavenging Activity
TAC: Total Anthocyanin Content
TVB-N: Total Volatile Base Nitrogen
WVP: Water Vapor Permeability
SEM: Scanning Electron Microscope
CFU:
Colony-Forming Units
PCA: Plate Count Agar
TVC: Total Viable Counts
Introduction
The growing global demand for safe and high-quality seafood has underscored the importance of effective preservation technologies. Shrimp, in particular, is highly perishable due to its high protein content and susceptibility to microbial and enzymatic spoilage, which leads to pH elevation and the formation of volatile nitrogen compounds during storage. Conventional packaging materials can provide temporary protection but often rely on synthetic polymers that raise environmental concerns. Consequently, increasing attention has been given to biodegradable and intelligent films as sustainable alternatives for food preservation and freshness monitoring [1].
There have been numerous studies on the development of intelligent films from various biopolymer sources, including carbohydrate-based polymers such as starch, chitosan, carrageenan, and cellulose derivatives; protein-based polymers such as gelatin, soy protein, and whey protein; and blended matrices combining polysaccharides and proteins. These studies demonstrated that incorporating natural pigments, particularly anthocyanins, can provide halochromic properties that enable films to act as freshness indicators. For example, starch-agar films with shikonin were used to monitor shrimp freshness [2], gelatin-bacterial cellulose composites loaded with anthocyanin/curcumin showed high pH sensitivity for pork [3], elderberry anthocyanin films were applied to shrimp [4], and dragon fruit peel anthocyanin films were developed as intelligent pH indicators [5]. Similarly, various anthocyanin-based films from hibiscus, butterfly pea, roselle, or black wolfberry have been reported for monitoring meat and seafood spoilage [6-10]. In addition, bioactive composite films incorporating extracts and essential oils have also been explored for multifunctional intelligent packaging [11,12]. These findings highlight the versatility of natural polymer matrices and anthocyanins in developing intelligent films with practical applications in food quality monitoring [13].
Among natural polymers, glucomannan derived from porang tubers (Amorphophallus muelleri) has attracted attention due to its distinctive physicochemical properties. As a soluble dietary fiber, glucomannan exhibits high viscosity, strong water-holding capacity, and excellent gelling ability [14-16]. It significantly contributes to the mechanical properties of biopolymer films, allowing for the development of biodegradable packaging that maintains food quality while reducing environmental impact [17]. In addition, glucomannan has been reported to exhibit antibacterial activity [18], an important functional property in extending shrimp shelf life.
Gelatin, a protein-based polymer, is another material widely applied in edible films due to its excellent film-forming capacity, transparency, and compatibility with polysaccharides [19,20]. Blending gelatin with polysaccharides such as glucomannan improves the structural and barrier properties of the resulting films and enables the incorporation of natural additives to enhance antimicrobial or antioxidant activity [21]. Therefore, the synergy of glucomannan and gelatin offers an effective biopolymer matrix for the fabrication of intelligent films.
Anthocyanins, widely distributed in fruits and flowers, are natural pigments known for their antioxidant properties and halochromic behavior under different pH conditions. Rambutan peel (Nephelium lappaceum), an abundant agro-industrial by-product in Indonesia, is particularly rich in anthocyanins, primarily cyanidin-3-glucoside and delphinidin-3-glucoside. When incorporated into film matrices, these pigments can act as visual pH indicators, shifting color during spoilage processes and thereby providing consumers with a simple tool to assess food safety [8,22]. Optimizing extraction and stabilization methods, such as co-pigmentation with gallic acid (GA), has further enhanced anthocyanin yield and color stability [23].
Despite extensive research on anthocyanin-based intelligent films, most studies have focused on pigments extracted from hibiscus, butterfly pea, dragon fruit, or other botanical sources [5,9,24], while limited studies have examined RPAE (Rambutan Peels Anthocyanin Extract) in combination with glucomannan–gelatin matrices. This represents a critical research gap, given the availability of rambutan peel waste in tropical regions and its potential for value-added applications. Furthermore, comprehensive evaluations of the physicochemical, mechanical, and functional properties of glucomannan-gelatin films enriched with RPAE, particularly for shrimp freshness monitoring, remain scarce.
During shrimp storage, microbial spoilage leads to the degradation of nitrogenous compounds and the accumulation of total volatile base nitrogen (TVB-N), which elevates pH and deteriorates sensory quality. These biochemical changes can be effectively detected through colorimetric shifts in anthocyanin-based intelligent films, as the pigment structure transforms with increasing pH. The relationship between microbial activity, TVB-N formation, and pH variation, therefore, provides a reliable scientific basis for freshness assessment in shrimp and other seafood products. Despite extensive research on anthocyanin-based intelligent films, most studies have focused on pigments extracted from hibiscus, butterfly pea, dragon fruit, and other botanical sources [6,9,25]. However, limited studies have examined the potential of RPAE incorporated into glucomannan–gelatin matrices. This represents a critical research gap, considering the abundant availability of rambutan peel waste in tropical regions and its potential for value-added applications. Furthermore, comprehensive evaluations of the physicochemical, mechanical, and functional properties of glucomannan-gelatin films enriched with RPAE, particularly their ability to monitor shrimp freshness through pH-driven color responses, remain scarce.
Therefore, this study aims to develop and characterize intelligent films based on porang (Amorphophallus oncophyllus) glucomannan and gelatin incorporated with copigmented RPAE for monitoring shrimp freshness. The integration of these natural components is expected to enhance film functionality, mechanical integrity, and color stability, enabling real-time freshness monitoring of shrimp through pH-dependent color transitions. This approach highlights the novelty of utilizing locally abundant rambutan peel waste and porang-derived biopolymers to produce sustainable, biodegradable, and intelligent food packaging materials.
Materials and methods
Material
Ripe red rambutan peel of the Binjai variety obtained in Bantul, Yogyakarta, porang glucomannan (85% glucomannan, Ikarie Organic, Bantul, Indonesia), fish gelatin (Redman, Singapore), glycerol, demineralized water, ethanol, methanol, distilled water, and Whatman No. 1 paper. All chemicals are analytical grade (Merck, Germany).
Extraction of anthocyanin from rambutan peels
The anthocyanin extraction process for rambutan peels follows the modified method [23]. The rambutan peels were washed with running water, then blanched in hot water at 50 °C for 2.5 min to inactivate enzymes that can degrade the color. Next, the rambutan peels were dried at 50 °C for 24 h using a cabinet dryer (LocknLock EJO316, LocknLock Co., Ltd., Seoul, South Korea) to a moisture content of 6%, ground into powder, and sieved using a 40-mesh sieve. The resulting rambutan peel powder was stored in aluminum foil pouches with silica gel and stored in a freezer at –20 °C.
The rambutan peel powder was then extracted using 96% ethanol containing 0.2% citric acid at a 1:20 ratio using the Ultrasound-Assisted Extraction (UAE) method (UP200St, Hielscher Ultrasonics GmbH, Teltow, Germany). Extraction was performed using a 7 mm diameter probe at 200 W power and 20 kHz frequency, with the temperature maintained at 50 °C for 20 min using a Frigiterm-TFT-10 water bath thermostat (J.P. Selecta S.A., Barcelona, Spain). After extraction, the extracted solution was centrifuged at 2,600×g for 15 min using a centrifuge (DLAB DM0636, DLAB Scientific Co., Ltd., Beijing, China) to obtain a supernatant containing anthocyanin extract. It was then filtered using Whatman No. 1 filter paper. The resulting extract was then concentrated using a rotary evaporator (IKA RV, IKA®-Werke GmbH & Co. KG, Staufen, Germany) and stored at −18 °C for analysis and copigmentation process (Figure 1).

Figure 1 Flowchart of Research.
Copigmentation of anthocyanin
The copigmentation steps for RPAE were carried out based on a modified method [23]. The molarity of the RPAE was first calculated to determine the concentration required for the copigmentation process. Copigmentation was carried out using a molarity ratio of 1:300 (RPAE: GA) and a volume ratio of 1:1. The GA solution was prepared with a molarity 300 times the molarity of the anthocyanin extract. The copigmentation process was carried out by mixing 10 mL of the anthocyanin extract and 10 mL of the GA solution, then stirring using a magnetic stirrer (Thermo Scientific Cimarec™ SP88857107, Thermo Fisher Scientific Inc., Waltham, MA, USA) at 750 rpm for 20 min. After stirring, the mixture was allowed to stand for 1 hour to allow the copigmentation process to occur. The resulting 20 mL of RPAE copigmented with GA was obtained.
Production of intelligent film
Intelligent packaging film was made by preparing 2 separate solutions. The 1 %(w/v) glucomannan solution was prepared by dissolving porang glucomannan in distilled water using a magnetic stirrer at 50 °C and 1,500 rpm for 3 h (A solution). The 2 %(w/v) gelatin solution was prepared by dissolving fish gelatin in distilled water using a magnetic stirrer at 50 °C and 1,500 rpm for 3 h (B solution).
The 2 solutions (A and B) were then mixed in a 4:1 ratio (80 mL of glucomannan solution and 20 mL of gelatin solution) to obtain a film solution volume of 100 mL using a magnetic stirrer at 1,500 rpm and 50 °C for 5 min. Next, 0.7 mL of glycerol was added and stirred for 5 min. Non-copigmented anthocyanin extract (A) and copigmented anthocyanin extract (K) were then added in different concentrations based on Table 1.
Table 1 Formulation of intelligent film.
No |
Sampel Code |
Material |
||||
Glucomanan 1 %w/v |
Gelatin 2 %w/v |
Glycerol |
Non Copigmented Anthocyanin 74.31 mg/L |
Copigmented Anthocyanin 74.31 mg/L |
||
1 |
C |
80 mL |
20 mL |
0.7 mL |
- |
- |
2 |
A1 |
80 mL |
20 mL |
0.7 mL |
1 mL |
- |
3 |
A2 |
80 mL |
20 mL |
0.7 mL |
2 mL |
- |
4 |
A3 |
80 mL |
20 mL |
0.7 mL |
3 mL |
- |
5 |
K1 |
80 mL |
20 mL |
0.7 mL |
- |
1 mL |
6 |
K2 |
80 mL |
20 mL |
0.7 mL |
- |
2 mL |
7 |
K3 |
80 mL |
20 mL |
0.7 mL |
- |
3 mL |
C = control film, A = film added with non copigmented anthocyanin, K = film added with copigmented anthocyanin.
Evaluation of TAC in rambutan peels
Total anthocyanin Content (TAC) analysis was performed using the pH differential method [23,26]. RPAE was diluted 25-fold with methanol PA. A 1 mL solution was taken and added to 4 mL of pH 1 buffer to obtain a total volume of 5 mL. The same procedure was repeated using a pH 4.5 buffer and homogenized using a vortex (Thermo Scientific MaxiMix™ II, Thermo Fisher Scientific Inc., Waltham, MA, USA). Measurements were made using a spectrophotometer (Thermo Scientific Genesys 10S UV-Vis, Germany). Absorbance readings were recorded at 520 and 700 nm, using distilled water as a calibration blank. The difference in absorbance between the pH values and wavelengths was calculated using the formula:
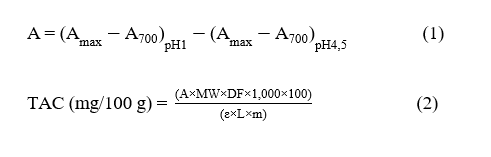
Characteristic evaluation of intelligent films
Thickness
Film thickness was measured using a digital micrometer (Mitutoyo 293-340-30, Mitutoyo Corporation, Kawasaki, Japan), which has an accuracy of 0.001 mm. Measurements were taken at 5 points, and the average value was taken [13].
Initial color of film
Initial color of film analysis was conducted using a chromameter (CR-400, Konica Minolta, Japan) equipped with a 0.8 cm aperture, and calibrated with a white plate (Y = 86.5, x = 0.3168, y = 0.3245). Intelligent packaging color parameters were L* (lightness), a* (red-green), and b* (yellow-blue) [23].
Tensile strength (TS) and elongation at break (EAB)
TS and EAB were tested using a Universal Testing Machine (UTM ZWICK/ZO, Germany). Film samples were prepared by cutting them into 10 × 10 cm sections with a 0.5 cm width in the center. The samples were secured in the UTM clamp and stretched by the machine. The maximum force (Fmax) recorded during the test was used to calculate the TS (MPa) and EAB (%) [27].
Water vapor permeability (WVP)
WVP of the films was determined using the desiccant cup method as described in ASTM E96-95, with modifications [27]. Briefly, silica gel was placed inside a glass cup to maintain 0% relative humidity (RH), and the film sample was sealed tightly over the mouth of the cup to avoid edge leakage. The assembly was then placed in a sealed desiccator containing saturated NaCl solution, which generates a constant atmosphere of approximately 75% RH at 25 °C. The cups were weighed at regular intervals using an analytical balance (OHAUS PR224, OHAUS Corporation, Parsippany, NJ, USA), and the increase in mass over time was plotted to obtain the water vapor transmission rate (WVTR, H20 g·m⁻2·h⁻1). WVP (H20 g·Pa⁻1·h⁻1·m⁻1) was calculated according to the equation.

where L is the average film thickness (m) and ΔP is the water vapor pressure difference across the film (Pa). All measurements were carried out in triplicate, and results were expressed as mean ± standard deviation.
RSA
DPPH assay
The DPPH assay was used to determine the radical scavenging activity (RSA) of the films, which represents their antioxidant capacity. Briefly, 1 mL of the film extract was mixed with 3 mL of 0.1 mM DPPH (2,2-diphenyl-1-picrylhydrazyl) solution prepared in methanol. The mixture was incubated in the dark at 25 °C for 30 min to allow the reaction to proceed, after which the absorbance was recorded at 517 nm using a spectrophotometer [23].
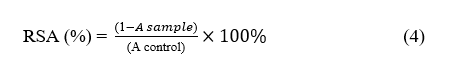
ABTS (2,2′-Azino-bis-(3-ethylbenzothiazoline-6-sulfonic acid) assay
A 7 mM ABTS solution was prepared by mixing it with 2.45 mM potassium persulfate (K₂S₂O₈) in a 1:1 ratio and allowing the reaction to proceed for 16 h at 25 °C in the dark [28]. The resulting solution was diluted with ethanol until an absorbance of approximately 0.70 at 734 nm was obtained, which was designated as A₀. Film samples (about 50 mg) were then immersed in 10 mL of the ABTS solution and incubated at 25 °C in the absence of light for 30 min. After incubation, the absorbance of the solution was measured at 734 nm (Aₜ), and the antioxidant activity of the films was determined using the following equation:
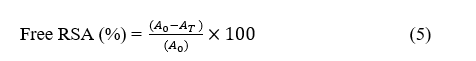
where A₀ and AT represent the absorbance values of the control and the film sample, respectively.
Color response to pH
The color response of the intelligent films to pH was evaluated by immersing film samples (2×2 cm2) in buffer solutions with pH values ranging from 1 to 14. After equilibration, the color of the films was measured using a chromameter (CR-400, Konica Minolta, Japan) equipped with a 0.8 cm aperture and calibrated against a standard white plate (Y = 86.5, x = 0.3168, y = 0.3245). The color parameters L* (lightness), a* (red-green), and b* (yellow-blue) were recorded, and the total color difference (ΔE) was calculated using the following equation [23].
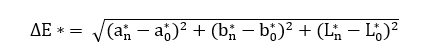
where
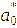 ,
,
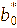 ,
and
,
and
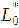 corresponded to the initial color parameters of the anthocyanin
extract without co-pigmentation and
corresponded to the initial color parameters of the anthocyanin
extract without co-pigmentation and
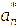 ,
,
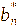 ,
and
,
and
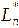 represented the color parameters of the anthocyanin extract after
co-pigmentation.
represented the color parameters of the anthocyanin extract after
co-pigmentation.
Intelligent film morphology
The film surface was observed using a JSM-6510LA Scanning Electron Microscope (SEM) with magnifications ranging from 10 to 300,000× and a resolution of 1 - 10 nm. Surface micrographs were recorded at magnifications of 100×, 500×, 1,000×, and 3,000× to ensure clear images.
Intelligent film functional groups
Functional groups were analyzed using a Fourier Transform Infra-Red (FTIR) (Perkin Elmer Spectrum Two System L160000A) equipped with an attenuated total reflectance (ATR) accessory. FTIR spectra were recorded in the range of 400 to 4,000 cm−1 with a resolution of 2 cm−1.
Intelligent packaging application on shrimp
The films were cut into 1.5×1.5 cm2 strips and affixed to the inner surface of the container lid to function as indicator sensors for black tiger shrimp (Penaeus monodon) packaging. Approximately 85 g of shrimp was placed into a 200 mL container. Color changes (∆E) of the films, along with the pH, TVB-N, and TVC of the shrimp, were monitored visually over a storage period of 3 days.
Color response of intelligent packaging during shrimp storage
The color of the films was measured using a chromameter (CR-400, Konica Minolta, Japan) equipped with a 0.8 cm aperture and calibrated against a standard white plate (Y = 86.5, x = 0.3168, y = 0.3245) [23].
pH
Samples (10.0 g) were weighed, ground, and suspended in 90 mL of distilled water in a beaker. The mixtures were homogenized and centrifuged at 500 rpm for 1 min at 25 °C. The resulting supernatants were collected and used for pH determination with an electronic pH meter (Mettler Toledo FiveEasy™ F20, Mettler Toledo GmbH, Greifensee, Switzerland) [2].
TVB-N
TVB-N content of shrimp samples was determined according to [29] for fishery products, which includes extraction, distillation, and titration steps. Approximately 10 g of homogenized shrimp flesh was extracted with 90 mL of 6% perchloric acid and filtered through Whatman filter paper. A 50 mL aliquot of the filtrate was placed into a distillation flask, mixed with 10 mL of 20% NaOH and a few drops of silicone antifoaming agent, and distilled for 5 - 10 min. The released volatile bases were collected in 100 mL of 3% boric acid solution containing Tashiro indicator, producing a greenish coloration, and the distillate was titrated with 0.01 N HCl to a reddish-purple endpoint. TVB-N values were expressed as mg N per 100 g of shrimp flesh.
TVC
The microbial quality of shrimp samples was analyzed according to the method of Ran et al. [30] with slight modifications. Approximately 10 g of shrimp flesh was homogenized and mixed with 90 mL of sterile physiological saline (0.85 g NaCl/L), followed by thorough shaking. Serial dilutions were prepared, and 1 mL aliquots from appropriate dilutions were inoculated onto Plate Count Agar (PCA) using the pour-plate method. The plates were incubated at 37 °C for 48 h, after which colony-forming units (CFU) were counted, and the total viable counts (TVC) were expressed as log CFU/g of shrimp.
Data analysis
All experimental measurements were conducted in triplicate to ensure reproducibility and reliability of the data. The obtained results were organized and tabulated using Microsoft Excel 2016 (Microsoft Corp., Redmond, WA, USA). Statistical analyses were carried out with IBM SPSS Statistics, Version 27.0 (IBM Corp., Armonk, NY, USA). A one-way analysis of variance (ANOVA) was employed to evaluate the significance of treatment effects, and when significant differences were detected (p < 0.05), Duncan’s Multiple Range Test (DMRT) was applied as a post-hoc comparison. The results are expressed as mean ± standard deviation (SD).
Results and discussion
Thickness of edible film
The thickness of intelligent films made from gelatin and glucomannan that are enriched with anthocyanins varies depending on the specific formulation and the method of preparation used. The results showed that intelligent films based on glucomannan and gelatin enriched with RPAE exhibited an increase in film thickness (Table 2). The increase in thickness was attributed to the higher amount of dissolved solids during the film-forming process. Intelligent films developed from gelatin incorporated with anthocyanins exhibited significant thickness variations based on the ratios of the components used [3]. Their study showed that the microstructure and properties of the films were influenced by the incorporation of curcumin and anthocyanins, which suggests variations in thickness as a function of formulation [3]. Additionally, variations in biopolymer or anthocyanin content, including those from polysaccharides like glucomannan, can affect physical dimensions and integrity of the films [4].
Table 2 Physical and mechanical characteristics of intelligent film.
80 |
Sample |
Thickness (mm) |
Color |
TS (MPa) |
EAB (%) |
WVP (g·m−1·Pa−1·h−1) |
||
L |
a |
b |
||||||
1 |
C |
0.12 ± 0.01b |
83.42 ± 0.60a |
1.59 ± 0.01f |
−1.89 ± 0.01f |
5.70 ± 0.65a |
21.80 ± 2.60a |
3.10×10−7 ± 6.34×10−9 d |
2 |
A1 |
0.15 ± 0.02ab |
64.85 ± 0.18b |
1.95 ± 0.09e |
20.18 ± 0.21c |
7.24 ± 1.40a |
21.76 ± 3.65a |
3.82×10−7 ± 1.32×10−9 bc |
3 |
A2 |
0.15 ± 0.02ab |
63.10 ± 0.35c |
2.40 ± 0.14d |
18.63 ± 0.48d |
7.07 ± 1.75a |
24.34 ± 1.38a |
4.09×10−7 ± 4.03×10−9 b |
4 |
A3 |
0.17 ± 0.01a |
54.56 ± 0.56f |
4.52 ± 0.17b |
17.12 ± 0.40e |
8.21 ± 1.59e |
31.90 ± 1.08a |
4.72×10-7 ± 3.70×10−9 a |
5 |
K1 |
0.14 ± 0.01ab |
60.35 ± 0.42d |
1.98 ± 0.06e |
17.57 ± 0.40e |
8.68 ± 1.72e |
25.61 ± 6.72a |
3.94×10−7 ± 2.54×10−9 bc |
6 |
K2 |
0.14 ± 0.01ab |
58.10 ± 0.45e |
3.12 ± 0.14c |
23.56 ± 0.28b |
9.26 ± 1.20b |
26.99 ± 2.56a |
3.56×10−7 ± 1.77×10−9 c |
7 |
K3 |
0.14 ± 0.01ab |
49.41 ± 0.74g |
6.09 ± 0.14a |
24.50 ± 0.31a |
9.37 ± 2.54a |
28.67 ± 7.71a |
3.10×10−7 ± 9.47×10−9 d |
a-d the same letter in the same column is not significantly different (p > 0.05). C = control film, A = film added with non copigmented anthocyanin, K = film added with copigmented anthocyanin.
The addition of anthocyanins impacted the physical properties of gelatin-based films, such as density and thickness, confirming that formulations could lead to considerable variations depending on the interactions with gelatin [22]. Furthermore, the incorporation of anthocyanins from specific plant sources enhanced the structural integrity of the films and suggested that these interactions could lead to thicker and more robust films [6]. In a broader context, various natural extracts, including anthocyanins, can influence the thickness and functional properties of gelatin films [7]. Although the exact thickness can differ, it is generally observed that the inclusion of anthocyanins contributes to the formation of thicker, more functional films. In conclusion, intelligent films made from gelatin and enriched with anthocyanins, along with glucomannan, can exhibit significant thickness variations, with specific studies suggesting values ranging from approximately 0.12 mm to over 0.17 μm depending on the specific formulation and additives used.
Color of edible film
The initial color of the intelligent film is a key parameter for evaluating its ability to indicate food freshness. This study, Table 2, showed a significant decrease in L (lightness) values (p < 0.005) with increasing concentrations of both native RPAE and GA-copigmented RPAE, ranging from 49.41 to 83.42. In contrast, the a (redness) and b* (yellowness) values of the intelligent films significantly increased (p < 0.005) with anthocyanin addition, with the highest a* and b* values observed in K3 films, 6.09 and 24.50 ± 0.31, respectively. These findings indicate that higher anthocyanin concentrations result in films that are darker, redder, and more yellowish.
Different anthocyanin sources yield distinct initial film colors depending on their molecular structures. The carrier matrix and the pH of the film-forming solution also play crucial roles in determining color appearance. At acidic pH, anthocyanins predominantly exist in the red flavylium cation form. Upon deprotonation, the flavylium cation converts into the purple quinoidal base, which can further deprotonate to form the anionic quinoidal base, exhibiting blue to green hues. In aqueous environments, the quinoidal base may hydrate to form the colorless carbinol pseudobase [23]. In the present study, the anthocyanin-based films exhibited a noticeable greenish-yellow coloration at pH 5 - 6, suggesting that quinoidal forms were dominant under these slightly acidic conditions. This color behavior reflects the intrinsic pH sensitivity of anthocyanins extracted from rambutan peel, which undergo structural transformations in response to environmental pH. The transformation of anthocyanins under varying pH conditions is a critical aspect of their functionality as colorimetric indicators. Particularly within the pH range of 5 to 6, quinoidal forms dominate, leading to notable spectral and ∆E. At lower pH levels, anthocyanins exist predominantly as flavylium cations, exhibiting vibrant red hues. As the pH increases, these compounds undergo structural transformations into quinoidal bases, then carbinol pseudobases, and finally chalcones, which display less color saturation and varied color profiles, ranging from purple to blue [31]. The quinoidal form is especially significant because it represents a stable intermediate that contributes to the characteristic color response of anthocyanins. This pH-dependent behavior highlights the molecular basis of anthocyanin sensitivity, enabling their application in intelligent packaging systems designed for freshness monitoring [32,33]. Such materials leverage the colorimetric potential of anthocyanins to visually indicate shifts in pH that correlate with food spoilage processes, thus offering a non-invasive means of assessing food quality [33]. Studies have demonstrated that the structural integrity and environmental conditions can significantly influence the stability and colorimetric properties of anthocyanins, solidifying their role as effective bioindicators in smart food packaging technologies [34,35].
Initial film colors depend on anthocyanin sources and matrix interactions. RPAE incorporated into pectin-chitosan films produced red films [23]. Fish gelatin films with butterfly pea flower anthocyanins showed a blue color, which shifted to green-yellow upon the incorporation of chitosan carbon dots [24]. Meanwhile, chitin-based intelligent films containing black wolfberry anthocyanins displayed a purple hue with sodium alginate, but appeared yellowish-brown when combined with gelatin [8]. In summary, the initial color of intelligent films is strongly influenced by the type of anthocyanin, film matrix, and pH conditions, which together determine their suitability and effectiveness as freshness indicators in intelligent packaging applications.
TS and EAB
The mechanical properties of intelligent films made from gelatin and glucomannan, particularly when enriched with anthocyanins, are critical for their performance in food packaging applications. Studies indicate that the TS and EAB of these films can be significantly influenced by the incorporation of anthocyanins. The results, Table 2, showed that the incorporation of both native and copigmented anthocyanins into the intelligent films significantly (p < 0.005) increased their TS, ranging from 5.70 to 9.37 MPa, while the EAB showed no significant differences, with values ranging from 21.80% to 28.67%.
For instance, the incorporation of anthocyanins into gelatin-based films can affect their TS, which may fluctuate based on the anthocyanin concentration used. Specifically, higher concentrations of anthocyanins were noted to reduce TS while enhancing EAB due to improved interfacial interactions within the film, which can increase flexibility [10]. In another study, it was found that films incorporated with anthocyanin extracts had a TS of approximately 12.91 MPa after adding specific proportions of plant extracts, showcasing varied mechanical strength depending on the formulation [4]. Moreover, the incorporation of anthocyanins into gelatin films led to an increase in the EAB from 91.19% to 107.86%, suggesting that this modification significantly enhances the practicality of the films by improving flexibility and resilience under tension [22]. The increase in EAB indicates that the films can withstand stretching without breaking, which is advantageous for handling during packaging processes. Additionally, adding anthocyanin extracts may reduce TS in gelatin films formulated with other agents like glucomannan. This may indicate that the interactions between the natural polymer network and the anthocyanins could enhance flexibility while compromising ultimate TS [9]. The observed values for TS and EAB are strongly dependent on the type and amount of additional components used in the film formulation.
To summarize, films synthesized from gelatin and glucomannan enriched with anthocyanins can exhibit variable TS, often measured around 9.37 MPa, with EAB values that can surpass 28.67%. Such mechanical properties can be markedly improved through careful formulation and selection of synergistic components, which can play a pivotal role in their application as intelligent packaging materials.
WVP
WVP is a critical parameter in intelligent films, as it determines their ability to regulate vapor transfer, including TVB-N, which directly affects the film’s sensitivity in monitoring food freshness. In this study, the WVP values ranged from 3.10×10−7 to 4.72×10−7 H2O g·Pa⁻1·h⁻1 (Table 2). The incorporation of both native and copigmented RPAE led to an increase in WVP, which can be ascribed to the hydrophilic nature of anthocyanins and GA. The presence of GA within the anthocyanin structure is known to enhance the affinity of the polymer matrix toward water molecules, thereby facilitating their diffusion through the film network.
These findings are consistent with previous research where the addition of grape seed extract into poly(butylene adipate-co-butylene terephthalate)/poly (lactic acid) films increased WVP, likely due to the hydrophilic nature of glycerol in the extract [11]. In contrast, other studies demonstrated opposite trends. For example, gelatin-based films incorporated with chitosan carbon dots and butterfly pea flower anthocyanins exhibited a significant reduction in WVP, with the decrease becoming more pronounced at higher anthocyanin concentrations [24]. This reduction was attributed to the uniform dispersion of carbon dots and enhanced interfacial interactions between the polymer matrices, resulting in a denser network structure that restricted water permeation. Similarly, carrageenan-PVA films enriched with anthocyanins and thyme oil showed a decrease in WVP, suggesting that the combined effect of phenolic compounds and essential oils contributed to a more compact microstructure and reduced water transport [12].
Taken together, these results highlight that the influence of anthocyanins on WVP is strongly dependent on the type of polymer matrix and the presence of other functional additives, which can either enhance hydrophilicity and water diffusion or reinforce structural compactness, thereby lowering permeability.
pH sensitivity of intelligent film
Protein-based foods, such as shrimp, typically undergo pH elevation during spoilage (Figure 2). Therefore, the color response of intelligent films to varying pH conditions is a critical parameter for freshness monitoring. In this study, the L* (lightness) values of intelligent films incorporated with both native and copigmented anthocyanins decreased progressively with increasing pH. The a* (redness) value decreased sharply at pH 2, followed by a gradual increase up to pH 14. Conversely, the b* (yellowness) value consistently increased with rising pH. These changes can be attributed to the structural transformation of RPAE. The total color difference (ΔE) was highest at pH 1, decreased until pH 4, and then increased again. This behavior suggests that the films are most stable around pH 5 - 6, as the observed color closely resembled the initial state, while more pronounced ∆E were evident under highly acidic and highly alkaline conditions. At acidic pH, anthocyanins predominantly exist in the red flavylium cation form. Upon deprotonation, the flavylium cation converts to the purple quinoidal base, which can further deprotonate into an anionic quinoidal base, yielding blue to green hues. In aqueous environments, hydration of the quinoidal base leads to the colorless carbinol pseudobase [23]. As the pH increases, these compounds undergo structural transformations into quinoidal bases, then carbinol pseudobases, and finally chalcones, which display less color saturation and varied color profiles, ranging from purple to blue [31]. The quinoidal form is especially significant because it represents a stable intermediate that contributes to the characteristic color response of anthocyanins. For instance, bilayer intelligent films based on konjac glucomannan, alginate, and alizarin displayed a color transition from yellowish to purple as pH increased [36]. Similarly, chitosan/gelatin films enriched with butterfly pea anthocyanins exhibited distinct transitions from pink to purple, blue, and green across the pH range of 2 - 12 [24]. Gelatin films incorporated with purple cabbage anthocyanins also demonstrated pH-dependent changes from pink to purple, blue, and green [33]. Moreover, starch–polyvinyl alcohol-chitosan films enriched with roselle anthocyanins displayed progressive color shifts under increasing alkaline nitrogen conditions, ranging from red to purple, green, and eventually yellow [37]. These variations highlight that the color transition profiles are strongly influenced by both the biopolymer matrix and the anthocyanin source. RPAE are predominantly composed of cyanidin-3-glucoside and delphinidin-3-glucoside [23], which likely explains the characteristic greenish-yellow hues observed in this study. In conclusion, the distinctive pH-dependent color response of RPAE underscores its potential as a reliable natural indicator for intelligent food packaging, particularly for monitoring the freshness and spoilage of protein-rich products.
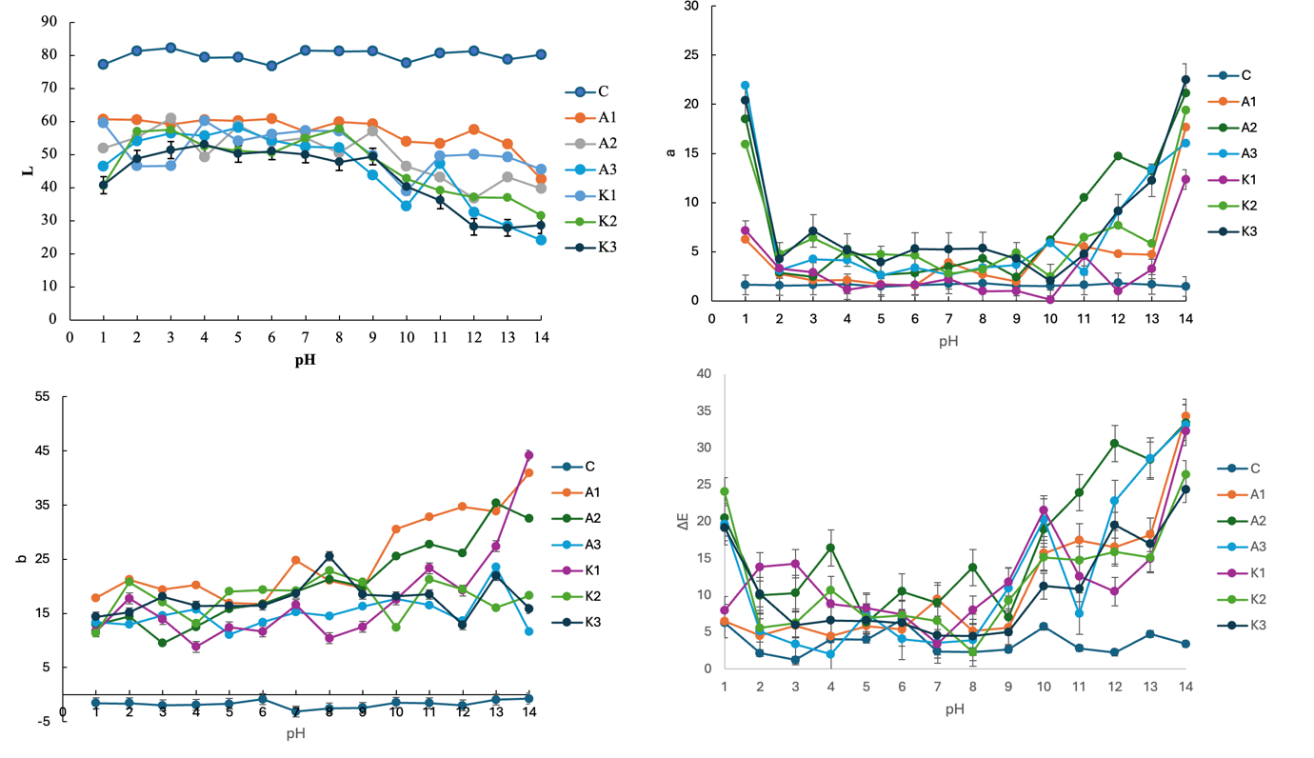
Figure 2 pH sensitivity of intelligent film. C = control film, A = film added with non copigmented anthocyanin, K = film added with copigmented anthocyanin.
RSA
The intelligent films developed in this study also exhibited potential antioxidant activity due to the incorporation of RPAE. Antioxidant activity was evaluated through RSA using DPPH and ABTS assays, as presented in Table 3. The results showed that films containing copigmented RPAE exhibited higher RSA values in both DPPH and ABTS assays compared to those containing native anthocyanins. The DPPH assay is based on the reduction of the stable purple DPPH radical to a yellow-colored product by hydrogen-donating antioxidants, while the ABTS assay measures the ability of antioxidants to quench the blue-green ABTS⁺ radical cation [23]. These results confirm that copigmentation enhances the antioxidant activity of anthocyanins in intelligent films.
Table 3 Antioxidant Activity of intelligent film.
No |
Sample |
Antioxidant Activity |
|
DPPH |
ABTS |
||
1 |
C |
4.35 ± 0.82e |
4.88 ± 0.94f |
2 |
A1 |
21.89 ± 1.82d |
13.41 ± 1.52e |
3 |
A2 |
59.89 ± 1.21c |
27.47 ± 0.77d |
4 |
A3 |
92.46 ± 0.70ab |
73.65 ± 1.13b |
5 |
K1 |
90.94 ± 0.41b |
64.42 ± 1.62a |
6 |
K2 |
93.41 ± 0.27ab |
77.64 ± 1.06b |
7 |
K3 |
94.71 ± 0.16a |
93.01 ± 2.35c |
a-f the same letter in the same column is not significantly different (p > 0.05)
C = control film, A = film added with non copigmented anthocyanin, K = film added with copigmented anthocyanin.
Several studies have demonstrated that the incorporation of anthocyanins into biopolymer films enhances their antioxidant potential. For instance, loading Clitoria ternatea anthocyanin-rich extract into sodium alginate films increased TPC and TAC, resulting in higher ABTS and DPPH activities [38]. Similarly, red cabbage anthocyanin extract significantly improved the DPPH scavenging activity of modified starch-based films [39], while black rice anthocyanins in pectin-chitosan films exhibited stronger antioxidant capacity than ascorbic acid. In line with these findings, RPAE incorporated into pectin-chitosan films markedly enhanced DPPH RSA (95.54%), attributed to their high phenolic content (3,439.63 mg/100 g) [23].
Free radicals are atoms or molecules containing unpaired electrons, typically generated through the homolytic cleavage of covalent bonds under external factors such as light or heat. In food systems, their concentration increases over time, leading to weakened cellular defense mechanisms and the formation of reactive compounds that accelerate microbial contamination and oxidative degradation [35]. Copigmented anthocyanins exhibit strong antioxidant activity, enabling them to scavenge free radicals and thereby inhibit spoilage. Consequently, intelligent films incorporating copigmented RPAE offer an effective strategy for extending food preservation.
Intelligent film morphology
The morphology of intelligent films formed from gelatin and glucomannan, particularly those enriched with anthocyanins, can be effectively characterized using Scanning SEM. The SEM analysis provides insights into the structural attributes and surface characteristics of these films, which are critical for their application in food packaging. As shown in Figure 3, the incorporation of copigmented RPAE into glucomannan-gelatin films (K3) improved the surface morphology by producing a smoother and more homogeneous structure, while also reducing cracks observed in the cross-section. This indicates improved polymer compatibility and structural integrity, likely resulting from hydrogen bonding and electrostatic interactions among glucomannan, gelatin, and anthocyanins. The hydroxyl and phenolic groups of glucomannan and anthocyanins can form intermolecular hydrogen bonds with the amine and carbonyl groups of gelatin, leading to a more compact and homogeneous microstructure with fewer voids. Such a dense and uniform structure is generally correlated with higher TS and lower Water Vapor Permeability (WVP), as reported in recent studies on hydrogen-bonded biopolymer blends [3]. These molecular interactions collectively contribute to the development of high-performance intelligent films with enhanced mechanical and barrier properties. These findings are consistent with previous studies, where anthocyanin incorporation similarly influenced the morphology and performance of biopolymer-based films.
For intelligent films that incorporate anthocyanins, the morphology of gelatin-based films becomes more heterogeneous, displaying variations in surface roughness and texture due to the interactions between anthocyanins and the gelatin matrix [10]. The SEM images presented in their research revealed an increase in surface irregularities and a less uniform distribution compared to control films, indicating that anthocyanin inclusion alters interface characteristics, potentially enhancing the functional properties of the films. Moreover, pH-responsive gelatin films indicated a noticeable variation in microstructure when anthocyanin and diatomite were added. The SEM analysis revealed that the addition of these compounds resulted in a roughened surface and influenced the mechanical properties of the films, highlighting the composite nature and its impact on functionality, such as gas barrier and mechanical strength [40]. Films from fish gelatin and Coleus scutellarioides Anthocyanins also supported these findings, noting that their SEM analyses showed a more compact and dense morphology in films containing anthocyanins compared to those without. This suggests enhanced compatibility between film components, which can improve the performance of the film as a barrier against moisture and gases, vital for food preservation [7]. The compatibility and morphological features of anthocyanin-containing films were favorable, as indicated by the smooth and bubble-free cross-sections visible through SEM analysis. This uniformity is indicative of successful interactions between gelatin, glucomannan, and anthocyanins, leading to better mechanical integrity and barrier performance in food packaging applications [41]. Overall, SEM analysis of gelatin and glucomannan films enriched with anthocyanins reveals significant influences on their morphology, enhancing their structural integrity and functionality, which are crucial for their applications in intelligent food packaging.
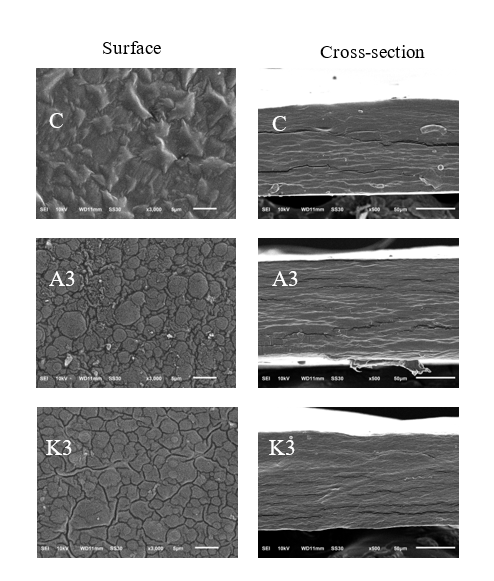
Figure 3 Morphology of intelligent packaging to pH. C = control film, A = film added with non copigmented anthocyanin, K = film added with copigmented anthocyanin.
Intelligent film functional group
FTIR spectroscopy provides essential insights into the functional groups present in intelligent films made from gelatin and glucomannan, especially when these films are enriched with anthocyanins. Through FTIR analysis, various characteristic peaks are identified, which correlate with specific functional groups responsible for the physical and chemical properties of these films.
As shown in Figure 4, the FTIR spectra of the films with native and copigmented anthocyanins exhibited peak shifts toward lower wavenumbers. The broad band associated with O–H and N–H stretching vibrations shifted from 3,287.21 to 3,285.48 cm⁻1, indicating the formation of hydrogen bonding interactions between anthocyanins and the glucomannan-gelatin matrix. Similarly, the peak at 1,645.18 cm⁻1, corresponding to C=O stretching (amide I region), shifted to 1,642.51 cm⁻1, suggesting possible interactions between the carbonyl groups of the protein backbone and anthocyanins. In addition, the shift from 1,032.54 to 1,031.66 cm⁻1 in the C–O stretching region further confirmed the involvement of hydroxyl groups of anthocyanins in hydrogen bonding. These peak shifts collectively demonstrate that the incorporation of anthocyanins enhances molecular interactions within the polymer network, thereby improving film stability and compatibility. FTIR analysis of intelligent films embedded with anthocyanins revealed significant peaks corresponding to amide functional groups. They found that the spectra exhibited peaks for Amide A, observed in the range of 3,377.07 - 3,281.73 cm⁻1 (related to N-H stretching vibrations) and Amide I at 1,625.25 - 1,632.40 cm⁻1 (C=O stretching vibrations) [23]. Furthermore, Amide II was noted at 1,539.17 - 1,552.67 cm⁻1, while Amide III appeared in the range of 1,235.17 - 1,239.65 cm⁻1, illustrating the presence of these integral structures within the gelatin matrix.
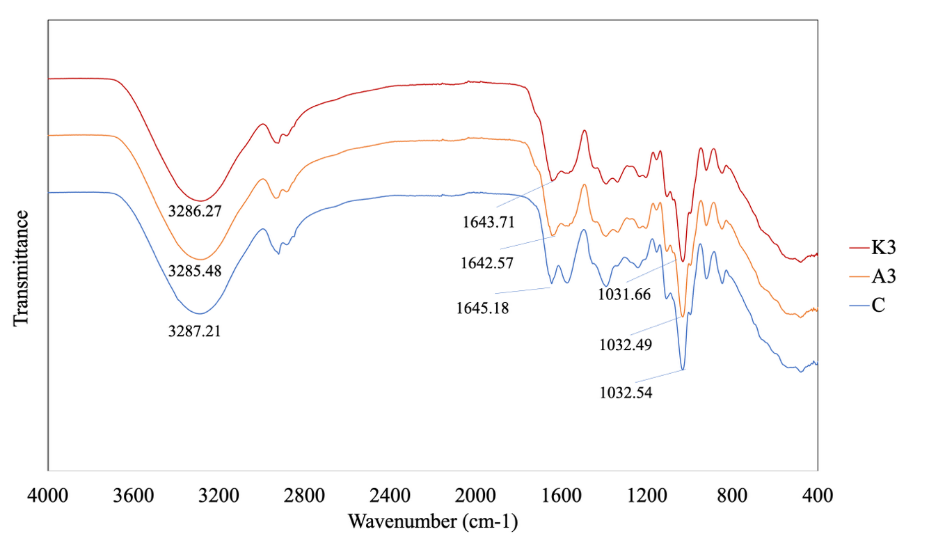
Figure 4 FTIR of intelligent packaging to pH. C = control film, A = film added with non copigmented anthocyanin, K = film added with copigmented anthocyanin.
Moreover, the absorption bands around 3,500 - 3,200 cm⁻1 indicated O-H stretching vibrations, which are crucial in the context of anthocyanins [32]. The addition of anthocyanins influenced the positioning and intensity of these peaks, highlighting interactions between the anthocyanins and the gelatin matrix, likely through hydrogen bonding mechanisms. It observed similar characterizations, noting that such interactions are significant for the morphological stability and barrier properties of the films [7].
Additionally, the incorporation of anthocyanins into polymeric systems such as polyvinyl alcohol (PVA) films showed characteristic peaks associated with hydroxyl groups, with a broad peak around 3,365 cm⁻1, confirming their presence within the film matrices [42]. These findings indicate a strong correlation between the molecular structure of anthocyanins and the resultant physical properties of the films. The spectral data around 2,930 - 2,934 cm⁻1 was observed. Reflected C–H stretching and –NH₃⁺ stretches, showing that gelatin maintains its characteristic bonding structures even when mixed with anthocyanins. This retention is critical for preserving the mechanical properties necessary for packaging applications [43]. In summary, FTIR analysis of intelligent films from gelatin and glucomannan enriched with anthocyanins reveals the presence of important functional groups such as amides (Amide A, B, I, II, III) and hydroxyls, confirming that these components interact significantly with the film’s matrix. This interaction is essential for improving the overall properties of these films, making them suitable for intelligent packaging applications.
Intelligent film application on shrimp
During refrigerated storage, shrimp quality deterioration was reflected by concordant changes in chemical and microbiological indices (Table 4). TVB-N rose from 28.11 ± 1.15 to 71.03 ± 1.79 mg N/100 g by 72 h, exceeding common freshness limits (30 - 35 mg N/100 g), while total TVC increased from 5.24 to 6.27 log CFU/g, approaching the 6 log CFU/g spoilage threshold. The pH showed a slight fluctuation (7.75 ± 0.02 → 7.43 ± 0.02), consistent with proteolysis-driven amine accumulation and buffering effects late in spoilage [44,45]. This modest pH decrease corresponds to an early post-mortem acidification phase, in which glycolysis and lactic acid accumulation lower muscle pH before a gradual rise occurs due to microbial deamination and volatile base formation [46,47]. To facilitate practical interpretation, film colors were mapped to freshness categories based on concurrent TVB-N/TVC values: Yellow = fresh (TVB-N ≤ 30 - 35 mg N/100 g; TVC < 6.0 log CFU/g), yellow-green = borderline (TVB-N 35 - 60 mg N/100 g or TVC approaching 6.0 log CFU/g), and green = spoiled (TVB-N > 60 mg N/100 g and/or TVC ≥ 6.0 log CFU/g).
Table 4 TVB-N, TVC, and pH shrimp during storage.
No |
Time (h) |
TVB-N (mg/100g) |
TVC (log CFU/g) |
pH |
1 |
0 |
28.11 ± 1.15 |
5.24 |
7.75 ± 0.02 |
2 |
24 |
38.82 ± 0.85 |
5.49 |
7.74 ± 0.03 |
3 |
48 |
46.79 ± 1.09 |
5.91 |
7.58 ± 0.03 |
4 |
72 |
71.03 ± 1.79 |
6.27 |
7.43 ± 0.02 |
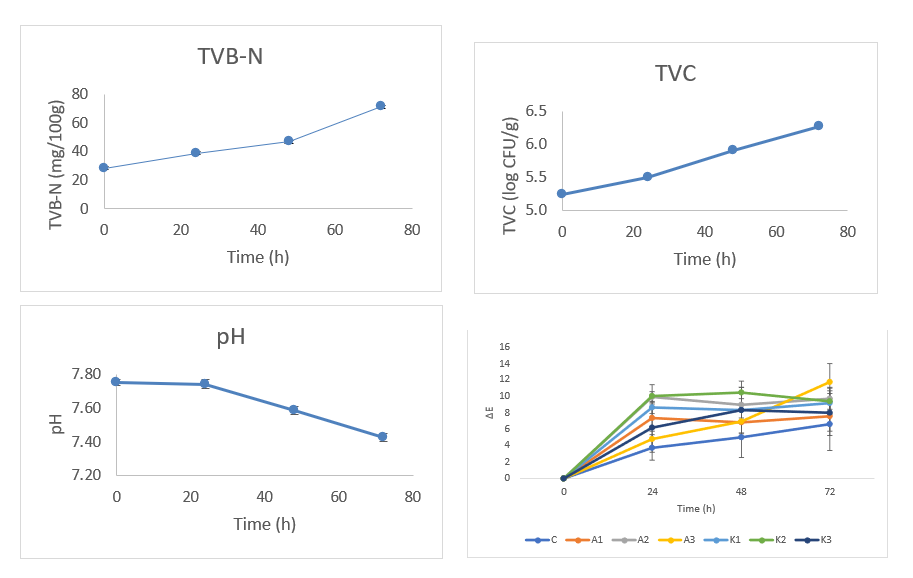
Figure 5 TVB-N, TVC, pH shrimp, and ΔE film.
Accordingly, the glucomannan-gelatin films loaded with RPAE exhibited a yellow-to-green transition over time with ΔE > 5, reflecting the pH-dependent structural interconversion of anthocyanins from flavylium to quinoidal and ultimately to chalcone within the biopolymer matrix (Figure 5). This pH-dependent behavior highlights the molecular basis of anthocyanin sensitivity, enabling their application in intelligent packaging systems designed for freshness monitoring [32,33]. Such materials leverage the colorimetric potential of anthocyanins to visually indicate shifts in pH that correlate with food spoilage processes, thus offering a non-invasive means of assessing food quality [33]. Studies have demonstrated that the structural integrity and environmental conditions can significantly influence the stability and colorimetric properties of anthocyanins, solidifying their role as effective bioindicators in smart food packaging technologies [34,35]. Similar abediindicator behavior color shifts that track increases in TVB-N/TVC has been reported for chitosan and gelatin-based anthocyanin films, where hydrogen-bonding interactions help sharpen the sensing signal [33,48-50]. Collectively, these parallel changes in TVB-N, TVC, and ΔE demonstrate that the developed films provide a reliable, non-invasive indicator of shrimp freshness suitable for intelligent packaging applications.

Figure 6 Film color during application. C = control film, A = film added with non copigmented anthocyanin, K = film added with copigmented anthocyanin.
Conclusions
This study demonstrates that intelligent films fabricated from porang-derived glucomannan and gelatin and enriched with RPAE, particularly in the copigmented form, offer a robust, sustainable platform for seafood freshness monitoring. The films showed improved functional performance, including TS up to ~9.37 MPa, EAB up to ~31.90%, and WVP in the range of ~3.10 - 4.72×10⁻7 g·m⁻1·Pa⁻1·h⁻1, along with enhanced RSA (DPPH/ABTS). FTIR analysis indicated hydrogen-bonding interactions among gelatin, glucomannan, and anthocyanins, while SEM revealed smoother, more homogeneous morphologies for copigmented systems, supporting better structural integrity. Critically, during shrimp storage, the films provided clear visual readouts (ΔE > 5) that tracked quality deterioration, aligning with increases in TVB-N and microbial load, thus enabling a non-invasive, real-time indication of spoilage (Figure 5). Leveraging locally abundant biopolymers and colorants, these films advance biodegradable, intelligent packaging with practical relevance to seafood safety and quality. Future work should validate performance across diverse cold-chain conditions, develop quantitative models linking ΔE with TVB-N/TVC for predictive decision thresholds, strengthen long-term color stability (e.g., optimized co-pigmentation/encapsulation), and assess scalability and regulatory aspects for industrial translation.
Acknowledgements
The authors wish to thank Kemendiktisaintek, Indonesia, for supporting the project under the PFR 2025 (Penelitian Fundamental Research 2025) contract number: 067/C3/DT.05.00/PL/2025; 2459/ UN1/DITLIT/Dit-Lit/PT.01.03/2025.
CRediT author statement
Ida Bagus Gede Mahatmananda: Formal Analysis, Investigation, Validation, Original Writing; Arum Widyastuti Perdani: Formal Analysis, Investigation, Validation, Methodology, Writing; Bambang Purwono: Conceptualization, Resources, Data Curation, Visualization; Writing - Reviewing and Editing, Supervision; Supriyadi: Conceptualization, Resources, Data Curation, Visualization; Writing - Reviewing and Editing, Supervision, Project Administration, Funding Acquisition; Andriati Ningrum: Conceptualization, Resources, Data Curation, Methodology Visualization; Writing - Reviewing and Editing, Supervision, Project Administration, Funding Acquisition.
References
[1] SF Hosseini and MC Gómez-Guillén. A state-of-the-art review on the elaboration of fish gelatin as bioactive packaging: Special emphasis on nanotechnology-based approaches. Trends in Food Science & Technology 2018; 79, 125-135.
[2] P Ezati and JW Rhim. Starch- and agar-based color-indicator films integrated with shikonin for smart packaging application of shrimp. ACS Food Science & Technology 2021; 1, 1963-1969.
[3] S Zhou, N Li, H Peng, X Yang and D Lin. The development of highly pH-sensitive bacterial cellulose nanofibers/gelatin-based intelligent films loaded with anthocyanin/curcumin for the fresh-keeping and freshness detection of fresh pork. Foods 2023; 12(20), 3719.
[4] N Guo, M Song, W Liu, F Zhang and G Zhu. Preparation of an elderberry anthocyanin film and fresh-keeping effect of its application on fresh shrimps. PLoS One 2023; 18(11), e0290650.
[5] NA Azlim, AM Nafchi, N Oladzadabbasabadi, F Ariffin, P Ghalambor, S Jafarzadeh and AA Al-Hassan. Fabrication and characterization of a pH-sensitive intelligent film incorporating dragon fruit skin extract. Food Science & Nutrition 2022; 10(2), 597-608.
[6] NF Latiff, MI Shaik, NJ Mohamad, WM Khairul, AI Daud, B Nurhadi and NM Sarbon. Recent advances in green halochromic smart film packaging: Investigating the natural colorant as biosensor in smart film packaging for real-time meat freshness monitoring. Food Control 2025; 172, 111157.
[7] F Hematian, H Baghaei, AM Nafchi and M Bolandi. Preparation and characterization of an intelligent film based on fish gelatin and Coleus scutellarioides anthocyanin to monitor the freshness of rainbow trout fish fillet. Food Science & Nutrition 2023; 11(1), 379-389.
[8] Y Zheng, X Li, Y Huang, H Li, L Chen and X Liu. Two colorimetric films based on chitin whiskers and sodium alginate/gelatin incorporated with anthocyanins for monitoring food freshness. Food Hydrocolloids 2022; 127, 107517.
[9] R Pramitasari, LN Gunawicahya and DSB Anugrah. Development of an indicator film based on cassava starch-chitosan incorporated with red dragon fruit peel anthocyanin extract. Polymers 2022; 14, 4142.
[10] NF Latiff, NF Sulaiman, MI Shaik, NJ Mohamad, WM Khairul, AI Daud and NM Sarbon. Halochromic smart film: a gelatin-based pH-sensitive film embedded with anthocyanin from roselle (Hibiscus sabdariffa) extracts for potential food spoilage indicator application. Journal of Food Science 2025; 90(3), e70134.
[11] X He, L Tang, C Li, J Hao and R Huang. Eco-friendly poly(butylene adipate-co-butylene terephthalate)/poly(lactic acid) packaging films with grape seed extract for extending the shelf life of strawberries and broccoli. LWT 2025; 227, 117990.
[12] O Dardari, MA Benzaouia, A El Idrissi, BE Channab, GR Benjellound, A El Gharrak, M El Ouardi and A El Kadib. Anthocyanin and thyme oil embedded carrageenan - PVA towards bioactive and pH-sensitive food-packaging materials. Journal of Biological Macromolecules 2025; 310, 143213.
[13] LT Wu, IL Tsai, YC Ho, YH Hang, C Lin, ML Tsai and FL Mi. Active and intelligent gellan gum-based packaging films for controlling anthocyanins release and monitoring food freshness. Carbohydrate Polymers 2021; 254, 117410.
[14] A Koga, F Tanaka, S Hagiwara, LP Wigati and F Tanaka. Functional properties of gelatin/konjac glucomannan-based coatings with Aomori hiba essential oil and their effect on ethylene production in peach fruit (Prunus persica (L.) Batsch cv. ‘Hikawa Hakuho’) during cold storage. Environmental Control in Biology 2024; 62, 17-28.
[15] SMB Hashemi and D Jafarpour. The efficacy of edible film from konjac glucomannan and saffron petal extract to improve shelf life of fresh-cut cucumber. Food Science & Nutrition 2020; 8(7), 3128-3137.
[16] HC Lai, AA Karim and CC Seow. Effects of water-glycerol and water-sorbitol interactions on the physical properties of konjac glucomannan films. Journal of Food Science 2006; 71(2), E62-E67.
[17] A Dirpan, AF Ainani and M Djalal. A review on biopolymer-based biodegradable film for food packaging: trends over the last decade and future research. Polymers 2023; 15(13), 2781.
[18] E Harmayani, NF Geraldo and A Murdiati. Physicochemical, nutritional and sensory properties of kluklui supplemented with porang glucomannan and banana flour. Indonesian Food and Nutrition Progress 2022; 18(1), 9-15.
[19] AS Sekarina, Supriyadi, HSH Munawaroh, E Susanto, PL Show and A Ningrum. Effects of edible coatings of chitosan–fish skin gelatine containing black tea extract on quality of minimally processed papaya during refrigerated storage. Carbohydrate Polymer Technologies and Applications 2023; 5, 100287.
[20] F Azizah, H Nursakti, A Ningrum and Supriyadi. Development of edible composite film from fish gelatin–pectin incorporated with lemongrass essential oil and its application in chicken meat. Polymers 2023; 15(9), 2075.
[21] PAV Freitas, RRA Siiva, TV de Oiiveira, RRA Soares and NFF Soares. Biodegradable film development by nisin Z addition into hydroxypropylmethylcellulose matrix for mozzarella cheese preservation. International Journal of Food Studies 2020; 9(2), 360-372.
[22] NA Azlim, AM Nafchi, N Oladzadabbasabadi, F Ariffin, P Ghalambor, S Jafarzadeh and AA Al-Hassan. Fabrication and characterization of a pH-sensitive intelligent film incorporating dragon fruit skin extract. Food Science & Nutrition 2021; 10, 597-608.
[23] AW Perdani, AD Setiowati, B Purwono and S Supriyadi. Color enhancement of rambutan peel anthocyanins extracts using co-pigmentation with gallic acid for pH-sensitives dye. Trends in Sciences 2025; 22, 9518.
[24] A Ponnusamy, A Khan, T Prodpran, JW Rhim and S Benjakul. Multifunctional fish gelatin film incorporated with chitosan carbon dots and butterfly pea flower anthocyanins for active/smart packaging of Pacific white shrimp. Food Bioscience 2024; 62, 105483.
[25] M Cissé, P Bohuon, F Sambe, C Kane, M Sakho and M Dornier. Aqueous extraction of anthocyanins from Hibiscus sabdariffa: Experimental kinetics and modeling. Journal of Food Engineering 2012; 109(1), 16-21.
[26] A Manosroi, W Ruksiriwanich, B Kietthanakorn, W Manosroi and J Manosroi. Relationship between biological activities and bioactive compounds in the fermented rice sap. Food Research International 2011; 44(9), 2757-2765.
[27] A Ningrum, AW Perdani, Supriyadi, H Siti Halimatul Munawaroh and S Aisyah. Characterization of tuna skin gelatin edible films with various plasticizers-essential oils and their effect on beef appearance. Journal of Food Processing and Preservation 2021; 45, 15701.
[28] MR Teruel, MD Garrido, MC Espinosa and MB Linares. Effect of different format-solvent rosemary extracts (Rosmarinus officinalis) on frozen chicken nuggets quality. Food Chemistry 2015; 172, 40-46.
[29] National Standardization Agency of Indonesia. Chemical testing method - Part 8: determination of total volatile base nitrogen (TVB-N) and trimethylamine nitrogen (TMA-N) in fishery products. National Standardization Agency of Indonesia, Jakarta, Indonesia, 2009.
[30] R Ran, T Zheng, P Tang, Y Xiong, C Yang, M Gu and G Li. Antioxidant and antimicrobial collagen films incorporating Pickering emulsions of cinnamon essential oil for pork preservation. Food Chemistry 2023; 420, 136108.
[31] D Verma, N Sharma and U Malhotra. Structural chemistry and stability of anthocyanins. The Pharma Innovation 2023; 12, 1366-1373.
[32] M Tavassoli, MA Sani, A Khezerlou, A Ehsani, GJ Khaniki and DJ McClements. Smart biopolymer-based nanocomposite materials containing pH-sensing colorimetric indicators for food freshness monitoring. Molecules 2022; 27, 3168.
[33] R Abedi-Firoozjah, S Yousefi, M Heydari, F Seyedfatehi, S Jafarzadeh, R Mohammadi, M Rouhi and F Garavand. Application of red cabbage anthocyanins as pH-sensitive pigments in smart food packaging and sensors. Polymers 2022; 14, 1629.
[34] S Duarte, GA Valencia and O Ferreiro. Biodegradable colorimetric indicative films based on kurugua (Sicana odorifera) peel powder. Polymers 2025; 17, 1167.
[35] HE Khoo, A Azlan, ST Tang and SM Lim. Anthocyanidins and anthocyanins: Colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food & Nutrition Research 2017; 61, 1361779.
[36] S Zhou, J Li, D Lin, X Feng, R Zhang, D Wang, A Zhao, H Tian and X Yang. Development of konjac glucomannan-based active–intelligent emulsion films loaded with different curcumin-metal chelates: stability, antioxidant, fresh-keeping and freshness detection properties. International Journal of Biological Macromolecules 2024; 282, 137231.
[37] J Zhang, X Zou, X Zhai, XW Huang, C Jiang and M Holmes. Preparation of an intelligent pH film based on biodegradable polymers and roselle anthocyanins for monitoring pork freshness. Food Chemistry 2019; 272, 306-312.
[38] E da Nóbrega Santos, TC de Albuquerque Sousa, DC de Santana Neto, CV Brandão Grisi, VC da Silva Ferreira and FA Pereira da Silva. Edible active film based on gelatin and Malpighia emarginata waste extract to inhibit lipid and protein oxidation in beef patties. LWT 2022; 154, 112837.
[39] OJ Esua, DW Sun, JH Cheng, H Wang and M Lv. Functional and bioactive properties of Larimichthys polyactis protein hydrolysates as influenced by plasma functionalized water-ultrasound hybrid treatments and enzyme types. Ultrasonics Sonochemistry 2022; 86, 106023.
[40] K Wu, H Wang, Z Zhang, H Cai, K Qian, W Ma, M Fan and L Li. pH-responsive gelatin/diatomite/rose anthocyanin composite films by melt extrusion: physicochemical properties and application in monitoring freshness of Litopenaeus vannamei. Food Science & Nutrition 2025; 38, 739-751.
[41] T Aowpitaya and R Sasanatayart. Properties of gelatin-based films incorporated with anthocyanins and curcuminoids and stability of antioxidant activity during in vitro digestion. Current Applied Science and Technology 2024; 24, e0259441.
[42] Y Qi and Y Li. Colorimetric films based on polyvinyl alcohol and anthocyanins extracted from purple tomato to monitor the freshness of shrimp. Polymers 2024; 16, 495.
[43] M Pouralkhas, M Kordjazi, SM Ojagh and OA Farsani. Physicochemical and functional characterization of gelatin edible film incorporated with fucoidan isolated from Sargassum tenerrimum. Food Science & Nutrition 2023; 11, 4124-4135.
[44] MZ Çoban and ÖE Çoban. Potency and use of chia mucilage coating containing propolis liquid extract for improves shelf-life of sea bass fillets. Acta Scientiarum Polonorum Technologia Alimentaria 2020; 19, 255-260.
[45] N Wannawisan, K Pathanasriwong, J Runglerdkriangkrai, P Wilaipun, A Sane and P Suppakul. Microbiological, chemical and physical attributes and mathematical models for total volatile basic nitrogen formation of Asian seabass (Lates calcarifer) fillets stored under refrigerated and temperature-abuse conditions. Agriculture and Natural Resources 2023; 57(3), 511-522.
[46] E Jaffrès, V Lalanne, S Macé, J Cornet, M Cardinal, T Sérot, X Dousset and J Joffraud. Sensory characteristics of spoilage and volatile compounds associated with bacteria isolated from cooked and peeled tropical shrimps using SPME-GC-MS analysis. International Journal of Food Microbiology 2011; 147(3), 195-202.
[47] S Yang, J Xie and Y Qian. Determination of spoilage microbiota of Pacific white shrimp during ambient and cold storage using next-generation sequencing and culture-dependent method. Journal of Food Science 2017; 82, 1178-1183.
[48] Y He, Y Yuan, Y Gao, M Chen, Y Li, Y Zou, L Liao, X Li, Z Wang, J Li and W Zhou. Enhancement of colorimetric pH-sensitive film incorporating Amomum tsao-ko essential oil as antibacterial for mantis shrimp spoilage tracking and fresh-keeping. Foods 2024; 13, 1638.
[49] D Li, Y Zhao, P Han, C Yang, X Liang, L Li and S Cai. Effect of chitosan-jicama starch coating on changes in qualities of fresh Nile tilapia (Oreochromis niloticus) fillets during ice storage. International Journal of Food Science and Technology 2018; 53(9), 2220-2228.
[50] M Aitboulahsen, H Chairi, A Laglaoui, S Zantar, A Arakrak, M Bakkali and MH Zerrouk. Gelatin/pectin-based film incorporated with essential oils: Functional characteristics and shelf life extension of tilapia fillets under refrigeration. Journal of Food Safety 2020; 40(3), e12774.