
Trends
Sci.
2026;
23(5):
11956
Claudin-4 Expression as a Prognostic Biomarker in Colorectal Adenocarcinoma: Correlation with Lymphovascular Invasion, Lymph Node Metastasis, and Pathological Stage
Ririn
Endah Puspitasari1,
![]() ,
Upik Anderiani Miskad1,2,*,
Muhammad Husni Cangara1,2,
,
Upik Anderiani Miskad1,2,*,
Muhammad Husni Cangara1,2,
![]() ,
Syarifuddin Wahid1,2,
Djumadi Achmad1,2,
Amalia Yamin1,3,
,
Syarifuddin Wahid1,2,
Djumadi Achmad1,2,
Amalia Yamin1,3,
![]() and Suryani Tawali4,
and Suryani Tawali4,
![]()
1Department of Anatomical Pathology, Faculty of Medicine, Hasanuddin University, Makassar, Indonesia
2Anatomical Pathology Laboratory, Hasanuddin University Hospital, Makassar, Indonesia
3Anatomical Pathology Laboratory, Dr. Wahidin Sudirohusodo General Hospital, Makassar, Indonesia
4Department of Public Health, Faculty of Medicine, Hasanuddin University, Makassar, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 18 September 2025, Revised: 28 October 2025, Accepted: 10 November 2025, Published: 5 January 2026
Abstract
Colorectal cancer (CRC) remains a leading cause of cancer mortality worldwide, with increasing incidence in Asia, including Indonesia. Claudin-4, a tight junction protein, plays a paradoxical role in epithelial barrier function and tumor progression. This study aimed to investigate the relationship between Claudin-4 expression and major prognostic parameters in colorectal adenocarcinoma. A total of 104 formalin-fixed paraffin-embedded tissue blocks of colorectal adenocarcinoma were analyzed immunohistochemically. Claudin-4 expression was categorized as strong (79.8%) or weak (20.2%) based on the Total Immunostaining Score (TIS). Statistical analysis revealed no significant association between Claudin-4 expression and age, sex, tumor location, or size. However, Claudin-4 expression was significantly negatively correlated with lymphovascular invasion (p = 0.020; OR = 0.314, 95% CI 0.114 - 0.861), lymph node metastasis (p = 0.011; OR = 0.283, 95% CI 0.103 - 0.778), and pathological stage (p = 0.028). No significant correlation was observed with distant metastasis. These findings suggest that Claudin-4 may serve as a prognostic biomarker reflecting tumor invasiveness, supporting its potential role in guiding risk stratification in CRC patients.
Keywords: Colorectal adenocarcinoma, Claudin-4, Lymphovascular invasion, Lymph node metastasis, Prognostic biomarker, Pathological stage
Introduction
Colorectal cancer (CRC) remains one of the leading causes of cancer-related death worldwide. According to GLOBOCAN 2022, CRC ranks third in incidence, representing 10% of all new cases, and second in mortality, accounting for approximately 9.3% of all cancer deaths [1]. In Indonesia, CRC is among the 5 most common malignancies and continues to rank among the top 5 causes of cancer mortality [2]. Its risk is shaped by complex interactions between genetic predisposition, environmental exposures, and lifestyle factors [2]. While surgery and radiotherapy remain the mainstay for early-stage CRC, advanced disease often requires systemic chemotherapy [3]. Colorectal adenocarcinoma (CRA) is the predominant histological subtype [4,5]. Its prognosis, however, is highly dependent on histopathological and molecular tumor characteristics [6]. Established prognostic indicators include histopathological grade, tumor budding, tumor infiltration (T), lymphovascular invasion (LVSI), perineural invasion, and lymph node metastasis (LNM) [6,7]. Among these, LVSI and LNM remain among the strongest predictors of metastatic potential and poor outcome [2].
Claudin-4 is a transmembrane protein and an essential component of tight junctions (TJs) that safeguard epithelial barrier integrity [8]. Claudin proteins, including Claudin-4, are expressed at different sites in epithelial cells, the apical membrane, basolateral membrane, and even the nucleus, each location reflecting distinct biological functions. At the apical membrane, Claudin-4 forms part of the TJ complex together with occludin, which connects to intracellular actin filaments to maintain intercellular adhesion [9]. At the basolateral membrane, Claudin-4 contributes to the activation of pro-MMP into MMP and interacts with integrins in focal adhesion complexes, thereby influencing critical signaling pathways such as MAPK [9].
Beyond its structural role, Claudin-4 also exerts multiple non-junctional functions that have attracted increasing attention in cancer biology [8]. Non-TJ Claudin-4 has been implicated in enhancing stemness, promoting anti-apoptotic effects, conferring drug resistance, and facilitating metastatic capacity in cancer cells [8,10,11]. Furthermore, Claudin-4 plays a role in epithelial mesenchymal transition (EMT) and metastatic colonization [8,9,11]. Interestingly, Claudin-4 demonstrates a paradoxical behavior in tumorigenesis. On the one hand, Claudin-4-mediated TJs maintain a tumor microenvironment enriched with growth factors such as VEGF and IL-8, which promotes angiogenesis and tumor progression [8,11-13]. On the other hand, dysregulation or downregulation of Claudin-4 may facilitate the loss of cell cohesion and enhance tumor cell motility, accelerating invasion and metastasis [12].
Although Claudin-4 has been widely studied in other malignancies, its prognostic significance in colorectal adenocarcinoma remains controversial and insufficiently characterized. Some studies link high Claudin-4 expression with tumor aggressiveness and EMT, whereas others report that decreased expression correlates with invasive features and advanced disease [8,11]. Importantly, data from Southeast Asia, particularly Indonesia, are still scarce, despite the unique population characteristics and increasing burden of CRC in the region [14].
Based on this paradoxical role, we hypothesized that Claudin-4 expression is closely related to tumor invasiveness in colorectal adenocarcinoma. Specifically, we expected strong Claudin-4 expression to be more common in early-stage tumors without LVSI or LNM, whereas reduced expression would correlate with more aggressive features, including LVSI, LNM, and advanced pathological stage.
This study was designed to investigate whether Claudin-4 can serve as a prognostic biomarker reflecting tumor invasiveness and regional metastasis in colorectal adenocarcinoma, or whether its role is more closely tied to tumorigenesis itself. By addressing this knowledge gap, particularly in the Indonesian population, we aim to provide evidence that contributes to a better understanding of CRC biology and to the development of more accurate risk stratification strategies for patient management. To the best of our knowledge, this is the first study from Indonesia to comprehensively evaluate Claudin-4 expression in colorectal adenocarcinoma and its correlation with key prognostic parameters, including lymphovascular invasion, lymph node metastasis, and pathological stage. In doing so, the study not only provides population-specific evidence but also contributes to the global understanding of the paradoxical role of Claudin-4 in tumor biology, highlighting its potential as an additional prognostic biomarker to support risk stratification and guide more tailored clinical decision-making in colorectal cancer.
Materials and methods
Study design and samples
This cross-sectional study was conducted between October 2024 and January 2025 at the Anatomic Pathology Laboratory, Faculty of Medicine, Hasanuddin University, Makassar, Indonesia. A total of 104 paraffin-embedded tissue specimens from patients diagnosed with colorectal adenocarcinoma were analyzed. These specimens were obtained from the archives of the Anatomic Pathology Laboratory, Dr. Wahidin Sudirohusodo General Hospital, and Hasanuddin University Hospital, collected between January 2022 and December 2024. All included cases had a histologically confirmed diagnosis of colorectal adenocarcinoma and complete clinicopathological data that fulfilled the study’s inclusion criteria.
Immunohistochemistry (IHC) and evaluation of claudin-4 expression
Claudin-4 expression was evaluated by immunohistochemistry (IHC) using a rabbit polyclonal anti-Claudin-4 antibody (E-AB-15673, Elabscience) at a dilution of 1:400. Sections were prepared from paraffin blocks at a thickness of 3 µm, deparaffinized, and subjected to IHC staining. Claudin-4 reactivity was observed in the membrane and/or cytoplasm of tumor cells under an Olympus CX-43 light microscope at 400× magnification. To minimize bias, all slides were independently evaluated by 2 blinded pathologists who were unaware of the patients’ clinical data. Claudin-4 expression was scored semiquantitatively using the Total Immunostaining Score (TIS), derived by multiplying the intensity and proportion scores, yielding a scale of 1 to 9. Intensity was graded as: 0 (negative, no staining), 1 (weak), 2 (moderate), and 3 (strong). The proportion of positive tumor cells was scored as: 0 (< 5%), 1 (5% - 25%), 2 (26% - 50%), and 3 (≥ 51%). Claudin-4 expression was categorized as weak when TIS < 3 and strong when TIS ≥ 3.
Assessment of clinicopathological characteristics
Clinicopathological data, including patient age, sex, tumor location (proximal, distal, rectum, rectosigmoid), tumor size, histopathological grade (low-grade and high-grade), tumor infiltration (T), lymphovascular invasion (LVSI), lymph node metastasis (LNM), N status (regional lymph node involvement), distant metastasis (M status), and pathological stage, were obtained from medical records. Histopathological grade, LVSI, LNM, and tumor infiltration were determined according to standard pathological criteria.
Statistical analysis
Statistical analyses were performed using SPSS version 30. Descriptive statistics were used to summarize the basic characteristics of the samples. Bivariate analyses were conducted using the Chi-square test, Mann-Whitney test, and Fisher’s exact test to evaluate the association between Claudin-4 expression (strong vs. weak) and categorical clinicopathological variables, including LVSI, LNM, histopathological grade, tumor location, tumor size, distant metastasis, pathological stage, age, and sex. A p-value ≤ 0.05 was considered statistically significant.
Ethical approval
This study was reviewed and approved by the Research Ethics Committee of the Faculty of Medicine, Hasanuddin University, Makassar, Indonesia (Approval No: Protocol # UH24A9AV23 - Registry 905/UN4.6.4.5.31/ PP36/ 2A24). All procedures were conducted in accordance with the ethical standards of the institutional and national research committees, as well as the Declaration of Helsinki.
Results and discussion
A total of 104 cases of colorectal adenocarcinoma were analyzed in this study. The clinical and pathological characteristics of the cohort are well described, encompassing a broad range of ages, sex distribution, tumor locations, sizes, grading, tumor infiltration, lymphovascular invasion, nodal involvement, metastatic status, and pathological stage (Table 1). This comprehensive sampling strengthens the generalizability of the findings and allows for robust subgroup analysis, as emphasized by previous epidemiological studies. By age distribution, the majority of patients were older than 50 years (73.1%), while those aged ≤ 50 years accounted for 26.9% of cases. In terms of sex, male patients were slightly more frequent (53.8%) than female patients (46.2%). Tumor location was most commonly in the rectum (34.6%), followed by the distal colon (30.8%), the proximal colon (28.8%), and least frequently in the rectosigmoid region (5.8%). Regarding tumor size, most cases measured 3 - 5 cm (56.7%), followed by tumors larger than 5 cm (32.7%), while tumors smaller than 3 cm were less common (10.6%). Histopathologically, low-grade tumors predominated (62.5%), whereas high-grade tumors were identified in 37.5% of cases. For tumor infiltration (T), most patients fell into categories T2 (39.4%) and T3 (41.3%), while only 19.2% were classified as T1. With respect to lymphovascular invasion (LVSI), more than half of the cases were negative (55.8%), while 44.2% were positive.
Table 1 Characteristics of colorectal adenocarcinoma samples examined.
Characteristics |
n (%) |
Age |
|
≤ 50 |
28 (26.9) |
> 50 |
76 (73.1) |
Sex |
|
Male |
56 (53.8) |
Female |
48 (46.2) |
Tumor Location |
|
Proximal |
30 (28.8) |
Distal |
32 (30.8) |
Rectum |
36 (34.6) |
Rectosigmoid |
6 (5.8) |
Tumor Size (cm) |
|
< 3 |
11 (10.6) |
3 - 5 |
59 (56.7) |
> 5 |
34 (32.7) |
Histopathological Grade |
|
Low |
65 (62.5) |
High |
39 (37.5) |
Tumor Infiltration (T) |
|
T1 (limited to the submucosa) |
20 (19.2) |
T2 (through muscularis propria) |
41 (39.4) |
T3 (through visceral peritoneum or other organs) |
43 (41.3) |
Lymphovascular Invasion |
|
Negative |
58 (55.8) |
Positive |
46 (44.2) |
Lymph Nodes Metastasis |
|
Negative |
60 (57.7) |
Positive |
44 (42.3) |
N status (Metastases to Regional Lymph Node) |
|
N0 (no regional lymph node metastases) |
60 (57.7) |
N1a (metastases in 1 regional lymph nodes) |
20 (19.2) |
N1b (metastases in 2 - 3 regional lymph nodes) |
15 (14.4) |
N2a (metastases in 4 - 6 regional lymph nodes) |
8 (7.7) |
N2b (metastases in > 6 regional lymph nodes) |
1 (1.0) |
Distant Organ Metastasis |
|
Negative |
81 (77.9) |
Positive |
23 (22.1) |
Liver Metastasis |
19 (18.3) |
Lung Metastasis |
2 (1.9) |
Bone Metastasis |
1 (1.0) |
Liver, Lung, and Bone Metastasis |
1 (1.0) |
M status |
|
M0 (No distant metastasis) |
81 (77.9) |
M1a (Metastasis to a single organ) |
21 (20.2) |
M1b (Metastasis to ≥ 2 organs or sites) |
2 (1.9) |
Pathological Stage |
|
Stage I |
34 (32.7) |
Stage IIA |
19 (18.3) |
Stage IIB |
1 (1.0) |
Stage IIIA |
12 (11.5) |
Stage IIIB |
19 (18.3) |
Stage IVA |
18 (17.3) |
Stage IVB |
1 (1.0) |
Claudin-4 Expression |
|
Strong |
83 (79.8) |
Weak |
21 (20.2) |
Lymph node metastasis was observed in 42.3% of cases. The distribution of nodal status showed N0 in 57.7%, N1a in 19.2%, N1b in 14.4%, N2a in 7.7%, and N2b in 1.0% of cases. For distant metastasis (M status), most patients were classified as M0 (77.9%), whereas M1a (20.2%) and M1b (1.9%) were found in a smaller proportion. The liver was the most frequent site of distant spread (18.3%), followed by the lungs (1.9%) and bones (1.0%). When analyzed by pathological stage, most cases were diagnosed at stage I (32.7%), followed by stage IIA (18.3%), stage IIIB (18.3%), stage IIIA (11.5%), stage IVA (17.3%), and stage IVB (1.0%). With respect to Claudin-4 expression, the majority of tumors showed strong expression (79.8%), while weak expression was observed in 20.2% of cases.
This study demonstrated that most patients with colorectal adenocarcinoma were older than 50 years. This finding is consistent with epidemiological studies reporting that colorectal cancer (CRC) occurs more frequently in individuals aged 50 years and above [15]. The main risk factors for CRC at older ages include overweight and obesity, smoking, heavy alcohol consumption, high intake of red or processed meat, and low levels of physical activity [14].
In this cohort, male patients predominated (53.8%), which aligns with previous reports indicating that men are at higher risk of developing CRC compared to women. This disparity has been linked to hormonal differences, alcohol intake, and smoking habits [16]. Men are considered more vulnerable to CRC due to lifestyle patterns that increase exposure to these risk factors, while in women, estrogen appears to play a protective role against CRC development [15,17]. This protective effect is not limited to endogenous estrogen but is also observed with oral contraceptive use and postmenopausal hormone replacement therapy, both of which have been associated with a reduced risk of CRC [18]. Mechanistically, estrogen is believed to enhance survival in women through regulation of genes involved in ion transport and epithelial-mesenchymal transition via crosstalk with the Wnt/β-catenin pathway. Furthermore, estrogen modulates proliferative responses of CRC cells under hypoxic conditions through G-protein-coupled estrogen receptor (GPER), HIF1A, and VEGF signaling pathways [17,19].
The predominance of tumors in the rectum (34.6%) is also in agreement with previous studies reporting higher proportions of rectal and distal colon cancers in Asian populations, a pattern thought to be associated with diets rich in animal fat and low in fiber [14,20]. Most tumors measured 3 - 5 cm at the time of diagnosis, suggesting that patients often presented with relatively large tumors, likely due to delayed detection [21].
From a histopathological perspective, low-grade tumors accounted for the majority of cases (62.5%), which is consistent with the general pattern of colorectal adenocarcinoma, where most tumors are still well- to moderately differentiated. However, the relatively high proportion of lymphovascular invasion (44.2%) and lymph node metastasis (42.3%) indicates that despite the predominance of low-grade tumors, many patients already presented with invasive disease that increases the risk of further dissemination [22].
The pattern of spread observed in this study showed that nearly half of the patients had lymph node involvement (42.3%), with most falling into the N1 category (33.6%), defined as metastasis to one to 3 regional lymph nodes. This finding is in line with the well-established concept that lymphatic dissemination represents one of the earliest steps in colorectal cancer progression before tumor cells enter the systemic circulation and metastasize to distant organs [7], [21]. Lymph node involvement has long been recognized as a key predictor of prognosis and serves as a cornerstone of the globally applied TNM staging system (UICC/AJCC) [13]. Ueno et al. [23] further demonstrated that the number of metastatic lymph nodes is directly correlated with recurrence risk and poorer survival outcomes.
Regarding distant metastasis, this study reaffirmed that the liver was the most common site involved (18.3%), which is consistent with the hematogenous spread through the portal venous system that makes the liver the first “filter” for disseminating cancer cells [24,25]. This proportion is comparable to the findings of Wangi et al. [24], who reported that more than half of CRC patients with distant metastasis had liver involvement, either as the sole metastatic site or in combination with other organs. By contrast, pulmonary (1.9%) and bone metastases (1.0%) were relatively rare, but their presence reflects advanced disease associated with poor prognosis. These findings are in agreement with the population-based study by Riihimäki et al. [26], which documented the typical metastatic pattern of CRC, with the liver as the dominant site, followed by the lungs and bones.
Clinically, the combination of lymph node and distant organ involvement highlights that many patients in this cohort were already diagnosed at a potentially aggressive stage of disease. The fact that nearly half of the cases had nodal metastasis and about one-fifth had liver involvement underscores the importance of early detection and structured screening programs in high-risk populations. Moreover, histopathological evaluation of nodal status, along with radiological assessment for distant spread, is essential not only for risk stratification but also for guiding therapeutic strategies, including decisions between curative resection and systemic chemotherapy.
When analyzed by pathological stage, more than half of the patients were diagnosed at an early stage (I-II). This is encouraging, as curative treatment offers a higher chance of long-term survival when CRC is detected early. Nevertheless, a considerable proportion of patients were already at advanced stages (III-IV), accounting for nearly half of all cases. This distribution mirrors reports from other Southeast Asian countries, where the stage at diagnosis varies widely depending on access to healthcare and the availability of systematic screening programs [14].
In terms of biomarker expression, Claudin-4 was strongly expressed in 79.8% of cases. This finding is consistent with previous reports that Claudin-4 is frequently overexpressed in colorectal adenocarcinoma and may be associated with tumor aggressiveness, deeper invasion, and EMT [11,27]. However, its role as a prognostic biomarker remains controversial, as some studies have linked high expression to more favorable outcomes. The present study therefore contributes to the growing body of evidence suggesting that Claudin-4 has potential as an additional prognostic marker, particularly when considered in combination with histopathological features and tumor staging.
The intensity of Claudin-4 immunohistochemistry staining can be observed in the membrane and/or cytoplasm of tumor cells to varying degrees. Figure 1 below shows the expression of Claudin-4 for each intensity score.
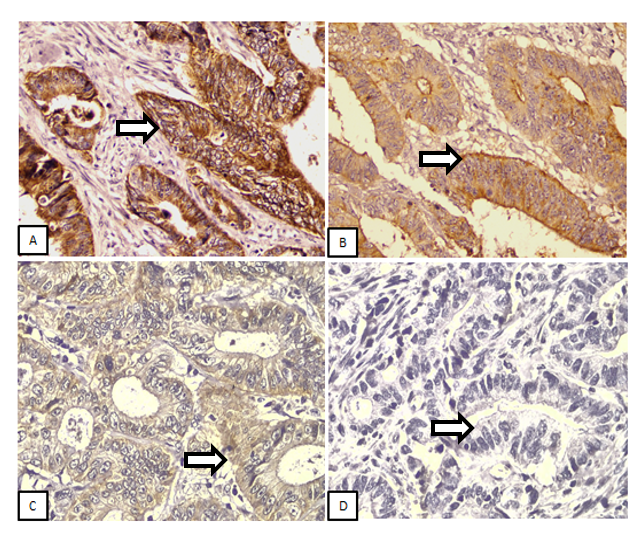
Figure 1 Claudin-4 Intensity in Colorectal Adenocarcinoma. A: Strong (+3); B: Moderate (+2); C: Weak (+1); D: Negative (0). The arrows highlight representative tumor cells demonstrating the respective levels of Claudin-4 staining intensity (IHC, 400× Magnification).
Table 2 Correlation of Claudin-4 expression with age, sex, tumor location, tumor size of colorectal adenocarcinoma samples.
Characteristics |
(n) |
Claudin-4 Expression |
p Valuea |
|
Weak n (%) |
Strong n (%) |
|||
Age |
|
|
||
≤ 50 |
28 |
6 (21.4) |
22 (78.6) |
0.849 |
> 50 |
76 |
15 (19.7) |
61 (80.3) |
|
Sex |
|
|
||
Male |
56 |
12 (21.4) |
44 (78.6) |
0.734 |
Female |
48 |
9 (18.8) |
39 (81.3) |
|
Tumor Location |
|
|
||
Proximal |
30 |
8 (26.7) |
22 (73.3) |
0.518b |
Distal |
32 |
1 (3.1) |
31 (96.9) |
|
Rectum |
36 |
10 (27.8) |
26 (72.2) |
|
Rectosigmoid |
5 |
2 (33.3) |
4 (66.7) |
|
Tumor Size (cm) |
|
|
|
|
< 3 |
11 |
1 (9.1) |
10 (90.9) |
|
3 - 5 |
59 |
11(18.6) |
48 (81.4) |
0.415 |
> 5 |
34 |
9 (26.5) |
25 (73.5) |
|
aChi-square test; bMann-Whitney test; *Significant p value ≤ 0.05.
Table 2 presents the analysis of the relationship between Claudin-4 expression and patient characteristics in colorectal adenocarcinoma. The findings demonstrate that there were no statistically significant differences across the variables examined. For age, strong Claudin-4 expression was observed in 78.6% of patients ≤ 50 years and 80.3% of patients > 50 years (p = 0.849). Regarding sex, strong expression was seen in 78.6% of male patients and 81.3% of female patients, with no statistically significant difference (p = 0.734). Tumor location showed some variability in strong Claudin-4 expression, with distal colon (96.9%), rectosigmoid (66.7%), rectum (72.2%), and proximal colon (73.3%); however, these differences were not statistically significant (p = 0.518). For tumor size, strong expression was most frequently observed in tumors measuring 3 - 5 cm (81.4%) compared to < 3 cm (90.9%) and > 5 cm (73.5%), but again, the differences were not statistically significant (p = 0.415). Collectively, these results suggest that Claudin-4 expression is not significantly associated with demographic factors (age and sex) or basic clinical variables (tumor location and tumor size). This supports the interpretation that Claudin-4 expression is more likely determined by biological and molecular characteristics of the tumor rather than by host-related or anatomical factors.
These findings are supported by previous studies. Süren et al. [28]; Kyuno et al. [29] reported that Claudin-4 expression in colorectal adenocarcinoma is more closely related to EMT and tumor progression than to patient demographics [28,29]. Similarly, Hana et al. [11]; Ersoz et al. [30]; Cox et al. [31] found that high Claudin-4 expression was correlated with tumor depth of invasion and lymph node metastasis, but not with age or sex. Although in this study higher expression was more frequently observed in distal colon tumors, the difference between proximal and distal locations was not statistically significant. Several studies have reported molecular distinctions between proximal and distal colorectal cancers, particularly the higher prevalence of microsatellite instability (MSI) in proximal tumors [7]. However, Claudin-4 expression did not appear to fully follow this pattern, suggesting that its role may be more universal as a marker of cell adhesion and EMT regulation.
Tumor size also showed no significant association with Claudin-4 expression. This finding is consistent with the study by Fujiwara-Tani et al. [8]; Kyuno et al. [29], which suggested that Claudin-4 expression does not directly correlate with tumor bulk growth but is more closely linked to invasive and migratory capacity of cancer cells [8,29]. Taken together, these findings imply that Claudin-4 may be more relevant in the context of microscopic invasion processes rather than macroscopic tumor expansion.
Table 3 Correlation of Claudin-4 expression with histopathological grading and tumor infiltration of colorectal adenocarcinoma samples.
Characteristics |
(n) |
Claudin-4 Expression |
p Valuea |
|
Weak n (%) |
Strong n (%) |
|||
Histopathological Grade |
|
|
|
|
Low |
65 |
16 (24.6) |
49 (75.4) |
0.147 |
High |
39 |
5 (12.8) |
34 (87.2) |
|
Tumor Infiltration |
|
|
|
|
T1 |
20 |
5 (25.0) |
15 (75.0) |
0.098 |
T2 |
41 |
4 (9.08) |
37 (90.2) |
|
T3 |
43 |
12 (27.9) |
31 (72.1) |
|
aChi-square test; *Significant p value ≤ 0.05
In this study, the analysis of Claudin-4 expression in relation to histopathological grading and tumor infiltration (T) revealed no statistically significant differences. In the low-grade group, strong Claudin-4 expression was observed in 75.4% of cases, while in the high-grade group it was slightly higher at 87.2% (p = 0.147). This observation is also consistent with the study by Tokuhara et al. [32] in gastric cancer, which found no significant correlation between Claudin-4 expression and histopathological grade. It is plausible that Claudin-4 plays a greater role in adhesion and invasion at the cellular level rather than directly affecting differentiation, which underlies grading systems [12]. Indeed, histopathological grade reflects how closely tumor cells resemble their normal counterparts, a process likely governed by distinct or more dominant signaling pathways than those regulated by Claudin-4 [8,33].
With regard to tumor infiltration, strong Claudin-4 expression was most frequently observed in T2 tumors (90.2%), followed by T1 (75.0%) and T3 (72.1%) (p = 0.098). This trend suggests that Claudin-4 may play a more prominent role in the early phase of invasion, particularly as the tumor penetrates the muscularis propria, but its expression tends to decline in deeper invasive stages. This phenomenon aligns with the findings of Fujiwara-Tani et al. [8]; Ueno et al. [23], who reported that Claudin-4 undergoes alterations in function and localization during colorectal cancer progression, particularly through epithelial–mesenchymal transition (EMT) [34-36]. In other words, Claudin-4 may participate in the dynamic transition of cancer cells from early invasive phases toward more aggressive phenotypes. Although the associations observed were not statistically significant, the tendency for stronger Claudin-4 expression in high-grade tumors and T2 infiltration is noteworthy. The lack of significance may be biological heterogeneity across cases. Therefore, further studies with larger cohorts and additional molecular approaches are needed to clarify the prognostic role of Claudin-4, especially when evaluated alongside other invasive markers.
Table 4 Correlation of Claudin-4 expression with lymphovascular invasion and lymph node metastasis of colorectal adenocarcinoma samples.
Characteristics |
(n) |
Claudin-4 Expression |
OR (CI 95%) |
p Valuea |
|
Weak n (%) |
Strong n (%) |
||||
Lymphovascular Invasion |
|
|
|
|
|
Negative |
58 |
7 (12.1) |
51 (87.9) |
0.314 (0.114 - 0.861) |
0.020* |
Positive |
46 |
14 (30.4) |
32 (69.6) |
||
Lymph Node Metastasis |
|
|
|
|
|
Negative |
60 |
7 (11.7) |
53 (88.3) |
0.283 (0.103 - 0.778) |
0.011* |
Positive |
44 |
14 (31.8) |
30 (68.2) |
||
aChi-square test; *Significant p value ≤ 0.05.
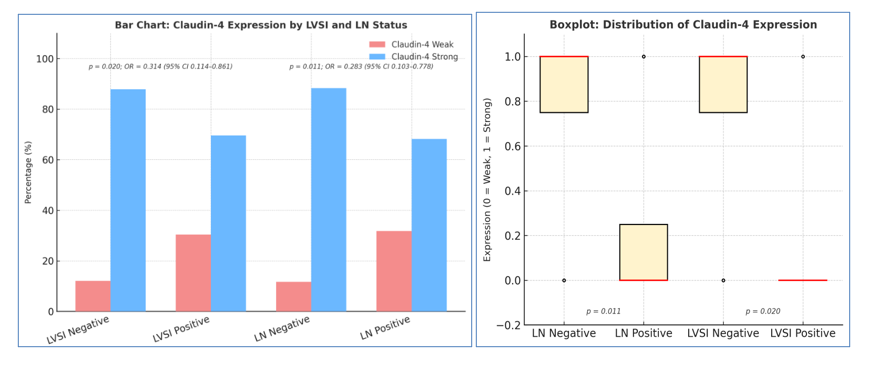
Figure
2
Claudin-4
expression according to lymphovascular invasion (LVSI) and lymph
node (LN) status in colorectal adenocarcinoma. (A)
Bar
chart
shows the proportion of weak and strong Claudin-4 expression in
relation to LVSI and LN status. Strong expression predominated in
non-invasive groups (LVSI−: 87.9%; LN−: 88.3%) compared to
invasive groups (LVSI+: 69.6%; LN+: 68.2%). Statistical analysis
demonstrated significant associations for LVSI (p
= 0.020; OR = 0.314; 95% CI 0.114 - 0.861) and LN status (p
= 0.011; OR = 0.283; 95% CI 0.103 - 0.778).
(B) Boxplots
illustrate the distribution of Claudin-4 expression (0 = weak, 1 =
strong) across groups. Non-invasive categories (LVSI− and LN−)
show tight distributions centered at strong expression, while
invasive categories (LVSI+ and LN+) display greater variability with
more cases of weak expression, indicating a shift in Claudin-4
patterns in invasive tumors.
In this study, a significant association was found between Claudin-4 expression and both lymphovascular invasion (LVSI) and lymph node metastasis. The majority of cases without LVSI demonstrated strong Claudin-4 expression (87.9%), whereas in cases with LVSI, the proportion of strong expression decreased to 69.6% (p = 0.020). A similar pattern was observed in nodal metastasis: patients with N0 status more frequently expressed strong Claudin-4 (88.3%), while in those with nodal involvement, the proportion declined to 68.2% (p = 0.011). These findings indicate that reduced Claudin-4 expression is more commonly associated with tumors exhibiting invasive behavior and a tendency toward regional metastasis.
This result is consistent with the studies by Ueda et al. [22]; Li et al. [37]; Nefedova et al. [38]; Wang et al. [39]; Fatimah [40], which reported a correlation between Claudin-4 expression, tumor invasion, and lymph node involvement in colorectal adenocarcinoma. These studies also emphasized the close association of Claudin-4 with epithelial–mesenchymal transition (EMT), a process that enhances cancer cell migration and invasion. Interestingly, although some previous studies have linked Claudin-4 overexpression with tumor aggressiveness, the present findings suggest that reduced expression is likewise associated with LVSI and nodal metastasis. This paradox may be explained by the dual nature of Claudin-4: In certain contexts, high expression may promote an invasive phenotype, whereas in more advanced stages, reduced expression facilitates the detachment of cancer cells into the lymphatic circulation.
Collectively, these findings indicate that Claudin-4 may exert a context-dependent role in colorectal cancer progression, functioning both to maintain epithelial cohesion and to facilitate cellular detachment required for metastasis. The observed association between Claudin-4 expression, lymphovascular invasion, and lymph node metastasis reinforces its value as a potential prognostic biomarker in colorectal adenocarcinoma. However, the underlying molecular mechanisms remain complex, warranting further investigation to clarify whether upregulation or downregulation more consistently reflects tumor aggressiveness. Overall, Claudin-4 appears to be predominantly active during the early phase of tumorigenesis and local invasion, preserving epithelial integrity, while its reduced expression may enable cellular detachment and dissemination at later stages.
Table 5 Correlation between distant organ metastasis, pathological stage, and claudin-4 expression in colorectal adenocarcinoma samples.
Characteristics |
(n) |
Claudin-4 Expression |
p Value |
|
Weak n (%) |
Strong n (%) |
|||
Distant Organ Metastasis |
|
|
||
Negative |
81 |
17 (21.0) |
64 (79.0) |
0.705a |
Positive |
23 |
4 (17.4) |
19 (82.6) |
|
Pathological Stage |
|
|
||
Stage I |
34 |
3 (8.8) |
31 (91.2) |
0.028*b |
Stage IIA |
19 |
3 (15.8) |
16 (84.2) |
|
Stage IIB |
1 |
0 (0.0) |
1 (100) |
|
Stage IIIA |
12 |
6 (50) |
6 (50) |
|
Stage IIIB |
19 |
5 (26.3) |
14 (73.7) |
|
Stage IVA |
18 |
3 (16.7) |
15 (83.3) |
|
Stage IVB |
1 |
1 (100) |
0 (0.0) |
|
aChi-square test; bFisher’s exact test *Significant p value ≤ 0.05
The analysis presented in Table 5 demonstrated that Claudin-4 expression was not significantly associated with distant metastasis, as most cases without organ metastasis exhibited strong expression (79.0%), with a comparable proportion observed among those with distant metastasis (82.6%; p = 0.705). In contrast, a significant association was found with pathological stage (p = 0.028), where strong Claudin-4 expression predominated in early stages, particularly Stage I (91.2%) and Stage IIA (84.2%), but progressively declined in advanced disease, decreasing in Stage IIIA (50.0%) and Stage IIIB (73.7%), with exclusively weak expression observed in Stage IVB (100%). These findings indicate that Claudin-4 expression is unrelated to distant metastasis and may instead play a more prominent role in local invasion and regional lymph-node involvement rather than systemic dissemination. Although no prior study has directly examined the relationship between Claudin-4 and pathological stage, earlier investigations by Ueda et al. [22]; Li et al. [37]; Nefedova et al. [38]; Wang et al. [39]; Fatimah [40] have reported associations between Claudin-4 expression and several parameters contributing to pathological staging. Consistent with these findings, our analysis revealed a negative correlation between pathological stage and Claudin-4 expression, suggesting that Claudin-4 downregulation occurs more frequently in advanced disease. This pattern may reflect its involvement in EMT, whereby the loss of tight-junction integrity facilitates local invasion and drives progression to higher stages of colorectal adenocarcinoma.
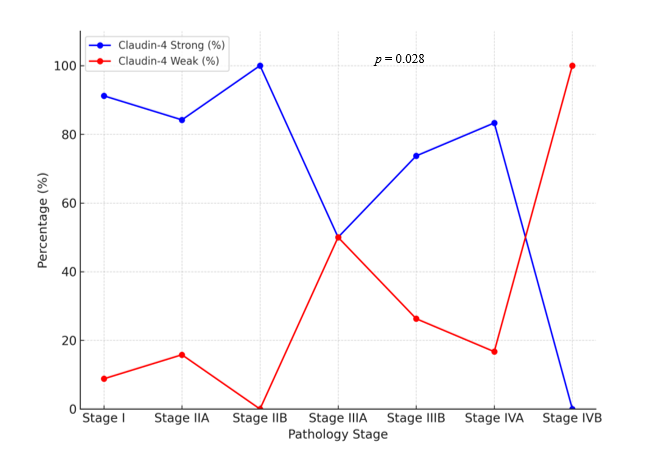
Figure 3 Trend of Claudin-4 expression across pathological stages of colorectal adenocarcinoma.
Line chart illustrates the distribution of strong (blue) and weak (red) Claudin-4 expression according to pathological stage (I - IVB). Strong expression predominated in early stages (Stage I - II, > 80%), but progressively declined in advanced stages. A marked drop was observed in Stage IIIA (50%), with further reduction in Stage IIIB and IVA, and complete loss of strong expression in Stage IVB (100% weak). Statistical analysis confirmed a significant association between Claudin-4 expression and pathological stage (p = 0.028, Fisher’s exact test), suggesting Claudin-4 downregulation is linked to tumor progression and advanced disease.
These observations are in line with recent reports describing the dual role of Claudin-4 in cancer biology: Fujiwara-Tani et al. [8] emphasized that Claudin-4 can function either as a tumor suppressor or as a promoter of invasiveness depending on the tumor context and microenvironment, while Cox et al. [31] observed that in colorectal cancer, high Claudin-4 expression is typically found in early stages, with reduced expression at the invasive front correlating with more aggressive behavior. This aligns with the findings of Li et al. [37], who emphasized the context-dependent function of Claudin-4 in cancer progression. In the early stages of tumorigenesis, Claudin-4 helps preserve epithelial integrity and tight-junction stability, whereas alterations in its expression facilitate local invasion. Furthermore, reduced expression is associated with tumor advancement and more aggressive disease behavior. In agreement with these studies, Claudin-4 expression in our cohort was strongly preserved in early-stage tumors (Stages I and IIA), implying its essential role in maintaining intercellular adhesion during initial tumor formation, whereas diminished expression in advanced, deeply invasive, or regionally metastatic tumors suggests a phenotypic transition toward enhanced motility and invasiveness through the EMT process [8,12]. Moreover, a significant association was identified between reduced Claudin-4 expression and the presence of lymphovascular invasion (LVSI) and lymph-node metastasis; patients without LVSI or nodal involvement generally exhibited strong expression, whereas those with invasion and metastasis displayed weaker staining. Nonetheless, no significant correlation was observed between Claudin-4 expression and distant organ metastases, including the liver, lungs, and bones, supporting the interpretation that Claudin-4 serves as a biomarker more closely related to local and regional invasion rather than systemic dissemination [22].
These results further reinforce the interpretation that Claudin-4 functions as a valuable complementary biomarker when assessed alongside conventional histopathological parameters. The integration of Claudin-4 immunoexpression with indicators such as lymphovascular invasion, lymph-node metastasis, and pathological stage may improve the reliability of prognostic evaluation in colorectal cancer. This combined, multimodal approach could enhance diagnostic precision and refine risk stratification, particularly in identifying early-stage patients who may benefit most from curative therapies, while distinguishing those at higher risk for local invasion or nodal involvement despite the absence of distant metastasis.
Although significant correlations were observed between Claudin-4 expression, lymphovascular invasion, lymph-node metastasis, and pathological stage, the current findings alone cannot conclusively establish the prognostic efficacy of Claudin-4 in colorectal adenocarcinoma. The cross-sectional design of this study limits its ability to assess longitudinal outcomes and causal relationships, as the data represent a single time-point observation rather than temporal dynamics. Therefore, further studies with larger cohorts, multicenter participation, and survival analyses are warranted to validate the prognostic significance and clinical utility of Claudin-4 [8,37,38]. Such investigations will help determine whether Claudin-4 can be reliably implemented in clinical practice for risk assessment, therapeutic decision-making, and outcome prediction. Until stronger evidence emerges, Claudin-4 should be regarded as a promising adjunct marker that is best interpreted in conjunction with established histopathological parameters.
Conclusions
The findings of this study support the hypothesis that Claudin-4 expression is correlated with prognostic parameters of tumor invasiveness in colorectal adenocarcinoma. Claudin-4 expression negative correlated significantly with lymphovascular invasion, lymph node metastasis, and pathological stage. Strong Claudin-4 expression was more frequently observed in cases without lymphovascular invasion, without lymph node metastasis, and in early pathological stages, whereas reduced expression correlated with more aggressive tumor behavior. This pattern suggests that Claudin-4 plays a role in maintaining cell adhesion during the early phase of tumorigenesis but undergoes downregulation as tumors progress, thereby contributing to tumor invasion and progression. Although no significant associations were found with patient demographics or distant metastasis, the significant link between Claudin-4 expression and pathological stage highlights its potential as a supplementary prognostic biomarker, particularly for predicting local invasion and regional progression. Further multicenter studies with larger cohorts and survival analyses are warranted to validate these findings and establish the long-term prognostic value of Claudin-4 in colorectal cancer.
Acknowledgements
We thank all volunteers who participated. We are grateful to the Anatomic Pathology Laboratory at Hasanuddin University Hospital for facilitating this study, and to Ni Ketut Sungowati, Juanita, and Imeldy Prihatni Purnama at Dr. Wahidin Sudirohusodo General Hospital for their assistance with sample collection. This research received some funding from Immuno-Oncology, Thematic Research Group Hasanuddin University, No15523/UN4.1/KEP/2024.
Declaration of generative AI in scientific writing
Generative AI tools (ChatGPT, OpenAI) were used to improve the readability and language of this manuscript under human oversight. The authors are fully responsible for the content.
CRediT author statement
Ririn Endah Puspitasari: Conceptualization; Methodology; Data curation; Formal analysis; Validation; Funding acquisition; Writing - original draft. Upik Anderiani Miskad: Conceptualization; Supervision; Methodology; Writing - Review & Editing. Muhammad Husni Cangara: Supervision; Methodology; Writing - Review & Editing. Syarifuddin Wahid: Supervision; Methodology; Writing - Review & Editing. Djumadi Achmad: Supervision; Methodology; Writing - Review & Editing. Amalia Yamin: Supervision; Methodology; Writing - Review & Editing. Suryani Tawali: Supervision; Methodology; Formal analysis.
References
[1] F Bray, M Laversanne, H Sung, J Ferlay, RL Siegel, I Soerjomataram and A Jemal. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians 2024; 74(3), 229-263.
[2] RL Siegel, KD Miller, AG Sauer, SA Fedewa, LF Butterly, JC Anderson, A Cercek, RA Smith and A Jemal. Colorectal cancer statistics, 2020. CA: A Cancer Journal for Clinicians 2020; 70(3), 145-164.
[3] P Tantimetta, T Rattanaarchanai, A Aroonkesorn, W Chanket, P Runsaeng, S Roytrakul, P Hutamekalin and S Obchoei. Recombinant bacteriocin rhamnosin suppresses malignant phenotypes of colorectal cancer cells. Trends in Sciences 2025; 22(8), 10057.
[4] AE Noffsinger. Fenoglio-Preiser’s gastrointestinal pathology. Lippincott Williams & Wilkins, New York, 2017.
[5] UA Miskad, MM Henry, CI Pardamean, A Budiarto, A Irwan, JW Baurley, I Yusuf and B Pardamean. Colorectal cancer in south sulawesi: A case-control study for nongenetic risk factors. Oncology Reviews 2025; 19, 1589655.
[6] X Wang, Y Cao, M Ding, J Liu, X Zuo, H Li and R Fan. Oncological and prognostic impact of lymphovascular invasion in colorectal cancer patients. International Journal of Medical Sciences 2021; 18(7), 1721.
[7] AM Rianti, MH Cangara, A Yamin, H Dahlan, MR Ilyasa and UA Miskad. FGFR2 as a prognostic and predictive marker in colorectal adenocarcinoma based on tils grade. The Indonesian Biomedical Journal 2025; 17(2), 188-196.
[8] R Fujiwara-Tani, S Mori, R Ogata, R Sasaki, A Ikemoto, S Kishi, M Kondoh and H Kuniyasu. Claudin-4: A new molecular target for epithelial cancer therapy. International Journal of Molecular Sciences 2023; 24(6), 5494.
[9] N Roehlen, AA Roca Suarez, H El Saghire, A Saviano, C Schuster, J Lupberger and TF Baumert. Tight junction proteins and the biology of hepatobiliary disease. International Journal of Molecular Sciences 2020; 21(3), 825.
[10] CD Hough, CA Sherman-Baust, ES Pizer, FJ Montz, DD Im, NB Rosenshein, KR Cho, GJ Riggins and PJ Morin. Large-scale serial analysis of gene expression reveals genes differentially expressed in ovarian cancer. Cancer Research 2000; 60(2), 6281-6287.
[11] C Hana, NN Thaw Dar, M Galo Venegas and M Vulfovich. Claudins in cancer: A current and future therapeutic target. International Journal of Molecular Sciences 2024; 25(9), 4634.
[12] J Li. Context-dependent roles of claudins in tumorigenesis. Frontiers in Oncology 2021; 11, 67681.
[13] OP Popova, AV Kuznetsova, SY Bogomazova and AA Ivanov. Claudins as biomarkers of differential diagnosis and prognosis of tumors. Journal of Cancer Research and Clinical Oncology 2021; 147(10), 2803-2817.
[14] H Sung, J Ferlay, RL Siegel, M Laversanne, I Soerjomataram, A Jemal and F Bray. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians 2021; 71(3), 209-249.
[15] T Sawicki, M Ruszkowska, A Danielewicz, E Niedźwiedzka, T Arłukowicz and KE Przybyłowicz. A review of colorectal cancer in terms of epidemiology, risk factors, development, symptoms and diagnosis. Cancers 2021; 13(9), 2025.
[16] M Ahmed. Colon cancer: A clinician’s perspective in 2019. Gastroenterology Research 2020; 13(1), 1.
[17] M Abancens, V Bustos, H Harvey, J McBryan and BJ Harvey. Sexual dimorphism in colon cancer. Frontiers in Oncology 2020; 10, 607909.
[18] O Majek, A Gondos, L Jansen, K Emrich, B Holleczek, A Katalinic, A Nennecke, A Eberle and H Brenner. Sex differences in colorectal cancer survival: Population-based analysis of 164,996 colorectal cancer patients in Germany. PLoS One 2013; 8(7), e68077.
[19] F Caiazza, EJ Ryan, G Doherty, DC Winter and K Sheahan. Estrogen receptors and their implications in colorectal carcinogenesis. Frontiers in Oncology 2015; 5, 19.
[20] P Rawla, T Sunkara and A Barsouk. Epidemiology of colorectal cancer: Incidence, mortality, survival, and risk factors. Gastroenterology Review/Przegląd Gastroenterologiczny 2019; 14(2), 89-103.
[21] I Theodora, IK Sudiana, VS Budipramana, F Erwin, S Dewi and BD Novita. Tumor differentiation is correlated with Estrogen Receptor Beta (ERβ) expression but not with Interleukin-6 (IL-6) expression in colorectal carcinoma. The Indonesian Biomedical Journal 2024; 16(5), 481-486.
[22] J Ueda, S Semba, H Chiba, N Sawada, Y Seo, M Kasuga and H Yokozaki. Heterogeneous expression of claudin-4 in human colorectal cancer: Decreased claudin-4 expression at the invasive front correlates cancer invasion and metastasis. Pathobiology 2007; 74(1), 32-41.
[23] H Ueno, H Mochizuki, Y Hashiguchi, H Shimazaki, S Aida, K Hase, S Matsukuma, T Kanai, H Kurihara, K Ozawa, K Yoshimura and S Bekku. Risk factors for an adverse outcome in early invasive colorectal carcinoma. Gastroenterology 2004; 127(2), 385-394.
[24] Y Wang, X Zhong, X He, Z Hu, H Huang, J Chen, K Chen, S Zhao, P Wei and D Li. Liver metastasis from colorectal cancer: Pathogenetic development, immune landscape of the tumour microenvironment and therapeutic approaches. Journal of Experimental & Clinical Cancer Research 2023; 42(1), 177.
[25] J Martin, A Petrillo, EC Smyth, N Shaida, S Khwaja, HK Cheow, A Duckworth, P Heister, R Praseedom, A Jah, A Balakrishnan, S Harper, S Liau, V Kosmoliaptsis and E Huguet. Colorectal liver metastases: Current management and future perspectives. World Journal of Clinical Oncology 2020; 11(10), 761-808.
[26] M Riihimäki, A Hemminki, J Sundquist and K Hemminki. Patterns of metastasis in colon and rectal cancer. Scientific Reports 2016; 6, 29765.
[27] R Fujiwara-Tani, T Sasaki, YI Luo, K Goto, I Kawahara, Y Nishiguchi, S Kishi, S Mori, H Ohmori, M Kondoh and H Kuniyasu. Anti-claudin-4 extracellular domain antibody enhances the antitumoral effects of chemotherapeutic and antibody drugs in colorectal cancer. Oncotarget 2018; 9(100), 37367.
[28] D Süren, M Yıldırım, V Kaya, AS Alikanoğlu, N Bülbüller, M Yıldız and C Sezer. Loss of tight junction proteins (Claudin 1, 4, and 7) correlates with aggressive behavior in colorectal carcinoma. Medical Science Monitor: International Medical Journal of Experimental and Clinical Research 2014; 20, 1255.
[29] D Kyuno, A Takasawa, S Kikuchi, I Takemasa, M Osanai and T Kojima. Role of tight junctions in the epithelial-to-mesenchymal transition of cancer cells. Biochimica et Biophysica Acta (BBA)-Biomembranes 2021; 1863(3), 183503.
[30] S Ersoz, S Mungan, U Cobanoglu, H Turgutalp and Y Ozoran. Prognostic importance of Claudin-1 and Claudin-4 expression in colon carcinomas. Pathology - Research and Practice 2011; 207(5), 285-289.
[31] KE Cox, S Liu, RM Hoffman, SK Batra, P Dhawan and M Bouvet. The expression of the claudin family of proteins in colorectal cancer. Biomolecules 2024; 207(5), 285-289.
[32] Y Tokuhara, T Morinishi, T Matsunaga, H Ohsaki, Y Kushida, R Haba and E Hirakawa. Claudin-1, but not claudin-4, exhibits differential expression patterns between well- to moderately-differentiated and poorly-differentiated gastric adenocarcinoma. Oncology Letters 2015; 10(1), 93-98.
[33] DW Wang, WH Zhang, G Danil, K Yang and JK Hu. The role and mechanism of claudins in cancer. Frontiers in Oncology 2022; 12, 1051497.
[34] B Baran, NM Ozupek, NY Tetik, E Acar, O Bekcioglu and Y Baskin. Difference between left-sided and right-sided colorectal cancer: A focused review of literature. Gastroenterology Research 2018; 11(4), 264.
[35] G Menon, A Recio-Boiles, S Lotfollahzadeh and B Cagir. Colon cancer. StatPearls Publishing, Florida, United States, 2023.
[36] J Yang, P Antin, G Berx, C Blanpain, T Brabletz, M Bronner, K Campbell, A Cano, J Casanova, G Christofori, S Dedhar, R Derynck, HL Ford, J Fuxe, AG de Herreros, GJ Goodall, AK Hadjantonakis, RYJ Huang and C Kalcheim. Guidelines and definitions for research on epithelial - mesenchymal transition. Nature Reviews Molecular Cell Biology 2020; 21(6), 341-352.
[37] W Li, SQ Zong, Q Shi, HJ Li, J Xu and F Hou. Hypoxia-induced vasculogenic mimicry formation in human colorectal cancer cells: Involvement of HIF-1a, Claudin-4, and E-cadherin and Vimentin. Scientific Reports 2016; 6(1), 37534.
[38] NА Nefedova, ОА Kharlova and PG Malkov. Еxpression of claudin-1, 3, and 4 in colorectal cancer and polyps. Arkhiv Patologii 2016; 78(3), 11-19.
[39] L Wang, SY Li, P An and HY Cai. Expression and clinical significance of Claudin-1 and Claudin-4 in colorectal cancer tissues. Zhonghua wei Chang wai ke za zhi= Chinese Journal of Gastrointestinal Surgery 2012; 15(10), 1073-1076.
[40] S Fatimah, AS Rahaju and A Rahniayu. Role of claudin-4 and matrix metalloproteinase-2 in tumor invasion of colorectal adenocarcinoma. Research Journal of Pharmacy and Technology 2021; 14(9), 4795-4800.