
Trends
Sci.
2026; 23(6): 13289
Biochar-Based Catalysts in Hydrogen Production: Mechanisms, Activation, and Efficiency
Ricky Andi Syahputra1,2, Muhammad Irvan1, Andriayani Andriayani1,3,
Basuki Wirjosentono1, Karna Wijaya4, Shiplu Sarker5 and Saharman Gea1,3,*
1Postgraduate School, Department of Chemistry, Faculty of Mathematics and Natural Sciences,
Universitas Sumatera Utara, Medan 20155, Indonesia
2Department of Chemistry, Faculty of Mathematics and Natural Sciences, Universitas Negeri Medan,
Medan 20221, Indonesia
3Cellulosic and Functional Materials Research Centre, Universitas Sumatera Utara, Medan 20155, Indonesia
4Department of Chemistry, Faculty of Mathematics and Natural Sciences, Gadjah Mada University,
Yogyakarta 55281, Indonesia
5Department of Civil and Manufacturing Engineering, Faculty of Engineering,
Norwegian University of Science and Technology, NTNU, Gjøvik 2815, Norway
(*Corresponding author’s e-mail: [email protected])
Received: 8 January 2026, Revised: 19 February 2026, Accepted: 26 February 2026, Published: 1 March 2026
Abstract
Biochar, a carbon-rich material derived from biomass, has emerged as a promising catalytic platform owing to its low cost, wide availability, structural tunability, and environmental compatibility. This review provides a critical and mechanistically integrated assessment of biochar-based catalysts for sustainable hydrogen production across thermochemical and photochemical pathways. Rather than cataloging reported yields, the analysis adopts a standardized normalization framework, expressing hydrogen productivity primarily as mmol H₂ g⁻¹ catalyst h⁻¹ to enable reliable cross-study comparison. The review introduces a unified structure–property–performance perspective linking feedstock chemistry, activation strategy, and metal–carbon interactions to pathway-specific hydrogen productivity. Evidence indicates that feedstock-derived chemistry often exerts a stronger influence on catalytic behavior than pyrolysis temperature alone, while controlled metal doping—particularly with Ni, Fe, and Co—enhances active-site accessibility and stability. Activation treatments modulate porosity and surface functionality, yet excessive modification may induce structural degradation or catalyst deactivation. Beyond catalytic efficiency, biochar-based systems offer sustainability advantages through biomass valorization, reduced reliance on critical raw materials, and compatibility with circular carbon strategies. By integrating mechanistic insight, normalization rigor, and sustainability considerations, this review positions biochar not merely as a low-cost alternative support, but as a tunable catalytic platform for next-generation hydrogen technologies.
Keywords: Biochar catalysts, Hydrogen production, Surface activation, Catalytic efficiency, Renewable energy
Introduction
The role of hydrogen in the energy landscape
Hydrogen is increasingly recognized as a strategic energy carrier in the global transition toward low-carbon systems, with applications spanning industry, transport, power generation, and chemical synthesis [1-3]. According to the International Energy Agency [178], global hydrogen demand reached approximately 95
million tonnes in 2023, yet over 95% of production remains fossil-based, generating nearly 900 million tonnes of CO₂ annually. Achieving net-zero emissions by 2050 will require rapid expansion of low-carbon hydrogen to an estimated 500 - 600 million tonnes per year, alongside advances in renewable integration, electrolyzer performance, and carbon management technologies.
Currently, steam methane reforming (SMR) dominates global hydrogen supply, accounting for the majority of production but emitting substantial CO₂. Mitigation strategies such as carbon capture (“blue hydrogen”) and methane pyrolysis reduce emissions to varying degrees; however, these approaches remain dependent on fossil feedstocks and involve additional cost or infrastructure requirements. Although green hydrogen via renewable-powered electrolysis is widely promoted, high capital costs and system integration challenges continue to limit large-scale deployment.
These constraints highlight the need for alternative catalytic systems that are sustainable, economically viable, and compatible with biomass valorization. Biochar-based catalysts derived from agricultural and forestry residues offer renewable carbon frameworks with tunable porosity and surface functionality. When modified with transition metals such as Ni or Fe, biochar can serve as an efficient and low-cost platform for reforming and gasification processes. By coupling waste biomass utilization with hydrogen production, this approach addresses both catalytic performance and circular bioeconomy objectives, positioning biochar as a promising material in the development of scalable, low-carbon hydrogen technologies.
Challenges in current catalytic methods
Catalysts are indispensable in facilitating hydrogen production by enhancing reaction rates, selectivity, and overall process efficiency. However, conventional catalytic systems are not without limitations. Across environmental and energy applications, catalytic methods encounter persistent challenges such as limited understanding of surface chemistry, low stability under harsh operating conditions, and difficulties in emissions control catalysis [10].
In particular, nickel-based catalysts, though widely used due to their affordability and availability, suffer from coke deposition, active site sintering, and sensitivity to sulfur and chlorine impurities, leading to rapid deactivation. Meanwhile, noble metal catalysts such as platinum (Pt), palladium (Pd), and ruthenium (Ru) exhibit superior activity and resistance to coking but remain economically unfeasible for large-scale deployment due to their high cost and limited reserves. These challenges have spurred the exploration of alternative catalytic materials that are cost-effective, efficient, and environmentally benign.
The direct functionalization of methane represents another significant hurdle due to complex mechanistic pathways, mass transfer limitations, and the need for robust catalyst design [11]. Similarly, gas-to-liquids (GTL) and steam reforming processes face bottlenecks in synthesis gas generation, Fischer–Tropsch selectivity, and product upgrading [12]. Thermochemical (e.g., pyrolysis, gasification) and photochemical (e.g., water splitting) methods are also highly catalyst-dependent for achieving high hydrogen yields and stable operation. A recent overview of catalysis challenges underscores the broad scope of innovation required—ranging from surface modification to nanostructured and bio-derived catalytic systems [13].
To align with sustainability principles and Environmental, Social, and Governance targets, the development of next-generation catalysts must emphasize resource efficiency, waste valorization, and carbon footprint reduction. In this regard, emerging catalysts based on biochar, perovskites, and transition metal carbides (e.g., Mo₂C, Ni–Fe composites) show strong potential to support the circular economy by utilizing renewable and waste-based precursors, thereby minimizing dependence on critical raw materials. The comparative features of conventional and emerging catalysts are summarized in Table 1.
Table 1 Comparison of Conventional vs. Emerging catalysts for hydrogen production.
Parameter |
Conventional catalysts (e.g., Ni, Pt, Pd) |
Emerging catalysts (e.g., Biochar-based, Mo₂C, Perovskite, Ni–Fe) |
Active phase |
Ni, Pt, Pd, Ru metals with high intrinsic activity but costly [186,187] |
Transition-metal oxides/carbides and bimetallic systems such as Ni–Fe, Mo₂C, or perovskites showing comparable activity [184,180] |
Feedstock source |
Derived from fossil or mineral ores [186] |
Biomass-derived, renewable, or waste-based precursors such as coconut-shell biochar [181,182] |
Catalytic activity |
High (Pt, Pd), moderate (Ni) but prone to sintering [187] |
Comparable or enhanced when doped or activated biochar is used [15,182] |
Coke resistance |
Moderate to low; Ni-based catalysts easily deactivate by carbon deposition [186] |
Improved through oxygenated or graphitic carbon supports and metal dispersion [185,181] |
Sulfur tolerance |
Low to moderate; sulfur poisons active sites [188] |
Higher tolerance due to surface modification or perovskite lattice flexibility [180,179] |
Thermal stability |
High at first, but sintering occurs at > 800 °C [187] |
Excellent due to carbon–metal or oxide–lattice synergy [184,183] |
Cost and scalability |
Noble metals are expensive and scarce [187] |
Biochar and transition-metal-based catalysts are low-cost and easily scalable [182,181] |
Sustainability / ESG impact |
High environmental footprint, non-renewable feedstocks [186] |
Low carbon footprint, renewable precursors, aligns with circular-economy principles [15,182,179] |
In conclusion, overcoming the technical and sustainability limitations of conventional catalysts is vital to achieving cleaner hydrogen production. The transition toward biochar-based and other emerging catalytic systems aligns directly with global decarbonization and Environmental, Social, and Governance-driven innovation goals, offering a dual advantage of environmental remediation and resource circularity. This paradigm shift from mineral to renewable catalyst design not only addresses technical bottlenecks such as coking and deactivation but also represents a crucial step toward the realization of a sustainable hydrogen economy.
Emergence of biochar as a sustainable alternative
Biochar has emerged as a promising candidate in the quest for sustainable catalytic materials. It is a carbon-rich material produced from the thermochemical conversion of biomass through processes such as pyrolysis, gasification, or torrefaction. Its abundant availability, low cost, and environmental benefits—such as carbon sequestration potential and waste valorization—position biochar as a viable alternative to traditional catalysts. Biochar-based catalysts are considered long-lasting and sustainable because they utilize waste biomass, supporting the circular economy and reducing carbon emissions [14]. These catalysts are cost-effective, have tunable surface chemistry, and exhibit high surface area and porosity, making them suitable for diverse catalytic applications including biodiesel production, syngas generation, and pollutant degradation [15,16].
The preparation method (e.g., pyrolysis or gasification temperature, heating rate, and activation treatment) significantly influences the physicochemical characteristics of biochar-based catalysts, including pore structure, surface functionality, and crystallinity, which in turn determine their catalytic activity and stability [17]. Although biochar-based catalysts demonstrate comparable performance to conventional catalysts in several reactions, challenges remain in optimizing their activity, selectivity, and reusability [17]. Recent studies have explored the use of machine learning and artificial intelligence to predict catalytic behavior and guide the design of high-performance biochar-based catalysts by correlating process parameters and target properties [14]. Future research should therefore focus on improving the structure–property–performance relationships and addressing environmental risks to fully realize the potential of biochar in sustainable catalysis [18].
Nickel-based catalysts are also widely used for hydrogen production from biomass pyrolysis and reforming because of their affordability and strong ability to cleave C–C and C–H bonds [19]. However, these catalysts suffer from coke deposition and deactivation, which are influenced by the type, location, and morphology of carbon species formed during reactions. Consequently, researchers are investigating biochar-supported nickel catalysts, which offer improved stability and lower susceptibility to coking while providing a highly porous carbon framework for Ni particle dispersion [20]. The porous structure, high surface area, and adjustable functional groups of biochar facilitate enhanced bio-oil reforming and tar cracking, while nickel loading further promotes H₂ production. The addition of water vapor also boosts syngas yield through the steam reforming of bio-derived carbon [20]. Novel Ni–biochar composite catalysts with enhanced metal dispersion, activity, and thermal stability are now being developed for reactions such as hydrodeoxygenation of lignin-derived bio-oil, gasification, and methane reforming.
Beyond catalysis, biochar serves as a multifunctional material for environmental and agricultural applications. Due to its adsorptive and ion-exchange properties, biochar can effectively remove heavy metals and organic pollutants from soil and water [21,22]. In agriculture, biochar enhances soil fertility, water retention, and nutrient availability, thereby improving crop yields and promoting sustainable land management [23,24]. Furthermore, biochar contributes to climate change mitigation by sequestering atmospheric carbon and reducing greenhouse gas emissions [23]. Its versatility extends to waste management, as it can be synthesized from a wide range of biomass residues, making it a flexible platform for resource recovery and sustainable material engineering [22].
Structure–Property–Performance relationship
The performance of biochar-based catalysts is governed by the intricate interplay between structure and properties:
Pore structure and surface area affect the diffusion of reactants and the accessibility of active sites, directly influencing catalytic turnover and conversion efficiency.
Surface functional groups (–OH, –COOH, –NH₂, etc.) modulate acid–base characteristics and improve metal anchoring and electron transfer, which enhance reaction selectivity and stability [15,17].
Degree of graphitization determines thermal conductivity and mechanical strength, leading to higher durability under harsh reforming or gasification conditions [20].
Metal dispersion and particle–support interaction govern coke resistance and regeneration ability, where biochar’s carbon matrix minimizes sintering and deactivation [19,20].
This structure–property–performance relationship highlights that the rational design of biochar—through controlled pyrolysis, doping, and activation—can yield tailored catalysts suitable for sustainable hydrogen and syngas production.
Environmental risks and mitigation strategies
Despite its environmental advantages, large-scale deployment of biochar-based catalysts raises several ecotoxicological concerns, particularly related to metal leaching and microchar pollution.
Toxic Metal Leaching: When biochar is impregnated with metals such as Ni, Cu, Fe, or Co, there is a potential risk of metal ion leaching into surrounding ecosystems. Factors such as pH, oxidation state, and production temperature influence the leaching rate. Lower pyrolysis temperatures (≤500 °C) generally lead to higher metal mobility due to incomplete carbonization [188,190]. Prolonged exposure to leached metals may induce aquatic toxicity, soil contamination, and bioaccumulation.
Microchar and Nanoparticle Pollution: Mechanical weathering, erosion, or catalytic abrasion can produce micro- and nano-sized biochar particles (“microchar”), which may persist in the environment. These particles can transport adsorbed contaminants or enter biological systems, leading to potential health risks and ecotoxicity [189].
Mitigation Strategies: Utilize high-temperature pyrolysis (>700 °C) to enhance carbonization and minimize leachable fractions. Apply surface coatings (e.g., oxide layers) to encapsulate metals and prevent release. Conduct life cycle assessment (LCA) to quantify overall environmental benefits versus potential risks [189].
Addressing these concerns through standardized safety protocols, toxicological evaluations, and field-scale testing is essential for the responsible application of biochar in sustainable catalysis.
Scope and objectives of the review
This review aims to provide a comprehensive overview of the current progress and challenges related to biochar-based catalysts for hydrogen production, focusing on their structural, mechanistic, and sustainability aspects. The review is structured to address six interconnected themes, each guided by a central research question (Table 2).
Table 2 Scope and key research questions of the review.
Section |
Focus area |
Guiding research questions |
Intended contribution |
1. Fundamentals of Biochar |
Production processes, structural features, and catalytic suitability. |
How do pyrolysis and gasification parameters influence the surface structure and catalytic activity of biochar? |
Establishes foundational understanding of biochar’s physicochemical and catalytic characteristics. |
2. Mechanistic Insights |
Reaction mechanisms in thermochemical and photochemical hydrogen production. |
What is the role of active sites, metal dispersion, and surface chemistry in enhancing H₂ yield? |
Clarifies how biochar participates in catalytic hydrogen formation pathways. |
3. Activation and Functionalization |
Physical, chemical, and thermal modification strategies. |
How do activation or doping methods (e.g., KOH, metal impregnation) affect hydrogen yield and catalyst stability? |
Identifies optimal modification techniques to enhance catalyst performance. |
4. Efficiency Enhancement Strategies |
Integration with metals, oxides, and reactor engineering. |
How can hybridization and reactor design optimization improve conversion efficiency? |
Links material design with engineering applications for scalable hydrogen production. |
5. Environmental and Economic Implications |
Sustainability, circular economy, and life-cycle impacts. |
What are the environmental and economic trade-offs of biochar-based catalysts compared with conventional systems? |
Assesses feasibility within ESG and carbon-neutral frameworks. |
6. Future Research Directions |
Gaps, emerging trends, and digital approaches. |
How can AI/ML tools accelerate the design of high-performance, sustainable biochar catalysts? |
Provides strategic guidance for future research and innovation. |
This structured framework enables a systematic synthesis of biochar-based hydrogen catalysis by bridging material science, catalytic engineering, and sustainability perspectives. While several recent reviews (2022 - 2024) have summarized catalyst preparation methods and hydrogen yield comparisons, they often treat structural properties, metal modification, and reaction pathways as independent variables. This fragmented approach has led to unresolved inconsistencies, particularly regarding feedstock versus temperature dominance and the relative roles of support effects and intrinsic active sites in metal-modified systems.
The present review advances the field by introducing a unified structure–property–performance framework that explicitly integrates carbon structural evolution, pore hierarchy, surface functionality, and metal–carbon interactions within pathway-specific hydrogen mechanisms. By reorganizing mechanistic understanding into surface activation, metal–carbon synergy, and failure modes, and by translating these insights into a decision-oriented catalyst design matrix with normalized performance metrics, this work moves beyond descriptive aggregation toward mechanistic reconciliation and rational catalyst engineering. In doing so, biochar is repositioned not merely as a low-cost alternative material, but as a tunable catalytic platform governed by interacting structural and stability constraints.
Methodology of literature review
A systematic literature review (SLR) approach was adopted following PRISMA-style principles to ensure transparency and reproducibility. The methodology integrates database selection, inclusion/exclusion screening, data extraction, normalization, and bias mitigation, as illustrated in Figure 1 and Table 3.
Literature search strategy
Searches were conducted in Scopus, Web of Science, ScienceDirect, and SpringerLink databases using combinations of keywords such as “biochar catalyst”, “hydrogen production”, “steam reforming”, “activation”, and “bimetallic Ni–Fe”. Boolean operators (AND, OR) were applied to refine results. Only peer-reviewed journal articles (2010–2025) were considered to ensure scientific rigor and contemporary relevance.
PRISMA-Style flow diagram
The PRISMA-based screening process (Figure 1) summarizes the sequential stages from initial retrieval to final inclusion.
Figure 1 PRISMA-style flowchart of literature selection process.
Figure 1 presents the PRISMA-based systematic selection process used in this review. The flowchart outlines four main stages—Identification, Screening, Eligibility, and Inclusion—that guided the filtering of literature on biochar-based catalysts for hydrogen production. Out of 1,142 initially identified records, duplicates and irrelevant articles were removed, resulting in 78 studies included in qualitative synthesis and 45 in quantitative comparison. Additional procedures such as reference list checking and forward citation tracking were incorporated to strengthen comprehensiveness and minimize publication bias.
Inclusion and exclusion criteria
Table 3 Inclusion and exclusion criteria used for selecting relevant studies in this systematic review. The criteria were designed to ensure scientific rigor, comparability, and relevance to hydrogen production catalysis using biochar-based materials.
Table 3 Inclusion and exclusion criteria for study selection.
Criteria type |
Inclusion criteria |
Exclusion criteria |
Rationale |
Publication Year |
2010 - 2025 |
Before 2010 |
Ensures focus on current technologies and modern analytical tools. |
Study Type |
Experimental, modeling, or LCA studies on biochar catalysts for H₂ production |
Reviews, opinion papers, or unrelated catalytic systems |
Captures original quantitative and mechanistic data. |
Methods |
Studies involving pyrolysis, gasification, reforming, or photochemical hydrogen production |
Studies unrelated to hydrogen generation (e.g., soil, adsorption only) |
Focuses on hydrogen-relevant catalysis. |
Catalyst Parameters |
Reports including surface area, pore volume, functional group, metal loading, and H₂ yield data |
Incomplete catalyst characterization |
Ensures data comparability and performance benchmarking. |
Language |
English |
Non-English |
Guarantees accessibility and standardized terminology. |
Data extraction and normalization
A structured data extraction matrix was employed to systematically capture (i) feedstock type, catalyst preparation and activation strategy, and metal incorporation approach; (ii) reaction conditions including temperature, pressure, and gas composition; and (iii) hydrogen productivity, selectivity, and catalyst stability indicators such as coke resistance.
To ensure cross-study comparability and minimize reporting bias, hydrogen productivity was standardized and primarily expressed as mmol H₂ g⁻¹ catalyst h⁻¹. Surface area and metal loading were converted into consistent units (m² g⁻¹ and wt%, respectively). When performance ranges were reported, mean ± standard deviation (SD) values were calculated where sufficient data were available.
If hydrogen production was originally reported in alternative units (e.g., mol kg⁻¹ biomass, volumetric concentration %, or percentage increase), values were recalculated into mmol H₂ g⁻¹ catalyst h⁻¹ whenever complete experimental parameters permitted conversion. When full normalization was not feasible due to incomplete reporting, original metrics were retained and explicitly identified as secondary descriptors. Gas-phase hydrogen concentration (%) was treated strictly as a compositional parameter rather than a yield metric.
Outliers were verified through cross-referencing original experimental datasets or recalculated using standardized unit conversion equations to maintain methodological consistency. This harmonized normalization framework enables reliable mechanistic comparison across thermochemical, biological, and electrochemical hydrogen pathways.
Bias mitigation and quality control
To reduce bias and enhance reliability:
Publication bias minimized by including both high-impact and regional journals.
Selection bias controlled through double-blind screening by two independent reviewers.
Data heterogeneity addressed via normalization and weighting by study size.
Methodological quality assessed using a modified GRADE (Grading of Recommendations Assessment, Development and Evaluation) scale, emphasizing reproducibility, control experiments, and statistical transparency.
Summary of the review workflow
This systematic methodology ensures comprehensive coverage and methodological rigor. By integrating quantitative normalization, quality assessment, and bias mitigation, the review synthesizes consistent insights on biochar-based catalysts for hydrogen production—linking structural properties, activation techniques, and catalytic performance across diverse studies.
Fundamental properties of biochar
Biochar is a carbon-rich substance that is made from biomass using thermochemical processes. It has gotten a lot of attention as a useful substance that has a lot of potential in catalysis, especially for making hydrogen. Biochar, a carbon-rich product of biomass pyrolysis, exhibits diverse properties depending on feedstock and production conditions. Its fundamental characteristics include carbonaceous form, pore structure, surface chemistry, and mineral content [32]. This section provides a detailed exploration of biochar’s production methods, physicochemical properties, and unique features that make it suitable for catalytic applications.
Biochars usually have nutrients, are alkaline, and have functional groups that contain oxygen that improve the ability to exchange cations [33]. Their high surface area, porosity, and adsorption capacity make them suitable for various applications, including soil improvement and contaminant remediation [34]. There are big differences between commercial biochars in terms of their fixed carbon (0% - 47.8%), volatile matter (28% - 74.1%), ash content (1.5% - 65.7%), and surface area (0.1 - 155.1 g/m²) [35]. Polycyclic aromatic hydrocarbon (PAH) content is affected by the production method; fast pyrolysis produces higher levels than gasification [35]. The fact that biochars have so many different properties shows how important it is to test them first before using them in certain situations and how their properties can be changed to make them work better [36].
Biochar production: Processes
Biochar production involves the thermal decomposition of biomass under limited oxygen conditions. Various technologies are available, including slow pyrolysis, fast pyrolysis, gasification, and torrefaction [34,37]. The choice of feedstock and production method profoundly influences the physicochemical characteristics of the resulting biochar, determining its catalytic properties. The choice of production method significantly influences the biochar's physicochemical properties and yield, with slower heating rates generally producing higher yields [34,38].
Biochar has numerous applications, including soil amendment, carbon sequestration, and greenhouse gas mitigation [34,38]. However, its effectiveness can vary depending on biochar type, application rate, soil type, and crop species [34].
To illustrate the state of research in this area, Table 1 summarizes several critical studies conducted on biochar-based catalysts for hydrogen production. As shown in Table 1, different synthesis and activation methods—such as pyrolysis, hydrothermal carbonization, co-pyrolysis, microwave-assisted synthesis, chemical activation, heteroatom doping, and mechanochemical activation—have been reported to significantly influence catalytic performance. For example, Ni-impregnated biochar from pyrolysis of agricultural waste demonstrated improved hydrogen yield and catalyst stability at higher pyrolysis temperatures [39]. Similarly, hydrothermally carbonized biochar doped with Fe-Ni bimetallic catalysts showed synergistic effects with 30% higher activity compared to mono-metallic systems [20].
Other approaches such as co-pyrolysis [40], microwave-assisted synthesis [18], chemical activation [41], nitrogen doping [42], and magnetic biochar preparation [14] further highlight the versatility of biochar catalysts in different hydrogen production pathways. Advanced methods, including mechanochemical activation [43], feedstock variation [44], and template-assisted synthesis for hierarchical porosity [34], also demonstrate promising outcomes in terms of catalyst stability, selectivity, and hydrogen yield.
Table 4 Critical studies done in the field of biochar-based catalysts in hydrogen production.
Study |
Strategy |
Key mechanistic insight |
Main outcomes |
[39] |
Pyrolysis-derived biochar (400 - 800 °C) with Ni loading |
Higher pyrolysis temperature improved structural stability; optimal 15 wt% Ni balanced dispersion and activity |
Enhanced H₂ yield, improved carbon conversion, increased catalyst stability |
[20] |
Hydrothermal carbonization + Fe–Ni bimetal doping |
Synergistic metal–carbon interaction reduced coking and enhanced redox activity |
~30% higher H₂ production rate, improved longevity |
[44] |
Feedstock-dependent biochar with varying Ni loading |
Biomass chemistry influenced catalytic behavior more than pyrolysis temperature |
Significant variation in H₂ yield and selectivity across feedstocks |
[41] |
KOH activation + bimetal Cu–Zn co-catalyst |
Activation increased porosity and improved metal dispersion; synergy enhanced water splitting |
72% higher H₂ production, improved light absorption and stability |
[42] |
N-doped biochar (metal-free / Fe–N–C system) |
Heteroatom doping created active sites with Pt-comparable HER activity at lower cost |
Reduced HER overpotential, improved exchange current density |
Overall, these studies (Table 4) confirm that optimization of biochar production, activation, and functionalization is crucial for enhancing hydrogen production efficiency. Research has particularly focused on engineering surface properties, optimizing metal loading, implementing heteroatom doping, and developing enhancement strategies such as defect engineering. Furthermore, life cycle analysis, energy evaluations, carbon footprint assessments, and economic viability studies are increasingly being considered to determine the feasibility of biochar-based catalytic systems for real-world hydrogen production. The complex relationships between production technologies, biochar physicochemical properties, and their catalytic performance still require further investigation [34,45].
Pyrolysis
Pyrolysis remains the most widely applied route for producing catalytically active biochar, particularly within the temperature range of 400 - 700 °C relevant to hydrogen reforming processes [46-49]. Rather than the general thermochemical description of pyrolysis, its importance in hydrogen catalysis lies in how temperature governs biochar structure. Moderate temperatures (400 - 600 °C) preserve oxygen-containing functional groups (–COOH, –OH, C=O), which enhance surface reactivity, water–gas interactions, and redox activity [38,50]. Increasing the temperature toward 600 - 700 °C promotes higher surface area, improved pore development, and enhanced aromaticity, contributing to greater thermal stability and resistance to structural collapse during reforming [38,48]. However, excessive carbon ordering at temperatures above ~700 °C reduces functional group density, diminishing surface-mediated hydrogen activation despite improved conductivity [36,51,52]. These trends indicate that pyrolysis temperature controls the balance between surface reactivity and thermal stability, which directly determines hydrogen formation pathways. Therefore, pyrolysis should be understood not as a generic production method, but as a structural tuning tool for optimizing biochar performance in hydrogen-related reactions.
Gasification
Gasification involves the partial oxidation of biomass at elevated temperatures (typically 700 - 1,000 °C), producing syngas and a thermally robust carbon residue [14,53,54]. Compared with pyrolysis, gasification generally yields biochars with greater structural ordering and enhanced thermal stability, owing to the higher reaction temperatures and oxidative environment [55-59]. Although detailed surface area and pore-volume PORE vary across feedstocks (see Supplementary Information) [55-62], gasification-derived biochars consistently exhibit improved resistance to structural degradation under severe reforming conditions. However, the high-temperature environment also reduces oxygen-containing functional groups and surface acidity, leading to lower inherent chemical reactivity compared with pyrolysis-derived biochars [63,64]. This distinction is catalytically significant: while pyrolysis biochars often perform better in low-to-moderate temperature hydrogen pathways due to richer surface functionality, gasification-derived biochars are more suitable for high-temperature reforming systems where structural durability and coke resistance are critical [65-70]. Gasification-derived biochars therefore favor thermal durability over surface reactivity, explaining their inconsistent performance in low-temperature hydrogen pathways but reliable behavior under severe thermochemical reforming conditions.
Hydrothermal Carbonization (HTC)
Hydrothermal carbonization (HTC) converts wet biomass into hydrochar under subcritical water conditions (typically 180 - 250 °C and elevated pressure), offering a low-temperature route for generating chemically functionalized carbon materials [70-75]. In the context of hydrogen catalysis, the significance of HTC lies not in process thermodynamics but in the chemical characteristics of the resulting hydrochar. Compared with high-temperature pyrolysis or gasification, HTC-derived biochars retain a high density of oxygen-containing functional groups, including hydroxyl, carbonyl, and carboxyl moieties, which enhance redox activity, proton transfer, and metal anchoring capacity [71-74]. These features facilitate improved dispersion of catalytically active metals and promote water–gas interactions in reforming or redox-driven hydrogen pathways. However, HTC biochars generally exhibit lower surface area and limited pore development relative to pyrolysis-derived carbons, restricting reactant accessibility and mass transfer [73-76]. As a result, additional activation or post-treatment is often required to achieve competitive catalytic performance. HTC therefore succeeds chemically but fails structurally unless followed by activation, underscoring that its catalytic relevance depends on integrating chemical functionality with engineered porosity for hydrogen-related applications.
Structure property performance relationships of biochar
The catalytic behavior of biochar in hydrogen-related reactions is governed by an interconnected structure–property–performance relationship rather than by isolated physicochemical parameters. The fundamental carbon structure—ranging from amorphous domains to partially graphitized microcrystalline regions—controls both electronic conductivity and thermal stability [92]. Increasing carbon ordering at higher treatment temperatures enhances structural robustness and resistance to sintering, but simultaneously reduces the abundance of surface oxygen functionalities that contribute to catalytic reactivity [36,58,111].
This carbon framework directly determines pore development and surface area. Feedstock type and thermal history influence the formation of micropores, mesopores, and macropores, which regulate mass transport and active-site accessibility [35,88,91]. Moderate carbonization typically produces balanced porosity and surface accessibility, whereas excessive graphitization reduces pore connectivity and surface polarity. As a result, surface area alone does not predict catalytic efficiency; instead, the distribution and accessibility of reactive sites become decisive.
Surface functional groups—including hydroxyl (–OH), carbonyl (–C=O), and carboxyl (–COOH)—further modulate adsorption, redox activity, and metal anchoring capacity [93,94,128,130]. Their concentration decreases with increasing temperature as O/C and H/C ratios decline [44,127], reflecting a shift from chemically reactive to structurally stable carbon matrices. This shift has direct catalytic implications: Oxygen-rich biochars promote water dissociation, redox cycling, and proton transfer in hydrogen evolution pathways, while more graphitized biochars offer improved durability under high-temperature reforming conditions.
Ultimately, hydrogen yield and catalyst longevity emerge from the balance between structural stability and surface reactivity. Biochars with hierarchical porosity and controlled oxygen functionality typically exhibit enhanced hydrogen productivity and reduced deactivation compared with highly ordered but chemically inert carbons [39,110,115]. Therefore, rational catalyst design should not prioritize surface area maximization alone, but rather optimize the coupling between carbon structure, accessible porosity, and functional group chemistry to sustain both activity and stability in hydrogen production systems.
Sustainability and deployment considerations
Beyond catalytic performance, the viability of biochar-based hydrogen systems depends on lifecycle emissions, resource efficiency, economic durability, and scalability. As a biomass-derived material, biochar aligns with circular economy principles and can contribute to carbon-neutral or carbon-negative integration when produced from agricultural or forestry residues [103,104]. Compared with mineral-derived supports, biochar synthesis generally requires lower energy input and generates reduced emissions under optimized pyrolysis conditions [21,39]. However, sustainability outcomes are strongly influenced by activation intensity and metal incorporation strategies, which may increase environmental burden if not carefully controlled.
From a techno-economic perspective, biochar offers substantial cost advantages over noble metal catalysts due to abundant feedstock availability and lower raw material costs [21,109]. Nevertheless, overall system economics are governed primarily by catalyst durability, regeneration efficiency, and resistance to sintering or coke deposition under thermochemical hydrogen conditions [100,102]. Excessive metal loading may enhance short-term activity but compromise long-term viability through accelerated deactivation.
Scalability further depends on reproducible synthesis protocols and feedstock consistency. Variations in biomass composition can alter pore architecture and surface chemistry, affecting pathway-dependent hydrogen performance. Therefore, sustainable deployment of biochar-based hydrogen catalysts requires integrating lifecycle assessment, mechanistic stability, and standardized performance metrics within a unified design framework rather than optimizing individual parameters in isolation.
Mechanistic pathways of biochar in hydrogen production
Biochar-mediated hydrogen production arises from coupled surface-driven, metal-assisted, and stability-dependent mechanisms. Rather than treating adsorption, reforming, and doping as separate phenomena, hydrogen formation should be understood as the result of coordinated interactions between carbon structure, surface chemistry, and metal–support dynamics across thermochemical and biological systems [8,110,111,115].
Surface activation
Surface activation governs the initial interaction between reactants and the carbon matrix. In thermochemical pathways—including methane reforming, biomass pyrolysis, and gasification—biochar facilitates bond cleavage through surface-mediated activation of CH₄, H₂O, and volatile intermediates [7,39,102]. Oxygen-containing functional groups (–OH, –COOH, –C=O) enhance water dissociation and promote water–gas interactions, particularly in moderate-temperature biochars [36,113,128,130]. Increasing carbonization temperature enhances aromatic ordering and thermal stability but reduces surface polarity and oxygen functionality, shifting the balance from chemically reactive to structurally robust carbons [44,111,127].
In fermentation-based hydrogen systems, the mechanism differs fundamentally. Biochar enhances hydrogen production by stabilizing redox-active intermediates, promoting microbial adhesion, and facilitating extracellular electron transfer [110,115]. Hydrogen yield increases (20% - 328%) are attributed not to catalytic cracking but to improved redox mediation and electron shuttling at biochar interfaces [115]. Thus, thermochemical pathways emphasize bond activation and structural resilience, whereas fermentation systems depend on redox-active functional groups and electron-transfer facilitation.
Metal–Carbon synergy
Metal incorporation fundamentally modifies reaction pathways through metal–support interactions. Transition metals such as Ni, Fe, and Co enhance hydrogen production by promoting C–H and C–O bond cleavage and accelerating water–gas shift reactions [19,20,100,102]. Nickel increases reforming kinetics, iron mitigates coke formation and enhances CO conversion, and cobalt improves steam reforming stability [19,20]. The carbon matrix acts as an electronically interactive support, stabilizing dispersed nanoparticles and enabling charge transfer between active sites and reactants.
The effectiveness of metal-assisted catalysis depends critically on dispersion and anchoring strength. Strong metal–oxygen–carbon interactions preserve nanoparticle stability and maintain accessible active sites. Weak interactions promote sintering and active-site loss under reforming conditions [100,102]. Metal oxide doping (e.g., CeO₂, MgO) further modifies oxygen storage capacity and surface basicity, enhancing redox cycling and cracking reactions [133].
In parallel, heteroatom-doped biochars (N-, S-, P-doped) demonstrate that hydrogen evolution can proceed via defect-rich carbon sites without metallic assistance [42,134]. Nitrogen doping increases electron density and modulates adsorption free energy, enabling metal-free hydrogen evolution pathways. This duality—metal-assisted versus metal-free catalysis—highlights the versatility of biochar as both an active catalytic matrix and a tunable support structure.
Distinguishing support effects, active sites, and synergistic interactions
To avoid mechanistic ambiguity, it is essential to distinguish three distinct roles of biochar in hydrogen catalysis. First, support effects refer to the structural and physicochemical contributions of the carbon matrix itself, including pore hierarchy, thermal stability, surface area, and functional-group anchoring capacity [36,92,111,128]. In this role, biochar regulates mass transfer, stabilizes dispersed metal particles, and mitigates sintering without necessarily serving as the primary catalytic center [100,102]. Surface oxygen functionalities further influence adsorption strength and water activation behavior in reforming reactions [113,130].
Second, active-site effects arise from catalytically active species that directly participate in bond activation. In metal-assisted systems, transition metals such as Ni, Fe, and Co function as primary sites for C–H bond cleavage, steam reforming, and water–gas shift reactions [19,20,100]. In metal-free systems, heteroatom-doped carbon defects (e.g., N-doped sites) can act as intrinsic catalytic centers for hydrogen evolution reactions through modulation of adsorption free energy and electron density [42,134]. In both cases, hydrogen formation occurs at these active centers rather than across the entire carbon matrix.
Third, synergistic metal–carbon mechanisms emerge when electronic interactions between metal particles and the carbon framework generate enhanced catalytic behavior beyond individual contributions. Strong metal–oxygen–carbon interactions improve nanoparticle dispersion, suppress coke formation, and modify adsorption energies at the metal interface [19,102,133]. Charge transfer between metal particles and defect-rich carbon domains can alter reaction pathways and turnover frequency, explaining why optimized biochar–metal composites often outperform unsupported metals or pristine biochar alone [39,110,115].
Distinguishing these roles clarifies why surface area enhancement alone does not guarantee catalytic improvement and why metal loading strategies must consider support interaction strength and electronic coupling rather than active-site density alone.
Failure modes and mechanistic constraints
Despite promising hydrogen yields, biochar-based catalytic systems are constrained by structural and chemical degradation mechanisms. In thermochemical reforming, prolonged exposure to temperatures above ~800 °C induces partial graphitization and loss of surface functional groups, reducing redox activity and catalytic turnover [111]. Excessive metal loading accelerates nanoparticle agglomeration, pore blockage, and coke deposition, ultimately decreasing hydrogen selectivity and catalyst lifetime [19,102]. These phenomena explain conflicting reports in which aggressive metal loading improves short-term yield but compromises long-term stability.
In biological systems, surface aging and functional group depletion reduce electron-transfer efficiency, limiting sustained hydrogen enhancement [115]. Similarly, over-activation treatments that maximize surface area may collapse pore networks or eliminate catalytically relevant oxygen species, diminishing performance despite increased BET values.
Collectively, these failure modes demonstrate that hydrogen productivity depends on maintaining equilibrium between surface reactivity, metal dispersion, and structural durability. Strategies that optimize only one parameter—surface area, metal loading, or graphitization—often destabilize the system. Sustainable hydrogen catalysis therefore requires integrated control of carbon structure, functional-group chemistry, and metal–support interactions across operating regimes.
To translate the mechanistic insights discussed above into actionable catalyst design guidance, Table 5 synthesizes the relationships among production route, structural characteristics, surface reactivity, metal modification strategy, and hydrogen pathway suitability. Rather than presenting isolated parameters, the matrix highlights the trade-offs that govern performance and stability across operating regimes.
Table 5 Decision-Matrix for designing biochar catalysts in hydrogen production.
Production route |
Structural characteristics |
Surface reactivity |
Suitable H₂ pathway |
Metal modification strategy |
Strength |
Limitation |
Pyrolysis (400 - 600°C) |
Moderate porosity, oxygen-rich groups |
High |
Fermentation, low–moderate T reforming |
Low–moderate Ni/Fe loading |
High reactivity |
Lower thermal stability |
Pyrolysis (600 - 700°C) |
Increased surface area, partial graphitization |
Balanced |
Steam reforming |
Optimized Ni dispersion |
Good activity–stability balance |
Functional group loss at higher T |
Gasification (700 - 1,000 °C) |
Highly ordered carbon, low functionality |
Low–moderate |
High-T reforming |
Metal stabilization required |
Excellent thermal durability |
Limited low-T reactivity |
HTC (180 - 250 °C) |
Oxygen-rich, low surface area |
High (chemical) |
Redox/biological systems |
Requires post-activation |
Strong metal anchoring |
Poor mass transfer |
Heteroatom-doped biochar |
Defect-rich carbon |
Moderate–High |
Metal-free HER |
N/S/P doping |
Lower cost |
Stability under harsh T |
The decision-matrix underscores that optimal hydrogen performance does not arise from maximizing a single parameter—such as surface area or metal loading—but from balancing structural stability, surface functionality, and metal–carbon interaction strength. This integrative perspective reconciles previously conflicting findings and provides a rational framework for designing robust biochar-based hydrogen catalysts.
Collectively, these insights demonstrate that hydrogen performance over biochar-based catalysts emerges from dynamic coupling between carbon structural evolution, surface functionality, and metal–support interactions rather than from isolated optimization of individual parameters. Apparent contradictions in feedstock selection, temperature control, and metal loading strategies can be reconciled within this unified mechanistic perspective. By integrating structural tuning, pathway-specific functionality, and durability constraints into a single conceptual model, this review establishes a rational foundation for next-generation biochar catalyst engineering.
Unified mechanistic framework of biochar in hydrogen production
Biochar functions as a tunable catalytic platform whose role varies across hydrogen production pathways. To reconcile fragmented interpretations in the literature, we propose a unified mechanistic framework that links structural evolution, surface chemistry, and metal interactions to pathway-dependent hydrogen formation.
At the structural level, feedstock composition and thermal treatment determine carbon ordering, pore hierarchy, and functional group density. These parameters collectively govern surface activation, including reactant adsorption, water dissociation, and redox mediation. In thermochemical pathways (e.g., steam reforming, methane reforming, gasification), biochar primarily acts as a thermally stable, porous support that facilitates bond cleavage and promotes metal-assisted reactions. In contrast, in fermentation-based systems, biochar functions as a redox mediator and electron-transfer facilitator, enhancing microbial hydrogen production through surface functional groups and defect sites.
Metal incorporation introduces an additional mechanistic dimension, where hydrogen formation is governed by synergistic interactions between metal active sites and the carbon matrix. However, excessive graphitization or aggressive metal loading leads to deactivation through sintering, pore blockage, or coke accumulation. Thus, hydrogen yield and stability emerge from balancing structural durability, surface reactivity, and metal–support interaction strength across operating regimes.
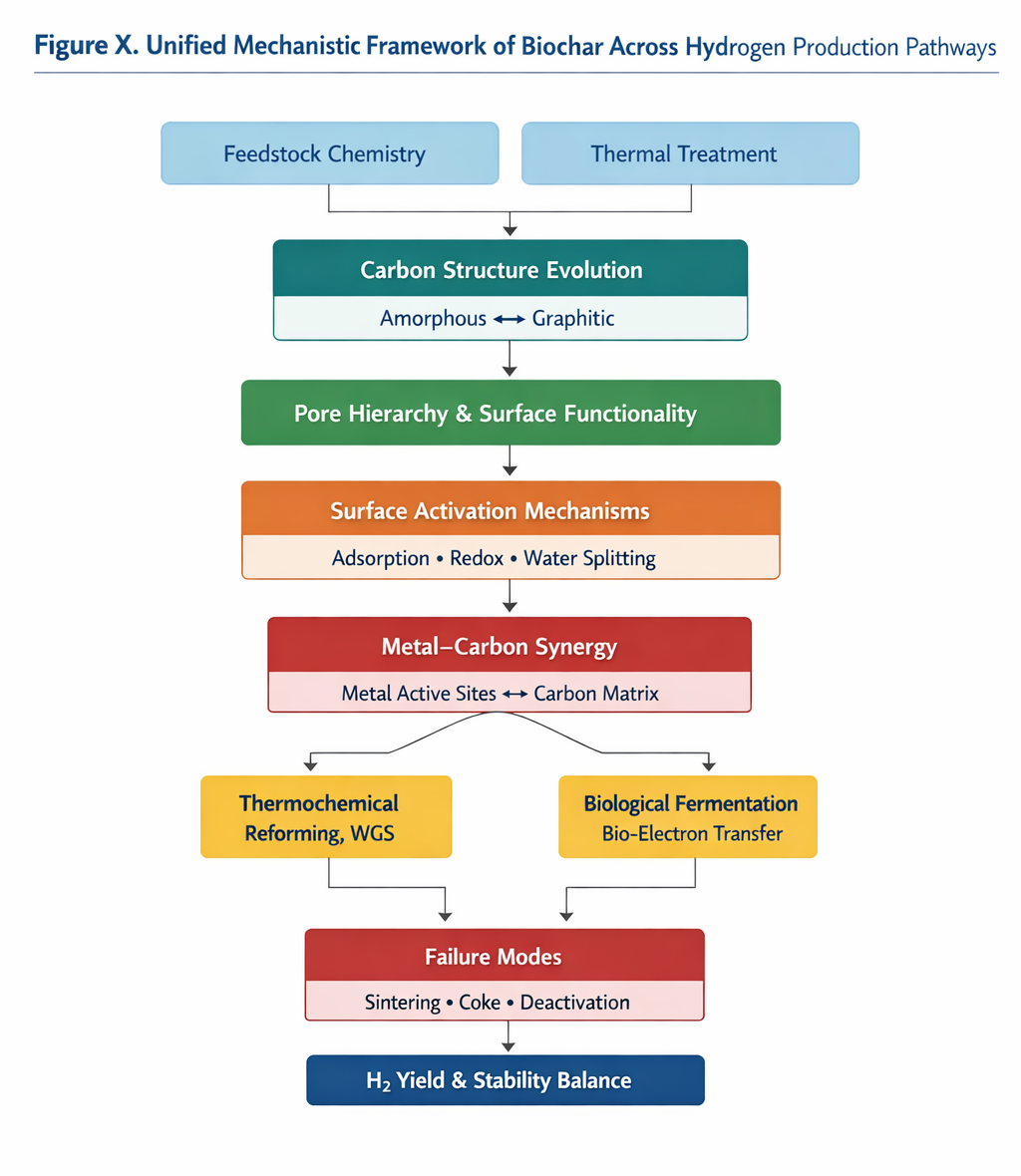
Figure 2 Unified mechanistic framework of biochar across hydrogen production pathways.
The mechanistic integration presented in this section demonstrates that hydrogen performance over biochar-based catalysts cannot be explained by isolated parameters such as surface area, metal loading, or carbonization temperature. Instead, catalytic behavior emerges from coordinated interactions among carbon structural evolution, pore hierarchy, surface functionality, and metal–support synergy under pathway-specific conditions. Conflicting findings in previous literature regarding feedstock dominance, temperature optimization, or metal enhancement can be reconciled when viewed through this unified framework. By explicitly linking structural evolution, metal interactions, and failure modes within a coherent mechanistic model, this review moves beyond cataloging catalyst performance and instead provides a rational roadmap for next-generation biochar catalyst design.
Activation and functionalization of biochar
Activation and functionalization are critical strategies for tailoring biochar properties toward hydrogen catalysis. Through controlled structural modification, activation enhances pore hierarchy, surface chemistry, and metal anchoring capacity, thereby influencing catalytic turnover and long-term stability [69].
Post-activation, biochar typically exhibits significant increases in surface area and pore volume, improving reactant diffusion and accessibility of active sites. Physical activation (e.g., steam or CO₂ treatment) promotes mesopore development and structural robustness, favoring thermochemical hydrogen pathways that require thermal stability and resistance to sintering [11,73]. In contrast, chemical activation (e.g., alkali or acid treatments) introduces abundant surface functionalities and heteroatom doping, enhancing metal dispersion and proton adsorption in hydrogen evolution reactions (HER) [10,147].
However, maximizing surface area alone does not guarantee improved hydrogen performance. Aggressive chemical activation may compromise structural integrity or introduce processing complexity, while excessive thermal treatment can diminish oxygen-containing functional groups essential for redox-mediated hydrogen pathways. Therefore, activation must be aligned with pathway-specific catalytic requirements rather than applied as a universal enhancement strategy.
Advanced approaches such as plasma and electrochemical activation offer localized surface modification with reduced structural damage, potentially improving metal–support interactions and resistance to coke formation. Nevertheless, their scalability and industrial readiness remain under evaluation.
Comparatively, activation strategies differ primarily in their trade-offs between porosity development, structural stability, and environmental footprint. Rather than selecting the most intensive treatment, optimal hydrogen catalyst design requires balancing functionalization efficiency with durability under prolonged operation. Moderate activation combined with controlled metal doping frequently yields more stable hydrogen performance than extreme surface area maximization.
Overall, activation and modification strategies are successful when they enhance accessible active sites without introducing failure modes such as pore collapse, metal sintering, or accelerated coke accumulation. These considerations reinforce that long-term stability, not initial activity alone, must guide rational biochar catalyst design for sustainable hydrogen production.
Efficiency optimization strategies
Efficiency optimization in biochar-based hydrogen systems requires coordinated control of catalyst structure, metal dispersion, reaction conditions, and reactor configuration rather than isolated improvement of individual parameters. Biochar-supported metal catalysts—particularly Ni- and Fe-based systems—have demonstrated enhanced hydrogen productivity across thermochemical and biochemical pathways due to improved metal anchoring, surface reactivity, and electron transfer characteristics [39,115,138].
In thermochemical processes, catalytic performance is strongly influenced by temperature management, pore hierarchy, and resistance to sintering and coke deposition. Optimized graphitic balance and controlled metal loading enhance catalytic turnover while preserving long-term structural integrity. In biological and electrochemical hydrogen pathways, biochar facilitates biofilm formation, redox mediation, and improved charge transfer, supporting stable hydrogen evolution under moderate conditions [110,115].
Hybrid biochar–metal composites further extend performance by integrating waste-derived oxides and multifunctional supports, aligning catalyst design with circular economy principles [10,29]. However, sustained efficiency depends less on peak hydrogen yield and more on durability, regeneration capability, and stability under repeated operation cycles. Reactor-scale considerations including heat transfer, gas–solid interaction, and feedstock consistency also play a decisive role in translating laboratory activity into industrial viability.
Emerging approaches integrating digital process control and machine-learning-assisted optimization offer new opportunities to dynamically adjust operating parameters and predict long-term catalyst behavior. Ultimately, efficiency optimization must balance activity enhancement with structural preservation and techno-economic feasibility, reinforcing the importance of stability-driven catalyst engineering over short-term performance maximization.
Environmental and deployment considerations
Biochar-based catalysts offer environmental advantages in hydrogen production due to their biomass origin, carbon sequestration potential, and reduced reliance on critical raw materials. Life cycle assessments generally indicate lower greenhouse gas emissions and energy intensity compared with mineral- or noble-metal-based catalysts, particularly when waste-derived feedstocks are employed [40,101]. Nevertheless, environmental performance depends strongly on activation intensity, feedstock logistics, and the energy source used during carbonization and reforming. Aggressive chemical treatments or fossil-based process heat may diminish sustainability gains, emphasizing the need for lifecycle-integrated design.
Economically, biochar benefits from feedstock diversity and scalability, with agricultural residues often providing a favorable cost–performance balance. However, techno-economic viability remains highly sensitive to catalyst durability, regeneration efficiency, plant scale, and energy pricing. Reported hydrogen cost projections should therefore be interpreted as scenario-dependent and contingent upon clearly defined boundary conditions. Stability-oriented catalyst engineering and reproducible synthesis protocols are essential to improving long-term cost competitiveness.
Industrial deployment requires coordinated optimization of catalyst structure, reactor configuration, and system integration. Strengthened metal–carbon interfaces, controlled porosity, and digital process monitoring can improve operational durability. Coupling biochar-based catalysts with renewable electricity, carbon management strategies, and circular biomass supply chains enhances their potential within low-carbon energy infrastructures. Ultimately, commercialization will depend on transparent techno-economic evaluation and standardized performance metrics aligned with mechanistic understanding.
Conclusions
This review synthesizes recent advances in biochar-based catalysts for hydrogen production within a unified structure–property–performance perspective. Standardizing hydrogen productivity to mmol H₂ g⁻¹ catalyst h⁻¹ improves cross-study comparability and clarifies inconsistencies arising from heterogeneous reporting metrics. Across thermochemical and biological pathways, hydrogen productivity is governed by coordinated interactions among pore accessibility, surface chemistry, and stabilized metal dispersion.
In biological systems such as dark fermentation, biochar enhances redox mediation and extracellular electron transfer, sustaining normalized hydrogen productivity. In thermochemical processes, particularly steam gasification, engineered biochar–metal interfaces facilitate tar cracking and steam–carbon reactions under elevated temperatures. Importantly, hydrogen concentration in product gas should not be conflated with catalytic productivity, underscoring the need for normalized yield metrics.
Activation and functionalization modulate active-site exposure and durability; however, overly aggressive treatments may accelerate deactivation through sintering or coke formation. Effective catalyst design therefore depends on pathway-aligned structural control rather than surface-area maximization alone.
Beyond catalytic performance, biochar contributes to biomass valorization and reduced reliance on critical raw materials, aligning hydrogen production with circular carbon strategies. Integration with renewable energy systems, carbon management frameworks, and digital optimization tools strengthens its potential for scalable deployment. Continued progress will require durability-oriented design, standardized performance reporting, and coupling of mechanistic insight with techno-economic validation to support industrial translation.
Future research directions
Advancing biochar-based hydrogen catalysis requires a shift from empirical optimization toward mechanism-informed design. Future development should prioritize structural control aligned with pathway-specific hydrogen chemistry rather than generalized surface-area maximization. In thermochemical reforming, hierarchical porosity combined with controlled graphitic ordering is essential to balance mass transport, heat tolerance, and coke resistance. In contrast, biologically mediated hydrogen systems benefit from oxygen-rich surface functionalities that facilitate redox mediation and extracellular electron transfer. Rational catalyst engineering must therefore integrate feedstock chemistry, carbonization parameters, and reaction environment within a unified structure–property–performance framework.
Durability enhancement represents a critical frontier. Strengthening metal–carbon interfaces through defect engineering, heteroatom doping, and controlled anchoring strategies can mitigate nanoparticle sintering and carbon deposition. Optimal performance depends not on maximal metal loading, but on stabilized dispersion and resistance to long-term deactivation. Stability-driven design principles should replace short-term activity maximization as the dominant benchmark for catalyst advancement.
A persistent limitation in current literature is the absence of standardized quantitative descriptors linking structural evolution to hydrogen productivity and catalyst lifetime. Establishing reproducible correlations between parameters such as O/C ratio, defect density, pore hierarchy, and metal dispersion will enable predictive catalyst engineering. Consistent normalization of hydrogen productivity (mmol H₂ g⁻¹ catalyst h⁻¹) is essential to facilitate meta-analytical comparison, reduce reporting bias, and strengthen mechanistic interpretation across hydrogen pathways.
Artificial intelligence and machine learning should transition from exploratory mention to structured implementation. Curated structure–property datasets can support predictive modeling of nonlinear relationships between synthesis variables and catalytic performance. AI-assisted optimization, when coupled with mechanistic understanding, offers the potential to accelerate catalyst discovery, anticipate deactivation pathways, and identify robust synthesis windows while minimizing experimental trial-and-error. Such digital integration may serve as a bridge between laboratory-scale innovation and scalable, durability-oriented hydrogen infrastructure.
Acknowledgements
We would like to acknowledge the Rector of Universitas Negeri Medan (UNIMED), Indonesia for the funding support in this study through the Applied Product Research scheme, under grant number 0148//UN33.8/PPKM/PPT/2025.
Declaration of Generative AI in Scientific Writing
The authors acknowledge the use of generative artificial intelligence (AI) tools, including ChatGPT (OpenAI) and QuillBot, during the preparation of this manuscript. These tools were used solely to assist with language editing, grammar improvement, and clarity of expression. No AI tools were used for data generation, analysis, interpretation of results, or scientific decision-making. All scientific content, interpretations, and conclusions presented in this article were developed and verified by the authors. The authors take full responsibility for the integrity, accuracy, and originality of the work reported in this manuscript.
CRediT Author Statement
Ricky Andi Syahputra: Conceptualization, Methodology, Investigation, Data curation, Writing original draft, Visualization. Muhammad Irvan: Methodology, Supervision, Validation, Writing review and editing. Andriayani Andriayani: Investigation, Data curation, Writing review and editing. Basuki Wirjosentono: Supervision, Resources, Writing review and editing. Karna Wijaya: Validation, Supervision, Writing – review and editing. Shiplu Sarker: Methodology, Validation, Writing – review and editing. Saharman Gea: Conceptualization, Supervision, Project administration, Funding acquisition, Writing review and editing.
References
[1] I Staffell, D Scamman, AV Abad, P Balcombe, PE Dodds, P Ekins, N Shahd and KR Warda. The role of hydrogen and fuel cells in the global energy system. Energy & Environmental Science 2019; 12, 463-491.
[2] KP Tinggi and P Penelitian. Peluncuran program penelitian dan pengabdian kepada masyarakat program prioritas riset dan pengembangan. Sekolah Pascasarjana UPI, Jawa Barat, Indonesia, 2025.
[3] S French. The role of zero and low carbon hydrogen in enabling the energy transition and the path to net zero greenhouse gas emissions with global policies and demonstration projects hydrogen can play a role in a net zero future. Johnson Matthey Technology Review 2020; 64(3), 357-370.
[4] X Cao, S Sun and R Sun. Application of biochar-based catalysts in biomass upgrading: A review. RSC Advances 2017; 7, 48793-48805.
[5] R Angelico, F Giametta, B Bianchi and P Catalano. Green hydrogen for energy transition: A critical perspective. Energies 2025; 18(2), 404.
[6] WB Noor and T Amin. Towards sustainable energy: A comprehensive review on hydrogen integration in renewable energy systems. Future Energy 2024; 3(4), 1-17.
[7] MH Amin. A Mini-Review on CO2 reforming of methane. Progress in Petrochemical Science 2018; 2(2), 161-165.
[8] GU Ingale, K Hyun-Min, S Jeong, D Park, W Kim, B Bang, L Young-Il, SW Kim, K Youn-Bae and U Lee. Assessment of greenhouse gas emissions from hydrogen production processes: Turquoise Hydrogen vs. Steam methane reforming. Energies 2022; 15(22), 8679.
[9] Y Sun, J He, G Yang, G Sun and V Sage. A review of the enhancement of bio-hydrogen generation by chemicals addition. Catalysts 2019; 9(4), 353.
[10] C Dong-Wan, K Yoon, Y Ahn, Y Sun, DCW Tsang, D Hou, YS Ok and H Song. Fabrication and environmental applications of multifunctional mixed metal-biochar composites (MMBC) from red mud and lignin wastes. Journal of Hazardous Materials 2019; 374, 412-419.
[11] F Suárez-García, JI Paredes, M Pérez-Mendoza, J Nauroy, A Martínez-Alonso and JMD Tascón. Porosity development in carbon nanofibers by physical and chemical activation. Journal of Nano Research 2012; 17, 211-227.
[12] J Alvarez, G Valderrama, E Pietri, MJ Pérez-Zurita, CU de Navarro, EF Sousa-Aguiar and MR Goldwasser. Ni-Nb-Based mixed oxides precursors for the dry reforming of methane. Topics in Catalysis 2011; 54, 170-178.
[13] PKR Panyam, B Atwi, F Ziegler, W Frey, M Nowakowski, M Bauer and MR Buchmeiser. Rh(I)/(III)-N-Heterocyclic carbene complexes: Effect of steric confinement upon immobilization on Regio- and Stereoselectivity in the hydrosilylation of alkynes. Chemistry 2021; 27(68), 17220-17229.
[14] M Han, Q Zhao, W Li, P Ciais, W Ying-Ping, DS Goll, L Zhu, Z Zhao, J Wang, Y Wei and F Wu. Global soil organic carbon changes and economic revenues with biochar application. GCB Bioenergy 2022; 14, 364-377.
[15] M Bartoli, M Giorcelli and A Tagliaferro. A comprehensive overview on Biochar-Based materials for catalytic applications. Catalysts 2023; 13(10), 1336.
[16] B Yang, J Dai, Y Zhao, J Wu, C Ji and Y Zhang. Advances in preparation, application in contaminant removal, and environmental risks of biochar-based catalysts: A review. Biochar 2022; 4, 51.
[17] LY Yee, QH Ng, SH Shuit, SKEA Rahim, DM Nawi and SC Low. Application of the novel pH-catalytic-magnetic tri-functionalities augmented bead for removal of organic dye pollutants. Environmental Progress & Sustainable Energy 2021; 40(6), e13699.
[18] H Yang, Y Cui, X Lu, T Han, L Sandström, PG Jönsson and W Yang. Evaluation of engineered Biochar-Based catalysts for syngas production in a biomass pyrolysis and catalytic reforming process. Energy & Fuels 2023; 37(8), 5942-5952.
[19] H Setiabudi, MAA Aziz, S Abdullah, LP Teh and R Jusoh. Hydrogen production from catalytic steam reforming of biomass pyrolysis oil or bio-oil derivatives: A review. International Journal of Hydrogen Energy 2020; 45(36), 18376-18397.
[20] AD Igalavithana, S You, L Zhang, J Shang, J Lehmann, X Wang, Z Yong-Guan, DCW Tsang, P Young-Kwon, D Hou and YS Ok. Progress, Barriers, and Prospects for achieving a ‘hydrogen Society’ and opportunities for biochar technology. ACS ES&T Engineering 2022; 2, 1987-2001.
[21] M Ahmad, AU Rajapaksha, JE Lim, M Zhang, N Bolan, D Mohan, M Vithanage, SS Lee and YS Ok. Biochar as a sorbent for contaminant management in soil and water: A review. Chemosphere 2014; 99, 19-33.
[22] N Khan, P Chowdhary, E Gnansounou and P Chaturvedi. Biochar and environmental sustainability: Emerging trends and techno-economic perspectives. Bioresource Technology 2021; 332, 125102.
[23] S Torabian, R Qin, C Noulas, Y Lu and G Wang. Biochar: An organic amendment to crops and an environmental solution. AIMS Agriculture and Food 2021; 6(1), 401-405.
[24] S Dwibedi, V Pandey and D Divyasree. Biochar: A potential soil ameliorant for sustainable land, agriculture and environmental development. Authorea 2021. https://doi.org/10.22541/au.161884210.06764839/v1
[25] Y Li, M Chen, B Liu, Y Zhang, X Liang and X Xia. Heteroatom doping: An effective way to boost sodium ion storage. Advanced Energy Materials 2020; 10(27), 2000927.
[26] M Patel, AK Chaubey, C Pittman and D Mohan. Aqueous ibuprofen sorption by using activated walnut shell biochar: Process optimization and cost estimation. Environmental Science Advances 2022; 1(4), 530-545.
[27] I Shafiq, S Shafique, P Akhter, W Yang and M Hussain. Recent developments in alumina supported hydrodesulfurization catalysts for the production of sulfur-free refinery products: A technical review. Catalysis Reviews 2022; 64(1), 1-86.
[28] L Fang, T Huang, H Lu, W Xi-Lin, Z Chen, H Yang, S Wang, Z Tang, Z Li, B Hu and X Wang. Biochar-based materials in environmental pollutant elimination, H2 production and CO2 capture applications. Biochar 2023; 5, 42.
[29] R Zhao, B Wang, BKG Theng, P Wu, F Liu, X Lee, M Chen and J Sun. Fabrication and environmental applications of metal-containing solid waste/biochar composites: A review. Science of The Total Environment 2021; 799, 149295.
[30] KN Palansooriya, J Li, PD Dissanayake, M Suvarna, L Li, X Yuan, B Sarkar, DCW Tsang, J Rinklebe, X Wang and YS Ok. Prediction of soil heavy metal immobilization by Biochar using machine learning. Environmental Science & Technology 2022; 56(7), 4187-4198.
[31] Y Liu, X Dai, J Li, S Cheng, J Zhang and Y Ma. Recent progress in TiO2-biochar-based photocatalysts for water contaminants treatment: Strategies to improve photocatalytic performance. RSC Advances 2024; 14(1), 478-491.
[32] B Sajjadi, WY Chen and NO Egiebor. A comprehensive review on physical activation of biochar for energy and environmental applications. Reviews in Chemical Engineering 2019; 35(6), 735-776.
[33] J Yuan and X Renkou. Progress of the research on the properties of biochars and their influence on soil environmental functions. Ecology and Environment 2011; 20(4), 779-785.
[34] D Wang, P Jiang, H Zhang and W Yuan. Biochar production and applications in agro and forestry systems: A review. Science of the Total Environment 2020; 723, 137775.
[35] EN Yargicoglu, BY Sadasivam, KR Reddy and K Spokas. Physical and chemical characterization of waste wood derived biochars. Waste Management 2015; 36, 256-268.
[36] H Lyu, Q Zhang and B Shen. Application of biochar and its composites in catalysis. Chemosphere 2020; 240, 124842.
[37] PD Phadtare and SR Kalbande. Biochar production technologies from agricultural waste, its utilization in agriculture and current global biochar market: A comprehensive review. International Journal of Environment and Climate Change 2022; 12(11), 1010-1031.
[38] F Ronsse, SV Hecke, D Dickinson and W Prins. Production and characterization of slow pyrolysis biochar: Influence of feedstock type and pyrolysis conditions. GCB Bioenergy 2013; 5(2), 104-115.
[39] D Yao, Q Hu, D Wang, H Yang, C Wu, X Wang and H Chen. Hydrogen production from biomass gasification using biochar as a catalyst/support. Bioresource Technology 2016; 216, 159-164.
[40] RS Frazier, E Jin and A Kumar. Life cycle assessment of biochar versus metal catalysts used in syngas cleaning. Energies 2015; 8(1), 621-644.
[41] JL Santos, C Megías-Sayago, S Ivanova, MÁ Centeno and JA Odriozola. Functionalized biochars as supports for Pd/C catalysts for efficient hydrogen production from formic acid. Applied Catalysis B: Environmental 2020; 282, 119615.
[42] O Norouzi, A Kheradmand, Y Jiang, FD Maria and O Masek. Superior activity of metal oxide biochar composite in hydrogen evolution under artificial solar irradiation: A promising alternative to conventional metal-based photocatalysts. International Journal of Hydrogen Energy 2019; 44(54), 28698-28708.
[43] M Farooq, L Romdhane, A Rehman, AKM Al-Alawi, WM Al-Busaidi, SA Asad and L Dong-Jin. Integration of seed priming and biochar application improves drought tolerance in cowpea. Journal of Plant Growth Regulation 2020; 40, 1972-7980.
[44] Z Liu, W Niu, H Chu, T Zhou and Z Niu. Effect of the carbonization temperature on the properties of biochar produced from the pyrolysis of crop residues. BioResources 2018; 13(2), 3429-3446.
[45] S Meyer, B Glaser and P Quicker. Technical, economical, and climate-related aspects of biochar production technologies: A literature review. Environmental Science & Technology 2011; 45(22), 9473-9483.
[46] I Drevin, BL Johansson and EL Son. Pyrolysis in biotechnology. Biotechnology and Genetic Engineering Reviews 2001; 18, 3-28.
[47] A Demirbas. Pyrolysis mechanisms of biomass materials. Energy Sources, Part A: Recovery, Utilization, and Environmental Effects 2009; 31(13), 1186-1193.
[48] A Demirbaş and G Arin. An overview of biomass pyrolysis. Energy Sources 2002; 24(5), 471-482.
[49] JA Ippolito, L Cui, C Kammann, N Wrage-Mönnig, JM Estavillo, T Fuertes-Mendizabal, ML Cayuela, G Sigua, J Novak, K Spokas and N Borchard. Feedstock choice, pyrolysis temperature and type influence biochar characteristics: A comprehensive meta-data analysis review. Biochar 2020; 2, 421-438.
[50] FHPV Velden, GM Kramer, V Frings, IA Nissen, ER Mulder, AJ de Langen, OS Hoekstra, EF Smit and R Boellaard. Repeatability of radiomic features in Non-Small-Cell lung cancer [18F]FDG-PET/CT Studies: Impact of reconstruction and delineation. Molecular Imaging and Biology 2016; 18, 788-795.
[51] JS Tumuluru, S Sokhansanj, CT Wright, RD Boardman and RJ Hess. Review on biomass torrefaction process and product properties and design of moving bed torrefaction system model development. American Society of Agricultural and Biological Engineers 2011. https://doi.org/10.13031/2013.37192
[52] D Nhuchhen, P Basu and B Acharya. A Comprehensive Review on Biomass Torrefaction. International Journal of Renewable Energy & Biofuels 2014; 2014, 506376.
[53] V Soni and V Naik. Gasification - A process for energy recovery and disposal of municipal solid waste. American Journal of Modern Energy 2016; 2(6), 38-42.
[54] G Xu, Y Lv, J Sun, H Shao and L Wei. Recent advances in biochar applications in agricultural soils: Benefits and environmental implications. CLEAN - Soil, Air, Water 2012; 40(10), 1093-1098.
[55] CE Brewer. 2012, Biochar characterization and engineering. Ph. D. Dissertation. Iowa State University, Ames, Iowa.
[56] S You and X Wang. On the carbon abatement potential and economic viability of biochar production systems: Cost-benefit and life cycle assessment. Elsevier Inc., New York, 2018.
[57] Z Qiao, Z Wang, C Zhang, S Yuan, Y Zhu and J Wang. PVAm–PIP/PS composite membrane with high performance for CO2/N2 separation. AIChE Journal 2012; 59(4), 215-228.
[58] X Wang, G Sun, P Routh, DH Kim, W Huang and P Chen. Heteroatom-doped graphene materials: Syntheses, properties and applications. Chemical Society Reviews 2014; 43(20), 7067-7098.
[59] NB Klinghoffer, MJ Castaldi and A Nzihou. Influence of char composition and inorganics on catalytic activity of char from biomass gasification. Fuel 2015; 157, 37-47.
[60] V Hansen, D Müller-Stöver, J Ahrenfeldt, JK Holm, UB Henriksen and H Hauggaard-Nielsen. Gasification biochar as a valuable by-product for carbon sequestration and soil amendment. Biomass and Bioenergy 2015; 72, 300-308.
[61] V Benedetti, F Patuzzi and M Baratieri. Characterization of char from biomass gasification and its similarities with activated carbon in adsorption applications. Applied Energy 2018; 227, 92-99.
[62] Y Wang, Y Hu, X Zhao, S Wang and G Xing. Comparisons of biochar properties from wood material and crop residues at different temperatures and residence times. Energy & Fuels 2013; 27(10), 5890-5899.
[63] L Qian, W Zhang, J Yan, L Han, W Gao, R Liu and M Chen. Effective removal of heavy metal by biochar colloids under different pyrolysis temperatures. Bioresource Technology 2016; 206, 217-224.
[64] T Zhang, Y Zhou, L Li, Y Zhao, MD Felici, RJ Reiter and W Shen. Melatonin protects prepuberal testis from deleterious effects of bisphenol A or diethylhexyl phthalate by preserving H3K9 methylation. Journal of Pineal Research 2018; 65(2), e12497.
[65] J Bastien and C Handler. Hydrogen production from renewable energy sources. IEEE Xplore 2006; 12(7), 1-9.
[66] T Marc, L Sarah, H Samuel, S Thomas, M Shannon, B Kyle, K Norma, M Hari, T Troy, W Mark, H Hannah and B Indrajit. Cost and performance baseline for fossil energy plants volume 1: Bituminous coal and natural gas to electricity. U.S. Department of Energy Office of Scientific and Technical Information 2025. https://doi.org/10.2172/2580491
[67] JS Rhodes and DW Keith. Engineering economic analysis of biomass IGCC with carbon capture and storage. Biomass and Bioenergy 2005; 29(6), 440-450.
[68] JP Ciferno and JJ Marano. Benchmarking biomass gasification technologies for fuels, chemicals and hydrogen production. U.S. Department of Energy National Energy Technology Laboratory, Pennsylvania, 2002.
[69] A Faaij, RV Ree, L Waldheim, E Olsson, A Oudhuis, AV Wijk, C Daey-Ouwens and W Turkenburg. Gasification of biomass wastes and residues for electricity production. Biomass and Bioenergy 1997; 12(6), 387-407.
[70] X Tan, Y Liu, G Zeng, X Wang, X Hu, Yg Gu and Z Yang. Application of biochar for the removal of pollutants from aqueous solutions. Chemosphere 2015; 125, 70-85.
[71] ND Berge, KS Ro, J Mao, JRV Flora, MA Chappell and S Bae. Hydrothermal carbonization of municipal waste streams. Environmental Science & Technology 2011: 45(13), 5696-5703.
[72] Y Zhang, Y Zheng, M Yang and Y Song. Effect of fuel origin on synergy during co-gasification of biomass and coal in CO2. Bioresource Technology 2016: 200, 789-794.
[73] J Mohammed, NS Nasri, MAA Zaini, UH Dadum and MM Ahmed. Comparison on the characteristics of bio-based porous carbons by physical and novel chemical activation. Applied Mechanics and Materials 2014; 554, 22-26.
[74] SJ Puetz. A relational database of global U–Pb ages. Geoscience Frontiers 2018: 9(3), 877-891.
[75] L Fiori, D Basso, D Castello and M Baratieri. Hydrothermal carbonization of biomass: Design of a batch reactor and preliminary experimental results. Chemical Engineering Transactions 2014; 37, 55-60.
[76] A Saba, K McGaughy and MT Reza. Techno-economic assessment of co-hydrothermal carbonization of a coal-Miscanthus blend. Energies 2019; 12(4), 630.
[77] Z Liu, W Demisie and M Zhang. Simulated degradation of biochar and its potential environmental implications. Environmental Pollution 2013: 179, 146-152.
[78] QV Bach and O Skreiberg. Upgrading biomass fuels via wet torrefaction: A review and comparison with dry torrefaction. Renewable and Sustainable Energy Reviews 2016; 54, 665-677.
[79] SK Hoekman, A Broch, L Felix and W Farthing. Hydrothermal carbonization (HTC) of loblolly pine using a continuous, reactive twin-screw extruder. Energy Conversion and Management 2017; 134, 247-259.
[80] JM Coronado, F Fresno, MD Hernández-Alonso and R Portela. Design of advanced photocatalytic materials for energy and environmental applications. Springer London, London, 2013.
[81] K Ghassemi-Golezani and S Rahimzadeh. Biochar modification and application to improve soil fertility and crop productivity. Agriculture 2022; 68(2), 45-61.
[82] D Reißmann, D Thrän and A Bezama. Hydrothermal processes as treatment paths for biogenic residues in Germany: A review of the technology, sustainability and legal aspects. Journal of Cleaner Production 2018; 172, 239-252.
[83] M Heidari, S Salaudeen, A Dutta and B Acharya. Effects of process water recycling and particle sizes on hydrothermal carbonization of biomass. Energy & Fuels 2018; 32(11), 11576-11586.
[84] B Wirth and J Mumme. Anaerobic digestion of waste water from hydrothermal carbonization of corn silage. Applied Bioenergy 2013; 1(1), 1-10.
[85] K Fakkaew, T Koottatep and C Polprasert. Effects of hydrolysis and carbonization reactions on hydrochar production. Bioresource Technology 2015; 192, 328-334.
[86] A Sharma, V Pareek and D Zhang. Biomass pyrolysis - A review of modelling, process parameters and catalytic studies. Renewable and Sustainable Energy Reviews 2015; 50, 1081-1096.
[87] JD Marin-Batista, JA Villamil, JJ Rodriguez, AF Mohedano and MADL Rubia. Valorization of microalgal biomass by hydrothermal carbonization and anaerobic digestion. Bioresource Technology 2019; 274, 395-402.
[88] A A Gryta, K Skic, A Adamczuk, A Skic, M Marciniak, G Józefaciuk and P Boguta. The importance of the targeted design of biochar physicochemical properties in microbial inoculation for improved agricultural productivity—A review. Agriculture 2023; 14(1), 37.
[89] A Tomczyk, Z Sokołowska and P Boguta. Biochar physicochemical properties: Pyrolysis temperature and feedstock kind effects. Reviews in Environmental Science and Biotechnology 2020; 19(1), 191-215.
[90] IS Mohammed, R Na, K Kushima and N Shimizu. Investigating the effect of processing parameters on the products of hydrothermal carbonization of corn stover. Sustainability 2020; 12(12), 5100.
[91] L Leng, Q Xiong, L Yang, H Li, Y Zhou, W Zhang, S Jiang, H Li and H Huang. An overview on engineering the surface area and porosity of biochar. Science of The Total Environment 2021; 763, 144204.
[92] M Mohammadi, SR Shadizadeh, AK Manshad and AH Mohammadi. Experimental study of the relationship between porosity and surface area of carbonate reservoir rocks. Journal of Petroleum Exploration and Production Technology 2020; 10(5), 1817-1834.
[93] J Struyf. Relating functional groups to the periodic table. Journal of Chemical Education 2009; 86(2), 190-193.
[94] N Villanueva-Rosales and M Dumontier. Describing chemical functional groups in OWL-DL for the classification of chemical compounds. CEUR Workshop Proceedings 2007; 258, 22.
[95] V Fung, G Hu, Z Wu and DE Jiang. Hydrogen in nanocatalysis. The Journal of Physical Chemistry Letters 2020; 11(17), 7049-7057.
[96] G Balmuk, M Videgain, JJ Manyà, G Duman and J Yanik. Effects of pyrolysis temperature and pressure on agronomic properties of biochar. Journal of Analytical and Applied Pyrolysis 2023; 169, 105858.
[97] Y Chen, Z Huang, J Yu, H Wang, Y Qin, L Xing and L Du. Research progress of Pt-Based catalysts toward cathodic oxygen reduction reactions for proton exchange membrane fuel cells. Catalysts 2024; 14(9), 569.
[98] Y Han, Q Zeng, Z Geng and Q Zhu. Energy management and optimization modeling based on a novel fuzzy extreme learning machine: Case study of complex petrochemical industries. Energy Conversion and Management 2018; 165, 163-171.
[99] W Xia, A Mahmood, Z Liang, R Zou and S Guo. Earth-Abundant nanomaterials for oxygen reduction. Angewandte Chemie International Edition 2016; 55(8), 2650-2676.
[100] F Cheng and X Li. Preparation and application of biochar-based catalysts for biofuel production. Catalysts 2018; 8(9), 346.
[101] J Lyu, L Niu, F Shen, J Wei, Y Xiang, Z Yu, G Zhang, C Ding, Y Huang and X Li. In situ hydrogen peroxide production for selective oxidation of benzyl alcohol over a Pd@Hierarchical titanium silicalite catalyst. ACS Omega 2020; 5(27), 16865-16874.
[102] N Tejwan, AK Saini, A Sharma, TA Singh, N Kumar and J Das. Metal-doped and hybrid carbon dots: A comprehensive review on their synthesis and biomedical applications. Journal of Controlled Release 2020; 330, 132-150.
[103] MRA Bhuiyan. Overcome the future environmental challenges through sustainable and renewable energy resources. Micro & Nano Letters 2022; 17(14), 402-416.
[104] S Harichandan and SK Kar. An empirical study on technology readiness level of industries to use green hydrogen in India: Role of policy interventions. International Journal of Energy Sector Management 2024; 18(6), 2115-2140.
[105] M Ayaz, D Feizienė, V Tilvikienė, K Akhtar, U Stulpinaitė and R Iqbal. Biochar role in the sustainability of agriculture and environment. Sustainability 2021; 13(3), 21.
[106] JA Ippolito, DA Laird and WJ Busscher. Environmental benefits of biochar. Journal of Environmental Quality2012; 41(4), 967-972.
[107] J Matuštík, T Hnátková and V Kočí. Life cycle assessment of biochar-to-soil systems: A review. Journal of Cleaner Production 2020; 259, 120998.
[108] ES Azzi, E Karltun and C Sundberg. Assessing the diverse environmental effects of biochar systems: An evaluation framework. Journal of Environmental Management 2021; 286, 112154.
[109] K Qian, A Kumar, H Zhang, D Bellmer and R Huhnke. Recent advances in utilization of biochar. Journal of Environmental Management 2015; 42, 1055-1064.
[110] W Li, C Cheng, L He, M Liu, G Cao, S Yang and N Ren. Effects of feedstock and pyrolysis temperature of biochar on promoting hydrogen production of ethanol-type fermentation. Science of The Total Environment 2021; 790, 148206.
[111] Y Chen, X Zhang, W Chen, H Yang and H Chen. The structure evolution of biochar from biomass pyrolysis and its correlation with gas pollutant adsorption performance. Bioresource Technology 2017; 246, 101-109.
[112] L Wu, L Yu, X Xiao, F Zhang, S Song, S Chen and Z Ren. Recent advances in Self-Supported layered double hydroxides for oxygen evolution reaction. Research 2020; 2020, 3976278.
[113] T Chen, R Liu and NR Scott. Characterization of energy carriers obtained from the pyrolysis of white ash, switchgrass and corn stover - Biochar, syngas and bio-oil. Fuel Processing Technology 2016; 142, 124-134.
[114] W Chen, S Shi, T Nguyen, M Chen and X Zhou. Effect of temperature on the evolution of physical structure and chemical properties of bio-char derived from co-pyrolysis of lignin with high-density polyethylene. BioResources 2016; 11(2), 3923-3936.
[115] SK Das, GK Ghosh, RK Avasthe and K Sinha. Compositional heterogeneity of different biochar: Effect of pyrolysis temperature and feedstocks. Journal of Environmental Management 2021; 278(P2), 111501.
[116] JM Coronado and A Bayón. Catalytic enhancement of production of solar thermochemical fuels: Opportunities and limitations. Physical Chemistry Chemical Physics 2023; 25(26), 17092-17106.
[117] SB Bentjen, DA Nelson, BJ Tarasevich and PC Rieke. The introduction of alkyl, ester, carboxylate, amino, hydroxyl, and phosphate functional groups to the surface of polyethylene. Journal of Applied Polymer Science 1992; 44(6), 965-980
[118] W Gadek, M Mlonka-Mȩdrala, M Prestipino, P Evangelopoulos, S Kalisz and W Yang. Gasification and pyrolysis of different biomasses in lab scale system: A comparative study. E3S Web of Conferences 2016; 10, 4-9.
[119] OM Larina, VA Sinelshchikov and GA Sytchev. Comparison of thermal conversion methods of different biomass types into gaseous fuel. Journal of Physics: Conference Series 2016; 774, 012137.
[120] Y Shen, D Ma and X Ge. CO2-looping in biomass pyrolysis or gasification. Sustainable Energy & Fuels 2017; 1(8), 1700-1729.
[121] MH Park, JH Li, A Kumar, G Li and Y Yang. Doping of the metal oxide nanostructure and its influence in organic electronics. Advanced Functional Materials 2009; 19(8), 1241-1246.
[122] TYA Fahmy, Y Fahmy, F Mobarak, M El-Sakhawy and RE Abou-Zeid. Biomass pyrolysis: Past, present, and future. Environment, Development and Sustainability 2020; 22(1), 17-32.
[123] D He, Y Luo and B Zhu. Feedstock and pyrolysis temperature influence biochar properties and its interactions with soil substances: Insights from a DFT calculation. Science of the Total Environment 2024; 922, 171259.
[124] VJ Inglezakis and AA Zorpas. Heat of adsorption, adsorption energy and activation energy in adsorption and ion exchange systems. Desalination and Water Treatment 2012; 39(1-3), 149-157.
[125] MN Alaya, MA Hourieh, AM Youssef and F El-Sejariah. Adsorption properties of activated carbons prepared from olive stones by chemical and physical activation. Adsorption Science & Technology 2000; 18(1), 27-42.
[126] H Kondo, Y Nishida and A Morikawa. Determination and quantitative analysis of surface functional groups of magnetic media. In: Proceedings of the Asia-Pacific Magnetic Recording Conference 2006, Singapore. 2006.
[127] M Ilić, H Franz-Hubert, A Lolić, Z Nedić, T Tosti, IS Ignjatović, A Linden, ND Jablonowski and H Hartmann. Surface functional groups and degree of carbonization of selected chars from different processes and feedstock. PLoS One 2022; 17(11), e0277365.
[128] RC Pereira, MC Arbestain, MV Sueiro and JA MacIá-Agulló. Assessment of the surface chemistry of wood-derived biochars using wet chemistry, Fourier transform infrared spectroscopy and X-ray photoelectron spectroscopy. Soil Research 2015; 53(7), 753-762.
[129] SL McArthur. Thin films of vanadium oxide grown on vanadium metal. Surface and Interface Analysis 2006; 38, 1380-1385.
[130] T Jamieson, E Sager and C Guéguen. Characterization of biochar-derived dissolved organic matter using UV-visible absorption and excitation-emission fluorescence spectroscopies. Chemosphere 2014; 103, 197-204.
[131] FG de Mendonça, IT da Cunha, RR Soares, JC Tristão and RM Lago. Tuning the surface properties of biochar by thermal treatment. Bioresource Technology 2017; 246, 28-33.
[132] D Avnir. Recent progress in the study of molecularly doped metals. Advanced Materials 2018; 30(41), e1706804.
[133] T Gatti, F Lamberti, R Mazzaro, I Kriegel, D Schlettwein, F Enrichi, N Lago, ED Maria, G Meneghesso, A Vomiero and S Gross. Opportunities from doping of Non-Critical metal oxides in last generation Light-Conversion devices. Advanced Energy Materials 2021; 11, 2101041.
[134] JP Paraknowitsch and A Thomas. Doping carbons beyond nitrogen: An overview of advanced heteroatom doped carbons with boron, sulphur and phosphorus for energy applications. Energy & Environmental Science 2013; 6(10), 2839-2855.
[135] X Xing, J Lin, Y Song, Q Hu, Y Zhou and S Mu. Optimization of hydrogen yield of a high-temperature electrolysis system with coordinated temperature and feed factors at various loading conditions: A model-based study. Applied Energy 2018; 232, 368-385.
[136] X Xiao, B Chen, Z Chen, L Zhu and JL Schnoor. Insight into multiple and multilevel structures of biochars and their potential environmental applications: A critical review. Environmental Science & Technology 2018; 52(9), 5027-5047.
[137] MA Murmura, S Cerbelli and MC Annesini. Modelling and optimization of hydrogen yield in membrane steam reforming reactors. The Canadian Journal of Chemical Engineering 2017; 95(9), 1676-1682.
[138] WQ Zeng, LJ Zhu and Q Wang. Steam gasification of biochar derived from fast pyrolysis for hydrogen-rich gas production. Advanced Materials Research 2014; 830, 477-480.
[139] F Niedermann, S Radeschütz and B Mitschang. Deep business optimization: A platform for automated process optimization. Regular Research Papers, Leipzig, 2011, p. 168-180.
[140] L Leng and H Huang. An overview of the effect of pyrolysis process parameters on biochar stability. Bioresource Technology 2018; 270, 627-642.
[141] L Luo, C Xu, Z Chen and S Zhang. Properties of biomass-derived biochars: Combined effects of operating conditions and biomass types. Bioresource Technology 2015; 192, 83-89.
[142] A Al-Rumaihi, M Shahbaz, G Mckay, H Mackey and T Al-Ansari. A review of pyrolysis technologies and feedstock: A blending approach for plastic and biomass towards optimum biochar yield. Renewable and Sustainable Energy Reviews 2022; 167, 112715.
[143] K Tekin, S Karagöz and S Bektaş. A review of hydrothermal biomass processing. Renewable and Sustainable Energy Reviews 2014; 40, 673-687.
[144] M Kumar, X Xiong, Y Sun, IKM Yu, DCW Tsang, D Hou, J Gupta, T Bhaskar and A Pandey. Critical review on Biochar-Supported catalysts for pollutant degradation and sustainable biorefinery. Advanced Sustainable Systems 2020; 4(10), 1900149.
[145] JL Field, CMH Keske, GL Birch, MW Defoort and MF Cotrufo. Distributed biochar and bioenergy coproduction: A regionally specific case study of environmental benefits and economic impacts. GCB Bioenergy 2013; 5(2), 177-191.
[146] L Wang, F Sun, J Gao, X Pi, Z Qu and G Zhao. Adjusting the porosity of Coal-Based activated carbons based on a catalytic physical activation process for gas and liquid adsorption. Energy and Fuels 2018; 32(2), 1255-1264.
[147] I Denmark, A Alam, R Ahsan, F Watanabe, T Viswanathan and N Siraj. Comparative study of chemical activation and physical activation approach to optimize biomass-based doped carbons for energy applications. ECS Journal of Solid State Science and Technology 2024; 13(6), 061003.
[148] HW Lee, YM Kim, S Kim, C Ryu, SH Park and YK Park. Review of the use of activated biochar for energy and environmental applications. Carbon Letters 2018; 26(1), 1-10.
[149] NTL Chi, S Anto, TS Ahamed, SS Kumar, S Shanmugam, MS Samuel, T Mathimani, K Brindhadevi and A Pugazhendhi. A review on biochar production techniques and biochar based catalyst for biofuel production from algae. Fuel 2021; 287, 119411.
[150] YD Chen, R Wang, X Duan, S Wang, NQ Ren and SH Ho. Production, properties, and catalytic applications of sludge derived biochar for environmental remediation. Water Research 2020; 187, 116390.
[151] R Ramos, VK Abdelkader‐fernández, R Matos, AF Peixoto and DM Fernandes. Metal‐Supported biochar catalysts for sustainable biorefinery, electrocatalysis and energy storage applications: A review. Catalysts 2022; 12, 207.
[152] R Wang and P Eng. Low carbon steam Reforming-Based hydrogen production. Gas Liquids Engineering Ltd., Alberta, Canada, 2020.
[153] United States Patent. (12) United states patent field of classification search. United States Patent, Virginia, 2014.
[154] P Zhao, Y Shen, S Ge, Z Chen and K Yoshikawa. Clean solid biofuel production from high moisture content waste biomass employing hydrothermal treatment. Applied Energy 2014; 131, 345-367.
[155] A Kumar, K Saini and T Bhaskar. Advances in design strategies for preparation of biochar based catalytic system for production of high value chemicals. Bioresource Technology 2020; 299, 122564.
[156] E Weidner, E Karbassiyazdi, A Altaee, T Jesionowski and F Ciesielczyk. Hybrid metal Oxide/Biochar materials for wastewater treatment technology: A review. ACS Omega 2022; 7(31), 27062-27078.
[157] NMS Sunyoto, M Zhu, Z Zhang and D Zhang. Effect of biochar addition and temperature on hydrogen production from the first phase of two-phase anaerobic digestion of carbohydrates food waste. Journal of Energy Resources Technology 2018; 140(6), 062204.
[158] D Pierson, N Anderson, J Brewen, N Clark, MC Hardy, D McCollum, FH McCormick, J Morisette, T Nicosia, D Page-Dumroese, C Rodriguez-Franco and J Tirocke. Beyond the basics: A perspective on barriers and opportunities for scaling up biochar production from forest slash. Biochar 2024; 6, 1.
[159] SE Ibitoye, C Loha, RM Mahamood, J Tien-Chien, M Alam, I Sarkar, P Das and ET Akinlabi. An overview of biochar production techniques and application in iron and steel industries. Bioresources and Bioprocessing 2024; 11, 65.
[160] TD Minh, J Song, A Deb, L Cha, V Srivastava and M Sillanpää. Biochar based catalysts for the abatement of emerging pollutants: A review. Chemical Engineering Journal 2019; 394, 124856.
[161] WJ Liu, H Jiang and HQ Yu. Development of Biochar-Based functional materials: Toward a sustainable platform carbon material. Chemical Reviews 2015; 115(22), 12251-12285.
[162] R Heijungs. Ecodesign - Carbon Footprint - Life Cycle Assessment - Life Cycle Sustainability Analysis. A flexible framework for a continuum of tools. Environmental and Climate Technologies 2010; 4(1), 42-46.
[163] T Haeldermans, L Campion, T Kuppens, K Vanreppelen, A Cuypers and S Schreurs. A comparative techno-economic assessment of biochar production from different residue streams using conventional and microwave pyrolysis. Bioresource Technology 2020; 318, 124083.
[164] JM Thomas and KDM Harris. Some of tomorrow’s catalysts for processing renewable and non-renewable feedstocks, diminishing anthropogenic carbon dioxide and increasing the production of energy. Energy & Environmental Science 2016; 9(3), 687-708.
[165] U Buinwi and JA Buinwi. The evolution of trade and industrial policies: Lessons from Cameroon. International Advances in Economic Research 2024; 6(7), 319-339.
[166] H Hirsch-Kreinsen, D Jacobson, S Laestadius, and KH Smith. Low and medium technology industries in the knowledge economy: The analytical issues. Low-tech Innovation in the Knowledge Economy, Frankfurt, Germany, 2023.
[167] J Pellegrin, ML Giorgetti, C Jensen and A Bolognini. EU Industrial policy: Assessment of recent developments and recommendations for future policies. European Parliament, France, 2015.
[168] AK Bhakta, R Fiorenza, K Jlassi, Z Mekhalif, AMA Ali and MM Chehimi. The emerging role of biochar in the carbon materials family for hydrogen production. Chemical Engineering Research and Design 2022; 188, 209-228.
[169] R Shan, J Han, J Gu, H Yuan, B Luo and Y Chen. A review of recent developments in catalytic applications of biochar-based materials. Resources, Conservation and Recycling 2020; 162, 105036.
[170] T Sizmur, T Fresno, G Akgül, H Frost and E Moreno-Jiménez. Biochar modification to enhance sorption of inorganics from water. Bioresource Technology 2017; 246, 34-47.
[171] A Ursúa, LM Gandía and P Sanchis. Hydrogen production from water electrolysis: Current status and future trends. Proceedings of the IEEE 2012; 100(2), 410-426.
[172] M Shakeel, K Mehmood, R Hayat and K Malik. Effect of biochar-based rhizobium biofertilizer on groundnut production. Advances in Agriculture and Biology 2023; 6, 47-53.
[173] P Basu. Biomass gasification and pyrolysis: Practical design and theory. Academic Press, Massachusetts, 2010.
[174] Q Lu, Z Zhang, C Dong and X Zhu. Catalytic reforming of biomass pyrolysis vapors for hydrogen-rich syngas production: A review. Catalysts 2022; 12(2), 215.
[175] TD Minh, J Song, A Deb, L Cha, V Srivastava and M Sillanpaa. Biochar based catalysts for the abatement of emerging pollutants: A review. Chemical Engineering Journal 2020; 394, 124856.
[176] X Zhou, B Liu, R Zhu and R Liu. New notion of biochar: A review on the mechanism of biochar applications in advanced oxidation processes. Chemical Engineering Journal 2021; 416, 129027.
[177] F Amalina, ASA Razak, S Krishnan, AW Zularisam and M Nasrullah. A comprehensive assessment of the method for producing biochar and its potential for soil application: A review. Environmental Technology & Innovation 2022; 27, 102775.
[178] International Energy Agency (IEA), Available at: https://www.iea.org/reports/global-hydrogen-review-2024, accessed October 2024.
[179] AG Al-Gamal, MA El-Sayed and OE Abdelsalam. Perovskite materials for hydrogen evolution: Processes, challenges, and opportunities. International Journal of Hydrogen Energy 2024. https://doi.org/10.1016/j.ijhydene.2024.07.021
[180] AG Georgiadis, ND Charisiou and MA Goula. A mini-review on lanthanum-nickel-based perovskite-derived catalysts for hydrogen production via the dry reforming of methane (DRM). Catalysts 2023; 13(10), 1357.
[181] Y Li, H Liu, W Zhang and J Cao. Catalytic biochar and refuse-derived char for the steam reforming of biomass tar: Towards hydrogen production. Industrial & Engineering Chemistry Research 2023; 62(35), 13514-13527.
[182] R Mishra, H Jena and JN Sahu. Progress and development of biochar as a catalyst for hydrogen production. Journal of Cleaner Production 2024; 456, 143378.
[183] T Wu, C Zhang and Z Zhao. Recent progress on perovskite-based electrocatalysts for CO₂ reduction and hydrogen evolution reactions. Nanomaterials 2023; 13(2), 265.
[184] J Xu and Q Zhu. Transition-metal carbides and nitrides as emerging catalysts for hydrogen production and CO₂ reforming. Applied Catalysis B: Environmental 2022; 307, 121168.
[185] K Harun, MT Rahman and D Rokhsana. Hydrogen production via thermocatalytic decomposition of methane using biochar catalysts. RSC Advances 2020; 10, 44069-44081.
[186] S Wang, Q Zhang and X Hu. Advances in Ni-based catalysts for reforming and decomposition reactions: Coking resistance and sulfur tolerance. Fuel Processing Technology 2023; 245, 107661.
[187] L Zhang, J Chen and Y Huang. Sustainable catalyst design for hydrogen production: From noble metals to earth-abundant alternatives. Energy & Fuels 2022; 36(12), 6697-6712.
[188] T Zhou, H Wang and X Liu. Metal leaching and stability assessment of Fe- and Ni-modified biochars in aquatic systems. Journal of Hazardous Materials 2023; 460, 132541.
[189] X Zhang, L Wu and J Peng. Microchar generation and environmental impacts: Emerging challenges of large-scale biochar application. Science of the Total Environment 2024; 928, 171002.
[190] C Lin, Y Yang and H Sun. Environmental risks of metal-doped biochar: Insights into leaching behavior and risk mitigation. Chemosphere 2024; 353, 142703.