
Trends
Sci.
2026;
23(6):
12319
Alginate-Coated Astaxanthin Liposomes: Enhance Antioxidant Activity and Storage Stability
Lusi Nurdianti1, Nur Rahayuningsih1, Maya Nurul Rahma1,2
Wulan Aprilia Susilawati1, Ardianes Firmansya1 and Fajar Setiawan1,*
1Department of Pharmaceutics, Faculty of Pharmacy, Bakti Tunas Husada University, Tasikmalaya 46115, Indonesia
2Department of Pharmaceutics and Pharmaceutical Technology, Padjadjaran University, West Java 45363, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 17 October 2025, Revised: 13 November 2025, Accepted: 20 November 2025, Published: 5 February 2026
Abstract
Reactive oxygen species (ROS) play a key role in aging and various degenerative diseases such as cardiovascular, neurodegenerative, and chronic inflammatory diseases. Astaxanthin (ATX) has antioxidant activity but is limited in terms of solubility and stability. Liposomes are promising drug carriers but suffer from instability and leakage issues in terms of stability that causes leakage in vesicles and also low entrapment efficiency. This study aims to investigate the potential of a liposome coating system containing astaxanthin using alginate to improve antioxidant activity and stability. Preparation ATX loaded liposomes (ATX-Lip) using thin film hydration and then modified with alginate coating. The formulation was characterized based on particle size, zeta potential, % entrapment efficiency (% EE), and morphology. Furthermore, in vitro release dynamics, antioxidant activity occurred and storage stability test. ATX-Lip with a ratio of soy lecithin: Cholesterol (9:1) gave the best characterization results so that it was continued with coating using alginate. Modification of alginate coating on ATX-Lip (NA-ATX-Lip) successfully increased the zeta potential value from –8.57 ± 0.61 to –44.90 ± 0.1 mV and an increase in %EE with a difference of 9.32%. This is in line with the results of in vitro antioxidant activity where there was a decrease in the IC50 value of ATX by 27.49 to 20.12 ppm in the NA-ATX-Lip formula. This study proves that the modification of the alginate layer on ATX was successful and NA-ATX-Lip was proven to increase antioxidant activity. Surface modification of alginate also enhanced the protective capacity of the system of ATX-Lip, reducing degradation and structural alterations during storage.
Keywords: Astaxanthin, Nanocarrier, Liposome, Coating, Alginate, Antioxidant, Stability, Storage
Introduction
Reactive oxygen species (ROS) play a key role in aging and various degenerative diseases such as cardiovascular, neurodegenerative, and chronic inflammatory diseases. Therefore, the search for natural antioxidants with high radical-neutralizing capacity has become a focus of global research. One of the most potent antioxidants known is astaxanthin (ATX), a lipophilic keto-xanthophyll carotenoid with a long poly-conjugated structure and polar groups at both ends of the molecule, which allow it to interact with lipid layers and biological systems [1-3].
Astaxanthin exhibits significantly stronger free radical scavenging and lipid oxidation inhibition abilities than other carotenoids. Its antioxidant activity include quenching, scavenging reactive oxygen radicals (ROS), and protecting lipid membranes due to the orientation of the ATX molecule in the lipid layer, a mechanism also associated with modulation of antioxidant signaling pathways such as Nrf2/ARE and inhibition of NF-κB [3-5]. Thus, the structural stability of ATX is crucial for the effectiveness of its antioxidant activity.
Despite its great biological potential, the practical application of ATX in pharmaceutical, food, and cosmetic products is limited due to its high chemical instability and very low water solubility [6-9]. ATX is prone to oxidation, cis/trans isomerization, and degradation when exposed to heat, light, oxygen, and variations in pH, leading to a significant reduction in its antioxidant efficacy [7-9].
To overcome these challenges, various encapsulation approaches have been developed, including microcapsulation (using polymers such as alginate, chitosan, or zein), inclusion complexes (cyclodextrins), emulsions, polymeric nanoparticles, and liposomal systems [10-13]. Among these, liposomes are particularly attractive because they mimic biological membranes, protect lipophilic compounds from oxidation, and improve their bioaccessibility [14-15]. Several in vitro studies have confirmed that liposomal ATX shows improved stability, color retention, and cellular uptake [16-18].
However, liposomes still face limitations such as fusion, aggregation, and instability under gastrointestinal conditions, leading to premature leakage of ATX [10-12]. To address this issue, coating the liposome surface with biopolymers like alginate, chitosan, or zein has been explored to improve physicochemical stability and controlled release [19-21]. Among these polymers, alginate is favorable due to its anionic nature and ability to form stable gels with divalent cations (Ca²⁺), which has been well described in structural studies of alginates [22]. However, even without ionic crosslinking, sodium alginate can interact electrostatically with positively charged liposomal surfaces, forming a stable adsorptive coating that enhances colloidal stability, modifies zeta potential, improves encapsulation efficiency, and protects ATX under acidic conditions [12,18,34].
In comparison, chitosan (a polycation) provides mucoadhesive properties and enhances controlled release, while zein (a hydrophobic corn protein) offers excellent water resistance and compatibility with lipophilic molecules. Nevertheless, alginate is more preferred for liposomal coating due to its biocompatibility, low toxicity, and simple gelation process, making it suitable for pharmaceutical and food applications [19].
Although several studies have reported the use of polymeric coatings for liposomes, the effect of alginate film thickness, crosslinking conditions, and ATX: Lipid ratio on antioxidant and isomeric stability has not been fully explained. There is also limited information linking atx chemical stability to its post-encapsulation antioxidant activity in alginate-coated systems.
Despite advances in nanocarrier systems, limited studies have examined the effect of alginate coating on the antioxidant preservation and release dynamics of ATX liposomes. This work is the first to systematically evaluate the impact of alginate surface coating on the antioxidant and storage stability of ATX liposomes. We hypothesized that alginate coating would improve structural stability, maintain antioxidant capacity, and enhance bioaccessibility of ATX. This research aims to provide scientific insight into the application of alginate-coated ATX liposomes as stable antioxidant delivery systems for food, pharmaceutical, and cosmetic uses [6,13].
Materials and methods
Material
Astaxanthin (Fuji Chemical Industries, Japan), soy lecithin, cholesterol (Sigma-Aldrich), sodium hydroxide (NaOH) (Sigma-Aldrich), potassium dihydrogen phosphate (KH2PO4) (Sigma-Aldrich), chloroform (Sigma-Aldrich), Tween 80 (Sigma-Aldrich), methanol (Sigma-Aldrich), sodium alginate (NA) (Sigma-Aldrich).
Method
Manufacturing of astaxanthin liposomes (ATX-Lip)
100 mg total lipid phases (soy lecithin: Cholesterol) 9:1 (F1), 8:2 (F2) and 7:3 (F3) and astaxanthin (ATX) of 30 mg were dissolved in 20 mL of a mixture of methanol and chloroform (1:1) and mixed to form a homogeneous solution. This solution was evaporated using a rotary evaporator at a temperature of 40 °C with a speed of 60 rpm. The film formed in the flask was then stored overnight in a desiccator. The next day, hydration was carried out using a phosphate buffer solution pH 7.4 with the addition of 0.1% tween 80 at a temperature of 60 °C with a speed of 200 rpm for 120 min, then sonication was carried out with a sonicator probe for 30 min using an amplitude of 50% [24].
Preparation of alginate-coated astaxanthin liposomes (NA-ATX-Lip)
Sodium alginate (NA) 0.1 % solution was dissolved in distilled water, then stirred at room temperature for 1 h. Then the optimum ATX-LIP formula was dropped into the NA solution with a volume ratio 1:1 and stirred at low speed at room temperature for 1 h to obtain NA-ATX-Lip. Then sonicated for 15 min and continued with centrifugation of 5,000 rpm for 30 min to separate the alginate residue [18].
Characterization of ATX-Lip and NA-ATX-Lip
Particle size analysis of ATX-Lip and NA-ATX-Lip was performed using PSA (Particle size analyzer). Approximately 1 mL of ATX-Lip or NA-ATX-Lip dissolved in distilled water, then the sample is stored in a flowcell cuvette. Zeta Potential is measured by placing the sample in a cuvette and placed on a zetasizer probe [16]. Morphological observations are tested with a negative staining system of a transmission electron microscope (TEM) set at 200 KV [17]. Determination of % entrapment efficiency (%EE) is carried out by inserting the sample into a viva spin tube, then centrifuged with ultra centrifugation at a temperature of 4 °C and a speed of 12,000 rpm for 1 h. The supernatant is taken to measure the levels of ATX that are not encapsulated by liposomes with a UV Vis spectrophotometer [9]. The calculation of %EE is done using the formula Eq. (1).

Qt is the theoretical amount of ATX added.
Qs is the amount of ATX detected in the supernatant.
In Vitro drug release
A total of 5 mL of sample and powder of ATX, ATX-Lip and NA-ATX-Lip as a comparison, each was placed into a dialysis bag (MWCO 14000) and then placed into the dissolution medium. The temperature was maintained at 37 ± 0.5 °C and stirring was carried out at a speed of 50 rpm. Samples were taken as much as 5 mL at hours 0, 2, 4, 6, 8, 12 and 24 [18].
Antioxidant activity test
The antioxidant activity of ATX-Lip and NA-ATXLip preparations was tested using the DPPH scavenging assay method. Each sample was prepared by diluting it with methanol pro analys in various concentrations of 20, 40, 60, 80, and 100 ppm. The test began with 1 mL of 0.2 mM DPPH in methanol solvent added to 1 mL of the sample. Then, incubation was carried out for 10 min under light-protected conditions, and absorbance was measured at a wavelength (λ) of 517 nm. All tests were carried out in triplicate. Furthermore, serial dilutions of ascorbic acid were also prepared to create a calibration curve. Ascorbic acid (vitamin C) was used as the reference antioxidant for comparison of radical scavenging activity.
Storage stability test
ATX-Lip and NA-ATX-Lip samples were stored at two different temperatures: Room temperature and 4 °C. On day 28, colloidal properties and adsorption efficiency were tested [28].
Data analysis
Data obtained from the characterization and evaluation results are presented as mean ± SD (standard deviation). One-way ANOVA analysis using SPSS software was performed to evaluate differences between groups. Statistical plots were used to study the effect of alginate coating on liposome characterization and evaluation. Results were considered significant if the p-value was < 0.05 [29].
Results and discussion
Manufacturing of astaxanthin liposomes (ATX-Lip)
Figure 1 illustrates the preparation of ATX-Lip using the thin film hydration method. The preparation of liposomes using this method involves 2 main stages, namely the preparation of a thin layer of phospholipids by solvent evaporation followed by a hydration process [30]. In this formula, soy lecithin acts as a phospholipid material and cholesterol acts as a stabilizer in the liposome vesicle system [31]. Astaxanthin, which is lipophilic and soluble in methanol, is combined in the lipid phase in the first stage. To produce a thin film, solvent evaporation is carried out at a temperature of 40 °C, which is the optimal temperature for evaporating organic solvents such as methanol and chloroform [32]. After the thin film dries, the film is left in a desiccator for 1 night to ensure that no organic solvent residue remains. The hydration process the next day was carried out using a phosphate buffer solution of pH 7.4 at a temperature of 60 °C, which is the temperature required for the change from gel phase vesicles to liquid crystal phase which then forms liposome vesicles [33]. The final stage is sonication to form liposomes on a nano scale [34].
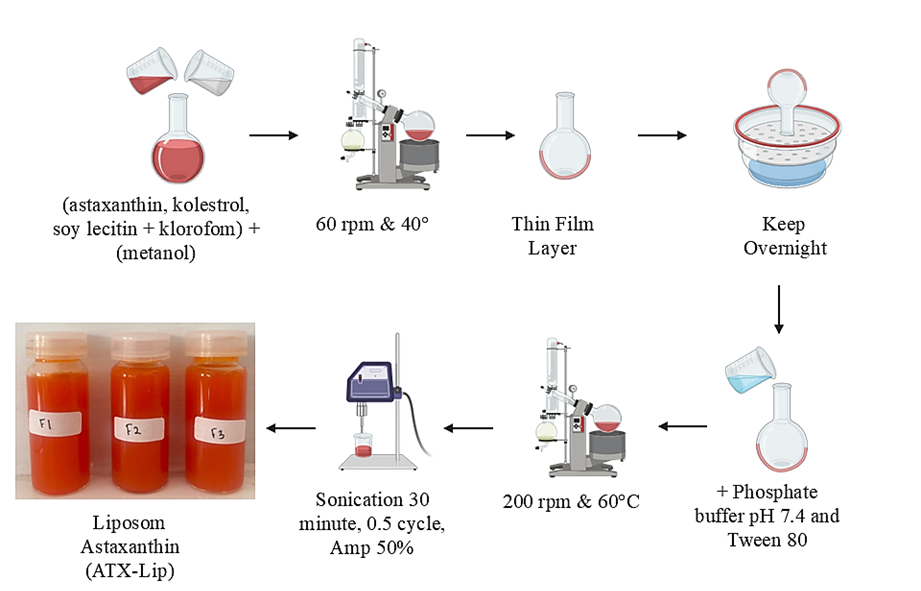
Figure 1 Astaxanthin liposomal formulation procedure.
Preparation of alginate-coated astaxanthin liposomes (NA-ATX-Lip)
Figure 2 illustrates the preparation of NA-ATX-Lip, where the ATX-Lip used is the best formula obtained from the previous procedure steps. Modification of the alginate layer around the liposome surface is known as monolayer modification. The process that occurs when the ATX-Lip suspension is dropped into a sodium alginate solution is polymer adsorption. This system does not require prior alginate functionalization, making it an efficient method of outer layer modification. The interaction between alginate and liposomes, as well as the arrangement of alginate on the ATX-Lip surface, will affect changes in charge, particle size, and stability of the NA-ATX-Lip system [35].
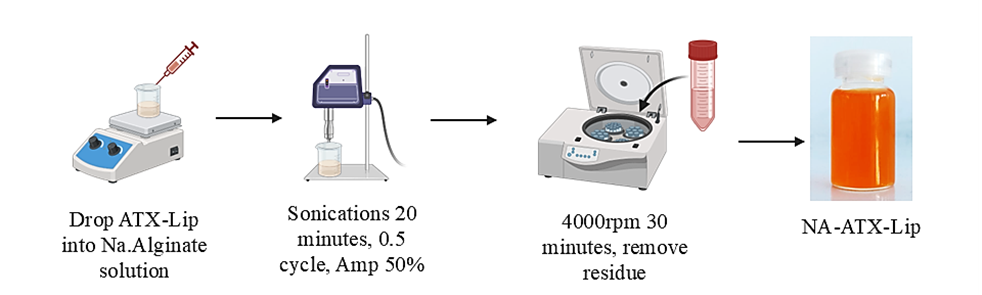
Figure 2 Alginate-coated astaxanthin liposomes formulation procedure.
Characterization of ATX-Lip and NA-ATX-Lip
Table 1 shows the results of the characterization of ATX-Lip with 3 variations of formula, with a total lipid phase of 100 mg and varied into 3 ratios of phospholipids and cholesterol. Overall, all formulas meet the requirements of nano formulations, namely particle sizes ranging from 1 - 1000, PDI values <0.7 and zeta potential values are in the range of > ± 10 mV [26]. However, when viewed from the %EE, the best results are shown in F3, namely 77.77 ± 0.37% with a ratio of soy lecithin: Cholesterol (9: 1). This formula is used as ATX-Lip which will be modified with an alginate layer.
Table 2 shows the changes in the characterization of colloidal properties before and after coating on ATX-Lip. The particle size increased from 48.97 ± 2.86 nm to 73.40 ± 1.36 nm proving that the alginate layer on the ATX-Lip surface was successful, the increase in particle size illustrates the presence of an additional layer of alginate surrounding ATX-Lip through the polymer adsorption mechanism [37]. The zeta potential value also increased from –18.57 ± 0.61 mV to –44.90 ± 0.1 mV. The increase in negative zeta potential after alginate coating can be attributed to the adsorption of –COO⁻ groups on the liposome surface [38]. This increase proves that the presence of an alginate layer can increase the stability of the liposome system by preventing the formation of aggregation between liposome particles and increasing the repulsive force between particles. This occurs with a decrease in the PDI value from 0.6343 ± 0.12 to 0.48 ± 0.01 which illustrates that the system made becomes more homogeneous due to the influence of the increase in the zeta potential value [39]. The %EE value of NA-ATX-Lip was seen to increase from 77.77 ± 0.37% to 87.03 ± 0.01%. This occurs because during the coating process using alginate, the ATX part outside the liposome vesicle was also coated when the suspension was dropped into the polymer solution [40].
From all aspects of characterization, NA-ATX-Lip was proven to be superior to ATX-Lip. Polymer adsorption was chosen over the layer-by-layer technique due to its simplicity, mild processing conditions, and ability to maintain the liposome structure to prevent vesicle leakage. This allows direct electrostatic interaction between the alginate and the liposome surface, resulting in adequate surface stabilization without the need for repeated washing steps required in LbL coating.
To validate the successful formation of the alginate monolayer, morphological analysis was carried out using TEM.
Table 1 Characterization results of particle size, PDI, Zeta potential and % EE of ATX-Lip.
Formula |
PSA value (nm) |
Polydispersity index (PI) |
Zeta potential (mV) |
%EE |
F1 |
48.97 ± 2.86 |
0.6343 ± 0.12 |
–18.57 ± 0.61 |
77.77 ± 0.37 |
F2 |
213.90 ± 9.21 |
0.6940 ± 0.04 |
–17.61 ± 0.24 |
62.89 ± 0.38 |
F3 |
333.37 ± 3.98 |
0.6675 ± 0.04 |
–14.25 ± 0.61 |
56.21 ± 0.55 |
Note: Table 1 presents the characterization of uncoated ATX-Lip formulations (F1 - F3), while Table 2 compares the optimized ATX-Lip formulation with its sodium alginate-coated counterpart (NA-ATX-Lip). Both tables are presented separately to maintain clarity due to different formulation categories.
Table 2 Characterization Results of Particle Size, PDI, Zeta Potential and % EE of NA-ATX-Lip.
Formula |
PSA value (nm) |
Polydispersity index (PI) |
Zeta potential (mV) |
%EE |
ATX-Lip |
48.97 ± 2.86 |
0.6343 ± 0.12 |
-18.57 ± 0.61 |
77.77 ± 0.37 |
NA-ATX-Lip |
73.40 ± 1.36* |
0.48 ± 0.01* |
-44.90 ± 0.1* |
87.03 ± 0.01* |
Note: Data are presented as mean ± standard deviation (n=3); *significance level of p-value < 0.05.

Figure 3 Results of morphological analysis (a) ATX-Lip and (b) NA-ATX-Lip calculated using transmission electron microscopy (TEM).
The structural morphology of the ATX-Lip and NA-ATX-Lip formulas (Figure 3) was described using a transmission electron microscope (TEM). ATX-Lip appeared spherical and distributed in the NA-ATX-Lip monodisperse system, showing a difference compared to ATX-Lip, with the presence of an additional layer indicating the success of the coating process with alginate [41]. The diameter observed through TEM was also close to the diameter results measured using a zeta sizer.
In Vitro drug release
In the release test (Figure 4), unmodified ATX produced the lowest release percentage. This is basically due to the classification of ATX which falls into BCS Class II which has low solubility and high permeability [42]. Meanwhile, ATX modified with liposome nanocarriers showed a much higher release. This increase can be caused by the nanoscale size of the liposome formulation [43]. With a smaller particle size, it will produce a larger surface area and can increase solubility, thereby increasing the permeability of ATX.
The higher release rate of ATX-Lip compared to NA-ATX-Lip is due to the thinner polyelectrolyte layer of ATX-Lip compared to NA-ATX-Lip backcoated with alginate. This thinner layer reduces the diffusion distance of ATX from the liposome core to the substrate, facilitating faster release. NA-ATX-Lip, on the other hand, exhibits a more sustained release profile, most likely due to the presence of a polymer layer surrounding the liposome. This layer acts as a barrier, which gradually releases the drug and supports a controlled release rate in NA-ATX-Lip [44].
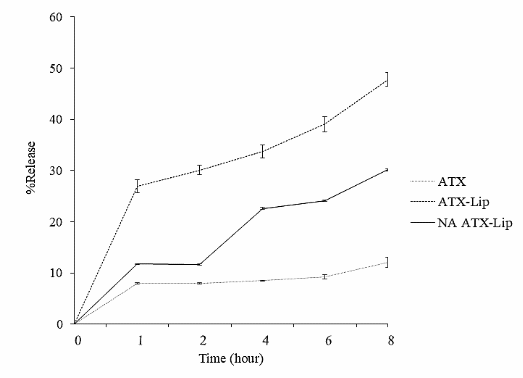
Figure 4 In vitro release of unloaded astaxanthin (ATX), astaxanthin liposomes (ATX-Lip), and alginate-coated astaxanthin liposomes (NA-ATX-Lip) in phosphate buffer (pH 7.4). Data are presented as mean ± standard deviation (n=3).
Table 3 The correlation coefficient of four release kinetic models of ATX, ATX-Lip and NA-ATX-Lip.
Model |
Formula |
||
ATX |
ATX-Lip |
NA-ATX-Lip |
|
Zero-order equation |
0.8161 |
0.9577 |
0.9018 |
First-order equation |
0.8122 |
0.9740 |
0.9257 |
Higuchi equation |
0.7192 |
0.9286 |
0.9717 |
Korsmeyer-Peppas |
0.6058 |
0.8534 |
0.8673 |
As shown in Table 3, ATX demonstrates the best fit with the zero-order model (R² = 0.8162), reflecting a constant linear release pattern, unaffected by the matrix development or polymeric structure processes. ATX-Lip, on the other hand, shows the best fit with the first-order model (R² = 0.9740), indicating that the drug release is governed by diffusion through the lipid matrix due to the microporous structure of the liposomes. For the NA-ATX-Lip formulation, the most appropriate model is the Higuchi equation (R² = 0.9717), which suggests that the release of astaxanthin from NA-ATX-Lip is predominantly driven by diffusion through the sodium alginate matrix, allowing for a more regulated and controlled release [45]. Therefore, the NA-ATX-Lip formulation shows the greatest potential for controlled-release applications, where a more consistent and sustained release is highly desired.
Antioxidant activity test
The IC50 value shown in Figure 5 shows that the best antioxidant activity is produced by NA-ATX-Lip with the smallest IC50 value. The effectiveness of NA-ATX-Lip can be attributed to the gradual release mechanism and increased solubility of ATX by the formation of nano-size. Encapsulation of ATX into liposomes, and followed by alginate coating can protect ATX from degradation [17]. Alginate contains hydroxyl groups (-OH) which can act as electron donors that can neutralize DPPH [46] The increase in antioxidant activity in the NA-ATX-Lip formula is also influenced by improvements in its physicochemical characteristics. A lower PDI value indicates a more uniform particle distribution, resulting in more efficient interaction with free radicals. An increased zeta potential value indicates better colloidal stability due to strong electrostatic repulsion, which helps prevent aggregation and degradation of antioxidants. Furthermore, an increase in entrapment efficiency of 9.32% indicates more astaxanthin is protected within the liposomal vesicles, resulting in higher and more stable antioxidant activity compared to ATX-Lip. Although NA-ATX-Lip exhibited higher IC₅₀ values than ascorbic acid, it demonstrated markedly improved antioxidant activity compared with the uncoated liposomes, indicating that alginate surface modification effectively enhanced the radical scavenging efficiency of astaxanthin.
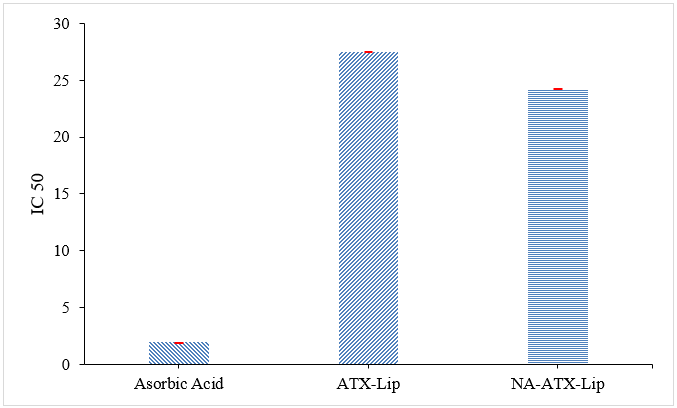
Figure 5 IC50 for antioxidant activity.
Storage stability test
Figure 6 illustrates the stability profiles of ATX-Lip and NA-ATX-Lip formulations, evaluated through various physicochemical parameters over 30 days of storage at room temperature (RT) and 4 °C. The particle size analysis showed that ATX-Lip experienced an increase in diameter after storage, particularly at room temperature, while NA-ATX-Lip remained more stable. Liposome degradation can result from the direct catalytic action of lipase, which hydrolyzes lipid molecules by cleaving the fatty acid chains from the lipid backbone, thereby increasing membrane permeability and causing vesicle leakage [47].
Zeta potential results revealed that both formulations carried negative surface charges, and the values tended to decrease especially under room temperature conditions indicating potential changes in the electrostatic stability of the liposomal systems. The polydispersity index (PDI) remained relatively consistent for both formulations, with only a slight increase observed after storage, particularly at RT. Since the PDI values stayed below 0.7, the liposomal dispersions can be considered to maintain good size uniformity. Meanwhile, the encapsulation efficiency (%EE) of ATX-Lip slightly decreased after 30 days at room temperature but was better preserved at 4 °C. In comparison, NA-ATX-Lip exhibited higher and more stable encapsulation efficiency across both conditions. Overall, these results indicate that storage at 4 °C is more favorable for maintaining the physical and chemical stability of the liposomal formulations. Moreover, the incorporation of alginate appears to enhance the protective capacity of the system, reducing degradation and structural alterations during storage. This is in line with previous studies, alginate-coated liposomes containing collagen peptides also showed increased storage stability within one month [18].
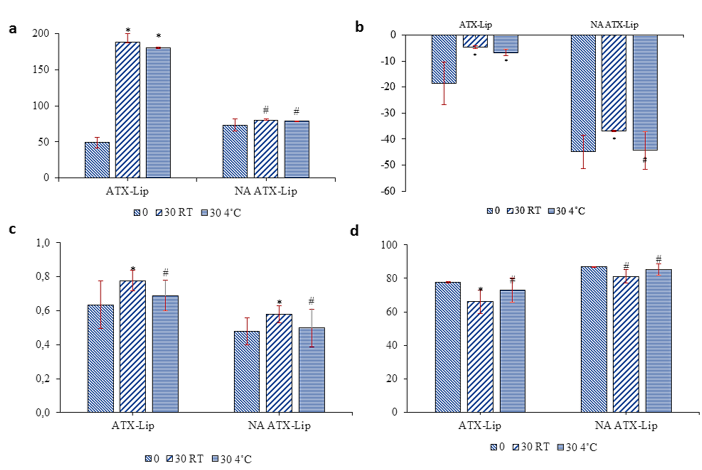
Figure 6 Stability test result of ATX-Lip and NA-ATX-Lip at Room Temperature (RT) and 4 °C for 28 Days (a) Particle Size (b) Zeta Potential (c) Polydispersity Index (d) % EE. Data are presented as mean ± standard deviation (n = 3); * significance level of p-value < 0.05; and # insignificance level of p-value > 0.05 compare with H0.
Conclusions
The present study demonstrated that surface modification of ATX-Lip with alginate (NA-ATX-Lip) effectively improved their physicochemical stability and antioxidant performance. The alginate-coated formulation exhibited an increased zeta potential from −18.57 ± 0.61 mV to −44.90 ± 0.10 mV, indicating enhanced electrostatic stabilization due to the negatively charged –COO⁻ groups of alginate. The encapsulation efficiency also increased from 77.77 ± 0.37% to 87.03 ± 0.01%, confirming stronger drug retention within the vesicles. Additionally, the lower polydispersity index (0.48 ± 0.01) of NA-ATX-Lip reflected improved uniformity and colloidal stability. These improvements correlated with the research objective of enhancing antioxidant stability and storage performance. Overall, alginate surface modification via polymer adsorption provided an efficient approach to strengthen the structural integrity of ATX-Lip during storage at 4 °C. Future studies should investigate the bioavailability and in vivo antioxidant performance of NA-ATX-Lip formulations.
Acknowledgements
This research was funded by the Ministry of Education, Culture, Science, and Technology. Year of 2025 (Grant Number: 0070/C3/AL.04/2025).
Declaration of Generative AI in Scientific Writing
The authors acknowledge the use of generative AI tools (e.g., QuillBot and Chat GPT) in the preparation of this manuscript, specifically for language editing and grammar correction. No content generation or data interpretation was performed by AI. The authors take full responsibility for the content and conclusions of this work.
CRediT Author Statement
Lusi Nurdianti: Conceptualization, Methodology, Software. Nur Rahayuningsih: Project administration. Maya Nurul Rahma: Data curation, Writing Original draft preparation. Wulan Aprilia Susilawati: Visualization, Investigation. Fajar Setiawan: Supervision, Reviewing and Editing. Ardianes Firmansya: Software, Validation.
References
[1] Y Dang, Z Li and F Yu. Recent advances in astaxanthin as an antioxidant in food applications. Antioxidants 2024; 13(7), 879
[2] Y Nishida, PC Berg, B Shakersain, K Hecht, A Takikawa, R Tao, Y Kakuta, C Uragami, H Hashimoto, N Misawa and T Maoka. Astaxanthin: Past, Present, and Future. Marine Drugs 2023; 21(10), 514.
[3] MX Chang and F Xiong. Astaxanthin and its effects in inflammatory responses and inflammation-associated diseases: Recent advances and future directions. Molecules 2020; 25(22), 5342.
[4] Y Ni, M Nagashimada, F Zhuge, L Zhan, N Nagata, A Tsutsui, Y Nakanuma, S Kaneko and T Ota. Astaxanthin prevents and reverses diet-induced insulin resistance and steatohepatitis in mice: A comparison with vitamin E. Scientific Reports 2015; 5, 17192.
[5] J Lee, MH Kim and H Kim. Anti-oxidant and anti-inflammatory effects of astaxanthin on gastrointestinal diseases. International Journal of Molecular Sciences 2022; 23(24), 15471.
[6] L Pan, H Meng, J Li, Z Liu, D Zhang, Z Liu, Q Zhao and F Xu. Enhancement of astaxanthin bioaccessibility by encapsulation in liposomes: An in vitro study. Molecules 2024; 29(8), 1687.
[7] L Pan, S Zhang, K Gu and N Zhang. Preparation of astaxanthin-loaded liposomes: Characterization, storage stability and antioxidant activity. CyTA - Journal Food 2018; 16(1), 607-618.
[8] HI Chang, CW Shao, E Huang and KY Huang. Development of astaxanthin-loaded nanosized liposomal formulation to improve bone health. Pharmaceuticals 2022; 15(4), 490.
[9] B Radwan, A Prabhakaran, S Rocchetti, E Matuszyk, TE Keyes and M Baranska. Uptake and anti-inflammatory effects of liposomal astaxanthin on endothelial cells tracked by Raman and fluorescence imaging. Mikrochimica Acta 2023; 190(8), 332.
[10] R Song, Y Qi, Z Jia, X Liu and R Wei. Astaxanthin-loaded zein/calcium alginate composite microparticles: Characterization, molecular interaction and release kinetics in fatty food simulant system. LWT 2020; 134, 110146.
[11] SF Lin, YC Chen, RN Chen, LC Chen, HO Ho, YH Tsung, MT Sheu and DZ Liu. Improving the stability of astaxanthin by microencapsulation in calcium alginate beads. PLoS One 2016; 11(4), e0153685.
[12] D Pasarin, AI Ghizdareanu, CE Enascuta, CB Matei, C Bilbie, L Paraschiv-Palada and PAVeres. Coating materials to increase the stability of liposomes. Polymers 2023; 15(3), 782.
[13] Z Jafari, A Bigham, S Sadeghi, SM Dehdashti, N Rabiee, A Abedivash, M Bagherzadeh, B Nasseri, H Karimi-Maleh, E Sharifi, RS Varma and P Makvandi. Nanotechnology-abetted astaxanthin formulations in multimodel therapeutic and biomedical applications. Journal of Medicinal Chemistry 2022; 65(1), 2-36.
[14] S Chen, J Wang, J Feng and R Xuan. Research progress of astaxanthin nano-based drug delivery system: Applications, prospects and challenges. Frontiers in Pharmacology 2023; 14, 11102888.
[15] YS Lee, SH Jeon, HJ Ham, HP Lee, MJ Song and JT Hong. Improved anti-inflammatory effects of liposomal astaxanthin on a phthalic anhydride-induced atopic dermatitis model. Frontiers in Immunology 2020; 11, 565285.
[16] Y Xu, Z Jia, J Wang, J Sun and R Song. Property and stability of astaxanthin emulsion based on pickering emulsion templating with zein and sodium alginate as stabilizer. International Journal of Molecular Sciences 2022; 23(16), 9386.
[17] I Panagiotakopoulos and C Nasopoulou. Extraction methods, encapsulation techniques, and health benefits of astaxanthin. Sustainability 2024; 16(24), 10859.
[18] P Wu, L Chen, M Chen, BS Chiou, F Xu, F Liu and F Zhong. Use of sodium alginate coatings to improve bioavailability of liposomes containing DPP-IV inhibitory collagen peptides. Food Chemistry 2023; 414, 135685.
[19] Ó Martínez-Álvarez, MM Calvo and J Gómez-Estaca. Recent advances in astaxanthin micro/nanoencapsulation to improve its stability and functionality as a food ingredient. Marine Drugs 2020; 18(8), 406.
[20] T Taksima, M Limpawattana and W Klaypradit. Astaxanthin encapsulated in beads using ultrasonic atomizer and application in yogurt as evaluated by consumer sensory profile. LWT - Food Science and Technology 2015; 62(1-2), 431-437.
[21] H Goshtasbi, ED Abdolahinia, M Fathi, A Movafeghi, H Omidian, J Barar and Y Omidi. Astaxanthin-loaded alginate-chitosan gel beads activate Nrf2 and pro-apoptotic signalling pathways against oxidative stress. Journal of Microencapsulation 2024; 41(2), 140-156.
[22] R Abka-khajouei, L Tounsi, N Shahabi, AK Patel, S Abdelkafi and P Michaud. Structures, properties and applications of alginates. Marine Drugs 2022; 20(6), 364.
[23] F Yu, J Chen, Z Wei, P Zhu, Q Qing, B Li, H Chen, W Lin, H Yang, Z Qi, X Hong and XD Chen. Preparation of carrier-free astaxanthin nanoparticles with improved antioxidant capacity. Frontiers in Nutrition 2022; 9, 1022323.
[24] E Sulastri, MN Rahma, Y Herdiana, KM Elamin, AFA Mohammed, SA Mahmoud and N Wathoni. Enhancing antifungal efficacy and stability of nystatin liposomes through chitosan and alginate layer-by-layer coating: In vitro studies against candida albicans. International Journal of Nanomedicine 2025; 20, 10739-10750.
[25] A Firmansya, F Setiawan, L Nurdianti and A Yuliana. Formulation and characterization of buccal film nanoemulsion apigenin as antidiabetic. Indonesian Journal of Pharmaceutical Science and Technology 2022; 1(1), 22.
[26] C Suhandi, G Wilar, R Lesmana, F Zulhendri, I Suharyani, N Hasan and N Wathoni. Propolis-based nanostructured lipid carriers for α-mangostin delivery: Formulation, characterization, and in vitro antioxidant activity evaluation. Molecules 2023; 28(16), 6057.
[27] N Dhas, HS Preetha, A Dubey, G Ravi, I Govindan, A Rama, A Naha and S Hebbar. Factorial design-based fabrication of biopolymer-functionalized asiatic acid-embedded liposomes: In-vitro characterization and evaluation. Journal of Applied Pharmaceutical Science 2022; 12(11), 071-081.
[28] X Zhang, Z Wu, W Zhang, L Wang, P Zhao, X Lv, P Guo and Ju Chen. Surface modification by chitosan for improving stability and antioxidative activity of astaxanthin-loaded liposomes. LWT 2024; 198, 116033.
[29] T Milanda, FRC Mo'o, AFA Mohammed, KM Elamin, G Wilar, I Suharyani and N Wathoni. Alginate/chitosan-based hydrogel film containing α-mangostin for recurrent aphthous stomatitis therapy in rats. Pharmaceutics 2022; 14(8), 1709.
[30] H Zhang. Thin-film hydration followed by extrusion method for liposome preparation. Methods in Molecular Biology 2023; 1522, 57-63.
[31] AA Mardhotillah and AA Musfa. Kajian metode dan jenis penyusun sediaan liposom dalam sistem penghantaran obat. Jurnal Riset Farmasi 2023; 3(2), 95-102.
[32] T Lee, AV Sanzogni, PL Burn and AE Mark. Evolution and morphology of thin films formed by solvent evaporation: an organic semiconductor case study. ACS Applied Materials & Interfaces 2020; 12(36), 40548-40557.
[33] D Putri, R Dwiastuti, M Hadimartono and A Nugroho. Optimization of mixing temperature and sonication duration in liposome preparation. Journal of Pharmaceutical Sciences and Community 2017; 14, 79-85.
[34] A Chaerunisaa, MK Dewi, S Sriwidodo, IM Joni and RF Dwiyana. Development of cathelicidin in liposome carrier using thin layer hydration method. International Journal of Applied Pharmaceutics 2022; 14(4), 178-185.
[35] X Tan, Y Liu, X Wu, M Geng and F Teng. Layer-by-layer self-assembled liposomes prepared using sodium alginate and chitosan: Insights into vesicle characteristics and physicochemical stability. Food Hydrocolloids 2024; 149; 109606.
[36] NA Jusril, SIA Bakar, KA Khalil, WMM Saad, NK Wen and MI Adenan. Development and optimization of nanoemulsion from ethanolic extract of centella asiatica (NanoSECA) using d-optimal mixture design to improve blood-brain barrier permeability. Evidence-based Complementary and Alternative Medicine 2022; 2022, 3483511.
[37] F Tello, RN Falfan-Cortés, F Martinez-Bustos, VMD Silva, MD Hubinger and C Grosso. Alginate and pectin-based particles coated with globular proteins: Production, characterization and anti-oxidative properties. Food Hydrocolloids 2015; 43, 670-678.
[38] P Hashemzadeh and M Dinari. Development of alginate/PVP hydrogel incorporating pomegranate seed nanoparticles for selective heavy metal removal. Desalination and Water Treatment 2025; 324, 101520.
[39] X Yi, S Gao, X Gao, X Zhang, G Xia, Z Liu, H Shi and X Shen. Glycolipids improve the stability of liposomes: The perspective of bilayer membrane structure. Food Chemistry 2023; 412, 135517.
[40] Y Gu, Z Zhao, F Xue and Y Zhang. Alginate-chitosan coated nanoliposomes as effective delivery systems for bamboo leaf flavonoids: Characterization in vitro release, skin permeation and anti-senescence activity. Antioxidants, 11(5), 1024.
[41] K Tai, M Rappolt, X He, Y Wei, S Zhu, J Zhang, L Mao, Y Gao and F Yuan. Effect of β-sitosterol on the curcumin-loaded liposomes: Vesicle characteristics, physicochemical stability, in vitro release and bioavailability. Food Chemistry 2019; 293, 92-102.
[42] P Kanaujia, P Poovizhi, WK Ng and RBH Tan. Amorphous formulations for dissolution and bioavailability enhancement of poorly soluble APIs. Powder Technology 2015; 285, 2-15.
[43] HW Chen, SD Chen, HT Wu, CH Cheng, CS Chiou and WT Chen. Improvement in Curcumin’s stability and release by formulation in flexible nano-liposomes. Nanomaterials 2024; 14(22), 1836.
[44] T Ramasamy, ZS Haidar, THiep Tran , JY Choi, JH Jeong, BS Shin, HG Choi, CS Yong and JO Kim. Layer-by-layer assembly of liposomal nanoparticles with PEGylated polyelectrolytes enhances systemic delivery of multiple anticancer drugs. Acta Biomaterialia 2014; 10(12), 5116-5127.
[45] F Tan, H Li, K Zhang, L Xu, D Zhang, Y Han and J Han. Sodium alginate/Chitosan-coated liposomes for oral delivery of hydroxy-α-sanshool: In vitro and in vivo evaluation. Pharmaceutics 2023; 15(7), 2010.
[46] SM Elbayomi, H Wang, TM Tamer and Y You. Enhancement of antioxidant and hydrophobic properties of alginate via aromatic derivatization: Preparation, characterization, and evaluation. Polymers 2021; 13(15), 2575.
[47] MM Virk and E Reimhult. Phospholipase A2-induced degradation and release from lipid-containing polymersomes. Langmuir 2018; 34(1), 395-405.