
Trends
Sci.
2026; 23(6): 12032
Effect of Non-thermal Atmospheric Pressure Plasma Activated Water Against Candida albicans Biofilms on Acrylic Surface
Pattarawan Singkharat1, Chanchai Dechthummarong2,
Chintana
Itthidecharon1
and Phenphichar Wanachantararak3,*,
![]()
1Department of Family and Community Dentistry, Faculty of Dentistry, Chiang Mai University,
Chiang Mai 50200, Thailand
2Department of Electrical Engineering, Faculty of Engineering, Rajamangala University of Technology Lanna,
Chiang Mai University, Chiang Mai 50300, Thailand
3Dentistry Research Center, Faculty of Dentistry, Chiang Mai University, Chiang Mai 50200, Thailand
(*Corresponding author’s e-mail: [email protected])
Received: 24 September 2025, Revised: 29 October 2025, Accepted: 5 November 2025, Published: 10 February 2026
Abstract
Denture stomatitis, often caused by C. albicans biofilm formation on acrylic surfaces, presents a challenge in dental care. This study investigated the optimal conditions for generating non-thermal atmospheric pressure plasma-activated water (PAW) and its antifungal efficacy. Through a series of optimization experiments, the optimal parameters for yielding the highest concentrations of hydrogen peroxide and nitrate were found to be a 500 W of power input and a reduced distilled water volume of 500 mL. In subsequent analysis of PAW’s effect on planktonic C. albicans, a 12-minutes PAW demonstrated yielded the highest percentage of C. albicans inhibition, although it was not significantly different from the inhibition achieved at the 10-minutes PAW. Furthermore, immersing C. albicans biofilms on an acrylic surface for 10 min in 10 and 12-minutes PAW resulted in comparable mean C. albicans inhibition percentages of 82.0 ± 1.61% and 82.0 ± 3.50%, respectively. While the anti-biofilm efficacy of PAW was lower than that of 1% sodium hypochlorite (NaOCl), which was 98.6 ± 1.82% (p < 0.005). The 10 and 12-minutes PAW both markedly reduced biofilm viability across all immersion times with no significant differences observed between them at shorter durations (10, 30 min and 1 h). However, after 8 h of immersion, the 12-minutes PAW demonstrated a significant difference in biofilm inhibition from the 10-minutes PAW (p < 0.05). PAW is a chemically gentler alternative and indicates a promising, non-corrosive approach for future denture cleaning agents. Subsequent investigations should concentrate on augmenting PAWʼs anti-biofilm characteristics and examining its viability for clinical implementation in the prevention and management of denture stomatitis.
Keywords: Non-thermal atmospheric pressure plasma, Plasma activated water, Candida albicans biofilms on acrylic surface
Introduction
Denture stomatitis presents a significant concern in oral health, with global prevalence rates ranging from 20% to 80% [1]. This inflammatory condition, characterized by erythematous tissue under removable dentures, has a complex aetiology, including inadequate denture fit, poor oral hygiene, an immunocompromised host, and colonization by Candida albicans. Current
prevalent management methods for denture stomatitis include adjusting the dentures, patient education on proper oral hygiene, topical antifungal agents, and antimicrobial disinfection of the dentures [2].
Heat-cured acrylic resin, or polymethyl methacrylate (PMMA), is a commonly used denture base material due to its inherent porosity and surface roughness. Research has shown that dental biofilms accumulate more readily on rough denture surfaces than on smooth ones. A study found that acrylic sheets polished with 600-grit sandpaper, which a surface roughness of approximately 0.65 µm, exhibited the highest C. albicans adherence [3]. Sodium hypochlorite is a prevalent disinfectant for dentures immersion due to its broad-spectrum pathogen elimination efficacy. It has demonstrated that a 10-minutes immersion in 1% NaOCl can remove over 90% of biofilm-forming cells [4]. Nevertheless, significant drawbacks such as the corrosion of metallic components in removable partial dentures and potential tissue toxicity necessitate the exploration of alternative antimicrobial treatments. Consequently, research has expanded to include alternative therapeutic modalities for example natural extracts, microwave irradiation, lasers and photodynamic therapy [5-8], and increasingly, non-thermal atmospheric pressure plasma (NTAPP) for microbial decontamination [9-11].
Discovered in 1879, NTAPP is an ionized gas referred to as the fourth state of matter. Unlike the conventional three states of matter (solid, liquid, and gas), plasma is created when a neutral gas is supplied with sufficient energy, such as high thermal energy or intense electromagnetic fields, leading to the ionization of atoms and the liberation of electrons. This produces an ionized gas containing both positive and negative charges [12]. In medicine, plasma can be applied in two ways: Direct exposure, where the plasma plume directly contacts cells and tissues, or indirect exposure. The latter involves generating a liquid medium in front of the plasma plume, resulting in PAW. PAW offers numerous advantages, including flexibility in application, capacity for mass production, and suitability for long-term storage [13]. During PAW production, the plasma plume emits free electrons and energized gas molecules. As these species traverse the atmosphere and interact with water, they induce the formation of various reactive oxygen and nitrogen species (RONS). This includes short-lived, highly reactive species such as hydroxyl radicals (⋅OH), nitric oxide radicals (⋅NO), peroxynitrite (ONOO‒), peroxynitrous acid (ONOOH), nitrous acid (HNO₂), nitric acid (HNO), and ozone (O3). While many reactive species possess extremely short half-lives, typically ranging from milliseconds to several seconds, more persistent and stable RONS, referred to long-lived RONS, such as hydrogen peroxide (H₂O₂), nitrate (NO-3), and nitrite (NO-2), remain in the PAW. The generation and concentration of these RONS are significantly correlated with parameters such as power input, water volume, working gas type, gas flow rate, liquid type, the distance between the plasma plume and the liquid, and activation time. These stable RONS eliminate microorganisms by compromising their cell membranes, altering or obliterating their DNA structure, and disrupting the extracellular matrix within microbial biofilms [14,15].
Numerous studies have demonstrated NTAPP’s efficacy against C. albicans, with both direct and indirect exposure [16-22]. C. albicans yeast in water appeared to be less susceptible to direct plasma, requiring 30 min for complete inactivation [23], whereas PAW could achieve complete inhibition within 1 h when used immediately after production [24]. Currently, research has not concentrated on the effectiveness of PAW against C. albicans biofilms specifically formed on acrylic substrates, which serve as the relevant model for its proposed use as a denture cleansing agent. This study hypothesizes that PAW effectively inhibits C. albicans biofilm on acrylic surfaces, with the potential to alter existing denture maintenance protocols. The implications extend beyond scientific inquiry to clinical application, potentially offering a novel, efficient, and safer approach to preventing and treating denture stomatitis, thereby improving oral health outcomes for the global denture-wearing population.
Materials and methods
Experimental setup for plasma activated water treatment
NTAPP employed in this schematic diagram was designed by the Faculty of Engineering, Rajamangala University of Technology Lanna is shown in Figure 1. The plasma generator consisted of a custom-designed high-voltage (HV) power supply connected to a high voltage electrode in a glass tube, which together generated a plasma discharge directly under the water. The HV power supply was configured with a controlled current of 500 mA to generate an output voltage up to 4 kVp. A stainless-steel disk with a diameter of 50 mm served as the ground electrode and was placed at the bottom of a 1,000 mL beaker. The vertical spacing between the high-voltage electrode and the water surface was precisely maintained at 5 cm. Ambient air, introduced into the glass tube at a flow rate of 3 liter per minute, served as the working gas for PAW production.
The study followed a multi-phase optimization approach. Initially, PAW was generated from 900 mL of distilled water using power input of 400, 500 and 700 W over various activation times (2, 4, 6, 8 and 10 min) to identify the optimal power setting for RONS production. The setting that produced the highest RONS concentration was then selected for next phase. Subsequently, the volume of distilled water was varied to 700 and 500 mL, while maintaining the optimal power setting constant. This was done to determine the water volume that resulted in the highest RONS production. Finally, once both the optimal power input and water volume were identified, extended plasma activation time were explored to further enhance RONS concentrations in the PAW.
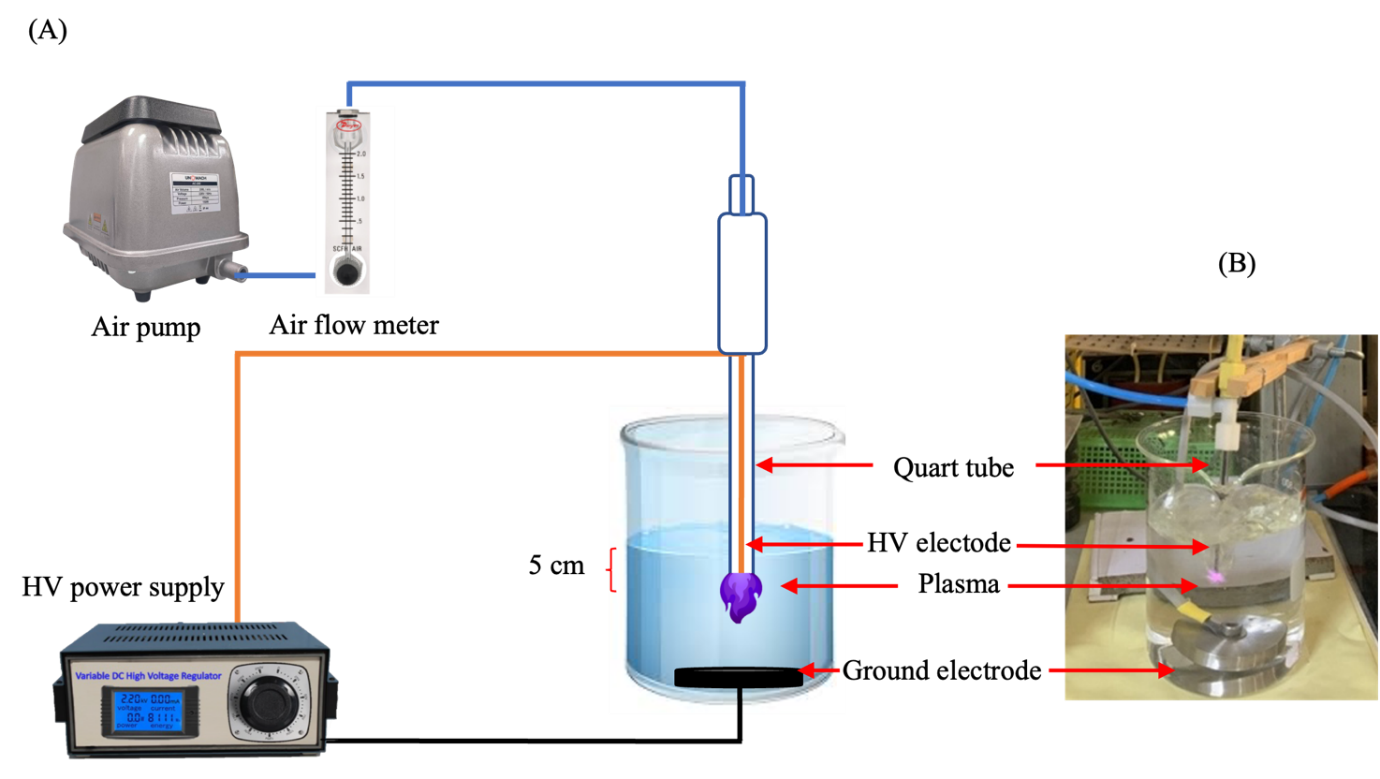
Figure 1 Schematic diagram showing the non-thermal atmospheric pressure plasma configuration (A) and (B) Illustration of PAW generators.
Reactive species concentration measurements
The concentration of long-lived RONS in PAW was determined using analytical methods. H₂O₂ concentrations were quantified using titanium oxysulfate colorimetry [25] (Sigma-Aldrich, Germany), where the reaction of H₂O₂ with titanium oxysulfate forms a yellow peroxotitanium complex. The absorbance of this complex was then measured spectrophotometrically at 407 nm, and concentrations were estimated using a standard calibration curve. (NO-3) concentrations were determined using a calibrated portable nitrate photometer [26] (HI 96786C, Hanna instrument, USA) via cadmium reduction method, followed by a Griess reaction. Meanwhile, (NO-2) concentrations were assessed using the Griess reagent method [27,28] (Thermo Fisher Scientific, USA) with the resulting azo compound measured spectrophotometrically at 540 nm for concentrations calculation using a standard calibration curve. The pH was measured using a pH meter (Hanna instrument, USA).
Effect of PAW on planktonic C. albicans
A reference strain of C. albicans ATCC 10231 (National Science and Technology Development Agency, Thailand) was cultured in Sabouraud Dextrose Broth (SDB; Merck, Germany) at 37 °C for 24 h. The PAW was assessed against C. albicans using microplate assay. The culture turbidity was then adjusted to 0.5 McFarland standard (approximately 1×106 CFU/mL) using spectrophotometer (sunrise TECAN, Austria) at 540 nm. A 10⁻² dilution of the C. albicans suspension was prepared, and 100 µL of this diluted suspension was added to each well of 96-well microplates. The experiment included six groups in triplicate: a no-treatment distilled water group (control) and five groups receiving of 200 µL 2, 4, 6, 8, and 10-minutes PAW, respectively. After incubation at 37 °C for 16 h, cell density was quantified by measuring optical density using spectrophotometer. The reduction of fungal concentration suspension was calculated using the formula [29]:
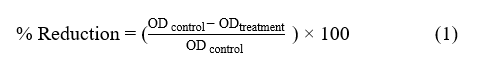
Where ODcontrol represents the optical density of the a no-treatment distilled water group ODtreatment represents the optical density of PAW groups.
Antifungal activity against C. albicans biofilms on acrylic surface
Cylindrical heat-cured acrylic plates (10 mm diameter×2 mm thickness) as shown in Figure 2 were prepared following the manufacturer’s instructions, with residual monomers removed by immersion in water at 37 °C for 48 h. Surface standardization was achieved using a metallographic polisher (MoPao 160E, China) with 600-grit wet sandpaper for 1 min per side. The average surface roughness (Ra) was quantified using a profilometer (Mitutoyo, Japan), and only plates with an average Ra between 0.6 to 0.9 µm were selected [30]. The plates were then sterilized with ethylene oxide gas [31]. C. albicans biofilm formation was initiated on the sterile plates. The sterilized filtered artificial saliva (Faculty of Dentistry, Chiang Mai University) was verified via culture testing. Initially, acrylic plates were exposed to 1 mL artificial saliva per well in 24-well plates at 37 °C for 2 h to facilitate pellicle formation. After washing twice with phosphate-buffered saline (PBS), a standardized C. albicans culture was introduced (1 mL/well) and incubated at 37 °C for 90 min to enable initial cellular adhesion. Non-adherent cells were removed via a twice PBS wash, and specimens were transferred to new 24-well plates containing fresh culture medium. To ensure mature biofilm formation, the culture medium was replaced at 24-hour intervals throughout a 48-hours maturation period, resulting in the establishment of a robust C. albicans biofilm.
Following biofilm maturation, specimens were subjected to antifungal treatment. The existing culture medium in each well was removed and replaced with 1 mL of PAW, which was generated using the optimal activation time determined from prior experimental. The specimens were divided into six groups (n = 3 per group); four PAW-treated groups exposed for varying exposure durations (10, 30 min, 1 and 8 h), 10-minutes immersion in 1% sodium hypochlorite group and a no-treatment distilled water (control) in each exposure durations.
After the designated immersion periods, the specimens were processed for colony enumeration to quantify biofilm efficacy. Each acrylic plate was transferred to a microcentrifuge tube containing 1 mL of culture medium, and the biofilms were dislodged via 5-minutes ultrasonic vibration (Biosonic UC125, USA). The resulting suspension was diluted to 10⁻² concentration, and 100 µL was plated in triplicate on Sabouraud Dextrose Agar (SDA; Merck, Germany). The plates were incubated at 37 °C for 24 h before the colonies were counted.
The percentage of biofilm inhibition was calculated using the formula:

Where Colonycontrol represents the colony count from no-treatment distilled water group and Colonyexperimental represents the colony count from PAW-treated groups or sodium hypochlorite-treated group.

Figure 2 Acrylic surface after polishing.
Statistical analysis
The data were shown as mean ± standard deviation (SD). The statistical analysis of data was performed by GraphPad Prism version 10. Normal distribution of data was tested by Shapiro-Wilk test. One-way and two-way analysis of variance (ANOVA) were used followed by Turkey’s multiple comparisons at a confidence level of 0.05, 0.01, and 0.005.
Results and discussion
Reactive species concentration and pH measurements
A plasma generator was used to generate PAW from 900 mL of distilled water, using a constant current of 500 mA, a potential difference of 4 kVp, and a gas flow rate of 3 liter per. PAW was generated with power input settings of 400, 500 and 700 W for activation times of 2, 4, 6, 8 and 10 min. The concentrations of hydrogen peroxide, nitrate, and nitrite, along with the pH, were measured for each condition. The findings demonstrated that the RONS concentrations varied significantly with both power input and activation time. Hydrogen peroxide concentrations consistently increased over times as shown in Figure 3(A), while nitrate levels initially rose, then slightly decreased before gradually increasing as shown in Figure 3(B). Nitrite concentrations were initially low and became undetectable thereafter as shown in Figure 3(C). Significant differences in pH were also observed at all power levels when comparing the 2-minutes PAW to the longer activation periods as shown in Figure 3(D).
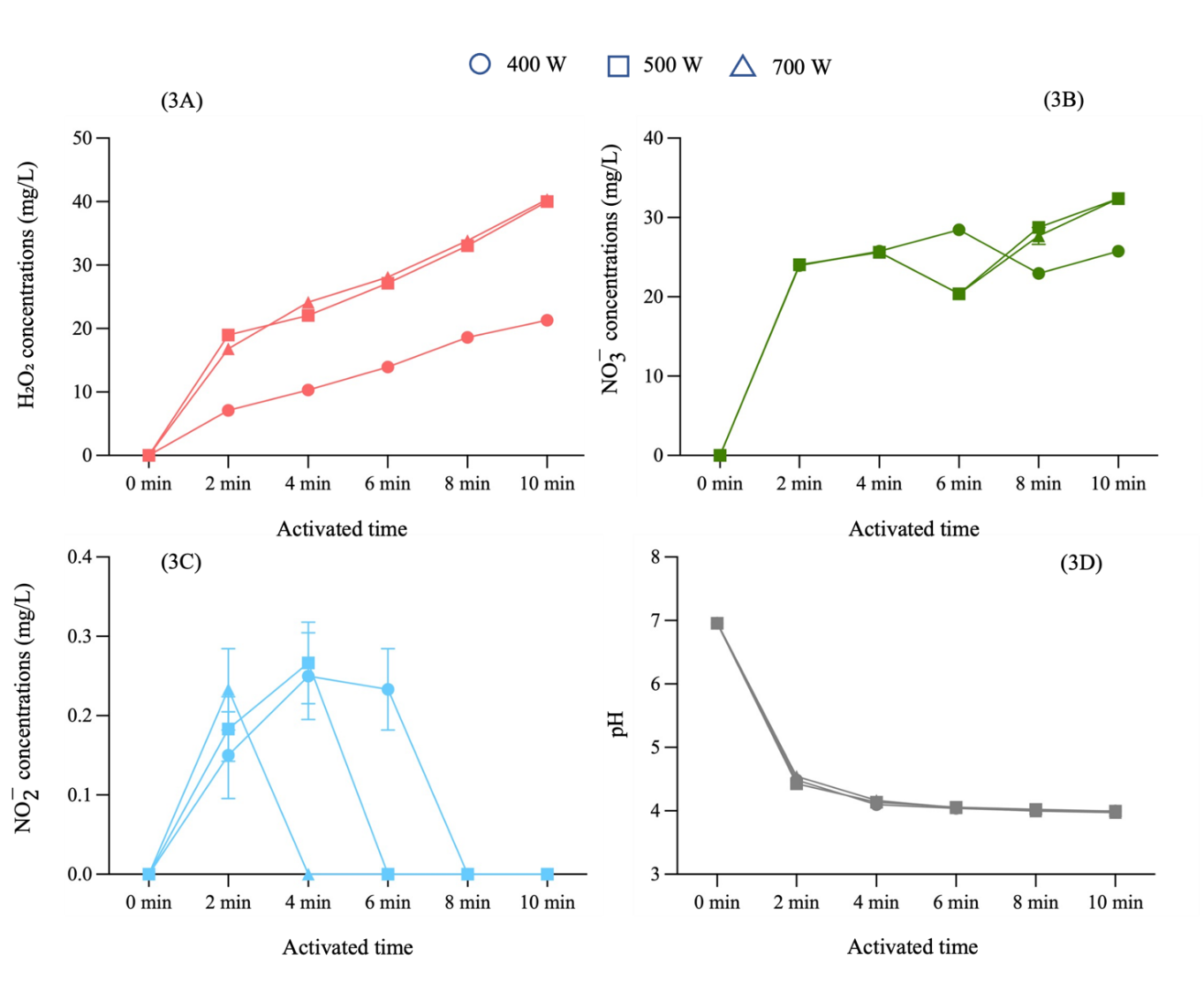
Figure 3 The concentrations of hydrogen peroxide, nitrate, nitrite and pH in PAW, with different power input settings at various activation times.
To optimize RONS production, we selected the 500 W power setting for subsequent experiments, as it yielded significant quantities of hydrogen peroxide and nitrate comparable to the 700 W setting, thus offering greater energy efficiency. We then varied the volume of distilled water to 700 and 500 mL while maintaining the optimized parameters. Figure 4 shows that the concentrations of hydrogen peroxide, nitrate, nitrite, and pH exhibit similar trends in relation to variations in power input. This showed that the PAW generated from 500 mL of distilled water with a 500 W power input produced the highest RONS concentrations. To further enhance RONS concentrations, the activation duration was prolonged to 12 min, which resulted in a continued rise in the mean concentrations of hydrogen peroxide and nitrate, while nitrite remained undetectable as shown in Table 1.
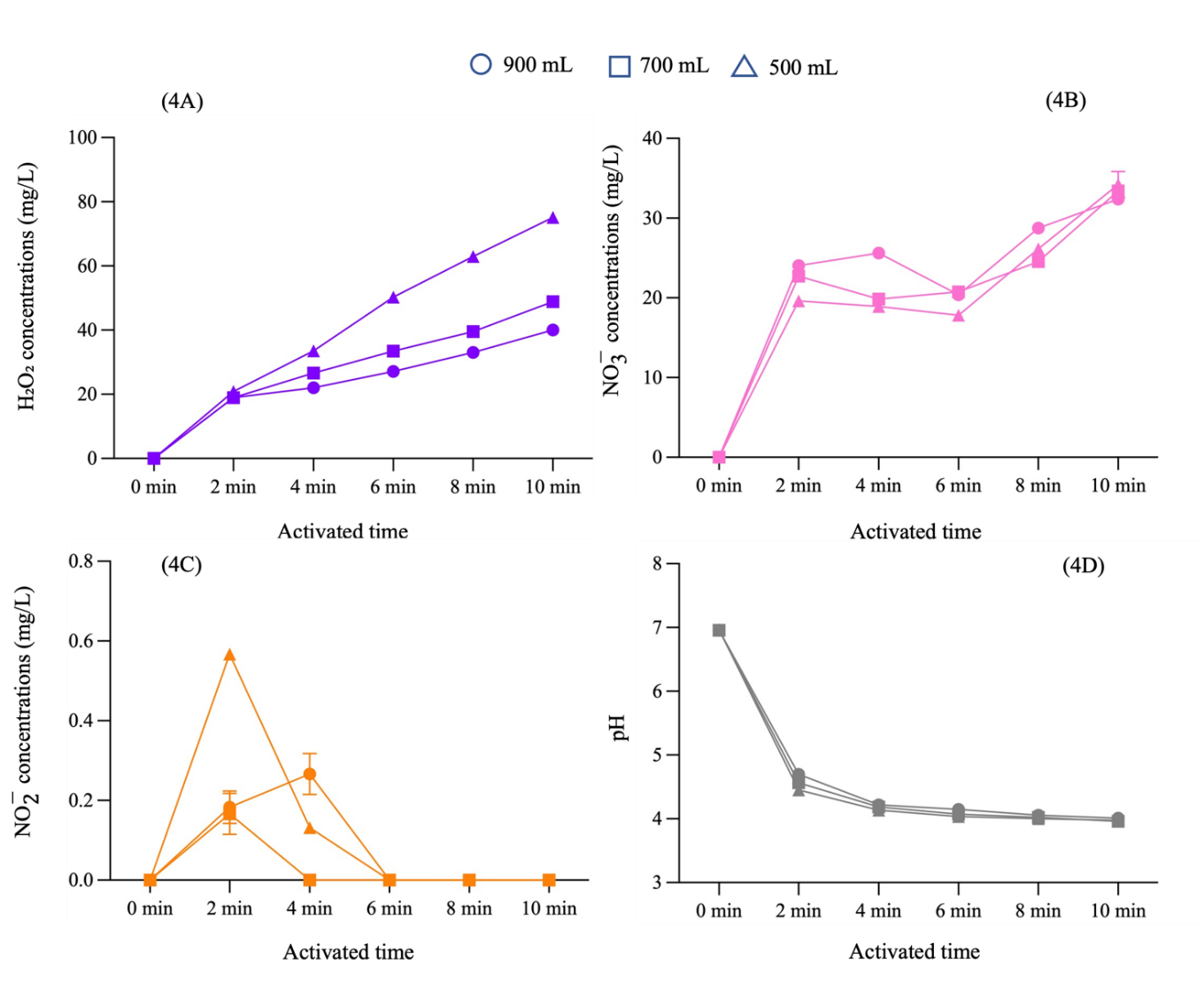
Figure 4 The concentrations of hydrogen peroxide, nitrate, nitrite and pH in PAW, with different volumes of distilled water at various activation times.
Table 1 Reactive species concentration in PAW generated from 500 mL of distilled water with a 500 W power input.
Activation time (min) |
RONS concentrations (mg/L) |
pH |
||
H₂O₂ |
|
|
||
2 |
20.9 ± 0.25 |
9.6 ± 0.39 |
0.6 ± 0.01 |
4.5 ± 0.15 |
4 |
33.6 ± 0.81 |
18.9 ± 0.51 |
0 |
4.1 ± 0.04 |
6 |
50.3 ± 0.72 |
17.8 ± 0.35 |
0 |
4.0 ± 0.02 |
8 |
62.9 ± 1.02 |
26.2 ± 0.44 |
0 |
4.1 ± 0.04 |
10 |
75.1 ± 1.76 |
34.2 ± 1.46 |
0 |
4.0 ± 0.03 |
12 |
89.7 ± 1.48 |
38.8 ± 1.73 |
0 |
4.0 ± 0.03 |
Experimental results demonstrated the direct correlation between NTAPP activation time, power input, and the concentrations of hydrogen peroxide and nitrate in PAW. Conversely, a decrease the water volume led to an increase in hydrogen peroxide and nitrate concentrations, which is attributable to a higher concentration of RONS per unit volume. These findings align with previous studies [32]. RONS primarily originate from the dissociation and excitation of electrons in ambient air molecules, notably hydroxyl radicals (⋅OH). Upon contact with water, these molecules recombine to form hydrogen peroxide through reactions such as ⋅OH+⋅OH→ H₂O₂ and ⋅OH+O*→ H₂O₂ +⋅H. Furthermore, the ultraviolet (UV) radiation emitted by NTAPP also stimulates increased hydrogen peroxide production. In our study has shown nitrite concentrations peaked at 2 min of activation and subsequently decreased to undetectable levels, indicating a dynamic equilibrium with other nitrogen species, particularly nitrate. This conversion of nitrite to nitrate in PAW is an oxidation process mediated by reactive oxygen species, including hydroxyl radicals (⋅OH) and atomic oxygen (O), which act as strong oxidizers, converting less stable nitrite into more stable nitrate. As NTAPP activation time increases, the accumulation of these reactive oxygen species enhances this conversion, leading to a decrease in nitrite and an increase in nitrate. This observation corresponds to research that similarly identified this pattern in their investigation on RONS analysis and the fungicidal effects of PAW against C. albicans using a dielectric barrier discharge plasma device with air as the working gas [33]. Consequently, in our research, the optimization of PAW production primarily focused on maximizing hydrogen peroxide and nitrate concentrations due to their greater stability compared to nitrite, which showed only a transient increase at 2 - 4 min before becoming undetectable. The plasma activation time in this study was limited to 12 min. Due to the limitations of this investigation, increasing the activation time to 14 min caused uneven plasma plume and burning damage to the high voltage electrode.
Effect of PAW on planktonic C. albicans
Initial findings indicated that varying the power input and activation time of PAW had a dose-dependent effect on C. albicans concentration reduction. At a power input of 400 W, no reduction was observed of across all tested activation times. The percentage concentration reduction of C. albicans by 10-minutes PAW at 500 W was found to be the highest and was not significantly different from that achieved 8-minutes PAW at 500 W or 10-minutes PAW at 700 W as shown in Figure 5. Based on these investigations, the power input was set at 500 W, and the volume of distilled water was varied to 900, 700, and 500 mL. PAW generated from 500 mL of distilled water for a duration of 10 min demonstrated the highest mean percentage concentration reduction of C. albicans as shown in Figure 6. Following the optimization of power input and water volume, extended plasma activation times were considered to further enhance RONS concentrations. The finding from this final phase showed that a 12-minutes PAW yielded the highest percentage of C. albicans concentration reduction, although it was not significantly different from the concentration reduction achieved at the 10-minutes PAW as shown in Figure 7.
The investigation assessed the turbidity of microbial cultures to evaluate PAW’s efficacy against C. albicans. The findings indicated a clear correlation between the PAW activation time and the reduction in C. albicans, with concentration reduction effects directly linked to elevated concentrations of RONS. This investigation demonstrates that PAW exhibits significant efficacy against C. albicans. The proposed mechanism of concentration reduction suggests that RONS generated by NTAPP disrupt the fungal cell wall, which is primarily composed of chitin, beta-(1,3)-glucan, and beta-(1,6)-glucan. The three substances are interconnected through hydrogen bonds, resulting in a robust and dense chitin polymer network. The reactive species have the capability to disrupt hydrogen bonds by extracting hydrogen atoms from the chitin polymer, subsequently converting them into stable water molecules [34]. In contrast, the pH of the PAW did not serve as a primary inhibitory factor, as this fungus is known to thrive in a wide pH range from below 2 to above 10 [35].
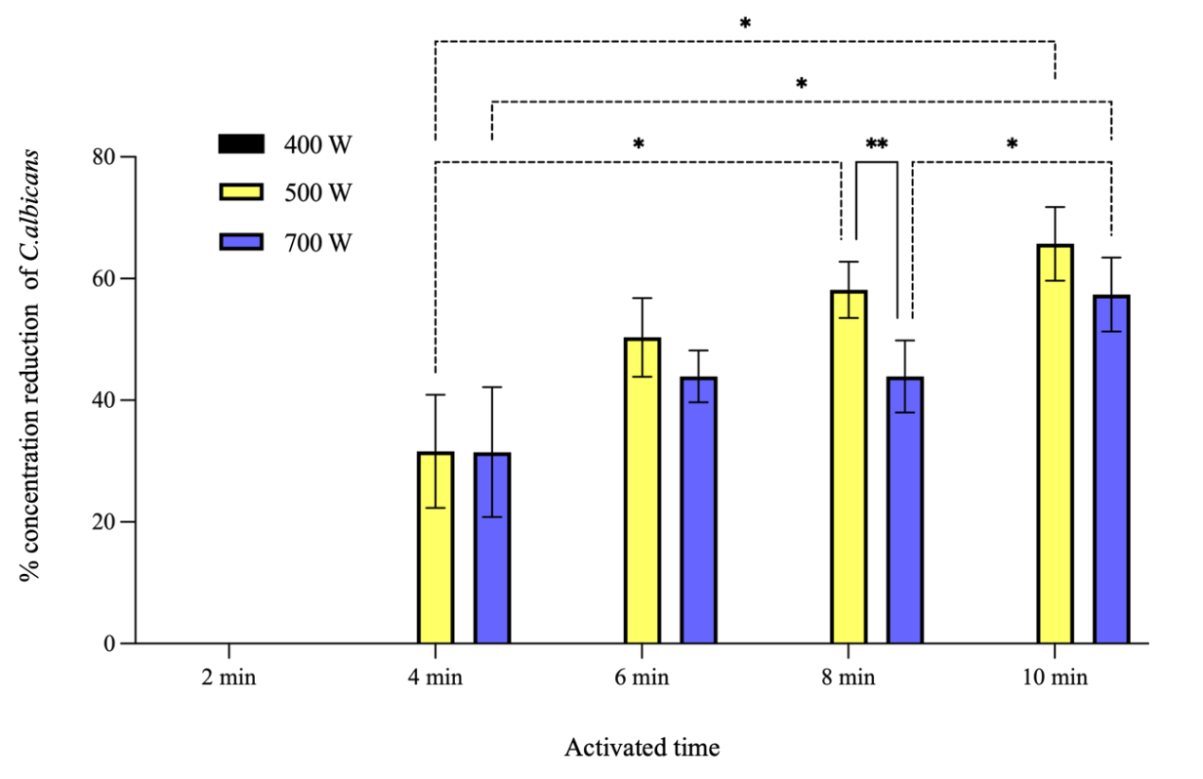
Figure 5 The percentage concentration reduction of C. albicans by PAW prepared from 900 mL of distilled water. PAW was generated at various activation times and with different power input settings. Symbols * and **: Indicate that the mean values are statistically significantly different at p < 0.05 and p < 0.01, respectively.
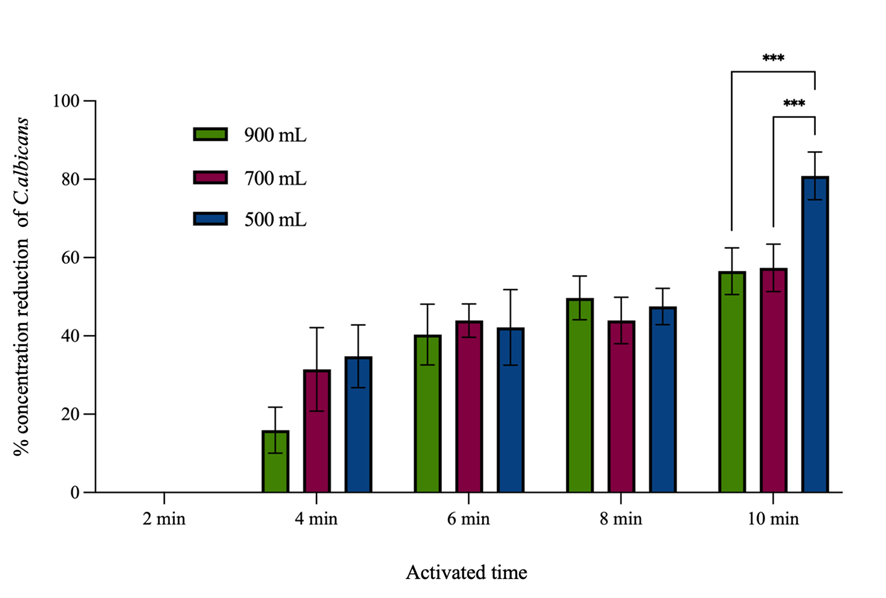
Figure 6 The percentage concentration reduction of C. albicans by PAW generated 500 W of power input at various activation times and with different distilled water volumes. Symbols ***: Indicate that the mean values are statistically significantly different at p < 0.005.
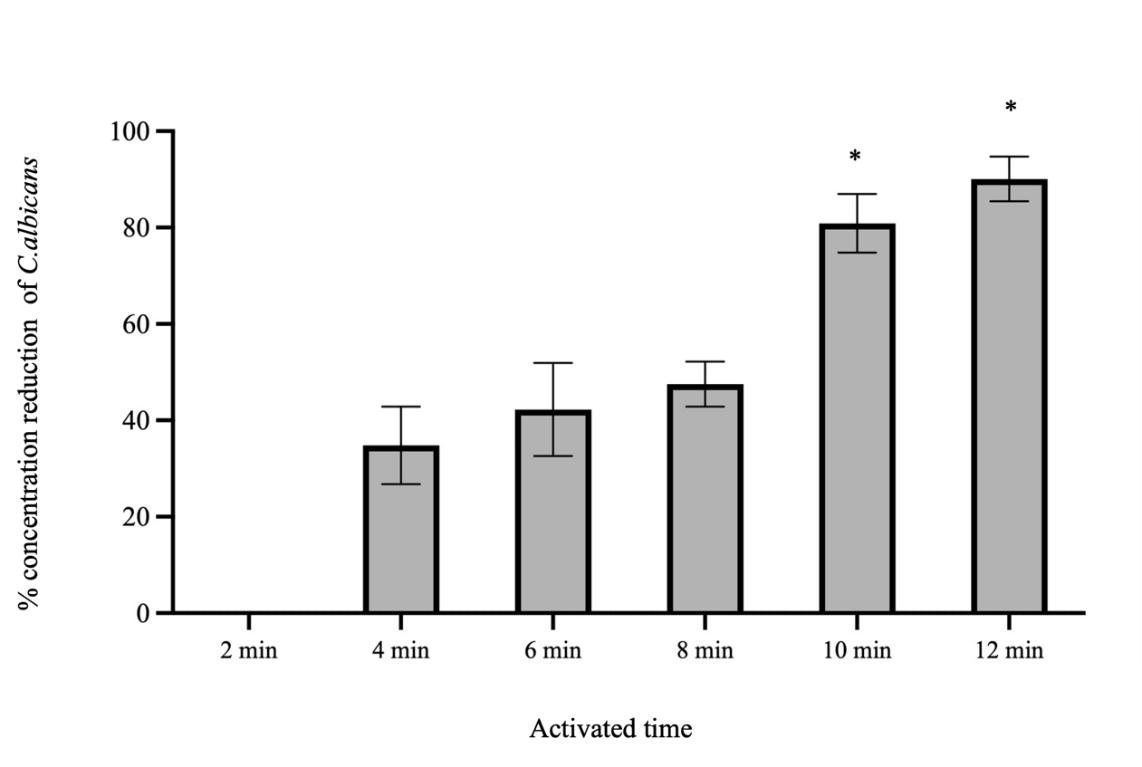
Figure 7 The percentage concentration reduction of C. albicans by PAW generated 500 W of power input from 500 mL of distilled water at various activation times. Symbols *: Indicates that the mean value of the group is statistically significantly different from the other groups p < 0.05.
Antifungal activity against C. albicans biofilms on acrylic surface
This study phase investigated the effect of PAW in eliminating C. albicans biofilms on acrylic surfaces. PAW was generated under the following parameters: 500 mL of distilled water, 500 W of power input, 500 mA current, 4 kVp potential difference, and ambient air as the working gas at a flow rate of 3 liter per minute. While 12-minutes PAW yielded higher hydrogen peroxide and nitrate concentrations than 10-minutes PAW, both were selected for this experiment due to their comparable efficacy in the prior effect of PAW on planktonic C. albicans test. By quantifying viable cells via colony count as shown in Table 2 and Figure 8, the findings indicated that PAW significantly diminished biofilm viability compared to the distilled water control. 10 and 12-minutes PAW both markedly reduced biofilm viability across all immersion times with no significant differences observed between them at shorter durations. However, after 8 h of immersion, 12-minutes PAW demonstrated a significant difference in biofilm inhibition from 10-minute PAW. These results imply that PAW possesses antifungal activity against C. albicans biofilms. Nevertheless, 10 and 12-minutes PAW was less effective than 1% sodium hypochlorite, a difference that was statistically significant (p < 0.001) as shown in Figure 8.
Table 2 Colony count of C. albicans biofilms at various immersion times.
Group |
Colony count (×104 CFU/mL ) at various immersion times |
|||
10 min |
30 min |
1 h |
8 h |
|
distilled water (control) |
74.7 ± 6.0 |
71.7 ± 6.9 |
50.7 ± 5.0 |
50.4 ± 2.9 |
10-minute PAW |
14.1 ± 1.6 |
14.6 ± 1.5 |
11.1 ± 7.4 |
11.3 ± 2.2 |
12-minute PAW |
15.9 ± 3.2 |
18.0 ± 3.0 |
12.4 ± 3.1 |
6.7 ± 1.5 |
NaOCl |
1.4 ± 1.6 |
- |
- |
- |
The experimental design utilized artificial saliva, composed of methylcellulose, glycerine, xylitol, and distilled water, to develop C. albicans biofilms. Candida albicans is capable of autonomous biofilm formation. Artificial saliva is used to increase the mediumʼs viscosity, and this method has been adopted in several studies for the purpose of creating biofilms [36-38]. This substitution improved experimental repeatability and consistency, which is essential for controlled scientific research. However, a key limitation is the biological difference between artificial and human saliva. Human saliva contains a complex mixture of proteins, enzymes, electrolytes, immunoglobulins, and its pH and viscosity can individually varied. These differences could potentially affect the biofilm’s physical structure, the initial cellular adhesion of C. albicans, and, most importantly, the diffusion of RONS in PAW. Accordingly, while artificial saliva enhances experimental control, this limitation makes it difficult to extrapolate the results directly to the complex human oral environment, necessitating further research in more physiologically relevant models.
The percentage inhibition of C. albicans of 10-minute PAW decreased slightly after 8 h of immersion, rendering it less effective than 12-minute PAW. The decreased percentage inhibition of C. albicans over the 8-hour period is likely attributable to the higher regrowth of C. albicans as well as the lower concentrations of hydrogen peroxide and nitrate in 10-minute PAW compared to 12-minute PAW. This result corresponds with a previous study that examined the effect of direct NTAPP exposure in removing clinical strains and C. albicans biofilms (ATCC 10231) from acrylic surfaces. It showed that biofilm levels were significantly reduced by using plasma jet for 8 min [39]. Therefore, variations in treatment duration and efficacy between studies can be attributed to differences in the experimental setup, such as the method of NTAPP application, power input setting, potential difference, working gas, and the mechanism of C. albicans biofilm formation.
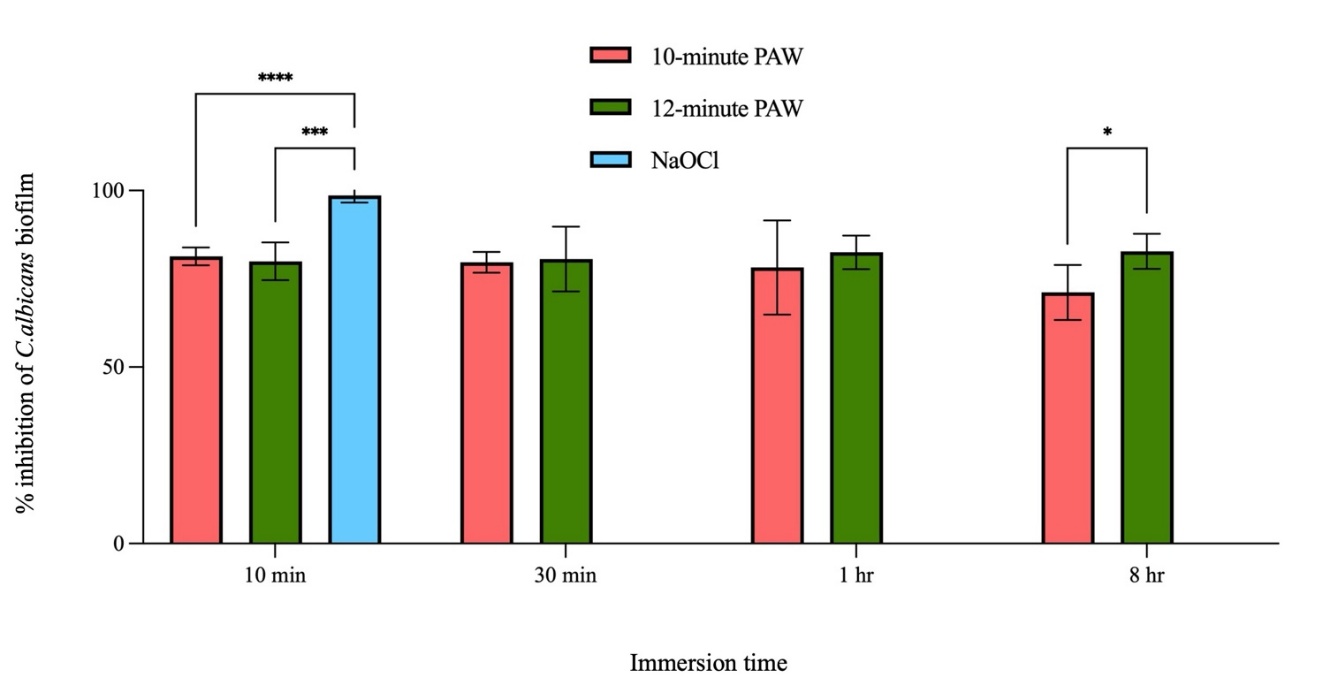
Figure 8 The percentage inhibition of C. albicans by 10 and 12-minutes PAW at various immersion times. * significant difference at p < 0.05 after comparison with treated groups, *** and **** significant difference at p < 0.005 and p < 0.001, respectively after comparison with control group.
A 10-minute immersion in 1% NaOCl in eliminating C. albicans biofilms compared to 10 and 12-minutes PAW can be attributed to the differing mechanisms of action and some components. NaOCl primarily eradicates pathogens by producing hypochlorous acid (HOCl), which rapidly and efficiently penetrates the extracellular matrix of biofilms. NaOCl is highly effective at degrading organic materials, thereby deconstructing crucial components of the C. albicans biofilm matrix, including polysaccharides, proteins, and DNA. In contrast, PAW functions as an antibacterial by generating RONS [40]. While these RONS can also harm microbial cells and decompose biofilm matrix, total concentration and stability in PAW may be less than the concentration of produced agents in NaOCl.
Complete inhibition of C. albicans biofilms is hindered by the distinctive structural features and fluid transport dynamics inherent to biofilms. A previous study demonstrated that bacterial biofilms, including those of Bacillus subtilis, contain internal water channels. This principle applies equally to biofilms of C. albicans, where the thick fluid transport channels are located within the center of the biofilm and extend to the exterior, facilitating the movement of fluids from the base to the outside. Upon contact with C. albicans biofilms, RONS accumulate at the base and subsequently traverse the internal channels to various regions of the biofilm. This disrupts the cell membrane, allowing the cell's contents to escape [41]. This ultimately undermines cellular integrity and may lead to the disassociation of cells from the biofilm matrix. Although PAW can inhibit C. albicans biofilms, its efficacy against planktonic cells is comparatively diminished. This is probably attributable to the inherently complex and protective nature of biofilm construction.
Recent research indicates that NaOCl is more effective than PAW in reducing C. albicans biofilms. Nonetheless, PAW possesses a significant benefit over NaOCl due to its considerably lower toxicity to host tissues. The use of NaOCl presents a significant issue, particularly in medical and dental environments that must take in to account patient safety and equipment longevity are important. Future research should focus on improving the efficiency of PAW manufacturing to get higher concentrations of more stable RONS, along with devising suitable storage techniques to maintain its activity. Exploring combination therapy that incorporates PAW with additional therapies, such as ultrasonic therapy, physical debridement, or reduced doses of conventional antibacterial drugs, is also advisable. These solutions may effectively eliminate biofilms, potentially beyond the efficacy of current chemical agents, while maintaining the inherent advantages of safety and environmental sustainability.
Conclusions
PAW effectively inhibits C. albicans through the generation of RONS. Our findings demonstrate that 12-minute PAW prepared from 500 mL of distilled water with a power of 500 W yielded the highest concentrations of hydrogen peroxide and nitrate. Furthermore, both 10 and 12-minutes PAW demonstrated comparable efficacy in inhibiting C. albicans suspension. When applied to C. albicans biofilms on acrylic surfaces, both the 10 and 12-minutes PAW also showed similar efficacy after a 10, 30-imnutes and 1-hour immersion except 8-hour immersion. While these results are promising, further clinical studies are necessary to fully explore the application of PAW against C. albicans on acrylic surfaces for the prevention and treatment of denture stomatitis.
Acknowledgements
The authors are grateful to the Research Fund for Postgraduate Students of the Faculty of Dentistry, Chiang Mai University, Chiang Mai 50200, Thailand and Royal Collage of Dental Surgeon, Nonthaburi 11000, Thailand for financial support.
Declaration of Generative AI in Scientific Writing
The authors used Generative AI (Gemini) and Quillbot to improve the language, readability, and structural flow of the manuscript. The AI was used solely for language enhancement; all data interpretation, literature synthesis, and conclusions were performed by the authors. The authors have meticulously reviewed all AI-assisted sections for technical accuracy.
CRediT Author Statement
Pattarawan Singkharat: Conceptualization, Methodology, Formal analysis, Investigation, and Writing - Original draft preparation. Chanchai Dechthummarong: Methodology, Investigation; Resources, and Supervision. Chintana Itthidecharon: Conceptualization, Visualization, and Supervision. Phenphichar Wanachantararak: Conceptualization, Methodology, Data curation, Investigation, Writing - Reviewing and Editing, Visualization, and Supervision.
References
[1] DE McReynolds, A Moorthy, JOC Moneley, MA Jabra-Rizk and AS Sultan. Denture stomatitis—an interdisciplinary clinical review. Journal of Prosthodontics 2023; 32(7), 560-570.
[2] A Rodriguez-Archilla and C Garcia-Galan. Etiological factors related to denture stomatitis: A Meta-analysis. Dentistry and Medical Research 2020; 8(2), 37.
[3] J Verran, GC Lees and AP Shakespeare. The effect of surface roughness on the adhesion of Candida albicans to acrylic. Biofouling 2009; 3(3), 183-191.
[4] PMB da Silva, EJTR Acosta, LDR Pinto, M Graeff, DMP Spolidorio, RS Almeida and VC Porto. Microscopical analysis of Candida albicans biofilms on heat-polymerised acrylic resin after chlorhexidine gluconate and sodium hypochlorite treatments. Mycoses 2011; 54(6), 712-717.
[5] Z Choudhry, S Malik, ZA Mirani, SA Khan, SMR Kazmi, WA Farooqui, MA Ahmed, KA AlAali, A Alshahrani, M Alrabiah, AH Albaqawi and T Abduljabbar. Antifungal efficacy of sodium perborate and microwave irradiation for surface disinfection of polymethyl methacrylate polymer. Applied Sciences 2022; 12(14), 7004.
[6] R Harintharanon, C Itthidecharon, P Wanachantararak and S Sookkhee. Anticandidal activity of cajuput and lemongrass essential oils supplemented in alcohol-free mouthwash against Candida albicans biofilm formation on 96-well plate and acrylic surfaces. Natural and Life Sciences Communications 2023; 22(4), 2023067
[7] EH Lee, YH Jeon, SJ An, YH Deng, HB Kwon, YJ Lim, H Kong and MJ Kim. Removal effect of Candida albicans biofilms from the PMMA resin surface by using a manganese oxide nanozyme-doped diatom microbubbler. Heliyon 2022; 8(12), 12290.
[8] E Merigo, M Chevalier, S Conti, T Ciociola, C Fornaini, M Manfredi, P Vescovi and A Doglio. Antimicrobial effect on Candida albicans biofilm by application of different wavelengths and dyes and the synthetic killer decapeptide KP. Laser Therapy 2019; 28(3), 180-186.
[9] S Cha and Y Parl. Plasma in dentistry. Clinical plasma medicine 2014; 2(1), 4-10.
[10] S Lata, S Chakravorty, T Mitra, PK Pradhan, S Mohanty, P Patel, E Jha, PK Panda, SK Verma and M Suar. Aurora borealis in dentistry: The applications of cold plasma in biomedicine. Materials Today Bio 2022; 13, 100200.
[11] AC Borges, KG Kostov, RS Pessoa, GMA de Abreu, GMG de Lima, LW Figueira and CY Koga-Ito. Applications of cold atmospheric pressure plasma in dentistry. Applied Sciences 2021; 11(5), 1975.
[12] A Rutscher, H Kersten, M Schmidt, KH Schoenbach. Low temperature plasmas: Fundamentals, technologies and techniques. Wiley, Germany, 2008.
[13] M Laroussi. Cold plasma in medicine and healthcare: The new Frontier in low temperature plasma applications. Frontiers in Physics 2020; 8, 74.
[14] NK Kaushik, B Ghimire, Y Li, M Adhikari, M Veerana, N Kaushik, N Jha, B Adhikari, SJ Lee, K Masur, T von Woedtke, KD Weltmann and EH Choi. Biological and medical applications of plasma-activated media, water and solutions. Biological Chemistry 2018; 400(1), 39-62.
[15] KS Wong, NSL Chew, M Low and MK Tan. Plasma-activated water: Physicochemical properties, generation techniques, and applications. Processes 2023; 11(7), 2213.
[16] M He, J Duan, J Xu, M Ma, B Chai, G He, L Gan, S Zhang, X Duan, X Lu and H Chen. Candida albicans biofilm inactivated by cold plasma treatment in vitro and in vivo. Plasma Processes and Polymers 2020; 17(4), 1900068.
[17] ACOC Doria, FR Figueira, JSB de Lima, JAN Figueira, AHR Castro, BN Sismanoglu, G Petraconi, HS Maciel, S Khouri and RS Pessoa. Inactivation of Candida albicans biofilms by atmospheric gliding arc plasma jet: Effect of gas chemistry/flow and plasma pulsing. Plasma Research Express 2019; 1, 015001.
[18] E Tyczkowska-Sieroń, T Kałużewski, M Grabiec and J Tyczkowski. Genotypic and phenotypic changes in Candida albicans as a result of cold plasma treatment. International Journal of Molecular Sciences 2020; 21(21), 8100.
[19] TMC Nishime, AC Borges, CY Koga-Ito, M Machida, LRO Hein and KG Kostov. Non-thermal atmospheric pressure plasma jet applied to inactivation of different microorganisms. Surface and Coatings Technology 2017; 312, 19-24.
[20] KG Kostov, AC Borges, CYK Ito, T Nishime, V Prysiazhnyi and R Honda. Inactivation of Candida albicans by cold atmospheric pressure plasma jet. IEEE Transactions on Plasma Science 2015; 43(3), 770-775.
[21] J Delben, C Zago, N Tyhovych, S Duarte and C Vergani. Effect of atmospheric-pressure cold plasma on pathogenic oral biofilms and in vitro reconstituted oral epithelium. PLoS One 2016; 11(5), 0155427.
[22] P Wanachantararak, P Suanpoot and M Nisoa. Inhibitory activity of cold atmospheric plasma on Candida albicans. Walailak Journal of Science and Technology 2019; 16(6), 401-408.
[23] V Scholtz, J Julák and V Kríha. The microbicidal effect of low-temperature plasma generated by corona discharge: Comparison of various microorganisms on an agar surface or in aqueous suspension. Plasma Processes and Polymers 2010; 7(3-4), 237-243.
[24] J Julák, V Scholtz, S Kotúčová and O Janoušková. The persistent microbicidal effect in water exposed to the corona discharge. Physica Medica 2012; 28(3), 230-239.
[25] R Hossain, JJ Dickinson, A Apblett and NF Materer. Detection of hydrogen peroxide in liquid and vapors using Titanium (IV)-Based test strips and low-cost hardware. Sensors 2022; 22(17), 6635.
[26] Z Rashmei, H Bornasi and M Ghoranneviss. Evaluation of treatment and disinfection of water using cold atmospheric plasma. Journal of Water and Health 2016; 14(4), 609-616.
[27] M Ali, JH Cheng and DW Sun. Effect of plasma activated water and buffer solution on fungicide degradation from tomato (Solanum lycopersicum) fruit. Food Chemistry 2021; 350, 129195.
[28] Y Zheng, S Wu, J Dang, S Wang, Z Liu, J Fang, P Han and J Zhang. Reduction of phoxim pesticide residues from grapes by atmospheric pressure non-thermal air plasma activated water. Journal of Hazardous Materials 2019; 377, 98-105.
[29] RO Adeyemo, IM Famuyide, JP Dzoyem and MGL Joy. Anti-biofilm, antibacterial, and anti-quorum sensing activities of selected south african plants traditionally used to treat diarrhoea. Evidence-Based Complementary and Alternative Medicine 2022; 2022, 1307801.
[30] MM Quezada, H Salgado, A Correia, C Fernandes and P Fonseca. Investigation of the effect of the same polishing protocol on the surface roughness of denture base acrylic resins. Biomedicines 2022; 10(8), 1971.
[31] W Namangkalakul, S Benjavongkulchai, T Pochana, A Promchai, W Satitviboon, S Howattanapanich, R Phuprasong, N Ungvijanpunya, D Supakanjanakanti, T Chaitrakoonthong, S Muangsawat, P Thanyasrisung and O Matangkasombut. Activity of chitosan antifungal denture adhesive against common Candida species and Candida albicans adherence on denture base acrylic resin. Journal of Prosthodontics Dentistry 2020; 123(1), 181.1-181.7.
[32] R Ono and T Oda. Measurement of hydroxyl radicals in pulsed corona discharge. Journal of Electrostatics 2002; 55(3-4), 333-342.
[33] R Laurita, D Barbieri, M Gherardi, V Colombo and P Lukes. Chemical analysis of reactive species and antimicrobial activity of water treated by nanosecond pulsed DBD air plasma. Clinical Plasma Medicine 2015; 3(2), 53-61.
[34] X Wang, L Pang, S Yang, L Zou, Y Zhang and T Zhao. Plasma-induced destruction of Candida albicans cell wall components: A reactive molecular dynamics simulation. Biochemical and Biophysical Research Communications 2021; 576, 53-58.
[35] HS Rane, SR Hayek, JE Frye, EL Abeyta, SM Bernardo, KJ Parra and SA Lee. Candida albicans Pma1p contributes to growth, pH homeostasis, and hyphal formation. Frontiers in Microbiology 2019; 10, 1012.
[36] MP Silva, JC Junior, AL Jorjão, AKDS Machado, LD de Oliveira, JC Junqueira and AOC Jorge. Influence of artificial saliva in biofilm formation of Candida albicans in vitro. Brazil Oral Research 2012; 26(1), 24-28.
[37] HRDS Schacher, MM Oliveira, R Medina-Silva and LM de Menezes. In vitro Streptococcus mutans and Candida albicans adhesion on clear orthodontic aligners exposed to artificial saliva. Dental Materials 2025; 41(10), 1332-1338.
[38] J Wu, F Zeng, Z Chen, Y Chen, L Ge, K Teng, Q Qu and L Li. Influence of Candida albicans-Streptococcus mutans cross-border interactions on the corrosion behaviour of Ti and Ti-Zr in artificial saliva. Corrosion Science 2024; 233, 112101.
[39] P Suwanampai, C Itthidecharon, P Wanachantararak and P Suanpoot. Efficiency of non-thermal atmospheric pressure plasma on killing Candida albicans biofilm. Chiang Mai Dental Journal 2019; 40(3), 113-123.
[40] S Fukuzaki. Mechanisms of actions of sodium hypochlorite in cleaning and disinfection processes. Biocontrol Science 2006; 11(4), 147-157.
[41] JN Wilking, V Zaburdaev, M de Volder, R Losick, MP Brenner and DA Weitz. Liquid transport facilitated by channels in Bacillus Subtilis biofilms. Proceedings of the National Academy of Sciences of the United States of America 2013; 110(3), 848-852.

