
Trends
Sci.
2026;
23(6):
11671
Optimized In Vitro Culture Media and Plant Growth Regulators Enhance Propagation and Steviol Glycoside Yield in Stevia rebaudiana Bertoni
Kittiya Thongjan, Tassanai Jaruwattanaphan and Benya Manochai*
Department of Horticulture, Faculty of Agriculture, Kasetsart University, Bangkok 10900, Thailand
(*Corresponding author’s e-mail: [email protected])
Received: 22 August 2025, Revised: 3 December 2025, Accepted: 10 December 2025, Published: 15 February 2026
Abstract
Thailand has great potential for growing Stevia rebaudiana Bertoni, a zero-calorie sweetener. Traditional stem cutting propagation is inefficient, laborious, and susceptible to disease. Microbial contamination, hyperhydration, and poor seed germination limit tissue culture’s efficiency. This study, conducted between January 2023 and November 2024 at Kasetsart University in Thailand. The goal was to evaluate the plant growth regulators (PGRs) affected in vitro culture conditions to improve seed germination, shoot multiplication, and plant performance. Seeds cultured on Murashige and Skoog (MS) medium with 3% sucrose achieved 9.5% germination within 4 weeks, which may reflect the small seed size and the self-incompatibility commonly reported in Stevia rebaudiana. Benzyl adenine (BA) supplementation at 2 mg/L increased shoot proliferation, creating robust plantlets with higher height and biomass potential. After one month on BA-enriched medium, plantlets were moved to hormone-free MS medium for roots and greenhouse adaptation, resulting in 100% survival. Mature plants yielded an average of 12 g fresh weight and 1.78 g dry weight per plant, with favorable steviol glycoside levels (stevioside 2.37%; rebaudioside A 2.10%), analyzed by high-performance liquid chromatography (HPLC) following the FAO (2010) method. These results demonstrate that BA supplementation is critical for efficient in vitro multiplication of stevia, while subsequent hormone-free rooting ensures high survival and stable growth. The developed protocol improves propagation efficiency, enhances plant quality, and supports reliable steviol glycoside accumulation, suggesting its potential application as a scalable method for commercial stevia production.
Keywords: Sweetener, Auxin, Cytokinin, Reb A, Propagation
Introduction
Stevia (Stevia rebaudiana Bertoni), a perennial herbaceous plant in the Asteraceae family, has gained significant attention as a natural source of high-intensity sweeteners in food and pharmaceutical industries [1-3]. This plant generates diterpenoid glycosides, stevioside and rebaudioside A (Reb A), which exhibit sweetness levels 200 - 300 times superior to sucrose [2,3]. Among these glycosides, Reb A has a favorable, sugar-like taste, whereas stevioside is frequently associated with a lingering bitter aftertaste [1,3].
However, large-scale cultivation in Thailand remains limited due to poor propagation efficiency and dependence on imported raw materials [4-6]. Conventional propagation methods such as seed germination and stem cutting are often inefficient, with
low success rates resulting from - self incompatibility [7] and high susceptibility to disease transmission. To overcome these limitations, tissue is a reliable approach for the rapid multiplication of genetically uniform and disease-free plants. Among the explant sources, nodal segments are consistently reported to be the most effective, exhibiting higher regeneration frequencies than leaf tissues [8]. Nodal explants cultured on Murashige and Skoog (MS) medium supplemented with appropriate combinations of plant growth regulators have demonstrated successful shoot multiplication, underscoring their suitability for efficient and large-scale propagation of stevia [9-12].
Plant growth regulators (PGRs) are essential for tissue culture work. The 6-benzylaminopurine (BA) is commonly used for promoting shoot growth. Auxins like naphthaleneacetic acid (NAA) and indole-3-butyric acid (IBA) promote root initiation, enhancing acclimation upon transplantation. The challenge is that the optimal PGR combinations and concentrations differ among species and genotypes. Consequently, plant responses are often inconsistent, leading to weak seedlings, hyperhydricity, and low survival rates.
Acclimatization, the transitional phase from in vitro to ex vitro environments, is a crucial factor in the success of tissue culture. Effective protocols have been shown to increase survival rates to 87.5% and shorten production cycles to just 51 days [13]. Furthermore, gradual hardening regimes have further improved survival to over 93.3%, along with increased chlorophyll fluorescence and antioxidant enzyme activity, indicating enhanced physiological adaptation to external environments [14].
The type and concentration of PGRs influence the root development, acclimatization, and overall vigor of plantlet. Chotikadachanarong and Dheeranupattana [15] found that low NAA concentration (0 - 0.1 mg/L) yielded the highest survival rate (80%) in stevia plantlets, while elevated concentrations (1.0 - 2.0 mg/L) decreased survival to 60%. The interaction between auxin concentration and media strength is also important. Thiyagarajan and Venkatachalam [16] reported that 0.4 mg/L NAA in half-strength MS medium promoted the highest rooting, with 65.8% survival after field transfer. Similarly, Yesmin [17] showed that 0.2 mg/L IBA in full-strength MS medium led to 84% survival and flowering within 40 days. These findings highlight the importance of optimizing both the type and concentration of auxins, as well as culture medium strength, to improve both rooting and post-transplant performance in stevia.
Beyond morphological growth parameters, PGRs also influence the biochemical composition of stevia plants. Aman et al. [18] indicated that culture media supplemented with cytokinins (BA and kinetin) reduced the accumulation of secondary metabolites, dulcoside-A and stevioside, in in vitro-grown tissues. Röck-Okuyucu et al. [19] also observed a reduction in stevioside and Reb A concentrations when culture media included diverse combinations of BA, kinetin, TDZ, IAA, and NAA. These results showed that while cytokinins help shoots grow, too much or too long of exposure could impair the commercial quality of stevia by lowering the levels of important bioactive components. Furthermore, high levels of cytokinins, especially BA, are closely related to hyperhydricity, a physiological disorder that restricts stem growth leading to in apical necrosis. This phenomenon may be related to cytokinin-induced ethylene biosynthesis [20]. To mitigate hyperhydricity and protect tissue integrity, various methods have been proposed, including reducing cytokinin concentrations, applying less potent alternatives like kinetin, or alternating between hormone-free and cytokinin-enriched media.
The addition of PGRs into tissue culture mediums has markedly improved stevia propagation efficiency. The challenges remain in refining hormonal regimes to simultaneously enhance shoot quality, sustain glycoside accumulation, diminish physiological diseases, and ensure superior post-transplant survival rates. Most previous studies have investigated the effects of cytokinin and auxins individually. However, the optimal hormonal balance that integrates propagation efficiency with biochemical stability has not been clearly established.
The establishment of clean and contamination-free cultures is challenging when employing field-gathered shoot explants, which are susceptible to hyperhydricity and necrosis post-sterilization. To address this constraint, seed-derived explants were used as initial materials for producing aseptic cultures and ensuring uniform growth. Solving the initial restriction is necessary to improving in vitro culture conditions and hormonal balance, which impacts propagation efficiency and steviol glycoside accumulation. Therefore, this study aims to investigate the combined effects of different PGR combinations in in vitro culture media on seed germination, shoot multiplication, glycoside accumulation, and plantlet performance. The findings are expected to refine existing micropropagation protocols and provide reproducible and commercially viable strategies for sustainable Stevia rebaudiana production.
Materials and methods
This study was conducted from January 2023 to November 2024 at the Department of Horticulture, Faculty of Agriculture, Kasetsart University, Bangkok, Thailand. The experiment comprised 2 main phases. In vitro culture experiments were performed at Plant Tissue Culture Laboratory, focusing on seed sterilization, germination and shoot multiplication. Acclimatization and greenhouse cultivation were carried out at the Horticultural Greenhouse, located within Kasetsart University (13°51'13"N, 100°34'08"E), to evaluate survival rate, plant growth performance, and steviol glycoside content.
Plant material and sterilization
Stevia rebaudiana seeds (Figure 1), collected from cultivated fields in Lampang Province, Thailand (18°19'32"N, 99°29'40"E), were used as the initial explant source. The seeds were harvested from healthy, mature plants and air-dried before use. For surface sterilization, a 2-step disinfection protocol was employed: (1) 15 %(v/v) sodium hypochlorite (NaOCl) for 15 min, followed by (2) 10 %(v/v) NaOCl for 10 min, with three rinses in sterile distilled water between steps to remove residual disinfectant.
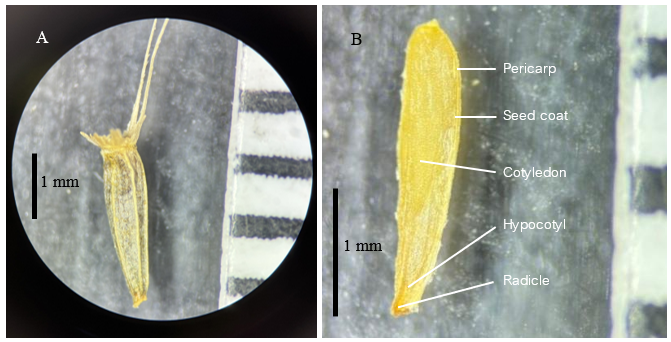
Figure 1 Microscopic view of a Stevia rebaudiana seed, showing detailed morphology including the seed coat and awn structure (A) and internal morphology (B). The scale bar represents millimeters for size reference.
Culture conditions
All experiments were conducted under aseptic conditions using gellan gum-solidified (2.5 g/L) media, pH-adjusted to 5.7 - 5.8. Cultures were maintained at 24 ± 2 °C under a 16-hours photoperiod (40 - 60 μmol m⁻² s⁻¹ light intensity).
Experiment 1: Optimization of medium and sucrose for stevia seed germination in vitro
Seeds were cultured on either half-strength or full-strength MS medium supplemented with 2% or 3% sucrose, resulting in 4 treatment combinations (Table 1). The experiment was arranged in a completely randomized design (CRD) with 4 treatments and 10 replications per treatment. Each replication consisted of a 125 mL culture bottle containing 25 mL of medium and 10 seeds, totaling 100 seeds per treatment. Germination rates were recorded biweekly over a 4-week period. Data were subjected to one-way analysis of variance (ANOVA), and treatment means were compared using Duncan’s multiple range test (DMRT) at a significance level of p < 0.05.
Table 1 Treatment conditions for optimizing medium and sucrose for stevia seed germination.
Treatment |
Culture medium |
Sucrose concentration (%) |
T1 |
½ MS medium |
2% |
T2 |
½ MS medium |
3% |
T3 |
MS medium |
2% |
T4 |
MS medium |
3% |
Experiment 2: Optimizing growth regulators for stevia micropropagation
Shoot tips and nodal segments (5 - 10 mm) from in vitro-grown seedlings (Experiment 1) were initially cultured on MS medium containing 30 g/L sucrose for 4 weeks. Subsequently, the explants were transferred to MS medium supplemented with 30 g/L sucrose, 2.5 g/L gellan gum, and 9 combinations of 6-benzylaminopurine (BA; 0.5 - 2.0 mg/L) and α-naphthaleneacetic acid (NAA; 0.1 - 0.5 mg/L) (Table 2). The pH of the medium was adjusted to 5.7 - 5.8 before autoclaving, and cultures were maintained under standard growth conditions. A 2×9 factorials experiment was arranged in a completely randomized design (CRD) with 15 replicates per treatment, with each replicate consisting of 4 explants. After 6 weeks, morphogenic responses, including shoot multiplication, shoot length, number of nodes, and root induction were recorded. Data were analyzed using 2-way ANOVA followed by Duncan’s multiple range test (DMRT) at a significance level of p < 0.01.
Table 2 Treatment conditions used to study the optimizing growth regulators for stevia micropropagation.
Treatment |
BA concentration |
NAA concentration |
|
0 mg/L |
0.0 mg/L |
|
0 mg/L |
0.5 mg/L |
|
0 mg/L |
1.0 mg/L |
|
1 mg/L |
0.0 mg/L |
|
1 mg/L |
0.5 mg/L |
|
1 mg/L |
1.0 mg/L |
|
2 mg/L |
0.0 mg/L |
|
2 mg/L |
0.5 mg/L |
|
2 mg/L |
1.0 mg/L |
Experiment 3: Effects of media formulations on stevia seedling vigor after greenhouse transfer
This experiment investigated the impact of nine different tissue culture media formulations (Table 2) on the vigor of Stevia rebaudiana seedlings following transfer from sterile conditions to a greenhouse environment. Shoot-tip explants were cultured on media formulations previously described in Experiment 2. After 4 weeks of in vitro growth, plantlets approximately 6 - 8 cm in height with 2 - 3 nodes were selected. These plantlets were carefully removed from culture vessels and inserted into sponge cubes for hydroponic cultivation. They were then transferred to plastic cups containing half-strength Enshi nutrient solution and acclimatized under in vitro conditions for one week. During this acclimatization period, the cups were covered to maintain high humidity. The 2 - 3 days before transplantation, the covers were gradually removed to expose the plantlets to ambient air. The environmental conditions outside the greenhouse during the experiment were as follows: Maximum temperature 31 °C, minimum temperature 24 °C, and average temperature 27 °C; relative humidity (RH) ranged from 67% (minimum) to 95% (maximum), with an average of 83%; and the average light intensity was 1,583 µmol m⁻² s⁻¹. These data represent the ambient climatic conditions outside the greenhouse and were obtained from the National Aeronautics and Space Administration (NASA) Langley Research Center (LaRC) Prediction of Worldwide Energy Resource (POWER) Project [21], funded through the NASA Earth Science/Applied Science Program.
The acclimated seedlings were transplanted into hydroponic channels operated under the Nutrient Film Technique (NFT) and supplied with the same half‑strength Enshi nutrient solution. During the first week, plastic covers were placed over the channels to maintain high humidity; they were then gradually removed. Plant survival was recorded every 2 weeks for a total of eight weeks, and plant height was measured at the same intervals. At week eight, the crop was harvested to determine final yield, and stevioside and rebaudioside A contents were quantified using the Food and Agriculture Organization method [22]. Antioxidant activity was assessed according to the procedure of Duvivier et al. [23]. The cultivation period spanned October to November 2024.
Results and discussion
Effect of culture medium and sucrose concentration for stevia seed germination
The results showed that Stevia rebaudiana seeds had a low germination rate, ranging from 5.00% to 9.5% (Figure 2), due to their diminutive achene size (about 3 mm) and restricted endosperm reserves (Figure 1). This poor germination rate may also be associated with self-incompatibility. Yadav et al. [7] reported that Stevia rebaudiana exhibits self-incompatibility, which reduces germination rates because of ineffective self-pollination and genetic incompatibility with closely related species. This trait is controlled by 1 or 2 gene pairs and is a characteristic frequently observed in members of the Asteraceae family.
Moreover, the result indicated that culturing stevia seeds in tissue culture medium did not enhance germination rates, likely due to the genetic control of this trait. However, germination was influenced by sucrose concentration. A sucrose concentration of 3% resulted in a higher germination rate (9.5%) compared to 2% (5.0% and 6.5% in MS and ½ MS media, respectively) (Figure 2). These findings align with Roni et al. [24], who reported sucrose-dependent germination in Eustoma seeds (0 - 3%). Higher sucrose concentrations (3%) also promoted more vigorous seedling development in both studies. Notably, basal medium (MS or ½ MS) did not significantly affect germination rates, ½ MS showed a slight trend toward improved germination. However, analysis of standard deviation (SD) values indicated that full-strength MS medium resulted in more consistent germination outcomes due to lower variability (Figure 2).
In summary, the results confirm that stevia seed germination was most successful when using MS medium supplemented with 3% sucrose. Due to the small size of stevia seeds and their limited endogenous nutrient reserves, an external supply of both nutrients and sugars is essential during early development. The ability of stevia seeds to absorb nutrients and sucrose at this concentration may be influenced by the presence of natural sweetening compounds within the seeds, which could enhance solute uptake. Therefore, a culture medium with relatively high concentrations of essential nutrients and sucrose is recommended to support optimal germination and early seedling growth in Stevia rebaudiana.

Figure 2 Germination rate of Stevia rebaudiana seeds in different culture media after 4 weeks. No significant differences was observed among treatments, and the data showed a coefficient of variation (CV) of 39.53%.
Effect of plant growth regulators on in vitro multiplication of stevia
Stevia rebaudiana plantlets were successfully propagated in vitro from seed-derived explants. Shoot-tip and nodal explants were grown on MS medium with various PGR concentrations. Explant type (Factor A) significantly impacted shoot length, internode number, and root number, but not shoot proliferation. In contrast, culture medium (Factor B) affected all measured traits (p < 0.01, Tables 3 and 4). There were significant Factor A×B interactions for shoot length, internode number, and root number, showing that explant source influenced medium response.
Among all variables, medium composition had the strongest influence. MS medium supplemented with 1, 2 or 2 mg/L BA combined with 1 mg/L NAA significantly enhanced shoot number, yielding 5.79, 6.08, and 5.61 shoots per explant, respectively (Table 3). These results are comparable to Ghose et al. [27], who reported 5.8 shoots per explant using 10 mg/L BAP, a concentration 5 times higher than that used in the present study. Conversely, media containing only NAA suppressed shoot formation, reaffirming the essential role of BA. These findings support the well-established principle that a high cytokinin-to-auxin ratio promotes shoot induction, while an auxin-dominant environment favors root development [28]. Comparable shoot yields under balanced PGR regimes have also been documented in Stevia [14] and Tinospora cordifolia, where 0.10 mg/L BA plus kinetin and NAA yielded 5.0 shoots per explant [29].
The exogenous BA induced de novo shoot formation even in the presence of endogenous auxin, suggesting limited cytokinin biosynthesis in stevia. In contrast, supplemental NAA disrupted the cytokinin-to-auxin balance and caused a dose reduction in both shoot and root development. Explants treated with 1 mg/L NAA developed purple, stunted shoots, which are indicative of auxin-induced ethylene stress (Figure 3). Elevated NAA likely enhances ACC synthase activity, increasing ethylene production, which in turn suppresses longitudinal growth and promotes radial swelling, resulting in thick, compact shoots [30]. Auxin oversaturation may also induce toxicity, further inhibiting rooting. By comparison, explants grown on hormone-free MS medium produced longer shoots, more nodes, and higher root numbers which resulted in the most vigorous plantlets (Tables 3 and 4; Figure 3).
The interaction between explant type and medium (Factor A×B) was significant for shoot length, internode number, and root number, but not for shoot number. This suggests that BA and NAA acted independently to regulate shoot proliferation yet interacted with tissue type in shaping overall morphology. BA predominantly facilitates shoot induction, whereas NAA is instrumental in root development. Within the concentration range, the effects of these hormones were near saturation, resulting in similar shoot numbers for both explants.
The hormone-free MS medium provided the high quality of plantlets, characterized by the longest shoots length (8.23 cm), greatest number of nodes (3.16), and highest root number (5.19) (Tables 3 and 4). In contrast, media containing BA and/or NAA significantly reduced these parameters. Shoot length showed negative relationship with NAA concentration, declining from 8.23 cm under hormone-free conditions to 1.64 cm (0.5 mg/L NAA) and 0.40 cm (1.0 mg/L NAA) in shoot tip explants. Nodal segments exhibited a similar trend. These finding suggest that shoot tip tissues, which already contain adequate endogenous auxin, are sensitive to oversaturation and then exhibit auxin toxicity when supplemented with NAA. Nodal explants, being initially auxin-deficient due to dormancy, are less affected by excess auxin and thus show a convergence in shoot length under high NAA concentrations.
In the 1BA0NAA treatment, shoot tip explants produced longer shoots (2.45 cm) than stem explants (2.16 cm), while the latter developed more internodes (2.30 vs. 1.95). This indicates that BA stimulated compact shoot clusters with different morphological characteristics depending on the explant. Shoot tips retained apical dominance through endogenous auxin, promoting elongation but limiting node formation. In contrast, the removal of apical meristems in stem explants increased the cytokinin-to-auxin ratio, triggering axillary bud outgrowth and nodal differentiation, although elongation was restricted. Similar patterns were observed in the 2BA1NAA treatment, which resulted in the longest shoots (2.95 cm in shoot tips), but also suppressed nodal development and root formation. These observations highlight that shoot elongation and nodal initiation are regulated by distinct, though overlapping, hormonal pathways, and that the response to PGRs depends on explant-specific hormonal profiles and sensitivity.
Root induction in Stevia rebaudiana explants was influenced by the local auxin-to-cytokinin ratio and the physiological state of the tissue (Table 4). In BA-free media or containing only low concentrations of NAA, shoot tip explants, enriched in endogenous IAA from their apical meristems, maintained a high auxin-to-cytokinin balance that promoted rapid root formation. As a result, they produced more roots than stem segments. This trend reversed in media containing high BA levels (1 - 2 mg/L) with or without added NAA. In these conditions, elevated BA increased cytokinin activity, and when combined with endogenous auxin, stimulated ethylene production in shoot tips.
This hormonal interaction suppressed rooting and increased auxin toxicity. In contrast, stem explants, which lack apical dominance and contain lower endogenous auxin levels, were less sensitive to ethylene stress. Additionally, the presence of NAA raised auxin levels to the optimal range for rooting without reaching toxic thresholds. Basal nodes, which often contain pre-formed root initials, responded favorably, allowing stem explants to outperform shoot tips in root number under high BA conditions. In low-cytokinin media, however, shoot tips retained their rooting advantage.
This study revealed that the type and concentration of PGRs had significant effects on Stevia rebaudiana in vitro propagation. PGR treatment with BA at 1 - 2 mg/L, either alone or combined with 1 mg/L NAA, markedly enhanced shoot multiplication (producing 5.61 - 6.08 shoots per explant) and elongation. The cytokinin-rich media performed better than the NAA-only media treatments and the hormone-free control, which confirms that a high cytokinin-to-auxin ratio is essential for shoot multiplication in Stevia. Although explant type did not significantly affect shoot number, shoot-tip explants produced plantlet better quality, particularly in terms of elongation, internode formation, and rooting.
Therefore, the practical method for in vitro multiplication of Stevia rebaudiana for commercial-scale propagation can follow this protocol. MS medium supplemented with 1 mg/L BA (1BA0NAA) or 2 mg/L BA plus 1 mg/L NAA (2BA1NAA) is recommended for shoot induction. Then, transferring the plantlets to hormone-free MS medium promotes root induction and prevents auxin-induced stress.

Figure 3 The growth response and morphology of stevia shoot explants vary depending on the concentration of BA and NAA in the medium at 4-week-old.
Table 3 Number of shoots and shoot length of Stevia explants grown on MS medium supplemented with different concentrations of BA and NAA after 4 weeks of culture. Values are represented as mean ± standard deviation.
Medium |
Number of shoots |
Shoot length (cm) |
||||
Shoot explant |
Stem explant |
Mean of medium (2) (Factor B) |
Shoot explant |
Stem explant |
Mean of medium (2) (Factor B) |
|
0BA0NAA |
1.30 ± 0.27 |
1.73 ± 0.61 |
1.52 ± 0.51 de |
8.23 ± 1.21 Aa |
1.63 ± 1.34 Ba |
4.86 ± 3.58 a |
0BA0.5NAA |
1.68 ± 0.78 |
1.85 ± 0.56 |
1.77 ± 0.67 d |
1.64 ± 1.68 Ab |
0.68 ± 0.62 Bc |
0.94 ± 1.34 c |
0BA1NAA |
0.88 ± 0.77 |
0.68 ± 0.55 |
0.78 ± 0.66 e |
0.40 ± 0.37 Ad |
0.23 ± 0.26 Bd |
0.34 ± 0.33 d |
1BA0NAA |
5.13 ± 1.31 |
6.45 ± 2.04 |
5.79 ± 1.81 a |
2.45 ± 0.58 Ab |
2.16 ± 0.50 Bb |
2.29 ± 0.55 b |
1BA0.5NAA |
3.08 ± 1.48 |
3.32 ± 1.46 |
3.20 ± 1.45 c |
1.75 ± 0.52 Ac |
1.05 ± 0.61 Bc |
1.44 ± 0.66 c |
1BA1NAA |
1.05 ± 0.78 |
1.13 ± 0.60 |
1.09 ± 0.68 de |
0.93 ± 0.71 Ad |
0.25 ± 0.20 Bd |
0.58 ± 0.62 d |
2BA0NAA |
5.70 ± 2.86 |
6.45 ± 2.02 |
6.08 ± 2.46 a |
1.94 ± 0.80 Ac |
1.23 ± 0.47 Bc |
1.62 ± 0.74 c |
2BA0.5NAA |
4.92 ± 2.58 |
3.87 ± 2.52 |
4.40 ± 2.56 b |
1.97 ± 0.44 Ac |
0.77 ± 0.46 Bc |
1.34 ± 0.75 c |
2BA1NAA |
5.40 ± 2.45 |
5.82 ± 2.76 |
5.61 ± 2.57 a |
2.95 ± 0.94 Ab |
2.17 ± 0.76 Bb |
2.60 ± 0.93 b |
Mean of Explant (1) (Factor A) |
3.24 ± 2.56 |
3.48 ± 2.72 |
– |
2.47 ± 2.33 A |
1.13 ± 0.95 B |
– |
Explant (Factor A) |
ns |
** |
||||
Medium (Factor B) |
** |
** |
||||
Explant x Medium |
ns |
** |
||||
C.V (%) |
50.72 |
43.72 |
||||
(1), (2) Means followed by the same letters in each column are not significantly different at p < 0.01 according to Duncan’s multiple range test (DMRT).
** indicates a significant effect of the factor at p < 0.01 according to one-way ANOVA; ns = not significant; C.V = coefficient of variation.
Table 4 Number of internodes and number of roots of Stevia rebaudiana explants cultured on MS media with various concentrations of BA and NAA after 4 weeks. Values are represented as mean ± standard deviation.
Medium |
Number of internode |
Number of root |
||||
Shoot explant |
Stem explant |
Mean of medium (2) (Factor B) |
Shoot explant |
Stem explant |
Mean of medium (2) (Factor B) |
|
0BA0NAA |
5.30 ± 1.37 Aa |
1.17 ± 0.87 Ba |
3.16 ± 2.39 a |
6.28 ± 1.88 Aa |
4.22 ± 1.93 Ba |
5.19 ± 2.15 a |
0BA0.5NAA |
0.80 ± 1.06 Adef |
0.32 ± 0.45 Bdef |
0.43 ± 0.84 def |
2.09 ± 2.04 Ab |
1.23 ± 1.24 Bb |
1.46 ± 1.72 b |
0BA1NAA |
0.10 ± 0.21 Af |
0.07 ± 0.11 Bf |
0.08 ± 0.17 f |
1.27 ± 1.87 Ac |
0.35 ± 0.67 Bc |
0.89 ± 1.46 c |
1BA0NAA |
1.95 ± 0.45 Ab |
2.30 ± 0.77 Bb |
2.13 ± 0.64 b |
0.00 ± 0.00 Ac |
0.00 ± 0.00 Bc |
0.00 ± 0.00 c |
1BA0.5NAA |
1.43 ± 0.49 Acd |
0.68 ± 0.70 Bcd |
1.06 ± 0.70 cd |
1.92 ± 1.14 Ab |
1.63 ± 1.39 Bb |
1.72 ± 1.26 b |
1BA1NAA |
0.42 ± 0.54 Aef |
0.00 ± 0.00 Bef |
0.23 ± 0.43 ef |
0.55 ± 0.81 Ac |
0.75 ± 0.63 Bc |
0.66 ± 0.72 c |
2BA0NAA |
1.52 ± 0.62 Ac |
0.88 ± 0.53 Bc |
1.23 ± 0.65 c |
0.05 ± 0.19 Ac |
0.13 ± 0.35 Bc |
0.11 ± 0.28 c |
2BA0.5NAA |
1.35 ± 0.63 Acde |
0.07 ± 0.26 Bcde |
0.74 ± 0.80 cde |
0.00 ± 0.00 Ac |
0.03 ± 0.09 Bc |
0.02 ± 0.06 c |
2BA1NAA |
2.33 ± 1.12 Ab |
1.52 ± 1.11 Bb |
1.95 ± 1.17 b |
0.10 ± 0.39 Ac |
0.27 ± 0.70 Bc |
0.21 ± 0.56 c |
Mean of Explant(1) (Factor A) |
1.69 ± 1.64 A |
0.78 ± 0.96 B |
– |
1.36 ± 2.25 A |
0.96 ± 1.59 B |
– |
Explant (Factor A) |
** |
** |
||||
Medium (Factor B) |
** |
** |
||||
Explant x Medium |
** |
** |
||||
C.V (%) |
58.72 |
95.42 |
||||
(1), (2) Means followed by the same letters in each column are not significantly different at p < 0.01 according to Duncan’s multiple range test (DMRT).
** indicates a significant effect of the factor at p < 0.01 according to 1-way ANOVA; C.V = coefficient of variation.
Effects of in vitro culture media on seedlings and quality traits after greenhouse transplantation
In this study, a structured acclimatization approach was employed to overcome this issue and successfully transition Stevia rebaudiana plantlets from in vitro to ex vitro conditions. Plantlets were screened for uniformity, plant height of 6 - 8 cm and healthy root. These plantlets were transferred into sponge cubes and placed in plastic cups containing half-strength Enshi nutrient solution (Figure 4(a)). To maintain a high-humidity microenvironment, the cups were sealed and kept in the tissue culture room for 2 weeks. A gradual hardening process was then initiated by removing the lids 2 - 3 days before transplantation, allowing for controlled exposure to ambient air (Figure 4(b)).
This protocol was effective, and all plantlets exhibiting a 100% survival after acclimatization. These results indicate that the transition stage plays an important role, especially when working with hormone-treated explants that can be more sensitive to environmental changes. With suitable acclimatization conditions, transplant shock can be reduced, which helps the seedlings establish more uniformly for hydroponic production.
Kaplan-Meier analysis (Figure 5) showed that plantlets grown on 2BA0NAA and 2BA1NAA media survived significantly better than those on any other formulation (log‑rank test, p < 0.01). After transplantation to the NFT system, these 2 treatments sustained 100 % survival during the first 14 days and remained alive through the eight‑week harvest, whereas all other treatments failed to establish (Figure 5). From Day 28 onward, plantlets in the 2BA1NAA group elongated more rapidly, reaching a final height of 41.0 cm by Day 56, compared with 31.0 cm in the 2BA0NAA group (Mann-Whitney U test, p < 0.05). Although 2BA0NAA promoted faster early growth, its rate of elongation declined at later stages (Figure 6). These results suggest that a culture medium supplemented with 2 mg/L BA, either alone or in combination with low NAA, can improve seedling vigor and post-transplantation resilience. The superior early-stage performance of 2BA0NAA-treated plants may be attributed to enhanced leaf development, as indicated by larger leaf size and greater leaf area (Figure 3), while the 2BA1NAA treatment appeared to support sustained growth through to harvest.
After eight weeks of greenhouse cultivation, significant differences in biomass accumulation were observed. Plants from the 2BA0NAA group yielded 12.00 g/plant fresh weight and 1.78 g/plant dry weight, while those from 2BA1NAA produced 6.38 g/plant fresh weight and 1.10 g/plant dry weight, representing an approximate 1.8-fold increase in productivity under the 2BA0NAA treatment (Figure 7). Morphologically, 2BA0NAA plants exhibited increased lateral branching and broader canopy architecture, in contrast to the upright, sparsely branched structure observed in 2BA1NAA-derived plants (Figure 8).
An analysis of the sweetener composition in stevia leaves revealed significant differences between treatments. Plants from the 2BA0NAA group contained 2.37% stevioside and 2.10% rebaudioside A (Reb A), whereas plants from the 2BA1NAA group exhibited 3.78% stevioside and only 0.32% Reb A (Figure 9(A)). The more balanced sweetness profile observed in 2BA0NAA can be attributed to its higher Reb A content, which is known to impart a cleaner and less bitter taste [1]. This composition was consistent with the concentrations per gram of dry leaf weight: 2BA0NAA plants contained 24.15 mg/g DW of stevioside and 20.23 mg/g DW of Reb A, compared to 38.21 mg/g DW of stevioside and only 2.61 mg/g DW of Reb A in the 2BA1NAA group (Figure 9(B)). Furthermore, compound yield calculations highlighted this advantage, with 2BA0NAA producing 41.57 mg of stevioside and 34.68 mg of Reb A per plant, versus 39.11 mg of stevioside and a mere 2.74 mg of Reb A in 2BA1NAA (Figure 9(C)).
These results indicate that the 2BA0NAA treatment promoted a more balanced accumulation of stevioside and Reb A. In contrast, the significantly reduced Reb A content in 2BA1NAA-treated plants may result from the suppressive effect of NAA on Reb A biosynthesis, likely mediated by auxin-cytokinin antagonism. Excessive NAA may disrupt Reb A production through 2 interrelated mechanisms. First, it may redirect the glucose donor (UDP-glucose) which is a key precursor toward root development, thereby limiting its availability for Reb A synthesis. Second, NAA may regulate the expression of the UGT76G1 gene, which encodes the enzyme essential for converting stevioside to Reb A [31,32]. This suppression may further involve interference with cytokinin activity, a hormone known to stimulate UGT76G1. Collectively, these mechanisms plausibly explain the decrease in Reb A and the corresponding accumulation of stevioside observed in 2BA1NAA-treated plants.
The results indicate that plant-growth regulator exposure during in vitro cultivation may create a persistent metabolic imprint lasting a minimum of eight weeks following transplantation to a nutrient-film technique (NFT) system. Additionally, auxins and cytokinins regulate the composition and concentration of secondary metabolites [33]. Therefore, the elevated stevioside levels observed in the 2BA1NAA treatment may arise from a similar auxin-mediated metabolic imprint that remains active after transplantation.
Antioxidant assays showed that plantlets grown on 2BA1NAA medium exhibited a lower EC50 (1.58 mg/mL) than those from the 2BA0NAA treatment (2.94 mg/mL), indicating stronger antioxidant activity (Figure 9(D)). This enhanced antioxidant activity is associated with the higher stevioside content in 2BA1NAA plantlets, as stevioside has been reported to exhibit more potent radical-scavenging activity than Reb A [34]. However, the lower Reb A content in 2BA1NAA plants may adversely affect the sensory quality of stevia products.
Collectively, these results demonstrate that optimizing cytokinin levels (2 mg/L BA without excess NAA) not only improves transplant success but also enhances biomass and produces a more desirable sweetener profile, providing a robust foundation for high-quality hydroponic stevia production.

Figure 4 Morphology of tissue-cultured stevia seedlings in the pre-planting stage (a) and seedlings grown in the NFT system within the greenhouse (b).
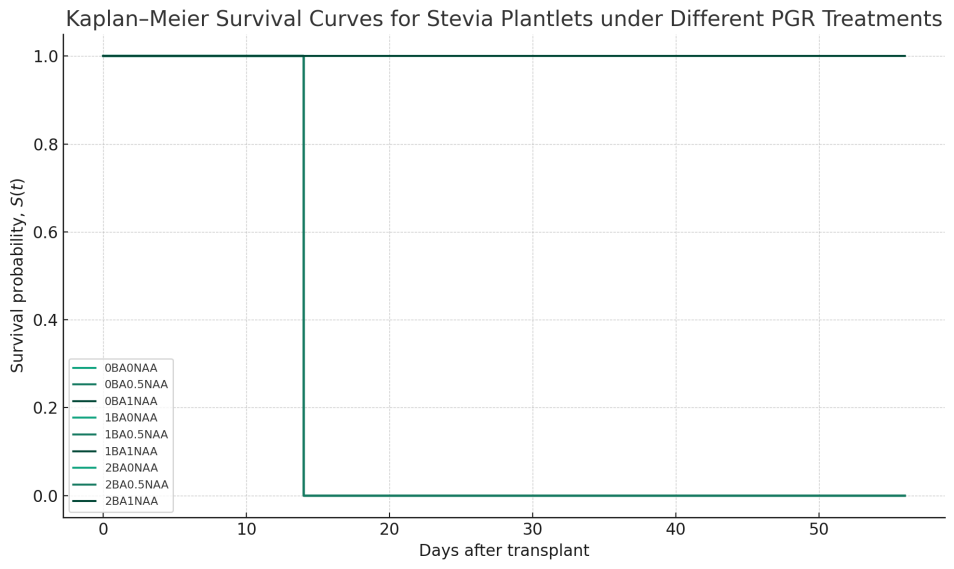
Figure 5 Kaplan-Meier survival curves for Stevia plantlets (n = 8 per treatment) over a 56‑day post‑transplant period. The y‑axis shows survival probability, S(t), and the x‑axis shows days after transplant. Most treatments exhibit a steep drop in survival probability from 1.0 to 0.0 around Day 14, indicating a high early mortality rate.
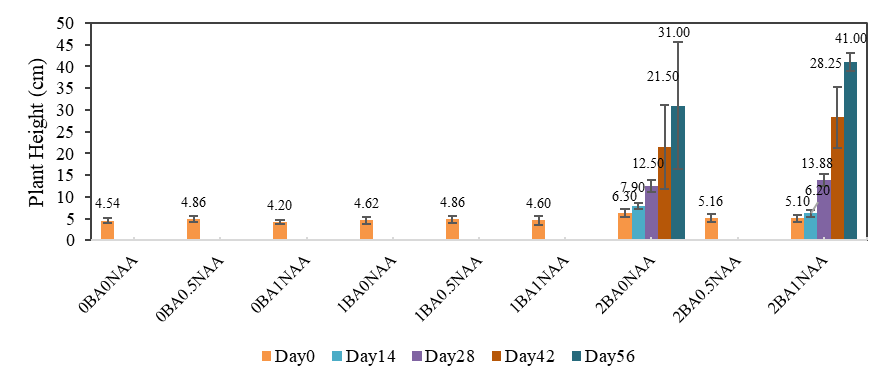
Figure 6 Changes in plant height of Stevia rebaudiana at 0, 14, 28, 42, and 56 days after transplanting to an NFT system. In vitro plantlets were pre‑treated on MS medium containing various BA + NAA concentrations and subsequently rooted on hormone‑free MS (3% sucrose) before transplanting. Plant height was measured on 4 randomly selected plants out of the eight transplanted per treatment.
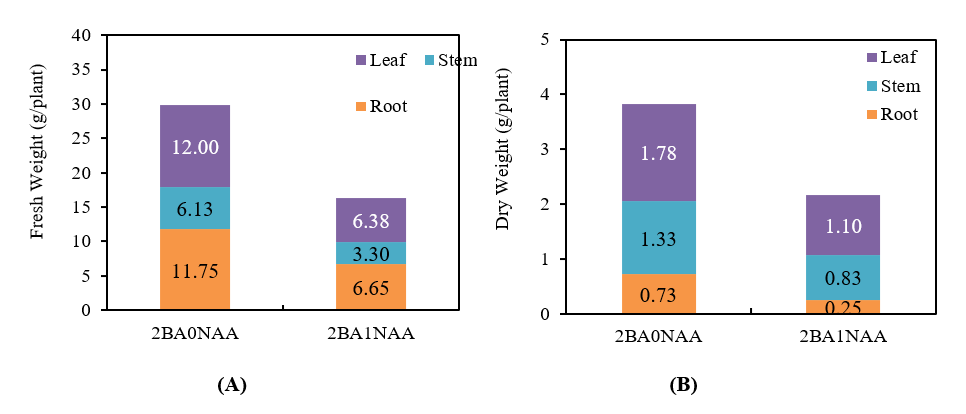
Figure 7 Effects of 2BA0NAA and 2BA1NAA on fresh weight (A) and dry weight (B) of Stevia rebaudiana plants. The biomass was categorized into leaf, stem, and root. Data are presented as mean values for each plant part under different treatments (n = 4).

Figure 8 Morphology of stevia grown on different media: (A) 2BA0NAA, (B) 2BA1NAA.
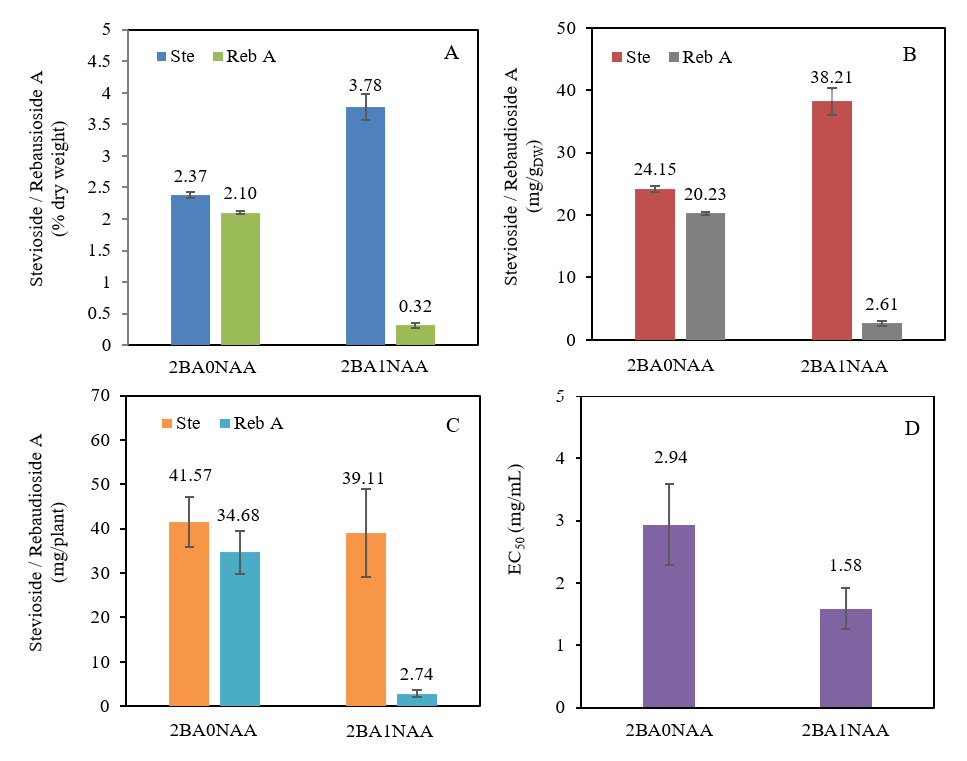
Figure 9 Effects of 2BA0NAA and 2BA1NAA on the bioactive properties of Stevia rebaudiana. (A) Percentage of stevioside (Ste) and rebaudioside A (Reb A) in leaves (% dry weight). (B) Active compound content per dry weight (mg/gDW). (C) Stevioside and rebaudioside A content per plant (mg/plant). (D) Antioxidant activity expressed as EC₅₀ (mg/mL). Values represent mean ± SD (n = 3). Treatments significantly affected compound contents and EC₅₀ values.
Conclusions
This study successfully developed an optimized in vitro culture protocol for Stevia rebaudiana, enhancing seed germination, shoot multiplication, and post-transplantation performance. Culturing seeds on full-strength Murashige and Skoog (MS) medium supplemented with 3% sucrose achieved a maximum germination rate of 9.5%. Shoot multiplication was significantly promoted by the addition of 2 mg/L benzyl adenine (BA) to the culture medium, while root formation was best supported by hormone-free MS medium. A structured acclimatization process ensured 100% survival of plantlets after greenhouse transplantation. Importantly, plants regenerated from media supplemented with 2 mg/L BA alone (2BA0NAA) exhibited superior biomass accumulation and a more favorable sweetener profile, characterized by a higher rebaudioside A content and a lower stevioside-to-rebaudioside A ratio, compared to those treated with combined BA and NAA. These findings highlight the crucial role of growth regulator optimization during micropropagation not only in improving propagation efficiency but also in enhancing the phytochemical quality of stevia plants. The established protocol offers a scalable and sustainable approach for commercial stevia production with improved plant survival, yield, and sweetener composition.
Declaration of Generative AI in Scientific Writing
In preparing this manuscript, the authors used a generative AI tool (e.g., ChatGPT) strictly for grammar checking and language editing. The AI was not involved in content generation or data interpretation. The authors take full responsibility for the content and conclusions of this article.
CRediT Author Statement
Kittiya Thongjan: Investigation; Formal analysis; Writing - Original Draft; Visualization. Tassanai Jaruwattanaphan: Methodology; Resources; Writing - Original draft preparation. Benya Manochai: Supervision, Conceptualization, Methodology, Validation; Writing - Reviewing and Editing; Funding acquisition.
References
[1] R Lemus-Mondaca, A Vega-Gálvez, L Zura-Bravo and K Ah-Hen. Stevia rebaudiana Bertoni, source of a high-potency natural sweetener: A comprehensive review on the biochemical, nutritional and functional aspects. Food Chemistry 2012. https://doi.org/10.1016/j.foodchem.2011.11.140
[2] SK Goyal, Samsher and RK Goyal. Stevia (Stevia rebaudiana) a bio-sweetener: A review. International Journal of Food Sciences and Nutrition 2010; 61(1), 1-10.
[3] AS Dacome, CC da Silva, CEM da Costa, JD Fontana, J Adelmann and SC da Costa. Sweet diterpenic glycosides balance of a new cultivar of Stevia rebaudiana (Bert.) Bertoni: Isolation and quantitative distribution by chromatographic, spectroscopic, and electrophoretic methods. Process Biochemistry 2005; 40(11), 3587-3594.
[4] Department of Agricultural Extension. Report on stevia production in Thailand (in Thai), Available at: http://www.agriinfo.doae.go.th/year63/plant/rortor/herb/หญ้าหวาน.pdf, accessed January 2025.
[5] Customs Department. Statistics of imports: Stevia extract (in Thai), Available at: https://www.customs.go.th, accessed January 2025.
[6] J Duangsong and S Theerathammakorn. Evaluation and reduction of the carbon footprints associated with steviol glycoside production. ASEAN Journal of Scientific and Technological Reports 2024; 27(3), e252464.
[7] A Yadav, S Singh, D Dhyani and PS Ahsuja. A review on the improvement of Stevia [Stevia rebaudiana (Bertoni)]. Canadian Journal of Plant Science 2011; 91(1), 1-27.
[8] R Žukienė, R Žūkienė and K Ramanauskienė. Factors affecting organogenesis of Stevia rebaudiana and in vitro accumulation of steviol glycosides. Zemdirbyste-Agriculture 2020; 107(2), 171-178.
[9] MR Islam, MT Jahan and MA Islam. Establishment of a suitable protocol for clonal propagation of Stevia rebaudiana and creating its advanced lines using gamma-ray. Journal of Science and Technology Research 2024; 6(1), 99-107.
[10] MSS Mim, H Jamal, S Jafrin, S Yasmin, KM Nasiruddin and F Khatun. Development of a protocol for in-vitro regeneration of Stevia rebaudiana Bertoni. Asian Journal of Biotechnology and Bioresource Technology 2024; 10(4), 133-141.
[11] LA Rodríguez-Páez, YY Pineda-Rodriguez, MF Pompelli, AM Jimenez-Ramirez, OJ Genes-Avilez, J Jaraba-Navas, A Jarma-Orozco, EM Combatt-Caballero, LE Oviedo Zumaqué, IE Suarez-Padron, MI Oloriz-Ortega and N Veitía Rodríguez. Micropropagation protocols for three elite genotypes of Stevia rebaudiana Bertoni. Horticulturae 2024; 10(4), 404
[12] N Jadid, S Anggraeni, MRN Ramadani, M Arieny and F Mas’ud. In vitro propagation of Indonesian Stevia rebaudiana genotype using axenic nodal segments. BMC Research Notes 2024; 17(1), 45.
[13] JR Acosta-Motos, L Noguera-Vera, G Barba-Espín, A Piqueras and JA Hernández. Antioxidant metabolism and chlorophyll fluorescence during the acclimatisation to ex vitro conditions of micro propagated Stevia rebaudiana Bertoni plants. Antioxidants 2019; 8(12), 615.
[14] M Rezvankhah, H Askari, M Tohidfar and H Rezadoost. Economic micropropagation of Stevia rebaudiana Bertoni and evaluation of in vitro cultures in order to improve steviol glycosides. Scientia Horticulturae 2022; 305, 111372.
[15] K Chotikadachanarong and S Dheeranupattana. Micropropagation and acclimatization of Stevia rebaudiana Bertoni. Pakistan Journal of Biological Sciences 2013; 16(17), 887-890.
[16] M Thiyagarajan and P Venkatachalam. Large scale in vitro propagation of Stevia rebaudiana (Bert.) for commercial application: Pharmaceutically important and antidiabetic medicinal herb. Industrial Crops and Products 2012; 37(1), 111-117.
[17] S Yesmin. In vitro micropropagation of Stevia rebaudiana Bertoni. Plant Tissue Culture & Biotechnology 2019; 57, 23-28.
[18] N Aman, F Hadi, SA Khalil, R Zamir and N Ahmad. Efficient regeneration for enhanced steviol glycosides production in Stevia rebaudiana (Bertoni). Comptes Rendus Biologies 2013; 336(10), 486-492.
[19] B Röck-Okuyucu, M Bayraktar, IH Akgun and A Gurel. Plant growth regulator effects on in vitro propagation and stevioside production in Stevia rebaudiana Bertoni. HortScience 2016; 51(12), 1573-1580.
[20] O Polivanova and V Bedarev. Hyperhydricity in plant tissue culture. Plants 2022; 11(23), 3313.
[21] NASA Langley Research Center. NASA Prediction of Worldwide Energy Resource (POWER) project. NASA Earth Science/Applied Science Program, 2024. Available at: https://power.larc.nasa.gov, accessed August 2025.
[22] Joint FAO/WHO Expert Committee on Food Additives. Compendium of food additive specifications (FAO JECFA Monographs 10), Available at: https://openknowledge.fao.org/server/api/core/bitstreams/b25ac484-96a0-4d84-b311-a6b1a66714e6/content, accessed August 2025.
[23] P Duvivier, PC Hsieh, PY Lai and AL Charles. Evaluation of drying methods on antioxidant activity, total phenolic and total carotenoid contents of sweet potato (Ipomoea batatas (L.) Lam.) var. Tainong 73. Journal of International Cooperation 2008; 3(2), 73-86.
[24] MZK Roni, MS Islam and K Shimasaki. In vitro seed germination and tracking the seedling growth of eustoma. New Zealand Journal of Crop and Horticultural Science 2018; 46(3), 224-242.
[25] S Pawar, V Khandagale, V Jambhale, A Jadhav and B Pawar. In vitro regeneration studies in Stevia through nodal segment and shoot tip. Bioscan 2015; 10(3), 1007-1010.
[26] S Amien, AZ Azhari, C Bakti and H Maulana. The effect of shoot explant types of eleven stevia (Stevia rebaudiana Bertoni) accessions on in vitro growth. Jurnal Agro 2021; 8(1), 142-152.
[27] AK Ghose, SNA Abdullah, MA Md Hatta and PEM Wahab. In vitro regeneration of Stevia (Stevia rebaudiana Bertoni) and evaluation of the impacts of growth media nutrients on the biosynthesis of steviol glycosides (SGs). Agronomy 2022; 12(8), 1957.
[28] M Raspor, V Motyka, AR Kaleri, S Ninković, L Tubić, A Cingel and T Ćosić. Integrating the roles for cytokinin and auxin in de novo shoot organogenesis: From hormone uptake to signaling outputs. International Journal of Molecular Sciences 2021; 22(16), 8554.
[29] KR Mridula, S Parthibhan, TS Kumar, AS Rao and MV Rao. In vitro micropropagation of Tinospora cordifolia (Willd.) Miers from shoot tip explants. Agriculture and Natural Resources 2019; 53(5), 449-456.
[30] H Hansen and K Grossmann. Auxin-induced ethylene triggers abscisic acid biosynthesis and growth inhibition in cleavers (Galium aparine). Plant Physiology 2000; 124(3), 1437-1448.
[31] S Verma, A Singh, A Kumari and R Sharma. Advances in steviol glycosides biosynthesis and their regulation in Stevia rebaudiana (Bertoni). Plants 2022; 105(13), 5367-5381.
[32] P Biswas, A Kumari, A Modi and N Kumar. Improvement and regulation of steviol glycoside biosynthesis in Stevia rebaudiana Bertoni. Gene 2024; 891, 147809.
[33] W Li, S Lin, R Wang, C Chen, L Ni and Z Liang. Regulation of plant hormones on the secondary metabolism of medicinal plants. Medicinal Plant Biology 2025; 4, e020.
[34] T Ouriagli, A Amnay, SM Raoui, F Errachidi, FO Chahdi, B Bennani, YK Rodi and R Chabir. Antioxidant activities of steviol glycosides from moroccan cultivated stevia Rebaudiana bertoni leaves: An in vitro study. BIO Web of Conferences 2024; 115, 07002.