
Trends
Sci.
2026;
23(6):
11942
Therapeutic Effects of Bromelain on Diabetic Wound Healing: Analysis of Cytokine Levels TNF-α, IL-10, Collagen, Epithelial Thickness, and Angiogenesis in Wistar Rats
Cemy
Nur Fitria1,4,*,
![]() ,
Soetrisno1,
,
Soetrisno1,
![]() ,
Ahmad Yunus2,
,
Ahmad Yunus2,
![]() ,
Brian Wasita1,
,
Brian Wasita1,
![]() ,
,
Vitri
Widyaningsih3,
![]() ,
Tatar Sumandjar1 and Paramasari
Dirgahayu1,
,
Tatar Sumandjar1 and Paramasari
Dirgahayu1,
![]()
1Medicine Sciences Doctoral Study Program, Faculty of Medicine, Universitas Sebelas Maret, Surakarta, Indonesia
2Plant Biotechnology study program, Faculty of Agrotechnology, Universitas Sebelas Maret, Surakarta, Indonesia
3Public Health Doctoral Study Program, Faculty of Medicine, Universitas Sebelas Maret, Surakarta, Indonesia
4Nursing Study Program, Faculty of Health Sciences, Universitas Muhammadiyah PKU, Surakarta, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 18 September 2025, Revised: 1 November 2025, Accepted: 10 November 2025, Published: 30 January 2026
Abstract
Introduction: Diabetes mellitus (DM) can cause resident microglial cells to become activated and transform into an amoeboid form. Activated microglial cells then produce reactive oxygen species (ROS), reactive nitrogen species, interleukin (IL)-1β, tumor necrosis factor (TNF)-α, and other pro-inflammatory and cytotoxic substances, significantly promoting the progression of DM. Bromelain, a proteolytic enzyme derived from Ananas comosus, has demonstrated anti-inflammatory and wound-healing properties, indicating its potential as a topical therapeutic agent for diabetic wounds. This study aimed to evaluate the effects of topical bromelain on diabetic wound healing in Wistar rats by assessing TNF-α, IL-10, collagen deposition, epithelial thickness, and angiogenesis. Materials and methods: A post-test-only controlled experimental design was employed. Twenty-four male Wistar rats (Rattus norvegicus), aged 2 - 3 months and weighing 150 - 200 g, were randomly assigned to 6 groups. Data were analyzed statistically, and hypothesis testing using 1-way analysis of variance (ANOVA). Results and discussion: The calculation of the average value of the DM group with bromelain enzyme cream intervention compared to the DM group without intervention which was used as a negative control resulted in a decrease in TNF-α of around 15.66% and an increase in IL-10 of around 31.26%. Histopathological analysis showed greater collagen deposition, thicker epithelium, and enhanced angiogenesis in bromelain-treated groups compared with controls (p < 0.05). These results indicate that bromelain accelerates diabetic wound healing through immunomodulation and tissue regeneration. Conclusions: Bromelain enzyme has potential as an alternative topical therapy for anti-inflammatory and wound healing in DM.
Keywords: Bromelain, Diabetic wound healing, TNF-α, IL-10, Collagen, Epithelial thickness, Angiogenesis
Introduction
Diabetes mellitus (DM) is a chronic, fatal disease and can worsen the quality of life of sufferers because it causes long-term complications such as coronary artery disease, diabetic neuropathy, nephropathy, retinopathy, cellulitis, and sexual dysfunction [1]. One of the complications caused by DM is diabetic foot ulcers (DFU), with a percentage of 25% affecting DM patients throughout their lifetime [2], where around 15% - 20% of DFU cases require amputation [3,4]. Diabetic foot ulcers (DFUs) are defined as ulcers identified through wounds on the feet of diabetic patients associated with nerve disorders (peripheral neuropathy) and blood circulation (macroangiopathy and microangiopathy) [5]. Wounds suffered by DM sufferers are further aggravated and become more chronic due to impaired wound healing due to dysregulation of standard repair mechanisms due to increased oxidative stress, prolonged inflammatory response, and impaired re-epithelialization [6].
DM occurs when the beta cells in the pancreas are damaged by an autoimmune process, preventing them from producing insulin. One of the hallmarks of DM is hyperglycemia, which is caused by the inability of insulin-dependent tissues to utilize glucose and, in part, by accelerated gluconeogenesis from amino acids derived from muscle protein [7]. Hyperglycemia can stimulate the activation of inflammatory pathways, oxidative stress, and apoptosis [8]. Hyperglycemia in diabetes accumulates advanced glycation end products (AGEs), complex compounds formed through the glycation of amino acids, lipids, and DNA molecules. AGEs interact with their receptor (RAGE) to increase cellular oxidant stress and exacerbate pro-inflammatory cytokine production. Thus, AGEs play a role in promoting the activation of inflammatory processes that can lead to tissue malfunction [6]. These pathological alterations contribute to chronic wound complications. Consequently, elucidating the underlying pathophysiological mechanisms and developing effective therapeutic strategies to enhance diabetic wound repair remain critical unmet needs in clinical research [9].
The primary challenge in diabetic wound healing lies in the impaired progression of the typical healing cascade, which encompasses the hemostasis, inflammatory, proliferative, and remodeling phases [10]. Following wound injury, neutrophils play a crucial role in infection control by activating adhesion molecules, secreting Growth Factors (GFs) to promote proliferation, and releasing proteases that facilitate Extracellular Matrix (ECM) degradation. Circulating monocytes differentiate into pro-inflammatory M1 macrophages, eliminating bacteria, apoptotic cells, and tissue debris via phagocytosis while expressing inflammatory mediators and cytokines. Mast cells rapidly respond to tissue damage by releasing cytokines and proteinases, thereby recruiting neutrophils and enhancing ECM breakdown. In the late inflammatory phase, T lymphocytes act as key modulators in tissue repair [3].
The inflammatory phase concludes when macrophages polarize into the anti-inflammatory phenotype (M2), facilitating the transition to the proliferative phase of wound healing. M2 macrophages secrete various anti-inflammatory mediators and protease inhibitors [3]. In diabetic foot ulcers (DFUs), persistent dominance of pro-inflammatory macrophages (M1) over the M2 phenotype indicates chronic inflammation [11]. Chronic inflammation in diabetic wounds creates a microenvironment conducive to bacterial colonization, leading to infection, tissue damage, and prolonged inflammatory responses. Furthermore, the rising prevalence of antibiotic-resistant bacteria has significantly diminished the efficacy of conventional antibiotic therapies [12].
Conventional management of diabetic wounds primarily involves medical interventions such as debridement (surgical removal of necrotic tissue) and maintenance of a moist wound environment with adequate blood supply. Necrotic tissue serves as a medium for bacterial proliferation, while persistent infection and tissue hypoxia are key contributors to chronic wound progression. Incomplete microbial eradication may prolong the inflammatory phase and impair healing [13]. Topical therapies, including localized antibiotic administration and advanced wound dressings (e.g., films, fibers, and hydrogels), play a critical role in infection control and microenvironment modulation [12].
A promising therapeutic alternative is bromelain, a multifunctional enzyme exhibiting proteolytic, antibacterial, anti-inflammatory, and anti-edema properties [14]. Bromelain with relatively high concentrations can be obtained through the extraction of pineapple stems and flesh [15]. Indonesia is one of the tropical countries that produces pineapples (Ananas comosus), which are members of the Bromeliaceae family. Bromelain can be absorbed in the intestine without degradation and without losing its biological activity [16]. Bromelain significantly reduced blood glucose, serum triglycerides and cholesterol, hepatic malondialdehyde, oxidized LDL (Oxi-LDL), hepatic lysophosphatidic acid (LPA), and the expression of lysophosphatidic acid receptor 1 (LPAR1) and beta-secretase (BACE1) proteins in hepatic tissue. Furthermore, bromelain also increased albumin and total serum protein levels, as well as wound healing rates [17].
Bromelain belongs to a group of protein-digesting enzymes, including cysteine proteinase, sulfhydryl protease, thiolendopeptidase, and several protease inhibitors. It also contains several other enzymes, such as β-glucosidase, cellulase, phosphatase, peroxidase, and β-escarase [18]. In vivo studies demonstrate that bromelain possesses broad-spectrum enzymatic activity, including nonspecific proteolysis, gelatinolytic and collagenase effects, fibrinolysis, as well as phosphatase, nuclease, and peroxidase functions [16]. Furthermore, bromelain exhibits antimicrobial efficacy against bacteria and fungi, highlighting its potential as an adjuvant therapy for wound healing and infection control [19].
Pineapple core extract exhibits a Minimum Inhibitory Concentration (MIC) of 1.25%. Among the tested gel formulations, Gel IV demonstrated the largest inhibition zone diameter (62.5 mm), along with stable viscosity and pH. Nanoparticle encapsulation of the extract may further enhance antimicrobial activity in topical gel formulations [20]. Experimental evidence indicates that bromelain exerts anti-inflammatory effects through multiple mechanisms: (1) upregulation of serum fibrinolytic activity, (2) reduction of plasma fibrinogen levels, and (3) suppression of bradykinin-induced vascular permeability, thereby mitigating edema and pain. Additionally, bromelain downregulates pro-inflammatory mediators, including prostaglandins (PGE2 and thromboxane A2), and modulates surface cell adhesion molecules involved in immune response [21,22].
Topical therapy using the enzyme bromelain has been used for celiac disease [23], polycystic ovary syndrome (PCOS) [24], and to inhibit inflammation in diabetic periodontitis [6]. However, to date, no experimental studies have evaluated the effects of topical bromelain on histopathological parameters of diabetic wound healing. Therefore, this study aims to investigate the effects of topical bromelain (Ananas comosus) on diabetic wound healing in Wistar rats by assessing inflammatory cytokines TNF-α, IL-10, collagen, epithelial thickness, and angiogenesis.
Materials and methods
Materials
The materials utilized in this study included pineapple stems (Ananas comosus), potassium dihydrogen phosphate (KH₂PO₄), sodium hydroxide (NaOH), ethanol, chloroform, diethyl ether, acetate buffer (pH 4), phosphate buffer (pH 7), copper (II) sulfate pentahydrate (CuSO₄·5H₂O), potassium sodium tartrate tetrahydrate (KNaC₄H₄O₆·4H₂O), bovine serum albumin (BSA), tyrosine, casein, and trichloroacetic acid (TCA).
Bromelain enzyme isolation
Pineapple stems (200 g) were cut into small pieces and then mashed with 200 ml of phosphate buffer solution at pH 7.0. This mashed mixture was then filtered to obtain the stem juice, which was then stored in the refrigerator for 24 h. Two layers formed: The top layer was water and the bottom layer was extract. The resulting extract was centrifuged at 3,500 rpm for 15 min to obtain 3 layers. The second layer, a colloid containing the bromelain enzyme, was removed and freeze-dried at −57 °C for 22 h to obtain the bromelain enzyme. The resulting mixture was then cooled to 4 °C for 24 h to prevent damage and maintain the stability of the enzyme, as well as to maintain the activity of the bromelain enzyme. Keeping the extraction process cold also protects the enzyme from denaturation and degradation. This is because enzymes are structurally very labile and highly susceptible to microbial degradation. Denaturation and degradation can be minimized by keeping the enzyme cold at 4 °C [25].
Protein concentration measurement
Evaluation of the bromelain enzyme protein concentration used the visible spectrophotometry (Biuret) method. A 10-mL volumetric flask containing 4 mL of 1% bromelain enzyme solution was added to 6 mL of Biuret reagent and allowed to stand for approximately 20 min until a purple color formed. The absorbance was then measured at a wavelength of 547 nm. The sample absorbance value was substituted into the Y value of the BSA standard curve to obtain the x value, which represents the sample protein concentration [26].
Determination of enzyme activity
Quantitative enzyme activity tests measure both cellular and extracellular activity by measuring the initial reaction velocity (V0). Under these conditions, the velocity is directly proportional to the enzyme being tested. To obtain initial velocity data, the substrate concentration must significantly exceed the enzyme concentration, ensuring the enzyme is saturated with substrate. The amount of substrate depleted during the test should be less than 10%, so that the remaining substrate concentration remains essentially unchanged.
Protease activity is determined by reacting a protease extract sample with a casein substrate. Protease activity is expressed as a concentration based on the amount of tyrosine released, which is read at 275 nm.

Preparation of topical bromelain cream 10%, 12.5% and 15%
Formulation of pineapple stem extract cream preparation (Ananas comosus [L] Merr. as follows [27]:
Table 1 Topical bromelain cream 10%, 12.5% and 15%.
Formula |
||||||
|
|
Negative control (%) |
F1 |
F2 |
F3 |
F4 |
Composition |
Function |
7.5 |
10 |
12.5 |
15 |
|
Pineapple stem extract |
Active ingredient |
- |
|
|
|
|
Oil phase (Phase A) |
||||||
Stearic acid |
Base |
15 |
15 |
15 |
15 |
15 |
Cera alba |
Emulsifier |
2 |
2 |
2 |
2 |
2 |
Vaseline alba |
Emollient |
8 |
8 |
8 |
8 |
8 |
Emulsifying Wax |
Emulsifier |
1.5 |
1.5 |
1.5 |
1.5 |
1.5 |
Nipasol |
Preservative |
0.015 |
0.015 |
0.015 |
0.015 |
0.015 |
Water phase (Phase B) |
||||||
Propylene glycol |
Humectant |
8 |
8 |
8 |
8 |
8 |
Nipagin |
Preservative |
0.1 |
0.1 |
0.1 |
0.1 |
0.1 |
Aquadest |
Solvent |
65.385 |
57.885 |
55.385 |
52.885 |
50.385 |
The study was conducted by making O/W cream using different concentrations of 7.5%, 10%, and 12.5%, then an evaluation of the cream preparation was carried out, including a hedonic test to see between the 4 formula concentrations with one of the formulas as a control. At room temperature, the preparation can be said to be physically stable. The cream is physically stable in terms of homogeneity and pH parameters [28].
Experimental design
The study began with the preparation of research equipment and materials, which lasted for 5 days, including initial care of the mice prior to streptozotocin induction. Wistar rats were kept in 6 groups of 4 rats each, fed 15 g per rat per day, and water was provided in a special container provided for each cage. For each contact with the rats, the researchers used sterile gloves for aseptic precautions.
The first stage of the study involved dividing the rats into 6 groups: one group of uninjected rats and 5 groups of STZ-induced rats. Diabetes was induced with a single intraperitoneal injection of STZ (45 mg/kg-BW) 15 min after intraperitoneal nicotinamide (110 mg/kg-BW) administration. Diabetes induction was confirmed after 72 h with a fasting blood glucose (FBG) level of ≥ 250 mg/dL [29]. NA (Nicotinamide) was dissolved in PBS (Phosphate Buffered Saline) and STZ was dissolved in citrate buffer with a pH of 4.5 [30]. Blood glucose levels were measured using a glucometer. Mice were diagnosed with diabetes if their blood glucose levels were ≥ 200 mg/dL [30]. Streptozotocin was injected intraperitoneally at a dose of 45 mg/kg body weight. Before being induced with STZ, the mice were fasted for 12 h and their blood glucose levels were measured [31].
This study investigated the therapeutic effects of topical bromelain cream, derived from pineapple (Ananas comosus), on wound healing in male Wistar rats (Rattus norvegicus). The animals, aged approximately 2 - 3 months and weighing 150 - 200 g, were divided into 6 groups (n = 4 per group), with an additional 20% sample size (total N = 24) to account for potential attrition. Randomization allocated the rats as follows: Group 1 labeled K (N) = Normal control (non-DM wounds without therapy), Group II labeled K (-) = negative control of type 2 DM wounds without treatment, Group III labeled K (+) = type 2 DM wounds + administration of glibendamide 0.09 mg/200 g + 10% Providone Iodine, Group IV labeled (P1) = type 2 DM wounds + administration of gkibenclamide 0.09 mg/200 g + 10% bromelain enzyme cream, Group V labeled (P2) = type 2 DM wounds + administration of glibendamide 0.09 mg/200 g + 12.5% bromelain enzyme cream, Group VI labeled (P3) = Group III = type 2 DM wounds + administration of glibendamide 0.09 mg/200 g + 15% bromelain enzyme cream.
Statistical analysis
Data were analyzed using Statistical Product and Service Solutions (SPSS) software, version 27.0 for Windows (IBM Corp., USA). Normal distribution was assessed using the Shapiro-Wilk test. For hypothesis testing, a 1-way analysis of variance (ANOVA) was applied.
Results and discussion
ELISA analysis of serum TNF-α and IL-10 levels
The ELISA results revealed that the highest mean serum TNF-α level was observed in the negative control group (untreated diabetic Wistar rats with wound injury). Similarly, the highest mean IL-10 concentration was also detected in the negative control group.
Table 2 Mean ± Standard Deviation of serum TNF-α and IL-10 levels.
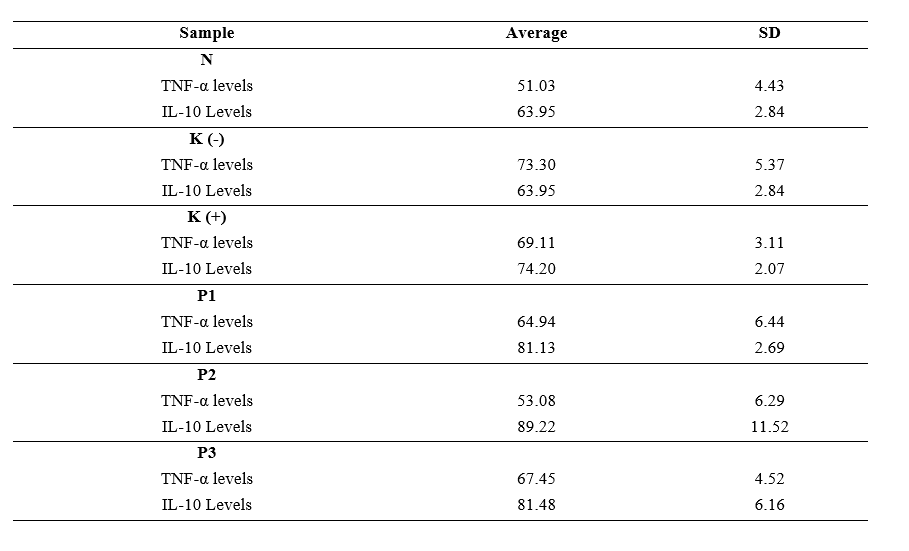
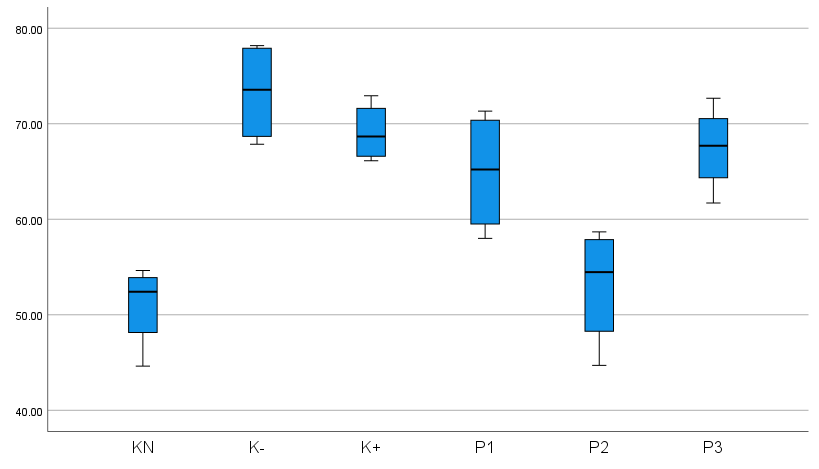
Figure 1 Differences in TNF-α levels between groups.
Based on Figure 1 and Table 2 above, it is known that the positive control group K (+) mice had TNF-α levels of 69.11 ± 3.11, in the negative control group K (-) mice, TNF-α levels were obtained with an average of 73.30 ± 4.43, in the non-diabetic wound control group K (N) mice, TNF-α levels were obtained with an average of 51.03 ± 4.43. In treatment group 1 (P1), mice had an average TNF-α level of 64.94 ± 6.44. In treatment group 2 (P2), mice had an average TNF-α level of 53.08 ± 6.29, and in treatment group 3 (P3), mice had an average TNF-α level of 67.45 ± 4.52. These results indicate that the average value of TNF-α levels is the highest in the control group K (-), specifically in Wistar mice with DM wounds that did not receive treatment.
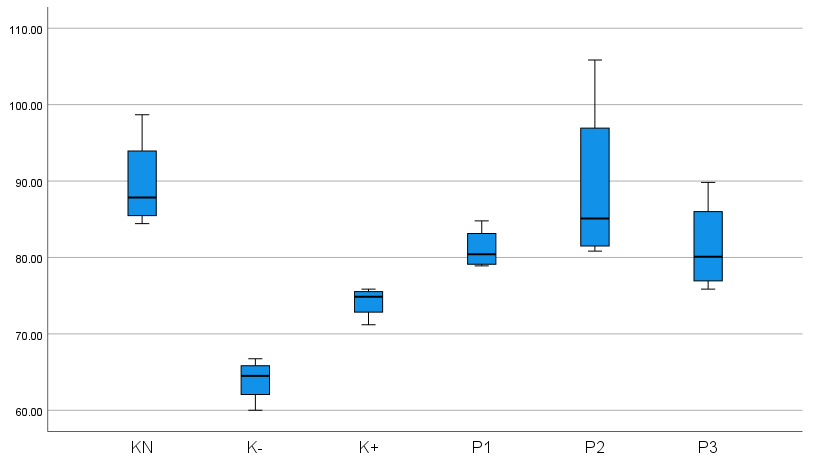
Figure 2 Differences in IL-10 levels between groups.
Based on Figure 2 and Table 2, it is known that the average value of IL-10 levels is the highest in the control group K (-), namely, Wistar rats with DM wounds that did not receive treatment. IL-10 levels in groups P1, P2, and P3 were significantly higher than in K (-). This shows that topical administration of bromelain enzyme cream and oral glibenclamide is more effective in increasing IL-10 levels in rats that have been made with a type 2 DM model, accompanied by diabetic wounds, than administration of 10% providone Iodine. IL-10 levels in P1, P2, and P3 showed that bromelain enzyme cream had an effect at doses of 10%, 12.5%, and 15% in type 2 DM model mice with diabetic wounds, by optimally increasing IL-10 levels.
Epidermal epithelial thickness
The
research data on the variable of epidermal thickness were the
thickness of the epidermis in each treatment group, examined
according to the pathological characteristics of the skin. Epidermal
thickness was observed and measured using morphometric methods,
using a microscope connected to an Optilab device with a 40×
objective lens magnification.
Figure 3 Histopathological examination of the thickness of the epidermal epithelium of wistar rats.
In Figure 3, A = K (N), B = K (-), C = K (+), D = (P1), E = (P2) and F = (P3). After 10 days of treatment, the thickness of the epidermal epithelium of the skin of the K (-) group was the lowest compared to that of K (N) group. The K (+), P1, P2, and P3 groups increased approaching the K (N) group. The results of the HE test of epidermal thickness of the epithelium showed that the K (N) group had an average value of 66.04 µm; the control group K (-) 22.08 µm; the control group K (+) 35.36 µm; the treatment group P1 with a dose of 10% bromelain enzyme cream had an average value of 42.99 µm; the treatment group P2 with a dose of 12.5% bromelain enzyme cream had an average value of 62.68 µm; and the treatment group P1 with a dose of 15% bromelain enzyme cream had an average value of 39.72 µm.
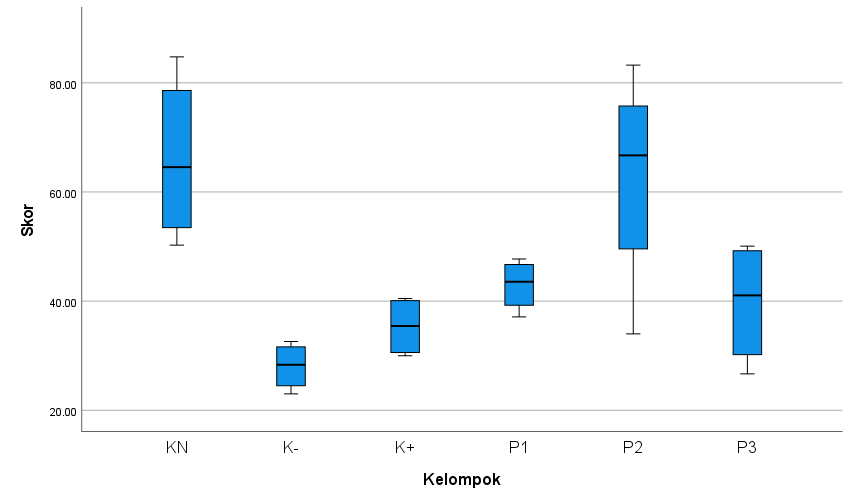
Figure 4 Differences in epidermal epithelial thickness between groups.
The mean epidermal thickness in the non-diabetic control group K (N) was significantly higher than in other groups, indicating enhanced epithelial regeneration in non-diabetic Wistar rats compared to diabetic wounds. In contrast, the untreated diabetic control group K (-) exhibited the lowest epithelial thickness, suggesting impaired wound healing in diabetic rats without intervention. Among the treatment groups, P2 (topical 12.5% bromelain cream + oral glibenclamid 0.09 mg/kg BW) demonstrated superior epithelial thickening compared to the positive control K (+), negative control K (-), P1, and P3, highlighting the optimal therapeutic effect of this combined treatment. Across all groups, the mean epithelial thickness scores ranged from 28.08 (minimum) to 66.04 (maximum), further supporting the efficacy of bromelain in promoting diabetic wound repair.
Collagen with trichome masson’s
Collagen deposition was assessed using masson’s trichrome staining. Prepared slides were evaluated by validated observers, with each examiner scoring 4 microscopic fields per slide. The scoring criteria included nuclear integrity, cytoplasmic staining uniformity, and color homogeneity. The final score for each slide was derived from the average of the 4 fields.
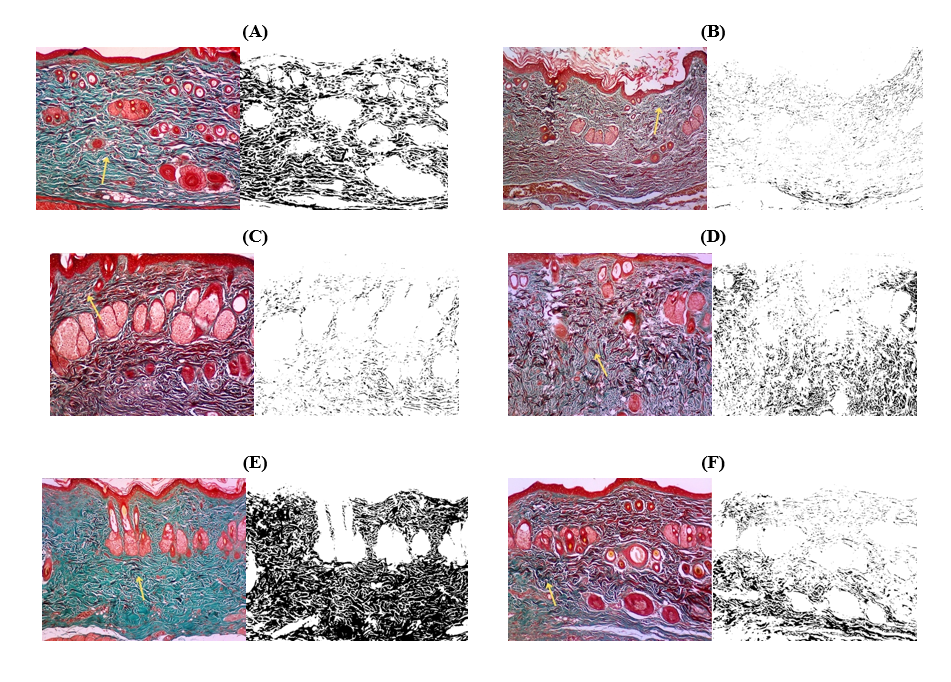
Figure 5 Overview of histopathological examination results of collagen with trichome masson’s.
The results of histological examination of collagen with trichome masson’s showed that the K (N) group had an average value of 26.27 µm, K (-) an average value of 4.10 µm, K (+) an average value of 5.44 µm, P1 an average value of 14.75 µm, P2 an average value of 22.21 µm, and P3 an average value of 8.17 µm.
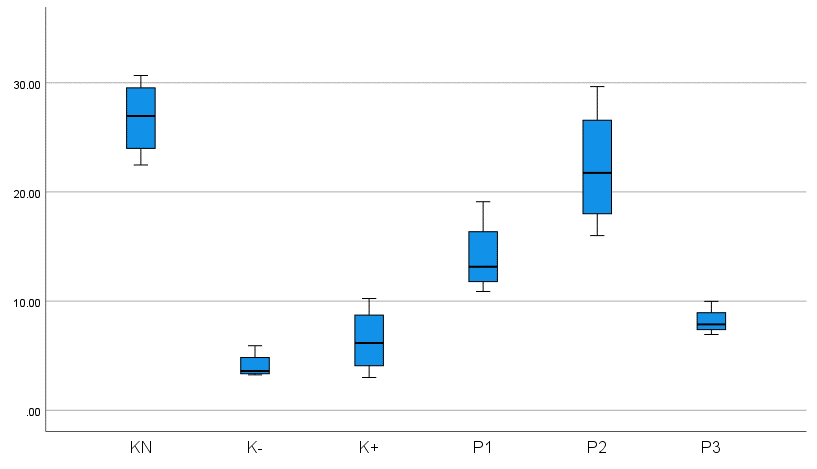
Figure 6 Differences in collagen between groups.
The non-diabetic wound group K (N) exhibited the highest mean collagen score, indicating significantly enhanced collagen synthesis in Wistar rats without diabetes. In contrast, the untreated diabetic control group K (-) displayed the lowest score, reflecting impaired collagen deposition. Notably, the P2 group (treated with topical 12.5% bromelain cream and oral glibenclamide [0.09 mg/kg body weight]) demonstrated superior collagen synthesis compared to the diabetic control K (+), untreated diabetic K (-), and other treatment groups (P1, P3). These findings suggest that the combined regimen of bromelain and glibenclamide optimally promotes collagen remodeling in diabetic wounds. The mean collagen density scores across all 6 experimental groups ranged from 28.08 (lowest) to 66.04 (highest), as quantified by histopathological assessment.
Based on the research results presented above, data consistency is seen, namely a decrease in TNF-α expression, an increase in IL-10 levels, and an increase in collagen and epidermal epithelial layer thickness, in all treatment groups, especially group P (2) which had a bromelain enzyme level of 12.5% with the most significant results. Therefore, the research data has met the assumption of homogeneity or normality. Next, a post-Hoc LSD test was carried out to analyze differences in TNF-α levels, IL-10 levels, and epithelial thickness.
Table 3 LSD on TNF-α levels, Hoc LSD on IL-10 levels, LSD on epithelial thickness.
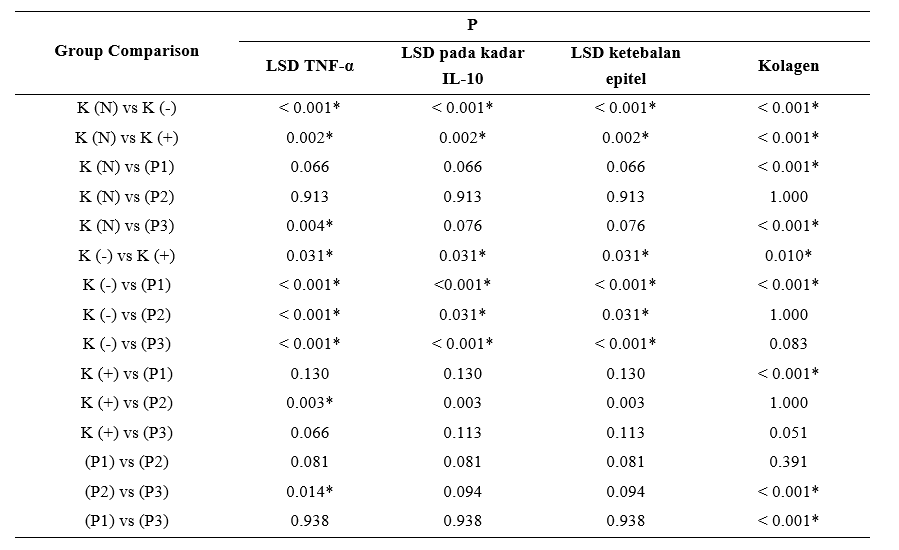
Based on the Post-Hoc test in the table above, it appears that the TNF-α levels of the K (+) and P1, P2, and P3 treatment groups were significantly different from the K (-) group. The TNF-α levels in the K (+) and P3 treatment groups were significantly different from the K (N) group, while the P2 group was significantly different from the K (+) group. This shows that the comparison of the average TNF-α levels in P1 and P3 was not significant, indicated by the p value of the LSD post hoc test above 0.05 (p > 0.05). The results of this LSD post hoc test show that the treatment given had a significant effect on the P2 group. There was a significant difference between the P2 and P3 groups, but not between the P1 and P2 groups. The TNF-α levels of K (-) were significantly higher than those of K (N). This indicates that the mouse model with wounds without type 2 DM exhibited the lowest TNF-α levels compared to K (+) and K (-), likely due to the absence of chronic inflammation, which allowed for a normal wound healing process. The IL-10 levels of the K (+) and P1, P2, and P3 treatment groups were significantly different from the K (-) group. The IL-10 levels in the P2 treatment group were significantly different from the K (+) group, while the P2 group was not significantly different from the K (N) group. This shows that the comparison of the average IL-10 levels in P1 and P3 was not significant, indicated by the p value of the LSD post hoc test above 0.05 (p > 0.05). The results of this LSD post hoc test showed that the treatment given had a significant effect on the P2 group. There was no significant difference in the P2 group compared to P3, and similarly, no significant difference was found in the P1 group compared to P2. The IL-10 levels in K (-) mice were significantly higher than in K (N) mice. This indicates that the mouse model with wounds without type 2 DM exhibited the highest IL-10 levels compared to K (+) and K (-) mice, likely due to the absence of chronic inflammation, which allowed for a normal wound healing process. The thickness of the epidermal epithelium of the K (+) and P1, P2, and P3 treatment groups was significantly different from that of the K (-) group. The thickness of the epidermal epithelium in the P2 treatment group was significantly different from the K (+) group, while the P2 group was not significantly different from the K (N) group. This shows that the comparison of the average thickness of the epidermal epithelium in P1 and P3 was not significant, indicated by the p value of the LSD post hoc test above 0.05 (p > 0,05). The results of this LSD post hoc test showed that the treatment given had a significant effect on the P2 group. There was no significant difference in the P2 group compared to P3, and similarly, no significant difference was found in the P1 group compared to P2. The thickness of the epidermal epithelium of K (-) was significantly higher than that of K (N) group. This indicates that the rat model with wounds without type 2 DM exhibited the highest epidermal epithelium thickness compared to K (+) and K (-), likely due to the absence of chronic inflammation, which allowed for a normal wound healing process.
Angiogenesis with CD31
Angiogenesis was assessed immunohistochemically using CD31 staining. Both the validator and the authors independently evaluated each specimen, analyzing 4 microscopic fields per slide. Scoring was based on nuclear integrity, cytoplasmic staining intensity, and color uniformity, with the final angiogenesis score derived from the average of the 4 highest-scoring fields across all 6 experimental groups.
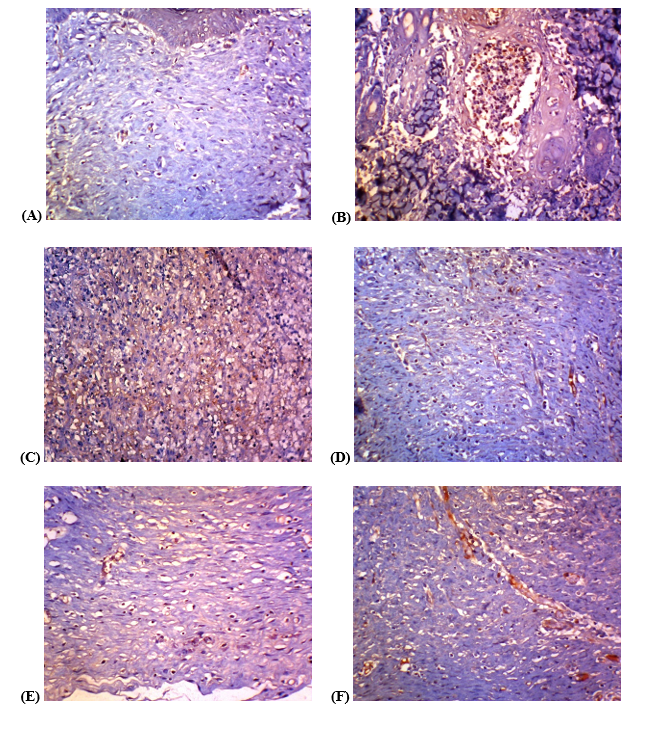
Figure 7 Histopathological examination results of Angiogenesis with CD31.
Angiogenesis test with CD 31, control group A = K (N) with a value of 25.12, control group B = K (-) with a value of 9.19, control group C = K (+) with a value of 10,81, treatment group D = P1 dose of 10% bromelain enzyme cream value of 15.49, treatment group E = P2 dose of 12.5% bromelain enzyme cream value of 21.76, treatment group F = P3 d dose of 15% bromelain enzyme cream value of 14.81.
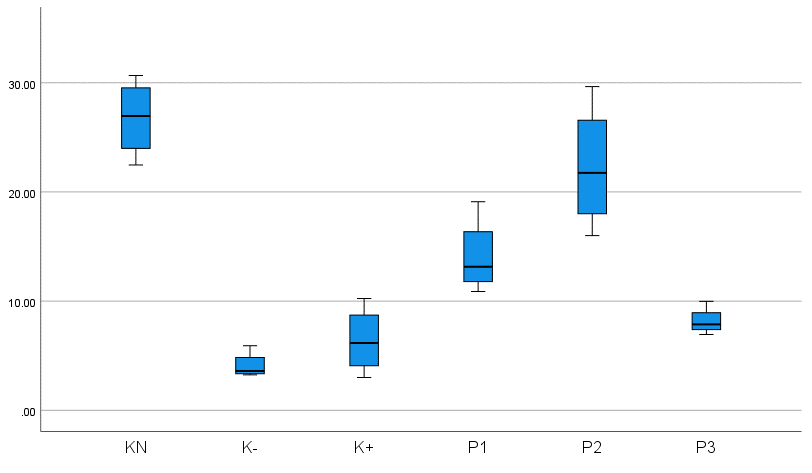
Figure 8 Differences in angiogenesis between groups.
The non-diabetic wound group K (N) exhibited the highest mean angiogenesis score, reflecting robust vascularization. In contrast, the untreated diabetic group K (-) showed the lowest score, indicating impaired angiogenesis. Notably, the P2 group (treated with 12.5% bromelain cream and 0.09 mg/kg oral glibenclamide) demonstrated significantly enhanced angiogenesis compared to the diabetic control K (+), untreated diabetic K (-), P1, and P3 groups, suggesting synergistic efficacy of topical bromelain and systemic glibenclamide in promoting microvascular formation.
Table 4 One-way anova hypothesis test results.


The results of Table 4 of the One Way Anova hypothesis test obtained a significant value of 0.014 (p < 0.05) so the hypothesis is accepted or can be interpreted that there is a substantial difference in the microscopic results of preparations on wound tissue that were tested for angiogenesis with CD31 in the K (N), K (-), K (+), P1, P2, P3 groups in Wistar rats with diabetic wounds. This is based on the results of statistical calculations of the preparations, indicating differences in the results among the 6 groups of staining of Wistar rat wound tissue preparations. This shows that the comparison of the average angiogenesis in P1 is not significant, that angiogenesis in K (-) is substantial to K (N), indicating that the rat model without type 2 DM, accompanied by wounds, has greater angiogenesis than K (-) because K (N) does not experience chronic inflammation, so it experiences a normal wound healing process. Angiogenesis in P1, P2, and P3 was significantly higher than in K (+) and K (-), indicating that bromelain enzyme administration and control with glibenclamide 0.09 mg/kgBW increased angiogenesis in type 2 diabetic rats with diabetic wounds at doses of 10%, 12.5%, and 15%.
Angiogenesis in group P2 was significantly higher than in group P1, P3, and K (+), indicating that topical bromelain enzyme cream and oral glibenclamide 0.09 mg/kgBW were more effective in reducing angiogenesis in rats modeled as type 2 diabetic rats with diabetic wounds at 12.5% than at 10% and 15%. Angiogenesis in P1 and P2 was higher than in P3, indicating that bromelain enzyme cream at doses of 10% and 12.5% was more effective in increasing angiogenesis in rats modeled as type 2 diabetic rats with diabetic wounds than at a dose of 15%. This shows that a 12.5% dose of bromelain enzyme cream with 0.09 mg/kg bw oral glibenclamide has an optimal effect.
DM can be caused by a patient's insulin deficiency or the development of insulin resistance in insulin receptors or at various metabolic stages affected by insulin. Without insulin, the liver lacks the enzymatic capacity to reduce excess blood glucose concentration, leading to hyperglycemia [32]. Excessive blood glucose concentrations can result in glycosylation reactions that can affect the activity, solubility, and degradation of a protein. In the case of HbA, glycosylation occurs through a non-enzymatic reaction between glucose and the N-terminal valine residue on the β-chain of hemoglobin. A Schiff base is formed between glucose and valine, followed by molecular rearrangement until a 1-deoxyfructose molecule binds to the valine. This reaction is enhanced by high glucose concentrations, resulting in HbA1c. Glycosylated hemoglobin produces HbA1c, which is formed spontaneously in erythrocytes through the combination of the terminal –NH2 group on the β-chain of hemoglobin with glucose. The aldehyde group of glucose forms a Schiff base with the –NH2 group. A Schiff base above can rearrange to create a more stable amino ketone bridge.
Non-enzymatic glycosylation reactions and molecular rearrangements occur spontaneously, known as Amadori rearrangements. HbA1c concentrations depend on blood glucose concentrations, and prolonged hyperglycemia can account for more than 12% of total hemoglobin. Patients with DM have high blood glucose concentrations and relatively high HbA1c levels. Glycosylation reactions can also occur in proteins, contributing to the symptoms of diabetes-related medical complications such as coronary heart disease, retinopathy, nephropathy, cataracts, and neuropathy. Increased glycosylation of lens proteins leads to cataracts. Collagen, laminin, vitronectin, and other matrix proteins can undergo glycosylation reactions, leading to impaired biological function, such as the inability to self-assemble to form complexes and the loss of binding ability to other matrix molecules. Glycosylated proteins and lipoproteins can also be captured by receptors on macrophages, leading to the formation of atherosclerotic plaques [7].
Hyperglycemia can stimulate the activation of inflammatory pathways, oxidative stress, and apoptosis. Hyperglycemia in diabetes accumulates advanced glycation end products (AGEs), complex compounds formed through the glycation of amino acids, lipids, and DNA molecules. AGEs interact with their receptor (RAGE) to increase cellular oxidant stress and exacerbate pro-inflammatory cytokine production. The accumulation of AGEs upregulates inflammatory cytokines, particularly interleukin 6 (IL-6) and interleukin 8 (IL-8). IL-6 is involved in the immune response and is responsible for the course of acute-phase inflammation, while IL-8 helps recruit and activate neutrophils to the site of acute inflammation. TNF-α (Tumor Necrosis Factor-alpha) is a key pro-inflammatory cytokine that initiates the inflammatory cascade. In diabetic wounds, dysregulated overexpression of TNF-α exacerbates chronic inflammation and impairs tissue regeneration [33].
Bromelain, a proteolytic enzyme derived from pineapple (Ananas comosus), exhibits significant anti-inflammatory properties, positioning it as a promising alternative to conventional anti-inflammatory drugs. Studies have demonstrated that bromelain effectively suppresses the secretion of key pro-inflammatory cytokines, including IL-1β, IL-6, and TNF-α, particularly under conditions of cytokine overproduction induced by inflammation [34]. Administration of bromelain enzyme has been shown to reduce serum pro-inflammatory IL-6 levels in people living with diabetes [6]. Bromelain enzymes have anti-inflammatory properties that can reduce the production of pro-inflammatory molecules such as TNFα by inhibiting the nuclear factor kappa-B and mitogen-activated protein kinase signaling pathways. Furthermore, bromelain enzymes also activate the SIRT1 pathway by reducing oxidative stress, thereby inhibiting oxidative stress, inflammation, and apoptosis through activation of the SIRT1/AMPK pathway [24].
Bromelain exhibits potent anti-inflammatory properties, mitigating swelling, pain, and inflammation associated with arthritis, sports injuries, acute wounds, and postoperative recovery. Its mechanism involves the suppression of key inflammatory mediators, including cytokines (e.g., IL-1β, TNF-α, IL-6), prostaglandins, and leukotrienes, which are critical in initiating and propagating inflammatory responses [6,35–37]. Furthermore, bromelain modulates the nuclear factor-kappa B (NF-κB) pathway, a central regulator of inflammation and immune responses. By inhibiting NF-κB activation, bromelain downregulates pro-inflammatory gene expression and attenuates inflammatory cascades. The accumulation of AGEs has been shown to upregulate NF-κB signaling. Activation of NF-κB signaling is known to be a key trigger of inflammation because it promotes the secretion of inflammatory cytokines.
Furthermore, pro-inflammatory mediators also influence NF-κB activation through a positive feedback loop, resulting in a repeating cycle that exacerbates inflammatory damage. The accumulation of AGEs also stimulates the phosphorylation of protein kinases such as MAPK and ERK, which are essential modulators of the pro-inflammatory response. Bromelain can reduce inflammation and accelerate wound healing in diabetic patients by inhibiting IL-6 and IL-8 through downregulation of the NF-κB and MAPK pathways [6]. Furthermore, the bromelain enzyme can also reduce the expression and function of NF-κB and cyclooxygenase 2 (COX-2), acting as an inflammatory mediator by converting arachidonic acid into the pro-inflammatory lipid prostaglandin E2, and can modulate other inflammatory mediators such as IFNγ, IFNγ-mediated nitric oxide, TNF-α, IL-1β, and IL-8 [23].
Conversely, IL-10 (Interleukin-10), an anti-inflammatory cytokine, suppresses pro-inflammatory cytokine production and promotes inflammatory resolution. Upregulation of IL-10 facilitates the transition from the inflammatory to the proliferative phase of wound healing [38]. IL-10 also inhibits cytokine production, vascular leakage, and inflammation during Th-1 cytokine-induced delayed-type hypersensitivity [39]. The dynamic interplay between TNF-α and IL-10 reflects a critical biological equilibrium between tissue destruction and repair, which is fundamental to successful wound healing. Molecularly, the bromelain enzyme has anti-inflammatory effects due to its ability to undergo glycosylation at Asn117. A study showed that a bromelain complex can be formed through glycosylation of the Asn117 residue in the bromelain enzyme with polysaccharides via a GlcNAc bonded to the second GlcNAc (β1-4) and a fucose (β1-3), and a mannose bonded to the second GlcNAc (β1-4) and a xylose (β1-2) [40]. This fact suggests that the bromelain enzyme can glycosylate polysaccharides, including glucose, to prevent glycosylation between glucose and hemoglobin, thus preventing hyperglycemia, which can activate inflammatory pathways [41].
Histopathological analysis provides a direct assessment of tissue status during wound healing, particularly regarding epithelial thickness and collagen deposition. Epithelial thickness serves as a critical indicator of re-epithelialization, wherein basal cells migrate and proliferate to achieve wound closure. In diabetic wounds, this phase is significantly delayed [42]. Collagen, predominantly types I and III, constitutes the primary structural protein in the dermis and is a key marker of the remodeling phase. Impaired collagen synthesis and disorganized deposition are hallmarks of chronic wound pathogenesis [43].
The bromelain enzyme can modulate the inflammatory system and the debridement properties of the skin, namely helping to clean wounds from necrotic tissue and bacteria so that the wound bed becomes clean [44]. The study quantified the levels of TNF-α (a pro-inflammatory cytokine) and IL-10 (an anti-inflammatory cytokine), reflecting the dynamic immune response during the inflammatory phase of wound healing. In diabetic wound models treated with topical bromelain cream, TNF-α levels were modulated during the proliferative phase, demonstrating bromelain’s anti-inflammatory role in suppressing excessive inflammation and facilitating tissue repair. This intervention promoted keratinocyte and fibroblast migration, enhancing granulation tissue formation [15,16,18].
Bromelain, a potent chemoresponsive proteolytic enzyme derived from pineapple stems, exhibits antibacterial activity by degrading bacterial membrane proteins [45]. Composed of several thiol endopeptidases, bromelain is extracted and purified through multiple methodologies, with its primary therapeutic application being anti-inflammatory modulation [46]. Its efficacy in soft tissue wound healing is attributed to escharase activity, which facilitates rapid eschar removal, reduces edema and bruising, and accelerates full-thickness wound repair through adequate biological debridement [37].
The proteolytic activity of bromelain facilitates the degradation of edema-associated proteins and extracellular matrix components, reducing fluid accumulation in tissues and alleviating inflammation-induced swelling. These properties underscore its therapeutic potential in conditions characterized by edema and inflammation, such as acute trauma, postoperative care, and joint disorders like arthritis [37]. The proteolytic activity of bromelain contributes to wound healing and tissue repair by facilitating the removal of necrotic tissue and promoting the proliferation of healthy cells [37,47]. Through accelerating inflammation resolution and supporting wounded tissue remodeling, bromelain enhances the body’s capacity to recover from injuries, surgical procedures, and other forms of tissue damage [48–50].
Histopathological examination of collagen was conducted using collagen protein scoring, which predominantly assesses connective tissue deposition in the skin. The Masson’s Trichrome staining method was employed to evaluate hemostatic capacity, platelet interactions, fibronectin activity, enhanced fluid exudation, cellular components, and growth factors, ultimately promoting fibroplasia and epidermal proliferation in diabetic wound models [51].
Histopathological evaluation of epithelial thickness and collagen density serves as a critical indicator of tissue regeneration and cutaneous remodeling processes. The final phase of wound healing involves extensive remodeling, characterized by the deposition and reorganization of collagen and elastin, alongside the differentiation of fibroblasts into myofibroblasts. Over time, myofibroblast activity and re-epithelialization determine the quality and elasticity of healed tissue, as well as the extent of scar formation, which is marked by irregular fibrous tissue proliferation and disorganized collagen fibers. The complex healing response aims to restore both dermal and epidermal structures, ultimately reestablishing skin barrier integrity, elasticity, and functionality. However, impaired healing may result in abnormal scarring, manifesting as hyperactive, erythematous, pruritic, or painful fibrotic tissue, clinically termed hypertrophic scars or keloids [52].
This study demonstrates that bromelain enzyme cream significantly accelerates wound healing by enhancing wound contraction rates, reducing epithelialization time, promoting collagen deposition, and stimulating granulation tissue formation. Effective wound management aims to achieve rapid tissue repair while minimizing secondary damage, ensuring adequate tissue perfusion, optimal oxygenation, and nutrient supply to the wound bed, thereby mitigating risk factors that impede healing and reducing the incidence of infected wounds. An ideal treatment should not only expedite wound closure but also support proper tissue regeneration.
Angiogenesis, a critical process in neovascularization, ensures the delivery of oxygen and nutrients to regenerating tissues during wound healing. In type 2 DM (T2DM), impaired angiogenesis—driven by endothelial dysfunction, chronic inflammation, and oxidative stress—severely compromises wound repair. Diabetic wounds exhibit persistent inflammatory responses, delayed tissue maturation, reduced tensile strength, and diminished angiogenic capacity. Notably, diabetic angiogenesis dysfunction manifests as impaired endothelial cell activity, aberrant vessel formation, and deficient pericyte recruitment [53].
Angiogenesis, the formation of new blood vessels, is a critical process in wound healing, facilitating oxygen and nutrient delivery to injured tissues. CD31 (Platelet Endothelial Cell Adhesion Molecule-1, PECAM-1) and Vascular Endothelial Growth Factor (VEGF) serve as key molecular markers in assessing angiogenic activity. In diabetic wounds, impaired angiogenesis is attributed to oxidative stress and dysregulation of growth factors, contributing to delayed tissue repair. Bromelain exhibits anti-inflammatory and pro-angiogenic properties, which are hypothesized to upregulate vascular endothelial growth factor (VEGF) expression and enhance neovascularization [54]. The pineapple (Ananas comosus) stem contains higher concentrations of bromelain—a proteolytic enzyme—compared to the fruit pulp, attributed to its role as the plant’s metabolic and growth center [55]. This elevated enzymatic activity correlates with increased protein content, underscoring the stem’s therapeutic potential.
The enzyme bromelain can control the number of macrophages infiltrating the wound site. This is done by producing several growth factors, including TGF-β, basic fibroblast growth factor (BFGF), platelet-derived growth factor (PDGF), and vascular endothelial growth factor (VEGF). These factors promote cell proliferation and the production of extracellular matrix molecules by local skin cells. Macrophages play distinct roles in the various phases of the repair response and coordinate the natural sequence of repair phases in the skin, which is necessary for restoring homeostasis and integrity of dense tissue after injury. In the middle phase of the repair response, macrophages are essential for stabilizing vascular structures and transforming granulation tissue into scar tissue. Previous research has shown that macrophages are activated in different ways during physiological healing. This results in the expression of VEGF and TGF-β, which promote wound angiogenesis and myofibroblast differentiation [8,56].
Conclusions
This study demonstrates that topical bromelain cream significantly reduces TNF-α levels while enhancing anti-inflammatory IL-10 cytokine expression in diabetic wound models of Wistar rats. Furthermore, bromelain treatment promotes collagen deposition, angiogenesis, and epithelial thickness, indicating its potential as a therapeutic agent for diabetic wound healing.
The novelty of this study lies in its scientific/biological substance, encompassing: (1) The application of topical bromelain isolated from pineapple (Ananas comosus) stems as a therapeutic agent for type 2 diabetic wounds in a rat model. This formulation remains underexplored, particularly in modulating histopathological tissue remodeling. (2) Comprehensive analysis of bromelain’s anti-inflammatory and antimicrobial effects via cytokine biomarker profiling (TNF-α and IL-10) using ELISA, elucidating its underlying molecular mechanisms. (3) Histopathological and immunohistochemical evaluation of skin/target organ tissues revealed significant improvements in tissue architecture, including enhanced epithelial thickness, collagen density, and reduced inflammatory cell infiltration. These findings provide robust morphological evidence supporting bromelain's therapeutic potential in diabetic wound healing.
Topical bromelain cream holds potential as a therapeutic agent for human wound care, pending further clinical validation. However, this study has limitations, including the absence of safety assessments regarding topical bromelain application. Additionally, the observation period was restricted to the inflammatory and proliferative phases (10 days), during which wounds in both rat groups were fully covered by granulation tissue. Consequently, the remodeling phase post-healing was not evaluated, leaving the potential for scar formation unaddressed. Future studies should investigate the long-term safety profile of bromelain cream and its effects on the remodeling phase to assess scar development.
Declaration of generative AI in scientific writing
The authors declare that no generative AI or AI-assisted technologies were used in the preparation of this manuscript.
CRediT author statement
Cemy Nur Fitria: Conceptualization; Methodology; Writing - Original draft preparation. Soetrisno: Supervision; Validation. Ahmad Yunus: Data curation; Formal analysis. Brian Wasita: Investigation; Resources. Vitri Widyaningsih: Software; Visualization. Tatar Sumandjar: Writing - Reviewing and Editing. Paramasari Dirgahayu: Supervision; Project administration.
References
[1] P Ramalingam, M Sumathi, J Jothi, N Hasika, K Ramanathan, SS Subramanian, F Alhalaiqa, G Samhan and L Abdelhadi. A study to assess the level of resilience and quality of life among patients with diabetes mellitus at selected villages under Rural Health and Training Centre. Retos 2025; 68, 1721-1728
[2] RB Boulton, VL Singleton, LF Bisson and RE Kunkee. Principles and practices of winemaking. Springer Science & Business Media, Heidelberg, Germany, 2013.
[3] D Baltzis, I Eleftheriadou and A Veves. Pathogenesis and treatment of impaired wound healing in diabetes mellitus: New insights. Advances in Therapy 2014; 31(8), 817-836.
[4] GM Tsaffo, J Djenguemtar, SPC Fodouop, H Merzouk, JB Sokoudjou, GT Kamsu, HBL Feudjio, N Kodjio, A Mebarki and D Gatsing. Resistance profile of bacteria isolated from diabetic wounds: Phytochemicals and antibacterial studies of eriosema robustum leaf extracts. Trends in Sciences 2024; 21(6), 7881.
[5] L Wijaya, A Budiyanto, I Astuti and Mustofa. Pathogenesis, evaluation, and recent management of diabetic foot ulcer. Journal of the Medical Sciences 2019; 51(1), 82-97.
[6] HC Lu, MY Ng, YW Liao, S Maekawa, T Lin and CC Yu. Bromelain inhibits the inflammation and senescence effect in diabetic periodontitis: A preliminary in vitro study. Journal of dental sciences 2023; 18(2), 659-665.
[7] M Nurkhozin and S Mulyanti. Biokimia: Struktur dan fungsi biomolekul (in Indonesian). Andi, Yogyakarta, Indonesia, 2017.
[8] A D’Angelo, F Lixi, L Vitiello, V Gagliardi, A Pellegrino and G Giannaccare. The role of diet and oral supplementation for the management of diabetic retinopathy and diabetic macular edema: A narrative review. BioMed Research International 2025; 2025(1), 6654976.
[9] W Xie, W Hu, Z Huang, M Li, H Zhang, X Huang and P Yao. Betulinic acid accelerates diabetic wound healing by modulating hyperglycemia-induced oxidative stress, inflammation and glucose intolerance. Burns & Trauma 2022; 10, tkac007.
[10] A Perez-Favila, ML Martinez-Fierro, JG Rodriguez-Lazalde, MA Cid-Baez, MJ Zamudio-Osuna, MR Martinez-Blanco, FE Mollinedo-Montaño, IP Rodriguez-Sanchez, R Castañeda-Miranda and I Garza-Veloz. Current therapeutic strategies in diabetic foot ulcers. Medicina 2019; 55(11), 714.
[11] C Lan, I Liu, A Fang, C Wen and C Wu. Hyperglycaemic conditions decrease cultured keratinocyte mobility: implications for impaired wound healing in patients with diabetes. British Journal of Dermatology 2008; 159(5), 1103-1115.
[12] MJ Malone-Povolny, SE Maloney and MH Schoenfisch. Nitric oxide therapy for diabetic wound healing. Advanced Healthcare Materials 2019; 8(12), 1801210.
[13] AVA Mariadoss, AS Sivakumar, CH Lee and SJ Kim. Diabetes mellitus and diabetic foot ulcer: Etiology, biochemical and molecular based treatment strategies via gene and nanotherapy. Biomedicine & Pharmacotherapy 2022; 151, 113134.
[14] K Aichele, M Bubel, G Deubel, T Pohlemann and M Oberringer. Bromelain down-regulates myofibroblast differentiation in an in vitro wound healing assay. Naunyn-Schmiedeberg's Archives of Pharmacology 2013; 386(10), 853-863.
[15] HI Aydin, A Eser, I Kaygusuz, S Yildirim, T Celik, S Gunduz and S Kalman. Adipokine, adropin and endothelin-1 levels in intrauterine growth restricted neonates and their mothers. Journal of Perinatal Medicine 2016; 44(6), 669-676.
[16] R Pavan, S Jain, Shraddha and A Kumar. Properties and therapeutic application of bromelain: A review. Biotechnology Research International 2012; 2012(1), 976203.
[17] NFA El-Magd, NM Ramadan and SM Eraky. The ameliorative effect of bromelain on STZ-induced type 1 diabetes in rats through Oxi-LDL/LPA/LPAR1 pathway. Life Sciences 2021; 285, 119982.
[18] A Mameli, V Natoli and C Casu. Bromelain: An overview of applications in medicine and dentistry. Biointerface Research in Applied Chemistry 2021; 11(1), 8165-8170.
[19] S Dutta and D Bhattacharyya. Enzymatic, antimicrobial and toxicity studies of the aqueous extract of Ananas comosus (pineapple) crown leaf. Journal of Ethnopharmacology 2013; 150(2), 451-457.
[20] D Rahmat, D Ratih, L Nurhidayati and MA Bathini. Peningkatan aktivitas antimikroba ekstrak nanas (Ananas comosus (L.). Merr) dengan pembentukan nanopartikel (in Indonesian).” Jurnal Sains dan Kesehatan 2016; 1(5), 236-244.
[21] JA Ataide, LC Cefali, FM Croisfelt, AAM Shimojo, L Oliveira-Nascimento and PG Mazzola. Natural actives for wound healing: A review. Phytotherapy Research 2018; 32(9), 1664-1674.
[22] S Brien, G Lewith, A Walker, SM Hicks and D Middleton. Bromelain as a treatment for osteoarthritis: A review of clinical studies. EEvidence‐Based Complementary and Alternative Medicine 2004; 1(3), 251-257.
[23] MS Mousavi Maleki, R Ebrahimi kiasari, SJ Seyed Mousavi, H Hashemi‐Moghaddam, AA Shabani, H Madanchi and S Sardari. Bromelain-loaded nanocomposites decrease inflammatory and cytotoxicity effects of gliadin on Caco-2 cells and peripheral blood mononuclear cells of celiac patients. Scientific Reports 2023; 13(1), 21180.
[24] MS Arab, DM Tahoon, AA El Saadany and SE Hedya. Ameliorating effects of bromelain with or without metformin on endocrine-metabolic disturbances in letrozole-induced polycystic ovary syndrome in female rats via targeting SIRT1, insulin resistance, and inflammatory axis. Naunyn-Schmiedeberg's Archives of Pharmacology 2025. https://doi.org/10.1007/s00210-025-04517-w
[25] N Herdyastuti. Isolasi dan karakterisasi ekstrak kasar enzim bromelin dari batang nanas (Ananas comusus L.merr) (in Indonesian). Berkala Penelitian Hayati 2006; 12(1), 75-77.
[26] RV Devakate, VV Patil, SS Waje and BN Thorat. Purification and drying of bromelain. Separation and Purification Technology 2009; 64(3), 259-264.
[27] I Ismiranda. 2022, Formulasi Sediaan krim ekstrak bonggol nanas madu (Ananas comosus (L.) Merr). (in Indonesian). Ph. D. Dissertation. Poltekkes Tanjungkarang, Lampung, Indonesia, 2022.
[28] CK
Tara. Formulasi
sediaan krim anti inflamasi sari bonggol nanas (Ananas comosus (L.)
Merr.)
(in
Indonesian).
Jurusan Farmasi, Jakarta, Indonesia, 2019.
[29] A Ghasemi, S Khalifi and S Jedi. Streptozotocin-nicotinamide-induced rat model of type 2 diabetes (review). Acta Physiologica Hungarica 2014; 101(4), 408-420.
[30] American Diabetes Association. Standards of medical care in diabetes 2019. American Diabetes Association, Virginia, United States, 2019.
[31] NT Saputra, IN Suartha and AAGO Dharmayudha. Agen diabetagonik streptozotocin untuk membuat tikus putih jantan diabetes mellitus (in Indonesian). Buletin Veteriner Udayana 2018; 10(2), 116.
[32] MA Melakhessou, SE Marref, N Benkiki, C Marref, I Becheker and L Khattabi. In vitro, acute and subchronic evaluation of the antidiabetic activity of Atractylis flava Desf n-butanol extract in alloxan-diabetic rats. Future Journal of Pharmaceutical Sciences 2021; 7(1), 206.
[33] G Kaur, PVM Lakshmi, A Rastogi, A Bhansali, S Jain, Y Teerawattananon, H Bano and S Prinja. Diagnostic accuracy of tests for type 2 diabetes and prediabetes: A systematic review and meta-analysis. PLoS One 2020; 15(11), e0242415.
[34] O Insuan, P Janchai, B Thongchuai, R Chaiwongsa, S Khamchun, S Saoin, W Insuan, P Pothacharoen, W Apiwatanapiwat, A Boondaeng and P Vaithanomsat. Anti-inflammatory effect of pineapple rhizome bromelain through downregulation of the NF-κB-and MAPKs-signaling pathways in lipopolysaccharide (LPS)-stimulated RAW264.7 cells. Current Issues in Molecular Biology 2021; 43(1), 93-106.
[35] RG Vivanco, ABS Sousa, V de de C Oliveira, MAC Sinhoreti and FCP Pires-de-Souza. Effect of the use of bromelain associated with bioactive glass-ceramic on dentin/adhesive interface. Clinical Oral Investigations 2024; 28(1), 106.
[36] AÖ Şehirli, S Sayiner, G Savtekin and A Velioğlu-Öğünç. Protective effect of bromelain on corrosive burn in rats. Burns 2021; 47(6), 1352-1358.
[37] U Kansakar, V Trimarco, MV Manzi, E Cervi, P Mone and G Santulli. Exploring the therapeutic potential of bromelain: Applications, benefits, and mechanisms. Nutrients 2024; 16(13), 2060.
[38] Y Zhao, Q Wang, S Yan, J Zhou, L Huang, H Zhu, F Ye, Y Zhang, L Chen and T Zheng. Bletilla striata polysaccharide promotes diabetic wound healing through inhibition of the NLRP3 inflammasome. Frontiers in Pharmacology 2021; 12, 659215.
[39] B Roep, N van den Engel, A van Halteren, G Duinkerken and S Martin. Modulation of autoimmunity to beta-cell antigens by proteases. Diabetologia 2002; 45(5), 686-692.
[40] M Azarkan, E Maquoi, F Delbrassine, R Herman, N M'Rabet, R Calvo Esposito, P Charlier and F Kerff. Structures of the free and inhibitors-bound forms of bromelain and ananain from Ananas comosus stem and in vitro study of their cytotoxicity. Scientific Reports 2020; 10(1), 19570.
[41] H Febriani, S Ilyas, R Silaban and E Elimasni. Exploring the therapeutic effects of Allium chinense G. Don extract on pancreatic histopathology and apoptosis biomarker modulation in diabetic rat models. Trends in Sciences 2024; 22(12), 10638.
[42] G Yuan, ML Wahlqvist, G He, M Yang and D Li. Natural products and anti-inflammatory activity. Asia Pacific Journal of Clinical Nutrition 2006; 15(2), 143-152.
[43] YY Han, Y Jiang and J Hu. Collagen incorporation into waterborne polyurethane improves breathability, mechanical property, and self-healing ability. Composites Part A: Applied Science and Manufacturing 2020; 133, 105854.
[44] JA Ataide, NM de Carvalho, MA Rebelo, MV Chaud, D Grotto, M Gerenutti, M Rai, PG Mazzola and AF Jozala. Bacterial nanocellulose loaded with bromelain: Assessment of antimicrobial, antioxidant and physical-chemical properties. Scientific Reports 2017; 7(1), 2-10.
[45] R
Wahyudin, Y Lukmayani and ER Sadiah. Penelusuran pustaka potensi
aktivitas antibakteri ekstrak kulit buah nanas (Ananas comosus L.
Merr) (in
Indonesian),
Available at:
https://www.
semanticscholar.org/paper/Penelusuran-Pustaka-Potensi-Aktivitas-Antibakteri-Wahyudin-Lukma
yani/3b067ac71def6d29d1206cb6a256e97b90eb687d,
accessed July 2025.
[46] AJ Chakraborty, S Mitra, TE Tallei, AM Tareq, F Nainu, D Cicia, K Dhama, TB Emran, J Simal-Gandara, J Simal-Gandara and R Capasso. Bromelain a potential bioactive compound: A comprehensive overview from a pharmacological perspective. Life 2021; 11(4), 317.
[47] AJ Chakraborty, S Mitra, TE Tallei, AM Tareq, F Nainu, D Cicia, K Dhama, TB Emran, J Simal-Gandara and R Capasso. Development of a contaminated ischemic porcine wound model and the evaluation of bromelain based enzymatic debridement. Burns 2018; 44(4), 896-904.
[48] S Chhabra, N Chhabra, A Kaur and N Gupta. Wound healing concepts in clinical practice of OMFS. Journal of Maxillofacial and Oral Surgery 2017; 16(4), 403-423.
[49] S Soheilifar, M Bidgoli, A Hooshyarfard, A Shahbazi, F Vahdatinia and F Khoshkhooie. Effect of oral bromelain on wound healing, pain, and bleeding at donor site following free gingival grafting: A clinical trial. Journal of Dentistry 2018; 15(5), 309.
[50] AN Fathi, MH Sakhaie, S Babaei, S Babaei, F Slimabad and S Babaei. Use of bromelain in cutaneous wound healing in streptozocin-induced diabetic rats: An experimental model. Journal of Wound Care 2020; 29(9), 488-495.
[51] H Afzali, M Khaksari, S Jeddi, K Kashfi, MA Abdollahifar and A Ghasemi. Acidified nitrite accelerates wound healing in type 2 diabetic male rats: A histological and stereological evaluation. Molecules 2021; 26(7), 1872.
[52] NA Thomas, M Taupik, FN Ramadhani, AH Hutuba and DRP Papeo. Penyembuhan luka bakar gel enzim bromelin secara in vivo. Journal Syifa Sciences and Clinical Research 2024; 6(1), 66-77.
[53] K Huang, B Mi, Y Xiong, Z Fu, W Zhou, W Liu, G Liu and G Dai. Angiogenesis during diabetic wound repair: From mechanism to therapy opportunity. Burns & Trauma 2025; 13, tkae052.
[54] HA Celik, N Gurbuz, E Turantepe, M Seçme and Y Dodurga. Profiling of toll-like receptors and related signaling mediators in the pathogenesis of morphea. Dermatology Practical & Conceptual 2024; 14(4), e2024219.
[55] R Pezzani, M Jimenez-Garcia, X Capo, ES Gürer, F Sharopov, TYL Rachel, DN Woutouoba, A Rescigno, S Peddio, P Zucca, PVT Fokou, M Martorell, Z Gulsunoglu-Konuskan, A Ydyrys, T Bekzat, T Gulmira, C Hano, J Sharifi-Rad and D Calina. Anticancer properties of bromelain: State-of-the-art and recent trends. Frontiers in Oncology 2023; 12, 1068778.
[56] M Patrick, WNWM Zohdi, S Abd Muid and E Omar. Alpha(α)-mangostin (xanthone of Garcinia mangostana L.): Augmenting macrophages activity for an effective diabetic wound healing. Trends in Sciences 2024; 21(10), 8254.