
Trends
Sci.
2026;
23(6):
12228
In Vivo Analysis of Anti-inflammatory, Analgesic, and Antipyretic Activities of Litsea elliptica Blume Leaf Extracts
Raudhatun Samsul1, May Goh2,3, Hussein Taha1,4 and Norhayati Ahmad1,2,4,*
1Environmental and Life Sciences, Faculty of Science, Universiti Brunei Darussalam,
Bandar Seri Begawan BE1410, Brunei
2Herbal Research Group, Universiti Brunei Darussalam, Bandar Seri Begawan BE1410, Brunei
3PAP Rashidah Sa’adatul Bolkiah Institute of Health Sciences, Universiti Brunei Darussalam,
Bandar Seri Begawan BE1410, Brunei
4Institute for Biodiversity & Environmental Research, Universiti Brunei Darussalam,
Bandar Seri Begawan BE1410, Brunei
(*Corresponding author’s e-mail: [email protected])
Received: 10 October 2025, Revised: 5 November 2025, Accepted: 12 November 2025, Published: 1 February 2026
Abstract
The leaves of Litsea elliptica have been traditionally claimed to be beneficial for the treatment of headaches, fever, itch, stomach ulcers, and cancer. This present study investigates the anti-inflammatory, analgesic, and antipyretic activities of the mature and young leaves of L. elliptica extracted via maceration (MACE) and microwave-assisted extraction (MAE) in animal models using adult male Wistar albino rats. Anti-inflammatory activity was assessed via carrageenan-induced paw oedema and xylene-induced ear oedema assays. Analgesic activity was determined using formalin-induced paw flinching test and acetic acid-induced writhing test. The brewer’s yeast-induced pyrexia assay was employed to determine the antipyretic properties of the extracts. The extraction methods did not influence the activity of mature leaf extracts across all the assays conducted. However, MAE seemed to enhance the anti-inflammatory and analgesic activities of young leaf extracts. Treatment with both mature and young leaf extracts attenuated inflammation, pain, and fever in the animal models at varying degrees of inhibition. Overall, our current findings highlighted the potential of L. elliptica leaves as novel anti-inflammatory, analgesic, and antipyretic agents.
Keywords: Litsea elliptica, Anti-inflammatory, Analgesic, Antipyretic, Animal models
Introduction
Traditional medicine is often viewed as pre-scientific and inferior to allopathic medicine [1]. However, the early framework of many medical innovations, drugs, and pharmaceuticals today was derived from traditional medicinal knowledge, including notable drugs such as aspirin [2], artemisinin [3], vinblastine, and vincristine [4]. This highlights the importance of traditional medicine and indigenous knowledge in guiding novel drug discovery. The local communities of Brunei Darussalam are not unfamiliar to the use of traditional medicine for the treatment of diseases. Despite access to free healthcare system, many
locals are still inclined to seek treatment from traditional healers known as “Orang pandai”, who specialise in “Ubat kampung” or “Berubat kampung”, which is a traditional Malay medicinal practice that combines the use of various medicinal plants and incantations as part of the healing process [5,6]. Several literatures have documented the medicinal plants employed in these practices [6,7], and Litsea elliptica Blume is among the listed species [8].
L. elliptica, locally known as “Pawas” in Brunei Darussalam, is a large tropical tree that can grow up to 45 m in height and 80 cm in diameter and is typically found in primary and secondary lowland forests of Southeast Asia [9]. The leaves are elliptic or elliptic oblong in shape, thinly coriaceous, midrib sunken above, and glabrous on both surfaces [10]. The lower surface of mature leaves appears whitish, while in young leaves it is reddish-brown (Figure 1). The aromatic scent of L. elliptica leaves has led to its use in certain culinary applications in Brunei and Thailand. The young leaves are often consumed raw or lightly blanched and are enjoyed by the Bruneians as a delectable side dish known as “Ulam” [11]. In Thailand, they are used as a flavoring ingredient in a local spicy dipping sauce called “Nam Prik” [12]. In addition to its culinary use, different parts of the plant have been utilised by various ethnic groups across the region to treat a wide range of ailments [13]. In Indonesia, the Talang Mamak ethnic group applied L. elliptica leaf paste on the forehead to alleviate headaches [14] whereas the local community of Melanau in Sarawak applied the leaf poultice over the abdominal areas to relieve bloating and flatulence in babies and children [15]. A similar practice was also observed in traditional Thai medicine, using the bark of L. elliptica instead to treat flatulence [16]. Nevertheless, in general, the leaves of L. elliptica were traditionally claimed to be beneficial for the treatment of headaches, fever, itch, stomach ulcers, and cancer [8,17].

Figure 1 Mature and young leaves of L. elliptica collected from Bukit Udal, Tutong, Brunei Darussalam. The leaves are elliptic to elliptic-oblong, thinly coriaceous, and glabrous on both surfaces, with the lower surface appearing whitish in mature leaves and reddish-brown in young leaves.
Despite the wide use of L. elliptica in traditional medicine, investigations on the potential therapeutic application of the species are relatively limited compared to those of the other species within the Litsea genus. Nevertheless, past investigations revealed some pharmacological activities for different parts of the plant, whereby its crude extracts and essential oils were shown to possess antimutagenicity [18], antioxidant [17,19-21], antimicrobial [19-22], and anti-alpha glucosidase properties [16]. Toxicity studies on the essential oil of L. elliptica leaves reported that the essential oil was of no toxicity concern [23-25].
The biological activities of plants are often associated with their phytochemical compositions, hence, recent studies on L. elliptica have focused on establishing the phytochemical profile of its extracts. The chemical constituents of different parts of L. elliptica vary significantly, with approximately 49 compounds identified thus far [16,17,26]. The major groups isolated from shoots, barks, branchlets, young leaves, and mixed leaves include flavonoid glycosides, alkaloids, fatty acids, fatty acid esters, phenolic compounds, coumaran, and vitamin E. These compounds have been associated with various health-promoting properties, including anti-inflammatory and analgesic activities [17], supporting its numerous ethno-pharmacological applications whilst forming a basis for further exploration. Nonetheless, the effects of mature and young leaf extracts of L. elliptica on inflammation, pain, and fever remain largely unknown.
Plant species under the genus Litsea have been valued for their therapeutic properties against inflammation, pain, and fever. Numerous studies have supported their anti-inflammatory and analgesic claims, such as L. cubeba, L. japonica, L. salicifolia, L. glutinosa, L. guatemalensis, L. monopetala, and L. glaucescens, as they have been revealed to have significant inhibitory effects in both in vitro and in vivo assays [27,28]. Although several species of Litsea, such as L. glutinosa, L. cubeba., L. pungens, L. elliptica, L. khasyana, and L. laeta have been claimed to treat fever [27,28], to date, only the crude leaf extract of L. glutinosa has been studied for its fever-reducing property [29]. Building upon the growing body of research on the Litsea genus, this present study aims to investigate the efficacy of L. elliptica mature and young leaf extracts in suppressing swellings in carrageenan- and xylene-induced oedema models, pain in formalin- and acetic acid-induced pain models, and fever in brewer’s yeast-induced pyrexia model.
Materials and methods
Chemicals
Ethyl alcohol, absolute (denatured) (6923) and formaldehyde solutions (3955) were purchased from Reagents Duksan. λ - carrageenan, plant mucopolysaccharide (22049-25G-F), acetic acid (glacial) (100063), and xylene (214736) were from Sigma Aldrich. Both diclofenac sodium (Voren ®) and paracetamol (Duopharma) were obtained from local pharmacies, whereas brewer’s yeast was obtained from a local supermarket. Ketamine and xylazine were purchased from local veterinaries.
Plant collection, identification, and drying
Mature and young leaves of L. elliptica Blume were collected in June 2023, from Bukit Udal, Tutong, Brunei Darussalam. The samples were identified and authenticated by the Assistant curator and Botanist from Universiti Brunei Darussalam Botanical Research Centre (UBD BRC), and a voucher specimen (Reference no: B019661, BRUN 5031) was deposited in IBER BRC Herbarium. The collected mature and young leaves were separated based on the size and the colour of the lower surface of the leaves. They were then thoroughly cleaned, shade-dried at room temperature (25 - 28 °C) until constant weight was achieved and pulverised using a domestic blender (Sharp EM-130 WH, Malaysia). The grounded coarse samples were stored at room temperature in airtight containers until use.
Preparation of ethanol extracts
Maceration (MACE)
The pulverised mature and young leaves of L. elliptica were soaked in 75% ethanol at a weight-to-volume (w/v) ratio of 1:25 for 3 days at room temperature with constant agitation at 140 rpm using an orbital shaker (Joan Lab OS-20, Indonesia) [30,31]. The resulting mixtures were vacuum filtered through Whatman No. 1 filter paper, and the ethanol was removed under reduced pressure using a rotary evaporator (IKA RVS, imLab, France) at 50 °C. The residual extracts were then oven-dried at 40 °C until a constant weight of crude extract was achieved. The dried extracts (Figure 2) were sealed and stored at 4 °C until further analysis.
Microwave-assisted extraction (MAE)
The powdered mature and young leaves were extracted using 75% ethanol at a ratio of 1:25 (w/v). The extractions were carried out using a domestic microwave oven (Toshiba ER-SGS20 (K), Malaysia) at 540 W for a total irradiation time of 15 min [30], consisting of 10 cycles of 1 min 30 s irradiation followed by 1 min cooling. The cooling was performed by submerging the extraction flask in a water bath at room temperature (25 - 28 °C). During each cooling interval, the temperature was monitored to ensure it remained below 70 °C. The mixture was then vacuum filtered through Whatman No. 1 filter paper, and the ethanol was evaporated from the filtrate under reduced pressure using a rotary evaporator at 50 °C. The collected residues were further dried in an oven at 40 °C until a constant weight of crude extract was achieved. The remaining extracts (Figure 2) were collected and stored at 4 °C until further use.

Figure 2 Crude ethanolic extracts of L. elliptica mature and young leaves obtained via MACE and MAE.
Extraction yield analysis
The weight of the total pulverised sample before extraction and the weight of the final crude extract after oven drying were recorded. The percentage yield of each extract was then determined using the following formula [32].
Percentage
yield of extract =
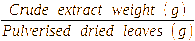 ×100
×100
Experimental animal housing and groupings
Adult male Wistar albino rats, aged 10 - 12 weeks and weighing between 300 - 400 g were obtained from the Universiti Brunei Darussalam animal facility. The animals were maintained under standard laboratory conditions (room temperature 25 ± 2 °C; relative humidity 55% - 60%, 12 h light/dark cycle). The rodents were fed a standard diet of rodent pellets (Altromin 1314, 10 mm pellets: Altromin Spexialfutter GmbH, Lage, Germany) and water ad libitum. The experimental study involving animals was approved by the Universiti Brunei Darussalam Animal Research Ethics Committee Ref file: UB/FOS/E2(J) dated 02 May 2023.
Repeated Measures ANOVA: Within-Between Interaction performed using GPower 3.1 was used to estimate the sample size for each assay. The power analysis indicated that at least 6 rats (n = 6) were required per group to achieve statistically reliable results. The rodents were randomly assigned to 10 treatment groups, each receiving their respective treatment orally. All dosages were calculated based on the animals’ body weight (b.w.), expressed in mL/100 g b.w. or mg/kg b.w. The treatment groups and their respective dosages were as follows:
Negative control (NC) : Received 1 mL/100 g b.w. of sterile saline.
Positive control (PC): Received standard reference drugs as described in the respective subsections for each assay.
MMACE400: Received 400 mg/kg b.w. of mature L. elliptica leaf extract obtained by MACE.
MMACE50: Received 50 mg/kg b.w. of mature L. elliptica leaf extract obtained via MACE.
MMAE 400: Received 400 mg/kg b.w. of mature L. elliptica leaf extract obtained via MAE.
MMAE50: Received 50 mg/kg b.w. of mature L. elliptica leaf extract obtained via MAE.
YMACE400: Received 400 mg/kg b.w. of young L. elliptica leaf extract obtained via MACE.
YMACE50: Received 50 mg/kg b.w. of young L. elliptica leaf extract obtained via MACE.
YMAE 400: Received 400 mg/kg b.w. of young L. elliptica leaf extract obtained via MAE.
YMAE50: Received 50 mg/kg b.w. of young L. elliptica leaf extract obtained via MAE.
Anti-inflammatory effect assessments
Carrageenan-induced paw oedema model
The carrageenan-induced paw oedema assay was performed as previously described by Mansouri et al. [33] with minor modifications. At 1 h post-treatment, paw oedema was induced by subcutaneously (s.c.) injecting 100 µL of 1% λ-carrageenan suspended in sterile saline into the plantar surface of the right hind paw. PC group received 10 mg/kg b.w. of diclofenac sodium. The progression of oedema was monitored by measuring paw volume at 0, 1, 2, 3, 4, and 5 h using a plethysmometer (Muromachi MK-101P, Japan). The degree of oedema (mL) was determined by calculating the difference in the volume of the right hind paw between 0 h and 1, 2, 3, 4, and 5 h following oedema induction. A reduced increase in paw volume compared to the NC group at each time point was interpreted as an anti-inflammatory activity.
Xylene-induced ear oedema model
An established method previously described by Soliman et al. [34] was employed in this study with slight modifications. At 1 h post-treatment, acute ear oedema was induced by topically applying 30 µL of absolute xylene to the right ear lobe, where 15 µL was applied to the anterior surface and another 15 µL to the posterior surface. The left ear lobe was left uninduced and served as the reference ear. PC group received diclofenac sodium at 150 mg/kg b.w. At 2 h post-oedema induction, the rats were anesthetized via intraperitoneal (i.p.) injection of a combination of ketamine (80 mg/kg b.w.) and xylazine (8 mg/kg b.w.) [35]. Circular punch biopsy specimens (6 mm diameter) were collected from approximately the same site on both ears using a single-hole punch and weighed using an analytical balance (Shimadzu ATX224, Japan). The degree of ear oedema was quantified by calculating the difference in tissue weight (mg) between the oedema-induced right ear and the uninduced reference left ear. A smaller weight difference compared to the NC group was taken as an indication of anti-inflammatory effects.
Analgesic effect assessments
Formalin-induced paw flinching model
The formalin-induced paw flinching assay described by Roca-Vinardell et al. [36] was adopted to evaluate the analgesic activity of mature and young L. elliptica leaf extracts [36]. To allow acclimatization, rats were individually placed in transparent chambers (26×17×16 cm3) for 1 h before treatment. At 30 min post-treatment, 50 µL of 5% formalin suspended in sterile saline was injected via s.c. route into the plantar surface of the right hind paw of the rats. The PC group received paracetamol at a dose of 150 mg/kg b.w [36]. The animals then immediately returned to the chamber for a 1 h observation period. A camera was positioned in front of each chamber to record the paw-flinching behaviour. The number of flinches of the affected paw was observed in 2 phases: The early phase (Phase 1), occurring from 0 to 10 min post-injection, and the late phase (Phase 2), from 10 - 60 min post-injection. A lower number of flinches relative to the NC group in either phase was considered indicative of analgesic activity.
Acetic acid-induced body writhing model
The analgesic activity of the extracts was evaluated using the acetic acid-induced body writhing assay, as described by Rashid et al. [37], with slight modifications. Rats were acclimatized in transparent chambers (26×17×16 cm3) for 1 h prior to treatment. At 30 min post-treatment, 1 mL/100 g b.w. of 0.7% acetic acid suspended in sterile saline was i.p. administered to induce pain-like behaviour, which manifested as characteristic body writhing. The animals were then returned to their chambers for observation. PC group received diclofenac sodium at 10 mg/kg b.w. A camera was positioned in front of the chamber to record the observations. The number of body writhes (n) was counted over a 15 min observation period, with a smaller number of writhes relative to the NC group reflecting analgesic activity.
Antipyretic effect assessments
Brewer’s yeast-induced pyrexia model
The
brewer’s yeast-induced pyrexia method was adopted in this study as
previously described by Forkuo et
al.
[38] with slight modifications. The normal rectal temperature (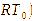 was measured using a lubricated digital thermometer (Omron MC-343F,
Japan) before induction. Pyrexia was induced by i.p. injection of
brewer’s yeast suspended in sterile saline at a dose of 135 mg/kg
b.w. After the induction, rectal temperature was measured at 1 h
intervals over a 4 h period to monitor the development of fever.
Rats that exhibited an increase in rectal temperature of
was measured using a lubricated digital thermometer (Omron MC-343F,
Japan) before induction. Pyrexia was induced by i.p. injection of
brewer’s yeast suspended in sterile saline at a dose of 135 mg/kg
b.w. After the induction, rectal temperature was measured at 1 h
intervals over a 4 h period to monitor the development of fever.
Rats that exhibited an increase in rectal temperature of
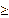 0.5 °C compared to their
0.5 °C compared to their
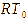 were considered pyretic, and only these animals were selected to
receive treatment. Following treatment administration, rectal
temperature was recorded at hourly intervals for 4 h (
were considered pyretic, and only these animals were selected to
receive treatment. Following treatment administration, rectal
temperature was recorded at hourly intervals for 4 h (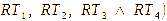 .
PC
group received 150 mg/kg b.w. of paracetamol. The change in rectal
temperature at each time point (
.
PC
group received 150 mg/kg b.w. of paracetamol. The change in rectal
temperature at each time point (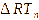 )
was determined using the following formula.
)
was determined using the following formula.
![]()
where,
∆RTn
= change in rectal temperature at hour n (n = 1, 2, 3 and 4),
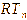 = rectal temperature at hour n (n = 1, 2, 3, and 4),
= rectal temperature at hour n (n = 1, 2, 3, and 4),
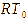 = normal rectal temperature. A smaller ∆RTn
relative to the NC group was interpreted as the antipyretic activity
of the treatment.
= normal rectal temperature. A smaller ∆RTn
relative to the NC group was interpreted as the antipyretic activity
of the treatment.
Statistical analysis
All
data in this study were statistically analysed using Origin 2024
(OriginLab Corporation Northampton, MA, USA) and expressed as mean ±
standard deviation (SD). One-way ANOVA followed by Tukey’s test
was carried out to compare the differences between treatment groups
at each time point or phase, whereby p
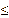 0.05
was considered significant.
0.05
was considered significant.
Results and discussion
In the present study, mature and young leaves of L. elliptica were extracted using 2 methods of extraction, MACE and MAE. To ensure comparability, identical parameters were applied to both methods, including solvent (ethanol), the concentration of solvent (75%), and the sample/solvent ratio (1:25). The extraction method did not appear to significantly influence the efficacy of mature leaf extracts, as both MACE and MAE yielded similar results and demonstrated a dose-dependent inhibition across all assays. In contrast, for young leaf extracts, MAE exhibited potent anti-inflammatory and analgesic effects, whereas MACE was more effective in the antipyretic assay. Interestingly, young leaf extracts generally did not demonstrate a consistent dose-dependent response and often demonstrated better efficacy at a lower dose. Nonetheless, treatment with mature and young leaf extracts of L. elliptica at varying degrees ameliorated oedema, reduced pain responses, and lowered fever in acute inflammation, analgesic, and pyretic models.
Anti-inflammatory activity of L. elliptica mature and young leaf extracts in acute inflammation models
Typical
manifestations of acute inflammation induced by 1% λ-carrageenan,
include redness (Figure
3)
and a time-dependent increase in paw volume (Figure
4).
These symptoms were observed in all treatment groups, indicating
successful development of oedema. A maximum degree of paw oedema was
recorded in NC at 0.46 ± 0.049 mL. Treatments with PC and L.
elliptica mature
and young leaf extracts mitigated inflammation, as indicated by
reduction in redness and paw volume compared to NC group. Rats
treated with mature leaf extracts showed a dose-dependent inhibition
(Figure
4(a)).
MMACE400 significantly (p
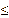 0.05)
impeded oedema progression from 2 h onwards, in which an
oedema degree of only 0.13 ± 0.072 mL was observed by the end of
the assay. MMACE50, MMAE400, and MMAE50 suppressed oedema
significantly (p
0.05)
impeded oedema progression from 2 h onwards, in which an
oedema degree of only 0.13 ± 0.072 mL was observed by the end of
the assay. MMACE50, MMAE400, and MMAE50 suppressed oedema
significantly (p
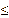 0.05)
from
4 h onwards. Rats that were treated with young leaf extracts, except
for YMACE50, significantly (p
0.05)
from
4 h onwards. Rats that were treated with young leaf extracts, except
for YMACE50, significantly (p
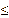 0.05)
hampered
oedema development from 3 h onwards (Figure
4(b)).
YMACE50 was only able to suppress oedema from 4 h onwards
significantly. Noteworthily, at 5 h post-induction, paw volume
reduction was markedly great in YMACE400 and YMAE50 groups,
measuring at only 0.11 ± 0.054 and 0.02 ± 0.012 mL, respectively.
Treatment with YMAE50 exhibited the most pronounced effect, reducing
paw oedema by 96% relative to NC, whereas PC achieved only a 63%
reduction by the end of the assay.
0.05)
hampered
oedema development from 3 h onwards (Figure
4(b)).
YMACE50 was only able to suppress oedema from 4 h onwards
significantly. Noteworthily, at 5 h post-induction, paw volume
reduction was markedly great in YMACE400 and YMAE50 groups,
measuring at only 0.11 ± 0.054 and 0.02 ± 0.012 mL, respectively.
Treatment with YMAE50 exhibited the most pronounced effect, reducing
paw oedema by 96% relative to NC, whereas PC achieved only a 63%
reduction by the end of the assay.
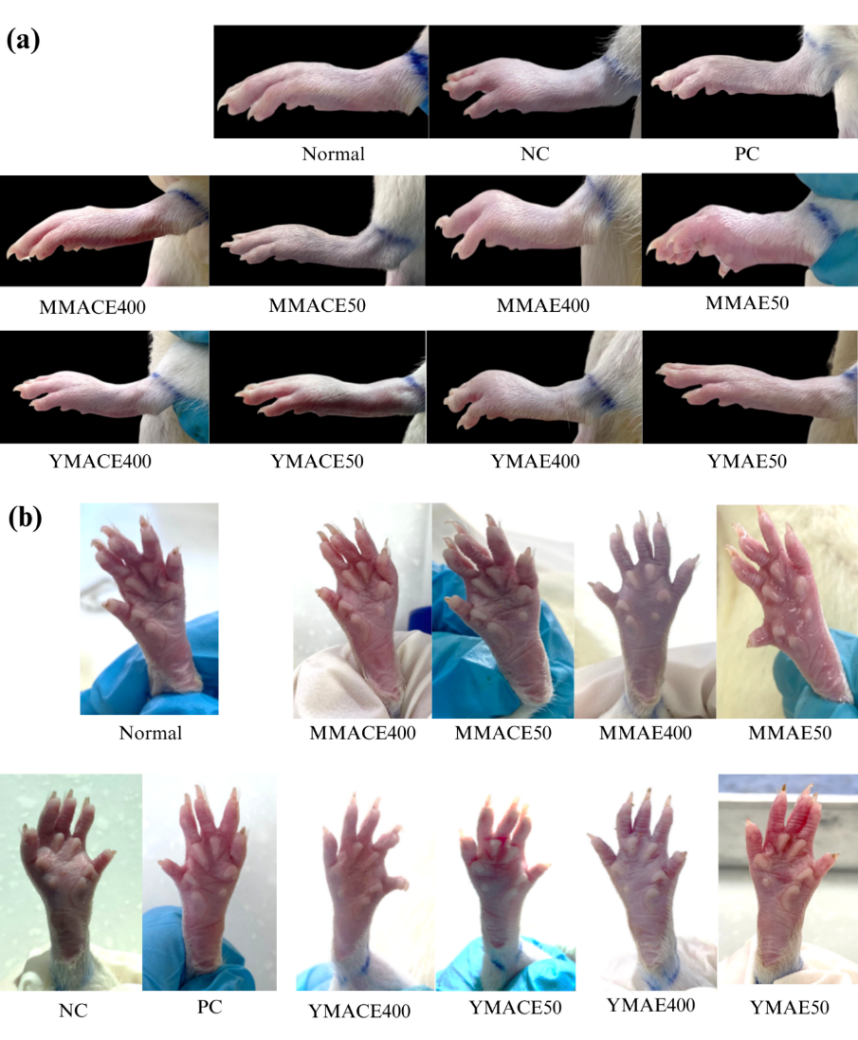
Figure 3 The anti-inflammatory effects of L. elliptica mature and young leaf extracts at 50 and 400 mg/kg b.w. on carrageenan-induced paw oedema in Wistar rats. (a) Lateral and (b) palmar views of paw swelling were captured at 5 h post-induction. Diclofenac sodium (10 mg/kg b.w.) served as PC, while sterile saline (1 mL/100 g b.w.) was administered as the NC.
Figure
4 Degree
of paw oedema (mL) following treatment with L.
elliptica (a)
mature leaf extracts and (b) young leaf extracts obtained by MACE
and MAE at 50 and 400 mg/kg b.w. Diclofenac sodium (10 mg/kg b.w.)
served as PC, while sterile saline (1 mL/100 g b.w.) was
administered as the NC. Values are presented as mean ± SD; *
indicates p
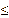 0.05
significant difference compared to NC as analysed using 1-way ANOVA
followed by Tukey’s test.
0.05
significant difference compared to NC as analysed using 1-way ANOVA
followed by Tukey’s test.
Topical
application
of xylene on the right ear lobe of the rats induced an immediate ear
scratching reaction, redness (Figure
5),
and an increase in ear oedema (Figure
6).
The development of these symptoms was observed in all treatment
groups, demonstrating the successful development of inflammation in
the animal model. The greatest degree of oedema was observed in NC
at 3.88 ± 0.76 mg, and treatments with mature (Figure
6(a))
and young (Figure
6(b))
L.
elliptica leaf
extracts showed a smaller weight difference in tissue weight
relative to NC. Mature leaf extracts exhibited a dose-dependent
inhibition, whereby MMACE400 (1.88 ± 0.42 mg) and MMAE400 (1.93 ±
0.65 mg) were more effective in attenuating ear oedema than MMACE50
(2.95 ± 0.69 mg) and MMAE50 (3.45 ± 0.78 mg). The increase in ear
weight was significantly (p
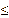 0.05) inhibited by YMAE400 (1.07 ± 0.23 mg), followed by YMACE400
(1.55 ± 0.51 mg), YMAE50 (1.63 ± 0.57 mg), and YMACE50 (1.93 ±
0.65 mg). Interestingly, treatments with young leaf extracts
resulted in a greater reduction in ear oedema compared to PC (2.17 ±
0.71 mg), which exhibited only a moderate ear oedema reduction,
highlighting the potentially stronger potency of young leaf extracts
in suppressing ear oedema.
0.05) inhibited by YMAE400 (1.07 ± 0.23 mg), followed by YMACE400
(1.55 ± 0.51 mg), YMAE50 (1.63 ± 0.57 mg), and YMACE50 (1.93 ±
0.65 mg). Interestingly, treatments with young leaf extracts
resulted in a greater reduction in ear oedema compared to PC (2.17 ±
0.71 mg), which exhibited only a moderate ear oedema reduction,
highlighting the potentially stronger potency of young leaf extracts
in suppressing ear oedema.
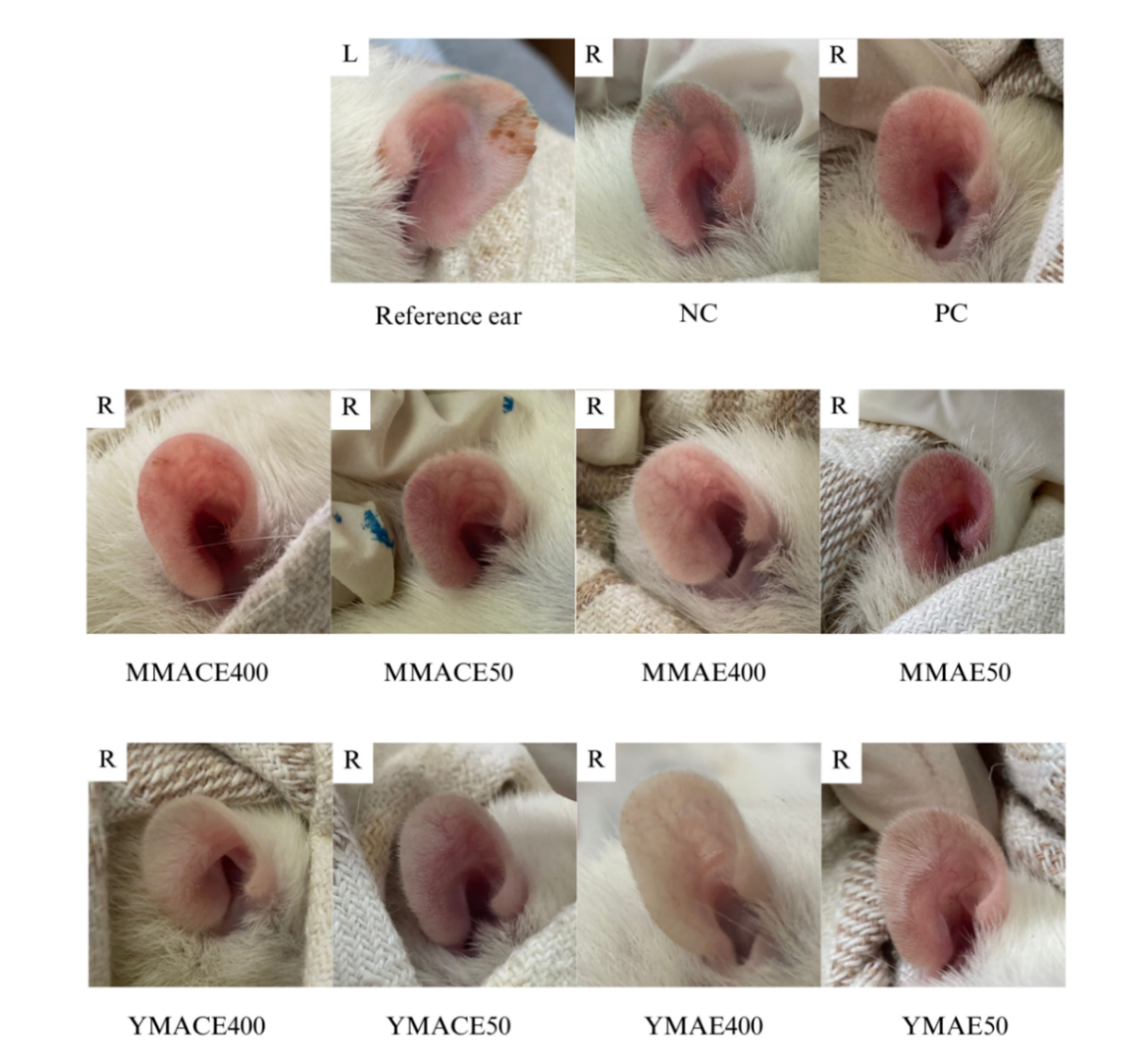
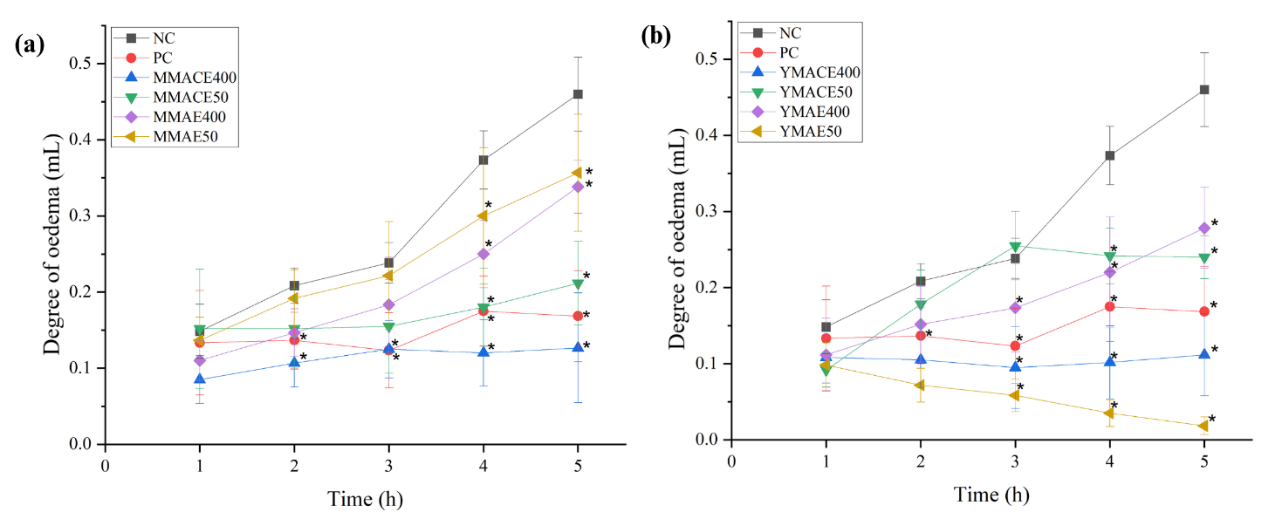
Figure 5 Anti-inflammatory effects of L. elliptica mature and young leaf extracts obtained by MACE and MAE at 400 and 50 mg/kg b.w. on xylene-induced ear oedema. Representative images of right (R) ear oedema in each treatment group at 2 h post-induction. Diclofenac sodium (150 mg/kg b.w.) served as PC and sterile saline (1 mL/100 g b.w.) was administered as NC.
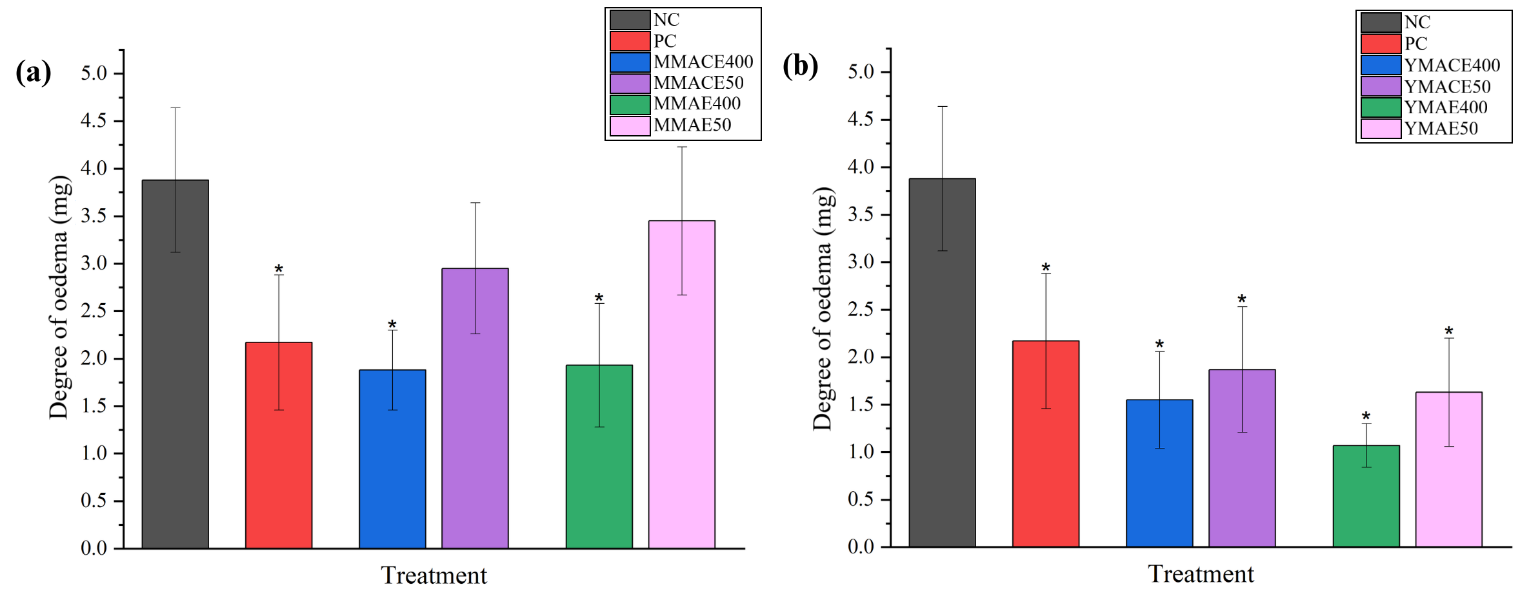
Figure
6 Degree
of ear oedema (mg) following treatment with L.
elliptica (a)
mature leaf extracts and (b) young leaf extracts obtained via MACE
and MAE at 400 and 50 mg/kg b.w. Diclofenac sodium (150 mg/kg b.w.)
was used as PC, while sterile saline (1 mL/ 100 g b.w.) served as
NC. Values are expressed as mean ± SD; * indicates p
 0.05
significant difference against NC as analysed using one-way ANOVA
followed by Tukey’s test.
0.05
significant difference against NC as analysed using one-way ANOVA
followed by Tukey’s test.
The anti-inflammatory activity observed in this study may result from the inhibition of key inflammatory mediators involved in the inflammation models employed. In the carrageenan-induced paw oedema model, the oedema develops in a biphasic manner, which is primarily triggered by the activation of the cyclooxygenase (COX) pathway [39]. The second phase, which occurs approximately 3 h post-carrageenan administration, is associated with the release of prostaglandins (PGs) through the activation of JAK2/STAT3 and NF-κB signaling pathways [39,40]. Topical application of xylene, on the other hand, induces neurogenous oedema, which is partially linked to the release of substance P (SP) from sensory neurons, causing vasodilation and plasma extravasations [41]. The secretion of SP has been reported to promote upregulation of COX-2 and prostaglandin (PGE2) expression via JAK2/STAT3 and NF-κB activation in human colonocytes [42,43]. Both mature and young leaf extracts ameliorated paw oedema, particularly in the second phase of carrageenan-induced paw oedema and in xylene-induced ear oedema. This suggests that L. elliptica leaf extracts may suppress the release of the later-stage mediators and interfere with JAK2/STAT3 and NF-κB activation, thereby reducing downstream inflammation-related mediators such as COX-2 and PGE2 [39,40]. About half of the phytochemicals identified by Goh et al. [8] in the young leaf extract of L. elliptica have been reported to exhibit anti-inflammatory activity, suggesting the presence of potent bioactive constituents, which aligns with the anti-inflammatory effects demonstrated in this study. Among the characterised components, phytol was found in relatively high concentration in the young L. elliptica leaf extract [8]. Both in vitro and in vivo studies have demonstrated that phytol inhibits egg albumin denaturation and reduces formalin-induced paw oedema. Furthermore, in silico analysis revealed that phytol interacts with COX-1, COX 2, NF-κB, and IL-1β, suggesting a potential role in mediating anti-inflammatory activity [44]. Phytol was also detected in the mature leaf extract, although at a relatively lower concentration, which may partially explain why the young leaf extract showed greater inhibition of paw and ear oedema compared to the mature leaf extract. Furthermore, the 2 models of inflammation are known to be sensitive to non-steroidal anti-inflammatory drugs (NSAIDs) such as diclofenac sodium, whose mechanism of action (MOA) involves targeting the NF-κB signaling pathway by non-selectively or selectively inhibiting COX-1 and COX-2 [45]. The comparable level of inhibition of mature and young L. elliptica leaf extracts to diclofenac sodium observed in this study suggests a similar MOA.
Although there is no direct evidence demonstrating the MOA of L. elliptica, a number of studies have reported interactions between the extracts and isolated compounds of other Litsea species, such as L. cubeba and L. japonica, with various inflammatory markers. L. cubeba has been valued for its therapeutic effects against rheumatic and inflammatory conditions in Traditional Chinese Medicine (TCM). Aligning with their traditional application [46], the crude extracts and compounds, namely reticuline, boldine, and neral, from the L. cubeba fruit and root have been demonstrated to reduce COX, 5-lipoxygenase (5-LOX), nitric oxide (NO), and inducible nitric oxide synthase (iNOS) expression, alongside pro-inflammatory cytokines such as TNF-α, IL-6, and IL-1β [40,47-49]. Similar suppression of inflammatory mediators and cytokines was also demonstrated by extracts and isolated compounds, such as litsenolide A2, from the fruits of L. japonica [50,51], where downregulation of pNF-κBp50/p65, pIκBα, pERK, pJNK, and p38 was also observed in lipopolysaccharide (LPS)-stimulated murine macrophages [50]. Likewise, L. salicifolia ethanolic bark extract has been reported to reduce COX-2, PGE2, NO, TNF-α, IL-6, and IL-1β levels in LPS-induced inflammation in RAW 267.7 cells [52]. The extract’s anti-inflammatory activity was further evaluated using Freund’s complete adjuvant (CFA) - induced arthritis model where they found that the bark extract, at 100 and 200 mg/kg b.w., lowered TLR4, NF-κB, COX-2, and iNOS levels while increasing anti-inflammatory mediators such as Nrf2, HO-1, and SIRT1 expression in paw tissues [52]. Taken together, we speculate that the anti-inflammatory activity of L. elliptica observed in this study may be ascribed to its bioactive phytochemicals that are involved in the inhibition of COX-2, PGE2, NO, TNF-α, IL-6, and IL-1β levels through the regulation of JAK2/STAT3 and NF-κB signaling pathways.
Analgesic activity of L. elliptica mature and young leaf extracts in acute pain models
Administration
of
5% formalin into the paw triggered an immediate paw flinching
response, marking the onset of Phase 1. In this phase, neither the
extracts nor PC significantly (p
 0.05)
attenuated the flinching behaviour relative to NC (Figure
7).
This suggested the limited effect of treatments on the direct
activation of nociceptors by formalin in the early phase.
Nevertheless, treatment with mature leaf extracts resulted in a
dose-dependent suppression in Phase
2. A reduction of approximately 40% - 46% (p
0.05)
attenuated the flinching behaviour relative to NC (Figure
7).
This suggested the limited effect of treatments on the direct
activation of nociceptors by formalin in the early phase.
Nevertheless, treatment with mature leaf extracts resulted in a
dose-dependent suppression in Phase
2. A reduction of approximately 40% - 46% (p
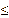 0.05)
relative to NC was recorded, although the differences between the
doses were relatively modest (Figure
7(a)).
YMAE400 demonstrated the greatest efficacy in Phase 2, reducing the
number of flinches by 50% (p
0.05)
relative to NC was recorded, although the differences between the
doses were relatively modest (Figure
7(a)).
YMAE400 demonstrated the greatest efficacy in Phase 2, reducing the
number of flinches by 50% (p
 0.05)
compared
to NC (Figure
7(b)),
followed by YMAE50, YMACE400, and YMACE50, in decreasing order of
potency. Notably, treatment with both mature and young L.
elliptica
leaf extracts resulted in a pronounced reduction in flinching
behaviour compared to PC.
0.05)
compared
to NC (Figure
7(b)),
followed by YMAE50, YMACE400, and YMACE50, in decreasing order of
potency. Notably, treatment with both mature and young L.
elliptica
leaf extracts resulted in a pronounced reduction in flinching
behaviour compared to PC.
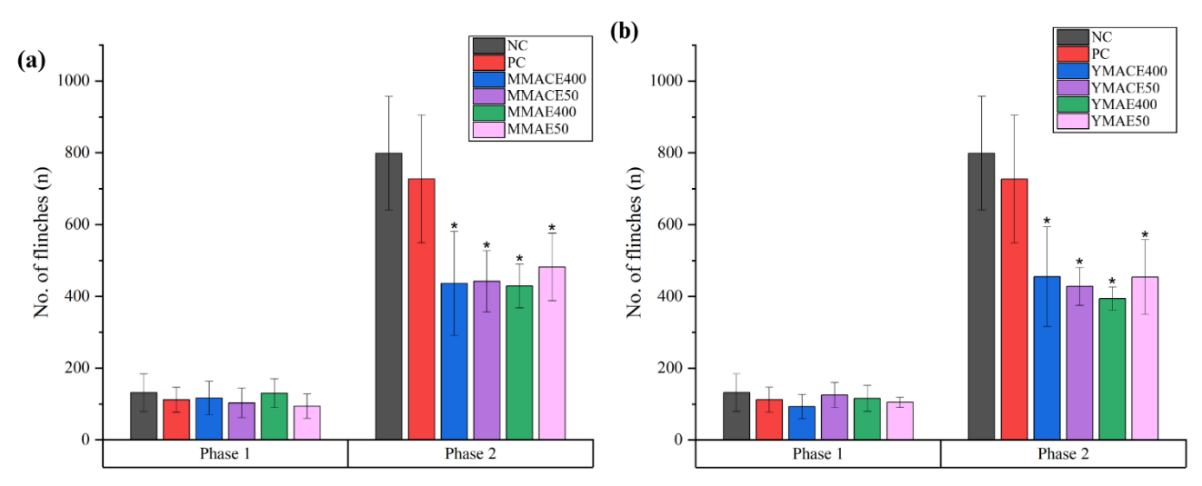
Figure 7 Analgesic effects of L. elliptica (a) mature leaf extracts and (b) young leaf extracts at 400 and 50 mg/kg b.w. obtained via MACE and MAE in Phase 1 (0 - 10 min) and Phase 2 (10 - 60 min) of the formalin-induced paw flinching model. Sterile saline at 1 mL/100 g b.w. (NC) and paracetamol at 150 mg/kg b.w. (PC) were used as the control groups. Values are expressed as mean number of flinches (n) ± SD; * indicates p ≤ 0.05 significant difference against NC analysed using 1-way ANOVA followed by Tukey’s test.
A dose-dependent trend of inhibition was observed in groups treated with mature leaf extracts in the acetic acid-induced body writhing assay. Both MMACE400 and MMAE400 significantly (p ≤ 0.05) reduced the number of body writhes by 35% and 50%, respectively, compared to NC (Figure 8 (a)). Remarkably, treatment with young leaf extracts led to a significant (p ≤ 0.05) suppression of pain-like behaviour in the rats, with reductions of approximately 83% - 98% in the number of body writhes relative to NC (Figure 8(b)). This level of inhibition was comparable to PC, which showed a 49% reduction, highlighting the strong potency of young leaf extracts in attenuating acetic acid-induced body writhing.
The inhibition of inflammatory mediators and cytokines has been an effective approach for not only treating inflammatory diseases but also managing pain [53]. Some of these mediators can directly stimulate the nociceptive pathway by acting on the nociceptors, leading to a lowered pain threshold to cause either hyperalgesia or allodynia [54]. Administration of low concentrations of acetic acid in rodents has been linked to an overproduction of analgesic mediators, particularly PGE₂ and prostacyclin (PGI₂), in peritoneal fluids [37], whereas exposure to formalin triggers a 2-phase pain response. In the initial nociceptive pathway, formalin directly activates the transient receptor potential ankyrin 1 (TRPA1) channel, which releases neurotransmitters such as SP and glutamate, causing sharp pain. This is followed by the inflammatory phase, where mediators including PGE₂, bradykinin, histamine, TNF-α, and IL-1β trigger phospholipase and COX pathways, sustaining the pain [55].
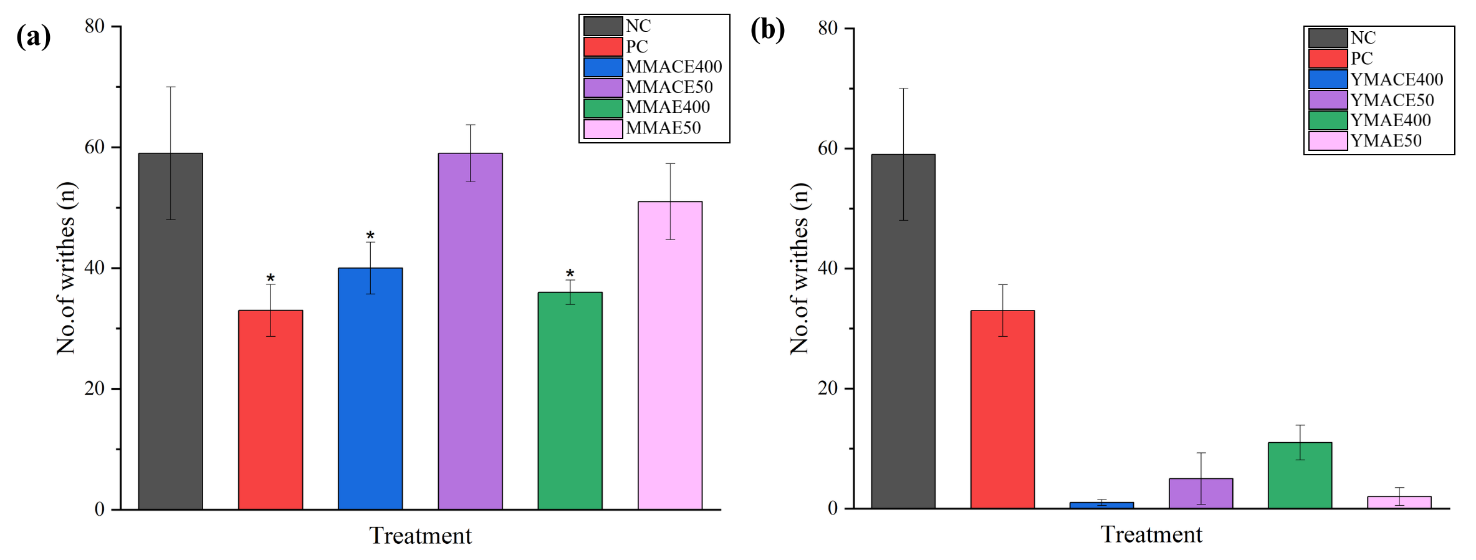
Figure 8 Analgesic effects of L. elliptica (a) mature leaf extracts and (b) young leaf extracts obtained by MACE and MAE at 400 and 50 mg/kg b.w. in the acetic acid-induced body writhing model. Values are expressed as mean number of body writhes (n) ± SD; * indicates p ≤ 0.05 significant difference against NC as analysed using 1-way ANOVA followed by Tukey’s test.
Generally, centrally acting drugs inhibit both phases equally, while peripherally acting drugs inhibit the second phase. For instance, pre-treatment with an intraperitoneal injection of celecoxib, a selective COX-2 inhibitor, 1 h before formalin injection was shown to induce dose-dependent inhibition of the inflammatory phase only [56]. The lack of significant reduction in paw flinching across all extract-treated groups in the initial phase suggested that the mature and young leaf extracts might have limited central analgesic property but instead are able to exert their effects peripherally to achieve pain relief. This selectivity implied that L. elliptica might be more beneficial for managing inflammatory pain rather than acute nociceptive pain. Therefore, we postulated that L. elliptica leaf extracts may impart their analgesic activity by inhibiting the production or action of inflammatory pain mediators. Goh et al. [27] summarised the analgesic potential of nine Litsea species, and six of them, L. cubeba, L. japonica, L. glutinosa, L. lancifolia, L. liyuyingi, and L. monopetala, demonstrated peripheral antinociceptive properties. The antinociceptive mechanism of L. cubeba and L. japonica has been primarily attributed to the regulatory effects of their bioactive compounds on inflammatory mediators [47,50,51,53]. While there are limited molecular studies on the inflammatory pain phase actions of the other Litsea species mentioned above, these species have shown similar potencies in the acetic acid-induced writhing models.
Antipyretic activity of L. elliptica mature and young leaf extracts in pyretic model
Antipyretic
activity of L.
elliptica
mature and young leaf extracts were evaluated by monitoring the
progression of ∆RT of pyretic rats following treatments. NC
demonstrated the largest ∆RT across all time points, with
temperature increases ranging from 2.05 to 2.15 °C, indicating a
persistent febrile response in the model. Rats treated with mature
leaf extracts showed significant (p
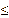 0.05) reduction in ∆RT relative to NC (Figure
9(a)).
By the end of the assay, ∆RT4
remained below 1 °C, comparable to that observed in PC, suggesting
a potent effect of mature leaf extracts against brewer’s
yeast-induced pyrexia. Treatment with YMACE50 showed significant (p
0.05) reduction in ∆RT relative to NC (Figure
9(a)).
By the end of the assay, ∆RT4
remained below 1 °C, comparable to that observed in PC, suggesting
a potent effect of mature leaf extracts against brewer’s
yeast-induced pyrexia. Treatment with YMACE50 showed significant (p
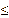 0.05) reductions at 1, 2, and 4 h, while YMAE400 showed significant
(p
0.05) reductions at 1, 2, and 4 h, while YMAE400 showed significant
(p
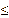 0.05) reductions only at 1 and 4 h relative to NC (Figure
9(b)).
Young leaf extracts demonstrated comparatively weaker potency and
less consistent antipyretic activity. Although
the SD of the ∆RT
appears numerically large (
0.05) reductions only at 1 and 4 h relative to NC (Figure
9(b)).
Young leaf extracts demonstrated comparatively weaker potency and
less consistent antipyretic activity. Although
the SD of the ∆RT
appears numerically large (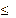 ±
0.6), such variation is also observed in other in vivo pyrexia
studies and reflects normal biological and thermoregulatory
differences among animals.
±
0.6), such variation is also observed in other in vivo pyrexia
studies and reflects normal biological and thermoregulatory
differences among animals.
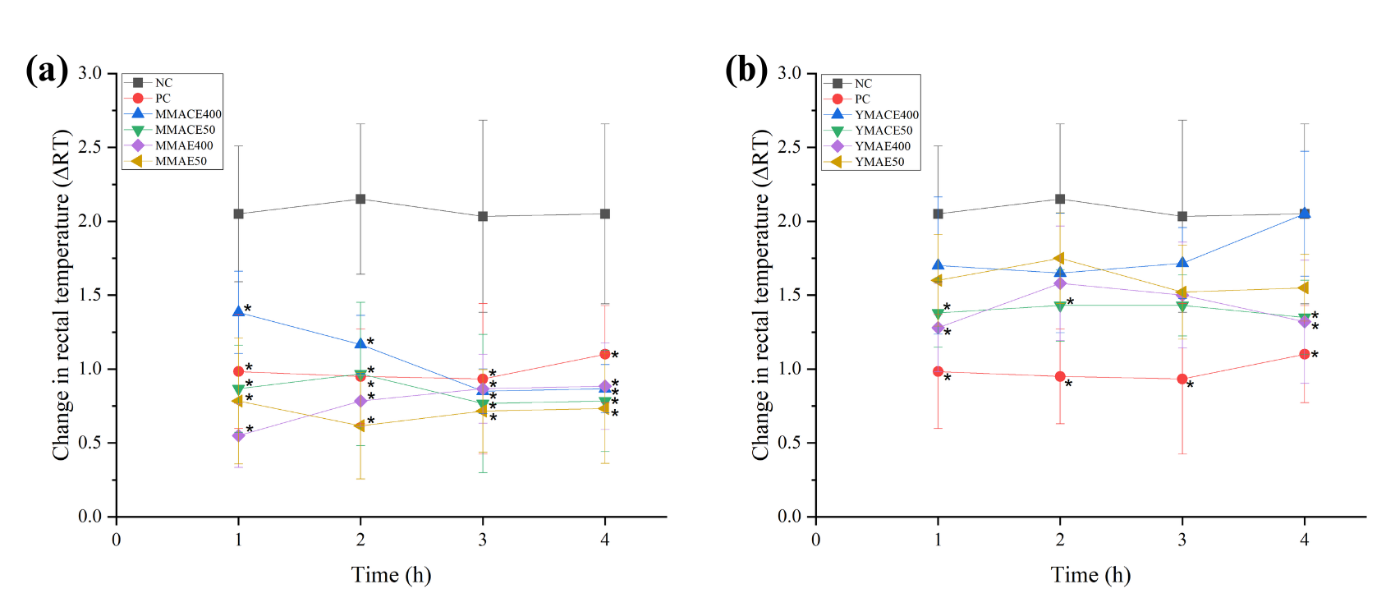
Figure 9 Changes in rectal temperature (∆RT) at each time point in pyretic rats following treatments with L. elliptica (a) mature leaf extracts and (b) young leaf extracts obtained by MACE and MAE at 400 and 50 mg/kg b.w. Sterile saline (1 mL/100 g b.w.) served as NC, whereas paracetamol (150 mg/kg b.w.) was used as PC. Values are expressed as mean ∆RTn ± SD; * indicates p ≤ 0.05 significant differences against NC analysed using one-way ANOVA followed by Tukey’s test.
Brewer’s yeast derived from Saccharomyces cerevisiae is a widely utilised fungal pyrogen for inducing pathogenic fever in rodents, especially in ethnopharmacological studies screening for potential antipyretic agents [57]. The results of this study indicated that L. elliptica mature and young leaf extracts exhibited strong to limited antipyretic activities, respectively. Notably, mature leaf extract showed comparable activity to paracetamol, suggesting that the antipyretic effects of the extracts might be mediated through the inhibition of COX-2 activity, thereby reducing the concentration of PGs in the hypothalamus. Given that the anti-inflammatory and analgesic effects of L. elliptica have been proposed to suppress the expression of COX-2, PGE2, NO, TNF-α, IL-6, and IL-1β by regulating JAK2/STAT3 and NF-κB signaling pathways, it is plausible that its antipyretic activity may also operate through these mechanisms. Several Litsea species, including L. cubeba, L. pungens, L. khasyana, L. laeta and L. guatemalensis, have been traditionally used in various Asian communities to treat fever [28]. However, as mentioned earlier, only L. glutinosa has been scientifically investigated for its antipyretic activity, whereby a similar speculation on the MOA of L. glutinosa leaf extracts has been suggested [29]. Direct evidence on the antipyretic mechanism of L. elliptica is still lacking, and studies exploring the antipyretic activity of Litsea species remain limited, despite their widespread traditional use for managing fever. Nevertheless, our findings indicated the promising potential of L. elliptica as an antipyretic agent.
Possible mechanisms underlying the effects of L. elliptica mature and young leaf extracts
The pathways underlying inflammation, pain, and fever are closely interconnected, with numerous common mediators such as COX-2, PGE2, TNF-α, IL-6, and IL-1β playing crucial roles in driving the development and progression of these responses [58,59]. Consequently, an agent that targets these shared mediators may simultaneously exert anti-inflammatory, analgesic, and antipyretic activities. In the present study, L. elliptica leaf extracts were found to modulate these 3 responses, albeit at variable efficacy. We postulated that the effects seen in L. elliptica may be due to its ability to act on these shared mediators. This suggestion can be supported by studies on the effects of phytochemical components in L. elliptica leaf extracts sourced from different plants on COX-2, PGE2, TNF-α, IL-6, and IL-1β expression. In addition to the abovementioned alkaloids, flavonoids such as quercetin and its derivatives have been shown to suppress the overexpression of COX-2, TNF-α, IL-6, and IL-1β in a number of cultured cells and animal models [60]. Xiao et al. [61] unveiled quercetin’s interesting capability of lowering COX-2 expression and PGE2 production through the inhibition of multiple key transactivators such as NF-κB, CREB2, C-Jun, and C/EBPβ in human breast cancer cells. This suppression was linked to quercetin's ability to inhibit p300 histone acetyltransferase (HAT) activity, thereby disrupting the binding of transcriptional coactivators to COX-2 promoter. Vitamin E, although it does not appear to influence COX-1 or COX-2 expression at the transcriptional level, supplementation studies have reported its ability to inhibit COX-2 enzymatic activity, leading to reduced PGE₂ synthesis [62,63]. This post-translational regulation was suggested to be mediated by the neutralisation of reactive oxygen species (ROS) such as peroxynitrite [64]. As mentioned above, several studies have reported the radical scavenging ability of different parts of L. elliptica [17,19-21]. This may further support its role in modulating the COX pathway, particularly through the neutralisation of ROS involved in COX-2 activation.
Seven fatty acids (FAs) have been identified from mixed leaf extracts using gas chromatography-mass spectrometry (GC-MS), including lauric acid, myristic acid, linoleic acid, and stearic acid [17]. Termer et al. (2011) studied COX-2 inhibition of Waltheria indica leaf extract, with α-linolenic acid and linoleic acid contributing up to 41% of the inhibition observed in the extracts [65]. Meanwhile, co-treatment with myristic acid and heptadecanoic acid has been shown to reduce the expression of TNF-α, IL-6, and IL-1β via NF-κB pathway in LPS-stimulated BV-2 microglial cells [66]. Interaction of FAs with the 2 binding sites - catalytic (Ecat) and allosteric (Eallo) subunits – of COX-1 and COX-2 have been discussed by Smith and Malkowski [67] where they showed that some FAs such as eicosapentaenoic acid (EPA), docosapentaenoic acid (DPA), and docosahexaenoic acid (DHA) could compete with arachidonic acid for Ecat on COX-1 and -2, thus, interfering the production of PGs. All these findings highlighted the potential contribution of phytochemical compounds found in L. elliptica extracts in targeting key mediators in the COX pathway across inflammation, pain, and fever pathways. Nonetheless, further research is required to confirm the effects of mature and young leaf extracts and their corresponding compounds on these mediators.
Conclusions
This study seeks to provide insights into the pharmacological potential of L. elliptica and contribute to a broader understanding of its role as a promising source of natural medicinal agents. Overall, our findings revealed that the mature and young leaf extracts of L. elliptica had significant anti-inflammatory, analgesic, and antipyretic activities when tested on various animal models. Bioactive compounds previously isolated from the leaf extracts, such as alkaloids, quercetin, vitamin E, and FAs, have the capability of modulating both upstream and downstream processes in the COX pathway. This warrants further investigation to explore its potential as a treatment for inflammation, pain, and fever.
Acknowledgements
The authors would like to acknowledge UBD BRC and Brunei National Herbarium for their support in the collection of plant samples. We also thank the technical staff from the Environmental and Life Sciences Programme, Faculty of Science, Universiti Brunei Darussalam for their assistance. This research was funded by the Universiti Brunei Darussalam Grant number: UBD/RSCH/URC/NIG/1.0/2022/008.
Declaration of Generative AI in Scientific Writing
The authors recognize that generative AI tools such as Grammarly and OpenAI’s ChatGPT were utilized during the preparation of this manuscript, exclusively for language refinement and grammar editing. No section of the content was generated, nor was any data interpreted, by AI. The authors take full responsibility for the content and conclusion of this work.
CRediT Author Statement
Raudhatun Samsul: Conceptualization; Methodology; Investigation; Formal analysis; Writing - Original Draft; Writing - Review & Editing; Visualization; Project administration. May Goh: Conceptualization; Methodology; Writing - Original Draft; Writing - Review & Editing; Visualization; Supervision; Project administration; Funding acquisition. Hussein Taha: Writing - Original Draft; Writing - Review & Editing. Norhayati Ahmad: Conceptualization; Methodology; Validation; Writing - Original Draft; Writing - Review & Editing; Supervision; Project administration; Funding acquisition.
[1] N Bakar, FM Franco and NH Hassan. The intersection of Kedayan folk medicine and traditional ecological calendar. In: FM Franco, M Knudsen and NH Hassan (Eds.). Case studies in biocultural diversity from Southeast Asia. Spinger, Singapore, 2022, p. 105-124.
[2] MJR Desborough and DM Keeling. The aspirin story - from willow to wonder drug. British Journal of Haematology 2017; 177(5), 674-683.
[3] Y Tu. Artemisinin - a gift from Traditional Chinese Medicine to the world (nobel lecture). Angewandte Chemie International Edition 2016; 55(35), 10210-10226.
[4] S Foster. From herbs to medicines: the Madagascar periwinkle’s impact on childhood leukemia: A serendipitous discovery for treatment. Alternative and Complementary Therapies 2010; 16(6), 347-350.
[5] N Kifli, Z Wint, J Nh and J Nh. Prevalence on the use of traditional medicine in Brunei Darussalam. Journal of Homeopathy & Ayurvedic Medicine 2014; 3(155), 2167-1206.
[6] V Roseberg. 2017, Brunei Malay traditional medicine: Persistence in the face of western medicine and Islamic orthodoxy. Ph. D. Dissertation. The University of Western Australia, Perth, Australia.
[7] K Kamsani, FM Franco and F Slik. A comparative account of the traditional healing practices of healers and non-healers in the Kiudang area of Brunei Darussalam. Journal of Ethnopharmacology 2020; 256, 112788.
[8] MPY Goh, AM Basri, H Yasin, H Taha and N Ahmad. Ethnobotanical review and pharmacological properties of selected medicinal plants in Brunei Darussalam: Litsea elliptica, Dillenia suffruticosa, Dillenia excelsa, Aidia racemosa, Vitex pinnata and Senna alata. Asian Pacific Journal of Tropical Biomedicine 2017; 7(2), 173-180.
[9] RPJD Kok. A revision of Litsea (Lauraceae) in Peninsular Malaysia and Singapore. Garden’s Bulletin Singapore 2021; 73(1), 81-178.
[10] N Chaisoung, H Balslev, R Suksathan, P Panyadee, C Long, C Ngernsaengsaruay, T Chaowasku and A Inta. Essential oil and phylogenetic positions of five medicinal Litsea species (Lauraceae). Diversity 2025; 17(3), 168.
[11] F Awang-Kanak and MF Abu Bakar. Traditional vegetable salad (ulam) of Borneo as source of functional food. Food Research 2019; 4(1), 1-12.
[12] H Kuspradini, MS Mala, AS Putri, NA Sa’adah and Kiswanto. Effect of leaf maturity and solvent extract on the antioxidant activity of Litsea elliptica. Journal of the Korean Wood Science and Technology 2024; 52(5), 450-458.
[13] S Jiwajinda, V Santisopasri, A Murakami, OK Kim, HW Kim and H Ohigashi. Suppressive effects of edible Thai plants on superoxide and nitric oxide generation. Asian Pacific Journal of Cancer Prevention 2002; 3(3), 215-223.
[14] PW Grosvenor, PK Gothard, NC McWilliam, A Supriono and DO Gray. Medicinal plants from Riau Province, Sumatra, Indonesia. part 1: Uses. Journal of Ethnopharmacology 1995; 45(2), 75-95.
[15] S Omar, M Latip and NH Ismail. The use of plants in the management of health of the Melanau community in Sarawak (in Malaysian). In: Proceedings of the 8th Antarabangsa Sains Sosial & Kemanusiaan, Selangor, Malaysia. 2023, p. 2811-4051.
[16] S Phoopha, C Wattanapiromsakul, T Pitakbut and S Dej-adisai. Chemical constituents of Litsea elliptica and their alpha-glucosidase inhibition with molecular docking. Pharmacognosy Magazine 2020; 16(70), 327-334.
[17] MPY Goh, AF Kamaluddin, TJL Tan, H Yasin, H Taha, A Jama and N Ahmad. An evaluation of the phytochemical composition, antioxidant and cytotoxicity of the leaves of Litsea elliptica Blume - An ethnomedicinal plant from Brunei Darussalam. Saudi Journal of Biological Sciences 2022; 29(1), 304-317.
[18] K Nakahara, G Trakoontivakorn, NS Alzoreky, H Ono, M Onishi-Kameyama and M Yoshida. Antimutagenicity of some edible Thai plants, and a bioactive carbazole alkaloid, mahanine, isolated from Micromelum minutum. Journal of Agricultural and Food Chemistry 2002; 50(17), 4796-4802.
[19] F Aryani, IW Kusuma, Y Meliana, NM Sari and H Kuspradini. Potential antibacterial and antioxidant activities of ten essential oils from East Kalimantan, Indonesia. Biodiversitas Journal of Biological Diversity 2023; 24(1), 665-672.
[20] N Chaichanawongsaroj, S Amonyingcharoen, P Pattiyathanee, R Vilaichone and Y Poovorawan. Anti-Helicobacter pylori and anti-internalization activities of Thai folk remedies used to treat gastric ailments. Journal of Medicinal Plants Research 2012; 6(8), 1389-1393.
[21] MH Wong, LF Lim, F Ahmad and Z Assim. Antioxidant and antimicrobial properties of Litsea elliptica Blume and Litsea resinosa Blume (Lauraceae). Asian Pacific Journal of Tropical Biomedicine 2014; 4(5), 386-392.
[22] T Lawal, C Slover, V Lee and GB Mahady. In vitro susceptibility of oral pathogens to traditional medicines used to treat gingivitis and peridontal infections. Planta Medica 2016; 82(5), PB29.
[23] SN Ain, S Masran, H Othman, SB Budin, S Nor, AS Masran, R Salji, SB Budin and IS Taib. Acute toxicity (oral) information of Litsea elliptica Blume essential oil. In: Proceedings of the International Conference on Bioscience, Biochemistry and Bioinformatics, Singapore. 2011, p. 399-403.
[24] SB Budin, SMSN Ain, B Omar, IS Taib and O Hidayatulfathi. Acute and subacute oral toxicity of Litsea elliptica Blume essential oil in rats. Journal of Zhejiang University - Science B 2012; 13(10), 783-790.
[25] IS Taib, SB Budin, SMSN Ain, J Mohamed, SR Louis, S Das, S Sallehudin, NF Rajab and O Hidayatulfathi. Toxic effects of Litsea elliptica Blume essential oil on red blood cells of Sprague-Dawley rats. Journal of Zhejiang University - Science B 2009; 10(11), 813-819.
[26] MK Pillai, F Yakop, N Metussin, MH Hamid, H Yasin, M Majid and DJ Young. Phytochemical characterization of essential oils from shoots, mature leaves and branchlets of Litsea elliptica (Lauraceae) collected in Brunei Darussalam. Scientia Bruneiana 2018; 17(2), 18-22.
[27] MPY Goh, RN Samsul, AW Mohaimin, HP Goh, NH Zaini, N Kifli and N Ahmad. The analgesic potential of Litsea species: A systemic review. Molecules 2024; 29(9), 2079.
[28] G Li, Z Li and Y Wang. The genus Litsea: A comprehensive review of traditional uses, phytochemistry, pharmacological activities and other studies. Journal of Ethnopharmacology 2024; 334, 118494.
[29] R Bhowmick, MS Sarwar, SMR Dewan, A Das, B Das, MMN Uddin, MS Islam and MS Islam. In vivo analgesic, antipyretic, and anti-inflammatory potential in Swiss albino mice and in vitro thrombolytic activity of hydroalcoholic extract from Litsea glutinosa leaves. Biological Research 2014; 47(1), 56.
[30] Z Liu, H Li, Y Qi, Z Zhu, D Huang, K Zhang, J Pan, L Wen and Z Zou. Cinnamomum camphora leaves as a source of proanthocyanidins separated using microwave-assisted extraction method and evaluation of their antioxidant activity in vitro. Arabian Journal of Chemistry 2021; 14(9), 103328.
[31] DGN Pastura, JDV Sousa, MC Silva, AF Matos, HLCD Santos, GL Pereira, EBD Rocha, RS Alves, LDO Bastos, LDO Pires, RN Castro, HR Borba and VMD Lima. Persea americana Mill.: Evaluation of cytogenotoxicity and phytochemical prospection of leaf extracts. Brazilian Journal of Pharmaceutical Sciences 2022; 58, e19261.
[32] NA Shafie, NA Suhaili, H Taha and N Ahmad. Evaluation of antioxidant, antibacterial and wound healing activities of Vitex pinnata. F1000Research 2020; 9, 187.
[33] MT Mansouri, AA Hemmati, B Naghizadeh, SA Mard, A Rezaie and B Ghorbanzadeh. A study of the mechanisms underlying the anti-inflammatory effect of ellagic acid in carrageenan-induced paw oedema in rats. Indian Journal of Pharmacology 2015; 47(3), 292-298.
[34] SM Soliman, MH Teaima, KO Rashwan, BM Ali, BR Jasti, MA El-Nabarawi and SM Abd El-Halim. The deleterious effect of xylene-induced ear oedema in rats: protective role of dexketoprofen trometamol transdermal invasomes via inhibiting the oxidative stress/NF-κB/COX-2 pathway. International Journal of Pharmaceutics 2023; 631, 122525.
[35] RI Erickson, MC Terzi, SM Jaber, FC Hankenson, A McKinstry-Wu, MB Kelz and JO Marx. Intraperitoneal continuous-rate for the maintenance of anesthesia infusion in laboratory mice (Mus musculus). Journal of the American Association for Laboratory Animal Science 2016; 55(5), 548-557.
[36] A Roca-Vinardell, E Berrocoso, M Llorca-Torralba, JA García-Partida, J Gibert-Rahola and JA Mico. Involvement of 5-HT1A/1B receptors in the antinociceptive effect of paracetamol in the rat formalin test. Neurobiology of Pain 2018; 3, 15-21.
[37] MMO Rashid, A Islam, MS Amran and MA Hossain. Evaluation of analgesic activity by acetic acid induced writhing method of crude extracts of Acacia nilotica. Scholars Academic Journal of Pharmacy 2017; 6(4), 126-138.
[38] AD Forkuo, KB Mensah, EO Ameyaw, AO Antwi, NK Kusi-Boadum and C Ansah. Antiplasmodial and antipyretic activity and safety evaluation of the methanolic leaf extract of Murraya exotica (L.). Journal of Parasitology Research 2020; 2020, 1308541.
[39] KR Patil, UB Mahajan, BS Unger, SN Goyal, S Belemkar, SJ Surana, S Ojha and CR Patil. Animal models of inflammation for screening of anti-inflammatory drugs: Implications for the discovery and development of phytopharmaceuticals. International Journal of Molecular Sciences 2019; 20(18), 4367.
[40] X Yang, X Gao, Y Cao, Q Guo, S Li, Z Zhu, Y Zhao, P Tu and X Chai. Anti-Inflammatory effects of boldine and reticuline isolated from Litsea cubeba through JAK2/STAT3 and NF-κB signaling pathways. Planta Medica 2018; 84(1), 20-25.
[41] M Eddouks, D Chattopadhyay, VD Feo and WC Cho. Medicinal plants in the prevention and treatment of chronic diseases. Evidence-Based Complementary and Alternative Medicine 2012; 2012, 458274.
[42] SD Douglas and SE Leeman. Neurokinin-1 receptor: Functional significance in the immune system in reference to selected infections and inflammation. Annals of the New York Academy of Sciences 2011; 1217(1), 83-95.
[43] HW Koon and C Pothoulakis. Immunomodulatory properties of substance P: The gastrointestinal system as a model. Annals of the New York Academy of Sciences 2006; 1088(1), 23-40.
[44] MT Islam, SA Ayatollahi, SMNK Zihad, N Sifat, MR Khan, A Paul, B Salehi, T Islam, MS Mubarak, N Martins and J Sharifi-Rad. Phytol anti-inflammatory activity: Pre-clinical assessment and possible mechanism of action elucidation. Cellular and Molecular Biology 2020; 66(4), 264-269.
[45] RA Abdel-Emam and AM Abd-Eldayem. Systemic and topical Ginkgo biloba leaf extract (Egb-761) ameliorated rat paw inflammation in comparison to dexamethasone. Journal of Ethnopharmacology 2022; 282, 114619.
[46] B Lin, H Zhang, XX Zhao, K Rahman, Y Wang, XQ Ma, CJ Zheng, QY Zhang, T Han and LP Qin. Inhibitory effects of the root extract of Litsea cubeba (lour.) pers. on adjuvant arthritis in rats. Journal of Ethnopharmacology 2013; 147(2), 327-334.
[47] EM Choi and JK Hwang. Effects of methanolic extract and fractions from Litsea cubeba bark on the production of inflammatory mediators in RAW264.7 cells. Fitoterapia 2004; 75(2), 141-148.
[48] PC Liao, TS Yang, JC Chou, J Chen, SC Lee, YH Kuo, CL Ho and LKP Chao. Anti-inflammatory activity of neral and geranial isolated from fruits of Litsea cubeba Lour. Journal of Functional Foods 2015; 19, 248-258.
[49] L Xia, R Li, T Tao, R Zhong, H Du, Z Liao, Z Sun and C Xu. Therapeutic potential of Litsea cubeba essential oil in modulating inflammation and the gut microbiome. Frontiers in Microbiology 2023; 14, 1233934.
[50] YM Ham, YJ Ko, SM Song, J Kim, KN Kim, JH Yun, JH Cho, G Ahn and WJ Yoon. Anti-inflammatory effect of litsenolide B2 isolated from Litsea japonica fruit via suppressing NF-κB and MAPK pathways in LPS-induced RAW264.7 cells. Journal of Functional Foods 2015; 13, 80-88.
[51] QMT Ngo, TQ Cao, PL Tran, JA Kim, ST Seo, JC Kim, MH Woo, JH Lee and BS Min. Lactones from the pericarps of Litsea japonica and their anti-inflammatory activities. Bioorganic & Medicinal Chemistry Letters 2018; 28(11), 2109-2115.
[52] ER Puppala, Md Abubakar, V Swamy, SS Yalamarthi, S NP, AN Prakash, JK Gangasani, D Chamundeswari and VGM Naidu. Litsea salicifolia Roxb. bark ethanolic extract attenuates rheumatoid arthritis by inhibiting TLR4/NF-κB and activating SIRT1-Nrf2/HO-1 signaling axis: In-vitro and in-vivo evaluations. Phytomedicine Plus 2023; 3(1), 100390.
[53] HJ Koo, WJ Yoon, EH Sohn, YM Ham, SA Jang, JE Kwon, YJ Jeong, JH Kwak, E Sohn, SY Park, KH Jang, S Namkoong, HS Han, YH Jung and SC Kang. The analgesic and anti-inflammatory effects of Litsea japonica fruit are mediated via suppression of NF-κB and JNK/p38 MAPK activation. International Immunopharmacology 2014; 22(1), 84-97.
[54] YH Zhang, D Adamo, H Liu, Q Wang, W Wu, YL Zheng and XQ Wang. Editorial: Inflammatory pain: Mechanisms, assessment, and intervention. Frontiers in Molecular Neuroscience 2023; 16, 1286215.
[55] MF Yam, YC Loh, CW Oo and R Basir. Overview of neurological mechanism of pain profile used for animal “pain-like” behavioral study with proposed analgesic pathways. International Journal of Molecular Sciences 2020; 21(12), 4355.
[56] YQ Zhao, HY Wang, JB Yin, Y Sun, Y Wang, JC Liang, XJ Guo, K Tang and YT Wang. The analgesic effects of celecoxib on the formalin-induced short- and long-term inflammatory pain. Pain Physician 2017; 20(4), E575-E584.
[57] R Dangarembizi, KH Erlwanger, C Rummel, J Roth, MT Madziva and LM Harden. Brewer’s yeast is a potent inducer of fever, sickness behavior and inflammation within the brain. Brain, Behavior, and Immunity 2018; 68, 211-223.
[58] CA Dinarello. Overview of the IL‐1 family in innate inflammation and acquired immunity. Immunological Reviews 2018; 281(1), 8-27.
[59] C Liu, D Chu, K Kalantar‐Zadeh, J George, HA Young and G Liu. Cytokines: From clinical significance to quantification. Advanced Science 2021; 8(15), 2004433.
[60] A Septembre-Malaterre, A Boumendjel, ALS Seteyen, C Boina, P Gasque, P Guiraud and J Sélambaro. Focus on the high therapeutic potentials of quercetin and its derivatives. Phytomedicine Plus 2022; 2(1), 100220.
[61] X Xiao, D Shi, L Liu, J Wang, X Xie, T Kang and W Deng. Quercetin suppresses cyclooxygenase-2 expression and angiogenesis through inactivation of P300 signaling. PLoS One 2011; 6(8), e22934.
[62] KA O’Leary, S de Pascual-Tereasa, PW Needs, YP Bao, NM O’Brien and G Williamson. Effect of flavonoids and vitamin E on cyclooxygenase-2 (COX-2) transcription. Mutation Research/Fundamental and Molecular Mechanisms of Mutagenesis 2004; 551(1-2), 245-254.
[63] NY Park, S Im and Q Jiang. Different forms of vitamin E and metabolite 13’-carboxychromanols inhibit cyclooxygenase-1 and its catalyzed thromboxane in platelets, and tocotrienols and 13’-carboxychromanols are competitive inhibitors of 5-lipoxygenase. The Journal of Nutritional Biochemistry 2022; 100, 108884.
[64] H Slika, H Mansour, N Wehbe, SA Nasser, R Iratni, G Nasrallah, A Shaito, T Ghaddar, F Kobeissy and AH Eid. Therapeutic potential of flavonoids in cancer: ROS-mediated mechanisms. Biomedicine & Pharmacotherapy 2022; 146, 112442.
[65] M Termer, C Carola, A Salazar, CM Keck, J Hemberger and J von Hagen. Activity-guided characterization of COX-2 inhibitory compounds in Waltheria indica L. extracts. Molecules 2021; 26(23), 7240.
[66] Q Huang, C Chen, Z Zhang and Q Xue. Anti-inflammatory effects of myristic acid mediated by the NF-κB pathway in lipopolysaccharide-induced BV-2 microglial cells. Molecular Omics 2023; 19(9), 726-734.
[67] WL Smith and MG Malkowski. Interactions of fatty acids, nonsteroidal anti-inflammatory drugs, and coxibs with the catalytic and allosteric subunits of cyclooxygenases-1 and -2. Journal of Biological Chemistry 2019; 294(5), 1697-1705.