
Trends
Sci.
2026;
23(6):
12723
Optimization of Laccase Production by Megasporoporia sp. for Bioremoval of Methyl Orange and Cr(VI)
Titan Dwikama Putra1,2, Wichanee Bankeeree2 and Sehanat Prasongsuk2,*
1Program in Biotechnology, Faculty of Science, Chulalongkorn University, Bangkok 10330, Thailand
2Plant Biomass Utilization Research Unit, Department of Botany, Faculty of Science, Chulalongkorn University, Bangkok 10330, Thailand
(*Corresponding author’s e-mail: sehanat.p@chula.ac.th)
Received: 20 November 2025, Revised: 9 December 2025, Accepted: 19 December 2025, Published: 15 February 2026
Abstract
Industrial expansion has accelerated the discharge of hazardous pollutants, particularly azo dyes such as methyl orange (MO) and heavy metals such as hexavalent chromium [Cr(VI)], which pose serious environmental and health risks, especially when both contaminants coexist in wastewater. Enzymatic bioremediation offers a sustainable alternative to conventional physicochemical treatments, and this study aimed to optimize laccase production by the white-rot fungus Megasporoporia sp. and evaluate its ability to simultaneously remove MO and Cr(VI). Laccase biosynthesis was optimized using a One-factor-at-a-time (OFAT) approach by systematically varying nutritional and physicochemical factors, including carbon sources, nitrogen sources, copper concentration, pH, temperature, agitation speed, and incubation time. The highest yield was achieved under optimal conditions: glucose or sucrose as the carbon source, a combination of yeast extract and ammonium sulfate as the nitrogen source, 1.5 - 2.0 mM CuSO4, pH 5.0, incubation at 28 °C for 5 days, and static cultivation. The crude enzyme exhibited strong pollutant removal in the single-pollutant systems, achieving 74.17% MO removal and 98.67% Cr(VI) removal, whereas its performance declined in the dual-pollutant system due to inhibitory interactions between the two contaminants (42.46% MO removal and 87.57% Cr(VI) removal). Phytotoxicity assays using Vigna radiata demonstrated that laccase substantially mitigated toxicity in the single-pollutant treatments, improving germination from 22% to 64% for MO and from 16% to 82% for Cr(VI), whereas only partial recovery was observed in the dual-pollutant system. These findings indicate that Megasporoporia sp. can efficiently produce high laccase yields under mild, low-cost conditions, and that the crude enzyme is highly effective for individual MO and Cr(VI) remediation, although simultaneous removal remains constrained by pollutant interactions. The study highlights the potential of laccase-based bioremediation for wastewater treatment while underscoring the need for further strategies, such as enzyme immobilization, to enhance performance in complex contaminant systems.
Keywords: Optimization, One-factor-at-a-time (OFAT), Laccase, Megasporoporia sp., Bioremoval, MO, Cr(VI), Simultaneous removal
Introduction
As industrialization has advanced, the global industrial sector has undergone rapid expansion to meet market demands. Examples of such industries include textiles, dye manufacturing, leather, and paper pulp production. Although this development contributes significantly to national economic growth, these industries also generate a massive amount of wastewater [1,2]. This issue becomes critical when waste management practices are inadequate, leading to the direct discharge of effluents into the environment. The resulting impact can lead to severe environmental problems, as industrial effluents generally contain hazardous substances that are persistent and harmful to living organisms. Among these pollutants, synthetic dyes and heavy metals are xenobiotic compounds frequently found in such wastewater [3-5].
Azo dyes are among the most widely used types of synthetic dyes in various industrial processes. At least 70% of dyes used in these industries are azo dyes [6]. One example of a commonly used azo dye is methyl orange (MO; 4-dimethylaminoazobenzene 4’-sulfonic acid sodium salt). Studies have shown that MO and its intermediates, such as dimethyl benzyl-1,4-diamine, sulfonamide, and 1,4-diaminobenzene, are carcinogenic, mutagenic, and teratogenic compounds [7,8]. Meanwhile, heavy metals such as hexavalent chromium [Cr(VI)] can also be found in leather industry. This is because chromium sulfate (Cr(OH)SO4) is usually used as a tanning agent. Although it is predominantly present as trivalent chromium [Cr(III)], a fraction of it can be oxidized to Cr(VI) under certain conditions [9]. Cr(VI) represents the most toxic oxidation state of chromium and is reported to be up to 100 times more toxic than Cr(III). Due to its structural similarity to sulfate ions, Cr(VI) can easily enter cells through sulfate channels, leading to cellular damage [10,11]. Moreover, MO and Cr(VI) have been reported to coexist in various types of wastewater, thereby enhancing its toxicity [12].
Several studies have developed various physicochemical methods for wastewater treatment containing MO and Cr(VI). These methods include adsorption [13], electrocoagulation [14,15], ion exchange [16,17], and catalytic reduction [18,19]. Although these approaches show promising results, they generally have several drawbacks, such as high operational costs, secondary sludge generation, incomplete pollutant removal, and limited applicability to large-scale systems [20]. In contrast, biological methods utilizing microorganisms have been considered as alternative approaches since they can overcome some of the limitations of physicochemical techniques. However, under actual wastewater conditions, nutrient limitations and extreme environmental factors can inhibit microbial growth, thereby reducing their remediation efficiency [21]. Considering these challenges, enzyme-based bioremediation has recently attracted increasing attention. Instead of employing whole microbial cells, this approach utilizes the enzymes produced by microorganisms to transform hazardous compounds into less toxic or non-toxic forms.
Laccase (EC 1.10.3.2) is an oxidoreductase class enzyme that mediates the oxidation of aromatic and non-aromatic substrates by reducing molecular oxygen to water [22]. Laccase has been reported to be produced by various microorganisms, although it is predominantly known to be synthesized by white-rot fungi (WRF) [23]. Among microbial enzymes, laccase holds significant economic value and has demonstrated great potential in wastewater treatment. The major advantage of this enzyme lies in its broad catalytic ability to degrade a wide range of pollutants [24,25]. Silveira et al. [26], reported that laccase derived from Marasmiellus palmivorus efficiently decolorizes MO. Another study also highlighted the potential of laccase produced by Ganoderma multipileum in reducing Cr(VI) [27]. Nevertheless, to the best our knowledge simultaneous enzymatic remediation of MO and Cr(VI) has not been previously reported. Therefore, this study aims to optimize laccase production from the WRF Megasporoporia sp. and to evaluate its potential to simultaneously remediate MO and Cr(VI).
Materials and methods
Fungal culture
The WRF used in this study, Megasporoporia sp., was obtained from the Plant Biomass Utilization Research Unit (PBURU), Department of Botany, Faculty of Science, Chulalongkorn University. The culture was maintained on Potato Dextrose Agar (PDA) containing (g/L): 200 potato infusion, 20 dextrose, and 15 agar, and stored at 4 °C. Prior to further experiments, the WRF was routinely subcultured on fresh PDA plates and incubated at room temperature for 1 week.
Laccase production and optimization
Megasporoporia sp. was previously reported by Lestari et al. [28] to produce laccase in a liquid medium containing (g/L): 10 glucose, 1 KH2PO4, 0.5 MgSO4, 0.14 CaCl2, 0.0025 thiamine, and 0.4 mM veratryl alcohol, adjusted to pH 5.0. However, the resulting laccase yield was relatively low. Therefore, in this study, laccase production by Megasporoporia sp. was optimized using a one-factor-at-a-time (OFAT) approach with a modified medium composition according to Othman et al. [29] (g/L): 10.5 glucose, 5 yeast extract, 2 (NH4)2SO4, 0.5 K2HPO4, 0.5 MgSO4, 0.02 FeSO4, 0.3 CaHPO4, 0.2 ZnSO4, 0.2 MnSO4, and 0.25 CuSO4, adjusted to pH 5.0. Five mycelial plugs (Ø ~1 cm) from a 1-week-old culture were inoculated into the liquid medium. Subsequently, the crude enzyme was obtained by separating the culture supernatant from the fungal biomass using Whatman No. 1 filter paper.
Several factors were evaluated to determine the optimal conditions for laccase production by Megasporoporia sp. These factors included the carbon sources (rice straw, sugarcane leaf, rice husk, glucose, sucrose, starch), organic nitrogen sources (beef extract (BE), meat extract (ME), peptone (PE), yeast extract (YE)) paired with inorganic nitrogen sources (ammonium chloride (AC), sodium nitrate (SN), potassium nitrate (PN), ammonium sulfate (AS)), copper concentration (0.5 - 2.0 mM), pH (3.0 - 8.0), temperature (25 - 30 °C), agitation speed (0 - 200 rpm), and incubation time (5 - 21 days). Each factor was evaluated independently to identify conditions that maximized laccase production by Megasporoporia sp.
Laccase activity and protein content assay
Laccase activity in the crude enzyme was determined spectrophotometrically based on the oxidation of 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) at 420 nm. The reaction mixture consisted of 50 μL of crude enzyme and 500 μL of 2 mM ABTS, with the final volume adjusted to 1000 μL using 0.1 M citrate buffer (pH 3.0). One unit (U) of laccase activity was defined as the amount of enzyme required to oxidize 1 μmol of ABTS per minute, calculated according to the formula described by Anita et al. [30] (Eq. (1)).
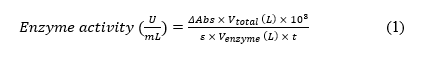
where ΔAbs is the difference between the final and initial absorbance, 103 is the correction factor (µmol/mol), ε is the molar absorptivity (3.6×104 M⁻¹ cm⁻¹), and t is the reaction time (min).
Meanwhile, the total protein content in the crude enzyme was determined using the method of Lowry et al. [31], with bovine serum albumin (BSA) used as the standard.
Bioremoval of MO and Cr(VI)
The laccase produced by Megasporoporia sp. was evaluated for its potential to remove MO and Cr(VI) from synthetic wastewater. In this study, crude enzyme with an initial activity of 1 U/mL was added to 5 mL of solution containing 50 mg/L of MO or Cr(VI) for a single-pollutant system, and 50 mg/L each of MO and Cr(VI) for a dual-pollutant system. The mixtures were then incubated at 28 °C and shaken at 150 rpm for 48 h in the dark to prevent photodegradation. Samples were collected periodically to determine the removal of MO and Cr(VI). The decolorization of MO was analyzed spectrophotometrically at 462 nm [32], while the reduction of Cr(VI) was measured using the 1,5-diphenylcarbazide (DPC) method, in which the resulting purple complex was quantified at 540 nm [33]. The bioremoval efficiencies of MO and Cr(VI) were calculated using the following formula (Eq. (2)):
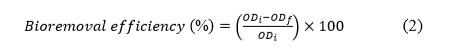
where ODi is the optical density before removal, and ODf is the optical density after removal.
Phytotoxicity evaluation
The phytotoxicity evaluation was conducted to determine whether the bioremoval products of MO and Cr(VI) by the laccase produced by Megasporoporia sp. still exhibited phytotoxic effects on plants. In this assay, mung bean (Vigna radiata) seeds were used as a plant model due to their sensitivity to azo dyes and heavy metals [34]. Prior to use, the seeds were surface-sterilized by soaking in 70% ethanol for 10 min, followed by several rinses with sterile deionized water (DI). Ten seeds were placed in a Petri dish containing pre-sterilized Whatman No. 1 filter paper, onto which 5 mL of the treated synthetic wastewater was added. DI water served as the negative control, while untreated synthetic wastewater served as the positive control. The plates were then incubated in the dark for 1 week [35]. The percent germination (Eq. (3)), germination index (Eq. (4)), and percent germination index (Eq. (5)) were calculated using the following formulas [36]:

where l is the total sum of root lengths of the germinated seeds in each replicate, and n is the number of germinated seeds.
Statistical analysis
All experiments were conducted in triplicate (n = 3), while the phytotoxicity evaluation was performed in 5 replicates (n = 5). Statistical differences among treatments were assessed using 1-way ANOVA with a significance level of p < 0.05. When significant differences were detected, Tukey’s HSD post-hoc test was applied for pairwise comparisons. Data visualization and statistical analyses were carried out using GraphPad Prism version 8 (GraphPad Software, San Diego, CA, USA) and SPSS Statistics version 23 (IBM Corp., Armonk, NY, USA).
Results and discussion
Laccase production and optimization
Effect of carbon source
The type of carbon source plays a significant role in laccase production by WRF. In this study, when glucose was used, Megasporoporia sp. exhibited the highest laccase activity. However, this value was not significantly different from that obtained with sucrose (26.22 U/mL), as shown in Figure 1. A similar observation was reported by Umar and Ahmed [37], who demonstrated that both the monosaccharide (glucose) and the disaccharide (sucrose) enhanced laccase activity in Ganoderma leucocontextum. These findings suggest that, in certain fungal species, glucose and sucrose may induce the expression of laccase-encoding genes more effectively than other carbon sources [38]. Based on the results of this study, sucrose may therefore serve as an alternative carbon source for laccase production by Megasporoporia sp., considering that it is more cost-effective than glucose [39].
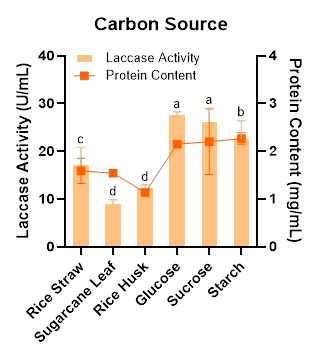
Figure 1 Effect of different carbon source on laccase activity and protein content produced by Megasporoporia sp.
Data are presented as mean ± SD (n = 3). Bars sharing different letters differ significantly in enzyme activity (p < 0.05; 1-way ANOVA followed by Tukey’s HSD test).
Effect of nitrogen source
Ardila-Leal et al. [40] explained that the simultaneous use of organic and inorganic nitrogen sources can enhance laccase activity due to nitrogen assimilation. In Figure 2, the combination of yeast extract and ammonium sulfate is shown to significantly increase laccase activity compared to other nitrogen combinations. Similar findings have also been reported in several other WRF species, such as Pleurotus ostreatus [41], Pleurotus sajor-caju [42], and Pycnoporus sanguineus [43], which demonstrated a positive effect of yeast extract supplementation on laccase productivity. This preference for yeast extract in fungal laccase production is likely due not only to its role as a nitrogen source but also to the presence of protein hydrolysates and vitamins that enhance laccase biosynthesis [44]. Meanwhile, Umar and Ahmed [37] also reported that ammonium sulfate can effectively stimulate laccase production in Ganoderma leucocontextum.
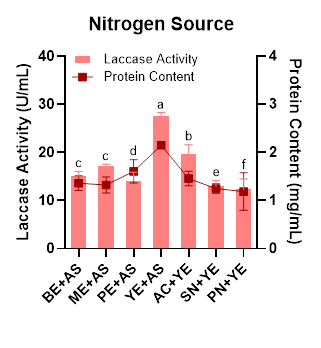
Figure 2 Effect of different nitrogen source on laccase activity and protein content produced by Megasporoporia sp.
Data are presented as mean ± SD (n = 3). Bars sharing different letters differ significantly in enzyme activity (p < 0.05; 1-way ANOVA followed by Tukey’s HSD test).
Effect of copper concentration
Since the catalytic active site of laccase contains copper atoms, the availability of copper in the culture medium plays a crucial role in regulating laccase biosynthesis. Copper acts as an inducer by activating the transcription of laccase-encoding genes. However, at higher concentrations, copper can exert toxic effects on fungal cells, impairing growth and consequently reducing enzyme productivity [45]. In this study, laccase activity increased proportionally with copper concentration and reached its maximum at 2.0 mM (Figure 3). A previous report also demonstrated that supplementation with 0.5 - 2.0 mM copper enhanced pel3 gene transcription and extracellular laccase activity in Pleurotus eryngii KS004 [46]. Despite this, the enzyme activity observed at 1.5 mM was not significantly different from that at 2.0 mM. Therefore, 1.5 mM can be considered the optimal concentration for inducing laccase production, as it provides high activity while being more cost-effective and reducing potential cytotoxic stress.
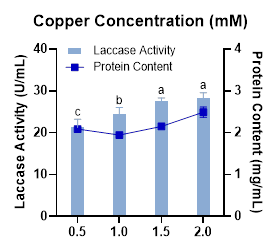
Figure 3 Effect of copper concentration on laccase activity and protein content produced by Megasporoporia sp.
Data are presented as mean ± SD (n = 3). Bars sharing different letters differ significantly in enzyme activity (p < 0.05; 1-way ANOVA followed by Tukey’s HSD test).
Effect of pH value
At the lowest growth medium pH (pH 3.0), the laccase activity was the lowest. However, as the pH slightly increased, laccase activity also increased, reaching its highest peak at pH 5.0 (Figure 4). The optimal pH for laccase production varies among fungi, although many species generally produce laccase most effectively within the pH range of 3.0 - 6.0 [47]. The results also show that further increases in pH led to a gradual decline in laccase activity. A similar pattern was reported for Pestalotiopsis sp. CDBT-F-G1, where laccase activity decreased when the growth medium pH exceeded 5.0 [48]. According to Salem et al. [49], the increased presence of OH⁻ ions at higher medium pH can interact with the type 2 and type 3 copper centers in laccase. These copper centers form the catalytic sites of the enzyme; thus, higher pH conditions can reduce the redox potential of the substrate and consequently decrease laccase activity.
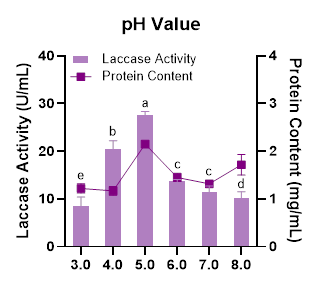
Figure 4 Effect of medium pH on laccase activity and protein content produced by Megasporoporia sp.
Data are presented as mean ± SD (n = 3). Bars sharing different letters differ significantly in enzyme activity (p < 0.05; 1-way ANOVA followed by Tukey’s HSD test).
Effect of temperature
Laccase production by Megasporoporia sp. was highest at 28 °C (Figure 5). The lowest enzyme activity occurred at 25 °C, which is likely associated with reduced metabolic and enzymatic reaction rates at lower temperatures. Temperatures that are too low or below the optimal range can slow fungal growth and consequently reduce its enzymatic activity [50]. Conversely, temperatures exceeding the optimum may lead to partial enzyme denaturation, thereby diminishing catalytic efficiency [51]. These results are consistent with findings by Edae and Alemu [52], who reported 28 °C as the optimal temperature for laccase production in Pleurotus sp. Similarly, Sura et al. [53] observed comparable temperature-dependent behavior in Trichoderma harzianum. In general, each fungal species has a specific temperature range that supports maximal enzyme biosynthesis, and most studies indicate that optimal laccase production commonly occurs between 20 - 30 °C [42]. Based on these results, it can be concluded that Megasporoporia sp. can produce high levels of laccase at room temperature, indicating that no additional temperature control is required to achieve optimal enzyme production.
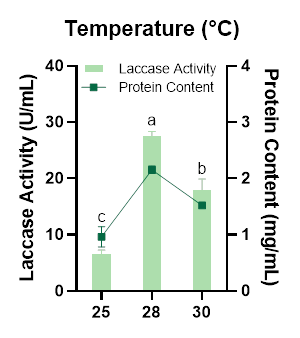
Figure 5 Effect of temperature on laccase activity and protein content produced by Megasporoporia sp.
Data are presented as mean ± SD (n = 3). Bars sharing different letters differ significantly in enzyme activity (p < 0.05; 1-way ANOVA followed by Tukey’s HSD test).
Effect of agitation speed
Laccase production under different agitation speeds was evaluated. Although previous reports generally indicate that shaking conditions enhance laccase production in filamentous fungi due to improved oxygen transfer and homogenized nutrient distribution [54,55], the present study demonstrated that Megasporoporia sp. exhibited higher laccase productivity under static cultivation (Figure 6). In contrast, cultures subjected to increased agitation speeds showed a pronounced decrease in laccase activity, with the most significant reduction observed at 200 rpm. A study by Krumova et al. [56] reported similar results in Trametes trogii, which produced higher laccase activity under static conditions compared to shaking conditions. It is likely that the cell walls of Megasporoporia sp. possess relatively low shear resistance. Under agitated conditions, hydrodynamic shear stress could negatively affect mycelial integrity and growth, and this suppression of biomass development would directly reduce laccase production [57,58]. Moreover, high agitation rates generate strong shear forces that can disrupt fungal physiological homeostasis, ultimately inhibiting enzyme biosynthesis [59].
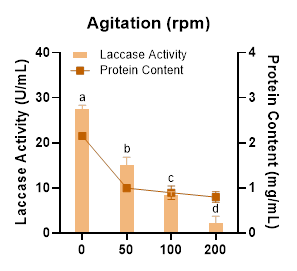
Figure 6 Effect of agitation speed on laccase activity and protein content produced by Megasporoporia sp.
Data are presented as mean ± SD (n = 3). Bars sharing different letters differ significantly in enzyme activity (p < 0.05; 1-way ANOVA followed by Tukey’s HSD test).
Effect of incubation time
Determining the optimal incubation time is necessary to identify the appropriate harvesting period for laccase. After 5 days of incubation, laccase activity reached its highest level at 30.57 U/mL (Figure 7). In the subsequent incubation intervals, the activity showed a slight decline, and by days 14 and 21, the laccase activity dropped sharply to 5.22 and 3.37 U/mL, respectively. Similar findings were reported for Aspergillus niger IBP2013 and Curvularia lunata MY3, which also exhibited an optimal incubation time of 5 days for laccase production [60,61]. Therefore, it can be concluded that Megasporoporia sp. is able to produce high laccase activity within a relatively short incubation period, which is advantageous for production efficiency.
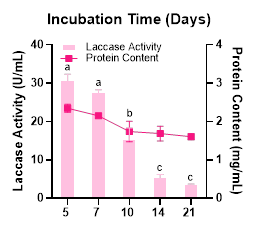
Figure 7 Effect of incubation time on laccase activity and protein content produced by Megasporoporia sp.
Data are presented as mean ± SD (n = 3). Bars sharing different letters differ significantly in enzyme activity (p < 0.05; 1-way ANOVA followed by Tukey’s HSD test).
Collectively, this study demonstrates that optimized conditions resulted in more than a 30-fold increase in laccase production compared to previous unpublished data obtained by Lestari et al. [28]. Although optimization using the OFAT approach successfully enhanced laccase production, this method is unable to reveal interactions among factors. Therefore, in future studies, the most influential factors (e.g., carbon source, nitrogen source, and copper concentration) should be further investigated to determine their interactive effects on laccase production by Megasporoporia sp., for example by applying response surface methodology (RSM) [62].
Bioremoval of MO and Cr(VI)
In the single-pollutant system, laccase showed remarkable efficiency in removing both MO and Cr(VI), achieving approximately 74.17% and 98.67% removal, respectively (Figures 8(a) and 8(b)). This strong performance highlights the enzyme’s versatility in acting on both metal and organic pollutants. The higher Cr(VI) removal can be explained by the enzyme’s ability to reduce toxic Cr(VI) to its less toxic Cr(III), a process that has been reported in previous studies. For example, the laccase produced by Ganoderma multipileum was found to eliminate over 94% of Cr(VI) [27]. Furthermore, the MO decolorization in this study showed a comparable result to that reported by Girelli et al. [63], where the application of laccase for MO decolorization demonstrated notably high catalytic efficiency, achieving around 71% decolorization under optimized conditions. MO decolorization involves oxidative cleavage of the azo (–N=N–) bond and breakdown of aromatic ring reactions that are often slower and less complete [64,65].
When both MO and Cr(VI) were present together (dual-pollutant system), the removal efficiency of each pollutant decreased slightly compared to the single-pollutant setup (Figure 8(c)). Cr(VI) removal remained relatively high (87.57%), whereas MO removal dropped more noticeably (42.46%). This pattern suggests that interactions between the 2 contaminants influence enzymatic activity and reaction kinetics. However, when pollutant concentration is high, combined toxicity may inhibit enzyme activity, leading to an overall reduction in removal performance [66]. Therefore, the observed differences between the single- and dual-pollutant systems likely reflect a balance between synergistic stimulation of enzyme activity and the inhibitory effects caused by the coexisting pollutants. To address this limitation, enzyme immobilization for further applications is highly recommended, as it can enhance both the stability and catalytic performance of the enzyme, thereby potentially improving its bioremoval efficiency toward pollutants [67].
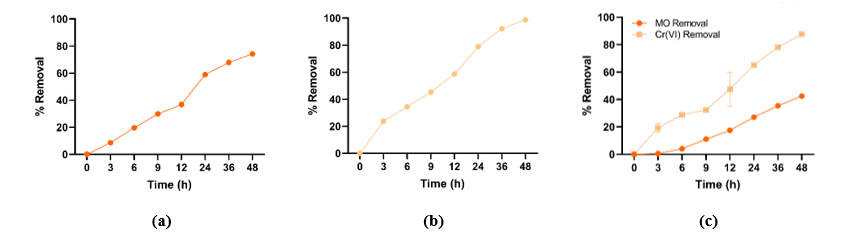
Figure 8 (a) MO removal in the single-pollutant system; (b) Cr(VI) removal in the single-pollutant system; and (c) Simultaneous removal in the dual-pollutant system (orange line: MO; yellow line: Cr(VI)).
Phytotoxicity evaluation
The phytotoxicity evaluation using V. radiata showed that both MO and Cr(VI) exhibited strong inhibitory effects on seed germination and growth, indicating their toxicity to plants (Table 1). The addition of laccase reduced this toxicity in both MO and Cr(VI) treatments, as reflected by higher germination (from 22% to 64% for MO and from 16% to 82% for Cr(VI)) and higher germination indices (from 2.24% to 37.57% for MO and from 0.83% to 68.50% for Cr(VI)), along with lower growth inhibition compared to the pollutants alone. This suggests that laccase effectively detoxifies these contaminants, likely by degrading MO’s azo bonds and reducing Cr(VI) to the less toxic Cr(III) form. Moreover, in the dual-pollutant system, phytotoxicity remained pronounced (8% germination and 0.17% GI), and although laccase treatment slightly improved germination (60%) and GI (31.60%), the enzyme’s protective effect was less substantial than in the single-pollutant systems. The higher toxicity of MO and Cr(VI), particularly in combination, is consistent with previous findings that both pollutants induce oxidative stress, inhibit nutrient uptake, and disrupt photosynthetic activity in plants [68, 69]. The coexistence of MO and Cr(VI) likely causes synergistic stress, increasing reactive oxygen species accumulation and cellular damage. Meanwhile, laccase’s partial detoxification effect aligns with its known ability to oxidize aromatic dyes and reduce metal toxicity [70].
Table 1 Germination, root elongation, and GI of V. radiata under MO, Cr(VI), and mixed-pollutant treatments.
Treatment |
% Germination |
Root elongations (cm) |
% GI |
DI Water |
100 ± 0.00a |
65.50 ± 0.35a |
100 ± 5.47a |
MO |
22 ± 8.36d |
5.94 ± 0.24d |
2.24 ± 1.51e |
Laccase-treated MO |
64 ± 8.94c |
38.02 ± 0.39c |
37.57 ± 8.62c |
Cr(VI) |
16 ± 11.4de |
2.56 ± 0.17d |
0.83 ± 0.76f |
Laccase-treated Cr(VI) |
82 ± 13.03b |
53.66 ± 0.93b |
68.50 ± 22.3b |
MO and Cr(VI) |
8 ± 4.47e |
1.20 ± 0.06e |
0.17 ± 0.11f |
Laccase-treated MO and Cr(VI) |
60 ± 10.0c |
33.80 ± 0.53c |
31.60 ± 10.06d |
Description: Different superscript letters within the same column indicate significant differences (p < 0.05; n = 5; 1-way ANOVA followed by Tukey’s HSD test).
Conclusions
The OFAT optimization of laccase production by Megasporoporia sp. resulted in the following optimal conditions: glucose or sucrose as the carbon source, a combination of yeast extract and ammonium sulfate as the nitrogen source, 1.5 - 2.0 mM CuSO4, pH 5.0, incubation at 28 °C for 5 days, and static cultivation. The crude enzyme produced by this fungus demonstrated considerable potential for MO and Cr(VI) removal in the single-pollutant system, achieving 74.17% MO removal and 98.67% Cr(VI) removal. Meanwhile, in the dual-pollutant system, the crude enzyme achieved 42.46% MO removal and 87.57% Cr(VI) removal. In addition, the phytotoxicity evaluation using V. radiata revealed that laccase treatment improved germination from 22% to 64% for MO and from 16% to 82% for Cr(VI), while the dual-pollutant system showed a more limited improvement (from 8% to 60% germination). These results indicate that laccase from Megasporoporia sp. holds promising potential as an eco-friendly and safe approach for treating wastewater containing MO and Cr(VI) in the future.
Acknowledgements
The authors gratefully acknowledge the financial support from Chulalongkorn University, Thailand through the ASEAN and Non-ASEAN Countries Scholarship for the completion of this study.
Declaration of generative AI in scientific writing
The authors admit the use of generative AI tool (ChatGPT by OpenAI) for language and grammatical editing during the arrangement of this manuscript. AI tool did not contribute to content or data interpretation. All authors remain the responsibility for the manuscript’s content and conclusions.
CRediT author statement
Titan Dwikama Putra: Methodology; Investigation; Data curation; Formal analysis; Writing - Original Draft. Wichanee Bankeeree: Project administration; Supervision; Writing - Review & Editing; Resources. Sehanat Prasongsuk: Conceptualization; Validation; Supervision; Funding acquisition; Project administration.
References
[1] A Azanaw, B Birlie, B Teshome and M Jemberie. Textile effluent treatment methods and eco-friendly resolution of textile wastewater. Case Studies in Chemical and Environmental Engineering 2022; 6, 100230.
[2] J Zhao, Q Wu, Y Tang, J Zhou and H Guo. Tannery wastewater treatment: Conventional and promising processes, an updated 20-year review. Journal of Leather Science and Engineering 2022; 4(1), 10.
[3] Arti and R Mehra. Analysis of heavy metals and toxicity level in the tannery effluent and the environs. Environmental Monitoring and Assessment 2023; 195(5), 554.
[4] R Etana, K Angassa and T Getu. Dye removal from textile wastewater using scoria-based of vertical subsurface flow constructed wetland system. Scientific Reports 2025; 15(1), 949.
[5] SH Fouda, ESA El-Halim and HAA Ghany. Radiological and chemical hazards of persistent organic pollutants in the textile sector. Scientific Reports 2025; 5(1), 20102.
[6] S Benkhaya, S M'Rabet and A El Harfi. Classifications, properties, recent synthesis and applications of azo dyes. Heliyon 2020; 6(1), e03271.
[7] Asranudin, AS Purnomo, Holilah, D Prasetyoko, N El Messaoudi, AA Rohmah, ARP Hidayat and R Subagyo. Adsorption and biodegradation of the azo dye methyl orange using Ralstonia pickettii immobilized in polyvinyl alcohol (PVA)-alginate-hectorite beads (BHec-RP). RSC Advances 2024; 14(26), 18277-18290.
[8] SK Dutta, MK Amin, J Ahmed, M Elias and M Mahiuddin. Removal of toxic methyl orange by a cost-free and eco-friendly adsorbent: Mechanism, phytotoxicity, thermodynamics, and kinetics. South African Journal of Chemical Engineering 2022; 40, 195-208.
[9] P Prete, S Acocella, A Intiso and R Cucciniello. Determination of Cr (VI) released by leather samples using smartphone-based colorimetry for on-site determination and miniaturization as greener preparation strategy. Green Analytical Chemistry 2025; 12, 100232.
[10] A Arishi and I Mashhour. Microbial mechanisms for remediation of hexavalent chromium and their large-scale applications; current research and future directions. Journal of Pure and Applied Microbiology 2021; 15(1), 53-67.
[11] X Wang, H Li, H Huang, H Luo, S Luo, L Jiang, S Feng and H Xu. Investigation on mechanism of Hexavalent chromium bioreduction by Escherichia sp. TH-1 and the stability of reduction products. Journal of Environmental Chemical Engineering 2022; 10(3), 6805.
[12] C Du, X Chen, H Wu, Z Pan, C Chen, G Zhong and C Cai. A novel cationic covalent organic framework as adsorbent for simultaneous removal of methyl orange and hexavalent chromium. RSC Advances 2023; 13(34), 24064-24070.
[13] U Ghani, K Hina, M Iqbal, MK Irshad, I Aslam, R Saeed and M Ibrahim. Kinetic and isotherms modeling of methyl orange and chromium (VI) onto hexagonal ZnO microstructures as a membrane for environmental remediation of wastewater. Chemosphere 2022; 309(2), 136681.
[14] SH Abbas, YM Younis, KH Rashid and AA Khadom. Removal of methyl orange dye from simulated wastewater by electrocoagulation technique using Taguchi method: Kinetics and optimization approaches. Reaction Kinetics, Mechanisms and Catalysis 2022; 135(5), 2663-2679.
[15] E Aguilar-Ascon, L Marrufo-Saldana and W Neyra-Ascon. Enhanced chromium removal from tannery wastewater through electrocoagulation with iron electrodes: Leveraging the Box-Behnken design for optimization. Heliyon 2024; 10(3), e24647.
[16] LC Flint, MS Arias-Paić and JA Korak. Removal of hexavalent chromium by anion exchange: Non-target anion behavior and practical implications. Environmental Science: Water Research & Technology 2021; 7(12), 2397-2413.
[17] MI Khan, A Shanableh, N Elboughdiri, MH Lashari, S Manzoor, S Shahida, N Farooq, Y Bouazzi, S Rejeb, Z Elleuch, K Kriaa and AU Rehman. Adsorption of methyl orange from an aqueous solution onto a BPPO-Based Anion Exchange Membrane. ACS Omega 2022; 7(30), 26788-26799.
[18] S Bawazeer. A potential eco-friendly degradation of methyl orange by water-ball (sodium polyacrylate) stabilized zero valent iron nanoparticles. Heliyon 2025; 11(1), e41226.
[19] R Kajala, SS Tomar, N Verma and KDP Nigam. Catalytic reduction of in-flow aqueous Cr(vi) using a slurry of activated carbon fiber-supported Ni nanoparticles in a coiled flow inverter. Reaction Chemistry & Engineering 2022; 7(12), 2508-2517.
[20] R Al-Tohamy, SS Ali, F Li, KM Okasha, YA Mahmoud, T Elsamahy, H Jiao, Y Fu and J Sun. A critical review on the treatment of dye-containing wastewater: Ecotoxicological and health concerns of textile dyes and possible remediation approaches for environmental safety. Ecotoxicology and Environmental Safety 2022; 231, 113160.
[21] N Thirumalaivasan, L Gnanasekaran, S Kumar, R Durvasulu, T Sundaram, S Rajendran, S Nangan and K Kanagaraj. Utilization of fungal and bacterial bioremediation techniques for the treatment of toxic waste and biowaste. Frontiers in Materials 2024; 11, 1416445.
[22] V Hahn. Potential of the enzyme laccase for the synthesis and derivatization of antimicrobial compounds. World Journal of Microbiology and Biotechnology 2023; 39(4), 107.
[23] LB Zhang, XG Qiu, TT Qiu, Z Cui, Y Zheng and C Meng. A complex metabolic network and its biomarkers regulate laccase production in white-rot fungus Cerrena unicolor 87613. Microbial Cell Factories 2024; 23(1), 167.
[24] L Arregui, M Ayala, X Gomez-Gil, G Gutierrez-Soto, CE Hernandez-Luna, MH de Los Santos, L Levin, A Rojo-Dominguez, D Romero-Martinez, MCN Saparrat, MA Trujillo-Roldan and NA Valdez-Cruz. Laccases: Structure, function, and potential application in water bioremediation. Microbial Cell Factories 2019; 18(1), 200.
[25] H Wang, LX Tang, YF Ye, JX Ma, X Li, J Si and BK Cui. Laccase immobilization and its degradation of emerging pollutants: A comprehensive review. Journal Environment Management 2024; 359, 120984.
[26] TR Silveira, CD Ebling, LD Magro, RC Rodrigues, WD Hahn Schneider, M Camassola, E Weber de Menezes, Á Meneguzzi and MP Klein. An efficient decolorization of methyl orange dye by laccase from Marasmiellus palmivorus immobilized on chitosan-coated magnetic particles. Biocatalysis and Agricultural Biotechnology 2020; 30, 101859.
[27] MA Alshiekheid, A Umar, F Ameen, SA Alyahya and L Dufossé. Biodegradation of chromium by laccase action of Ganoderma multipileum. Journal of King Saud University - Science 2023; 35(10), 102948.
[28] IP Lestari, S Prasongsuk, P Lotrakul, H Punnapayak and W Bankeeree. Decolorization of remazol brilliant blue R with immobilized laccase from Megasporoporia sp. onto spent coffee ground biochar. Asia-Pacific Journal of Science and Technology 2024; 29(06), 06-16.
[29] AM Othman, M Mahmoud, M Abdelraof, GSA Abdel Karim and AM Elsayed. Enhancement of laccase production from a newly isolated Trichoderma harzianum S7113 using submerged fermentation: Optimization of production medium via central composite design and its application for hydroquinone degradation. International Journal of Biological Macromolecules 2021; 192, 219-231.
[30] SH Anita, FC Ardiati, M Oktaviani, FP Sari, OD Nurhayat, KP Ramadhan and DHY Yanto. Immobilization of laccase from Trametes hirsuta EDN 082 in light expanded clay aggregate for decolorization of Remazol Brilliant Blue R dye. Bioresource Technology Reports 2020; 12, 100602.
[31] O Lowry, N Rosebrough, AL Farr and R Randall. Protein measurement with the folin phenol reagent. Journal of Biological Chemistry 1951; 193(1), 265-275.
[32] G Du, Y Ding, C Li, L Zhang, J Li, M Li, W Zhu and C He. Preparation of Cu/Cu(2)O/BC and its performance in adsorption-photocatalytic degradation of methyl orange in water. Materials 2024; 17(17), 4306.
[33] S Akkurt, M Oguz and A Alkan Uckun. Bioreduction and bioremoval of hexavalent chromium by genetically engineered strains (Escherichia coli MT2A and Escherichia coli MT3). World Journal of Microbiology and Biotechnology 2022; 38(3), 45.
[34] Y Bilal, T Sarwar, E Mazhar, F Mahmood, T Shahzad, DA Al-Farraj, I Alzaidi, MS Elshikh, TW Chen and S Hussain. Reduction in phytotoxicity of a textile wastewater against Vigna radiata using Citrobacter sp. M41 in a bioaugmented packed bed column bioreactor. Journal of King Saud University - Science 2024; 36(2), 103030.
[35] D Pandey, A Daverey, K Dutta and K Arunachalam. Bioremoval of toxic malachite green from water through simultaneous decolorization and degradation using laccase immobilized biochar. Chemosphere 2022; 297, 134126.
[36] F Bandini, E Taskin, F Vaccari, M Soldano, S Piccinini, A Frache, S Remelli, C Menta, PS Cocconcelli and E Puglisi. Anaerobic digestion and aerobic composting of rigid biopolymers in bio-waste treatment: Fate and effects on the final compost. Bioresource Technology 2022; 351, 126934.
[37] A Umar and S Ahmed. Optimization, purification and characterization of laccase from Ganoderma leucocontextum along with its phylogenetic relationship. Scientific Reports 2022; 12(1), 2416.
[38] P Vasudhevan, D Kalaimurugan, S Ganesan, N Akbar, S Dixit and S Pu. Enhanced biocatalytic laccase production using agricultural waste in solid-state fermentation by Aspergillus oryzae for p-chlorophenol degradation. International Journal of Biological Macromolecules 2024; 281(3), 136460.
[39] D Ni, Z Chen, Y Tian, W Xu, W Zhang, BG Kim and W Mu. Comprehensive utilization of sucrose resources via chemical and biotechnological processes: A review. Biotechnology Advances 2022; 60, 107990.
[40] LD Ardila-Leal, MF Alvarado-Ramirez, IS Gutierrez-Rojas, RA Poutou-Pinales, B Quevedo-Hidalgo, A Perez-Florez and AM Pedroza-Rodriguez. Low-cost media statistical design for laccase rPOXA 1B production in P. pastoris. Heliyon 2020; 6(4), e03852.
[41] C Zhu, G Bao and S Huang. Optimization of laccase production in the white-rot fungus Pleurotus ostreatus (ACCC 52857) induced through yeast extract and copper. Biotechnology & Biotechnological Equipment 2016; 30(2), 270-276.
[42] S Sharma and DS Murty. Enhancement of laccase production by optimizing the cultural conditions for Pleurotus sajor-caju in solid-state fermentation. Journal of Pure and Applied Microbiology 2021; 15(2), 958-967.
[43] E González-Bautista, E Alarcón-Gutierrez, N Dupuy, I Gaime-Perraud, F Ziarelli and AM Farnet-da-Silva. Influence of yeast extract enrichment and Pycnoporus sanguineus inoculum on the dephenolisation of sugar-cane bagasse for production of second-generation ethanol. Fuel 2020; 260, 116370.
[44] C Velásquez-Quintero, A Merino-Restrepo and A Hormaza-Anaguano. Production, extraction, and quantification of laccase obtained from an optimized solid-state fermentation of corncob with white-rot fungi. Journal of Cleaner Production 2022; 370, 133598.
[45] F Wang, X Yu, Z Yu, Y Cui, L Xu, S Huo, Z Ding, L Zhao, L Du and Y Qiu. Improved laccase production by Trametes versicolor using Copper-Glycyl-L-Histidyl-L-Lysine as a novel and high-efficient inducer. Front Bioeng Biotechnol 2023; 11, 1176352.
[46] S Sharghi, FS Ahmadi, AM Kakhki and M Farsi. Copper increases laccase gene transcription and extracellular laccase activity in Pleurotus eryngii KS004. Brazilian Journal of Microbiology 2024; 55(1), 111-116.
[47] A Gałązka, U Jankiewicz and A Szczepkowski. Biochemical characteristics of laccases and their practical application in the removal of xenobiotics from water. Applied Sciences 2023; 13(7), 4394.
[48] M Yadav, G Bista, R Maharjan, P Poudyal, M Mainali, L Sreerama and J Joshi. Secretory laccase from Pestalotiopsis Species CDBT-F-G1 fungal strain isolated from high altitude: Optimization of its production and characterization. Applied Sciences 2019; 9(2), 340.
[49] MM Salem, TM Mohamed, AM Shaban, YA Mahmoud, MA Eid and NA El-Zawawy. Optimization, purification and characterization of laccase from a new endophytic Trichoderma harzianum AUMC14897 isolated from Opuntia ficus-indica and its applications in dye decolorization and wastewater treatment. Microbial Cell Factories 2024; 23(1), 266.
[50] L Tian, Y Li, J Yang, L Qin, H Ma, L Zhu and Z Yin. Promoting the achievement of wastewater treatment by microalgae-based co-culture systems: From interactions mechanisms to pollution control performance. Energy Conversion and Management 2024; 320, 118981.
[51] NR Oloye, BA Oyedeji, MO Taiwo, SO Adebajo, AK Akintokun, JA Akamo and BT Folarin. Multifaceted screening and optimization of laccase-producing rhizospheric yeast for enhanced biosynthesis of laccase enzyme. The Microbe 2025; 7, 100419.
[52] T Edae and M Alemu. Selection and optimization of lignocellulosic substrate for laccase production from Pleurotus species. International Journal of Biotechnology and Molecular Biology Research 2017; 8(4), 38-48.
[53] NI Sura, AJ Hashim and WM Abood. Biodegradation of some environmental pollutants by laccase produced from Trichoderma harzianum using solid state fermentation. Iraqi Journal of Agricultural Sciences 2022; 53(3), 522- 533.
[54] D Yao, Y Ma, J Ran, J Wang, U Kues, J Liu, D Zhou, X Zhang, Z Fang and Y Xiao. Enhanced extracellular production of laccase in Coprinopsis cinerea by silencing chitinase gene. Applied Microbiology and Biotechnology 2024; 108(1), 324.
[55] AJ Fernandes, VP Shibukawa, AMR Prata, F Segato, JC Dos Santos, A Ferraz and AMF Milagres. Using low-shear aerated and agitated bioreactor for producing two specific laccases by Trametes versicolor cultures induced by 2,5-xylidine: Process development and economic analysis. Bioresource Technology 2024; 401, 130737.
[56] E Krumova, N Kostadinova, J Miteva-Staleva, G Stoyancheva, B Spassova, R Abrashev and M Angelova. Potential of ligninolytic enzymatic complex produced by white-rot fungi from genus Trametes isolated from Bulgarian forest soil. Engineering in Life Sciences 2018; 18(9), 692-701. .
[57] Y Adamian, L Lonappan, K Alokpa, SN Agathos and H Cabana. Recent developments in the immobilization of laccase on carbonaceous supports for environmental applications - a critical review. Frontiers in Bioengineering and Biotechnology 2021; 9, 778239.
[58] F Wang, M Wang, M Wang, L Xu, J Qian, G Guan and B Xu. Clarification of sugarcane juice catalyzed by magnetic immobilized laccase intensified by alternating magnetic field. Foods 2025; 14(3),444.
[59] A Umar, I Abid, MS Elshikh, L Dufosse, AM Abdel-Azeem and I Ali. Agitation role (Dissolved Oxygen) in production of laccase from newly identified Ganoderma multistipitatum sp. nov. and its effect on mycelium morphology. BMC Microbiology 2023; 23(1), 280.
[60] AA Hamed, AM Abd-Elaziz, MME Ghanem, ME ElAwady and MS Abdel-Aziz. Production of laccase enzyme from Curvularia lunata MY3: Purification and characterization. Folia Microbiologica 2024; 69(1), 221-234.
[61] S Hasan, Z Anwar, W Khalid, F Afzal, M Zafar, U Ali, MY Refai, M Afifi, A Al-Farga and MO Aljobair. Laccase production from local biomass using solid state fermentation. Fermentation 2023; 9(2), 179.
[62] MA Abdel-Rahman, SED Hassan, MN El-Din, MS Azab, EF El-Belely, HMA Alrefaey and T Elsakhawy. One-factor-at-a-time and response surface statistical designs for improved lactic acid production from beet molasses by Enterococcus hirae ds10. SN Applied Sciences 2020; 2(4), 573.
[63] AM Girelli, E Pambianco and FR Scuto. Sustainable recycling of spent grain for laccase immobilization as dyes removal tool. Journal of Environmental Chemical Engineering 2021; 9(6), 106653.
[64] M Arif. Catalytic reduction/degradation of methyl orange by metal nanoparticle containing systems: a critical review. RSC Advances 2025; 15(34), 27668-27684.
[65] Z Li, Q Zhu, Z Liu, L Sha and Z Chen. Improved performance of immobilized laccase for catalytic degradation of synthetic dyes using redox mediators. New Journal of Chemistry 2022; 46(20), 9792-9798.
[66] BE Igiri, SIR Okoduwa, GO Idoko, EP Akabuogu, AO Adeyi and IK Ejiogu. Toxicity and bioremediation of heavy metals contaminated ecosystem from tannery wastewater: A review. Journal of Toxicology 2018; 2018, 2568038.
[67] W Zhang, Y Zhang, Z Lu, B Nian, S Yang and Y Hu. Enhanced stability and catalytic performance of laccase immobilized on magnetic graphene oxide modified with ionic liquids. Journal Environment Management 2023; 346, 118975.
[68] P Kumar and N Thakur. Examination of seed germination process and assess phytotoxicity in Vigna radiata (Mung bean) and Allium cepa (Onion) using triple-doped green hematite nanoparticles and their photocatalytic potential. Environmental Science and Pollution Research 2025. https://doi.org/10.1007/s11356-025-36735-3
[69] D Srivastava, M Tiwari, P Dutta, P Singh, K Chawda, M Kumari and D Chakrabarty. Chromium Stress in Plants: Toxicity, Tolerance and Phytoremediation. Sustainability 2021; 13(9), 4629.
[70] S Morsy, A Ahmad Tajudin, MSM Ali and FM Shariff. Current development in decolorization of synthetic dyes by immobilized laccases. Frontiers in Microbiology 2020; 11, 572309.