
Trends
Sci.
2026;
23(6):
12479
Unravelling the Bifunctional Potential of Phylloplane Yeasts from Rose Flowers: Zinc Solubilization and Biocontrol of Fungal Phytopathogens
Passorn Jongkaewvijit1, Noramon Panchanon1, Nawarat Hongthong1,
Sopitar Soponputtaporn1, Kriengsak Thaipong2, Janpen Tangjitjaroenkun3,
Moltira Srithaworn1 and Orawan Chunhachart1,4,*
1Division of Microbiology, Department of Science and Bioinnovation, Faculty of Liberal Arts and Science,
Kasetsart
University, Kamphaeng Saen Campus, Nakhon Pathom
73140,
Thailand
2Department
of Horticulture, Faculty of Agriculture, Kamphaeng Saen, Kasetsart
University, Kamphaeng Saen Campus, Nakhon Pathom 73140, Thailand
3
Department
of Resources and Environment, Faculty of Science at Sriracha,
Kasetsart University,
Sriracha Campus, Chonburi 20230, Thailand
4Microbes for Agriculture Research Unit, Faculty of Liberal Arts and Science, Kasetsart University,
Kamphaeng Saen Campus, Nakhon Pathom 73140, Thailand
(*Corresponding author’s e-mail: [email protected])
Received: 31 October 2025, Revised: 1 December 2025, Accepted: 10 December 2025, Published: 15 February 2026
Abstract
Plants cultivated on calcareous soils frequently show symptoms of zinc deficiency, which can increase their susceptibility to fungal phytopathogen infections and adversely impact plant growth and productivity. One potential approach to mitigate these issues is the application of zinc-solubilizing microbes with antagonistic activity against fungal phytopathogens. Therefore, the objective of this study was to isolate and screen yeasts from rose flowers for their ability to solubilize zinc and suppress fungal phytopathogens. Ninety-two phylloplane yeasts were screened for zinc-solubilizing efficiency using modified Pikovskaya’s agar individually supplemented with 0.1% ZnO, ZnCO₃, and ZnS. Among these isolates, we selected 5 zinc-solubilizing yeasts that exhibited high zinc-solubilizing efficiency (ZSE), ranging from 3.73 to 5.38 and from 3.80 to 5.12 on media supplemented with 0.1% ZnO and ZnCO₃, respectively. These strains were identified as Kurtzmaniella quercitrusa and Hanseniaspora opuntiae based on the D1/D2 domain of LSU rDNA sequence analysis. Their growth, zinc solubilization at alkaline pH, acid production, and traits for promoting plant growth were investigated. Kurtzmaniella quercitrusa KPR1006 grew well in alkaline conditions and had the highest amount of soluble zinc at 73.64 mg/L at pH 7.0, followed by 53.04 mg/L at pH 8.0 and 29.75 mg/L at pH 9.0. The analysis of acids produced by K. quercitrusa KPR1006 indicated that citric acid, succinic acid, malic acid, and acetic acid were present in the culture filtrate. The cell-free supernatant from Hanseniaspora opuntiae KPR2060 significantly inhibited the hyphal growth of Sclerotium sp. and Phytophthora sp. M01 by 54.35 ± 1.54% and 81.65 ± 1.93%, respectively, and it also suppressed the germination of sclerotia and conidia. While K. quercitrusa KPR1006 inhibited the hyphal growth of Sclerotium sp. and Phytophthora sp. M01 by 29.88% and 76.94%, respectively. These phylloplane yeasts have demonstrated significant potential in simultaneously enhancing zinc solubilization and functioning as yeast-based bio-fungicides.
Keywords: Phyllosphere, Biocontrol, Zinc solubilization, Calcareous soil, Phytopathogenic fungi, Phylloplane yeast
Introduction
Zinc is an essential micronutrient for plant growth and development, playing critical roles in enzyme function, protein synthesis, and hormonal regulation [1,2]. Zn is an essential component of many biomolecules, including lipids, proteins, and cofactors of auxins. Zn plays an important role in the metabolism of carbohydrates, synthesis of proteins and chlorophyll, protection of membrane lipids from reactive oxygen species, and biosynthesis of plant growth hormones such as auxin [3]. However, in calcareous soils, zinc often exists in insoluble forms, limiting its availability to plants and resulting in widespread zinc deficiency. Zinc deficiency is a prevalent agricultural problem in Thailand, particularly in areas where calcareous soils are commonly used for growing essential crops such as corn (Zea mays L.) and mung bean (Vigna radiata L.) [4,5]. Young leaves with interveinal chlorosis, older leaves with spots, smaller leaves, and stunted development are signs of zinc deficiency in plants [6]. Therefore, zinc deficiency adversely affects crop productivity and nutritional quality, posing significant challenges for sustainable agriculture. The bioavailability of zinc in these soils is often limited due to its adsorption onto calcium carbonate particles [7]. Calcareous soils contain high levels of calcium carbonate (CaCO₃) and Ca(OH)₂, which carbonate and hydroxide ions can react with zinc, forming insoluble zinc compounds such as zinc carbonate (ZnCO₃) and zinc hydroxide (Zn(OH)₂) [4,5]. These forms are unavailable to plants. Moreover, zinc deficiency in crops can result from multiple contributing factors, such as inherently low soil zinc content, limited solubility of zinc compounds, and the transformation of zinc into insoluble forms that plants cannot uptake efficiently [8]. Thailand possesses approximately 800,000 rai of alkaline or calcareous soil, predominantly located in the upper central region, including the provinces of Lopburi, Suphan Buri, Saraburi, and Nakhon Sawan, where such soils are prevalent in limestone mountainous areas. These regions cultivate economic crops, including mung beans and cassava [9]. Zinc fertilizers such as zinc sulfate (ZnSO₄) have been applied into the soil, but zinc ions are quickly adsorbed onto soil particles or precipitated as insoluble compounds [10,11]. Additionally, reports suggest that chelated zinc fertilizers, such as Zn-EDTA, and foliar zinc application are more effective in addressing these issues [12,13]. These methods enhance the availability of zinc to plants, ensuring better absorption and utilization [13]. Although foliar application of zinc is more effective for providing immediate availability of zinc to plants, soil-applied zinc remains in use due to its residual benefits, practicality, and cost-effectiveness [10].
Zinc-deficient plants exhibit increased susceptibility to fungal phytopathogen infections due to compromised structural, biochemical, and immunological defense mechanisms [1]. Zinc plays a pivotal role in maintaining cell wall integrity [14], activating antioxidant enzymes such as superoxide dismutase [2], and regulating the expression of pathogenesis-related proteins such as phytoalexins [1] and defense hormones [15]. Zinc is important for auxin metabolism and other hormone-regulated pathways that influence growth and immune responses [16]. Deficiency in zinc leads to weakened cell walls [17], impaired oxidative stress response [18], and disrupted signaling pathways [19], collectively reducing the plant’s ability to mount effective defenses against fungal invasion. Fungal phytopathogens such as Sclerotium sp. and Phytophthora sp. are major threats to crop cultivation, including leguminous crops, causing diseases that result in substantial yield losses [20,21]. Phytophthora sojae, the causal agent of Phytophthora root and stem rot, can infect soybean (Glycine max L.) at any developmental stage, from seed germination to plant maturity [22]. While Sclerotium rolfsii commonly causes southern blight disease on beans [23]. Conventional control methods, including chemical fungicides, raise concerns about environmental safety, resistance development, and regulatory restrictions [24]. Application of fungicides seriously affects the microflora of different ecosystems, with destructive effects on beneficial microorganisms, including epiphytic and endophytic fungi and bacteria [25]. Thus, there is an increasing demand for sustainable and environmentally friendly solutions that can simultaneously improve plant nutrition and protect against phytopathogens [26].
Currently, the application of plant growth-promoting microorganisms (PGPMs) tends to focus on the use of strains that possess multiple beneficial traits [27]. These include direct growth promotion mechanisms, such as the production of indole-3-acetic acid (IAA) [27], as well as the ability to suppress plant pathogenic microorganisms and mitigate abiotic stress of plants [28]. This can involve either the use of a single microbial strain or a combination of multiple strains in consortia [29]. Zinc-solubilizing bacteria and actinomycetes demonstrated the ability to mitigate zinc deficiency and promote plant growth [30,31]. Several zinc-solubilizing bacteria, such as Pseudomonas protegens RY2 [32], Bacillus megaterium [33], and Bacillus altitudinis [34], have been recognized as plant growth-promoting bacteria due to the production of plant hormones and growth factors and making zinc available to plants, which is beneficial for plant growth [29]. Zinc-solubilizing bacteria have the ability to convert these insoluble zinc compounds into bioavailable forms through the secretion of organic acids and other chelating agents [35]. Furthermore, Enterobacter cloacae ZSB14, a zinc-solubilizing strain, possesses Zn-regulated transporters and iron (Fe)-regulated transporter-like protein (ZIP) genes, which are upregulated under zinc deficiency. These genes are important for the transport and accumulation of Zn in rice in iron-deficient conditions [36]. Strains of zinc-solubilizing Bacillus spp. can be inoculated into the soil alone or in combination with chemical fertilizers to increase the soil zinc availability for crop uptake [37-39]. It has also been suggested that seed priming or coating with zinc-soluble Bacillus is an alternative method for promoting plant development [33,34]. Zinc-solubilizing microorganisms might not only solubilize zinc, an essential nutrient for plants, but also have biocontrol capabilities against various fungal diseases [40]. This dual action can make them particularly valuable in integrated pest management strategies. Phylloplane yeasts, which reside on the aerial surfaces of plants, have emerged as promising candidates for dual-function agricultural applications. These microorganisms can exhibit both plant growth-promoting traits and antagonistic activity against plant pathogens [41]. Rose flowers produce nectar and other exudates that are rich in sugars, amino acids, and organic acids, creating a favorable microenvironment for diverse microorganisms, including yeasts [42,43]. Generally, yeast favors acidic pH for growth, and they can produce several organic acids, which would benefit metal solubilization. Nectar and pollen of rose flowers contain essential minerals such as potassium, calcium, magnesium, and trace elements such as zinc and phosphorus [44]. These nutrients support the growth of microbes that might have specialized roles, such as zinc solubilization. These microbes can help in nutrient acquisition, and in return, the plant provides a hospitable habitat. Zinc-solubilizing yeasts are among the microorganisms that may colonize rose flowers, particularly the nectar and pollen, where they play a functional role in micronutrient dynamics, pollen viability, flower development, and overall reproductive success [45,46].
In this study, we aimed to isolate bifunctional phylloplane yeasts from rose (Rosa spp.) flowers that possess the ability to solubilize insoluble zinc compounds and inhibit the growth of fungal plant pathogens. Growth and solubilization of insoluble zinc compounds under alkaline conditions and acid production were examined. Further, the potent yeast strains were tested for their ability to inhibit the growth of Sclerotium sp. and Phytophthora sp.
Materials and methods
Isolation and screening of zinc-solubilizing yeast strains
Roses were collected from Jira Rose farm, Chonburi province and a rose farm of the Department of Horticulture, Faculty of Kasetsart Kamphaeng Saen, Kasetsart University, Kamphaeng Saen campus, Nakhon Pathom province, Thailand, for the isolation and screening of Zn-solubilizing yeast (ZSY). The samples were collected from September 2022 to February 2023. Roses from various types were collected in sterile polythene bags, kept in a cooling box, and transported to the laboratory for further analysis within 12 h after collection. The rose samples were surface sterilized by using 0.9% sodium hypochlorite for 1 min, then washed with 70% ethyl alcohol for 1 min, and washed 3 times with sterile distilled water. The rose samples were put in an Erlenmeyer flask containing 50 mL of yeast extract peptone dextrose broth (YPD broth). Streptomycin at a concentration of 0.02% (w/v) was used to inhibit bacterial growth. The sample was shaken at 150 rpm at 25 ± 2 °C for 48 h. After that, the samples were cross-streaked on YPD agar plate and incubated at 25 ± 2 °C for 48 h. The growing colonies were inspected and purified using YPD agar streaking.
The ability to solubilize zinc was evaluated on modified Pikoskaya’s agar, and the pH was maintained at 6.8 [47]. Different sources of insoluble zinc salts, consisting of zinc oxide (ZnO), zinc carbonate (ZnCO3), and zinc sulfide (ZnS), were supplemented individually at a final concentration of 0.1% to the medium, and the resulting medium was sterilized at 121 °C for 15 min. All tested strains were point inoculated onto the medium, and the plates were incubated at 25 ± 2 °C for 10 days. Strains with a distinct zone around the colony were termed zinc-solubilizing. The halo zone was measured, and the zinc solubilization efficiency (ZSE) of the strains was calculated using the following equation [46]:
ZSE = HZ/C, where SE is the Zn solubilization efficiency, HZ is the diameter of the solubilization halo zone, and C is the diameter of the colony.
Quantitative analysis of soluble Zn
The quantitative measurement of soluble zinc was performed in a 250 mL conical flask containing 50 mL of Pikovskaya’s medium (pH 7.0) and 0.1% ZnO. Cell suspension of yeast with an OD600 of 1.0 was used as inoculum (10% inoculum). The suspension was incubated at 25 ± 2 °C for 10 days in static conditions. Uninoculated medium was used as a control. After 10 days of cultivation, cells were removed by centrifugation at 8,000 rpm for 10 min. The supernatant was collected and digested with HNO3 and HClO4 (1:4 v/v). To measure the quantity of soluble zinc, the digested sample was analyzed using an atomic absorption spectrophotometer (Agilent Technologies 200 Series AA, USA).
Determination of growth and soluble Zn in alkaline media
The 5 yeast isolates exhibiting the highest zinc oxide solubilization efficiency were selected for growth kinetics analysis. Each isolate was initially cultured on YPD agar at 25 ± 2 °C for 48 h. A single loopful of each isolate was then transferred into YPD broth and incubated at 25 ± 2 °C for an additional 48 h with shaking at 150 rpm. The resulting yeast suspensions were adjusted to an optical density (OD₆₀₀) of 0.5. Subsequently, 500 µL of yeast suspension was inoculated into 250 mL Erlenmeyer flasks containing 50 mL of fresh YPD broth with pH 7.0, 8.0, and 9.0. The cultures were incubated at 25 ± 2 °C with agitation at 150 rpm. Each isolate was tested in triplicate. Samples (1 mL) were collected at 3-hour intervals for 60 h. Cell growth was monitored by measuring optical density at 600 nm. A growth curve was generated based on the OD₆₀₀ values to evaluate growth rates over the incubation period.
Analysis of the acid production of zinc-solubilizing yeast
The yeast isolate KPR1006, demonstrating the highest efficiency in zinc oxide solubilization, was selected for analysis of types of acid production. The isolate was cultured in YPD broth and incubated at 25 ± 2 °C for 48 h with continuous shaking at 150 rpm. The resulting yeast suspensions were adjusted to an OD₆₀₀ value of 1.0. Subsequently, 5 mL of yeast suspension was inoculated into 250 mL Erlenmeyer flasks containing 50 mL of Pikovskaya’s broth supplemented with 0.1% ZnO. The pH of the medium was adjusted to 7.0, 8.0, and 9.0. Each pH condition was tested in triplicate. After incubation, the culture broths were prepared for high-performance liquid chromatography (HPLC) analysis. For sample preparation, the cultures were first filtered using a sterile syringe filter with a pore size of 0.45 µm. The filtrates were then diluted 5-fold using HPLC-grade water and subsequently filtered again through nylon membrane filters with a pore size of 0.22 µm. HPLC analysis was performed using a Rezex ROA-Organic Acid H⁺ (8%) column (300×7.8 mm2 i.d.) (Phenomenex Inc., USA). The mobile phase was 0.005 N sulfuric acid at a flow rate of 0.8 mL/min. The column temperature was maintained at 40 °C, and detection was conducted at a wavelength of 210 nm. Quantification of organic acids was performed by comparison with standards of 4 organic acids: Acetic acid, citric acid, malic acid, and succinic acid.
Screening of plant growth promotion traits
Indole-3-acetic acid (IAA) production was analyzed both with and without 1% L-tryptophan supplementation. Zn-solubilizing yeast was inoculated into YPD broth with and without 0.1% L-tryptophan, and the culture was grown for 5 days at 150 rpm and 28 ± 2 C. Cells were separated from the suspension by centrifugation at 10,000 rpm for 10 min. Then, Salkowski's reagent was added to the supernatant at a ratio of 1:1 (v/v), and the mixture was stored in the dark for 25 min. For quantitative analysis of IAA, the absorbance at 530 nm was measured using a UV-vis double beam spectrophotometer (GENESYS 10S UV-VIS, Thermo Scientific Inc., USA) and calibrated with a standard curve of pure IAA [28].
To measure the solubilization of inorganic phosphate, yeast strain cultures were inoculated on Pikovskaya’s agar supplemented with 0.1% of calcium phosphate and incubated at 25 ± 2 C for 10 days. Strains showing a halo zone around the colony were identified as P-solubilizing strains. Chrome azurol S (CAS) medium was used for siderophore production according to a method described previously [48]. In brief, zinc-solubilizing yeast was grown on YPD agar at 25 ± 2 C for 48 h and then point-inoculated onto CAS agar. The cultures were incubated at 25 ± 2 C for 5 days. A colony surrounded by light orange zones was considered to possess siderophore production ability [28].
The production of ammonia was evaluated by inoculating bacterial strains into 4% peptone broth and incubating them at 25 ± 2 °C for 5 days. One milliliter of Nessler's reagent was added after incubation. A positive result for NH3-producing strains was the development of a yellow to dark brown hue [46].
Dual culture assay
Yeast inoculum was prepared through cultivation on a YPD agar plate incubated at 25 ± 2 °C for 48 h. The inoculum was streaked orthogonally on a YPD agar and carrot agar, 3 cm away from the rim of the Petri dish (90 mm diameter). Fungal inoculum was prepared by cultivating the fungi on PDA and incubating the plates at 25 ± 2 °C for 3 days for Sclerotium sp. and 7 days for Phytophthora sp. Following a 5 mm2 mycelium disk of Sclerotium sp., or Phytophthora sp. M01 was placed 3 cm from the opposite side, and the plates were incubated at 25 ± 2 °C for 7 days. The plates only inoculated with the mold were used as controls. When the mold reached the dish border, the mycelial growth towards the yeast strip was measured using a ruler. The mycelial growth reduction was calculated as
Inhibition (%) = [(C – T)/C]×100
where C was the mycelial growth in the control (cm), and T was the mycelial growth of pathogenic fungi in the presence of yeast cultures (cm). The experiment was carried out in triplicate [49].
Evaluation of antifungal activity of yeast cell-free supernatant
Yeast isolates were cultured in YPD broth using a standard inoculum (1 loopful) and incubated at 25 ± 2 °C for 24 h with shaking at 150 rpm. Following incubation, 0.5 mL of the cultured yeast suspension was transferred into a 250 mL Erlenmeyer flask containing 50 mL of fresh YPD broth and incubated under the same conditions (25 ± 2 °C, 150 rpm) for 48 h. After cultivation, the cultures were centrifuged at 8,000 rpm for 10 min. The resulting supernatant was collected and filtered through a 0.45 µm pore-size membrane filter to obtain a sterile, cell-free supernatant. The antifungal assay was performed using Sclerotium sp. and Phytophthora sp. M01. For Sclerotium sp., potato dextrose agar (PDA) was used, while carrot agar was used for Phytophthora sp. The culture media were mixed with the yeast cell-free supernatant at a ratio of 10 mL of media to 10 mL of supernatant. The mixture was poured into sterile Petri dishes and allowed to solidify. Fungal inoculation was performed by cutting a 5 mm diameter mycelial disc from the actively growing edge of each fungal culture using a sterile cork borer. The disc was placed at the center of the prepared medium containing the yeast-derived supernatant. Plates were incubated at 25 ± 2 °C for 7 days. Culture media supplemented with 0.5% carbendazim and 0.5% metalaxyl were used as positive controls. Whereas unsupplemented culture media were used as a negative control. At the end of the incubation period, the diameter of fungal colonies was measured. The percentage of growth inhibition was calculated according to the method described by Dissanayake [50] using the formula:
Inhibition (%) = {(C − T)/C}×100
where C was the colony diameter in the control (cm),
and T was the colony diameter in the treatment (cm)
Evaluation of conidial and sclerotia germination inhibition by yeast cell-free supernatant
Conidial germination inhibition of Phytophthora sp. M01 was assessed following the method of Cabañas et al. [51], with some modifications. Briefly, 100 µL of a Phytophthora sp. suspension (10⁵ conidia/mL) was evenly spread onto carrot agar plates. After the surface dried, 5 µL of yeast cell-free supernatant (10⁵ CFU/mL) was applied as a drop onto the agar surface. Plates were incubated at 28 ± 2 °C for 7 days. Spore germination was evaluated microscopically and compared to control plates without supernatant. The experiment was conducted in triplicate. For sclerotia germination inhibition [52], 1 mL of yeast cell-free supernatant was added to 4 mL of YPD broth in sterile test tubes. Three sclerotia of Sclerotium sp. were introduced per tube. All treatments were performed in triplicate and incubated at 25 ± 2 °C for 7 days. YPD broth alone served as a negative control, while 0.5% carbendazim and 0.5% metalaxyl were included as positive controls. Germination and mycelial growth were monitored using a light microscope after incubation.
Production of extracellular hydrolytic enzymes
Cellulase activity was determined on solid medium containing sodium carboxymethylcellulose (5 g/L) and mineral salts, as mentioned by Kumar et al. [53]. Chitinase activity was evaluated on solid medium supplemented with colloidal chitin [54].
Biofilm formation by zinc-solubilizing yeast
Biofilm formation was evaluated following a previously described procedure [55]. with some modifications. Yeast strains were grown overnight at 25 ± 2 °C in YPD broth. Cells were then harvested by centrifugation at 8,000 rpm for 10 min, washed twice with 0.1 M phosphate buffer (pH 7.0), and resuspended in YPD broth to 105 CFU/mL. Aliquots (200 μL) were inoculated in triplicate into wells of a 96-well polystyrene plate and incubated at 28 ± 2 °C for 2 days. As a control, ten wells were handled identically, except that no yeast suspensions were added. After incubation, wells were washed twice with water, and the adherent biofilm layer was stained with an aqueous solution of 1% (w/v) crystal violet for 20 min, rinsed twice with water, and air-dried. The stained biofilm layer was eluted from each well with 200 μL of 95% ethanol, and 100 μL of the eluted solution was transferred to a new polystyrene 96-well plate. The amount of crystal violet in the solution was measured at 590 nm. The absorbance (A) values for the controls were subtracted from the values for the test well to minimize background interference. Biofilm formation was considered positive in a well where the mean A of the treatment was higher than the mean A of the negative control. All assays were carried out 3 times.
Identification of the selected strains
Morphological characteristics were examined for preliminary identification, followed by molecular identification of the selected yeast strains. A 1 loopful of yeast was cultured in YPD broth at 25 ± 2 °C for 16 h with shaking at 150 rpm. After incubation, 1,000 µL of the yeast cell suspension was transferred into a microcentrifuge tube and centrifuged at 10,000 rpm for 1 min to pellet the cells. Genomic DNA was extracted from the cell pellet using the GF-1 Plant DNA Extraction Kit (Vivantis, Malaysia) following the manufacturer’s protocol. The extracted DNA was transferred to a new microcentrifuge tube and stored at –20°C until further use. PCR amplification of the D1/D2 domain of the 26S rRNA gene was performed using the primers NL-1 (5´-GCATATCAATAAGCGGAGGAAAAG-3´) and NL-4 (5´-GGTCCGTGTTTCAAGACGG-3´), as described by Kurtzman and Robnett [56]. The PCR products were verified by agarose gel electrophoresis using a 1.5% agarose gel containing GelRed nucleic acid stain. The PCR products were subsequently purified using the GF-1 AmbiClean Kit (Vivantis, Malaysia) according to the manufacturer’s instructions. The nucleotide sequences of the D1/D2 region of the 26S rRNA gene were compared with reference yeast sequences available in the GenBank database (http://www.ncbi.nlm.nih.gov/blast) using the BLASTN (Basic Local Alignment Search Tool for nucleotides) homology search program [57]. Species identification was based on the criteria proposed by Kurtzman and Robnett [56]. Phylogenetic analysis was conducted by using BioEdit Sequence Alignment Editor version 7.2.5. A phylogenetic tree was then constructed using the Maximum Likelihood method implemented in MEGA software version 11.0.11 [58].
Statistical analysis
Statistical analysis was carried out with SPSS 16.0 (SPSS Inc., Illinois, USA). Data were examined using 1-way analysis of variance, followed by Duncan’s New Multiple Range Test, to determine significant differences between the treatments at p < 0.05.
Results and discussion
Zinc-solubilizing yeast
In the present study, zinc-solubilizing yeast were isolated from rose flowers growing in Chonburi and Nakhon Pathom Provinces, Thailand. The ability of all isolates to solubilize zinc was tested using agar media supplemented with 3 types of insoluble zinc compounds: ZnO, ZnCO₃, and ZnS. The formation of a clear halo zone surrounding the yeast colonies indicated zinc solubilization. All the selected strains can effectively solubilize insoluble Zn compounds, notably ZnO and ZnCO₃, under the conditions of the assay. None of the isolates showed any ZnS solubilization (data not shown). The halo zone diameter and zinc-solubilization efficiency (ZSE) for the selected zinc-solubilizing yeast are summarized in Table 1. Out of the 92 isolates, only 5 (which is 5.4%) showed a zinc-solubilization efficiency (ZSE) on both ZnO and ZnCO3 higher than 3.00, which are KPR1006, KPR2047, KPR2056, KPR2060, and KPR2062, so these were chosen for further study. The isolate KPR2047 exhibited the highest ZSE in ZnO-supplemented medium (5.38 ± 0.46). The isolate KPR2060 showed the highest ZSE in ZnCO₃-supplemented medium (5.12 ± 0.32). Flowers are typically habitats for yeasts because the nectar and pollen provide a nutrient-rich substrate that supports their growth and metabolism [59]. When these yeasts inhabit nectar or pollen, they may contribute to the local bioavailability of zinc, both for the plant and for other microorganisms or visiting pollinators that rely on zinc as a micronutrient. This relationship can be mutually beneficial for both yeast and the rose flower, the presence of zinc-solubilizing yeast may enhance zinc nutrition, which is important for pollen viability, flower development, and overall reproductive success [46]. In previous studies, zinc solubilization efficiency varied among microorganisms [30,46,60]. According to the zinc solubilization efficiency (ZSE) data, none of the isolates exhibited consistent solubilization efficiency for all 3 zinc compounds. Unlike earlier studies that found the best zinc solubilization efficiency for zinc oxide or zinc sulfate, this study shows that both ZnO and ZnCO3 also have solubilization efficiency. Saravanan reported that Bacillus sp. isolated from zinc ore sphalerite can solubilize ZnO, ZnCO₃, and Zn [61]. Pseudomonas oleovorans ZSB13 isolated from contaminated soil efficiently solubilize ZnO more than ZnCO3, ZnS and Zn(PO4)2 by organic acids production [62]. However, the finding that none of the yeast isolates in this investigation solubilized ZnS might be attributable to variations in the environments in which they were isolated. Zinc solubilizing efficiency on solid medium is sometimes different from in liquid medium [63]. Therefore, quantitative analysis of soluble zinc in Pikovskaya’s broth supplemented with 0.1% ZnO at pH 7.0 was performed. The yeast isolates solubilized insoluble ZnO in liquid medium, producing soluble zinc at concentrations ranging from 14.32 ± 6.76 to 72.77 ± 5.76 mg/L (Table 2). Although the isolate KPR1006 did not show high ZSE on solid medium, it showed the maximum solubilization of Zn (72.77 ± 5.7 mg/L) in liquid medium and the pH of the culture medium did not significantly alter. There are several strategies for dissolving zinc, including the excretion of metabolites such as organic acids, proton expulsion, and the production of chelating agents. Several microbes generate organic acids in the soil, which bind zinc cations and lower the pH of the surrounding soil. Furthermore, the anions can chelate zinc and improve zinc solubility. It has been reported that lactic, acetic, succinic, formic, isobutyric, and isovaleric acid generation by microbial isolates is a key solubilization process [64]. Several reports have demonstrated that the decrease in the pH of the culture medium was proportional to the quantity of soluble Zn produced by zinc-solubilizing microorganisms [53]. Furthermore, the formation of siderophores has been documented to facilitate metal solubilization, including Fe³⁺, and other studies have shown that phylloplane yeast can synthesize siderophores. All selected yeasts in this investigation are capable of producing siderophores (Table 5). Consequently, it may serve as a mechanism to facilitate zinc solubilization [28,76].
Table 1 Zinc solubilizing efficiency of ZSY isolates on solid medium using insoluble zinc compounds.
Isolates |
Zn solubilization |
|||||
ZnO |
ZnCO3 |
|||||
Colony diameter (cm) |
Zone of clearance (cm) |
ZSE |
Colony diameter (cm) |
Zone of clearance (cm) |
ZSE |
|
KPR1006 |
0.53 ± 0.01 |
2.28 ± 0.06 |
4.28 ± 0.06bc |
0.54 ± 0.02 |
2.04 ± 0.15 |
3.80 ± 0.16c |
KPR2047 |
0.16 ± 0.03 |
0.86 ± 0.09 |
5.38 ± 0.46a |
0.20 ± 0.03 |
0.87 ± 0.09 |
4.38 ± 0.16b |
KPR2056 |
0.22 ± 0.02 |
0.82 ± 0.05 |
3.73 ± 0.38c |
0.24 ± 0.04 |
0.94 ± 0.10 |
4.05 ± 0.32bc |
KPR2060 |
0.21 ± 0.02 |
0.95 ± 0.07 |
4.46 ± 0.36b |
0.16 ± 0.02 |
0.80 ± 0.07 |
5.12 ± 0.32a |
KPR2062 |
0.23 ± 0.02 |
0.99 ± 0.17 |
4.26 ± 0.43bc |
0.13 ± 0.03 |
0.66 ± 0.08 |
4.91 ± 0.27a |
*Data are statistically analyzed using 1-way ANOVA followed by Duncan’s New Multiple Range Test. Mean ± SD values with different lowercase superscripts are significantly (p < 0.05) different.
Table 2 Zinc solubilizing efficiency of different ZSY isolates in Pikovskaya’s broth supplemented with 0.1% ZnO.
Isolates |
Soluble zinc (mg/L) |
Final pH of culture broth |
KPR1006 |
72.77 ± 5.76a |
6.96 ± 0.07 |
KPR2047 |
14.32 ± 6.76c |
7.05 ± 0.05 |
KPR2060 |
16.31 ± 2.74c |
7.12 ± 0.03 |
KPR2062 |
18.46 ± 2.44c |
7.15 ± 0.03 |
*Data are statistically analyzed using one-way ANOVA followed by Duncan's New Multiple Range Test. Mean ± SD values with different lowercase superscripts are significantly (p < 0.05) different.
Effect of alkaline environment on growth and zinc-solubilizing efficiency of ZSY
Since zinc deficiency in plants is commonly found in calcareous or alkaline soils, it is essential to evaluate the survival and growth of zinc-solubilizing yeasts under alkaline conditions. Such an assessment is necessary to determine whether these yeasts can tolerate alkaline environments and maintain their zinc-solubilizing ability under such conditions. All yeast isolates exhibited an increased lag phase when cultured under alkaline conditions (Figure 1(A)). The results showed that the yeast isolate KPR1006 maintained moderate growth at pH 8.0 and 9.0, with only a slight reduction compared to its growth at pH 7.0, whereas the growth of the other 4 isolates was markedly suppressed under alkaline conditions. These findings suggest that KPR1006 possesses a degree of alkali tolerance that supports its survival and partial metabolic activity in such environments. When evaluating the amount of soluble zinc produced by the yeast isolate KPR1006 cultured in Pikovskaya’s broth supplemented with 0.1% ZnO at pH 8.0 and 9.0, it was found that the concentration of soluble zinc decreased compared to that at pH 7.0 (73.64 ± 4.31 mg/L). At pH 8.0 and 9.0, the concentrations of soluble zinc were 53.04 ± 1.93 mg/L and 29.75 ± 6.36 mg/L, respectively (Table 3). Under alkaline conditions, zinc solubilization efficiency decreased by 27.98% at pH 8.0 and 59.60% at pH 9.0 compared to pH 7.0, indicating that elevated pH levels impair the yeast’s ability to mobilize zinc effectively. Notably, the final pH of the culture broth, initially adjusted to pH 8.0 and 9.0, decreased obviously, suggesting substantial acid production by the yeast in response to alkaline stress. However, the concentration of soluble zinc was lower under these conditions. This suggests that the acids produced by the yeast were primarily utilized to neutralize the alkaline environment, thereby creating conditions more favorable for growth or survival, rather than directly contributing to zinc solubilization. Yeast possesses multiple mechanisms to respond to alkaline stress, including activating proton pumps such as the plasma membrane H⁺-ATPase (Pma1p) to expel excess OH⁻ ions and maintain optimal cytosolic pH [65]. In alkaline conditions, cells rapidly absorb calcium ions from the outside surrounding environment through the Mid1-Cch1 protein complex. This activates calcineurin, a calcium/calmodulin-dependent phosphatase. Calcineurin dephosphorylates the zinc-finger transcription factor Crz1, which allows it to enter the nucleus. Crz1 binds to particular DNA sequences called Calcineurin-Dependent Response Elements (CDREs) in the promoter regions of calcineurin-responsive genes, including ENA1, which codes for the Na⁺-ATPase enzyme [65]. The Rim101 pathway has also been associated with the way yeast reacts to alkaline environments. Rim101 serves as a repressor, controlling the degree to which NRG1 (Negative Regulator of Glucose-Repressed Genes) is expressed. Snf1 regulates Nrg1 further in the Snf1 signaling pathway. When the pH increases, Snf1 is activated via phosphorylation, which prevents Nrg1 from functioning. This inhibition reduces the repression on ENA1, which promotes the expression of the gene that codes for the Na⁺-ATPase enzyme, which is very important for keeping ions in balance when the pH becomes elevated [65,66]. Based on these results, the ability of yeast isolate KPR1006 to grow and solubilize zinc under alkaline conditions highlights its potential application in biofertilization strategies for calcareous soils, where zinc deficiency is prevalent.
Table 3 Zinc solubilizing efficiency of KPR1006 in alkaline conditions using Pikovskaya’s broth supplemented with 0.1% ZnO.
Initial pH of culture broth |
Soluble zinc (mg/L) |
Relative zinc solubilizing efficiency (%) |
Final pH of culture broth |
7.0 |
73.64 ± 4.31 |
100 |
6.91 ± 0.10 |
8.0 |
53.04 ± 1.93 |
72.98 |
7.19 ± 0.14 |
9.0 |
29.75 ± 6.36 |
40.40 |
7.28 ± 0.04 |
Analysis of organic acids production by ZSY under alkaline stress
The yeast isolate KPR1006 produced citric acid, acetic acid, succinic acid, and malic acid when cells were grown in Pikovskaya’s broth adjusted to pH 7.0, 8.0, and 9.0 with supplementation of 0.1% ZnO. An increase in the production of malic acid and acetic acid was observed at pH 8.0 and 9.0, while the levels of citric acid and succinic acid decreased under these conditions. These shifts are consistent with previous studies showing that yeasts adapt to alkaline stress by producing acids that help neutralize the external pH [67,68]. The increase in malic and acetic acid production is likely a physiological response aimed at maintaining intracellular pH homeostasis and enabling cell survival under unfavorable pH conditions. Acetic acid production also plays a role in redox balance and energy conservation through the pyruvate-acetyl-CoA pathway, which is often upregulated under stress conditions. In contrast, the reduced accumulation of citric and succinic acids may reflect partial downregulation of the tricarboxylic acid (TCA) cycle, as certain enzymes (e.g., citrate synthase, succinate dehydrogenase) are pH-sensitive and less active at higher pH. Additionally, the metabolic flux may be redirected away from these intermediates toward pathways that produce acids with stronger acidifying effects or that support more immediate survival needs [69]. Alkaline environments cause an elevated level of mitochondrial proteins involved in the tricarboxylic acid cycle (TCA cycle), according to proteomic investigations. Malate dehydrogenase levels notably rise, indicating an enhanced flux through the TCA cycle to support biosynthetic processes and energy demands under stress [70].
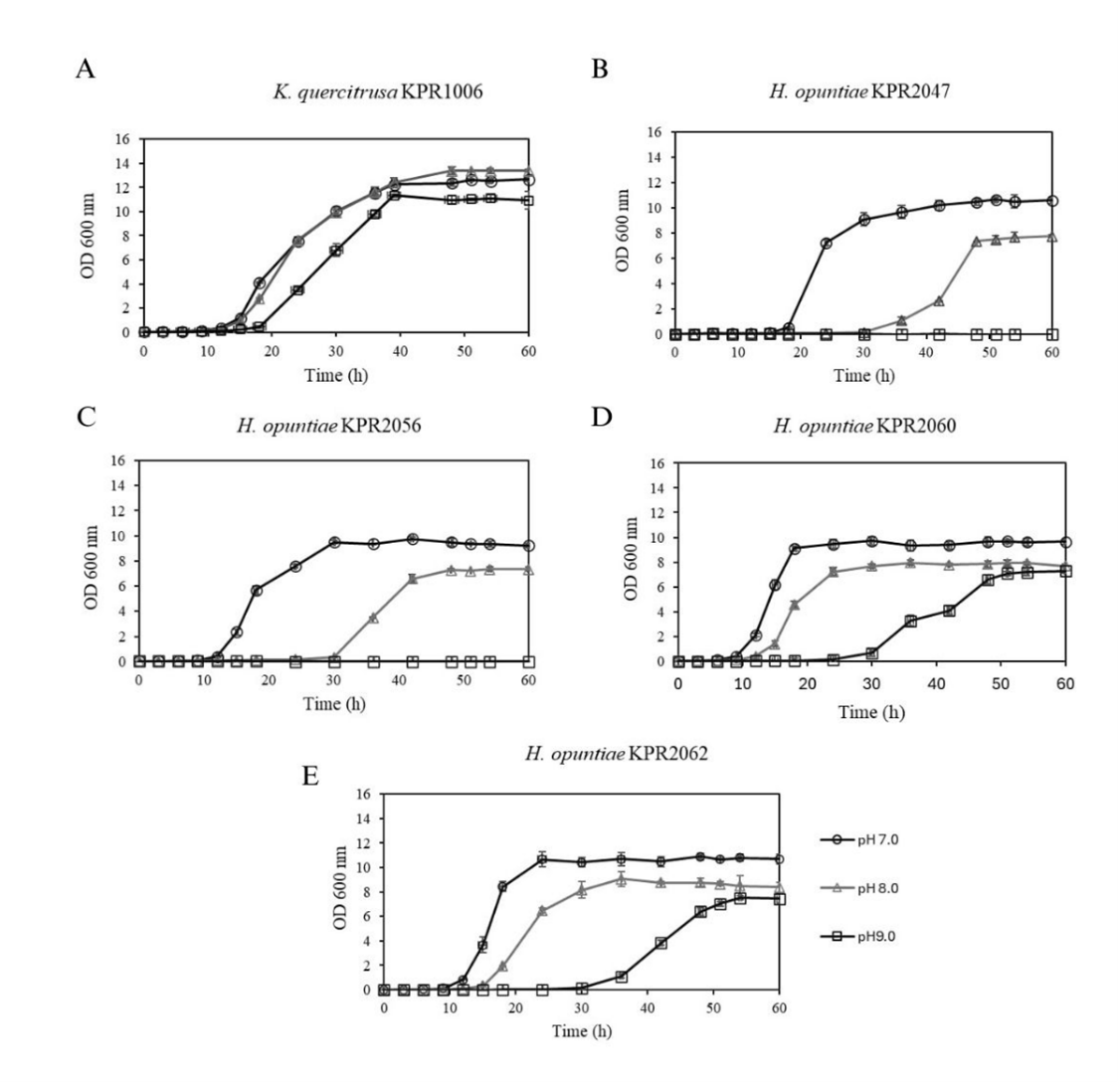
Figure 1 Effect of pH on the growth of zinc-solubilizing yeast.
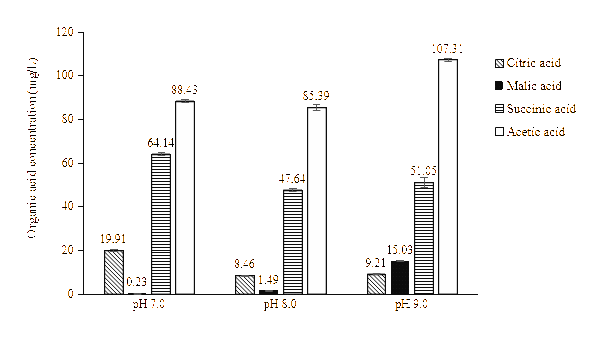
Figure 2 Acid profile of the yeast isolate KPR1006.
Biocontrol of soil fungal phytopathogens by zinc-solubilizing yeast
High zinc-solubilizing yeasts were evaluated for antifungal activity against Sclerotium sp. and Phytophthora sp. M01. Variability in the inhibition of Sclerotium sp. and Phytophthora sp. M01 was observed in the dual culture assay (Figure 3(A)). While the cell-free supernatant of ZSY remarkably inhibited both Sclerotium sp. and Phytophthora sp. M01. A significant suppression was detected with Sclerotium sp.; the cell-free supernatant of yeast isolates KPR2047, KPR2056, and KPR2060 inhibited fungal growth by 54-58%, surpassing the efficacy of 0.5% carbendazim and metalaxyl (Figure 3(B); Table 4). The cell-free supernatant from yeast isolates KPR1006, KPR2047, KPR2060, and KPR2062 inhibited the growth of Phytophthora sp. M01 by over 70%, as seen in Table 4. Inhibition of sclerotia germination was clearly observed by yeast cell-free culture broth of the isolate KPR2047 and KPR2056. The hyphal growth originating from the sclerotia, indicative of a filamentous form of Sclerotium sp., demonstrated a significantly lowered growth rate relative to the hyphae in the control and 0.5% metalaxyl treatments (Figure 4(A)). Inhibition of Phytophthora sp. M01 spore germination by yeast cell suspensions was observed as shown in Figure 4(B). A considerable variety of yeast species have been proven to be antagonistic agents against fungal plant diseases [55,71]. Certain yeast strains entirely inhibited the expansion of mycelial development. The limitation of mycelial growth and the establishment of the inhibitory zone in dual cultures may be ascribed to the release of diffusible antifungal compounds by the antagonistic yeasts [72]. The cell-free culture broth of ZSY showed significant antifungal activity in this investigation. Prior reports have documented the production of cell wall-degrading enzymes (chitinase, cellulase, and pectinase) by fungal biocontrol yeasts. Nonetheless, all ZSY examined in this investigation were unable to produce cellulase and chitinase (data not shown), indicating that these traits were strain-specific. Based on previously reported findings [73], it was noted that the most effective antagonistic strain against the examined fungi cannot produce glucanase, chitinase, or cellulase. Consequently, while the production of hydrolytic enzymes from fungal cell walls has demonstrated efficacy in reducing pathogen growth, our findings suggest it is an important yet non-essential aspect of yeast biocontrol activity.
In our research, the biocontrol effect appears to be facilitated by various yeast metabolic components or their combinations. The pH of the culture medium was approximately 5.0; thus, pH might be one factor that affects fungal growth and germination of spores. Consequently, the production of biofilms and the suppression of spore and sclerotia germination were investigated. Despite biofilm formation being one of the least understood mechanisms of antagonism, this ability has been associated with wound colonization in plants, leading to fast proliferation and coverage of the affected area [74]. Our results demonstrate that ZSY exhibited minimal biofilm formation, with the exception of KPR1006 (Figure 4(C)). A high level of biofilm production is not necessary for wound protection, and the most effective yeast strains against Botrytis cinerea were very low biofilm producers [55]. Concerning the capacity to suppress sclerotia germination, only KPR2047 and KPR2056 demonstrated significant inhibition, while other ZSY strains, such as KPR2060 and KPR2062, exhibited substantial inhibition of fungal growth but failed to inhibit sclerotia germination. This suggests that the inhibition of germination is not critical for yeast to regulate fungal hyphal growth, although it may contribute to a broader spectrum of antagonistic effects. Our results support the hypothesis that antifungal properties are not based on a single activity of yeasts, but instead involved in combination of nutrient competition, production of secondary metabolites, formation of biofilm or inhibition of spore germination [75]. These particular ZSY may serve as an effective biocontrol agent; nevertheless, additional mechanisms, secondary metabolites and killer toxins that can directly inhibit the growth or germination of fungal pathogens, require examination.
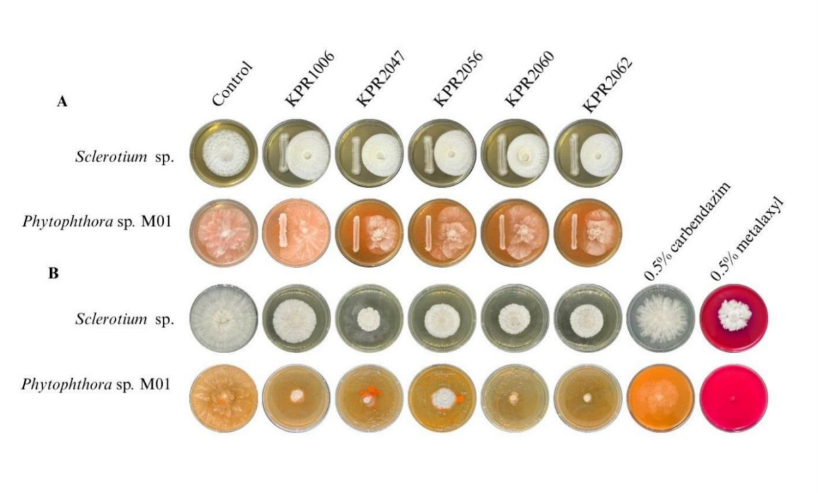
Figure 3 In vitro antagonistic activity of zinc-solubilizing yeasts against Sclerotium sp. and Phytophthora sp. M01; (A) Dual culture assay and (B) Poisoned food technique assay.
Table 4 Inhibition of mycelial growth of fungal phytopathogen by ZSY.
Isolates |
Dual culture inhibition (%) |
Mycelial growth inhibition by cell-free supernatant (%) |
||
Sclerotium sp. |
Phytophthora sp. M01 |
Sclerotium sp. |
Phytophthora sp. M01 |
|
KPR1006 |
1.41 ± 1.34 c |
35.04 ± 0.44 a |
29.88 ± 6.47 d |
76.94 ± 6.24 b |
KPR2047 |
15.51 ± 0.85 b |
33.66 ± 3.69 a |
58.82 ± 3.93 a |
72.39 ± 4.67 b |
KPR2056 |
37.56 ± 0.58 a |
29.87 ± 4.49 a |
54.51 ± 2.94 ab |
58.59 ± 4.90 c |
KPR2060 |
36.69 ± 0.20 a |
33.45 ± 1.73 a |
54.35 ± 1.54 ab |
81.65 ± 1.93 ab |
KPR2062 |
37.56 ± 0.52 a |
32.09 ± 3.39 a |
48.55 ± 2.58 b |
82.43 ± 0.75 ab |
0.5% Carbendazim |
NA |
NA |
25.41 ± 1.47 d |
35.69 ± 3.40 d |
0.5% Metalaxyl |
NA |
NA |
37.30 ± 2.03 c |
89.45 ± 0.47 a |
Different superscripts in a column differ significantly (p < 0.05) according to Duncan’s multiple range test.
NA = Not analysis
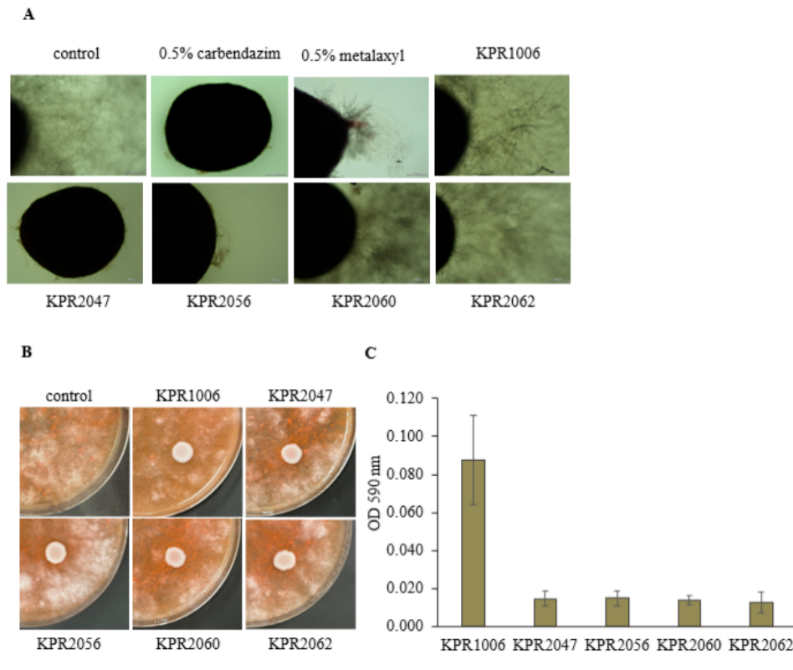
Figure 4 Characterization of the fungal antagonistic activity of zinc-solubilizing yeast; (A) Inhibition of sclerotia germination by yeast cell-free culture broth and (B) Inhibition of Phytophthora sp. M01 spore germination by yeast cell-free supernatant and (C) Biofilm formation capacity of yeast.
Characterization of plant growth promoting activity and identification of ZSY
These 5 isolates of yeast capable of zinc dissolution were examined for plant growth-promoting activities and molecular identification at the species level, as shown in Table 5. All of them produce indole-3-acetic acid (IAA) and siderophore. None of them can solubilize inorganic phosphate, and only the isolate KPR1006 can produce ammonia. Phylloplane yeasts have been reported to produce IAA and possess other plant growth-promoting traits [28,76]. The molecular identification results indicated that all 5 yeast isolates were classified under the phylum Ascomycota and the family Saccharomycetaceae. The isolate KPR1006 revealed over 99% nucleotide sequence similarity with Kurtzmaniella quercitrusa, while isolates KPR2047, KPR2056, KPR2060, and KPR2062 displayed over 99% similarity with Hanseniaspora opuntiae (Table 5). Based on molecular identification, all 5 isolates had nucleotide alterations of less than 1%; hence, they are not likely to represent new yeast species. A phylogenetic tree based on the evolutionary connections in the nucleotide sequences of these zinc solubilizing yeasts are presented in Figure 5. Our findings indicate that Kurtzmaniella quercitrusa KPR1006 showed outstanding growth and zinc solubilization in alkaline conditions, whereas Hanseniaspora opuntiae KPR2047, KPR2056, KPR2060, and KPR2062 displayed significant inhibitory effects against Sclerotium sp. and Phytophthora sp. M01. Therefore, the potential individual efficacy of the strain or co-inoculation of these strains needs to be investigated. However, the interactions across strains in the consortium need to be evaluated. The efficacy of these ZSY strains in mitigating zinc deficiencies in plants cultivated in calcareous soil must be assessed under field conditions.
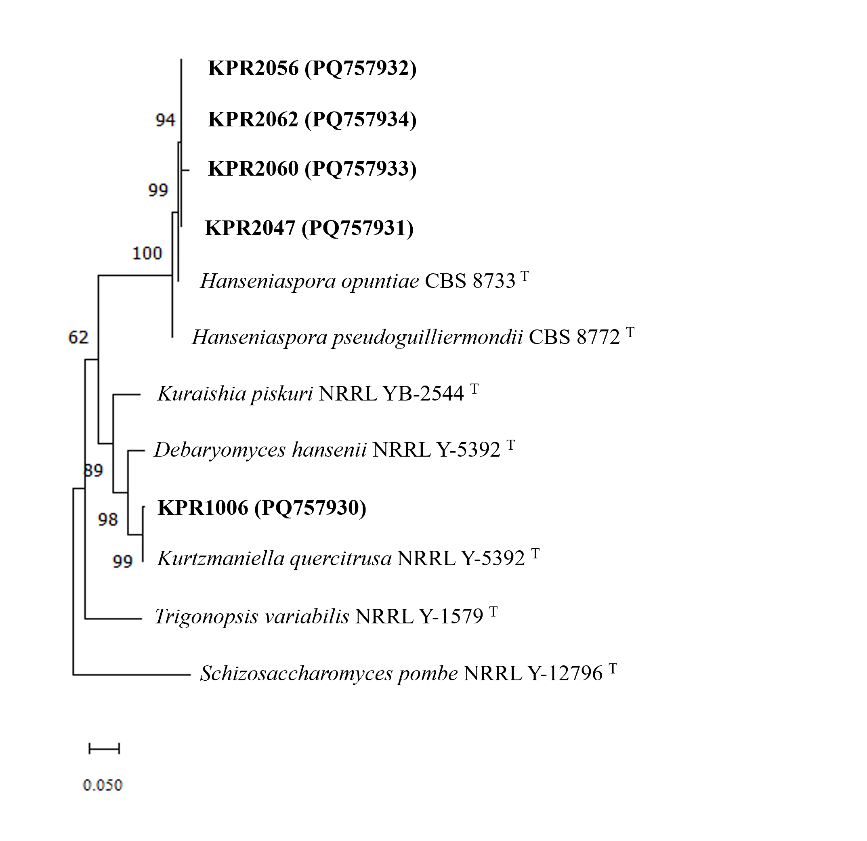
Figure 5 Phylogenetic tree of yeasts isolated from rose flowers and related species constructed by the maximum likelihood technique based on the D1/D2 domains of LSU rRNA gene.
Table 5 Plant growth promotion activities and identification of ZSY.
Isolate |
IAA production with L-Tryp (mg/g DW) |
Phosphate a solubilization (SE) |
Siderophore b production (mm)
|
NH3 c production |
Closest species |
% Similarity |
Result of identification (Accession no.) |
Roses |
KPR1006 |
1.60 ± 0.38 |
- |
0.6 ± 0.4 |
+ |
Kurtzmaniella quercitrusa (NRRL Y-5392T) |
99.81% |
Kurtzmaniella quercitrusa (PQ757930) |
Honey Dijon
|
KPR2047 |
12.94 ± 1.63 |
- |
2.8 ± 0.8 |
- |
Hanseniaspora opuntiae (CBS 8733T) |
99.63% |
Hanseniaspora opuntiae (PQ757931) |
Jessika |
KPR2056 |
12.59 ± 1.48 |
- |
2.6 ± 0.6 |
- |
Hanseniaspora opuntiae (CBS 8733T) |
99.45% |
Hanseniaspora opuntiae (PQ757932) |
Wedgewood |
KPR2060 |
12.20 ± 0.63 |
- |
1.9 ± 0.3 |
- |
Hanseniaspora opuntiae (CBS 8733T) |
99.73% |
Hanseniaspora opuntiae (PQ757933) |
Wedgewood |
KPR2062 |
12.90 ± 1.01 |
- |
3.1 ± 0.4 |
- |
Hanseniaspora opuntiae (CBS 8733T) |
99.73% |
Hanseniaspora opuntiae (PQ757934) |
Wedgewood |
Conclusions
In conclusion, zinc-solubilizing phylloplane yeasts were isolated from rose flowers. The investigation focused on the capacity of bifunctional ZSY to solubilize insoluble zinc in alkaline conditions and suppress Sclerotium sp. and Phytophthora sp. M01, significant fungal phytopathogens affecting mungbean and soybean plants. Their growth and capacity to solubilize ZnO in alkaline conditions were examined. The study revealed that Kurtzmaniella quercitrusa KPR1006 exhibited outstanding growth and zinc solubilization (53.04 ± 1.93 mg/L) under alkaline conditions. The zinc solubilizing efficiency remained stable at 72.98% at pH 8.0, with a noticeable decline in the pH value of the culture broth. The decrease in pH is attributable to the production of acids, including acetic, malic, succinic, and citric acid. These ZSY exhibited biocontrol activity against both fungal pathogens. The significant inhibitory effects against Sclerotium sp. and Phytophthora sp. M01 was found from dual culture assay and cell-free culture broth of Hanseniaspora opuntiae KPR2047, KPR2056, KPR2060, and KPR2062. These findings demonstrate the potential application of these yeasts as bioinoculants in mitigating zinc deficiency and providing yeast based biofingicide in plants, especially mungbean grown under alkaline soil conditions. Additional research will be conducted on the development of a consortium bioinoculant comprising these ZSY, assessing their capacity for zinc solubilization and their efficacy in controlling fungal diseases in mungbean cultivated in alkaline soil in field trials.
Acknowledgements
This research was supported from Microbiology Division, the Department of Science and Bioinnovation, Faculty of Liberal Arts and Science, Kasetsart University (Grant Year: 2025).
Declaration of generative AI in scientific writing
We would like to declare that generative AI (QuiltBot) was used solely for assistant in checking and refining in English language in this manuscript, including minor translations. The authors entirely generated the contents, ideas, and findings presented in the manuscript without AI assistance. After language editing, the authors reviewed and validated the final version to ensure its accuracy and integrity.
CRediT Author Statement
Passorn Jongkaewvijit: Data curation, Formal analysis, Investigation, Validation, and Writing –original draft. Noramon Panchanon: Data curation, Formal analysis, and Investigation. Nawarat Hongthong: Data curation, Formal analysis, Investigation, and Validation. Sopitar Soponputtaporn: Data curation and Visualization. Kriengsak Thaipong: Resources, Supervision, and Validation. Janpen Tangjitjaroenkun: Resources, Supervision and Validation. Moltira Srithaworn: Methodology, Supervision, and Validation. Orawan Chunhachart: Conceptualization, Funding acquisition, Methodology, Project administration, Supervision, Validation, and Writing –original draft.
References
C Cabot, S Martos, M Llugany, B Gallego, R Tolrà and C Poschenrieder. A role for zinc in plant defense against pathogens and herbivores. Frontiers in Plant Science 2019; 10, 1171.
M Umair Hassan, M Aamer, M Umer Chattha, T Haiying, B Shahzad, L Barbanti, M Nawaz, A Rasheed, A Afzal, Y Liu and H Guoqin. The critical role of zinc in plants facing the drought stress. Agriculture 2020; 10(9), 369-389.
T Tsonev and FJC Lidon. Zinc in plants - an overview. Emirates Journal of Food and Agriculture 2012; 24(4), 322-333.
N Takrattanasaran, J Chanchareonsook, S Thongpae and E Sarobol. Evaluation of mehlich 3 and ammonium bicarbonate-DTPA extractants for prediction of available zinc in calcareous soils in central Thailand. Kasetsart Journal 2010; 44, 824-829.
N Chittamart, J Inkam, D Ketrot and T Darunsontaya. Geochemical fractionation and adsorption characteristics of zinc in Thai major calcareous soils. Communication in Soil Science and Plant Analysis 2016; 47(20), 2348-2363.
EM Mattiello, HA Ruiz, JC Neves, MC Ventrella and WL Araujo. Zinc deficiency affects physiological and anatomical characteristics in maize leaves. Journal Plant Physiology 2015; 183, 138-143.
N Takrattanasaran, J Chanchareonsook, PG Johnson, S Thongpae and E Sarobol. Amelioration of zinc deficiency of corn in calcareous soils of Thailand: zinc sources and application methods. Journal of Plant Nutrition 2013; 36(8), 1275-1286.
A Sharma, B Patni, D Shankhdhar and SC Shankhdhar. Zinc - an indispensable micronutrient. Physiology Molecular Biological Plants 2013; 19(1), 11-20.
Land Development Department, Available at: https://www.ldd.go.th, accessed July 2025.
S Hussain, MA Maqsood and Rahmatullah. Increasing grain zinc and yield of wheat for the developing world: A review. Emirates Journal of Food and Agriculture 2010; 22(5), 326-339.
AQ Zhao, XH Tian, YL Chen and S Li. Application of ZnSO4 or Zn-EDTA fertilizer to a calcareous soil: Zn diffusion in soil and its uptake by wheat plants. Journal of Science Food and Agriculture 2015; 96(5), 1484-1491.
SS Dhaliwal, V Sharma, AK Shukla, J Kaur, V Verma, M Kaur, P Singh, A Gaber and A Hossain. Zinc-based mineral (ZnSO4·7H2O) and chelated (Zn-EDTA) fertilizers improve the productivity, quality and efficiency indices of field pea (Pisum sativum L.) through biofortification. Journal of Trace Elements and Minerals 2022; 2, 10033.
CL Doolette, TL Read, C Li, KG Scheckel, E Donner, PM Kopittke, JK Schjoerring and E Lombi. Foliar application of zinc sulphate and zinc EDTA to wheat leaves: Differences in mobility, distribution, and speciation. Journal of Experimental Botany 2018; 69(18), 4469-4481.
M Hamzah Saleem, K Usman, M Rizwan, H Al Jabri and M Alsafran. Functions and strategies for enhancing zinc availability in plants for sustainable agriculture. Frontiers of Plant Science 2022; 13, 1033092.
K Yuce and B Tileklioglu. Effect of plant hormones on the cambial activity of Cerasus vulgaris miller under stress conditions with Zn. Journal of Plant Protection Research 2019; 59(3), 304-323.
I Cakmak. Tansley review No. 111: Possible roles of zinc in protecting plant cells from damage by reactive oxygen species. The New Phytologist 2000; 146, 185-205.
N Natasha, M Shahid, I Bibi, J Iqbal, S Khalid, B Murtaza, HF Bakhat, ABU Farooq, M Amjad, HM Hammad, NK Niazi and M Arshad. Zinc in soil-plant-human system: A data-analysis review. Science of The Total Environment 2022; 808, 152024.
G Feigl, N Lehotai, A Molnár, A Ӧrdӧg, M Rodríguez-Ruiz, JM Palma, FJ Corpas, L Erdei and Z Kolbert. Zinc induces distinct changes in the metabolism of reactive oxygen and nitrogen species (ROS and RNS) in the roots of two Brassica species with different sensitivity to zinc stress. Annals of Botany 2015; 116(4), 613-625.
BG Forde and PJ Lea. Glutamate in plants: Metabolism, regulation, and signaling. Journal of Experimental Botany 2007; 58(9), 2339-2358.
GK Patra, GK Acharya, J Panigrahi, AK Mukherjee and GR Rout. The soil-borne fungal pathogen Athelia rolfsii: Past, present, and future concern in legumes. Folia Microbiologica 2023; 68(5), 677-690.
F Sun, S Sun, Y Yang, B Zhou, C Duan, W Shan and Z Zhu. A novel disease of mung bean, phytophthora stem rot caused by a new forma specialis of Phytophthora vignae. Plant Disease 2021; 105(8), 2160-2168.
AG McCoy, AM Byrne, JL Jacobs, G Anderson, JE Kurle, DEP Telenko and MI Chilvers. Oomicide treated soybean seeds reduce early season stand loss to Phytophthora sojae. Crop Protection 2022; 157, 105984.
P Paparu, A Acur, F Kato, C Acam, J Nakibuule, A Nkuboye, S Musoke and C Mukankusi. Morphological and pathogenic characterization of Sclerotium rolfsii, the causal agent of southern blight disease on common bean in Uganda. Plant Disease 2020; 104(8), 2130-2137.
MF Ahmad, FA Ahmad, AA Alsayegh, M Zeyaullah, AM AlShahrani, K Muzammil, AA Saati, S Wahab, EY Elbendary, N Kambal, MH Abdelrahman and S Hussain. Pesticides impact on human health and the environment with their mechanisms of action and possible countermeasures. Heliyon 2024; 10(7), e29128.
MC Fisher, NJ Hawkins, D Sanglard and SJ Gurr. Worldwide emergence of resistance to antifungal drugs challenges human health and food security. Science 2018; 360(6390), 739-742.
SF Syed Ab Rahman, E Singh, CMJ Pieterse and PM Schenk. Emerging microbial biocontrol strategies for plant pathogens. Plant Science 2018; 267, 102-111.
A Ansabayeva, M Makhambetov, NY Rebouh, M Abdelkader, HS Saudy, KM Hassan, MA Nasser, MAA Ali and M Ebrahim. Plant growth-promoting microbes for resilient farming systems: Mitigating environmental stressors and boosting crop productivity - a review. Horticulturae 2025; 11(3), 260-290.
S Soponputtaporn, M Srithaworn, Y Promnuan, P Srirat and O Chunhachart. Indole-3-acetic acid producing yeasts in the phyllosphere of legumes: Benefits for chili growth. Trends in Sciences 2024; 21(3), 7335-7360.
J Garcia and J Kao-Kniffin. Microbial group dynamics in plant rhizospheres and their implications on nutrient cycling. Frontiers in Microbiology 2018; 9, 1516-1523.
M Srithaworn, J Jaroenthanyakorn, J Tangjitjaroenkun, C Suriyachadkun and O Chunhachart. Zinc solubilizing bacteria and their potential as bioinoculant for growth promotion of green soybean (Glycine max L. Merr.). PeerJ 2023; 11, e15128.
VK Upadhayay, AV Singh, A Khan and A Sharma. Contemplating the role of zinc-solubilizing bacteria in crop biofortification: An approach for sustainable bioeconomy. Frontiers in Agronomy 2022; 4, 903321.
R Yasmin, S Hussain, MH Rasool, MH Siddique and S Muzammil. Isolation, characterization of Zn solubilizing bacterium (Pseudomonas protegens RY2) and its contribution in growth of chickpea (Cicer arietinum L.) as deciphered by improved growth parameters and Zn content. Dose Response 2021; 19(3), 15593258211036791.
K Bhatt and DK Maheshwari. Zinc solubilizing bacteria (Bacillus megaterium) with multifarious plant growth promoting activities alleviates growth in Capsicum annuum L. 3 Biotech 2020; 10(2), 36-46.
P Kushwaha, R Srivastava, K Pandiyan, A Singh, H Chakdar, PL Kashyap, AK Bhardwaj, K Murugan, N Karthikeyan, SY Bagul, AK Srivastava and AK Saxena. Enhancement in plant growth and zinc biofortification of chickpea (Cicer arietinum L.) by Bacillus altitudinis. Journal of Soil Science and Plant Nutrition 2021; 21(2), 922-935.
VK Upadhayay, S Gangola, G Taj, K Gaurav, A Rani, S Kumar, S Garg, G Gupta, H Ali, S Siddiqui, SAM Alamri, A Mittal, SA Alrumman and M Pandey. Zinc-solubilizing bacterial consortia: A promising approach for zinc biofortification of crops. Frontiers in Microbiology 2025; 16, 1575514.
S Krithika and D Balachandar. Expression of zinc transporter genes in rice as influenced by zinc-solubilizing Enterobacter cloacae strain ZSB14. Frontiers in Plant Science 2016; 7, 446.
M Ali, I Ahmed, H Tariq, S Abbas, MH Zia, A Mumtaz and M Sharif. Growth improvement of wheat (Triticum aestivum) and zinc biofortification using potent zinc-solubilizing bacteria. Frontiers in Plant Science 2023; 14, 1140454.
RC Yadav, SK Sharma, A Varma, UB Singh, A Kumar, I Bhupenchandra, JP Rai, PK Sharma and HV Singh. Zinc-solubilizing Bacillus spp. in conjunction with chemical fertilizers enhance growth, yield, nutrient content, and zinc biofortification in wheat crop. Frontiers in Microbiology 2023; 14, 1210938.
R Khande, SK Sharma, A Ramesh and MP Sharma. Zinc solubilizing Bacillus strains that modulate growth, yield and zinc biofortification of soybean and wheat. Rhizosphere 2017; 4, 126-138.
G Sethi, KK Behera, R Sayyed, V Adarsh, BS Sipra, L Singh, AA Alamro and M Behera. Enhancing soil health and crop productivity: the role of zinc-solubilizing bacteria in sustainable agriculture. Plant Growth Regulations 2025; 105, 601-617.
KA Nimsi, K Manjusha, K Kathiresan and H Arya. Plant growth-promoting yeasts (PGPY), the latest entrant for use in sustainable agriculture: A review. Journal of Applied Microbiology 2023; 134, 1-11.
M Rebolleda-Gómez, NJ Forrester, AL Russell, N Wei, AM Fetters, JD Stephens and TL Ashman. Gazing into the anthosphere: considering how microbes influence floral evolution. New Phytologist 2019; 224(3), 1012-1020.
S Baudino, P Sun, JC Caissard, B Nairaud, S Moja, JL Magnard, A Bony, F Jullien, RC Schuurink, P Vergne, A Dubois, O Raymond, M Bendahmane, L Hibrand-Saint Oyant, J Jeauffre, J Clotault, T Thouroude, F Foucher and B Blerot. Rose floral scent. Acta Horticulturae 2019; 1232, 69-80.
O Rop, J Mlcek, T Jurikova, J Neugebauerova and J Vabkova. Edible flowers-a new promising source of mineral elements in human nutrition. Molecules 2012; 17, 6672-6683.
T Hisatomi and K Toyomura. Isolation, identification, and characterization of wild budding yeasts from rose flowers in Fukuyama city, Hiroshima, Japan, and their application in bread and wine production. Mycoscience 2021; 62, 382-389.
SF Fu, PF Sun, HY Lu, JY Wei, HS Xiao, WT Fang, BY Cheng and JY Chou. Plant growth-promoting traits of yeasts isolated from the phyllosphere and rhizosphere of Drosera spatulata Lab. Fungal Biology 2016; 120, 433-448.
Tripti, V Kumar and Anshumali. Phosphate solubilizing activity of some bacterial strains isolated from chemical pesticide exposed agriculture soil. International Journal of Engineering Research and Development 2012; 3(9), 1-6.
B Schwyn and JB Neilands. Universal chemical assay for the detection and determination of siderophores. Analytical Biochemistry 1987; 160, 47-56.
S Öztekin and F Karbancioglu-Guler. Biological control of green mould on mandarin fruit through the combined use of antagonistic yeasts. Biological Control 2023; 180, 105186.
M Dissanayake. Inhibitory Effect of selected medicinal plant extracts on phytopathogenic fungus Fusarium oxysporum. Annual Research and Review in Biology 2014; 4(1), 133-142.
CM Cabañas, A Hernández, A Martínez, P Tejero, M Vázquez-Hernández, A Martín and S Ruiz-Moyano. Control of Penicillium glabrum by indigenous antagonistic yeast from vineyards. Foods 2020; 9(12), 1864.
DAS Gebily, GAM Ghanem, MM Ragab, AM Ali, NE-dK Soliman and TH Abd El-Moity. Characterization and potential antifungal activities of three Streptomyces spp. as biocontrol agents against Sclerotinia sclerotiorum (Lib.) de bary infecting green bean. Egyptian Journal of Biological Pest Control 2021; 31(1), 1-15.
CMS Kumar, S D’Silva, R Praveena, A Kaprakkaden, LR Athira Krishnan, M Balaji Rajkumar, V Srinivasan and R Dinesh. Zinc solubilization and organic acid production by the entomopathogenic fungus, Metarhizium pingshaense sheds light on its key ecological role in the environment. Science of The Total Environment 2024; 923, 171348.
T Agrawal and AS Kotasthane. Chitinolytic assay of indigenous Trichoderma isolates collected from different geographical locations of Chhattisgarh in Central India. SpringerPlus 2012; 1(a73), 1-10.
A Fernandez-San Millan, L Larraya, I Farran, M Ancin and J Veramendi. Successful biocontrol of major postharvest and soil-borne plant pathogenic fungi by antagonistic yeasts. Biological Control 2021; 160, 104683
CP Kurtzman and CJ Robnett. Identification and phylogeny of ascomycetous yeasts from analysis of nuclear large subunit (26S) ribosomal DNA partial sequences. Antonie van Leewenhoek 1998; 73, 331-371.
SF Altschul, TL Madden, AA Schäffer, J Zhang, Z Zhang, W Miller and DJ Lipman. Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Research 1997; 25(17), 3389-3402.
S Kumar, G Stecher, M Li, C Knyaz and K Tamura. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Molecular Biology and Evolution 2018; 35(6), 1547-1549.
CC Rering, AB Rudolph and JJ Beck. Pollen and yeast change nectar aroma and nutritional content alone and together, but honey bee foraging reflects only the avoidance of yeast. Environmental Microbiology 2021; 23(8), 4141-4150.
C Suriyachadkun, O Chunhachart, M Srithaworn, R Tangchitcharoenkhul and J Tangjitjareonkun. Zinc-solubilizing Streptomyces spp. as bioinoculants for promoting the growth of soybean (Glycine max (L.) Merrill). Journal of Microbiology and Biotechnology 2022; 32(11), 1435-1446.
VS Saravanan, SR Subramoniam and SA Raj. Assessing in vitro solubilization potential of different zinc solubilizing bacterial (ZSB) isolates. Brazilian Journal of Microbiology 2004; 35(1-2), 121-125.
HF Rehman, A Ashraf, S Muzammil, MH Siddique and T Ali. Assessment of zinc solubilization potential of zinc-resistant Pseudomonas oleovorans strain ZSB13 isolated from contaminated soil. Brazilain Journal of Biology 2021; 83, e240015.
S S Shaikh and M S Saraf. Optimization of growth conditions for zinc-solubilizing plant growth-associated bacteria and fungi. Journal of Advanced Research in Biotechnology 2017; 2(1), 1-9.
MZ Mumtaz, KM Barry, AL Baker, DS Nichols, M Ahmad, ZA Zahir and ML Britz. Production of lactic and acetic acids by Bacillus sp. ZM20 and Bacillus cereus following exposure to zinc oxide: A possible mechanism for Zn solubilization. Rhizosphere 2019; 12, 100170.
A Serra-Cardona, D Canadell and J Arino. Coordinate responses to alkaline pH stress in budding yeast. Microbial Cell 2015; 2 (6), 182-196.
J Arino. Integrative responses to high pH stress in S. cerevisiae. OMICS 2010; 14(5), 517-523
ZP Guo and L Olsson. Physiological responses to acid stress by Saccharomyces cerevisiae when applying high initial cell density. FEMS Yeast Research 2016; 16(7), 1-11.
K Zhang, W Wang and Q Yang. Transcriptome analysis reveals the regulation of Aureobasidium pullulans under different pH stress. International Journal of Molecular Sciences 2023; 24(22), 16103.
PA Lund, D De Biase, O Liran, O Scheler, NP Mira, Z Cetecioglu, EN Fernandez, S Bover-Cid, R Hall, M Sauer and C O’Byrne. Understanding how microorganisms respond to acid pH is central to their control and successful exploitation. Frontiers in Microbiology 2020; 11, 556140.
VY Sekova, LI Kovalyov, MA Kovalyova, NN Gessler, MA Danilova, EP Isakova and YI Deryabina. Proteomics readjustment of the Yarrowia lipolytica yeast in response to increased temperature and alkaline stress. Microorganisms 2021; 9(12), 2619.
X Zhang, Y Yao, S Dhanasekaran, J Li, GL Ngolong Ngea, X Gu, B Li, L Zhao and H Zhang. Controlling black spot of postharvest broccoli by Meyerozyma guilliermondii and its regulation on ROS metabolism of broccoli. Biological Control 2022; 170, 104938.
J Li, T Yang, F Yuan, X Lv and Y Zhou. Inhibitory effect and potential antagonistic mechanism of isolated epiphytic yeasts against Botrytis cinerea and Alternaria alternata in postharvest blueberry fruits. Foods 2024; 13(9), 1334.
A Fernandez-San Millan, I Farran, L Larraya, M Ancin, LM Arregui and J Veramendi. Plant growth-promoting traits of yeasts isolated from Spanish vineyards: benefits for seedling development. Microbiological Research 2020; 237, 126480.
G Zara, M Budroni, I Mannazzu, F Fancello and S Zara. Yeast biofilm in food realms: Occurrence and control. World Journal of Microbiology and Biotechnology 2020; 36(9), 134-144.
S Muccilli and C Restuccia. Bioprotective role of yeasts. Microorganisms 2015; 3(4), 588-611.
G Muthukrishanan, J Munisamy, SK Gopalasubramaniam, KS Subramanian, R Dharmaraj, DJ Nath, P Dutta and AK Devarajan. Impact of foliar application of phyllosphere yeast strains combined with soil fertilizer application on rice growth and yield. Environmental Microbiome 2024; 19(1), 102.