
Trends
Sci.
2026;
23(6):
12450
Effect of Sonneratia alba Fruit Extract on LOX-1 and Nrf2 Expression in the Aortic Arches of High Fat Diet Induced Rats
Huriatul
Masdar1,2,
![]() ,
Nur Indrawaty Lipoeto3,*,
,
Nur Indrawaty Lipoeto3,*,
![]() ,
Gusti Revilla4,
,
Gusti Revilla4,
![]() ,
,
Eka
Fithra Elfi5,6,
![]() and Eryati Darwin7,
and Eryati Darwin7,
![]()
1Biomedical Science Doctoral Program, Faculty of Medicine, University of Andalas, Padang 25171, Indonesia 2Departement of Histology, Faculty of Medicine, University of Riau, Pekanbaru 28133, Indonesia
3Departement of Nutrition, Faculty of Medicine, University of Andalas, Padang 25171, Indonesia
4Departement of Anatomy, Faculty of Medicine, University of Andalas, Padang 25163, Indonesia
5Departemen of Cardiology and Vascular Medicine, Faculty of Medicine, University of Andalas,
Padang 25171, Indonesia
6Departement of Cardiology and Vascular Medicine, M. Djamil General Hospital, Padang 25171, Indonesia
7Departement of Histology, Faculty of Medicine, University of Andalas, Padang 25163, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 1 November 2025, Revised: 8 December 2025, Accepted: 15 December 2025, Published: 15 February 2026
Abstract
Atherosclerosis, the primary cause of ischemic heart disease, is a major contributor to global mortality. This highlights the urgent need for therapeutic strategies that are not only beneficial and safe but also economically sustainable, such as those derived from natural bioactive compounds. This study investigated the anti-atherogenic effect of mangrove Sonneratia alba, a source of bioactive compounds with known antioxidant and anti-inflammatory properties. The objective was to determine the extract’s effect on the pro-atherogenic receptor LOX-1 and its subsequent effect on foam cell formation and the endogenous antioxidant axis Nrf2-MnSOD in the aortic arch of male Wistar rats exposed to a 10-week high-fat diet. A total of twenty-four rats were randomly divided into six groups; normal control, HFD control, atorvastatin control (10 mg/day) and HFD groups treated with S. alba extract at 200, 400 and 800 mg/kg/day. The expression of LOX-1, Nrf2, and MnSOD was assessed using immunohistochemistry, while foam cells were identified through H&E staining. The results showed S. alba fruit extract at the dose of 800 mg/kg/day significantly reduced circulating LDL-C levels. Within the aortic arch, the S. alba extract at the same dose significantly suppressed LOX-1 expression, significantly reduced approximately 82% of foam cell formation and significantly elevated Nrf2 expression. The expression of MnSOD was enhanced however this change did not reach statistical significance. These data collectively suggest that S. alba fruit extract ameliorates early atherogenic features, associated with the modulation of key regulatory proteins LOX-1 and Nrf2, and subsequent reduction in foam cell formation. Thus, the extract seems promising as a natural agent for vascular protection, but further functional investigations are needed to definitively confirm the underlying mechanism.
Keywords: Atherosclerosis, Antioxidant, Foam cells, Hyperlipidemia, LOX-1, Nrf2, MnSOD, Sonneratia alba
Introduction
Atherosclerosis, a chronic inflammatory disease of the arterial wall is the primary pathological basis for ischemic heart disease, that persists the leading cause of morbidity and mortality globally [1]. Despite the availability and efficacy of atherosclerosis therapies,
particularly those focused in lipid control, the global incidence of this disease continues to rise [2]. Furthermore, the mortality rate associated with this disease has been reported to increase from 5.367 million in 1990 to 8.992 million in 2021 [3]. The early stages of atherosclerosis are annotated by endothelial dysfunction, retention of low-density lipoprotein (LDL) in the sub endothelial space and its subsequent modification, particularly through oxidation. Oxidized low-density lipoprotein (oxLDL) serve as a critical driver of the disease’s progression [4]. It is recognized by scavenger receptors on macrophages, most notably the lectin-like oxidized low-density lipoprotein receptor-1 (LOX-1), that modulates the uncontrolled uptake of lipids and drives the transformation of macrophages into foam cells. The accumulation of foam cells in the arterial intima is a hallmark lesion of early atherogenesis [5]. Therefore, interrupting the oxLDL/LOX-1 pathway and mitigating foam cells growth represents one of major therapeutic strategies against atherosclerosis [6]. Despite numerous studies targeting LOX-1 have been conducted using either natural or synthetic compounds, research in this area remains limited and requires further investigation regarding its efficacy and safety [7].
Oxidative stress plays a pivotal role in this cascade, not only by generating oxLDL but also by overwhelming the intrinsic antioxidant defenses of the vascular wall [8]. The excessive production of reactive oxygen species (ROS) driven by enzymes like NADPH oxidase (NOX) and mitochondrial disfunction is what fundamentally causes the oxidation of LDL [9]. Macrophages via its scavenger receptors uptake the oxidized LDL, turning them into lipid laden foam cells that accumulate in arterial walls. This process triggers inflammation and endothelial disfunction, recruits more immune cells, induces vascular smooth muscle cells phenotype switching and migration which leads to plaque formation and vicious cycle of oxidative stress [10]. A key cellular defense mechanism against oxidative stress is the nuclear factor erythroid 2-related factor 2 (Nrf2) signaling pathway. Under normal condition, Nrf2 is sequestered in the cytoplasm; however, upon exposure to ROS, it is translocated into the nucleus. There, Nrf2 binds to the antioxidant response element (ARE) and induces the expression of numerous cytoprotective genes, including antioxidant enzymes such as manganese superoxide dismutase (MnSOD) [11]. Activating the Nrf2 pathway is hypothesized to attenuate the pro-atherogenic environment by neutralizing ROS and potentially inhibiting the formulation of oxLDL [12]. Thus, Nrf2 represents an effective therapeutic target for atherosclerosis, complementing the existing anti-cholesterol therapies [13].
Natural product, especially those derived from medicinal plants, serve as a rich source of bioactive compounds with potential anti-atherosclerotic effects, often related to their strong antioxidant and anti-inflammatory activities [14]. Sonneratia alba (L.f.) Sm., a widely distributed mangrove species, is traditionally used for its therapeutic benefits. Phytochemical analyses reveal that its fruits are particularly rich in polyphenols, flavonoids, triterpenoid and steroids that are well known for their potent radical-scavenging activities [15,16]. Other studies have identified several important phytoconstituents in S. alba, including oleanolic acid, ursolic acid, α- dan β- amyrin cinnamate, stigmasterol, β-stigmasterol, lupeol, and squalene [15,16]. Oleanolic acid is recognized significant capability to lower total cholesterol, triglyceride and LDL-C levels, and increase high-density lipoprotein cholesterol (HDL-C) levels, prevent aortic endothelial thickening and inhibit atherosclerotic plaque formation in high fat diet animal model [17,18]. Moreover, it inhibits type 1 collagen formation and enhance the expression of Nrf2 and heme-oxygenase 1 (HO-1) in LOX-1 knockout human umbilical vein endothelial cells (HUVEC) that is induced by oxLDL [19]. Lupeol, another identified compound, demonstrates antioxidant properties and significantly elevates nitric oxide (NO), superoxide dismutase (SOD), catalase and glutathione (GSH) in cardiac injury animal model [20], promotes macrophages polarization to an inflammatory phenotype, thereby preventing inflammatory signal transmission and diminishing intracellular lipid accumulation, subsequently prevents atheroma plaque formation [21]. Lupeol seems to protect against myocardial ischemia-reperfusion injury by regulating nuclear factor kappa B (NF-κB) and Nrf2 signaling pathways [22]. Stigmasterol significantly suppresses foam cell formation by activating AMP activated protein kinase (AMPK) pathway and inhibiting NFkB/NLRP3 signaling axis, thus inhibiting macrophage polarization into inflammatory cells and limiting the uptake of oxLDL [23]. Previous studies on the S. alba fruit extract have demonstrated its capacity to suppress LDL-C and triglyceride (TG) as well as to reduce the atherogenic index plasma in vivo [24]. Additionally, the S. alba fruit extract inhibits foam cells formation, prevents microvascular structural alterations toward atherosclerosis and diminishes tumor growth factor-β (TGF-β) expression in high fat diet animal model [24,25]. In silico analysis indicates that S. alba fruit possesses active metabolites that can interact with three ROS proteins through hydrogen bonding [27].
The existing literatures, however, remain limited to the phytochemical compounds and antioxidant capacity of S. alba fruit. In our knowledge, there is no research has thoroughly investigated the potential of S. alba fruit extract in preventing atherogenesis, particularly its effects on LOX-1 expression, as well as the cellular antioxidant response mediated by Nrf2 and its downstream antioxidant MnSOD. This research examined the effect of S. alba fruit extract on atherosclerosis progression in high-fat diet-induced rat model, focusing on the alterations in the aortic arch. Spastically we aimed to determine whether the extract could modulate the expression of the pro-atherogenic receptor LOX-1 and its downstream consequence, foam cell formation. Furthermore, we sought to elucidate the expression of the critical antioxidant transcription factor Nrf2 and one of its key target enzymes, MnSOD. We hypothesized that S. alba fruit extract could inhibit early atherogenesis effect by concurrently reducing LOX-1 expression and elevating the Nrf2 mediated antioxidant defense system.
Materials and methods
Study design and ethical approval
This study performed a post-test control only group design using experimental animals to evaluate the effect of S. alba fruit extract. The research strictly adhered to the ethical guidelines for the animal experimentation. Ethical approval for the study was obtained from Research Ethical Committee of the Faculty of Medicine, University of Andalas, Padang, Indonesia under approval number 231/UN.16.2/KEP-FK/2024.
Preparation of Sonneratia alba fruit extract
Unripe fruit of Sonneratia alba measuring 2 - 3 cm in diameters were collected from the coastal area of Sungai Apit Regency, Siak Sri Indrapura District, Riau Province, Indonesia. The species was authenticated by a botanist from Faculty of Mathematic and Natural Sciences, University of Riau, Pekanbaru, Indonesia with voucher specimen number 583/UN19.5.1.1.3-4.1/TU.00.01/2025. After removing its calyxes, the fruits were thoroughly washed, oven-dried at a degree of 40 - 45 °C, and ground into a coarse powder. From 4 kg fruit, approximately 670 g crude powder were obtained, corresponding to an estimated yield of 16.75%. The powdered material was subjected to maceration by 96% ethanol (1 gram per 10 mL solvent) for 72 h, with intermittent stirring. The solvent was then filtered and stored in a dark room at room temperature. The maceration was done twice. The filtrate was concentrated in vacuo using a rotary evaporator at 40 °C to yield the crude ethanol extract. The extract was stored in refrigerator until used. Qualitative phytochemical analysis of the S. alba fruit extract confirmed the presence of flavonoids, phenolics, saponins and steroids. The extract was further quantified for its secondary metabolites. The total phenolic content (TPC), determined using the Folin-Ciocalteu assay [16], was measured at 171.99 ± 1.53 mg GAE/g extract. The total flavonoid content (TFC), assessed by the aluminum chloride colorimetric method [16], was found to be 14.49 ± 0.31 mg QE/g extract. The antioxidant capacity of the extract was evaluated using the DPPH (2,2-diphenyl-1-picrylhydrazyl) radical inhibition assay, yielding an IC50 value of 61.42 ppm. This result indicate that the extract possesses strong antioxidant activity.
Experimental animal and atherogenesis induction
A total of 24 male Wistar rats (Rattus norvegicus), aged 8 - 10 weeks and weighing 170 - 190 g, were procured from the Faculty of Pharmacy, University of Andalas, Padang. The sample size was determined using the Resource Equation method. The animals were housed under standard laboratory condition with a 12-hours light/dark cycle, 22 ± 2 °C and ad libitum access to water.
Atherosclerosis was induced by feeding rats a high fat diet (HFD) of 20 g per day. This HFD protocol was adapted from Ismawati et al. [28] with two primary modifications; the high-dose administration of vitamin D3 (700.000 IU/kg) was omitted to avoid the masking effect of high-dose vitamin D3 effect on protein targets expression, and the diet duration was extended to 10 weeks, following the established publication to induce hyperlipidemia and early atherogenesis with high fat diet in rats [29,30]. This modified protocol was established through a preliminary study to optimize and confirm the diet duration and effectiveness in reliably inducing early atherogenesis in rats, characterized by significant LDL-C elevation and observable foam cell accumulation in aortic arch of rat model (data not shown). The HFD formulation consisted of standard chow Vivo512 (Charoen Pokphand Indonesia Tbk, Medan, Indonesia) supplemented by 2% cholesterol-derived from egg yolk, 5% goat fat, and 0.2% cholic acid (Sigma Aldrich, St. Louis, USA). The feeding and treatment protocols were conducted for a total duration of 10 weeks.
Rats were randomly divided into six group (n = 4 per group); normal control (NC) fed standard chow Vivo512; high-fat diet control (HFD-C) fed HFD; high-fat diet + atorvastatin (HFD-A) fed HFD and administrated atorvastatin (KalbeMED, Jakarta, Indonesia) at a converted dose of 10 mg/day by oral gavage; high-fat diet + extract 200 (HFD-E200) fed HFD and administrated S. alba fruit extract at a dose of 200 mg/kg body weight/day by oral gavage; high-fat diet + extract 400 (HFD-E400) fed HFD and administrated S. alba fruit extract at a dose of 400 mg/kg body weight/day by oral gavage; and high-fat diet + extract 800 (HFD-E800) fed HFD and administrated S. alba fruit extract at a dose of 800 mg/kg body weight/day by oral gavage.
At the end of the 10-week treatment, the animals were fasted overnight and then deeply anesthetized using an intra peritoneal injection of ketamine (70 mg/kg) and xylazine (0.7 mg/kg). Blood samples were collected via cardiac puncture for LDL-C and HDL-C plasma levels analysis. Following perfusion with normal saline to remove residual blood, the aortic arch was carefully excised from each rat and immediately fixed in 10% neutral buffered formalin for subsequent histological and immunohistochemical analysis.
Histological and immunohistochemical analysis
Fixed aortic arch tissues were routinely processed and embedded in paraffin blocks. Section of 3 μm thickness were cut and stained with hematoxylin and eosin (H&E) to appraise general morphology and detect foam cells. The foam cells were identified in the intimal and medial layer of aortic arch by using a light microscope (Leica IC550HD, Leica Mycrosystem, Danaher Life Sciences, Germany). Photomicrographs were captured in all microscopic fields of 400x magnification by using Leica Application Suite Las Ez v3.4.0 (build:272) (Leica Mycrosystem, Switzerland) and the foam cells were quantified by using ImageJ v1.54g software (Wayne Rasband, National Institutes of Health, USA).
Immunohistochemistry was performed on 3 μm paraffin-embedded aortic arch section to assess the expression indexes of LOX-1, Nrf2 and MnSOD. Briefly, the sections were deparaffinized, rehydrated and subjected to antigen retrieval using a citrate buffer. Endogenous peroxidase activity was quenched with 3% H2O2. The sections were then incubated for one hour at room temperature with primary antibodies anti-LOX-1 1:50 (Cat PA5-102452, Thermo Fisher Scientific, USA), anti-Nrf2 1:50 (SAB4501984, Sigma-Aldrich, Darmstadt, Germany) and anti-MnSOD 1:300 (BT Lab, Zhijiang, China). Following washing, an appropriate biotinylated secondary antibody and a streptavidin-HRP complex were applied. Immunoreactivity was visualized using 3,3’-diaminobenzidine (DAB) substrate, resulting in brown precipitates at the site of antigen binding. The sections were then counterstained with Mayer’s hematoxylin.
The expression indexes of LOX-1, Nrf2, and MnSOD were quantified using Colour Deconvolution v2 plugin integrated into ImageJ v1.54g software, across eight random high-power field photomicrographs (400x magnification). The expression index was calculated as Σ (intensity x percentage of positive cells). The intensity was obtained from mean grey value and the percentage of positive cells were obtained from percentage area measured in ImageJ.
Statistical analysis
All quantitative data were expressed as the mean and Standard Error of Mean (SEM). The normality of the data distribution was assessed using the Saphiro-Wilk test. Differences among the six groups were analyzed using One-Way Analysis of Variance (ANOVA) or Kruskal Wallis-test, followed by Tukey’s Post Hoc test or Dunn’s test for multiple pairwise comparisons. A p - value of less than 0.05 was considered statistically significant. All statistical analysis was performed using Graphpad Prism v9.0.0 software (Graphpad Software, Boston, MA, USA).
Results and discussion
Effect of Sonneratia alba fruit extract on plasma lipid profile
The impact of the high-fat diet and the subsequent treatment with S. alba fruit extract on plasma low-density lipoprotein cholesterol (LDL-C) and high-density lipoprotein cholesterol (HDL-C) levels is presented in Table 1. The HFD regimen effectively induced hypercholesterolemia as demonstrated by a highly significant increase in plasma LDL-C level relative to the normal control (NC) group (~3.3 fold increase, p < 0.05), confirming the successful induction of hyperlipidemia and the pro-atherogenic state. Treatment with a high dose of S. alba fruit extract (HFD-E800) significantly showed a potent hypolipidemic effect, achieving a statistically significant reduction in LDL-C that nearly normalizing the levels to those of the NC group. Moreover, the efficacy of the 800 mg/kg/day extract dose was comparable to the positive control, Atorvastatin (HFD-A). This indicates that the extract exerts a significant potential to modulate this key atherogenic risk factor. Conversely, the lower dosages of S. alba (HFD-E200 and HFD-E400) exhibited a modest, dose-dependent tendency towards reduction, while the difference was not statistically significant compared to HFD-C.
In contrast to LDL-C, the concentrations of HDL-C plasma levels showed no statistically significant difference across all six experimental groups. The HFD-C group had a minimal, non -significant reduction compared to the NC group. Treatment with S. alba extract, even at the highest dosage, did not significantly restore or modulate this cardio-protective lipid fraction. This finding indicates that the primary hypolipidemic effect of the S. alba fruit extract is mediated through the diminishing of atherogenic lipoproteins (LDL-C) rather than by the augmenting of cardio-protective lipoproteins (HDL-C).
Table 1 Plasma LDL-C and HDL-C levels across experimental groups (means ± SEM).
Group |
LDL-C (mg/dL) ± SEM |
HDL-C (mg/dL) ± SEM |
Normal control (NC) |
46.63 ± 5.41### |
51.30 ± 3.02 |
HFD control (HFD-C) |
155.40 ± 14.61 |
46.90 ± 4.95 |
HFD + atorvastatin (HFD-A) |
81.00 ± 16.35# |
45.35 ± 1.75 |
HFD + extract 200 mg (HFD-E200) |
101.20 ± 7.39 |
47.00 ± 3.62 |
HFD + extract 400 mg (HFD-E400) |
94.68 ± 23.31 |
46.50 ± 4.18 |
HFD + extract 800 mg (HFD-E800) |
59.38 ± 4.67## |
49.15 ± 1.30 |
p - value |
0.0005* |
0.7509 |
*indicates a significant different across the experimental groups (p < 0.05). #, ## and ### indicate a significant different compared to HFD-C group with p < 0.05, p < 0.01 and p < 0.001, respectively.
The significant decrease in LDL-C plasma levels, particularly by the 800 mg/kg/day dose of S. alba fruit extract is a crucial finding as hypercholesterolemia is the fundamental risk factor for atherogenesis initiation. This preliminary observation highlights the extract’s potential as a natural agent for controlling hyperlipidemia. The mechanism responsible for this hypolipidemic effect remains hypothetical, as specific biochemical assays such as enzyme activity or receptor quantification were beyond the scope of this study. The hypolipidemic effect may be associated with the phytoconstituents such as polyphenol, flavonoid, sterols and triterpenoids previously identified in Sonneratia species [16,17]. From mechanistic point of view, phytocompounds may interfere with the cholesterol absorption in the intestine, possible by binding to bile acids and thus increase fecal excretion, modulate key enzymes in hepatic lipogenesis, such as 3-hydroxy-3-methyl-glutaryl-coenzyme A (HMG-CoA) reductase that is functioning similarly to statins like Atorvastatin. Subsequently, the extract may facilitate to enhance circulating lipids catabolism by modulating hepatic LDL receptor (LDL-R) expression, therefore augmenting the clearance of LDL-C from the plasma or boosting cholesterol-7α-hydroxylase (CYP7A1) enzyme involved in converting cholesterol into bile acids [31-33]. Polyphenols reduce LDL-C level by enhancing the activity of liver X receptor α (LXRα), suppressing hepatocyte nuclear factor 1 α (HNF-1α), consequently diminishing the mRNA expression of proprotein convertase subtilisin/kexin type 9 (PCSK9). This mechanism ultimately results in increased LDLR expression and enhanced LDL-C uptake [34]. These pathways suggest a direct effect on the clearance of LDL-C without necessarily affecting the synthesis or reverse cholesterol transport function of HDL-C as observed in a hypercholesterolemic male rabbits fed 0.2% curcumin [35].
The lack of significant effect of HDL might be explained by the high complexity of HDL metabolism. However, this result is consistent with the existing hyperlipidemia therapies, in which high-efficacy therapeutic classes such as statin and cholesterol absorption inhibitors provide major clinical benefits despite minimal HDL-C augmentation [36]. The metabolism of HDL-C involves various regulatory steps, including the function of cholesteryl ester transfer protein (CETP), lecithin-cholesterol acyltransferase (LCAT) and the expression of ATP-binding cassette transporter A1 (ABCA1) for cholesterol binding. The phytocompounds do not seem to strongly interfere with this specific enzymes or transporters that control HDL [37]. Elevated plasma HDL concentration is not a reliable surrogate for low hyperlipidemia risk, instead, it may reflect increased HDL turnover or functional demand in response to high circulating LDL-C burden. Consequently, determining atherosclerosis risk merely by quantifying HDL-C concentration is insufficient [38]. Further investigation is warranted to assess HDL functional capacity, such as its cholesterol efflux capability. It will determine whether the extract preserves the qualitative protective properties of HDL even without altering its plasma concentration.
The significant decrease in LDL-C levels necessitates an examination of the downstream events occurring within the arterial wall. The essential process in atherogenesis is the transformation of LDL into oxidized LDL which serve as the primary ligand for LOX-1 receptor and the key trigger for the inflammatory cascade [39]. Therefore, we hypothesized that the extract efficacy in reducing LDL-C would result in a tangible reduction in vascular pathology. The expression of pro-atherogenic receptor LOX-1 and its downstream consequent foam cell formation are discussed in the following section.
LOX-1 expression and foam cell formation in aortic arch
Lectin-like oxidized low-density lipoprotein receptor 1 (LOX-1) is a key molecular driver of early atherogenesis, and its expression in the aortic arch of rats was analyzed using immunohistochemical staining (Figure 1). In the normal control (NC) group, minimal LOX-1-expression was detected. In contrast, the high-fat diet control (HFD-C) group exhibited a pronounced and diffuse upregulation of LOX-1 expression, predominantly localized in the endothelial cells lining the intima and subendothelial cells likely infiltrating macrophages and/or transdifferentiated vascular smooth muscles (VSMCs). This confirmed the pathological activation of the pro-atherogenic state induced by HFD. Quantitative analysis (Table 2) confirmed that the HFD-C group displayed a more than threefold increase in LOX-1 expression compared to NC group.
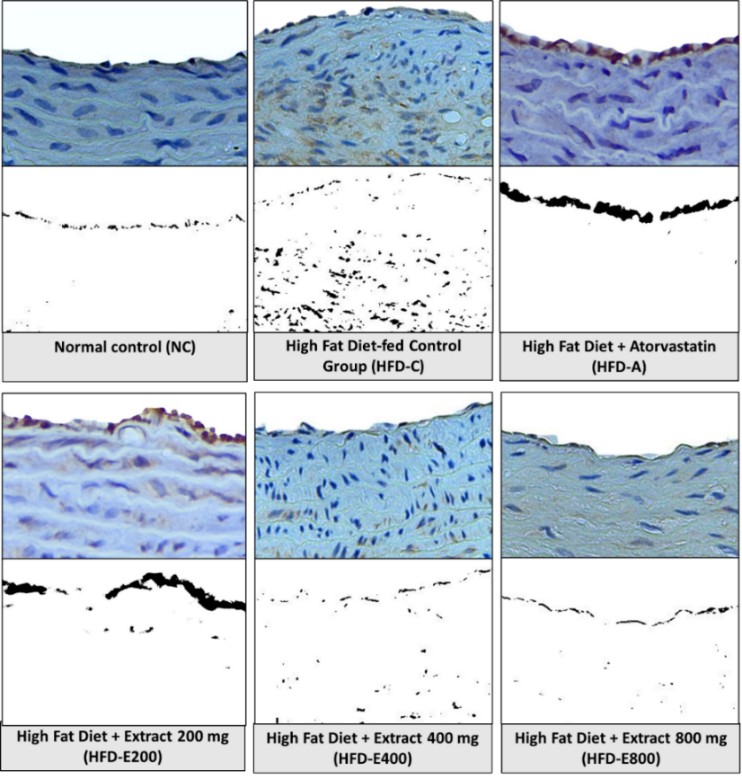
Figure 1 Immunohistochemical analysis of LOX-1 expression in the aortic arches of experimental groups. Representative images of immunohistochemical staining for LOX-1 (brown chromogen) in the intimal and medial layers of the rat aortic arch (top row of each panel) and the corresponding processed images highlighting positive staining using Colour Deconvolution 2 plugin integrated in ImageJ software (bottom row of each panel) are revealed at 400x magnification.
Treatment with Sonneratia alba fruit extract demonstrated a clear, dose-dependent decreasing in LOX-1 expression, with significant difference observed across the experimental group (Table 2). The HFD-E400 group showed a significant reduction of LOX-1 expression compared to the HFD-C group. The HFD-E800 group achieved the most pronounced attenuation, approximately 63% reduction compared to HFD-C group, resulting in a statistically significant reduction that returned LOX-1 expression to levels closely resembling those of the NC group. This indicates a powerful suppression effect of S. alba extract on the expression of LOX-1 receptor that is responsible for oxLDL uptake. Although the positive control (HFD-A) also showed a reduction in LOX-1 expression, however, its effect was not statistically significant compared to HFD-C group (Table 2).
Table 2 Expression index of LOX-1 and foam cell number in aortic arch of experimental groups.
Group |
LOX-1 expression index ± SEM |
Foam cell number ± SEM |
Normal control (NC) |
51.08 ± 2.32 |
275.5 ± 123.9 |
HFD control (HFD-C) |
160.30 ± 19.03 |
932.5 ± 238.5 |
HFD + atorvastatin (HFD-A) |
105.20 ± 21.08 |
481 ± 188.9 |
HFD + extract 200 mg (HFD-E200) |
94.15 ± 11.14 |
433 ± 78.52 |
HFD + extract 400 mg (HFD-E400) |
83.43 ± 20.95# |
358 ± 162.4 |
HFD + extract 800 mg (HFD-E800) |
58.96 ± 15.80## |
168 ± 42.44# |
p - value |
0.0051* |
0.0398* |
*indicates a significant different across the experimental groups (p < 0.05). # and ## indicate a significant different compared to HFD-C group with p < 0.05 and p < 0.01, respectively.
The histological quantification of lipid-laden cells or foam cells in the aortic arch supported the molecular findings regarding reduction of LOX-1 expression (Table 2). The HFD-C group demonstrated the highest accumulation of foam cells, serving as a definitive morphological indicator of early atherogenic lesions in the model. Histological examination revealed these cells distributed across both the intimal and medial layers of the aorta in HFD groups (Figure 2), suggesting participation from both infiltrating macrophages and trans-differentiated vascular smooth muscle cells (VSMCs) in the complex lesion morphology.
The administration of S. alba fruit extract resulted in a significant and dose-dependent reduction of foam cells formation (p = 0.0398). The highest dosage group, HFD-E800, exhibited the most significant effect on reducing foam cells accumulation. This reduction was statistically significant compared to the HFD-C group (p = 0.0256), demonstrating a remarkable anti-atherogenic influence. The HFD-E400 group also exhibited a prominent reduction, outperforming the atorvastatin group, however, this reduction was not statistically significant compared to HFD-C. Moreover, the expression of LOX-1 significantly correlated with accumulation of foam cells (p = 0.0005, R2 = 0.4462), suggesting this direct morphological evidence confirms that the extract interferes the pathological uptake and retention of lipids by vascular cells.
The quantity of foam cells observed in this study has a high variability in almost all groups. This observed variability may be a reflection of biological heterogeneity to this specific in vivo model of early atherogenesis, which is commonly reported in diet-induced rodent models. This variation reflects the heterogenous metabolic and inflammatory responses of individual rats to the chronic HFD-induced stress and hyperlipidemia [38]. Moreover, foam cells are the manifestation of highly localized and scattered early lesions or fatty streaks [40]. By analyzing a limited number of tissue sections, in this case aortic arch, inherent non-uniformity in lesion distribution throughout the entire aortic tress in evitable contributes to a larger standard deviation.
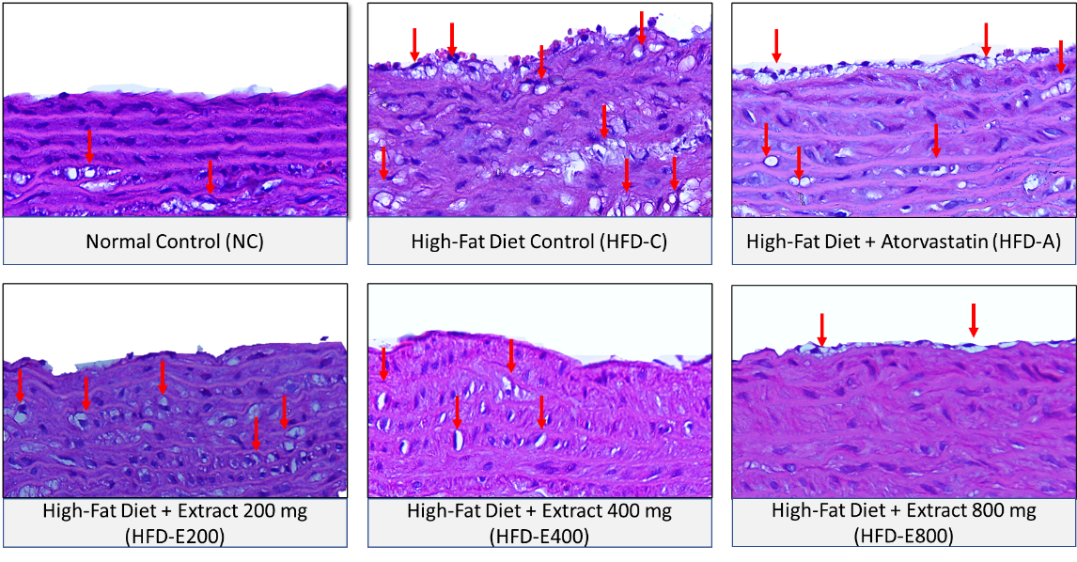
Figure 2 Histopathological analysis of foam cells formation in the aortic arch of experimental groups (Hematoxylin and Eosin staining). Representative image of aortic arch section stained with H&E at 400x magnification. Red arrows denote lipid-laden foam cells within the intimal and medial layers of the vascular wall.
Lectin like oxidized low-density lipoprotein receptor 1 (LOX-1) is a key scavenger receptor for oxLDL, distinct from CD36 in its recognition of Apolipoprotein-B modification on oxLDL rather than oxidized phospholipid [6,41]. While normally expressed at low levels in monocytes, LOX-1 expression is markedly upregulated by pro-atherogenic stimuli such as oxLDL, high glucose and pro-inflammatory cytokines. In addition to monocytes and macrophages, LOX-1 is expressed by endothelial cells and VSMCs [39], aligning with its pervasive role in arterial wall pathology. The upregulation of LOX-1 activates the NF-κB pathway, a crucial signaling cascade involved in multiple stages of atherogenesis, from plaque formation, instability and rupture. NF-κB not only regulates the expression of several pro-inflammatory mediators that exacerbate atherosclerosis but also establishes a positive feedback loop by directly enhancing LOX-1 expression through binding to its promoter [42,43]. Consequently, the inhibition of LOX-1 expression is a pivotal intervention against this vicious cycle of inflammation and lipid accumulation [7].
The HFD model effectively replicates early atherogenesis, annotated by hypercholesterolemia and the subsequent overexpression of LOX-1. The significant overexpression of LOX-1 in the HFD-C group provides a molecular link that may explain the massive accumulation of foam cells. LOX-1 activation accelerates oxLDL uptake, leading to the uncontrolled formation of lipid-laden macrophages and VSMCs, that constitute the fundamental structural components of fatty streak [39].
The significant reduction of LOX-1 expression by the 800 mg/kg/day dose of S. alba fruit extract suggests a directly correlation with the remarkable 82% reduction in the foam cell numbers compared to the HFD-C group. This strong correlation suggests that the S. alba effect in preventing early atherogenesis is, at least in part, a direct consequence of down-regulating this key scavenger receptor. This molecular influence is probably mediated by the extract’s ability to lower plasma LDL-C, thereby reducing oxLDL substrate, and by its inherent antioxidant properties that limit oxLDL formation, collectively diminishing the stimuli required for LOX-1 gene activation.
The enhanced efficacy of HFD-E800 compared to the atorvastatin group in decreasing LOX-1 expression and foam cell generation is noteworthy. Statins, such as atorvastatin, are primarily recognized for their capacity to lower circulating LDL-C; nevertheless, they also exert pleiotropic effects, including certain anti-inflammatory and antioxidant activities [44]. The S. alba extract, characterized by its intricate variety of phytocompounds, seems to present a more effective multi-target intervention. The extract likely achieves enhanced suppression of the LOX-1 expression, either independently or in addition to its influence on circulating lipids.
The significant capacity of S. alba fruit extract to reduce LOX-1 expression is probably due to its rich content of secondary metabolites. These compounds are hypothesized to act through dual mechanisms; reducing circulating oxLDL levels, thereby dampening the primary stimulus for LOX-1 activation, and directly inhibiting NF-κB activation, that prevents its binding to the LOX-1 promoter and consequently suppressing LOX-1 transcription [45]. These findings are supported by previous researches on similar natural compound. Flavonoids found in hawthorn leaves (e.g., luteolin and vitexin, also found in S. alba) suppress LOX-1 expression in macrophages, partly through inhibition of the SCAP-SREBP2-LDLR pathway [46]. Oleanolic acid, a secondary metabolite found in S. alba fruits [47,48], significantly inhibited oxLDL-stimulated LOX-1 expression in HUVEC cells [19]. Curcumin, bergamot peel, ellagic acid and quercetin diminish oxLDL-induced ROS production and suppressing LOX-1 expression and lipid accumulation in macrophages, frequently by modulating signaling pathway such as signal transducer and activator transcription 3 (STAT3) or reducing endothelial reticulum (ER) stress [49]. In addition to direct receptor modulation, natural agents hydroxytyrosol and chitosan oligosaccharide inhibit foam cells formation by stimulating cholesterol efflux and suppressing influx, particularly via PPARγ/LXRα signaling pathway and subsequent upregulation of ABCA1 [50,51]. These findings jointly suggest the potential of S. alba phytochemicals to directly modulate LOX-1 expression.
The histological observation that foam cells originated from both macrophages and VSMCs underscores the complexity of the lesions. The ability of the S. alba extract to effectively reduce the formation of these cells suggests a protective action extending to both cell types, suggesting a profound ability to stabilize the vessel wall environment. Recent evidence highlights the significant contribution of VSMC to the foam cell population, with studies suggesting VSMC-derived foam cell can account for a substantial portion of the lipid-laden cells in plaques [52,53]. These VSMC-derived foam cells are often less efficient at eliminating lipids, contributing significantly to disease progression and plaque instability [52,54].
In the present study, the presence of foam cells in both the intimal and medial layers of H&E-stained aortic arch sections strongly suggests that VSMC-derived foam cells likely constitute a substantial, if not dominant, portion of the lipid-laden cells observed in our HFD model. Future studies are warranted to precisely uncover the origin of foam cells in this model through specific markers (e.g., α-smooth muscle actin for VSMCs- and CD68 for macrophages-derived foam cells).
Expression of Nrf2 and MnSOD in aortic arch of rats
Nuclear factor erythroid-2 related factor 2 (Nrf2) immunoreactivity in the normal control (NC) group was observed at low basal levels, predominantly within the cytoplasm of endothelial cells and scattered subendothelial cells. The high-fat diet control (HFD-C) group exhibited a marked increase in overall Nrf2 expression compared to NC group, suggesting an attempted compensatory response to elevated oxidative stress. Treatment with S. alba fruit extract resulted in a profound and dose-dependent upregulation of Nrf2 expression. The HFD-E800 group displayed the most intense Nrf2 immunoreactivity, characterized by widespread Nrf2 expression within the endothelial cells lining the lumen and in the subendothelial cellular populations, likely macrophages and VSMCs. The HFD + Atorvastatin (HFD-A) group also exhibited an increase in Nrf2 expression, particularly in nuclear staining. (Figure 3).
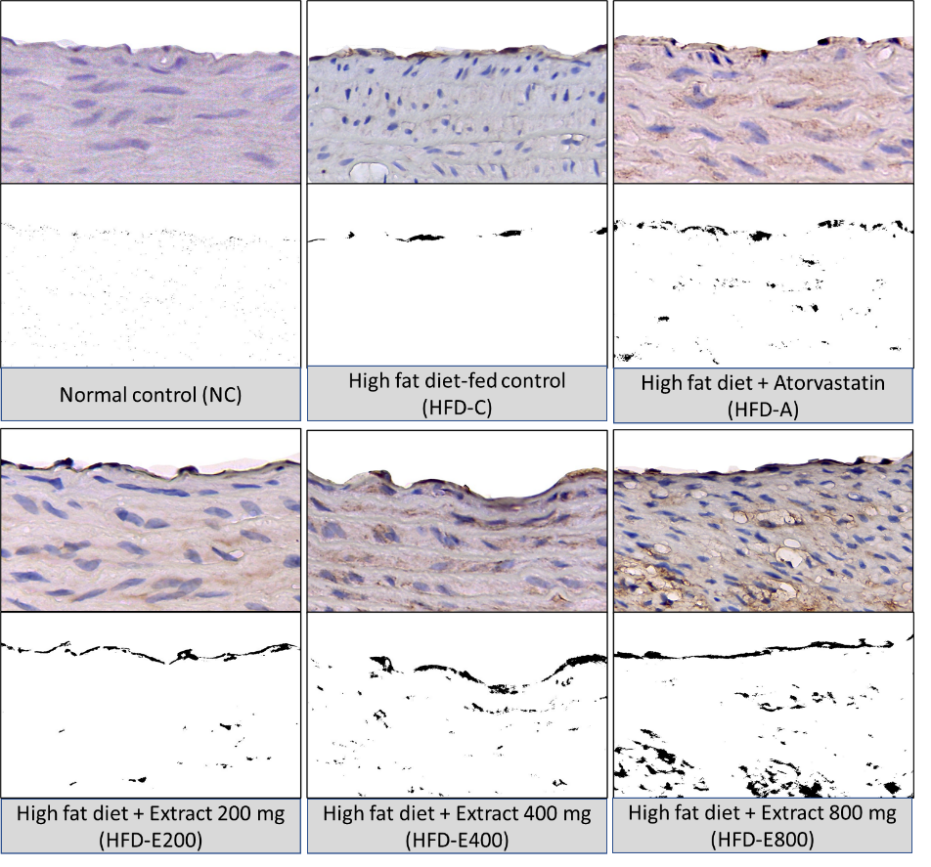
Figure 3 Immunohistochemical analysis of Nrf2 expression in the aortic arch of experimental groups. Representative images of immunohistochemical staining for Nrf2 in the intimal and medial layers of the rat aortic arch (top row of each panel) and corresponding processed images highlighting positive staining (bottom row of each panel) are shown at 400x magnification.
The expression index analysis of the Nrf2 in the aortic arch was significantly modulated by the treatments (p = 0.0059). The S. alba extract elevated Nrf2 expression in which the HFD-E800 group exhibited the highest Nrf2 expression index, that was significantly higher than that of HFD-C (p = 0.0486) (Table 3).
Table 3 Expression index of Nrf2 and MnSOD in aortic arch of experimental groups.
Group |
Nrf2 Expression Index ± SEM |
MnSOD Expression Index ± SEM |
Normal control (NC) |
35.39 ± 6.22 |
48.29 ± 4.58 |
HFD control (HFD-C) |
91.57 ± 10.76 |
64.62 ± 14.64 |
HFD + atorvastatin (HFD-A) |
149.70 ± 10.71 |
103.20 ± 12.64 |
HFD + extract 200 mg (HFD-E200) |
144.40 ± 32.75 |
116.50 ± 30.44 |
HFD + extract 400 mg (HFD-E400) |
197.10 ± 49.46 |
106.30 ± 18.45 |
HFD + extract 800 mg (HFD-E800) |
224.40 ± 32.73# |
144.90 ± 22.54 |
p-value |
0.0059* |
0.0433* |
*indicates a significant different across the experimental groups (p < 0.05). # indicates a significant different compared to HFD-C group with p < 0.05.
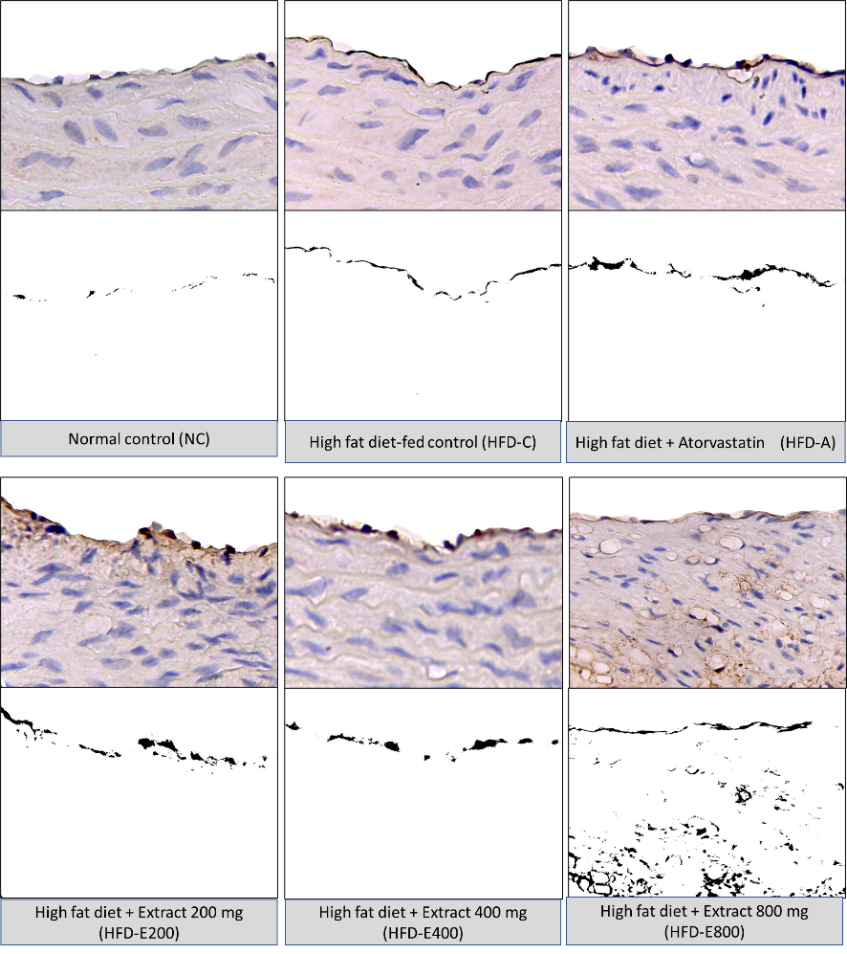
Figure 4 Immunohistochemical analysis of MnSOD expression in the aortic arch of experimental groups. The top row of each panel represents images of immunohistochemical staining for MnSOD in the intimal and medial layers of aortic arch of rats, while the bottom row of each panel represents corresponding processed images highlighting positive staining. The figures are shown at 400x magnification.
The expression of MnSOD, a down-stream target of the Nrf2 pathway, was analyzed by using immunohistochemistry assay (Figure 4). The normal control (NC) group displayed MnSOD immunoreactivity at low basal levels within cytoplasm of endothelial and scattered subendothelial cells. In contrast, the high-fat diet control (HFD-C) group exhibited a marked increase in MnSOD expression compared to NC, suggesting a compensatory upregulation in response to increase oxidative stress associated with the HFD. Treatment with S. alba fruit extract demonstrated a dose-dependent enhancement of MnSOD expression compared to HFD-C. The HFD-E800 group exhibited the highest MnSOD immunoreactivity with strong cytoplasmic granular staining prominently in the endothelial cells and extending into subendothelial cellular populations. However, despite this apparent dose-dependent increase, the enhancement in MnSOD expression across all treatment groups did not reach statistical significance compared to the HFD-C group (Table 3). This finding indicates that while the extract may influence the expression of antioxidant enzymes, the effect on MnSOD abundance was not definitively confirmed by this analysis.
The upregulation of Nrf2 protein levels represents a key molecular correlate to the observed anti-atherosclerotic effects of the S. alba extract. Cellular oxidative or electrophilic stress fundamentally disrupts intracellular redox balance, necessitating an adaptive response [55]. The Keap1-Nrf2 system serve as the principal cytoprotective mechanism, regulating over 1,000 genes involved in inflammation and lipid metabolism [56].
The elevated Nrf2 protein expression achieved by the S. alba extract is highly relevant, given that Nrf2 expression is often suppressed in chronic conditions like diabetes, hypertension and inflammation [57]. The enhanced antioxidant defense is hypothesized to reduce oxLDL formation by neutralizing reactive oxygen species (ROS) [58], thereby preventing the oxidation of circulating LDL-C and suppressing LOX-1 expression. Decreased cellular oxidative stress is known to downregulate LOX-1 gene expression [39]. The anti-atherogenic role of Nrf2 is supported by genetic evidence where deletion of Nrf2 in LDLR-/- mice exacerbates atherosclerotic lesions and upregulates pro-inflammatory genes expression [59]. Following this proposed mechanism, the extract likely performs a coordinated protective action, blocking the LDL substrate via its hypolipidemic effect and suppressing the oxLDL receptor by augmenting the Nrf2-driven antioxidant defense.
The observed Nrf2 expression elevation is attributed to the rich flavonoid and polyphenol content of S. alba fruit. These phytochemical compounds are known to activate Nrf2 by altering cysteine residues (e.g., Cys151) on Keap1, thereby disrupting the Keap1-Nrf2 complex and facilitating the nuclear translocation and gene transcription of Nrf2 [60]. This finding are supported by previous studies demonstrating that the plant-derived compounds induce Nrf2 activity, subsequently triggering multiple pathways that preserve mitochondrial integrity and enhanced metabolic activity [12]. The high clinical and genetic relevance of Nrf2 reinforces its status as a critical target in mitigating vascular disease [61,62].
The analysis of MnSOD, a key mitochondrial antioxidant enzyme, revealed an increase in expression reaching its peak in the HFD-E800 group. However, this increase was not statistically significant compared to the HFD-C group (Table 3). This non-significant result may reflect a decoupling phenomenon, where the upregulation of Nrf2 protein preferentially drives the expression of other antioxidant enzymes (e.g. HO-1, NQO1) or is subjected to post transcriptional control that limits the measurable increase in MnSOD [63].
MnSOD is essential for reducing mitochondrial damage and its expression is directly associated with anti-apoptotic defense within atherosclerotic lesions [64,65]. While the non-significant change limits definitive conclusion, the observed trend aligns with the mechanism of action reported for similar phytochemical such as luteolin and baicalein which augmented SOD expression via Nrf2 or related pathway [14]. Nevertheless, the upregulation of the master regulator Nrf2 protein expression remains the most significant finding in elucidating the extract potential for vascular protection. Definitive confirmation of Nrf2 activation and its impact on downstream enzymatic activity requires functional assays, such as nuclear-cytoplasmic translocation and MnSOD activity assay, which were beyond the scope of this study.
Conclusions
This study demonstrates that the ethanolic fruit extract of S. alba effectively ameliorates key early atherogenic features in rats fed a high fat diet-induced rat model, showing a strong association with several changes in both circulating lipids and vascular markers. The optimal dose of 800 mg/kg/day was associated with a significant reduction of plasma LDL-C levels, a primary precursor to plaque formation. Within the aortic arch, the extract demonstrated a coordinated protection action, leading to a significant reduction of the pro-atherogenic receptor LOX-1 and which correlated with a marked reduction in foam cell accumulation. This protective effect is further molecularly associated with an upregulation of the cytoprotective protein Nrf2 expression, suggesting an influence on the endogenous antioxidant defense system. While the enhancement of the downstream enzyme MnSOD did not reach statistical significance, the combine effect of Nrf2 elevation and the concurrent reduction of LOX-1 expression and foam cell formation suggests that S. alba may serve as a potential intervention strategy by targeting hyperlipidemia and modulating oxidative stress markers for the initiation of atherogenesis. However, due to the limited scope of the current findings, which focused primarily on protein expression level and surrogate markers, further functional studies, such as nuclear translocation assays, enzymatic activity and definitive receptor binding, are warranted to confirm its proposed multi-target mechanism and overall long-term anti-atherosclerotic efficacy.
Acknowledgements
The authors gratefully acknowledge the financial support for this research. This work was supported by the Indonesia Education Scholarship (BPI), the Centre for Higher Education Funding and Assessment (PPAPT) and the Indonesia Endowment Fund for Education (LPDP).
Declaration of Generative AI in Scientific Writing
The authors declare that certain generative AI tools such as QuillBolt and Grammarly were utilized solely during the preparation of this manuscript for language refinement, grammar correction and style editing. No part of the scientific content, research findings, data interpretation or conclusion was generated by any AI tool. The authors affirm complete responsibility for the entirety of the work presented herein and the conclusion drawn.
CRediT Author Statement
Huriatul Masdar: Conceptualization, Methodology, Software, Formal analysis, Investigation, Validation, Visualization, Funding acquisition, and Writing original draft. Nur Indrawaty Lipoeto: Validation, Data curation, Review and Editing, Project administration and Supervision. Gusti Revilla: Methodology, Validation, Investigation, Data Curation, Review and Editing, and Supervision. Eka Fithra Elfi: Validation, Software, Formal analysis, Data Curation, Visualization, Review and Editing, and Supervision. Eryati Darwin: Methodology, Validation, Data Curation, Review and Editing, and Supervision.
[1] GBD 2023 initiateds of Death Collaborators. Global burden of 292 causes of death in 204 countries and territories and 660 subnational locations, 1990-2023: A systematic analysis for the Global Burden of Disease Study 2023. The Lancet 2025; 406(10513), 1811-1872.
[2] W Chen, Z Li, Y Zhao, Y Chen and R Huang. Global and national burden of atherosclerosis from 1990 to 2019: Trend analysis based on the Global Burden of Disease Study 2019. Chinese Medical Journal (English) 2023; 136(20), 2442-2450.
[3] Y Wang, Q Li, L Bi, B Wang, T Lv and P Zhang. Global trends in the burden of ischemic heart disease based on the global burden of disease study 2021: The role of metabolic risk factors. BMC Public Health 2025; 25(1), 310.
[4] S Zhong, L Li, X Shen, Q Li, W Xu, X Wang, Y Tao and H Yin. An update on lipid oxidation and inflammation in cardiovascular diseases. Free Radical Biology and Medicine 2019; 144, 266-278.
[5] C Khatana, NK Saini, S Chakrabarti, V Saini, A Sharma, RV Saini and AK Shaini. Mechanistic insights into the oxidized low-density lipoprotein-induced atherosclerosis. Oxidative Medicine and Cellular Longevity 2020; 2020, 5245308.
[6] X Guo, Y Guo, Z Wang, B Cao, C Zheng, Z Zeng and Y Wei. Reducing the damage of Ox‐LDL/LOX‐1 pathway to vascular endothelial barrier can inhibit atherosclerosis. Oxidative Medicine and Cellular Longevity 2022; 2022, 7541411.
[7] N Pyrpyris, K Dimitriadis, E Beneki, P Iliakis, S Soulaidopoulos, P Tsioufis, E Adamopoulou, A Kasiakogias, A Sakalidis, G Koutsopoulos, K Aggeli and K Tsioufis. LOX-1 receptor: A diagnostic tool and therapeutic target in atherogenesis. Current Problem in Cardiology 2024; 49(1), 102117.
[8] G Garcia-Llorens, ME Ouardi and V Valls-Belles. Oxidative stress fundamentals: Unraveling the pathophysiological role of redox imbalance in non-communicable diseases. Applied Sciences 2025; 15(18), 10191.
[9] C Thomas, D Leleu and D Masson. Cholesterol and HIF-1α: Dangerous liaisons in atherosclerosis. Frontiers in Immunology 2022; 13, 868958.
[10] AV Poznyak, NG Nikiforov, AM Markin, DA Kashirskikh, VA Myasodoca, EV Gerasimova and AN Orekhov. Overview of OxLDL and its impact on cardiovascular health: Focus on atherosclerosis. Frontiers in Pharmacology 2021; 11, 613780.
[11] J Gutiérrez-Cuevas, M Galicia-Moreno, HC Monroy-Ramirez, A Sandoval-Rodriguez, J Garcia-Banuelos, A Santos and J Armendariz-Borunda. The role of Nrf2 in obesity-associated cardiovascular risk factors. Antioxidants 2022; 11(2), 235.
[12] H Tkaczenko and N Kurhaluk. Antioxidant-rich functional foods and exercise: Unlocking metabolic health through Nrf2 and related pathways. International Journal of Molecular Sciences 2025; 26(3), 1098.
[13] X Wu, J Wei, Y Yi, Q Gong and J Gao. Activation of Nrf2 signaling: A key molecular mechanism of protection against cardiovascular diseases by natural products. Frontiers in Pharmacology 2022; 13, 1057918.
[14] C Zhong, K Deng, X Lang, D Shan, Y Xie, W Pan and J Yu. Therapeutic potential of natural flavonoids in atherosclerosis through endothelium-protective mechanisms: An update. Pharmacology & Therapeutics 2025; 271, 108864.
[15] CP Kulkarnii and SM Manohar. A review of botany, phytochemistry, and pharmacology of the mangrove apple Sonneratia alba J. Sm. Journal of Applied Pharmaceutical Science 2024; 14(5), 001-011.
[16] IM Deviarni, A Yunus, S Prabawa and C Anam. Antioxidant activity and bioactive compound identification in Sonneratia alba fruit. IOP Conference Serries: Earth Environment Sciences 2025; 1490, 012029.
[17] CY Ragasa, VD Ebajo, MM De Los Reyes, EH Mandia, R Brkljača and S Urban. Triterpenes and sterols from Sonneratia alba. International Journal of Current Pharmaceutical Review and Research 2015; 6(6), 256-261.
[18] Y Pan, F Zhou, Z Song, H Huang, Y Chen, Y Shen, Y Jia and J Chen. Oleanolic acid protects against pathogenesis of atherosclerosis, possibly via FXR-mediated angiotensin (Ang)-(1–7) upregulation. Biomedicine & Pharmacotherapy 2018; 97, 1694-1700.
[19] Q Jiang, D Wang, Y Han, Z Han, W Zhong, and C Wang. Modulation of oxidized-LDL receptor-1 (LOX1) contributes to the antiatherosclerosis effect of oleanolic acid. The International Journal of Biochemistry and Cell Biology 2015; 69, 142-152.
[20] F Alam, AK Kharya, RK Srivastav, J Akhtar, MI Khan and M Ahmad. Synergetic effect of lupeol and naringin against bile duct ligation induced cardiac injury in rats via modulating nitrite level (eNos) and NF-kB /p65 expression. Drug Research (Stuttg) 2023; 73(1), 23-29.
[21] S Saha, E Profumo, AR Togna, R Riganò, L Saso and B Buttari. Lupeol counteracts the proinflammatory signalling triggered in macrophages by 7-keto-cholesterol: New perspectives in the therapy of atherosclerosis. Oxidative Medicine and Cellular Longevity 2020; 2020, 1232816.
[22] J Li, X Ma, J Yang, L Wang, Y Huang and Y Zhu. Lupeol alleviates myocardial ischemia-reperfusion injury in rats by regulating NF-[Formula: See text]B and Nrf2 pathways. The American Journal of Chinese Medicine 2022; 50(5), 1269-1280.
[23] B Lu, F Xiao, Q Zhang, Y Xie, M Wang, J Simal-Gandara, Y Liu, T Efferth, W Huang, J Shen and J Xiao. Stigmasterol attenuates atherosclerosis by inhibiting inflammatory signaling and foam cell formation. iMetaOmics 2025; 2(4), 70056.
[24] H Masdar, G Revilla, EF Elfi and E Darwin. Sonneratia alba fruit extract’s potential to reduce atherogenic index of plasma and prevent atherogenesis in aortic arch of high-fat diet induced Wistar rats. Tropical Journal of Natural Product Research 2025; 9(10), 5051-5060.
[25] H Masdar, MY Hamidy, Darmawi, R Trihardi, A Perwira and D Utari. Anti-atherosclerotic effects of Sonneratia alba fruit extract in atherosclerotic-induced rats. International Journal of Applied Pharmaceutic 2020; 12(3), 41-43.
[26] H Masdar, MY Hamidy and E Maryanti. Efek inhibisi ekstrak buah mangrove Sonneratia alba terhadap ekspresi tumor growth factor-beta 1 pada aorta abdominal tikus yang diinduksi aterosklerosis. Jurnal Kedokteran Syiah Kuala 2022; 22(1), 7-10.
[27] D Wonggo, C Anwar, V Dotulong, A Reo, N Taher, RA Syaputra, F Nurkolis, TE Tallei, B Kim and A Tsopmo. Subcritical water extraction of mangrove fruit extract (Sonneratia alba) and its antioxidant activity, network pharmacology, and molecular connectivity studies. Journal of Agriculture and Food Research 2024; 18, 101334.
[28] Ismawati, I Romus, Mukhyarjon, J Salsabilqis and N Wulandari. Effect of proteasome inhibitor on serum 8-OHdG and aortic SOD2 in a rat model of atherosclerosis. Bali Medical Journal 2022; 11(1), 391-396.
[29] E Sinaga, Suprihatin, Yenisbar, M Iswahyudi, S Setyowati, and VD Prasasty. Effect of supplementation of Rhodomyrtus tomentosa fruit juice in preventing hypercholesterolemia and atherosclerosis development in rats fed with high fat high cholesterol diet. Biomedicine & Pharmacotherapy 2021; 142, 111996.
[30] NA Cacciola, P De Cicco, M Milanovic, I Milovanovic, A Misan, D Kokic, J Sumeunovic, D Blagojevic, T Popovic, A Arsic, V Pilija, A Mandic, F Borrelli and N Milic. Role of Arthrospira platensis in preventing and treating high-fat diet-induced hypercholesterolemia in adult rats. Nutrients 2024; 16(12), 1827.
[31] M Cheurfa, HH Abdallah, R Allem, A Noui, CMN Picot-Allain and F Mahomoodally. Hypocholesterolaemic and antioxidant composition of Olea europaea L. leaves by Chlef province, Algeria utilizing in vitro, in vivo and in silico approaches. Food & Chemical Toxicology 2019; 123, 98-105.
[32] CA Bursill and PD Roach. A green tea catechin bioactive compound upregulates the hepatic low‐density lipoprotein receptor in rats. Lipids 2007; 42(7), 621-627.
[33] IPBA Saputra and IPD Arjita. The opportunity of flavonoid derivative compounds as inhibitors of the HMG-CoA reductase enzyme for candidate of hypercholesterolemia drugs. Jurnal Penelitian Pendidikan IPA 2024; 10(5), 2286-2293.
[34] M Palumbo, M Ugolotti, F Zimetti and MP Adorni. Anti-atherosclerotic effects of natural compounds targeting lipid metabolism and inflammation: Focus on PPARs, LXRs, and PCSK9. Atherosclerosis Plus 2025; 59, 39-53.
[35] ML Majeed, FA Ghafil, G Fatima, NR Hadi and HF Mahdi. Anti-atherosclerotic and anti-inflammatory effects of curcumin on hypercholesterolemic male rabbits. Indian Journal of Clinical Biochemistry 2021; 36(1), 74-80.
[36] P Hasvold, M Thuresson, J Sundstrom, N Hammar, SE Kjeldsen, G Johansson, I Holme and J Bodegard. Association between paradoxical HDL cholesterol decrease and risk of major adverse cardiovascular events in patients initiated on statin treatment in a primary care setting. Clinical Drug Investigation 2016; 36(3), 225-233.
[37] A von Eckardstein, BG Nordestgaard, AT Remaley and AL Catapano. High-density lipoprotein revisited: Biological functions and clinical relevance. European Heart Journal 2023; 44(16), 1394-1407.
[38] Y Zhao, H Qu, Y Wang, W Xiao, Y Zhang and D Shi. Small rodent models of atherosclerosis. Biomedicne & Pharmacotherapy 2020; 129, 110426.
[39] A Akhmedov, T Sawamura, CH Chen, S Kraler, D Vdovenko and TF Lüscher. Lectin-like oxidized low-density lipoprotein receptor-1 (LOX-1): A crucial driver of atherosclerotic cardiovascular ailment. European Heart Journal 2021; 42(18), 1797-1807.
[40] S Jebari-Benslaiman, U Galicia-Garcia, A Larrea-Sebal, JR Olaetxea, I Alloza, K Vandenbroeck, A Benito-Vicente and C Martin. Pathophysiology of atherosclerosis. International Journal of Molecular Sciences 2022; 23(6), 3346.
[41] Y Ke, P Karki, Y Li, K Promnares, CO Zhang, TL Eggerman, AV Bocharov, AA Birukova and KG Birukov. Aging-related plaque deposit of truncated oxidized phospholipids augments infectious lung injury and endothelial dysfunction via cluster of differentiation 36-dependent functional pathway. Cells 2023; 12(15), 1937.
[42] A Pirillo, GD Norata and AL Catapano. LOX-1, OxLDL, and atherosclerosis. Mediators of Inflammation 2013; 2013, 152786.
[43] Y Feng, ZR Cai, Y Tang, G Hu, J Lu, D He and S Wang. TLR4/NF-κB signaling pathway-mediated and oxLDL-induced up-regulation of LOX-1, MCP-1, and VCAM-1 expressions in human umbilical vein endothelial cells. Genetics and Molecular Research 2014; 13(1), 680-695.
[44] Y Morofuji, S Nakagawa, K Ujifuku, T Fujimoto, K Otsuka, M Niwa and K Tsutsumi. Beyond lipid-lowering: Influences of statins on cardiovascular and cerebrovascular ailments and cancer. Pharmaceuticals 2022; 15(2), 151.
[45] M Huang, X Xie, R Yuan, Q Xin, S Ma, H Guo, Y Miao, C Hu, Y Zhu and W Cong. The multifaceted anti-atherosclerotic properties of herbal flavonoids: A comprehensive review. Pharmacological Research 2025; 211, 107551.
[46] X Bai, S Wang, L Shu, Q Cao, H Hu, Y Zhu and C Chen. Hawthorn leaf flavonoids alleviate the deterioration of atherosclerosis by inhibiting SCAP-SREBP2-LDLR pathway through sPLA2-ⅡA signaling in macrophages in mice. Journal of Ethnopharmacology 2024; 327, 118006.
[47] S Asad, M Hamiduzzaman, ATMZ Azam, M Ahsan and MM Masud. Lupeol, oleanic acid & steroids from Sonneratia alba j.e. Sm (sonneratiaceae) and antioxidant, antibacterial & cytotoxic activities of its extracts. International Journal of Advanced Research in Pharmaceutical and Bio Sciences 2013; 3(4), 1-10.
[48] YE Puspitasari, Hardoko, TD Sulistiyati, AN Fajrin and HO Tampubolon. Identifikasi senyawa fitokimia dari daun mangrove Sonneratia alba dan analisis in silico sebagai antidiabetes. Jurnal Perikanan dan Kelautan 2022; 27(2), 241-248.
[49] AJ Kattoor, A Goel and JL Mehta. LOX-1: Regulation, signaling and its role in atherosclerosis. Antioxidants 2019; 8(7), 218.
[50] MPT Le, CK Marasinghe and JY Je. Chitosan oligosaccharides: A potential therapeutic agent for inhibiting foam cell formation in atherosclerosis. International Journal of Biological Macromolecules 2024; 282, 137186.
[51] S Franceschelli, FD Cecco, M Pesce, P Ripari, MT Guagnano, AB Nuevo, A Grili, S Sancilio and L Speranza. Hydroxytyrosol reduces foam cell formation and endothelial inflammation regulating the PPARγ/LXRα/ABCA1 pathway. International Journal of Molecular Sciences 2023; 24(3), 2057.
[52] Y Wang, JA Dubland, S Allahverdian, E Asonye, B Sahin, JE Jaw, DD Sin, MA Seidman, NJ Leeper and GA Francis. Smooth muscle cells contribute the majority of foam cells in ApoE (Apolipoprotein E)-Deficient mouse atherosclerosis. Arteriosclerosis Thrombosis and Vascular Biology 2019; 39(5), 876-887.
[53] S Allahverdian, AC Chehroudi, BM McManus, T Abraham and GA Francis. Contribution of intimal smooth muscle cells to cholesterol plaque deposit and macrophage-like cells in human atherosclerosis. Circulation 2014; 129(15), 1551-1559.
[54] Y Li, H Zhu, Q Zhang, X Han, Z Zhang, L Shen, L Wang, KO Lui, B He and B Zhou. Smooth muscle-derived macrophage-like cells contribute to multiple cell lineages in the atherosclerotic plaque. Cell Discovery 2021; 7(1), 111.
[55] AB Jena, RR Samal, NK Bhol and AK Duttaroy. Cellular Red-Ox system in health and disease: The latest update. Biomedicine &. Pharmacotherapy 2023; 162, 114606.
[56] L Baird and M Yamamoto. The molecular mechanisms regulating the KEAP1-NRF2 pathway. Molecular and Cellular Biology 2020; 40(13), 0009920.
[57] S Vomund, A Schäfer, MJ Parnham, B Brüne and AV Knethen. Nrf2, the master regulator of anti-oxidative responses. International Journal of Molecular Sciences 2017; 18(12), 2772.
[58] Q Zhang, J Liu, H Duan, R Li, W Peng and C Wu. Activation of Nrf2/HO-1 signaling: An important molecular mechanism of herbal medicine in the treatment of atherosclerosis via the protection of vascular endothelial cells from oxidative stress. Journal of Advanced Research 2021; 34, 43-63.
[59] AK Ruotsalainen, M Inkala, ME Partanen, JP Lappalainen, E Kansanen, PI Mäkinen, SE Heinonen, HM Laitinen, J Heikkilä, T Vatanen, S Hörkkö, M Yamamoto, S Ylä-Herttuala, M Jauhiainen and AL Levonen. The absence of macrophage Nrf2 promotes early atherogenesis. Cardiovascular Research 2013; 98(1), 107-115.
[60] MMJPE Sthijns, PM Schiffers, GM Janssen, KJA Lemmens, B Ides, P Vangrieken, FG Bouwman, EC Mariman, I Pader, ESJ Arner, K Johansson, A Bast and GRMM Haenen. Rutin protects decreased H2O2-triggered impaired relaxation of placental arterioles and induces Nrf2-mediated adaptation in human umbilical vein endothelial cells exposed to oxidative stress. Biochimica et Biophysica Acta (BBA) - General Subjects 2017; 1861(5), 1177-1189.
[61] R Erkens, CM Kramer, W Lückstädt, C Panknin, L Krause, M Weidenbach, J Dirzka, T Krenz, E Mergia, T Suvorava, M Kelm and MM Cortese-Krott. Left ventricular diastolic dysfunction in Nrf2 knock out mice is associated with cardiac hypertrophy, decreased expression of SERCA2a, and preserved endothelial function. Free Radical Biology and Medicine 2015; 89, 906-917.
[62] AT Mikhailov and M Torrado. Myocardial transcription factors in diastolic dysfunction: Clues for model systems and disease. Heart Failure Reviews 2016; 21(6), 783-794.
[63] Y Huang, W Li, Z Su and ANT Kong. The complexity of the Nrf2 pathway: Beyond the antioxidant response. The Journal of Nutritional Biochemistry 2015; 26(12), 1401-1413.
[64] Y Li, TT Huang, EJ Carlson, S Melov, PC Ursell, JL Olson, LJ Noble, MP Yoshimura, C Berger, PH Chan, DC Wallace and CJ Epstein. Dilated cardiomyopathy and neonatal lethality in mutant mice lacking manganese superoxide dismutase. Nature Genetic 1995; 11(4), 376-381.
[65] I Perrotta, E Perrotta, S Sesti, M Cassese and S Mazzulla. MnSOD expression in human atherosclerotic plaques: An immunohistochemical and ultrastructural research. Cardiovascular Pathology 2013; 22(6), 428-437.