
Trends
Sci.
2026;
23(6):
12246
Natural Deep Eutectic Solvent (NADES) Extract of Curcuma Xanthorrhiza Roxb Attenuates Physical Fatigue and Increases Exercise Tolerance in Mice
Adelina
Simamora1,2,
![]() ,
Heri Setiawan2,3,
,
Heri Setiawan2,3,
![]() ,
Kris Herawan Timotius1,
,
Kris Herawan Timotius1,
![]() ,
,
Peni
Ahmadi2,4,
![]() ,
Mukerrem Betul Yerer5,
,
Mukerrem Betul Yerer5,
![]() and Abdul Munim2,6,*
and Abdul Munim2,6,*
1Department of Biochemistry, Faculty of Medicine and Health Sciences, Krida Wacana Christian University,
Jakarta 11510, Indonesia
2National Metabolomics Collaborative Research Center, Faculty of Pharmacy, Universitas Indonesia,
Depok 16424, Indonesia
3Department of Pharmacology, Faculty of Pharmacy, Universitas Indonesia, Depok 16424, Indonesia
4Research Center for Vaccine and Drugs, Research Organization for Health,
National Research and Innovation Agency (BRIN), Cibinong 16911, Indonesia
5Department of Pharmacology, Faculty of Pharmacy, University of Erciyes, Kayseri 38039, Turkey
6Department of Pharmacognosy-Phytochemistry, Faculty of Pharmacy, Universitas Indonesia, Depok 16424, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 12 October 2025, Revised: 8 December 2025, Accepted: 15 December 2025, Published: 20 February 2026
Abstract
Background: Fatigue, a common problem in modern society, reduces quality of life and productivity, highlighting the need for natural remedies. Curcuma xanthorrhiza Roxb., a well-known Indonesian plant medicine, is recognized for its diverse bioactivities, mainly attributed to its marker compound xanthorrhizol (XTZ). Aim: This study investigated the antifatigue activity of C. xanthorrhiza extracts extracted by an eco-friendly natural deep eutectic solvent (NADES), composed of choline chloride and sucrose (CC-Suc 3:1, 10% water). Methods: Male Balb/c mice were randomly divided into seven groups (n = 4). Each group was orally administered for 28 days vehicles (carboxymethyl cellulose and CC-Suc), the positive control (taurine 700 mg/kg/BW), C. xanthorrhiza extracted by CC-Suc or ethanol equivalent to 10 or 25 mg XTZ/kg BW. The effect of treatment on exercise performance was evaluated by forced swimming test (FST) and changes in fatigue-related parameters. Results: C. xanthorrhiza extracts prolonged FST time in a dose-dependent manner, increased muscle ATP and adenosine 5-monophosphate- (AMP-) activated protein kinase (AMPK) levels. Treatment with ethanol extracts of C. xanthorrhiza reduced serum urea nitrogen in a dose dependent trend compared with the vehicle group. No significant differences were observed in hepatic alanine aminotransferase (ALT) and aspartate aminotransferase (AST), indicating absence of liver toxicity. Conclusions: These findings demonstrate that NADES-based C. xanthorrhiza extract improves exercise endurance, possibly through modulation of energy metabolism. Moreover, the choline chloride–sucrose NADES serves as a safe and sustainable extraction solvent and delivery medium, supporting potential use of C. xanthorrhiza as a functional food ingredient against fatigue.
Keywords: AMPK, ATP, Choline chloride-sucrose, Deep eutectic solvent, Fatigue, Taurine, Xanthorrhizol
Introduction
Fatigue has become a major public health problem that affects the quality of life and work efficiency. Fatigue is also a common symptom of diseases, such as HIV/AIDS, cancer, and diabetes. Global reports reveal
that the prevalence of adults experiencing fatigue for more than six months was 20% of the population [1]. Fatigue relates to various mechanisms, including the endocrine system, central and peripheral nervous system, metabolic activity, immune system, and oxidative stress [2]. Therapeutic regimens for fatigue, however, are limited due to adverse side effects, including addiction and excitability [3]. Herbal sources are rich in antifatigue agents [4]. Literatures have reported the antifatigue effect of extracts or compounds of plant origin by different mechanisms as reported by molecular, animal, and clinical studies [5,6]. In Indonesian traditional medicine, C. xanthorrhiza has been used to eliminate fatigue and maintain body wellness [7]. Despite traditional uses, scientific reports are insufficient to support these claims.
Phytochemical studies on C. xanthorrhiza reported that xanthorrhizol and curcumin are among the main markers in the rhizomes [8]. Studies have demonstrated that curcumin reduced fatigue through several mechanisms as seen by animal and molecular models [6]. Curcumin supplementation increased swimming time in mice in a dose-dependent manner, possibly by increasing glycogen storage in mice muscle and reducing fatigue markers including blood urea nitrogen (BUN), alanine aminotransferase (ALT), aspartate aminotransferase (AST), and creatinine kinase [6]. Another study showed that curcumin improved exercise performance by the reduction of oxidative stress related to fatigue, as demonstrated by an increase in liver superoxide dismutase (SOD) and a decrease in malondialdehyde (MDA) levels in mice [9]. At the molecular level, it was observed that curcumin treatment upregulated mitochondria biogenesis, activated adenosine monophosphate-activated protein kinase (AMPK), and increased the expression of sirtuin-1 (SIRT1) [6], which are all associated with energy production. In addition, Kim et al. [10] found that water extract of C. longa improved exercise endurance in mice and promoted intramuscular mitochondrial biogenesis [10]. Despite the various antifatigue studies of curcumin and C. longa, to date, no study reported on the antifatigue effect of C. xanthorrhiza.
Extraction methods that currently exist for C. xanthorrhiza use organic solvents, such as hexane, ethanol, and ethyl acetate [11]. However, they suffer from some limitations, including a long extraction process to remove toxic extraction solvents and the presence of residual solvents in the extract. To obtain cleaner, non-toxic, and more environmentally friendly extracts, natural deep eutectic solvents (NADES) – a new generation of solvents, have emerged as an alternative to conventional solvents. NADES are formed of hydrogen bond acceptor and hydrogen bond donor compounds of natural origins, which upon mixing produce liquid with a low melting point, lower than their forming components [12]. Previously, we have reported that NADES consisting of glucose: Lactic acid (1:3) succeeded in selectively extracting a higher yield of xanthorrhizol than using an ethanol solvent [13].
In this paper, we investigated the antifatigue effect of C. xanthorrhiza extract prepared by choline chloride: Sucrose (CC-Suc) using a forced swimming test in mice. The effect of extract administration was investigated on energy metabolisms and fatigue-related markers. NADES composed of CC and Suc have been shown to extract bioactive compounds more effectively compared to conventional solvents [14]. An addition of water into CC-Suc (10%) was required to reduce viscosity that is typically high in sugar based-NADES, while still maintaining the hydrogen bond network in NADES [15]. Choline chloride and sucrose are inexpensive, low-toxic, and food-grade building blocks, supporting a safer route to nutraceutical extracts [16]. In addition, NADES can serve as a delivery matrix, thus simplifying formulation for oral application [16]. To our knowledge, this is the first report to apply CC-Suc NADES as both extraction solvent and oral-compatible vehicle for C. xanthorrhiza. The study provides a basis for antifatigue mechanisms of C.xanthorrhiza and indicates its potential application for health supplementation.
Materials and methods
Plant material and chemicals
Rhizomes of C. xanthorrhiza were obtained from Balittro (Research Institute for Herbs and Spices) Bogor, Indonesia (-6.576893345305293, 106.78633149414519). The plant was identified by Dr. Anang Setiawan Achmadi of the Research Center for Biosystematics and Evolution, Indonesia. A specimen was deposited at the Herbarium Bogoriense (voucher number HB B-609.V.D1.0507). C. xanthorrhiza oil was kindly gifted from PT. Phytochemindo Reksa. Choline chloride (CC) was purchased from Xi’an Rongsheng Biotechnology (Xi’an, China). Sucrose (Suc) were obtained from Merck (Darmstadt, Germany). Taurine (TAU) was purchased from Sigma Aldrich (St. Louis, MO, USA). Total AMP-Activated Protein Kinase ELISA kit was obtained from Bioenzy (CN: BZ-22143759-CPEB). The following assay kits were obtained from Elabscience (Houston, Texas, USA): ATP colorimetric assay kit (Cat No E-BC-K157-S), alanine aminotransferase (ALT) (Cat No E-BC-K235-M) and aspartate aminotransferase (AST) (Cat No E-BC-K236-M) activity assay kits, urea (BUN) colorimetric assay kit (E-BC-K183-M). Bovine serum albumin was obtained from Vivantis (Selangor, Malaysia). Bradford protein assay dye reagent was bought from Bio-Rad (Hercules, USA). All buffer reagents were of analytical grade. Xanthorrhizol was purchased from MarkHerb (Bandung, Indonesia). Organic solvents used for HPLC experiment were of HPLC grade, and obtained from SmartLab (Tanggerang, Indonesia). Bidistilled water pro injection, used for sample dilution and HPLC separation, was obtained from IkaPharmindo Putramas (Jakarta, Indonesia).
Preparation of choline chloride: Sucrose (CC-Suc) extract of C. xanthorrhiza
NADES composed of choline chloride (CC): Sucrose (Suc) (CC-Suc, 3:1, 10% water) was used in this study. The mole ratio and percentage of water addition was optimized by response surface methodology as reported previously [17]. CC-Suc was prepared according to a method described previously [13]. CC and Suc in a mole ratio of 3:1 was heated at 70 °C and stirred at 300 rpm. A clear liquid was obtained after 10 min of stirring. While continuously stirred, 10% v/v of water was added to the mixture. The mixture was further stirred for 20 min. CC-Suc was let to cool at room temperature and kept in a closed bottle for further use.
Extraction of C. xanthorrhiza using CC-Suc was carried out by ultrasound-assisted technique. The procedure was previously optimized regarding solid-to-liquid ratio, water content in the preparation of CC-Suc, and duration of sonication [17]. C.xanthorrhiza oil was used for extraction with CC-Suc, in order to obtain maximum content of the marker compound XTZ. In a typical preparation, 1 g of C.xanthorrhiza oil (ρ = 0.95 g/mL) was dissolved in 25 mL CC-Suc. The mixture was ultrasonicated for 20 min in an ultrasonic bath at 45 °C (40 kHz) (Bandelin Sonorex Digitec ultrasonic bath, Berlin, Germany). After extraction, the mixture was centrifuged at 7,000 rpm for 10 mins. The oil residue was removed from the liquid phase by filtration. The resulting filtrate was collected and kept in dark at room temperature until use.
For comparison, a conventional extraction of C.xanthorrhiza was conducted using ethanol. According to the Indonesia herbal pharmacopoeia, rhizome pulverized powder was macerated in ethanol (1:10) for 24 h [18]. The residue was separated and re-macerated exhaustively. The filtrate was pooled, and the solvent was removed under reduced pressure by a Buchi vacuum evaporator (Flawil, Switzerland). The resulting dry extract was stored at 4 C until use.
HPLC analysis of XTZ in CC-Suc extracts
HPLC analysis of XTZ in C. xanthorrhiza extracts was carried out by A Schambeck system with an S 9425 pump and an S 8515 vacuum degasser. The HPLC system was equipped with an S 4245 UV-Vis detector to analyze XTZ at 275 nm. Samples were run on a Zodiac C18 column (150×4.6 mm2, 5 μm) (Zodiac Life Sciences, Hyderabad, India). A gradient elution consisting of acetonitrile (eluent A) and formic acid 0.07% in water (eluent B) was used as a mobile phase at a flowrate of 0.6 mL/min at room temperature, with conditions as follows: 0 - 5 min, 45% - 50% A; 5 - 10 min, 50% - 55% A; 10 - 25 min, 55% - 60% A, 25 - 35 min, 60% - 65% A, 35 - 45 min, 65% - 70% A, 45 - 60 min, 70% - 100% A and 60 - 65 min 100% A.
Animal experiments
The study was performed after obtaining ethical approval from the Ethics Committee of the Faculty of Medicine, University of Indonesia – Cipto Mangunkusumo Hospital (Ref No. 211/UN2.F1/ETIK/PPM.00.02/2023). All animal procedures were conducted in accordance with animal welfare as stated in ARRIVE 2.0 guidelines. In this study, 28 male Balb/c mice strains were included, which were assigned into seven groups, each consisted of four mice. The number was in accordance with the resource equation approach [19], which was commonly used for preclinical animal study. The effect size (measured as partial η2) and power were analyzed for FST and fatigue related biochemical endpoints (SPSS v23), obtaining the following values, respectively: FST (0.462 and 0.931), ATP (0.599 and 0.971), AMPK (0.486 and 0.840), SUN (0.692 and 0.997), ALT (0.330 and 0.519), and AST (0.311 and 0.479). The effect size for all endpoints were > 0.140, indicating large treatment effect for the sample size (n = 4). Similarly, the observed power for most of parameter were > 0.800, except for ALT and AST obtaining moderate power, indicating adequate statistical power for the sample size (n = 4). These results indicate the large treatment effect between groups.
All mice were pathogen-free which were purchased from PT. Biofarma Breeding Facility (Bandung, Indonesia), aged 5 - 6 weeks old, weighing 20 - 30 g. Mice were treated according to the protocol guidelines of animal welfare. Mice were provided with a standard mice diet and distilled water, both ad libitum. A 12-hour light/dark cycle was maintained. The temperature (25 °C) of the animal house was kept constant. The mice bedding was changed twice weekly.
High and low dosages of NADES extract and ethanol extract of C. xanthorrhiza were selected based on a study by Kim et al. [20]. The high dosage of NADES extract (Nad-H) referred to dosing containing 25 mg XTZ/kg BW of C. xanthorrhiza oil extracted by CC-Suc, whereas the low dosage NADES (Nad-L) containing 10 mg XTZ/kg BW. Similarly, the high dosage of ethanol extract (CXM-H) referred to a dosing containing 25 mg XTZ/kg BW of C.xanthorrhiza ethanol extract, and low dosage of ethanol extract (CXM-L) containing 10 mg XTZ/kg BW.
The dosage for the positive control (TAU) was based on a previous report on antifatigue effect of TAU by Miyazaki et al. [21], i.e. 500 mg/kg BW in rats. Dosing conversion calculation to mice obtained 700 mg/kg BW. The vehicle groups, i.e. NADES alone (Nad group) and Na-CMC 1% (CMC group) received 0.5 mL/40 g BW as per guideline from IQ 3Rs (IQ Consortium, 2018).
Experimental design of antifatigue activity evaluation
The experimental protocol for antifatigue activity testing is described in Figure 1. Mice were allowed to acclimatize for two weeks prior to the experiment to familiarize themselves with the environment and food. Following acclimatization, an initial forced swimming test was carried out to determine baseline swimming time. Mice were grouped according to their BW and assigned to the following groups (n = 4): Nad-H, Nad-L, Nad, CXM-H, CXM-L, TAU, and CMC groups. The mice were treated with controls or standardized extracts once daily using oral gavage for 28 consecutive days [9]. On the last day of treatment (day-28), 30 min after treatment, a final forced swimming test was conducted. The mice were then dried and sacrificed. The muscle from rear extremities, liver, and blood from submandibular duct were collected. It is worth noting that during 28 days of treatment, no visible crystallization or phase separation was observed in the CC-Suc extract. The color and odor of the extract also remained unchanged, suggesting minimal degradation.
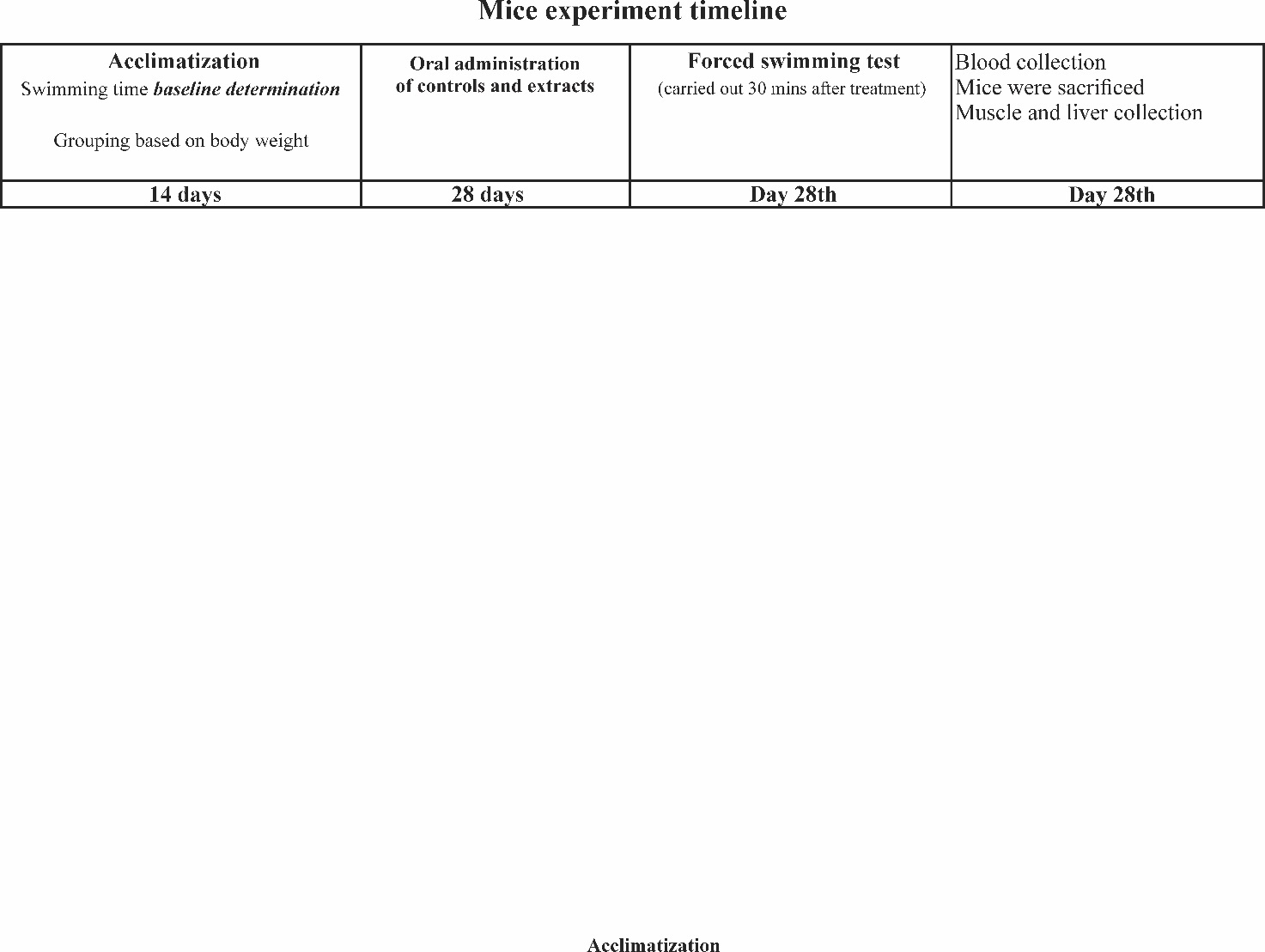
Figure 1 The timeline of mice experiment.
Forced swimming test of mice
As mentioned above, mice performed a forced swimming test initially before treatment, and finally after treatment. A forced swimming test was conducted based on previous methods [22]. The initial swimming test was performed at the end of the acclimatization period. The final swimming exercise was conducted 30 min after the last administration on day 28. Mice were placed individually in an acrylic swimming pool (20×20×40 cm3) filled with water at temperature of 30 - 35 °C at 30 cm depth. Time was recorded from the start of swimming to the immobility time for 7 s i.e. when mice lose coordinated movement, stop struggling, and only float motionless in the water, making only trivial movements necessary to keep their head above water for 7 s [10]. After swimming, the mice were dried and kept warm.
Homogenate preparation
After undergoing the final forced swimming exercise, mice were immediately anesthetized and sacrificed. Blood from the submandibular duct was collected in heparinized tubes. The serum was separated by centrifugation at 5,000 rpm for 10 min (BG-SGM-015 Biogear centrifuge) and stored at –80 °C for the measurement of serum urea nitrogen content. The liver and skeletal muscles from the rear and front legs (both dextral and sinistral extremities) were immediately excised. They were washed in a cold physiological saline solution. The liver and muscles were stored at –80 °C for further analysis. Homogenates of the liver and mouse skeletal muscle were prepared for biochemical testing as follows. After thawing, the tissue was separated from the fat. Around 200 - 300 mg tissue was dissected into small pieces, placed in a small tube, and added with 1 mL of saline water, and homogenized with a mechanical grinder (MS-100 Tomy micro smash). After centrifugation at 4 °C, for 5 min at 50×100 G2 (MX-307 Tomy microcentrifuge), the supernatant was taken out and stored as aliquots at –20 °C for further testing.
Measurement of ATP Metabolisms and fatigue-related metabolites in mice
Muscle homogenate was tested for ATP level using an ATP colorimetric assay kit (Elabscience) and for AMPK level using an AMP-Activated Protein Kinase ELISA kit (Bioenzy). SUN content was determined using a urea (BUN) colorimetric assay kit (Elabscience, Texas, USA). The determination was conducted according to the manufacturer’s instructions. The liver homogenate was tested for ALT and AST by ALT and AST activity ELISA assay kits (Elabscience). Procedures provided by the manufacturer were followed.
Statistical analysis
Data were processed using SPSS v25.0 (IBM Statistics) and were shown as mean ± SD (n = 4). The acquired data were analyzed for normality and homogeneity of variance by the Shapiro-Wilk and the Levine tests, respectively. For normal and homogenous data, the significant differences between groups were analyzed using One-way ANOVA, followed by Tukey’s Post Hoc test. Non-normal data were analyzed by the Kruskal-Wallis test, followed by the Dunn multiple comparison Post Hoc test to analyze the significance of differences. Meanwhile, for normal but non-homogenous data, the Welch test was used, followed by the Games-Howell Post Hoc test for multiple comparisons. p-value < 0.05 was considered significant.
Results and discussion
Content of XTZ in CC-Suc Extract of C.xanthorrhiza
XTZ content in the extracts was used to calculate the dosage of extract treatment. Analysis of XTZ is also required to confirm the quality of CC-Suc extracts of C. xanthorrhiza. Low and high dosages of 10 and 25 mg XTZ/kgBW, respectively, were selected based on a previous study by Kim et al. [20]. For this purpose, extracts were quantified for the content of XTZ by the HPLC analysis method described previously [13].
Figure 2 shows the chromatogram of XTZ analysis in NADES extract. When detected at 275 nm, the chromatograms of CC-Suc extract and ethanol extract exhibited XTZ peak (peak 4) at retention times of 44.76 and 43.54 min, respectively, similar to that of the reference standard XTZ. BDMC, DMC, and Cur were also identified at 275 nm, and appeared as peaks 1, 2, and 3, respectively. XTZ content was calculated by the calibration curve of XTZ (y=4.42x + 9.0517) [13].
This result highlights the ability of CC-Suc to extract important markers of C. xanthorrhiza. Recent reviews reported that NADES including choline chloride and sugar systems improve extraction of bioactive compounds and can act as biocompatible delivery media [23].
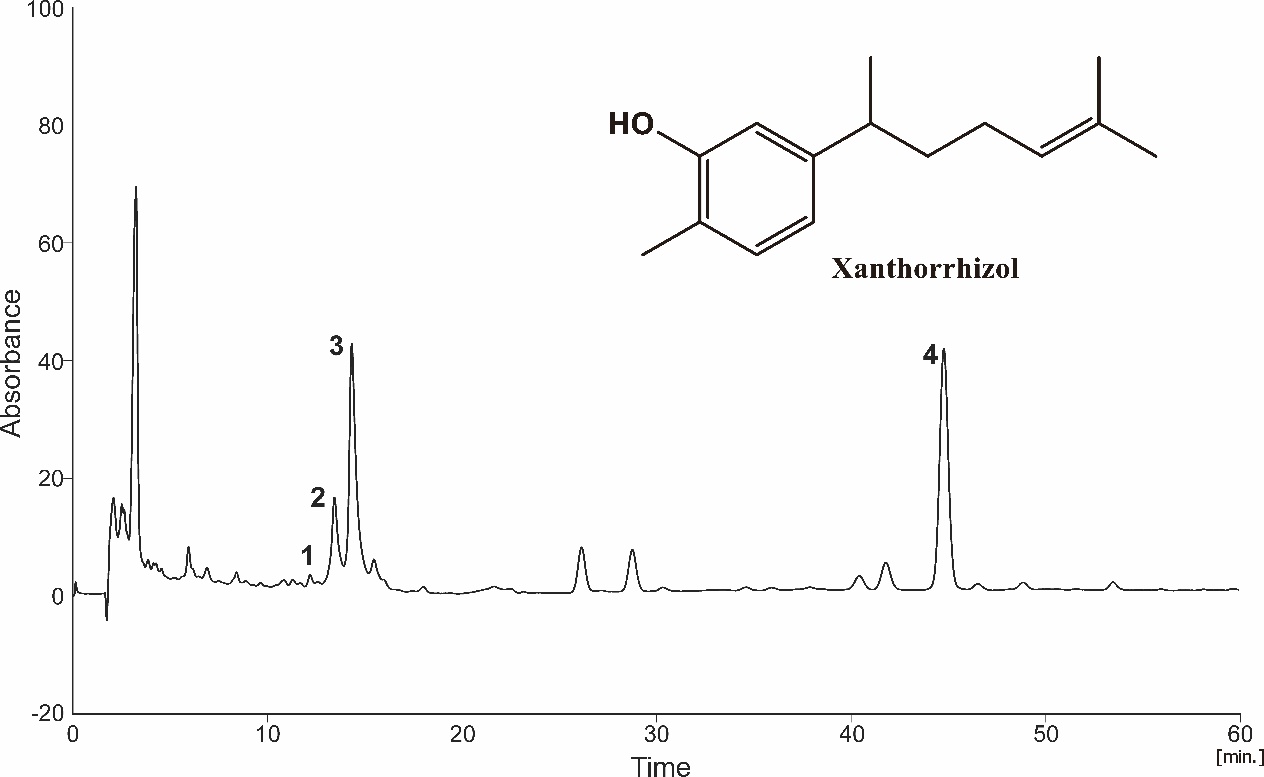
Figure 2 The chromatogram of (CC-Suc extract of C.xanthorrhiza oil recorded at 275 nm, showing (1) bisdemethoxycurcumin (BDMC), (2) demethoxycurcumin (DMC), (3) curcumin (Cur), and (4) xanthorrhizol (XTZ), at retention times of 12.20, 13.44, 14.34, and 44.76, respectively.
Effect of CC-Suc extract of C. xanthorrhiza on mice body weight
In this study, mouse body weight (BW) was recorded regularly once a week, starting from one week prior to treatment (Week 0), and then every week during the experimental period (Week 1, 2, 3, and 4). Mice BW was scaled during treatment because changes in BW may reflect the effect of treatment samples on the health status and appetite of mice [24]. In addition, regular check on BW was required for adjustment of dosing volume of the samples given to mice. BW data was tabulated and statistically analyzed (SPSS v25) to evaluate the differences between groups on each week of treatment.
Figure 3 displays changes in the weekly average of mouse body weight during experimental weeks. In general, the BW of mice gradually increased following continuous administration of C. xanthorrhiza extracts. Analysis of BW further evaluated the significance of BW changes during treatment.
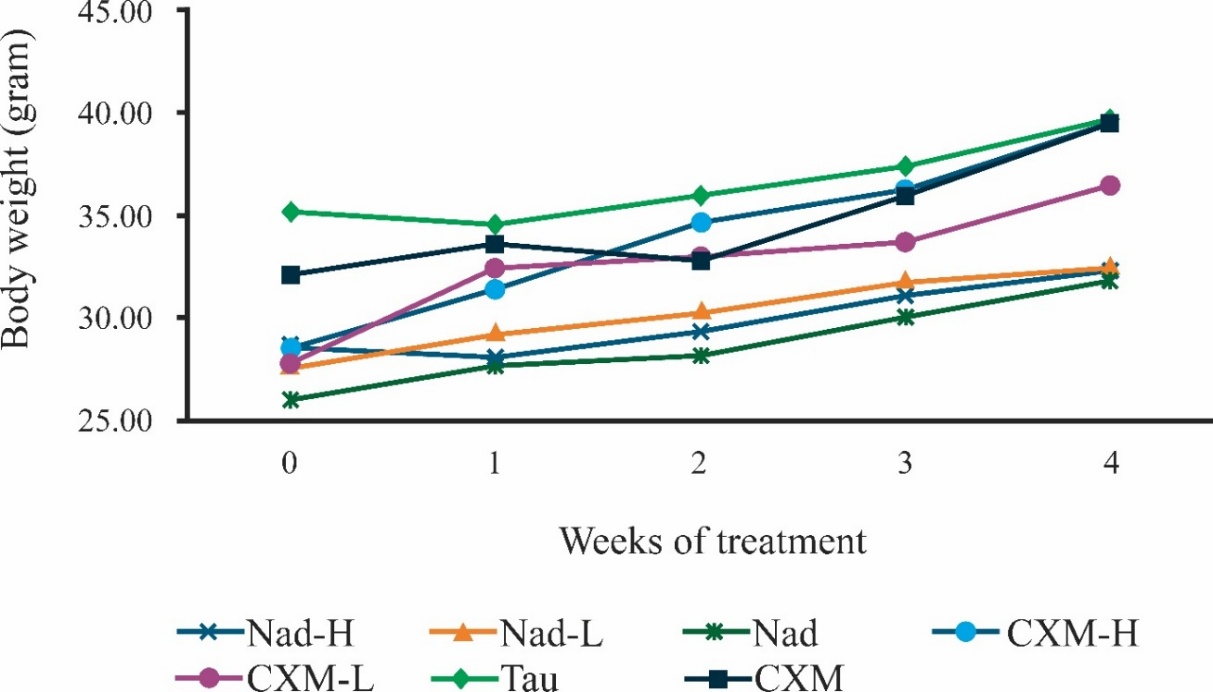
Figure 3 Body weight changes in mice over 28 days of extract administration. Week 0 was mice body weight recorded on the week before treatment, whereas weeks 1, 2, 3, and 4 were body weight measured on the following weeks during treatment. Nad-H, Nad-L, Nad, CXM-H, CXM-L, TAU, and CMC refer to mice treated with, respectively, high dose of NADES extract, low dose of NADES extract, NADES only, high dose of ethanol extract, low dose of ethanol extract, taurine, and CMC (Na-CMC, 1%). Data points represent the mean of four mice.
One-way ANOVA was conducted to evaluate the differences between groups and found that BW was significantly different between groups on all weeks (weeks 0, 1, 2, and 4, with p-values of 0.005, 0.029, 0.026, and 0.040, respectively), except for BW on week 3 (p-value = 0.125). It could be that mice were nearing growth plateau on week 3, thus their weight difference was less pronounced. Close observation of mice BW on week 0 showed that the average BW for mice of groups Nad-H, Nad-L, Nad, CXM-H, and CXM-L were 28.60 ± 2.35, 27.70 ± 0.77, 26.06 ± 1.42, 28.56 ± 4.10, and 27.75 ± 5.38 g, suggesting similar BW. Meanwhile, the BW of groups TAU and CMC were 35.18 ± 1.63 and 32.12 ± 2.29 g. Mice were allowed to access food and drink freely (ad libitum). This condition led to mice gaining weight significantly throughout the experiment period. However, close observation of mice BW on the last week of treatment (week 4) shows that mice in Nad-H, Nad-L, and Nad groups had similar BW i.e. 32.24 ± 3.62, 32.36 ± 2.81, and 31.71 ± 4.34 g, respectively. Meanwhile, similar BW were also observed for mice in CXM-H, TAU, and CMC groups, i.e. 39.52 ± 2.89, 39.70 ± 2.52, and 39.47 ± 8.91 g. Mice in CXM-L obtained 36.52 ± 3.02 g by week 4 of treatment.
3.3. Effect of CC-Suc Extract of C. xanthorrhiza on mice swimming time
Endurance exercise reflects the antifatigue ability of the body [24]. The ability to improve exercise endurance is a strong manifestation of antifatigue effect of drugs. In this case, the length of swimming time may reflect the fatigue degree of animals. In this experiment, a forced swimming test (FST) was used to evaluate the antifatigue effect of C.xanthorrhiza extract. FST using mouse or rat models has been widely used for the evaluation of the antifatigue effect of novel compounds [25,26]. In the FST experiment, time was recorded from the start of swimming to the immobility time for 7 s i.e. when mice lost coordinated movement, stopped to struggle, floated motionless in the water, making only movements necessary to keep their head above water for 7 s [25].
Force swimming test (FST) was conducted twice for every group. Prior to sample treatment, initial FST was conducted, in order to record baseline swimming time for each mouse. In addition to initial FST, final FST was conducted on the last day (on day 28 of treatment), 30 min after treatment. Analysis was performed for the initial FST, final FST, and pair-wise comparison between the initial and final FST for each group. Values of p-value < 0.05 were considered significant (IBM SPSS v25).
Figure 4 shows comparisons of the average initial and final swimming time for every group. For the average initial swimming time, the data fulfilled the assumptions of normal and homogenous data (p-value = 0.152 and p-value = 0.159, respectively). One-way ANOVA test obtained no significant differences between groups (p-value = 0.427), indicating that mice had similar swimming performance before treatment.
Analysis
was also conducted for the average final swimming time for every
group. It is clear that treatment with C.
xanthorrhiza
extracts (both NADES and ethanol extracts) succeeded in prolonging
swimming time when compared to the negative control CMC group. A P
value = 0.006 by the non-parametric Kruskal-Wallis test was
calculated, indicating significant differences in the final FST time
between groups. Moreover, there was a trend, in the length of
swimming time with the doses of extracts administered, in both NADES
and ethanol extracts. In NADES extracts, increases by around 6
times, 3.8 times, and 0.7 times longer in swimming time were noted
in Nad-H, Nad-L, and Nad groups compared to the CMC group. A P
value = 0.020 by the Dunn
Post Hoc test was calculated for the
difference of
Nad-H – CMC. Meanwhile, treatment with ethanol
extracts obtained increased swimming time by 2.8 times and 2.2 times
when compared to the swimming time of the CMC group. As expected,
treatment with TAU significantly increased swimming time (p-value
= 0.025 by the Dunn Post Hoc test) with an increase of 6.7 times
compared to the CMC-treated group. By comparison, the final swimming
time of Nad-H-treated group was not significantly different with
TAU-treated group.
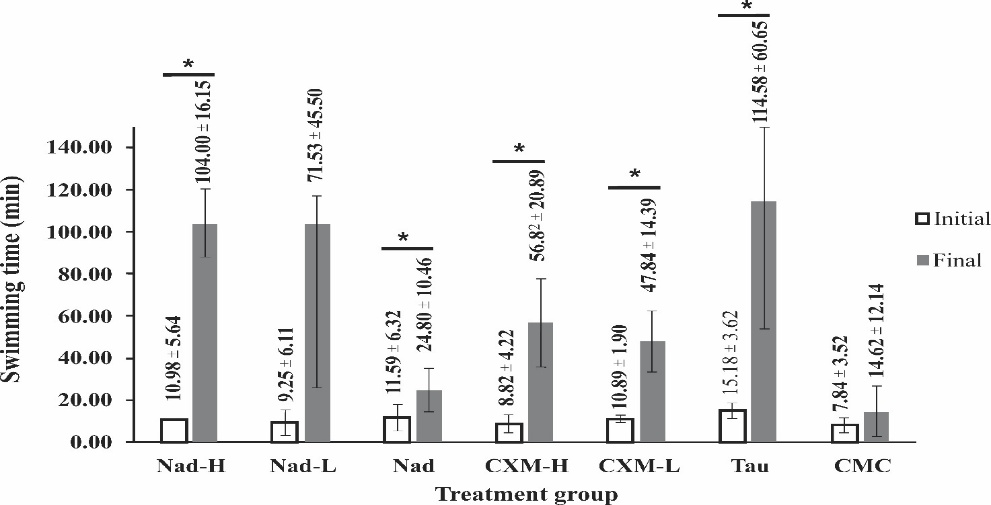
Figure 4 Pairwise comparison of before and after treatment for each group. Data mean ±SD (n = 4). Nad-H in mice treated with a high dose of NADES extract, Nad-L is a low dose of NADES extract, Nad is NADES only, CXM-H is a high dose of ethanol extract, CXM-L is a low dose of ethanol extract, TAU is taurine, and CMC is Na-CMC (1%). The sign * indicates a significant difference at p-value < 0.05 (paired t-test).
As
seen in Figure
4,
the administration of mice with Nad-H, Nad, CXM-H, CXM-L, and TAU
for 28 days could significantly extend the swimming time of mice
(p-value
= 0.00, p-value
= 0.03,
p-value
= 0.03,
p-value
= 0.01, and p-value
= 0.05, respectively) compared to before treatment. These findings
indicate that treatment with these samples produced pharmacological
effects in increasing tolerance to fatigue conditions. The TAU
effect is as expected in increasing exercise tolerance. Previously,
TAU was reported to increase exercise time in rats in a
dose-dependent manner [21]. In this report, TAU was administered to
rats with an equivalent dose conversion given to mice (500 mg/kg BW
rats) and reached running time on a treadmill by 115 min. Nowadays,
TAU is widely recognized as a supplement given to increase exercise
performance among athletes [27]. The antifatigue effect observed for
Nad-H, CXM-H, and CXM-L indicates that C.
xanthorrhiza
extract rich with XTZ may have caused the antifatigue activity. The
results may also indicate that the antifatigue effect of Nad-H is
not only caused by bioactive compounds extracted by NADES but may
also be sourced from the NADES components. It is likely that
administration of NADES 30 min before final swimming may increase
glucose blood level. Sucrose in NADES is hydrolyzed into glucose and
fructose. In the cellular level, glucose is the entry substrate to
the TCA cycle, and subsequently to electron transport chain in the
production of ATP. Previously, Gatward et
al.
found that elevation by 40% in blood glucose levels in mice were
observed within 60 min after glucose administration [28]. It is
essential that the effect of sucrose supplementation be included in
future study to confirm its contribution to energy metabolism.
Different cases were observed for group of Nad-L administration. Although an increase in swimming time can be noticed following administration with Nad-L for four weeks (Figure 4), the increase in swimming time was of no significant difference (p-value = 0.06). This result may likely to be due to one or more mice in this group which were weaker than others.
Taken together, the result indicated that C. xanthorrhiza extract administration could enhance exercise endurance through prolonged forced swimming time and the anti-fatigue ability of mice.
Effect of CC-Suc extract of C.xanthorrhiza on energy metabolism and fatigue-related markers
Effect of CC-Suc Extract of C.xanthorrhiza on ATP level in mice muscles
In the present study, muscle ATP levels were determined and compared, following a forced swimming test. For this purpose, mice were sacrificed after 28 days of treatment, and limb muscles were homogenized to obtain the homogenate for ATP testing.
As shown in Figure 5, treatment with C. xanthorrhiza extracts elevated the level of ATP in the skeletal muscle of mice. After 28 days of treatment with TAU, the positive control group was found to have a significant increase in skeletal ATP level, by 51.27% compared to CMC group (p-value = 0.02). In the NADES extracts groups, a trend of dose-dependent response was observed for groups treated with Nad-H, Nad-L, and Nad, with increases of 60.00%, 25.50%, and 12.00%, respectively for Nad-H, Nad-L, and Nad. When compared with CMC-treated group, treatment with Nad-H resulted in a significant increment in ATP level (p-value = 0.004). A similar trend of dose-dependent response was also noted in CXM-H and CXM-L-treated groups, although no significant difference was obtained. In this case, increases of 38.90% and 16%, respectively for CXM-H and CXM-L were noted. CMC treated group shows the lowest level of ATP after forced swimming test compared to other treatment groups. No significant difference was observed when CMC group was compared with Nad group (p-value = 0.974, by a Post hoc Tukey test), suggesting that at administered dose, sucrose containing-NADES did not exert increase in muscle AMPK. It is important to note that the ATP levels due to treatment with Nad-H and CXM-H were not significantly different with treatment with TAU (p-value = 0.99 and p-value = 0.97, respectively). It was noted that when compared with CXM-H, treatment with Nad-H resulted in a higher level of ATP by 13.18%, although no significant difference was observed.
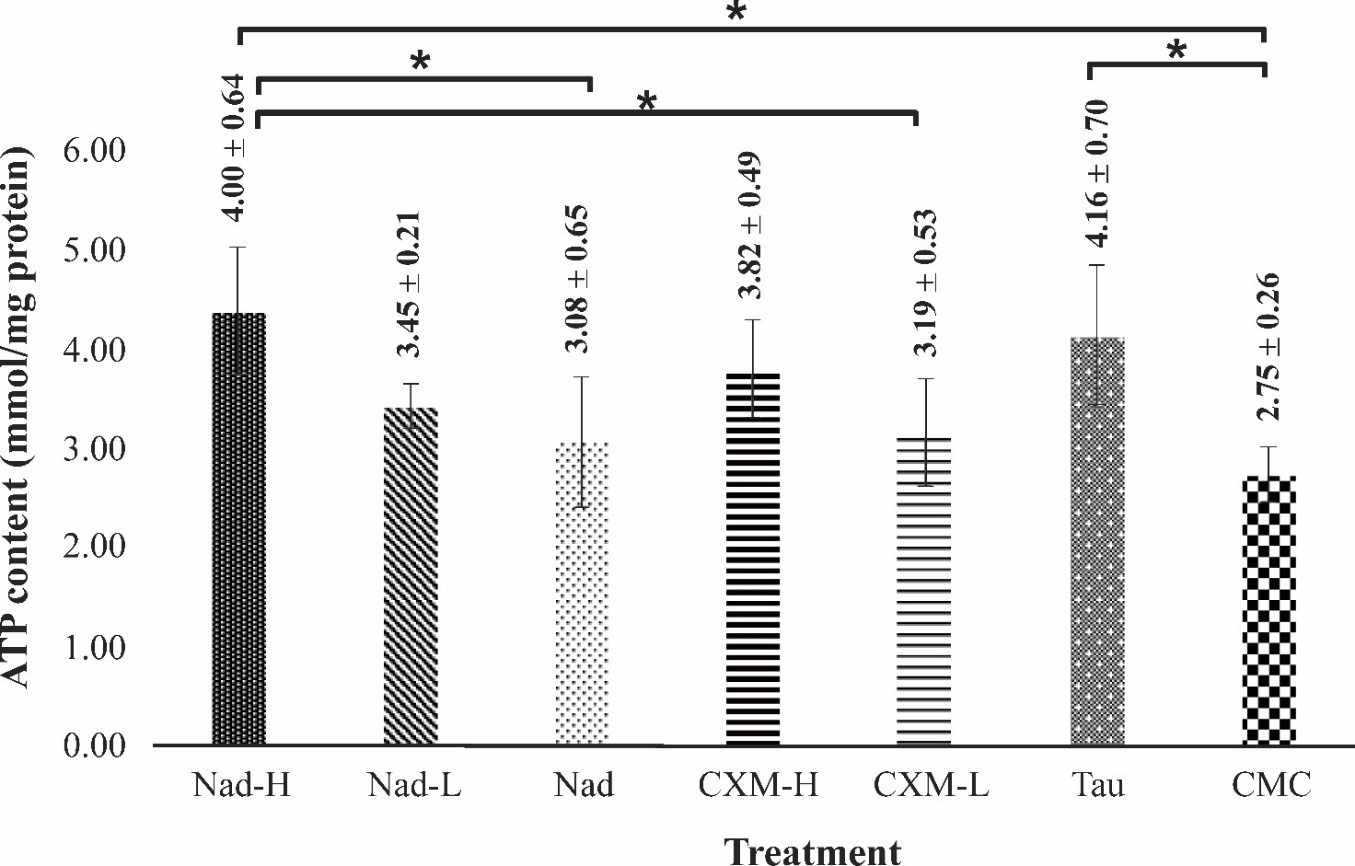
Figure 5 Effect of administration of C. xanthorrhiza extracts on ATP level in mice muscle. The measurements were taken following forced swimming tests, after 28 days of extract treatment. Data are presented as mean ± SD (n = 4). Nad-H is mice treated with a high dose of NADES extract, Nad-L is a low dose of NADES extract, Nad is NADES only, CXM-H is high dose of ethanol extract, CXM-L is a low dose of ethanol extract, TAU is taurine, and CMC is Na-CMC (1%). The sign * denotes a significant difference at p-value < 0.05 (ANOVA with a post hoc Tukey test).
ATP is a rapid energy source, by which chemical energy is released through hydrolysis of phosphoanhydride bonds in ATP into subsequent ADP, AMP, and P. Insufficient ATP in the muscle during exercise leads to physical fatigue, therefore ATP levels in muscle may reflect fatigue degree. High levels of ATP indicate the availability of energy to sustain physical activity, thus relieving fatigue. In the present study, fatigue muscle was signified by low ATP level in the muscle, as seen in the negative control group. This was in contrast with the increased ATP level in the TAU group. TAU was previously reported to enhance ATP levels in rats by maintaining mitochondrial enzyme activities involved in the electron transport chain, including complexes I, II, III [29]. Furthermore, it was reported that TAU increased ATP production by conjugating to NADH-ubiquinone oxidoreductase chain 6 (ND6), a subunit in complex I that is required for maximal complex I activity [30]. In the present study, it was found that TAU treatment induced an increase in ATP levels.
The association between increased ATP content in muscle and increased exercise performance was reported previously by other researchers [31]. It should be highlighted that the ATP levels were increased in all C. xanthorrhiza-treated groups compared with the CMC control group. These results supported findings in the swimming test, whereby treatment with C. xanthorrhiza extracts (NADES and ethanol extracts) both succeeded in increasing swimming time. Similar to the results observed of the final swimming test, a trend of dose-dependent response was observed, not only in NADES extract supplementations but also in ethanol extract treatment. Comparisons among treatment groups showed that Nad-H-treated group obtained the highest level of ATP, in accordance with the result obtained in the final forced swimming test, by which the longest physical endurance was observed for the Nad-H group (Figure 4). Nad-L group exerted lower ATP level than Nad-H. It is worth noting that NADES supplementation (CC-Suc alone) elevated ATP content in muscle as well. The dose dependent trend observed for Nad-H > Nad-L > Nad indicates that ATP increase cannot be solely attributed to bioactive compounds extracted by NADES, but NADES components may also contribute to the activity. Hydrolysis of sucrose produces fructose and glucose; the latter enhance energy availability during exercise.
Taken together, the above results further demonstrate the antifatigue effect of C. xanthorrhiza extracts (both NADES and ethanol extracts). C. xanthorrhiza extracts could improve energy metabolism in mice’s skeletal muscle and therefore could increase exercise endurance.
Effect of CC-Suc Extract of C.xanthorrhiza on AMPK level in mice muscles
In this study, the total AMPK level in fatigued mice muscle was determined to evaluate the effect of C. xanthorrhiza supplementation on improving the fatigue condition of mice. Figure 6 shows the levels of AMPK in the skeletal muscle of different groups after 28 days of treatment, followed by a forced swimming test. As compared to the case of the CMC group, treatment with NADES extracts and NADES resulted in increases in AMPK levels in a dose-dependent trend, by 46.62%, 36.52%, and 30.33% respectively for Nad-H, Nad-L, and Nad. Upon treatment with CXM-H, an increment of 25.28% in the AMPK level was observed compared to the CMC group. In contrast, CXM-L treatment did not significantly increase the AMPK level. In this case, treatment with CXM-L resulted in a significantly lower level of AMPK compared to the TAU group (p-value = 0.02). On the other hand, TAU administration showed a significantly higher AMPK level (by 62.36%) when compared to the negative control (CMC group) (p-value = 0.04). By a Post Hoc Tukey test, treatment with Nad-H, Nad-L, Nad, and CXM-H gave no significant difference in AMPK increases compared to TAU-group (Tukey Post Hoc test, p-value = 0.96, p-value = 0.72, p-value = 0.50, p-value = 0.33, respectively).
AMPK is a key mediator of cell signaling pathways associated with muscle function and energy metabolism [32]. AMPK is activated by increased ratios of AMP/ATP and/or ADP/ATP. Its activation plays an important role in regulating ATP metabolism in muscle during exercise [33]. The total AMPK level therefore is a measure of fatigue state in mice.
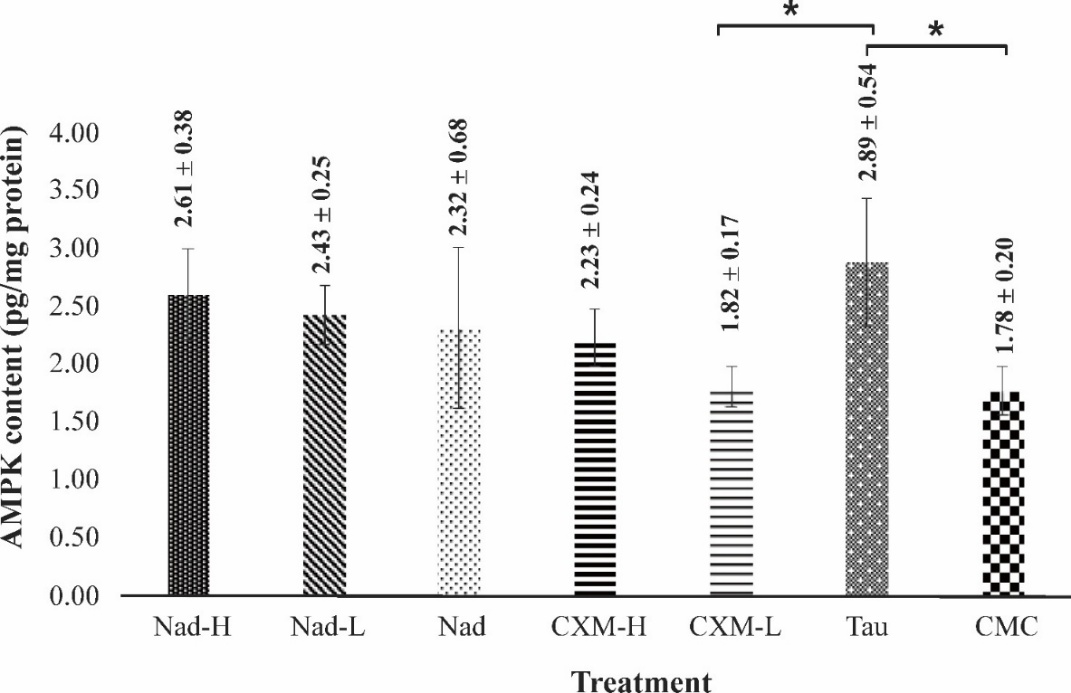
Figure 6 Effect of administration of C. xanthorrhiza extracts on AMPK levels in mice muscle. The measurements were taken following a forced swimming test, after 28 days of extract treatment. Data are presented as mean ± SD (n = 4). Nad-H in mice treated with a high dose of NADES extract, Nad-L is a low dose of NADES extract, Nad is NADES only, CXM-H is a high dose of ethanol extract, CXM-L is a low dose of ethanol extract, TAU is taurine, and CMC is Na-CMC (1%). The sign * denotes a significant difference at p-value < 0.05 (ANOVA with a Post hoc Tukey test).
Skeletal muscle shows high adaptability to drastic energy turnover during exercise and rest. AMPK is an important
signaling molecule that functions as a sensor for intracellular energy status [33]. Studies showed that AMPK regulates energy metabolism in skeletal muscle at several levels. Upon activation, AMPK increases ATP production, mainly through stimulation of glycolysis. However, previous studies showed that reduced activity of AMPK impaired the absorption of glucose in skeletal muscle, accelerated ATP degradation, and impaired mitochondrial function by disrupting Complexes I and IV of the electron transport chain [34]. A study reported that a lack of skeletal muscle AMPK reduced the effect of increased ADP to stimulate mitochondrial oxidative phosphorylation [35]. Further, AMPK stimulates peroxisome proliferator-activated receptor-gamma coactivator 1alpha (PGC-1 ), which regulates genes of glucose transport protein-4 (GLUT4) [4]. With different metabolic regulatory functions as mentioned above, AMPK levels in skeletal muscle reflect the ability of skeletal muscle to energetic adaptation in energy stress conditions such as during exercise.
In general, the present study found that treatments with C. xanthorrhiza extracts, in particular, Nad-H, Nad-L, Nad, and CXM-H increased AMPK levels in those groups, like that observed in the TAU-treated group. Kim et al. reported that, after 30 days of TAU administration, the level of phosphorylated-AMPK (an activated form of AMPK) increased in mice challenged with a forced swimming test [36]. The resulting prolonged swimming was noted in this report. The present study found an increment in the unphosphorylated AMPK level due to TAU administration. Increase in unphosphorylated AMPK level may reflect cell capacity or readiness to respond to energy stress. It is known that AMPK is activated by rises in AMP/ADP ratio. In the case of CMC group, this group showed the lowest level of AMPK when compared to other treatment groups, which may reflect fatigue state in the CMC group.
These results herein may suggest that C. xanthorrhiza extracts (NADES extract and ethanol extract) delay fatigue by modulating AMPK. The determination of AMPK levels supported that treatment with the extracts increases the ATP levels in mice muscle (Figure 5), which is likely associated with the increase in AMPK level. However, detailed mechanisms on the regulation of C. xanthorrhiza extract supplementation on the AMPK signal pathway are not clear and need further study.
Effect of CC-Suc extract of C. xanthorrhiza on serum urea nitrogen (SUN)
In the previous section, the study has proven the beneficial effect of C. xanthorrhiza extracts on prolonging swimming time after 28 days of C. xanthorrhiza extracts supplementation. The fatigue state induced by forced swimming exercise causes biochemical changes in response to the fatigue condition. Accumulation of biochemical-related parameters such as urea nitrogen, ALT and AST are indicators associated with body fatigue.
Figure 7 shows SUN content in mice after a forced swimming test, following treatment for 28 days with C. xanthorrhiza extracts.
Heavy and long exercises such as in forced swimming tests induced the formation of serum urea nitrogen. The vehicle (CMC) group reflected this condition as identified by high serum urea nitrogen content. However, the forced swimming test performed by mice in the TAU-treated group resulted in a significantly lower SUN level by 21.56%, compared to the negative control CMC group (p-value = 0.02).
In
cases of extract-treated groups, treatment with NADES extracts and
ethanol extracts, except for CXM-L, resulted in lower values of SUN
compared to the CMC group. It was noted that Nad-H supplementation
caused a significantly lower SUN value of 28.31% than the CMC group
(p-value
= 0.00). Meanwhile, Nad-L treatment caused a similar SUN lowering
effect as Nad-H by 28.68%. Treatment with Nad resulted in lower
value by 22.48% than CMC group. CXM-H treatment decreased SUN level
by 21.29%. In contrast, CXM-L had no effect on lower SUN levels. It
should be noted that the SUN-lowering effect of Nad-H, Nad-L, and
CXM-H was not significantly different from the effect exerted by TAU
supplementation (p-value
= 0.42,
p-value
= 0.38
p-value
= 0.99, respectively). This suggests that extracts used in this
study, and TAU possessed similar ability in removing SUN. It was
also noted that there was no significant difference between Nad
group and CMC group (p-value
=
0.073, by a Post hoc Tukey test).
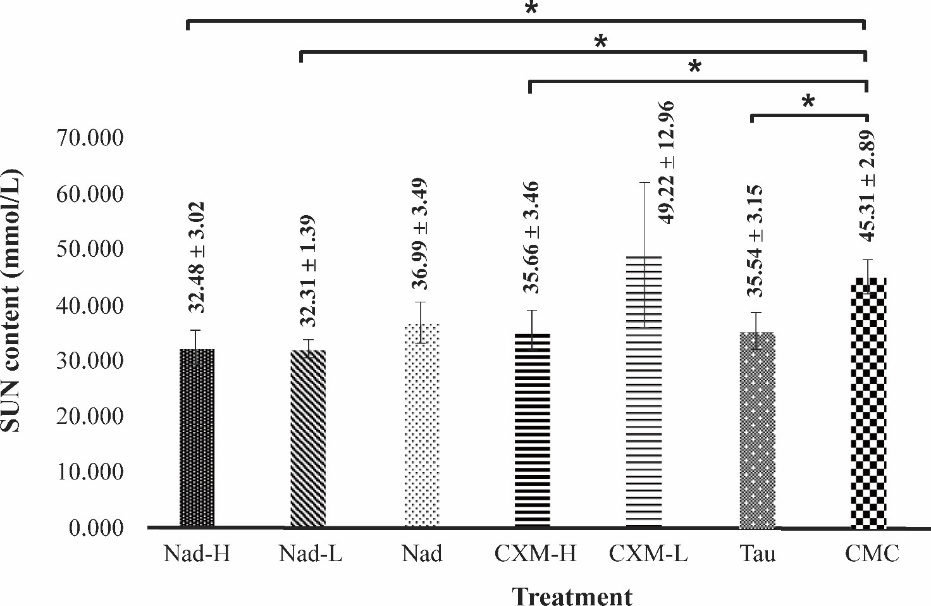
Figure 7 Effect of C. xanthorrhiza extracts on SUN levels in mice after forced swimming test. Data are taken after 28 days of extract treatment and presented as mean ± SD (n = 4). Nad: NADES extract of C. xanthorrhiza, CXM: C. xanthorrhiza maceration extracts, TAU: Taurine, CMC: Na- carboxymethyl cellulose. The sign * denotes a significant difference (p-value < 0.05, ANOVA with a post hoc Tukey test).
Fatigue condition following muscle exercise can be evaluated by several biochemical markers, including blood or serum urea nitrogen (SUN) [37]. Previous study have reported that TAU, the positive control used in this study, possessed the ability to lower the formation of blood urea nitrogen after exercise in mice [36]. Recently, curcumin, a marker compound found in C. xanthorrhiza was reported to improve physical performance and physiological fatigue, including lowering BUN levels in mice [37]. In another study, the SUN-lowering activity was shown by xanthorrhizol which is an important marker of C. xanthorrhiza. Pre-treatment with xanthorrhizol led to a lower level of urea nitrogen in cisplatin-induced rats [38]. In the current study, it was demonstrated that C. xanthorrhiza extracts could improve swimming performance by lowering SUN levels.
SUN is an important indicator of protein metabolism levels. SUN can reflect the fatigue status of working muscles. In intense working muscle conditions, in which energy produced from carbohydrate and lipid metabolisms is not sufficient to fulfill energy needs, protein is catabolized to provide energy for the muscle [4]. Various reports confirm increases in blood ammonia levels after exercise [39]. Physiologically, ammonia is produced in large amounts in muscle during strenuous muscle activity. The immediate source of ammonia in the muscle and blood is the deamination of AMP (adenosine monophosphate) into IMP (inosine monophosphate) by AMP deaminase. The utilization of ATP during intense exercise increases the supply of its degradation products (ADP and AMP), causing more formation of ammonia [40]. During strenuous exercise, the body requires a large quantity of energy. The energy derived from the catabolism of carbohydrates and fat can provide energy within 30 min of performing exercise. However, at longer exercise duration, catabolism of protein and amino acid is activated to compensate for energy consumption. Accumulation of ammonia in the blood and muscle causes a deleterious effect on the nervous system. Temporary detoxification of ammonia by glutamine prevents the toxic effect before the final route of detoxification via urea synthesis in the liver. Therefore, accumulation of urea nitrogen in the blood is a sensitive marker of fatigue and reflects the ATP metabolism.
Taken together, results herein suggest the beneficial effect of C. xanthorrhiza extracts administration in attenuating SUN levels in fatigue mice, similar to TAU. The removal of this fatigue marker subsequently increases fatigue tolerance, as noted by increases in swimming time.
Effect of CC-Suc extract of C. xanthorrhiza on ALT and AST
The evaluation of hepatic ALT and AST is crucial to study the effect of strenuous exercise on the liver function. In addition, these measurements can also assess the physiological changes due to the long treatment of C. xanthorrhiza extracts. Figures 8 and 9 show the effect of extracts administration on hepatic ALT and AST levels, after the forced swimming test.
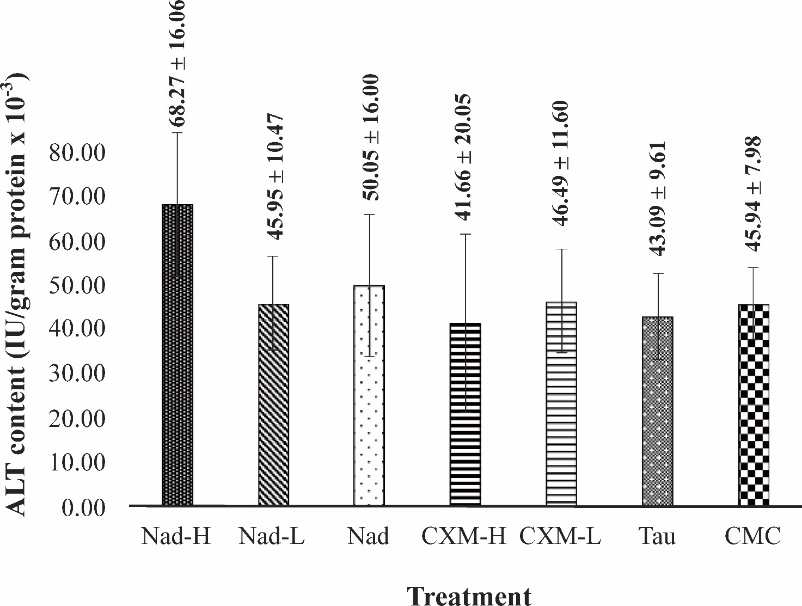
Figure
8
Effect of C.
xanthorrhiza
extracts on hepatic ALT levels in mice after forced swimming test.
Data are taken after 28 days of extract treatment and presented as
mean ± SD (n = 4). Nad: NADES extract of C.
xanthorrhiza,
CXM:
C.
xanthorrhiza
maceration extracts, TAU: Taurine, CMC: Na- carboxymethyl cellulose,
(no significant differences among groups, p-value
= 0.165, ANOVA).
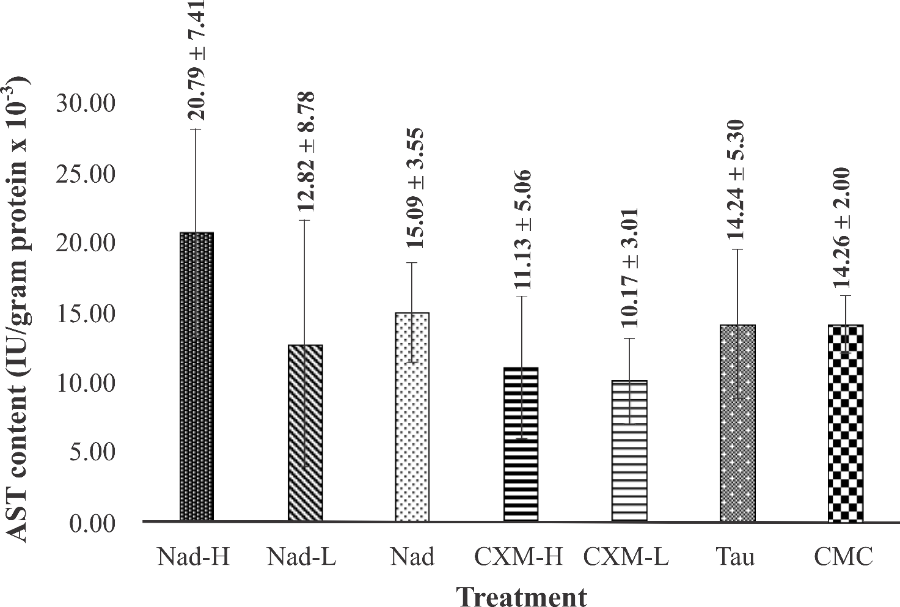
Figure 9 Effect of C. xanthorrhiza extracts on hepatic AST levels in mice after forced swimming test. Data are taken after 28 days of extracts treatment and presented as mean ± SD (n = 4). Nad: NADES extract of C.xanthorrhiza, CXM: C.xanthorrhiza maceration extracts, TAU: Taurine, CMC: Na- carboxymethyl cellulose (no significant differences among groups, p-value = 0.20, ANOVA).
As seen in Figures 8 and 9, fatigue condition induced by forced swimming test did not elevate liver ALT and AST levels in the vehicle (CMC) group, indicating that strenuous exercise did not cause liver injury. The same result was observed in mice treated with TAU. Administration of TAU for 28 consecutive days followed by exhaustive swimming did not significantly change the liver ALT and AST levels. No significant increase was observed in mice treated with Nad-L, Nad, CXM-H, and CXM-L (p-value = 165), following forced swimming test. In the case of mice treated with Nad-H, although forced swimming was seen to raise the hepatic ALT and AST levels, the increase was not significantly different from those observed for TAU and other C. xanthorrhiza extracts (p-value = 0.20).
ALT and AST are sensitive predictors of hepatocyte injury due to strenuous physical activity [41]. Intense exercise can lead to the contraction of liver cells, thus elevation in hepatic ALT and AST values. In addition, ALT and AST are important biochemical markers for assessing toxicity of drugs on the liver [42]. Based on the result in CMC-group, intense exercise did not cause liver injury in fatigue mice. Meanwhile, treatment with TAU for 28 consecutive days followed by forced swimming test did not elevate liver ALT and AST levels in the same way as in CMC group. This result indicates that TAU is nontoxic to the liver. Similarly, treatment with Nad-H, Nad-L, Nad, CXM-H, and CXM-L for 28 consecutive days did not increase ALT and AST levels, suggesting that the continuous treatment did not cause a deleterious effect on the liver.
TAU has been widely reported for its antifatigue effect and has been generally considered safe [43]. A previous study showed TAU treatment did not elevate hepatic ALT and AST levels in exercise muscles [44], similar to those observed in the current study.
In cases of heavy physical activity, previous studies have reported that forced swimming exercise in mice injured the liver, as shown by elevated values of hepatic ALT and AST [22]. However, the current study found that treatment with C. xanthorrhiza extracts did not increase liver ALT and AST. Traditionally, C. xanthorrhiza has long been used to treat liver disorders. C. xanthorrhiza extract was previously reported to have a protective effect on the liver. Pre-treatment with the extract prevents the rise of the ALT and AST levels in mice and rats induced by CCl4 dan ethanol [45]. XTZ was also reported to protect the liver when pretreated to cisplatin-induced hepatotoxicity in mice. Concerning curcumin, it was reported that curcumin treatment reduced liver ALT and AST in fatigue induced mice [22]. The results herein serve as initial screening for potential liver injury due to repeated treatment of extracts and exercise-related damage. However, comprehensive subacute safety of C. xanthorrhiza and histopathological examination of the liver and kidney are necessary to confirm safety.
Comparison and proposed mechanimsms
As summarized in Table 1, treatment with C. xanthorrhiza extracts (NADES and ethanol extracts) prolonged the swimming time of mice compared with the CMC group, confirming their antifatigue activity. A dose dependent trend was observed in both NADES and ethanol extracts. However, stronger effect was observed in NADES extract treated groups compared to ethanol extract groups.
Among all groups, the NADES extract at high dose (Nad-H) produced the longest swimming time, comparable to the positive control TAU, and greater than the ethanol extract at the same xanthorrhizol dose (CXM-H). A dose-dependent trend was observed for NADES extracts (Nad-H > Nad-L > Nad), suggesting that the NADES vehicle contributed modestly to performance enhancement, likely due to sucrose component in NADES.
Consistent with the improved physical endurance, Nad-H treatment yielded the highest muscle ATP and AMPK levels, similar to TAU, and exceeded those of CXM-H. Conversely, serum urea nitrogen (SUN), a fatigue marker, was markedly reduced in all extract-treated groups, with the lowest levels observed in TAU and Nad-H mice. Hepatic ALT and AST levels remained within normal range across all groups, indicating no liver toxicity.
Overall, the NADES extract (Nad-H) showed more efficacy than the ethanol extract by producing greater endurance performance, enhanced energy metabolism (ATP and AMPK), and lower fatigue biomarkers (SUN) without adverse hepatic effects.
It should be noted, however, several limitations of the present study. Firstly, the sample size for each group was relatively small that may limit generalizability. Within the constraints of our study, the detected antifatigue effects are statistically reliable for the main pharmacological endpoint, as seen by the post hoc effect size and power values. However, the small sample size limits the representativeness to the populations. Secondly, only male mice were included in the study, thus potential sex-related metabolic and hormonal differences cannot be addressed. It is known that female mice utilize skeletal muscle lipid as energy source during exercise greater than male mice [46]. However, recent study showed that male and female mice gave similar response in the activation of muscle AMPK [47]. Estrous cycle in female mice may cause variability in fatigue and treatment response, although a meta-analyses study concluded that female mice are not inherently more variable than males [48]. Nevertheless, it is important for future studies to include female mice in antifatigue study, to investigate whether fatigue resistance differs by sex. Thirdly, long term safety evaluation is necessary to confirm non-toxicity of NADES extract and thus should be included in future study.
To the best of our knowledge, the present study is the first report on the antifatigue activity of C. xanthorrhiza, While C. xanthorrhiza is known for its antioxidant and hepatoprotective properties [49], its role in improving physical endurance and energy metabolism has not been previously explored. In addition, the comparative evaluation between NADES and ethanol extracts highlights the potency of the NADES system. NADES CC-Suc can serve dual roles, i.e. as extraction media for important markers, and oral vehicle that simplify formulation for antifatigue agent. These findings contribute to the new insights into both green extraction technology and the nutraceutical potential of C. xanthorrhiza.
Table 1 Comparison of treatment effects on swimming time and biochemical parameters.
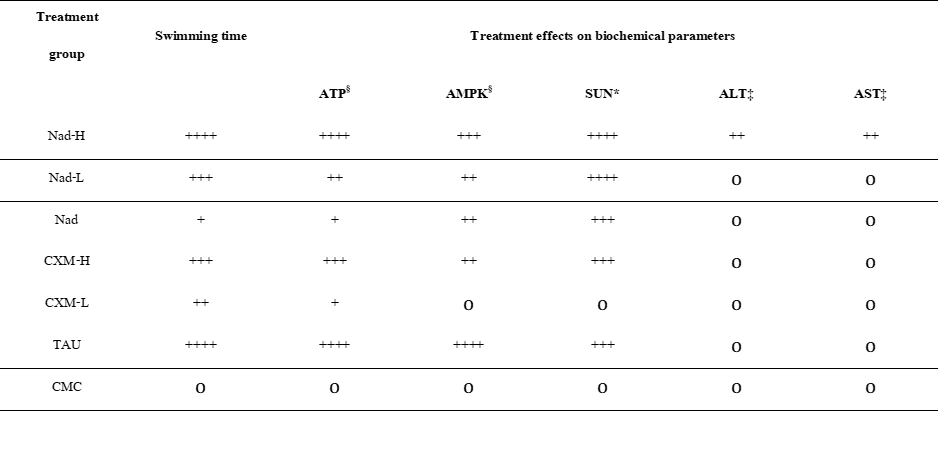
§Increased level of ATP or AMPK compared to TAU
*Decreased level of SUN compared to TAU
‡Increased level of ALT or AST compared to TAU
++++ Very strong effect
+++ Strong effect
++ Moderate effect
+ Weak effect
о No effect
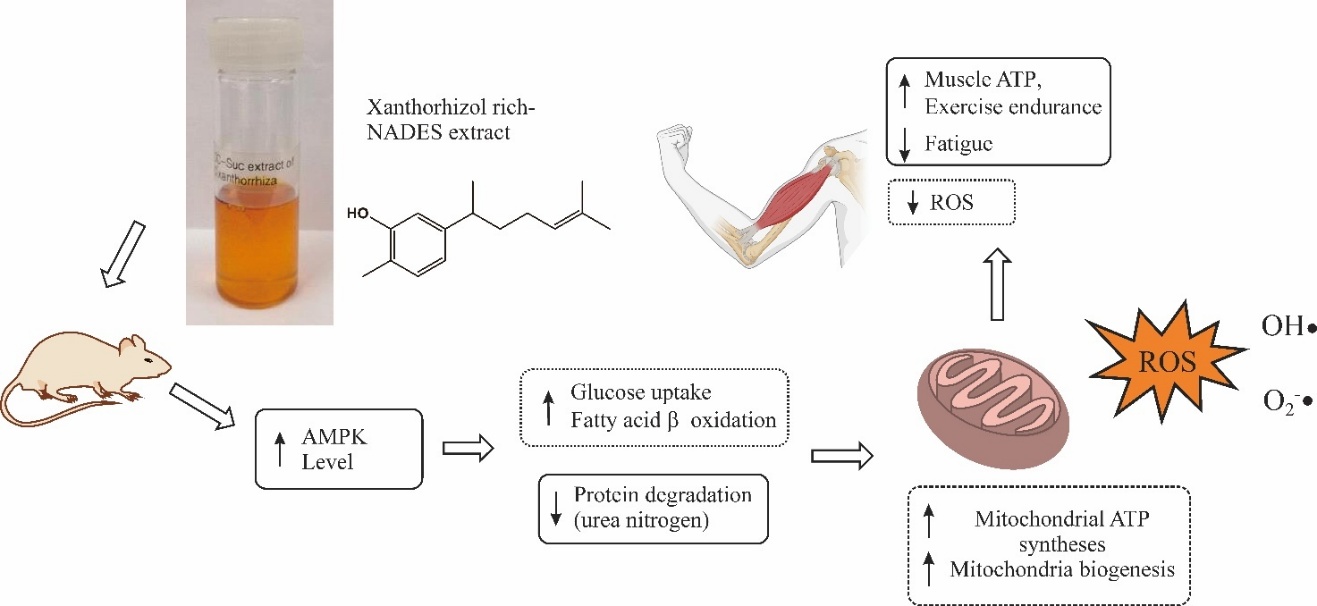
Figure 10 Proposed mechanism underlying the antifatigue effect of Curcuma xanthorrhiza NADES extracts. Solid rectangles indicate findings demonstrated in the present study, whereas dashed rectangles represent proposed mechanisms that were not directly investigated.
Figure 10 shows the proposed mechanisms underlying the antifatigue effect of C. xanthorrhiza extracts. The observed elevation of muscle ATP and AMPK levels in extract-treated mice indicate regulation on energy metabolism. The antifatigue activity of C. xanthorrhiza extract may likely be mediated through modulation of cellular energy metabolism via the AMP-activated protein kinase (AMPK) pathway. Activation of AMPK serves as a key metabolic switch that promotes ATP generation through increased glucose uptake, fatty acid oxidation, and mitochondrial biogenesis. In addition, exercise-induced fatigue is closely associated with excessive production of reactive oxygen species (ROS), which impairs mitochondrial function and disrupts redox homeostasis. Furthermore, the NADES system composed of choline chloride and sucrose may enhance XTZ solubilization, thereby potentiating its bioactivity.
Conclusions
Our results demonstrated the proof of concept of the antifatigue effect of C. xanthorrhiza extracts. Both low and high dosages of C. xanthorrhiza extracts could prolong swimming time in fatigue mice compared to vehicle in a dose dependent trend. The strongest effect was observed in the high dosage NADES extract group, comparable to that observed for the positive control TAU. Improvement of swimming performance may be enhanced by elevation muscle ATP and AMPK levels and decrease in the level of serum urea nitrogen. Treatment with NADES and ethanol extracts did not significantly change the levels of hepatic ALT and AST, as also seen in TAU and CMC groups. These results highlight the dual advantage of the NADES system—serving not only as a green extraction medium but also as an oral vehicle. The eco-friendly and low-cost NADES extraction offers potential industrial relevance as extraction media for bioactive compounds, to allow future development of sustainable functional food and nutraceutical products.
Future studies with larger samples and inclusion of female mice are warranted to assess sex-related differences in fatigue and metabolic regulation. Further, long-term toxicity assessment is required for the development of antifatigue agent. Further studies should also include pharmacokinetic investigation for the bioavailability of xanthorrhizol. In the present study, increases in muscle ATP and AMPK were observed following treatment with C. xanthorrhiza extracts, which indicate regulation in energy metabolism. However, downstream AMPK-mediated signaling pathway should be further investigated, for example increased expression of PGC-1 and SIRT1. This confirmation will better elucidate how C. xanthorrhiza and its major compound xanthorrhizol enhance energy metabolism and prevent fatigue.
Acknowledgements
Adelina Simamora acknowledged kind support from Krida Wacana Christian University, Indonesia for research facilities in animal experimentation. This work was supported by the grant from Badan Riset dan Inovasi Nasional through the National Metabolomics Collaborative Research Centre (03/PKR.LJ/PPK.02/DFRI/2024 and 270/PKS/ WRIII/UI/2024).
Conflict of interest
The authors report there are no competing interests to declare.
Declaration of Generative AI in Scientific Writing
Grammarly was used to improve the readability and language of this manuscript.
CRediT Author Statement
Adelina Simamora: Conceptualization; Methodology; Investigation; Writing - Original Draft, and Editing. Heri Setiawan: Methodology; Supervision; Writing – Review & Editing. Kris Herawan Timotius: Conceptualization; Methodology; Writing - Review & Editing; Supervision. Peni Ahmadi: Supervision; Writing – Review & Editing. Mukerrem Betul Yerer: Conceptualization; Writing - Review & Editing. Abdul Mun’im: Conceptualization; Methodology; Supervision; Resources; Funding Acquisition; Writing - Review & Editing. All authors agree to the final version of the manuscript.
References
[1] JH Yoon, NH Park, YE Kang, YC Ahn, EJ Lee and CG Son. The demographic features of fatigue in the general population worldwide: A systematic review and meta-analysis. Frontiers in Public Health 2023; 11, 1192121.
[2] Y Zhou, Z Chu, Y Luo, F Yang, F Cao, F Luo and Q Lin. Dietary polysaccharides exert anti-fatigue functions via the gut-muscle axis: Advances and prospectives. Foods 2023; 12(16), 3083.
[3] F Torri, P Lopriore, V Montano, G Siciliano, M Mancuso and G Ricci. Pathophysiology and management of fatigue in neuromuscular diseases. International Journal of Molecular Sciences 2023; 24(5), 5005.
[4] W Yu, C Song, Z Lei, Y Li, X He, J Yu and X Yang. Anti-fatigue effect of traditional chinese medicines: A review. Saudi Pharmaceutical Journal 2023; 31(4), 597-604.
[5] G Lu, Z Liu, X Wang and C Wang. Recent advances in panax ginseng c.A. Meyer as a herb for anti-fatigue: An effects and mechanisms review. Foods 2021; 10(5), 1030.
[6] H Zhang, R Kang, T Song, F Ren, J Liu and J Wang. Advances in relieving exercise fatigue for curcumin: Molecular targets, bioavailability, and potential mechanism. Journal of Food Science 2024; 89(8), 4604-4619.
[7] BF Wahidah, N Hayati, UN Khusna, TPD Rahmani, R Khasanah, I Kamal, F Husain and AI Setiawan. The ethnobotany of zingibraceae as the traditional medicine ingredients utilized by colo muria mountain villagers, central java. Journal of Physics: Conference Series 2021; 1796(1), 012113.
[8] ME Klau, E Rohaeti, M Rafi, IM Artika, L Ambarsari and W Nurcholis. Metabolite profiling of curcuma xanthorriza varieties grown in different regions using uhplc-q-orbitrap-hrms and chemometrics analysis. Biointerface Research in Applied Chemistry 2023; 13(1), 1-13.
[9] S Jinyuan. Effects of curcumin on physical fatigue and oxidative damage in forced swimming mice. E3S web of conferences. E3S Web of Conferences 2020; 189, 02021.
[10] S Kim, K Kim, J Park and W Jun. Curcuma longa l. Water extract enhances endurance exercise capacity by promoting intramuscular mitochondrial biogenesis in mice. Journal of Medicinal Food 2022; 25(2), 138-145.
[11] W Nurcholis, AA Munshif and L Ambarsari. Xanthorrhizol contents, α-glucosidase inhibition, and cytotoxic activities in ethyl acetate fraction of curcuma zanthorrhiza accessions from indonesia. Revista Brasileira de Farmacognosia 2018; 28(1), 44-49.
[12] C Cannavacciuolo, S Pagliari, J Frigerio, CM Giustra, M Labra and L Campone. Natural deep eutectic solvents (nadess) combined with sustainable extraction techniques: A review of the green chemistry approach in food analysis. Foods 2023; 12(1), 56-64.
[13] A Simamora, KH Timotius, H Setiawan, MY Putra and A Mun’Im. Natural deep eutectic solvent extraction of xanthorrhizol and curcuminoids from curcuma xanthorrhiza roxb and simultaneous determination by high-performance liquid chromatography. Journal of Pharmacy & Pharmacognosy Research, 2023; 11(6), 1056-1071.
[14] I Rukavina, MJ Rodrigues, CG Pereira, I Mansinhos, A Romano, S Slusarczyk, A Matkowski and L Custódio. Greener is better: First approach for the use of natural deep eutectic solvents (nades) to extract antioxidants from the medicinal halophyte polygonum maritimum l. Molecules 2021; 26(20), 6136.
[15] Y Dai, GJ Witkamp, R Verpoorte and YH Choi. Tailoring properties of natural deep eutectic solvents with water to facilitate their applications. Food Chemistry 2015; 187, 14-19.
[16] C Villa, D Caviglia, FS Robustelli Della Cuna, G Zuccari and E Russo. Nades application in cosmetic and pharmaceutical fields: An overview. Gels 2024; 10(2), 107.
[17] AS Amartya. 2022, The use of choline chloride sugar based- natural deep eutectic solvents for extraction of curcuma xanthorrhiza roxb. Bachelor Thesis, Universitas Indonesia, Jawa Barat, Indonesia.
[18] Kementerian Kesehatan Republik Indonesia and Kemenkes. Farmakope herbal indonesia edisI II. Kementerian Kesehatan Republik Indonesia and Kemenkes, Jakarta, Indonesia, 2017.
[19] WN Arifin and WM Zahiruddin. Sample size calculation in animal studies using resource equation approach. Malaysian Journal of Medical Science 2017; 24(5), 101-105.
[20] MB Kim, C Kim, Y Song and JK Hwang. Antihyperglycemic and anti-inflammatory effects of standardized curcuma xanthorrhiza roxb. Extract and its active compound xanthorrhizol in high-fat diet-induced obese mice. Evidence-based Complementary and Alternative Medicine 2014; 2014, 205915.
[21] T Miyazaki, Y Matsuzaki, T Ikegami, S Miyakawa, M Doy, N Tanaka and B Bouscarel. Optimal and effective oral dose of taurine to prolong exercise performance in rat. Amino Acids 2004; 27(3-4), 291-298.
[22] WC Huang, WC Chiu, HL Chuang, DW Tang, ZM Lee, L Wei, FA Chen and CC Huang. Effect of curcumin supplementation on physiological fatigue and physical performance in mice. Nutrients 2015; 7(2), 905-921.
[23] P Jauregi, L Esnal-Yeregi and J Labidi. Natural deep eutectic solvents (nades) for the extraction of bioactives: Emerging opportunities in biorefinery applications. PeerJ Analytical Chemistry 2024; 6(8), e32.
[24] J Chen, X Lu, P Chen, Y Shen, B Zheng and Z Guo. Anti-fatigue effect of glycoprotein from hairtail (trichiurus lepturus) by-products in a behavioral mouse model. Food Chemistry: X 2023; 18, 100645.
[25] SH Kim, HJ Kim, S Kim, JS Kang, YT Koo, SH Lee and DH Paik. A comparative study of antifatigue effects of taurine and vitamin c on chronic fatigue syndrome. 2022; 13(8), 300-312.
[26] MA Jung, A Jo, J Shin, H Kang, Y Kim, DR Oh and CY Choi. Anti-fatigue effects of elaeagnus multiflora fruit extracts in mice. Journal of Applied Biological Chemistry 2020; 63(1), 69-74.
[27] JA Kurtz, TA VanDusseldorp, JA Doyle and JS Otis. Taurine in sports and exercise. Journal of the International Society of Sports Nutrition 2021; 18(1), 39.
[28] LFD Gatward, MR Kennard, LIF Smith and AJF King. The use of mice in diabetes research: The impact of physiological characteristics, choice of model and husbandry practices. Diabetic Medicine 2021; 38(12), e14711.
[29] CJ Jong, J Azuma and S Schaffer. Mechanism underlying the antioxidant activity of taurine: Prevention of mitochondrial oxidant production. Amino Acids 2012; 42(6), 2223-2232.
[30] SW Schaffer, CJ Jong, T Ito and J Azuma. Role of taurine in the pathologies of melas and merrf. Amino Acids 2014; 46(1), 47-56.
[31] WD Shen, XY Li, YY Deng, XQ Zha, LH Pan, QM Li and JP Luo. Polygonatum cyrtonema hua polysaccharide exhibits anti-fatigue activity via regulating osteocalcin signaling. International Journal of Biological Macromolecules 2021; 175, 235-241.
[32] HR Spaulding and Z Yan. Ampk and the adaptation to exercise. Annual Review of Physiology 2022; 84, 209-227.
[33] GR Steinberg and DG Hardie. New insights into activation and function of the ampk. Nature Reviews Molecular Cell Biology 2023; 24(4), 255-272.
[34] RS Lee-Young, SR Griffee, SE Lynes, DP Bracy, JE Ayala, OP McGuinness and DH Wasserman. Skeletal muscle amp-activated protein kinase is essential for the metabolic response to exercise in vivo. Journal of Biological Chemistry 2009; 284(36), 23925-23934.
[35] HJ Kim, YJ Kim and JK Seong. Amp-activated protein kinase activation in skeletal muscle modulates exercise-induced uncoupled protein 1 expression in brown adipocyte in mouse model. The Journal of Physiology 2022; 600(10), 2359-2376.
[36] J Kim, S Beak, S Ahn, BS Moon, BS Kim, SJ Lee, S Jun Oh, HY Park, SH Kwon, CH Shin, K Lim and KP Lee. Effects of taurine and ginseng extracts on energy metabolism during exercise and their anti-fatigue properties in mice. Nutrition Research and Practice 2021; 16(1), 33-45.
[37] Z Li, H Zhu, H Hua, C Liu, Y Cheng, Y Guo, P Du and H Qian. Anti-fatigue activity of brassica rapa l. Extract and correlation among biochemical changes in forced swimming mice. Food Bioscience 2022, 47, 101633.
[38] DJ Crona, A Faso, TF Nishijima, KA McGraw, MD Galsky and MI Milowsky. A systematic review of strategies to prevent cisplatin-induced nephrotoxicity. The Oncologist 2017; 22(5), 609-619.
[39] S Chen, Y Minegishi, T Hasumura, A Shimotoyodome and N Ota. Involvement of ammonia metabolism in the improvement of endurance performance by tea catechins in mice. Scientific Reports 2020; 10(1), 6065.
[40] M Holeček. Muscle amino acid and adenine nucleotide metabolism during exercise and in liver cirrhosis: Speculations on how to reduce the harmful effects of ammonia. Metabolites 2022; 12(10), 971.
[41] LLD Santos, JBPD Castro, DG Linhares, AOBD Santos, LDS Cordeiro, CJB Pinheiro and RGDS Vale. Effects of physical exercise on hepatic biomarkers in adult individuals: A systematic review and meta-analysis. Retos: Nuevas Tendencias en Educación Física, Deporte y Recreación 2023; (49), 762-774.
[42] S Thakur, V Kumar, R Das, V Sharma and DK Mehta. Biomarkers of hepatic toxicity: An overview. Current Therapeutic Research 2024; 100, 100737.
[43] PF Surai, K Earle-Payne and MT Kidd. Taurine as a natural antioxidant: From direct antioxidant effects to protective action in various toxicological models. Antioxidants 2021; 10(12), 1876.
[44] Y Wang, T Xu, H Zhao, C Gu and Z Li. Effect of taurine in muscle damage markers and inflammatory cytokines in running exercise. Frontiers in Physiology 2022;13, 1008060.
[45] INE Lister, L Chiuman, MS Mutia, H Hartono, E Girsang, AF Sutendi, HSW Kusuma, DS Hadiprasetyo and W Widowati. Hepatoprotective effects of curcuma xanthorrhiza roxb. Extract via free radical scavenger, inhibiting apoptosis and inflammation mechanisms in acetaminophen-induced liver injury. Iranian Journal of Basic Medical Sciences 2025; 28(8), 1100-1106.
[46] LE Holcomb, P Rowe, CC O’Neill, EA DeWitt and SC Kolwicz. Sex differences in endurance exercise capacity and skeletal muscle lipid metabolism in mice. Physiological Reports 2022; 10(3), e15174.
[47] PS Hafen, AS Law, C Matias, SG Miller and JJ Brault. Skeletal muscle contraction kinetics and ampk responses are modulated by the adenine nucleotide degrading enzyme ampd1. Journal of Applied Physiology 2022; 133(5), 1055-1066.
[48] AM Kaluve, JT Le and BM Graham. Female rodents are not more variable than male rodents: A meta-analysis of preclinical studies of fear and anxiety. Neuroscience & Biobehavioral Reviews 2022; 143, 104962.
[49] E Rahmat, J Lee and Y Kang. Javanese turmeric (curcuma xanthorrhiza roxb.): Ethnobotany, phytochemistry, biotechnology, and pharmacological activities. Evidence-Based Complementary Alternative Medicine 2021; 2021, 9960813.