
Trends
Sci.
2026;
23(6):
12635
In vitro Inhibition of Protein Glycation, Methylglyoxal, α-Glucosidase, and Metal Ions by a Thai Polyherbal Formulation
Papada
Nadee1,2,
![]() ,
Chinnaphat
Chaloemram2,
,
Chinnaphat
Chaloemram2,
![]() ,
Nuttapong
Wichai2,
,
Nuttapong
Wichai2,
![]() ,
,
Achida
Jaruchotikamol2,
![]() and Benjamart
Cushnie2,*
and Benjamart
Cushnie2,*
1Department of Thai traditional medicine, Faculty of Allied Health Sciences, Pathumthani University,
Pathumthani 12000, Thailand
2Pharmaceutical Chemistry and Natural Products Research Unit, Faculty of Pharmacy, Mahasarakham University, Maha Sarakham 44150, Thailand
(*Corresponding author’s e-mail: [email protected])
Received: 14 November 2025, Revised: 11 December 2025, Accepted: 21 December 2025, Published: 20 February 2026
Abstract
Glycation processes generate advanced glycation end products (AGEs) and intermediates, such as methylglyoxal (MGO), which contribute to non-communicable diseases like diabetes and cardiovascular and cerebrovascular disorders. This study explores the antiglycation, carbohydrate-hydrolyzing enzyme inhibition, MGO-trapping, and metal-chelating properties of hydroethanolic extracts of Benchalokawichian (BLW) and its individual herbal components. The total flavonoid content was assessed using the aluminum chloride method, revealing Tiliacora triandra and Ficus racemosa as rich flavonoid sources. Antiglycation activity was evaluated using bovine serum albumin (BSA)/fructose and BSA/MGO models, with aminoguanidine (AG) as a positive control. All extracts demonstrated concentration-dependent antiglycation effects. Notably, Harrisonia perforata at 1 mg/mL significantly inhibited AGE formation by 73.79 ± 1.55% in BSA/fructose and 99.58 ± 0.11% in BSA/MGO models, and exhibited a substantial MGO-trapping efficiency of 98.19 ± 0.02%, surpassing the standard agent AG at 92.24 ± 0.12%. The extracts’ capacity to inhibit carbohydrate-hydrolyzing enzymes was assessed through α-glucosidase activity. H. perforata and F. racemosa showed significant inhibitory effects, with IC50 values of 36.44 ± 8.38 and 81.67 ± 16.74 μg/mL, respectively, outperforming the standard drug acarbose (IC50 104 ± 17.91 μg/mL). Metal chelation capacity, measured through an iron-ferrozine assay, was consistently lower than that of the standard chelating agent EDTA across all tested extracts. In conclusion, H. perforata demonstrates promising potential in reducing glycation and inhibiting α-glucosidase enzymes involved in glucose metabolism, indicating its possible application in functional foods or as a therapeutic agent for diabetes management. Further research is warranted to explore its clinical benefits.
Keywords: Benchalokawichian remedy, Flavonoids, α-glucosidase, Metal chelation, Antiglycation, Methylglyoxal-trapping
Introduction
Glycation, a non-enzymatic reaction involving the covalent attachment of reducing sugars to biomolecules, initially forms reversible Amadori products such as fructosamine, which serve as key indicators of short-term glycemic control. These products are precursors to the formation of irreversible advanced glycation end products (AGEs). The accumulation of AGEs is strongly linked to the pathogenesis of various chronic non-communicable diseases (NCDs), including diabetes mellitus, cardiovascular diseases, and cancer [1]. Methylglyoxal (MGO), a highly reactive dicarbonyl compound primarily produced during glycolysis, is a crucial intermediate in this process. Due to its high reactivity, MGO rapidly modifies proteins and nucleic acids, accelerating early AGE formation and resulting in cellular dysfunction. Elevated MGO levels are associated with increased oxidative stress and inflammatory responses, contributing to tissue damage and disease progression [2-4]. In diabetes, chronic hyperglycemia significantly enhances AGE production, contributing to vascular damage and nephropathy. Thus, therapeutic strategies that inhibit glycation and scavenge reactive intermediates like MGO are essential for preventing related complications [5]. Additionally, suppressing carbohydrate-hydrolyzing enzymes, such as α-glucosidase, is important as it slows down the digestion and absorption of carbohydrates, thereby managing postprandial hyperglycemia, a significant driver of glycation processes [6]. Moreover, transition metal ions can significantly contribute to the formation of irreversible AGEs by catalyzing oxidative reactions (glycoxidation), leading to increased reactive oxygen species and enhanced cross-linking of proteins [7]. Therefore, targeting these various pathways is crucial for effective antiglycation interventions.
The Benchalokawichian (BLW) remedy, also known as Ya-Ha-Rak, is a traditional polyherbal formulation approved by the Thai Ministry of Public Health since 2012 for the treatment of fever [8]. This remedy comprises five equally proportioned plant roots: Clerodendrum indicum (CI), Capparis micracantha (CM), Ficus racemosa (FR), Harrisonia perforata (HP), and Tiliacora triandra (TT) [9]. Pharmacological investigations have documented BLW’s various bioactivities, including antioxidant properties (anti-lipid peroxidation and free radical scavenging) and anti-inflammatory effects [10,11]. Clinical and animal studies consistently report a favorable safety profile [12,13]. BLW is rich in bioactive compounds such as phenolics, flavonoids, and terpenoids [10], which contribute to its antioxidant, anti-inflammatory, and enzyme-inhibitory properties. Ethnobotanical evidence suggests that these herbal combinations may exert antiglycation effects by trapping reactive carbonyl intermediates, preventing the formation of AGEs, and regulating carbohydrate-digesting enzymes.
While ethnobotanical evidence suggests the antiglycation effects of BLW, a comprehensive and mechanistic understanding of this polyherbal formulation and its individual components in combating glycation-related pathways is notably lacking. Previous pharmacological investigations have primarily focused on its antioxidant and anti-inflammatory properties, but the specific evaluation of its ability to directly inhibit AGE formation, trap highly reactive methylglyoxal, modulate α-glucosidase activity, and chelate metal ions has not been systematically addressed. Therefore, this study aims to fill this critical knowledge gap by providing a detailed scientific evaluation of the antiglycation potential of BLW and each of its five constituent herbs. By elucidating these multifaceted mechanisms, this research offers novel insights into the specific contributions of each component and the synergistic effects within the formulation, thereby scientifically validating traditional Thai medicine within the context of modern metabolic disease management. The findings are expected to support the development of functional foods or nutraceuticals incorporating these herbs, potentially aiding in the prevention of NCD progression and enhancing metabolic health.
Materials and Methods
Plant materials and extraction
The roots of five medicinal plants used in the formulation of Benchalokawichian (BLW) - CI, CM, FR, HP, and TT, were collected from Roi Et province, Thailand (16°18'0.22'' N, 103°58'40.30'' E). A botanist authenticated the species, and voucher specimens were deposited at the Faculty of Pharmacy, Mahasarakham University. The reference numbers and yield percentages for these samples are listed in Table 1. The plant materials were meticulously cleaned, dried in a hot air oven at 70 °C for 72 h, and ground into a fine powder.
To prepare the ethanolic extract of BLW, equal portions (100 g each) of the five root powders were combined to yield 500 g. This mixture underwent maceration in 2.5 L of 80% (v/v) ethanol at room temperature, performed twice over 7 days. After maceration, the extract was filtered through Whatman filter paper. The solvent was then evaporated using a rotary evaporator (Rotavapor R-300, Buchi™, Switzerland). The concentrated extract was freeze-dried with a Scanvac CoolSafe 110-4 lyophilizer (Labogene™, Denmark) and stored at −20 °C until further analysis.
Chemicals and reagents
Aminoguanidine hydrochloride was purchased from Thermo Fisher Scientific, Belgium. Aluminum chloride hexahydrate, D-fructose, sodium azide, ethylenediaminetetraacetic acid (EDTA), and anhydrous sodium carbonate were sourced from Carlo Erba, Italy. Alpha-glucosidase enzyme type I from Saccharomyces cerevisiae, acarbose, nitroblue tetrazolium chloride, and p-nitrophenyl-α-D-glucopyranoside (p-NPG) were obtained from Sigma-Aldrich, USA. Quercetin hydrate and ferrozine iron reagent-hydrate were acquired from Acros Organics, Belgium. Bovine serum albumin (BSA) was purchased from Himedia, Belgium. Methylglyoxal (40% solution in water), o-phenylenediamine (OPDA), and 5-methylquinoxaline (5-MQ) were also purchased from Sigma-Aldrich Co. (St. Louis, MO, USA).
Evaluation of total flavonoid content
Total flavonoid content (TFC) was determined using a modified aluminum chloride colorimetric assay, as described by Jirasripongpun et al. [14]. Briefly, 500 μL of the extract (1 mg/mL concentration) was mixed with 75 μL of 5% (w/v) sodium nitrite solution and 150 μL of 10% (w/v) aluminum chloride solution. The mixture was incubated in the dark at room temperature for 5 min. Subsequently, 500 μL of 1 M sodium hydroxide and 275 μL of distilled water were added. Quercetin was used as the standard flavonoid for calibration. After an additional 15-min incubation, absorbance was measured at 510 nm using a microplate reader (Varioskan LUX, Thermo Scientific™, USA). The TFC was calculated based on a standard curve constructed with quercetin, and results were expressed as milligrams of quercetin equivalents per gram of dry extract (mg QE/g).
In vitro glycation of bovine serum albumin (BSA) by fructose (BSA/Fr model)
Preparation of glycated samples
The antiglycation activity of the extract was evaluated using an in vitro glycation model involving BSA and fructose, as described by Zuo et al. [15]. BSA and fructose solutions were prepared in 0.1 M phosphate-buffered saline (PBS) with a pH of 7.4, supplemented with 0.02% (w/v) sodium azide to inhibit microbial growth. To generate the glycated samples, 350 µL of a 1.1 M fructose solution was combined with 600 µL of a BSA solution at 50 mg/mL, and 50 µL of the extract at various concentrations ranging from 100 to 1,000 µg/mL. The mixtures were incubated at 37 °C for 14 days. Aminoguanidine (AG), a synthetic antiglycation agent, served as a positive control at a concentration of 1 mg/mL.
In vitro glycation of bovine serum albumin (BSA) by methylglyoxal (BSA/MGO model)
The antiglycation activity of the extract was evaluated using a simulated glycation system comprising BSA and methylglyoxal (MGO), following established protocols [16]. Initially, solutions of BSA and MGO were prepared in 0.1 M PBS at pH 7.4, with 0.02% (w/v) sodium azide added to prevent microbial contamination. The glycation reaction mixture included 350 µL of 50 mM MGO solution, 600 µL of BSA solution, and 50 µL of the extract at 1 mg/mL. The mixture was then incubated at 37 °C for 24 h. Aminoguanidine (AG) served as a positive control at a concentration of 1 mg/mL.
Quantification of fluorescent AGEs
Fluorescent AGEs in the glycated samples from both the BSA/Fr and BSA/MGO assays were quantified by transferring 200 μL of each sample into black 96-well plates. Using a Varioskan LUX microplate reader (Thermo Scientific™, USA), fluorescence intensity was measured at excitation and emission wavelengths of 355 and 460 nm, respectively. The percentage inhibition of glycation was subsequently calculated using Eq. (1).
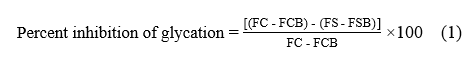
where FC represents the fluorescence measurement of the glycated control without any extract, and FCB refers to the fluorescence of the blank control, consisting of BSA without fructose. FS represents the fluorescence of the glycated sample, while FSB indicates the fluorescence of the blank sample, composed of BSA and the extract in the absence of fructose.
Quantification of fructosamine
Fructosamine levels in glycated samples from the BSA/fructose assay were determined using the nitroblue tetrazolium (NBT) assay, following the protocol by Ali et al. [17] with slight modifications. After incubating for 14 days, 180 µL of a 500 mM NBT solution, dissolved in a 100 mM carbonate buffer at pH 10.4, was thoroughly mixed with 20 µL of the glycated sample. The mixture was incubated in the dark for 15 min. Absorbance was measured at 530 nm using a microplate reader. Fructosamine concentrations were calculated based on a standard curve generated from varying concentrations of 1-deoxy-1-morpholino-fructose (1-DMF). Results were expressed as millimolar (mM) fructosamine equivalents in DMF units.
Evaluation of MGO-trapping capacity
The methylglyoxal (MGO) trapping capacity was evaluated through the reduction of carbonyl compounds, following the method outlined by Chayaratanasin et al. [18]. Fresh solutions were prepared with 1 mM methylglyoxal, 100 mM OPDA as a derivatization reagent, and 10 mM 5-methylquinoxaline (5-MQ) as an internal standard, all in 0.1 M phosphate buffer at pH 7.4. To assess the trapping capacity, we mixed 200 µL of the MGO solution with either 200 µL of phosphate buffer (as a blank control) or 200 µL of 1 mM aminoguanidine (AG) as a positive control. For evaluating inhibitory effects from various samples, 200 µL of each extract, at a concentration of 2 mg/mL, was added to 200 µL of 1 mM MGO solution. The mixtures were incubated at 37 °C for 60 min to enable the trapping reaction.
After incubation, 100 µL of OPDA and 20 µL of the internal standard 5-MQ were added to each sample, and the solutions were maintained at room temperature for 30 min to complete the derivatization reaction. The remaining MGO, which was converted to its 2-methylquinoxaline (2-MQ) derivative, was quantified using high-performance liquid chromatography with diode array detection (HPLC-DAD). The analyses were conducted on an Agilent 1260 Infinity II Prime system (Agilent Technologies, USA) using ChemStation software. Derivative separation was achieved with a Kromosil C18 column (5 µm, 100 Å, 250×4.6 mm2). The mobile phase comprised solvent A (water) and solvent B (methanol), with a flow rate of 1.2 mL/min. An aliquot of 20 µL from each sample was injected, with isocratic elution at 70% solvent B maintained for 10 min per run. Detection occurred at 315 nm, monitoring peaks of 2-MQ and 5-MQ. Quantitative analysis was conducted by comparing the ratio of peak areas of 2-MQ to 5-MQ. The percentage reduction of MGO was calculated using a specific Eq. (2).
![]()
Determination of α-glucosidase inhibition
The inhibitory activity against α-glucosidase from Saccharomyces cerevisiae was evaluated with slight modifications to a previously published method [19]. In this assay, 50 μL of the sample or acarbose, the standard inhibitor, was combined with 100 μL of α-glucosidase enzyme solution at a concentration of 0.05 units/mL in phosphate buffer at pH 6.8. The mixture was incubated at 37 °C for 10 min. Thereafter, 50 μL of 0.1 M p-nitrophenyl-α-D-glucopyranoside (p-NPG) substrate was added, and the incubation was extended for another 20 min at 37 °C. The reaction was stopped by adding 50 μL of 0.1 M sodium bicarbonate solution. Absorbance was subsequently measured at 405 nm using a Varioskan LUX microplate reader (Thermo Scientific™, USA). The percentage inhibition of α-glucosidase activity was calculated using Eq. (3).
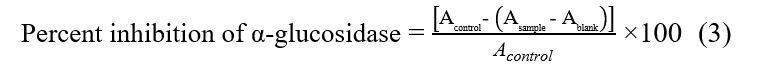
Metal-chelating assay
The chelating ability of ferrous iron was assessed using a method based on Gulcin et al. [20], with minor modifications. Briefly, 50 μL of either the sample (at final concentrations of 1 to 3 mg/mL) or the standard EDTA (at final concentrations of 10 to 240 μg/mL) was combined with 50 μL of 1 mM FeSO₄. The reaction mixtures were incubated at room temperature for 10 min. Following this, 100 μL of 0.2 mM ferrozine was added to each mixture, and the incubation continued for an additional 10 min at room temperature. Absorbance was then measured at 562 nm using a Varioskan LUX microplate reader (Thermo Scientific™, USA). The percentage inhibition of Fe²⁺-ferrozine complex formation was calculated using Eq. (3).
Statistical analysis
Data are presented as the mean ± standard error of the mean (SEM), whereas IC₅₀ values are expressed as the mean ± standard deviation (SD), based on three independent experiments. We performed statistical analysis using one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test for multiple comparisons. A p-value less than 0.05 was considered statistically significant. All analyses were conducted using GraphPad Prism software (version 9).
Results and discussion
Extract yield and total flavonoid content
The concentration of ethanol for the extraction was based on previous studies [10] that consistently demonstrated 80% ethanol as an optimal solvent for extracting a broad spectrum of phytochemicals and achieving high total phenolic content from similar herbal matrices. This approach was crucial as these compounds are often associated with bioactivities relevant to our current investigation into glycation reactions, thereby enabling for preparation of an extract expected to exhibit significant anti-glycation potential. The percentage yield and total flavonoid content (TFC) of 80% ethanolic extracts procured from the BLW remedy and its individual herbal components are shown in Table 1. TFC was measured using the aluminum chloride colorimetric assay and expressed as milligrams of quercetin equivalents per gram of dry extract (mg QE/g). TT exhibited the highest TFC at 124.41 ± 1.79 mg QE/g, followed by FR with 53.28 ± 0.18 mg QE/g. The BLW formulation displayed a moderate flavonoid level of 34.36 ± 0.64 mg QE/g, similar to the level observed in HP at 37.88 ± 2.42 mg QE/g. The significantly elevated TFC in TT and FR suggests that these two plants significantly contribute to the antioxidant and antiglycation potential of the BLW formulation. Flavonoids are known for their ability to scavenge free radicals and inhibit glycation pathways [21].
Prior research conducted by our group using the same ethanolic extract of TT showed a maximum total phenolic content (TPC) of 55.97 ± 0.49 mg GAE/g. In parallel, the BLW extract demonstrated an intermediate TPC of 28.67 ± 0.43 mg GAE/g, approximately half the amount found in TT [10]. This finding is consistent with the highest TFC recorded in TT compared to other individual herb extracts. A positive correlation between TPC and TFC is typical of herbs because flavonoids are a subset of phenolic compounds; thus, higher flavonoid concentrations inherently raise total phenolic levels [22]. However, despite TT’s high TFC, it is crucial to recognize that flavonoid type may be a more significant factor than quantity. The bioactivity of different flavonoids can vary substantially, with the A-ring serving as the functional group for MGO capture, while the hydroxyl group at the C3 position on the C-ring may hinder adduct formation [21]. Further research is needed to identify the specific flavonoids present in each extract and their individual contributions to the observed antiglycation, MGO-trapping, and α-glucosidase inhibitory effects.
Table 1 Percent yields and total flavonoid content in ethanolic extracts of the BLW remedy and its plant ingredients.
Root extracts |
Abbreviations |
Voucher number |
Percent yield |
Total flavonoid content (mg QE/g dry extract) |
Benchalokawichian remedy |
BLW |
PH-BLW01 |
7.25 |
34.36 ± 0.64a |
Clerodendrum indicum |
CI |
PH-CI01 |
10.36 |
20.55 ± 1.32b |
Capparis micracantha |
CM |
PH-CM01 |
6.91 |
23.96 ± 1.02c |
Ficus racemosa |
FR |
PH-FR01 |
5.57 |
53.28 ± 0.18d |
Harrisonia perforata |
HP |
PH-HP01 |
5.50 |
37.88 ± 2.42a |
Tiliacora triandra |
TT |
PH-TT01 |
10.00 |
124.41 ± 1.79e |
Note: Different letters (a-e) denote statistically significant differences (p < 0.05).
AGE inhibition assay
AGEs are major contributors to NCDs and diabetic complications, leading to tissue damage and inflammation. Inhibiting AGE formation is therefore essential. We utilized both BSA/fructose and BSA/MGO glycation models to comprehensively evaluate the anti-AGE potential of BLW extracts, which target different AGE-generating pathways.
In the BSA/fructose model, the concentration-dependent inhibition of AGE formation by the BLW remedy and its constituent plant extracts is depicted in Figure 1. After a 14-day incubation at 37 °C, extracts were tested at concentrations of 100, 250, 500, and 1,000 µg/mL. These extracts demonstrated statistically significant variations in inhibitory efficacy (p < 0.05). Notably, BLW, FR, HP, and TT showed substantial AGE inhibition at higher concentrations, such as 1,000 µg/mL, while CI and CM exhibited comparatively lower activity.
Figure 2 presents results from the BSA/MGO model, where all extracts were tested at a fixed concentration of 1 mg/mL for 24 h. All extracts, including the AG positive control, significantly inhibited AGE formation (p < 0.05). Differential inhibition was observed; AG, HP, and BLW demonstrated superior activity, whereas CI, CM, FR, and TT showed relatively lower inhibition. HP’s superior capacity might be partly attributed to its high flavonoid luteolin content (143.53 ± 1.58 mg/g extract), which has been reported to have potent anti-inflammatory effects [23]. Furthermore, luteolin is known to stabilize protein structures and prevent protein cross-linking, particularly in BSA, thereby directly enhancing its antiglycation activity [24].
The efficacy of the extracts differed between the two models. In the BSA/fructose model (Figure 1), HP, BLW, and FR demonstrated potent activity at higher concentrations. Conversely, in the BSA/MGO model (Figure 2), HP and BLW consistently exhibited greater effectiveness. These results suggest that HP and BLW are capable of inhibiting AGE formation through both glycation pathways. The BSA/fructose model represents the complete, slower Maillard reaction pathway, a more chronic glycation process. In contrast, the BSA/MGO model primarily addresses the later, faster intermediate stages that involve the highly reactive dicarbonyl species MGO [25]. The observed differences in activity imply that the BLW extracts contain compounds that target various stages or mechanisms within these pathways. The consistent effectiveness of HP and BLW extracts in both models (BSA/fructose at higher concentrations and BSA/MGO) suggests these extracts possess a broad spectrum of antiglycation compounds capable of acting through multiple mechanisms. This is particularly significant given the complex nature of glycation in vivo, where both slow, sugar-mediated glycation and rapid MGO-induced glycation contribute to AGE formation [26].
FR’s potent activity in the BSA/fructose model (Figure 1), as opposed to its relatively lower activity in the BSA/MGO model (Figure 2), suggests that FR may be particularly effective in interfering with the earlier stages of glycation. This could be due to its ability to compete with fructose for binding to proteins or to prevent the formation of reactive intermediates before MGO is produced. Additionally, a previous study demonstrated that the FR extract showed significant superoxide radical scavenging activity, with 63.77 ± 1.12% inhibition at 1 mg/mL. This ability to scavenge radicals supports its potential to mitigate glycation, considering the recognized role of oxidative stress in glycation progression [10]. In contrast, the comparatively lower activity of CM in both models suggests that this extract may contain fewer or less potent antiglycation compounds than other extracts, or that its primary mechanisms are not effectively assessed by these in vitro assays. This finding aligns with prior research reporting that CM extract exhibited the lowest superoxide radical scavenging activity [10].
The integrated findings from the BSA/fructose and BSA/MGO models suggest that the BLW remedy serves as a multi-faceted antiglycation agent. By incorporating ingredients effective at various stages of the glycation pathway, BLW may provide a comprehensive strategy for preventing AGE formation and mitigating the long-term complications of diabetes and other NCDs [27]. These results support the overall hypothesis, highlighting the value of BLW as a complex formulation.
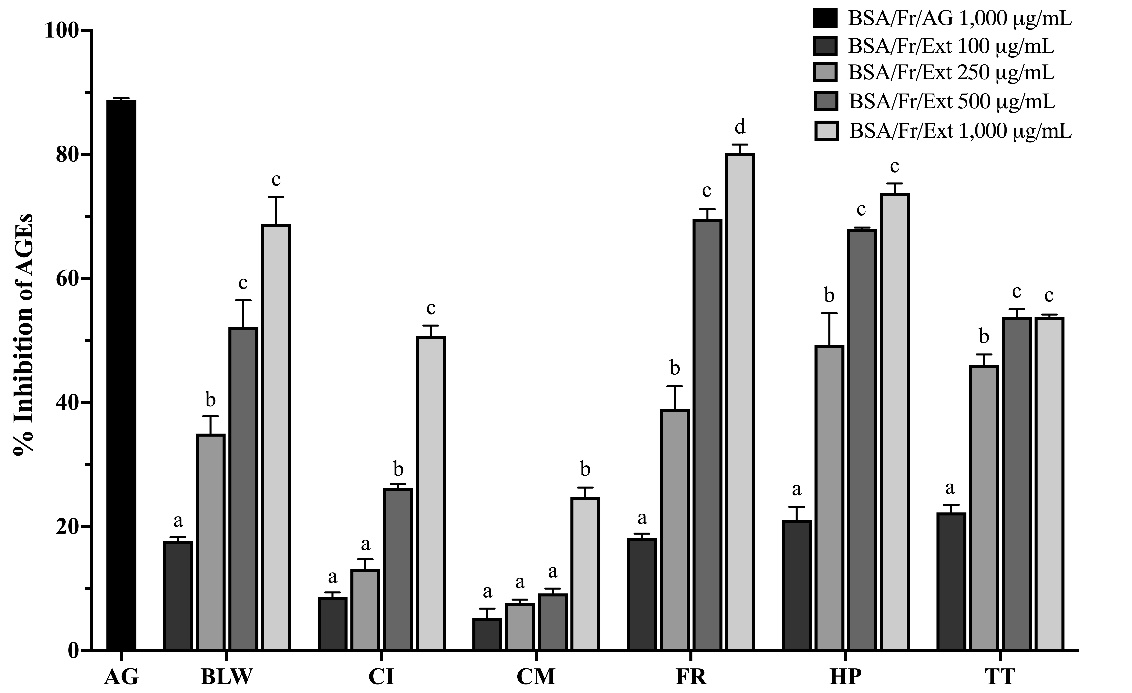 Figure
1 Inhibition
of advanced glycation end-products (AGEs) formation by BLW
extracts in a bovine serum albumin (BSA)/fructose glycation
model. Percentage inhibition of AGE formation by ethanolic
extracts of the BLW remedy and its plant components in a
BSA/fructose model after 14 days. Extracts were tested at
concentrations ranging from 100 to 1000 µg/mL. Data represent
mean ± SEM (n=3). Bars with different letters indicate
significant differences (p
< 0.05) within each group. Abbreviations: AG: aminoguanidine;
BLW: Benchalokawichian; CI: Clerodendrum
indicum;
CM: Capparis
micracantha;
FR: Ficus
racemosa;
HP: Harrisonia
perforata;
TT: Tiliacora
triandra.
Figure
1 Inhibition
of advanced glycation end-products (AGEs) formation by BLW
extracts in a bovine serum albumin (BSA)/fructose glycation
model. Percentage inhibition of AGE formation by ethanolic
extracts of the BLW remedy and its plant components in a
BSA/fructose model after 14 days. Extracts were tested at
concentrations ranging from 100 to 1000 µg/mL. Data represent
mean ± SEM (n=3). Bars with different letters indicate
significant differences (p
< 0.05) within each group. Abbreviations: AG: aminoguanidine;
BLW: Benchalokawichian; CI: Clerodendrum
indicum;
CM: Capparis
micracantha;
FR: Ficus
racemosa;
HP: Harrisonia
perforata;
TT: Tiliacora
triandra.
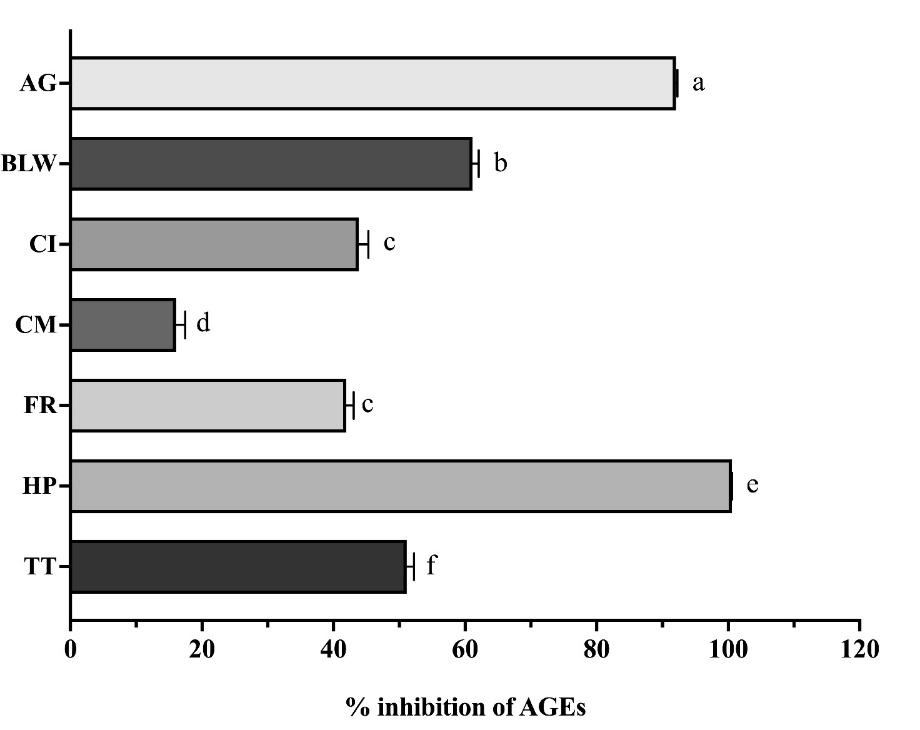 Figure
2 Inhibition
of advanced glycation end-products (AGEs) formation in a
methylglyoxal (MGO)-induced model. Percentage inhibition of AGEs
by ethanolic extracts (1 mg/mL) of the BLW remedy and its
constituent plants in an MGO-induced glycation model after 24-h
incubation. Data are presented as mean ± SEM (n = 3). Different
letters above bars indicate significant differences (p
< 0.05) as determined by one-way ANOVA with Tukey’s post-hoc
test.
Figure
2 Inhibition
of advanced glycation end-products (AGEs) formation in a
methylglyoxal (MGO)-induced model. Percentage inhibition of AGEs
by ethanolic extracts (1 mg/mL) of the BLW remedy and its
constituent plants in an MGO-induced glycation model after 24-h
incubation. Data are presented as mean ± SEM (n = 3). Different
letters above bars indicate significant differences (p
< 0.05) as determined by one-way ANOVA with Tukey’s post-hoc
test.
Abbreviations: AG: aminoguanidine; BLW: Benchalokawichian; CI: Clerodendrum indicum; CM: Capparis micracantha; FR: Ficus racemosa; HP: Harrisonia perforata; TT: Tiliacora triandra.
Fructosamine inhibition assay
Fructosamine, an early glycation product, is a critical marker for assessing short-term glycemic control and predicting the formation of AGEs. Reducing fructosamine levels is essential for preventing diabetic complications. In this study, a concentration of 1 mg/mL was selected for the extracts based on their proven efficacy in inhibiting glycation during preliminary assessments. Using a BSA/fructose glycation model and following a 14-day incubation at 37 °C, all tested extracts, including the positive control AG, significantly reduced fructosamine levels compared to the untreated control (p < 0.05) (Figure 3). The observation that the extracts achieved similar reductions in fructosamine levels indicates their clinically relevant antiglycation properties. AG, a synthetic antiglycation agent, has shown promise in clinical trials [28], and the ability of BLW extracts to produce similar effects merits further investigation. Although the other extracts are not superior to AG, they remain significant. Despite these encouraging in vitro results, it is crucial to recognize the limitations of the BSA/fructose glycation model. This model offers a simplified system, and the observed effects may not completely translate to in vivo conditions. Future studies should explore the antiglycation activity of BLW extracts in more complex models, such as cell-based assays or animal studies, and aim to identify the specific compounds responsible for the observed effects [29].
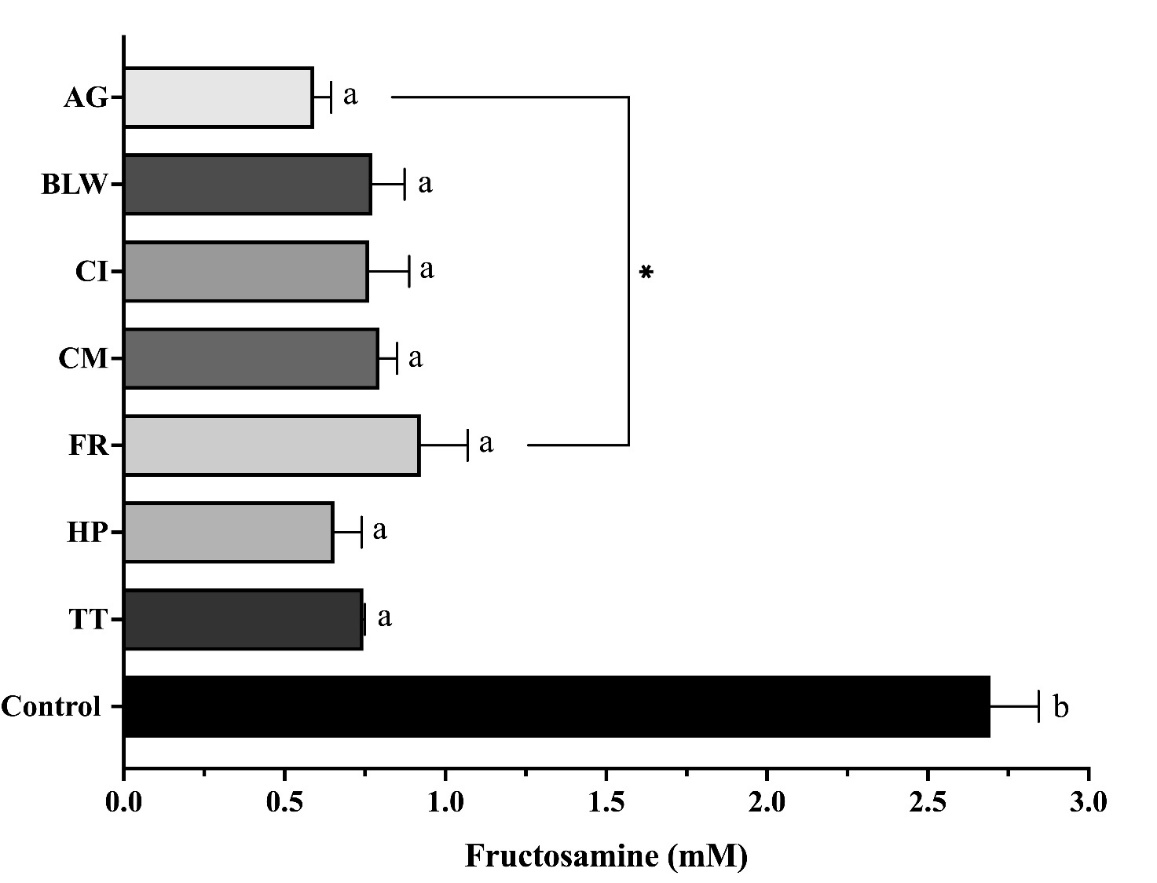
Figure 3 Impact of ethanolic extracts of the BLW remedy and its plant compositions on fructosamine formation in a bovine serum albumin (BSA)/fructose glycation model. Extracts (1 mg/mL) were incubated at 37 °C for 14 days in a BSA/fructose model. Data are expressed as mean ± SEM (n = 3). Statistical significance: a p < 0.05 compared to the BSA/Fr control; * p < 0.05 compared to AG. Abbreviations: AG: aminoguanidine; BLW: Benchalokawichian; CI: Clerodendrum indicum; CM: Capparis micracantha; FR: Ficus racemosa; HP: Harrisonia perforata; TT: Tiliacora triandra.
Methylglyoxal-trapping capacity assay
Assessing methylglyoxal (MGO) trapping capacity is essential for understanding how compounds neutralize this reactive dicarbonyl and prevent the formation of AGEs. MGO-trapping constitutes a vital detoxification mechanism that mitigates protein damage. We utilized high-performance liquid chromatography (HPLC) to quantify the MGO-trapping capacity of BLW extracts, thereby providing insights into their antiglycation mechanisms. In this study, extracts of BLW and individual herbs at a concentration of 1 mg/mL were directly incubated with 1 mM MGO for 60 min. Figure 4 illustrates the HPLC chromatograms from the MGO-trapping assay, evaluating the ability of the extracts and a control substance to sequester or react with MGO, thus reducing its presence. The chromatograms display peaks corresponding to the MGO derivative 2-methylquinoxaline (2-MQ) and the internal standard 5-methylquinoxaline (5-MQ). The inclusion of 5-MQ as an internal standard ensured accurate quantification. A reduction in the 2-MQ peak area relative to the control indicates effective MGO-trapping or reaction by the tested substance, thereby inhibiting its conversion to 2-MQ. The AG demonstrated a significant reduction in the 2-MQ peak relative to the control.
Table 2 quantifies the percentage reduction in MGO. To normalize the data, the ratio of the 2-MQ peak area to the 5-MQ peak area was calculated. Higher percentages suggest a greater capacity for trapping MGO. The control, with 0% MGO reduction, serves as the baseline. The AG positive control exhibited a significant MGO reduction of 92.486 ± 1.083%. Among the extracts, the HP extract achieved the highest reduction at 98.191 ± 0.186%, whereas the TT extract had the lowest reduction at 36.645 ± 0.152%. The visual decrease in the 2-MQ peak area within the chromatograms shown in Figure 4 corresponds to the elevated MGO reduction percentages reported in Table 2.
The finding that the HP extract exhibited the highest MGO reduction, surpassing even the positive control AG, strongly suggests that HP contains highly effective MGO-trapping compounds. This ability to directly sequester MGO is a crucial mechanism for preventing AGE formation and mitigating the damaging effects of this reactive dicarbonyl species [30]. The differing degrees of MGO reduction observed among the BLW extracts indicate significant variations in their MGO-trapping capabilities (p < 0.05). This variability may reflect differences in the concentration or types of active compounds in each extract, as well as their mechanisms of action. For instance, gallic acid is reported to exhibit a synergistic mechanism involving free radical scavenging, MGO-trapping, and protein binding. This synergistic action, shared by related natural antioxidants like resveratrol and epicatechin gallate, underlies their ability to inhibit protein glycation and aggregation processes implicated in diseases such as diabetes and amyloid disorders. These properties position these compounds as potential therapeutic agents for both preventing and treating these conditions [31]. As previously noted, the high luteolin content in HP may significantly contribute to its potent antiglycation effects [24]. Consequently, further investigation is warranted to identify the specific compounds responsible for MGO-trapping across all tested extracts. Conversely, the relatively low MGO reduction observed for the TT extract suggests that TT is either less effective at directly trapping MGO or that its antiglycation activity is primarily mediated through other mechanisms, such as inhibiting the formation of MGO itself or interfering with later stages of AGE formation.
These findings indicate that the BLW remedy holds promise as a multi-targeted antiglycation agent. Notably, specific components - particularly HP - play a significant role in trapping MGO. Future research should aim to isolate and identify the specific MGO-trapping compounds within HP, and explore their potential therapeutic application in managing diabetes and its related complications [25].
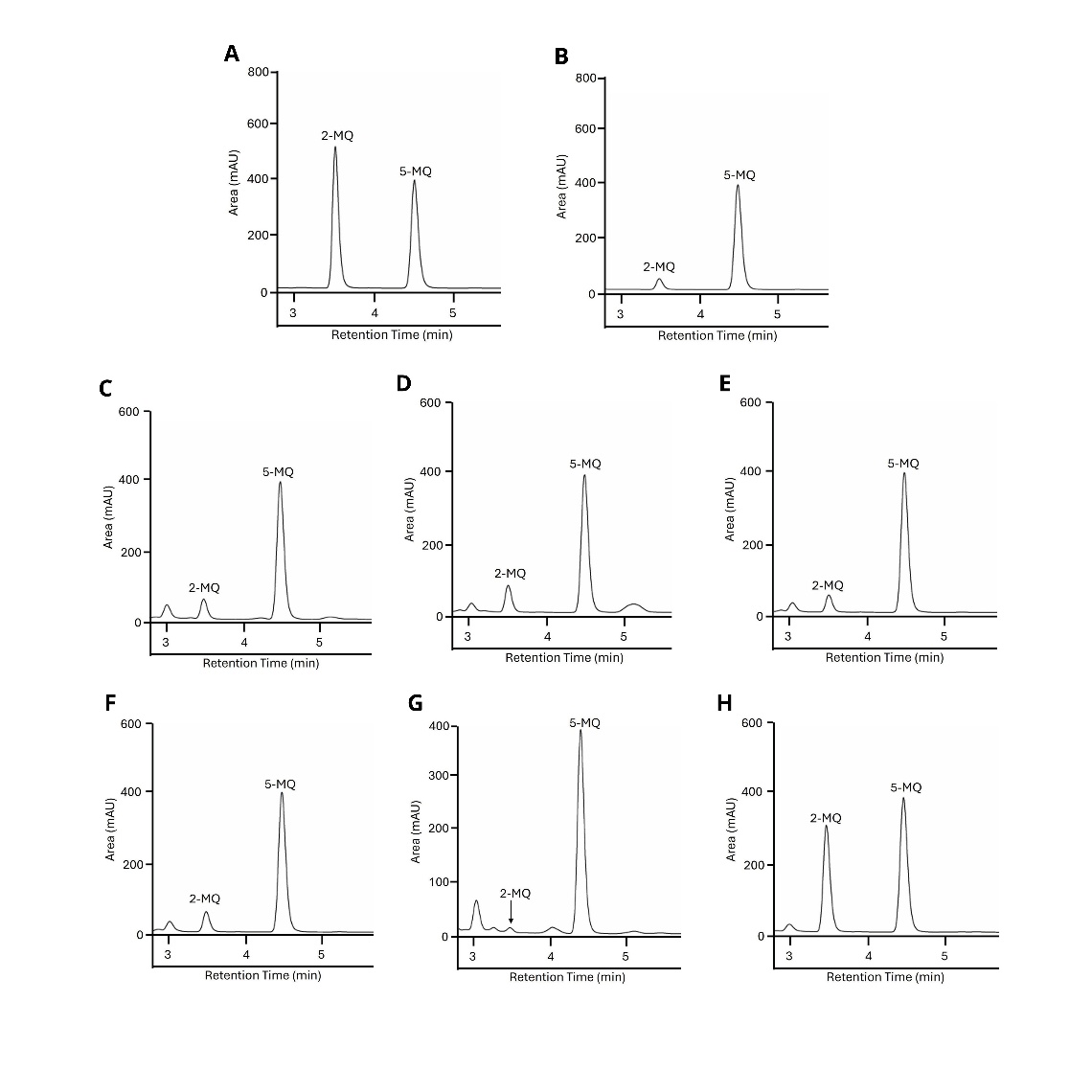
Figure 4 MGO trapping assay by HPLC: Impact of BLW extracts. The figure shows representative HPLC chromatograms from an MGO-trapping assay. (A) Control: 1 mM MGO plus phosphate buffer. (B-H) Treatments: 1 mM MGO solutions incubated with (B) aminoguanidine (AG), (C) Benchalokawichian (BLW), (D) Clerodendrum indicum (CI), (E) Capparis micracantha (CM), (F) Ficus racemosa (FR), (G) Harrisonia perforata (HP), and (H) Tiliacora triandra (TT). Peaks indicate the MGO derivative 2-methylquinoxaline (2-MQ) and the internal standard 5-methylquinoxaline (5-MQ).
Table 2 Percentage methylglyoxal (MGO) reduction by the BLW remedy and its constituent plants (1 mg/mL) after a 60-min incubation.
Samples |
HPLC peak area ratio: 2-MQ/5-MQ |
Percent MGO reduction |
Control AG BLW CI CM FR HP TT |
1.131 ± 0.019 0.088 ± 0.012 0.129 ± 0.031 0.175 ± 0.025 0.111 ± 0.015 0.130 ± 0.011 0.021 ± 0.021 0.717 ± 0.017 |
0 92.486 ± 1.083a 88.592 ± 0.278b 84.570 ± 0.221c 90.155 ± 0.137d 88.535 ± 0.101b 98.191 ± 0.186e 36.645 ± 0.152f |
Data are presented as mean ± SEM (n = 3). Within each column, values with different superscript letters indicate significant differences (p < 0.05) by 1-way ANOVA with Tukey’s post hoc test. Different letters (a-f) denote statistically significant differences (p < 0.05). Abbreviations: BLW: Benchalokawichian; CI: Clerodendrum indicum; CM: Capparis micracantha; FR: Ficus racemosa; HP: Harrisonia perforata; TT: Tiliacora triandra; AG: aminoguanidine was used as a positive control.
Alpha-glucosidase inhibitory activity
The antidiabetic potential of the extracts was evaluated by assessing their α-glucosidase inhibitory activity (Table 3). Acarbose, used as a positive control, demonstrated an IC50 value of 104.00 ± 17.91 µg/mL. The HP and FR extracts exhibited high inhibitory potency, with low IC50 values of 36.44 ± 8.38 and 81.67 ± 16.74 μg/mL, respectively, indicating significantly superior (p < 0.05) α-glucosidase inhibitory capacity compared to acarbose. In contrast, the BLW, CI, CM, and TT extracts showed IC50 values exceeding 1 mg/mL, suggesting a comparatively weaker α-glucosidase inhibitory effect than that of HP and FR. These results indicate that, although the BLW extract and its constituents possess α-glucosidase inhibitory activity, their potencies vary considerably among the different herbal components.
The remarkably low IC50 values demonstrated by HP and FR in this assay indicate a significantly greater inhibitory capacity than that of the positive control, acarbose. This suggests that these extracts contain potent α-glucosidase inhibitors. Clinically, α-glucosidase inhibitors like acarbose, miglitol, and voglibose can effectively delay carbohydrate digestion and reduce postprandial hyperglycemia [32]. The potent activity of HP and FR suggests their potential for managing blood sugar spikes after meals, particularly for individuals with type 2 diabetes who struggle to control postprandial glucose levels. Sakpakdeejaroen et al. [8] previously quantified pectolinarigenin at 18.50 mg/g of extract in the BLW remedy extract using a reversed-phase HPLC (RP-HPLC) method; however, no data were provided for HP or FR. Pectolinarigenin is especially noteworthy as a natural flavonoid with established α-glucosidase inhibitory activity, demonstrating an IC50 of 230 µM, which is significantly more potent than acarbose (IC50 996.02 µM) [33].
While the BLW, CI, CM, and TT extracts demonstrate relatively weaker α-glucosidase inhibitory effects (IC50 > 1 mg/mL), the antidiabetic potential of the BLW remedy may be amplified by the presence of HP and FR. These potent α-glucosidase inhibitors, in combination with other compounds in the BLW formulation that target various aspects of glucose metabolism - such as antiglycation and MGO-trapping - may produce a synergistic effect [34]. Future research should aim to identify the specific compounds responsible for the significant α-glucosidase inhibitory activity in HP and FR. Understanding the structure-activity relationships of these compounds could facilitate the development of novel and more effective α-glucosidase inhibitors for diabetes treatment [35]. Additionally, in vivo studies are necessary to validate these findings and to evaluate the bioavailability and efficacy of these extracts in a more complex biological system.
Table 3 Alpha-glucosidase inhibitory activity of ethanolic extracts of the BLW remedy and its herbal components.
Samples |
Concentration (µg/mL) |
Maximal inhibition (%) |
IC50 values (µg/mL) |
BLW CI CM FR HP TT Acarbose |
2,500 2,500 2,500 750 200 2,500 750 |
45.66 ± 0.87a,b 54.00 ± 2.56b 32.69 ± 0.36a 93.46 ± 5.14c,d 98.24 ± 0.54c 64.46 ± 5.74b,d 79.19 ± 0.24d |
n.a. n.a. n.a. 81.67 ± 16.74a 36.44 ± 8.38b n.a. 104.00 ± 17.91c |
Data are presented as mean ± SEM (n=3). Within each column, values with different superscript letters indicate significant differences (p < 0.05) as determined by one-way ANOVA with Tukey’s post hoc test. Abbreviations: BLW: Benchalokawichian; CI: Clerodendrum indicum; CM: Capparis micracantha; FR: Ficus racemosa; HP: Harrisonia perforata; TT: Tiliacora triandra. Note: Different letters (a-e) denote statistically significant differences (p < 0.05); n.a. is not applicable, with IC50 values > 1 mg/mL.
Metal-chelating capacity
Metal ions, particularly iron and copper, catalyze glycation and promote oxidative stress, thereby accelerating the formation of AGEs. Consequently, metal chelation presents a strategic approach to indirectly inhibit AGEs and reduce related damage [7]. This study examines the metal-chelating activity of BLW extracts, investigating their potential to mitigate glycation through metal ion sequestration. Figure 5(A) shows the metal-chelating activity of the standard inhibitor EDTA, which increases in a concentration-dependent manner. Figure 5(B) depicts the metal-chelating activity of the extracts at concentrations of 1, 2, and 3 mg/mL. At 3 mg/mL, TT and FR exhibited the highest metal-chelating activity, significantly exceeding that of the other extracts (p < 0.05). These results suggest that TT and FR likely contain a higher concentration of metal-chelating compounds or possess compounds with greater affinity for metal ions. This enhanced metal-chelating capacity may contribute to the antioxidant properties of these extracts [10,36].
While the metal-chelating activity of the BLW extracts was consistently lower than that of EDTA, a non-selective chelating agent with a high affinity for metal ions, it is crucial to note that EDTA may not be directly comparable to the natural chelating compounds in the extracts. The relatively weaker chelation activity of the extracts could still hold physiological significance, possibly targeting specific metal ions or acting locally within tissues to reduce oxidative stress without depleting essential metals systemically [37]. Thus, despite their lower potency compared to EDTA, the extracts’ chelating activity may be meaningful and potentially safer for biological applications.
Future research should aim to identify the specific metal-chelating compounds in TT, FR, and other active BLW extracts, as well as assess their efficacy in chelating various metal ions such as iron, copper, and zinc. Furthermore, in vitro and in vivo studies are necessary to evaluate the impact of these extracts on oxidative stress and inflammation in the context of diabetes and related complications [38].
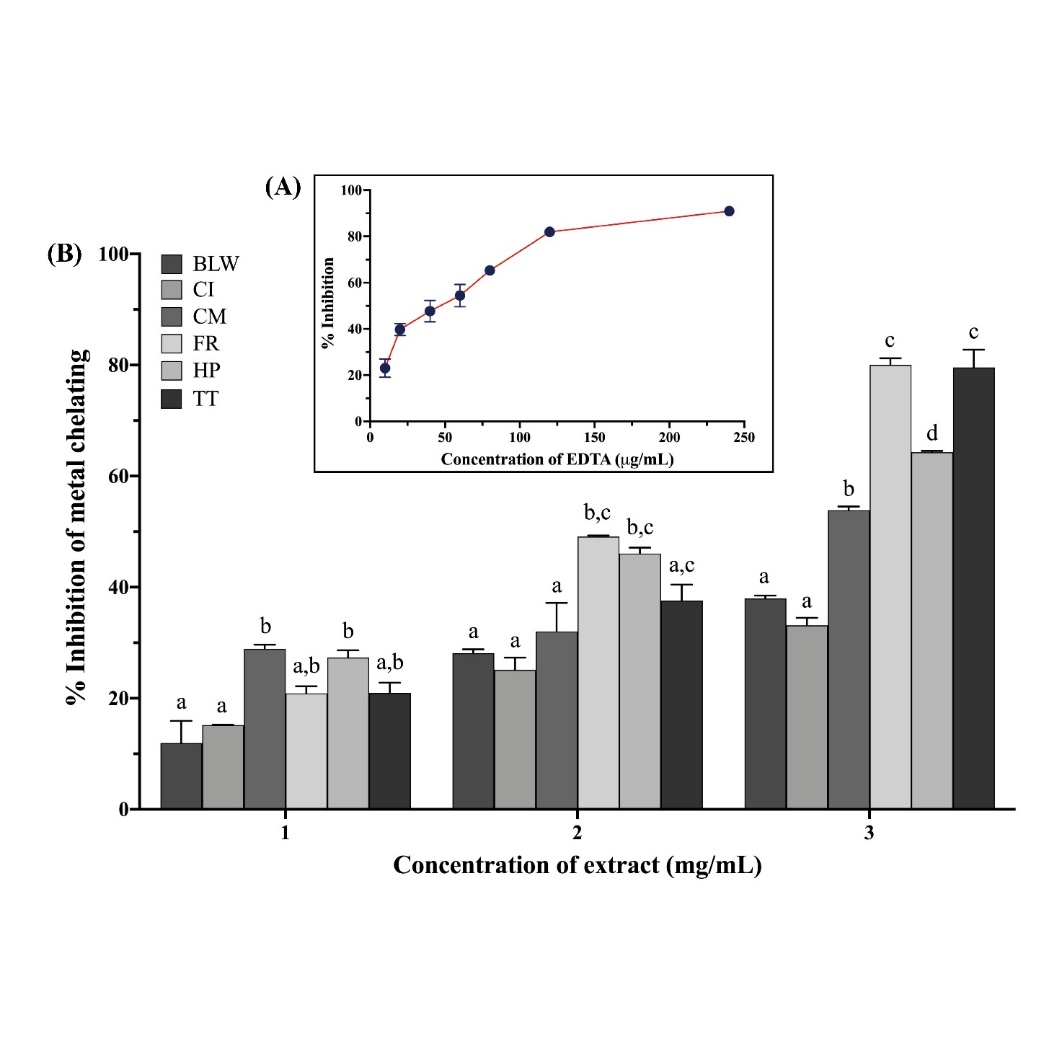
Figure 5 In vitro metal chelating activity of EDTA, BLW formulation, and its herbal components. (A) Metal chelation by EDTA, a standard chelating agent, is shown for comparison. (B) Percentage inhibition of metal chelation by the BLW formulation and its constituent herbal extracts at varying concentrations (1 - 3 mg/mL). Data are expressed as mean ± SEM (n = 3). Within each concentration, bars with different letters (a-d) indicate statistically significant differences (p < 0.05) as determined by 1-way ANOVA with Tukey’s post-hoc test. Abbreviations: EDTA, ethylenediaminetetraacetic acid; BLW, Benchalokawichian; CI, Clerodendrum indicum; CM, Capparis micracantha; FR, Ficus racemosa; HP, Harrisonia perforata; TT, Tiliacora triandra.
Conclusions
This study affirms the multi-targeted antiglycation and antidiabetic potential of the Thai polyherbal formulation Benchalokawichian (BLW) and its components. BLW and one of its components, HP, consistently inhibited glycation, such as fructosamine, and AGEs, across various models. Notably, HP demonstrated superior methylglyoxal-trapping capabilities, even surpassing those of aminoguanidine. Both HP and another component, FR, proved to be potent α-glucosidase inhibitors, exhibiting greater efficacy than acarbose. Although metal chelation activity was primarily observed in TT and FR, these activities were modest. These findings indicate a promising potential for developing functional foods or therapeutic agents to manage diabetes based on BLW and its specific components. However, it is crucial to recognize the preliminary nature of this investigation. As an in vitro study, its findings provide foundational insights but require validation in more complex biological systems. While previous animal and clinical studies have consistently indicated a favorable safety profile for BLW (e.g., [12,13] as cited in the Introduction), comprehensive toxicity assessments specific to the potential therapeutic dosages and long-term administration derived from these anti-glycation findings remain an essential area for future research. Therefore, future research should not only focus on isolating and identifying the specific bioactive compounds responsible for these effects and conducting in vivo studies to confirm efficacy and clarify underlying molecular mechanisms but also explore its potential in human clinical trials. These steps are vital to fully realize BLW's potential in NCD prevention and metabolic health improvement.
Acknowledgements
This research project was financially supported by Faculty of Pharmacy at Mahasarakham University Fund, Thailand (S#3/2568). The authors gratefully acknowledge the Pharmaceutical Chemistry and Natural Product Research Unit (PCNRU), Faculty of Pharmacy, Mahasarakham University, Thailand, for providing laboratory equipment.
Declaration of generative AI in scientific writing
The authors confirm that no Generative AI tools were employed in the preparation of this article.
CRediT author statement
Papada Nadee: Investigation; Methodology; Formal analysis; Writing - Original draft preparation, Reviewing & Editing; visualization. Chinnaphat Chaloemram: Investigation; Validation; Writing - visualization. Nuttapong Wichai: Supervision; Methodology, Validation. Achida Jaruchotikamol: Conceptualization; Supervision. Benjamart Cushnie: Conceptualization; Methodology; Formal analysis; Writing – Reviewing & Editing; Supervision; Funding acquisition.
References
[1] E Qaed, W Aldahmash and MA Mahyoub. Advanced glycation end products (AGEs) and their role in diabetes mellitus and related complications: Mechanisms and therapeutic insights. Glycoconjugate Journal 2025; 42, 209-223.
[2] MI Khan, F Ashfaq, AA Alsayegh, A Hamouda, F Khatoon, TN Altamimi, FS Alhodieb and MMA Beg. Advanced glycation end product signaling and metabolic complications: Dietary approach. World Journal of Diabetes 2023; 14(7), 995-1012.
[3] AL Oliveira, MG de Oliveira, FZ Mónica and E Antunes. Methylglyoxal and advanced glycation end products (AGEs): Targets for the prevention and treatment of diabetes-associated bladder dysfunction? Biomedicines 2024; 12(5), 939.
[4] CG Schalkwijk and CDA Stehouwer. Methylglyoxal, a highly reactive dicarbonyl compound, in diabetes, its vascular complications, and other age-related diseases. Physiological Reviews 2020; 100(1), 407-461.
[5] P Vangrieken, JLJM Scheijen, PMH Schiffers, MPH Van De Waarenburg, S Foulquier and CCG Schalkwijk. Modelling the effects of elevated methylglyoxal levels on vascular and metabolic complications. Scientific Reports 2025; 15(1), 6025.
[6] Q Song, J Liu, L Dong, X Wang and X Zhang. Novel advances in inhibiting advanced glycation end product formation using natural compounds. Biomedicine & Pharmacotherapy 2021; 140, 111750.
[7] NT Moldogazieva, IM Mokhosoev, TI Mel’nikova, YB Porozov and AA Terentiev. Oxidative stress and advanced lipoxidation and glycation end products (ALEs and AGEs) in aging and age-related diseases. Oxidative Medicine and Cellular Longevity 2019; 2019, 3085756.
[8] I Sakpakdeejaroen, T Juckmeta and A Itharat. Development and validation of RP-HPLC method to determine anti-allergic compound in Thai traditional remedy called Benjalokawichian. Journal of the Medical Association of Thailand 2014; 97(S8), S76-S80.
[9] C Singharachai, C Palanuvej, H Kiyohara, H Yamada and N Ruangrungsi. Pharmacognostic specification of five root species in Thai traditional medicine remedy: Ben-Cha-Lo-Ka-Wi-Chian. Pharmacognosy Journal 2011; 3(21), 1-11.
[10] P Nadee, A Jaruchotikamol and B Cushnie. Antioxidant capacity and phytochemical analysis of the traditional Thai remedy Benchalokawichian. Tropical Journal of Natural Product Research 2024; 8(8), 8054-8060.
[11] T Juckmeta and A Itharat. Anti-inflammatory and antioxidant activities of Thai traditional remedy called “Ya-ha-rak”. Journal of Health Research 2012; 26(4), 205-210.
[12] W Chandranipapongse. Study the effect of an antipyretic drug, Thai herbal Ha-Rak formula on platelet aggregation in healthy Thai volunteers: A randomized, placebo - controlled trial. Siriraj Medical Journal 2017; 69(5), 283-289.
[13] N Srichaipor, P Pongcharoen, P Kanokkangsadal and A Itharat. Skin irritation and allergic testing of Thai herbal extracts (Ha-Rak with Turmeric) in healthy volunteers. Thammasat Medical Journal 2020; 20(2), 165-174.
[14] K Jirasripongpun, N Jirakanjanakit, S Pola and K Obsuwan. Antioxidation and anti-melanogenesis of three colored flowers of Dendrobium hybrids. Science, Engineering and Health Studies 2022; 16, 22030006.
[15] GL Zuo, HY Kim, YN Guillen Quispe, ZQ Wang, SH Hwang, KO Shin and SS Lim. Efficient separation of phytochemicals from Muehlenbeckia volcanica (Benth.) Endl. by polarity-stepwise elution counter-current chromatography and their antioxidant, antiglycation, and aldose reductase inhibition potentials. Molecules 2021; 26(1), 224.
[16] I Fecka, K Bednarska and A Kowalczyk. In vitro antiglycation and methylglyoxal trapping effect of Peppermint Leaf (Mentha × piperita L.) and its polyphenols. Molecules 2023; 28(6), 2865.
[17] A Ali, R Shahu, P Balyan, S Kumari, R Ghodmare, R Jobby and P Jha. Antioxidation and antiglycation properties of a natural sweetener: Stevia rebaudiana. Sugar Tech 2022; 24(2), 563-575.
[18] P Chayaratanasin, S Adisakwattana and T Thilavech. Protective role of Clitoria ternatea L. flower extract on methylglyoxal-induced protein glycation and oxidative damage to DNA. BMC Complementary Medicine and Therapies 2021; 21(1), 80.
[19] S Amin, B Ullah, M Ali, A Rauf, H Khan, E Uriarte and E Sobarzo-Sánchez. Potent in vitro α-glucosidase inhibition of secondary metabolites derived from Dryopteris cycadina. Molecules 2019; 24(3), 427.
[20] İ Gulcin and SH Alwasel. Metal ions, metal chelators and metal chelating assay as antioxidant method. Processes 2022; 10(1), 132.
[21] LF Li, MD Wang, CY Zhang, MY Jin, HL Chen, H Luo, TY Hou, ZJ Zhang and H Li. Influence of hydroxyl substitution on the inhibition of flavonoids in advanced glycation end-products formation in glucose-lysine-arginine maillard reaction models. Food Research International 2025; 207, 116068.
[22] Z Khiya, Y Oualcadi, A Gamar, F Berrekhis, T Zair and FE Hilali. Correlation of total polyphenolic content with antioxidant activity of hydromethanolic extract and their fractions of the Salvia officinalis leaves from different regions of Morocco. Journal of Chemistry 2021; 2021(1), 8585313.
[23] W Semmarath, P Arjsri, K Srisawad, S Umsumarng and P Dejkriengkraikul. Luteolin-rich extract from Harrisonia perforata (Blanco) Merr. root alleviates SARS-CoV-2 spike protein-stimulated lung inflammation via inhibition of MAPK/NLRP3 inflammasome signaling pathways. Life 2025; 15(7), 1077.
[24] FM Djeujo, V Stablum, E Pangrazzi, E Ragazzi and G Froldi. Luteolin and Vernodalol as bioactive compounds of leaf and root Vernonia amygdalina extracts: Effects on α-glucosidase, glycation, ROS, cell viability, and in silico ADMET parameters. Pharmaceutics 2023; 15(5), 1541.
[25] N Yadav, JD Palkhede and SY Kim. Anti-glucotoxicity effect of phytoconstituents via inhibiting MGO-AGEs formation and breaking MGO-AGEs. International Journal of Molecular Sciences 2023; 24(8), 7672.
[26] AB Uceda, L Mariño, R Casasnovas and M Adrover. An overview on glycation: Molecular mechanisms, impact on proteins, pathogenesis, and inhibition. Biophysical Reveiws 2024; 16(2), 189-218.
[27] W Jia, R Ma, R Zhang, Z Fan and L Shi. Synthetic-free compounds as the potential glycation inhibitors performed in in vitro chemical models: Molecular mechanisms and structure requirements. Trends in Food Science & Technology 2022; 128, 147-159.
[28] VP Reddy, P Aryal and EK Darkwah. Advanced glycation end products in health and disease. Microorganisms 2022; 10(9), 1848.
[29] L Mou, P Hu, X Cao, Y Chen, Y Xu, T He, Y Wei and R He. Comparison of bovine serum albumin glycation by ribose and fructose in vitro and in vivo. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease 2022; 1868(1), 166283.
[30] B Stratmann. Dicarbonyl stress in diabetic vascular disease. International Journal of Molecular Sciences 2022; 23(11), 6186.
[31] H Liu, X Huo, S Wang and Z Yin. The inhibitory effects of natural antioxidants on protein glycation as well as aggregation induced by methylglyoxal and underlying mechanisms. Colloids and Surfaces B: Biointerfaces 2022; 212, 112360.
[32] AM Dirir, M Daou, AF Yousef and LF Yousef. A review of α-glucosidase inhibitors from plants as potential candidates for the treatment of type-2 diabetes. Phytochemistry Reviews 2022; 21(4), 1049-1079.
[33] D Hadiarti, W Haryadi, S Matsjeh, RT Swasono and N Awaliyah. Profiling of α-glucosidase inhibitors from ethyl acetate fraction of Buas-buas (Premna serratifolia) leaves using UHPLC-Q-Orbitrap HRMS and protein-ligand interaction with molecular docking. Journal of Applied Pharmaceutical Science 2022; 13(2), 89-98.
[34] S Lv, J Li, Y Hu, J Cai, G Nan, Y Kong, X Shen, L Zhu, S Yang and C Dong. Multi-target therapeutic potential of Arctii Fructus Lignans in diabetes mellitus and its complications: A mechanistic review. Pharmaceuticals 2025; 18(10), 1569.
[35] SA Halim, S Jabeen, A Khan and A Al-Harrasi. Rational design of novel inhibitors of α-glucosidase: An application of quantitative structure activity relationship and structure-based virtual screening. Pharmaceuticals 2021; 14(5), 482.
[36] N Pradubyat, F Madaka, T Songsak and S Jongrungruangchok. In vitro biological activity of Tiliacora triandra (Colebr.) Diels root extract. Journal of Current Science and Technology 2024; 15(1), 77.
[37] R Evstatiev, A Cervenka, T Austerlitz, G Deim, M Baumgartner, A Beer, A Krnjic, C Gmainer, M Lang, A Frick, H Schachner, V Khare and C Gasche. The food additive EDTA aggravates colitis and colon carcinogenesis in mouse models. Scientific Reports 2021; 11(1), 5188.
[38] P Mucha, A Skoczyńska, M Małecka, P Hikisz and E Budzisz. Overview of the antioxidant and anti-inflammatory activities of selected plant compounds and their metal ions complexes. Molecules 2021; 26(16), 4886.