
Trends
Sci.
2026;
23(6):
12140
Enhancing the Functional and Sensory Properties of Yogurt with Red Fruit Oil (Pandanus conoideus L.): Antioxidant and α-Glucosidase Inhibitory Activities
Nur Haerati, Rimbawan Rimbawan*, Eny Palupi and Ribka Tande
Department of Nutrition Science, Faculty of Human Ecology, IPB University, Bogor 16680, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 2 October 2025, Revised: 18 November 2025, Accepted: 28 November 2025, Published: 5 February 2026
Abstract
Red fruit oil (Pandanus conoideus L.) is rich in bioactive compounds with antioxidant and antidiabetic activity. However, its application in food remains limited due to its unfavorable taste and aroma. Meanwhile, yogurt is a widely consumed fermented dairy product known for its distinctive physical and sensory characteristics. This study aimed to evaluate the effect of red fruit oil addition on the physical properties, sensory quality, bioactive compound content, antioxidant activity, and α-glucosidase inhibitory activity of yogurt. Four formulations (F0, F1, F2 and F3) were prepared by adding red fruit oil at levels of 0%, 5%, 10% and 15%, respectively. Parameters analyzed included pH, viscosity, titratable acidity (TA), color, hedonic test, QDA, antioxidant activity (CUPRAC), total flavonoid and phenolic contents, and in vitro α-glucosidase inhibition. Yogurt without red fruit oil (F0) showed antioxidant activity of 130.99 ± 7.48 μmol TE/g, flavonoid content of 20.04 ± 0.95 mg QE/g, phenolic content of 24.08 ± 0.36 mg GAE/g, and α-glucosidase inhibition of 64.46 ± 1.56%. The addition of 10% red fruit oil (F2) significantly increased these values to 369.47 μmol TE/g, 29.00 ± 0.62 mg QE/g, 155.54 ± 0.95 mg GAE/g, and 78.08 ± 0.37%, respectively. The addition of the oil also improved the sensory perception of red fruit oil itself, making it more organoleptically acceptable. F2 exhibited a balance between biological functionality and sensory acceptance, with an overall liking score of 5.38 ± 1.7 and the highest color score of 7.30 ± 1.4. These findings support the potential of red fruit oil yogurt as a functional food with antioxidant and antidiabetic effects.
Keywords: Yogurt, Red fruit oil, Bioactive compounds, Antioxidant, α-glucosidase, Sensory quality
Introduction
Diabetes mellitus is one of the most prevalent degenerative diseases worldwide with a steadily increasing trend. According to the International Diabetes Federation (IDF), approximately 537 million people worldwide are living with diabetes by 2021, and this number is projected to rise to 783 million by 2045 [1]. In Indonesia, the growing prevalence of diabetes is accompanied by chronic complications such as cardiovascular disease and nephropathy, posing significant challenges to the national healthcare system [2,3]. Type 2 diabetes is characterized by hyperglycemia due to insulin resistance and pancreatic β-cell dysfunction. One therapeutic approach involves inhibition of α-glucosidase, a key enzyme in carbohydrate digestion that influences postprandial blood glucose spikes [4,5].
With increasing public awareness of healthy food, yogurt has emerged as a widely consumed fermented dairy product that is recognized for its functional food potential. Yogurt contains lactic acid bacteria and metabolic compounds that promote gut health and shows antidiabetic potential through enzymatic inhibition and modulation of blood glucose levels [6,7]. Enhancing the functional value of yogurt by incorporating natural bioactive ingredients is an innovative strategy alignsigned with current trends in natural-based functional food development.
Red fruit oil (Pandanus conoideus L.), a local commodity from Papua, is rich in flavonoids, phenolics, β-carotene, and α-tocopherol [8-10]. These compounds have been reported to exhibit strong antioxidant activity and contribute to blood glucose reduction through β-cell regeneration and α-glucosidase inhibition [11-13]. However, its application in food systems, particularly in fermented dairy products such as yogurt remains limited. Considering the potential of red fruit oil in mitigating diabetes-related complications, the purpose of this study was to explore its integration into yogurt and evaluate its effects on physical and sensory qualities, bioactive compound content, and α-glucosidase inhibitory activity.
Materials and methods
Red fruit oil was extracted using a pressing method. The fresh red fruits were halved, and the cores were removed. The flesh
Extraction of red fruit oil
Red fruit oil was extracted using a pressing method. The fresh red fruits were halved, and the cores were removed. The flesh of three kilograms of red fruit was steamed at 100 °C for 15 min. Subsequently, 1,500 mL of hot water (80 °C) was added before pressing the mixture at a pressure of 4,000 - 4,500 psi. The oil fraction formed in the upper layer was collected as red fruit oil. Water was evaporated at 50 °C for 15 min to concentrate the oil extract.
Extraction of bioactive compounds from Yogurt
Bioactive compounds were extracted using a multistep solvent-extraction method. Yogurt (20 g) was weighed in a centrifuge tube, mixed with 20 mL 70% ethanol, and vortexed. The mixture was stored for 30 min and centrifuged at 4,000 rpm at 4 °C for 20 min. The supernatant was then transferred to an amber vial. The pellet was then mixed with 20 mL of hexane, vortexed, incubated for 30 min, and centrifuged again. The hexane supernatant was then combined with the ethanol extract. The combined extracts were first subjected to rotary evaporation to remove hexane, followed by ethanol removal. The resulting extracts were stored in foil-wrapped amber vials at 4 °C until further analysis.
Yogurt formulation
Yogurt was prepared by heating cow’s milk to 85 - 90 °C for 15 min and then cooling it to 42 °C. A 3% mixed starter culture (Streptococcus thermophilus and Lactobacillus bulgaricus), 3% granulated sugar, and 10% skim milk powder were added. The mixture was then incubated at 37 °C for approximately 16 h. After fermentation, the yogurt was divided into four groups and enriched with red fruit oil at concentrations of 0% (F0), 5% (F1), 10% (F2) and 15% (F3). All samples were homogenized and stored at 4 °C prior to analysis. The formulation followed the method described by Jonathan et al. [14] with dosage modifications based on the method of Lestari et al. [13].
Physical properties
The pH was measured using a calibrated digital pH meter. A 25 mL yogurt sample was poured into a beaker, and the electrode was immersed until the pH stabilized [15]. Titratable Acidity (TA) was determined by titration, as described by Jonathan et al. [14]. Ten grams of yogurt was diluted 100 times, and 10 mL of the dilution was titrated with 0.1 N NaOH using phenolphthalein indicator. The results are expressed as the percentage of lactic acid. The viscosity was measured using a viscometer with spindle No.4 at 30 rpm. A 150 mL yogurt sample was placed in a beaker and the viscosity was recorded on the device display. Color was analyzed using a chromameter to determine the L* (lightness), a* (red-green), and b* (yellow-blue) values.
Sensory evaluation
This study was approved by the Ethics Committee for Research Involving Human Subjects of IPB University (No. 1640/IT3. KEPMSM-IPB/SK/2025). All panelists signed an informed consent form before the tests. Hedonic Testing was conducted with 30 semi-trained panelists to evaluate their preferences for color, aroma, taste, mouthfeel, aftertaste, and overall acceptability on a 9-point scale (1 = extremely dislike, 9 = extremely like). An overall score of > 4.5 was considered acceptable. The inclusion criteria were age 18 - 40 years, no olfactory/taste disorders, no milk allergy, and willingness to follow the protocol. The exclusion criteria were diabetes, digestive disorders, medications, special diets, smoking, and conditions affecting sensory perception.
Quantitative Descriptive Analysis (QDA) was performed by trained panelists to quantify the sensory attributes of red fruit oil yogurt. Panelists were selected based on the inclusion and exclusion criteria. The training included focus group discussions (FGD), standard reference calibration, and scoring practices (0 - 15 scale). Attributes were developed using FGD and confirmed using real samples. The panelists rated the samples individually in a controlled environment using randomized 3-digit codes. QDA was conducted for the F0 - F3 samples.
Antioxidant activity (CUPRAC method)
Antioxidant capacity was assessed using the Cupric Reducing Antioxidant Capacity (CUPRAC) method, as described by Nurcholis et al. [16]. A 50 μL sample extract was added to a microplate well containing 50 μL of 0.01 M CuCl₂.2H₂O, 50 μL of 0.0075 M neocuproine, and 50 μL of 1 M ammonium acetate buffer (pH 7). The mixture was incubated in the dark for 30 min at room temperature, and absorbance was measured at 450 nm using a microplate reader. The results are expressed as μmol Trolox equivalents per gram of extract (μmol TE/g). Calibration was based on Trolox standards (0 - 400 μmol), and all analyses were performed in triplicate.

Selected formula
The optimal yogurt formulation was determined based on the combination of two main criteria: Antioxidant activity and sensory properties. The formulation with the highest antioxidant activity was selected and evaluated for its acceptability. Although sensory preference is important, functional benefits were prioritized in this study given the aim of developing a functional food to support blood glucose control. The selected formula was used for the bioactivity analyses.
Total flavonoid content
A 0.01 g sample was dissolved in 10 mL of a mixture of 1 mL DMSO and ethanol to obtain a 1,000 ppm solution, which was homogenized using a vortex. Then, 0.5 mL of the solution was mixed with 0.15 mL of 15% NaNO₂, incubated for 6 min, followed by 0.15 mL of AlCl₃, incubated for 60 min, and then 2 mL of NaOH and 2 mL of distilled water were added. The mixture was incubated for 15 min and the absorbance was measured at 420 nm using a UV-Vis spectrophotometer. Quercetin was used as a standard (0 - 200 μg/mL), and the results are expressed as mg quercetin equivalent per gram sample (mg QE/g).
Total phenolic content
The total phenolic content was determined using the Folin-Ciocalteu method. A 0.01 g sample was dissolved in 10 mL of a DMSO-water mixture to yield 1,000 ppm, vortexed, and 100 μL was reacted with 2 mL of 7% sodium carbonate. After 5 min in the dark, 1 mL of diluted Folin–Ciocalteu reagent (1:10) was added. The mixture was incubated for 30 min in the dark at room temperature and the absorbance was measured at 780 nm. Gallic acid (0 - 200 μg/mL) was used for the calibration. The results are expressed as mg gallic acid equivalents per gram of sample (mg GAE/g). All measurements were performed in triplicates.
α-Glucosidase inhibitory activity
In vitro α-glucosidase inhibition was tested in yogurt samples enriched with red fruit oil and compared with that of acarbose, a positive control. The procedure followed the methods of Chaerunnisa et al. [17] and Agustin et al. [6], with modifications. The assay involved a sample solution (S1), control (S0), blank (B1), and blank control (B0). The substrate used was p-nitrophenyl α-D-glucopyranoside (pNPG) in 0.1 M phosphate buffer (pH 7.0). The enzyme was prepared by dissolving 1 mg of α-glucosidase in 100 mL of phosphate buffer to obtain 0.04 an activity. reaction mixture: 10 µL of sample, 50 µL of phosphate buffer, 25 µL of enzyme solution, and 25 µL of 10 mM pNPG and incubated at 37 °C for 30 min. The reaction was stopped with 100 µL of 0.2 M sodium carbonate, and the absorbance was measured at 410 nm using a microplate reader. All tests were performed in triplicate. The percentage of inhibition was calculated as follows:

Statistical analysis
Data were analyzed using IBM SPSS Statistics (version 26.0; IBM Corp., Armonk, NY, USA) and are presented as mean ± standard deviation (SD). One-way ANOVA was used to evaluate differences among the groups. If significant differences were found (p < 0.05), Duncan’s Multiple Range Test (DMRT) was used for post-hoc analysis. Independent t-tests were performed for comparisons between two groups (e.g., bioactive content or enzyme inhibition).
Results and discussion
The following results describe the effects of red fruit oil addition on yogurt, as a continuation of the extraction and formulation stages described previously.
Physicochemical and biological activities of red fruit oil
The red fruit oil used in this study exhibited characteristics consistent with previous reports. Chemical and biological analyses revealed that the oil is rich in bioactive compounds with high functional capacity. In addition, the physicochemical parameters of red fruit oil included a pH of 5.81 ± 0.10, viscosity of 6.86 ± 0.06 cP, and ash content of 0.11 ± 0.003%, indicating good oil quality. These findings strengthen the evidence that red fruit oil is not only a source of lipids but also contains bioactive compounds that play a crucial role in antioxidant and antidiabetic activities.
Table 1 Physicochemical and biological activities of red fruit oil.
Category |
Parameter |
Value (mean ± SD) |
Physicochemical |
pH |
5.81 ± 0.10 |
Viscosity (cP) |
6.86 ± 0.06 |
|
Ash content (%) |
0.11 ± 0.003 |
|
Biological activities |
ɑ-Glukosidase inhibitory activity (1,000 ppm, %) |
88.72 ± 0.34 |
α-Glucosidase inhibitory activity of red fruit oil (88.72 ± 0.34%) demonstrated stronger inhibition compared with acarbose at 1 ppm (67.21 ± 1.8%), highlighting its potential as a natural α-glucosidase inhibitor.
Physical characteristics of Yogurt
Each yogurt formulation exhibited significant differences (p < 0.05) in terms of physical and chemical characteristics. The analyzed parameters were pH, viscosity (cP), titratable acidity (TA), and color. The physical properties of each formulation are shown in Table 2 and Figure 1.
Table 2 Effect of red fruit oil addition on the physical and chemical properties of yogurt.
Attribute |
Formulation |
SNI 2009 |
|||
F0 (0%) |
F1 (5%) |
F2 (10%) |
F3 (15%) |
||
pH value |
4.60 ± 0.01b |
4.69 ± 0.01ab |
4.72 ± 0.01a |
4.70 ± 0.01a |
3.8 - 4.8 |
Viscosity (cP) (1×102) |
25.69 ± 0.15a |
23.76 ± 0.21ab |
20.77 ± 0.12bc |
15.33 ± 0.10c |
- |
Titratable Acidity (TA) (%) |
1.35 ± 0.0a |
1.35 ± 0.0ab |
0.90 ± 0.0b |
0.90 ± 0.0b |
0.5 - 2.0 |
Color Parameters |
|
|
|
|
|
L* (lightness) |
87.31 ± 1.98a |
56.60 ± 0.31ab |
48.01 ± 0.06bc |
39.33 ± 0.33c |
- |
a* (red-green) |
0.81 ± 0.03b |
36.0 ± 0.08ab |
35.98 ± 0.05ab |
38.16 ± 1.20a |
- |
b* (yellow-blue) |
11.13 ± 0.88bc |
5.96 ± 0.27b |
14.85 ± 0.10ac |
17.60 ± 0.52a |
- |
image |
|
|
|
|
- |
Description: Values are expressed as mean ± standard deviation. Means within the same row followed by different superscript letters (a, b, and c) indicate significant differences at p < 0.05. F0 = Yogurt with 0% red fruit oil; F1 = 5%; F2 = 10%; F3 = 15%. TA = Titratable Acidity. SNI 2009 = Indonesian National Standard for yogurt (SNI 01-2981-2009).

Figure 1 The Visual Appearance of Yogurt Supplemented with Different Concentrations of Red Fruit Oil (Pandanus conoideus Lam.).
The addition of red fruit oil significantly influenced the acidity profile of yogurt, particularly pH and titratable acidity (TA). As the oil concentration increased, the pH increased, while the TA decreased. This phenomenon may be attributed to the high fat content of red fruit oil, which can suppress the metabolic activity of lactic acid bacteria, thereby reducing lactic acid production during fermentation [18]. Dilution effects and possible interactions between oil compounds and bacterial cell membranes may also contribute to a reduced acidification rate [19].
The viscosity decreased with higher concentrations of red fruit oil. This is likely due to the disruption of the protein gel network formation caused by the high lipid content. Fatty acids, such as oleic and palmitic acids, may interfere with the hydrophobic interactions between casein micelles, weakening the gel structure and increasing the risk of syneresis [20]. An unbalanced fat-to-protein ratio has been reported to impair gel stability and increase whey separation in fermented dairy products [21].
Color is a critical parameter in consumer perception, and is closely associated with visual appeal and taste expectations [20]. Yogurt color was measured using the Hunter L*, a*, and b* color systems, where L* indicates lightness, a* represents the red-green axis, and b* indicates the yellow-blue axis. Red fruit oil addition significantly altered yogurt color parameters (Table 1). The highest L* value was found in the control (F0), which progressively decreased with increasing oil concentrations in F1, F2 and F3. This trend aligns with the deep red hue of red fruit oil, which reduces the product brightness.
The a* value (redness) significantly increased with increasing oil concentration. F0 showed the lowest a*, whereas F3 exhibited the highest a *, reflecting the contribution of carotenoid pigments, particularly β-carotene, which is naturally present in red fruit oil [18]. However, no significant difference was observed between F2 and F3, suggesting that the saturation point of red pigmentation was reached at 10% concentration.
Similarly, the b* value (yellowness) increased significantly in F2 and F3 compared to that in F1 and F0. The lack of a significant difference between F2 and F3 indicates that carotenoid intensity peaked at 10%, and further oil addition did not enhance the yellowness. Overall, the color changes observed in F1 - F3 compared to F0 were attributed to the natural carotenoid pigments in red fruit oil, imparting a reddish-yellow hue. Variations among the formulations were primarily due to differences in the composition and concentration of the red fruit oil.
In conclusion, the addition of red fruit oil increased the pH and color intensity but decreased the viscosity and titratable acidity. These effects highlight the potential of bioactive ingredients to modulate the physical characteristics of yogurt. Further studies are recommended to determine the optimal concentration that balances physicochemical properties, sensory acceptance, and functional benefits.
Sensory characteristics
Hedonic testing
The hedonic test was designed to assess consumer acceptability of yogurt enriched with red fruit oil. Conducted with 33 semi-trained panelists, each sample was evaluated independently according to Indonesian National Standard (SNI 01-2346-2006) guidelines. The results revealed a significant difference (p < 0.05) in the color attribute, whereas other attributes showed no significant differences, although variations in the mean scores were observed among samples (Table 3).
Table 3 Hedonic test results of red fruit oil yogurt.
Attribute |
Formula |
||
F1 (5%) |
F2 (10%) |
F3 (15%) |
|
Appearance |
7.03 ± 1.4ab |
7.30 ± 1.4ab |
6.33 ± 1.5b |
Aroma |
6.58 ± 1.4a |
6.39 ± 1.4ab |
5.70 ± 1.8b |
Texture |
6.64 ± 1.5a |
6.88 ± 1.4a |
6.21 ± 1.4a |
Taste |
5.52 ± 1.6a |
4.94 ± 1.9a |
4.39 ± 1.7a |
Mouthfeel |
5.39 ± 1.6a |
4.91 ± 2a |
4.58 ± 1.8a |
Aftertaste |
5.00 ± 1.7a |
4.79 ± 2a |
4.48 ± 1.8a |
Overall |
5.97 ± 1.5a |
5.38 ± 1.7ab |
4.85 ± 1.7b |
Description: Values are expressed as mean ± standard deviation. Different superscript letters within a row indicate significant differences (p < 0.05). F0 = Yogurt with 0% red fruit oil; F1 = 5%; F2 = 10%; F3 = 15%.
Color is a key sensory parameter that influences consumer perceptions. The addition of red fruit oil provided a natural pink hue owing to its high β-carotene content, which is a major carotenoid with antioxidant properties [18]. F2 had the highest color score (7.30), which was significantly higher than that of F3 (6.33), which was perceived as overly intense and unnatural (p < 0.05), indicating that excessive β-carotene may reduce visual acceptability [19].
Yogurt aroma is influenced by fermentation-derived volatiles and aromatic compounds from red fruit oil, including aldehydes, free fatty acids, and phenols [18,19]. F1 had the highest aroma score (6.58), whereas F3 had the lowest (5.70), although it was not significantly different. This decline may be due to the high levels of free fatty acids (e.g., palmitic and linoleic acids) and free phenolics, which can produce sharp or unfamiliar aromas at high concentrations [20,24].
Texture preferences favored soft and thick consistencies. F2 scored (6.88), and F3 scored (6.21). High lipid content, particularly oleic and palmitic acids, can interfere with protein gel stability, cause syneresis, and result in thinner, less homogeneous textures [20].
The taste scores declined as the oil concentration increased. F1 received the highest score (5.52), whereas F3 received the lowest score (4.39). This may be due to dominant compounds, such as β-carotene, free fatty acids, and phenolics, which contribute astringent, bitter, or foreign tastes uncommon in traditional yogurt at high levels [20,25]. Additionally, the oxidizable nature of red fruit oil may lead to rancidity if not properly stabilized.
Mouthfeel, which is associated with creaminess and smoothness, was highest in F1 (5.39) and lowest in F3 (4.58). A high lipid content (99.7 g/100 g oil), mostly unsaturated fatty acids, can create an oily layer in the mouth and diminish perceived creaminess [18,20].
The aftertastes followed a similar trend. F1 had the highest score (5.00), whereas F3 had the lowest (4.48). Bioactive compounds, such as β-carotene, free phenolics, and phytosterols, can cause lingering bitter, greasy, or astringent sensations after consumption [20].
The overall liking score was highest in F1 (5.97), followed by F2 (5.38) and F3 (4.85). This decline was likely due to an imbalance in sensory attributes caused by excessive bioactive compound content. While compounds such as β-carotene, tocopherol, and phenolics provide health benefits, excessive levels may negatively impact sensory quality [20,26].
Quantitative Descriptive Analysis (QDA)
Quantitative Descriptive Analysis (QDA) was performed by 12 trained panelists aged 24 - 39 years (3 males and 4 females) from diverse regions in Indonesia. The results indicated that red fruit oil significantly affected the sensory profile of the yogurt (p < 0.05). The yogurt-specific aroma and taste intensity decreased with increasing oil concentration, while the red fruit-specific attributes (aroma, taste, and aftertaste) increased linearly from F1 to F3. The addition of red fruit oil also reduced the thickness of the mouthfeel and visual viscosity while increasing the oily sensation and astringency. F3 displayed the highest intensity of red fruit characteristics but also had the strongest astringent aftertaste and oily mouthfeel, which may lower overall acceptability. F0 showed the most classic yogurt profile, whereas F1 and F2 provided a balanced sensory profile between yogurt characteristics and red fruit oil enhancement.
Table 4 Results of Focus Group Discussion (FGD) on sensory attributes and their definitions for yogurt product with red fruit oil.
Sensory attributes |
Description |
Yogurt-specific aroma |
Characteristic sour aroma resulting from lactic acid fermentation, resembling the natural aroma of plain yogurt. |
Milky aroma |
Fresh milk-like aroma described as creamy, smooth, and slightly sweet. |
Cheesy aroma |
Cheese-like aroma, associated with fermentation-derived notes and a slight mustiness. |
Rancid aroma |
Oxidized fat aroma, similar to the smell of reused or poorly stored cooking oil. |
Red fruit-specific aroma |
Distinctive aroma of red fruit (Pandanus conoideus L.), recognizable and specific to the added ingredient. |
Sweet taste |
Sweetness sensation perceived on the tongue. |
Sour taste |
Sourness sensation, typical of fermented dairy products. |
Yogurt specific taste |
Combined taste of sour and slightly creamy, characteristic of yogurt. |
Milky taste |
Creamy, fresh, smooth, and mildly sweet taste, representing the sensory profile of fresh milk. |
Red fruit-specific taste |
Typical taste of red fruit (Pandanus conoideus L.), contributing a unique fruity note. |
Thick mouthfeel |
Thickness or viscosity sensation, giving a sense of heaviness during swallowing. |
Oily mouthfeel |
Slick and slightly oily mouth-coating sensation, resembling the texture of oil. |
Astringent mouthfeel |
Astringent or dry sensation in the mouth, often felt as puckering. |
Sour aftertaste |
Lingering sourness, defined as the acidic taste that persists in the mouth approximately 30 seconds after swallowing the sample. |
Sweet aftertaste |
Persistent sweetness, defined as the intensity of sweet taste perceived approximately 30 seconds after swallowing the sample. |
Astringent aftertaste |
Residual astringent sensation, referring to the tactile dryness felt on the tongue and inner cheeks 30 seconds post-ingestion. |
Red fruit-specific aftertaste |
Residual red fruit aftertaste, defined as the characteristic flavor of red fruit (Pandanus conoideus L.) that remains in the mouth 30 seconds post-ingestion. |
Table 5 Sensory attributes of red fruit oil yogurt evaluated using Quantitative Descriptive Analysis (QDA)
Sensory attributes |
F0 |
F1 |
F2 |
F3 |
Yogurt-specific aroma |
8.62 ± 0.32a |
6.55 ± 0.49b |
4.75 ± 0.48c |
3.54 ± 0.47c |
Milky aroma |
4.63 ± 0.43a |
2.63 ± 0.37b |
0.00c |
0.00c |
Cheesy aroma |
5.43 ± 0.55a |
0.71 ± 1.08b |
0.09 ± 0.17b |
0.08 ± 0.18b |
Rancid aroma |
0.00a |
0.05 ± 0.08a |
0.06 ± 0.15a |
0.09 ± 0.18a |
Red fruit-specific aroma |
0.00d |
3.79 ± 0.27c |
4.74 ± 0.50b |
5.85 ± 0.43a |
Sweet taste |
7.47 ± 0.34a |
1.67 ± 0.40b |
0.00c |
0.00c |
Sour taste |
5.88 ± 0.45a |
3.47 ± 0.47b |
2.90 ± 0.43c |
0.00d |
Yogurt specific taste |
7.44 ± 0.47a |
3.47 ± 0.28b |
2.56 ± 0.38c |
1.81 ± 0.28d |
Milky taste |
5.62 ± 0.35a |
0.00b |
0.00b |
0.00b |
Red fruit-specific taste |
0.00d |
9.71 ± 0.43c |
11.58 ± 0.35b |
12.63 ± 0.36a |
Thick mouthfeel |
12.47 ± 0.54d |
11.53 ± 0.50c |
10.50 ± 0.56b |
9.67 ± 0.36a |
Oily mouthfeel |
0.00d |
2.64 ± 0.49c |
4.67 ± 0.37b |
6.53 ± 0.45a |
Astringent mouthfeel |
0.00c |
3.45 ± 0.70b |
5.77 ± 0.69a |
7.28 ± 0.75a |
Sour aftertaste |
4.63 ± 0.41a |
2.66 ± 0.37b |
1.77 ± 0.25c |
0.93 ± 0.32d |
Sweet aftertaste |
4.77 ± 0.49a |
0.00b |
0.00b |
0.00b |
Astringent aftertaste |
0.00d |
1.55 ± 0.47c |
3.58 ± 0.49b |
5.44 ± 0.54a |
Red fruit-specific aftertaste |
0.00d |
1.78 ± 0.28c |
3.79 ± 0.26b |
6.65 ± 0.48a |
White appearance |
6.54 ± 0.50a |
0.00b |
0.00b |
0.00b |
Reddish appearance |
0.00d |
3.67 ± 0.32c |
5.66 ± 0.30b |
7.63 ± 0.34a |
Yellowish appearance |
0.00b |
0.79 ± 0.24a |
0.00b |
0.00b |
Viscosity appearance |
7.61 ± 0.35a |
5.43 ± 0.37b |
4.47 ± 0.35c |
3.62 ± 0.26d |
Description: Values are presented as mean ± standard deviation. Different superscript letters in the same row indicate significant differences (p < 0.05). F0 = Yogurt without red fruit oil. F1 = 5%, F2 = 10%, and F3 = 15% red fruit oil enrichment. A score of 0 indicates that the attribute was not detected by the panelists in the sample.
The selection of sensory evaluation methods in this study was based on both national and international standards. The hedonic test was applied to assess consumer preference and acceptance levels, following the Indonesian National Standard SNI 01-2346-2006, which recommend the 9-point scale (1 = extremely dislike, 9 = extremely like) as the most reliable tool for quantifying consumer liking in dairy products. Meanwhile, the Quantitative Descriptive Analysis (QDA) followed ISO 13299:2016, involving trained panelists using a 15-point intensity scale to describe sensory attributes such as aroma, taste, texture, and aftertaste. This combined use of Hedonic and QDA methods enables a comprehensive sensory evaluation, hedonic testing reflects consumer perception, whereas QDA provides a detailed understanding of attribute intensity influencing acceptability. Such integration has been widely used in recent dairy product studies [21,27-29].
Antioxidant activity (CUPRAC method)
Following the evaluation of yogurt physical and sensory characteristics, this section discusses its functional potential based on antioxidant activity, which was measured using the CUPRAC method. The cupric-reducing antioxidant capacity (CUPRAC) method evaluates the ability of yogurt to neutralize free radicals by reducing Cu²⁺ ions to Cu⁺ ions. The absorbance was measured at 450 nm. This method was selected because of its sensitivity to both polar and non-polar antioxidant compounds, making it suitable for yogurt systems containing oils [16,30]. The antioxidant activity values, expressed as μmol Trolox equivalents (TE) per gram, are presented in Figure 2.
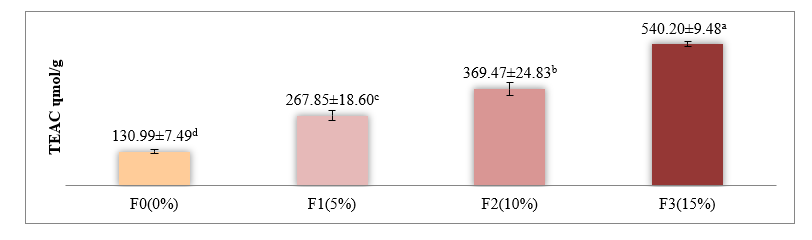
Figure 2 Antioxidant activity of yogurt with red fruit oil using the CUPRAC method (μmol TE/g).
Description: Values are presented as the mean ± standard deviation. Different superscript letters within the same row indicate significant differences (p < 0.05). F0 = yogurt without red fruit oil; F1 = 5%; F2 = 10%; F3 = 15%. CUPRAC, Cupric Reducing Antioxidant Capacity; TE = Trolox Equivalent.
The results showed that The add ition of red fruit oil significantly increased the antioxidant activity of yogurt (p < 0.05). The highest value was observed in F3 (15%) at 540.20 ± 9.48 μmol TE/g extract (equivalent to 615.83 μmol TE/g sample), followed by F2 at 369.47 μmol TE/g, and F1 at 267.55 μmol TE/g. The control group, F0, had the lowest antioxidant activity at 130.99 ± 7.48 μmol TE/g. Post hoc Duncan’s Multiple Range Test (DMRT) showed no significant difference between F3 and F2, but both were significantly higher than F1 and F0. This trend indicates a positive correlation between red fruit oil concentration and antioxidant capacity up to a certain saturation point.
The increase in antioxidant activity is closely related to the presence of bioactive compounds in red fruit oil. These compounds include flavonoids, phenolics, β-carotene, total carotenoids, tocopherol, and α-tocopherol [18,22,24]. The CUPRAC assay is based on the electron-donating ability of antioxidant compounds, especially those containing hydroxyl groups or double bonds conjugated to Cu²⁺ ions. The higher the concentration of these compounds, the greater the antioxidant activity.
Phenolic compounds play an important role in the redox capacity because their aromatic structure allows stable hydrogen atom donation to neutralize free radicals. In addition to their antioxidant effects, phenolics exhibit antimicrobial activity and contribute to the characteristic aroma of the product [23]. Król et al. [7] and Tarchi et al. [31] also reported that increasing the phenolic content in yogurt, for example, through the addition of olive leaf extract, was positively correlated with antioxidant capacity due to synergistic mechanisms. Phenolics are generally more stable when added after fermentation, which was the approach used in this study. In addition to phenolics, red fruit oil contains high levels of tocopherol (502 ± 2 ppm) and carotenoids (2,003 ± 75 ppm) [20].
Tocopherol is a primary antioxidant that sacrifices itself during oxidative stress and protects cellular lipids and proteins. Carotenoids help neutralize singlet oxygen and stabilize free radicals through electron transfer [32]. Flavonoids also contribute to total antioxidant capacity. However, studies have shown that the relationship between the flavonoid content and antioxidant activity is not always linear. This is due to variations in stability and interactions with the food matrix, which can influence their effectiveness [16].
The selection of the CUPRAC method in this study was grounded on its theoretical advantages over other commonly used antioxidant assays such as DPPH, FRAP, and ABTS. Unlike radical-based assays, the CUPRAC method operates under a redox mechanism that involves the reduction of Cu²⁺–neocuproine complexes by antioxidant compounds, allowing simultaneous detection of both hydrophilic and lipophilic antioxidants [16,33]. This characteristic is particularly relevant for yogurt systems containing oil-based ingredients like red fruit oil, which harbor both polar (phenolics, flavonoids) and non-polar (tocopherols, carotenoids) antioxidant fractions.
In addition, the CUPRAC assay provides more stable absorbance values and higher reproducibility under near-neutral pH conditions compared to acidic or radical-mediated assays. Therefore, the enhanced antioxidant activity observed in red fruit oil yogurt may not solely reflect the concentration of bioactive compounds but also the methodological suitability of CUPRAC in accurately quantifying total antioxidant potential in complex dairy matrices. This theoretical consideration reinforces the validity of the CUPRAC results and supports the reliability of interpreting the functional properties of red fruit oil-enriched yogurt.
Compared to previous studies, such as that by Durmus et al. [34] on yogurt with mulberry addition (5.73 μmol TE/g) and Arruda et al. [35] using araticum fruit (91.02 μmol TE/g), the antioxidant activity values observed in this study were considerably higher. This highlights the potential of red fruit oil as a natural antioxidant for yogurt products. These findings are supported by those of previous studies that have used different methods. For instance, antioxidant activity measured by DPPH showed an IC₅₀ of 14.45 ppm and 81.02% inhibition [9], while the FRAP method reported 1.392×10⁻³ g ATE/g extract [22]. Based on these results, it can be concluded that red fruit oil significantly improved the antioxidant activity of yogurt. This improvement was primarily attributed to the contribution of phenolic compounds, carotenoids, and tocopherols. Higher antioxidant activity not only indicates better protection against oxidative stress but also suggests potential antidiabetic properties, such as α-glucosidase inhibition, which will be discussed in the next section.
Selected formula
The selection of the optimal yogurt formulation for this study was based on two primary criteria: antioxidant activity and consumer acceptance as measured by hedonic testing. Formula F3 (15%) showed the highest antioxidant activity, reaching 540.20 μmol TE/g. However, it had the lowest sensory acceptance, particularly for taste, mouthfeel, aftertaste, and the overall attributes. Conversely, F1 (5%) had the highest hedonic scores, but exhibited considerably lower antioxidant activity. Formula F2 (10%) was selected as the best formulation because it provided an optimal balance between the functional efficacy and sensory acceptance. Its antioxidant activity reached 369.47 μmol TE/g and was not significantly different from that of F3. In the hedonic test, F2 achieved the highest score for color (7.30 ± 1.4) and an overall liking score of 5.38 ± 1.7, which fell within the acceptable range. In addition, F2 exhibited balanced physical and sensory characteristics, including an attractive pink color, homogeneous consistency, and a moderate intensity of red fruit flavor. Although F2’s hedonic scores were slightly lower than those of F1, this formulation still met the primary objective of developing yogurt as a functional food. It offers health benefits without compromising consumers’ preferences. Therefore, F2 was selected as the optimal formulation and used for further analysis of total flavonoid content, total phenolic content, and α-glucosidase inhibitory activity.
Bioactive compound content
To further investigate the functional potential of the red fruit oil yogurt, the levels of key bioactive compounds, total flavonoids, and total phenolics were analyzed. These compounds are widely recognized for their antioxidant and antidiabetic properties, particularly their ability to inhibit oxidative stress and regulate postprandial glucose levels. The quantitative results of total flavonoid and phenolic contents in yogurt with and without red fruit oil addition are presented in Figure 3.
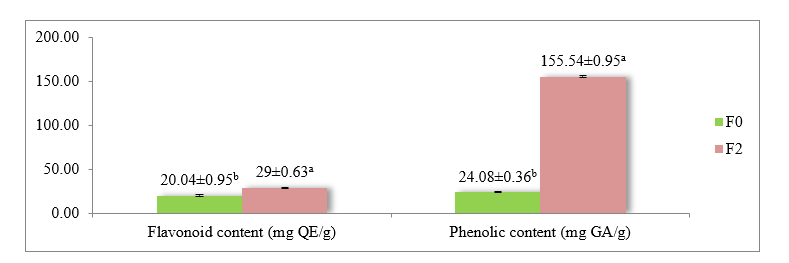
Figure 3 Total flavonoid and phenolic contents of yogurt.
Description: Values are presented as mean ± standard deviation (SD). Different superscript letters (a, b) in the same column indicate statistically significant (p < 0.05). F0 = Yogurt formulation with 0% red fruit oil. F2 = Yogurt formulation with 10% red fruit oil.
The total phenolic content (TPC) was determined using the Folin-Ciocalteu spectrophotometric method, a well-established and sensitive approach for evaluating phenolics in dairy matrices. This method is based on the redox reaction between phenolic hydroxyl groups and the Folin-Ciocalteu reagent, producing a blue complex measured at 765 nm using a UV-Vis spectrophotometer [7].
Similarly, the total flavonoid content (TFC) was quantified using the aluminum chloride (AlCl₃) spectrophotometric method, in which flavonoid molecules form a stable yellow complex with Al³⁺ ions measurable at 510 nm. This method provides high selectivity for flavones and flavonols, making it suitable for evaluating lipid-enriched systems such as red fruit oil yogurt [21]. Quercetin and gallic acid were used as standard compounds for TFC and TPC, respectively, and results were expressed as mg QE/g and mg GAE/g.
As shown in Figure 3, yogurt enriched with red fruit oil (F2) contained significantly higher levels of both flavonoids and phenolics than the control (F0) (p < 0.05). The flavonoid content increased from 20.04 ± 0.95 mg QE/g in F0 to 29.00 ± 0.62 mg QE/g in F2. Meanwhile, total phenolic content rose markedly from 24.08 ± 0.36 mg GAE/g in F0 to 155.54 ± 0.95 mg GAE/g in F2. This substantial increase was attributed to the presence of bioactive compounds in red fruit oil (Pandanus conoideus Lam.), including flavonoids, phenolics, tocopherols, and carotenoids [22,36]. Flavonoids contain aromatic hydroxyl groups that function as hydrogen or electron donors, whereas phenolic compounds neutralize free radicals by stabilizing their aromatic ring structures. These compounds synergistically contribute to both antioxidant mechanisms and α-glucosidase inhibition. In addition to the added ingredients, the fermentation process involving lactic acid bacteria (LAB) increases the concentration of bioactive compounds. LAB-produced β-glucosidase enzymes can hydrolyze glycosylated compounds into more active aglycones, thereby enhancing the bioavailability of both flavonoids and phenolics [37]. The observed increase in bioactive compounds following the addition of red fruit oil is consistent with the results of previous studies. Shori and Baba [38]reported increases in both flavonoid and phenolic contents in yogurt enriched with Allium sativum. Similarly, Hamed et al. [27] demonstrated a significant increase in phenolic compounds in yogurt after addition of Pelargonium graveolens oil. These findings reinforce the effectiveness of combining antioxidant-rich functional ingredients, such as red fruit oil, with fermentation, as a promising approach to enhance yogurt as a functional food with high biological activity.
α-Glucosidase inhibitory activity
To evaluate the potential of yogurt as an anti-hyperglycemic functional food, this study employed an in vitro method to assess the α-glucosidase inhibitory activity. The α-glucosidase inhibitory assay was chosen because it is one of the most widely accepted in vitro screening methods to evaluate the antidiabetic potential of food matrices through the inhibition of carbohydrate-hydrolyzing enzymes. This method enables quantitative determination of enzyme inhibition by monitoring the hydrolysis of p-nitrophenyl-α-D-glucopyranoside (pNPG) into p-nitrophenol, which can be spectrophotometrically measured at 405 nm. Compared to other enzymatic assays such as α-amylase inhibition, the α-glucosidase assay provides a more direct representation of postprandial glucose regulation since α-glucosidase catalyzes the terminal step of carbohydrate digestion [32]. Moreover, this assay exhibits high sensitivity and reproducibility for both polar and non-polar inhibitors, making it suitable for complex food systems like red fruit oil yogurt that contain both hydrophilic phenolics and lipophilic antioxidants [33].
This enzyme was selected because of its role in catalyzing the breakdown of carbohydrates into glucose. The inhibition of α-glucosidase can reduce postprandial blood glucose spikes, thereby preventing hyperglycemia and the onset of type 2 diabetes. Yogurt samples, with and without red fruit oil, were compared for their inhibitory activity against α-glucosidase. Acarbose was used as the positive control. The results of the α-glucosidase inhibition by F0, F2, and acarbose are presented in Figure 4.
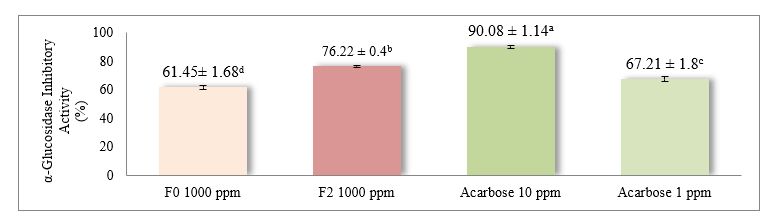
Figure 4 α-Glucosidase inhibitory activity of yogurt and acarbose.
Description: Values are expressed as mean ± standard deviation (SD). Different superscript letters (a, b, c and d) indicate statistically significant differences at p < 0.05, based on an independent sample t-test. F0 = Yogurt without red fruit oil (0%), F2 = Yogurt with 10% red fruit oil, Acarbose = Positive control at 1 ppm and 10 ppm concentrations.
The results showed that F2 yogurt (10% red fruit oil) had significantly higher α-glucosidase inhibitory activity than F0 yogurt. The inhibitory effect of F2 was 78.08 ± 0.37%, whereas F0 was 64.46 ± 1.56%. The inhibition level of F0 at 1,000 ppm was slightly lower than that of acarbose at 1 ppm (67.21 ± 1.8%). Notably, F2 exceeded the inhibitory effect of acarbose at 1 ppm but was still lower than that of acarbose at 10 ppm (90.08 ± 1.14%).
In this study, acarbose was used as a positive control at two reference concentrations (1 ppm and 10 ppm) to represent low and high levels of inhibitory activity, respectively. These levels were selected to ensure the measurement remained within the linear response range of the assay and to allow clear differentiation between moderate and strong inhibition. Although these exact concentrations were determined empirically in this work, they are within the general range commonly applied in in-vitro α-glucosidase inhibitory assays [41]. All acarbose solutions were freshly prepared and analyzed under the same assay conditions as the yogurt samples to maintain consistency.
The assay used 10 mM p-nitrophenyl-α-d-glucopyranoside (p-NPG) as the substrate, α-glucosidase (0.04 U/mL), phosphate buffer (pH 7.0), and 200 mM sodium carbonate. Absorbance was measured using an ELISA reader, and inhibition was calculated based on the reduction in enzyme activity as determined by the color intensity resulting from p-NPG hydrolysis. These results demonstrate that F0, without red fruit oil, exhibited promising anti-hyperglycemic potential, with an inhibitory effect similar to that of low-dose acarbose. The relatively high inhibitory activity of F0 may be attributed to bioactive compounds formed during milk fermentation. Lactic acid bacteria (LAB) produce bioactive peptides, organic acids, and β-glucosidase, which can convert glycosylated phenolic compounds into more active aglycones. Previous studies have indicated that the presence of phenolic compounds and peptides during fermentation can enhance enzyme inhibitory capacity [37,42]. The addition of red fruit oil (F2) further enhanced α-glucosidase inhibition. This effect is associated with its bioactive content, particularly that of flavonoids, phenolics, and tocopherols [36]. Several in vivo studies have confirmed that red fruit oil can lower the blood glucose levels in animal models [11-13,43]. Some bioactive compounds in red fruit oil, such as flavonoids and phenolics, are polar and can interact directly with the active sites of α-glucosidase. Their hydroxyl groups may bind to the active site, thereby competing with the substrate (p-NPG) and resulting in competitive inhibition [39,40,44]. In contrast, non-polar compounds in red fruit oil, such as tocopherols, β-carotene, and unsaturated fatty acids, can exert competitive, non-competitive, or indirect inhibitory effects.
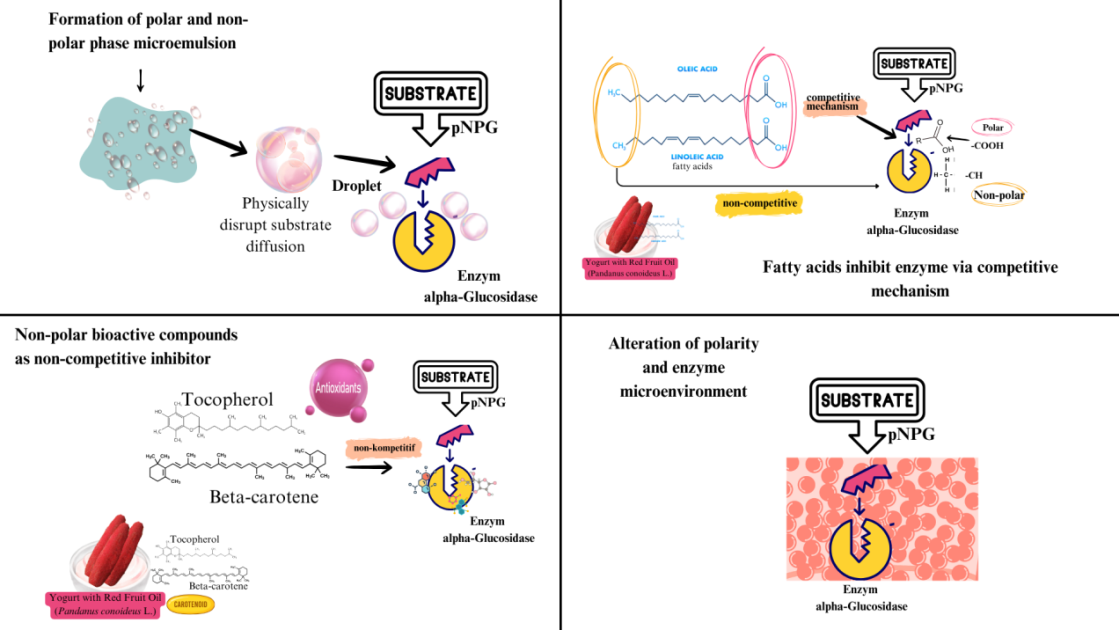
Figure 5 Mechanism of non-polar compounds inhibiting α-glucosidase.
In polar systems, nonpolar and polar phases may form microemulsions containing lipid droplets that are insoluble in water. These droplets can physically hinder substrate access to the enzyme active site. A previous study reported that certain wall materials used in microencapsulation might contribute to enzyme inhibition through indirect mechanisms [45]. For example, butyric oil alone did not exhibit any inhibitory effects. However, when encapsulated in quinoa protein, its inhibitory activity increased significantly. This indicates that encapsulation can disrupt or delay substrate diffusion toward the active site of the enzyme. Red fruit oil contains oleic and linoleic acids [46], which may inhibit α-glucosidase via two mechanisms.
The polar carboxyl groups (–COOH) of these fatty acids can bind to positively charged or polar sites on enzymes, resulting in competitive inhibition. Moreover, their long non-polar hydrocarbon chains (–CH) can bind to the surface of the enzyme, contributing to non-competitive inhibition. Su et al. [47] showed that these fatty acids compete with the substrate for the enzyme active site. Kinetic analysis indicated an increase in Km without a change in Vmax, suggesting a reduced substrate affinity. The Ki value of oleic acid was lower than that of linoleic acid, indicating stronger inhibitory potential. Although the secondary structure of the enzyme remained unchanged, microstructural modifications that could affect catalytic activity were observed. Furthermore, nonpolar bioactive compounds in red fruit oil, such as tocopherols, β-carotene, and carotenoids [24], may inhibit α-glucosidase activity through interactions with allosteric sites. Kinetic and molecular docking studies by Tshiyoyo et al. [48] revealed that large, branched, and non-polar molecules could bind to these sites, leading to enzyme inhibition.
This mechanism involves altering enzyme polarity and the surrounding microenvironment, which represents an indirect mode of inhibition that is distinct from competitive or non-competitive models. When non-polar compounds enter an enzyme system that typically functions in a polar environment, they can disrupt the structural and dynamic equilibrium, reducing the ability of the enzyme to interact with its substrate [40,49]. Skjold-Jørgensen et al. [49] also demonstrated that under polar conditions, the lid structure of lipase enzymes remains closed, preventing substrate access and reducing enzymatic activity. This supports the idea that polarity mismatch can negatively affect enzyme performance. In summary, yogurt with red fruit oil exhibited stronger α-glucosidase inhibitory activity than the control and low-dose acarbose. This reinforces its potential as an anti-hyperglycemic functional food. These findings are consistent with those of previous studies that reported increased enzyme inhibition when plant extracts were added to yogurt. For example, Shori and Baba [38] found that plain cow’s milk yogurt showed only 11.3 ± 0.4% inhibition, while yogurt enriched with Allium sativum reached 15.2 ± 0.4%.
Conclusions
This study demonstrated that the addition of red fruit oil (Pandanus conoideus L.) to yogurt significantly enhanced its functional properties, including antioxidant activity, total flavonoid and phenolic contents, and α-glucosidase inhibitory activity. The formulation containing 10% red fruit oil (F2) was selected as the optimal formula because it exhibited a balance between biological efficacy and sensory acceptability. F2 showed antioxidant activity of 369.47 μmol TE/g, flavonoid content of 29.00 mg QE/g, phenolic content of 155.54 mg GAE/g, and an α-glucosidase inhibitory activity of 78.08%, surpassing that of 1 ppm acarbose. From a sensory perspective, F2 had an appealing color, acceptable texture, and distinctive attributes that remained favorable to panelists. These findings support the potential of red fruit oil yogurt as a functional anti-hyperglycemic food product with good sensory quality, and highlight the possibility of utilizing local bioactive ingredients in the development of nutraceutical products.
Acknowledgements
The authors express their sincere gratitude to the Directorate General of Research and Development, Ministry of Higher Education, Science, and Technology (KEMDIKTISAINTEK), for providing funding support through the 2025 Fiscal Year Research Program Implementation Contract No. 006/C3/DT.05.00/PL/2025. Appreciation was also extended to the laboratory team at IPB University, West Java 16680, Indonesia for the support and facilities provided during the research process. The authors also thank the sensory panelists and all parties who contributed to the success of this study.
Declaration of generative AI in scientific writing
The authors acknowledge the use of generative AI tools (ChatGPT by OpenAI and Paperpal) in the preparation of this manuscript, specifically for language editing and grammar correction. No content generation, data analysis, or interpretation was performed by AI. The authors take full responsibility for the content and conclusions of this work.
CRediT author statement
Nur Haerati: Conceptualization; Methodology; Data Curation; Investigation; Formal analysis; Writing - Original Draft; Writing - Review & Editing; Funding acquisition. Rimbawan: Conceptualization; Methodology; Supervision; Funding acquisition. Eny Palupi: Conceptualization; Methodology; Supervision; Funding acquisition. Ribka Tande: Conceptualization; Funding acquisition.
References
[1] DJ Magliano and EJ Boyko. IDF Diabetes Atlas 10th Edition. International Diabetes Federation, Belgium, 2021.
[2] RI Kemenkes. Laporan Tahunan Direktorat P2PTM 2021. Scribd, Indonesia, 2021.
[3] World Health Organization. Available at: http://apps.who.int/bookorders, accessed 2022.
[4] G Pan, Y Lu, Z Wei, Y Li, L Li and X Pan. A review on the in vitro and in vivo screening of α-glucosidase inhibitors. Heliyon 2024; 10(18), 37467.
[5] Rahmayanti, M Mursyid and C Astari. Formulasi yogurt sari buah belimbing wuluh (Averrhoa bilimbi L.) dan pengaruhnya terhadap penurunan kadar glukosa darah mencit (Mus musculus). Jurnal Mandala Pharmacon Indonesia 2024; 10(1), 181-190.
[6] AT Agustin, A Sulaeman and S Budi. Available at: https://repository.ipb.ac.id/handle/123456789/ 134758, accessed July 2025.
[7] J Król, A Brodziak, L Ślusarczyk, A Matwijczuk, M Chwil and R Matraszek-Gawron. Yogurt with cornflower (Centaurea cyanus L.) petals as a source of antioxidant compounds and dietary fiber: Physicochemical and spectroscopic research during storage. Journal of Dairy Science 2025; 108(3), 2243-2263.
[8] IA Palupi and M Martosupomo. Buah merah: Potensi dan manfaatnya sebagai antioksidan. Jurnal Tumbuhan Obat Indonesia 2009; 1279, 42-48.
[9] SA Sangkala, MR Jura and IM Tangkas. Uji aktivitas antioksidan ekstrak buah merah (Pandanus baccari L) di daerah poso sulawesi tengah. Jurnal Akademika Kimia 2014; 3(4), 198-205.
[10] AK Umar. Flavonoid compounds of buah merah (Pandanus conoideus Lamk) as a potent SARS-CoV-2 main protease inhibitor: In silico approach. Future Journal of Pharmaceutical Sciences 2021; 7(1), 158.
[11] AWMD Diah, NKA Diani and MR Jura. Efektivitas ekstrak buah merah (Pandanus conoideus De Vriese) asal kabupaten poso sebagai alternatif penurun kadar gula darah. Jurnal Akademika Kimia 2016; 5(4), 185-190.
[12] LV Hermawan, SH Fabanyo, MS Khoiruzaman, W Nurwahida, WN Fadilah and Irwandi. Pengaruh buah merah (Pandanus Conoideus Lam) asal Sorong Papua Barat terhadap antihiperglikemia pada mencit (Mus Musculus). Biolearning Journal 2023; 10(2), 41-44.
[13] IT Lestari, K Anggadiredja and AN Garmana. Red fruit (Pandanus conoideus Lam) oil ameliorates streptozotocin-induced diabetic peripheral neuropathy by targeting the oxidative and inflammatory pathways in the spinal cord in a rat model. PHARMACIA 2024; 71, 1-13.
[14] HA Jonathan, IN Fitriawati, II Arief, MS Soenarno and RH Mulyono. Fisikokimia, Mikrobiologi dan Organoleptik Yogurt Probiotik dengan Penambahan Buah merah (Pandanus conodeous L.). Jurnal Ilmu Produksi dan Teknologi Hasil Peternakan 2022; 10(1), 34-41.
[15] AOAC. Official Methods of Analysis. 18th eds. 2005.
[16] W Nurcholis, DN Sya'bani Putri, H Husnawati, SI Aisyah and BP Priosoeryanto. Total flavonoid content and antioxidant activity of ethanol and ethyl acetate extracts from accessions of Amomum compactum fruits. Annals of Agricultural Sciences 2021; 66(1), 58-62.
[17] NN Chaerunnisa, “Pengaruh Rasio Mucilage Dan Puree Okra Hijau (Abelmoschus esculentus) Pada Jelly Stick Terhadap Profil Fisikokimia, Organoleptik, Dan Penghambatan a-Glukosidase,” Available at: https://repository.ipb.ac.id/handle/123456789/134749, accessed July 2025.
[18] MS Sirait, E Warsiki and D Setyaningsih. Potential of red fruit oil (Pandanus conoideus Lam.) as an antioxidant active packaging: A review. IOP Conference Series: Earth and Environmental Science 2021; 749, 012008.
[19] C Galarreta-Morales, F Ludena-Urquizo, M Castillo-Arispe, J Carrasco-Macedo, K Ocampo-Ocampo, G Farfán-Saico, S Gutierrez-Estrada and J Liñan-Pérez. Sensory development of whey-based yogurt with Cushuro (Nostoc sphaericum), through novel consumer-based sensory evaluation methods. International Dairy Journal 2025; 166, 106254.
[20] R Qadir, WNAWM Nasir, AB Azmi, S Fatima, N Mehmood and ASM Hussin. An insight into plant-based yogurts: Physicochemical, organoleptic properties and functional food aspects. Journal of Food Composition and Analysis 2025; 143, 107578.
[21] SY Kim, O Hyeonbin, PL and YS Kim. The quality characteristics, antioxidant activity, and sensory evaluation of reduced-fat yogurt and nonfat yogurt supplemented with basil seed gum as a fat substitute. Journal of Dairy Science 2020; 103(2), 1324-1336.
[22] RA Wabula, S Seniwati and H Widiastuti. Aktivitas antioksidan ekstrak etanol buah merah (Pandanus conoideus Lam.) dengan metode Ferric Reducing Antioxidant Power FRAP. Window of Health: Jurnal Kesehatan 2019; 2(4), 329-337.
[23] G Bucalossi, G Fia, C Dinnella, AD Toffoli, V Canuti, B Zanoni, M Servili, E Pagliarini, TG Toschi and E Monteleone. Functional and sensory properties of phenolic compounds from unripe grapes in vegetable food prototypes. Food Chemistry 2020; 315, 126291.
[24] DV Ponglabba, ZL Sarungallo and B Santoso. Komposisi kimia dan stabilitas Puree buah merah (Pandanus conoideus Lamk.) selama penyimpanan. Jurnal Agroteknologi 2022; 16(1), 15-28.
[25] BY Espinaco, I Niizawa, F Cuffia, SE Zorrilla and GA Sihufe. Astaxanthin and chia oil encapsulated in gel beads: Evaluation of the impact of their addition in a commercial yogurt by a consumer-based sensory analysis. International Dairy Journal 2025; 160, 106080.
[26] VL Botta-Arias, F Ramos-Escudero, AM Muñoz and M Anticona. Nutritional composition, phenolic compounds, and sensory evaluation of osmosonicated orange peel snacks impregnated with plant extracts. Applied Food Research 2024; 4(2), 100486.
[27] AM Hamed, AAA El-Maksoud, MA Hassan, E Tsakali, JFMV Impe, HA Ahmed and AA Nassrallah. Enhancing functional buffalo yogurt: Improving physicochemical properties, biological activities, and shelf life using marjoram and geranium essential oils. Journal of Dairy Science 2024; 107(9), 6437-6450.
[28] International Organization for Standardization. ISO 13299:2016 - Sensory analysis - Methodology - General guidance for establishing a sensory profile iteh standard preview iteh standard preview. ISO, Switzerland, 2016.
[29] Badan Standardisasi Nasional. SNI 01-2346-2006 - Petunjuk pengujian organoleptik dan atau sensori. Standar Nasional Indonesia, Indonesia, 2006.
[30] A Sintya, AI Made and N Waras. Available at: http://repository.ipb.ac.id/handle/123456789/109216, accessed July 2025.
[31] I Tarchi, M Koubaa, F Ozogul, M Bouaziz and A Aït-Kaddour. Influence of olive leaf extract on the physicochemical properties of yogurts made from cow, sheep, and goat milk. Food Bioscience 2025; 63(8), 105728.
[32] RRD Atmaja, VK Annadiyah, M Thoyyibah, S Maimunah, B Maʼarif, R Mutiah, IM Budi, M Amiruddin and FR Inayatilah. The effect of red fruit oil (Pandanus conoideus Lamk.) emulgel on angiogenesis and collagen density in incisive wound healing in mice (Mus musculus). Journal of Advanced Pharmaceutical Technology & Research 2023; 14(4), 311-316.
[33] IG Munteanu and C Apetrei. Analytical methods used in determining antioxidant activity: A review. International Journal of Molecular Sciences 2021; 22(7), 3380.
[34] N Durmus, E Capanoglu and M Kilic-Akyilmaz. Activity and bioaccessibility of antioxidants in yoghurt enriched with black mulberry as affected by fermentation and stage of fruit addition. International Dairy Journal 2021; 117, 105018.
[35] HS Arruda, FT Borsoi, ACHF Sawaya, GM Pastore and MR Marostica Junior. The behavior of phenolic compounds, sugars, and antioxidants from araticum fruit (Annona crassiflora Mart.) during gastrointestinal digestion: An in vitro and in silico approach. Food Chemistry Advances 2025; 7, 101021.
[36] A Mulyani, W Warsidah, MSJ Sofiana, S Minsas, R Linda and I Safitri. Activity alpha-glucosidase enzyme and toxicity from ethanol extract of Caulerpa Seaweed. Jurnal Ilmiah Perikanan dan Kelautan 2022; 4(2), 187-192.
[37] T Pantoa, K Wadeesirisak, S Saah and P Saithong. In vitro protein digestibility and bioactive activities of yogurt from black soybean (Glycine max L.) milk. Future Foods 2025; 11, 100636.
[38] AB Shori and AS Baba. Comparative antioxidant activity, proteolysis and in vitro α-amylase and α-glucosidase inhibition of Allium sativum-yogurts made from cow and camel milk. Journal of Saudi Chemical Society 2014; 18(5), 456-463.
[39] TT Le, MT Ha, TQ Cao, JA Kim, JS Choi and BS Min. 1,5-Anhydro-D-glucitol derivative and galloylated flavonoids isolated from the leaves of Acer ginnala Maxim. as dual inhibitors of PTP1B and α-glucosidase enzymes: In vitro and in silico studies. Phytochemistry 2023; 213, 113769.
[40] J Pan, M Nawaz, J Liu, H Liu, Z Lv, W Yang, Z Jiao and Q Zhang. Exploring synergistic inhibitory mechanisms of flavonoid mixtures on α-glucosidase by experimental analysis and molecular dynamics simulation. Food Chemistry 2025; 464(1), 141560.
[41] M Daou, NA Elnaker, MA Ochsenkühn,SA Amin, AF Yousef and LF Yousef. In vitro α -glucosidase inhibitory activity of Tamarix nilotica shoot extracts and fractions. PLoS One 2022; 17(3), 0264969.
[42] M Yang, X Yang, X Chen, J Wang, Z Liao, L Wang, Q Zhong and X Fang. Effect of kefir on soybean isoflavone aglycone content in soymilk kefir. Frontiers in Nutrition 2020; 7 587665.
[43] YS Mokosuli, N Migau and M Wurarah. Bioactivity of papua red fruit extract (Pandanus conoideus L.) against superoxide dismutase, malondialdehyde and blood glucose of rat (Rattus norvegicus L.) hyperglycemia. Molekul 2024; 19(1), 86-97.
[44] RR Sunagar and YN Sreerama. Implication of solvent polarities on browntop millet (Urochloa ramosa) phenolic antioxidants and their ability to protect oxidative DNA damage and inhibit α-amylase and α-glucosidase enzymes. Food Chemistry 2023; 411, 135474.
[45] N Lingiardi, NS Morais, VM Rodrigues, SMG Moreira, M Galante, D Spelzini, CF de Assis and FC de Sousa Júnior. Quinoa protein-based Buriti oil nanoparticles: Enhancement of antioxidant activity and inhibition of digestive enzymes. Food Research International 2025; 214, 116693.
[46] ZL Sarungallo, P Hariyadi, N Andarwulan and EH Purnomo. Pengaruh metode ekstraksi terhadap mutu kimia dan komposisi asam lemak minyak buah merah (Pandanus conoideus). Jurnal Teknologi Industri Pertanian 2014; 24(3), 209-217.
[47] CH Su, CH Hsu and LT Ng. Inhibitory potential of fatty acids on key enzymes related to type 2 diabetes. BioFactors 2013; 39(4), 415-421.
[48] KS Tshiyoyo, A Rabbad, AA Yusuf and S Malgas. Combination of citrus peel-derived essential oils with acarbose to inhibit amylolytic enzymes: A potential type II diabetes treatment approach. International Journal of Biological Macromolecules 2025; 306(3), 141504.
[49] J Skjold-Jørgensen, VK Bhatia, J Vind, A Svendsen, MJ Bjerrum and D Farrens. The enzymatic activity of lipases correlates with polarity-induced conformational changes: A trp-induced quenching fluorescence study. Biochemistry 2015; 54(27), 4186-4196.



