
Trends
Sci.
2026;
23(6):
13035
Measurement of Alcohol Type and Concentration in Hand Gel Sanitizers During the COVID-19 Era: A Review Article for the Future World Health Pandemic Situation
Panya Khaenamkaew
Department of Basic Science and Physical Education, Faculty of Science at Sriracha, Kasetsart University,
Sriracha Campus, Chonburi 20230, Thailand
(Corresponding author’s e-mail: [email protected], [email protected])
Received: 15 December 2025, Revised: 11 January 2026, Accepted: 21 January 2026, Published: 5 February 2026
Abstract
The COVID-19 pandemic drastically altered public health practices worldwide, highlighting the critical role of hand hygiene in controlling viral transmission. In response, alcohol-based hand sanitizers (ABHS) became essential preventive tools, prompting unprecedented global demand, regulatory adaptations, and scientific scrutiny. This review critically examines the types and concentrations of alcohol used in hand sanitizers, the analytical techniques for their measurement, and the evolving regulatory frameworks that govern their production and use. It synthesizes current knowledge on ethanol and isopropanol efficacy, explores safety considerations such as skin irritation and ingestion risks, and evaluates global compliance challenges, particularly in the wake of counterfeit and substandard products. The review also compares advanced measurement techniques - including GC, HPLC, FTIR, and NMR - for their precision, cost, and usability in different settings. Additionally, it discusses market dynamics during the pandemic, shifting consumer preferences, and key research gaps, notably in standardized testing and long-term safety. By integrating multidisciplinary insights from analytical chemistry, public health, regulatory science, and consumer behavior, this paper offers forward-looking recommendations for improving hand sanitizer quality, safety, and pandemic preparedness. The findings aim to support researchers, manufacturers, and policymakers in developing more robust, effective, and trusted hand hygiene solutions for future global health emergencies.
Keywords: Alcohol-based hand sanitizers, COVID-19 pandemic, Alcohol concentration measurement, Regulatory compliance, Public health safety, Analytical techniques (GC, HPLC, FTIR, NMR)
Introduction
The COVID-19 pandemic, caused by SARS-CoV-2, began in December 2019. It rapidly escalated into a global health crisis, with the World Health Organization (WHO) declaring it a public health emergency on January 30, 2020, and a pandemic on March 11, 2020. By mid-2023, over 775 million confirmed cases and approximately 7 million reported deaths had been recorded worldwide [1]. Countries responded differently to the pandemic. The United States, India, and Brazil were among the hardest hit, with the U.S. surpassing 1 million deaths by early 2023 [2]. In contrast, Australia and New Zealand were praised for their effective management strategies, including strict border controls and early lockdowns [3].
The WHO played a pivotal role in coordinating global responses, providing guidelines, and facilitating vaccine distribution through initiatives like COVAX. However, disparities in vaccination rates persisted, particularly in low-income countries and regions like Africa [4]. The pandemic had profound social and economic impacts. The WHO reported a decline in global life expectancy by 1.8 years from 2019 to 2021 [5]. Economically, it triggered the largest global recession since the Great Depression, leading to widespread unemployment and supply chain disruptions [6]. During these challenging times, alongside COVAX efforts, disease prevention measures like hand sanitizers have been essential, especially for healthcare workers, in reducing the spread of the virus. The pandemic significantly altered public health practices and consumer behavior, particularly regarding hand hygiene. As the WHO urged individuals to stay at home, wear masks, and wash hands frequently, the demand for alcohol-based hand sanitizers surged, leading to widespread shortages [7]. In response, the WHO provided guidelines on hand sanitizer formulation, advocating for the use of at least 60% ethanol or 70% isopropanol [8]. These guidelines have been crucial in ensuring product safety and effectiveness. Various health organizations, including the WHO and the Food and Drug Administration (FDA), have established regulatory guidelines to ensure the efficacy and safety of alcohol-based hand sanitizers [9,10]. However, it is crucial to note that hand sanitizers may also contain other constituents, such as thickeners, humectants, and fragrances, which can potentially cause contact allergies [11]. Various countries recommended that specific businesses and pharmacies temporarily produce hand sanitizers. However, this rapid production expansion raised concerns about consumer safety, particularly regarding methanol contamination and inconsistencies in alcohol concentration [12].
The COVID-19 pandemic has underscored the critical importance of hand hygiene in preventing the spread of infectious diseases. Hand sanitizers, particularly alcohol-based formulations, play an important role in public health initiatives aimed at reducing viral transmission. When properly formulated and used according to recommended guidelines, alcohol-based hand sanitizers represent an essential tool in infection control [13,14]. The effectiveness of these sanitizers in eliminating or reducing the presence of the virus on hands, especially after contact with potentially contaminated surfaces, has been well-documented [15].
The quality and composition of hand sanitizers have become subjects of increased scrutiny during the pandemic. Methanol adulteration has been reported, emphasizing the need for stringent monitoring and public awareness campaigns [14]. Furthermore, research has focused on developing formulations with efficient polymers for alcohol-based gel hand sanitizers, showcasing innovation in enhancing their rheological properties for improved usability and effectiveness [16].
This critical review aims to systematically examine the measurement of alcohol type and concentration in hand gel sanitizers within the context of the COVID-19 pandemic. Through a rigorous analysis of current literature, regulatory guidelines, and emerging research, we provide a comprehensive synthesis of the pivotal role of alcohol content in hand sanitizers, the diverse methodologies employed for its quantification, and the far-reaching implications for public health and safety.
This comprehensive review is structured into 9 interconnected sections: (1) Introduction and Background; (2) Key Components of Hand Gel Sanitizers: A Chemical Perspective; (3) Analytical Techniques for Alcohol Quantification in Hand Sanitizers: Methodological Comparisons and Critical Evaluation; (4) Regulatory Frameworks and Compliance: A Global Perspective; (5) Efficacy and Safety Considerations: Balancing Antimicrobial Activity and User Health; (6) Market Dynamics of Hand Sanitizers in the COVID-19 Era: Trends and Economic Implications; (7) Research Gaps and Future Directions: Opportunities for Innovation and Improvement; (8) Synthesis of Findings: A Holistic View of Alcohol Content Measurement in Hand Sanitizers; (9) Implications and Recommendations: Towards Enhanced Public Health Practices and Product Safety.
Literature search strategy and article selection
To ensure a comprehensive and balanced review, the literature included in this article was identified through systematic searches of major scientific databases, including Scopus, Web of Science, PubMed, and Google Scholar. The search was conducted using combinations of keywords such as “alcohol-based hand sanitizer,” “hand gel sanitizer,” “ethanol,” “isopropanol,” “alcohol concentration,” “analytical methods,” “GC,” “FTIR,” “HPLC,” and “regulatory standards.”
Publications from approximately 2000 to 2024 were considered, with particular emphasis on studies published during and after the COVID-19 pandemic due to the rapid expansion of sanitizer production and regulatory scrutiny during this period. Peer-reviewed journal articles, international guidelines (e.g., WHO, FDA, EN standards), and authoritative reports were prioritized.
Studies were selected based on their relevance to the measurement of alcohol type and concentration in hand gel sanitizers, including analytical methodology, validation performance, regulatory compliance, and safety considerations. Articles focusing exclusively on unrelated matrices (e.g., biological fluids or beverages) were included only when their analytical principles were directly transferable to hand gel formulations.
Although numerous studies and reviews have addressed alcohol-based hand sanitizers from perspectives such as formulation efficacy, microbiological performance, or regulatory compliance, a focused synthesis centered on the measurement of alcohol type and concentration in hand gel sanitizers remains limited. Existing reviews often discuss analytical techniques in isolation or within broader contexts that include biological, food, or forensic matrices.
The present review uniquely integrates analytical chemistry techniques, regulatory standards, and practical challenges specifically related to alcohol-based hand gel sanitizers, with emphasis on method selection, performance characteristics, and applicability to gel formulations. By systematically comparing commonly used analytical approaches and linking them to regulatory requirements and real-world manufacturing conditions, this review provides a targeted reference for researchers, regulators, and quality-control laboratories involved in sanitizer evaluation during and beyond global health emergencies.
Key components of hand gel sanitizers
The primary components of hand gel sanitizers include alcohol, thickeners, moisturizers, and sometimes fragrances or additional active ingredients. These components not only define the efficacy of the sanitizer but also affect its safety and user experience [17].
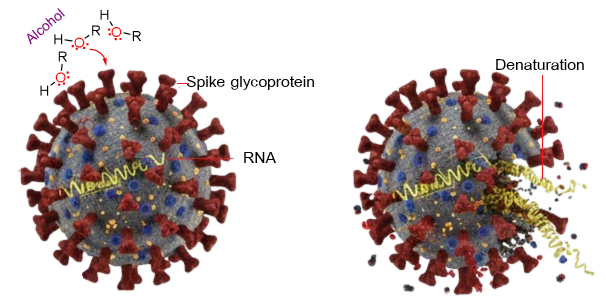
Figure 1 Antimicrobial mechanisms of ethanol against pathogens.
Alcohol content: The active ingredient
The primary active components in hand sanitizers are alcohols such as ethanol, isopropanol, and n-propanol, used alone or in combination. Their germicidal action, effective against a wide range of pathogens including bacteria and enveloped viruses like SARS-CoV-2, works by denaturing proteins and disrupting cell membranes. For reliable efficacy, health authorities recommend a minimum alcohol concentration of 60% (Figure 1). Ethanol is most effective at 60% - 85% and isopropanol at 60% - 80% [8,18,19,20]. Solutions below this threshold may only inhibit growth rather than kill microbes, while those with excessively high alcohol content are also less effective due to the necessity of water for the denaturation process [11,18]. Isopropanol, often used in surface disinfectants, evaporates faster and can be less irritating to the skin. However, alcohol concentrations below the recommended threshold may fail to effectively kill pathogens, while concentrations above 90% can lead to rapid evaporation, diminishing the contact time necessary for full microbial inactivation [20]. Additionally, the inclusion of alcohol in formulations must be balanced with skin safety considerations, as frequent use can cause skin dryness and irritation.
Thickeners and gel consistency
Thickeners are used in hand sanitizers to modify the product’s texture and enhance its usability. Common thickeners include carbomers, xanthan gum, and hydroxyethyl cellulose. These ingredients help achieve a gel-like consistency, making the sanitizer easy to apply and less likely to spill. The gel formulation also ensures that the alcohol remains in contact with the skin for an adequate amount of time, allowing for effective sanitization. The consistency of the gel can also influence user experience, with thicker gels often preferred for their slower evaporation rate and improved application control [15].
Moisturizers and skin conditioning agents
Frequent use of alcohol-based sanitizers can lead to dry and irritated skin due to alcohol’s ability to strip natural oils. To mitigate this, many hand sanitizers include moisturizers such as glycerin, aloe vera, or propylene glycol. These ingredients act as humectants, which attract moisture to the skin, improving comfort and preventing dryness [20]. The inclusion of moisturizers is especially important for users who frequently sanitize their hands, such as healthcare workers, as it helps maintain skin integrity and reduce the risk of dermatitis or eczema [16,21].
While moisturizers can improve the user experience, their presence must be balanced carefully. Excessive use of certain ingredients can reduce the sanitizer’s antimicrobial efficacy, highlighting the need for formulations that maintain both effectiveness and skin safety.
Fragrances and additional additives
Fragrances and other additives are often incorporated into hand sanitizers to improve their sensory appeal. Common fragrances include citrus, floral, and herbal scents, which mask the strong odor of alcohol and enhance the overall user experience. However, these additives can sometimes cause skin sensitivities or allergic reactions in some individuals [21]. Consequently, fragrance-free formulations are available for those with sensitive skin or allergies.
In addition to fragrances, some sanitizers include antibacterial agents like chlorhexidine or hydrogen peroxide. These ingredients can enhance the product’s effectiveness, though they must be carefully balanced to avoid irritation or adverse reactions [18]. The ethanol content and ingredients of the commercial hand sanitizers analyzed are listed in Table 1. However, these additional ingredients are not always necessary, and excessive additives can compromise the product’s safety and effectiveness.
Table 1 Ingredients and ethanol percentage of 6 commercial hand gel sanitizers collected from the drug stores [22].
Sample No. |
Ethanol (%v/v) |
Expire date |
Ingredients (as label) |
a) Com1_70% (ALSOFF) |
70.0 |
08.05.2022 |
Alcohol, Aqua, Glycerin, Acrylates/C10-30 Alkyl Acrylate Crosspolymer, Triethanolamine, Tocopheryl Acetate, Disodium EDTA, Aloe Barbadensis Leaf Juice, Citric Acid, Sodium Benzoate |
b) Com2_72.4% (SURE)
|
72.4 |
21.03.2022 |
Alcohol, Aqua, Propylene Glycol, Carbomer, Triethanolamine, Fragrance, Allantoin, Butylene Glycol, Glycerin, Aloe Barbadensis Leaf Juice, etc. |
c) Com3_75% (MUKO) |
75.0 |
13.03.2022 |
Ethyl Alcohol, Aqua, Propylene, Glycerin, Carbomer, Tocopherol, Fragrance, etc. |
d)
Com4_75% |
75.0 |
09.04.2022 |
Ethanol, Aqua, Propylene Glycol, Acrylates/C10-30 alkyl acrylate crosspolymer, Glycerin, Methylparaben, Propylparaben, Mentha piperita oil, Triethanolamine |
e) Com5_75% (Rabbit) |
75.0 |
24.04.2022 |
Ethyl Alcohol, Aqua, Acrylates/C10-30 Alkyl Methacrylates copolymer, Triethanolamine, Aloe Barbadensis Leaf Extract, Glycerin, Fragrance Free |
f)
Com6_76% |
76.0 |
08.04.2023 |
Ethanol, Water, Carbomer, Triethanolamine, Propylene Glycol, Fragrance, CI 420-90 |
Figure 2 representative commercial gel-based hand sanitizer products examined in this review, illustrating variability in formulation type, labeled alcohol content, packaging size, and intended use. The selected samples include both consumer-grade and industrial-use hand sanitizers with different ethanol concentrations and gel compositions (data compiled from Khaenamkaew et al. [22]).

Figure 2 Commercial alcohol-based hand gel sanitizer products analyzed in the Khaenamkaew et al. [22].
Regulatory compliance and formulation standards
Regulatory bodies such as the WHO and FDA set standards for the formulation of hand sanitizers, primarily focusing on alcohol concentration and the exclusion of harmful additives. For example, the WHO guidelines recommend formulations containing 80% ethanol or 75% isopropanol to ensure the product’s virucidal activity [8]. Furthermore, the FDA has established specific requirements for labeling and safety to ensure consumer protection [19]. As the market for hand sanitizers continues to expand, adherence to these regulatory guidelines is crucial to maintaining product safety and efficacy.
Measurement techniques
The measurement techniques for hand gel sanitizers involve various methods to assess their type and concentration. One common approach is to evaluate the physical and chemical properties of the gel, such as viscosity, pH, and alcohol content. Adugo et al. [23] conducted a physicochemical evaluation of alcohol-based hand sanitizers, quantifying the effects of temperature on their potency and concentrations after exposure to different environmental conditions. Effectiveness comparisons are another crucial aspect of measurement techniques. Prajapati et al. [24] compared the effectiveness of hand washing with water, antiseptic soap, hand sanitizer gel, and alcohol 70% against the amount of bacteria on the hands, finding that alcohol 70% showed the most significant decrease in bacterial colonies. The use of natural ingredients and emollients can impact the texture and application of the gel. Booq et al. [25] studied the preparation and evaluation of ethanol-free hand sanitizers, finding that the addition of moisturizers like glycerin improved the comfort and usability of the products. Similarly, Fallica et al. [26] investigated the formulation and effectiveness of hand sanitizer gels produced using Salam bark extract, determining the minimum inhibitory concentration (MIC) of the extract and evaluating the physical stability of the gels over time.
The measurement techniques for hand gel sanitizers involve a combination of physical and chemical assessments, as well as the evaluation of their efficacy and user acceptance. These techniques help in understanding the optimal balance between efficacy, safety, and compatibility with the skin. A concise comparison of analytical techniques, including accuracy, cost, and time considerations, is provided in Table 2.
Analytical methods
Gas chromatography (GC)
Gas Chromatography (GC) is a highly accurate and reliable method for quantifying alcohol content in hand sanitizers. This technique involves separating volatile compounds, including alcohols, from a sample using a column and a carrier gas, followed by detection and quantification using a detector such as a flame ionization detector (FID) or a mass spectrometer (MS) [27].
GC offers several advantages over other measurement techniques: (1) Accuracy and Precision: GC provides precise measurements of alcohol concentrations, capable of detecting and quantifying even trace amounts of ethanol in the sample [28]; (2) Separation and Identification: The technique separates volatile compounds, allowing for accurate identification and quantification of specific alcohols, such as ethanol, which is the primary active ingredient in most hand sanitizers [27]; (3) Sensitivity: GC is highly sensitive, making it crucial for quality control and ensuring consistency of alcohol content across different batches of hand sanitizers [29]; (4) Versatility: It can analyze various types of hand sanitizers, including gels, sprays, and wipes [28]; (5) Detection of Impurities: GC can detect and quantify impurities in hand sanitizers, such as acetaldehyde and ethyl acetate, which are common contaminants in ethanol-based products [29].
Studies
have demonstrated the effectiveness of GC in analyzing hand
sanitizers. For instance, 1
one study achieved high linearity (R2
> 0.998) in the range of 0.25 to 2.5% (w/w) ethanol when
quantifying ethanol content in hand sanitizer gels [28]. Another
study found that the majority of hand sanitizer samples analyzed
fell within the WHO recommendation range, with ethanol levels
between 66% and 76% [27].
In conclusion, gas chromatography has proven to be a highly accurate and reliable technique for quantifying alcohol content across various applications. Its ability to separate and quantify complex mixtures of alcohols, coupled with its sensitivity and selectivity, makes GC an indispensable tool in the beverage industry, forensics, toxicology, and environmental analysis. Ongoing research is expected to further enhance the accuracy and efficiency of alcohol quantification using GC technology.
Fourier transform infrared spectroscopy (FTIR)
Fourier transform infrared spectroscopy (FTIR) offers a rapid and non-destructive approach for the screening and semi-quantitative analysis of alcohol content in hand gel sanitizers. The technique relies on characteristic infrared absorption bands associated with O–H stretching and C–O vibrations of alcohol molecules, enabling differentiation between ethanol- and isopropanol-based formulations.
For hand gel sanitizer analysis, FTIR is particularly attractive because it often requires minimal sample preparation, allowing direct measurement of gel formulations using attenuated total reflectance (ATR) accessories. This feature makes FTIR suitable for high-throughput screening, counterfeit detection, and preliminary compliance assessment.
Despite these advantages, FTIR generally exhibits lower sensitivity and selectivity compared with chromatographic techniques, especially when complex matrices or overlapping spectral features are present. Consequently, FTIR is best positioned as a screening tool, with confirmatory analysis performed using GC or other quantitative methods when precise alcohol concentration determination is required.
Recent studies have demonstrated its effectiveness in various applications, from quality control to biomedical research. A study compared 2 portable spectrometers for detecting toxic contaminants in alcohol-based hand sanitizers. The researchers found excellent correlation coefficients (R2 up to 0.99) and low root mean square errors of prediction (as low as 0.34% v/v), highlighting IR spectroscopy’s potential for rapid, inexpensive, and portable testing [30]. As shown in Figure 3, the FTIR spectra provide rapid qualitative and semi-quantitative insight into alcohols in hand gel sanitizers, revealing characteristic O–H stretching and C–O vibration bands, with spectral variations reflecting differences in alcohol type and formulation; thus, FTIR serves as an effective screening tool prior to confirmatory chromatographic analysis [22]. Another study explored using IR spectroscopy for simultaneous in vivo measurement of ethanol and its metabolites. The researchers demonstrated the capability of quantifying concentrations of ethanol, acetaldehyde, and acetic acid simultaneously using transmission mode IR and ATR. They also explored direct measurement using evanescent-field IR spectroscopy with hydrophobic polymer coatings, showing potential for improved sensitivity [31,32].
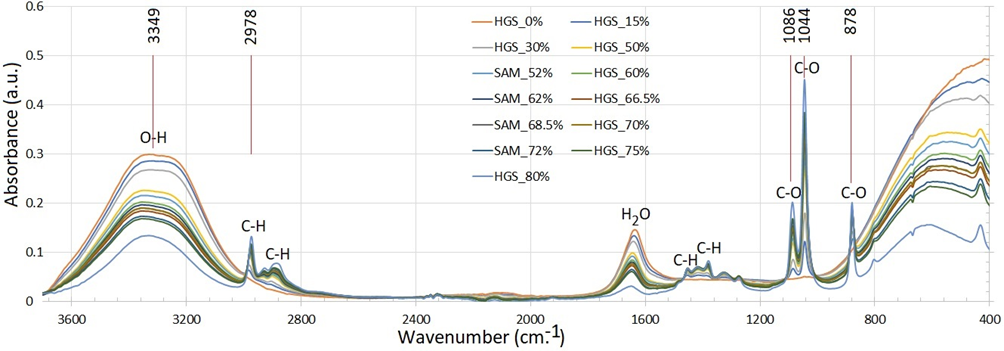
Figure 3 FTIR spectra of selected hand gel sanitizer (HGS) samples, showing spectral differences associated with variations in alcohol type and formulation. Abbreviations: HGS = hand gel sanitizer; SAM = standard additional method sample [22].
Ultrafast IR spectroscopy has been used to study the structural dynamics of highly concentrated lithium chloride and lithium bromide aqueous solutions. This technique revealed distinct water components and measured frequency-dependent water orientational relaxation, providing insights into molecular dynamics not observable with traditional Fourier transform IR (FT-IR) spectroscopy [33]. Frequency-domain near-infrared spectroscopy (FD-NIRS) has been applied to measure absolute optical properties of brain tissue, enabling the determination of absolute concentrations of oxyhemoglobin and deoxyhemoglobin. This technique has been used in functional brain studies focusing on both slow hemodynamic responses and faster optical signals linked to neuronal activation [34]. A recent development in IR spectroscopy is mid-infrared (MIR) dispersion spectroscopy, which uses a tunable quantum cascade laser and Mach-Zehnder interferometer. This method detects refractive index changes instead of intensity attenuation, resulting in improved sensitivity and increased analytical path length compared to FT-IR [35]. Researchers have used this technique to monitor enzyme kinetics and sugar mutarotation, demonstrating its potential for liquid-phase analysis [36].
The application of FT-IR spectroscopy for measuring alcohol concentration, particularly ethanol, has garnered significant attention in recent years, especially in the context of hand sanitizer analysis during the COVID-19 pandemic. FT-IR spectroscopy offers numerous advantages, including rapid analysis, minimal sample preparation, and the ability to provide both qualitative and quantitative data simultaneously. The technique relies on analyzing specific absorption bands associated with ethanol’s molecular vibrations, notably the C-O stretching vibrations observed around 1,086 and 1,044 cm−1 (Figure 3) [22]. However, the accuracy and reliability of these measurements are contingent upon a comprehensive understanding and estimation of measurement uncertainty. This aspect has become increasingly crucial, as evidenced by the growing body of literature addressing the subject. The estimation of measurement uncertainty in FTIR analysis of ethanol concentration encompasses several key components, including calibration uncertainty, sample preparation variability, instrumental factors, and environmental conditions. Calibration uncertainty is derived from the variability in the measured response when generating calibration curves using standard ethanol solutions. Sample preparation introduces potential errors through pipetting and dilution processes, emphasizing the need for consistent and standardized procedures. Instrumental factors, such as resolution and noise, can affect measurement precision, necessitating regular maintenance and calibration of FTIR instruments. Environmental conditions, including temperature and humidity, can also influence measurements, underscoring the importance of controlling these variables during analysis. The Nordtest guidelines and ISO/IEC 17025 standards provide a systematic approach to uncertainty estimation, offering a framework for researchers to enhance the reliability of their measurements. Recent studies have applied these principles to real-world scenarios, such as the assessment of ethanol concentration in hand sanitizers. Khaenamkaew et al. [22] reported an expanded uncertainty of approximately ±1.14 %v/v at a 95% confidence interval for ethanol measurements in hand sanitizers, highlighting the critical role of uncertainty estimation in ensuring product quality and safety. Complementing these findings, the National Institute of Standards and Technology (NIST) conducted a comprehensive evaluation of various instrumental methods, including FTIR, for measuring ethanol and impurities in hand sanitizers. Their research confirmed FTIR as a quick and effective method for analyzing complex samples, though noting potential discrepancies with gas chromatography results, particularly in the presence of methanol [37]. The collective body of research underscores the value of FTIR spectroscopy in measuring ethanol concentration across diverse applications, while simultaneously emphasizing the paramount importance of estimating measurement uncertainty. By adhering to established guidelines and methodologies, researchers can significantly enhance the reliability and comparability of their results across different laboratories. As the field continues to evolve, future research directions should focus on refining measurement techniques and improving uncertainty estimation methodologies to better support public health and safety standards. This ongoing work will be crucial in addressing the challenges posed by emerging public health crises and ensuring the efficacy and safety of alcohol-based products in various contexts.
High-performance liquid chromatography (HPLC)
High-Performance Liquid Chromatography (HPLC) is a versatile and powerful analytical technique particularly useful for the detailed analysis of complex formulations, including hand gel sanitizers. This method allows for the separation, identification, and quantification of multiple components in a single sample, providing a comprehensive understanding of the formulation’s composition and properties. HPLC involves using a liquid mobile phase and a stationary phase, typically a column packed with small particles or a capillary column [38]. The sample is injected into the system, and components are separated based on their interactions with the stationary and mobile phases. Separated components are then detected and quantified using various detectors, such as ultraviolet (UV), refractive index (RI), or mass spectrometers (MS) [39].
HPLC excels in analyzing complex mixtures like hand sanitizers, which often contain multiple ingredients including alcohols, gelling agents, moisturizers, and fragrances. For instance, Bedner et al. [37] used HPLC to quantify ethanol content and detect impurities like acetaldehyde and ethyl acetate in alcohol-based hand sanitizers. HPLC is also useful for analyzing natural ingredients added to hand sanitizers. HPLC is an essential tool for ensuring the quality, safety, and efficacy of hand sanitizers and other complex formulations. Its ability to separate, identify, and quantify multiple components in a single sample makes it invaluable in both research and quality control applications.
Nuclear Magnetic Resonance (NMR)
Nuclear Magnetic Resonance (NMR): Provides structural information about alcohol types. Nuclear Magnetic Resonance (NMR) spectroscopy is a crucial analytical technique for the structural elucidation of organic compounds, including various alcohols. This method provides detailed structural information by observing the interaction of nuclear spins with an external magnetic field, offering insights into molecular structure, dynamics, and environment.
Recent developments in NMR technology have expanded its applicability to complex mixture analysis, particularly in natural products and alcohols [40]. Two-dimensional NMR techniques, such as J-resolved spectroscopy and diffusion-ordered spectroscopy (DOSY), allow for the separation and identification of different alcohol types within complex mixtures without prior purification. NMR’s non-destructive nature makes it particularly advantageous for studying alcohols in various matrices, including biological samples. This capability has significant implications for fields like pharmacology and toxicology, where understanding alcohol metabolism and effects is crucial [41].
The integration of NMR with other analytical techniques, such as mass spectrometry, provides complementary data on molecular weight and fragmentation patterns, enriching the structural information available [37]. High-resolution magic angle spinning (HR-MAS) NMR allows for the examination of solid and semi-solid samples, making it particularly useful for studying alcohols in food products and beverages. The application of cryo-probes has further enhanced sensitivity, enabling the detection of low-concentration alcohols in complex matrices [42]. Comprehensive databases and software tools facilitate spectral interpretation, enabling researchers to compare experimental spectra with known standards. The use of machine learning algorithms in conjunction with NMR data is an emerging trend that promises to enhance the accuracy and efficiency of alcohol identification and quantification [37].
NMR spectroscopy serves as a robust tool for providing structural information about various alcohol types, facilitating rapid and accurate analysis in both research and industrial applications. Its ability to analyze complex mixtures directly, coupled with advancements in technology and methodology, positions NMR as an indispensable technique in the characterization of alcohols, contributing significantly to our understanding of their chemical properties and biological implications.
Advantages and limitations
The importance of measurement accuracy in regulatory compliance
Accurate measurements are crucial for ensuring compliance with regulatory standards across various fields, including pharmaceuticals, environmental monitoring, food safety, and clinical diagnostics. Regulatory bodies such as the FDA, EPA, and ISO establish stringent guidelines that require precise and reproducible measurement techniques [43].
Pharmaceutical Industry: In the pharmaceutical sector, accurate dosage measurements are critical for ensuring therapeutic efficacy and patient safety. Regulatory agencies require pharmaceutical companies to implement robust quality control measures, including validated analytical methods like HPLC and GC, to verify measurement accuracy throughout the manufacturing process [44].
Environmental Monitoring: Accurate measurements of pollutants and contaminants are essential for compliance with environmental regulations. Research has highlighted the need for standardized measurement protocols and calibration procedures to enhance the accuracy of environmental data. Implementing QA/QC protocols in environmental sampling and analysis is vital for ensuring reliable and compliant measurements [45].
Clinical Diagnostics: In clinical diagnostics, measurement accuracy is paramount for patient care. Regulatory standards, such as those set by CLIA, mandate high levels of accuracy in laboratory testing processes. Studies have shown that inaccuracies in diagnostic measurements can lead to misdiagnosis and inappropriate treatment decisions. The integration of advanced technologies, such as point-of-care testing and automated laboratory systems, has been shown to improve measurement accuracy and enhance compliance with regulatory requirements [46].
Standardized Reporting Guidelines: The development of standardized reporting guidelines, such as STARD, plays a crucial role in improving the accuracy of measurement reporting. These guidelines provide a framework for transparent reporting of study design, methodology, and results. However, a systematic review revealed inconsistent adherence to these reporting standards, highlighting the need for greater enforcement and education within the research community [47,48].
Implications of Inaccurate Measurements: Inaccuracies in measurement can lead to non-compliance, resulting in severe implications, including product recalls, legal penalties, and damage to reputations. In environmental monitoring, inaccurate measurements can lead to underestimating risks associated with environmental hazards, potentially resulting in adverse health effects and ecological damage [49].
The accuracy of measurement is essential for ensuring compliance with regulatory standards across various sectors. Continuous efforts to improve measurement techniques, implement standardized protocols, and adhere to reporting guidelines are necessary to enhance measurement accuracy and support compliance with regulatory requirements. As industries evolve and new technologies emerge, ongoing research and development in measurement accuracy will be vital for addressing compliance challenges and ensuring the safety and efficacy of products and services.
Sensitivity: Important for detecting low concentrations of alcohol
Sensitivity is crucial in detecting low concentrations of alcohol, particularly in applications where accurate quantification is essential for public health and safety. Developing highly sensitive analytical techniques is vital for measuring alcohol levels in various matrices, including biological fluids, food products, and environmental samples [50].
Table 2 Key analytical techniques used for hand sanitizer analysis during the COVID-19 pandemic.
Techniques |
Accuracy |
Sensitivity |
Specificity |
Cost |
Time Consumption |
Worker Skill Level |
Gas Chromatography (GC) [31] |
High; effectively quantifies ethanol and detects impurities like methanol (National Institute of Standards and Technology |
High; detects low levels of impurities (µg/g) per FDA limits [28]
|
High; specifically identifies alcohols and impurities [41]
|
Moderate; equipment is expensive but widely used [31]
|
Moderate; requires sample preparation and calibration
|
Moderate; requires training in chromatography techniques |
High-Performance Liquid Chromatography (HPLC) [31] |
High; effective for quantifying alcohols in complex matrices [37] |
High; detects low concentrations of components [37,38] |
High; good specificity for various compounds [37] |
High; typically more expensive than GC due to equipment costs |
Moderate to high; can be time-consuming depending on method |
High; requires skilled personnel for operation and interpretation |
Fourier Transform Infrared Spectroscopy (FTIR)
|
Moderate; good for qualitative analysis but less precise than GC or HPLC for quantification [36] |
Moderate; can detect functional groups but less sensitive than GC |
Moderate; effective for identifying specific compounds but can miss others |
Moderate; generally less expensive than GC and HPLC
|
Fast; typically quicker than chromatography methods |
Moderate; requires understanding of spectral interpretation |
Nuclear Magnetic Resonance (NMR)
|
High; provides detailed structural information and quantification |
Moderate; less sensitive than GC for trace impurities [37,40] |
High; very specific for molecular structure identification |
High; equipment is costly and requires maintenance |
Moderate; sample preparation and analysis can take time |
High; requires advanced training to operate and analyze results |
These techniques offer varying levels of accuracy, sensitivity, specificity, cost, time consumption, and required worker skill. The choice of method depends on the specific analytical needs and available resources. A key challenge in measuring low alcohol concentrations is potential interference from other compounds in the sample matrix. To address this, researchers have developed advanced analytical techniques combining high sensitivity with high selectivity. Gas chromatography-mass spectrometry (GC-MS) and liquid chromatography-tandem mass spectrometry (LC-MS/MS) are widely used for detecting and quantifying alcohol and its metabolites in blood, urine, and breath samples, offering excellent sensitivity with detection limits in the nanogram per milliliter range [28,32,50].
Alternative approaches
Researchers have explored alternative approaches to enhance alcohol detection sensitivity:
Infrared (IR) Spectroscopy: This technique offers rapid analysis, reagent-free operation, and the ability to measure multiple analytes simultaneously. Studies have demonstrated its feasibility for measuring ethanol, acetaldehyde, and acetic acid in aqueous solutions [30,51].
Advanced Sampling Techniques: Solid-phase microextraction (SPME) and headspace sampling allow for selective extraction and concentration of alcohol from complex matrices. SPME combined with GC-MS has been used to detect volatile organic compounds, including alcohols, in environmental samples, with detection limits in the parts per trillion range [50].
Regulatory Compliance: The sensitivity of alcohol detection is crucial for complying with regulatory standards and guidelines. Many countries have established legal limits for alcohol consumption, particularly for drivers, and law enforcement agencies rely on highly sensitive analytical techniques to enforce these laws. In the food and beverage industry, sensitive alcohol detection is essential for quality control and product safety, with regulatory bodies like the FDA and EFSA establishing guidelines for maximum allowable alcohol levels in various food products [19,52,53].
The sensitivity of measurement is critical in detecting low alcohol concentrations, with significant implications for public health, safety, and regulatory compliance. Ongoing research and development in analytical techniques continue to improve sensitivity and selectivity, enabling reliable alcohol detection and quantification even in complex matrices. As new challenges emerge and regulatory standards evolve, the need for highly sensitive and accurate alcohol detection methods will drive further innovation in this field.
Cost: Considerations for laboratories and manufacturers
The cost of measurement is a crucial consideration for laboratories and manufacturers, influencing the choice of analytical techniques and the overall feasibility of compliance with regulatory standards. As industries increasingly prioritize accuracy and reliability in their measurements, the financial implications of adopting advanced technologies become paramount. Low-cost sensors (LCS) have emerged as a viable alternative for monitoring various environmental parameters, including air quality. These sensors offer the advantage of increased spatial coverage and accessibility, particularly in urban and remote areas, with costs ranging from several hundred to several thousand euros [28]. However, while LCS can provide cost-effective solutions, studies have shown that their performance can be inconsistent, often affected by environmental conditions and cross-sensitivities, which may compromise measurement accuracy [38].
In the context of laboratory settings, the development of simple and low-cost measurement setups has been explored. For instance, a study introduced a low-cost densitometer capable of measuring the densities of various liquids, including water-ethanol mixtures, with acceptable relative errors of less than ±3.0% [29]. This approach demonstrates that cost-effective measurement solutions can be both reliable and educational, reinforcing fundamental concepts in physical chemistry while minimizing financial burdens on educational institutions and laboratories.
Moreover, the financial implications of measurement accuracy extend to regulatory compliance, where the cost of non-compliance can be significantly higher than the investment in accurate measurement technologies. In the pharmaceutical and food industries, for example, manufacturers face stringent regulations that require precise quantification of active ingredients and contaminants. Non-compliance can result in product recalls, legal penalties, and reputational damage, underscoring the importance of investing in reliable measurement systems [54].
Digital assessment tools (DATs) have also been evaluated for their cost-effectiveness in quantifying alcohol consumption, which is essential for public health interventions. A systematic review identified various DATs that utilize animation features to enhance user engagement and improve the accuracy of alcohol consumption reporting [2]. While these tools are often freely available, their effectiveness in providing accurate data can influence the overall costs associated with alcohol-related health interventions, highlighting the need for careful evaluation of both financial and operational aspects.
In summary, the cost of measurement is a multifaceted issue that laboratories and manufacturers must navigate to ensure compliance with regulatory standards while maintaining accuracy and reliability. The balance between cost and performance is critical, as low-cost solutions may not always yield the desired measurement accuracy, potentially leading to greater costs in the long run due to non-compliance. As industries continue to evolve, the development of innovative, cost-effective measurement technologies will be essential for meeting regulatory requirements and enhancing operational efficiency.
Time efficiency: The speed of results can impact quality control processes
Time efficiency is a critical factor in analytical techniques, significantly impacting quality control processes across various industries. This efficiency is particularly crucial in sectors where rapid decision-making is essential, as it enables timely interventions and corrective actions. Research has explored the importance of time efficiency in different applications, highlighting its effects on product quality, process optimization, and regulatory compliance.
The assessment of time efficiency in analytical techniques such as FTIR spectroscopy, GC, HPLC, and NMR is critical for optimizing quality control processes across various industries. Each method possesses unique advantages and limitations regarding speed, accuracy, and applicability, significantly impacting the overall efficiency of quality control operations. FTIR spectroscopy stands out for its rapid analysis capabilities, providing results within minutes and allowing for quick identification and quantification of functional groups in samples. This technique’s ability to analyze samples with minimal preparation contributes to its time efficiency, enabling immediate decision-making in production environments, particularly in pharmaceutical and food product quality control [55,56]. However, while FTIR excels in speed, it may lack the specificity required for complex mixtures, potentially necessitating confirmatory analyses using other methods [32]. GC is another widely employed technique that excels in separating volatile compounds, typically delivering results within 15 to 30 min per sample, depending on mixture complexity and column selection. The speed of GC makes it an excellent choice for routine analysis in quality control laboratories, particularly in the petrochemical and food industries [37]. Nevertheless, GC requires careful sample preparation and can be limited by analyte volatility, sometimes necessitating derivatization techniques to enhance detection. HPLC is renowned for its versatility and ability to analyze a wide range of compounds, including non-volatile and thermally unstable substances. While HPLC typically requires longer analysis times than both FTIR and GC, often exceeding 30 min, it compensates with high resolution and accuracy. HPLC is particularly valuable in pharmaceutical quality control, where precise quantification of active ingredients is critical [38]. The development of faster HPLC methods, such as ultra-high-performance liquid chromatography (UHPLC), has further improved time efficiency, allowing for shorter run times without compromising resolution. NMR spectroscopy, while providing detailed structural information about compounds, generally requires more time than the other techniques mentioned. NMR analysis can take anywhere from 30 min to several hours, depending on sample complexity and the type of NMR experiment performed. Despite its longer analysis time, NMR offers unique advantages in terms of providing comprehensive structural elucidation and quantitative analysis, making it invaluable in research and development settings [40]. Its ability to analyze samples in their native state without extensive preparation is a significant benefit, though the time required for data acquisition and processing can be a drawback in high-throughput environments. The choice of analytical technique in quality control processes ultimately hinges on balancing time efficiency with the need for accuracy and specificity. FTIR and GC offer rapid analysis suitable for routine applications, while HPLC provides high-resolution data essential for complex mixtures. NMR, though slower, delivers unmatched structural insights that can guide product development and formulation. Understanding the strengths and limitations of each method is essential for optimizing quality control processes and ensuring the reliability of analytical results. As industries continue to seek faster and more accurate analytical methods, the development of hybrid techniques and improvements in existing technologies are likely to further enhance the time efficiency and accuracy of quality control processes. For instance, the integration of mass spectrometry with chromatographic techniques (GC-MS and LC-MS) has significantly improved the specificity and sensitivity of analyses, allowing for more rapid identification and quantification of compounds in complex mixtures. Similarly, advancements in NMR technology, such as the development of benchtop NMR spectrometers and the application of hyperpolarization techniques, are making NMR analysis more accessible and time-efficient for routine quality control applications. These technological advancements, coupled with the ongoing refinement of data processing algorithms and automation in sample handling, are continuously pushing the boundaries of what is possible in terms of speed and accuracy in analytical chemistry. The future of quality control processes will likely see an increased emphasis on multi-modal approaches, where complementary techniques are used in tandem to provide a more comprehensive and rapid analysis of complex samples. This approach could involve the use of FTIR or Raman spectroscopy for initial screening, followed by targeted analysis using GC, HPLC, or NMR for specific compounds of interest. Such strategies could significantly reduce overall analysis times while maintaining the high levels of accuracy and specificity required in modern quality control environments. Additionally, the integration of artificial intelligence and machine learning algorithms in data analysis and interpretation is expected to play a crucial role in further improving the time efficiency of analytical processes. These advanced computational methods can help in rapidly identifying patterns, predicting outcomes, and automating decision-making processes based on complex analytical data, thereby reducing the time required for manual data interpretation and enhancing the overall efficiency of quality control operations. As industries continue to face increasing pressure to improve productivity and maintain high quality standards, the ongoing development and optimization of analytical techniques will remain a critical area of focus. The ideal analytical method for quality control should strike a balance between speed, accuracy, and ease of use, while also being adaptable to a wide range of sample types and industrial applications. Achieving this balance requires a deep understanding of the principles underlying each analytical technique, as well as a keen awareness of the specific requirements and challenges faced in different industrial sectors. By continuously refining existing methods and exploring new analytical approaches, researchers and quality control professionals can work towards developing more efficient, reliable, and cost-effective solutions for ensuring product quality and safety across various industries.
Regulatory standards and compliance
The regulatory landscape for alcohol-based hand sanitizers (ABHS) has evolved significantly in response to the global health crisis triggered by the COVID-19 pandemic. Regulatory standards and compliance protocols are essential for ensuring the safety, efficacy, and quality of hand sanitizers, given their widespread use in public health initiatives. This section discusses the global guidelines set by regulatory bodies such as the WHO, the U.S. FDA, and the European Union (EU), as well as compliance challenges and ongoing efforts to address issues like substandard products and counterfeit sanitizers.
Global guidelines
WHO recommendations
The WHO has provided comprehensive guidelines for the formulation and production of alcohol-based hand sanitizers to ensure their effectiveness in preventing the transmission of infectious diseases. These recommendations emphasize the importance of a minimum alcohol concentration of 60% ethanol or 70% isopropanol for ensuring proper microbial inactivation. In addition to alcohol concentration, WHO guidelines stress the inclusion of emollients like glycerol to prevent skin dryness, a common side effect of alcohol-based sanitizers [8]. The guidelines also recommend strict manufacturing practices to ensure consistency and quality control across production facilities. Despite these recommendations, challenges in global adherence persist, particularly in low- and middle-income countries (LMICs), where limited regulatory enforcement has led to concerns about product quality and safety [4,10].
FDA regulations
In the United States, the FDA has established regulations for alcohol-based hand sanitizers under its Temporary Policy for Preparation of Certain Alcohol-Based Hand Sanitizer Products During the Public Health Emergency [19]. These regulations specify the required labeling for hand sanitizers, including the active ingredient, alcohol concentration, and intended use. According to FDA standards, ethanol and isopropyl alcohol are the only acceptable active ingredients, with a minimum concentration of 60% ethanol or 70% isopropyl alcohol required to effectively inactivate microorganisms [19]. The FDA also monitors compliance with Good Manufacturing Practices (GMP), conducting inspections and issuing recalls for products that fail to meet regulatory standards. Given the rapid rise in hand sanitizer production during the pandemic, the FDA’s regulatory role has become critical in ensuring the safety and efficacy of these products.
EU standards
In the European Union, the Biocidal Products Regulation (BPR) No 528/2012 governs the use of hand sanitizers as biocidal products, requiring manufacturers to demonstrate that their products are safe and effective through rigorous testing and evaluations [41]. Hand sanitizers must comply with the EU’s General Product Safety Directive (GPSD) 2001/95/EC, which mandates that all consumer products, including hand sanitizers, are safe for use under normal conditions [47]. Additionally, the EN 14476 standard, developed by the European Committee for Standardization (CEN), sets the requirements and testing methods for evaluating the virucidal activity of disinfectants, including alcohol-based hand sanitizers [53,57]. This standard is crucial for ensuring that hand sanitizers are effective against viral pathogens such as SARS-CoV-2, the virus responsible for COVID-19.
Compliance and quality control
As the global demand for hand sanitizers surged during the pandemic, concerns over product quality, including the risk of methanol contamination and non-compliance with alcohol concentration standards, became more pronounced. The proliferation of counterfeit and substandard products, often containing dangerous levels of methanol or other toxic substances, poses significant health risks [4,10]. Regulatory agencies have responded by implementing stricter enforcement measures, including more frequent inspections and monitoring of manufacturing processes. However, significant challenges remain, particularly in regions with limited regulatory infrastructure.
Required alcohol concentrations
The minimum alcohol concentration required for effective hand sanitization is a key regulatory standard that ensures the efficacy of the product. Research and guidelines from WHO and FDA stipulate a minimum of 60% ethanol or 70% isopropanol for alcohol-based hand sanitizers to effectively inactivate viral particles [8,19]. These standards are based on extensive research showing that alcohol concentrations below this threshold significantly reduce antimicrobial activity. However, concentrations above 90% may be less effective due to rapid evaporation, which reduces contact time [20].
Testing protocols for compliance
To ensure compliance with regulatory standards, hand sanitizers must undergo testing to verify their alcohol content and microbial efficacy. Microbiological testing protocols, such as EN 1500 (for hygienic hand rubs) and EN 14476 (for virucidal efficacy), are commonly used to evaluate the effectiveness of hand sanitizers in reducing microbial loads [57]. These protocols standardize the procedures for determining whether a product is effective against pathogens like bacteria and viruses. Additionally, safety assessments are conducted to evaluate the potential for skin irritation or sensitization, particularly in formulations that contain additional ingredients like fragrances or preservatives.
Regulatory agencies, such as the FDA and WHO, rely on these testing protocols to ensure that hand sanitizers meet the required safety and efficacy standards. Manufacturers are required to adhere to these standards, and non-compliance can result in recalls or bans on the sale of their products.
Challenges in regulatory compliance
While regulatory frameworks like those established by WHO, FDA, and EU are essential in ensuring the safety and effectiveness of hand sanitizers, challenges in implementation remain. A significant issue is the inconsistency in alcohol concentrations and the presence of harmful additives, such as methanol, in some sanitizers. Reports of substandard products have raised concerns about the reliability of hand sanitizers in preventing disease transmission [4,10]. Additionally, the increasing number of counterfeit products on the market has created difficulties for regulatory bodies in maintaining quality control, particularly as online sales and unverified supply chains proliferate.
Efforts to address these challenges include enhancing regulatory oversight and increasing public awareness about the risks of counterfeit hand sanitizers. However, continued collaboration between regulatory bodies, manufacturers, and consumers is essential for ensuring that hand sanitizers meet the required safety and efficacy standards.
Effectiveness and safety
The effectiveness and safety of ABHS are critical factors in their widespread adoption during public health emergencies, such as the COVID-19 pandemic. This section evaluates the antimicrobial efficacy of hand sanitizers, focusing on the role of alcohol concentration and the type of alcohol used, while also addressing safety concerns related to skin irritation, toxicity, and prolonged use. The balance between antimicrobial efficacy and safety is crucial for ensuring that hand sanitizers fulfill their intended purpose without causing harm to users.
Efficacy of different alcohol types
Antimicrobial activity of ethanol vs. isopropanol
Alcohol-based hand sanitizers rely primarily on ethanol and isopropanol as their active ingredients due to their broad-spectrum antimicrobial properties. Ethanol is particularly effective against a wide range of pathogens, including bacteria, fungi, and enveloped viruses such as coronaviruses and influenza [20]. The antimicrobial mechanism of alcohols, discussed in detail in the previous section, is primarily attributed to protein denaturation and membrane disruption.
Isopropanol,
while also effective against many pathogens, has a slightly
different antimicrobial profile. Studies suggest that isopropanol
may be less effective against certain bacterial strains compared to
ethanol at the same concentrations [18]. However, isopropanol is
preferred for some disinfectant applications, particularly surface
disinfection, due to its faster evaporation rate and lower risk of
skin irritation compared to ethanol [20].
Both alcohols are effective against enveloped viruses, but ethanol
is generally more potent in inactivating viruses like SARS-CoV-2
[20].
Impact of concentration on effectiveness
The concentration of alcohol in hand sanitizers is a key determinant of their antimicrobial effectiveness. Recommended minimum alcohol concentrations, as defined by WHO and FDA guidelines, are referenced throughout this review where analytically relevant [8,19]. Research shows that alcohol concentrations between 70% and 80% are optimal for reducing microbial load, with concentrations above 90% offering diminishing returns due to rapid evaporation, which reduces the contact time necessary for effective sanitization [20].
Alcohol concentrations below 60% are significantly less effective at killing pathogens and may fail to provide adequate protection against infections [8]. This underscores the importance of adhering to recommended alcohol content standards to ensure both safety and efficacy.
Safety considerations
Skin irritation and toxicity
While alcohol-based hand sanitizers are highly effective at reducing microbial load, their frequent use can lead to skin irritation and dryness. Ethanol and isopropanol can strip the skin of its natural oils, weakening the skin barrier and increasing susceptibility to irritation, dermatitis, and eczema [21]. Healthcare workers, who frequently use hand sanitizers, are particularly vulnerable to these adverse skin reactions [16,21].
Formulation ingredients, such as fragrances, preservatives, and colorants, can exacerbate these skin issues. Some preservatives, like parabens, and fragrances may cause allergic reactions or contact dermatitis in sensitive individuals [16,21]. To mitigate these effects, many hand sanitizers include emollients, such as glycerol or aloe vera, which help to moisturize the skin and counteract the drying effects of alcohol [12]. Products formulated with these moisturizing agents are generally better tolerated, enhancing user compliance with hand hygiene recommendations.
Ingestion risks
Ingestion of alcohol-based hand sanitizers is a significant safety concern, particularly in households with children. High alcohol concentrations, typically ranging from 60% to 95%, pose a risk of alcohol poisoning if ingested. Ethanol, the active ingredient in many hand sanitizers, can lead to hypoglycemia, metabolic acidosis, and central nervous system depression in large quantities. Isopropanol is even more toxic, with ingestion potentially leading to hypotension, respiratory failure, and even death [18].
To minimize the risk of accidental ingestion, it is essential for manufacturers to use child-resistant packaging and clear labeling. Public health campaigns should focus on educating caregivers and parents about the risks of ingestion and the importance of storing hand sanitizers out of reach of children [12,58,59].
Prolonged use and dermatological health
The
prolonged use of alcohol-based hand sanitizers, particularly during
public health emergencies like the COVID-19 pandemic, may lead to
cumulative effects on skin health. Repeated exposure to alcohol can
cause skin irritation, redness, and peeling, especially in
individuals with sensitive skin or pre-existing skin conditions
[20]. A systematic review by Vuppu et
al.
[60] highlighted that frequent use of hand sanitizers by healthcare
workers often leads to dermatitis, particularly in high-risk areas
like the hands and forearms [60].
To address these concerns, further research is needed to assess the long-term impact of regular hand sanitizer use on dermatological health. Investigating the cumulative effects, including the interaction between alcohol-based sanitizers and other skin-care products, will be crucial in developing guidelines that ensure safe and effective hand hygiene practices without compromising skin integrity [16,21].
Balancing effectiveness and safety
For hand sanitizers to be effective tools in public health, it is crucial to find a balance between their antimicrobial efficacy and their safety for users. Manufacturers must ensure that formulations contain sufficient alcohol concentrations to kill pathogens while minimizing the risk of skin irritation or toxicity. The addition of moisturizing agents, the use of safe preservatives, and proper packaging can help reduce adverse effects and improve the user experience. Moreover, public health campaigns should promote safe and responsible usage, emphasizing the need for proper hand hygiene while avoiding overuse or ingestion.
In summary, the effectiveness and safety of hand sanitizers are central to their role in infection control, particularly during pandemics. Ethanol and isopropanol, when used at appropriate concentrations, are highly effective against a wide range of pathogens. However, safety concerns, such as skin irritation, toxicity from ingestion, and the potential long-term effects of prolonged use, highlight the need for balanced formulations and informed public health strategies. Ensuring that hand sanitizers meet regulatory standards and are used correctly will help maximize their public health benefits while minimizing risks to users.
Market analysis during the COVID-19 era
The COVID-19 pandemic has significantly disrupted the global market for personal hygiene products, particularly ABHS. What was once a niche product became an essential tool in the fight against viral transmission, driving a dramatic surge in demand and reshaping the market dynamics. This section explores the trends, demand fluctuations, challenges, and economic implications of the hand sanitizer market during the COVID-19 era.
Trends and demand
Prior to the pandemic, the global hand sanitizer market was relatively stable, growing at an annual rate of 5-7% [20]. The market was characterized by a few established brands with a limited range of formulations. However, the onset of the COVID-19 pandemic caused a rapid and unparalleled increase in demand for hand sanitizers, with both consumers and healthcare institutions scrambling to secure sufficient supplies. Public health campaigns and government mandates advocating frequent hand hygiene further escalated the demand for ABHS worldwide [6,49].
The surge in demand prompted new players to enter the market, including distilleries, cosmetics manufacturers, and even non-pharmaceutical companies, who repurposed their production lines to meet the urgent need for hand sanitizers. A study conducted in India found that 98.73% of hand sanitizers available during the pandemic were alcohol-based, with ethyl alcohol being the most common active ingredient [15,47]. The pandemic also led to an increase in product variety, with a wide range of fragrances, moisturizers, and other additives being introduced to cater to changing consumer preferences [6,60,61].
Despite the increased production, global supply chains were disrupted, leading to shortages and price hikes. The high demand and limited supply resulted in market volatility, with some regions facing severe shortages while others saw excess stock [19,58,62]. Consequently, the market was characterized by both rapid expansion and instability, which presented unique challenges for manufacturers, distributors, and regulators.
Surge in production and variety of products
The COVID-19 pandemic triggered an explosion in the variety and volume of hand sanitizers produced. In response to the sudden surge in demand, many manufacturers outside the traditional hygiene and pharmaceutical sectors entered the market. Distilleries, cosmetic manufacturers, and other non-pharmaceutical companies quickly repurposed their facilities to produce hand sanitizers, resulting in a dramatic increase in available products [20]. This increase in production also led to a variety of new formulations. In addition to traditional alcohol-based sanitizers, formulations with added fragrances, aloe vera, glycerin, and other skin-conditioning agents gained popularity.
Consumers, increasingly concerned with both hygiene and skin care, began to prioritize sensory aspects such as scent and moisturizing properties in their hand sanitizers. This shift in consumer expectations pushed manufacturers to innovate and diversify their product offerings. The availability of alcohol-free alternatives, for example, increased during the pandemic, although these products did not achieve the same level of effectiveness against pathogens as alcohol-based sanitizers [20].
However, this rapid increase in production was accompanied by challenges regarding quality control and regulatory compliance. With so many new entrants into the market, many of whom lacked experience in manufacturing hygiene products, concerns about product safety and efficacy became more pronounced. Substandard products, including those with inadequate alcohol concentrations or harmful additives like methanol, surfaced in the market, raising serious public health concerns [20].
Consumer preferences and perceptions
As the pandemic progressed, consumer expectations surrounding hand sanitizers evolved. While effectiveness remained the primary concern, other factors such as sensory attributes, packaging, and brand trust gained prominence. The demand for sanitizers with pleasant fragrances, moisturizing properties, and skin-friendly formulations grew, with many consumers seeking products that provided a balance of efficacy and comfort [63-65].
The rising consumer focus on sustainability and ethical considerations also influenced purchasing decisions. Many consumers began prioritizing eco-friendly packaging, cruelty-free products, and brands that emphasized socially responsible manufacturing processes. This shift in consumer preferences has prompted brands to adjust their strategies, aligning their offerings with consumer values to remain competitive in a crowded market.
Furthermore, e-commerce platforms became a primary distribution channel during the pandemic, especially with the closure of physical stores or restrictions on in-person shopping. Online sales of hand sanitizers surged as consumers turned to digital platforms for convenience and safety. Small manufacturers, many of whom had limited physical retail presence, successfully reached a broader audience through online sales, further diversifying the market [10,63,65].
Challenges and issues
The surge in hand sanitizer demand during the COVID-19 pandemic presented a host of challenges for manufacturers, regulators, and consumers. These challenges ranged from ensuring product quality and regulatory compliance to combating counterfeit and substandard products in the market.
Quality control issues
One of the primary challenges resulting from the rapid expansion of the hand sanitizer market was maintaining consistent product quality. As new manufacturers entered the market, quality control became a significant concern. Many new producers lacked experience in manufacturing pharmaceutical-grade hygiene products, leading to variations in alcohol concentration, improper labeling, and the inclusion of harmful additives in some formulations [15,32].
A study conducted in Ethiopia revealed that many locally produced hand sanitizers did not meet the required alcohol concentration, highlighting the risks posed by substandard products [10]. Methanol contamination, previously discussed in the regulatory context, is further considered here from a market surveillance perspective. Methanol, a toxic alcohol, has been found in some hand sanitizers, posing serious health risks to consumers [19].
As a result, the importance of stringent quality control measures and regulatory oversight has never been more apparent. Regular inspections, adherence to GMP, and transparency in labeling are essential to ensure that hand sanitizers meet safety and efficacy standards.
Adulteration and counterfeit products
The rise of counterfeit and adulterated hand sanitizers has been one of the most concerning issues during the pandemic. Counterfeit hand sanitizers often contain harmful or ineffective ingredients, including methanol, which can lead to severe health complications, including poisoning and death. The rush to meet the demand for hand sanitizers created an environment ripe for exploitation by fraudulent actors [19,23,66].
Counterfeit products, which are often marketed as legitimate but contain substandard or dangerous ingredients, have proliferated, especially on e-commerce platforms. Consumers who are unaware of the risks associated with purchasing from unverified sources are at increased risk of exposure to these unsafe products.
To combat this issue, regulators have intensified their efforts to monitor the market and have issued warnings and recalls for products found to be non-compliant with safety standards. Moreover, public health campaigns aimed at educating consumers on how to identify counterfeit products and purchase from trusted sources are crucial to mitigating the risks associated with fraudulent hand sanitizers.
Economic implications
The hand sanitizer market’s rapid expansion during the COVID-19 pandemic has had significant economic implications. On one hand, the surge in demand has led to a boon for manufacturers and suppliers, with profits rising substantially in certain regions. On the other hand, the challenges posed by inconsistent product quality and counterfeit goods have created significant costs for public health systems and regulatory bodies, who must manage the fallout from substandard products.
Additionally, the economic strain caused by the pandemic has resulted in higher production costs, particularly due to supply chain disruptions. The cost of raw materials, including alcohol, has fluctuated, and the need for rigorous quality control testing and certification has further increased operational costs for manufacturers [3,6].
The future stability of the market will depend on the ability of manufacturers and regulators to ensure consistent product quality, combat counterfeit goods, and respond to changing consumer preferences. As the pandemic subsides, it is expected that the demand for hand sanitizers will normalize, although it is likely to remain higher than pre-pandemic levels due to sustained public health awareness.
Research gaps and future directions
The COVID-19 pandemic has highlighted the critical role of hand sanitizers in public health, yet it has also revealed significant gaps in our understanding of these products. While much progress has been made in measuring their efficacy, ensuring their safety, and improving formulations, there remains a need for further research to address the evolving challenges in this area. This section outlines the key research gaps identified from the existing literature and suggests future directions for continued improvement in the field of hand sanitization.
Identified gaps
Limited studies on long-term safety: The need for research on the effects of prolonged use of hand sanitizers on skin health
One of the most significant gaps in research is the long-term impact of regular hand sanitizer use, particularly concerning skin health. While short-term effects such as dryness, irritation, and dermatological conditions (e.g., eczema) are well-documented, the cumulative consequences of frequent hand sanitizer use remain underexplored. Studies have primarily focused on the immediate effects of alcohol-based sanitizers, but there is limited research on how continuous, long-term use may affect skin integrity over extended periods.
Frequent use of alcohol-based hand sanitizers, especially those with higher concentrations of ethanol or isopropanol, can strip natural oils from the skin, leading to compromised skin barriers and increased susceptibility to irritation. This issue is particularly relevant in healthcare settings where frequent use is necessary and among vulnerable populations, including children and individuals with pre-existing skin conditions [20]. Future research should explore the long-term effects of various sanitizer formulations on skin health, accounting for frequency of use, individual skin types, and environmental factors. Understanding these long-term effects will help inform public health recommendations and lead to safer hand sanitizers that balance antimicrobial efficacy with skin protection.
Need for Standardized Testing Methods: Establishing Uniform Protocols for Evaluating Hand Sanitizer Efficacy
Another key gap identified is the lack of standardized testing methods for evaluating hand sanitizer efficacy. While guidelines from regulatory bodies like the WHO and FDA set minimum standards for alcohol concentration and efficacy, discrepancies in testing methods and reporting still exist across different regions and manufacturers. This variability makes it difficult to accurately assess product performance and ensure consistency across products on the market [20].
Current testing protocols, such as the EN 1500 method for hygienic hand rubs and the EN 14476 method for virucidal activity, are not universally adopted or consistently applied. As a result, consumers and healthcare professionals may be misled about the efficacy of certain products. The development and adoption of standardized testing protocols across the global market are essential for ensuring the safety, consistency, and efficacy of hand sanitizers. This research should aim to establish uniform, internationally recognized testing procedures that can be implemented by manufacturers and regulators alike.
Future research directions
Innovations in Formulation: Exploring new ingredients and technologies to enhance effectiveness and user experience
Future research should explore innovative approaches to hand sanitizer formulations, focusing on enhancing both effectiveness and user experience. Although alcohol remains the most widely used active ingredient, alternatives or adjuncts, such as novel antimicrobial agents (e.g., benzalkonium chloride or chlorhexidine), could offer enhanced protection while minimizing skin irritation [20]. The development of formulations that balance efficacy with skin safety is crucial, particularly given the growing consumer demand for products that are both effective and gentle on the skin.
Research into alternative delivery systems, such as sprays, foams, or wipes, could improve usability, particularly in settings where traditional gel formulations are impractical. Moreover, integrating technologies like microencapsulation for controlled release of active ingredients could extend the sanitizer’s efficacy without requiring frequent reapplication. Such innovations could lead to more convenient and user-friendly hand sanitizers, which are essential for increasing compliance with hand hygiene practices.
In addition to antimicrobial agents, the inclusion of skin-conditioning ingredients such as aloe vera, glycerin, or other moisturizers could mitigate the drying effects of alcohol. These additions would improve consumer satisfaction and ensure that hand sanitizers are more comfortable to use, especially during prolonged periods.
Impact of prolonged use: Investigating the long-term effects of regular hand sanitizer use on skin and health
As discussed, the long-term effects of regular hand sanitizer use on skin health represent a critical area for future research. Research should aim to investigate the cumulative effects of prolonged alcohol exposure, including the risk of developing chronic skin conditions such as dermatitis or eczema. This research could also explore the impact of different formulations - specifically those containing skin conditioners - on skin health over extended use.
Moreover, research could examine whether prolonged use of alcohol-based sanitizers has systemic effects. Although alcohol is primarily absorbed through the skin in small amounts, frequent use could lead to cumulative absorption, which may have unintended health consequences, especially if the product contains additional additives. Studies that focus on the systemic effects of these products, especially for individuals with pre-existing health conditions, would provide essential insights for improving hand hygiene safety.
Consumer Behavior: Investigating factors influencing the purchase of counterfeit and substandard products
With the rise of counterfeit and substandard hand sanitizers during the COVID-19 pandemic, understanding the factors that drive consumers to purchase such products is crucial. Research in this area could investigate consumer behavior, including motivations behind purchasing counterfeit sanitizers despite the associated risks. Factors such as price sensitivity, product availability, and trust in brand names play significant roles in consumer purchasing decisions [27,60,67].
Additionally, understanding consumer awareness regarding the dangers of counterfeit products could guide the development of public health campaigns aimed at educating the public about safe hand sanitizer use. This research could also help identify how to better communicate the risks of counterfeit products to consumers and encourage them to purchase from reputable sources.
Innovations in detection methods for counterfeit products
As counterfeit products continue to pose a significant threat to public health, developing reliable and rapid detection methods for identifying counterfeit hand sanitizers is critical. Current methods of detection, such as laboratory-based testing, are not feasible for widespread use in retail settings. Future research should explore the potential for on-site detection technologies that can identify counterfeit or substandard products in real time. Techniques such as portable infrared spectroscopy or colorimetric assays could be used to quickly assess the authenticity and alcohol concentration of hand sanitizers in the field.
The integration of emerging technologies, such as blockchain, to track the authenticity of hand sanitizer products from production to point of sale, could also enhance transparency and trust in the market. Research in this area could also explore partnerships with e-commerce platforms to ensure that counterfeit products are effectively filtered out of online marketplaces.
Role of healthcare professionals in counterfeit detection
Pharmacists and other healthcare professionals play a key role in ensuring the safety and efficacy of hand sanitizers. Research should explore the knowledge gaps in healthcare professionals regarding counterfeit products and their ability to detect substandard sanitizers. Assessing the current training and education levels of healthcare professionals, as well as identifying the most effective methods for improving their ability to recognize counterfeit products, is vital for improving public health safety.
Educational programs targeted at pharmacists could be developed to enhance their capacity to identify counterfeit hand sanitizers and educate consumers about the dangers of using substandard products.
The COVID-19 pandemic has exposed several significant research gaps in the hand sanitizer market, particularly concerning long-term safety, standardized testing, formulation innovation, and the rise of counterfeit products. Addressing these gaps will require interdisciplinary research that includes public health, consumer behavior, regulatory science, and innovation in product development. By focusing on these areas, the research community can contribute to safer, more effective hand sanitizers and help mitigate the risks associated with substandard products. Through continued collaboration between researchers, manufacturers, and regulators, the next generation of hand sanitizers can be developed to better serve public health needs in future global health crises.
Conclusion and summary of findings
The COVID-19 pandemic has significantly heightened the importance of hand sanitizers as essential tools in public health strategies, especially in preventing the spread of infectious diseases. This review has examined the key components, measurement techniques, regulatory frameworks, and safety concerns related to hand sanitizers, particularly alcohol-based formulations. The findings from the literature emphasize the vital role that hand sanitizers play in reducing microbial load, particularly against enveloped viruses like SARS-CoV-2, and the critical need for standardized protocols to ensure their effectiveness and safety.
Overview of key insights
One of the primary findings of this review is the established efficacy of alcohol-based hand sanitizers in reducing microbial contamination, especially when they contain alcohol concentrations between 60% and 95%. Ethanol and isopropanol have been shown to be effective against a broad spectrum of pathogens, including bacteria and viruses, making them indispensable in the fight against infectious diseases [20]. The literature confirms that these products are particularly effective in preventing the transmission of viruses such as SARS-CoV-2, underscoring their importance in controlling viral outbreaks.
However, the surge in hand sanitizer production due to the pandemic has raised significant concerns about product quality and safety. Reports of substandard products, including those with insufficient alcohol content and the presence of harmful additives like methanol, have highlighted the need for improved regulatory oversight and quality control [4,10]. The increased production of hand sanitizers, coupled with the entry of new, often unregulated manufacturers, has led to inconsistencies in product formulations. This variability presents challenges for public health, as ineffective or dangerous products could undermine the broader goal of infection control.
The review also emphasizes the importance of regulatory standards in ensuring the safety and efficacy of hand sanitizers. Regulatory bodies such as the WHO, U.S. FDA, and EU have set guidelines for the formulation, labeling, and testing of hand sanitizers, yet implementation remains inconsistent. The lack of standardized testing protocols for measuring hand sanitizer efficacy complicates the assessment of their performance and creates opportunities for counterfeit or substandard products to enter the market [20].
Future directions
The findings from this review point to several important areas where future research and policy development are needed. One key area is the long-term impact of hand sanitizer use on skin health. While the immediate effects, such as skin irritation and dryness, are well-documented, the cumulative effects of prolonged use remain understudied. Research should investigate the dermatological consequences of long-term hand sanitizer use, particularly in healthcare settings where frequent application is necessary.
Another critical gap identified is the need for standardized testing methods to assess the efficacy of hand sanitizers. Variability in testing protocols and methodologies across regions and manufacturers makes it difficult to ensure consistency and reliability in product performance. Future research should focus on developing universal testing standards that can be adopted globally to ensure that all hand sanitizers meet minimum safety and efficacy requirements.
Innovation in product formulation is also essential for improving the user experience while maintaining antimicrobial effectiveness. Research into alternative antimicrobial agents, skin-conditioning additives, and user-friendly delivery systems will be critical as consumer preferences evolve. Additionally, the development of eco-friendly and sustainable hand sanitizers could address growing concerns about environmental impact, while still maintaining high standards of hygiene and safety.
In conclusion, alcohol-based hand sanitizers have proven to be an essential tool in preventing the spread of infectious diseases, particularly during the COVID-19 pandemic. However, challenges related to product quality, safety, and regulatory compliance highlight the need for continuous improvement in manufacturing practices, testing standards, and regulatory enforcement. Future research must address gaps in understanding the long-term effects of hand sanitizer use, particularly on skin health, and establish standardized protocols for efficacy testing to ensure consistency and reliability. By addressing these challenges, stakeholders can develop safer, more effective hand sanitizers and enhance public confidence in these vital hygiene products. This review has examined the key components, measurement techniques, regulatory frameworks, and safety concerns related to hand sanitizers, particularly alcohol-based formulations. The findings from the literature emphasize the vital role that hand sanitizers play in reducing microbial load, particularly against enveloped viruses like SARS-CoV-2, and the critical need for standardized protocols to ensure their effectiveness and safety.
Implications
The findings from the literature review on hand sanitizers have broad implications for public health policy, manufacturers, and regulatory bodies. As the importance of hand sanitizers has grown, particularly during the COVID-19 pandemic, it is critical that policymakers, manufacturers, and regulators work together to ensure the safety, efficacy, and quality of these products. This section discusses the implications for each of these stakeholders and outlines recommendations for improving the overall effectiveness of hand sanitizers.
For public health policy: Recommendations for guidelines and practices
The review underscores the essential role that hand sanitizers play in reducing the transmission of infectious diseases, particularly in healthcare settings and community environments. However, the variability in product quality and efficacy highlights the need for stronger public health policies and guidelines to ensure the proper use of these products.
Recommendations for Public Health Policy: Establish Clear and Stringent Guidelines: Regulatory bodies, including the WHO and the U.S. FDA, should continue to reinforce the importance of standardized formulations. Clear minimum alcohol concentration thresholds (60-95%) must be enforced globally to ensure product efficacy.
Enhance Public Awareness: Public health campaigns should focus on educating the public about proper hand hygiene practices, the correct use of hand sanitizers, and how to identify safe and effective products. Such initiatives are especially important in areas with limited access to water and soap.
Combat Counterfeit Products: With the proliferation of counterfeit and substandard hand sanitizers, there is a need for ongoing consumer education on how to avoid fraudulent products. Public health efforts should inform consumers about how to distinguish authentic, compliant hand sanitizers from potentially harmful alternatives.
Ongoing Monitoring and Surveillance: Regulatory agencies must implement routine market surveillance to assess hand sanitizers for compliance with safety and efficacy standards. Regular inspections of production facilities, coupled with random sampling of products in the market, will help identify substandard products and remove them from circulation before they harm public health.
For manufacturers and regulators: Insights into quality control and product development
The findings emphasize the need for manufacturers to adhere to quality control measures to ensure the consistency and safety of hand sanitizers. The rapid expansion of hand sanitizer production during the COVID-19 pandemic has increased the risk of substandard products entering the market.
Implications for Manufacturers: GMP: Manufacturers should adopt rigorous GMP to ensure the consistent quality of hand sanitizers. This includes strict quality assurance procedures for raw materials, production processes, and final product testing. Manufacturers must also prioritize transparency, ensuring that their products consistently meet established alcohol concentration and safety standards.
Innovation in Formulation: Manufacturers should invest in research and development to improve product formulations. This includes incorporating skin-conditioning agents to reduce irritation, exploring alternative antimicrobial agents, and experimenting with new delivery methods (e.g., sprays or wipes) to enhance user experience.
Product Labeling and Transparency: Clear and accurate product labeling is vital. Manufacturers should include all relevant information on product packaging, such as alcohol concentration, active ingredients, potential allergens, and safety warnings. Transparent labeling helps consumers make informed decisions and ensures compliance with regulatory requirements.
Implications for Regulators: Standardization of Testing Methods: Regulatory bodies must work to establish and enforce standardized testing protocols for evaluating the efficacy of hand sanitizers. Current variability in testing methods leads to inconsistent results, making it difficult for consumers and healthcare professionals to reliably assess the effectiveness of these products. By developing universal testing methods, regulators can ensure that all hand sanitizers meet minimum standards.
Tackling Counterfeit and Substandard Products: Regulators must step up their efforts to identify and eliminate counterfeit products. Stronger enforcement measures, including increased inspections of manufacturers and distributors, as well as collaboration with law enforcement agencies to combat fraud, are essential to safeguard public health. Regulatory bodies must also collaborate with global organizations to ensure that guidelines are uniformly followed worldwide.
In conclusion, the findings from this review highlight both the critical role of hand sanitizers in public health and the need for more rigorous quality control, innovation, and standardized testing to ensure their safety and effectiveness. For public health policy, a comprehensive approach that includes clear guidelines, public education, and continuous surveillance is necessary. Manufacturers must prioritize quality assurance and invest in product innovation to meet the evolving needs of consumers, while regulatory bodies must enforce and standardize testing to ensure the consistency and reliability of hand sanitizers across markets.
By addressing these implications, stakeholders can work collaboratively to improve public health outcomes, enhance consumer confidence, and ensure that hand sanitizers continue to play a vital role in controlling the spread of infectious diseases worldwide.
Acknowledgements
The author is grateful to the Faculty of Science at Sriracha, Kasetsart University, Sriracha Campus, for their valuable support and resources that contributed to the completion of this manuscript.
Declaration of generative AI in scientific writing
The author acknowledges the use of generative AI tools (e.g., QuillBot and ChatGPT by OpenAI) in the preparation of this manuscript, specifically for improving language clarity and grammar. In addition, Google Gemini was used to generate the graphical abstract. These tools were used under human oversight and did not contribute to content generation, data interpretation, or scientific analysis. The authors take full responsibility for the content and conclusions of this work.
CRediT author statement
Panya Khaenamkaew: Conceptualization; Methodology; Data curation; Writing - Original draft preparation; Visualization; Investigation; Supervision; Validation; Writing - Reviewing and Editing.
References
[1] MP Cruz, E Santos, MAV Cervantes and ML Juárez. COVID-19, a worldwide public health emergency. Revista Clínica Española 2021; 221(1), 55-61.
[2] G Battineni, N Chintalapudi and F Amenta. Forecasting of COVID-19 epidemic size in four high hitting nations (USA, Brazil, India and Russia) by Fb-Prophet machine learning model. Applied Computing and Informatics 2025; 21, 2-11.
[3] A Blair, M de Pasquale, V Gabeff, M Rufi and A Flahault. The end of the elimination strategy: Decisive factors towards sustainable management of COVID-19 in New Zealand. Epidemiologia 2022; 3(1), 135-147.
[4] S Dalasile, E Itoba Tombo, BS Madonsela, PP Mpungose, N Mshicileli and M Menziwa. Alcohol-based hand sanitizers used for COVID-19 prevention in the informal settlements of Cape Town, South Africa. COVID 2024; 4, 1655-1675.
[5] RK Masters, LY Aron and SH Woolf. Life expectancy changes during the COVID-19 pandemic, 2019-2021: Highly racialized deaths in young and middle adulthood in the United States as compared with other high-income countries. American Journal of Epidemiology 2024; 193(1), 26-35.
[6] M Meier and E Pinto.COVID-19 supply chain disruptions. European Economic Review 2020; 48(1), 139-170.
[7] KO Ahmed. Impact of COVID-19 pandemic on hand washing process and water consumption. Eurasian Journal of Science & Engineering 2021; 7(1), 228-245.
[8] World Health Organization. Guide to local production: WHO-recommended handrub formulations, Available at: https://www.who.int/gpsc/5may/Guide_to_Local_Production.pdf, accessed November 2025.
[9] HV Annegowda, M Zuber, JC Darshan, S Ghosh and P Paul. Hand sanitizer: A comprehensive narrative review. International Journal of Pharmaceutical Science 2021; 66(1), 109-114.
[10] MN Selam. Hand sanitizers marketed in the streets of Addis Ababa, Ethiopia, in the era of COVID-19: A quality concern. Risk Management and Healthcare Policy 2021; 66(1), 109-114.
[11] AP Golin, D Choi and A Ghahary. Hand sanitizers: A review of ingredients, mechanisms of action, modes of delivery, and efficacy against coronaviruses. American Journal of Infection Control 2020; 48(9), 1062-1067.
[12] M Islam, K Shahin Ahmed, R Karim, BD Nath, S Prosad Moulick, R Islam, SM Hassan, H Hossain, M Moniruzzaman, S Jahan, AA Shaikh and PE Georghiou. Alcohol‐based hand sanitizers amid COVID‐19: Chemical formulation, analysis, safety. ChemistrySelect 2022; 7(45), e202203290.
[13] MA Misri, KH Qadr and MA Rahmatullah. SEIR mathematical model with the use of hand sanitizers to prevent the spread of covid-19 disease. CAUCHY: Jurnal Matematika Murni dan Aplikasi 2024; 9, 138-154.
[14] MA Cook and N Brooke. Event-based surveillance of poisonings and potentially hazardous exposures over 12 months of the COVID-19 pandemic. International Journal of Environmental Research and Public Health 2021; 18(21), 11133.
[15] KG Piparva, S Dutta, S Singhal and R Shah. A critical evaluation of contents, labeling, and cost of hand sanitizers marketed in India during COVID-19 pandemic. Journal of Family Medicine and Primary Care 2024; 13, 2289-2294.
[16] JLJ Jing, T Pei Yi, RJC Bose, JR McCarthy, N Tharmalingam and T Madheswaran. Hand sanitizers: A review on formulation aspects, adverse effects, and regulations. International Journal of Environmental Research and Public Health 2020; 17(9), 3326.
[17] D Singh, K Joshi, A Samuel, J Patra and N Mahindroo. Alcohol-based hand sanitisers as first line of defence against SARS-CoV-2: A review of biology, chemistry and formulations. Epidemiology & Infection 2020; 148, e229.
[18] G McDonnell and AD Russell. Antiseptics and disinfectants: Activity, action, and resistance. Clinical Microbiology Reviews 2020; 12, 147-179.
[19] U.S. Food and Drug Administration. Temporary policy for preparation of certain alcohol-based hand sanitizer products during the public health emergency (COVID-19) guidance for industry. Center for Drug Evaluation and Research (CDER), Food and Drug Administration, Maryland, USA.
[20] G Kampf and A Kramer. Epidemiologic background of hand hygiene and evaluation of the most important agents for scrubs and rubs. Clinical Microbiology Reviews 2004; 17(4), 863-893.
[21] A Mahmood, M Eqan, S Pervez, HA Alghamdi, AB Tabinda, A Yasar, K Brindhadevi and A Pugazhendhi. COVID-19 and frequent use of hand sanitizers; human health and environmental hazards by exposure pathways. Science of the Total Environment 2020; 742, 140561.
[22] P Khaenamkaew, C Tanghengjaroen, D Manop, WPN Ayuthaya, K Wongsorntam and N Noulkhow. Assessment of uncertainty estimation for measurement of ethanol concentration in COVID-19 hand sanitizer using FTIR spectroscopy. Journal of Chemical Metrology 2023; 17(1), 1-14.
[23] M Adugo, SJ Bunu, NT Jonathan and D Ogoina. Physicochemical evaluation of the potency and viability of alcohol-based hand sanitizers. EAS Journal of Parasitology and Infectious Diseases 2023; 5(6), 61-65.
[24] P Prajapati, H Desai and C Chandarana. Hand sanitizers as a preventive measure in COVID-19 pandemic, its characteristics, and harmful effects: A review. Journal of the Egyptian Public Health Association 2022; 97(1), 6.
[25] RY Booq, AA Alshehri, FA Almughem, NM Zaidan, WS Aburayan, AA Bakr, SH Kabli, HA Alshaya, MS Alsuabeyl, EJ Alyamani and EA Tawfik. Formulation and evaluation of alcohol-free hand sanitizer gels to prevent the spread of infections during pandemics. International Journal of Environmental Research and Public Health 2021; 18(12), 6252.
[26] F Fallica, C Leonardi, V Toscano, D Santonocito, P Leonardi and C Puglia. Assessment of Alcohol-based hand sanitizers for long-term use, formulated with addition of natural ingredients in comparison to WHO formulation 1. Pharmaceutics 2021; 13(4), 571.
[27] NQ Thang and LV Tan. Determination of methanol, acetaldehyde, and ethyl acetate in thousand folds of ethanol sample by headspace gas chromatography with mass spectrometry. International Journal of Analytical Chemistry 2023; 2023(1), 8851265.
[28] C Quan, HM Li, T Huang, W Zhang, ZT Ding and YX Shen. High-precision analysis of ethanol in bioethanol by gas chromatography with flame ionization detector. Accreditation and Quality Assurance 2012; 17, 535-541.
[29] ND Nisbar, SK Jamal Khair, NB Bujang and AY Mohd Yusop. Determination of ethanol, isopropyl alcohol and methanol in alcohol-based hand sanitiser to ensure product quality, safety and efficacy. Scientific Reports 2023; 13(1), 9478.
[30] DI Ellis, J Ellis, H Muhamadali, Y Xu, AB Horn and R Goodacre. Rapid, high-throughput, and quantitative determination of orange juice adulteration by Fourier-transform infrared spectroscopy. Analytical Methods 2016; 8(28), 5581-5586.
[31] S Banik, SK Melanthota, AA Vannathan, KK Mahato, SS Mal and N Mazumder. Spectroscopic methods for assessment of hand sanitizers. Chemical Papers 2022; 76(8), 4907-4918.
[32] AR Chowdhury, U Burney, D King, TA Lee, D Hutter and T Hutter. Detection of toxic contaminants in alcohol-based hand sanitizers using infrared spectroscopy. Applied Spectroscopy Practica 2023; 1(2), 27551857231204630.
[33] SA Roget, TR Heck, KA Carter-Fenk and MD Fayer. Ion/water network structural dynamics in highly concentrated lithium chloride and lithium bromide solutions probed with ultrafast infrared spectroscopy. The Journal of Physical Chemistry B 2023; 127(20), 4532-4543.
[34] S Fantini and A Sassaroli. Frequency-domain techniques for cerebral and functional near-infrared spectroscopy. Frontiers in Neuroscience 2020; 14, 300.
[35] MA Butt, M Juchniewicz, M Słowikowski, Ł Kozłowski and R Piramidowicz. Mid-infrared photonic sensors: exploring fundamentals, advanced materials, and cutting-edge applications. Sensors 2025; 25(4), 1102.
[36] A Dabrowska, A Schwaighofer and B Lendl. Mid-infrared dispersion spectroscopy as a tool for monitoring time-resolved chemical reactions on the examples of enzyme kinetics and mutarotation of sugars. Applied Spectroscopy 2024; 78(9), 982-992.
[37] M Bedner, M Bedner, JA Murray, AA Urbas, WA MacCrehan and WB Wilson. A comparison of measurement methods for alcohol-based hand sanitizers (NISTIR 8342). US Department of Commerce, National Institute of Standards and Technology, Maryland, United States.
[38] A Gupta, SP Stratton and PB Myrdal. An HPLC method for quantitation of perillyl alcohol in a topical pharmaceutical cream formulation. Journal of Pharmaceutical and Biomedical Analysis 2005; 37(3), 447-452.
[39] R Bachhav, P Bachhav, M Bhamare, R Bachhav and G Sonawane. A concise review on high-per-formance liquid chromatography. Journal of Pharmaceutical Research 2023; 8(2), 340.
[40] HJR Caleja-Ballesteros, JI Ballesteros and MC Villena. Non-deuterated 1H NMR spectrometric method for the rapid and direct analysis of alcohol content in hand sanitizers. Philippine Journal of Science 2022; 151(3), 937-344.
[41] AE AISBL, G Graß and P Madsen. Application of regulation (EU) No 528/2012 (“BPR”) to disinfectant biocidal active substances generated in situ by devices. Aqua Europa AISBL, Brussels, Belgium, 2017.
[42] C Prakash and R Mahar. Chemical profiling and quality assessment of food products employing magnetic resonance technologies. Foods 2025; 14(14), 2417.
[43] PK Kumar, PB Dudhe and SS Nilewar. Analytical method validation in pharmaceutical quality control: Current guidelines and challenges. Journal of Pharmaceutical Research and Integrated Medical Sciences 2025; 2025, 26-39.
[44] MMS Alanazi, YSJ Alruwaili, MMR Alruwaili, HMR Alrwayli, MJN Alruwaili, FHBF Alanazi, SGS Alanazi, BGS Alanazi6, AKD Alanazi and AIA Alenezi. Advancements in analytical techniques for pharmaceutical quality control. Journal of International Crisis and Risk Communication Research 2024; 7(S10), 51-72.
[45] O Pani and T Górecki. Comprehensive 2-dimensional gas chromatography (GC×GC) in environmental analysis and monitoring. Analytical and Bioanalytical Chemistry 2006; 386(4), 1013-1023.
[46] ISA Alsharari, AMDA Alsharari and F Alsharari. Quality control of laboratory medicine: Preventing errors for diagnostic reliability. Saudi Journal of Medicine and Public Health 2024; 1(1), 114-123.
[47] J Ruohonen. A review of product safety regulations in the European Union. International Cybersecurity Law Review 2024; 1(1), 114-123.
[48] KO Hogan and GR Fraga. Compliance with standards for STARD 2015 reporting recommendations in pathology. American Journal of Clinical Pathology 2020; 154(6), 828-836.
[49] A Kramer, M Arvand, B Christiansen, S Dancer, M Eggers, M Exner, D Müller, NT Mutters, I Schwebke and D Pittet. Ethanol is indispensable for virucidal hand antisepsis: memorandum from the alcohol-based hand rub (ABHR) Task Force, WHO Collaborating Centre on Patient Safety, and the Commission for Hospital Hygiene and Infection Prevention (KRINKO), Robert Koch Institute, Berlin, Germany. Antimicrobial Resistance & Infection Control 2022; 11, 93.
[50] N Abrigo, C Ruzicka, P Faustino, N Stiber, A NguyenPho, T O’Connor and D Shakleya. Development and validation of a headspace GC-MS method to evaluate the interconversion of impurities and the product quality of liquid hand sanitizers. AAPS Open 2022; 8(1), 1.
[51] F De Ridder, R Braspenning, JS Ordonez, G Klarenbeek, P Lauwers, KJ Ledeganck, D Delbeke and CD Block. Early feasibility study with an implantable near-infrared spectroscopy sensor for glucose, ketones, lactate and ethanol. Plos One 2024; 19, e0301041.
[52] The British Standards Institution. Chemical disinfectants and antiseptics-hygienic handrub-test method and requirements (phase 2, step 2). BSI Standards, London, 1997.
[53] DS Ramkumar. EN 14476 Standard for infection control. pp. Swiss CRO and testing lab, Available at: https://microbe-investigations.com/en-14476-standard-for-infection-control, accessed November 2025.
[54] ML Wang, YM Choong, NW Su and MS Lee. A rapid method for determination of ethanol in alcoholic beverages using capillary gas chromatography. Journal of Food and Drug Analysis 2003; 11(2), 3.
[55] MMS Alanazi, YSJ Alruwaili, MMR Alruwaili, HMR Alrwayli, MJN Alruwaili, FHBF Alanazi, SGS Alanazi, BGS Alanazi6, AKD Alanazi and AIA Alenezi. Advancements in analytical techniques for pharmaceutical quality control. Journal of International Crisis and Risk Communication Research 2024; 7(S10), 51-72.
[56] Riyanto and DANP Sumira. Validation method and quality control of the hand sanitizer for Covid-19 using gas chromatography. AIP Conference Proceedings 2022; 2645(1), 030008.
[57] CDC Handwashing. Clean hands save lives. Centers for Disease Control and Prevention, Atlanta, United States, 2020.
[58] FN Faizah, AN Khabibah and S Afidah. Panic buying in the era of covid-19 pandemic: Consumer behavior perspective of Monzer Khaf. ISLAMICONOMIC: Jurnal Ekonomi Islam 2022; 13, 23-40.
[59] A Afework and A Tamene. Uncovering the obstacles: A comprehensive analysis of barriers to hand hygiene adherence among healthcare providers: A systematic review. BMC Infectious Diseases 2025; 25(1), 502.
[60] S Vuppu, T Mishra and A Chinamgari. Use of hand sanitizers in COVID-19 prevention: A comprehensive overview. Pharmacoepidemiology 2023; 2(3), 257-271.
[61] CA Enwuru, OO Awoderu and E Emelda. Evaluation of antibacterial efficacy of randomly selected alcohol based hand sanitizers sourced from grocery shops within lagos metropolis on some local bacterial strains in COVID-19 era. Microbiology & Infectious Diseases 2021; 5(3), 1-9.
[62] DD Gummin, JB Mowry, MC Beuhler, DA Spyker, AC Bronstein, LJ Rivers, BS Nathaniel, PT Pham and J Weber. 2020 annual report of the American association of poison control centers’ national poison data system (NPDS): 38th annual report. Clinical Toxicology 2021; 59, 1282-1501.
[63] RA Leslie, SS Zhou and DR Macinga. Inactivation of SARS-CoV-2 by commercially available alcohol-based hand sanitizers. American Journal of Infection Control 2021; 49(3), 401-402.
[64] Organisation for Economic Co-operation and Development. Local lymph node assay. OECD guidelines for the testing of chemicals. Organisation for Economic Co-operation and Development, Paris, France, 2002.
[65] M Pomffyová and L Veselovská. The impact of COVID-19 on purchase behavior changes in smart regions. Computers 2023; 12(2), 38.
[66] D Cucinotta and M Vanelli. WHO declares COVID-19 a pandemic. Acta Biomedica 2020; 19, 157-160.
[67] F Torko. 2025, The efficacy of foam-based hand sanitizers for the control of enveloped and non-enveloped virus transmission. Master’s Thesis. University of Arkansas, Arkansas, United States.