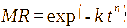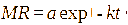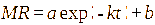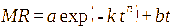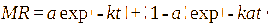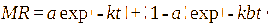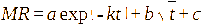Trends
Sci.
2026;
23(6):
12279
Development and Comparison of Drying Tropical Herbal Strategies for Annona muricata Leaves: Integrating of Effective Moisture Diffusivity Using Antioxidant Activity, FTIR Ratios, and Color Attributes
Dessy
Agustina Sari1,2,
![]() ,
Moh Djaeni1,*,
,
Moh Djaeni1,*,
![]() ,
Devi Yuni Susanti3,
,
Devi Yuni Susanti3,
![]() ,
,
Joko
Nugroho Wahyu Karyadi4,
![]() ,
Olly Sanny Hutabarat5,
,
Olly Sanny Hutabarat5,
![]() ,
Setia Budi Sasongko1,
,
Setia Budi Sasongko1,
![]() ,
,
Aji
Prasetyaningrum1,
![]() and Ching Lik Hii6,
and Ching Lik Hii6,
![]()
1Department of Chemical Engineering, Universitas Diponegoro, Semarang 50275, Indonesia
2Chemical Engineering Program, Universitas Singaperbangsa Karawang, Karawang 41361, Indonesia
3Department of Food and Agriculture Products Technology, Universitas Gadjah Mada, Yogyakarta 55281, Indonesia
4Department of Agricultural Engineering and Biosystems, Universitas Gadjah Mada, Yogyakarta 55281, Indonesia
5Department of Agricultural Technology, Hasanuddin University, Makassar 90245, Indonesia
6Department of Chemical and Environmental Engineering, University of Nottingham,
Selangor Darul Ehsan 43500, Malaysia
(*Corresponding author’s e-mail: [email protected])
Received: 14 October 2025, Revised: 27 October 2025, Accepted: 3 November 2025, Published: 10 January 2026
Abstract
Drying strategy employed exerts a significant influence on the kinetics and functional quality of Annona muricata (soursop) leaves. However, the current literature offers a paucity of practical, non-destructive indicators to inform process decisions. The present study sets out to compare sun drying (SD), room-temperature drying (RTD), convective tray drying (CTD; 40 - 60 °C), and microwave drying (MWD; 120 - 380 W), models thin-layer curves, and integrates effective moisture diffusivity (Deff) with quality metrics - antioxidant activity (IC50, DPPH), FTIR ratios (RQ, R1 - R3), and color attributes (ΔE, a*). Multi-parameter models have been shown to outperform simpler forms. The Midilli model provided the most precise global fit (R2 > 0.95; RMSE < 0.05), while Jenna-Das performed well in specific convective subsets. As the temperature/power were increased, Deff increased and reached a peak at an MWD of 380 W. This resulted in an approximate acceleration of ~225× compared to the CTD 50 °C and a drying time of approximately ≈ 4 min. A clear trade-off emerged: CTD 40 °C exhibited a preserved appearance (ΔE ≈ 2.7) but under-retained phenolics (weaker IC50), whereas CTD 60 °C and MWD 380 W produced higher ΔE (> 8) yet superior FTIR ratios (RQ, R1 - R3) and stronger antioxidant activity; mid-power MWD (120 - 250 W) was detrimental. The colorimetric - spectroscopic linkages were found to be quantitative, with ΔE-IC50 exhibiting a weak-moderate relationship, and a* demonstrating a strong colleration with RQ (R2 ≈ 0.73). Chemometrics (PLSR with VIP) identified RQ/R3 as dominant predictors (R2{LOOCV} ≈ 0.33), thereby converting FTIR from descriptive readout to an actionable inline/at-line QC. Collectively, these results establish a predictive quality-control framework - using ΔE and a* with RQ - for efficient selection, development, and implementation of tropical herbal drying technologies, and provide actionable set-points (optimal: MWD 380 W; convective alternative: CTD 60 °C) that balance speed and bioactive retention.
Keywords: Soursop leaves, Effective moisture diffusivity, IC50, Thin-layer drying, FTIR ratio
Introduction
The utilization of tropical herbal leaves in functional foods and phytopharmaceuticals has
increased in response to the demand for natural and sustainable products. While the drying process has been demonstrated to extend shelf life, this stage can concomitantly result in a decline in phenolics and antioxidant capacity due to pigment conversion, oxidation, and structural degradation [1-6]. It has been demonstrated that microwave and freeze-drying techniques result in superior preservation of bioactivity when compared to conventional ovens. However, sun drying remains a viable option in tropical settings despite the high variability observed [7-12]. Consequently, there is a practical need for efficient drying strategies and non-destructive, predictive indicators that bridge laboratory analysis and industrial decision-making.
Annona muricata (soursop) leaves have been found to be rich in phenolics (e.g., gallic acid, catechin, quercetin, rutin) and flavonoids, supporting robust antioxidant activity and reported cytotoxic/anti-inflammatory effects that motivate their use in herbal teas and extracts [13-16]. However, post-harvest processing is fragmented: convective, solar, microwave, and other modalities have yielded variable outcomes for phenolics, antioxidant capacity, and color [17-19]. Consequently, a concise, integrative approach is imperative to balance process efficiency, speed, and bioactivity retention.
The quality of tropical herbal materials is commonly assessed via colorimetry (ΔE, a*) as markers of pigment/phenolic degradation, Fourier-transform infrared spectroscopy (FTIR) to track functional-group shifts associated with oxidation, and IC50 (DPPH) as a global indicator of radical-scavenging capacity [5,20-23]. Prior studies have frequently focused on a single indicator or a single drying method, thereby limiting comparative insight and rapid translation [24-27]. Emerging literature suggest that integrating color-FTIR-bioactivity may facilitate rapid, non-destructive prediction of herbal quality, particularly when combined with chemometrics [28,29,35,36]. However, such systematic integration has rarely been applied to A. muricata leaves. Three concise gaps emerge from this analysis: (i) limited cross-method evaluation of drying; (ii) insufficient integration of colorimetry and FTIR with bioactivity; and (iii) lack of rapid parameters suitable for real-time quality control.
In order to address these gaps, the present study introduces a multiscale integration of drying kinetics and modelling (sun/room drying; convective 40 - 60 °C; microwave 120 - 380 W) using established thin-layer models (Midilli, Page, Henderson-Pabis), with effective moisture diffusivity (Deff) as a process-efficiency descriptor [30-34]. This study proposes a novel approach to predicting IC50 trends, offering a rapid and non-destructive surrogate for decision-making process. This study utilizes a combination of simple colorimetric indicators (ΔE, a*) with informative FTIR ratios (RQ, R1 - R3) to achieve this objective, aligning with green processing principles and scalable quality control [28,29,35-39].
The objective of this research is 2-fold: (i) to evaluate the effects of temperature, power, and drying method on drying kinetics (thin-layer models, Deff) and (ii) to assess product quality (IC50, FTIR ratios, and color). Additionally, the study aims to quantify the structure–function relationships linking colorimetric parameters (ΔE, a*) and FTIR indices (RQ, R1 - R3) with antioxidant capacity (IC50). The results of this study are expected to (iii) enable a predictive quality control framework in which ΔE and a*, in combination with RQ, serve as rapid and non-destructive indicators suitable for real-time process monitoring, and (iv) to provide actionable drying guidelines-comparing convective and microwave approaches - that balance process efficiency and phenolic retention for tropical herbal processing.
Materials and methods
Materials
Plant materials
Fresh soursop leaves (Annona muricata; Figure 1) were obtained from the cultivation garden of Diponegoro University, Tembalang, Semarang, Indonesia. The identification process was conducted in the Process Laboratory, and voucher specimens were stored for the purpose of taxonomic authentication. Prior to the commencement of the treatment, the leaf stalks were meticulously separated, the leaves were thoroughly washed and then stored at a temperature of 4 °C for 24 h. The samples were weighed (0.7 - 1.6 g on average) and their thickness was measured by means of a micrometer screw gauge (Mitutoyo, Japan; accuracy 0.1 mm). In each drying treatment, 30 g of fresh leaves were utilized (based on wet weight) with 3 replicates (n = 3).
Chemicals
Analytical-grade solvents (ethanol, methanol, n-hexane) were obtained from Merck (Germany). The standards of gallic acid, quercetin, and DPPH (2,2-diphenyl-1-picrylhydrazil) were obtained from Sigma-Aldrich (USA) and potassium bromide (KBr, ≥99%) was utilized for the preparation of FTIR pellets.

Figure 1 Annona muricata: (a) fruits, (b) fresh leaves, and (c) dry leaves.
Drying process
The drying treatments (Figure 2) comprised sun drying (SD), room temperature drying (RTD), convective tray drying (CTD), and microwave drying (MWD).
Sun drying (SD). It was conducted in an outdoor environment between 9:00 a.m. and 3:00 p.m. A total of 30 g of the sample was placed into a thin cloth bag (30×23 cm2) and placed on a stainless-steel tray (40×30 cm2).
Room temperature drying (RTD): It was conducted at room temperature between 27.5 and 31.1 °C with a relative humidity of 57.4% - 68.5%, in an indoor environment. The experiment was conducted from 08:00 a.m. to 16:00 p.m., and the temperature and humidity levels were monitored at regular intervals. The experiment was conducted in a way ascertaining protection from direct sunlight.
Convective tray drying (CTD). It utilized a Sharp EO-35ST oven (Tokyo, Japan; 1000 W with maximum temperature at 250 °C). The oven was operated at temperatures of 40, 50, and 60 °C, with 75 mm between trays.
Microwave drying (MWD). It was carried out using a Samsung MS20A3010AL microwave (Seoul, Korea; maximum 700 W) operated at 120, 250, and 380 W. A total of 30 g of the sample was spread evenly on a glass tray (205×205 mm2).
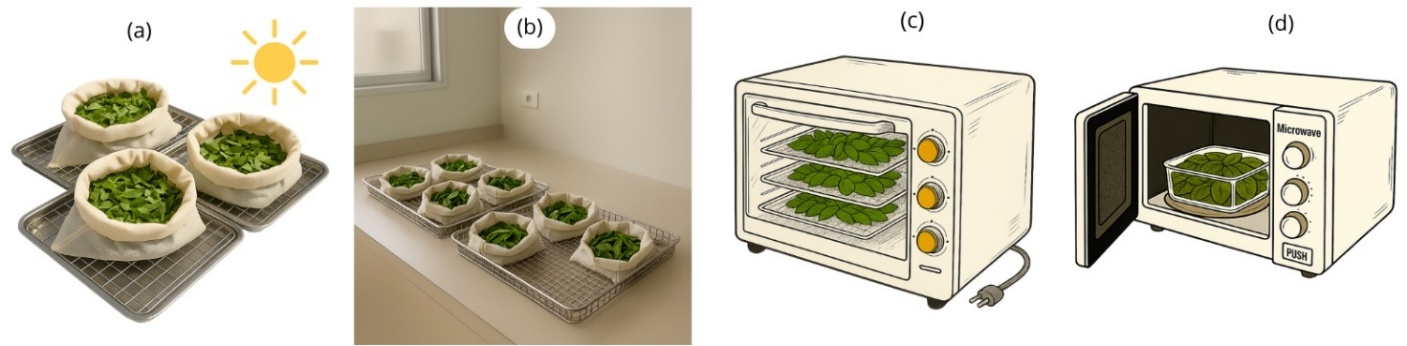
Figure 2 Drying methods utilized: (a) SD, (b) RTD, (c) CTD, and (d) MWD.
During the drying process, the samples were weighed at regular intervals using electronic scales (D2B, PT Arta Joil Tappa, China; accuracy (0.01 g) until a constant weight was achieved (3 consecutive weighing with a variation of <0.1%). Time stamps were synchronized for subsequent MR-time analysis.
The modelling of the drying kinetics
The moisture ratio (MR)
The drying characteristics of the samples were evaluated using the moisture ratio (MR; Eq. (1)):

with Xt = moisture at time t, X0 = initial moisture and Xe = equilibrium moisture [40].
Drying rate
The instantaneous rate was computed as Eq. (2):

Thin-layer drying models
This study involved the evaluation of seven models: Page, Henderson-Pabis, Logarithmic, Midilli, 2-term exponential, Diffusion approach, dan Jenna-Das [41-43]. The detailed equations are presented in Table 1 with constants (a, b, c, k, k₀, k₁, n) estimated using non-linear regression.
Table 1 Thin-layer drying model.
Model |
Equation |
Page |
|
Henderson and Pabis |
|
Logarithmic |
|
Midilli |
|
2-term exponential |
|
Diffusion approach |
|
Jenna-Das |
|
Effective moisture diffusivity (Deff)
The falling-rate period was modeled using Fick’s Second Law for flat geometry (Eq. (3)):
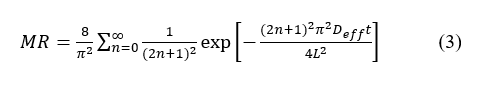
where L refers to the average thickness of leaves and t denotes time. In practice, the first-term approximation was applied where appropriate to obtain stable Deff estimates, while full-series fitting was checked for sensitivity.
Model adequacy and validation
The suitability of the model was evaluated by means of the coefficient of determination (R2), root mean square error (RMSE), sum of squares error (SSE), and total sum of squares (TSS), with adequacy criteria of R2 >0.95 and RMSE < 0.05 [44,45].
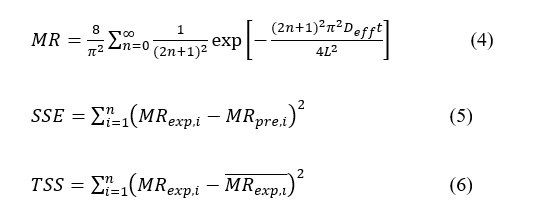
Residuals were screened for systematic deviation (runs/curvature). Models failing adequacy criteria were not advanced to comparative discussion.
Analysis of quality
Antioxidant activity (DPPH assay)
DPPH (2,2-diphenyl-1-picrylhydrazyl) is a stable radical used to assess radical-scavenging capacity. IC50 denotes the extract concentration (µg/mL) required to reduce DPPH absorbance by 50% - a smaller IC50 indicates stronger activity. With minor modifications to [46,47], leaf powder (1 g) was extracted in 20 mL 80% methanol using a digital sonicator (Elmasonic S30H, Germany) for 30 min, then filtered (Whatman No.1, 0.45 µm). An aliquot (2 mL extract) was mixed with 2 mL 0.1 mM DPPH, incubated 30 min in the dark, and absorbance was recorded at 517 nm (UV-Vis Shimadzu UV-1900, Japan). Inhibition (%) was computed by Eq. (7):
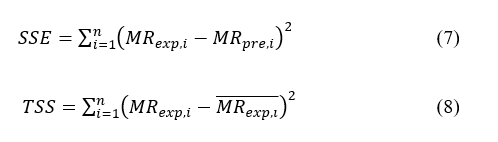
where A0 = control absorbance and A1 = sample absorbance. The value of IC50 was determined from the regression curves between the percentage of inhibition towards concentration.
FTIR spectroscopy
The spectrum was obtained using a PerkinElmer UATR Spectrum Two (US) in the range of 4,000 - 400 cm−1, with a resolution of 4 cm−1, scanned 32 times. The sample (in dry powder or fresh leaf pieces) was then placed directly on the ATR crystal. The main bands included O–H stretching, aliphatic C–H, ester/γ-lactone C=O, aromatic C=C, phenolic/ester C–O, and glycosidic C–O–C. Ratio indices R1 - R3 and the composite RQ were computed from integrated band areas, modified from the Tepe method [48]. A consistent baseline and pathlength normalization were employed across runs.
Color analysis
The color coordinates (L, a*, and b*) were measured using a HunterLab ColorFlex EZ Colorimeter (USA) in accordance with CIE standards [49,50]. Derived indices such as ΔE, C, and h° were calculated for further analysis.
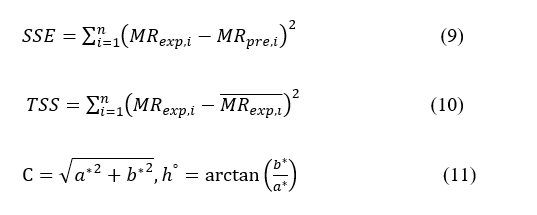
Statistical analysis and software
All experiments were performed in triplicate (n = 3) with randomized drying order to minimize bias, and the results were presented as the mean ± standard deviation. Nonlinear regression on the drying curve was performed using Polymath 6.10 (USA), while IC50 values and color parameters were analyzed by means of one-way ANOVA followed by Tukey’s test at a significance level of p < 0.05 (Minitab, Minitab LLC, USA). Treatment effects were also evaluated using Hedges’ g effect size and linear and non-parametric relationships were tested with OLS regression and Spearman’s correlation. To correlate rapid indicators with bioactivity, Partial Least Squares Regression (PLSR) (MATLAB R2024a, MathWorks, USA) was performed between FTIR indices (R1 - R3, RQ) and IC50. Leave-one-out cross-validation (LOOCV) was used to estimate predictive ability (reporting R²{LOOCV} and RMSE), and Variable Importance in Projection (VIP) was computed to rank spectral predictors [51,52]. Chemometric adequacy was judged by stable VIP ranking (VIP ≳ 1 considered influential) and the absence of pathological leverage.
Results and discussion
Drying process
Drying behavior and the factors of temperature/power
The MR-time curve (Figure 3) demonstrated that an augmentation in convective temperature (40 - 60 °C), microwave power (120 - 380 W), and sun-drying (SD) conditions significantly accelerated moisture release. RTD (29 °C) and CTD 40 °C were found at the slowest, requiring ≈ 30 - 33 h to reach MR = 0.10, while CTD 60 °C reached it in ≈ 4 h (≈ 64% faster than CTD 50 °C). SD appeared faster (~2.7 h) yet highly variable due to sunlight fluctuation. MWD 380 W has been observed as the most significant intensification, reducing drying time to ≈ 4 min (97% - 99% shorter than CTD 50 °C). The mean falling-rate slope (MR 0.60 - 0.20) substantiated this gradient: 0.025 MR·h−1 (CTD 40 °C) to 0.199 MR·h−1 (CTD 60 °C), 0.313 MR·h−1 (SD), and 0.036 - 0.226 MR·min−1 (MWD 120 - 380 W) (Figure 4; Table 2 summarizes statistics).
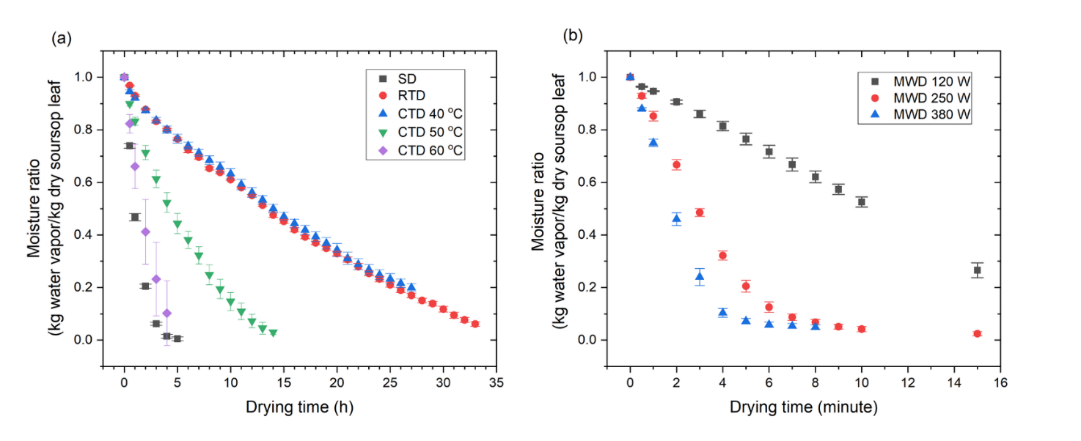
Figure 3 The moisture ratio during the drying process of soursop leaf.
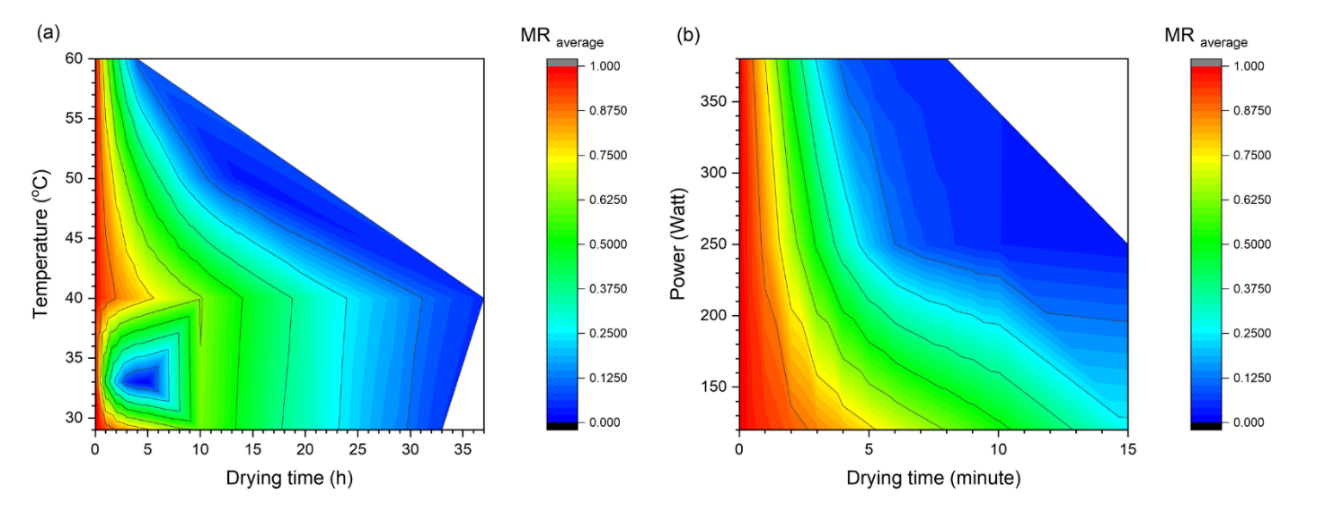
Figure 4 The plot of mean moisture ratio: (a) SD, RTD, CTD, and (b) MWD.
It is evident that all drying conditions follow a dominant falling-rate phase without a constant rate stage, which is typical of leafy matrices with robust internal diffusion resistance [42,53,54]. The enhancement observed at elevated CTD temperatures is consistent with findings reported for Moringa oleifera and holy basil [55,56]. The unreliability of SD is analogous to that observed in Plantago lanceolata and Alcea rosea [57,58]. In contrast, MWD generates volumetric heating (dipole rotation/ionic conduction), creating steep internal vapor-pressure gradients and microstructural disruption, as evidenced by celery and coriander [59,60]. It is noteworthy that an acceleration of up to ~225× (MWD 380 W vs. CTD 50 °C) is rarely reported for tropical medicinal leaves. However, it should be noted that each method has its limitations. The reliability of SD is compromised by the influence of UV/wind resulting in low reproducibility. MWD is susceptible to local overheating when the load is inadequate. RTD/CTD necessitate extended exposure and are vulnerable to phenolic degradation.
Thin-layer modelling
The multi-parameter nonlinear models (Midilli, Jenna-Das) demonstrated the highest level of accuracy (R2 ≥ 0.994; RMSE 0.006 - 0.026; Table 2; Figure 5). Jenna-Das demonstrated proficiency at RTD and at CTD 50 °C, while Midilli exhibited superiority at 40 °C; both exhibited comparable performance at 60 °C. Across MWD 120 - 380 W, Midilli was consistently superior (R2 > 0.995). Drying constants increased with temperature/power (0.028 - 0.387 CTD; 0.020 - 0.311 MWD), while n decreased modestly (1.68 - 1.36).
Table 2 Constants-coefficients for fitted model and its statistical parameter for drying of soursop leaves.
Model |
Statistical Parameter |
SD, 33 °C |
RTD, 29 °C |
Temperature |
Power MWD |
||||
CTD 40 °C |
CTD 50 °C |
CTD 60 °C |
120 W |
250 W |
380 W |
||||
Page |
k |
0.7252 |
0.0303 |
0.0284 |
0.1419 |
0.3866 |
0.0200 |
0.1568 |
0.3106 |
n |
1.2223 |
1.2318 |
1.2475 |
1.1174 |
1.1974 |
1.5543 |
1.4108 |
1.3491 |
|
R2 |
0.9081 |
0.9878 |
0.9922 |
0.9935 |
0.9933 |
0.9893 |
0.9985 |
0.9935 |
|
RMSE |
0.1009 |
0.0322 |
0.0265 |
0.0250 |
0.0295 |
0.0429 |
0.0133 |
0.0260 |
|
Henderson and Pabis |
k |
0.8076 |
0.0614 |
0.0613 |
0.1785 |
0.4762 |
0.0815 |
0.2907 |
0.4523 |
a |
1.0322 |
1.0333 |
1.0370 |
1.0099 |
1.0324 |
1.0708 |
1.0697 |
1.0619 |
|
R2 |
0.9052 |
0.9767 |
0.9799 |
0.9904 |
0.9847 |
0.9703 |
0.9875 |
0.9868 |
|
RMSE |
0.1024 |
0.0445 |
0.0424 |
0.0305 |
0.0446 |
0.0714 |
0.0387 |
0.0371 |
|
Logarithmic |
k |
0.5580 |
0.0288 |
0.0353 |
0.1239 |
0.3570 |
0.0690 |
0.2792 |
0.4666 |
a |
1.1136 |
1.4875 |
1.2934 |
1.1620 |
1.1429 |
1.1569 |
1.0861 |
1.0539 |
|
b |
−0.1262 |
−0.5140 |
−0.3100 |
−0.1833 |
−0.1347 |
−0.0980 |
−0.0195 |
0.0115 |
|
R2 |
0.9840 |
0.9991 |
0.9989 |
0.9991 |
0.9962 |
0.9860 |
0.9877 |
0.9874 |
|
RMSE |
0.0421 |
8.54 × 10⁻3 |
9.90 × 10⁻3 |
9.51 × 10⁻3 |
0.0221 |
0.0491 |
0.0385 |
0.0361 |
|
Midilli |
k |
0.7046 |
0.0511 |
0.0314 |
0.1577 |
0.3789 |
0.0135 |
0.1511 |
0.3103 |
n |
1.0385 |
0.8637 |
1.1290 |
0.9206 |
1.1018 |
1.6776 |
1.4358 |
1.3647 |
|
a |
1.0093 |
0.9940 |
0.9660 |
0.9928 |
0.9980 |
0.9612 |
0.9934 |
1.0017 |
|
b |
−0.0117 |
−8.83 × 10⁻3 |
−2.96 × 10⁻3 |
−0.0109 |
−9.92 × 10⁻3 |
−6.89 × 10⁻4 |
2.90 × 10⁻4 |
7.62 × 10⁻4 |
|
R2 |
0.9939 |
0.9995 |
0.9991 |
0.9993 |
0.9965 |
0.9979 |
0.9988 |
0.9952 |
|
RMSE |
0.0260 |
6.56 × 10⁻3 |
9.15 × 10⁻3 |
8.11 × 10⁻3 |
0.0212 |
0.0188 |
0.0120 |
0.0224 |
|
Two-term exponential |
k |
1.0999 |
0.0584 |
0.0814 |
0.2232 |
0.6227 |
0.1256 |
0.4201 |
0.6491 |
a |
1.7808 |
1.0000 |
1.7631 |
1.6088 |
1.7233 |
1.9691 |
1.9411 |
1.9176 |
|
R2 |
0.9080 |
0.9747 |
0.9919 |
0.9943 |
0.9929 |
0.9861 |
0.9986 |
0.9943 |
|
RMSE |
0.1009 |
0.0464 |
0.0269 |
0.0235 |
0.0305 |
0.0488 |
0.0131 |
0.0243 |
|
Diffusion approach |
k |
0.7672 |
0.0237 |
0.0357 |
0.1205 |
0.3589 |
0.0595 |
0.2459 |
0.4515 |
a |
−2.9799 |
−2.9800 |
2.1046 |
−2.6285 |
−4.9600 |
−6.3755 |
0.9999 |
1.0000 |
|
b |
0.8263 |
0.8317 |
0.4777 |
1.2740 |
0.9466 |
0.8726 |
0.9998 |
0.9996 |
|
R2 |
0.9080 |
0.9887 |
0.9986 |
0.9982 |
0.9862 |
0.9872 |
0.9257 |
0.9818 |
|
RMSE |
0.1009 |
0.0311 |
0.0112 |
0.0133 |
0.0424 |
0.0468 |
0.0945 |
0.0435 |
|
Jenna-Das |
k |
0.7672 |
0.0237 |
0.0357 |
0.1205 |
0.3589 |
0.0595 |
0.2459 |
0.4515 |
a |
0.8696 |
1.3176 |
1.3543 |
0.9498 |
1.3192 |
1.4092 |
1.2501 |
1.0795 |
|
b |
−0.1000 |
−0.0301 |
6.92 × 10⁻3 |
−0.0584 |
0.0536 |
0.0209 |
0.0293 |
6.30 × 10⁻3 |
|
c |
0.1567 |
−0.3158 |
−0.3780 |
0.0477 |
−0.3206 |
−0.3795 |
−0.2015 |
−0.0181 |
|
R2 |
0.9940 |
0.9996 |
0.9989 |
0.9995 |
0.9966 |
0.9896 |
0.9899 |
0.9878 |
|
RMSE |
0.0257 |
5.83 × 10⁻3 |
9.79 × 10⁻3 |
6.81 × 10⁻3 |
0.0211 |
0.0423 |
0.0348 |
0.0355 |
|
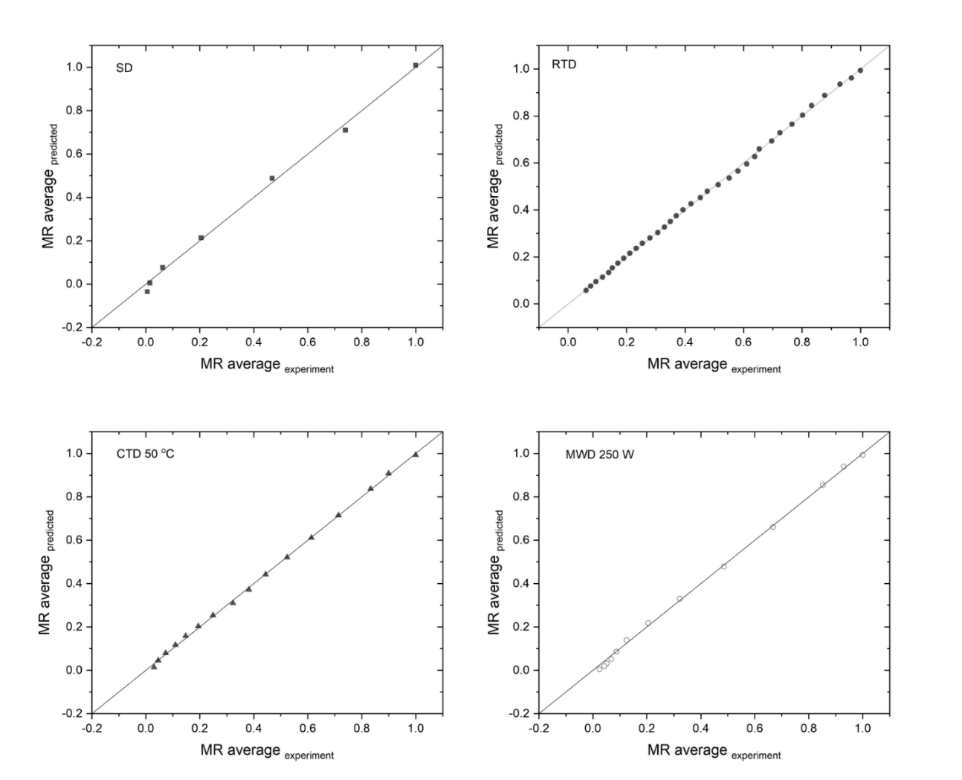
Figure 5 Comparison of MR experiment and Midilli modelling in the drying process of soursop leaf.
These findings emphasized the Midilli model as a universal predictive tool suitable for scale-up, while the Jenna-Das model remains more appropriate for convective subsets, consistent with the behavior observed in Cosmos caudatus and Vernonia amygdalina [61,62]. However, as empirical constructs, these models do not explicitly account for morphology or porosity evolution during drying process. Consequently, cross-batch validation is imperative, with particular consideration for variations in leaf thickness and ambient relative humidity, is required to ensure the adequacy of the input range and maintain predictive reliability.
Effective moisture diffusivity (Deff)
As demonstrated in Table 3, Deff exhibited a marked increase with both temperature and microwave power: From 1.92×10⁻15 m2/s (RTD) and 2.34×10⁻15 m2/s (CTD 40 °C) to 1.26×10⁻13 m2/s (CTD 60 °C); SD yielded 2.14×10⁻13 m2/s; MWD 120 - 380 W reached 2.07×10⁻13 to 3.40×10⁻12 m2/s (≈ 8.1 - 225×CTD 50 °C). The increase is indicative of lower viscosity, higher vapor pressure, and micro-cracks formation under higher thermal/microwave energy.
Table 3 Effective moisture diffusivity of soursop leaf.
Sample |
Deff, m2/s |
R2 |
Relative to CTD 50 °C (fold) |
SD |
2.142×10⁻13 |
0.989 |
14.19 |
RTD |
1.924×10⁻15 |
0.951 |
0.13 |
CTD 40 °C |
2.343×10⁻15 |
0.958 |
0.16 |
CTD 50 °C |
1.509×10⁻14 |
0.959 |
1.00 |
CTD 60 °C |
1.263×10⁻13 |
0.983 |
8.37 |
MWD 120 W |
2.067×10⁻13 |
0.940 |
13.70 |
MWD 250 W |
1.225×10⁻12 |
0.947 |
81.15 |
MWD 380 W |
3.400×10⁻12 |
0.942 |
225.13 |
At 380 W, the effective moisture diffusivity (Deff) exhibited a 2 to 3 orders of magnitude increase compared to the CTD, a finding consistent with observations reported for C. nardus and C. roseus [33,63]. Its strong correlation with the falling-rate slope and the time required to reach MR = 0.10 highlights Deff as a practical descriptor of drying efficiency. As demonstrated in relevant literature, elevated Deff values under MWD conditions have been shown to be associated with enhanced phenolic retention and superior color preservation [64,65]. However, the accuracy of Deff estimation is contingent upon the accuracy of geometry and thickness assumptions, and the reliability of the estimation is potentially compromised by the presence of uncontrolled external factors in the SD conditions.
Antioxidant activity (the assay of DPPH, IC50)
IC50 exhibited significant variation among treatments (Figure 6(a)), confirming a strong drying-method effect (ANOVA, F = 1,557.83; p < 0.001; η2 = 0.999; Table 4). Fresh leaves demonstrated the lowest antioxidant activity (≈ 546 µg/mL), whereas MWD 380 W exhibited the strongest potential (≈ 107 µg/mL; ~5-fold improvement). The CTD 60 °C (~183 µg/mL) demonstrated superior performance in comparison to CTD 40 - 50 °C (~210 - 236 µg/mL), while RTD exhibited the weakest performance (~292 µg/mL). Tukey analysis categorized treatments into distinct groups, and Hedges’ g revealed substantial contrasts, particularly between MWD 380 W and other methods (Table 5). A salient observation is that medium microwave power (120 - 250 W) yielded unanticipated IC50 values (≈ 310 - 333 µg/mL), indicating that mid-level energy prolongs exposure and expedite oxidative degradation, resulting in a U-shaped response. Conversely, high microwave intensity (i.e. ≥ 380 W) generates sufficient volumetric heating to reduce residence time and preserve bioactivity (Figure 6(a)). The IC₅₀-ln(Deff) relationship was weak and insignificant (R2 = 0.06; ρ = −0.14; Figure 6(b); Table 6), indicating that kinetic intensification alone is inadequate to predict antioxidant preservation.
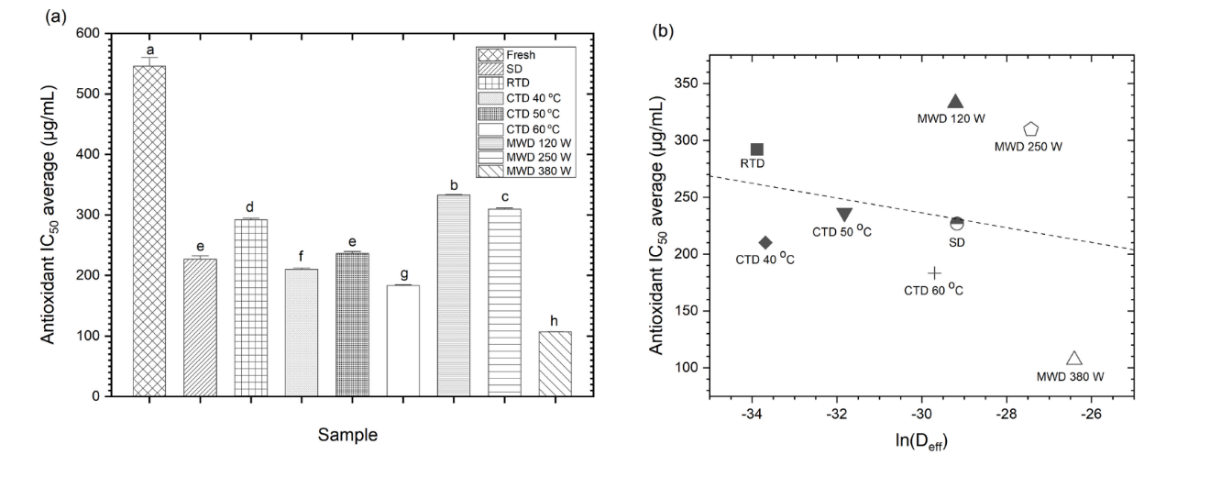
Figure 6 Soursop leaf: (a) anti-radical activity value, and (b) correlation of IC50-ln(Deff).
Table 4 ANOVA results regarding the effects of drying method on soursop leaf.
Source of variation |
SS |
df |
MS |
F |
p-value |
F-crit |
Between Groups |
368,170.4211 |
8 |
46,021.30263 |
1,557.8299 |
5.9164E-24 |
2.51015789 |
Within Groups |
531.7549217 |
18 |
29.54194009 |
|
|
|
Total |
368,702.176 |
26 |
|
|
|
|
Table 5 Level of Hedges’ effects g (CI 95%) for the contrast of main IC50 among treatments.
Contrast |
Hedges’ g |
95% CI |
Interpretation |
MWD 380 W vs Fresh |
−35.09 |
[−59.45, −10.74] |
Very large, significant |
MWD 380 W vs SD |
−23.28 |
[−39.46, −7.10] |
Large, significant |
MWD 380 W vs RTD |
−85.42 |
[−114.63, −26.21] |
Very large, significant |
MWD 380 W vs CTD 40 °C |
−55.51 |
[−94.00, −17.02] |
Very large, significant |
MWD 380 W vs CTD 50 °C |
−41.78 |
[−70.76, −12.80] |
Very large, significant |
MWD 380 W vs CTD 60 °C |
−48.58 |
[−82.27, −14.89] |
Very large, significant |
MWD 380 W vs MWD 120 W |
−198.36 |
[−335.83, −60.90] |
Extremely large, significant |
MWD 380 W vs MWD 250 W |
−97.35 |
[−164.82, −29.88] |
Extremely large, significant |
CTD 60 °C vs Fresh |
−28.78 |
[−48.77, −8.80] |
Very large, significant |
CTD 60 °C vs SD |
−8.12 |
[−13.89, −2.35] |
Moderate, significant |
CTD 60 °C vs RTD |
−41.51 |
[−70.31, −12.72] |
Very large, significant |
CTD 60 °C vs CTD 40 °C |
−11.29 |
[−19.21, −3.36] |
Large, significant |
CTD 60 °C vs CTD 50 °C |
−15.47 |
[−26.27, −4.67] |
Large, significant |
SD vs Fresh |
−23.60 |
[−40.00, −7.20] |
Large, significant |
SD vs CTD 50 °C |
−1.60 |
[−3.29, 0.10] |
Small, not significant |
Table 6 Correlation of IC50 to ln(Deff) using OLS regression and Spearman correlation.
Analysis |
Parameter |
Value |
Note |
OLS regression |
Intercept (β₀) |
41.33 |
± 322.47 (SE) |
|
Slope (β₁) |
−6.50 |
± 10.65 (SE) |
|
t(β₁) |
−0.610 |
df = 6 |
|
p-value |
0.564 |
Not significant |
|
R2 |
0.058 |
Weak |
|
R2adj |
−0.099 |
Negative (small n or poor fit) |
Spearman correlation |
ρ |
−0.142 |
Weak negative |
|
t |
−0.353 |
df = 6 |
|
p-value |
0.736 |
Not significant |
This nonlinear response has been documented in phenolic-rich matrices, where mid-level MWD induces oxidative stress, while higher microwave power enhances retention [66,67]. Similarly, moderate CTD temperatures (50 - 70 °C) often better preserve polyphenols compared to prolonged low-temperature drying or sun drying [5,68-70]. However, the DPPH assay only reflects overall antioxidant capacity and does not differentiate specific molecular contributors; thus, identifying individual compounds such as flavonoids or acetogenins requires further analysis using HPLC or LC-MS techniques.
FTIR analysis of phytochemical retention
FTIR spectra (Figure 7) displayed characteristic lignocellulosic-phenolic bands: O–H (3,280 - 3,300 cm⁻1), aliphatic C–H (2,920/2,850 cm⁻1), aromatic C=C (1,595 - 1,600 cm⁻1), C–O (1,050 - 1,030 cm⁻1), and glycosidic C–O–C (1,236 - 1,240 cm⁻1). In comparison with fresh leaves (O–H at 3,289 cm⁻1), dried samples exhibited a red shift (3,269 - 3,295 cm⁻1) and diminished O–H/C–O intensities, indicating weakened H-bonding and partial phenolic loss. A distinct C=O band (~1,734 cm⁻1) emerged exclusively after drying (SD–MWD 380 W), thereby indicating oxidation/esterification. At 380 W, the weakening at 1,236 - 1,240 cm⁻1 signified partial polysaccharide depolymerization.
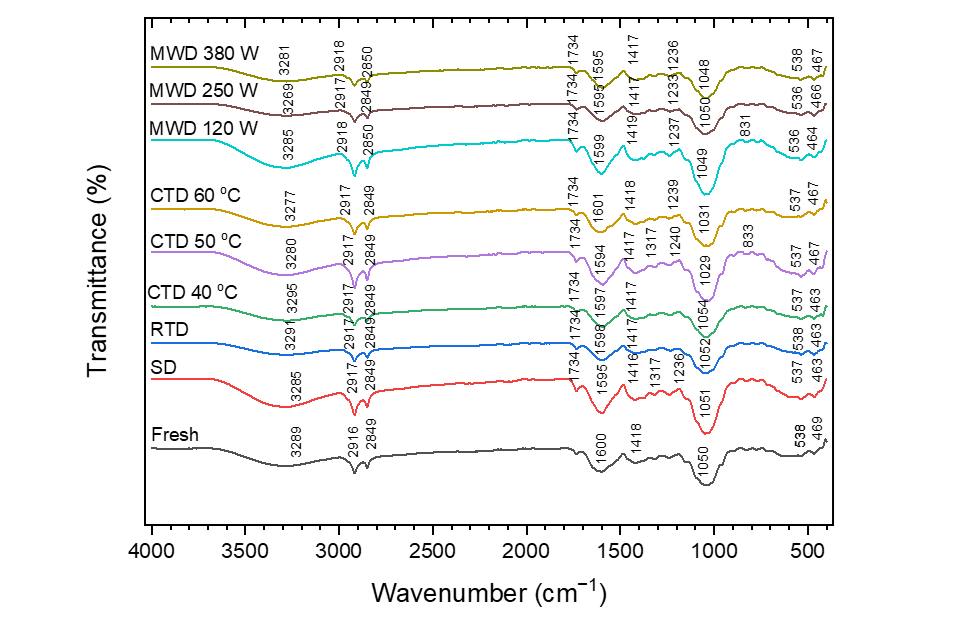
Figure 7 FTIR spectra of soursop leaf.
To capture these shifts quantitatively, ratios R1–R4 and a composite RQ were proposed (Figures 8(a) and 8(b)). RQ demonstrated the strongest consistency with IC50 and outperformed single-band indicators, thereby transforming FTIR analysis from a purely descriptive tool into an in-line quality control (QC) approach. Analogous correlations between FTIR indices and antioxidant capacity have been reported for Centella asiatica, Moringa oleifera, black carrot, dates, and apples [48,71-76]. Integration with chemometric modeling further reinforced this framework: PLS regression analysis between FTIR predictors and IC50 yielded R2{LOOCV} = 0.325 and RMSE = 95.9 µg/mL. VIP scores identified R3 and RQ as dominant predictors (Figures 8(c) - 8(d)), thereby confirming that the phenolic-carbonyl equilibrium plays a pivotal role in antioxidant retention [77-82]. However, FTIR provides only spectral proxies; LC-MS or HPLC remains necessary to identify specific phenolic subclasses, and PLSR is sensitive to preprocessing steps such as baseline correction and normalization, warranting multi-batch replication for validation.
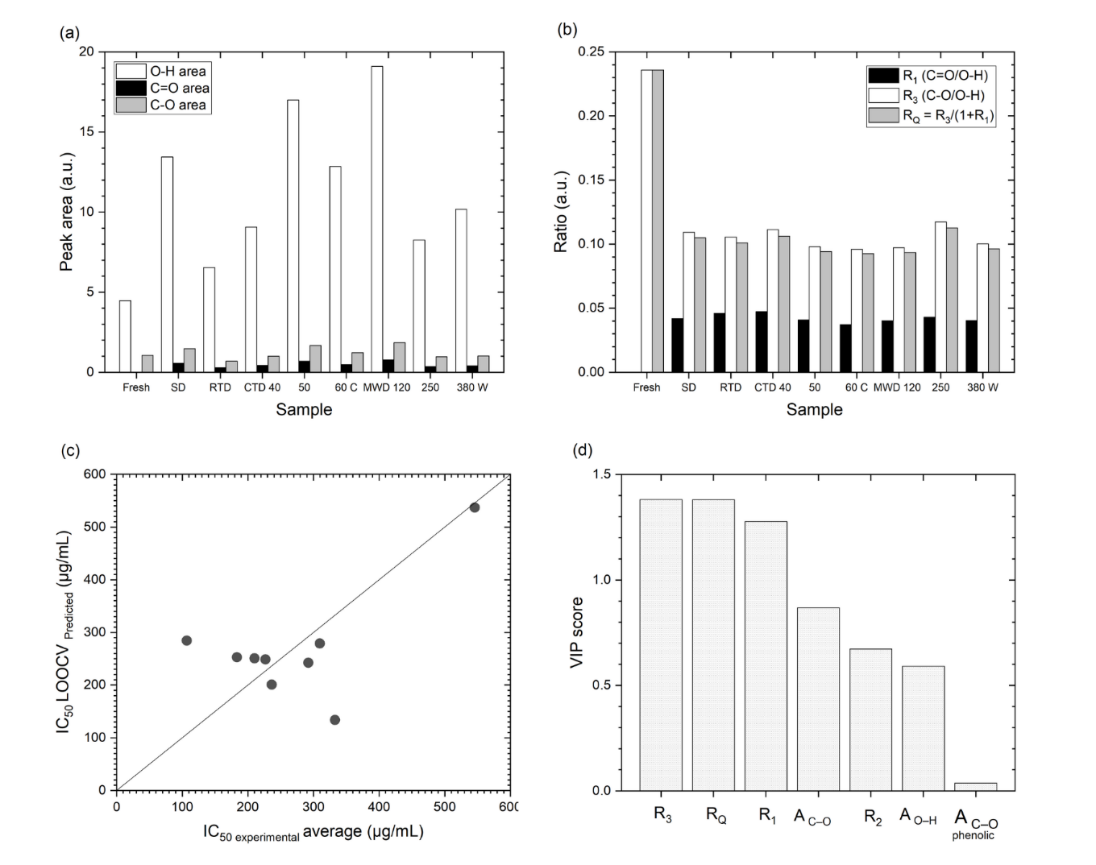
Figure 8 Quantitative FTIR analysis: (a) peak areas of O–H, C=O, and C–O; (b) ratio of R1 to RQ; (c) regression of PLS FTIR vs. antioxidant activity; and (d) FTIR ratio vs. VIP score.
Color attributes (L, a*, b*, ∆E, C and h°)
Fresh leaves exhibited low brightness and strong greenness (Table 7 for details; L = 24.05 ± 0.04; a* = −6.72 ± 0.04; b* = 7.01 ± 0.03). Drying resulted in significant alterations across coordinates: L increased (highest at CTD 60 °C and MWD 250 W), a* shifted towards 0 (smallest loss at CTD 40 °C), and b* increased (maximum at CTD 60 °C). ΔE was smallest for CTD 40 °C (≈ 2.71; below the ~3 perceptual threshold) and highest for RTD; MWD was generally high.
Table 7 Changes in the color attributes of soursop leaves using various drying methods.
Sample |
L |
a* |
b* |
∆E |
C (Chroma) |
h° (hue angle) |
Fresh |
24.05 ± 0.04d |
−6.72 ± 0.04h |
7.01 ± 0.03d |
- |
9.71 ± 0.05a |
−46.22 ± 0.06a |
SD |
25.84 ± 0.02c |
−1.58 ± 0.03c |
4.93 ± 0.02g |
5.83 ± 0.07f |
5.17 ± 0.04g |
−72.22 ± 0.18e |
RTD |
15.61 ± 0.02f |
−1.72 ± 0.04d |
5.34 ± 0.02e |
9.95 ± 0.04a |
5.61 ± 0.03f |
−72.13 ± 0.28e |
CTD 40 °C |
24.08 ± 0.59d |
−4.19 ± 0.02g |
7.86 ± 0.05b |
2.71 ± 0.04g |
8.91 ± 0.05c |
−61.91 ± 0.09b |
CTD 50 °C |
19.35 ± 0.01e |
−2.53 ± 0.06f |
4.94 ± 0.05g |
6.63 ± 0.05e |
5.56 ± 0.07f |
−62.87 ± 0.27c |
CTD 60 °C |
32.37 ± 0.03a |
−1.96 ± 0.02e |
8.87 ± 0.02a |
9.76 ± 0.03b |
9.08 ± 0.02b |
−77.52 ± 0.14f |
MWD 120 W |
29.82 ± 0.03b |
−0.24 ± 0.03a |
5.05 ± 0.04f |
8.89 ± 0.02c |
5.06 ± 0.04g |
−87.24 ± 0.34h |
MWD 250 W |
32.94 ± 0.05a |
−2.62 ± 0.04f |
7.50 ± 0.02c |
9.79 ± 0.09b |
7.94 ± 0.03d |
−70.71 ± 0.23d |
MWD 380 W |
30.39 ± 0.03b |
−1.12 ± 0.06b |
6.95 ± 0.04d |
8.45 ± 0.02d |
7.04 ± 0.03e |
−80.82 ± 0.53g |
Note: Fresh samples were excluded from ANOVA ΔE due to 0 variance. Values are expressed as mean ± SD with n = 3; p < 0,05, Tukey’s test.
The ΔE-IC50 plot demonstrated a weak-to-moderate correlation (R2 = 0.265; Figure 9), indicating that visual color changes alone cannot fully explain antioxidant variation [83-85]. In contrast, the a* parameter exhibited a robust correlation with the FTIR RQ ratio (R2 = 0.733), where the loss of greenness (a* approaching 0) corresponded to a decrease in C–O/O–H (RQ) and an increase in C=O (~1,734 cm⁻1), reflecting phenolic depletion and carbonyl accumulation. These findings are consistent with those reported for Strobilanthes crispus and Allium ursinum [5,7,86-88]. In practice, CTD 40 °C was found to be capable of maintaining visual appearance (ΔE ≈ 2.7) but did not optimize phenolic preservation. Conversely, CTD 60 °C and MWD 380 W produced higher ΔE values (a noticeable sensory shift [84,89,90]) yet achieved superior phenolic retention and IC50 performance. However, it should be noted that color is determined by illumination conditions and instrument calibration, and it is unable to differentiate chlorophyll from phenolic degradation pathways. Therefore, cross-device color-space standardization is recommended.
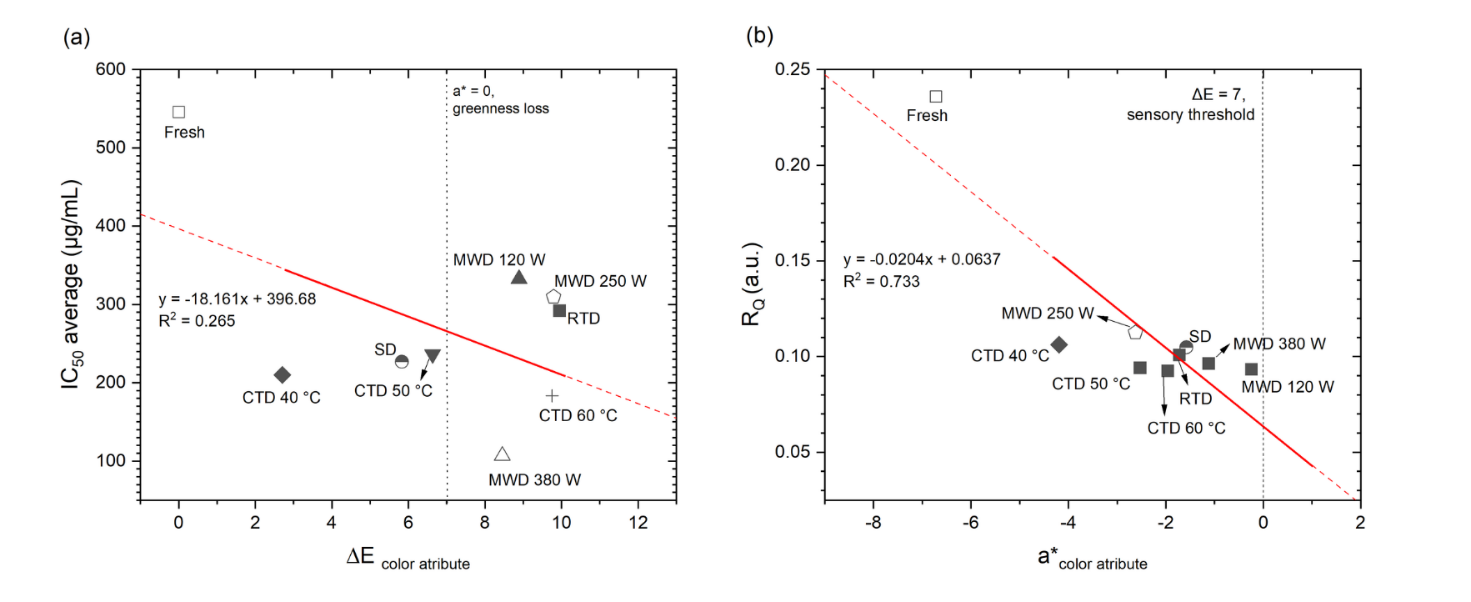
Figure 9 Correlation of color and function in soursop leaf.
Synthesis and practical implications
Overall, MWD 380 W represents an optimal process-product compromise: ≈ 4 min drying, low IC50, and FTIR signatures consistent with phenolic retention; CTD 60 °C is a viable convective compromise. Deff emerges as a kinetic surrogate, whereas bioactive quality should be predicted via FTIR ratios (RQ/R3) and, when required, a* as a rapid indicator. The integration of FTIR analysis with PLSR and VIP framework demonstrated potential for in-line quality control in tropical herbal processing.
Conclusions
This present study systematically integrated drying kinetics, spectroscopic indicators, and antioxidant response to establish a predictive framework for optimizing Annona muricata leaf drying. The findings can be summarized as follows: The first point to consider is that of kinetics and quality (scientific). The drying methods employed had a significant impact on both the kinetics and the quality of the product; the Midilli model yielded the optimal thin-layer fit (R2 > 0.95; RMSE < 0.05), while the Jenna-Das model demonstrated efficacy under specific convective conditions. The effective moisture diffusivity (Deff) exhibited an increase in accordance with both temperature and power, reaching a maximum at MWD 380 W, which resulted in an acceleration of up to ~225× in comparison to CTD 50 °C. A trade-off was observed, where CTD 40 °C maintained appearance (ΔE ≈ 2.7) but reduced phenolic retention, whereas CTD 60 °C and MWD 380 W yielded higher ΔE (> 8) yet superior FTIR ratios (RQ, R1 - R3) and IC50 values; mid-power MWD (120 - 250 W) was detrimental. (ii) Structure-function relationships (scientific): The ΔE-IC50 correlation was weak to moderate, but a* exhibited a strong correlation with RQ (R2 ≈ 0.73). Furthermore, PLSR between FTIR indices and IC50 yielded R2{LOOCV} ≈ 0.33 with VIP emphasizing R3 and RQ. This validates FTIR ratios as non-destructive predictors of antioxidant preservation. In the third instance, the practical implementation of predictive quality control was demonstrated. The combination of the parameters ΔE and a* in conjunction with RQ, functioned as expeditious indicators for real-time guidance. The integration of FTIR analysis with Partial Least Squares Regression (PLSR) and Variable Importance in Projection (VIP) transformed FTIR from a descriptive technique into an actionable inline or at-line quality control tool, thereby minimizing reliance on laborious assays and supporting green processing. The provision of actionable guidance on the practical implementation of drying processes is of paramount importance. MWD 380 W represented the optimal process-product compromise (≈ 4 min, low IC50, favorable FTIR ratios), while CTD 60 °C was a reliable convective alternative, SD was rapid but inconsistent, and mid-power MWD should be avoided. In practice, Deff can function as a kinetic gate, with RQ and a* providing rapid bioactivity verification. It is recommended that future research endeavours extend the validation process to pilot and industrial scales. Additionally, there is a need to explore hybrid drying configurations, and integrate advanced chemometrics for real-time control.
Acknowledgements
The authors acknowledged the financial support from Universitas Diponegoro, Indonesia, 2025.
Declaration of Generative AI in Scientific Writing
The authors declare we have not used Artificial Intelligence (AI) tools in the creation of this.
CRediT Author Statement
Dessy Agustina Sari: Methodology, Investigation, Data curation, Writing - original draft, Visualization, Writing - review & editing; Moh Djaeni: Conceptualization, Supervision, Validation, Writing-review & editing; Devi Yuni Susanti: Formal analysis; Joko Nugroho Wahyu Karyadi: Visualization; Olly Sanny Hutabarat: Software; Setia Budi Sasongko: Software, Visualization; Aji Prasetyaningrum: Formal analysis; Ching Lik Hii: Methodology, Writing - review & Editing.
[1] B Bhattacharjee, K Sandhanam, S Ghose, D Barman and RK Sahu. Market overview of herbal medicines for lifestyle diseases. In: AK Dhara and SC Mandal (Eds.). Role of herbal medicines: Management of lifestyle diseases. Springer Nature, Singapore, 2024, p. 597-614.
[2] H Kaur, S Singh, SG Kanagala, V Gupta, MA Patel and R Jain. Herbal medicine - A friend or a foe of cardiovascular disease. Cardiovascular and Hematological Agents in Medicinal Chemistry 2024; 22(2), 101-105.
[3] D Buziak, SM Garland, DS Nichols, I Hunt and DC Close. Astragalus membranaceus: Impact of postharvest drying processes on yield and components of bioactive compounds. Industrial Crops and Products 2024; 209, 117918.
[4] DH Ryu, JY Cho, M Hamayun, SH Lee, HH Cha, JH Jung and HY Kim. Optimizing drying time for Centella asiatica (L.) Urban: Metabolomic insights into dehydration effects on primary and secondary metabolites. Chemical and Biological Technologies in Agriculture 2025; 12(1), 26.
[5] J Lukinac and M Jukić. Influence of drying temperature on the organoleptic properties, antioxidant activity and polyphenol content in dried leaves of Allium ursinum L. subsp. ucrainicum. Ukrainian Food Journal 2022; 11(1), 2-26.
[6] HKF Oh, LF Siow and YY Lim. Approach to preserve phenolics in Thunbergia laurifolia leaves by different drying treatments. Journal of Food Biochemistry 2019; 43(7), e12856.
[7] INS Norasmadi, NN Zulkipli, S Lob, WZW Abdullah, MF Jusoh and A Mubarak. Effect of different drying methods on colour, total phenolic content, flavonoid content, and antioxidant activity retention of Strobilanthes crispus leaves. Pertanika Journal of Tropical Agricultural Science 2024; 47(4), 1157-1174.
[8] SB Rad, H Mumivand, S Mollaei and A Khadivi. Effect of drying methods on phenolic compounds and antioxidant activity of Capparis spinosa L. fruits. BMC Plant Biology 2025; 25(1), 133.
[9] K Mouhoubi, L Boulekbache-Makhlouf, K Madani, A Palatzidi, J Perez-Jimenez, I Mateos-Aparicio and A Garcia-Alonso. Phenolic compounds and antioxidant activity are differentially affected by drying processes in celery, coriander and parsley leaves. International Journal of Food Science & Technology 2022; 57(6), 3467-3476.
[10] A Stupar, Ž Kevrešan, A Bajić, J Tomić, T Radusin, V Travičić and J Mastilović. Enhanced preservation of bioactives in wild garlic (Allium ursinum L.) through advanced primary processing. Horticulturae 2024; 10(4), 316.
[11] S Salehinia, F Didaran, S Aliniaeifard, S Zohrabi, S MacPherson and M Lefsrud. Green light enhances the phytochemical preservation of lettuce during postharvest cold storage. PLoS One 2024; 19(11), e0311100.
[12] M Kaveh, F Sharifian, A Alirezalu, S Keramat, B Gheisary, N Farhadi, MD Bachejak, E Rahmati and NN Balestani. Effect of ethanol and ultrasonic pretreatments on drying kinetics, quality, and physicochemical attributes of dried Salvia officinalis. Biomass Conversion and Biorefinery 2025; 15, 30907-30923.
[13] GA Hernandez-Fuentes, OG Delgado-Enciso, EG Larios-Cedeño, JM Sánchez-Galindo, SG Ceballos-Magaña, K Pineda-Urbina, MA Alcalá-Pérez, NE Magaña-Vergara, J Delgado-Enciso, U Díaz-Llerenas, J Diaz-Martinez, I Garza-Veloz, ML Martinez-Fierro, IP Rodriguez-Sanchez and I Delgado-Enciso. Comparative analysis of infusions and ethanolic extracts of Annona muricata leaves from Colima, Mexico: Phytochemical profile and antioxidant activity. Life 2024; 14(12), 1702.
[14] S Oufquir, F Agouram, H Kabdy, MA Laaradia, K Oubella, B Abdelmoniim, A Rachida, S Garzoli and A Chait. Novel insights into the bioactive profile and therapeutic potentials of Indonesian Annona muricata leaves. Chemistry & Biodiversity 2025; 22(12), e01606.
[15] N Ismail, H Rajaratinam, KC Ghazali, SFM Salleh, WSFAW Ibrahim, MM Yahya and WZW Zain. The phytochemical components of Kelantan grown Annona muricata leaves and its anti-proliferative properties on MCF-7 breast cancer cells. Sains Malaysiana 2023; 52(10), 2785-2801.
[16] AG Peña, MR Alvarez, K Delica, PG Moreno, R Abogado, SJ Grijaldo, EL Salac, FM Deniega, M Basingan, CM Ravidas, F Heralde, GC Completo, I Padolina and R Nacario. Antioxidant and anticancer activities of Annona muricata L. and Antidesma bunius L. leaves, and molecular networking analysis using LC-MS/MS metabolomics. South African Journal of Botany 2022; 151, 559-566.
[17] FHD de Andrade, RS de Araújo Batista, TBL Melo, FHA Fernandes, RO Macedo, FS de Souza and AG Wanderley. Characterization and compatibility of dry extract from Annona muricata L. and pharmaceutical excipients. Journal of Thermal Analysis and Calorimetry 2021; 143(1), 237-246.
[18] JS Mahesh, K Ramalakshmi and R Balakrishnaraja. Investigation of mass transfer parameters, phytochemical analysis of dried ripen graviola fruit (Annona muricata) and its comparative approach by using statistical tools: ANN and mathematical modelling. Chemical Papers 2023; 77(11), 7085-7098.
[19] J Shafana, S Kandasamy, M Shaji and K Kavivarshini. A study on the effect of microwave drying of herbs for determination of antioxidant activity. AIP Conference Proceedings 2021; 2387, 020004.
[20] R ElGamal, OA Hamed, AM Rayan, C Liu, S Kishk, S Al-Rejaie and G ElMasry. Effect of convective and vacuum drying on some physicochemical and phytochemical characteristics of peppermint leaves. AIMS Agriculture and Food 2025; 10(1), 17-39.
[21] S Caliskan, H Oldenhof, R Brogna, B Rashidfarokhi, H Sieme and WF Wolkers. Spectroscopic assessment of oxidative damage in biomolecules and tissues. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy 2021; 246, 119003.
[22] A Światły-Błaszkiewicz, J Gębalski, T Kaczmarek, D Załuski and B Kupcewicz. Exploring ED-XRF and ATR-FTIR spectroscopic techniques in medicinal plant research: A pilot study with Scutellaria baicalensis roots. Journal of Pharmaceutical and Biomedical Analysis 2025; 265, 117008.
[23] F Martinez-Morales, AJ Alonso-Castro, JR Zapata-Morales, C Carranza-Álvarez and OH Aragon-Martinez. Use of standardized units for a correct interpretation of IC50 values obtained from the inhibition of the DPPH radical by natural antioxidants. Chemical Papers 2020; 74(10), 3325-3334.
[24] W Hou, Z Duan and Y Yi. Effect of microwave drying on the phenolic content and color changes in different regions of persimmon slices. Modern Food Science and Technology 2024; 40(10), 259-269.
[25] JY Yap, CL Hii, SP Ong, KH Lim, F Abas and KY Pin. Degradation kinetics of carpaine and antioxidant properties of dried Carica papaya leaves as affected by drying methods. International Journal of Food Engineering 2022; 18(8-9), 593-602.
[26] S Kayacan-Cakmakoglu, I Atik, P Akman, I Doymaz, O Sagdıc and S Karasu. Effect of the different infrared levels on some properties of sage leaves. Chemical Industry and Chemical Engineering Quarterly 2023; 29(3), 235-242.
[27] M Pasławska, K Sala, A Nawirska-Olszańska, B Stępień and E Pląskowska. Effect of different drying techniques on dehydration kinetics, physical properties, and chemical composition of lemon thyme. Natural Product Communications 2020; 15(2), 1934578X2090452.
[28] J Deng, Z Chen, H Jiang and Q Chen. High-precision detection of dibutyl hydroxytoluene in edible oil via convolutional autoencoder compressed Fourier-transform near-infrared spectroscopy. Food Control 2025; 167, 110808.
[29] AP Kusumadewi, R Martien, S Pramono, AA Setyawan, A Windarsih and A Rohman. Application of FTIR spectroscopy and chemometrics for correlation of antioxidant activities, phenolics and flavonoid contents of Indonesian Curcuma xanthorrhiza. International Journal of Food Properties 2022; 25(1), 2364-2372.
[30] NAZ Jinin, MAZ Benjamin and MA Awang. Drying kinetics and quality assessment of noodles from Piper sarmentosum Roxb. (Kaduk) leaves. Malaysian Journal of Fundamental and Applied Sciences 2024; 20(4), 835-851.
[31] KÇ Selvi. Investigating the influence of infrared drying method on Linden (Tilia platyphyllos Scop.) leaves: Kinetics, color, projected area, modeling, total phenolic, and flavonoid content. Plants 2020; 9(7), 916.
[32] S Verma, AB Usenov, DU Sobirova, SA Sultonova and JE Safarov. Mathematical description of the drying process of mulberry leaves. IOP Conference Series: Earth and Environmental Science 2022; 1112(1), 012012.
[33] RK Rout, A Kumar and PS Rao. A comparative assessment of drying kinetics, energy consumption, mathematical modeling, and multivariate analysis of Indian borage (Plectranthus amboinicus) leaves. Journal of Food Process Engineering 2024; 47(5), e14630.
[34] S Tan, Y Wang, W Fu, Y Luo, S Cheng and W Li. Drying kinetics and physicochemical properties of kumquat under hot air and air-impingement jet dryings. Food Science and Biotechnology 2022; 31(6), 711-719.
[35] Q Liu, X Jiang, F Wang, S Fan, B Zhu, L Yan, Y Chen, Y Wei and W Chen. Evaluation and process monitoring of jujube hot air drying using hyperspectral imaging technology and deep learning for quality parameters. Food Chemistry 2025; 467, 141999.
[36] H Zhang, WZ Wang, XY Hu, J Wang, YY Han, XM Wang, XM Zhang, XY Guo, XY Huan, J Zhao, N Li, YF Wang and ZS Wu. Research on dynamic on-line monitoring method of moisture attribute in 3 honey-processed Chinese herbal slice based on in-situ general model. Yaoxue Xuebao 2023; 58(10), 2890-2899.
[37] T Belwal, C Cravotto, MA Prieto, PR Venskutonis, M Daglia, HP Devkota, A Baldi, SM Ezzat, L Gómez-Gómez, MM Salama, L Campone, L Rastrelli, J Echave, SM Jafari and G Cravotto. Effects of different drying techniques on the quality and bioactive compounds of plant-based products: A critical review on current trends. Drying Technology 2022; 40(8), 1539-1561.
[38] AS Eapen, YK Bhosale and S Roy. A review on novel techniques used for drying medicinal plants and its applications. International Journal of Biomaterials 2025; 2025(1), 4533070.
[39] B Kumar, SD Patle and SK Dewangan. Solar drying of ginger (Zingiber officinale) and turmeric (Curcuma longa): A comprehensive review. Solar Energy 2025; 299, 113748.
[40] S Chokngamvong and C Suvanjumrat. Study of drying kinetics and activation energy for drying a pineapple piece in the crossflow dehydrator. Case Studies in Thermal Engineering 2023; 49, 103351.
[41] CL Recio-Colmenares, RB Recio-Colmenares, RF Conchas-Cedano, I Pilatowsky-Figueroa and CAG García. Data-driven hybrid CNN-LSTM neural networks for predicting drying kinetics of green tomatoes (Physalis ixocarpa) integrating mathematical models. IEEE Access 2025; 13, 57413-57425.
[42] MI Maamar, M Badraoui, M Mazouzi and L Mouakkir. Mathematical modeling on vacuum drying of olive pomace. Trends in Sciences 2022; 20(2), 3822.
[43] FD Utari, DA Sari, L Kurniasari, AC Kumoro, M Djaeni and CL Hii. The enhancement of sappanwood extract drying with foaming agent under different temperature. AIMS Agriculture and Food 2023; 8(1), 214-235.
[44] O Dajbych, A Kabutey, Č Mizera and D Herák. Investigation of the effects of infrared and hot air oven drying methods on drying behaviour and colour parameters of red delicious apple slices. Processes 2023; 11(10), 3027.
[45] CA Osagiede and TM Stephen. Kinetic modelling of the drying behaviour of palm kernel (Dura species). Journal of Science and Technology Research 2024; 6(2), 237-249.
[46] S Baliyan, R Mukherjee, A Priyadarshini, A Vibhuti, A Gupta, RP Pandey and CM Chang. Determination of antioxidants by DPPH radical scavenging activity and quantitative phytochemical analysis of Ficus religiosa. Molecules 2022; 27(4), 1326.
[47] İ Gulcin and SH Alwasel. DPPH radical scavenging assay. Processes 2023; 11(8), 2248.
[48] TK Tepe. Convective drying of golden delicious apple enhancement: Drying characteristics, artificial neural network modeling, chemical and ATR-FTIR analysis of quality parameters. Biomass Conversion and Biorefinery 2024; 14(12), 13513-13531.
[49] K Altay, SN Dirim and AA Hayaloglu. Effects of different drying processes on the quality changes in Arapgir purple basil (Ocimum basilicum L.) leaves and drying‐induced changes in bioactive and volatile compounds and essential oils. Journal of Food Science 2024; 89(12), 9088-9107.
[50] G Kumar, J Joshi, PS Rao and P Manchikanti. Effect of thin layer drying conditions on the retention of bioactive components in Malabar spinach (Basella alba) leaves. Food Chemistry Advances 2023; 3, 100419.
[51] J Magnanensi, M Maumy-Bertrand, N Meyer and F Bertrand. New developments in sparse PLS regression. Frontiers in Applied Mathematics and Statistics 2021; 7, 693126.
[52] T Mehmood, S Sæbø and KH Liland. Comparison of variable selection methods in partial least squares regression. Journal of Chemometrics 2020; 34(6), e3226.
[53] NN Mbegbu, CO Nwajinka and DO Amaefule. Thin layer drying models and characteristics of scent leaves (Ocimum gratissimum) and lemon basil leaves (Ocimum africanum). Heliyon 2021; 7(1), e05945.
[54] EE Wisdom, T Karikarisei and AH Cyrus. The dehydration kinetics of curry leaves (Murraya koenigii rataceae). Indian Journal of Engineering 2023; 20(53), e1ije1001.
[55] CM de Alcântara, IDS Moreira, MT Cavalcanti, RP Lima, HV Moura, RDS Neves, CAL Cassimiro, JJA Martins, FRDC Batista and EM Pereira. Mathematical modeling of drying kinetics and technological and chemical properties of Pereskia sp. leaf powders. Processes 2024; 12(10), 2077.
[56] OM Ferreira, VS Mendoza, THB Ferreira, RA Jordan and EJS Argandoña. Drying of Xanthosoma sagittifolium Schott leaves by heat pump and conventional dryer: And evaluation of bioactive compounds. Journal of the Science of Food and Agriculture 2025; 105(8), 4379-4387.
[57] F Ergün. Effects of drying methods on amounts of phenolic and flavonoid compounds and antioxidant capacity of Plantago lanceolata L. The Journal of Animal and Plant Sciences 2023; 33(1), 159-165.
[58] S Wahdaningsih, S Rizkifani, EK Untari and W Rinaldi. Effect of drying method on levels of antioxidant activity, total flavonoid levels, and total phenol levels in ethanol extract of Bawang Dayak (Eleutherine americana) leaves. Majalah Obat Tradisional 2023; 28(1), 37-39.
[59] O Bualuang, DI Onwude and S Prangsri-Aroon. The effect of various drying strategies on the greenness, chlorophyll, bioactive compounds, antioxidant activity, and anti-tyrosinase of dried Acanthus ilicifolius L. leaves. International Food Research Journal 2022; 29(2), 416-432.
[60] K Mouhoubi, L Boulekbache-Makhlouf, W Mehaba, H Himed-Idir and K Madani. Convective and microwave drying of coriander leaves: Kinetics characteristics and modeling, phenolic contents, antioxidant activity, and principal component analysis. Journal of Food Process Engineering 2022; 45(1), e13932.
[61] A Hasizah, M Djalal, AA Mochtar and S Salengke. Fluidized bed drying characteristics of moringa leaves and the effects of drying on macronutrients. Food Science and Technology 2022; 42, e103721.
[62] T Phull, P Phian and M Gupta. Kinetic simulation of convection drying for its effect on nutritional, functional, and phytochemical profiling of Colocasia antiquorum and Nasturtium officinale leaves of the Himalayan region. Journal of Food Process Engineering 2024; 47(2), e14548.
[63] A Kasara, OA Babar, A Tarafdar, T Senthilkumar, R Sirohi and VK Arora. Thin-layer drying of sadabahar (Catharanthus roseus) leaves using different drying techniques and fate of bioactive compounds. Journal of Food Processing and Preservation 2021; 45(2), e15140.
[64] A Ali, BL Chua, YH Chow and LH Tee. Quality and energy efficiency evaluation of Rosmarinus officinalis L. by intermittent and continuous microwave drying: Polyphenol composition, bioactive compounds quantification, antioxidant properties, physical characteristics, and energy consumption. Journal of Food Process Engineering 2023; 46(12), e14453.
[65] JG Yazar and E Demiray. Effects of conductive drying on drying kinetics and retention of bioactive compounds in mistletoe leaves. Chemical Engineering and Processing - Process Intensification 2023; 192, 109490.
[66] S Hazra, N Nahar, SK Saha and R Chakraborty. Analysis of the influence of different drying processes on the quality attributes of orange peel. Biocatalysis and Agricultural Biotechnology 2024; 62, 103429.
[67] M Ozdemir and S Karagoz. Effects of microwave drying on physicochemical characteristics, microstructure, and antioxidant properties of propolis extract. Journal of the Science of Food and Agriculture 2024; 104(4), 2189-2197.
[68] AM Castro, LE Díaz, MX Quintanilla-Carvajal, EY Mayorga and FL Moreno. Convective drying of feijoa (Acca sellowiana Berg): A study on bioactivity, quality, and drying parameters. LWT 2023; 186, 115209.
[69] A Ghorbani, G Eghlima, M Farzaneh and A Rezghiyan. Effect of drying methods on mucilage, anthocyanin content, and antioxidant activity of black hollyhock (Alcea rosea var. nigra). BMC Plant Biology 2025; 25(1), 478.
[70] D Stephen, KJ Antony, PM Munusamy and T Deivanayagame. Impact of drying methods on the quality of bioactive components in tree tomato (Cyphomandra betacae). Trends in Sciences 2022; 19(2), 2060.
[71] SJ Fan, XY Zhang, Y Cheng, YX Qiu, YY Hu, T Yu, WZ Qian, DJ Zhang and S Gao. Extraction optimization of phenolic compounds from Triadica sebifera leaves: Identification, characterization and antioxidant activity. Molecules 2024; 29(14), 3266.
[72] Y Noui, A Lekbir, H Moussa, M Haffas, A Bousselma, S Hameurlaine, H Tahraoui, J Zhang, F Boufahja, H Bendif, A Amrane and W Elfalleh. Effects of microwave drying on physicochemical characteristics, microstructure and antioxidant activities of date fruit powders. Journal of Food Measurement and Characterization 2025; 19(8), 5554-5571.
[73] AG Nomi, H Handayani, RH Khuluk, AH Karomah, L Wulansari, ND Yuliana, E Rohaeti and M Rafi. Antioxidant activity and metabolite changes in Centella asiatica with different drying methods using FTIR- and quantitative HPLC-based metabolomics. International Food Research Journal 2024; 31(1), 228-238.
[74] P Thakur, R Saini, P Suthar, A Dhiman and S Kumar. Comparative evaluation of tray and microwave drying of black carrot (Daucus carota L.): Effects on physicochemical, phytochemical and techno-functional properties. Journal of the Indian Chemical Society 2024; 101(10), 101327.
[75] JB Johnson, KB Walsh, M Naiker and K Ameer. The use of infrared spectroscopy for the quantification of bioactive compounds in food: A review. Molecules 2023; 28(7), 3215.
[76] AADL Santos, GF Leal, MR Marques, LCC Reis, JRDJ Junqueira, LL Macedo and JLG Corrêa. Emerging drying technologies and their impact on bioactive compounds: A systematic and bibliometric review. Applied Sciences 2025; 15(12), 6653.
[77] DC Fatmarahmi, RA Susidarti, RT Swasono and A Rohman. A development method of FTIR spectroscopy coupled with chemometrics for detection of synthetic drug adulterants of herbal products in quaternary mixture. Journal of Applied Pharmaceutical Science 2022; 12(3), 191-201.
[78] ZM Hamza, SA Kadhim and HH Hussain. Study of spectra physical for some samples of medical herbal by FTIR-ATR spectroscopy. Journal of Pharmaceutical Negative Results 2022; 13(1), 49-55.
[79] M Rafi, DA Tohib, A Saputra, Z Aziz and AH Karomah. FTIR-fingerprinting spectra combined with chemometrics analysis for distinguishing Strobilanthes phyllostachya leaves extracts and correlation with their antioxidant activity. HAYATI Journal of Biosciences 2025; 32(6), 1584-1591.
[80] R Ruslin, Y Yamin, I Irnawati, A Windarsih and A Rohman. FTIR spectra-based fingerprinting and chemometrics for rapid investigation of antioxidant activities of medicinal plants. Journal of Research in Pharmacy 2025; 29(4), 1522-1531.
[81] AA Bunaciu, S Fleschin and HY Aboul-Enein. FTIR spectroscopy used for study the thermal degradation of lard. Egyptian Pharmaceutical Journal 2021; 20(2), 166-172.
[82] T Chumroenphat, A Dechakhamphu, S Saensouk and S Siriamornpun. Drying conditions affected chemical profiles and health promoting properties of Castanopsis piriformis Hickel & A.Camus. Food Chemistry: X 2025; 29, 102874.
[83] TCB Rigolon, FARD Barros, ÉNR Vieira and PC Stringheta. Prediction of total phenolics, anthocyanins and antioxidant capacity of blackberry (Rubus sp.), blueberry (Vaccinium sp.) and jaboticaba (Plinia cauliflora (Mart.) Kausel) skin using colorimetric parameters. Food Science and Technology 2020; 40(S2), 620-625.
[84] W Li, Y Zhang, H Deng, H Yuan, X Fan, H Yang and S Tan. In vitro and in vivo bioaccessibility, antioxidant activity, and color of red radish anthocyanins as influenced by different drying methods. Food Chemistry: X 2023; 18, 100633.
[85] ZHT Cakmak, SK Cakmakoglu, E Avcı, O Sagdic and S Karasu. Ultrasound-assisted vacuum drying as alternative drying method to increase drying rate and bioactive compounds retention of raspberry. Journal of Food Processing and Preservation 2021; 45(12), e16044.
[86] J Zhang, J Zhang, L Zhang, Y Xue and K Zhang. Mechanistic insights into vegetable color stability: Discoloration pathways and emerging protective strategies. Foods 2025; 14(13), 2222.
[87] Y Huang, X Wang, Y Lyu, Y Li, R He and H Chen. Metabolomics analysis reveals the non-enzymatic browning mechanism of green peppers (Piper nigrum L.) during the hot-air drying process. Food Chemistry 2025; 464, 141654.
[88] W Luo, J Guo, J Zhou, M Yang, and Y Wang. Mechanistic elucidation and establishment of drying kinetic models of differential metabolite regulation in Rheum palmatum during natural sun drying: An integrated physiology, untargeted metabolomics, and enzymology study. Biology 2025; 14(8), 963.
[89] L Zhang, C Zhang, Z Wei, W Huang, Z Yan, Z Luo, T Beta and X Xu. Effects of four drying methods on the quality, antioxidant activity and anthocyanin components of blueberry pomace. Food Production, Processing and Nutrition 2023; 5(1), 35.
[90] S Baibuch, P Zema, E Bonifazi, G Cabrera, ADCM Portocarrero, C Campos and L Malec. Effect of the drying method and optimization of extraction on antioxidant activity and phenolic of rose petals. Antioxidants 2023; 12(3), 681.