
Trends
Sci.
2026;
23(6):
12293
Investigation of Aluminum in Symplocaceae: Total Content, Histological Structure, and Enzyme Activities from Different Leaf Ages
Dewi
Puspita Sari1,3,
![]() ,
Bambang Retnoaji2,
,
Bambang Retnoaji2,
![]() ,
Nastiti Wijayanti2,*,
,
Nastiti Wijayanti2,*,
![]() and Purnomo2,
and Purnomo2,
![]()
1Doctoral Program, Faculty of Biology, Universitas Gadjah Mada, Yogyakarta 55281, Indonesia
2Faculty of Biology, Universitas Gadjah Mada, Yogyakarta 55281, Indonesia
3Biology Education, Faculty of Teacher Training and Education, Universitas Sebelas Maret,
Surakarta 57126, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 16 October 2025, Revised: 17 November 2025, Accepted: 1 December 2025, Published: 1 February 2026
Abstract
Since Symplocaceae is known to be a hyperaccumulator of Aluminum (Al), studies on its accumulation capacity and physiological responses have not been widely investigated. The objectives of this research are to investigate the Al content of different leaf ages of Symplocos fasciculata and Symplocos cochinchinensis using Inductively Coupled Plasma Optical Emission Spectrometry (ICP OES). Moreover, the Al localization within tissues was determined using hematoxylin staining, followed by Scanning Electron Microscopy coupled with Energy-Dispersive X-ray spectroscopy (SEM-EDX) mapping to address the ultrastructural form and determine elemental distribution in the cross-section of the leaf. Additionally, confirmation of the mechanism of Al toxicity in relation to stress-induced oxidative enzymes, including Catalase (CAT), Superoxide Dismutase (SOD), and Peroxidase (POD), as well as the lipid peroxidation product Malondialdehyde (MDA) and Hydrogen peroxide (H2O2), serves as stress markers. ICP OES assay reveals a significant influence of leaf age on Al content. S. fasciculata old leaves had the highest Al concentration (39,001 ± 343.48 mg kg−1), while S. cochinchinensis young leaves had the lowest (16,788 ± 187.98 mg kg−1). The histological and SEM-EDX mapping evidence shows Al accumulation in S. fasciculata and S. cochinchinensis increases significantly with leaf age. Al3+ accumulation is found in cell walls, epidermal tissue, and the palisade layer, with the highest Al distribution in the old leaf lamina. The leaf ages of both Symplocos species exhibit a significant influence on CAT, POD, MDA, and H2O2, whereas SOD activity shows no significant variation. A positive correlation is observed between SOD, POD, MDA, and H2O2 in S. fasciculata (r > 0.7), similar to S. cochinchinensis, except for MDA (r = −0.6). This research presents a valuable investigation into the Al accumulation strategies and physiological responses of 2 Symplocos species.
Keywords: Symplocos, ICP OES, Leaves histology, SEM-EDX mapping, Oxidative stress
Introduction
Aluminum (Al) is relatively stable in neutral pH soil, existing in the form of aluminosilicates and aluminum oxides. Both forms are insoluble, ensuring they do not pose a significant threat to plants and organisms [1-4]. In acidic environments, aluminosilicates and other Al compounds are dissolved into the soil solution as Al ions (Al3+), which are harmful to plants [4]. Several plant species have evolved strategies for Al avoidance or tolerance. Excluder plants limit Al uptake by secreting organic acids that bind with metal ions, forming a barrier layer to prevent entry into plant tissue [3,4]. On the other hand, Al-resistant and accumulator plants form a chelate of organic acids with Al3+, converting them into a harmless form that inhibits their spread and stores them in the vacuole [3,5-7].
Symplocos is a remarkable hyperaccumulator plant known for storing large amounts of Al in its stems and leaves [5]. Based on previous research by Zheng et al. [7], Symplocos sumuntia leaf contains high concentrations of organic acids and multiple metal ions, particularly Al3+, whose content can reach 23,071.98 ± 11.19 mg kg⁻¹, confirming it as an Al hyperaccumulator plant. Metal and metalloid content were determined by inductively coupled plasma optical emission spectrometry (ICP OES) [8-10]. ICP OES allows precise quantification of metal ions in solutions before and after bioaccumulation, providing complementary data to SEM-EDX analysis [11]. Furthermore, the detection of Al in tissues and organs may vary depending on the method employed. Histochemical techniques typically identify specific compounds through their binding to chemical elements within biological tissues [12]. Al accumulation in leaves, as visualized using hematoxylin staining, results in a purplish hue in several tissues [12,13]. Although the hematoxylin staining method is well-established for detecting aluminum accumulation in plant tissues, it does have limitations, particularly in the scoring range [14]. Scanning electron microscopy and energy-dispersive X-ray spectroscopy (SEM-EDX) can provide more comprehensive insights into the structure and distribution of metal compounds stored within the tissues of hyperaccumulator plants [15]. This method benefits practical applications by determining the elemental composition, making it a vital analytical tool for the morphological and chemical characterization of various materials [16].
Al accumulation in plants at certain levels does not interfere with plant physiological processes; several studies have shown the benefits of low Al levels [3]. In maize, low levels of Al support leaf growth, whereas in tea, Al maximizes macronutrient absorption, increases root elongation, and accelerates metabolic rates, thereby increasing plant biomass levels [17,18]. Nonetheless, high Al levels exert antagonistic effects, notably inhibiting root growth and water absorption, and impairing the uptake of several essential elements, including potassium (K), calcium (Ca), and magnesium (Mg). Additionally, excess lipid peroxidase increases oxidative stress, leading to cell death [18-20]. Moreover, plants under Al stress upregulate their oxidative stress metabolism to counter Al toxicity [12]. Plants maintain their metabolism through enzymatic and non-enzymatic antioxidant defenses to mitigate the imbalance of Reactive oxygen species (ROS) mediated oxidative stress, considering the sophisticated strategies plants deploy to counteract a spectrum of combined abiotic stresses [21,22]. Some plant species, however, exhibit enhancement of their intrinsic antioxidant defenses to counterbalance stress induced by ROS accumulation and consequent oxidative damage [23-26]. Balancing ROS in cellular delivery through 2 pathways, including enzymatic antioxidants such as catalase (CAT), superoxide dismutase (SOD), and peroxidase (POD), is essential for mitigating reactive oxygen species (ROS) in cells [27]. Moreover, non-enzymatic antioxidants, including vitamins, flavonoids, tocopherols, carotenoids, and phenolic compounds, play crucial roles in maintaining redox balance and protecting against oxidative stress [11,27,28]. The accumulation strategies and physiological responses of Al in Symplocos leaves remains limited. Therefore, understanding the mechanisms of Al tolerance is deemed essential and requires a multidisciplinary approach. To compare the plant responses to Al in 2 Symplocos species across various leaf ages, this study employed a novel method that combines elemental analysis, histology, and enzymology.
Materials and methods
Study sites and species
Fieldwork was conducted from August to November 2024 at the Baturraden Botanical Garden, Mount Slamet, Central Java, Indonesia. Symplocos exhibits a shrubby to tree habit, reaching heights up to 20 m and is characterized by their hairy twigs. The young leaves of S. cochinchinensis are hairy and arranged in an alternate spiral pattern with leaf sheaths. In contrast, S. fasciculata also exhibits an alternate arrangement but in odd numbers. Both species possess elliptical or oval leaves with pointed apices, opposite venation, serrate margins, and a paracytic stomata. The primary difference lies in the leaf bases; S. fasciculata has rounded leaf bases, while S. cochinchinensis has narrowed bases. The general characteristics of the areas where each plant species was sampled, including S. fasciculata, located at 7°18’19” S - 109°13’57” E at 1,076 m, lower altitude 7°18’50” S - 109°13’58” E at 1,010 m, is the sampling area for S. cochinchinensis. The annual precipitation level record at the site was 5.198.96 mm, with 90% humidity levels for both species. However, these sites still exhibited different light intensities due to the presence of various organisms. S. fasciculata site area had a temperature of 23.7 ℃ with an average light intensity of 2.931 Cd, posses pH soil at 6.41, while S. cochinchinensis had a warmer temperature at 24.1 ℃ with an average light intensity of 5.658 C, posses pH soil at 6.19.
Collection of leaf material
Leaf samples were classified into 3 developmental stages - young, mature, and old - based on position and color [6,29]. Leaves were classified based on their position on a single twig, which indicated their increasing age from apex to base. Young leaves, which are still at an early developmental stage, exhibited a shoot position (the first leaves of apex) with a reddish to light green color. In contrast to the middle (4th leaves from apex to 5th leaves before basal) branches, dark green color showed mature leaves, and the older leaves were generally at the bottom (4 last leaves of base) with a yellowish green to yellow color [30]. The primary difference lies in the leaf bases; S. fasciculata has rounded leaf bases, while S. cochinchinensis has narrowed ones. Moreover, the average leaf size of S. cochinchinensis is larger than that of S. fasciculata, providing a clear visual distinction between the 2 species (Figure 1).

Figure 1 Description of leaf age categories in 2 Symplocos species: (A) Symplocos cochinchinensis; (B) Symplocos fasciculata.
Total Al content by ICP OES assay
The leaves were washed with a 3% hydrochloric acid solution [8], then rinsed sequentially with tap water and deionized water to reduce the potential risk of contamination. The leaves were placed in paper bags and oven-dried (WTC Binder 7200, Tutlingen, Germany) at 60 °C for 72 h. The dried leaves were ground into a fine powder and filtered through 74 µm [31] for subsequent chemical analysis. Powder samples of 1.0 g were transferred into a vessel, HNO3 was added, and the mixture was allowed to react for 15 min. The mixture was subsequently subjected to microwave digestion. The resulting digested solution was transferred into a 50 mL measuring flask, and 100 mg of yttrium (Y) was added as an internal standard. Distilled water was added to the flask, and the solution was thoroughly homogenized. The solution was filtered through a syringe filter 0.20 µm and measured using an ICP OES (Agilent 5800, Agilent Technologies, USA). The absorbance used for Al detection was 396.152 nm, and for yttrium (Y), 371.029 nm.
Histological preparation by the embedding technique
Fresh leaf samples were cut into 0.2×0.5 cm2 pieces and fixed with FAA (5% from formaldehyde 37%, 5% glacial acetic acid, 70% ethanol) for 24 h. Samples were dehydrated through a graded ethanol series and cleared in an ethanol–xylene mixture. Infiltrated for 24 h at 57 °C (Memmert oven Incubator, Western Germany) and embedded in paraplast (Merck, CAS No: 64742-51-4, Darmstadt, Germany). Sectioning using a rotary microtome (KD 1508A, Zhejiang Jinhua Kedee Instrumental Equipment Co., Ltd, China) at 8 µm. Remove the paraplast with xylene, then rehydrate it to decrease the ethanol. Furthermore, a solution of 2.0 g of hematoxylin (Merck, C.I. 75290, Darmstadt, Germany) and 0.2 g of KIO3 [12] was applied at room temperature to detect Al decomposition in tissue. Samples were mounted using Entellan (Bio Optica, Biomount HM). Samples were examined using an optical microscope (Nikon Eclipse 50i with Nikon camera DS-Fi1, Tokyo, Japan). Color quality from optical microscope capture was accessed by the colorimeter software (Colorimeter vers. 2.25.19, Lab Tools). Color grading was evaluated using the L*a*b* values and the L* value represents light intensity, with a range of 0 to 100, indicating that a higher L* value corresponds to a softer color [32].
Distribution pattern of element by SEM EDX
The fresh leaves were cut fragments measuring 0.5 mm thick×0.5 cm lenght and immersed in FAA (Formaldehyde-Alcohol-Acetic Acid) fixative for 24 h, then dehydrated through graded ethanol, mounted on carbon tape, and loaded into a JEOL JEC-3000FC Auto Fine Coater (Japan), and coated with gold for 120 s at 20 mA under a pressure of approximately 3.2 Pa. After coating, the samples were loaded into a JEOL JSM-6510LA SEM (Japan) and vacuumed for 60 s. The samples were then imaged using an electron beam optimized for the specific element being examined.
Enzymatic assay
Fresh leaves, having been sorted and cleaned, were weighed to a mass of 0.5 g and then crushed by adding 0.1 g of liquid nitrogen. The sample was diluted with 500 µL of 50 mM PBS (pH 7.0). Centrifugation was performed at 10,000 rpm for 15 min at 4 °C to obtain a supernatant. SOD activity was assessed by monitoring the enzyme's ability to inhibit pyrogallol auto-oxidation, a reaction mediated by superoxide free radicals, at 325 nm [33]. CAT activity was evaluated spectrophotometrically by measuring H₂O₂ decomposition at 240 nm every 15 s for 1 - 2 min [34]. POD activity was measured by monitoring guaiacol oxidation at 420 nm [35], with readings taken every 30 s for 2 - 3 min. Enzyme activities were measured using a UV-Vis Spectrophotometer (Genesys UV10, Thermo Scientific, USA). The Thiobarbituric Acid Reactive Substances (TBARS) method was used to quantify MDA levels. This method relies on the reaction between MDA and thiobarbituric acid (TBA), which formed a pink complex measurable with a Microplate Spectrophotometer (Multiskan SkyHigh, Thermo Fisher Scientific, USA) at 532 nm [36].
Statistical analysis
All experiments were conducted in triplicate (n = 3), and the data were presented as the mean ± standard deviation (SD). Statistical analyses were performed using GraphPad Prism version 8.0.2 (GraphPad Software Inc., San Diego, CA, USA). Two-way analysis of variance (ANOVA) followed by Tukey’s post hoc test was employed to determine statistical significance between species Symplocos and leaf ages. Differences were statistically considered significant at p < 0.05. The correlation was examined using Pearson’s correlation analysis to investigate the relationship between Al content, L* value, and each enzymatic activity. The value of strong positive correlation was r ≥ 0.7.
Results and discussion
Total Al content
The total Al content of S. fasciculata and S. cochinchinensis was analyzed using ICP OES, revealing a significant influence of leaf age on Al content. A significant difference (p = 0.002) in Al content was observed between the oldest and youngest leaves in both species. The old leaves of the 2 Symplocos species have significantly accumulated more Al than the young leaves. Additionally, the Al element was reported to be deposited and immobilized in older tissues. A non-significant difference was demonstrated in young and mature leaves (p = 0.06) vs mature and old leaves (p = 0.09) in S. fasciculata (Figure 2). This condition is assumed to cause the immobility of aluminum once it is bound within the cell walls [2]. There was a lack of a biological mechanism for its efficient remobilization.
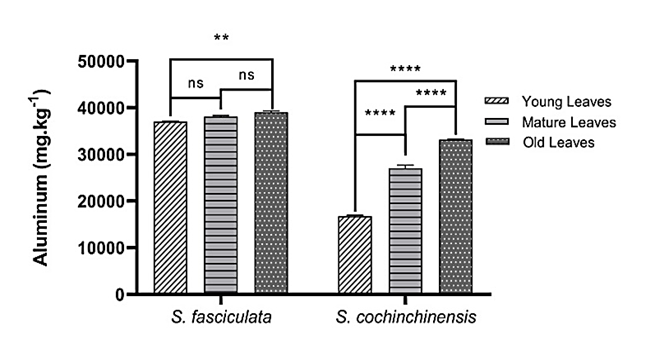
Figure 2 Aluminum (Al) content in 3 different leaf ages from 2 species of Symplocos was measured by ICP OES. Data are presented as mean ± SD, p < 0.05, ** p = 0.02, ****p < 0.001.
The typical Al concentration in the tissue of generally non-accumulating plant species is less than 100 mg kg−1 of dry weight [6]. Research findings revealed that the highest Al content was observed in the old leaves of S. fasciculata, with 39,001 ± 343.48 mg kg−1, followed by the old leaves of S. cochinchinensis, with 33,150 ± 147.84 mg kg−1. Furthermore, the mature leaves of S. fasciculata contained 38,045 ± 283.39 mg kg−1, whereas those of S. cochinchinensis contained 27,013 ± 660.45 mg kg−1. Additionally, the young leaves of S. fasciculata contained 37,002 ± 160.66 mg kg−1, and the lowest content among all samples was found in the young leaves of S. cochinchinensis 16,788 ± 187.98 mg kg−1.
Leaf age is a key parameter for detecting Al accumulation in tissue [6,12]. This finding aligns with the existing data that the old leaves of Symplocos are the highest accumulators of Al. Schmitt et al. [6] highlight that the highest Al concentrations were found in old leaves (24,180 ± 7,236 mg kg−1 dry weight, mean ± SD). In contrast, young leaves had significantly lower Al levels (20,708 ± 7,025 mg kg−1) in 3 different age groups of S. odoratissima, S. ophirensis, and S. ambangensis at 3 montane rainforest sites in Central Sulawesi [6]. Furthermore, our results for the old leaves demonstrated a similar trend to those from S. cochinchinensis and S. fasciculata in East Nusa Tenggara, Indonesia, which have Al contents of 39,311 and 49,775 ppm, respectively [37]. These results provide information on Al content in Symplocos leaves in the Indonesian montane rainforest, particularly on Java Island, as well as on the Sulawesi and East Nusa Tenggara Island.
Leaves histology assay
Histological assay revealed Al was detected in the cuticle, epidermis cell walls, palisade mesophyll, and chloroplasts (Figures 3(A) - 3(C)). Lamina of old leaves S. fasciculata (Figure 3(C)) showed dark purple (L* = 33.9 ± 0.854) at epidermal adaxial tissue compared to other leaf ages. This result led to the assumption that most of the Al was stored in old leaves. On the other hand, epidermal adaxial tissue of the young lamina of S. cochinchinensis appears light brown (L* = 70.1 ± 0.265) due to low Al content (Figure 3(D)). The results were in line with evidence that old leaves are Al storage in hyperaccumulator species [5,28,38,39]. The increasing color that develops from young to older leaves at both species (p < 0.0001) as they age suggests that Al3+ binds with pectin or hemicellulose, which constitute major components of the cell wall [12,15]. Functional cell walls act as a barrier to prevent Al3+ from penetrating the cell compartment and triggering overproduction of ROS within the cell [13,19].
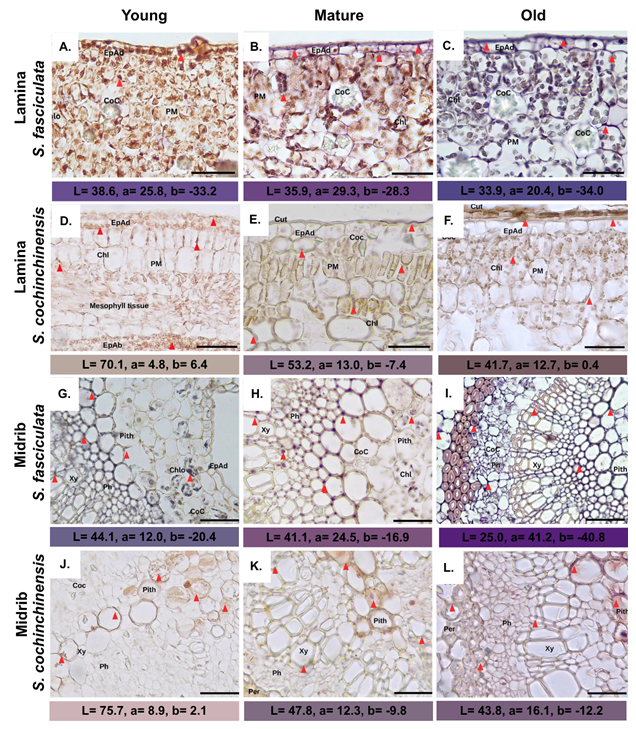
Figure 3 Symplocos leaf transverse sections with hematoxylin staining. Abbreviations: Chlo: Chloroplast; COC: Calcium Oxalate Crystal; Coll: Collenchyma; EpAd: Adaxial Epidermis; PM: Palisade Mesophyll; Ph: Phloem; Pith: Pith; Xy: Xylem; Red triangle: Al accumulation. Mag: 10×, scale bar: 200 µm.
The polar nature of the dye and metal cations will form a positively charged hematoxylin - Al (III) bond, allowing interactions with negatively charged components, such as basophilic components, carboxylate groups, and nucleic acids, to produce a brownish to dark purple color [40]. Furthermore, the Al accumulation in Symplocos lamina resembles that of Al complexes observed on the blade surface of Camelia major [13]. The accumulation of Al in hyperaccumulator plants is more abundant in the epidermal cells of old leaves, supporting the observation that leaf Al content increases with age (p <0.0001), where the primary distribution is in cell walls, especially in pectin or hemicellulose [12,15]. This condition causes the cell walls to act as a barrier, immobilizing Al from penetrating the cell compartment [36,37]. In addition to being in the cell walls, the epidermis and mesophyll parts of the leaves, particularly within the palisade mesophyll tissue, have staining that indicates Al accumulation [3,12,15]. However, sponge cells of both species did not exhibit any dye uptake except for limited staining observed in certain cell walls (Figures 3(A) -3(F)). These results contradict those of de Andrande et al. [13], who reported purple coloration in the cell walls of sponge parenchyma and absent in palisade mesophyll and chloroplasts. In contrast, palisade parenchyma tissue and chloroplasts were intensely stained purple by hematoxylin staining, as observed in our recent study of both leaves throughout their lifespan [41], as observed in our recent study of both leaves throughout their lifespan (Figures 3(A) - 3(F)), consistent with the results of of Zheng et al. [7]; de Andrande et al. [13]. The results showed that chloroplasts were capable of absorbing purple coloration, but no damage was observed. Differences in chloroplast size and shape were not observed, indicating that Al did not damage the chloroplasts [13]. Nevertheless, the role of Al in chloroplast metabolism warrants further study.
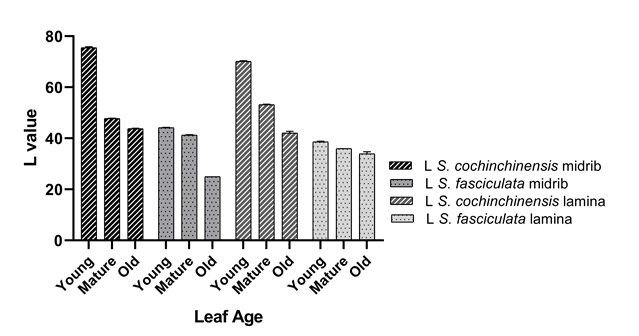
Figure 4 The comparison of L* value ranges from 0 to 100 indicates the light intensity of color as a histological measurement between S. fasciculata and S. cochinchinensis on the lamina and midrib, performed using a colorimeter. p-value < 0.0001.
In the midrib of both older leaves, hematoxylin was clearly visible in the phloem, xylem walls, and cortical parenchyma (pith), presenting a pale brown to purplish hue with red triangles (Figures 3(G) - 3(L)). The pericycle S. fasciculata (Figures 3(G) - 3(I)) presents different shades from light purple (L* = 44.1 ± 0.158) to darkened purple (L*= 25.0 ± 0.112). Moreover, the red triangle indicates the space between cells, which is mainly present outside the vascular bundle and pith. Accordingly, this is also observed in S. cochinchinensis; however, the L* value is lighter (Figure 4), ranging from nude brown (L* = 75.7 ± 0.141) to light purple (L* = 43.8 ± 0.190), indicating a specific reaction with Al [32]. These results suggest that the distribution of Al throughout the plant occurs through symplastic transport via the vascular tissue, from the root to the shoot, by forming ligand-Al complexes [42,43]. This finding is also in line with the research from de Andrade et al. [16], which reported the presence of Al complexes in the vascular bundles, particularly in the xylem region [13]. However, in contrast to the absence of staining in the vascular system.
An inverse correlation between aluminum (Al) content and lightness intensity, as represented by the L* value, has been observed across different leaf ages in S. fasciculata lamina (r = −0.998) and midrib (r = −0.919). Similarly, a phenomenon is also present in the L* value of S. cochinchinensis lamina (r = −0.996) and midrib (r = −0.965). These findings indicate that increasing aluminum content with leaf age is associated with a reduction in the L* value, resulting in a darker color intensity in histological preparations.
SEM EDX mapping assay
The distribution pattern of aluminum accumulation is represented by blue coloration that scatters in the midrib and lamina regions (Figures 5(A) and 5(B)). Notably, the abundant distribution occurs in the epidermis, penetrating the upper and lower epidermal layers, with partial presence in the mesophyll (Figures 5(A) and 5(B)). These findings are consistent with previous research on C. sinensis leaves, where aluminum was predominantly identified in the upper epidermis, with lower concentrations in the lower epidermis, as assessed through proton beam analysis [15]. Conversely, earlier study reported aluminum accumulation in the epidermis cell walls, with minimal detection in the mesophyll cell walls, using X-ray fluorescence [44]. Furthermore, elemental mapping also demonstrates the presence of calcium, represented by green coloration, showing localization in distinct clusters throughout the midrib to the lamina of older S. fasciculata leaves (Figure 5(A)). In comparison, calcium distribution in older S. cochinchinensis leaves is considerably less (Figure 5(B)). Additionally, Al is abundant in old leaves, indicating that both species utilize a range of mechanisms, including various minerals, to combat metal stress.
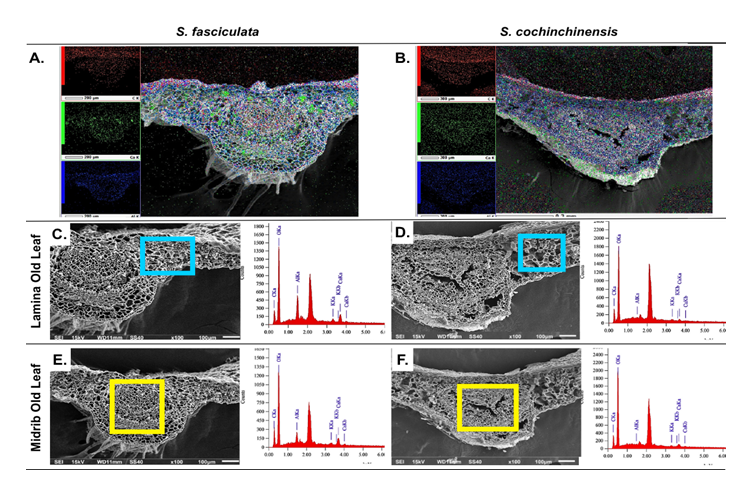
Figure 5 Scanning electron microscope (SEM) mapping element presenting distribution of Al, Ca, and O in old leaves of (A) S. fasciculata and (B) S. cochinchinensis. Energy-dispersive X-ray spectroscopy (EDX) elements through transverse sections in: (C,E) S. fasciculata (D,F) S.cochinchinensis. Mag: 150×, scale bar: 200 µm (A); Mag: 100×, scale bar: 300 µm (B); Mag: 100×, scale bar: 100 µm (C-F).
Table 1 EDX mass value (%) of the trace elements from various leaves in midrib (mid) and lamina (lam) of S. fasciculata and S. cochinchinensis.
Element |
S. fasciculata |
S. cochinchinensis |
||||||||||
Young |
Mature |
Old |
Young |
Mature |
Old |
|||||||
Mid |
Lam |
Mid |
Lam |
Mid |
Lam |
Mid |
Lam |
Mid |
Lam |
Mid |
Lam |
|
C |
40.90 |
40.65 |
18.97 |
13.80 |
17.08 |
12.87 |
50.46 |
48.31 |
15.16 |
12.01 |
10.55 |
11.83 |
O |
45.80 |
37.45 |
72.93 |
70.06 |
72.94 |
71.45 |
48.33 |
49.25 |
76.08 |
80.68 |
80.12 |
79.50 |
Al |
1.24 |
2.45 |
2.56 |
6.99 |
3.47 |
7.96 |
0.04 |
0.38 |
0.55 |
0.99 |
0.67 |
1.01 |
K |
0.02 |
0.02 |
0.74 |
1.57 |
1.06 |
1.75 |
0.08 |
0.06 |
2.03 |
1.61 |
2.34 |
2.06 |
Ca |
12.04 |
19.43 |
4.80 |
7.58 |
5.45 |
5.97 |
1.09 |
2.00 |
6.18 |
4.71 |
6.32 |
|
The Al element exhibited a higher tendency for accumulation in S. fasciculata, particularly in the lamina of the old leaves (7.96%) (Table 1). Meanwhile, in S. cochinchinensis, Al was also most abundant in the lamina of the old leaves (1.01%). Higher Al reflects differences in metal accumulation capacity or physiological adaptations between the 2 species to their environment [15]. The research indicates that Al uptake by Al-accumulating plants is not constrained by the saturation status of other mineral elements, such as Ca, K, and Mg in the soil [45,46]. However, in some cases, Al causes an imbalance in ion homeostasis by increasing the efflux of potassium and an abundant amount of calcium in the cytoplasm [45-47]. Potassium (K), a crucial element in osmotic regulation and metabolic processes [10], exhibited its highest concentration in the midrib of old S. cochinchinensis leaves (2.34%).
In contrast to the finding in S. fasciculata, the highest potassium content was observed in the lamina of old leaves. The influx of Ca2+ into the cytosol helps activate antioxidant enzymes, such as SOD, CAT, and APX [48]. In leaves of Symplocos, calcium was predominantly found in the lamina of young leaves of S. fasciculata (19.43%), differing from S. cochinchinensis, which has the highest calcium observed in the midrib of mature leaves. Aluminum and calcium ions experience competition in binding to the cell wall surface; Ca2+ interact with the cell surface, and Donnan's free space in the roots will inhibit the movement of Al3+ to the cytoplasm [46]. In addition, Ca2+ ions play a pivotal role in increasing the glycolysis process, resulting in an increase in pyruvate and acetyl-CoA, both of which contribute to reducing Al toxicity in cells [47].
Enzymatic assay
Symplocos is commonly known as a hyperaccumulator plant; however, high levels of aluminum (Al) within the cells may not consistently yield positive effects, particularly from a physiological perspective [21]. The presence of aluminum in cells acts as a catalyst that triggers the production of reactive oxygen species (ROS), notably hydrogen peroxide (H2O2), superoxide (O2−), and hydroxyl ions (OH−), resulting in the significant amounts of radicals [49,50]. An imbalance in the production of reactive oxygen species (ROS) can lead to oxidative stress, which subsequently causes lipid peroxidation [17-19].
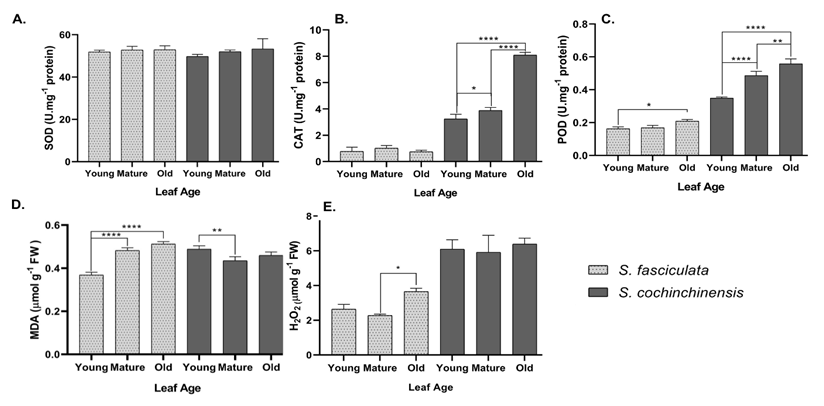
Figure 6 Enzyme activity profiles in S. cochinchinensis and S. fasciculata leaves at different leaf ages. (A) SOD enzyme activity, (B) CAT enzyme activity, (C) POD enzyme activity, (D) MDA concentration, (E) H202 concentration. Abbreviations: Light gray: S. fasciculata, Dark gray: S. cochinchinensis, ****: p < 0.001, ***: p < 0.01, **: p < 0.03, *: p < 0.05.
Superoxide dismutase (SOD) serves as the primary cellular defense mechanism in stressful oxidative conditions [50]. The results established an increase in SOD activity across leaf ages in both species (Figure 6(A)). In contrast to the results of previous studies at the same aluminum accumulator plant, where SOD activity is dominant in young leaves [12], both Symplocos species exhibited the highest activity in older leaves [12]. The abundant mass of Al in older leaves has been suggested to influence SOD activity, leading to a significant increase as a response to the mass production of ROS (p < 0.05). On the other hand, CAT activity indicated fluctuating activity between the 2 species (Figure 6(B)). Both exhibited distinct responses to catalyzed H2O2; however, significant differences in CAT activity were detected across leaf ages in S. cochinchinensis (p < 0.0001). As the presence of Al3+ triggers ROS production, younger leaves experience oxidative stress and develop mechanisms to prevent cellular damage [12]. However, although stress levels increase with leaf age, the associated stress-response mechanisms, indicated by elevated catalase activity in mature leaves, appear to remain stable in older leaves of S. fasciculata (p > 0.05).
Moreover, in high-stress conditions, POD plays a crucial role as a scavenger agent for ROS molecules. Both species demonstrated a significant increase (p < 0.0001) in POD activity from young leaves to older leaves (Figure 6(C)). S. fasciculata exhibited lower POD activity than S. cochinchinensis, with activity differing significantly from mature to older leaves (p = 0.0398). A subtle defense mechanism in this species may be associated with the activity of other enzymes, such as CAT [12]. In Vigna radiata, old leaves exhibit increased oxidative stress, as evidenced by the production of ROS, which in turn leads to higher peroxidase activity and lipid peroxidation [49]. Inversely, vigorous peroxidase activity is recorded in S. cochinchinensis, especially in young to mature leaves, then gradually increases to a stable point in old leaves (p < 0.0001). According to Lu et al. [12], the activity of peroxidase (POD) increases linearly with leaf age. Besides its role as a scavenger enzyme for the H2O2 substrate, POD contributes to the lignification of cell walls, where Al3+ is mainly stored [51].
Metal stress, in this case, Al, induces lipid peroxidation within the cell, as Al3+ binds to the phospholipid surface, replacing other cations such as Ca2+ and Mg2+, thereby disrupting the stability of plasma membrane homeostasis [43,44,47]. Furthermore, the degradation of lipids leads to an increasing production of hydrogen peroxide (H2O2) by peroxisomes and glyoxisomes under oxidative stress conditions [52]. The H2O2 dismutase reaction is a parameter used to assess oxidative stress rates by measuring the catalase activity of CAT and the peroxidation activity of POD, thereby reducing it into water molecules [53,54]. In this research, young leaves exhibited high concentrations of H2O2 (Figure 6(E)). Over time, the plant develops effective defense mechanisms through enzymatic activity, resulting in a decrease in the concentration in mature leaves [12]. Furthermore, this concentration then increased significantly at its peak in older leaves of both species (p < 0.0001). Elevated concentration in old leaves suggests greater Al accumulation, which is associated with heightened oxidative stress [12,15].
Additionally, the product of lipid peroxide, called MDA, is commonly used as a marker for lipid peroxide activity during oxidative stress conditions [22]. In Vigna radiata leaves, increasing ROS levels lead to higher peroxidase activity and lipid peroxidation [49]. The MDA concentration values exhibited a different pattern between the 2 species, depicting an increasing trend from young to mature leaves in S. fasciculata (Figure 6(D)), which suggests an accumulation of oxidative stress with leaf age (p < 0.0001). Otherwise, in S. cochinchinensis (Figure 6(D)) exhibited a pattern of higher concentration in young leaves, followed by a decline in mature leaves and stabilization thereafter. Consistent with this pattern, POD and CAT activities increased efficiently from mature to old leaves (p < 0.0001), decomposing high concentrations of H2O2 in older leaves and thereby reducing lipid peroxidation [50-52]. This suggests that the detoxification mechanism against metal stress in both species is more effective in older leaves. Furthermore, a study by Shahnaz et al. [55] indicated that aluminum significantly increased MDA levels, alongside peroxidase and other aldehyde compounds [54,55]. These observations suggest that aluminum stress can adversely affect metabolism and the activity rate of enzymes involved in breaking down ROS in cells [51,54,55].
Many external factors may disrupt the activity of antioxidant enzymes, such as replacing metal cofactors with aluminum [56]. However, the correlation between aluminum content, enzyme activity (SOD, CAT and POD), lipid peroxidation product (MDA), and oxidative markers (H2O2) across leaf age in 2 species is incredibly varied (range r = −0.6 – r = > 0.7). A high aluminum content, which significantly increases with leaf age (p = 0.002), exhibits a strong positive correlation with SOD activity in S. fasciculata (r = +0.9156), playing a crucial role as the first line of defense [12]. However, a peculiar result shows a very weak relationship between aluminum content and CAT activity (r = +0.0247), as CAT activity across leaf age in S. fasciculata yields no significant results ( p > 0.05). The consequence of this is a robust correlation between aluminum content and MDA levels (r = +0.9830), similar to H2O2 (r = +0.7240), indicating a high level of oxidative stress in S. fasciculata. Such elevated stress likely constrains the ability of other enzymes [51-53], such as SOD and POD (r = +0.9045), to effectively mitigate the damage. Moreover, a strong defense mechanism against aluminum stress was displayed by S. cochinchinensis. The correlation between aluminum content and enzyme activity in this species demonstrates a highly positive correlation for all enzymes SOD (r = +0.9998), CAT (r = +0.9998), and POD (r = +0.8327), and an inverse relationship with the oxidative stress marker MDA, which exhibits a moderate negative correlation (r = −0.6245). On the other hand, H2O2 concentrations exhibit a mild positive correlation with aluminum content (r = +0.5427), indicating that enzyme activity has a practical impact due to its high correlation to maintain oxidative stress in a stable condition (SOD - CAT: r = +0.8319, SOD - POD: r = +0.9997, CAT - POD: r = +0.8429). These mechanisms lead to a speculation pattern that S. cochinchinensis adaptively responds to aluminum stress by performing dynamic enzyme activity.
Conclusions
This investigation confirms S. fasciculata and S. cochinchinensis are Al Hyperaccumulator plants. Accumulating Al in the old leaves of S. fasciculata is higher than in S. cochinchinensis. The histological assay shows Al accumulates in cell walls, epidermal tissue, mesophyll, and chloroplasts. An inverse correlation between Al content and L* value, indicating that higher al content tends to result in a darker color, approaching purple. Confirming this data, SEM-EDX and mapping clearly show higher Al accumulation in the lamina. Both species exhibit increased SOD activity, CAT activity was significantly different across leaf ages in S. cochinchinensis, yet not significantly different in S. fasciculata. Additionally, POD activity increases in all leaves from both species. Furthermore, H2O2 fluctuates and then stabilizes in old leaves, resulting in MDA consistently increasing in S. fasciculata. However, significant difference between young and mature leaves in S. cochinchinensis, suggesting that young leaves experience a high level of lipid peroxidation. The correlation between aluminum content and enzymatic activity, lipid peroxidation products, and stress markers shows a strong positive relationship in S. fasciculata for SOD, POD, MDA, and H2O2; however, a weak correlation with CAT activity suggests a stable enzymatic adaptation. Lastly, S. cochinchinensis shows a strong correlation for SOD, CAT, POD, and H2O2, except for MDA, which portrays a negative correlation due to highly dynamic stress management.
Acknowledgements
This research was supported by the Doctoral Dissertation Research Scheme of Universitas Sebelas Maret on behalf of Dewi Puspita Sari, S.Pd., M.Sc. under contract number 369/UN27.22/PT.01.03/2025. Finally, we thank Secretariat of Scientific Authority for Biodiversity on the research permit granted with number B-8447/IV/KS.00/9/2024 and Baturraden Botanical Garden in Central Java, Indonesia for the research opportunity.
Declaration of generative AI in scientific writing
The authors declared that generative AI tools, including Grammarly Edu, were utilized solely to support the writing process, specifically language refinement and grammar correction. These tools were not involved in generating original content, interpreting results, or analyzing data. The authors have thoroughly reviewed the entire manuscript and take full responsibility for its accuracy and conclusions.
CRediT author statement
Dewi Puspita Sari: Investigation, Visualization, Writing - Original draft preparation. Bambang Retnoaji: Data curation; Software. Nastiti Wijayanti: Conceptualization, Supervision, Reviewing, and Editing. Purnomo: Reviewing and Language editing.
References
[1] R Ofoe, RH Thomas, SK Asiedu, G Wang-Pruski, B Fofana and L Abbey. Aluminum in plant: Benefits, toxicity and tolerance mechanisms. Frontiers in Plant Science 2023; 13, 1085998.
[2] J Chandra and S Keshavkant. Mechanisms underlying the phytotoxicity and genotoxicity of aluminum and their alleviation strategies: A review. Chemosphere 2021; 278, 130384.
[3] E Bojórquez-Quintal, C Escalante-Magaña, I Echevarría-Machado and M Martínez-Estévez. Aluminum, a friend or foe of higher plants in acid soils. Frontiers in Plant Science 2017; 8, 1767.
[4] LV Kochian, OA Hoekenga and MA Pineros. How do crop plants tolerate acid soils? Mechanisms of aluminum tolerance and phosphorous efficiency. Annual Review of Plant Biology 2004; 55(1), 459-493.
[5] E Maejima, S Hiradate, S Jansen, M Osaki and T Watanabe. Comparative analysis of aluminum accumulation in leaves of three angiosperm species. Botany 2014; 92(5), 327-331.
[6] M Schmitt, S Boras, A Tjoa, T Watanabe and S Jansen. Aluminium Accumulation and intra-tree distribution patterns in three Arbor aluminosa (Symplocos) species from Central Sulawesi. PLoS One 2016; 11(2), e0149078.
[7] M Zheng, M Zhou, H Liu, J Shen, Y Zhang and SX Zeng. Evaluation of extracts from Symplocos sumuntia leaves used as a natural coagulant for hakka tofu. Current Topics in Nutraceutical Research 2018; 1, 29-36.
[8] L da Silva Rodrigues, DS Ferreira, ER Pereira-Filho and FMV Pereira. Mineral and trace element analysis of non-conventional food plants using ICP OES and chemometric techniques. Food Chemistry 2025; 472, 142854.
[9] LC Werdemberg dos Santos, D Granja Arakaki, E Silva de Pádua Melo and VA Nascimento. Health hazard assessment due to slimming medicinal plant intake. Biological Trace Element Research 2022; 200, 1442-1154.
[10] V D'Eusanio, E Frignani, A Marchetti, L Pigani, M Rivi and F Roncaglia. Long-term variability in the content of some metals and metalloids in aesculus flowers: A four-year study using ICP OES and PCA analysis. Molecules 2025; 30(4), 908.
[11] P Boyd and DR Hyde. Iron contributes to photoreceptor degeneration and Müller glia proliferation in the zebrafish light-treated retina. Experimental Eye Research 2022; 216, 108947.
[12] J Lu, X Qu, J Li, Z Li and J Yuan. Characteristics of aluminum accumulation and distribution in leaves of different ages in Camellia oleifera. Forests 2023; 14(12), 2322.
[13] LRM De Andrade, LMG Barros, GF Echevarria, LIV Do Amaral, MG Cotta, DR Rossatto, M Haridasan and A Franco. Al-hyperaccumulator Vochysiaceae from the Brazilian Cerrado store aluminum in their chloroplasts without apparent damage. Environmental and Experimental Botany 2011; 70(1), 37-42.
[14] NV Campos, TAR Pereira, MF Machado, MBB Guerra, GS Tolentino, JS Araujo, MQ Rezende, M Carolina, NA Da Silva and CEGR Schaefer. Evaluation of micro-energy dispersive X-ray fluorescence and histochemical tests for aluminium detection in plants from High Altitude Rocky Complexes, Southeast Brazil. Anais da Academia Brasileira de Ciencias 2014; 86(1), 285-296.
[15] Y Haruyama, T Fujiwara, K Yasuda, M Saito and K Suzuki. Localization of aluminum in epidermal cells of mature tea leaves. Quantum Beam Science 2019; 3(2), 9.
[16] IA Alaraidh, MM Ibrahim and GA El-Gaaly. Evaluation of green synthesis of Ag nanoparticles using Eruca sativa and Spinacia oleracea leaf extracts and their antimicrobial activity. Iranian Journal of Biotechnology 2014; 12(1), 8-13.
[17] S Singh, DK Tripathi, S Singh, S Sharma, NK Dubey, DK Chauhan and M Vaculík. Toxicity of aluminium on various levels of plant cells and organism: A review. Environmental and Experimental Botany 2017; 137(1), 177-193.
[18] JA Siqueira, JAS Barros, M Dal-Bianco, SCV Martins, PC Magalhães, DM Ribeiro, FM DaMatta, WL Araújo and C Ribeiro. Metabolic and physiological adjustments of maize leaves in response to aluminum stress. Theoretical and Experimental Plant Physiology 2020; 32(2), 133-145.
[19] A Bizoń, J Chojdak-Łukasiewicz, S Budrewicz, A Pokryszko-Dragan and A Piwowar. Exploring the relationship between antioxidant enzymes, oxidative stress markers, and clinical profile in relapsing-remitting multiple sclerosis. Antioxidants 2023; 12(8), 1638.
[20] M Haridasan. Nutritional adaptations of native plants of the cerrado biome in acid soils. Brazilian Journal of Plant Physiology 2008; 20(3), 183-195.
[21] SU Rahman, JC Han, M Ahmad, MN Ashraf, MA Khaliq, M Yousaf, Y Wang, G Yasin, MF Nawaz, KA Khan and Z Du. Aluminum phytotoxicity in acidic environments: A comprehensive review of plant tolerance and adaptation strategies. Ecotoxicology and Environmental Safety 2024; 269, 115791.
[22] BS dos Santos, TC Ferreira, PF Rosalem, MLG Olivio, VP Bossardi, AR Martins, LA Souza and LS de Camargos. Aluminum stress in Crotalaria spp.: anatomic changes, antioxidative response and nutrition. Plant Stress 2025; 17, 100967.
[23] M Ozturk, M Metin, V Altay, T Kawano, A Gul, B ÜNAL, D Ünal, R Bhat, M Dervash, K Toderich, E Koç, PG Caparros and A Bukhari. Aluminum toxicity: A case study on tobacco (Nicotiana tabacum L.). Phyton-International Journal of Experimental Botany 2023; 92(1), 165-192.
[24] A Choudhary, S Sharma, H Kaur, N Sharma, MM Gadewar, S Mehta and A Husen. Plant system, abiotic stress resilience, reactive oxygen species, and coordination of engineered nanomaterials: A review. South African Journal of Botany 2024; 171, 45-59.
[25] MM Rahman, PK Ghosh, M Akter, MM Al Noor, A Rahman, SS Keya, S Roni, A Biswas and M Bulle. Green vanguards: Harnessing the power of plant antioxidants, signal catalysts, and genetic engineering to combat reactive oxygen species under multiple abiotic stresses. Plant Stress 2024; 13, 100547.
[26] MC Romero-Puertas, LC Terron-Camero, MA Pelaez-Vico, A Olmedilla and LM Sandalio. Reactive oxygen and nitrogen species as key indicators of plant responses to Cd stress. Environmental and Experimental Botany 2019; 161, 107-119.
[27] S Samanta, CS Seth and A Roychoudhury. The molecular paradigm of reactive oxygen species (ROS) and reactive nitrogen species (RNS) with different phytohormone signaling pathways during drought stress in plants. Plant Physiology and Biochemistry 2024; 206, 108259.
[28] P Wang, WC Liu, C Han, S Wang, MY Bai and CP Song. Reactive oxygen species: Multidimensional regulators of plant adaptation to abiotic stress and development. Journal of Integrative Plant Biology 2024; 66(3), 30-67.
[29] Q Xu, Y Wang, Z Ding, L Song, Y Li, D Ma, Y Wang, J Shen, S Jia, H Sun and H Zhang. Aluminum induced metabolic responses in two tea cultivars. Plant Physiology and Biochemistry 2016; 101, 162-172.
[30] KAMA Rahman and D Ibrahim. Screening of antibacterial activity of endophytic fungi isolated from different leaf ages of Curcuma mangga using different growth media. International Journal of Research in Medical and Health Sciences 2015; 4, 1-10.
[31] K Anwar, L Triyasmono and MI Rizki. The influence of leaf age on total phenolic, flavonoids, and free radical scavenging capacity of Aquilaria beccariana. Research Journal of Pharmaceutical Biological and Chemical Sciences 2017; 8, 129-133.
[32] M Frankowski, A Zioła-Frankowska and J Siepak. From soil to leaves - Aluminum fractionation by single step extraction procedures in polluted and protected areas. Journal of Environmental Management 2013; 127, 1-9.
[33] MJ Akhter, S Akhter, S Islam, MSH Sarker and SMK Hasan. Varietal influence on bioactive compounds and antioxidant activity in chilies during development stages. Heliyon 2024; 10, 37406.
[34] S Marklund and G Marklund. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. European Journal of Biochemistry 1974; 47(3), 469-474.
[35] LM Ellerby and DE Bredesen. Measurement of cellular oxidation, reactive oxygen species, and antioxidant enzymes during apoptosis. Methods in Enzymology 2000; 322, 413-421.
[36] EH Alici and G Arabaci. Determination of SOD, POD, PPO and cat enzyme activities in Rumex obtusifolius L. Annual Research and Review in Biology 2016; 11(3), 1-7.
[37] Nurkhasanah, MS Bachri and S Yuliani. Antioksidan dan stres oksidatif (in Indonesian). UAD Press, Yogyakarta, Indonesia, 2023.
[38] D Hadi and S Siswadi. Aluminum levels on leaves, bark, and root of two Loba species: Loba wawi (Symplocos fasciculata Zoll.) and Loba manu (Symplocos cochinchinensis). International Conference of Indonesia Forestry Reseacher 2011; 6, 72-74.
[39] R Shen, J Ma and M Kyo. Compartmentation of aluminium in leaves of an Al-accumulator, Fagopyrum esculentum Moench. Planta 2002; 215(3), 394-398.
[40] S Saberi, MIE Halmi, NA Ramle and K Mahmud. Metals accumulation of tropical shrub Melastoma malabathricum L. (Melastomataceae) populations and their relation to soil edaphic factor. Malaysian Applied Biology 2024; 53(1), 113-125.
[41] D Li, Z Shu, X Ye, J Zhu, J Pan, W Wang, P Chang, C Cui, J Shen, W Fang, X Zhu and Y Wang. Cell wall pectin methyl-esterification and organic acids of root tips involve in aluminum tolerance in Camellia sinensis. Plant Physiology and Biochemistry 2017; 119(2), 65-74.
[42] JR Baker. Experiments on the action of mordants 2. aluminum - haematein. Journal of Cell Science 1962; 103(4), 493-517.
[43] QL Zeng, RF Chen, XQ Zhao, HY Wang and RF Shen. Aluminium uptake and accumulation in the hyperaccumulator Camellia oleifera abel. Pedosphere 2011; 21(3), 358-364.
[44] R Hajiboland, CK Panda, O Lastochkina, MA Gavassi, G Habermann and JF Pereira. Aluminum toxicity in plants: Present and future. Journal of Plant Growth Regulation 2023; 42(7), 3967-3999.
[45] R Tolrà, S Martos, R Hajiboland and C Poschenrieder. Aluminium alters mineral composition and polyphenol metabolism in leaves of tea plants (Camellia sinensis). Journal of Inorganic Biochemistry 2020; 204, 110956.
[46] I Brunner and C Sperisen. Aluminum exclusion and aluminum tolerance in woody plants. Frontiers in Plant Science 2013; 4, 1-12.
[47] MA Rahman, SH Lee, HC Ji, AH Kabir, CS Jones and KW Lee. Importance of mineral nutrition for mitigating aluminum toxicity in plants on acidic soils: Current status and opportunities. International Journal of Molecular Sciences 2018; 19(10), 3073.
[48] V Munyaneza, W Zhang, S Haider, F Xu, C Wang and G Ding. Strategies for alleviating aluminum toxicity in soils and plants. Plant and Soil 2024; 504(1), 167-190.
[49] GJ Ahammed, Z Li, J Chen, Y Dong, K Qu, T Guo, F Wang, A Liu, S Chen and X Li. Reactive oxygen species signaling in melatonin-mediated plant stress response. Plant Physiology and Biochemistry 2024; 207, 108398.
[50] M Naz, MR Afzal, MA Raza, S Pandey, S Qi, Z Dai and D Du. Calcium (Ca2+) signaling in plants: A plant stress perspective. South African Journal of Botany 2024; 169(4), 464-485.
[51] U Chowra, E Yanase, H Koyama and SK Panda. Aluminium-induced excessive ROS causes cellular damage and metabolic shifts in black gram Vigna mungo (L.) Hepper. Protoplasma 2017; 254(1), 293-302.
[52] SK Jalmi, PK Bhagat, D Verma, S Noryang, S Tayyeba, K Singh, D Sharma and AK Sinha. Traversing the links between heavy metal stress and plant signaling. Frontiers in Plant Science 2018; 9, 12.
[53] R Berni, M Luyckx, X Xu, S Legay, K Sergeant, JF Hausman, S Lutts, G Cai and G Guerriero. Reactive oxygen species and heavy metal stress in plants: Impact on the cell wall and secondary metabolism. Environmental and Experimental Botany 2019; 161, 98-106.
[54] P García-Pérez, S Losada-Barreiro, C Bravo-Díaz, PP Gallego and L Lucini. Plant oxidative stress and specialized metabolites: a holistic physicochemical and metabolomics perspective. Plant Physiology and Biochemistry 2025; 227, 110093.
[55] T Guo, G Zhang, M Zhou, F Wu and J Chen. Effects of aluminum and cadmium toxicity on growth and antioxidant enzyme activities of two barley genotypes with different Al resistance. Plant and Soil 2004; 258, 241-248.
[56] R Kouki, I Bankaji, S Hidouri, H Bouzahouane, I Caçador, RM Pérez-Clemente and N Sleimi. Physiological behavior and antioxidant responses of Abelmoschus esculentus (L.) exposed to different concentrations of aluminum and barium. Horticulturae 2024; 10(12), 1338.
[57] G Shahnaz, E Shekoofeh, D Kourosh and B Moohamadbagher. Interactive effects of silicon and aluminum on the malondialdehyde (MDA), proline, protein, and phenolic compounds in Borago officinalis L. Journal of Medicinal Plants Research 2011; 5(58), 18-27.
[58] NG Nidhi Gupta, SS Gaurav and AK Ashwani Kumar. Molecular basis of aluminium toxicity in plants: A review. American Journal of Plant Sciences 2013; 4, 21-37.