
Trends
Sci.
2026;
23(6):
12592
i-PRF–Derived Exosomes Promote Hair Regeneration by Modulating Inflammatory, Oxidative, Angiogenic, and Regenerative Pathways in an Androgenetic Alopecia Mouse Model
Adniana Nareswari1,2,*, Paramasari Dirgahayu1, Didik Gunawan Tamtomo1,
Harijono Kariosentono1,2, Ratih Puspita Febrinasari1,3 and Muhammad Eko Irawanto2
1Doctoral Program of Medical Sciences, Faculty of Medicine, Universitas Sebelas Maret, Central Java, Indonesia
2Department of Dermatology Venereology, Faculty of Medicine, Universitas Sebelas Maret, Central Java, Indonesia
3Department of Pharmacology, Faculty of Medicine, Universitas Sebelas Maret, Central Java, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 9 November 2025, Revised: 15 December 2025, Accepted: 25 December 2025, Published: 1 February 2026
Abstract
Androgenetic alopecia (AGA) is a progressive hair loss disorder characterized by hair follicle miniaturization resulting from androgenic stimulation, inflammation, oxidative stress, and apoptosis. Exosomes derived from injectable Platelet-Rich Fibrin (i-PRF) contain growth factors and microRNAs that may restore the follicular microenvironment and promote regeneration. This study aimed to evaluate the therapeutic effects of i-PRF-derived exosomes on inflammatory, oxidative, apoptotic, and regenerative markers in a testosterone-induced AGA mouse model. Thirty male C57BL/6 mice were randomly allocated into six groups: normal control (K), negative control (KN), positive control treated with 5% minoxidil (KP), i-PRF exosome 0.1 mL (P1), i-PRF exosome 0.2 mL (P2), and a combination of i-PRF exosome 0.2 mL with 5% minoxidil (P3). Treatments were administered for 14 days following testosterone induction. Serum analyses demonstrated that all i-PRF-treated groups showed significant reductions in serum TNF-α, IL-6, MDA, and Caspase-3 levels compared with the negative control (all p-value < 0.05), confirming anti-inflammatory, antioxidant, and anti-apoptotic activities. Immunohistochemistry revealed increased VEGF and β-catenin expression across i-PRF-treated groups, with the most prominent upregulation observed in the combination (P3) group, indicating enhanced angiogenesis and Wnt/β-catenin pathway activation. Histological evaluation further demonstrated increased dermal thickness and hair follicle density, most prominently in the combination group. These findings suggest that i-PRF-derived exosomes effectively mitigate inflammation, oxidative stress, and apoptosis while promoting angiogenesis and follicular regeneration in AGA. The synergistic combination of i-PRF-derived exosomes and minoxidil produced the most substantial regenerative outcomes, highlighting their potential as a safe, autologous, and biocompatible nanotherapy for androgenetic alopecia.
Keywords: Androgenetic alopecia, Injectable platelet-rich fibrin, Exosome therapy, Inflammation, Oxidative stress, Apoptosis, VEGF, β-catenin, Hair follicle regeneration
Introduction
Androgenetic alopecia (AGA) is a progressive hair thinning disorder characterized by patterned follicular miniaturization influenced by genetic and hormonal factors. In men, it typically manifests as frontotemporal recession and vertex thinning, while in women, it presents as diffuse crown thinning with preservation of the frontal hairline [1]. The global prevalence of AGA increases with age, affecting approximately 50% of men and 30% of women after the age of 50 years [2]. In Asian populations, prevalence ranges from 30 - 40% in men aged 20 - 30 years and nearly 100% after 80 years, while in Indonesia, AGA affects about 31.2% of adults, with an earlier mean onset at 29 years [1,3,4].
Current therapeutic approaches for AGA include oral and topical agents such as finasteride and minoxidil, hormonal modulation, nutraceuticals, platelet-rich plasma (PRP), exosome therapy, microneedling, and surgical transplantation [2,5,6]. However, these modalities show variable efficacy, transient effects, and potential adverse reactions [2,7,8]. The exploration of autologous regenerative options has therefore gained increasing interest, particularly platelet-derived bioproducts.
Platelet-rich Fibrin (PRF), a second-generation platelet concentrate, offers distinct advantages over PRP because it forms a natural fibrin matrix that allows gradual degradation and sustained release of bioactive molecules for up to 14 days [9,10]. PRF contains numerous growth factors and cytokines—such as Vascular Endothelial Growth Factor (VEGF), Platelet-Derived Growth Factor (PDGF), Transforming Growth Factor-β (TGF-β), and Insulin-Like Growth Factor (IGF)—that support tissue regeneration and angiogenesis, potentially improving the hair cycle and follicular health. In addition, exosomes nanosized vesicles (30 - 150 nm) secreted by various cell types mediate intercellular communication by transferring proteins, mRNAs, and microRNAs that regulate cellular repair and signaling. Exosomes derived from PRF are thought to enhance the regenerative efficacy of platelet concentrate through anti-inflammatory, angiogenic, and folliculogenic mechanisms [11-14].
AGA pathogenesis involves a complex interplay between androgenic stimulation, inflammatory cytokines, oxidative stress, and apoptosis, leading to disrupted follicular homeostasis and impaired Wnt/β-catenin signaling [15]. In this study, injectable PRF (i-PRF) and its exosomal fraction were investigated in a testosterone-induced AGA mouse model. The analysis focused on inflammatory (TNF-α, IL-6), oxidative (MDA), and apoptotic (Caspase-3) biomarkers, alongside angiogenic (VEGF) and regenerative (β-catenin) markers, with dermal thickness and hair follicle density as histological outcomes.
Materials and methods
This in vivo experimental study employed a randomized controlled trial (RCT) design and was conducted from March to August 2025. The research was collaboratively performed at the Dermama Biotechnological Clinical Laboratory, Surakarta; the Food and Nutrition Study Center Laboratory, Universitas Gadjah Mada, Yogyakarta; and the Department of Anatomical Pathology, Faculty of Medicine, Universitas Sebelas Maret, Surakarta. All experimental procedures were approved by the Health Research Ethics Committee of Dr. Moewardi General Hospital, under approval number 471/III/HREC/2025, and were conducted in accordance with international ethical standards for animal experimentation.
Subjects
Male C57BL/6 mice were used as experimental subjects and obtained from the Animal Laboratory, Kemuning, Karanganyar. A purposive sampling technique was applied based on predefined inclusion and exclusion criteria. The inclusion criteria were healthy male mice aged 7 - 8 weeks, weighing 20 - 30 g, and exhibiting normal physical activity and behavior. The exclusion criteria included the presence of skin lesions or physical deformities, body weight gain exceeding 10% during the 7-day acclimatization period, or abnormal behaviors such as reduced exploratory activity, piloerection, dehydration, or distress vocalization. Mice that died during acclimatization were also excluded. The sample size was determined using the Federer formula for six experimental groups, with an additional 10% added to anticipate potential dropouts, resulting in a total of 30 male C57BL/6 mice included in the study.
AGA animal model induction
An androgenetic alopecia (AGA) model was established in male C57BL/6 mice (8 - 10 weeks old, 20 - 30 g) through subcutaneous administration of testosterone propionate (TP). Each mouse received TP at a dose of 1.5 mg/day for 14 consecutive days, injected subcutaneously into the dorsal skin. This dosage corresponded to approximately 50 - 75 mg/kg/day, consistent with validated protocols reported in previous AGA induction studies. Preliminary titration using doses of 0.5, 1.0, and 1.5 mg/day identified 1.5 mg/day as the optimal regimen, producing consistent androgen-induced alopecia features such as delayed hair regrowth, follicular miniaturization, and dermal thinning, without systemic toxicity. All injections were administered at a single 2×2 cm depilated dorsal site under brief isoflurane anesthesia to minimize stress. The procedure was performed once daily using a 29 G insulin syringe by the same trained investigator to ensure reproducibility and standardization of the induction process.
A pilot validation study (n = 3 per group) was first performed to determine the optimal TP dose and duration. Twenty-one mice were randomized into seven groups: One control (K) and six experimental groups (P1-P6) receiving TP at 0.5, 1.0, or 1.5 mg/day for 7 or 14 days. Dose- and time-dependent effects were observed, with progressive dermal thinning, follicular miniaturization, and suppression of β-catenin expression as testosterone exposure increased. The 1.5 mg/day for 14 days protocol (P6) produced the most consistent AGA phenotype, showing complete inhibition of hair regrowth, marked dermal thinning (367.87 ± 45.51 µm), decreased follicle density (31.00 ± 6.57 follicles/mm²), and significant β-catenin downregulation (2.80 ± 1.40) compared with controls (p-value < 0.05).
Statistical analysis confirmed significant reductions in dermal thickness (p-value < 0.001), follicle density (p-value = 0.028), and β-catenin (p-value = 0.017) across the higher-dose group (Table 1). These pilot results validated that subcutaneous TP at 1.5 mg/day for 14 days reliably induced a reproducible AGA phenotype in C57BL/6 mice characterized by clinical alopecia, dermal atrophy, follicular regression, and downregulating Wnt/β-catenin signaling (Figure 1). This optimized induction protocol was subsequently applied in the main therapeutic study using i-PRF-derived exosomes.
Table 1 Dermal thickness, hair follicle density, and β-catenin expression in C57BL/6 mice after testosterone propionate induction.
Group |
Clinical hair regrowth |
Dermal Thickness |
Hair Follicle Density |
β-catenin Expression |
||||||
Mean |
± SD |
p-value |
Mean |
± SD |
p-value |
Mean |
± SD |
p-value |
||
K (Control) |
Full regrowth |
785.57 |
± 50.64 |
< 0.001* |
132.67 |
± 38.90 |
0.028* |
14.59 |
± 3.69 |
0.017* |
P1 (0.5 mg, 7 days) |
Partial |
731.10 |
± 44.94 |
|
91.42 |
± 4.54 |
|
13.74 |
± 3.68 |
|
P2 (1.0 mg, 7 days) |
Minimal |
702.69 |
± 12.29 |
|
46.17 |
± 5.70 |
|
13.42 |
± 1.85 |
|
P3 (1.5 mg, 7 days) |
None |
464.51 |
± 126.30 |
|
37.50 |
± 9.93 |
|
5.05 |
± 0.90 |
|
P4 (0.5 mg, 14 days) |
Sparse |
524.91 |
± 124.09 |
|
62.92 |
± 16.59 |
|
13.00 |
± 6.70 |
|
P5 (1.0 mg, 14 days) |
Delayed |
485.05 |
± 94.26 |
|
43.42 |
± 38.98 |
|
8.88 |
± 2.08 |
|
P6 (1.5 mg, 14 days) |
None |
367.87 |
± 45.51 |
|
31.00 |
± 6.57 |
|
2.80 |
± 1.40 |
|
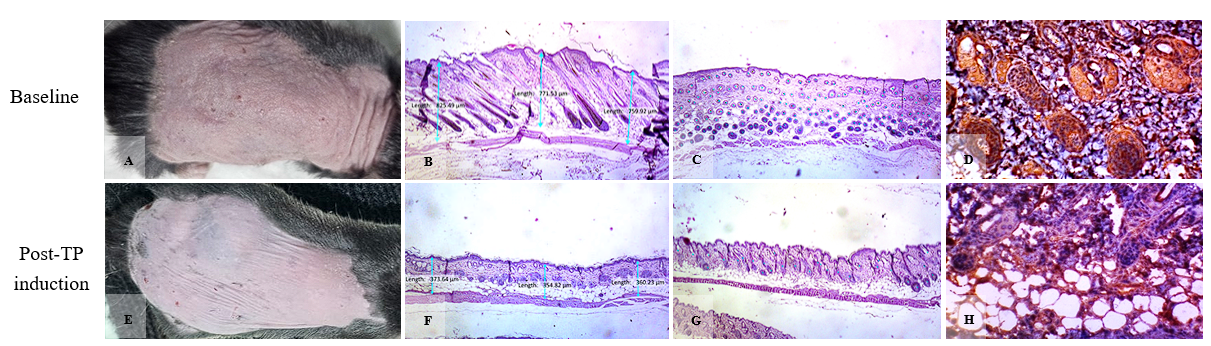
Figure 1 Validation of the testosterone-induced androgenetic alopecia (AGA) model in C57BL/6 mice. (A - D) Baseline (pre-induction): Normal hair coverage and textured skin; H&E shows a thick dermis with abundant, large anagen hair follicles; strong β-catenin staining in follicular epithelium. (E–H) After TP (1.5 mg/day s.c., 14 consecutive days): Clinical failure of hair regrowth with smoother, thinner skin; H&E demonstrates dermal thinning and marked reduction/miniaturization of hair follicles; β-catenin staining is weak, indicating down-regulated Wnt/β-catenin activity.
Stains: H&E and β-catenin IHC.
Grouping
A total of 30 male C57BL/6 were randomly assigned into six groups, with five animals per group. Randomization was performed using a computer-generated random sequence (Microsoft Excel RAND function) prepared by an independent assistant. Cages were coded to conceal treatment identity, and all investigators conducting ELISA, histopathological, and immunohistochemical (IHC) analyses were blinded to group allocation until completion of data analysis.
The study design included one normal control group, one negative control group, one positive control group, and three treatment groups. The normal control (K) group comprised healthy mice without testosterone induction, receiving only topical vaseline album (62.5 mg/day) from day 1 to day 21. The negative control (KN) group consisted of AGA model mice induced with testosterone propionate at a dose of 1.5 mg/day from day 1 to day 21 without receiving any therapeutic intervention. The positive control (KP) group comprised AGA model mice treated topically with 5% minoxidil for 14 consecutive days (day 8 - day 21) following testosterone induction.
The treatment groups were defined as follows: P1 received intradermal i-PRF exosome injections of 0.1 mL/week on days 8 and 15; P2 received intradermal i-PRF exosome injections of 0.2 mL/week on the same schedule; and P3 received combination therapy consisting of 5% minoxidil (8 - 21 days) and intradermal i-PRF exosome injections of 0.2 mL/week (days 8 and 15). In all testosterone-treated groups, androgenetic alopecia induction was achieved by subcutaneous administration of testosterone propionate (1.5 mg/day) to the depilated dorsal area from day 1 to day 21.
Exosomes from platelet-rich fibrin (Exos-PRF)
Platelet-rich fibrin (PRF) was prepared from venous blood obtained from healthy volunteers under predetermined donor eligibility criteria. Human blood collection was approved by the Health Research Ethics Committee of Dr. Moewardi Hospital, Surakarta, Indonesia (Approval No. 471/UNS/KEPK/2025). Written informed consent was obtained from all donors after a detailed explanation of the study objectives, procedures, and potential minimal risks. Eligible donors were non-smokers aged 20 - 35 years, with no history of systemic disease, infection, or recent medication affecting platelet function. Blood samples were collected exclusively for research use and processed under biosafety level 2 (BSL-2) conditions. All samples were pooled and processed under sterile conditions to obtain PRF intended solely for animal experimental use. No clinical human data or interventions were involved in this study.
Injectable PRF (i-PRF) was prepared by centrifuging whole blood without anticoagulant at 700 rpm for 8 min. Exosomes were then isolated from i-PRF using size-exclusion chromatography (SEC, qEV2 Izon Science®, New Zealand). Quantitative analysis by nanoparticle tracking (ViewSizer 3000, HORIBA Scientific) showed fraction 12 contained the highest concentration (1.4×1011 particles/mL, 125 - 142 nm). Flow cytometry confirmed low CD45+ contamination (10.63%) and high expression of CD63+/CD81+ (95.35%). These findings validate SEC as an effective method to obtain pure PRF-derived exosomes (Exo-PRF) with typical exosomal size and marker profiles suitable for downstream biological studies.
The dosage of PRF-derived exosomes used in this study was determined based on the quantified particle concentration obtained from prior characterization and supported by previous in vivo studies employing regenerative exosome therapy. Quantitative analysis using the Izon qEV system at the Dermama Laboratory revealed that the exosome fraction isolated from PRF by size-exclusion chromatography (SEC) contained approximately 1.4×1011 particles/mL.
According to published literature, the effective therapeutic range of exosomes in small-animal models typically lies between 1×108 and 1×1010 particles per injection, depending on the treatment objective and route of administration. Two working doses were therefore selected: A low dose (0.1 mL) and a high dose (0.2 mL) per mouse, corresponding to approximately 1.4×1010 and 2.8×1010 particles, respectively. These doses fall within the safe and biologically active range reported in previous studies, capable of exerting regenerative effects on hair follicles without cytotoxicity.
Exosome suspensions were administered locally by intradermal injection to the dorsal alopecic area, with repeated applications up to day 14 post-induction. The use of two dose levels was intended to evaluate potential dose-dependent effects of PRF-derived exosomes on hair follicle regeneration, dermal structural restoration, and modulation of inflammatory, oxidative, and angiogenic biomarkers. The results are expected to provide preliminary insight into the optimal therapeutic dosage for further translational research and potential clinical application.
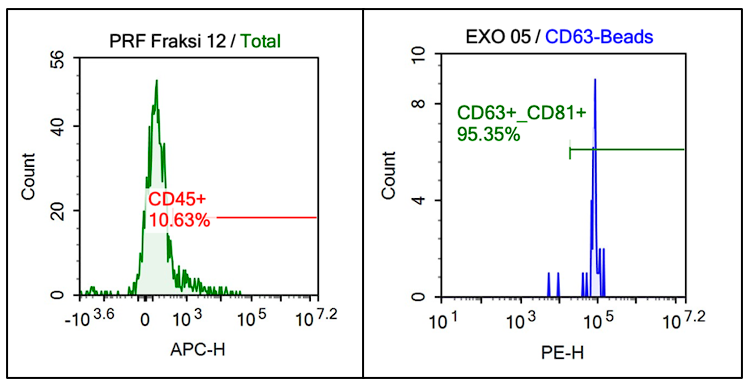
Figure 2 Flow cytometry analysis of PRF-derived exosomes.
PRF fraction 12 showed low CD45⁺ expression (10.63%), indicating minimal leukocyte contamination, while exosomes captured with CD63-beads exhibited strong CD63⁺/CD81⁺ expression (95.35%), confirming high exosomal purity.
Minoxidil
Topical 5% minoxidil was used in this study at a dosage of 100 μL (0.1 mL) per day. The dose was selected based on a previously published study in the Journal of Drugs in Dermatology, which demonstrated the effectiveness of topical minoxidil for androgenetic alopecia, including a validated mouse model. The study reported that daily application of 5% minoxidil in mice produced comparable outcomes to twice-daily application at the same concentration in humans, supporting its translational relevance and therapeutic efficacy across different experimental settings.
Procedure
C57BL/6 mice were acclimatized for 7 days under controlled laboratory conditions, receiving standard pellet feed, ad libitum access to water, a 12-hour light–dark cycle, a room temperature of 25 ± 2°C, and relative humidity of 50 ± 5%. On day 1, the dorsal hair of each mouse was carefully shaved to prepare the treatment area. Mice were anesthetized using an intramuscular injection of 0.1 mL per 10 g body weight of an anesthetic cocktail containing ketamine (80 mg/kg), xylazine (12.5 mg/kg), and acepromazine (3 mg/kg). Prior to treatment, the dorsal fur was completely removed using a Veet® depilatory cream until the skin surface was clearly visible.
Clinical assessment of hair regrowth was performed by serial digital photography on days 0, 7, 14, and 21 for all groups. On day 7, all mice were photographed, and blood samples were collected for baseline biochemical evaluation. On day 21, after final clinical documentation and blood collection, all mice were euthanized by cervical dislocation under deep anesthesia. Following euthanasia, dorsal skin tissue samples were excised using sterile surgical forceps and scissors, then immediately immersed in 10% buffer neutral formalin (BNF) for fixation and subsequent histological analysis.
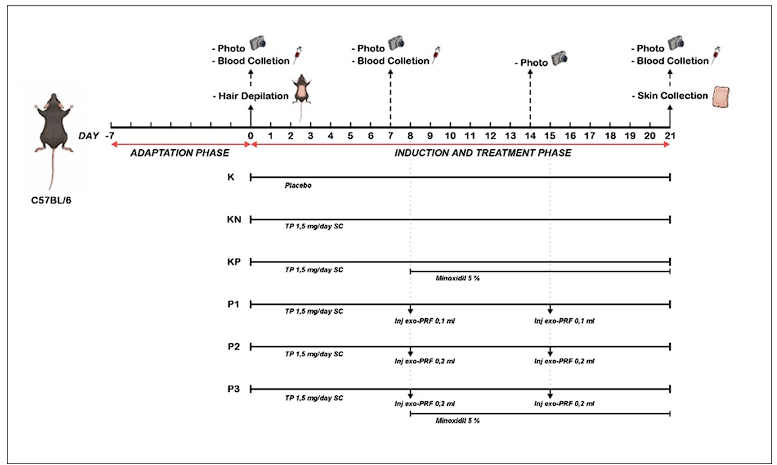
Figure 3 Experimental design and treatment timeline of AGA mouse model.
Outcome evaluation
Serum levels of tumor necrosis factor alpha (TNF-α), interleukin-6 (IL-6), malondialdehyde (MDA), and Caspase-3 were quantified using enzyme-linked immunosorbent assay (ELISA) from blood samples collected before and after treatment. Measurements were performed spectrophotometrically and expressed in ng/mL.
The expression of vascular endothelial growth factor (VEGF) and β-catenin was analyzed by immunohistochemical (IHC) staining on paraffin-embedded dorsal skin sections. Staining intensity was quantified using ImageJ software (NIH, USA) by calculating the percentage of positively stained area (%Area) obtained from color-thresholded binary images. All image analyses were independently performed by two blinded observers to ensure accuracy and reproducibility.
Dermal thickness was measured morphometrically on hematoxylin and eosin (H&E)-stained sections as the perpendicular distance from the stratum basale to the dermis-subcutis boundary across at least five standardized microscopic fields per section and three representative sections per sample. Measurements were expressed in micrometers (µm). Hair follicle density was determined histologically from H&E-stained sections and expressed as the number of follicles per millimeter (follicles/mm).
In summary, serum biomarkers (ELISA parameters) were evaluated both before and after treatment, whereas histopathological and immunohistochemical parameters were analyzed post-treatment to assess structural and molecular regeneration outcomes.
Statistical analysis
All statistical analyses were performed using Statistical Package for the Social Sciences (SPSS) software version 26.0 for Windows (IBM Corp., Armonk, NY, USA). The significance level (α) was set at 0.05. Data normality was assessed using the Shapiro-Wilk test, and homogeneity of variance was evaluated using Levene's test. For data that were normally distributed and homogenous, one-way analysis of variance (ANOVA) was applied, followed by the Least Significant Difference (LSD) post hoc test to determine intergroup differences. For non-normally distributed data, the Kruskal-Wallis test was used as a non-parametric alternative, followed by pairwise comparisons with the Mann-Whitney U test. All quantitative results were expressed as mean ± standard deviation (SD) and presented in both tabular and graphical formats. Statistical significance was accepted at p-value < 0.05.
Results and discussion
Thirty male C57BL/6 mice aged 7 - 8 weeks and weighing 20 - 30 g were used in this study. All animals underwent a 7-day acclimatization period under controlled environmental conditions: A 12 h light/dark cycle, ambient temperature of 25 ± 2 °C, and relative humidity of 50 ± 5%, with ad libitum access to a standard pellet diet and water. Mice were weighed before and after acclimatization to ensure health stability.
Eligible mice were randomly assigned into six groups (five animals per group) by two independent laboratory assistants. Each group was marked with a distinct waterproof color for identification. Daily health monitoring was performed, and animals showing signs of illness—such as > 20% body weight loss, reduced exploratory activity, abnormal posture, piloerection, or aggressive behavior—were excluded. Body weight changes throughout the experiment are summarized in Figure 3.
To confirm successful AGA model establishment, serum biomarkers including: TNF-α, IL-6, MDA, and Caspase-3 were analyzed for significant differences between testosterone-induced groups (KN, KP, P1, P2, and P3) and the normal control group (K).
Effects of testosterone propionate induction on serum TNF-α, IL-6, MDA, and Caspase-3 Levels in C57BL/6 mice prior to treatment
Baseline analysis showed no significant difference in serum levels of TNF-α, IL-6, MDA, and Caspase-3 among all groups (p-value > 0.05), indicating comparable pre-induction conditions. Following 14 days of subcutaneous testosterone propionate (TP) administration, one-way ANOVA demonstrated significant elevations in all four biomarkers compared with the non-induced control group (p-value < 0.001).
Post hoc analysis confirmed that testosterone-treated groups (KN, KP, P1, P2, and P3) exhibited markedly higher serum TNF-α, IL-6, MDA, and Caspase-3 concentrations, while inter-group differences among testosterone-treated mice were not statistically significant (p-value > 0.05). These results indicate that TP induction effectively triggered systemic inflammatory, oxidative, and apoptotic responses characteristic of AGA pathophysiology.
In summary, testosterone propionate administration significantly increased serum TNF-α, IL-6, MDA, and Caspase-3 levels compared to baseline, validating the successful establishment of a reproducible AGA mouse model for subsequent therapeutic evaluation.
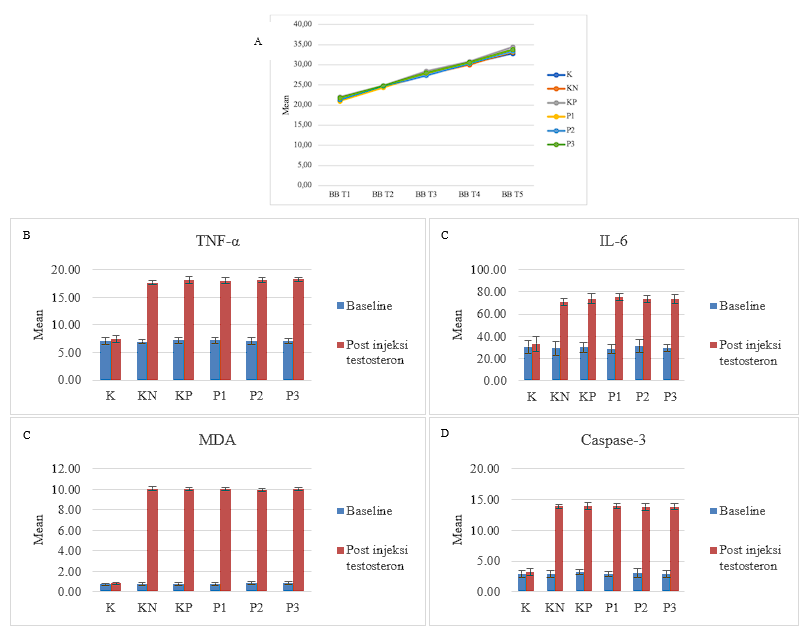
Figure 4 Changes in Body Weight and Serum Biomarkers Following Testosterone Propionate Induction in C57BL/6 Mice. (A) Line graph showing the progressive increase in mean body weight (g) of C57BL/6 mice throughout the experimental period, demonstrating maintained physiological stability across all groups. (B–E) Bar graphs comparing baseline and post–testosterone propionate (TP) injection levels of (B) TNF-α, (C) IL-6, (D) MDA, and (E) Caspase-3. All testosterone-treated groups (KN, KP, P1, P2, and P3) exhibited a significant increase in these serum markers compared to the untreated control (K) (p < 0.001), indicating successful establishment of the androgenetic alopecia (AGA) model characterized by elevated inflammatory, oxidative, and apoptotic activity.
Trends in serum inflammatory, oxidative, and apoptotic markers following therapy
Paired t-test analysis demonstrated significant reductions in serum TNF-α, IL-6, MDA, and Caspase-3 levels after treatment in all testosterone-induced AGA groups (p-value < 0.05). The combination therapy group (P3: 0.2 mL i-PRF-derived exosomes + 5% minoxidil) exhibited the greatest decrease in all four parameters, followed by the high-dose exosomes group (P2) and the positive control (KP; 5% minoxidil alone). The low-dose exosome group (P1) showed the smallest, yet still significant, reduction compared with the negative control (KN).
Overall, these findings indicate that i-PRF-derived exosomes effectively attenuated inflammatory (TNF-α, IL-6), oxidative (MDA), and apoptotic (Caspase-3) responses in the testosterone-induced AGA model. The reduction was dose-dependent and further enhanced by combination therapy with minoxidil, supporting the synergistic anti-inflammatory and cytoprotective potential of exosome-based treatment in restoring follicular homeostasis.
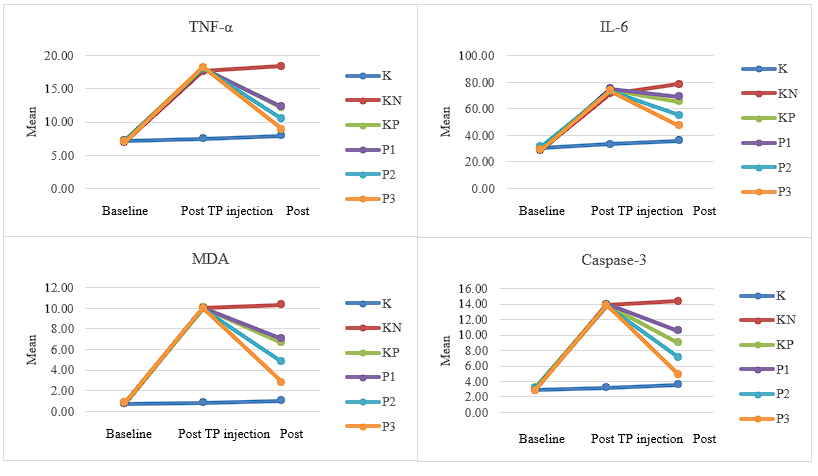
Figure 5. Trends in serum inflammatory, oxidative, and apoptotic biomarkers (TNF-α, IL-6, MDA, and Caspase-3) at baseline, after testosterone propionate (TP) induction, and post-therapy in C57BL/6 mice.
Post-treatment biochemical, IHC, and histological outcomes
After treatment, significant differences were observed in serum TNF-α, IL-6, MDA, and Caspase-3 levels among groups (one-way ANOVA, all p-value < 0.001; Figure 6). All treated AGA groups showed lower mean values than the testosterone-only group (KN). The most marked reductions were observed in group P3 (i-PRF-derived exosomes combined with 5% minoxidil), followed by P2 (0.2 mL exosomes), KP (5% minoxidil), and P1 (0,1 mL exosomes). Post hoc comparisons confirmed significant decreases in inflammatory, oxidative, and apoptotic markers in P3 compared with KN (p-value < 0.05), indicating a dose-dependent and synergistic therapeutic effect.
Immunohistochemical analysis demonstrated significant differences in VEGF (p-value = 0.012) and β-catenin (p-value = 0.021) expression among groups (Table 2 and Figure 7). VEGF expression was highest in group P3 and significantly higher than both KN (p-value = 0.002) and KP (p-value = 0.004). Similarly, β-catenin expression was significantly elevated in P3 compared with KN (p-value = 0.005) and KP (p-value = 0.005), whereas P1 and P2 did not differ significantly from KN. These findings suggest that the combination of PRF-derived exosomes and minoxidil enhanced angiogenesis and Wnt/β-catenin signaling more effectively than single therapies.
Histopathological evaluation revealed that dermal thickness differed significantly among groups (ANOVA, p-value = 0.005); Table 2 and Figure 7). All treated groups exhibited thicker dermis than KN (p-value < 0.05), with the greatest thickness observed in P3, followed by P2, KP, and P1. Hair follicle density also varied significantly (Kruskal-Wallis, p = 0.014; Table 4 and Figure 7). Post hoc analysis showed that KP, P2, and P3 had higher follicle counts than KN (p-value ≤ 0.026), while P1 did not differ significantly.
Overall, these findings indicate that treatment with PRF-derived exosomes improved inflammatory, oxidative, apoptotic, and regenerative parameters. The combination of PRF-derived exosomes with 5% minoxidil produced the most consistent and significant improvements, supporting its potential as an effective therapeutic strategy for AGA.
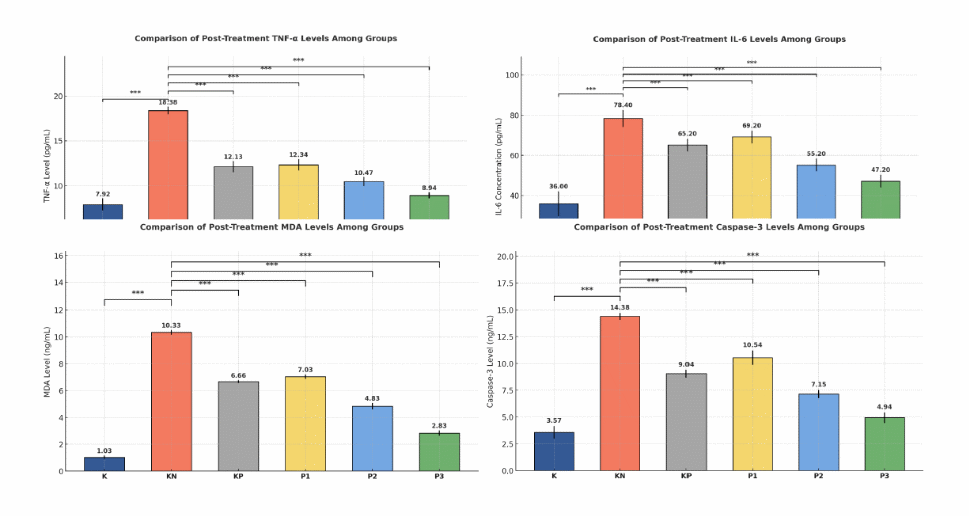
Figure 6 Comparison of post-treatment serum levels of TNF-α, IL-6, MDA, and Caspase-3 among experimental groups. Data are presented as mean ± SD (n = 5). K: Normal control; KN: Negative control (testosterone only); KP: Positive control (testosterone + 5% minoxidil); P1: Testosterone + PRF exosomes (0.1 mL); P2: Testosterone + PRF exosomes (0.2 mL); P3: Testosterone + 5% minoxidil + PRF exosomes (0.2 mL). Statistical analysis was performed using one-way ANOVA followed by the LSD post hoc test. Asterisks indicate significant differences compared with the negative-control group (KN) (*p-value < 0.05, **p-value < 0.01, ***p-value < 0.001).
Table 2 Post-treatment VEGF and β-catenin expression, dermal thickness, and hair follicle density across groups.
|
VEGF (%Area) |
β-catenin (%Area) |
Dermal thickness (µm) |
Hair follicle density (follicles/mm) |
||||||||||
Group |
Mean |
SD |
p-value |
Mean |
SD |
p-value |
Mean |
SD |
p-value |
Mean |
SD |
p-value |
|
|
K |
13.73 |
4.41 |
0.012* |
10.46 |
11.48 |
0.021* |
1168.67 |
316.34 |
0.005* |
43.10 |
18.34 |
0.014* |
|
|
KN |
6.19 |
1.40 |
|
2.84 |
0.94 |
|
517.25 |
165.11 |
|
13.40 |
4.29 |
|
|
|
KP |
7.33 |
5.74 |
|
2.90 |
1.05 |
|
853.13 |
213.14 |
|
46.40 |
21.96 |
|
|
|
P1 |
8.86 |
1.53 |
|
3.67 |
1.58 |
|
828.68 |
238.50 |
|
27.05 |
12.18 |
|
|
|
P2 |
10.92 |
5.47 |
|
5.01 |
2.05 |
|
864.52 |
169.18 |
|
36.45 |
25.74 |
|
|
|
P3 |
16.94 |
6.92 |
|
6.51 |
0.75 |
|
948.44 |
205.76 |
|
48.00 |
26.18 |
|
|
|
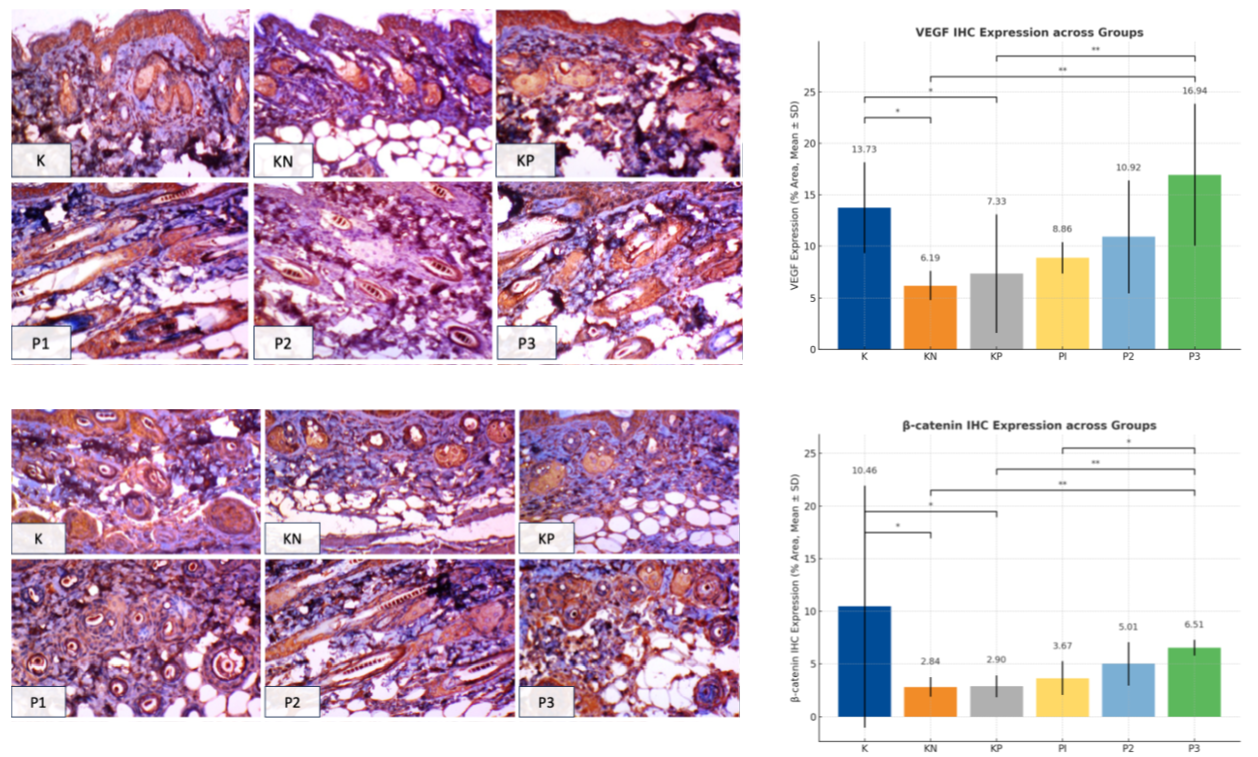
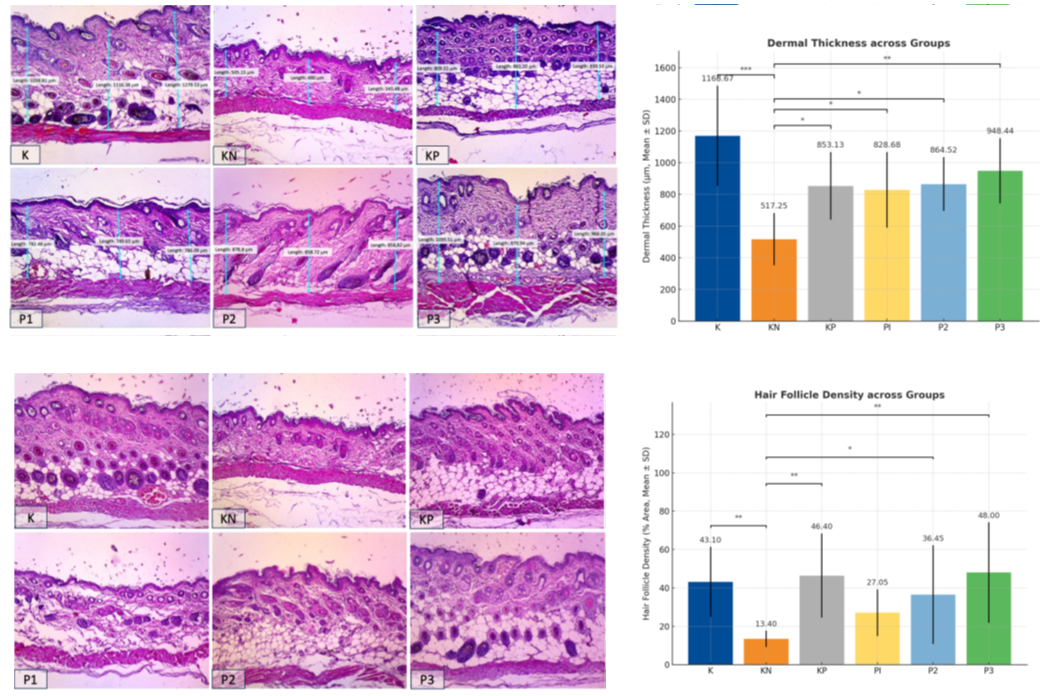
Figure 7 Representative IHC and histological findings across groups. (A) VEGF IHC showing increased angiogenic expression in treated groups, most notably in P3 (exosome + minoxidil). (B) β-catenin IHC demonstrating enhanced follicular signaling, highest in P2 and P3. (C) H&E staining illustrating dermal thickening in treated groups, with P3 showing the most pronounced restoration. (D) H&E staining revealing increased hair follicle density, greatest in P3. Right panels: Quantitative analyses (Mean ± SD) of VEGF, β-catenin, dermal thickness, and follicle density. Statistical significance: p-value < 0.05 (*), p-value < 0.01 (**), p-value < 0.001 (***).
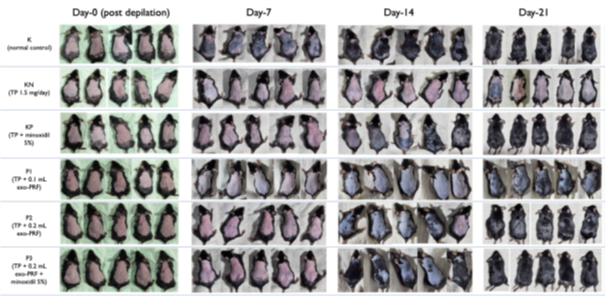
Figure 8 Representative clinical photographs of dorsal hair regrowth in C57BL/6 mice on days 0, 7, 14, and 21 after treatment.
Tumor Necrosis Factor-α (TNF-α)
Administration of i-PRF-derived exosomes significantly reduced serum TNF-α levels in the C57BL/6 mouse model of AGA, demonstrating their anti-inflammatory and regenerative properties. Exosomes derived from PRF are enriched with bioactive growth factors such as TGF-β, VEGF, PDGF, and IGF, as well as regulatory microRNAs, which collectively contribute to modulation of inflammatory signaling. Mechanistically, PRF-derived exosomes facilitate the polarization of macrophages from the pro-inflammatory M1 phenotype to the anti-inflammatory M2 phenotype in damaged tissues, thereby suppressing chronic inflammation. In addition, exosomal components attenuate the NF-κB signaling cascade, leading to downregulation of TNF-α and other pro-inflammatory cytokines within the hair follicle microenvironment. This shift helps restore the immune privilege and microvascular stability of hair follicles [16]. Moreover, PRF exosomes promote fibroblast proliferation, extracellular matrix remodeling, and angiogenesis, while simultaneously reducing oxidative stress and restoring cytokine balance. Specific microRNAs such as miR-146a and miR-21 have been shown to directly target the TNF-α/NF-κB axis, effectively dampening the inflammatory response associated with follicular miniaturization and premature transition to the catagen phase in AGA. Collectively, these findings indicate that PRF-derived exosomes exert potent anti-inflammatory effects by modulating both cellular and molecular pathways, thereby creating a favorable microenvironment for hair follicle regeneration [17-23].
Current evidence suggests that PRF-derived exosomes play a pivotal role in activating the Wnt/ β-catenin signaling pathway within anagen-phase hair follicle stem cells. Activation of this pathway leads to enhanced cellular proliferation and suppression of pro-inflammatory signaling cascades, including the TNF-α axis. In AGA, elevated dihydrotestosterone (DHT) levels trigger the release of pro-inflammatory cytokines and increase oxidative stress, ultimately promoting apoptosis and follicular regression. PRF-derived exosomes counteract these effects by delivering antioxidant enzymes and anti-apoptotic factors, thereby reducing TNF-α expression and supporting follicular cell survival and regeneration [17-23]. Shimizu et al. [18] reported that platelet-derived exosome therapy in AGA mice significantly suppressed TNF-α levels and increased the proportion of anagen follicles, mediated by microRNA-driven NF-κB inhibition [18]. Similarly, Yang et al. [23] demonstrated that PRP-derived exosomes enhanced hair regeneration through the SIRT1/FoxO3a-mediated oxidative stress suppression pathway, indirectly contributing to TNF-α downregulation and improved follicular health. Collectively, these findings support the hypothesis that PRF-derived exosomes mitigate DHT-induced follicular inflammation and apoptosis through integrated modulation of Wnt/ β-catenin, NF-κB, and SIRT1/FoxO3a pathways, thereby facilitating anagen maintenance and hair follicle regeneration.
Interleukin-6
The anti-inflammatory and regenerative mechanisms mediated by i-PRF-derived exosomes in suppressing serum IL-6 levels are consistent with their effects on TNF-α regulation in the AGA mouse model. i-PRF-derived exosomes are enriched with regulatory microRNAs (including miR-146a and miR-1246) and protein mediators that inhibit IL-6 transcription and translation within dermal papilla cells and immune effector cells. These exosomal microRNAs suppress the NF-κB signaling pathway, resulting in downregulation of IL-6 and attenuation of inflammatory signaling in the perifollicular microenvironment. Furthermore, i-PRF-derived exosomes induce a phenotypic switch of macrophages from the pro-inflammatory M1 to the anti-inflammatory M2 subtype, which contributes to the reduction of IL-6 secretion around hair follicles. Concomitantly, the upregulation of regenerative growth factors such as IGF-1, VEGF, and FGF-7 promotes angiogenesis and follicular proliferation, counteracting the catagen-inducing effects of IL-6 [22,24-26]. The observed reduction of TNF-α following i-PRF exosome administration further contributes to secondary downregulation of IL-6, restoring follicular homeostasis, preventing dermal papilla cell apoptosis, and facilitating the transition to the anagen phase. In parallel, exosomal bioactive contents were shown to suppress androgen receptor (AR) expression, thereby reducing androgen-mediated upregulation of TNF-α and IL-6. Activation of the Wnt/β-catenin reparative pathway by PRF exosomes enhances dermal papilla cell proliferation and microvascularization, which collectively diminish pro-inflammatory signals and promote follicular regeneration [22,24-26]. Ellistasari et al. [27] demonstrated that administration of PRP-derived exosomes in an AGA mouse model markedly reduced IL-6 levels compared to untreated AGA controls. Similarly, Oh et al. [24] reported that exosome treatment suppressed mRNA expression of AR, TGF-β, and IL-6 upregulated by testosterone through reactivation of the AR-related Wnt/β-catenin pathway and enhanced GSK3β phosphorylation.
Malondialdehyde
Administration of i-PRF-derived exosomes significantly reduced serum malondialdehyde (MDA) levels in C57BL/6 mice with AGA, primarily by mitigating oxidative stress and lipid peroxidation pathways. i-PRF exosomes contain abundant growth factors—including VEGF, IGF-1, and bFGF—as well as regulatory microRNAs (miR-21 and miR-146a) that stimulate dermal papilla cells and keratinocytes to enhance the production of endogenous antioxidant enzymes such as superoxide dismutase (SOD), catalase, and glutathione peroxidase (GPx). The upregulation of these enzymes enhances reactive oxygen species (ROS) neutralization, limiting membrane lipid peroxidation and consequently reducing MDA accumulation. Exosomal microRNAs and growth factors also activate the Nrf2 signaling pathway, a master regulator of cellular antioxidant defense. Upon activation, Nrf2 translocates to the nucleus and promotes the transcription of genes encoding detoxifying and ROS-scavenging enzymes, leading to decreased MDA levels in serum and local follicular tissues. Furthermore, i-PRF exosomes act as dual-function modulators, simultaneously exerting anti-inflammatory and antioxidant effects. By downregulating inflammatory cytokines and inhibiting ROS-producing enzymes such as NADPH oxidase, they indirectly suppress lipid peroxidation and protect cellular membranes from oxidative injury [23,24,26,28]. In addition, VEGF, IGF-1, and FGF-2 within PRF exosomes stimulate tissue regeneration and cellular repair processes, safeguarding follicular cells against oxidative damage and apoptosis, both of which are associated with elevated MDA levels. Overall, i-PRF exosome therapy restores redox homeostasis within the hair follicle microenvironment, preventing chronic oxidative stress that contributes to follicular miniaturization and hair loss in AGA [23,24,26,28].
Jiao et al. [29] demonstrated in a murine sepsis model that platelet administration decreased MDA levels while increasing SOD and GSH activity through modulation of the AKT/mTOR signaling pathways, known to regulate inflammation, oxidative stress, apoptosis, and autophagy. Similarly, Yang et al. [23] reported that platelet-derived exosomes act as potent antioxidants by activating the SIRT1/FoxO3a pathway, wherein AMPK-mediated activation of SIRT1 leads to deacetylation and activation of FoxO3a pathway, wherein AMPK-mediated activation of SIRT1 leads to deacetylation and activation of FoxO3a during oxidative stress. Moreover, Li et al. [30] summarized that platelets exert antioxidant effects through Nrf2 activation, miRNA-mediated ROS suppression, inhibition of TLR4/NF-κB/STAT3/Akt inflammatory cascades, elevation of SOD3 levels, and restoration of mitochondrial function.
Caspase-3
Administration of i-PRF-derived exosomes in the AGA mouse model significantly suppressed serum Caspase-3 levels, indicating potent anti-apoptotic and regenerative activity. The growth factors and microRNAs contained within PRF exosomes activate the PI3K/Akt signaling cascade in dermal papilla and follicular cells, promoting cell survival and proliferation [20,23,31-33]. Among these, VEGF not only enhances angiogenesis and improves follicular perfusion but also exerts direct cytoprotective effects on follicular keratinocytes through the same PI3K/Akt-mediated pathway. Exosomes also deliver microRNAs and antioxidant molecules that mitigate ROS generation and oxidative mitochondrial stress in follicular cells. This prevents loss of mitochondrial membrane potential and subsequent cytochrome-c release, critical upstream events in the caspase-9/caspase-3 apoptotic cascade. Furthermore, platelet-derived exosomes have been shown to attenuate endoplasmic reticulum (ER) stress by inhibiting PERK/CHOP signaling, thereby suppressing both intrinsic and extrinsic apoptotic pathways [20,23,31-33].
Wang et al. [19] reported that AGA model mice treated with combined exosome and minoxidil therapy exhibited significantly lower Caspase-3 expression, which correlated with increased VEGF and Akt activation. Similarly, Liu et al. [34] demonstrated in an inflammatory cartilage defect model that platelet-derived exosomes inhibited apoptosis through PI3K/Akt pathway regulation, accompanied by downregulation of MMP-3, TNF-α, IL1-β, and IL-6.
Vascular endothelial growth factor
PRF-derived exosomes are enriched with VEGF, PDGF, IGF-1, bFGF, and miRNAs. Following intradermal administration, they are rapidly internalized by dermal papilla and endothelial cells, delivering potent pro-angiogenic cues that enhance local VEGF transcription and translation. These effects are mediated in part through activation of the PI3K/Akt/mTOR signaling axis, which induces nuclear translocation of hypoxia-inducible factor-1α (HIF-1α). HIF-1α binds to the VEGF promoter region, markedly upregulating VEGF gene expression and counteracting androgen-induced suppression of follicular angiogenesis [19,23,26,30].
Growth factors such as PDGF and FGF within PRF exosomes further stimulate mesenchymal and consequent rise in VEGF expression aligns with the anti-apoptotic and anti-inflammatory actions of PRF exosomes—specifically, suppression of Caspase-3, Il-6, and TNF-α—which fosters a pro-regenerative microenvironment that sustains angiogenic signaling.
i-PRF exosomes can also activate the IL-6/STAT3 pathway, leading to STAT3 phosphorylation and nuclear translocation, which upregulates VEGF and other pro-angiogenic genes, thereby reinforcing hair-follicle vascularization and cycling. When combined with 5% minoxidil, PRF exosomes further augment VEGF levels; minoxidil independently elevates VEGF via potassium-channel opening and membrane hyperpolarization, while exosomes amplify these molecular signals, producing a synergistic, super-additive effect on angiogenesis and follicular perfusion [19,23,26,30].
Shams et al. [35] demonstrated that platelet-derived exosomes at concentrations of 25, 50, and 100 µg/mL enhanced dermal papilla cell migration and secretion of VEGF and PDGF. Similarly, Sun et al. (2019) reported that platelet-derived exosome administration in coronary artery syndrome (ACS) patients improved endothelial repair and significantly increased VEGF, bFGF, and TGF-β1 levels —an effect associated with miR-126 upregulation, a key driver of angiogenesis [36]. Furthermore, Asrianti et al. [37] observed elevated VEGF expression following platelet-derived exosome treatment in pulp-tissue regeneration, corroborating the broad angiogenic potential of platelet-origin exosomes.
β-Catenin
β-catenin is a pivotal intracellular effector of the canonical Wnt signaling pathway, which plays a central role in hair follicle morphogenesis, cycling, and regeneration. Activation of Wnt/β-catenin signaling stimulates hair follicle stem cell proliferation, triggers anagen initiation, and supports new follicle formation. The observed upregulation of β-catenin following i-PRF-derived exosome administration is attributed to growth factors and microRNAs within the exosomal cargo that activate Wnt signaling, leading to β-catenin accumulation and nuclear translocation in hair follicle progenitor and dermal papilla cells. Once in the nucleus, β-catenin associates with TCF/LEF transcription factors to promote the expression of genes governing follicular proliferation and anagen re-entry. In contrast, androgen (DHT/testosterone) exposure in the AGA model suppresses Wnt/β-catenin signaling, resulting in follicular miniaturization and shortening of the anagen phase. i-PRF exosome therapy is hypothesized to counteract androgen-induced suppression and restore Wnt/β-catenin signaling, thereby re-establishing follicular regenerative activity [19,20,23,26].
Moreover, i-PRF exosomes influence the PI3K/Akt/GSK3β signaling axis, another critical regulatory pathway of β-catenin stability. Platelet-derived exosomes can activate PI3K/Akt in dermal papilla and follicular cells, where Akt phosphorylation inactivates GSK3β, a kinase responsible for β-catenin phosphorylation and degradation. When GSK3β is inhibited, β-catenin escapes proteasomal degradation, accumulates in the cytoplasm, and translocates to the nucleus to induce pro-survival and proliferative gene expression. Chronic oxidative stress and inflammation are known to downregulate Wnt/β-catenin signaling through NF-κB activation and related inhibitory cascades. PRF exosome therapy mitigates these effects by reducing oxidative mediators and inflammatory cytokines, thereby indirectly stabilizing Wnt/β-catenin signaling. Additionally, growth factors within PRF exosomes can activate MAPK/ERK signaling, which regulates β-catenin via phosphorylation and transcriptional co-activation, further promoting follicular proliferation and tissue regeneration [19,20,23,26].
Lu et al. [26] demonstrated that platelet-derived exosome administration activated Wnt/β-catenin signaling while concurrently suppressing SFRP1 expression, a known Wnt inhibitor. Liu et al. [38] similarly reported that platelet-derived exosomes suppressed IL-1β and TNF-α, concomitant with increased β-catenin expression in osteoarthritis models. Furthermore, Wang et al. [39] described a bidirectional relationship between exosomes and the Wnt/β-catenin pathway, where Wnt agonist administration increased exosome release by 1.5-fold in a diabetic wound-healing model. While the current study did not investigate the SIRT1/FoxO3a, PI3K/Akt, or Wnt pathway components in detail, the observed β-catenin upregulation and associated regenerative effects are consistent with previous mechanistic evidence from platelet-derived exosome studies, suggesting a shared molecular framework underlying hair follicle regeneration and repair.
Dermal thickness
i-PRF-derived exosomes are rich in bioactive proteins, lipids, growth factors—including VEGF, TGF-β, PDGF, and IGF-1—and regulatory microRNAs that collectively modulate cellular function and the tissue microenvironment. Platelet-rich fibrin, as an autologous biomaterial, not only serves as a platelet and fibrin concentrate but also acts as a reservoir of exosomes that promote skin regeneration and wound healing. The growth factor-rich exosomal fraction of PRF stimulates the synthesis of extracellular matrix (ECM) components—particularly collagen types I and III, elastin, and fibronectin—which are essential for dermal remodeling and structural restoration. The pro-angiogenic content of i-PRF exosomes enhances oxygen and nutrient delivery, supporting tissue repair and neovascularization. The miRNA cargo of i-PRF exosomes plays a crucial regulatory role in maintaining dermal homeostasis and regeneration. Specifically, miR-21 and miR-146a enhance fibroblast proliferation and inhibit apoptosis during the healing phase, whereas miR-29 regulates collagen synthesis and deposition, influencing dermal architecture and tensile strength. In addition, miR-223 modulates inflammatory signaling, reduces fibrotic response, and promotes regenerative pathway activation [18,26,33,40-44].
The anti-inflammatory milieu created by PRF exosomes further facilitates optimal tissue regeneration. Their immunomodulatory effects efficiently shift the wound-healing process from the inflammatory phase to the proliferative and remodelling phases. PRF also provides a natural fibrin scaffold that enhances local exosome retention and allows sustained release of growth factors over time. This prolonged bioavailability ensures continuous stimulation of dermal fibroblasts, endothelial cells, and skin stem/progenitor cells, preventing rapid clearance and promoting sustained regenerative outcomes, as evidenced by the increase in dermal thickness observed in treated groups [18,26,33,40-44].
Mechanistically, i-PRF exosomes increase dermal thickness through activation of the Wnt/β-catenin pathway and upregulation of the Lef-1 transcription factor, as demonstrated by Lu et al. [26]. This activation is accompanied by suppression of the Wnt antagonist SFRP1 and induction of cell-cycle progression in dermal fibroblasts. Furthermore, PRF exosomes carry glycolytic enzymes such as PKM2 and GLUT1, which enhance glycolytic metabolism and ATP production, supporting fibroblast proliferation, migration, and ECM synthesis. These metabolic and molecular mechanisms help prevent fibroblast senescence, maintain a youthful, proliferative fibroblast population, and sustain continuous ECM regeneration [18,33,40-44].
i-PRF exosomes also activate local skin-derived stem and progenitor cells by delivering pro-survival and pro-differentiation signals, leading to the formation of new fibroblasts and ECM deposition that enhance dermal volume and integrity. In the context of AGA, these effects restore stromal support and functional fibroblast activity, counteracting tissue thinning and follicular degeneration [18,26,33,40-44].
He et al. [45] reported that platelet-derived exosome therapy improved skin healing in diabetic ulcers by accelerating macrophage polarization toward the M2 phenotype, highlighting their anti-inflammatory and reparative potential. A systemic review by Prasai et al. [46] corroborated these findings, attributing skin improvement after exosome therapy to miRNA-mediated regenerative mechanisms. Likewise, Guo et al. [47] demonstrated that platelet-derived exosomes enhanced skin repair and re-epithelialization in chronic wounds through activation of the YAP (Yes-associated protein) signaling pathway.
Collectively, these findings indicate that PRF exosomes promote dermal thickening through a multifactorial mechanism involving ECM remodeling, metabolic activation, Wnt/β-catenin signaling, and immunomodulation, leading to improved dermal architecture and structural resilience in AGA.
Hair follicle density
The increase in hair follicle density observed following i-PRF-derived exosome administration represents an integrated outcome of the exosomes' anti-inflammatory, antioxidant, anti-apoptotic, and pro-regenerative mechanisms. PRF exosomes are enriched with growth factors and regulatory microRNAs capable of activating the Wnt/β-catenin signaling pathway in follicular stem and dermal papilla cells. Nuclear translocation of β-catenin subsequently induces proliferative gene expression, stimulating hair-cycle re-entry into the anagen phase, and promotes new follicle formation, thereby enhancing hair density [19,23,26,33].
Exosomal VEGF, PDGF, and FGF also play critical roles by promoting angiogenesis and vascular remodeling, ensuring optimal oxygen and nutrient delivery to regenerating follicles, and supporting cell survival and proliferation. In parallel, activation of the PI3K/Akt pathway enhances anti-apoptotic signaling and suppresses pro-apoptotic markers, maintaining follicular cell viability within the androgen-deprived scalp microenvironment. The ability of PRF exosomes to modulate inflammation and oxidative stress further contributes to the creation of a pro-reparative microenvironment that facilitates follicular regeneration and restoration of hair density [19,23,26,33].
Experimental evidence supports these mechanistic pathways. Shams et al. [35] demonstrated that platelet-derived exosomes at a dose of 100 μg/mL increased dermal papilla cell proliferation, migration, and the expression of alkaline phosphatase (ALP), versican, and α-SMA, which are crucial markers for hair induction and follicular morphogenesis. Penha et al. [48] similarly reported increased hair follicle density following exosome administration, primarily through angiogenic stimulation that enhanced follicular nutrient and blood supply. In clinical study, Ersan et al. [49] evaluated 30 male patients with Norwood-Hamilton grade III-VI AGA and found a significant increase in hair density at weeks 4 and 12 post-exosome therapy (p-value < 0.05), with no adverse effects. Hair density improved from 149.7 ± 13.7 hairs/cm2 at baseline to 153.6 ± 16.8 hairs/cm2 at week 4 (p-value = 0.043) and 157 ± 18.3 hairs/cm2 at week 12 (p-value = 0.002) [49]. Mulianto et al. [50] further reported that platelet-derived exosome treatment achieved higher follicular density compared to minoxidil (68.61 ± 7.64 vs. 40.14 ± 11.60; p-value = 0.007). When combined with minoxidil, i-PRF exosomes amplify regenerative signaling. Minoxidil promotes VEGF upregulation through potassium-channel opening and hyperpolarization, while PRF exosomes potentiate these angiogenic and survival pathways, resulting in synergistic enhancement of hair regrowth and follicle preservation. This additive interaction maximizes follicle retention even under persistent androgenic stress [19,23,26,33].
Previous studies using PRP-derived exosomes have consistently demonstrated hair-growth-promoting effects via Wnt/β-catenin activation and dermal papilla stimulation. Lu et al. [26] showed that PRP-derived exosomes upregulated β-catenin expression and enhanced dermal papilla proliferation both in vivo and in ex vivo hair follicle organ cultures. Yang et al. [23] further confirmed that PRP exosomes promoted follicular regeneration and VEGF upregulation in testosterone-induced alopecia mice. The present study extend these findings by utilizing PRF as the exosome source. Unlike PRP, PRF is a second-generation platelet concentrate that incorporates a natural fibrin matrix and releases growth factors gradually without the use of anticoagulants. The compositional difference likely contributes to distinct exosomal cargo profiles and potentially more sustained biological activity. Thus, while our findings align with prior PRP-exosome studies demonstrating anti-inflammatory and pro-regenerative effects, they also suggest that PRF-derived exosomes represent a novel, autologous, and anticoagulant-free approach with enhanced therapeutic potential for hair restoration.
This study has several limitations. The parameters of β-catenin, VEGF, dermal thickness, and hair follicle density were evaluated only at the post-treatment stage, so the longitudinal dynamics of these markers remain unknown. Serum biomarkers were analyzed by ELISA, whereas tissue biomarkers were assessed semi-quantitatively by IHC; however, no molecular validation (e.g., qPCR or Western blot) of key signaling targets such as AR, DKK1, and Wnt/β-catenin was performed. Consequently, early transient pathway activation peaks may have been missed. Future investigation should include larger cohorts, extended treatment duration, and molecular confirmation of pathway activity (e.g., AR, DKK1, Wnt targets) to validate these findings. Because this was a preclinical study, aspects such as safety, optimal dosing intervals, and long-term effects have not yet been evaluated, warranting controlled clinical trials before therapeutic translation.
Conclusions
This study demonstrates that i-PRF-derived exosomes exert multifaceted therapeutic effects in a C57BL/6 mice model of AGA through integrated anti-inflammatory, antioxidant, anti-apoptotic, and pro-regenerative mechanisms. Administration of PRF exosomes significantly reduced serum TNF-α, IL-6, MDA, and Caspase-3 levels while upregulating VEGF and β-catenin expression, increasing dermal thickness, and improving hair follicle density. The combination of PRF exosomes and 5% minoxidil yielded the most consistent enhancement across the parameters, indicating a synergistic effect that promotes follicular regeneration, angiogenesis, and hair-cycle reactivation.
Mechanistically, the observed benefits appear to involve activation of PI3K/Akt, Wnt/β-catenin, and Nrf2 pathways, modulation of oxidative and inflammatory cascades, and stimulation of ECM synthesis and fibroblast proliferation within the dermal compartment. These results collectively support the potential of PRF-derived exosomes as a biologically active and autologous nanotherapeutic for hair restoration.
Given the simplicity of PRF preparation, its anticoagulant-free composition, and the sustained release of growth factors, PRF represents a practical and clinically feasible exosome source for regenerative dermatology applications. Further dose optimization, molecular validation, and controlled clinical trials are warranted to confirm efficacy and safety in human subjects. The current findings provide a preclinical foundation for future translational studies, advancing PRF-derived exosomes toward evidence-based, regenerative treatment protocols for AGA.
References
[1] S Ntshingila, O Oputu, AT Arowolo and NP Khumalo. Androgenetic alopecia: An update. Journal of the American Academy of Dermatology International 2023; 13, 150-158.
[2] D Sen, O Ezemma, KJ Kelley, E Stratton and M Senna. Androgenetic alopecia: Therapy update. Drugs 2023; 83(8), 701-715.
[3] P Pudasaini, GC Sagar, E Sujakhu, MB Pradhan, B Thapa and E Shrestha, R Bhatta and Sushil Paude. Prevalence and clinical characteristics of androgenetic alopecia in Nepalese patients: A cross-sectional observational study. Civil Medical Journal 2023; 1(1), 29-34.
[4] L Legiawati, LS Suseno, IBS Sitohang and AI Pratama. Hair disorder in Dr. Cipto Mangunkusumo cosmetic dermatology and venereology outpatient clinic, Jakarta: A socio-demographic and clinical evaluation. Dermatology Reports 2022; 14(3), 9341.
[5] L Fus-Mazurkiewicz, J Sak, M Nowiński, D Król and W Mazurkiewicz. Review of current knowledge and management of androgenetic alopecia. Polish Journal of Applied Sciences 2023; 9(3), 32-35.
[6] MS Nestor, G Ablon, A Gade, H Han and DL Fischer. Treatment options for androgenetic alopecia: Efficacy, side effects, compliance, financial considerations, and ethics. Journal of Cosmetic Dermatology 2021; 20(12), 3759-3781.
[7] A Asilian, A Farmani and M Saber. Clinical efficacy and safety of low-dose oral minoxidil versus topical solution in androgenetic alopecia: A randomized controlled trial. Journal of Cosmetic Dermatology 2024; 23(3), 949-957.
[8] S Sattur, A Talathi, G Shetty, S Arsiwala, R Pereira and D Dhoot. Comparative clinical study evaluating the efficacy and safety of topical 5% cetosomal minoxidil and alcohol-based minoxidil in Indian men. Cureus Journal of Medical Science 2023; 15(10), e46568.
[9] AG Jamjoom. From healing to regeneration: A comprehensive review of the efficacy of platelet-rich fibrin in periodontal plastic surgery procedures. Cureus Journal of Medical Science 2024; 16(9), e69287.
[10] RJ Miron, J Chai, P Zhang, Y Li, Y Wang and CFDAB Mourão, A Sculean, MF Kobayashi and Y Zhang. A novel method for harvesting concentrated platelet-rich fibrin (C-PRF) with a 10-fold increase in platelet and leukocyte yields. Clinical Oral Investigations 2020; 24(8), 2819-2828.
[11] F Du, J Li, S Zhang, X Zeng, J Nie and Z Li. Oxidative stress in hair follicle development and hair growth: Signalling pathways, intervening mechanisms and potential of natural antioxidants. Journal of Cellular and Molecular Medicine 2024; 28(12), e18486.
[12] SA Mohale, PV Thakare, SS Gaurkar, G Bharadia and S Acharya. Effectiveness of injectable platelet-rich fibrin therapy in alopecia and facial rejuvenation: A systematic review. Cureus Journal of Medical Science 2024; 16(6), e62198.
[13] B Shashank and M Bhushan. Injectable platelet-rich fibrin (PRF): The newest biomaterial and its use in various dermatological conditions in our practice: A case series. Journal of Cosmetic Dermatology 2021; 20(5), 1421-1426.
[14] S Shrestha, R Pathak, GC Jagat and J Donovan. Platelet-rich fibrin: A review of its role as a new treatment in androgenetic alopecia. Journal of Cosmetic Dermatology 2024; 23(8), 2524-2530.
[15] X Chen, B Liu, Y Li, L Han, X Tang and W Deng, W Lai and M Wan. Dihydrotestosterone regulates hair growth through the Wnt/β-Catenin pathway in C57BL/6 mice and in vitro organ culture. Frontiers in Pharmacology 2019; 10, 1528.
[16] DI Jang, AH Lee, HY Shin, HR Song, JH Park and TB Kang, SR Lee and SH Yang. The role of tumor necrosis factor alpha (TNF-α) in autoimmune disease and current TNF-α inhibitors in therapeutics. International Journal of Molecular Sciences 2021; 22(5), 2719.
[17] Y Li, G Wang, Q Wang, Y Zhang, L Cui and X Huang. Exosomes secreted from adipose-derived stem cells are a potential treatment agent for immune-mediated alopecia. Journal of Immunology Research 2022; 2022, 7471246.
[18] Y Shimizu, EH Ntege, H Sunami and Y Inoue. Regenerative medicine strategies for hair growth and regeneration: A narrative review of literature. Regenerative Therapy 2022; 21, 527-539.
[19] G Wang, Z Wang, J Zhang, Y Shen, X Hou and L Su, W Chen, J Chen, X Guo and H Song. Treatment of androgenetic alopecia by exosomes secreted from hair papilla cells and the intervention effect of lactotransferrin. Journal of Cosmetic Dermatology 2023; 22(11), 2996-3007.
[20] M Cheng, C Ma, HD Chen, Y Wu and XG Xu. The roles of exosomes in regulating hair follicle growth. Clinical, Cosmetic and Investigational Dermatology 2024; 17, 1603-1612.
[21] JY Zhang, XN Xiang, X Yu, Y Liu, HY Jiang and JL Peng, CQ He and HC He. Mechanisms and applications of the regenerative capacity of platelets-based therapy in knee osteoarthritis. Biomedicine and Pharmacotherapy 2024; 178, 117226.
[22] Y Liu, Y Liu, J Zhao, T Deng, Y Ben and R Lu, X Zhou, R Yan, X Chen, JV Zhang and G Zhou. Subcutaneous injection of genetically engineered exosomes for androgenic alopecia treatment. Frontiers in Bioengineering and Biotechnology 2025; 13, 1614090.
[23] X Yang, K Wang, G Liu, Y Chen, Y Wang and Y Cheng, Y Wang, Y Cheng and C Zhang. Exosomes derived from platelet-rich plasma promote hair regeneration by regulating the SIRT1/FoxO3a pathway to alleviate oxidative stress. Regenerative Therapy 2025; 30, 710-718.
[24] HG Oh, M Jung, SY Jeong, J Kim, S Han and H Kim, S Lee, Y Lee, H You, S Park, EA Kim, TM Kim and S Kim. Improvement of androgenic alopecia by extracellular vesicles secreted from hyaluronic acid-stimulated induced mesenchymal stem cells. Stem Cell Research and Therapy 2024; 15(1), 287.
[25] J Zheng, B Yang, S Liu, Z Xu, Z Ding and M Mo. Applications of exosomal miRNAs from mesenchymal stem cells as skin boosters. Biomolecules 2024; 14(4), 459.
[26] C Lu, Y Ding, R Zhang, Y Du, L Bi, M Zhao, C Wang, Q Wu, H Jing and W Fan. Platelet-rich plasma-derived exosomes stimulate hair follicle growth through activation of the Wnt/β-Catenin signaling pathway. Regenerative Therapy 2025; 29, 435-446.
[27] EY Ellistasari, S Widhiati, I Julianto, E Rosmarwati, PW Monica and A Yasmin. Reduction of interleukin-6 after PRP-Exosomes treatment in a mouse model of androgenetic alopecia. International Summit on Science, Technology and Humanity 2024; 1122-1129.
[28] F Norouzi, S Aghajani, N Vosoughi, S Sharif, K Ghahremanzadeh and Z Mokhtari and J Verdi. Exosomes derived stem cells as a modern therapeutic approach for skin rejuvenation and hair regrowth. Regenerative Therapy 2024; 26, 1124-1137.
[29] Y Jiao, Q Zhang, J Zhang, Y Zha, J Wang and Y Li and S Zhang. Platelet-rich plasma ameliorates lipopolysaccharide-induced cardiac injury by inflammation and ferroptosis regulation. Frontiers in Pharmacology 2022; 13, 1026641.
[30] Y Li, Z Zhu, S Li, X Xie, L Qin, Q Zhang, Y Yang, T Wang and Y Zhang. Exosomes: Compositions, biogenesis, and mechanisms in diabetic wound healing. Journal of Nanobiotechnology 2024; 22(1), 398.
[31] A Liang, Y Fang, L Ye, J Meng, X Wang and J Chen and X Xu. Signaling pathways in hair aging. Frontiers in Cell and Developmental Biology 2023; 11, 1278278.
[32] G Wang, Z Wang, J Zhang, Y Shen, X Hou, L Su, W Chen, J Chen, X Guo and H Song. Treatment of androgenetic alopecia by exosomes secreted from hair papilla cells and the intervention effect of lactotransferrin. Journal of Cosmetic Dermatology 2023; 22(11), 2996-3007.
[33] N Poddar, A Aratikatla and A Gupta. Therapeutic potential of stem cell-derived exosomes in hair regeneration: A systematic review. World Journal of Stem Cells 2025; 17(7), 108519.
[34] X Liu, R Chen, G Cui, R Feng and K Liu. Exosomes derived from platelet-rich plasma present a novel potential in repairing knee articular cartilage defect combined with cyclic peptide-modified β-TCP scaffold. Journal of Orthopaedic Surgery and Research 2024; 19(1), 718.
[35] SF Shams, M Javan and F Shahriyari. The role of platelet-derived exosomes in regenerative medicine. Nanomedicine Journal 2025; 12(1), 1-14.
[36] Y Sun, XL Liu, D Zhang, F Liu, YJ Cheng, Y Ma, YJ Zhou and YX Zhao. Platelet-derived exosomes affect the proliferation and migration of human umbilical vein endothelial cells via miR-126. Current Vascular Pharmacology 2019; 17(4), 379-387.
[37] D Asrianti. 2021, The potential ability of platelet-rich plasma (PRP) exosome in inducing dental pulp regeneration: An in-vitro analysis. Ph. D. Dissertation. Universitas Indonesia, Jakarta, Indonesia.
[38] X Liu, L Wang, C Ma, G Wang, Y Zhang and S Sun. Exosomes derived from platelet-rich plasma present a novel potential in alleviating knee osteoarthritis by promoting proliferation and inhibiting apoptosis of chondrocyte via Wnt/β-Catenin signaling pathway. Journal of Orthopaedic Surgery and Research 2019; 14(1), 470.
[39] L Wang, J Chen, J Song, Y Xiang, M Yang and L Xia, J Yang, X Hou, L Chen and L Wang. Activation of the Wnt/β-Catenin signalling pathway enhances exosome production by hucMSCs and improves their capability to promote diabetic wound healing. Journal of Nanobiotechnology 2024; 22(1), 373.
[40] RC Mehta, K Kadoya, P Maitra, LT Goberdhan, ET Makino, GK Naughton and MP Goldman. Fibroblast‐derived growth factors and exosomes as cosmeceuticals. Dermatology Review 2023; 4(6), 250-259.
[41] AY Park, J Lee, Y Jang, YJ Kim, JM Lee, SY Kim, BJ Kim and KH Yoo. Exosomes derived from human dermal fibroblasts protect against UVB-induced skin photoaging. International Journal of Molecular Medicine 2023; 52(6), 120.
[42] C Liang, Y Yi, J Li, S Aghayants, X Chen, W Cao and Q Zhang. Unveiling exosomes in combating skin aging: Insights into resources, mechanisms and challenges. Stem Cell Research and Therapy 2025; 16(1), 474.
[43] S Schaffer, L Tehrani, B Koechle, P Chandramohan, B Hilburn, KC Aoki and RJ Jacobs. A scoping review of exosome delivery applications in hair loss. Cureus Journal of Medical Science 2025; 17(3), e81152.
[44] X Zhang, W Wang, W Fan, Y Zhai, J Fu and J Cai, D Zhao and F Sun. Alteration of hair growth in mice by circulating exosomes from Xinji fine-wool and small-tailed Han sheep. Frontiers in Animal Science 2025; 6, 1562392.
[45] L He, N Zhao, X Chen, W Zhang, K Lv and Y Xu. Platelet-rich plasma-derived exosomes accelerate the healing of diabetic foot ulcers by promoting macrophage polarization toward the M2 phenotype. Clinical and Experimental Medicine 2025; 25(1), 163.
[46] A Prasai, JW Jay, D Jupiter, SE Wolf and AE Ayadi. Role of exosomes in dermal wound healing: A systematic review. Journal of Investigative Dermatology 2022; 142(3A), 662-670.
[47] SC Guo, SC Tao, WJ Yin, X Qi, T Yuan and CQ Zhang. Exosomes derived from platelet-rich plasma promote the re-epithelization of chronic cutaneous wounds via activation of YAP in a diabetic rat model. Theranostics 2017; 7(1), 81-96.
[48] FA Penha, GA Penha and HMC Acerbi. A comprehensive literature review: The potential of exosomes in hair growth and transplantation. International Society of Hair Restoration Surgery 2025; 35(3), 73-84.
[49] M Ersan, E Ozer, O Akin, PN Tasli and F Sahin. Effectiveness of exosome treatment in androgenetic alopecia: Outcomes of a prospective study. Aesthetic Plastic Surgery 2024; 48(21), 4262-4271.
[50] N Mulianto, I Julianto, ME Irawanto, N Dharmawan, PW Monica and T Oktaviani. Comparative study on therapeutic efficacy of minoxidil and platelet-rich plasma exosome in androgenetic alopecia mice models. International Journal of Applied Pharmaceutics 2025; 15, 89-92.