
Trends
Sci.
2026;
23(7):
13160
Platelet Concentrates in Regenerative Medicine: A Comparative Review of Platelet-Rich Plasma, Platelet-Rich Fibrin, and Concentrated Growth Factors
Nguyen Cao Nguyen1,2 and Phat Duc Huynh1,3,*
1Viet Nam National University, Ho Chi Minh City, Viet Nam
2Research Center of Infectious Diseases, International University, Ho Chi Minh City, Viet Nam
3VNUHCM-US Stem Cell Institute, University of Science Ho Chi Minh City, Viet Nam
(*Corresponding author’s e-mail: [email protected])
Received: 25 December 2025, Revised: 16 January 2026, Accepted: 26 January 2026, Published: 5 March 2026
Abstract
Platelet concentrates such as platelet-rich plasma (PRP), platelet-rich fibrin (PRF), and concentrated growth factors (CGF) are the Autologous Biomaterials of interest in regenerative medicine for their high growth factor content and simplicity of preparation. These products, which are obtained from peripheral blood by differential centrifugation protocols, represent a low-cost, biocompatible solution to tissue repair and regeneration for an extensive variety of clinical specialties from orthopedics, dentistry, and dermatology. This review contrasts and compares PRP, PRF, and CGF based on preparation protocols, cellular and molecular contents, growth factor release kinetics, and clinical applications. PRP, the first-generation concentrate, contains high platelet density but requires the addition of exogenous anticoagulants. PRF is a second-generation product with a fibrin matrix to provide slow growth factor release without additives. CGF, the newest development, uses variable centrifugation speeds to produce a denser fibrin matrix with a potentially higher regenerative potential. Current evidence suggests that the variations in preparation influence not just the biological activity, but also the clinical efficacy of each concentrate. While the most common, PRP is being progressively supplanted by PRF and CGF due to the fact that they are simple to use and additive-free. Nevertheless, standardization guidelines and comparative clinical studies are lacking, and therefore the optimization of treatment results is also challenging. In general, PRP, PRF, and CGF all possess distinct strengths in regenerative medicine. Additional understanding of their biologic profiles and handling during clinical practice is needed to individualize them in patient-tailored therapy and also to develop evidence-based guidelines for future application.
Keywords: Platelet-Rich Plasma (PRP), Platelet-Rich Fibrin (PRF), Concentrated Growth Factors (CGF), Platelet concentrates, Regenerative medicine, Fibrin matrix, Autologous therapy
Introduction
One of the main objectives of contemporary tissue engineering and regenerative medicine is the restoration of damaged or dysfunctional tissues resulting from degenerative disease, trauma, or reconstructive surgery [1,2]. The main drawback with the conventional therapies - including grafting, synthetic biomaterials, and exogenous growth factor delivery systems - are their high cost, risk of rejection, and relatively poor compatibility with the host tissues [3,4]. It is with this background that the need for regenerative medicine arose.
Growth factors play a central role in the coordination of tissue regeneration, including fundamental cell activities like cell division, migration, angiogenesis, and differentiation. Of the natural sources of growth factors, platelets stand out as a rich source of biological molecules like transforming growth factor-β (TGF-β), platelet-derived growth factor (PDGF), and vascular endothelial growth factor (VEGF) [5]. It is because of this reason that autologous platelet-derived biomaterials have emerged with popularity as regenerative materials owing to their biocompatibility, safety, and ease of preparation. Platelet concentrates are isolated from peripheral blood by differential centrifugation [6]. They possess a unique combination of a high concentration of growth factors and a fibrin network that regulates the spatial and temporal release of bioactive signals at the wound site. Therapeutic efficacy depends not only on growth factor concentration but also on fibrin architecture and release kinetics [7]. This has led the field of tissue engineering to focus on optimizing the composition based on platelets.
Historically, Max S first described the components of white blood cells in 1865 and identified small anucleate bodies in the blood, which were subsequently recognized as platelets [8]. Nearly 2 decades later, Bizzozero [9] built upon this work and provided an accurate account of the platelet role, emphasizing their central role in the process of blood coagulation. But it was not until 1969 that a method of storing platelets with maintenance of their hemostatic function was accomplished, a major achievement in transfusion medicine and clinical application of platelets [10]. After that, the development of platelet concentrates has developed over the past few decades, keeping pace with the development of regenerative medicine and biomaterials. PRP was the first generation of such products, which were initially launched in the 1970s [11] and more popularly in the 1990s [12] with its platelet and associated growth factor concentration. It is widely used in numerous areas such as orthopedics, sports medicine, and dentistry. But PRP requires the use of anticoagulants and often causes transient release of bioactive molecules, which limits its regenerative potential in some cases [13]. Because of these limitations, PRF was developed in the early 2000s by Choukroun et al. [14] as a second-generation concentrate. PRF is independent of anticoagulants and contains a fibrin matrix with a feature for slow and prolonged release of growth factors. More recently, concentrated growth factors (CGF), developed by Sacco [15], are the third generation. CGF is developed from a alternating speed rates centrifugation protocol, varied speed of spinning, with higher-density fibrin matrix. The evolution from PRP to CGF represents a move towards increasingly simple, additive-free, and highly biologically active preparations most closely resembling the physiological mechanisms of healing.
However, to date, despite its increasing application in clinical settings, evidence is still piecemeal. In particular, although PRP, PRF, and CGF are described in the literature as if they were equivalent products, they differ widely in their production techniques, cellular component, fibrin mesh structure, and release kinetics of growth factors. In addition to this lack of standardization in centrifugation techniques and lack of standardization in reporting platelet and leucocyte concentrations, it is particularly significant to point out that randomized head-to-head comparison studies of PRP, PRF, and CGF are limited. In response to these limitations, this narrative review aims to provide a structured comparison of PRP, PRF, and CGF by integrating existing evidence on their preparation methods, biological properties, fibrin network architecture, growth factor release kinetics, and clinical applications. For this narrative comparison, a structured search on the literature using the search engines of PubMed, Scopus, and Web of Science is employed, encompassing literature published between 2010 and 2025. In order to find the most related literature on platelet concentrates, a combination of keywords such as: Platelet rich plasma, platelet rich fibrin, concentrated growth factor, regenerative medicine, tissue engineering, and growth factor release kinetics is employed. For the synthesis of this narrative comparison, peer-reviewed research papers, clinical and preclinical studies, and high-quality narrative reviews published in English are employed.
Biological background
Platelets or thrombocytes are minute fragments of cells that are absent of nuclei; instead, they contain cytoplasmic components such as α-granules, dense granules, and lysosomes that are made up of numerous active molecules that are required in hemostasis. They are derived from megakaryocytes found in the bone marrow. They are discoid shaped and are supported by microtubules (Figure 1), which is similar to the cytoskeleton found in other cell types that provides them with shape and structure. When these platelets get activated due to injury to the blood vessel walls and contact with blood, among other factors such as encountering collagen surfaces, then these tiny particles change shape to be adhesive with the capacity to release these α-granules to the environment surrounding them. The α-granules are endowed with numerous growth factors such as PDGF, TGF-β, VEGF, and IGF-1, making these granules particularly useful within regenerative medicine [13,14]. These factors are particularly important in various cellular processes that include angiogenesis, cell migration, cell division, and degradation of extracellular matrices. Other than their function within hemostasis processes in the blood, these platelets are today found to take part in processes that include affecting wound healing and regulation within the immune processes within the organism. They are set to take an advantageous application within autologous treatments; these applications particularly include the use of PRP, PRF, and CGF [13,14].
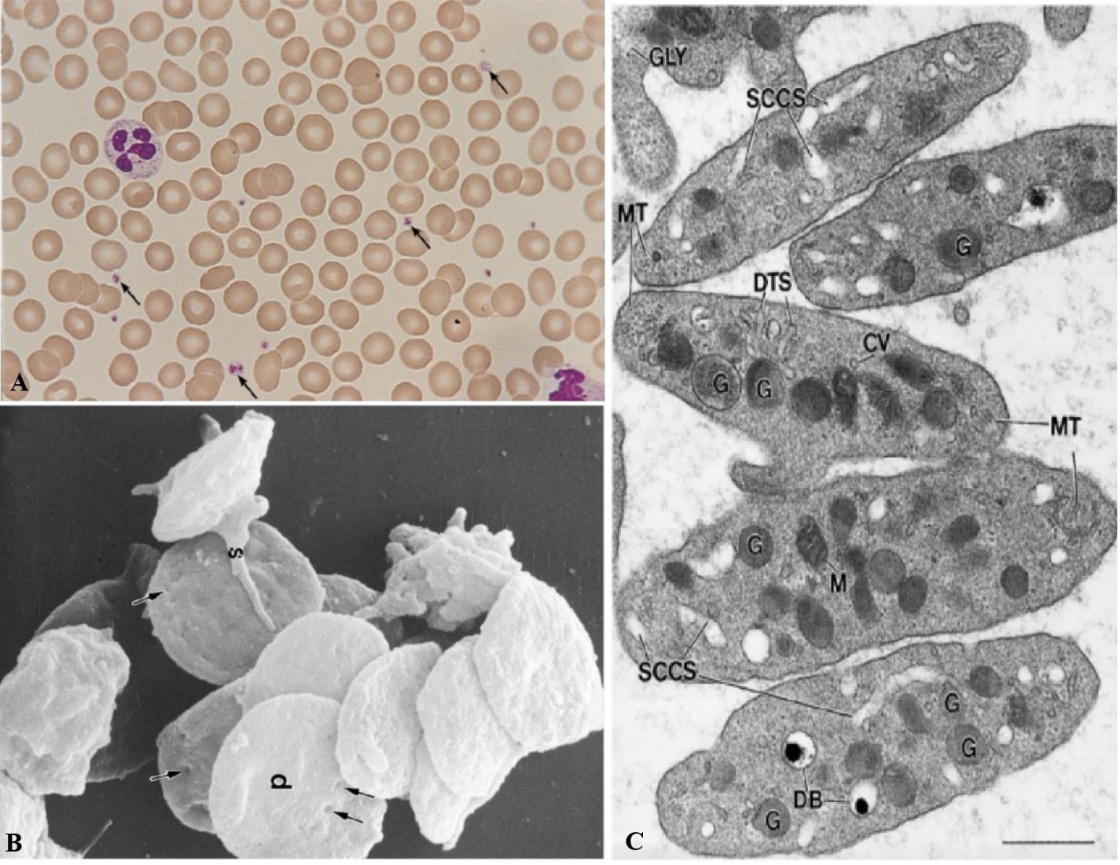
Figure 1 Representative Images of Platelets. (A) A peripheral blood smear from a human subject stained using Wright-Giemsa reveals platelets (arrows) scattered among red blood cells and leukocytes. The platelets display a pale grayish-blue cytoplasm with distinct purple-red granules. The original image was taken from a 35-mm slide (100× magnification) [16]. (B) A scanning electron microscope image shows resting (unstimulated) human platelets. Most exhibit a discoid shape (d), with numerous surface indentations (arrows) corresponding to openings of the surface-connected canalicular system, which communicates with the external environment (15,000× magnification) [17]. (C) An ultrastructural view of resting platelets highlights various internal components. Visible structures include the SCCS, dense tubular system (DTS), mitochondria (M), α-granules (G), dense bodies (DB), coated vesicles (CV), glycogen particles (GLY), and microtubules (MT), which appear in both cross-sectional and longitudinal orientations at the edges of the discoid platelet. Image captured at 46,000× magnification; scale bar represents 0.5 µm [16].
Stimulated, more so after coagulation or mechanical injury (e.g., centrifugation), platelets secrete their signaling molecules, in the form of α-granules, into the extracellular matrix. Perhaps one of the most potent growth factors is PDGF since it will induce proliferation and migration of fibroblasts, smooth muscle cells, and mesenchymal cells-a most crucial step towards tissue remodeling. Transforming growth factor-beta (TGF-β) is a 2-fold agonist, inducing synthesis of extracellular matrix (ECM) and modulating inflammation, a very crucial point in homeostatic wound healing. Vascular endothelial growth factor is a potent angiogenic stimulus, promoting the formation of new vessels for oxygen and nutrient supply to regenerating tissue [18]. Additionally, insulin-like growth factor-1 (IGF-1) plays a role in cell survival and differentiation, particularly in muscle and bone [19,20], whereas EGF induces proliferation of epithelial and endothelial cells to enhance repair of the skin and mucosae [21]. Basic fibroblast growth factor (bFGF) [22] and HGF [23] are other molecules that induce these repair processes by inducing mobilization of stem cells and tissue-specific repair pathways. The synergistic activity of these growth factors makes platelet concentrates extremely effective in triggering as well as sustaining multifactorial biological cascades related to healing. Interestingly, the concentration and release patterns of these factors in PRP, PRF, and CGF differ with the preparation technique. Clarification of individual and combined roles of these growth factors is mandatory in order to leverage the best possible therapeutic efficacy of platelet based treatments in clinical settings.
Platelet-mediated tissue repair is a multi-step, complex process mediated by platelet activation within injured tissue. Platelets adhere to and are activated by collagen exposed on vascular injury, and they release a cascade of growth factors, cytokines, and chemokines from their α-granules. These bioactive molecules initiate the first phase of healing, hemostasis, by stimulating fibrin clot formation, a provisional matrix and migration scaffold for cells. Then, the growth factors that are secreted reach and activate the fibroblasts, endothelial cells, and the mesenchymal stem cells to migrate towards the wound. Growth factors PDGF and TGF-β promote cell proliferation and cell production of ECM, whereas VEGF promotes angiogenesis to facilitate proper oxygenation and nutrition [24]. IGF and EGF regulate epithelialization and cell differentiation, which is important as the tissue is remodelling. Platelets also regulate local immunity by regulating macrophage polarization and preventing excessive inflammation to render the environment regenerative, and not fibrotic [25]. In platelet concentrates like PRP, PRF, and CGF, the fibrin network acts as a depot for controlled release of growth factors to extend the timeframe of the biological response beyond that of the short-lived post-trauma phase. Regulated release mimics physiological healing cascades and induces regeneration in hard and soft tissues. Platelets thus initiate healing and orchestrate the regenerative microenvironment in a way critical to successful tissue repair.
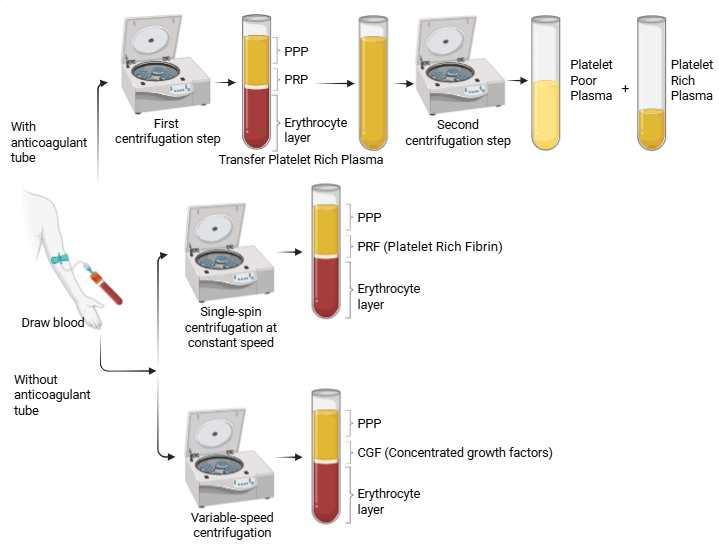
Figure 2 The preparation protocols for 3 autologous blood-derived products: PRP, PRF, and CGF. While all are obtained through centrifugation of whole blood, they differ in centrifugation speed, duration, and tube type. PRP typically involves anticoagulant use and 1 or 2 centrifugation steps to concentrate platelets. PRF is produced without anticoagulants using a single, fixed-speed spin, allowing fibrin clot formation. CGF requires a variable-speed centrifugation program to yield a denser fibrin matrix enriched with growth factors. (Drawn with Biorender)
Source: Authors.
Table 1 Main types of PRP, PRF, and CGF products. This is a summary of the most common and principal types, though several other variations also exist.
Abbreviation |
Full |
Highlights |
P-PRP or Pure-PRP |
Pure Platelet Rich Plasma |
Characterized by high platelet concentration, along with a lack or shortage of leukocytes and a low-density network of fibrin; often used in cosmetic treatments and therapies. |
LP-PRP |
Leukocyte-Poor Platelet-Rich Plasma |
Consists of leukocytes (low) and platelets suspended within a low-density fibrin network; suited to soft tissue trauma where a controlled inflammatory response can assist healing. |
LR-PRP |
Leukocyte-Rich Platelet-Rich Plasma |
LR-PRP contains both platelets and leukocyte (high). It is particularly beneficial in treating pressure ulcers and diabetic foot ulcers, conditions that require rapid tissue regeneration and reduced risk of infection. |
P-PRF |
Pure Platelet-Rich Fibrin |
P-PRF is a variation of PRF with a focus on high platelet and fibrin concentrations and the deliberate exclusion of leukocytes. P-PRF is thus considered suitable for soft tissue procedures where improved healing and minimized inflammation are of the highest priority. |
L-PRF |
Leukocyte-Platelet Rich Fibrin |
L-PRF is a valuable method that is used in periodontal surgeries. This is obtained using a gradual process of centrifugation that effectively concentrates leukocytes, fibrin, and platelets within a clot matrix. Leukocytes contribute to the healing process and angiogenesis. |
i-PRF |
Injectable Platelet Rich Fibrin |
i-PRF is the liquid form of PRF that is obtained with a lower centrifugation rate and a shorter duration, producing a viscous liquid. This makes it easily injectable into soft tissue or mixable with a different grafting material to enhance tissue regeneration outcomes. |
CGF |
Concentrated Growth Factor |
A thick fibrin matrix rich in platelets, leukocytes, and growth factors is used for extended tissue regeneration, especially in the dental and orthopedic disciplines. |
Source: Authors
Platelet-rich plasma
Platelet-Rich Plasma is defined as an autologous blood derivative containing a platelet concentration above baseline levels [26]. PRP was first introduced in 1954 by Kingsley [27]. To address variability in PRP preparations, classification systems have been developed to categorize PRP based on content and method of preparation. One of the most widely accepted is the classification by Ehrenfest [28], which divides PRP into 4 main types based on the presence or absence of leukocytes and fibrin architecture. To date, PRP has been further classified into multiple types, with the third generation represented by CGF (Table 1). PRP preparation methods vary significantly, resulting in different concentrations of blood components and growth factors [29,30]. Key growth factors in PRP include PDGF, TGF-β, VEGF, and EGF, which promote tissue regeneration and healing [31]. PRP has shown efficacy in treating various conditions, including knee osteoarthritis, tendinopathies, and wound healing [31]. However, the optimal composition and preparation method for specific clinical applications remain unclear [32]. Standardization of PRP preparation protocols and reporting is crucial for comparing study results and determining the most effective formulations for different treatments[30].
Centrifugation is one of the key steps in the preparation of PRP and influences platelet concentration and purity. Single-spin and double-spin are 2 commonly used protocols. Low-speed single centrifugation in single-spin protocol spins the blood apart into buffy coat, plasma, and red cells. Platelet-rich fraction is recovered over the buffy coat but generates more diluted levels of platelets and can include a higher leukocyte count or plasma. Double-spin, in contrast, is a 2-stage process: First spin for the separation of red cells and plasma/buffy coat and second spin at higher speed for concentrating the platelets. The method is more prone to provide more platelets and the facility to alter leukocyte content but takes more effort and time. Selection of protocol depends upon clinical need, demands of platelet and leukocytes, and availability of equipment. Standardization is difficult because protocols vary (Figure 2). Currently there are still many versions of manual PRP acquisition, with no uniformity (Table 2). In recent years, novel classification frameworks have emerged beyond the Dohan Ehrenfest system, incorporating not only leukocyte and fibrin content but also quantitative platelet concentration, growth factor profiling, and activation status[33]. Moreover, the concept of “personalized PRP” has been introduced, emphasizing the need to tailor PRP formulations to patient-specific biological parameters and clinical indications [34].
The addition of separation gel is a great improvement in PRP formulation, leading to enhanced uniformity, as well as purity, of the end product. Biologically inert, this polymer reduces inflammatory responses, leading to an improvement in PRP quality. Importantly, it is easy to adjust the density of the separation gel in order to yield PRP containing only platelets or a given population of leukocytes, depending on what therapy it has been formulated for. Moreover, separation gel reduces procedural heterogeneity in manipulation as well as allows for consistent reproduction of identical preparations, especially in clinics with limited cell-separation technologies (Table 2; No. 4 and 5). However, it is important to closely choose the right type of gel, as different commercial preparations have a potential for compromising platelet function or interfering with plasma constituents.
Table 2 Various protocols for PRP isolation from peripheral blood (Source: Authors).
No. |
Type |
Author |
Method |
% Recovery |
Note |
1 |
2 steps |
Segabinazzi and Podico [35] |
Step 1: 400 g/15 min Step 2: 1,000 g/10 min |
28 ± 13% |
Tradition |
2 |
2 steps |
Machado et al. [36] |
Step 1: 200 g/12 min Step 2: 1,600 g/8 min |
40 ± 19% |
Tradition |
3 |
2 steps |
Carvalho et al. [37] |
Step 1: 300 g/10 min Step 2: 700 g/15 min |
51.29% - 78.73% |
Tradition |
4 |
2 steps |
Muthu et al. [38] |
Step 1: 100 g/15 min Step 2: 1,600 g/20 min |
86% - 99 % |
Gel |
5 |
1 step |
Author |
Step 1: 1,500g/10 min |
> 90% |
Gel (Biothérapie Pure-PRP Kit) |
PRP therapy has numerous advantages making it an ideal candidate in regenerative medicine. First, PRP is autologous; therefore, there are no worries about immune rejection and disease transmission. PRP holds significant amounts of growth factors such as PDGF, TGF-β, and VEGF that stimulate cell growth and angiogenesis [1]. Additionally, PRP can be isolated in a rather straightforward process from peripheral blood. In essence, PRP has shown potential applications in regenerative medicine with promising results in sports injuries to joints, wounds that are unresponsive to treatments, hair loss disorders (alopecia), and reconstructive surgery. Despite these numerous benefits and potential uses, PRP has some key shortcomings as well. The most significant key implication is heterogeneity in preparation techniques; therefore, heterogeneity among subjects and among studies with regard to platelet count per appointment, cell count per appointment, and release of growth factors. This implies that one cannot compare results with regard to efficacy. Furthermore, PRP efficacy has been questioned with regard to various applications due to contradictory results from RCTs. Additionally, there are intrinsic (age-related factors in patients such as baseline platelet count and other con) and technical (factors such as centrifugation speed and activator use) factors that render it impossible to produce similar results. Another con (shortcoming) is that there would be immediate release of growth factors from PRP that might be limiting its use if aimed to be therapeutic in the medium to distant term without incorporating matrices/carriers like other cell types. Also, PRP must be made with the aid of other substances such as thrombin that might be potential indicators for immune reactions that may lead to possible contamination. Nevertheless, PRP is highly therapeutic; therefore, it needs significant research aimed at optimizing its efficacy and application in medicine.
Platelet-rich fibrin
Choukroun et al. [39] developed PRF, the second-generation platelet concentrates. PRF is a second-generation autologous platelet concentrate derived from human blood through centrifugation [40,41]. It contains growth factors and cytokines entrapped in a fibrin matrix, promoting wound healing and tissue regeneration [42]. PRF has diverse applications in dentistry, orthopedics, and aesthetic medicine, including hair restoration, skin rejuvenation, and scar revision [43]. Its advantages include ease of preparation, cost-effectiveness, and prolonged release of growth factors [40, 41]. PRF offers superior advantages compared to PRP, with no need for anticoagulants or exogenous compounds.
Recent studies have suggested that centrifugation methods are of importance in establishing the yield and quality of PRF used in regenerative medicine. Lower relative centrifugation forces (RCF) are associated with higher concentrations of platelets, leukocytes, and growth factors in PRF products [44,45]. Low-speed centrifugation (LSCC) has been proposed to improve PRF quality [44]. Moreover, centrifuge characteristics, such as vibration levels, also influence the composition and integrity of PRF [46]. Diversification of protocols has led to the development of various PRF types, including L-PRF, A-PRF, and I-PRF, each with distinct cellular and growth factor profiles [47] (Table 1). Standardization of PRF preparation protocols is needed to provide reproducibility and clinical efficacy [48]. The centrifugation time, force applied, and tube properties greatly impact the resulting PRF product [49,50], highlighting the need for optimized protocols in the field of regenerative medicine. Many different protocols have been developed over time; the table below summarizes them (Table 3).
Table 3 Standard protocols for PRF preparation (Source: Authors).
No. |
Type |
Authors |
Protocol |
1 |
L-PRF |
Choukroun et al. [39] |
2,700 rpm×12 min |
2 |
A-PRF |
Ghanaati et al. [51] |
1,500 rpm×14 min |
3 |
A-PRF+ |
Fujioka-Kobayashi et al. [52] |
1,300 rpm×8 min |
4 |
I-PRF |
Mourão et al. [53] |
3,300 rpm×2 min |
Over the years, several methodologies for PRF preparation have been developed with the aim to enhance cellular composition, fibrin architecture, and the release of growth factors. The original L-PRF protocol, developed by Choukroun et al. [39], employs a centrifugation of 2,700 rpm for 12 min, thus setting the stage for subsequent methodologies. Ghanaati et al. [51] introduced the A-PRF protocol, which employs a lower centrifugal force of 1,500 rpm for 14 min, with the aim to improve the entrapment and stabilization of leukocytes in the fibrin matrix. Later, Fujioka-Kobayashi et al. [52] took it a step further by developing A-PRF+, which features an even lower speed and time (1,300 rpm for 8 min) to improve the preservation of monocytes and accelerate the polymerization of fibrin. The latest innovation is the injectable i-PRF, which was developed by Mourão et al. [53] using a centrifugation speed of 3,300 rpm for only 2 min, allowing the final product to be retained in a liquid form before clotting. Compared to solid PRF forms, i-PRF offers unique advantages for integration with grafting materials and for therapeutic administration via injection[53]. Each method has specific advantages depending on the clinical goal-solid scaffolds for surgical application versus liquid preparations for injection-highlighting the need for a personalized approach in regenerative medicine.
Concentrated growth factors
CGF as proposed by Sacco [15], represent the third generation of autologous platelet concentrates and are processed by a unique protocol involving variable-speed centrifugation. Unlike PRP and PRF, which follow fixed-speed protocols, CGF is prepared by variating the speeds and acceleration phases with specialized centrifuges like the Medifuge (Silfradent, Italy). The long-established protocol yields a denser and more networked fibrin matrix with an increased concentration of plateletstogether with a high level of associated leukocytes and CD34+ stem-like cells [54]. The biological architecture of CGF allows for long-term release of growth factors like PDGF, TGF-β, and VEGF with the ability to enhance angiogenesis, fibroblic proliferation and remodeling of the extracellular matrix [15]. Compared to PRP, requiring the addition of anticoagulants and characterizied by rapid but transient factor release; and PRF, yielding a stable matrix with slow release kinetics, CGF offers a synergistic partnership between facile application and form and strength and healing potential. More importantly, CGF also requires the introduction of no external substance, implements a one-step processing protocol and yields a long-lasting biologically active matrix favorable for long-term healing. CGF is obtained using a pre‑programmed variable‑speed protocol, including 30 s acceleration, 2 min at 2,700 rpm, 4 min at 2,400 rpm, 4 min at 2,700 rpm, 3 min at 3,000 rpm, and ~36 s deceleration, for a total of approximately 12 -13 min [15].
Numerous studies have demonstrated the concentrations of growth factors in CGF and PRF to be equal to or higher than those in classical PRP [55,56]. CGF and PRF have equal potential in bone healing and tissue regeneration as demonstrated by extensive clinical and preclinical evidence [57]. Of note is the higher tensile strength and total volume of growth factors in CGF as compared to PRF [58], features expected to improve its physical strength and extent of biological action. Comparative analysis has demonstrated differences in the 3-dimensional architecture of the platelets, leukocytes, and bioactive entities in CGF and new generations of PRF such as A-PRF [59]. Both CGF and A-PRF facilitate cellular proliferation and serve as potent biological scaffolds and reservoirs for long-term release of the growth factors and thereby create a conducive microenvironment for healing of tissues.
Biological properties and release kinetics
Platelet concentrates such as PRP, PRF, and CGF exert their regenerative potential primarily through the localized delivery of biologically active molecules, particularly growth factors and cytokines. The concentration, spatial distribution, and temporal release of these molecules are critical in determining the biological efficacy of each preparation.
Growth factor content and platelet concentration
Numerous studies have quantified the levels of growth factors across PRP, PRF, and CGF preparations. PRP, by design, yields the highest initial concentration of platelets, often exceeding 3 - 5 times the baseline level. This corresponds with elevated immediate release of PDGF, TGF-β, VEGF, and EGF. However, due to the absence of a dense fibrin scaffold, these factors are released in a rapid burst phase, peaking within the first few hours post-application [55,56].
In contrast, PRF and CGF contain slightly lower total platelet counts but benefit from a more structured fibrin network. This architecture serves as a natural reservoir, enabling sustained and gradual release of growth factors over 7 - 14 days[59]. Comparative studies have shown that CGF, in particular, may contain higher cumulative levels of TGF-β and VEGF than PRF due to its denser and more cross-linked fibrin matrix [60].
Release kinetics
PRP exhibits a burst-type release profile with up to 90% of stored GFs released within the first 24 h [59]. This may be beneficial in acute injuries where immediate stimulation is desirable. However, it lacks the long-term release needed for sustained tissue remodeling.
PRF, especially advanced forms like A-PRF and A-PRF+, demonstrates a slow-release profile due to its fibrin entrapment. Studies have confirmed that these matrices maintain steady GF release for over a week [52]. CGF mirrors this behavior but extends the duration further. Its denser fibrin framework allows for the gradual elution of GFs, supporting prolonged angiogenesis, stem cell recruitment, and ECM synthesis.
Biological effects
The sustained release kinetics of PRF and CGF directly influence cellular responses. TGF-β promotes myofibroblast activation and ECM production; PDGF recruits fibroblasts and smooth muscle cells; VEGF induces robust neovascularization; and IGF-1 supports cell survival and differentiation. The presence of leukocytes and CD34+ cells in CGF may further enhance immunomodulation and tissue remodeling [59].
Taken together, while PRP delivers a high-dose, short-lived stimulus ideal for initial inflammatory phase, PRF and CGF provide a more physiological, time-controlled delivery suitable for orchestrating later phases of healing. CGF’s superior tensile strength and cellular content make it particularly advantageous in long-term regenerative contexts.
Table 4 Comparative summary of platelet concentrates (Sources: Authors).
Parameter |
PRP |
PRF |
CGF |
Platelet Concentration |
+++ |
++ |
++ |
Growth Factor Peak |
High (0 - 24 h) |
Moderate (3 - 7d) |
Moderate-High (5 - 14d) |
Release Kinetics |
Burst |
Sustained |
Extended Sustained |
Scaffold Density |
Low |
Moderate |
High |
Leukocyte Content |
Variable-Present |
Present |
High (with CD34+) |
Main Application |
Acute injury |
Soft tissue healing |
Long-term regeneration |
Note: +++ indicates highest relative levels.
Despite the growing body of literature supporting the clinical use of platelet concentrates, comparative evidence among platelet-rich plasma, platelet-rich fibrin, and concentrated growth factors remains inconsistent. Reported therapeutic outcomes vary substantially due to heterogeneity in preparation protocols, centrifugation parameters, platelet and leukocyte concentrations, and outcome measures across studies. In particular, direct head-to-head randomized controlled trials comparing PRP, PRF, and CGF under standardized conditions are scarce. As a result, differences in clinical efficacy are often inferred from indirect comparisons rather than robust comparative data. These limitations complicate evidence-based decision-making and underscore the need for standardized preparation protocols and well-designed comparative clinical studies.
Clinical applications
Dentistry and oral surgery
These preparations are rich in growth factors that stimulate tissue repair, with applications including socket preservation, implant surgery, bone regeneration, and managing conditions like bisphosphonate-related osteonecrosis [52]. In dentistry and implantology, these 3 are utilized to facilitate bone and soft-tissue repairing (for sinus lifts, ridge augmentations, periodontal defects). PRP has an abundant amount of GFs (PDGF, TGF-β, VEGF) concentrated in a liquid that facilitates quicker grafting; however, the results are transitory. PRF offers a fibrin sponge that releases GFs after days to weeks; these facilitate improved periodontal repairing and implant osseointegration; meta-analyses found PRF to result in significantly higher bone filling and attachment increments compared to other devices [61]. CGF has an even more compact fibrin matrix with more platelets and CD34⁺ progenitor cells; these facilitate angiogenesis and secondary bone formation. CGF was shown to significantly improve bone repairing and implant implantation in the maxillary sinus augmentation model compared to controls [62]. Though these are significant breakthroughs, efficacy still varies; most studies stress that various techniques that pair platelet concentrates with bone grafting (“sticky bone” approaches) result in maximally superior results [61,62].
Dermatology and aesthetic medicine
In dermatology and aesthetic medicine, PRP has the most established concentrate form. Clinical research and systematic reviews confirm PRP (often injected or microneedled) promotes the production of collagen and enhances skin texture and density besides raising hair density and circumference in patients with androgenetic alopecia [52]. PRF (particularly injectable i-PRF) has attracted interest due to possibly prolonged dermal fibroblast activation by the slower release fibrin mesh. Initial experiences with i-PRF indicate that it might optimize the efficacy of acne scars and alopecia; however, rigorous testing is still required. The concentrated GF and cytokines in CGF indicate possible prolonged activation of fibroblasts to renew skins; evidence is still evolving. In hair regrowth, PRP has reliably been shown to raise hair density and caliber [52]; PRF and CGF are still investigational; theoretically, though, these might prolong treatment success due to the fibrin scaffold that supports fibroblast activation.
Orthopedics and sports medicine
In orthopedics and sports medicine, PRP injections are commonly administered within the joint space to manage injuries due to degradation and overuse. Recent meta-analyses indicate that PRP results in significant pain relief and functional recovery in patients with knee arthritis and that PRP often surpasses HA in efficacy [63]. Additionally, conjoining PRP with HA results in greater improvements in pain and function compared with PRP alone [63]. PRP has also regenerative applications in managing tendinopathies (lateral epicondylitis and Achilles tendinopathy). In 2024, PRP injections resulted in superior pain relief with prolonged effectiveness (more than 6 months) over corticosteroid injections in managing tennis elbow [64]. PRF and CGF are more infrequently administered as isolated intra-articular injections. Furthermore, these are mostly investigated within surgical applications as matrices within repairs (supplementing tendon or ligament transplants). In PRF and CGF matrices, there might be certain cell infiltration supported by the fibrin structure. Yet, current conclusive data within musculoskeletal repairs with PRF and CGF remain mostly within the realm of ongoing investigations. Importantly, PRF and CGF can be more beneficial with application within musculoskeletal reconstructive implants/matrix biomaterials due to structural and immunomodulatory function.
Regenerative medicine and tissue engineering
PRF and CGF are utilized as autologous biomaterial substrates in regenerative medicine and tissue engineering applications. The 3D fibrin network in PRF and CGF acts as an extracellular matrix analog to facilitate cell adhesion and migration [65]. he PRF membranes intended for mesenchymal stem cell (MSC) transplantation improve osteogenic and angiogenic processes. The higher GF and progenitor cell contents in CGF create a microenvironment conducive to cell growth. These matrices are utilized in more sophisticated platforms such as hydrogel matrices, collagen sponge substrates, and 3D printed bio-inks. The application of PRF and CGF matrices with EVs and biofabrication has potential “smart” regenerative applications. In these therapeutic applications, PRF and CGF matrices function as biomimetic carriers to release GFs over time; PRP provides an initial GF release with subsequent PRF/CGF release and structural support [65].
Table 5 Clinical applications across specialties (Sources: Authors).
Field |
PRP |
PRF |
CGF |
Dentistry & Oral Surgery |
High platelet/GF dose for bone grafts and sinus lifts; accelerates bone regeneration and soft-tissue healing. |
3D fibrin scaffold with sustained GF release; improves periodontal regeneration, socket preservation, implant healing [61]. |
Dense fibrin matrix rich in CD34⁺ cells; enhances angiogenesis and osteogenesis (e.g. in infected-socket implant placement) [62]. |
Dermatology & Aesthetic |
Proven for skin rejuvenation and acne scar remodeling; significantly increases hair density and thickness in alopecia [66] |
Injectable i-PRF yields longer-lasting GF delivery for dermal remodeling; early evidence of benefit in skin and hair restoration [67]. |
Experimental in aesthetics; rich in GFs/cytokines may support prolonged skin regeneration (ongoing studies). |
Orthopedics & Sports Medicine |
Improves knee osteoarthritis and chronic tendinopathies; intra-articular injections yield pain relief and functional gains [64] |
Used experimentally as a scaffold in tendon/ligament repair and cartilage engineering; provides natural matrix support [65]. |
Under investigation for bone and soft tissue healing; high GF/Stem cell content suggests potential in tissue repair. |
Regenerative Medicine |
Delivers a rapid GF burst but lacks structural scaffold (less suited as standalone tissue scaffold). |
3D fibrin matrix supports MSC engraftment, angiogenesis, and controlled GF release [65,68]. Ideal for grafting and bioprinted constructs. |
Rich reservoir of GFs and progenitor cells; promising as an advanced biomaterial for stem-cell therapies and 3D biofabrication (e.g. bioinks). |
Limitations and challenges
However promising platelet concentrates are, they are plagued with inherent scientific deficiencies. Specifically, there is a remarkable divergence on the preparation protocols. Centrifugal speed and duration of centrifugation vary significantly. Moreover, the choice of anticoagulants or activators is divergent [69,70]. Consequently, the platelet concentration, number of leukocytes, and release of growth factors are significantly different across PRP, PRF, and CGF. This inherent diversity makes it difficult to compare results or interpret them. In fact, it has been pointed out that “absence of unified standards for PRP preparations, classifications and clinical applications” confuses the matter. There is a dearth of comparative clinical data. Very few small-scale randomized trials have comparatively evaluated the effectiveness of PRP, PRF, and CGF. Most of the available data are pilot or case series trials [71,72]. Therefore, the combined outcome is based on indirect comparisons and not on direct comparative trials. Moreover, the inherent variability due to the donor is yet another confounding factor. Age of the donor, initial platelet count, and the donor’s health condition significantly affect the final preparation. For instance, one study has shown that each decade of the donor’s age resulted in a reduction of 32,000 platelets/μL of PRP. Also, a unit increase in the initial platelet count increased the platelet output of PRP by 3.8× [73]. Such inherent variations render 2 different preparations bearing the same name to possess different biology [74]. In other words, inherent scientific deficiencies preclude the drawing of evidence-based conclusions on which preparation is the best preparation for which condition.
In the clinic, these scientific uncertainties are reflected as real-world challenges. There is no data to support which is the “best” concentrate to administer in a given patient, as there are no head-to-head studies of efficacy [75]. For example, while there is some data suggesting that PRP is good for rapid healing of soft tissues, while CGF/PRF is better suited to bone healing [76], there are no standardized guidelines, with some studies suggesting that CGF is better than PRF, while others suggest that CGF = PRF [57,77,78]. There are also unpredictable patient responses, with some responding very well, while others respond poorly, or not at all, which is most likely due to variability in product composition, as discussed. There are also issues with reproducibility. Even within a given practice, small variations in centrifugation or technique can lead to variability in product [79]. And variability between practices makes it even more difficult to interpret outcome studies, as what was considered to be PRF or CGF in one trial may or may not be similar to what was considered to be those things in another, making meta-analysis very difficult[80]. There are also some inherent limitations of these products. Both PRF and CGF produce solid blood clots that must be used immediately, as they cannot be molded or stored. They also are of very low tensile strength, which makes them unable to support heavy loads, as they are best suited to augmenting soft tissues or filling non-load bearing defects [81]. And, lastly, many of these are issues of cost. Good preparation sets and centrifuges are costly, as are good techniques, which are not always widely available. Moreover, in most healthcare delivery systems, procedures using these products are not covered by insurance, which makes them less widely available.
On the translational side, platelet concentrates are in an unclear regulatory environment globally. In most countries, PRP, PRF, and CGF are labeled as “minimally manipulated” autologous blood products, which puts them beyond the purview of drug regulations [82]. But there is no standardized global classification system. Even in Europe, there are variations in regulations in different countries. In one case, PRP was labeled a “non-standardized medicinal product” in Swiss law, while in other countries, PRP was labeled differently based on variations in blood product directives [83]. This creates a situation where there is no standardized global system for clinical trials or approval of platelet concentrates (PCs). Then there are quality control and production systems. PCs are mostly produced in a bedside setting. So, there are no “industry-scale” batches with standardized specifications [84]. Few institutions follow GMP-like standards for autologous concentrates. EU directives for blood components are more oriented toward safety and quality [83]. But it is “practically difficult” for manufacturers and clinicians to bring platelet concentrate preparation fully under these standards. This makes it difficult to ensure scalability in terms of potency and purity. From a practical standpoint, these are mostly manual procedures with variations in equipment and methods.
Among the frontier areas, some show great potential, but also many questions remain. A promising new application of PRP is the use of platelet-derived extracellular vesicles (EVs): Nano-sized exosomes derived from activated platelets [74,85]. Recent reviews indicate that PRP-EVs contain the same growth factors and microRNAs as conventional PRP, and may even have an advantage in terms of penetration and immune system interaction. Theoretically, EVs might be developed into an off-the-shelf biologic [86]. Yet, the development of EVs from pure, isolated EVs remains in its infancy, with challenges in isolation, dosage, and a lack of large, confirmatory studies in human patients. Similarly, PRF and CGF are being explored for use in bioactive scaffolding in the emerging field of tissue engineering. PRF/CGF scaffolds, for instance, have been incorporated into bioinks in 3-dimensional bioprinting of custom-made grafts [87]. Such constructs may facilitate the healing of either bone or cartilage, again with efficacy established in small studies only [88,89]. But in addition, broad questions exist in the development of platelet concentrates themselves. Should concentrates be customized on the basis of the starting level of a patient’s platelets/GFs? How should leukocytes best be concentrated? Can ‘good’ versus ‘bad’ platelet concentrates be distinguished, depending on the application? The answers, of course, would require better biomarkers, and a standardized set of endpoints. But, in a welcome development, the field has begun to come together in terms of proposed solutions, such as a ‘PAW’ system of product definition (Platelet count, Activation strategy, White cell concentration)[90]. Some also urge the establishment of a ‘minimal set’ of information in studies, describing the exact preparation and composition, and the conduct of ‘indication-specific’ randomized controlled trials [91]. The conclusion, then, would be that, despite the great scientific potential of the new applications of platelet concentrates (EVs, bioengineering scaffolds, stem cell mixtures, and so on), these also indicate a need for rigorous validation.
Future perspectives
In addition to traditional uses, platelet concentrates are currently being increasingly redefined as flexible platforms in the field of regeneration, and not as static components derived from blood. Research in biomaterials science and bioengineering is currently enabling enhanced precision in the control of platelet-derived bioactivity and delivery rates [92]. There is a shift towards moving from empirically based autologous concentrates to rationally based function-oriented strategies in regeneration [93]. Another area with great potential is personalized platelet therapy. Instead of a universal strategy, doctors will soon be able to personalize PRP therapy according to individual patient profiles [94]. This may involve personalizing the platelet dose or supplementing it with individual patient profiles of specific cytokines based on patient levels of either platelets or growth factors. Point-of-care testing kits are being developed to determine individual biomarkers and provide guidance on the best possible composition of the concentrate. Recent literature reports the potential of multi-omics studies (genomics, proteomics, and metabolomics) to predict PRP efficacy and develop personalized treatments [69,70].
Notably, platelet-derived bioengineered products are on the horizon. Scientists are working on developing ‘off-the-shelf’ platelet biologics of defined potency. This can be achieved by bioengineering/synthetic modification of platelet exosomes for the delivery of specific molecular cargo, and the use of gene-editing in progenitor cells for the secretion of targeted growth factors and anti-inflammatory signals [95,96]. The potential of platelets in biomimetic platforms for regeneration has been utilized in these approaches. Thus, the current focus of the field of platelet research has transformed from the use of autologous blood fractions to complex, customized platforms for regeneration. Although these new technologies are promising, their translation into mainstream practice depends on standardization and rigorous clinical studies for validation of safety and efficacy.
Conclusions
Although all 3 are sourced from the same biological origin, PRP, PRF, and CGF are known to be different regeneration modalities with considerable variations in terms of preparation procedures, fibrin network morphology, cellular contents, and release profiles of growth factors. These variations are associated with different biological and clinical outcomes, thus indicating the need to treat PRP and PRF/CGF as different therapeutic agents with their own sets of clinical utilities and limitations. Although considerable progress has been made in understanding their overall regeneration abilities in various medical and dental fields, the area is currently hindered by considerable variations in preparation procedures and lack of adequate comparative studies in clinical settings. Thus, drawing concrete inferences about their relative efficacies and choice in various clinical settings is currently not possible and will depend on future advances in terms of improved comparative studies and strategic combinations with newer biomaterials and/or platelet-derived extracellular vesicles and/or personalized regeneration strategies in the future.
Acknowledgments
We would like to thank Nhien Lam Huynh, and Yen Lam Huynh for their general supports during the work. The study was supported from VNUHCM-US Stem Cell Institute.
CRediT author statement
Phat Duc Huynh conceptualized the review topic, developed the outline, and finalized the manuscript. Nguyen Cao Nguyen contributed by preparing tables and figures, drafting responses to reviewers, thoroughly reviewing the manuscript, and managing reference formatting. All authors contributed to the critical revision of the work and approved the final version for submission.
References
[1] PD Huynh, QX Tran, ST Nguyen, VQ Nguyen and NB Vu. Mesenchymal stem cell therapy for wound healing: An update to 2022. Biomedical Research and Therapy 2022; 9(12), 5437-5449.
[2] N Saikia. Inorganic-based nanoparticles and biomaterials as biocompatible scaffolds for regenerative medicine and tissue engineering: Current advances and trends of development. Inorganics 2024; 12(11), 292.
[3] V Bhutambare, C Kamble, S Khilari, D Bhalekar, P Gawari and A Kanase. Innovative strategies in regenerative medicine: Bridging science and clinical practice. International Journal of Advanced Research in Science Communication and Technology 2024; 4(3), 186-195.
[4] ATM Nguyen. Regulatory T cells: From nobel prize foundations to engineered tolerance and next-generation immunotherapies. Innovative Medical and Biological Sciences Journal 2025; 1(1), 1-11.
[5] J Ramos-Torrecillas, E de Luna-Bertos, O García-Martínez and C Ruiz. Clinical utility of growth factors and platelet-rich plasma in tissue regeneration: A review. Wounds: A Compendium of Clinical Research and Practice 2014; 26, 7207-7213.
[6] CE Martínez, PC Smith and VAP Alvarado. The influence of platelet-derived products on angiogenesis and tissue repair: A concise update. Frontiers in Physiology 2015; 6, 290.
[7] X Wang, MR Fok, G Pelekos and L Jin. In vitro and ex vivo kinetic release profile of growth factors and cytokines from leucocyte- and platelet-rich fibrin (L-PRF) preparations. Cells 2022; 11(13), 290.
[8] M Schultze. Ein heizbarer objecttisch und seine verwendung bei untersuchungen des blutes (in Deutsch). Archiv für Mikroskopische Anatomie 1865; 1(1), 1-42.
[9] G Bizzozero. Di un nuovo elemento morfologico del sangue e della sua importanza nella trombosi e nella coagulazione (in Italian). Fr. Wallardi, Italy, 1883.
[10] S Murphy and FH Gardner. Platelet preservation. Effect of storage temperature on maintenance of platelet viability--deleterious effect of refrigerated storage. New England Journal of Medicine 1969; 280(20), 1094-1098.
[11] CJ Oon and JR Hobbs. Clinical applications of the continuous flow blood separator machine. Clinical & Experimental Immunology 1975; 20(1), 1-16.
[12] M Ferrari, S Zia, M Valbonesi, F Henriquet, G Venere, S Spagnolo, MA Grasso and I Panzani. A new technique for hemodilution, preparation of autologous platelet-rich plasma and intraoperative blood salvage in cardiac surgery. The International Journal of Artificial Organs 1987; 10(1), 47-50.
[13] M Yang, B Deng, W Hao, X Jiang, Y Chen, M Wang, Y Yuan, M Chen, X Wu, C Du, DG Armstrong, L Guo, W Deng and H Wang. Platelet concentrates in diabetic foot ulcers: A comparative review of PRP, PRF, and CGF with case insights. Regenerative Therapy 2025; 28, 625-632.
[14] J Choukroun, A Diss, A Simonpieri, MO Girard, C Schoeffler, SL Dohan, AJ Dohan, J Mouhyi and DM Dohan. Platelet-rich fibrin (PRF): A second-generation platelet concentrate. Part IV: Clinical effects on tissue healing. Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology 2006; 101(3), e56-e60.
[15] SL Sacco. International Academy of implant prosthesis and osteoconnection. Lecture 2006; 12, 4.
[16] SS Smyth. Platelet structure and function in hemostasis and thrombosis. In: JP Greer and DA Arber (Eds.). Wintrobe's Clinical hematology. Lippincott Williams & Wilkins, Philadelphia, United States, 2013, p. 389-410.
[17] PE Stenberg, MA Shuman, SP Levine and DF Bainton. Optimal techniques for the immunocytochemical demonstration of beta-thromboglobulin, platelet factor 4, and fibrinogen in the alpha granules of unstimulated platelets. Journal of Histochemistry & Cytochemistry 1984; 16(9), 983-1001.
[18] KE Johnson and TA Wilgus. Vascular endothelial growth factor and angiogenesis in the regulation of cutaneous wound repair. Advances in Wound Care 2014; 3, 647 - 661.
[19] J Fang, X Zhang, X Chen, Z Wang, S Zheng, Y Cheng, S Liu and L Hao. The role of insulin-like growth factor-1 in bone remodeling: A review. International Journal of Biological Macromolecules 2023; 238, 124125.
[20] X Zhang, FY Hu, J Li, L Chen, YF Mao, Q Li, C Nie, C Lin and J Xiao. IGF-1 inhibits inflammation and accelerates angiogenesis via Ras/PI3K/IKK/NF-κB signaling pathways to promote wound healing. European Journal of Pharmaceutical Sciences 2024; 200, 106847.
[21] SH Shin, YG Koh, WG Lee, J Seok and KY Park. The use of epidermal growth factor in dermatological practice. International Wound Journal 2023; 20(6), 2414-2423.
[22] M Farooq, AW Khan, MS Kim and S Choi. The role of Fibroblast Growth Factor (FGF) signaling in tissue repair and regeneration. Cells 2021; 10(11), 3242.
[23] JT Rodgers, MD Schroeder, C Ma and TA Rando. HGFA is an injury-regulated systemic factor that induces the transition of stem cells into GAlert. Cell Reports 2017; 19(3), 479-486.
[24] P Chandra, M Faizan, M Porwal, H Sharma and N Sachan. An overview and review of growth factors in wound healing: Emerging trends and innovations. Current Diabetes Reviews 2025. https://doi.org/10.2174/0115733998332692241202072249
[25] H Luo, W Liu, Y Zhou, X Jiang, Y Liu, Q Yang and L Shao. Concentrated growth factor regulates the macrophage-mediated immune response. Regenerative Biomaterials 2021; 8(6), rbab049.
[26] P Harrison and J Alsousou. The state of the art and future of PRP therapy. Platelets 2021; 32(2), 150-151.
[27] CS Kingsley. Blood coagulation; evidence of an antagonist to factor VI in platelet-rich human plasma. Nature 1954; 173(4407), 723-724.
[28] DM Dohan Ehrenfest, L Rasmusson and T Albrektsson. Classification of platelet concentrates: From pure platelet-rich plasma (P-PRP) to leucocyte- and platelet-rich fibrin (L-PRF). Trends in Biotechnology 2009; 27(3), 158-167.
[29] M Dejnek, J Witkowski, H Moreira, S Płaczkowska, P Morasiewicz, P Reichert and A Królikowska. Content of blood cell components, inflammatory cytokines and growth factors in autologous platelet-rich plasma obtained by various methods. World Journal of Orthopedics 2022; 13(6), 587-602.
[30] BW Oudelaar, JC Peerbooms, R Huis In't Veld and AJH Vochteloo. Concentrations of blood components in commercial platelet-rich plasma separation systems: A review of the literature. The American Journal of Sports Medicine 2019; 47(2), 479-487.
[31] AJ Sheean, AW Anz and JP Bradley. Platelet-rich plasma: Fundamentals and clinical applications. Arthroscopy 2021; 37(9), 2732-2734.
[32] L Atkinson and F Martin. Intraovarian injection of platelet-rich plasma in assisted reproduction: Too much too soon? Human Reproduction 2021; 36(7), 1737-1750.
[33] K Mautner, GA Malanga, J Smith, B Shiple, V Ibrahim, S Sampson and JE Bowen. A call for a standard classification system for future biologic research: Trationale for new PRP nomenclature. PM&R 2015; 7(4), S53-S59.
[34] JC Riboh, BM Saltzman, AB Yanke, L Fortier and BJ Cole. Effect of leukocyte concentration on the efficacy of platelet-rich plasma in the treatment of knee osteoarthritis. The American Journal of Sports Medicine 2016; 44(3), 792-800.
[35] L Segabinazzi and G Podico. Three manual noncommercial methods to prepare equine platelet-rich plasma. Animals 2021; 11(6), 1478.
[36] ES Machado, FP Soares, RS Yamaguchi, WK Felipone, R Meves, TAC Souza, R Topolniak, JP Caldas, EV Abreu, LS Rabelo Neto, PVS Pinchemel, and M Bredemeier. A simple double-spin closed method for preparing platelet-rich plasma. Cureus 2022; 14(1), e20899.
[37] A Carvalho, AF Ferreira, M Soares, S Santos, P Tomé, J Machado-Simões and AS Pais. Optimization of platelet-rich plasma preparation for regenerative medicine: Comparison of different anticoagulants and resuspension media. Bioengineering 2024; 11(3), 209.
[38] S Muthu, A Krishnan and KR Ramanathan. Standardization and validation of a conventional high yield platelet-rich plasma preparation protocol. Annals of Medicine and Surgery 2022; 82, 104593.
[39] J Choukroun, F Adda, C Schoeffer and A Vervelle. PRF: An opportunity in perio-implantology. Implantodontie 2000; 42, 55-62.
[40] TW Neal, SR Sullivan, S Cannon, W Spresser and T Schlieve. Platelet rich fibrin: A literature review of applications in oral and maxillofacial surgery. Journal of Oral and Maxillofacial Anesthesia 2024; 3(1), 1-10.
[41] V Pavlovic, M Ciric, V Jovanovic, M Trandafilovic and P Stojanovic. Platelet-rich fibrin: Basics of biological actions and protocol modifications. The Open Medicine Journal 2021; 16(1), 446-454.
[42] P Goswami, V Chaudhary, A Arya, R Verma, G Vijayakumar and M Bhavani. Platelet-Rich Fibrin (PRF) and its application in dentistry: A literature review. Journal of Pharmacy and Bioallied Sciences 2024; 16(S1), S5-S7.
[43] K Karimi and H Rockwell. The benefits of platelet-rich fibrin. Facial Plastic Surgery Clinics of North America 2019; 27(3), 331-340.
[44] J Choukroun and S Ghanaati. Reduction of relative centrifugation force within injectable platelet-rich-fibrin (PRF) concentrates advances patients' own inflammatory cells, platelets and growth factors: The first introduction to the low speed centrifugation concept. European Journal of Trauma and Emergency Surgery 2018; 44(1), 87-95.
[45] RJ Miron, H Xu, J Chai, J Wang, S Zheng, M Feng, X Zhang, Y Wei, Y Chen, C Mourão, A Sculean and Y Zhang. Comparison of platelet-rich fibrin (PRF) produced using 3 commercially available centrifuges at both high (~ 700 g) and low (~ 200 g) relative centrifugation forces. Clinical Oral Investigations 2020; 24(3), 1171-1182.
[46] DMD Ehrenfest, NR Pinto, A Pereda, P Jiménez, MD Corso, BS Kang, M Nally, N Lanata, HL Wang and M Quirynen. The impact of the centrifuge characteristics and centrifugation protocols on the cells, growth factors, and fibrin architecture of a leukocyte- and platelet-rich fibrin (L-PRF) clot and membrane. Platelets 2018; 29(2), 171-184.
[47] RFD Santos, JAA Peres and MS Queiroz. Advances in separation methods for the use of platelet-rich fibrin in tissue repair: an integrative review. General Dentistry 2023; 71(2), 65-69.
[48] PR Amable, RB Carias, MV Teixeira, I da Cruz Pacheco, RJ Corrêa do Amaral, JM Granjeiro and R Borojevic. Platelet-rich plasma preparation for regenerative medicine: Optimization and quantification of cytokines and growth factors. Stem Cell Research & Therapy 2013; 4(3), 67.
[49] DA Gutierrez, CA Alfonso, S Jaramillo-Isaza, LA Gomez and AL Muñoz. Evaluation of platelet rich fibrin obtained using different centrifugation parameters as a tool for regenerative medicine. Clinical Laboratory 2021; 67(11), 2462-2468.
[50] K Sneha, AJ Rani, RV Chandra, SP Kumar, RN Jannu and S Muppirala. The G-force conundrum in platelet-rich fibrin generation: Management of a problem hidden in plain sight. Contemporary Clinical Dentistry 2022; 13(2), 150-155.
[51] S Ghanaati, P Booms, A Orlowska, A Kubesch, J Lorenz, J Rutkowski, C Landes, R Sader, C Kirkpatrick and J Choukroun. Advanced platelet-rich fibrin: A new concept for cell-based tissue engineering by means of inflammatory cells. Journal of Oral Implantology 2014; 40(6), 679-689.
[52] M Fujioka-Kobayashi, RJ Miron, M Hernandez, U Kandalam, Y Zhang and J Choukroun. Optimized platelet-rich fibrin with the low-speed concept: Growth factor release, biocompatibility, and cellular response. Journal of Periodontology 2017; 88(1), 112-121.
[53] CF Mourão, H Valiense, ER Melo, NB Mourão and MD Maia. Obtention of injectable platelets rich-fibrin (i-PRF) and its polymerization with bone graft: Technical note. Revista do Colégio Brasileiro de Cirurgiões 2015; 42(6), 421-423.
[54] LF Rodella, G Favero, R Boninsegna, B Buffoli, M Labanca, G Scarì, L Sacco, T Batani and R Rezzani. Growth factors, CD34 positive cells, and fibrin network analysis in concentrated growth factors fraction. Microscopy Research and Technique 2011; 74(8), 772-777.
[55] J Qiao, N An and X Ouyang. Quantification of growth factors in different platelet concentrates. Platelets 2017; 28, 774-778.
[56] H Masuki, T Okudera, T Watanebe, M Suzuki, K Nishiyama, H Okudera, K Nakata, K Uematsu, CY Su and T Kawase. Growth factor and pro-inflammatory cytokine contents in platelet-rich plasma (PRP), plasma rich in growth factors (PRGF), advanced platelet-rich fibrin (A-PRF), and concentrated growth factors (CGF). International Journal of Implant Dentistry 2016; 2(1), 19.
[57] SA Khalifah, M Noureldin and M Eldibany. A comparative study between the effect of platelet rich fibrin and concentrated growth factors on osseointegration of immediate implants (a randomized clinical trial). EKB Journal Management System 2023; 48(2), 39-45.
[58] R Narayanaswamy, BP Patro, N Jeyaraman, P Gangadaran, RL Rajendran, A Nallakumarasamy, M Jeyaraman, P Ramani and BC Ahn. Evolution and clinical advances of platelet-rich fibrin in musculoskeletal regeneration. Bioengineering 2023; 10(1), 58.
[59] A Takahashi, T Tsujino, S Yamaguchi, K Isobe, T Watanabe, Y Kitamura, K Okuda, K Nakata and T Kawase. Distribution of platelets, transforming growth factor-β1, platelet-derived growth factor-BB, vascular endothelial growth factor and matrix metalloprotease-9 in advanced platelet-rich fibrin and concentrated growth factor matrices. Journal of Investigative and Clinical Dentistry 2019; 10(4), e12458.
[60] LH Lei, Y Yu, J Han, D Shi, W Sun, D Zhang and L Chen. Quantification of growth factors in advanced platelet-rich fibrin and concentrated growth factors and their clinical efficiency as adjunctive to the GTR procedure in periodontal intrabony defects. Journal of Periodontology 2020; 91(4), 462-472.
[61] WJ Park, SH Han, NJ Kim and JB Park. 2024, the efficacy of utilizing platelet-rich fibrin for managing periodontal intrabony defects in conjunction with graft material: A systematic review and meta-analysis. Applied Sciences 2024; 14(8), 3371.
[62] OA Al-Aroomi, Y Ou, KA Sakran, H Chen, Y Lin, Y Gao, Q Cai and J Chen. Effectiveness of concentrated growth factors with or without grafting materials in maxillary sinus augmentation: A systematic review. BMC Oral Health 2024; 24(1), 1275.
[63] D Du and Y Liang. A meta-analysis and systematic review of the clinical efficacy and safety of platelet-rich plasma combined with hyaluronic acid (PRP + HA) versus PRP monotherapy for knee osteoarthritis (KOA). Journal of Orthopaedic Surgery and Research. 2025; 20(1), 57.
[64] Y Xu, T Li, L Wang, L Yao, J Li and X Tang. Platelet-rich plasma has better results for long-term functional improvement and pain relief for lateral epicondylitis: A systematic review and meta-analysis of randomized controlled trials. The American Journal of Sports Medicine 2024; 52(10), 2646-2656.
[65] Y Li, H You, C Ou, H Zhu, B Cheng and J Tian. The evolution of 3 generations of platelet concentrates products: A leap from classical formulations to the era of extracellular vesicles. Frontiers in Bioengineering and Biotechnology 2025; 13, 1628565.
[66] R Lopes-Silva, M Santos, ML Sequeira, A Silva, T Antunes, P Valejo-Coelho and M Neiva-Sousa. Platelet-rich plasma effectiveness in treating androgenetic alopecia: A comprehensive evaluation. Cureus 2025; 17(1), e77371.
[67] N Farshidfar, MA Amiri, NE Estrin, P Ahmad, A Sculean and Y Zhang. Platelet-rich plasma (PRP) versus injectable platelet-rich fibrin (i-PRF): A systematic review across all fields of medicine. Periodontology 2000; 2025, 1-31.
[68] J Zhang, W Xu, Y Xiao, D Su, Y He, H Yang, Y Xie, X Wang, RH Xu, S Lei and D Wu. Accurate delivery of mesenchymal stem cell spheroids with platelet-rich fibrin shield: Enhancing survival and repair functions of Sp-MSCs in diabetic wound healing. Advanced Science 2025; 12(25), e2413430.
[69] H Zhou, Q Huang, Y Chen, J Wang and H Jiang. Biological mechanisms and clinical challenges of platelet-rich plasma in chronic musculoskeletal pain: From standardized preparation to multi-omics-guided precision therapy. Journal of Pain Research 2025; 18, 5931-5939.
[70] RV Tey and P Haldankar. Variability in platelet-rich plasma preparations used in regenerative medicine: A comparative analysis. 2022; 2022(1), 3852898.
[71] M Kavitha, S Shakthipriya, D Arunaraj, R Hemamalini, S Velayudham and B Bakthavatchalam. Comparative evaluation of platelet-rich fibrin and concentrated growth factor as scaffolds in regenerative endodontic procedure: A randomized controlled clinical trial. The Journal of Contemporary Dental Practice 2022; 23(12), 1211-1217.
[72] AT Ulusoy, I Turedi, M Cimen and ZC Cehreli. Evaluation of blood clot, platelet‐rich plasma, platelet‐rich fibrin, and platelet pellet as scaffolds in regenerative endodontic treatment: A prospective randomized trial. Journal of Endodontics 2019; 45, 560-566.
[73] L Rossi, M Ranalletta, I Pasqualini, JP Zicaro, MC Paz, P Camino and NS Piuzzi. Substantial variability in platelet-rich plasma composition is based on patient age and baseline platelet count. Arthroscopy, Sports Medicine, and Rehabilitation 2023; 5(3), e853-e858.
[74] J Wu, Y Piao and Q Liu. Platelet-rich plasma-derived extracellular vesicles: A superior alternative in regenerative medicine? Cell Proliferation 2021; 54(12), e13123.
[75] M Mozzati, G Gallesio, M Tumedei and M Del Fabbro. Concentrated growth factors vs. leukocyte-and-platelet-rich fibrin for enhancing postextraction socket healing. A longitudinal comparative study. Applied Sciences 2020; 10(22), 8256.
[76] HC Park, SG Kim, JS Oh, JS You, JS Kim, SC Lim, MA Jeong, JS Kim, C Jung, YS Kwon and H Ji. Early bone formation at a femur defect using CGF and PRF grafts in adult dogs: A comparative study. Implant Dentistry 2016; 25, 387-393.
[77] TH Kim, SH Kim, GK Sándor and YD Kim. Comparison of platelet-rich plasma (PRP), platelet-rich fibrin (PRF), and concentrated growth factor (CGF) in rabbit-skull defect healing. Archives of Oral Biology 2014; 59(5), 550-558.
[78] A Azadi, P Eftekhari-Moghadam, F Atarbashi-Moghadam, P Hazrati, AA Baghban and R Amid. Adjunctive therapy for root coverage with concentrated growth factor versus platelet-rich fibrin membranes: A systematic review and bayesian network meta-analysis. Clinical Oral Investigations 2024; 28(12), 654.
[79] CC Zumarán, MV Parra, SA Olate, EG Fernández, FT Muñoz and ZS Haidar. The 3 R's for platelet-rich fibrin: A "super" tri-dimensional biomaterial for contemporary naturally-guided oro-maxillo-facial soft and hard tissue repair, reconstruction and regeneration. Materials 2018; 11(8), 1293.
[80] A Al-Badran, S Bierbaum and C Wolf-Brandstetter. Does the choice of preparation protocol for platelet-rich fibrin have consequences for healing and alveolar ridge preservation after tooth extraction? A meta-analysis. Journal of Oral and Maxillofacial Surgery : Official Journal of the American Association of Oral and Maxillofacial Surgeons 2023; 11(8), 1293.
[81] K Monisha. Role of bone grafts and Prf/Cgf in different specialities of dentistry - a systematic review. International Journal of Scientific Development and Research 2021; 6(4), 603-607.
[82] ME Kramer and TC Keaney. Systematic review of platelet‐rich plasma (PRP) preparation and composition for the treatment of androgenetic alopecia. Journal of Cosmetic Dermatology 2018; 17, 666 - 671.
[83] P Sebbagh, A Cannone, G Gremion, V Gremeaux and W Raffoul. Current status of PRP manufacturing requirements & european regulatory frameworks: Practical tools for the appropriate implementation of PRP therapies in musculoskeletal regenerative medicine. Bioengineering 2023; 10(3), 292.
[84] M Rath, J Spinnen, LD Kuhrt, E Priglinger, P Seika, D Runge, S Schubring, D Laue, M Wickert, M Erdem, W Ertel and LK Shopperly. Platelet-rich plasma - a comprehensive review of isolation, activation, and application. Acta Biomaterialia 2025; 204, 52-75.
[85] SJ Awan, N Idrees, T Asif, T Munawar, M Munawar and N Naeem. Platelet-rich plasma-derived exosomes as novel frontier in regenerative medicine. Current Cancer Therapy Reviews 2025. https://doi.org/10.2174/0115733947399426250911115018
[86] Y Hou, X Wen, L Zhou, and X Fang. The value of platelet-rich plasma-derived extracellular vesicles in modern medicine. Annals of Medicine 2023; 55(2), 2287705.
[87] N Faramarzi, IK Yazdi, M Nabavinia, A Gemma, A Fanelli, A Caizzone, LM Ptaszek, I Sinha, A Khademhosseini, JN Ruskin and A Tamayol. Patient-specific bioinks for 3D bioprinting of tissue engineering scaffolds. Advanced Healthcare Materials 2018; 7(11), e1701347.
[88] K Yi, Q Li, X Lian, Y Wang and Z Tang. Utilizing 3D bioprinted platelet-rich fibrin-based materials to promote the regeneration of oral soft tissue. Regenerative Biomaterials 2022; 9, rbac021.
[89] Z Li, X Zhang, T Yuan, Y Zhang, C Luo, J Zhang, Y Liu and W Fan. Addition of platelet-rich plasma to silk fibroin hydrogel bioprinting for cartilage regeneration. Tissue Engineering Part A 2020; 26, 886-895.
[90] JM DeLong, RP Russell and AD Mazzocca. Platelet-rich plasma: The PAW classification system. Arthroscopy : The Journal of Arthroscopic & Related Surgery: Official Publication of the Arthroscopy Association of North America and the International Arthroscopy Association 2012; 28(7), 998-1009.
[91] T Buchheit, CL Hunt, JS Eldrige, Y Eshraghi and D Souza. Product characteristics should be reported in all biological therapy publications. Regional Anesthesia & Pain Medicine 2022; 47, 449.
[92] S Ren, H Wang, S Ma, J Zhou, J Zhai, Y Zhu, S Chen, S Chen, K Jia, W Xu and Y Zhou. New strategy of personalized tissue regeneration: When autologous platelet concentrates encounter biomaterials. Frontiers in Bioengineering and Biotechnology 2023; 11, 1297357.
[93] A Grzelak, A Hnydka and J Higuchi. Recent achievements in the development of biomaterials improved with platelet concentrates for soft and hard tissue engineering applications. International Journal of Molecular Sciences 2024; 25(3), 1525.
[94] M Niemann, M Ort, L Lauterbach, M Streitz, A Wilhelm, G Grütz, FN Fleckenstein, F Graef, A Blankenstein, S Reinke, U Stöckle, C Perka, GN Duda, S Geißler, T Winkler and T Maleitzke. Individual immune cell and cytokine profiles determine platelet-rich plasma composition. Arthritis Research & Therapy 2023; 25(1), 6.
[95] F Islam, SB Javdan, MR Lewis, JD Craig, H Wu and TL Deans. Programming megakaryocytes to produce engineered platelets for delivering non-native proteins. Communications Biology 2025; 8(1), 638.
[96] T Burnouf, P Strengers and MP Busch. Beyond transfusion: Platelet-derived therapeutic products as a new frontier for blood establishments and transfusion medicine. Vox Sanguinis 2026; 121(2), 114-123.