
Trends
Sci.
2026;
23(7):
12748
Synergistic Enhancement of Nisin Production through Metabolic and Regulatory Engineering of Lactococcus lactis ATCC 11454: Thermal and pH Stability and Application in Food Model System
Yuli Haryani1,2,*, Hanan Hasan3,4, Rudi Hendra1,2, Benni Iskandar5, Yulia Andriana1, Yuwanda1, Eliza Khoirunnisa1, Nadrah Abdul Halid3, Suriana Sabri6,
Mahmud Ab Rashid Nor-Khaizura3 and Muhammad Asyraf Md Hatta7
1Department of Chemistry, Faculty of Mathematics and Natural Sciences, Universitas Riau,
Riau 28293, Indonesia
2Centre of Biological Innovation for Regenerative and Natural Applications, Universitas Riau,
Riau 28293, Indonesia
3Department of Food Science, Faculty of Food Science and Technology, Universiti Putra Malaysia,
Selangor 43400, Malaysia
4Laboratory of Halal Science Research, Halal Research Product Institute, Universiti Putra Malaysia,
Selangor 43400, Malaysia
5Department of Pharmaceutical Technology, Sekolah Tinggi Ilmu Farmasi, Riau 28293, Indonesia
6Department of Microbiology, Faculty of Biotechnology and Biomolecular Sciences, Universiti Putra Malaysia, Selangor 43400, Malaysia
7Department of Agriculture Technology, Faculty of Agriculture, Universiti Putra Malaysia, Selangor 43400, Malaysia
(*Corresponding author’s e-mail: [email protected])
Received: 22 November 2025, Revised: 13 December 2025, Accepted: 23 December 2025, Published: 20 February 2026
Abstract
Nisin is one of the most studied bacteriocins due to its broad antimicrobial spectrum and Generally Recognized as Safe status. In this study, nisRK was overexpressed in lactic acid-deficient Lactococcus lactis ATCC 11454, generating a recombinant Lactococcus lactis ATCC 11454 Δldh carrying pMG36e-P8-nisRK (ldhΔnisRKOE), to enhance nisin biosynthesis. Growth kinetics (OD600 monitoring) and antimicrobial activity agar (well diffusion assay) were compared among wildtype, ldh∆, nisRKOE, and double mutant strains. The ldhΔnisRKOE strain exhibited significantly higher antimicrobial activity than all other strains, with inhibition zones increased by up to 107% against Gram-positive and 78% against Gram-negative bacteria, compared with the wildtype. Thermal and pH stability tests showed that antimicrobial activity was well preserved at low temperatures (0 -4 °C) and across a wide pH range (2 - 10), with optimal performance near pH 6. Activity declined by 26% - 34% following heat treatment at 100 - 121 °C. Application in raw chicken demonstrated that CFS from the engineered strain slowed microbial growth and extended shelf life. After seven days at 4 °C, treated samples maintained lower bacterial counts (8.41 ± 0.02 log CFU/g) compared with controls (9.10 ± 0.07 log CFU/g). At 30 °C, untreated samples spoiled within 72 h, while treated meats remained stable until day 7. These findings highlight that combining metabolic redirection with regulatory overexpression can substantially improves nisin yield and antimicrobial activity. L. lactis ATCC 11454-ldhΔnisRKOE strain holds strong potential as a microbial cell factory for bacteriocin-based applications in food preservation and biotechnology.
Keywords: Antimicrobial activity, Engineered lactic acid bacteria, Nisin, Preservation, Stability
Introduction
Nisin, a ribosomally synthesized and post-translationally modified peptide (RiPP), is one of the most studied bacteriocins due to its broad antimicrobial spectrum and its Generally Recognized as Safe (GRAS) status [1,2]. Produced by Lactococcus lactis, nisin has been used in the food industry for a long time as a natural preservative. It helps inhibit spoilage microorganisms and foodborne pathogens [3,4]. In addition to food preservation, nisin and other lantibiotics are gaining interest in biomedical and biotechnological applications, including combating antibiotic-resistant pathogens and serving as model systems to study complex biosynthetic pathways [5].
The NisRK two-component system precisely controls how nisin is produced. NisK is a histidine kinase that is linked to the membrane and detects nisin outside the cell. NisR is the response regulator that turns on the transcription of the genes that make nisin [6,7]. Studies have demonstrated that overexpressing nisRK can increase the production of nisin by increasing the transcription of the structural gene nisA and other biosynthetic parts [8,9]. Another study reported that the simultaneous overexpression of nisA, nisRK, and nisFEG in L. lactis LS01 resulted in the highest enhancement of bioactive nisin production relative to the parent strain [10]. However, despite its essential regulatory role, the potential of nisRK overexpression as a strategy for yield enhancement has not been thoroughly examined in metabolically modified L. lactis strains.
Several genetic strategies have been explored to increase nisin yield, including redirecting carbon fluxes, overexpressing biosynthetic genes, and optimising fermentation processes [11-14]. For instance, introducing multicopy plasmids with nisRK increased the expression of the nisin pre-peptide, thereby boosting production [8,9]. Nevertheless, while these strategies improve yield individually, the synergistic effects of regulatory engineering (nisRK overexpression) and metabolic rewiring are inadequately elucidated, leaving an important gap in understanding how these approaches may collaboratively optimise bacteriocin biosynthesis.
Our earlier study addressed this question by applying CRISPR/Cas9-mediated deletion of the ldh gene in L. lactis ATCC 11454 and other LAB strains. Knockout ldh redirected carbon flux away from lactic acid formation towards the production of bacteriocin-like inhibitory substances (BLIS) and other antimicrobial metabolites. The resulting ldhΔ strains exhibited significantly higher antimicrobial activity, up to 78% greater than their wild-type counterparts, particularly against Gram-negative pathogens [15]. This metabolic rewiring not only reduced acidification of the culture environment but also promoted the biosynthesis of secondary metabolites with potential biotechnological applications.
In the present study, we extend our previous work by introducing nisRK overexpression into the ldhΔ background of L. lactis ATCC 11454. The aim of the dual alteration is to synergistically combine metabolic flux redirection with enhanced transcriptional activation of the nisin biosynthetic cluster. By comparing wild-type, ldhΔ, nisRKOE, and ldhΔ-nisRKOE strains, we examine how these genetic modifications affect cell growth, antimicrobial yield, and the physicochemical stability of the resulting bacteriocins at different pH and temperature levels. Finally, recognizing that bacteriocins may behave differently in complex food matrices, we assess their performance in a meat system, using raw chicken as a highly perishable model food prone to microbial spoilage. This method directly connects metabolic and regulatory engineering to functional results that are important for preserving food.
Materials and methods
Bacterial strains, plasmids, and culture media
Table 1 lists the plasmids and bacterial strains used in this study for nisRK overexpression. Lactococcus lactis ATCC 11454 was grown in M17 broth (Oxoid) at 30 °C. The genetically engineered L. lactis ATCC 11454-Δldh strain, generated from our previous study [15], was cultured in M17 broth with 5 µg/mL erythromycin and 10 µg/mL chloramphenicol at 30 °C. Escherichia coli TOP10 was grown in Luria-Bertani broth (Merck, Darmstadt, Germany) at 37 °C, with the addition of 150 µg/mL erythromycin for propagation of E. coli TOP10 harboring the nisRK plasmid. Pathogenic bacteria used as indicator strains in the antibacterial assays, namely Listeria monocytogenes ATCC 7644, Staphylococcus aureus ATCC 6538, Bacillus subtilis ATCC 19659, Vibrio parahaemolyticus ATCC 17802, and Escherichia coli ATCC 8739, were grown overnight in Luria-Bertani broth at 37 °C. Cell growth was monitored by measuring the optical density at 600 nm (OD600) using a UV/Vis spectrophotometer (Techcomp UV1000, Thailand).
Table 1 Strains and plasmids used in nisRK overexpression.
Strain or plasmid |
Relevant characteristics |
Reference or source |
Strains |
|
|
E. coli TOP10 |
Cloning host |
Laboratory stock |
L. lactis ATCC11454 |
Nisin-producing strain |
Laboratory stock |
L. lactis ATCC11454- Δldh |
ldh knockout mutant of L. lactis ATCC11454 |
Laboratory stock |
L. paracasei K2003- Δldh |
ldh knockout mutant of L. paracasei K2003 |
Laboratory stock |
L. plantarum Y1002- Δldh |
ldh knockout mutant of L. plantarum Y1002 |
Laboratory stock |
L. rhamnosus MK2003- Δldh |
ldh knockout mutant of L. rhamnosus MK2003 |
Laboratory stock |
E. faecalis GM1003- Δldh |
ldh knockout mutant of E. faecalis GM1003 |
Laboratory stock |
Plasmids |
|
|
pMG36e |
L. lactis constitutively expressed plasmid, Er |
Laboratory stock |
pMG36e-P8 |
pMG36e derived, P32 promoter replaced by the P8 promoter |
Laboratory stock |
nisRK-pMG36e-P8 |
pMG36e-P8 derived, carrying nisRK |
This study |
Construction of the nisRK-expressing plasmid
Plasmid P8-pMG36e was employed as the vector backbone to drive nisRK expression under the control of the strong constitutive P8 promoter [15]. Propagation was carried out in E. coli TOP10. The nisRK sequence (derived from the complete Lactococcus lactis subsp. lactis nisin biosynthetic gene cluster (GenBank: HM219853.1) was synthesized and assembled into P8-pMG36e via PstI and SphI restriction sites to generate nisRK-P8-pMG36e. The recombinant plasmid was verified by DNA sequencing (GenScript, China) and by restriction enzyme digestion (PstI and SphI in a 37 °C water bath for 40 min).
Preparation of electrocompetent cells and transformation
L. lactis ATCC11454 and L. lactis ATCC11454-ldh∆ were made electrocompetent [15]. Immediately prior to transformation, 40 µL of each electrocompetent strain was mixed with 5 µL of nisRK-P8-pMG36e, and incubated on ice for at least 10 min. Each suspension was transferred to an ice-cooled electroporation cuvette (2-mm electrode gap) and subjected to a single electrical pulse at 2.3 kV using a Bio-Rad Xcell Gene Pulser (Bio-Rad Laboratories). The setup had a capacitance of 25 µF and a resistance of 200 Ω.
The electrotransformed cells were diluted in 960 µL of ice-cold SGM17 supplemented with 20 mM MgCl2 and 2 mM CaCl2, and incubated at 30 °C. After 4 h, 200 µL of the L. lactis ATCC11454 suspension was spread plated onto antibiotic-free M17 agar and M17 agar with 5 µg/mL erythromycin; besides, 200 µL of the L. lactis ATCC11454-ldh∆ suspension was spread plated onto antibiotic-free M17 agar and M17 with 5 µg/mL erythromycin and 10 µg/mL chloramphenicol. The plates were incubated for 24 - 48 h at 30 °C. Transformants were picked, stored as glycerol stocks, and tested for antimicrobial activity assay. As a control, and to verify nisRK overexpression, nisRK-P8-pMG36e plasmid vector was also transformed into our collection of ldh-deficient non nisin-producer LAB including L. paracasei K2003-ldh∆, L. plantarum Y1002-ldh∆, L. rhamnosus MK2003-ldh∆, and E. faecalis GM1003-ldh∆. The effect of nisRK overexpression on the growth of the nisRK-overexpressed (nisRKOE) and double mutant (ldh∆nisRKOE) strains of L. lactis ATCC11454 was determined by measuring OD600 of and medium pH over a 48-hours incubation. Meanwhile, the impact of the genetic modification on nisin production was examined by comparing the antimicrobial activity of the engineered strains with that of the wildtype (WT).
Preparation of cell-free supernatant (CFS) and antimicrobial activity assay
The
wildtype, ldhΔ,
nisRKOE,
and ldhΔ-nisRKOE
strain of L.
lactis
ATCC11454 were grown overnight up to 9
log CFU/mL. An aliquot (10%) of each suspension was added to new MRS
broth, incubated for
24 h at 35 °C, centrifuged (4.000×g,
15 min), and filter sterilised using 0.22
 m
membrane.
m
membrane.
Antimicrobial activity was assessed using the agar well diffusion assay against Gram-positive and Gram-negative pathogens. Wells (6 mm diameter) were punched in the Mueller Hinton agar plates seeded with 100 µL of the test pathogens (OD600 0.1). Each well was filled with 100 µL of CFS from the test strains, 100 µL of MRS broth (negative control), or 100 µL of ampicillin 150 µg/ml (positive control). Plates were incubated at 37 °C for 24 h, after which inhibition zone diameter (mm) surrounding the wells were measured with a caliper. The strength of activity was classified as strong for inhibition zone diameters ≥ 20 mm, moderate for diameters ranging from 10 to 19 mm and weak for diameters less than 9 mm [16].
Thermal and pH stability assessment of nisin antimicrobial activity
The effect of temperature on antimicrobial activity of nisin was evaluated by incubating 1 mL of CFS in eppendorf tube at different temperatures: 0, 4, 35, 60 and 100 °C for 2 h, and at 121 °C at 15 lbs (autoclave) for 15 min [17]. For pH stability assessment, the pH of CFS (initial pH 6.2) was adjusted to values ranging from 2 to 13 by adding either 1 M NaOH or 1 m HCl, followed by overnight incubation at 35 °C. After each treatment, the antimicrobial activity of nisin was assessed against S. aureus ATCC 6538, according to the method previously described in Section 2.4.
Stability of nisin antimicrobial activity in raw chicken meat
Chicken meat preparation
Raw chicken breast samples were purchased from a local supermarket (Jumbo Mart, Pekanbaru city, Indonesia), stored in an icebox, and transported on ice and cut into pieces of approximately 2.0 cm. Experimental samples were treated with CFS derived from ldhΔ-nisRKOE strain, while the negative control was treated with sterile MRS broth by immersion in the respective treatment solutions (1:1, w/v) for 30 min, and then drained for 10 mins. To evaluate CFS stability in chicken meat environment under different storage conditions, 2 sets of treated and control samples were prepared: One stored at room temperature and the other under refrigeration. Samples were collected after 0, 1, 3, 5 and 7 days of incubation for physicochemical and microbiological analysis.
pH analysis
Following incubation, 5 g of chicken meat from each treatment group was homogenized in 45 mL of distilled water. The pH of the resulting suspension was measured using a calibrated pH meter at 0, 1, 3, 5, and 7 days of storage.
Bacterial cell count analysis
The total viable cell count was determined to evaluate nisin antimicrobial effectivity and microbiological quality of treated/untreated chicken meat samples, following the method of Jo et al. [18] with minor modification. Briefly, 25 g of each sample was homogenised in 225 mL Buffered Peptone Water (pH 7.2), serially diluted up to 10-7, and 100 μL of each suspension was spread plated onto Plate Count Agar. Plates were incubated at 37 ℃ for 24 h, after which colonies were enumerated and expressed as colony forming unit (CFU).
Data analysis
Antimicrobial activity was measured as the diameter of the inhibition zone, reported as mean ± standard deviation from triplicate experiments. One-way ANOVA followed by Tukey’s post hoc test (p < 0.05) were used to assess for statistical significance with Minitab 19 (Pennsylvania, USA). A two tailed t-test (p < 0.05) was applied to compare the antimicrobial activity of the nisin control (without chicken meat) and the treatment group (with chicken meat).
Results and discussion
Construction of recombinant plasmids and the effect of nisRK overexpression
Figure 1 shows the digestion profile of the recombinant plasmid nisRK_P8-pMG36e (5569 bp) on 1% agarose gel. The observed fragment sizes matched the expected pattern, confirming successful insertion of the nisRK gene into the P8-pMG36e vector. Sequencing confirmed correct nisRK insertion, and transformants displayed stable growth on selective media. No growth differences were observed between control strains harboring the empty plasmid and those carrying nisRK, suggesting that plasmid maintenance imposed no major metabolic burden.
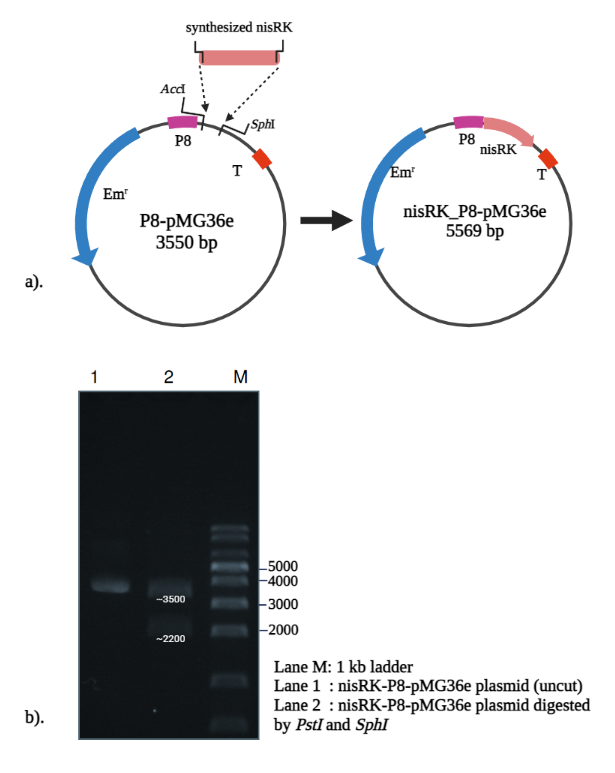
Figure 1 Construction of nisRK plasmid from P8-pMG36e as a vector backbone. (a) Insertion of synthesized nisRK gene, and (b) RE digestion to verify the construct.
The generated plasmid was introduced into both WT and ldh∆-strains of L. lactis ATCC11454, generating nisRK-overexpressed (nisRKOE) and double mutant (ldh∆nisRKOE). Figure 2(a) depicts medium pH changes over 48 h incubation. The pH of nisRKOE and ldh∆nisRKOE-strains was not much different from the initial pH of the growth medium throughout the fermentation period. In WT cells, pH dropped to 4.7 due to lactic acid accumulation, reaching pH 4.6 after 48 h, which inhibited growth. By contrast, the nisRKOE and ldhΔnisRKOE strains maintained a stable pH of ~ 6.0, close to their optimal growth range (6.3 - 6.9). This suggests that nisRK overexpression redirected carbon flux away from lactate production.
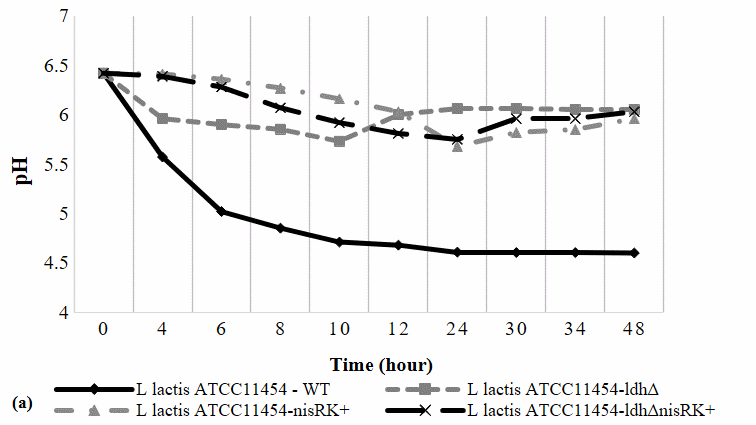
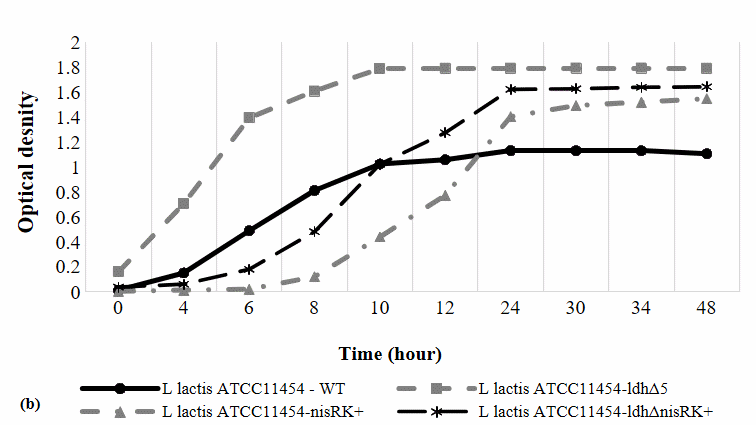
Figure 2 The effect of nisRK overexpression in the cell growth and pH of the growth medium.
The OD600 growth profile (Figure 2(b)) shows distinct growth dynamics among the strains. The nisRKOE, and ldhΔ-nisRKOE strains exhibited a noticeably prolonged lag phase lasting approximately 0 - 6 h, likely due to the additional metabolic load associated with nisRK overexpression. Despite the delayed onset, the nisRKOE strains entered a robust exponential phase between 6 - 12 h and ultimately achieved comparable or higher final cell densities, indicating that nisRK overexpression did not exert a toxic effect. The WT and ldhΔ strains reached the stationary phase earlier, at approximately 10 h, whereas the nisRKOE and ldhΔnisRKOE strains reached stationary phase later, around 24 h. Since bacteriocin synthesis typically peaks during the late logarithmic and early stationary phases [19], sample collection was aligned with these physiological stages: At 10 h for WT and ldhΔ strains, and at 24 h for the nisRKOE and ldhΔnisRKOE strains [20], ensuring representative metabolite production profiles.
Antimicrobial activity of engineered L. lactis ATCC 11454
Nisin is known for its potent antimicrobial properties, low cell toxicity, and slow resistance development. Its biosynthesis is tightly regulated by the two-component NisRK system, which balances production and immunity [21,22]. The impact of overexpressed nisR and nisK proteins in nisin production was determined using an antibacterial assay against representative Gram-positive and Gram-negative indicator strains (Table 2). The results demonstrated clear differences among strains. Based on the established classification criteria, the WT strain exhibited only weak inhibition. The ldhΔ strain produced noticeably larger zones (up to 45% wider than WT), which can be classified as moderate inhibition, suggesting that redirecting pyruvate flux away from lactate enhances nisin synthesis. The nisRKOE strain produced intermediate improvements as they showed intermediate to strong inhibition, indicating that upregulating the regulatory module alone positively affects bacteriocin output, albeit not to the same extent as ldh deletion. The strongest inhibition was recorded for the double mutant (ldhΔnisRKOE), which produced very strong and consistently well-defined clear zones, exhibited the strongest activity, increasing inhibition by 65% against V. parahaemolyticus, 78% against E. coli, and over 100% against B. subtilis and S. aureus. These trends indicate that although nisRK overexpression enhances nisin production compared to WT, deletion of ldh alone has a stronger effect, and the combination of both modifications yields the highest inhibition levels.
Table 2 Antimicrobial activity of the metabolically engineered L. lactis ATCC 11454.
No |
LAB strains |
Inhibition zone (mm) |
||||
Gram-positive |
Gram-negative |
|||||
SA *) |
BS |
LM |
EC |
VP |
||
1 |
L. lactis-WT |
8.17 ± 0.29d |
9.3 ± 0.58c |
10 ± 0c |
7.7±0.58b |
9.7±0.58c |
2 |
L. lactis-ldhΔ |
14.2 ± 0.29b |
15.7 ± 0.58b |
14.2 ± 0.29ab |
13.3±0.58a |
14.8±0.29a |
3 |
L. lactis-nisRKox |
13 ± 0c |
15 ± 0b |
13.3 ± 0.5b |
13±0a |
13.5±0.5b |
4 |
L. lactis -ldhΔ- nisRKox |
16.8 ± 0.29a |
19.3 ± 0.58 a |
14.5 ± 0a |
13.7 ± 0.58a |
16 ± 0.5a |
a-d : Means in the same column with different lowercase letters differed significantly (p ˂ 0.05)
*): SA = Staphylococcus aureus ATCC 6538; Bacillus subtilis ATCC 19659; LM = Listeria monocytogenes ATCC 7644; EC = Escherichia coli ATCC 8739; VP = Vibrio parahaemolyticus ATCC 17802.
In the WT strain, strong acidification suppressed growth. Furthermore, under low pH conditions, the energy of sugar metabolism is mostly utilised to produce different proteins associated with different acid tolerance mechanisms [23]. These conditions affected the lower flux toward nisin production, explaining its weak antimicrobial activity. By comparison, the near-neutral pH of nisRKOE strain supports their normal growth rate during the late stationary phase of fermentation. Since nisin biosynthesis is regulated in a cell-density-dependent manner [8], the elevated growth rate of nisRKOE strain promoted higher nisin production and consequently enhanced inhibitory activity against the tested organisms.
Additionally, at this pH, nisRKOE cells obtained a greater amount of energy from sugar catabolism, as they did not require the synthesis of various proteins associated with acid tolerance mechanisms [23]. This extra energy might be redirected toward other metabolites, including the synthesis of serine and threonine, the precursors for nisin biosynthesis. NisB dehydrates serine and threonine residues within the core peptide during post-translational modification to generate dehydroalanine and dehydrobutyrine, respectively. NisC subsequently catalyses intramolecular addition reactions between cysteine residues and the double bonds of dehydroalanine and dehydrobutyrine, resulting in the formation of lanthionine and methyllanthionine. The modified nisin, still containing the leader peptide, is transported by NisT, after which the N-terminal leader sequence is cleaved by NisP, an extracellular serine protease, releasing the mature and active nisin (21). Thus, increased precursor availability could enhance the production of mature nisin, which in turn induces transcription of the nisin biosynthetic gene cluster through the NisR-NisK system [24,25].
Furthermore, the overexpression of nisR and nisK which are central regulators of nisin biosynthesis, might be responsible for the increased antibacterial activity. Nisin-inducible promoters, including the nisZBTCIPRK and nisFEG operons, are activated by the nisRK system [26]. Thus, overexpression of these genes holds strong potential for improving overall nisin yield. The ldhΔnisRKOE strain exhibits a significantly greater inhibitory effect in the cell-free supernatant (CFS) due to the synergistic contribution of ldh disruption and nisRK overexpression.
The double mutant strain demonstrated the most significant enhancement in inhibition against the Gram-positive S. aureus ATCC 6538 and B. subtilis ATCC 19659, showing increases of 106.12% and 107.14%, respectively. In comparison to the WT, significant increases were also observed against Gram-negative isolates, including E. coli ATCC 8739 (78.26%), and V. parahaemolyticus ATCC 17802 (65.52%). Nisinʼs potency against Gram-positive bacteria was greater than that against Gram-negative bacteria, as anticipated, in accordance with its established mechanism of action [15]. Nonetheless, the enhanced activity of the double mutant against both groups underscores the potential of integrating metabolic and regulatory engineering as a novel strategy to optimise bacteriocin production.
Thermal and pH Stability of Nisin antimicrobial activity
The stability of CFS from L. lactis ATCC 11454-ldhΔnisRKOE under different thermal and pH conditions was evaluated to determine the robustness of nisin in variable environments.
Thermal stability
The antimicrobial activity of the CFS was effectively maintained during storage at low temperatures (0 and 4 °C), as demonstrated in Figure 3(a). The inhibition zones remained close to those of the untreated control (n-CFS; pH 6.0; 17.9 mm). In contrast, heating resulted in a substantial decrease in activity: the inhibition zone was reduced by 26% at 100 °C (11.63 mm) and by up to 34% at 121 °C (10.3 mm). This suggests that activity was more effectively retained at 100 °C than at 121 °C, indicating a threshold effect when exposure exceeded the boiling point. This thermal stability is in accordance with published reports that describe nisin as capable of maintaining antimicrobial activity after short-term high-temperature treatments, such as autoclaving and high-temperature short-time pasteurisation [27]. The sharper decline observed above 100 °C supports previous findings that nisin remains stable between 0 °C and 100 °C but loses activity under harsher conditions [28].
Disrupting ldh alters cellular metabolism, which can affect the post-translational modifications, folding and maturation processes, as well as the local chemical environment of nisin, thereby altering its stability. Nisin is a lantibiotic, a small antimicrobial peptide comprised of unusual amino acids, including lanthionine and dehydroalanine. Compared to larger proteins, its small size, simple structure, and strong covalent bonds confer a higher resistance to denaturation. Furthermore, the presence of lanthionine and β-methyllanthionine residues results in the formation of thioether bridges, which provide a rigid and stable conformation that prevents denaturation and unfolding at high temperatures [29].
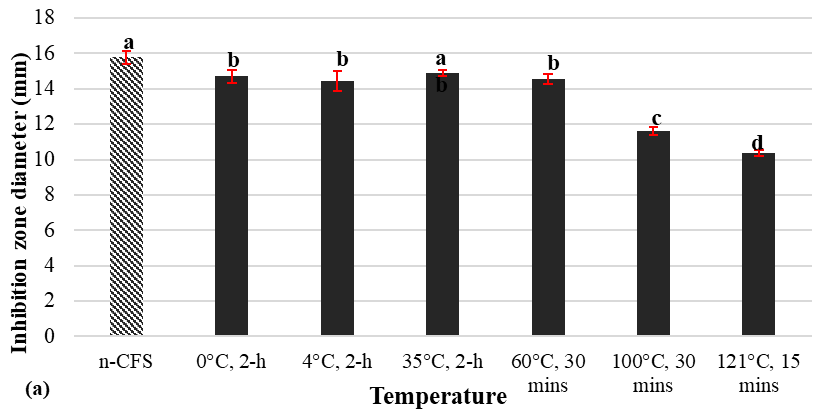
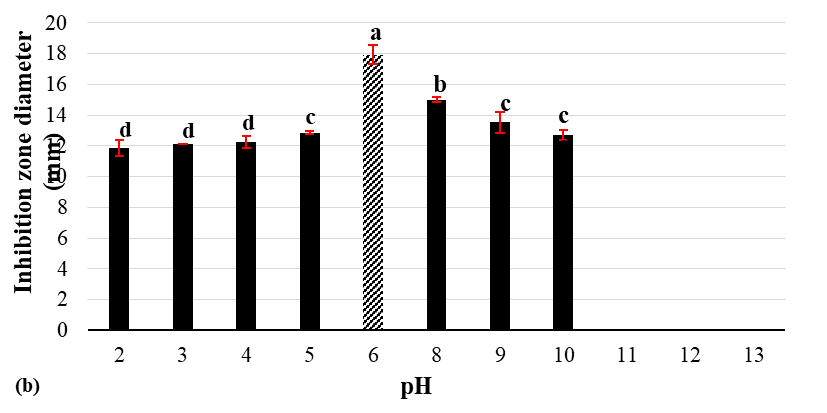
Figure 3 Effect of temperature (a) and pH (b) on antimicrobial activity of CFS from L. lactis ATCC 114545-ldhΔnisRKOE.
pH stability
The effect of pH variation on CFS activity is presented in Figure 3(b). Antimicrobial activity was retained over a wide range from pH 2 to pH 10, although the inhibition zones varied significantly. The highest activity was observed at pH 6 (17.9 mm), corresponding to the fermentation medium, consistent with the optimal conditions for nisin synthesis [29]. At strongly acidic pH (1 - 3), inhibition zones were notably smaller (9 - 12 mm), indicating partial inactivation under extreme acidity. Activity improved at pH 4 - 5 (12 - 13 mm) and remained moderate under alkaline conditions at pH 8 -10 (13 - 15 mm), although inhibition declined relative to neutral pH. Interestingly, the relatively high activity at pH 10 contrasted with earlier reports that alkaline environments reduce nisin solubility due to nucleophilic attack on dehydro residues by hydroxide ions, deprotonated amines, and hydroxyl groups, leading to aggregation [30]. This observation suggests that nisin and other metabolites present in the CFS from the double mutant strain may act protectively, minimizing modification of the nisin dehydro residues and thereby maintaining antimicrobial activity.
Stability and antimicrobial performance of nisin in raw chicken meat
Antimicrobial stability during storage
The antimicrobial activity of CFS against S. aureus ATCC 29213 was evaluated during seven days of storage at 4 °C, both in the absence and presence of chicken meats (Table 3). Over the storage period, a gradual decline in inhibition zones was observed in both treatments. The decline in antibacterial activity of nisin-producing LAB CFS during storage at 4 °C can be attributed to aggregation, reduced solubility, conformational changes, and chemical degradation. Nisin molecules tend to aggregate at low temperatures, reducing their capacity to inhibit bacterial growth and compromising their stability [31]. At 4 °C, the solubility also decreases, which further diminishes the efficacy. Antimicrobial activity is dependant upon structural integrity, however, conformational modifications affecting bioactivity and stability may results from low temperatures. Although refrigeration delays degradation, it does not entirely prevent it; nisin undergoes chemical breakdown over time, even when stored in cold conditions [32,33].
Table 3 Changes in nisin/BLIS antimicrobial activity during seven days of storage at 4 °C in the presence and absence of chicken meats.
Storage time (Days) |
Inhibition zones (mm)*) |
|
Without chicken meat |
With chicken meats |
|
0 |
14.67 ± 0.15a |
12.93 ± 0.12a |
1 |
14.45 ± 0.30ab |
12.43 ± 0.18ab |
2 |
14.10 ± 0.05b |
12.37 ± 0.21b |
3 |
12.40 ± 0.05c |
11.45 ± 0.22c |
4 |
12.38 ± 0.08c |
11.08 ± 0.29c |
5 |
12.32 ± 0.15c |
9.58 ± 0.13d |
6 |
12.20 ± 0.10c |
9.20 ± 0.17de |
7 |
12.13 ± 0.13c |
8.82 ± 0.20e |
*Mean values ± standard deviation from three replicates. Superscript letters within the same column indicate no significant differences (α = 0.05) according to Tukey’s test.
The antimicrobial activity of the control CFS, which was devoid of chicken meat, decreased moderately by 17.31%. However, the reduction in the chicken meat environment was more pronounced, reaching 31.79%. Significant reductions (p < 0.05) were observed in the control after day 2 and in the chicken meat samples after day 1, indicating that the inactivation process was faster when interacting with the meat matrix. The sharper decline in antimicrobial activity within the chicken meat environment can be attributed to physicochemical interactions between nisin and food components. Proteins and lipids in meat may bind or inactivate nisin through hydrophobic and electrostatic interactions, thereby decreasing its solubility and bioavailability. Previous studies have reported that fat globules adsorb nisin, thereby decreasing its capacity to penetrate bacterial membranes [34,35]. Furthermore, lipids may establish protective barriers around bacterial cells, thereby protecting them from the pore-forming action of nisin [36,37]. These results are consistent with previous observations that nisin stability and activity decrease more swiftly in complex food matrices than in simple laboratory media. This is attributed to the presence of proteins, fats, and other macromolecules [38]. The present results highlight the importance of considering food composition when applying bacteriocins as natural preservatives. Although nisin retained partial activity after seven days in chicken meats, the reduced inhibition zone suggests the need for formulation strategies such as encapsulation or combination with other antimicrobials to improve stability and efficacy in meat products.
Total bacterial counts and shelf-life extension
Table 4 illustrates the impact of CFS coating on the total bacterial counts of chicken meats stored at 4 and 30 °C. In comparison to untreated controls, bacterial proliferation was significantly reduced in CFS-treated samples at refrigeration temperature (4 °C). The untreated controls reached 8.58 ± 0.03 log CFU/g by day 3, whereas the treated samples remained significantly lower at 7.52 ± 0.07 log CFU/g (p < 0.05). After seven days, bacterial counts in controls were 9.10 ± 0.07 log CFU/g, while in treated samples they were 8.41 ± 0.02 log CFU/g. This represents a 0.69 log reduction that can be attributed to the CFS. These findings indicate that CFS application effectively slowed microbial proliferation, thereby extending the microbiological shelf life of chicken meats in chilled storage.
Table 4 Total bacterial counts of chicken fillets with and without CFS treatment during storage at 4 °C and 30 °C
Storage time (Days) |
Total bacterial count (log CFU/g)1) |
|||
Stored at 4 °C |
Stored at room temperature (~30 °C) |
|||
Untreated sample |
CFS-treated sample |
Untreated sample |
CFS-treated sample |
|
0 |
7.82 ± 0.07a |
7.16 ± 0.11a |
7.82 ± 0.07a |
7.16 ± 0.11a |
1 |
7.87 ± 0.02a |
7.38 ± 0.01b |
8.64 ± 0.03b |
7.96 ± 0.04b |
3 |
8.58 ± 0.03b |
7.52 ± 0.07b |
NA2) |
8.84 ± 0.10c |
5 |
8.87 ± 0.06c |
7.97 ± 0.00c |
NA |
9.78 ± 0.02d |
7 |
9.10 ± 0.07d |
8.41 ± 0.02d |
NA |
10.18 ± 0.01e |
1) Mean values ± standard deviation from three replicates. Superscript letters within the same column indicate no significant differences (α = 0.05) according to Tukey’s test.
2) NA = data not available (chicken fillet samples spoiled).
At room temperature (30 °C), spoilage occurred much more rapidly. The bacterial counts of control meats increased significantly, reaching 8.64 ± 0.03 log CFU/g after only 24 h. By day 3, visible deterioration was observed, rendering further measurements unavailable. On the other hand, CFS-treated samples demonstrated delayed microbial growth, rising from 7.16 ± 0.11 log CFU/g on day 0 to 7.96 ± 0.04 log CFU/g on day 1, and progressively reaching 10.18 ± 0.01 log CFU/g by day 7. The treatment effectively extended the utilisation of the product in comparison to the untreated controls, despite the fact that microbial growth was not entirely suppressed. In the untreated controls, spoilage occurred within 72 h, indicated by the strong odor, surface slime formation, and loss of structural integrity. At this stage, the meat matrix became highly degraded due to the activity of spoilage-associated microorganisms, and excessive microbial overgrowth, resulting in confluent colonies on agar plates and colony numbers exceeding the countable range. Because the samples no longer met the criteria for reliable enumeration, further quantitative measurements were not possible. This type of matrix breakdown and uncountable growth are typical indicators of advanced spoilage in high-protein perishable foods such as poultry.
The activity of nisin present in the CFS is responsible for the antimicrobial effect that was observed. Nisin inhibits cell wall biosynthesis by binding to lipid II and forming cavities in bacterial membranes, resulting in the leakage of cytoplasmic contents [39,40]. Nevertheless, the progressive deterioration of efficacy over time underscores the intricacy of implementing bacteriocins in actual food systems. For instance, glutathione, naturally present in meat, can inactivate nisin via glutathione S-transferase [35].
The relationship between nisin antibacterial activity and bacterial proliferation in chicken meats stored at 4 °C is illustrated in Figure 4. The inhibition zone was 12.93 mm in diameter at day 0, and the total bacterial count was 7.2 log CFU/g. Over the storage period, inhibition zones slowly decreased, dropping to 8.9 mm by day 7, whereas bacterial counts increased steadily to 8.4 log CFU/g. This inverse relationship indicates that the gradual loss of antimicrobial activity directly corresponded with the rise in bacterial populations.
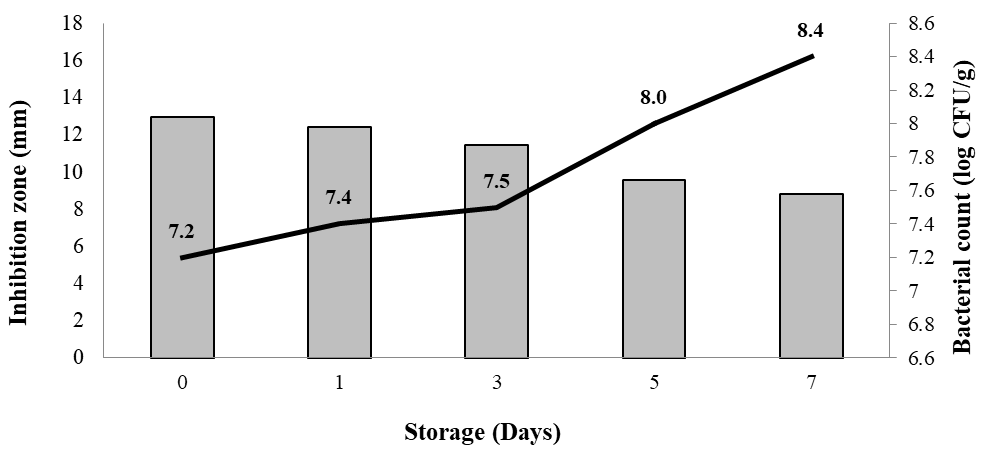
Figure 4 Relationship between nisin/BLIS antibacterial activity and bacterial proliferation in chicken fillets stored at 4 °C
The reduction in inhibition zones suggests a progressive decline in the bioactivity of nisin. Several mechanisms may contribute to this loss, including adsorption of nisin to meat proteins and lipids, enzymatic inactivation by endogenous compounds such as glutathione, and decreased bioavailability as storage progressed [34,35]. As a result, bacterial growth was only partially suppressed, particularly after day 3 when inhibition zones fell below 12 mm. Despite this decline, CFS treatment effectively slowed bacterial growth compared with untreated controls (Table 4).
pH changes during storage
The pH of chicken meats fluctuated over the storage period, as shown in Figure 5. Meats stored at 4 °C consistently exhibited lower pH values than those at 30 °C, mirroring microbial counts. At both 4 and 30°C, pH increased up to day 5, correlating with bacterial growth and the accumulation of biogenic amines produced by amino acid decarboxylation [41,42]. The pH increase was more pronounced at 30 °C, which is consistent with the higher rate of microbial proliferation. By day 7, a slight pH decrease was observed, which is likely the result of the accumulation of acidic metabolites and the onset of spoilage [43].
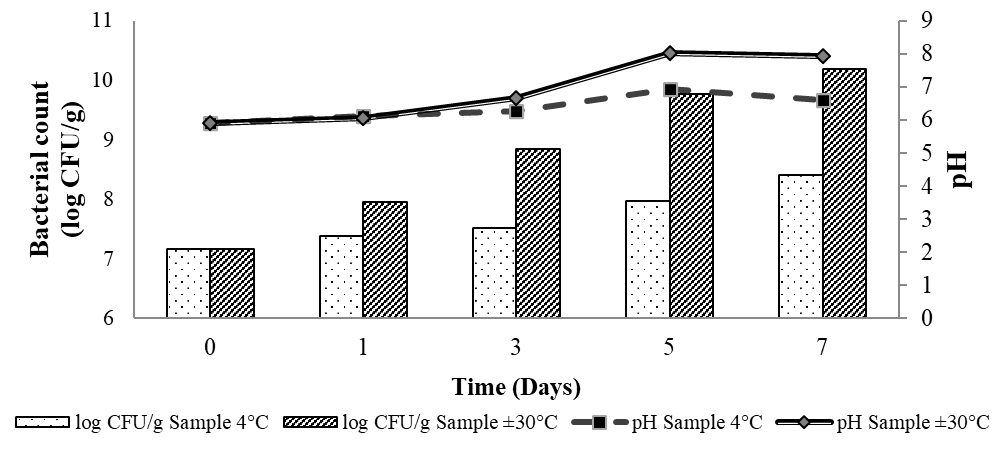
Figure 5 Changes in bacterial count and pH of chicken meats during storage at 4 and 30 °C
Overall, chicken meats coated with CFS demonstrated a longer shelf life and delayed microbial growth in comparison to uncoated controls, particularly under refrigeration. Given that raw chicken is highly susceptible to contamination by pathogens such as Salmonella spp., Campylobacter spp., Staphylococcus aureus, Escherichia coli, and Listeria spp. [44], these findings underscore the potential of nisin as a biopreservative in meat systems. Additionally, the broader antimicrobial spectrum observed with the engineered L. lactis ATCC 11454 ldhΔnisRKOE compared with wild-type strains emphasizes its promise for food applications. Beyond direct application, nisin can also be incorporated into active packaging systems, where it has been shown to inhibit microbial growth and extend shelf life in dairy and meat products [45,46].
Conclusions
This study demonstrates that combining lactate dehydrogenase knockout with nisRK overexpression in Lactococcus lactis ATCC 11454 significantly enhances nisin biosynthesis and antimicrobial activity. The double mutant (ldhΔnisRKOE) exhibited superior inhibitory effects against both Gram-positive and Gram-negative bacteria, outperforming strains engineered with single modifications. Stability tests confirmed that nisin produced by the engineered strain retained substantial antimicrobial activity across a wide pH spectrum and under short-term heat stress, highlighting its robustness for industrial applications.
Importantly, application of the engineered CFS to raw chicken meats revealed its capacity to reduce microbial proliferation and extend shelf life, particularly under refrigeration. These findings bridge laboratory-scale engineering with practical food preservation outcomes, underscoring the value of integrating metabolic and regulatory approaches to optimize bacteriocin production. Future studies should focus on scaling up fermentation, characterizing molecular interactions in complex food matrices, and exploring delivery systems such as active packaging to further expand the utility of engineered nisin-producing strains in food safety and preservation.
Acknowledgements
Fundamental Research Grant 2025, Directorate of Research and Community Service (DPPM), Ministry of Higher Education, Science, and Technology, Indonesia, under Contract No. 19608/UN19.5.1.3/AL.04/2025, through The Institute for Research and Community Service of Universitas Riau, Riau 28293, Indonesia.
Declaration of generative AI in scientific writing
Generative AI tools were used solely for editorial assistance, including grammar correction, phrasing improvements, and formatting refinement. All intellectual content, experimental design, data interpretation, and scientific conclusions were developed entirely by the authors. After using these tools, the authors carefully reviewed and thoroughly edited the entire manuscript to ensure accuracy, clarity, and proper scientific representation. The authors take full responsibility for the content, integrity, and conclusions presented in this publication.
CRediT author statement
Yuli Haryani: Supervision; Conceptualization; Software; Original draft preparation. Hanan Hasan: Supervision; Conceptualization; Methodology; Data Curation. Rudi Hendra: Formal analysis; Funding acquisition. Benni Iskandar: Visualization; Project administration. Yulia Andriana: Resources; Investigation. Yuwanda: Investigation; Validation. Eliza Khoirunnisa: Resources; Investigation. Nadrah Abdul Halid: Writing - Reviewing and Editing. Suriana Sabri: Methodology; Validation. Mahmud Ab Rashid Nor-Khaizura: Writing - Reviewing and Editing. Muhammad Asyraf Md Hatta: Formal analysis; Validation.
References
[1] AEC Olivos, A Linares‐Castañeda, LJ Corzo‐Ríos, M Márquez‐Lemus and C Jiménez‐Martínez. Nisin as a food preservative: Physicochemical, sensory properties and antimicrobial activity in Mexican tomato sauce. International Journal of Food Science & Technology 2024; 59(5), 3020-3030.
[2] L Yuan, S Wu, K Tian, S Wang, H Wu and J Qiao. Nisin-relevant antimicrobial peptides: Synthesis strategies and applications. Food & Function 2024; 15(19), 9662-9677.
[3] GH Choi, JII Fugaban, CM Dioso, JEV Bucheli, WH Holzapfel and SD Todorov. Antimicrobial peptides (Bacteriocins) produced by Lactococcus lactis and Pediococcus pentosaceus strains with activity against clinical and food-borne pathogens. Probiotics and Antimicrobial Proteins 2025; 17(3), 1171-1192.
[4] MP Mokoena, CA Omatola and AO Olaniran. Applications of lactic acid bacteria and their bacteriocins against food spoilage microorganisms and foodborne pathogens. Molecules 2021; 26(22), 7055.
[5] AM Charest, E Reed, S Bozorgzadeh, L Hernandez, NV Getsey, L Smith, A Galperina, HE Beauregard, HA Charest, M Mitchell and MA Riley. Nisin inhibition of gram-negative bacteria. Microorganisms 2024; 12(6), 1230.
[6] C García-Ausencio, F Guzmán-Chávez, R Rodríguez-Sanoja and S Sánchez. Promiscuity of lanthipeptide enzymes: New challenges and applications. World Journal of Microbiology and Biotechnology 2025; 41(8), 1-20.
[7] OP Kuipers, MM Beerthuyzen, PGGA de Ruyter, EJ Luesink and WM de Vos. Autoregulation of nisin biosynthesis in Lactococcus lactis by signal transduction. Journal of Biological Chemistry 1995; 270(45), 27299-27304.
[8] CI Cheigh, H Park, HJ Choi and YR Pyun. Enhanced nisin production by increasing genes involved in nisin Z biosynthesis in Lactococcus lactis subsp. lactis A164. Biotechnology Letters 2005; 27(3), 155-160.
[9] CI Cheigh, HJ Choi, H Park, SB Kim, MC Kook, TS Kim, JK Hwang and YR Pyun. Influence of growth conditions on the production of a nisin-like bacteriocin by Lactococcus lactis subsp. lactis A164 isolated from kimchi. Journal of Biotechnology 2002; 95(3), 225-235.
[10] ZJ Ni, XY Zhang, F Liu, M Wang, RH Hao, PX Ling and XQ Zhu. Effect of co-overexpression of nisin key genes on nisin production improvement in Lactococcus lactis LS01. Probiotics and Antimicrobial Proteins 2017; 9(2), 204-212.
[11] C Liu, L Zhang, H Liu and K Cheng. Delivery strategies of the CRISPR-Cas9 gene-editing system for therapeutic applications. Journal of Controlled Release 2017; 266, 17-26.
[12] NHM Rasid, NA Halid, AS Ai-Lian, S Sabri, MHA Rahim, N Saari and H Hasan. Rerouting the metabolic pathway in Lactococcus lactis ATCC 11454 to improve nisin productivity by disrupting lactate dehydrogenase (ldh). Food Biotechnology 2024; 38(2), 180-194.
[13] R Portieles, H Xu, F Chen, J Gao, L Du, X Gao, CB Nordelo, Q Yue, L Zhao, NP Gonzales, RS Bermudez and O Borrás-Hidalgo. Bioengineering of a Lactococcus lactis subsp. lactis strain enhances nisin production and bioactivity. PLoS One 2023; 18(4), 0281175.
[14] YF Zhang, SY Liu, YH Du, WJ Feng, JH Liu and JJ Qiao. Genome shuffling of Lactococcus lactis subsp. lactis YF11 for improving nisin Z production and comparative analysis. Journal of Dairy Science 2014; 97(5), 2528-2541.
[15] Y Haryani, NA Halid, SG Guat, MAR Nor-Khaizura, MA Hatta, S Sabri, S Radu and H Hasan. Efficient metabolic pathway modification in various strains of lactic acid bacteria using CRISPR/Cas9 system for elevated synthesis of antimicrobial compounds. Journal of Biotechnology 2024; 395, 53-63.
[16] Y Haryani, NA Halid, GS Guat, NK MAR, A Hatta, S Sabri, S Radu and H Hasan. Characterisation, molecular identification, and antimicrobial activity of lactic acid bacteria isolated from selected fermented foods and beverages in Malaysia. FEMS Microbiology Letters 2023; 370, 023.
[17] NL Sidek, M Halim, JS Tan, S Abbasiliasi, S Mustafa and AB Ariff. Stability of bacteriocin-like inhibitory substance (BLIS) produced by Pediococcus acidilactici KP10 at different extreme conditions. BioMed Research International 2018; 2018, 5973484.
[18] DM Jo, SK Park, F Khan, MG Kang, JH Lee and YM Kim. An approach to extend the shelf life of ribbonfish fillet using lactic acid bacteria cell-free culture supernatant. Food Control 2021; 123, 107731.
[19] MP Zacharof and RW Lovitt. Bacteriocins produced by lactic acid bacteria: A review. APCBEE Procedia 2012; 2, 50-56.
[20] JP van Pijkeren and RA Britton. High-efficiency recombineering in lactic acid bacteria. Nucleic Acids Research 2012; 40(10), 76.
[21] D Field, PD Cotter, RP Ross and C Hill. Bioengineering of the model lantibiotic nisin. Bioengineered 2015; 6(4), 187-192.
[22] JN Hansen and PWE Sandine. Nisin as a model food preservative. Critical Reviews in Food Science and Nutrition 1996; 34(1), 69-93.
[23] C Wang, Y Cui and X Qu. Mechanisms and improvement of acid resistance in lactic acid bacteria. Archives of Microbiology 2018; 200(2), 195-201.
[24] A Frelet-Barrand. Lactococcus lactis, an attractive cell factory for the expression of functional membrane proteins. Biomolecules 2022; 12(2), 180.
[25] XX Zhou, WF Li, GX Ma and YJ Pan. The nisin-controlled gene expression system: Construction, application and improvements. Biotechnology Advances 2006; 24(3), 285-295.
[26] J Wu, M Zang, S Wang, B Zhao, J Bai, C Xu, Y Shi and X Qiao. Nisin: From a structural and meat preservation perspective. Food Microbiology 2023; 111, 104207.
[27] S Yang, X Li, W Cang, D Mu, S Ji, R Wu and J Wu. Combined ε-polylysine and thermosonication treatment to inactivate Bacillus cereus and application in pasteurized milk. Food Bioscience 2023; 55, 102896.
[28] GM de Almeida, CMN Mendonça, A Converti and RP de Souza Oliveira. Kinetic and thermodynamic parameters of nisin thermoinactivation. Journal of Food Engineering 2020; 280, 109986.
[29] S Khelissa, NE Chihib and A Gharsallaoui. Conditions of nisin production by Lactococcus lactis subsp. lactis and its main uses as a food preservative. Archives of Microbiology 2021; 203(2), 465-480.
[30] W Liu and JN Hansen. Some chemical and physical properties of nisin, a small-protein antibiotic produced by Lactococcus lactis. Applied and Environmental Microbiology 1990; 56(8), 2551-2558.
[31] T Musafija-Jeknic. 1996, Structural analysis of thermally inactivated nisin. PhD Thesis. Oregon State University, Corvallis, USA.
[32] P Holcapkova, ZK Raskova, M Hrabalikova, A Salakova, J Drbohlav and V Sedlarik. Isolation and thermal stabilization of bacteriocin nisin derived from whey for antimicrobial modifications of polymers. International Journal of Polymer Science 2017; 2017(1), 3072582.
[33] S Senan, HA El-Aal, R Dave and A Hassan. Production and stability of nisin in whey protein concentrate. LWT - Food Science and Technology 2016; 71, 125-129.
[34] DS Jung, FW Bodyfelt and MA Daeschel. Influence of fat and emulsifiers on nisin efficacy against Listeria monocytogenes in fluid milk. Journal of Dairy Science 1992; 75(2), 387-393.
[35] D Lahiri, M Nag, B Dutta, T Sarkar, S Pati, D Basu, ZA Kari, LS Wei, S Smaoui, KW Goh and RR Ray. Bacteriocin: A natural approach for food safety and food security. Frontiers in Bioengineering and Biotechnology 2022; 10, 1005918.
[36] X Liu, H Yao, X Zhao and C Ge. Biofilm formation and control of foodborne pathogenic bacteria. Molecules 2023; 28(6), 2432.
[37] BD Needham and MS Trent. Fortifying the barrier: The impact of lipid A remodeling on bacterial pathogenesis. Nature Reviews Microbiology 2013; 11(7), 467-481.
[38] JL Webber, R Namivandi-Zangeneh, S Drozdek, KA Wilk, C Boyer, EHH Wong, BH Bradshaw-Hajek, M Krasowska and DA Beattie. Incorporation and antimicrobial activity of nisin Z within carrageenan/chitosan multilayers. Scientific Reports 2021; 11(1), 1690.
[39] I Wiedemann, E Breukink, C van Kraaij, OP Kuipers, G Bierbaum, B de Kruijff and HG Sahl. Specific binding of nisin to the peptidoglycan precursor lipid II combines pore formation and inhibition of cell wall biosynthesis for potent antibiotic activity. Journal of Biological Chemistry 2001; 276(3), 1772-1779.
[40] H Zhou, J Fang, Y Tian and XY Lu. Mechanisms of nisin resistance in Gram-positive bacteria. Annals of Microbiology 2013; 64(2), 413-420.
[41] M Yıldırım-Yalçın, H Sadıkoğlu and M Şeker. Characterization of edible film based on grape juice and cross-linked maize starch and its effects on the storage quality of chicken breast fillets. LWT 2021; 142, 111012.
[42] W Wójcik, M Łukasiewicz and K Puppel. Biogenic amines: Formation, action and toxicity - a review. Journal of the Science of Food and Agriculture 2021; 101(7), 2634-2640.
[43] Y Lei, Y Zhang, Y Cheng, J Huang and M Huang. Monitoring and identification of spoilage-related microorganisms in braised chicken with modified atmosphere packaging during refrigerated storage. Food Science and Human Wellness 2023; 12(1), 28-34.
[44] DK Wardhana, AEP Haskito, MTE Purnama, DA Safitri and S Annisa. Detection of microbial contamination in chicken meat from local markets in Surabaya, East Java, Indonesia. Veterinary World 2021; 14(12), 3138-3143.
[45] EE Popa, AC Miteluț, M Râpă, PA Popescu, MC Drăghici, M Geicu-Cristea and ME Popa. Antimicrobial active packaging containing nisin for preservation of animal-origin products: An overview. Foods 2022; 11(23), 3820.
[46] L Settier-Ramírez, G López-Carballo, R Gavara and P Hernández-Muñoz. Broadening the antimicrobial spectrum of nisin-producing Lactococcus lactis subsp. Lactis to Gram-negative bacteria by means of active packaging. International Journal of Food Microbiology 2021; 339, 109007.