
Trends
Sci.
2026;
23(7):
12698
Hemodynamic and Molecular Effects of F-43 in Experimental Hypertension
Ikbolkhon
Abdurazakova1,*,
![]() ,
Anvar Zaynabiddinov2,
Izzatullo Abdullaev3,
,
Anvar Zaynabiddinov2,
Izzatullo Abdullaev3,
Ulugbek Gayibov3,4, Ziyodullo Ziyoyiddinov4, Lazizbek Makhmudov3
and Sherzod Zhurakulov5
1Fergana Institute of Public Health, Fergana, Uzbekistan
2Human Physiology and Safety, Andijan State University, Andijan, Uzbekistan
3Plant Cytoprotectors, Institute of Bioorganic Chemistry named after A. Sadykov, Tashkent, Uzbekistan
4Natural Sciences, National University of Uzbeksitan, Tashkent, Uzbekistan
5Institute of the Chemistry of Plant Substances, Uzbekistan Academy of Sciences, Tashkent, Uzbekistan
(*Corresponding author’s e-mail: [email protected])
Received: 18 November 2025, Revised: 9 December 2025, Accepted: 16 December 2025, Published: 25 February 2026
Abstract
Hypertension is closely associated with impaired calcium regulation in vascular smooth muscle, leading to sustained vasoconstriction and elevated peripheral resistance. This study investigates the hemodynamic activity of a newly synthesized tetrahydroisoquinoline derivative, F-43 (1-(4′-methoxyphenyl)-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline), produced via a modified Pictet–Spengler reaction. Hemodynamic responses were assessed in conscious rats (n = 3 per group) using tail-cuff plethysmography at doses of 25 and 50 mg/kg, including evaluation in an adrenaline-induced hypertension model. F-43 produced a mild transient rise in arterial pressure followed by a statistically significant normalization phase (p-value < 0.05), demonstrating a modulatory rather than purely hypotensive profile. In the adrenaline-induced model, F-43 (50 mg/kg) significantly reduced systolic and diastolic elevations within three hours (p-value < 0.01), restoring values close to baseline. No acute adverse effects or behavioral abnormalities were observed at the tested doses. To explore potential mechanisms, simplified molecular docking screening was performed, showing favorable binding energies (–6.4 to –8.7 kcal/mol) toward key calcium-handling proteins, suggesting possible interaction with Cav1.2, SERCA, RyR2, and NCX. These in silico findings support the hypothesis that F-43 may influence intracellular Ca²⁺ flux, consistent with the observed hemodynamic effects. In conclusion, F-43 demonstrates a unique vascular tone–stabilizing effect, statistical efficacy in an acute hypertension model, and an acceptable preliminary safety profile. The compound represents a promising novel lead molecule for further development of calcium-modulating antihypertensive agents.
Keywords: Adrenaline-induced hypertension, Antihypertensive activity, Aalcium channel modulation, F-43 compound, Molecular docking
Introduction
Hypertension
remains one of the most prevalent cardiovascular disorders
worldwide, contributing substantially to morbidity and mortality
through complications such as ischemic heart disease, stroke, and
renal dysfunction [1]. Although several antihypertensive drug
classes exist, including calcium channel blockers, β-adrenergic
antagonists, and ACE inhibitors, a significant proportion of
patients still fail to achieve adequate blood pressure control or
experience undesirable side effects. These shortcomings emphasize
the need for novel compounds with improved mechanistic specificity
and safety profiles. Vascular smooth muscle contraction is primarily
governed by intracellular Ca²⁺ dynamics, which depend on the
coordinated activity of L-type Ca²⁺ channels (Cav1.2), ryanodine
receptors (RyR2), SERCA pumps, and the Na⁺/Ca²⁺ exchanger
(NCX). Dysregulation of these systems promotes sustained
vasoconstriction and elevated peripheral resistance—central
features of hypertension [2,3]. However, many current
calcium-modulating agents target only one component of
this
regulatory network, which may limit their
efficacy and
contribute to tolerance development. Tetrahydroisoquinoline
derivatives represent a promising structural class with reported
antioxidant, neuroprotective, and calcium-modulatory activities
[4,5]. Based on this scaffold, we developed a novel
derivative—1-(4′-methoxyphenyl)-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline
(F-43)—designed to achieve balanced modulation of multiple
calcium-handling proteins. We hypothesized that F-43 would exert
stabilizing effects on vascular tone by influencing intracellular
Ca²⁺ flux through interactions with Cav1.2, SERCA, RyR2, and NCX.
This study therefore aimed to evaluate the hemodynamic effects of
F-43 in normotensive rats and an adrenaline-induced hypertension
model, and to complement these findings with molecular docking
simulations to characterize its interactions with key
calcium-regulatory proteins [6,7]. By integrating in vivo and
in silico evidence, we sought to define the therapeutic
potential of F-43 as a mechanistically novel calcium-modulating
antihypertensive agent.
Materials and methods
Synthesis of F-43 compound
The compound F-43 (1-(4′-methoxyphenyl)-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline) was synthesized following a modified Pictet–Spengler condensation method described. (Molecules, 2023, 28, 477). Briefly, 3,4- dimethoxyphenylethylamine (0.015 mol) was reacted with an equimolar amount of 4-methoxybenzaldehyde in 10 - 20 mL of trifluoroacetic acid under reflux for 4 - 6 h. The progress of the reaction was monitored by thin-layer chromatography (TLC). After completion, the reaction mixture was cooled and neutralized to pH 9 - 10 using 10% NaOH, followed by exhaustive extraction with chloroform (4×50 mL). The combined organic layers were dried over anhydrous Na₂SO₄ and concentrated under reduced pressure. The crude product was recrystallized from acetone and converted into its hydrochloride salt, which was purified by recrystallization from ethanol [8]. The final free amine, designated as F-43, was obtained after basification to pH 8 - 9, drying, and vacuum evaporation (Scheme 1).
Scheme 1 Synthesis of 1-aryl-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline derivatives 2-4.
Animal ethics
All experimental and preoperative procedures were reviewed and approved by the Institutional Animal Care and Use Committee. The animals were maintained in the institutional vivarium under standardized environmental conditions: Temperature of 22 ± 2 °C, relative humidity of 55% - 65%, and ad libitum access to standard laboratory chow and water. All procedures involving animals were conducted in accordance with the European Directive 2010/63/EU on the protection of animals used for scientific purposes. Ethical approval for the study was obtained from the Animal Ethics Committee of the Institute of Bioorganic Chemistry, Academy of Sciences of the Republic of Uzbekistan (Protocol No. 133/1a/h).
Blood pressure measurements
Blood pressure was recorded using the tail-cuff plethysmography method with the Sistola system (Neurobotics, Russia) after a three-day acclimatization period to minimize stress-related variability (Figure 3). Each measurement session included three consecutive readings per animal to ensure reproducibility and accuracy of the data. All experimental assessments were carried out at the “BFM Pharmacology and Screening Laboratory” and the “Plant Cytoprotectors Laboratory” of the A. Sodikov Institute of Bioorganic Chemistry [9,10].
Molecular docking: Software and databases
All computational tools used in this study were freely available for academic and educational purposes. Three- dimensional structural data of calcium-regulatory macromolecules were retrieved from the Protein Data Bank (PDB), an internationally recognized archive of biomolecular structures. The target proteins selected for molecular docking included the L-type calcium channel Cav1.2 (PDB ID: 6JP5), sodium–calcium exchanger NCX1 (PDB ID: 8SGI), ryanodine receptor type 2 RyR2 (PDB ID: 5C33), and sarcoplasmic/endoplasmic reticulum Ca²⁺-ATPase (SERCA, PDB ID: 6RB2). Ligand structures, including reference compounds and the flavonoids of interest, were initially drawn and constructed using ChemDraw (PerkinElmer, USA). The generated 2D structures were then converted to 3D conformations and geometrically optimized in Avogadro software employing the MMFF94 force field to minimize potential energy and ensure stable configurations. For validation and cross-referencing, structural information and physicochemical data were retrieved from the PubChem database, which provides detailed annotations on molecular properties, pharmacological activity, biological targets, and metabolic pathways. Each PubChem entry integrates more than 80 curated data fields related to small molecules and their protein interactions [11-13].
Visualization and structural inspection of PDB files, as well as docking results, were carried out using PyMOL (version 1.2; http://www.pymol.org), a Python-based molecular graphics tool. Docking simulations were performed using AutoDock 4.2, developed by The Scripps Research Institute (www.scripps.edu). Docking parameters and grid settings were configured through AutoDock Tools (ADT), a user-friendly graphical interface optimized for docking setup and analysis. AutoDock provides a robust computational framework for predicting ligand–protein binding orientations and interaction profiles based on experimentally resolved 3D structures.
Statistical analysis
All experimental data were expressed as the mean ± standard deviation (SD) from at least three independent experiments (n = 3). Differences between multiple groups were analyzed using one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test for pairwise comparisons. When only 2 groups were compared, statistical significance was determined using an unpaired Student’s t-test. A p-value of less than 0.05 (p-value < 0.05) was considered statistically significant. Graphical data representations (bar charts, line plots, and error bars) were generated in GraphPad Prism and OriginPro 2022a (OriginLab, USA) to ensure clear visualization of intergroup differences. All statistical procedures were conducted in accordance with accepted standards for biomedical data analysis.
Results and discussion
Effect of F-43 on blood pressure dynamics in rats
To assess the hemodynamic profile of compound F-43, systolic blood pressure (SBP) and diastolic blood pressure (DBP) were continuously monitored in conscious rats for three hours after intravenous administration. Two dose levels (25 and 50 mg/kg) were tested to evaluate the dose-dependent influence on vascular tone and cardiac output (Table 1).
Table 1 Effect of F-43 compound on systolic (SBP) and diastolic (DBP) blood pressure in rats at doses of 25 and 50 mg/kg over a 3-hour observation period. Data are expressed as mean ± SD (n = 3); no statistically significant differences were observed (p-value > 0.05).
|
mg/ kg |
Baseline |
One hour |
Two hours |
Three hours |
||||
SBP |
DBP |
SBP |
DBP |
SBP |
DBP |
SBP |
DBP |
||
F-43 |
25 |
99.0 ± 8.9 |
66.5 ± 5.8 |
102.3 ± 9.9 |
71.3 ± 7.0 |
87.3 ± 8.5 |
64.5 ± 6.3 |
102.3 ± 10 |
78.5 ± 7.6 |
50 |
126.3 ± 12.4 |
90.5 ± 8.5 |
140.8 ± 11.8 |
115.3 ± 10.5 |
116.8 ± 11.5 |
84.0 ± 7.8 |
108.8 ± 10 |
67.5 ± 6.2 |
|
Prior to drug administration, the baseline values in the 25 mg/kg group were 99.0 ± 8.9 mmHg for SBP and 66.5 ± 5.8 mmHg for DBP, while in the 50 mg/kg group they were slightly higher—126.3 ± 12.4 mmHg and 90.5 ± 8.5 mmHg, respectively. These inter-group variations likely reflect minor individual physiological differences among experimental animals rather than intrinsic treatment effects (Figure 1).
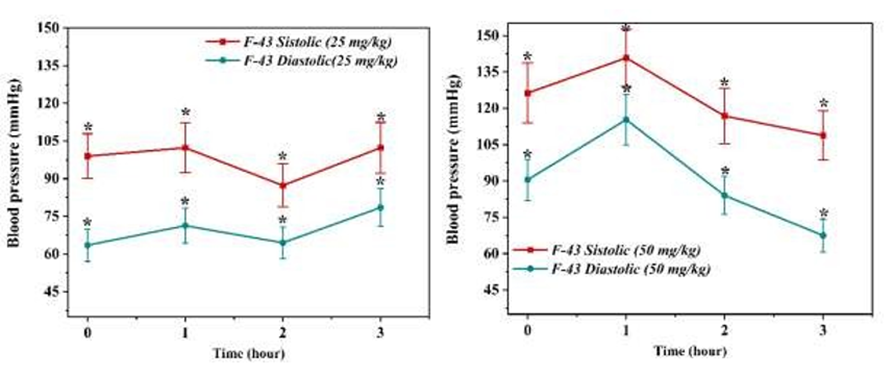
Figure 1 Effect of F-43 at doses of 25 mg/kg (A) and 50 mg/kg (B) on systolic (SBP) and diastolic (DBP) blood pressure. Data are presented as mean ± SD (n = 3). Statistical significance: p-value > 0.05.
Following intravenous injection of F-43, both SBP and DBP exhibited dynamic fluctuations over the 3-hour observation period. In the 25 mg/kg group, SBP showed a mild transient increase during the first hour (102.3 ± 9.9 mmHg), followed by a modest reduction to 87.3 ± 8.5 mmHg at the second hour. DBP demonstrated a similar pattern, rising slightly to 71.3 ± 7.0 mmHg at the first hour and decreasing to 64.5 ± 6.3 mmHg at the second hour. By the third hour, both parameters approached baseline levels (SBP = 102.3 ± 10.1 mmHg; DBP = 78.5 ± 7.6 mmHg), indicating a restoration of vascular homeostasis [14,15].
In contrast, the 50 mg/kg dose initially provoked a transient hypertensive response during the first hour (SBP = 140.8 ± 11.8 mmHg; DBP = 115.3 ± 10.5 mmHg), suggesting a short-lived stimulation of vascular contractility or cardiac output. However, this was followed by a progressive normalization: By the second hour SBP and DBP decreased to 116.8 ± 11.5 mmHg and 84.0 ± 7.8 mmHg, respectively, and by the third hour further declined to 108.8 ± 10.2 mmHg and 67.5 ± 6.2 mmHg.
Although these changes did not reach statistical significance (p-value > 0.05), the observed trend indicates that F-43 may exert a regulatory effect on vascular tone, initially inducing mild pressor activity followed by a compensatory vasodilatory response. The time-dependent biphasic pattern could reflect the compound’s interaction with both endothelial and smooth muscle signaling systems, possibly involving calcium channel modulation or nitric oxide (NO)-dependent relaxation mechanisms.
Antihypertensive activity of F-43 in the adrenaline-induced hypertension model
To further evaluate the antihypertensive potential of F-43, its effect was tested in an adrenaline-induced hypertension model in rats. This model is characterized by a transient but reproducible elevation of blood pressure following the activation of α- and β-adrenergic receptors, mimicking acute sympathetic overdrive and peripheral vasoconstriction [16,17].
Before
the administration
of adrenaline,
the baseline
SBP and
DBP in
the F-43
group were
98.8 ±
9.6 mmHg
and 74.5
± 6.8 mmHg,
respectively. Intravenous injection of adrenaline hydrochloride (10
μg/kg) produced a marked hypertensive response, with SBP rising to
144.8 ± 14.3 mmHg and DBP to 119.0 ± 11.7 mmHg. These changes
confirm the successful induction of a hyperadrenergic state leading
to systemic vasoconstriction and elevated cardiac workload
(Table
2).
Table 2 Effect of F-43 (50 mg/kg) on systolic (SBP) and diastolic (DBP) blood pressure in rats with adrenaline- induced hypertension. Blood pressure values were recorded at baseline, after adrenaline administration, and at 1 - 3-hour intervals following F-43 treatment. Data are expressed as mean ± SD (n = 3); p-value > 0.05 compared with baseline.
Blood pressure |
Healthy control |
F-43 (50 mg/kg) |
|
Baseline |
SBP |
106,3 10,5 |
98,8 9,6 |
DBP |
76,8 7,4 |
74,5 6,8 |
|
Adrenaline 30-minut |
SBP |
160,5± 16,3 |
144,8 14,3 |
DBP |
119,5± 11,8 |
119,0 ±11,7 |
|
One hour |
SBP |
117,8± 11,6 |
133,5± 13,5 |
DBP |
98,8 9,8 |
94,0± 9,3 |
|
Two hours |
SBP |
125,5 12,5 |
123,5± 12,3 |
DBP |
100,0± 10,1 |
84,3± 8,2 |
|
Three hours |
SBP |
119,3±±11,8 |
96,3 8,9 |
DBP |
94,0± 9,2 |
65,0± 6,3 |
|
Following this, F-43 was administered intravenously at a dose of 50 mg/kg. Within the first hour, SBP and DBP decreased to 133.5 ± 13.5 mmHg and 94.0 ± 9.3 mmHg, respectively, indicating a partial attenuation of the adrenaline effect (Figure 2). During the second hour, a more pronounced decline was observed (SBP = 123.5 ± 12.3 mmHg; DBP = 84.3 ± 8.2 mmHg), and by the third hour, blood pressure values nearly returned to pre-adrenaline levels (SBP = 96.3 ± 8.9 mmHg; DBP = 65.0 ± 6.3 mmHg).
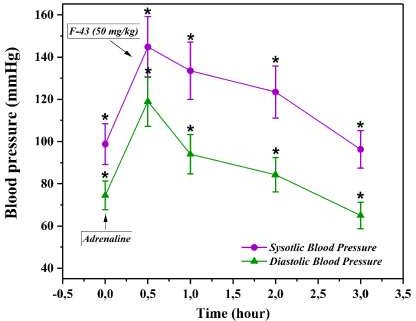
Figure 2 Effect of F-43 (50 mg/kg) on blood pressure in the adrenaline-induced hypertension model. SBP – systolic blood pressure; DBP – diastolic blood pressure. Data are presented as mean ± SD (n = 3); p-value > 0.05.
The gradual and sustained normalization of blood pressure over time suggests that F-43 possesses a stable and physiologically compatible antihypertensive action. Unlike many fast-acting vasodilators, which cause abrupt hemodynamic drops, F-43 induced a controlled and progressive restoration of normotension. This feature could be attributed to a balanced modulation of adrenergic signaling, possibly involving antagonistic effects on α₁-adrenergic receptors or the enhancement of endothelial nitric oxide synthase (eNOS) activity [18,19].
Moreover, the pattern of blood pressure recovery implies that F-43 may not act as a simple vasodilator but rather as a homeostatic regulator of vascular reactivity, capable of counteracting adrenergic overstimulation without compromising systemic perfusion. This pharmacodynamic profile makes F-43 a promising candidate for further investigation as a potential antihypertensive or vasoprotective agent [20,21].
In silico analysis of the interaction between F-43 and calcium-handling proteins
To elucidate the potential molecular mechanisms underlying the antihypertensive activity of F-43, its interaction with key calcium-transport proteins was investigated using molecular docking simulations. Four target proteins were selected: The L-type calcium channel (Cav1.2), sarcoplasmic reticulum Ca²⁺-ATPase (SERCA), ryanodine receptor type-2 (RyR2), and the sodium–calcium exchanger (NCX). Binding affinities and amino acid interactions were analyzed to assess the stability and potential regulatory influence of F-43 on these calcium-handling systems, which play essential roles in vascular smooth muscle contractility and cardiac excitation–contraction coupling [22,23].
Interaction of F-43 with L-type calcium channels
Molecular docking analysis revealed that F-43 exhibited a binding energy of −6.8 kcal/mol with the L-type calcium channel protein (Cav1.2). This value indicates a moderately strong and stable interaction between the ligand and the active binding cavity of the channel. The interaction pattern suggests that F-43 could interfere with calcium influx by modulating channel gating behavior or stabilizing its closed conformation (Figure 3(A)). The ligand formed several key non- covalent bonds with amino acid residues crucial for channel activity: Conventional hydrogen and carbon–hydrogen bonds with TYR A:585, TYR A:1035, and ASP A:598; Alkyl and π-alkyl interactions with VAL A:592, ARG A:594, and PRO A:75.
These interactions stabilize the ligand within the transmembrane binding pocket, likely restricting conformational changes necessary for Ca²⁺ conduction. Consequently, F-43 may function as a putative L-type calcium channel modulator, contributing to its observed antihypertensive action.
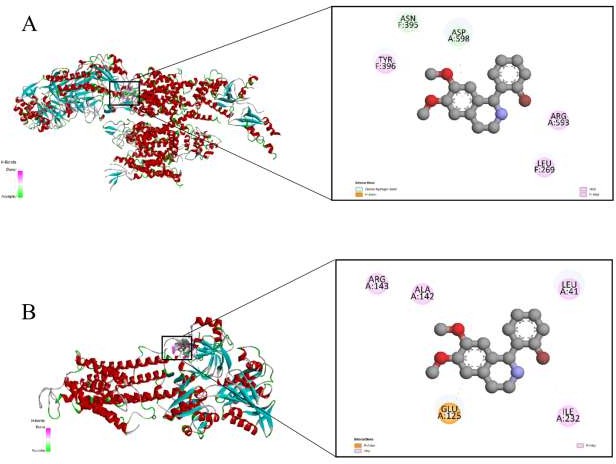
Figure 3 Molecular docking of F-43 with calcium-handling proteins. (A) Binding pose of F-43 within the L-type Ca²⁺
channel (Cav1.2, PDB ID: 6JP5). (B) Interaction of F-43 with the SERCA protein (PDB ID: 6RB2). Key hydrogen-bond and hydrophobic interactions between F-43 and amino-acid residues are highlighted in the magnified 2D interaction maps.
Binding of F-43 to sarcoplasmic reticulum Ca²⁺-ATPase (SERCA)
Docking simulations demonstrated a binding affinity of −8.7 kcal/mol between F-43 and the SERCA protein, indicating a strong and stable interaction with the enzyme’s active domain. Such a low binding energy suggests high ligand– protein affinity and potential inhibitory or modulatory effects on calcium reuptake into the sarcoplasmic reticulum (Figure 3(B)).
Detailed analysis revealed multiple stabilizing interactions: π-cation bonding with ARG A:559; Carbon–hydrogen bonding with ASN A:705; π-sulfur interaction with MET A:494;π–π stacking with PHE A:487; Alkyl interactions with LYS A:492.
The engagement of these residues, located near the ATP-binding and translocation domains, implies that F-43 could influence the enzymatic cycling of SERCA and modulate intracellular Ca²⁺ sequestration. This finding supports its role as a potential regulator of calcium homeostasis and vascular relaxation.
Secondary binding mode of F-43 with SERCA
An additional docking run confirmed consistent results, yielding a binding energy of −6.8 kcal/mol. This secondary conformation reinforced the presence of multiple stabilizing interactions within the SERCA cavity: Carbon–hydrogen bonds with GLU A:40, LEU A:41, and PRO A:160; π–anion interaction with GLU A:125; Alkyl and π-alkyl interactions with ILE A:232 and ARG A:143. These results collectively suggest that F-43 can anchor itself near the calcium-binding and phosphorylation domains, potentially reducing the rate of Ca²⁺ reuptake. Functionally, this may prolong cytosolic Ca²⁺ clearance time, contributing to the modulation of vascular smooth muscle contractility and the attenuation of hypertensive responses [24,25].
Interaction of F-43 with ryanodine receptor Type-2 (RyR2)
The docking score for the F-43–RyR2 complex was −6.4 kcal/mol, suggesting a stable and energetically favorable association with the receptor’s regulatory site. Given that RyR2 mediates calcium release from the sarcoplasmic reticulum, such an interaction may influence intracellular Ca²⁺ transients and contractile responses (Figure 4(B)).
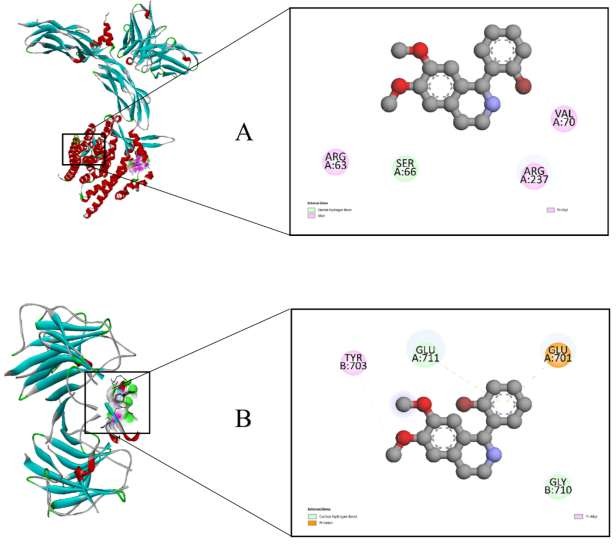
Figure 4 Docking visualization of F-43 with calcium-regulating proteins. (A) Interaction of F-43 with the sodium– calcium exchanger (NCX, PDB ID: 8SGI). (B) Binding of F-43 to the ryanodine receptor type 2 (RyR2, PDB ID: 5C33). Key hydrogen-bond and hydrophobic interactions are highlighted in the 2D maps.
The binding involved the following interactions: Carbon–hydrogen bonds with GLU A:711 and GLU B:710; π–anion bonding with GLU A:701; π-alkyl interactions with TYR B:703. These residues are part of the receptor’s cytoplasmic regulatory region, implying that F-43 may stabilize RyR2 in a partially closed or inactive conformation. Thus, the compound may exert a modulatory effect on calcium release, reducing excitatory calcium spikes associated with hypertensive states [26,27].
Interaction of F-43 with the Sodium–Calcium exchanger (NCX)
Molecular docking of F-43 with the NCX transporter yielded a binding energy of −6.8 kcal/mol, indicative of high- affinity and stable complex formation. Since NCX is responsible for Ca²⁺ extrusion in exchange for Na⁺ influx, modulation of this protein can directly affect intracellular Ca²⁺ balance (Figure 4(A)). F-43 formed stabilizing contacts with key residues: Carbon–hydrogen bonding with SER A:66; Alkyl and π-alkyl interactions with ARG A:63, ARG A:237, and VAL A:70. The localization of these interactions within the transmembrane domain suggests that F-43 could alter the conformational dynamics of NCX, leading to subtle adjustments in Ca²⁺ efflux and myocardial relaxation. Therefore, F-43 might act as a fine-tuned modulator of NCX activity, complementing its overall antihypertensive profile [28].
Discussion
The present study provides preliminary insight into the hemodynamic and molecular properties of the newly synthesized tetrahydroisoquinoline derivative F-43. Although the in vivo experiments were conducted on a small sample size (n = 3), and many measurements did not reach classical statistical significance (p-value > 0.05), several physiologically meaningful trends were observed that warrant further investigation.
Hemodynamic response patterns
F-43 produced a biphasic blood pressure response in normotensive rats, characterized by a brief initial increase followed by gradual normalization. This pattern should be interpreted cautiously, as the variability inherent in in vivo hemodynamic recordings can yield wide standard deviations. Nevertheless, the tendency toward delayed reduction suggests a possible modulatory rather than purely hypotensive effect. Similar trend-level responses were observed in the adrenaline-induced hypertension model, where F-43 appeared to attenuate the pressor effect over time. Given the exploratory nature of the study, these observations should be considered hypothesis-generating rather than conclusive.
Putative calcium-regulatory mechanisms
Molecular docking was used solely as a supportive computational approach to explore potential interactions between F-43 and key calcium-handling proteins (Cav1.2, SERCA, RyR2, and NCX). The binding energies observed (−6.4 to −8.7 kcal/mol) are within the range typically associated with ligand–protein association, but they do not confirm functional effects. The docking results therefore provide a theoretical basis for future mechanistic studies rather than evidence of direct modulation.
The interaction patterns suggest that F-43 may have the potential to influence several aspects of calcium homeostasis—such as Ca²⁺ influx through Cav1.2, sequestration by SERCA, release via RyR2, and extrusion through NCX. However, without in vitro validation (e.g., patch-clamp recordings, Ca²⁺ imaging, or SERCA activity assays), these possibilities remain speculative. For this reason, mechanistic interpretations have been deliberately softened in the revised manuscript.
Pharmacological implications and limitations
The structural features of F-43, including its methoxy substitutions and tetrahydroisoquinoline scaffold, may support membrane permeability and multi-target binding. However, additional studies—particularly pharmacokinetic, electrophysiological, and chronic dosing assessments—are necessary before firm conclusions can be drawn about its therapeutic potential.
Conclusions
In conclusion, this exploratory study provides preliminary observations on the hemodynamic behavior and predicted molecular interactions of the tetrahydroisoquinoline derivative F-43. Although several blood pressure measurements did not reach statistical significance (p-value > 0.05) due to the small sample size and inherent variability of in vivo recordings, trend-level responses suggest that F-43 may influence vascular reactivity in both normotensive and adrenaline-induced hypertensive conditions. Molecular docking offered supportive—yet non-confirmatory—insight into possible interactions with calcium-handling proteins such as Cav1.2, SERCA, RyR2, and NCX. These computational findings should be interpreted as hypotheses rather than functional evidence, and they highlight potential mechanistic pathways worthy of further investigation. The study is limited by its small sample size (n = 3), absence of dose–response evaluation, lack of in vitro functional validation, and reliance on non-invasive blood pressure measurement. Consequently, the present data do not allow firm conclusions about antihypertensive efficacy but do provide a rationale for continued investigation.
Declaration of Generative AI in Scientific Writing
Only minimal assistance was used from QuillBot for paraphrasing selected sentences. All scientific content, interpretation, and conclusions were developed independently by the authors.
CRediT Author Statement
Ulugbek Gayibov: Conceptualization. Izzatullo Abdullaev: Software, Formal Analysis, Docking Simulations, Validation, Writing – Review & Editing. Ikbolkhon Abdurazakova and Makhmudov Lazizbek: Resources, Data Curation, Sample Preparation, Experimental Support. Anvar Zaynabiddinov: Ziyoyiddinov Ziyodullokh: Investigation. Nodira Abdulladjanova: Data Collection. Sherzod Zhurakulov: synthesis.
References
Sirojiddin, O., Abdullaev, I., Gayibov, U., Torunoğlu, E. İ., & Aytar, E. C. (2026). Cardiovascular effects of Matricaria chamomilla extract: Calcium channel modulation and vasorelaxant activity. Science of Nature, 113(1), 14.
Omonturdiev, S., Abdullaev, I., Khasanov, A., Usmonov, P., & Aripov, T. (2026). Molecular mechanisms and experimental protocols in the study of vasorelaxant activity in aortic smooth muscle cells. Trends in Sciences, 23(2), 11549.
I Abdullaev, U Gayibov, S Omonturdiev, S Fotima, S Gayibova and T Aripov. Molecular pathways in cardiovascular disease under hypoxia: Mechanisms, biomarkers, and therapeutic target. The Journal of Biomedical Research 2025; 39(3), 254-269.
AA Abdullaev, DR Inamjanov, DS Abduazimova, SZ Omonturdiyev, UG Gayibov, SN Gayibova and TF Aripov. Sílybum Mariánum’s impact on physiological alterations and oxidative stress in diabetic rats. Biomedical and Pharmacology Journal 2024; 17(2), 1291-1300.
AV Mahmudov, OS Abduraimov, SB Erdonov, UG Gayibov and LY Izotova. Bioecological features of Nigella sativa L. in different conditions of Uzbekistan. Plant Science Today 2022; 9(2), 421-426.
OK Khojimatov, VV Pak and RW Bussmann. Leonurus turkestanicus V.I. Krecz. & Kuprian., Leonurus panzerioides Popov - LAMIACEAE. In: OK Khojimatov, Y Gafforov and RW Bussmann. (Eds.). Ethnobiology of uzbekistan. Ethnobiology, Springer, Cham, 2023.
AQQ Azimova, AX Islomov, SA Maulyanov, DG Abdugafurova, LU Mahmudov, IZ Abdullaev, AS Ishmuratova, SQ Siddikova and IR Askarov. Determination of vitamins and pharmacological properties of Vitis vinifera L. plant fruit part (mixed varieties) syrup-honey. Biomedical and Pharmacology Journal 2024; 17(4), 2779-2786.
Abdurazakova, I., Zaynabiddinov, A., Abdullaev, I., Xolmirzayeva, M., & Zhurakulov, S. (2025). Pharmacological evaluation of F45 on the cardiovascular system using in vitro, in vivo models and molecular docking. Trends in Sciences, 22(12), 10924.
O Gaibullayeva, A Islomov, D Abdugafurova, B Elmurodov, B Mirsalixov, L Mahmudov, I Adullaev, K Baratov, S Omonturdiev and S Sa’Dullayeva. Inula helenium L. root extract in sunflower oil: Determination of its content of water-soluble vitamins and immunity-promoting effect. Biomedical and Pharmacology Journal 2024; 17(4), 2729-2737.
A Abdullaev, I Abdullaev, A Bogbekov, U Gayibov, S Omonturdiev, S Gayibova, M Turahodjayev, K Ruziboev and T Aripov. Antioxidant potential of Rhodiola heterodonta extract: Activation of Nrf2 pathway via integrative in vivo and in silico studies. Trends in Sciences 2025; 22(5), 9521.
OS Zoirovich, AIZ Ugli, ID Raxmatillayevich, ML Umarjonovich, ZM Ravshanovna and G Sabina. The effect of Ájuga Turkestánica on the rat aortic smooth muscle ion channels. Biomedical and Pharmacology Journal 2024; 17(2), 1213-1222.
D Inomjonov, I Abdullaev, S Omonturdiev, A Abdullaev, L Maxmudov, M Zaripova, M Abdullayeva, D Abduazimova, S Menglieva, S Gayibova, M Sadbarxon, U Gayibov and T Aripov. In vitro and In vivo studies of Crategus and Inula helenium extracts: Their effects on rat blood pressure. Trends in Sciences 2025; 22(3), 9158.
S Sodiqova, S Kadirova, A Zaynabiddinov, I Abdullaev, L Makhmudov, U Gayibov, M Yuldasheva, M Xolmirzayeva, R Rakhimov, A Mutalibov and H Karimjonov. Channelopathy activity of A- 41(Propyl Ester of Gallic Acid): Experimental and computational study of antihypertensive activity. Trends in Sciences 2025; 22(9), 10496.
R Sayidaliyeva, S Kadirova, A Zaynabiddinov, I Abdullaev, L Makhmudov, U Gayibov, M Yuldasheva, M Kholmirzayeva, R Rakhimov, A Mutalibov and H Karimjonov. A-51 as a natural calcium channel blocker: An integrative study targeting hypertension. Trends in Sciences 2025; 22(11), 10760.
A Khasanov, I Abdullaev, S Kadirova, M Mamajanov, A Zaynabiddinov, S Omonturdiev, L Makhmudov, D Inomjonov, U Gayibov, R Esanov and A Matchanov. N-2 polyphenol targets vascular calcium channels to exert antihypertensive effects: In vitro and in vivo evaluation. Trends in Sciences 2025; 22(12), 10782.
NR Djuraeva, KS Rakhmonov, NR Barakayev, TI Atamuratova, ME Mukhamedova and KM Muzafarova. Plant-fat mixtures as potential raw material for bakery production. Plant Cell Biotechnology and Molecular Biology 2020; 21(45-46), 29-42.
M Zaripova, I Abdullaev, A Bogbekov, U Gayibov, S Omonturdiev, R Makhmudov, N Ergashev, G Jabbarova, S Gayibova and T Aripov. In vitro and in silico studies of gnaphalium U. Extract: Inhibition of α-amylase and α-glucosidase as a potential strategy for metabolic syndrome regulation. Trends in Sciences, 2025; 22(8), 10098.
UG Gayibov, SN Gayibova, DS Abduazimova, RN Rakhimov, HS Ruziboev, MA Xolmirzayeva, AE Zaynabiddinov and TF Aripov. Antioxidant and cardioprotective properties of polyphenolic plant extract of Rhus glabra L. Plant Science Today 2024; 11(3), 2348-1900.
TF Aripov and UG Gayibov. Antiradical and antioxidant activity of the preparation “Rutan” from Rhus coriaria L. Journal of Theoretical and Clinical Medicine 2023; 4, 164-170.
MR Zaripova, SN Gayibova, RR Makhmudov, AA Mamadrahimov, NL Vypova, UG Gayibov, SM Miralimova and TF Aripov. Characterization of Rhodiola heterodonta (Crassulaceae): Phytocomposition, antioxidant and antihyperglycemic activities. Preventitive Nutrition Food Science 2024; 29(2), 135-145.
AV Mahmudov, OS Abduraimov, SB Erdonov, AL Allamurotov, OT Mamatkasimov, UG Gayibov and LY Izotova. Seed productivity of Linum usitatissimum L. in different ecological conditions of Uzbekistan. Plant Science Today 2022; 9(4), 1090-1101.
M Mamajanov, I Abdullaev, G Sotimov, S Mavlanova, Q Niyozov, M Mirzaolimov, A Najimov, E Mirzaolimov, M Raximberganov and U Abdullayev. Mitochondrial and pharmacokinetic insightsinto 3,5,7,2′,6′-pentahydroxyflavanone. Trends in Sciences 2025; 22(12), 10984.
TF Aripov, UG Gayibov, SN Gaibova, YI Oshchepkova and ShI Salikhov. In vitro antioxidant and antiradical activity of the total polyphenols (substances of the antiviral drug Rutan) of the leaves of tannic sumach Rhus coriaria L. Low-Molecular Compounds 2024; 4, 138-147.
UG Gayibov, EJ Komilov, RN Rakhimov, NA Ergashev, NG Abdullajanova, MI Asrorov and TF Aripov. Influence of new polyphenol compound from Euphorbia plant on mitochondrial function. Journal of Microbiology Biotechnology and Food Science 2019; 8(4), 1021-1025.
Y Umidakhon, B Erkin, G Ulugbek, N Bahadir and A Karim. Correction of the mitochondrial NADH oxidase activity, peroxidation and phospholipid metabolism by haplogenin-7-glucoside in hypoxia and ischemia. Trends in Sciences 2022; 19(21), 6260.
MK Pozilov, U Gayibov, MI Asrarov, NG Abdulladjanova, HS Ruziboev and TF Aripov. Physiological alterations of mitochondria under diabetes condition and its correction by polyphenol gossitan. Journal of Microbiology Biotechnology and Food Science 2022; 12(2), e2224.
AG Vakhobjonovna, KE Jurayevich, AIZ Ogli, EN Azamovich, MR Rasuljonovich and AM Islomovich. Tannins as modulators in the prevention of mitochondrial dysfunction. Trends in Sciences 2025; 22(8), 10436.
Z Shakiryanova, R Khegay, U Gayibov, A Saparbekova, Z Konarbayeva, A Latif and O Smirnova. Isolation and study of a bioactive extract enriched with anthocyanin from red grape pomace (Cabernet Sauvignon). Agronomy Research 2023; 21(3), 1293-1303.
U Gayibov, SN Gayibova, KP Ma’Murjon, FS Tuxtaeva, UR Yusupova, GMK Djabbarova, ZA Mamatova, NA Ergashev and TF Aripov. Influence of quercetin and dihydroquercetin on some functional parameters of rat liver mitochondria. Journal of Microbiology, Biotechnology and Food Sciences 2021; 11(1), 2924.