
Trends
Sci. 2026; 23(7): 12540
First Spatial Resistance Mapping of Fall Armyworm in Jambi, Indonesia
Ratna
Rubiana1,2,
Witjaksono Witjaksono1,
![]() ,
,
Suputa
Suputa1,
![]() and Y. Andi Trisyono1,*,
and Y. Andi Trisyono1,*,
![]()
1Department of Plant Protection, Faculty of Agriculture, Gadjah Mada University, Yogyakarta 55281, Indonesia
2Food Crop Research Center, National Research and Innovation Agency, West Java 16911, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 5 November 2025, Revised: 11 December 2025, Accepted: 18 December 2025, Published: 5 March 2026
Abstract
1)
Introduction: The fall armyworm (Spodoptera frugiperda) has
rapidly expanded across Indonesia since its first detection in 2019
and continues to threaten maize production. Reliance on repeated
applications of chemical insecticides raises concern for emerging
resistance, yet spatial patterns of resistance remain poorly
documented and no spatially integrated resistance monitoring has
been conducted in Indonesia. 2) Materials and methods: Eleven field
populations from Jambi Province and one susceptible laboratory
population were assessed for susceptibility to four commonly used
insecticides: Chlorantraniliprole, deltamethrin, emamectin benzoate,
and spinetoram. Diet-dipping bioassays were performed,
and concentration mortality responses were analyzed using probit
regression to estimate LC₅₀ values and resistance ratios.
Spatial visualization was conducted in RStudio (version 2024.12.0
Build 467) using sf and ggplot2 packages to generate choropleth maps
showing variation in resistance intensity across administrative
regions. 3) Results and discussion: Resistance varied widely among
locations and insecticides. Chlorantraniliprole showed low–moderate
resistance (RR 0.17 - 22.95), highest in Kerinci. Deltamethrin
resistance was widespread (RR 1.26 - 7.45), especially in Jambi City
and Sarolangun. Emamectin benzoate showed extreme resistance (RR
17.44 - 8415.88), with hotspots in Tanjab Barat and Sungai Penuh.
Spinetoram remained mostly effective (RR 0.01 - 3.12). Spatial
patterns indicated localized selection pressure. Spatial patterns
indicated localized selection pressure. 4) Conclusions: The findings
confirm emerging resistance risks and spatially structured
resistance hotspots across Jambi. These results underscore the need
for region-specific insecticide rotation, routine resistance
surveillance, and integration of chemical control with broader IPM
and IRM strategies to maintain long term effectiveness and slow
resistance evolution.
Keywords: Bioassay, Choropleth map, Resistance management, Probit analysis, Fall armyworm
Introduction
The fall armyworm, Spodoptera frugiperda is an invasive lepidopteran pest capable of damaging more than 350 host plants [1]. Since its first report in Indonesia in 2019, infestation has caused severe maize defoliation and significant yield losses [2]. Current field management relies predominantly on repeated applications of insecticides [3], particularly chlorantraniliprole (diamides), deltamethrin (pyrethroids), emamectin benzoate (avermectins), and spinetoram (spinosyns) [4,5]. However, intensive and often unregulated use of these active ingredients has resulted in rapid insecticide resistance development in
several countries, including Brazil, China, India, and parts of Africa [6,7]. Resistance not only reduces field efficacy but also increase production costs and threatens long term crop protection sustainability.
Studies from established fall armyworm regions demonstrate that resistance can evolve rapidly under strong selection pressure and varies across landscapes due to differences in cropping systems, insecticide access, and management behavior [8-10]. Similar warning signs have been observed in Indonesia, including farmer-reported loss of efficacy, increased spray frequency, and heavy dependence on insecticides [3]. However, existing information remains fragmented, largely anecdotal, and rarely supported by standardized laboratory bioassays. Critically, no spatially explicit resistance monitoring has been conducted in Indonesia, making it unclear whether reduced field performance reflects true resistance, misapplication, or environmental variation.
Given Indonesia’s recent invasion history and heavy reliance on chemical control [3], early resistance monitoring is essential. Resistance evolution is rarely uniform across regions; instead, hotspots typically emerge where local selection pressure is highest [11]. Therefore, developing regional susceptibility baselines and spatially resolved resistance profiles is crucial to support evidence-based insect resistance management (IRM) strategies and prevent accelerated resistance development.
This
study aims to quantify susceptibility of fall armyworm
populations in Jambi Province to four commonly applied insecticides
using standardized
diet-dipping bioassays; and generate the
first spatially resolved resistance distribution map to identify
emerging resistance hotspots. We hypothesize that susceptibility
will vary among populations and that resistance will be detectable
in areas with intensive insecticide exposure. By establishing a
provincial-scale resistance baseline, this research provides
essential evidence to support region-specific IRM and integrated
pest management (IPM) strategies and offers an early warning
framework for preventing widespread resistance development in
Indonesia.
Materials and methods
Study area and insect collection
Fall armyworm populations were collected from maize field across 11 administrative regions in Jambi Province, Indonesia, between Mei 2023 and Oktober 2024 (Figure 1). Approximately 100 larvae per field population were sampled. The F₂ generation was used for all bioassays to minimize maternal field effects. A susceptible laboratory reference strain (F₉₅) was obtained from the Pesticide Toxicology Laboratory, Universitas Gadjah Mada. All larvae were reared on fresh maize leaves under controlled laboratory conditions (25 ± 2 °C, 65 ± 10% RH, 14:10 h L:D).
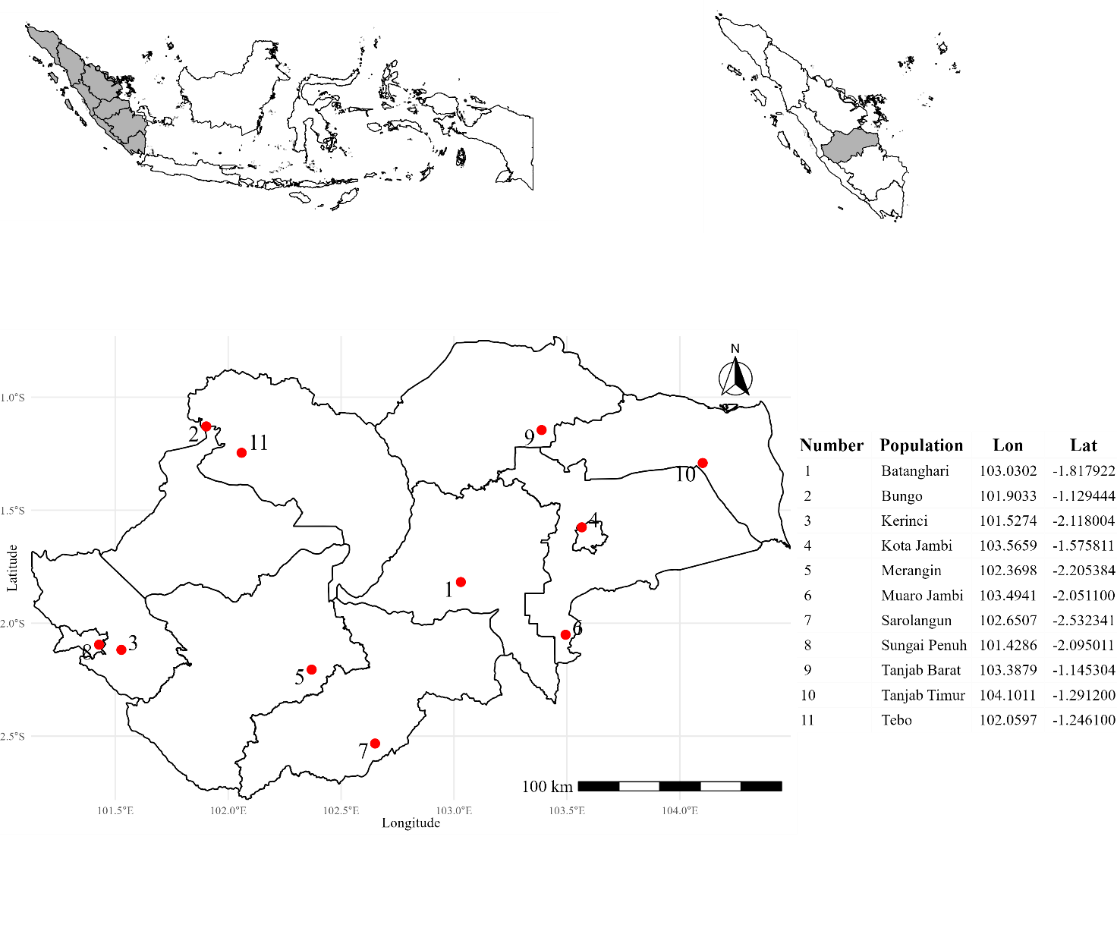
Figure 1 Geographic distribution of collection sites for fall armyworm (Spodoptera frugiperda) in Jambi Province.
Insecticide active ingredients
Four commonly used insecticides were tested: Chlorantraniliprole (diamides), deltamethrin (pyrethroids), emamectin benzoate (avermectins), and spinetoram (spinosyns). Stock solutions were prepared in distilled water, expressed in mg a.i L⁻¹, and serially diluted for bioassays.
Bioassay procedure
Larval
susceptibility was assessed using a
diet-dipping (baby-corn
cob) toxicity bioassay following the IRAC protocol for Spodoptera
species, with adaptations based on established fall armyworm
resistance studies [12]. Bioassays were conducted in two phases. The
preliminary range-finding assay included six concentrations plus a
control, with 10 second-instar larvae per concentration and three
replicates (180 larvae per insecticide), to identify concentration
ranges yielding 15% - 95% mortality. These refined concentrations
were used in the definitive assay, consisting of six concentrations
plus a control with 10 larvae per replicate and five replicates (300
larvae per insecticide×population). Treated baby-corn diet was
replaced every 48 h, and mortality was recorded at 96 h. Control
mortality exceeding 10% was corrected using Abbott’s formula.
Statistical analysis
Concentration–response
data were analyzed using probit regression to estimate LC₅₀ and
LC₉₀ values with 95% fiducial limits [12]. Analyses were
performed using PoloPlus [16] and the ecotox package in
R
(version 4.3.1) [17]. Model fit was evaluated using χ² values
(acceptable when χ²/df < 3) and heterogeneity values
(acceptable when Het. < 3.54). Resistance ratios (RR) were
calculated by comparing LC₅₀ values of field populations with
the susceptible strain. Resistance categories followed established
criteria: RR < 1 (more susceptible), 1 - 5 (low), 5 - 10
(moderate), and >10 (high) [9]. These categories are consistent
with interpretations used in other studies on fall armyworm and
chlorantraniliprole resistance [13,14], in which RR >10 was
considered indicative of a significant reduction in susceptibility.
Differences in generation (F₂ vs. F₉₅) were considered when
interpreting RR and addressed in the Discussion.
Spatial analysis
Resistance patterns (RR values) were visualized using R (packages: Sf, raster, ggplot2). Because the objective was to represent measured values rather than predict unsampled locations, no spatial interpolation (e.g., IDW or kriging) was applied. Outputs were presented as choropleth maps aggregated by administrative boundaries.
Results and discussion
Variation in insecticide toxicity
The toxicity of chlorantraniliprole, deltamethrin, emamectin benzoate, and spinetoram varied across populations (Tables 2 and 3, Figures 2 and 3), indicating heterogeneous susceptibility in fall armyworm across Jambi Province.
Table 2 Range of effective insecticide concentrations against fall armyworm (Spodoptera frugiperda) populations.
Active ingredient |
IRAC Group |
Formulation |
Label concentration (mg L−1) |
Field population concentration (mg L−1) |
Susceptible population concentration (mg L−1) |
Mode of action |
|
||||
Chlorantraniliprole |
28 |
50 SC |
5,000 |
0.4 - 250 |
0.1 - 63 |
Ryanodine receptor modulators |
|
|
|
|
|
Deltamethrin |
3A |
25 EC |
2,500 |
2.500 - 40.000 |
225 - 2.000 |
Sodium channel modulators |
|
|
|
|
|
Emamectin benzoate |
6 |
5,7 WG |
5,700 |
4.56 - 2.850 |
0.29 - 180 |
Glutamate-gated chloride channel (GluCl) activators |
|
|
|
|
|
Spinetoram |
5 |
120 SC |
12,000 |
0.24 - 150 |
0.6 - 375 |
Nicotinic acetylcholine receptor (nAChR) allosteric modulators – Site 1 |
|
|
|
||
Table 3 LC₅₀ values and resistance ratios for four insecticides across Spodoptera frugiperda field populations.
Population |
Chlorantraniliprole |
Deltamethrin |
Emamectin benzoat |
Spinetoram |
||||
LC₅₀ (mg L−1) |
RR |
LC₅₀ (mg L−1) |
RR |
LC₅₀ (mg L−1) |
RR |
LC₅₀ (mg L−1) |
RR |
|
Batanghari |
11.35 |
10.46 |
290 |
1.26 |
14.48 |
42.59 |
9.084 |
2.27 |
Bungo |
14.6 |
13.46 |
412.5 |
1.8 |
17.21 |
50.62 |
8.184 |
2.05 |
Jambi (City) |
0.65 |
0.6 |
1,707.5 |
7.45 |
10.66 |
31.35 |
0.245 |
0.06 |
Kerinci |
24.9 |
22.95 |
662.5 |
2.89 |
5.93 |
17.44 |
0.445 |
0.11 |
Merangin |
0.186 |
0.17 |
1,165 |
5.08 |
86.07 |
253.15 |
7.476 |
1.87 |
Muaro Jambi |
0.6 |
0.55 |
540 |
2.36 |
14.48 |
42.59 |
0.0251 |
0.01 |
Sarolangun |
3.345 |
3.08 |
1,692.5 |
7.38 |
179 |
526.47 |
4.548 |
1.14 |
Sungai Penuh |
0.78 |
0.72 |
765 |
3.34 |
1,191.3 |
3,503.82 |
12.48 |
3.12 |
Tjb Barat |
1.97 |
1.82 |
1,045 |
4.56 |
2,861.4 |
8,415.88 |
0.757 |
0.19 |
Tjb Timur |
1.665 |
1.53 |
765 |
3.34 |
77.52 |
228 |
0.461 |
0.12 |
Tebo |
5.8 |
5.35 |
1,162.5 |
5.07 |
25.88 |
76.12 |
11.748 |
2.94 |
Laboratory |
1.085 |
1 |
229.25 |
1 |
0.34 |
1 |
3.996 |
1 |
LC₅₀ = median lethal concentration; RR = resistance ratio. Full probit analysis output (regression equations, R², slope ± SE, 95% CL, χ², df, Het, and p-values) is available in Supplementary Table S1.
Chlorantraniliprole. LC₅₀ values ranged from 0.186 mg L⁻¹ (Merangin) to 24.9 mg L⁻¹ (Jambi City), corresponding to 0.17 - 25.95-fold resistance ratios. Populations from Muaro Jambi, Batanghari, and Tebo remained susceptible, while those from Merangin and Bungo showed moderate to high resistance. This west-to-east gradient likely reflects stronger selection pressure in highland regions, where diamide-based insecticides are intensively used on corn and vegetable crops. Similar localized resistance patterns have been reported in Pakistan, where continuous selection with chlorantraniliprole and flubendiamide resulted in 198.7- and 136.5-fold resistance, respectively [15]. Deltamethrin LC₅₀ values were markedly higher than those of the laboratory reference strain, ranging from 478.21 mg L⁻¹ (Sarolangun) to 1,916.52 mg L⁻¹ (Sungai Penuh). The corresponding resistance ratios (RR = 2.09 - 8.36) classify most populations within the low-to-moderate resistance category. Although no population reached the high resistance threshold (RR > 10), the elevated LC₅₀ values, particularly in Sungai Penuh and Tanjab Timur, suggest reduced field sensitivity and emerging tolerance trends.
These results are consistent with global reports where intensive pyrethroid use selected for kdr sodium channel mutations and metabolic detoxification mechanisms [16,17]. While mechanisms were not examined in this study, phenotypic patterns align with previously described resistance pathways and support the need for continued monitoring and insecticide rotation.
Emamectin benzoate. Emamectin benzoate exhibited the greatest variability in toxicity among the tested insecticides, with LC₅₀ values ranging from 0.34 mg L⁻¹ (laboratory strain) to 2,861.4 mg L⁻¹ (Sungai Penuh). Resistance ratios spanned from 0.14 to 8,415.88, identifying two major resistance hotspots: Sungai Penuh and Tanjab Barat. These values represent extreme resistance and demonstrate that emamectin benzoate efficacy is already severely compromised in parts of the province. Similar patterns have been reported in China and India, where intensive reliance on avermectins selected for GluCl target-site mutations and metabolic detoxification pathways [18]. Given that emamectin benzoate remains a primary control option in Southeast Asia, the magnitude of resistance observed here warrants urgent intervention and region-specific IRM planning [19].
Spinetoram. Spinetoram remained the most effective among the tested insecticides, with LC₅₀ values of 0.03 - 1.17 mg L⁻¹ and RR values ranging from 0.01 to 3.12. Although no population exhibited resistance (RR > 4), slightly elevated LC₅₀ values in Bungo, Tebo, and Sungai Penuh indicate potential early selection pressure. Continued monitoring is therefore recommended to prevent resistance development [3,17].
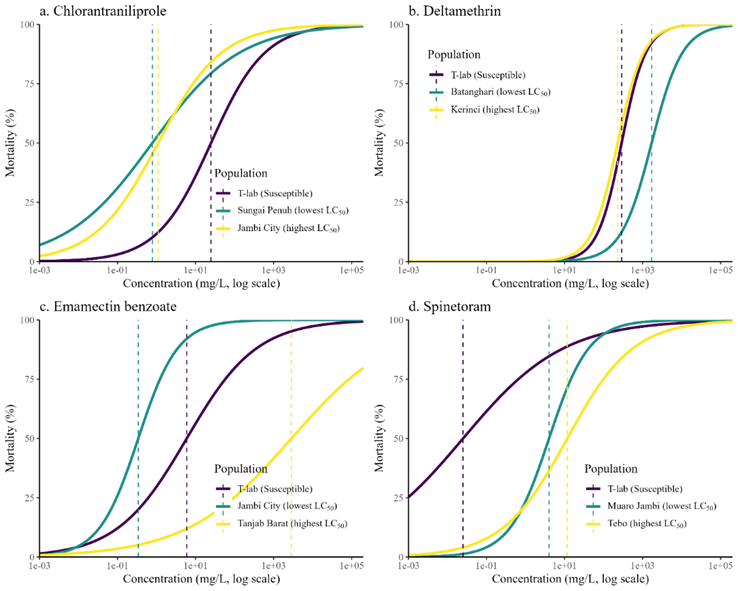
Figure 2 Probit regression curves of four insecticides against fall armyworm, showing the susceptible population (T-lab), lowest LC₅₀, and highest LC₅₀ populations.
Comparison of toxicity among active ingredients
Bioassays of four insecticidal active ingredients against eleven field populations of Spodoptera frugiperda from Jambi Province and one laboratory population revealed marked variations in susceptibility (Figure 3). Overall, the LC₅₀ values showed that spinetoram and emamectin benzoate were considerably more toxic than chlorantraniliprole and particularly deltamethrin. This finding suggests that spinetoram and emamectin benzoate remain effective options for managing S. frugiperda populations in Jambi Province, consistent with observations reported in Central Java [20].
The LC₅₀ values for spinetoram ranged from 0.0251 to 12.48 mg L⁻¹, with the Muaro Jambi population being the most susceptible and the Tebo and Sungai Penuh populations showing relatively higher tolerance. Emamectin benzoate exhibited a broader LC₅₀ range (0.34 - 2,861 mg L⁻¹), where the laboratory population showed the highest susceptibility, whereas the Sungai Penuh population displayed an exceptionally high LC₅₀, suggesting a possible reduction in sensitivity in that area. As commonly observed, laboratory populations typically exhibit high susceptibility (low LC₅₀ values), while field populations frequently exposed to emamectin benzoate tend to show increased LC₅₀ values often by several orders of magnitude, indicating the emergence of resistance [21].
By contrast, chlorantraniliprole and deltamethrin had substantially higher LC₅₀ values. Chlorantraniliprole resistance ranged from low to moderate (RR 1 - 25.95), while deltamethrin exhibited consistent low-to-moderate resistance (RR 2.09 - 8.36). The high LC₅₀ values of deltamethrin, despite RR remaining below the high-resistance threshold suggest reduced field sensitivity. Similar resistance patterns have been documented in other tropical regions where prolonged pyrethroid use selected for sodium channel (kdr) mutations and enhanced metabolic detoxification [8].
Differences in toxicity among the four insecticides likely reflect their contrasting modes of action. Spinetoram interferes with nicotinic acetylcholine and GABA-gated ion channels, while emamectin benzoate modulates glutamate-gated chloride channels [22,23]. These mechanisms differ from chlorantraniliprole, which targets ryanodine receptors, and deltamethrin, which affects voltage-gated sodium channel. Such functional divergence may contribute to the absence of cross-resistance between spinetoram or emamectin benzoate and pyrethroids or diamides in several field populations [24].
In summary, spinetoram and emamectin benzoate currently remain the most effective chemical options among the tested compounds, whereas chlorantraniliprole and deltamethrin demonstrated reduced efficacy in multiple locations. Implementing rotation strategies based on mode of action is therefore recommended to delay resistance escalation and maintain long term control efficacy in fall armyworm management programs.
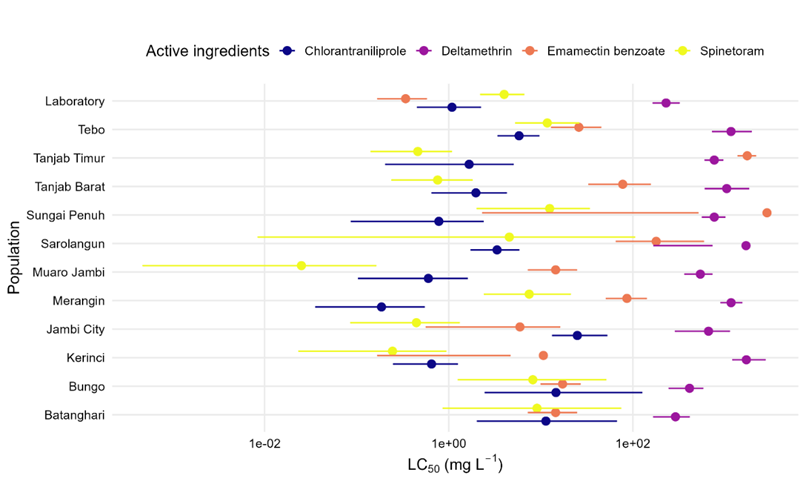
Figure 3 Comparison of LC₅₀ values of Spodoptera frugiperda to four insecticidal active ingredients across different populations in Jambi Province.
Spatial variation in resistance
Choropleth visualization of resistance ratio (RR) revealed spatially heterogeneous resistance patterns across Jambi Province (Figure 4). Darker colors indicated higher RR, highlighting distinct resistance hotspots for each active ingredient. Chlorantraniliprole resistance ranged from low to high depending on the region, with elevated levels occurring in Bungo, Batanghari, and Tebo (RR 5 - 22.95), while Muaro Jambi and Merangin remained largely susceptible (RR < 1). Four populations: Kerinci, Merangin, Sarolangun, and Tebo, exhibited moderate resistance to deltamethrin (RR 5 - 10), with no population exceeding the high-resistance threshold (RR > 10). Resistance to emamectin benzoate was highly localized and extreme (RR > 8,000) in Sungai Penuh and Tanjab Barat, whereas most other areas showed low-to-moderate resistance. In contrast, spinetoram resistance remained low throughout the province (RR < 4), although tolerance signals were detectable in three populations.
No population showed uniform resistance across all insecticides, suggesting that resistance evolution is influenced by localized spray regimes, cropping patterns, and agroecological conditions [7]. These patterns support the need for region-specific insect resistance management (IRM). Areas with high resistance to emamectin benzoate such as Tanjab Timur and Sungai Penuh; should prioritize reduced reliance on Group 6 insecticides, incorporate rotation across distinct IRAC groups, and integrate non-chemical strategies. Landscape-level resistance stewardship combining biological, cultural, and chemical tools will be essential to sustain long-term pest control efficacy [25,26].
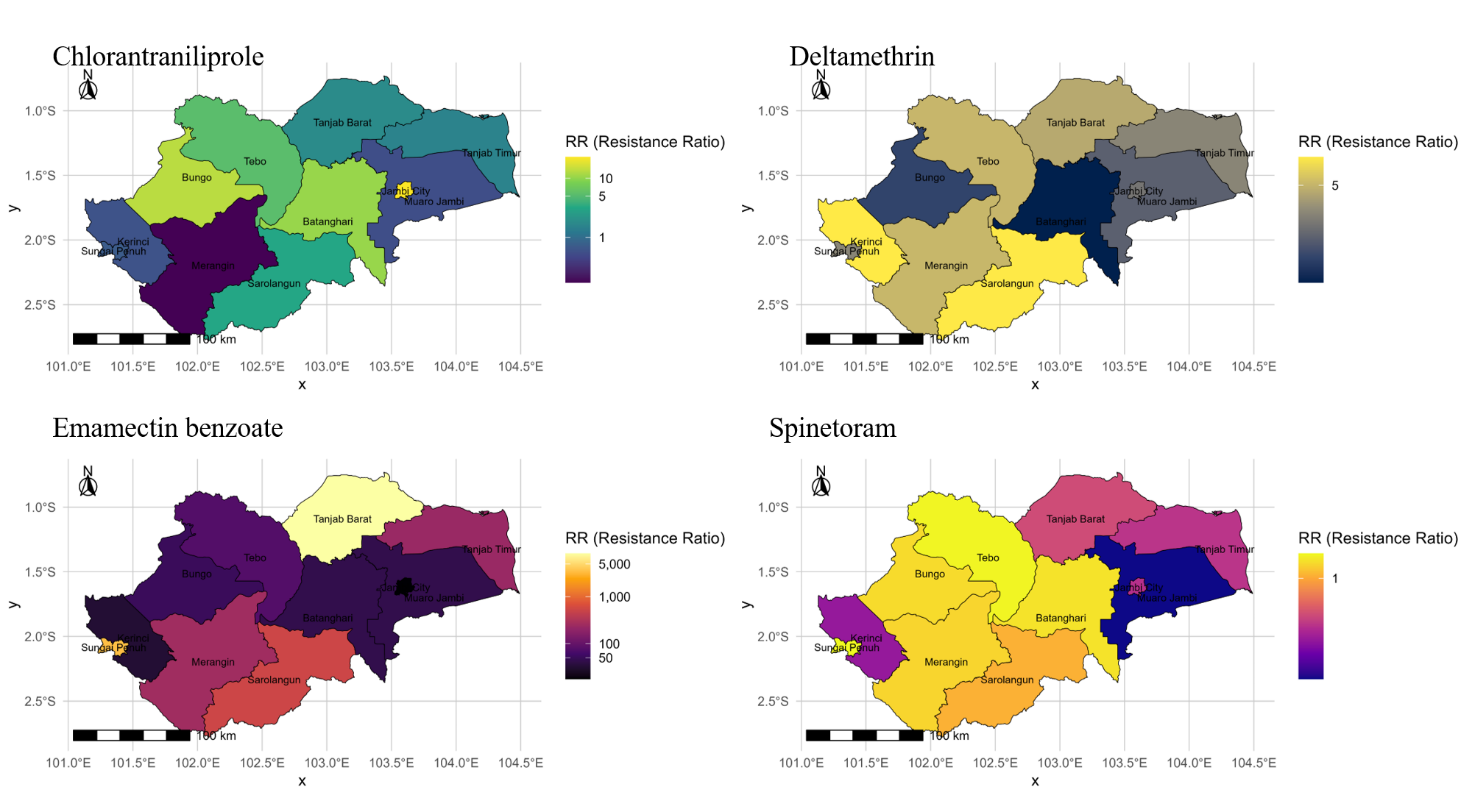
Figure 4 Spatial distribution of resistance ratios (RR) to chlorantraniliprole, deltamethrin, emamectin benzoate, and spinetoram in Spodoptera frugiperda populations across Jambi Province. Colors represent observed RR values using a color-blind-safe palette. The maps are displayed as administrative-level choropleth layers without interpolation. Scale bar and north arrow are included for geographic reference. RR categories follow IRAC-based interpretation thresholds (Susceptible < 1; Low 1 - 5; Moderate 5 - 10; High > 10)
Limitations
The use of F₂ field populations were intentional to retain resistance characteristics reflective of field conditions, as higher laboratory generations may lose resistance traits through relaxation of selection pressure and laboratory adaptation. Likewise, the highly susceptible reference strain (F₉₅) ensured a stable baseline for LC₅₀ comparison due to its long-term maintenance without insecticide exposure. However, differences in generational status (F₂ vs. F₉₅) may introduce minor variability in resistance ratio estimates, and future studies incorporating multiple susceptible reference colonies could further validate these comparisons. Second, while resistance levels were quantified using probit-based LC₅₀ estimates, no molecular or biochemical assays were performed to confirm the underlying mechanisms such as VGSC (kdr), GluCl mutations, or detoxification enzyme overexpression. Third, the diet-dipping bioassay method may differ from leaf-dip or field-exposure conditions, potentially affecting the comparability of values across studies. Finally, the spatial resistance patterns presented in the choropleth maps represent observed rather than model-predicted distributions, and therefore should not be interpreted as continuous regional predictions. These limitations highlight the need for complementary molecular validation, standardized exposure protocols, and expanded spatial monitoring in future work.
Conclusions
Fall armyworm populations across Jambi Province exhibited clear spatial variation in susceptibility to chlorantraniliprole, deltamethrin, emamectin benzoate, and spinetoram. Resistance to chlorantraniliprole remained generally low in most areas, whereas deltamethrin resistance was more widespread. Emamectin benzoate resistance was highly localized, and spinetoram remained largely effective, although early signs of reduced sensitivity were observed in some regions. Spatial analysis confirmed that resistance patterns were heterogeneous and influenced by local pest management practices.
Based on these resistance patterns, an IRAC mode-of-action rotation strategy is recommended, prioritizing Group 5 (spinetoram) and Group 6 (emamectin benzoate) as first-line options in areas with confirmed susceptibility. In regions where resistance ratios exceed the moderate threshold (RR > 10), chlorantraniliprole may be used as an alternative, while deltamethrin should be suspended where LC₅₀ values exceed 500 mg L⁻¹ or where field performance is reduced. A two-to-three season rotation schedule with non-cross-resistant MoA groups, reduced spray frequency, and integration of biological control and cultural tactics is recommended to slow further resistance development.
Acknowledgements
The author would like to thank the National Research and Innovation Agency (BRIN), Indonesia for supporting the doctoral education program in Department of Plant Protection, Faculty of Agriculture, Gadjah Mada University, Yogyakarta, Indonesia.
Declaration of Generative AI in Scientific Writing
The authors acknowledge the use of ChatGPT (OpenAI. USA) to improve the readability and grammar of the manuscript. The authors reviewed and edited the content as needed and take full responsibility for the final version of the manuscript.
CRediT Author Statement
Ratna Rubiana: Conceptualization; Methodology; Data curation; Investigation; Visualization; Software; Validation; Writing – Original draft. Y. Andi Trisyono: Conceptualization; Methodology; Supervision; Writing – Review & Editing. Suputa Suputa: Supervision. Witjaksono Witjaksono: Supervision.
References
[1] I Rwomushana. Spodoptera frugiperda (Fall armyworm). CABI Compendium 2019. https://doi.org/10.1079/cabicompendium.29810
[2] YA Trisyono, S Suputa, VEF Aryuwandari, M Hartaman and J Jumari. Occurrence of heavy infestation by the fall armyworm Spodoptera frugiperda, a new alien invasive pest, in corn Lampung Indonesia. Jurnal Perlindungan Tanaman Indonesia 2019; 23(1), 156.
[3] N Nonci, S Pakki and A Muis. Field testing of synthetic insecticides on fall armyworm (Spodoptera frugiperda J.E. Smith) in corn plant. IOP Conference Series: Earth and Environmental Science 2021; 911, 012059.
[4] MK Kansiime, I Mugambi, I Rwomushana, W Nunda, JL Godwin, H Rware, NA Phiri, G Chipabika, M Ndlovu and R Day. Farmer perception of fall armyworm (Spodoptera frugiperda J.E. Smith) and farm-level management practices in Zambia. Pest Management Science 2019; 75(10), 2840-2850.
[5] B Sisay, T Tefera, M Wakgari, G Ayalew and E Mendesil. The efficacy of selected synthetic insecticides and botanicals against fall armyworm Spodoptera frugiperda in maize. Insects 2019; 10(2), 45.
[6] Y Cai, H Chen, M Hu, X Wang and L Zhang. Discovery of novel potential insecticide-resistance mutations in Spodoptera frugiperda. Insects 2024; 15, 186.
[7] L Zhang, B Liu, W Zheng, C Liu, D Zhang, S Zhao, Z Li, P Xu, K Wilson, A Withers, CM Jones, JA Smith, G Chipabika, DL Kachigamba, K Nam, E D’Alençon, B Liu, X Liang, M Jin, C Wu, S Chakrabarty, X Yang, Y Jiang, J Liu, X Liu, W Quan, G Wang, W Fan, W Qian, K Wu and Y Xiao. Genetic structure and insecticide resistance characteristics of fall armyworm populations invading China. Molecular Ecology Resources 2020; 20(6), 1682-1696.
[8] RA Carvalho, C Omoto, LM Field, MS Williamson and C Bass. Investigating the molecular mechanisms of organophosphate and pyrethroid resistance in the fall armyworm Spodoptera frugiperda. PLoS One 2013; 8(4), e62268.
[9] HL Chen, A Hasnain, QH Cheng, LJ Xia, YH Cai, R Hu, CW Gong, XM Liu, J Pu, L Zhang and XG Wang. Resistance monitoring and mechanism in the fall armyworm Spodoptera frugiperda for chlorantraniliprole from Sichuan Province, China. Frontiers in Physiology 2023; 14, 1180655.
[10] V Matti, CP Mallapur, DN Kambrekar, UK Hulihalli and SI Harlapur. Laboratory evaluation of newer insecticides and biorationals against Spodoptera frugiperda (J.E. Smith). Journal of Experimental Zoology India 2023; 26(2), 1791-1796.
[11] A Meilin, R Rubiana, J Jumakir, K Suheiti, WS Murni, R Rustam and J Bobihoe. Study of pest attacks on maize plantation in oil palm replanting land of Jambi Province. IOP Conference Series: Earth and Environmental Science 2021; 752, 012008.
[12] IRAC Brazil Guidelines. Insec&cide resistance management guidelines for insect pests of corn, soybean and cotton in Brazil. IRAC Brazil Guidelines, Brazil, 2017.
[13] YX Zhao, JM Huang, H Ni, D Guo, FX Yang, X Wang, SF Wu and CF Gao. Susceptibility of fall armyworm Spodoptera frugiperda to eight insecticides in China, with special reference to lambda-cyhalothrin. Pesticide Biochemistry and Physiology 2020; 168, 104623.
[14] JT Hardke, GM Lorenz and BR Leonard. Fall armyworm (Lepidoptera: Noctuidae) ecology in Southeastern cotton. Journal of Integrated Pest Management 2015; 6(1), 10.
[15] R Salamat, B Siddique, MBS Afzal, A Banazeer, JE Serrao, MI Akram, A Sajjad, M Younus and AA Sial. Resistance of Spodoptera frugiperda to the diamides chlorantraniliprole and flubendiamide: Resistance development, heritability, inheritance and implications for management. Journal of Economic Entomology 2025; 118(6), 3157-3172.
[16] OA Posos-Parra, BR Pittendrigh, JC Wise, C DiFonzo, E Petterson and DM Sanchez. Inheritance and resistance mechanisms of field-evolved resistance to pyrethroids in a fall armyworm strain from Puerto Rico. Insects 2024; 15(12), 912.
[17] RK Thumar, MB Zala, HS Varma, CB Dhobi, BN Patel and PK Borad. Evaluation of insecticides against fall armyworm Spodoptera frugiperda infesting maize. International Journal of Chemical Studies 2020; 8(4), 100-104.
[18] M Oplopoiou, J Elias, R Slater, C Bass and CT Zimmer. Characterization of emamectin benzoate resistance in Plutella xylostella. Pest Management Science 2023; 80(2), 498-507.
[19] J Zhang, J Jiang, K Wang, Y Zhang, Z Liu and N Yu. A binary mixture of emamectin benzoate and chlorantraniliprole supplemented with an adjuvant effectively controls Spodoptera frugiperda. Insects 2022; 13(12), 1157.
[20] JN Suryani, YA Trisyono and E Martono. Susceptibility of Spodoptera frugiperda collected from central java province to emamectin benzoate. Jurnal Perlindungan Tanaman Indonesia 2022; 26(2), 159-166.
[21] DS Muraro, E Salmeron, JVS Cruz, FSA Amaral, AS Guidolin, ARB Nascimento, JB Malaquias, O Berbardi and C Omoto. Evidence of field-evolved resistance in Spodoptera frugiperda to emamectin benzoate in Brazil. Crop Protection 2022; 162, 106071.
[22] L Depalo, A Masetti, J Avilla, D Bosch and E Pasqualini. Toxicity and residual activity of spinetoram to neonate larvae of Grapholita molesta (Busck) and Cydia pomonella (L.) (Lepidoptera: Tortricidae): Semi-field and laboratory trials. Crop Protection 2016; 89, 32-37.
[23] XZ Wang, JS Chen, W Wang, DB Niu, HZ Wu, SR Palli, HQ Cao and CW Sheng. Knockdown of the glutamate-gated chloride channel gene decreases emamectin benzoate susceptibility in the fall armyworm, Spodoptera frugiperda. Pesticide Biochemistry and Physiology 2023; 196, 105636.
[24] A Mahajan, M Devi, P Mehra, A Kaur and S Kaur. Impact of chlorantraniliprole exposure on susceptible and resistant populations of Spodoptera litura. Journal of Applied Entomology 2025; 149(3), 1090-1104.
[25] BM Prasanna, A Bruce, Y Beyene, D Makumbi, M Gowda, M Asi, S Martinelli, GP Head and S Parimi. Host plant resistance for fall armyworm management in maize: Relevance, status and prospects in Africa and Asia. Theoretical and Applied Genetics 2022; 135(11), 3897-3916.
[26] PM Matova, CN Kamutando, C Magorokosho, D Kutywayo, F Gutsa and M Labuschagne. Fall armyworm invasion: Control practices and resistance breeding in Sub-Saharan Africa. Crop Science 2020; 60(6), 2951-2970.
Supplementary Material
This study provides one of the first spatially resolved resistance profiles for fall armyworm in Sumatra, offering evidence based guidance for region specific insecticide stewardship and decision making. Future work should focus on long-term resistance surveillance, integration of molecular diagnostics to identify emerging resistance alleles, and evaluation of non-chemical control options to support a more durable and sustainable IPM program. Supplementary material caption.
Table S1 Probit regression parameters for four insecticides tested against fall armyworm (Spodoptera frugiperda).
Insecticide |
Regression equations |
R2 |
Slope(±SE) |
LC50 (95% CL) mg L−1 |
Chi² |
df |
Het. |
P-Value |
Population |
||||||||
Chlorantraniliprole |
||||||||
Batanghari |
𝑦 = 1.200𝑥 + 1.970 |
0.9589 |
1.200 (± 0.1280) |
11.35 (2.055 - 66) |
10.600 |
3 |
3.54 |
0.003578 |
Bungo |
𝑦 = 0.760𝑥 + 1.100 |
0.9261 |
0.760 (± 0.0790) |
14.6 (2.495 - 124.5) |
8.950 |
3 |
2.98 |
0.008721 |
Kerinci |
𝑦 = 0.786𝑥 + 2.270 |
0.9747 |
0.786 (± 0.1160) |
0.65 (0.252 - 1.235) |
1.620 |
3 |
1 |
0.001724 |
Jambi City |
𝑦 = 0.629𝑥 + 0.820 |
0.9173 |
0.629 (± 0.0916) |
24.9 (13.45 - 52) |
5.030 |
3 |
1 |
0.010350 |
Merangin |
𝑦 = 0.791𝑥 + 0.987 |
0.9933 |
0.791 (± 0.0996) |
0.186 (0.036 - 0.54) |
0.550 |
3 |
1 |
0.000232 |
Muaro Jambi |
𝑦 = 0.481𝑥 + 1.400 |
0.8407 |
0.481 (± 0.0921) |
0.6 (0.105 - 1.585) |
5.030 |
3 |
1 |
0.028400 |
Sarolangun |
𝑦 = 0.732𝑥 + 1.590 |
0.9985 |
0.732 (± 0.0968) |
3.345 (1.75 - 5.75) |
0.102 |
3 |
1 |
0.000024 |
Sungai Penuh |
𝑦 = 0.389𝑥 + 1.090 |
0.9494 |
0.389 (± 0.0870) |
0.78 (0.088 - 2.35) |
1.110 |
3 |
1 |
0.004910 |
Tanjab Barat |
𝑦 = 0.523𝑥 + 1.260 |
0.9460 |
0.523 (± 0.0895) |
1.97 (0.66 - 4.205) |
2.280 |
3 |
1 |
0.005418 |
Tanjab Timur |
𝑦 = 0.345𝑥 + 0.854 |
0.9445 |
0.345 (± 0.0843) |
1.665 (0.207 - 5) |
0.994 |
3 |
1 |
0.005638 |
Tebo |
𝑦 = 0.826𝑥 + 1.600 |
0.9520 |
0.826 (± 0.0993) |
5.8 (3.44 - 9.5) |
3.390 |
3 |
1 |
0.004536 |
Laboratory |
𝑦 = 0.534𝑥 + 1.420 |
0.9331 |
0.534 (± 0.0880) |
1.085 (0.459 - 2.205) |
2.780 |
3 |
1 |
0.007501 |
Deltamethrin |
||||||||
Batanghari |
𝑦 = 1.460𝑥 - 0.963 |
0.9332 |
1.460 (± 0.2380) |
290 (168.5 - 407.5) |
2.360 |
3 |
1 |
0.000897 |
Bungo |
𝑦 = 1.240𝑥 - 0.268 |
0.8966 |
1.240 (± 0.2060) |
412.5 (248 - 572.5) |
4.790 |
3 |
1 |
0.002080 |
Kerinci |
𝑦 = 1.120𝑥 - 0.933 |
0.9271 |
1.120 (± 0.2010) |
1,707.5 (1,217.5 - 2,725) |
2.670 |
3 |
1 |
0.002363 |
Jambi City |
𝑦 = 0.707𝑥 - 0.299 |
0.9414 |
0.707 (± 0.1920) |
662.5 (290 - 1115) |
0.881 |
3 |
1 |
0.002620 |
Merangin |
𝑦 = 1.570𝑥 - 1.050 |
0.9313 |
1.570 (± 0.2140) |
1,165 (907.5 - 1,520) |
3.940 |
3 |
1 |
0.003394 |
Muaro Jambi |
𝑦 = 1.370𝑥 - 0.458 |
0.9097 |
0.137 (± 0.2130) |
540 (367.5 - 720) |
4.980 |
3 |
1 |
0.006735 |
Sarolangun |
𝑦 = 1.190𝑥 - 0.986 |
0.7366 |
1.190 (± 0.1990) |
1,692.5 (170 - 72) |
12.300 |
3 |
1 |
0.001163 |
Sungai Penuh |
𝑦 = 1.470𝑥 - 0.715 |
0.9298 |
1.471 (± 0.2060) |
765 (570 - 992.5) |
4.560 |
3 |
1 |
0.001016 |
Tanjab Barat |
𝑦 = 0.763𝑥 - 0.474 |
0.9019 |
0.763 (± 0.1870) |
1,045 (612.5 - 1,800) |
1.830 |
3 |
1 |
0.024190 |
Tanjab Timur |
𝑦 = 1.940𝑥 - 0.941 |
0.9445 |
1.940 (± 0.2280) |
765 (610 - 942.5) |
0.129 |
3 |
1 |
0.012300 |
Tebo |
𝑦 = 2.150𝑥 - 1.440 |
0.9520 |
2.50 (± 0.0242) |
1,162.5 (735 - 1,925) |
5.460 |
3 |
1 |
0.000324 |
Laboratory |
𝑦 = 1.300𝑥 + 0.049 |
0.9331 |
1.300 (± 0.2490) |
229.25 (166.25 - 317.5) |
1.870 |
3 |
1 |
0.002408 |
Emamectin benzoate |
||||||||
Batanghari |
𝑦 = 0.876𝑥 + 1.400 |
0.9836 |
0.876 (± 0.1150) |
14.48 (7.35 - 24.34) |
1.830 |
3 |
1 |
0.000897 |
Bungo |
𝑦 = 𝑥 + 0.268 |
0.9713 |
1.240 (± 0.1210) |
17.21 (10.09 - 26.62) |
3.530 |
3 |
1 |
0.002080 |
Kerinci |
𝑦 = 0.265𝑥 + 0.458 |
0.9688 |
0.265 (± 0.0831) |
10.66 (0.17 - 4.62) |
0.352 |
3 |
1 |
0.002363 |
Jambi City |
𝑦 = 0.484𝑥 + 0.961 |
0.9666 |
0.484 (± 0.0930) |
5.93 (0.57 - 15.96) |
0.974 |
3 |
1 |
0.002620 |
Merangin |
𝑦 = 0.837𝑥 + 0.688 |
0.9603 |
0.837 (± 0.0993) |
86.07 (51.76 - 139.65) |
3.640 |
3 |
1 |
0.003394 |
Muaro Jambi |
𝑦 = 0.876𝑥 + 1.400 |
0.9377 |
0.876 (± 0.1150) |
14.48 (7.35 - 24.34) |
1.340 |
3 |
1 |
0.006735 |
Sarolangun |
𝑦 = 0.382𝑥 – 0.192 |
0.9822 |
0.382 (± 0.0839) |
179 (66.12 - 582.0) |
0.437 |
3 |
1 |
0.001163 |
Sungai Penuh |
𝑦 = 0.607𝑥 + 4.806 |
0.9822 |
0.607 (± 0.0970) |
2,861.4 (2.34 - 506.7) |
0.777 |
3 |
1 |
0.001016 |
Tanjab Barat |
𝑦 = 0.323𝑥 - 0.226 |
0.8564 |
0.323 (± 0.1110) |
77.52 (33.40 - 154.0) |
1.470 |
3 |
1 |
0.024190 |
Tanjab Timur |
𝑦 = 0.566𝑥 + 0.492 |
0.9075 |
0.566 (± 0.0970) |
1,744.2 (1,390.8 - 2,148.9) |
1.440 |
3 |
1 |
0.012300 |
Tebo |
𝑦 = 0.761𝑥 +1.020 |
0.9917 |
0.761 (± 0.1010) |
25.88 (13.11 - 44.57) |
0.702 |
3 |
1 |
0.000324 |
Laboratory |
𝑦 = 0.857𝑥 + 2.760 |
0.9684 |
0.857 (± 0.1150) |
0.34 (0.17 - 0.57) |
3.650 |
3 |
1 |
0.002408 |
Spinetoram |
||||||||
Batanghari |
𝑦 = 0.642𝑥 + 1.360 |
0.9016 |
0.642 (± 0.0707) |
9.084 (0.871 - 73.44) |
8.80 |
3 |
1 |
0.000897 |
Bungo |
𝑦 = 0.857𝑥 + 1.860 |
0.9522 |
0.857 (± 0.0855) |
8.184 (1.272 - 50.64) |
9.87 |
3 |
1 |
0.002080 |
Kerinci |
𝑦 = 0.346𝑥 + 1.280 |
0.9724 |
0.346 (± 0.0646) |
0.245 (0.0236 - 0.926) |
0.83 |
3 |
1 |
0.002363 |
Jambi City |
𝑦 = 0.412𝑥 + 1.410 |
0.9300 |
0.412 (± 0.0668) |
0.445 (0.0863 - 1.296) |
3.36 |
3 |
1 |
0.002620 |
Merangin |
𝑦 = 0.368𝑥 + 0.812 |
0.9384 |
0.368 (± 0.0609) |
7.476 (2.436 - 20.88) |
2.77 |
3 |
1 |
0.003394 |
Muaro Jambi |
𝑦 = 0.337𝑥 + 1.580 |
0.8780 |
0.337 (± 0.0706) |
0.0251 (0.00048 - 0.161) |
3.78 |
3 |
1 |
0.006735 |
Sarolangun |
𝑦 = 0.338𝑥 + 0.940 |
0.8394 |
0.338 (± 0.0611) |
4.548 (0.00852 - 104.28) |
7.79 |
3 |
1 |
0.001163 |
Sungai Penuh |
𝑦 = 0.484𝑥 + 0.961 |
0.9683 |
0.484 (± 0.0930) |
12.48 (2.04 - 33.60) |
0.97 |
3 |
1 |
0.001016 |
Tanjab Barat |
𝑦 = 0.506𝑥 + 1.620 |
0.9240 |
0.506 (± 0.0723) |
0.757 (0.242 - 1.788) |
1.99 |
3 |
1 |
0.024190 |
Tanjab Timur |
𝑦 = 0.551𝑥 + 1.880 |
0.9835 |
0.551 (± 0.0769) |
0.461 (0.144 - 1.066) |
1.38 |
3 |
1 |
0.012300 |
Tebo |
𝑦 = 0.516𝑥 + 1.040 |
0.9649 |
0.516 (± 0.0661) |
11.748 (5.352 - 25.80) |
2.47 |
3 |
1 |
0.000324 |
Laboratory |
𝑦 = 0.852𝑥 + 2.110 |
0.9648 |
0.852 (± 0.1050) |
3.996 (2.232 - 6.516) |
3.33 |
3 |
1 |
0.002408 |
Regression equations. R² values. slope (± SE). LC50 with 95% confidence limits. Chi² values. degrees of freedom (df). heterogeneity values. and p-values are presented.