
Trends
Sci.
2026;
23(7):
12673
Chlorogenic Acid Ameliorates Testicular Damage and Hormonal Imbalance in Diabetic Male Wistar Rats
Eko
Naning Sofyanita1,2,
![]() ,
Eka Chandra Herlina3,
and
Achmad Zulfa Juniarto4,*
,
Eka Chandra Herlina3,
and
Achmad Zulfa Juniarto4,*
1Doctoral Study Program of Medical and Health Science, Universitas Diponegoro, Jawa Tengah 50275, Indonesia
2Department of Medical Laboratory Technology, Poltekkes Kemenkes Semarang, Jawa Tengah 50275, Indonesia
3Faculty of Medicine, Universitas Diponegoro, Jawa Tengah 50275, Indonesia
4Department of Medical Biology, Faculty of Medicine, Universitas Diponegoro, Jawa Tengah 50275, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 16 November 2025, Revised: 20 December 2025, Accepted: 27 December 2025, Published: 5 March 2026
Abstract
Diabetes mellitus (DM) is a metabolic disorder characterized by hyperglycemia that may lead to multiple complications, including male reproductive dysfunction. This study aimed to evaluate the effects of chlorogenic acid (CGA) administration on blood glucose levels (BGLs), reproductive hormones, testicular histology, and spermatogenesis in male Wistar rats with type 2 diabetes mellitus (T2DM). Thirty rats were divided into a healthy control group (n = 5) and a diabetic group (n = 25). T2DM was induced using a high-fat diet (HFD) combined with streptozotocin (STZ) and nicotinamide (NA). The diabetic rats were further divided into five subgroups: Untreated diabetic (DM), diabetic + metformin 500 mg/kg BW (DMet), diabetic + CGA 6.25 mg/kg BW (DMCGA 6.25mg), diabetic + CGA 12.5 mg/kg BW (DMCGA 12.5mg), and diabetic + CGA 25 mg/kg BW (DMCGA 25mg). CGA was administered intraperitoneally (i.p.) once daily for 31 days. Serum of luteinizing hormone (LH), follicle-stimulating hormone (FSH), and testosterone were measured using ELISA, and testicular structure and spermatogenesis were evaluated histologically and scored using the Johnsen system. CGA administration significantly reduced BGLs (p < 0.05), increased LH, FSH, and testosterone levels, and improved seminiferous tubule diameter, epithelial thickness, and Johnsen score. The CGA dose of 25 mg/kg BW showed the most pronounced effect, reducing blood glucose to 93.80 ± 5.31 mg/dL, comparable to the metformin group (104.40 ± 6.11 mg/dL), and increasing testosterone levels to 1,642.57 ± 38.66 pg/mL, approaching the normal control (1,763.86 ± 23.05 pg/mL). The 25 mg/kg BW CGA group demonstrated the most pronounced restorative effect, comparable to the metformin group. In conclusion, CGA exhibits antidiabetic and testicular protective effects by enhancing endocrine and spermatogenic functions in diabetic male rats.
Keywords: Chlorogenic acid, Diabetes mellitus, Luteinizing hormone, Follicle-stimulating hormone, Testosterone, Spermatogenesis, Johnsen score
Introduction
Diabetes mellitus (DM) is a metabolic disease that is generally characterized by hyperglycemia or high blood glucose levels (BGLs), caused by impaired insulin secretion or insulin performance, or both [1,2]. Persistent hyperglycemia disrupts carbohydrate, protein, and lipid metabolism and leads to various systemic complications [3]. DM is a rapidly growing global health problem [4]. The prevalence of DM worldwide in 2021 is estimated at 537 million people aged 20 to 79
years. DM cases will continue to increase to 643 million in 2030 and to around 783 million cases worldwide in 2045 [5-7]. As many as 79.2% of men with diabetes experience erectile dysfunction, 35% - 50% experience ejaculation disorders, and 16% of men with DM experience decreased libido. Reproductive disorders in women with DM affect around 32.3%, including low sexual desire (81.8%), orgasm problems (47.5%), and resolution disorders (35.1%) [8,9]. In women with type 1 diabetes, infertility affects 24.5% of them, with various complications such as polycystic ovary syndrome, micro-vascular issues, and primary hypothyroidism, which are also significant problems that can exacerbate reproductive disorders in women with diabetes [10,11].
Beyond chronic hyperglycemia in diabetes mellitus, stress hyperglycemia has emerged as a clinically significant metabolic condition in humans, particularly in hospitalized patients, individuals experiencing acute illness, trauma, infection, or psychological stress. Stress hyperglycemia can occur even in individuals without a prior history of diabetes and is associated with excessive activation of counter-regulatory hormones, systemic inflammation, and oxidative stress. Clinical evidence indicates that stress-induced hyperglycemia is strongly associated with adverse outcomes, including hormonal dysregulation, endothelial dysfunction, and impaired organ recovery. Importantly, stress hyperglycemia shares key pathophysiological mechanisms with diabetes-related hyperglycemia, including increased reactive oxygen species (ROS) production, activation of inflammatory pathways, and disruption of endocrine signaling [12].
DM disrupts the hypothalamic–pituitary–gonadal (HPG) axis, leading to hormonal imbalance and impaired spermatogenesis [13,14]. A decrease in luteinizing hormone (LH) and follicle-stimulating hormone (FSH) will disrupt Sertoli cell and Leydig cell function [15,16]. Leydig cells function in the secretion of testosterone, while Sertoli cells, together with testosterone, play a role in regulating spermatogenesis [15,17]. Sperm cell development is also affected by oxidative stress, inflammation, and mitochondrial dysfunction caused by diabetes, which can ultimately damage the testicles [18]. Endocrine disorders and increased severity of diabetic neuropathy will affect reproductive hormones in the next stage, which will be associated with the pathophysiology of infertility, especially in men [19].
Metformin is widely used as a first-line therapy for T2DM due to its efficacy in lowering blood BGLs through AMPK activation. However, accumulating evidence suggests that metformin has limited capacity to directly protect male reproductive tissues from oxidative and inflammatory damage, particularly under chronic hyperglycemic conditions [20]. Meanwhile, CGA is a natural polyphenolic compound that exhibits antihyperglycemic, antioxidant, and anti-inflammatory properties [21]. Despite these promising characteristics, evidence regarding the specific role of CGA in improving spermatogenesis and testicular histology under diabetic conditions remains limited. Therefore, this study aimed to investigate the effects of CGA on glycemic control, reproductive hormones, testicular histology, and spermatogenesis in diabetic male rats. This study hypothesized that CGA administration improves spermatogenesis in a dose-dependent manner by reducing BGLs and modulating hormonal and testicular structural alterations associated with diabetes.
Materials and methods
Animal preparation
This study was conducted using Wistar rats weighing 150 - 200 g in healthy conditions. The rats were kept in standardized cages with a 12-h light and dark cycle, room temperature of 23 ± 2 °C, and relative humidity of 50% - 60% with ad libitum feeding and drinking. The rats underwent adaptation for 7 days.
The number of rat samples in this study was 30, divided into six groups, so that each group consisted of 5 rats. Group 1 (N) – Healthy rats; Group II (DM) – Diabetic rats without treatment; Group III (DMet) – Diabetic rats given Metformin 500 mg/kg BW; Group IV (DMCGA 6.25mg) – Diabetic rats given CGA 6.25 mg/kg BW; Group V (DMCGA 12.5mg) – Diabetic rats given CGA 12.5 mg/kg BW; Group VI (DMCGA 25mg) – Diabetic rats given CGA 25mg/kg BW. CGA with a purity of ≥ 95% was obtained from Sigma-Aldrich (St. Louis, MO 63103, USA; product number C3878; batch number WXBF3738V). CGA is prepared freshly before administration and dissolved in phosphate buffered saline solution pH 7.4. CGA was given intraperitoneally (i.p.) for 31 days. The intraperitoneal route was selected to ensure precise dose delivery and consistent systemic bioavailability, while minimizing variability associated with gastrointestinal absorption and first-pass hepatic metabolism. This approach has been widely applied in mechanistic experimental studies investigating the effects of polyphenolic compounds, including chlorogenic acid, on metabolic regulation and male reproductive function in diabetic and testicular injury animal models [4,22,23]. The CGA dose was selected based on Sari et al. [24] rats in the diabetes group were fed an HFD for 2 weeks (20 g/rat/day) with the following composition: Comfeed Par S (Japfa Comfeed Indonesia 60%), flour (27.8%), cholesterol (2%), folic acid (0.2%), and pork fat (10%). After 2 weeks, the rats in the group were induced with diabetes by fasting for 12 h or overnight, then given a single injection of streptozotocin (STZ) 45mg/kg (Sigma-Aldrich, USA) dissolved in 0.1 M PH 4.5 citrate buffer and 110 mg/kg nicotinamide (Solarbio N8070, China). BGLs were measured 3 and 7 days after treatment using a glucometer (EasyTouch®, GCU Model: ET-301, Bioptik Technology, Taiwan) to ensure stable diabetic conditions in the rats. Rats with BGLs above 200 mg/dl were considered diabetic. In this study, the experimental procedures were approved by the Health Research Ethics Committee, Ministry of Health, Semarang Health Polytechnic (No. 836/EA/F.XXIII.38/2025).
Analysis of LH, FSH, and testosterone levels
Blood samples were collected in EDTA-free vials from cardiac puncture. After 30 min of storage at room temperature, the blood samples were centrifuged for 15 min at 3,000 rpm. LH, FSH, and testosterone hormones were examined on day 32 using an ELISA kit (FineTest®, Wuhan National Bio-industry Base, China). Testosterone EU0400, LH ER1123, and FSH ER0960). The procedure for analyzing testosterone, LH, and FSH hormone levels was performed according to the ELISA kit instructions. After the ELISA test was performed, the results were read using a Medical Pro reader-96 (Germany).
Testis organ preparation
The testis is fixed in 10% Neutral Buffered Formalin, then cut and placed in a tissue cassette. Next, dehydration is carried out sequentially using alcohol with increasing concentrations of 70%, 80%, 90%, Ethanol I, and Ethanol II in sequence in jars for 2 h. Next is clearing, where the tissue is cleaned with xylol and then embedded in a paraffin block, so that the preparation is printed in a block and stored in a refrigerator for 24 h. After that, the block containing the tissue is cut using a microtome to a thickness of 4 - 5 µm. The cut results are floated in warm water at 60 °C for 24 h to prevent folding. The preparation is then lifted and placed in a glass object and stained with Hematoxylin and Eosin (HE). Next, it is examined under a microscope.
Histological examination
Histological preparation was performed using histotechnical methods based on Dunn et al. [25]. HE staining was examined to evaluate histopathological changes in the seminiferous tubules and epithelial thickness. Spermatogenesis was examined based on Johnsen’s criteria [26]. 10: Normal tubule epithelium with complete spermatogenesis, open lumen, spermatozoa ≥ 10. 9: Damaged epithelium, closed lumen, spermatozoa ≥ 10. 8: Fewer than 10 spermatozoa. 7: No spermatozoa, but ≥ 10 spermatids. 6: No spermatozoa, < 10 spermatids. 5: No spermatozoa or spermatids, but ≥ 5 spermatocytes. 4: No spermatozoa or spermatids, < 5 spermatocytes. 3: Spermatogenic cells only spermatogonia. 2: No spermatogenic cells, only Sertoli cells. 1: Tubules are empty, no cells. Scale bars were calibrated using ImageJ software based on the microscope imaging system, and all histological images were analyzed and presented at identical magnification settings.
Statistical analysis
The data are presented as mean ± Standard Deviation (SD). Statistical tests were performed using SPSS version 27 (IBM Corp., USA). Differences between groups were analyzed using a one-way ANOVA and followed by the Tukey HSD test. Significant differences between mean values were considered at p < 0.05. To determine the relationship between variables, Pearson’s correlation test was performed and the results were analyzed using linear regression. The diagrams were processed using GraphPad Prism version 10.6.1 software and the regression path using SmartPLS version 4.1.1.4. Partial Least Squares Structural Equation Modeling (PLS-SEM) using SmartPLS is particularly suitable for exploratory biological and preclinical studies involving complex interrelated variables, small sample sizes, non-normal data distribution, and potential multicollinearity, where conventional covariance-based SEM assumptions may not be fully met [27].
Results and discussion
The results of data analysis show that administration of CGA at doses of 6.25, 12.5, and 25 mg/kg BW reduced BGLs and increased LH, FSH, and testosterone concentrations, as well as seminiferous tubule diameter, epithelial thickness, and spermatogenesis scores in diabetic male rats in a dose-dependent manner. The SmartPLS model shows that hormonal balance has a more substantial effect on spermatogenic improvement than structural recovery.
Hyperglycemia-induced oxidative stress represents a central pathological mechanism underlying diabetes-related male reproductive dysfunction. Chronic elevation of BGLs increases the production of ROS which disrupt mitochondrial function, damage cellular lipids and DNA, and activate pro-inflammatory signaling pathways. In testicular tissue, excessive ROS accumulation impairs Leydig and Sertoli cell function, disrupts the blood–testis barrier, and suppresses steroidogenic enzyme activity, ultimately leading to hormonal imbalance and impaired spermatogenesis. Thus, oxidative stress serves as a unifying link between metabolic dysregulation, endocrine disturbance, and structural damage in diabetic testes.
Blood Glucose Levels (BGLs) in diabetic-induced rats
Five groups of rats induced with STZ resulted in rats with diabetic mellitus (DM). In (Figure 1), rats with DM without therapy showed the highest average BGLs (298.60 ± 24.98 mg/dL, p < 0.05), followed by the DMCGA 6.25mg group (115.60 ± 5.68 mg/dL), DMet group (104.40 ± 6.11 mg/dL), DMCG 12.5 mg group (102.20 ± 2.95 mg/dL), and DMCGA 25 mg group (93.80 ± 5.31 mg/dL). The lowest BGLs were found in the healthy rat group (N), which was 85.20 ± 7.16 mg/dL. In the intergroup difference test using One Way ANOVA, the results showed a significant p-value of 0.000 (p < 0.05), and the Tukey test comparing the intervention groups versus the DM group yielded p = 0.000 (p < 0.05), indicating a significant difference.
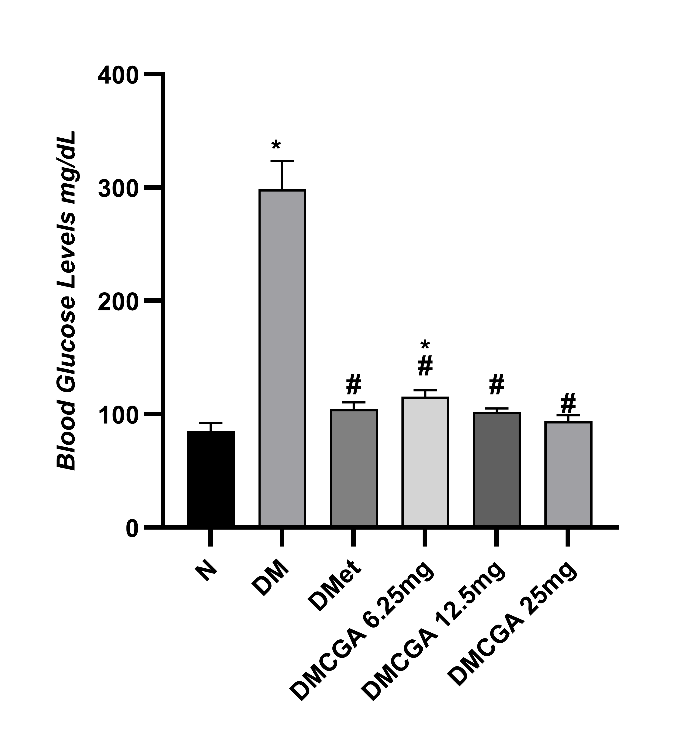
Figure 1 The Mean ± SD BGLs in DM model rats. There is a significant difference among the treatment groups (p = 0.001). Note: N (healthy rats); DM (rats with diabetes mellitus); DMet (diabetic rats + 500 mg/kg BW of metformin); DMCGA 6.25 mg (diabetic rats + injection of CGA at a dose of 6.25 mg/kg BW); DMCGA 12.5 mg (diabetic rats + injection of CGA at a dose of 12.5 mg/kg BW). DMCGA 25 mg (diabetic rats + injection of CGA at a dose of 25 mg/kg BW). N (85.20 ± 7.16mg/dL), DM (298.60 ± 24.98 mg/dL), DMet (104.40 ± 6.11 mg/dL), DMCGA 6.25mg (115.60 ± 5.68 mg/dL), DMCGA 12.5 mg (102.20 ± 2.95 mg/dL), DMCGA 25 mg (93.80 ± 5.31 mg/dL). * = p < 0.05 vs N, #= p < 0.05 vs DM.
In the normal rat group (N), the BGLs were shown to be normal at 85.20 ± 7.16 mg/dL, according to the study by Nazarudin et al. [28] rats given streptozotocin at 45 mg/kg BW developed high BGLs > 135 mg/dL, indicating that the rats were hyperglycemic. Consistent with this study, the BGLs in the DM group were 298.60 ± 24.98 mg/dL (Figure 1). The elevated BGLs caused by STZ injection induce a diabetic condition through a mechanism that damages pancreatic beta cells responsible for insulin production. STZ enters cells via the glucose transporter type 2 (GLUT2) and damages deoxyribonucleic acid (DNA), ultimately leading to cell necrosis due to depletion of nicotinamide adenine dinucleotide (NAD+) and adenosine triphosphate (ATP) [29,30]. The injection of NA in the animal model of diabetes aims to protect some pancreatic cells from the cytotoxic effects of STZ [31].
Rats given metformin and CGA had significantly lower BGLs than the DM group (p < 0.05). Metformin known as the first-line drug for type 2 diabetes, works by activating AMPK, which suppresses hepatic gluconeogenesis, increases glucose uptake by muscles, and inhibits the activity of mitochondrial respiratory chain complex I [32,33]. Activation of AMPK also stimulates fatty acid oxidation, reduces triglyceride accumulation in the liver, and enhances insulin sensitivity. Additionally, metformin decreases the expression of the glucose-6-phosphatase (G6Pase) gene and increases the number of GLUT-4 transporters in peripheral tissues. Regarding oxidative stress mechanisms, metformin acts as a mitochondrial stabilizer capable of reducing ROS formation and improving the NADH/NAD⁺ ratio. These findings align with previous research by Sianturi et al. [33] which reported that metformin lowers glucose and malondialdehid (MDA) levels while increasing SOD. In this study, the metformin group showed a significant decrease in glucose levels approaching normal values after 31 days of administration, confirming the important role of metformin in glycemic control through metabolic and antioxidant mechanisms.
CGA administration also showed comparable glucose lowering effects to metformin. CGA activates AMPK and Nrf2, which function to increase the expression of antioxidant enzymes such as SOD, GPx, and catalase. Activation of Nrf2 suppresses ROS formation in the pancreas and protects β cells from DNA damage caused by STZ. CGA also inhibits α-glucosidase and G6Pase, thereby reducing endogenous glucose production in the liver [34]. This is consistent with the research by Nguyen et al. [35] which reported a decrease in fasting glucose and an increase in insulin sensitivity after CGA administration. CGA is also known to improve mitochondrial biogenesis and suppress inflammation through NF-κB inhibition, making it not only antidiabetic but also a potent antioxidant that protects pancreatic tissue from apoptosis. The administration of CGA at a dose of 25 mg/kg BW in this study significantly lowered BGLs, approaching normal, indicating mark improvement metabolic parameters.
In the DM group, DMet and DMCGA show very distinct mechanisms and biological effects that complement each other. The DM group exhibits high BGLs due to pancreatic cell damage and uncontrolled insulin resistance. Metformin primarily lowers glucose by increasing insulin sensitivity through AMPK activation, while CGA acts dually: Through the AMPK pathway and via Nrf2 activation, enabling it to suppress systemic oxidative stress. CGA has a biological advantage as a natural phytochemical that can act on various metabolic tissues, including the liver, pancreas, muscles, and testes [36].
LH, FSH, and testosterone hormones levels in diabetic-induced rats
The results of one way ANOVA analysis showed significant differences in LH, FSH, and testosterone levels among treatment groups (p < 0.05). LH levels decreased in the DM group (4.43 ± 0.28 mIU/mL) compared to the normal group (14.49 ± 0.21 mIU/mL), but increased after metformin therapy (9.98 ± 0.21) and CGA, especially at a dose of 25 mg/kg BW (13.39 ± 0.34). FSH levels also decreased in the DM group (24.38 ± 2.17) compared to the normal group (105.38 ± 5.60) and increased after CGA administration (80.93 ± 4.08 at a dose of 25 mg/kg BW). Testosterone levels dropped drastically in the DM group (699.14 ± 41.66 pg/mL) compared to the normal group (1,763.86 ± 23.05 pg/mL). Testosterone levels increased after administration of CGA at a dose of 25 mg/kg BW (1,642.57 ± 38.66 pg/mL). The post hoc Tukey test indicated that all treatment groups differed significantly from the DM group (p < 0.05), with the highest increase observed in the DMCGA 25 mg group, but there was no significant difference between the DMCGA 25 mg group and the normal group in LH and testosterone levels (p > 0.05). (Figure 2).
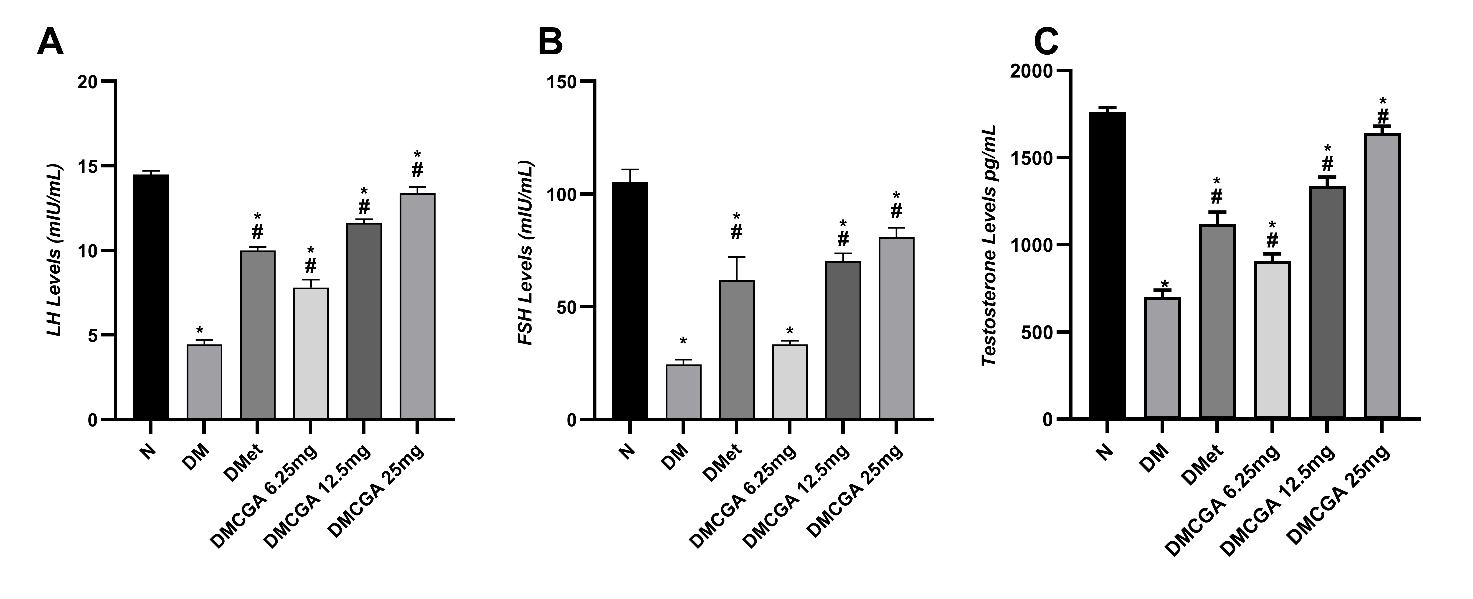
Figure 2 (A) LH levels, (B) FSH levels, and (C) Testosterone levels. The effect of CGA intervention for 31 days on the levels of LH, FSH, and testosterone in male diabetic rats in each group: N = normal; DM = diabetes rats; DMet = diabetes rats + metformin; DMCGA = diabetes rats + CGA at doses of 6.25, 12.5, and 25 mg/kg BW. Data are presented as mean ± SD (n = 5). DM caused a significant decrease in LH (4.43 ± 0.28 mIU/mL), FSH (24.38 ± 2.17 mIU/mL), and testosterone (699.14 ± 41.66 pg/mL) compared to the normal group (LH 14.49 ± 0.21; FSH 105.38 ± 5.60; testosterone 1763.86 ± 23.05). Administration of metformin or CGA appears to increase the levels of these three hormones as the dose increases, with the highest mean hormone levels observed in the DMCGA 25 mg group: LH 13.39 ± 0.34; FSH 80.93 ± 4.08; testosterone 1642.57 ± 38.66. Notes: p < 0.05 vs N (*); p < 0.05 vs DM (#); ns = not significant (p > 0.05). Statistical analysis was performed using one-way ANOVA followed by Tukey HSD post hoc test.
STZ-NA induction worsens the condition by damaging pancreatic β cells, decreasing insulin secretion, and suppressing insulin-like growth factor 1 (IGF-1) levels, which are important for testosterone production. This imbalance triggers gonadotropin hormone feedback disruption in the hypothalamus, resulting in low levels of LH and FSH, indicating hypogonadotropic hypogonadism [28]. Increased levels of inflammatory cytokines, such as tumor necrosis factor-alpha (TNF-α) and interleukin-6 (IL-6), also contribute to inhibiting GnRH secretion, thereby worsening reproductive hormone disturbances caused by diabetes [37].
Administration of metformin in diabetic rats shows partial recovery of LH, FSH, and testosterone levels through increased insulin sensitivity and activation of AMPK. AMPK activation stimulates the expression of Steroidogenic Acute Regulatory (StAR) in Leydig cells, thereby increasing cholesterol transport for testosterone synthesis, while oxidative stress is suppressed through decreased ROS production in the mitochondrial electron transport chain [38]. He et al. [39] reported that AMPK activation improves steroidogenesis and suppresses testicular apoptosis in diabetic rats. Similar results were observed in this study after 31 days of metformin administration, which increased levels of LH, FSH, and testosterone and restored the positive feedback of the HPG axis. The effects of metformin are more focused on glucose regulation rather than direct stimulation of androgen receptors, so the improvement in gonadal function has not yet been fully optimal. Compared to other groups, the diabetic rat group showed a very drastic decrease in reproductive hormones due to oxidative stress and inflammation. Metformin improves hormone levels by improving glucose metabolism, but its effects are limited to glycemic control, unlike CGA, which shows a broader effect because, in addition to lowering glucose, it also normalizes hormonal pathways through antioxidant and anti-inflammatory mechanisms. It can be concluded that administering CGA not only shares the same mechanism as metformin but also extends it through the protection of reproductive tissues.
In addition to oxidative stress, chronic inflammation plays a critical role in diabetes-induced male reproductive dysfunction. Persistent hyperglycemia promotes immune dysregulation characterized by increased activation and infiltration of T lymphocytes, particularly CD4⁺ and CD8⁺ cells. Clinical and experimental evidence indicates that diabetes is associated with elevated circulating pro-inflammatory cytokines such as tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and interferon-γ (IFN-γ), which are predominantly produced by activated T lymphocytes. The increase in CD4⁺ helper T cells amplifies inflammatory signaling, while cytotoxic CD8⁺ T cells contribute to tissue injury through direct cellular damage and cytokine-mediated apoptosis [40].
Diameter of the seminiferous tubules and the thickness of the germinal epithelium in diabetic-induced rats
The results of the analysis of seminiferous tubule diameter and epithelial thickness in this study showed that the results were generally not very significant (Figure 3). Average diameter of seminiferous tubules (µm): N = 40.72 ± 0.55; DM = 25.73 ± 1.46; DMet = 31.06 ± 0.83; DMCGA 6.25 mg = 31.44 ± 1.07; DMCGA 12.5 mg = 33.48 ± 0.87; DMCGA 25 mg = 37.37 ± 0.88. Average germinal epithelium thickness (µm): N = 10.04 ± 1.12; DM = 5.54 ± 0.34; DMet = 6.40 ± 0.15; DMCGA 6.25 mg = 5.94 ± 0.72; DMCGA 12.5 mg = 6.76 ± 0.61; DMCGA 25 mg = 8.48 ± 0.90.
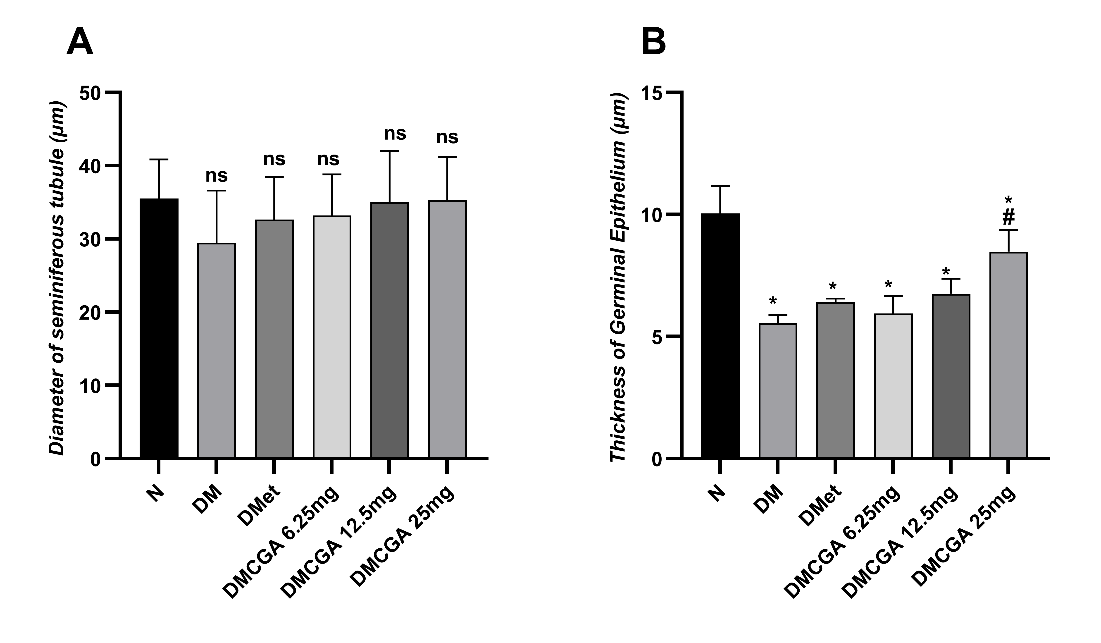
Figure 3 Diameter of the seminiferous tubules (left) and germinal epithelium thickness (right) in male rats from various treatment groups. Groups: N = normal; DM = diabetes without therapy; DMet = diabetes + metformin; DMCGA = diabetes + chlorogenic acid at doses of 6.25, 12.5, and 25 mg/kg body weight. Data are presented as mean ± SD. Notes: *significant compared to group N (p < 0.05), #significant compared to group DM (p < 0.05), and “ns” indicates not significant between groups N and DM (p > 0.05).
Figure 3 shows that the diameter of the seminiferous tubules among groups did not differ significantly in the Post hoc Tukey test (p > 0.05), as indicated by the label “ns” on the diagram. This suggests that diabetes induction and intervention administration have not yet caused significant changes in the lumen size of the tubules. The thickness of the germinal epithelium decreased in the diabetic group compared to the normal group (p < 0.05). Administration of metformin and CGA gradually increased the epithelial thickness, with the most notable improvement at a dose of 25 mg/kg BW (8.48 ± 0.90 µm). The post hoc Tukey test showed that the DMCGA 25 mg group differed significantly from the diabetic group but did not differ significantly from the normal group (p > 0.05).
DM increases the formation of free radicals such as superoxide and hydroxyl radicals, which attack the spermatogenic and Sertoli cell membranes. The decrease in the blood-testis barrier (BTB) function hampers nutrient delivery from Sertoli cells to spermatocytes, which can cause thinning of the germinal epithelium [41]. Research by Tian et al. [42] Shows that oxidative stress induced by diabetes activates destructive autophagy, worsening testicular epithelial degeneration and reducing testosterone synthesis by disrupting the functional interactions between Leydig and Sertoli cells. In this study, the reduction in tubule diameter and epithelial thickness in the diabetic group reflects the cumulative effects of oxidative stress and inflammation induced by diabetes.
The seminiferous tubules are the main part of the testis, accounting for about 80%, and are the site of spermatogenesis. Histological examination of the testis with HE staining shows a significant difference between groups p < 0.05. The largest seminiferous tubules are observed in the healthy rat group (N). A decrease in testosterone levels directly impacts the inhibition of spermatogenesis because testosterone plays a crucial role in the maturation of spermatogenic cells and provides nutritional support from Sertoli cells [43]. As a result, the number of spermatogenic cells within the lumen of the seminiferous tubules decreases, which histologically appears as a narrowing of the tubule diameter [44]. In the testes, excessive infiltration and activation of CD4⁺ and CD8⁺ T lymphocytes disrupt the immune-privileged environment, impair Sertoli cell function, and compromise the integrity of the blood–testis barrier. This inflammatory milieu suppresses steroidogenic enzyme expression in Leydig cells and interferes with gonadotropin signaling, thereby contributing to reduced testosterone production and impaired spermatogenesis. The administration of CGA in the present study may attenuate these inflammatory processes through its anti-inflammatory properties, including inhibition of NF-κB signaling and downregulation of pro-inflammatory cytokine production. By reducing T-cell–mediated inflammatory responses, CGA potentially supports the restoration of hormonal balance and preservation of testicular structure under diabetic conditions, thereby complementing its antihyperglycemic and antioxidant effects [40]. CGA is able to penetrate the testicular tissue, resulting in an increase in epithelial thickness that can be observed in the diabetic group given graded doses of CGA.
CGA has a stronger histological improvement effect compared to metformin (p < 0.05) because it works directly on antioxidant and anti-inflammatory pathways. CGA activates the transcription factor Nrf2, which increases the transcription of antioxidant genes such as Heme Oxygenase-1 (HO-1) and NAD(P)H Quinone Oxidoreductase 1 (NQO1). Activation of this pathway protects the integrity of the BTB and decreases caspase-3 expression, thereby preventing germinal epithelial apoptosis [45]. CGA increases SOD and catalase activity, improves microcirculation in the testes, and normalizes the morphology of the seminiferous tubules [46]. In this study, the group receiving 25 mg/kg BW of CGA showed tubule diameter and epithelial thickness close to normal, indicating optimal testicular tissue regeneration. These effects are also related to the suppression of the NF-κB and TNF-α pathways, which play roles in testicular degeneration due to hyperglycemia [47].
Spermatogenesis in rats with diabetes
Spermatogenesis examination using the Johnsen score criteria showed a decrease in the DM group. In the treatment groups with the administration of metformin and CGA, the scores increased with increasing doses. The DMCGA 6.25 mg group had a score of 6.92 ± 0.36, the DMCGA 12.5 mg group had a score of 8.68 ± 0.50, while the DMCGA 25mg group achieved a score close to 9.80 ± 0.24, indicating complete spermatogenesis with spermatozoa in the lumen (Figure 4). The results of the one-way ANOVA test on the spermatogenesis score variable showed a significant difference between groups (p < 0.05). The post hoc Tukey test indicated that the DM, DMet, DMCGA 65mg, and DMCGA 12.5 mg groups differed significantly from the normal group, with p < 0.05. However, only the DMCGA 25mg group showed a non-significant difference when compared to the N group.
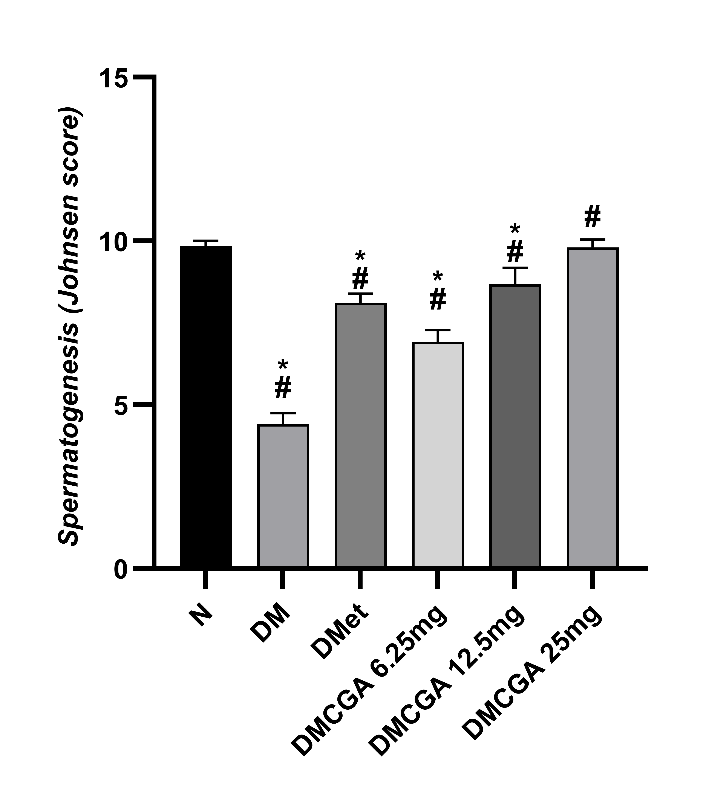
Figure 4 Spermatogenesis score (Johnsen score) in male rats after 31 days of treatment. The average value (Mean ± SD): N = 9.84 ± 0.17; DM = 4.40 ± 0.35; DMet = 8.12 ± 0.27; DMCGA 6.25m g = 6.92 ± 0.36; DMCGA 12.5 mg = 8.68 ± 0.50; and DMCGA 25 mg = 9.80 ± 0.24. *Significantly different compared to the normal group (N) (p < 0.05) and # significantly different compared to the DM group (p < 0.05).
The increase in BGLs causes oxidative stress and accumulation of ROS in testicular tissue. ROS damages the DNA of spermatogenic cells, thereby disrupting mitochondrial activity that is essential for meiosis division, as well as reducing cellular energy needed during spermiogenesis. The increase in AGEs alters protein structure and inhibits spermatid differentiation. Lipid peroxidation caused by HFD worsens this damage by triggering inflammation and germ cell apoptosis. The decrease in the expression of antioxidant genes such as SOD1 and GPx worsens the redox imbalance within the testis, leading to a reduction in the number of spermatocytes and spermatids, as reflected in a low Johnsen score [48]. The administration of metformin provides partial improvement of spermatogenesis through metabolic and antioxidant mechanisms. Activation of AMPK by metformin increases insulin sensitivity and improves energy balance in spermatogenic cells, thereby enhancing ATP synthesis, which is essential for meiosis and spermiogenesis processes. Metformin also inhibits the activation of the NF-κB inflammatory pathway, reducing the expression of proapoptotic genes such as Bax and caspase-9 [49].
In the DMCGA group, especially with a dose of 25 mg/kg BW, a stronger protective and regenerative effect was observed compared to metformin. CGA improves mitochondrial function and reduces ROS through activation of the Nrf2/HO-1 pathway, which increases the expression of antioxidant enzymes. CGA also decreases Bax expression and increases Bcl-2 inhibiting apoptosis in spermatogenic cells [50]. CGA regulates mitochondrial homeostasis and suppresses NLRP3 inflammasome activation, thus maintaining the energy balance of germ cells [18]. In this study, the DMCGA 25 mg/kg BW group showed a Johnsen score of ≥ 9, approaching the normal value. This indicates that DMCGA 25 mg/kg BW group was improvement in tubular morphology and spermatogenic features compared to the diabetic group. Additionally, CGA enhances Sertoli cell function by increasing the production of androgen-binding protein (ABP) and nutritional factors that support spermatid maturation. Activation of Nrf2 by CGA also suppresses inflammation by reducing TNF-α and IL-1β levels in testicular tissue. These results align with the findings of Al-Megrin et al. [46] who reported that green coffee extract with high CGA content increased testosterone levels and improved Johnsen scores in diabetic rats, as well as Demir et al. [51] who found that CGA protects the testes from oxidative damage through increased expression of Nrf2 and Bcl-2 In the DM group, tissue showed a drastic decrease in the spermatogenic layer, tubule diameter, and the number of mature spermatids. CGA demonstrated overall improvement compared to metformin, from the molecular level to morphology, with restoration of the germinal epithelial structure to that of the normal group. The study by Owumi et al. [52] Supports these findings by showing that CGA increases the number of spermatids and mature sperm by enhancing the activity of antioxidant enzymes.
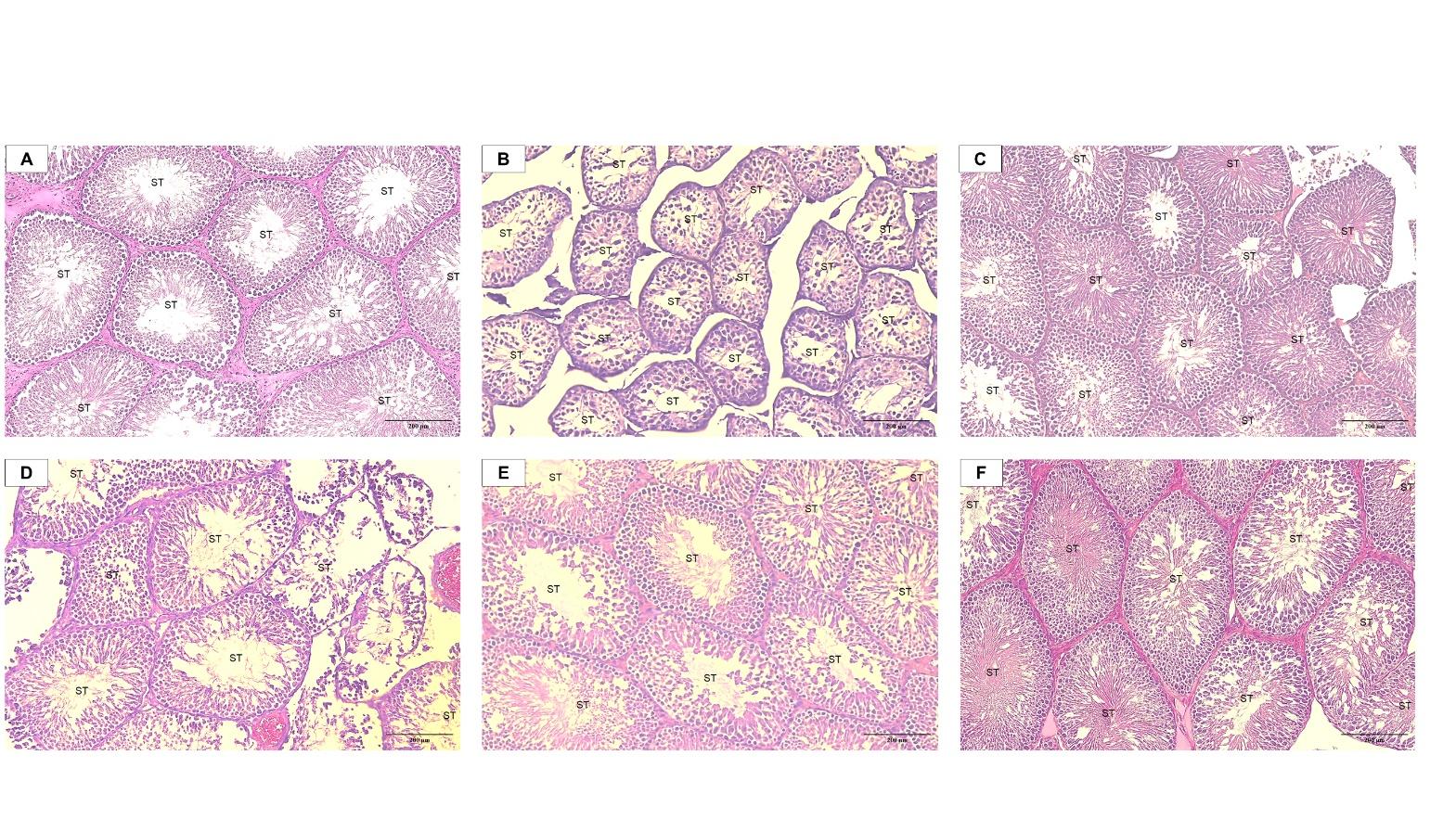
Figure 5 Representative photomicrographs of rat testicular tissue stained with hematoxylin and eosin (H&E). (A) Normal control group (N) showing intact and well-organized seminiferous tubules (ST) with complete spermatogenic layers. (B) Diabetic control group (DM) showing tubular atrophy, epithelial disorganization, and reduced spermatogenic cell layers. (C) Diabetic rats treated with metformin (DMet) showing partial improvement in seminiferous tubule architecture and germinal epithelium thickness. (D) Diabetic rats treated with chlorogenic acid (CGA) at 6.25 mg/kg BW showing mild preservation of tubular structure with limited spermatogenic organization. (E) Diabetic rats treated with CGA at 12.5 mg/kg BW showing improved spermatogenic organization and increased epithelial thickness. (F) Diabetic rats treated with CGA at 25 mg/kg BW showing marked improvement in seminiferous tubule morphology, characterized by a dense germinal epithelium and increased presence of spermatozoa within the lumen compared to the diabetic group. All images were captured at the same magnification (200×; scale bar = 200 µm).
Figure 5 illustrates the histological differences in testicular structure among the experimental groups. The normal control group exhibited intact seminiferous tubules with thick germinal epithelium and complete spermatogenic layers. In contrast, the diabetic control group showed severe histological alterations, including tubular atrophy, epithelial thinning, lumen collapse, and reduced spermatogenic cell populations, corresponding to low Johnsen scores (3 - 4). Treatment with metformin resulted in partial restoration of tubular architecture, characterized by improved epithelial thickness and the reappearance of developing germ cells, with Johnsen scores ranging from 7 to 8. The DMCGA 6.25 mg/kg BW group demonstrated mild histological improvement, with preservation of basic tubular structure, limited regeneration of spermatogenic cells, and Johnsen scores of 5 - 6. More pronounced improvements were observed in the DMCGA 12.5 (7 - 8 scores) and 25 mg/kg BW (9 - 10 scores) groups, which exhibited increased epithelial thickness, better-organized spermatogenic layers, and higher Johnsen scores approaching those of the normal control group.
DM has been proven to disrupt the male reproductive system through chronic hyperglycemia, which triggers oxidative stress, apoptosis, and germ cell dysfunction. A study by Pavlova et al. [53] shows that DM causes a significant decrease in spermatogenic cell populations and the diameter of seminiferous tubules in male rats. Zhang et al. [14] add that DM triggers damage to the HPG axis which can inhibiting spermatid differentiation and reducing sperm quality. These findings are consistent, with the DM group showing lumen collapse, thinning epithelium, and a Johnsen score of 3 - 4. Such damage reflects an atrophic phase due to the loss of hormonal stimulation from the HPG axis.
CGA is a natural polyphenol compound with strong antioxidant activity, in line with the research by Demir et al. [51] reported that CGA protects the testes from damage caused by cisplatin by decreasing MDA and increasing SOD and CAT. CGA also plays a role in lowering BGLs, improving lipid metabolism, and stabilizing gonadal hormones. In this study, CGA at a dose of 6.25 mg/kg body weight showed a mild improvement, with the epithelium beginning to thicken and some round spermatids appearing, although the lumen remained empty. This effect aligns with the mechanism of CGA working through the activation of Nrf and the inhibition of NF-κB [54]. Impaired spermatogenesis in diabetic rats, as indicated by reduced Johnsen scores, reflects disrupted germ cell maturation and decreased spermatozoa formation. Restoration of spermatogenesis following CGA administration is closely associated with improved hormonal support and preservation of testicular microarchitecture. The highest CGA dose specifically 12.5 and 25 mg/kg BW resulted in spermatogenesis scores approaching those of the normal control group, indicating substantial functional recovery.
Correlation between BGLs, LH, FSH, testosterone levels, diameter of seminiferous tubule, epithelial thickness, and spermatogenesis score
The relationship between BGLS variables, reproductive hormones, testis histology, and spermatogenesis scores is presented in Figure 6.
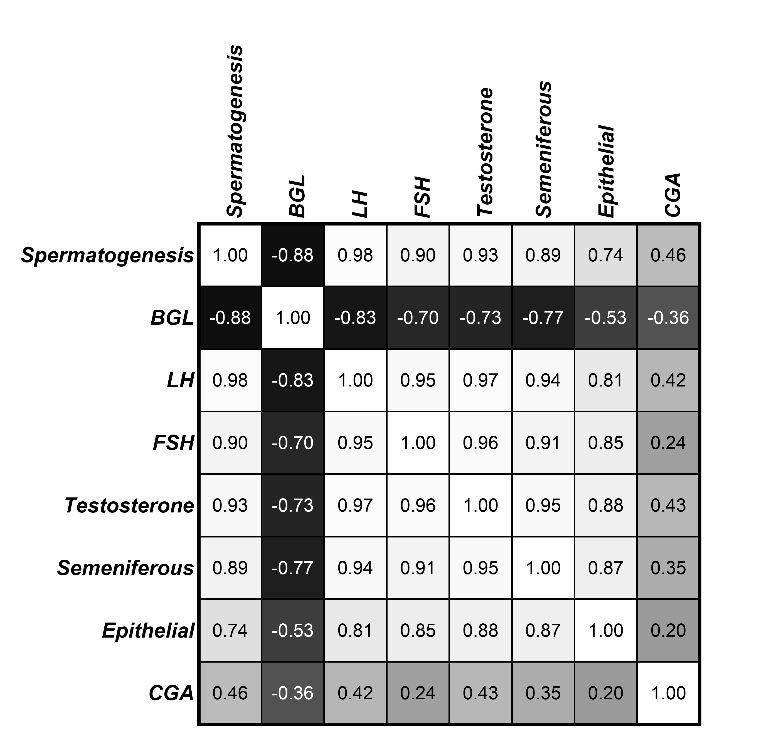
Figure 6 The heatmap shows the Pearson correlation between variables: Spermatogenesis, blood glucose level (BGLs), luteinizing hormone (LH), follicle-stimulating hormone (FSH), Testosterone, diameter of seminiferous tubules, germinal epithelium thickness, and dose of chlorogenic acid (CGA). Darker colors indicate strong correlations, while lighter colors indicate weak or insignificant correlations (p < 0.01).
Pearson correlation analysis revealed that BGLs exhibited a strong negative correlation with
spermatogenesis
(r = −0.88; p
< 0.001), luteinizing hormone (LH; r = −0.83; p
< 0.001), follicle-stimulating hormone (FSH; r = −0.70; p
<
0.01), testosterone (r = −0.73; p
< 0.001), seminiferous tubule diameter (r = −0.77;
p
< 0.001), and epithelial thickness
(r = −0.53; p
< 0.05). Spermatogenesis was strongly and positively correlated
with LH (r = 0.98), FSH (r = 0.90), and testosterone (r = 0.93), as
well as with seminiferous tubule diameter (r = 0.89) and epithelial
thickness (r = 0.74). Among testicular histological parameters,
testosterone showed the highest correlation with seminiferous tubule
diameter (r = 0.95) and epithelial thickness (r = 0.88), indicating
that androgen activity plays a crucial role in maintaining tubular
integrity. CGA showed a weak but positive association with
spermatogenesis (r = 0.46) and testosterone (r = 0.43), suggesting
that CGA may indirectly improve reproductive function by modulating
hormonal balance and testicular histology. Overall, these findings
indicate that elevated BGLs impair the HPG axis and spermatogenic
process, while CGA supplementation tends to be associated with
improvements in hormonal parameters and testicular histological
features. All correlations were statistically significant at the 99%
confidence level (p
< 0.01).
This result is consistent with the study by Zhang et al. [55] which shows that activation of AMPK and Nrf2 by CGA reduces systemic oxidative stress and improves HPG axis communication in DM animals. The mechanism of diabetes involves increased BGLs, which trigger oxidative stress and the accumulation of AGEs These activate the PKC–NFκB and TNF-α pathways, ultimately causing apoptosis of Leydig and Sertoli cells. A meta-analysis study by Tehrani et al. [56] showed that serum FSH levels are significantly lower in men with T2DM compared to healthy controls. Clinical research in adult men indicates a high prevalence of hypogonadism in T2DM patients. It also disrupts the HPG axis, decreasing LH, FSH, and testosterone levels, and suppresses the expression of steroidogenesis enzymes such as StAR and P450scc [57]. Venditti et al. [22] states that diabetes affects steroidogenesis and testicular tissue through ROS, AGEs, inflammation, mitochondrial dysfunction, and the NLRP3 inflammatory pathway. This condition worsens the decline in testosterone and hampers nutritional support by Sertoli cells to spermatogonia. This study shows a negative correlation between BGLs and reproductive hormones, which is a combination of metabolic and oxidative effects impacting the entire HPG axis.
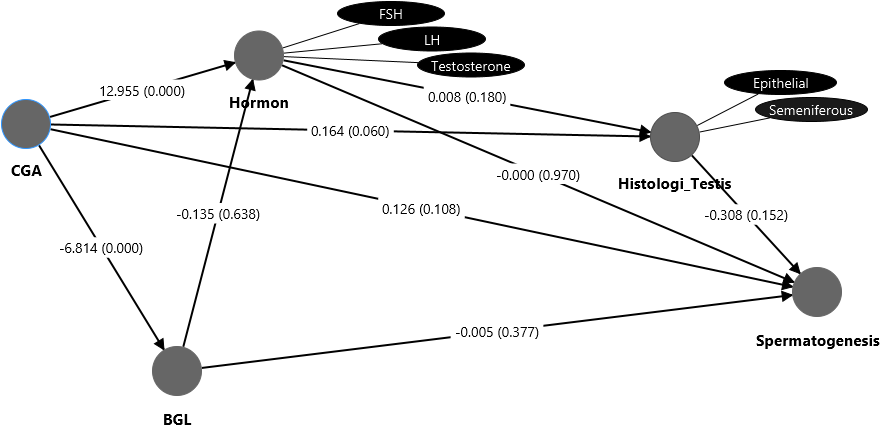
Figure 7 The SmartPLS pathway diagram displays the structural relationships among chlorogenic acid (CGA), blood glucose level (BGLs), hormone (LH, FSH, testosterone levels), testis histology, and spermatogenesis score. Each arrow indicates the direction of causal influence between variables. The values on each arrow represent the path coefficient (O), which indicates the direction and strength of the influence, as well as the significance value (p-value) that shows whether the relationship is statistically meaningful. A positive O value indicates a direct relationship (an increase in variable X followed by an increase in variable Y), while a negative O value indicates an inverse relationship. Relationships are considered significant if p < 0.05 and not significant if p > 0.05.
The structural relationships among CGA, BGLs, reproductive hormones, testicular histology, and
spermatogenesis are illustrated in Figure 7. This pathway model summarizes the direct and indirect effects of CGA on metabolic and reproductive parameters under diabetic conditions. The structural model results show that CGA has a very strong and significant effect on BGLs with a large negative path coefficient (O = −6.814; p = 0.000), as well as a significant positive effect on reproductive hormones such as LH, FSH, and testosterone (O = 12.955; p = 0.000). These results indicate that administering CGA is effective in lowering BGLs and increasing hormonal activity through stimulation of the HPG axis. The path between CGA and testicular histology shows a positive direction but is not significant (O = 0.164; p = 0.060), suggesting that improvements in testicular histological structure tend to occur indirectly through increased hormone levels and decreased blood glucose. The path between BGLs and spermatogenesis (O = −0.005; p = 0.377) is also not significant, indicating that the effect of hyperglycemia on spermatogenesis is mediated by decreases in hormone levels and histology, rather than directly. Additionally, the relationship between testicular histology and spermatogenesis (O = −0.308; p = 0.152) shows a non-significant negative correlation, reflecting that microscopic changes in testicular tissue do not always directly lead to improved spermatogenesis quality within this treatment duration.
The CGA pathway in the SmartPLS model can be considered the main causal relationship between metabolic improvement and reproductive function. This CGA not only acts as an antioxidant but also as a metabolic-endocrine modulator that simultaneously improves glucose and hormonal balance. On the CGA to testicular histology pathway, such as diameter of seminiferous tubules and epithelial thickness (O = 0.164; p = 0.060), indicates that CGA also has a direct effect on testicular histology. Gül et al. [58] confirms that CGA activates Nrf2/HO-1 and suppresses inflammation by inhibiting the NLRP3 inflammasome. Although in this study the Histology to Spermatogenesis pathway (O = −0.308; p = 0.152) was not significantly different, the positive histological improvement still plays an important role in germ cell regeneration. The hormonal pathway to spermatogenesis (O = 0.008; p = 0.180) indicates that reproductive hormones are the main mediators between the effects of CGA and the recovery of spermatogenic function. Although the p-value is slightly above the significance threshold, the large coefficient suggests a physiologically relevant strength of the relationship. The study by Owumi et al. [52] supports these results which shows the administration of CGA restores testosterone levels and improves spermatogenesis in male rats experiencing reproductive toxicity caused by tamoxifen. The proposed mechanism involves increased expression of Bcl-2 and decreased Bax and caspase-3, which aligns with increased testosterone levels.
The results from SmartPLS indicate that the mechanism of CGA acts through a multifactorial and interconnected process, starting with the reduction of BGLs via AMPK activation, followed by Nrf2 activation, increased endogenous antioxidants, hormonal improvements, and ultimately testicular morphological improvements that lead to enhanced spermatogenesis. Nrf2 activation decreases testicular cell apoptosis and improves spermatogenesis in diabetic rats [22]. These findings are also consistent with Cortez et al. [59] which explains that CGA acts as a protective phytochemical with antioxidant, anti-inflammatory, and genetic modulation effects on the AMPK/Nrf2 pathway. SmartPLS not only confirms the statistical relationships among variables but also validates the tangible biological basis that CGA works through an integrated metabolic endocrine histological mechanism. Based on the analysis results from this study, CGA can be developed as a promising phytopharmaceutical for managing male reproductive complications associated with diabetes mellitus. The increase in reproductive hormones mediated by CGA is the most effective mediator for improving spermatogenesis, while testicular histological recovery requires a longer duration like a of ≥ 52 days because that is approaching one cycle of rat spermatogenesis [60,61].
To integrate the metabolic, hormonal, and histological findings of this study, a graphical summary of the mechanisms is presented (Figure 8). CGA administration was associated with improved glycemic control, modulation of reproductive hormone levels, preservation of testicular histological features, and enhanced spermatogenesis in diabetic rats. This schematic represents a hypothesis-generating model based on the measured parameters of the present study.
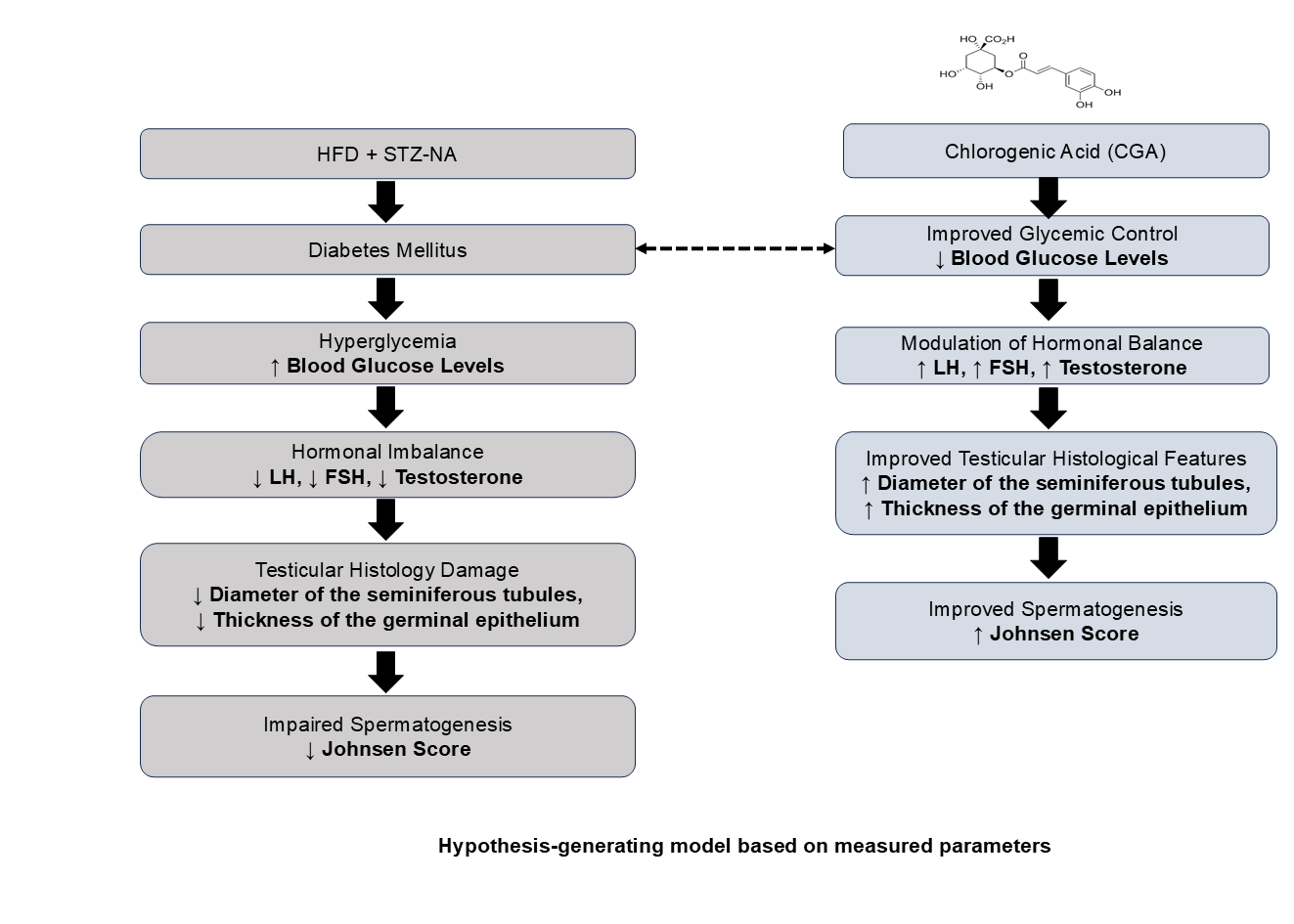
Figure 8 Graphical mechanism summary illustrating the proposed pathways by which chlorogenic acid (CGA) may ameliorate diabetes-induced testicular dysfunction.
CGA administration was associated with improved glycemic control and hormonal regulation. Based on these observations, CGA is suggested to support spermatogenesis primarily through metabolic improvement and hormonal regulation rather than through direct molecular pathways that were not assessed in this study. Accordingly, the graphical summary is presented as a hypothesis-generating model that reflects the measured parameters and provides a conceptual framework for future mechanistic investigations.
This study has several limitations that should be acknowledged. First, the sample size in each experimental group was relatively small (n = 5), which is commonly applied in controlled preclinical animal studies with comparable experimental designs. Several experimental studies investigating induced diabetes in rats have used comparable sample sizes and reported consistent biological and statistical results. Although the present sample size is modest, the observed dose-dependent effects across metabolic, hormonal, histological, and spermatogenic parameters support the reliability of the findings. Nevertheless, future studies with larger sample sizes and extent [62,63].
Conclusions
This study demonstrates that CGA is associated with improvements in glycemic control, reproductive hormone levels, testicular histological features, and spermatogenesis in a diabetic male rat model in a dose-dependent manner. These findings suggest a potential protective role of CGA on endocrine and testicular function under diabetic conditions. However, the relatively small sample size, the use of an animal model, and the intraperitoneal route of administration limit direct extrapolation to clinical settings. Future studies should focus on longer treatment durations, oral administration, and molecular-level investigations to further elucidate the mechanisms underlying CGA-mediated effects and to evaluate its use in combination with standard antidiabetic therapies. Although CGA demonstrates promising preclinical potential, further experimental and clinical validation is required before any clinical application can be considered.
Acknowledgements
This research was supported by Diponegoro University, Indonesia, which funded this research. The Institute for Research and Community Service of Diponegoro University, Indonesia funded this research through the Research Implementation Assignment Letter for the International Publication Research Scheme (No. 222-410/UN7.D2/PP/IV/2025).
Declaration of Generative AI in Scientific Writing
The authors state that Grammarly and other generative AI technologies were employed to aid in the writing process, particularly for grammar correction and language improvement. The original content, data analysis, and result interpretation were all done without the use of these technologies. The manuscript has been thoroughly examined by the authors, who accept full responsibility for its accuracy and conclusions.
CRediT Author Statement
Eko Naning Sofyanita: Conceptualization; Methodology; Software; Resources; Data Curation; Writing - Original Draft; Writing - Review & Editing. Eka Chandra Herlina: Formal analysis; Investigation; Visualization; Supervision. Achmad Zulfa Juniarto: Conceptualization; Methodology; Data Curation; Validation; Project administration; Funding acquisition.
References
[1] NE Asmar, BM Arafah and CK Smith. Diabetes mellitus. In: PM Paulman, RB Taylor, AA Paulman and LS Nasir (Eds.). Family Medicine. Springer, Cham, 2022, p. 173-187.
[2] A Monjed. Diabetes Mmellitus. In: W Al-Mohaimeed, M Al-Bishri and M Al-Agha (Eds.). Skills in Rheumatology. Springer, Singapore, 2021, p. 445-460.
[3] M Younes, G Aquilina, L Castle, GH Degen, KH Engel, PJ Fowler, P Fürst, R Gürtler, J König, C Lambré, A Mortensen, C Schlatter and D Arcella. Flavouring Group Evaluation 413 (FGE.413): Naringenin. EFSA Journal 2024; 22(4), e8747.
[4] MF El-Khadragy, WA Al-Megrin, S Alomar, AF Alkhuriji, DM Metwally, S Mahgoub, SS Abdel-Maksoud and MA Arisha. Chlorogenic acid abates male reproductive dysfunction in arsenic-exposed mice via attenuation of testicular oxido-inflammatory stress and apoptotic responses. Chemico-Biological Interactions 2021; 333, 109333.
[5] A Kumar, R Gangwar, AA Zargar, R Kumar and A Sharma. Prevalence of diabetes in India: A review of IDF Diabetes Atlas 10th edition. Current Diabetes Reviews 2024; 20(1), 105-114.
[6] PR Roshni and B Baby. Amylin in diabetes mellitus. In: A Kumar and M Siddiqui (Eds.). Diabetes mellitus: A Review. Elsevier, Amsterdam, 2025.
[7] P Pandey, V Jakhmola, S Saha and A Gaurav. Recent developments in diabetes management: Exploring receptors, pathways and compounds. BIO Integration 2025; 6(1), 1-28.
[8] O Ameen, RM Samaka and RAA Abo-Elsoud. Metformin alleviates neurocognitive impairment in aging via activation of AMPK/BDNF/PI3K pathway. Scientific Reports 2022; 12(1), 17084.
[9] E Rahmanian, N Salari, M Mohammadi and R Jalali. Evaluation of sexual dysfunction and female sexual dysfunction indicators in women with type 2 diabetes: A systematic review and meta-analysis. Diabetology & Metabolic Syndrome 2019; 11, 73.
[10] EN Sofyanita, AZ Juniarto, A Suwondo and H Nugroho. The effect of chlorogenic acid content in coffee can reduce malondialdehyde (MDA) and increase testosterone hormone levels in DM conditions. Gema Lingkungan Kesehatan 2025; 23(1), 20-26.
[11] N Abedpour, MZ Javanmard, M Karimipour and AP Liqvan. Effect of chlorogenic acid on follicular development, hormonal status and biomarkers of oxidative stress in rats with polycystic ovary syndrome. Veterinary Research Forum 2022; 13(4), 513-520.
[12] N Gusti, T Heriansyah and I Saputra. Association between admission blood sugar levels and length of stay among patients with acute heart failure: A cross-sectional study in Aceh, Indonesia. Narra X 2023; 1(2), e75.
[13] J Rong, X Leng, K Jiang, J Tan and M Dong. Systemic impacts of diabetes on spermatogenesis and intervention strategies: Multilayered mechanism analysis and cutting-edge therapeutic approaches. Reproductive Biology and Endocrinology 2025; 23(1), 122.
[14] W Zhang, L Tong, B Jin and D Sun. Diabetic testicular dysfunction and spermatogenesis impairment: Mechanisms and therapeutic prospects. Frontiers in Endocrinology 2025; 16, 1653975.
[15] OC Badejogbin, OP Akano, OEB Julius, MO Agunloye, MV Olubiyi, OE Chijioke-Agu, MA Obekpa, AP Arikawe and KS Olaniyi. Potential role of CREM in diabetes-associated testicular dysfunction: Current evidence and future perspectives. Reproductive Medicine and Biology 2025; 24(1), e12661.
[18] H Shoorei, A Khaki, M Shokoohi, AA Khaki, A Alihemmati, M Moghimian and SH Abtahi-Eivary. The ameliorative effect of carvacrol on oxidative stress and germ cell apoptosis in testicular tissue of adult diabetic rats. Biomedicine & Pharmacotherapy 2019; 111, 568-578.
[17] H Shoorei, A Khaki, M Shokoohi, AA Khaki, A Alihemmati, M Moghimian and SH Abtahi-Eivary. Evaluation of carvacrol on pituitary and sexual hormones and their receptors in the testicle of male diabetic rats. Human & Experimental Toxicology 2020; 39(8), 1019-1030.
[18] ZC Jia, SJ Liu, TF Chen, ZZ Shi, XL Li, ZW Gao, Q Zhang and CF Zhong. Chlorogenic acid can improve spermatogenic dysfunction in rats with varicocele by regulating mitochondrial homeostasis and inhibiting the activation of NLRP3 inflammasomes by oxidative mitochondrial DNA and cGAS/STING pathway. Bioorganic Chemistry 2024; 150, 107571.
[19] AK Singh and AK Pandey. Alleviation of diabetes mellitus–induced reproductive dysfunction by chlorogenic acid in male rats via combating redox imbalance. Indian Journal of Clinical Biochemistry 2025; 40(1), 1-9.
[20] YMA El-Hakim, AAR Mohamed, SI Khater, AH Arisha, MMM Metwally, MA Nassan and ME Hassan. Chitosan-stabilized selenium nanoparticles and metformin synergistically rescue testicular oxidative damage and steroidogenesis-related genes dysregulation in high-fat diet/streptozotocin-induced diabetic rats. Antioxidants 2021; 10(1), 17.
[21] CY Liu, TC Chang, SH Lin, ST Wu, TL Cha and CW Tsao. Metformin ameliorates testicular function and spermatogenesis in male mice with high-fat and high-cholesterol diet-induced obesity. Nutrients 2020; 12(7), 1932.
[22] M Venditti, MZ Romano, S Boccella, A Haddadi, A Biasi, S Maione and S Minucci. Type 1 diabetes impairs the activity of rat testicular somatic and germ cells through NRF2/NLRP3 pathway-mediated oxidative stress. Frontiers in Endocrinology 2024; 15, 1399256.
[23] IO Kazaz, S Demir, G Kerimoglu, F Colak, NT Alemdar, SY Dogan and S Bostan. Chlorogenic acid ameliorates torsion/detorsion-induced testicular injury via decreasing endoplasmic reticulum stress. Journal of Pediatric Urology 2022; 18(3), 289.e1-289.e7.
[24] I Sari, DM Rizal and RA Syarif. The effect of chlorogenic acid on endoplasmic reticulum stress and steroidogenesis in the testes of diabetic rats: Study of mRNA Expressions of GRP78, XBP1s, 3β-HSD, and 17β-HSD. BIO Web of Conferences 2022; 49, 01001.
[25] C Dunn, D Brettle, M Cockroft, E Keating, C Revie and D Treanor. Quantitative assessment of H & E staining for pathology: Development and clinical evaluation of a novel system. Diagnostic Pathology 2024; 19(1), 42.
[26] REP Köprülü, ME Okur, B Kolbaşi, İ Keskin and H Ozbek. Effects of vincamine on testicular dysfunction in alloxan-induced diabetic male rats. Iranian Journal of Pharmaceutical Research 2022; 21(1), e132265.
[27] JF Hair, GTM Hult, CM Ringle and M Sarstedt. A primer on partial least squares structural equation modeling (PLS-SEM). Sage Publications Inc., Thousand Oaks, 2017.
[28] N Nazaruddin, TN Siregar, A Sutriana, S Wahyuni and T Armansyah. The impact of streptozotocin-induced diabetes on testosterone hormone and androgen receptor expression and correlation with sperm quality impairment in Sprague Dawley rats. Trends in Sciences 2025; 22(9), 10409.
[29] F Marino, N Salerno, M Scalise, L Salerno, A Torella, C Molinaro, A Chiefalo, A Filardo, C Siracusa, G Panuccio, C Ferravante, G Giurato, F Rizzo, M Torella, M Donniacuo, AD Angelis, G Viglietto, K Urbanek, A Weisz, D Torella and E Cianflone. Streptozotocin-induced type 1 and 2 diabetes mellitus mouse models show different functional, cellular and molecular patterns of diabetic cardiomyopathy. International journal of molecular sciences 2023; 24(2), 1132.
[30] S Ramachandran, R Nandhini and R Felix. Streptozotocin-induced diabetes: Mechanisms, toxicity and experimental considerations. In: Advances in Experimental Pharmacology. Springer, Singapore, 2022, p. 87-112.
[31] KI Jung, JS Han and CK Park. Neuroprotective effects of nicotinamide (vitamin b₃) on neurodegeneration in diabetic rat retinas. Nutrients 2022; 14(6), 1162.
[32] LJ Yan. The nicotinamide/streptozotocin rodent model of type 2 diabetes: Renal pathophysiology and redox imbalance features. Biomolecules 2022; 12(9), 1225.
[33] M Sianturi, N Susilaningsih, H Nugroho, N Suci, TN Kristina and M Suryani. Effect of lycopene and metformin combination on phagocytosis, glycemic control, and oxidative stress in rats with type 2 diabetes. Medical Journal of Indonesia 2023; 32(1), 1-6.
[34] F Zamani-Garmsiri, G Ghasempour, M Aliabadi, SMR Hashemnia, S Emamgholipour and R Meshkani. Combination of metformin and chlorogenic acid attenuates hepatic steatosis and inflammation in high-fat diet fed mice. IUBMB Life 2021;73(1), 252-263.
[35] V Nguyen, EG Taine, D Meng, T Cui and W Tan. Chlorogenic acid: A systematic review on the biological functions, mechanistic actions, and therapeutic potentials. Nutrients 2024; 16(7), 924.
[36] D Pan, L Xu and M Guo. The role of protein kinase C in diabetic microvascular complications. Frontiers in Endocrinology 2022; 13, 973058.
[37] EN Sofyanita, MR Maulana, P Lestari and Burhannudin. The effect of robusta coffee (Coffea canephora) on the expression of sod and nrf-2 in diabetes mellitus condition (animal model). Jurnal Ilmu Kesehatan 2024; 18(3), 256-262.
[38] M Du, S Chen, Y Chen, X Yuan and H Dong. Testicular fat deposition attenuates reproductive performance via decreased follicle-stimulating hormone level and sperm meiosis and testosterone synthesis in mouse. Animal Bioscience 2024; 37(1), 50-60.
[39] W He, H Liu, L Hu, Y Wang, L Huang, A Liang, X Wang, Q Zhang, Y Chen, Y Cao, S Li, J Wang and X Lei. Icariin improves testicular dysfunction via enhancing proliferation and inhibiting mitochondria-dependent apoptosis pathway in high-fat diet and streptozotocin-induced diabetic rats. Reproductive Biology and Endocrinology 2021; 19(1), 168.
[40] B Muchtaromah, AM Firdaus, AN Ansori, MR Duhita, EB Minarno, A Hayati, M Ahmad and I Analisa. Effect of pegagan (Centella Asiatica) nanoparticle coated with chitosan on the cytokine profile of chronic diabetic mice. Narra J 2024; 4(1), e697.
[41] JA Kanaley, SR Colberg, MH Corcoran, SK Malin, NR Rodriguez, CJ Crespo, JP Kirwan and JR Zierath. Exercise/physical activity in individuals with type 2 diabetes: A consensus statement from the American College of Sports Medicine. Medicine and Science in Sports and Exercise 2022; 54(2), 353-368.
[42] Y Tian, W Song, D Xu, X Chen, X Li and Y Zhao. Autophagy induced by ROS aggravates testis oxidative damage in diabetes via breaking the feedforward loop linking p62 and Nrf2. Oxidative Medicine and Cellular Longevity 2020; 2020, 7156579.
[43] S Nikbin, A Derakhshideh, SK Jafari, A Mirzahamedani, A Moslehi, S Ourzamani, E Barati, F Amini, FS Zolfaghari and MA Azarbayjani. Investigating the protective effect of aerobic exercise on oxidative stress and histological damages of testicular tissue associated with chlorpyrifos in male rats. Andrologia 2020; 52(2), e13468.
[44] RA Septiani, IS Hamid, EK Sabdoningrum, A Ma’Ruf, EP Hestianah and M Mafruchati. Tomato (Lycopersicon esculentum Mill.) juice restored the number of Leydig cells and the diameter of the seminiferous tubules of mice (Mus musculus) exposed to lead acetate. Ovozoa Journal of Animal Reproduction 2022; 11(3), 123-129.
[45] M Yahayu, Z Begam, S Hamid, H Adawi, DJ Dailin, RA Malek, S Zulaiha Hanapi, A Boumehira, N Sulaiman, I Yusoff, T Ho and H El-Enshasy. Antioxidant activity in green and roasted coffee: A critical review. Bioscience Research 2020; 17(3), 2249-2263.
[46] WA Al-Megrin, MF El-Khadragy, MH Hussein, S Mahgoub, DM Abdel-Mohsen, H Taha, AAA Bakkar, AEA Moneim and HK Amin. Green coffea arabica extract ameliorates testicular injury in high-fat diet/streptozotocin-induced diabetes in rats. Journal of Diabetes Research 2020; 2020, 6762709.
[47] A Mansour, MR Mohajeri-Tehrani, M Samadi, M Qorbani, S Merat, H Adibi and H Poustchi. Effects of supplementation with main coffee components including caffeine and/or chlorogenic acid on hepatic, metabolic, and inflammatory indices in patients with non-alcoholic fatty liver disease and type 2 diabetes: A randomized, double-blind, placebo-c. Nutrition Journal 2021; 20, 35.
[48] SY Guo, XY Zhang, YX Yu, LQ Xie and CQ Chang. Effects of chlorogenic acid on glucose tolerance and its curve characteristics in high-fat diet-induced obesity rats. Journal of Peking University Health Sciences 2020; 52(2), 269-274.
[49] F Namvarjah, HS Afra, HM Sardareh, RB Khorzoughi, P Pasalar, G Panahi and R Meshkani. Chlorogenic acid improves anti-lipogenic activity of metformin by positive regulating of AMPK signaling in HepG2 cells. Cell Biochemistry and Biophysics 2022; 80, 537-545.
[50] E Hermawati, N Arfian, Mustofa and G Partadiredja. Chlorogenic acid ameliorates memory loss and hippocampal cell death after transient global ischemia. European Journal of Neuroscience 2020; 51(2), 651-669.
[51] EA Demir, S Demir, SA Mungan, NT Alemdar, A Menteşe and Y Aliyazıcıoğlu. Chlorogenic acid protects against cisplatin-induced testicular damage: A biochemical and histological study. Institute for Medical Research and Occupational Health 2025; 76(2), 130-137.
[52] SE Owumi, RA Anaikor, UO Arunsi, OA Adaramoye and AK Oyelere. Chlorogenic acid co-administration abates tamoxifen-mediated reproductive toxicities in male rats: An experimental approach. Journal of Food Biochemistry 2021; 45(2), e13615.
[53] E Pavlova, R Ivanov, D Abadjieva, Y Gluhcheva, E Petrova, I Vladov, E Lakova and N Atanassova. Evaluation of rat testicular cell populations in experimental condition of diabetes induced in early postnatal life. Cells 2025; 14(21), 1714.
[54] RF Alenezi, A Abdelkhalek, G El-Sayed, I Pet, M Ahmadi, ESE Sherbini, D Pușcașiu and AH Arisha. A natural polyphenol, chlorogenic acid, attenuates obesity-related metabolic disorders in male rats via miR-146a–IRAK1–TRAF6 and NRF2-mediated antioxidant pathways. Biomolecules 2025; 15(8), 1086.
[55] Z Zhang, C Shi and Z Wang. Therapeutic effects and molecular mechanism of chlorogenic acid on polycystic ovarian syndrome: Role of HIF-1alpha. Nutrients 2023; 15(3), 2833.
[56] FR Tehrani, V Ghasemi and MSG Naz. A systematic review and meta ‑ analysis of follicle ‑ stimulating hormone levels among men with type 2 diabetes. Basic and Clinical Andrology 2025; 35(1), 11.
[57] S Kumar, J Jain and A Jain. Biochemical correlation of sex hormone profile with diabetes mellitus type 2 in indian men- a case-control study. Medical and Health Science Journal 2023; 7(1), 9-20.
[58] M Gül, GI Russo, H Kandil, F Boitrelle, R Saleh, E Chung, P Kavoussi, T Mostafa, R Shah and A Agarwal. Male infertility: New developments, current challenges, and future directions. The World Journal of Men’s Health 2024; 42(3), 502-517.
[59] N Cortez, C Villegas, V Burgos, L Ortiz, JRC Pardo and C Paz. Therapeutic potential of chlorogenic acid in chemoresistance and chemoprotection in cancer treatment. International Journal of Molecular Sciences 2024; 25(10), 5189.
[60] O Golovinskaia and CK Wang. The hypoglycemic potential of phenolics from functional foods and their mechanisms. Food Science and Human Wellness 2023; 12(4), 986-1007.
[61] EN Sofyanita, A Suwondo, H Nugroho and AJ Juniarto. Global research trends on the role of chlorogenic acid on antidiabetic mechanisms and reproductive hormone regulation (2015-2025): A bibliometric analysis. Media Publikasi Promosi Kesehatan Indonesia 2025; 8(12), 1617-1632.
[62] F Dimitriadis, M Papaioannou, I Sokolakiset, F Aikaterini, H Dimitrios and A Apostolos. The effect of low-intensity extracorporeal shockwave treatment on the urinary bladder in an experimental diabetic rat model. International Neurourology Journal 2021; 25(1), 34-41.
[63] MB Ekong, FN Odinukaeze, AC Nwonu, CC Mbadugha and AA Nwakanma. Brain activities of streptozotocin-induced diabetic Wistar rats treated with gliclazide: Behavioural, biochemical and histomorphology studies. IBRO Neuroscience Reports 2022; 12, 271-279.