
Trends
Sci.
2026;
23(7):
12767
Valorization of Desalted Duck Egg White through Enzymatic Hydrolysis: Gastric Digestion Behavior and Antioxidant Responses
Sudarat Jiamyangyuen1, Naruemon Tosuk2, Sonthaya Numthuam3,
Chayanid Sringarm4, Thunnalin Winuprasith5 and Saowaluk Rungchang1,2,*
1Division of Food Science and Technology, Faculty of Agro-Industry, Chiang Mai University,
Chiang Mai 50100, Thailand
2Department of Agro-Industry, Faculty of Agriculture Natural Resources and Environment, Naresuan University, Phitsanulok 65000, Thailand
3Department of Agricultural Science, Faculty of Agriculture Natural Resources and Environment, Naresuan University, Phitsanulok 65000, Thailand
4Department of Agro-Industrial, Food, and Environmental Technology, Faculty of Applied Science,
King Mongkut’s University of Technology North Bangkok, Bangkok 10800, Thailand
5Institute of Nutrition, Mahidol University, Nakhon Pathom 73170, Thailand
(*Corresponding author’s e-mail: [email protected])
Received: 23 November 2025, Revised: 16 December 2025, Accepted: 26 December 2025, Published: 5 March 2026
Abstract
This study investigated the gastric-phase digestion behavior and antioxidant responses of enzymatic protein hydrolysates produced from desalted duck egg white (DS-DEW) and evaluated their potential for value-added food ingredient development. Duck egg white proteins were subjected to enzymatic hydrolysis, and a modified INFOGEST static in vitro digestion model focusing exclusively on the gastric phase was applied to native duck egg white (DEW), desalted duck egg white (DS-DEW), duck egg white hydrolysate (DEWH), desalted duck egg white hydrolysate (DS-DEWH), and commercial egg white powder (EWP). Proteolysis during digestion was assessed by acid consumption kinetics, while free amino acid (FAA) release and antioxidant responses were evaluated using HPLC, DPPH radical-scavenging, and ferric-reducing antioxidant power (FRAP) assays. Native and non-hydrolyzed samples (DEW, DS-DEW, and EWP) exhibited high acid uptake during gastric digestion (90% - 95%), whereas pre-hydrolyzed samples (DEWH and DS-DEWH) showed markedly lower acid consumption (< 20%), reflecting extensive peptide bond cleavage prior to gastric digestion rather than reduced digestibility. DS-DEWH exhibited the highest absolute FAA content after digestion (303.25 ± 4.38 mg/g), approximately 22-fold higher than EWP, indicating greater availability of hydrolysis products under acidic, pepsin-driven conditions. Antioxidant evaluation using chemical assays showed that DS-DEWH displayed significantly higher DPPH radical-scavenging activity (42.67%) and FRAP values (3.71 ± 0.69 µmol TE/g) than DEW and DS-DEW (p < 0.05). Due to analytical constraints, EWP was excluded from antioxidant assays. Overall, enzymatic hydrolysis altered gastric-phase digestion behavior and enhanced free amino acid availability and antioxidant responses of desalted duck egg white at the chemical-assay level, supporting its valorization as a sustainable food protein ingredient.
Keywords: Desalted duck egg white, Enzymatic hydrolysis, in vitro gastric digestion, Protein valorization, Antioxidant activity, Food ingredient development
Abbreviation
ANOVA Analysis
of variance;
DEW Duck egg white;
DEWH Duck egg white
hydrolysate;
DI Distilled water;
DMRT Duncan’s
multiple range
test;
DPPH 2,2-diphenyl-1-picrylhydrazyl;
DS-DEW Desalted
duck egg white;
DS-DEWH Desalted duck egg white
hydrolysate;
EWP Egg white powder;
FAA Free amino
acids;
FRAP Ferric reducing antioxidant
power;
GI Gastrointestinal;
GL30 Thermoase
GL30;
HCl Hydrochloric acid;
HPLC High-performance
liquid chromatography;
INFOGEST International Network on Food
Digestion;
NaCl Sodium chloride;
R² Coefficient of
determination;
SD Standard deviation;
SGF Simulated
gastric fluid;
SSF Simulated salivary
fluid;
TPTZ 2,4,6-tris(2-pyridyl)-s-triazine;
UV-Vis Ultraviolet-visible
spectrophotometry.
Introduction
Bioactive components derived from food proteins have attracted increasing attention in food science and nutrition due to their potential antioxidant, antihypertensive, and anti-inflammatory properties [1]. These components are commonly generated through enzymatic hydrolysis, which converts native proteins into smaller hydrolysis products with altered functional and digestive properties [2,3]. Their effectiveness depends not only on amino acid composition but also on stability during processing and gastrointestinal digestion. In particular, hydrolysis products enriched in hydrophobic, aromatic, or sulfur-containing amino acids - such as phenylalanine, tyrosine, tryptophan, and methionine - are frequently associated with enhanced antioxidant responses in chemical assays due to their radical-scavenging and metal-chelating abilities [4-7].
Among the various functional attributes of protein hydrolysates, antioxidant activity is especially important because it contributes to oxidative stability in food systems and may reduce oxidative stress. Antioxidant responses are influenced by factors such as the degree of hydrolysis, protease specificity, and the overall composition of hydrolysis products [8]. While extensive studies have been conducted on hydrolysates from plant and dairy proteins such as soy, rice, casein, and whey, investigations focusing on animal-derived hydrolysates particularly duck egg white remain limited.
Duck egg white (DEW) is a rich source of globular proteins, including ovalbumin, ovotransferrin, ovoglobulin, and lysozyme, which provide desirable functional properties such as foaming, emulsifying, and gelling [9,10]. Compared with chicken egg white, DEW contains higher total protein levels, a greater proportion of ovalbumin and ovotransferrin, and higher contents of aromatic and sulfur-containing amino acids that are commonly linked to antioxidant activity [12-14]. DEW also exhibits higher ovomucin content and a distinct albumin-to-globulin ratio, contributing to increased viscosity and gel strength [12,15]. These compositional characteristics suggest that DEW is a promising substrate for producing antioxidant-active protein hydrolysates.
Salted egg production, widely practiced in many Asian countries including Thailand, generates large quantities of salted egg white as a by-product. Traditional curing methods, such as brine immersion or clay-coating, induce extensive salt penetration and protein denaturation, rendering the by-product unsuitable for direct food applications and leading to disposal-related environmental and economic concerns. Desalting of this by-product yields desalted duck egg white (DS-DEW), which retains valuable proteins and represents a sustainable raw material for further processing [16,17]. However, prolonged exposure to high salt concentrations during curing can induce conformational changes that may reduce enzyme accessibility and influence subsequent digestion behavior [4,18]. Understanding how these salt-induced modifications interact with enzymatic hydrolysis is therefore critical for effective valorization of DS-DEW.
Although antioxidant-active hydrolysates from fresh egg white and other animal proteins have been reported [19,20] systematic investigations focusing on DS-DEW remain scarce. In particular, the combined effects of desalting and dual-enzyme hydrolysis on gastric-phase digestion behavior, free amino acid release, and antioxidant responses under standardized digestion models have not been clearly elucidated. Addressing this gap is essential for understanding how salt-induced structural modifications interact with enzymatic processing and pepsin-driven digestion, and for guiding the valorization of salted egg by-products.
Therefore, the aim of this study was to produce enzymatic protein hydrolysates from desalted duck egg white using a dual-enzyme system consisting of Protease M “Amano” SD and Thermoase GL30, and to evaluate their gastric-phase digestion behavior, free amino acid availability, and antioxidant responses under a modified INFOGEST model. Based on the composition of DEW and the anticipated synergistic effects of desalting and dual-enzyme hydrolysis, it was hypothesized that desalted duck egg white hydrolysate (DS-DEWH) would exhibit enhanced gastric-phase stability against further pepsin cleavage, greater free amino acid availability, and stronger antioxidant responses than non-hydrolyzed samples. This work provides a functional assessment of DS-DEWH as a value-added hydrolysate derived from a salted egg by-product, supporting its potential for sustainable food ingredient development.
Materials and methods
Chemicals and materials
Salted duck egg white was purchased from a local salted egg industry (Nakhon Pathom, Thailand) and stored at −20 °C until use. Fresh duck eggs were obtained from a convenience store (Phitsanulok, Thailand). Commercial egg white powder (EWP) was purchased from Krungthepchemi Co., Ltd. (Bangkok, Thailand). Protease M “Amano” SD (≥ 40,000 U/g) and Thermoase Gacid L30 (≥ 300,000 U/mL) were obtained from Amano Enzyme Asia Pacific Co., Ltd. (Pathum Thani, Thailand). DPPH (2,2-diphenyl-1-picrylhydrazyl, ≥ 95%), Trolox (≥ 97%), acetic acid, porcine mucin, and pepsin (Product code SA-P7000, ≥ 250 U/mg protein) were purchased from Sigma-Aldrich (St. Louis, MO, USA). All reagents and solvents were analytical grade (≥ 99.8%).
Sample preparation
Salted duck egg white was desalted by boiling in water at 80 - 100 °C for 3 - 5 cycles of 10 min each until the salt content was < 0.2% (w/w), as determined by the Mohr titration method [14]. The desalting procedure involved repeated boiling cycles (80 - 100 °C, 10 min per cycle), which may induce thermal denaturation of egg white proteins. However, heat treatment alone does not cleave peptide bonds but primarily alters protein conformation, facilitating salt diffusion and potentially increasing enzyme accessibility during subsequent hydrolysis. Similar thermal effects on egg white protein structure have been reported previously [21]. The desalted sample (DS-DEW) was cooled to room temperature, centrifuged at 10,000 rpm for 10 min at 4 °C, and the clear supernatant was collected. Fresh duck egg white (DEW) was separated manually and heated in boiling water for 10 min (80 - 100 °C) to obtain a thermally treated state comparable to DS-DEW. All prepared samples were stored at −20 °C until enzymatic hydrolysis.
Enzymatic hydrolysis preparation
DEW or DS-DEW was mixed with distilled water at a 1:0.5 ratio (w/v) and adjusted to pH 7.0 using 5% (v/v) acetic acid. Hydrolysis was carried out at 50 °C. A 1% (w/w, enzyme-to-substrate ratio) mixture of Protease M “Amano” SD and Thermoase GL30 (1:1, w/w) was added. The reaction proceeded for up to 6 h in a shaking water bath. Enzymatic activity was terminated by heating at 95 °C for 10 min, followed by centrifugation at 10,000 rpm for 10 min at 4 °C. The supernatant was collected and stored at −20 °C. Spray drying (Mini Spray Dryer S-300, BUCHI, Thailand) was performed at 170 °C inlet and 100 °C outlet with 650 L/h spray gas to obtain the hydrolysates DEWH and DS-DEWH. All hydrolysis experiments were performed in triplicate (n = 3).
In vitro gastrointestinal digestion of proteins
Five samples (DEW, DEWH, DS-DEW, DS-DEWH and EWP) were subjected to simulated oral and gastric digestion following the International Network on Food Digestion (INFOGEST) protocol with modifications [22,23]. The intestinal phase was intentionally omitted to isolate pepsin-driven gastric digestion. Samples were diluted to achieve a nitrogen concentration of 0.1% (w/v). Aliquots (5 mL) were transferred into 50 mL beakers. Distilled water served as the digestion blank.
Oral phase
Simulated salivary fluid (SSF) containing mucin (0.015 g/5 mL) was preheated to 37 °C. Each sample was mixed with SSF at a 1:1 (v/v) ratio, adjusted to pH 6.8, and incubated at 37 °C and 100 rpm for 2 min.
Gastric phases
Simulated gastric fluid (SGF) was prepared following INFOGEST ionic composition and contained NaCl and HCl to reach pH 3.0. Oral digesta (10 mL) were mixed with 10 mL SGF containing 64 mg pepsin and incubated at 37 °C for 180 min. A pH-stat titrator (Metrohm USA Inc.) maintained the pH at 3.0 using 0.1 M HCl. Digesta were cooled in ice, centrifuged at 5,500 rpm for 10 min, and the supernatants collected for free amino acid (FAA) and antioxidant analyses. Only DS-DEWH and EWP were subjected to FAA analysis due to sample volume limitations and to directly compare the unhydrolyzed powder with the optimized hydrolysate.
In this study, percentage of free amino acid analysis (%FAA) refers to the percentage of free amino acids released relative to the maximum free amino acid concentration measured during the digestion experiment for each sample. It does not represent the percentage of total protein or total nitrogen converted into free amino acids, but rather the relative extent of amino-acid liberation during gastric digestion. For %FAA, only DS-DEWH and EWP were analyzed to provide a direct comparison between the original sample and the final optimized hydrolysate. The other samples (DEW, DS-DEW, and DEWH) were excluded due to limited sample volumes and because the main objective was to evaluate the effect of desalting and enzyme combination (synergistic hydrolysis) rather than to compare all individual enzyme treatments.
Although the present work followed the standardized INFOGEST framework, the intestinal digestion phase was intentionally omitted to isolate and examine pepsin-driven hydrolysis of desalted duck egg white proteins. This decision reflects the study’s specific objectives of characterizing early gastric stability, cleavage behavior, and FAA release of DS-DEWH under acidic conditions (pH 3.0), prior to the more complex enzymatic environment of the small intestine. However, it must be acknowledged that this modification represents a methodological limitation. Pancreatic proteases (trypsin, chymotrypsin, elastase and carboxypeptidases) are responsible for the majority of peptide bond cleavage in the gastrointestinal tract, and many bioactive peptides are generated predominantly during intestinal digestion rather than the gastric phase. Excluding the intestinal phase may therefore underestimate total peptide liberation, alter FAA profiles, and limit interpretation of antioxidant activity, which often increases markedly following intestinal digestion. As such, the findings of this study should be interpreted as representative of gastric digestion only and not the complete gastrointestinal process.
Protein content
Total nitrogen content (%) was traced using a high-temperature combustion protein analyzer (Vario MAX CN; Elementar Analysensystememe GmbH, Langenselbold, Germany). A 6.25 multiplication was calculated to produce crude protein content from the total nitrogen content.
Free amino acids analysis
FAA analysis was performed according to Siddiqi et al. [24] with modifications. Samples from 0- and 180-minute gastric digestion were immediately cooled, filtered through 0.22 µm membranes, and analyzed using an HPLC system (Waters Alliance 2695, Milford, MA, USA) equipped with a Jasco FP2020 fluorescence detector. An AccQ-Tag Ultra C18 column (100×4.6 mm2, 2.5 µm; Osaka Soda, Japan) was used at 35 °C with a flow rate of 1.2 mL/min. The mobile phases were sodium acetate buffer (A) and acetonitrile (B). Injection volume was 5 µL. Each sample was analyzed in triplicate (n = 3).
Antioxidant activity
DPPH radical-scavenging activity was measured following Cui et al. [25]. Sample extract (100 µL) was mixed with 100 µL of 0.2 mM DPPH solution in 95% ethanol and incubated for 30 min. Absorbance at 517 nm was recorded in a microplate reader. Inhibition (%) was calculated as:
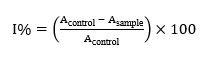

where, I%: The percentage of inhibition; Acontrol: Absorbance of DPPH solution without extract; Asample: Absorbance in the presence of extract or standard.
FRAP reagent was prepared from acetate buffer (300 mM, pH 3.6), TPTZ solution (10 mM), and FeCl₃·6H₂O (20 mM) at a 10:1:1 ratio. Sample (30 µL) was mixed with 3 mL FRAP reagent, incubated for 4 - 6 min at 37 °C (dark), and measured at 593 nm. Results were expressed as µmol Trolox equivalent (TE)/g dry sample. Due to high turbidity and insolubility in aqueous buffer systems, commercial egg white protein (EWP) could not be evaluated using DPPH or FRAP assays. Therefore, antioxidant activity was assessed only for DEW, DS-DEW, and DS-DEWH, and no direct comparisons with EWP were made.
Method validation
All assays were validated prior to data collection. Acceptance criteria included linearity (R² ≥ 0.995), repeatability (%RSD ≤ 5%), intermediate precision (%RSD ≤ 10%), spike-recovery 80% - 120%, and system suitability checks. Pepsin activity was verified using a casein substrate. pH electrodes were calibrated daily. Titrant (0.1 M HCl) was standardized against primary-standard Na₂CO₃. Digestion controls included a blank, an undigested matrix, and a commercial EWP positive control. FAA (HPLC) validation followed 7-point calibration curves, LOD/LOQ estimation (3.3σ/S; 10σ/S), injection precision, retention-time stability, and carryover checks. Antioxidant assays included Trolox calibration, reagent blanks, plate-to-plate QC, and sample clarification steps to minimize turbidity interference.
Statistical analysis
Data were analyzed using one-way ANOVA, and means were separated using Duncan’s multiple range test at p < 0.05. Paired t-tests were applied when comparing 2 related conditions. Statistical analysis was performed using SPSS version 16.
Results and discussion
Protein digestibility after in vitro gastrointestinal digestion
Gastric-phase protein digestibility was evaluated to determine how prior desalting and enzymatic hydrolysis influenced pepsin susceptibility under acidic conditions. In this study, digestibility was interpreted based on acid consumption kinetics monitored by the pH-stat system, which reflects the generation of newly exposed ionizable groups during peptide bond cleavage. This approach allows comparison of structural accessibility and cleavage behavior among native and pre-hydrolyzed protein systems during simulated gastric digestion. Because the intestinal phase was intentionally excluded, the results presented here specifically describe pepsin-driven hydrolysis and early-stage digestion behavior rather than complete gastrointestinal digestibility.
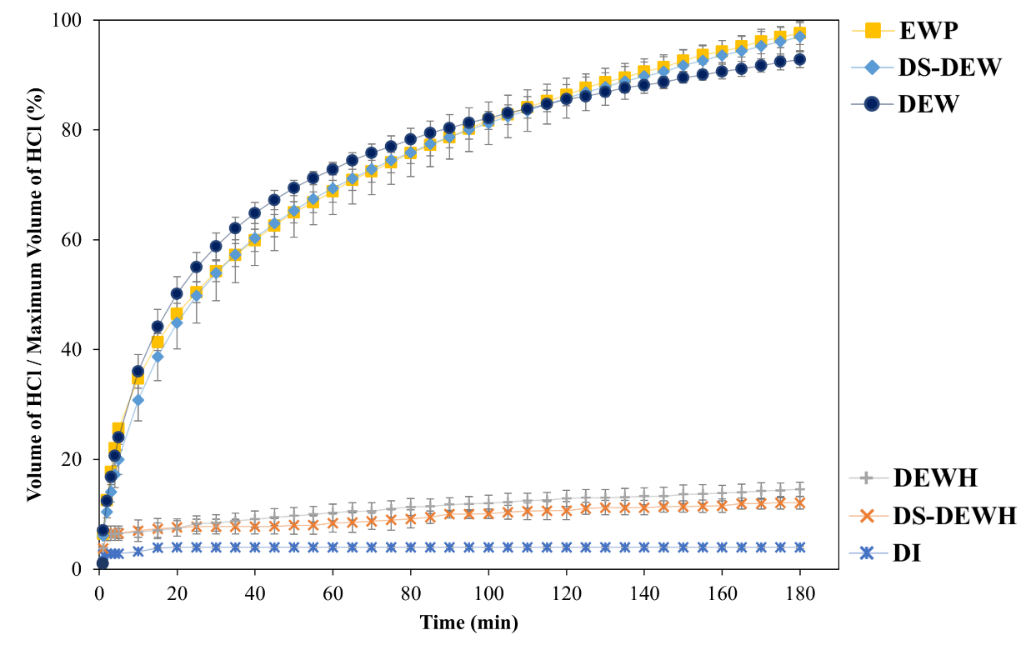
Figure 1 Acid consumption kinetics during protein hydrolysis during 180 min of gastric phase digestion of protein samples. DI water (Control), Duck egg white (DEW), Duck egg white hydrolysate (DEWH), Desalted-Duck egg white (DS-DEW), Desalted-Duck egg white hydrolysate (DS-DEWH), Egg white powder (EWP).
Protein hydrolysis behavior during gastric phase digestion
Figure 1 presents the acid consumption profiles obtained by pH-stat monitoring during 180 min of simulated gastric digestion. Acid consumption reflects the release of ionizable groups generated during pepsin-mediated peptide bond cleavage and is therefore used as an indirect indicator of gastric protein hydrolysis.
Native protein samples, including duck egg white (DEW), desalted duck egg white (DS-DEW), and commercial egg white powder (EWP), exhibited pronounced acid consumption, reaching approximately 90% - 100% of their maximum values after 180 min. All 3 samples showed a rapid initial increase within the first 20 - 30 min, followed by a gradual plateau phase, which is characteristic of pepsin-catalyzed digestion as accessible cleavage sites become progressively depleted. The comparable acid uptake profiles among DEW, DS-DEW, and EWP indicate similar susceptibility to pepsin digestion despite differences in processing history.
In contrast, enzymatically pre-hydrolyzed samples (DEWH and DS-DEWH) showed markedly lower acid consumption throughout the digestion period, remaining below 15% of the maximum HCl volume. This behavior indicates that a substantial proportion of peptide bonds had already been cleaved prior to gastric digestion, resulting in limited availability of further pepsin-cleavable sites. Low acid uptake in hydrolysates reflects fewer remaining peptide bonds, not reduced digestibility. Distilled water (DI) exhibited negligible acid consumption, confirming that the titration response was specific to protein-containing substrates.
Overall, these results demonstrate that prior enzymatic hydrolysis strongly alters gastric hydrolysis kinetics by reducing acid demand during pH stabilization. The observed differences reflect variations in protein structural state and peptide bond availability rather than overall nutritional digestibility. Because this model simulates only the gastric phase, the findings should be interpreted as comparative pepsin susceptibility rather than complete gastrointestinal protein digestibility.
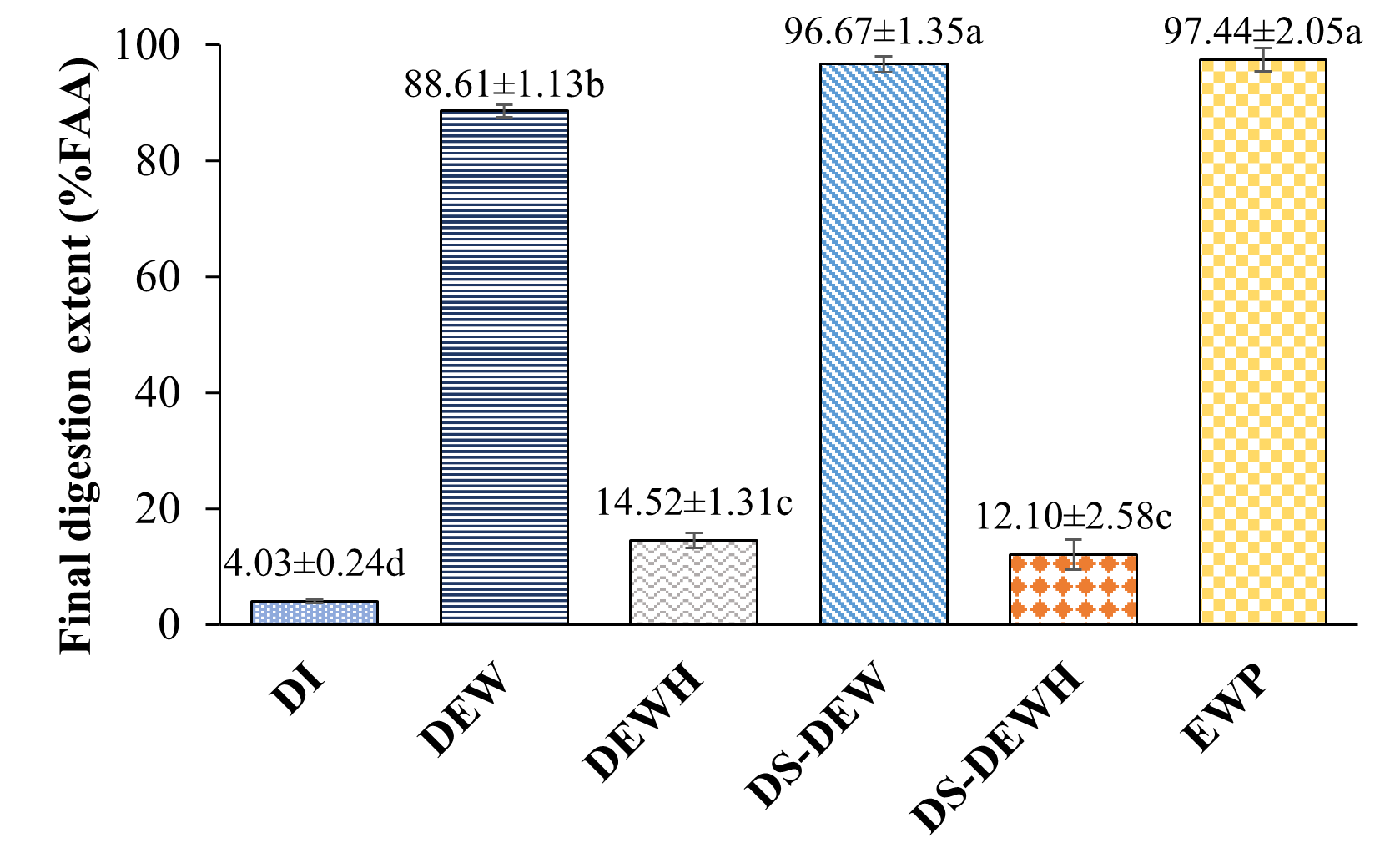
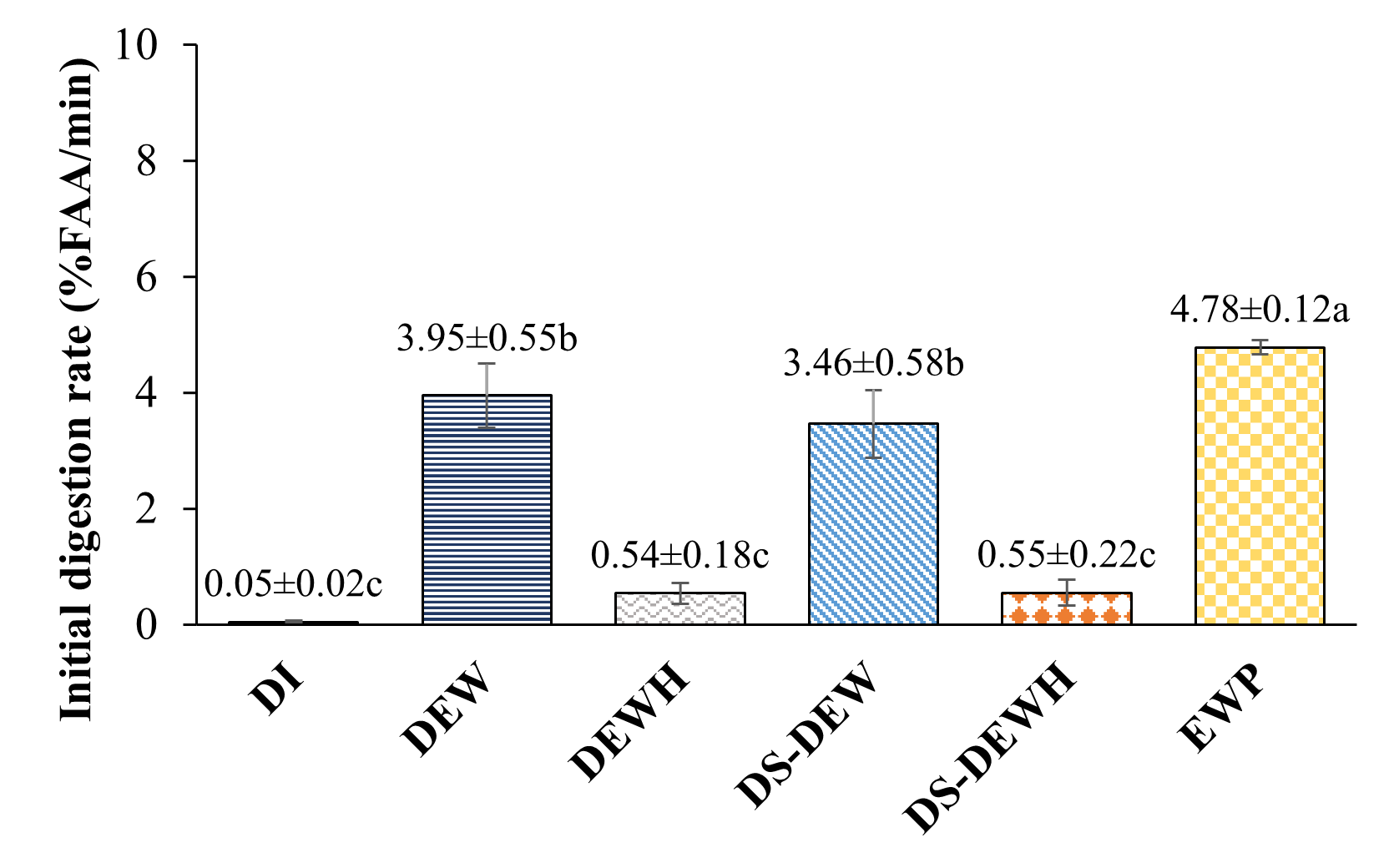
Figure 2 Initial at 0 - 5 min. digestion rate (Top) final at 175 - 180 min. digestion extent (Bottom) of protein samples. Different letters indicate significantly different (p < 0.05) by Duncan’s test (n = 3). DI water (Control), Duck egg white (DEW), Duck egg white hydrolysate (DEWH), Desalted-Duck egg white (DS-DEW), Desalted-Duck egg white hydrolysate (DS-DEWH), Egg white powder (EWP).
Free amino acid release kinetics during gastric digestion
Figure 2 presents the initial digestion rate (0 - 5 min) and final digestion extent (175 - 180 min) based on free amino acid (FAA) release during simulated gastric digestion. Significant differences among protein samples were observed for both parameters (p < 0.05). EWP exhibited the highest initial FAA release rate (4.78% FAA/min), followed by DEW (3.95% FAA/min) and DS-DEW (3.46% FAA/min), indicating rapid pepsin-mediated cleavage during the early stage of gastric digestion. In contrast, the enzymatically pre-hydrolyzed samples (DEWH and DS-DEWH) showed significantly lower initial digestion rates (approximately 0.54% - 0.55% FAA/min), while distilled water (DI) exhibited negligible FAA release.
A similar trend was observed for final digestion extent. EWP and DS-DEW reached the highest final FAA values after 180 min (97.44% and 96.67%, respectively), followed by DEW (88.61%). In contrast, DEWH and DS-DEWH exhibited significantly lower final FAA release (14.52% and 12.10%, respectively).
In this study, %FAA represents the relative increase in free amino acids released during gastric digestion, normalized to the maximum FAA concentration measured for each sample, and does not indicate the fraction of total protein hydrolyzed. Accordingly, the low %FAA values observed for the pre-hydrolyzed samples (DEWH and DS-DEWH) do not reflect poor protein digestibility, but rather indicate limited additional FAA generation during the 0 - 180 min gastric phase because these samples already contained substantial levels of free amino acids and short peptides prior to digestion. This interpretation is consistent with the acid consumption profiles shown in Figure 1, where low acid demand reflects a reduced availability of remaining pepsin-cleavable peptide bonds following enzymatic pre-hydrolysis. Thus, acid consumption and %FAA describe complementary aspects of gastric proteolysis, and the combination of low acid uptake with low incremental %FAA increases reflects extensive prior hydrolysis rather than impaired nutritional digestibility, while still being associated with higher absolute FAA contents.
Differences between native and desalted egg white proteins may be partially attributed to salt-induced structural modifications occurring during curing. High salt exposure can promote partial protein unfolding and aggregation, which may influence enzyme accessibility during subsequent digestion. Although desalting removes most salt ions, residual conformational changes may persist. However, these interpretations are based on indirect functional evidence, as direct structural analyses were not performed in the present study.
Overall, Figure 2 demonstrates that enzymatic pre-hydrolysis markedly alters gastric digestion behavior by limiting further conversion of peptides into free amino acids. These results highlight that FAA release during gastric digestion reflects gastric stability of protein hydrolysates during digestion and pepsin susceptibility rather than overall nutritional digestibility.
Free amino acids profile (FAAs profile)
Analysis of individual free amino acid (FAA) profiles was conducted to provide a more detailed characterization of digestion products beyond relative %FAA values. While %FAA reflects the incremental release of amino acids during gastric digestion, absolute FAA composition (mg/g dry sample) provides insight into the qualitative and quantitative distribution of amino acids present before and after digestion. This distinction is particularly important for pre-hydrolyzed systems, where substantial amounts of free amino acids may already exist prior to gastric exposure. Therefore, profiling individual FAAs enables clearer interpretation of how desalting and enzymatic treatment influenced amino acid availability and functional properties.
Table 1 Individual free amino acid content before and after in vitro gastrointestinal digestion of EWP and DS-DEWH.
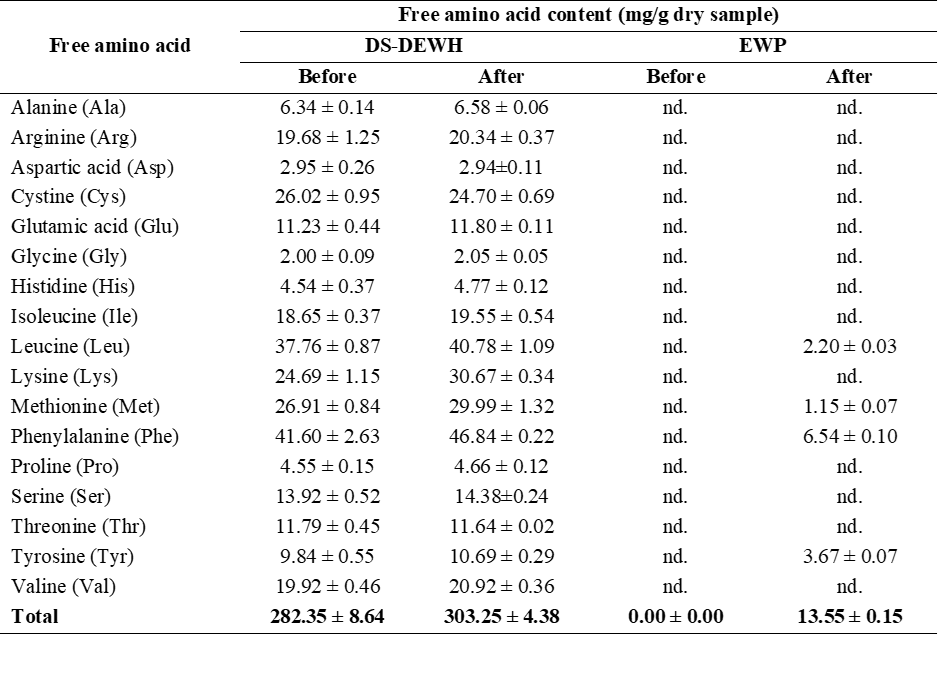
nd = not detected
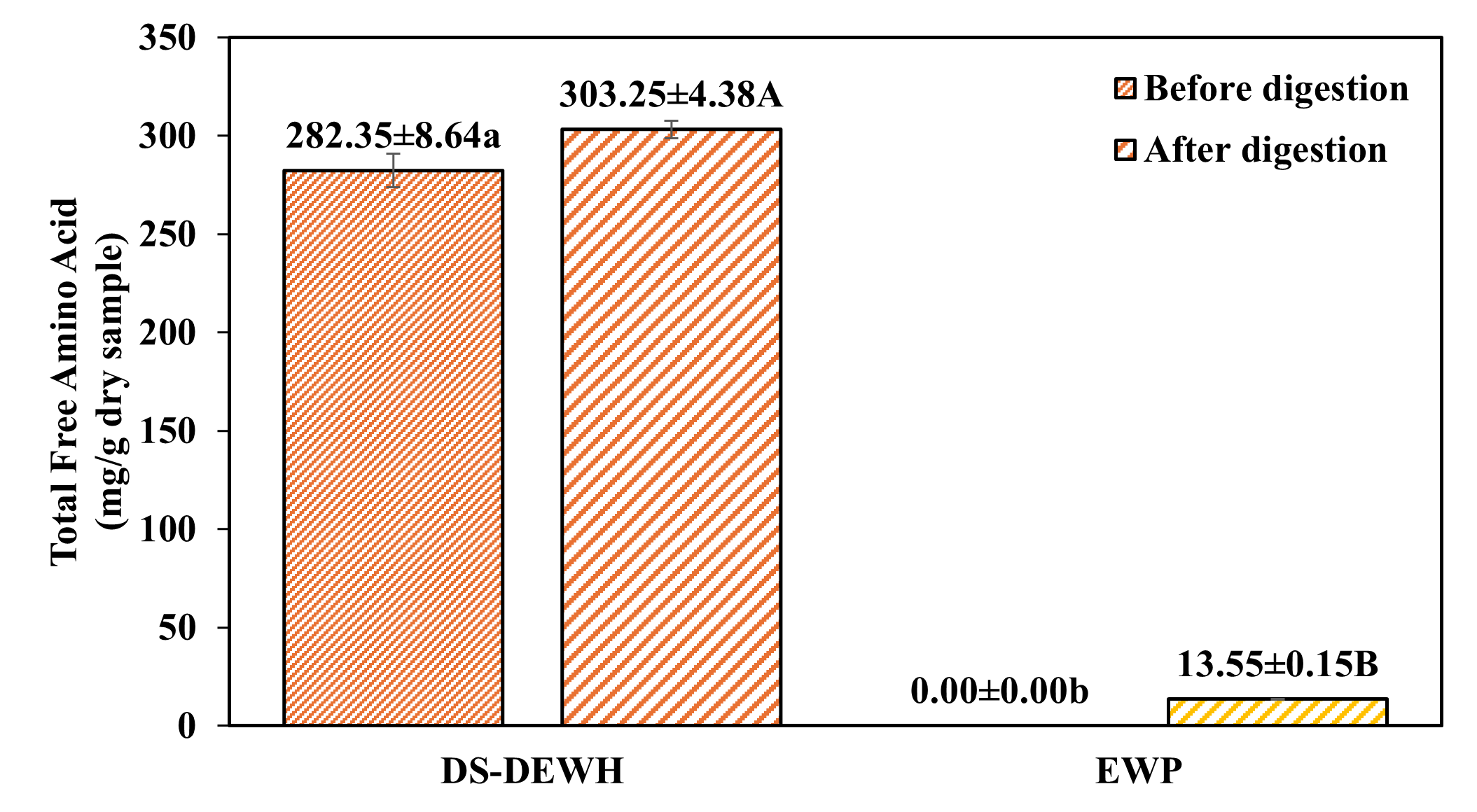
Figure 3 Total free amino acid content before and after in vitro gastrointestinal digestion of EWP and DS-DEWH.
Different letters indicate statistically significant differences (p < 0.05). Lowercase letters (a, b, c) compare samples within the “before digestion” group, and uppercase letters (A, B, C) compare samples within the “after digestion” group. Desalted duck egg white hydrolysate (DS-DEWH), Egg white powder (EWP).
Free amino acid profiles after gastric digestion
To further evaluate the effects of desalting and enzymatic hydrolysis on protein breakdown, individual and total free amino acids (FAAs) released from commercial egg white powder (EWP) and desalted duck egg white hydrolysate, (DS-DEWH) were analyzed before and after in vitro gastric digestion. Individual FAA profiles are presented in Table 1, while total FAA contents are summarized in Figure 3.
As shown in Table 1, DS-DEWH exhibited a broad spectrum of free amino acids both before and after gastric digestion, with individual FAA contents ranging from approximately 2 to 47 mg/g dry sample. In contrast, most free amino acids were not detected in EWP before digestion and were present only at low levels after digestion. The most abundant amino acids released from DS-DEWH included phenylalanine, leucine, methionine, cystine, and lysine, which are commonly associated with egg white protein composition and pepsin cleavage preference. Similar dominant amino acids have been reported previously for duck egg white proteins [26], supporting the consistency of the observed profiles.
Figure 3 illustrates the total FAA contents before and after gastric digestion. DS-DEWH showed significantly higher total FAA levels than EWP under both conditions (p < 0.05). Total FAA content in DS-DEWH increased from 282.35 ± 8.64 to 303.25 ± 4.38 mg/g after digestion, indicating further peptide breakdown during the gastric phase. In contrast, EWP showed no detectable FAAs before digestion and only a limited increase after digestion (13.55 ± 0.15 mg/g). Although EWP exhibited a high %FAA due to a large relative increase from an almost zero baseline, its absolute FAA concentration after digestion (mg/g) remained low. In contrast, DS-DEWH contained substantial baseline FAA prior to digestion, resulting in a lower relative %FAA increase but markedly higher absolute FAA levels.
The higher FAA release from DS-DEWH reflects the combined effects of desalting and enzymatic pre-hydrolysis. In comparison, the compact globular structure of EWP limited the
extent of FAA release during gastric digestion. These findings are consistent with the acid consumption and FAA kinetics observed in Figures 1 and 2, which demonstrated reduced pepsin-cleavable sites in pre-hydrolyzed samples but greater overall availability of short peptides and free amino acids. Consistent with the digestion indicators discussed above, the low incremental %FAA observed for pre-hydrolyzed samples reflects limited remaining pepsin-cleavable peptide bonds rather than impaired digestibility. Despite this low relative increase, DS-DEWH maintained substantially higher absolute FAA contents, highlighting differences in digestion product profiles rather than overall protein utilization.
Because the present study evaluated digestion using the gastric phase only, the total free amino acid (FAA) profiles reported here reflect early peptide breakdown driven by pepsin activity and do not account for additional peptide and amino acid release that would occur during intestinal digestion. As pancreatic enzymes play a major role in generating low-molecular-weight peptides and free amino acids, the absence of the intestinal phase may lead to an underestimation of total FAA production and an incomplete assessment of digestibility-related functional outcomes. Therefore, incorporation of the full INFOGEST protocol in future studies is necessary to determine how DS-DEWH peptide profiles evolve under intestinal conditions and to better predict potential bioavailability and physiological relevance.
DPPH radical scavenging activity and FRAP
The antioxidant activities of DEW, DS-DEW, and DS-DEWH were evaluated using DPPH radical-scavenging and FRAP assays (Figure 4). Commercial EWP was excluded from antioxidant analysis due to severe turbidity that caused unstable absorbance measurements, even after centrifugation and filtration [27]. Therefore, all interpretations in this section refer exclusively to DEW, DS-DEW, and DS-DEWH. Before digestion, DS-DEWH exhibited higher DPPH radical-scavenging activity (33.64%) than DS-DEW (3.16%) and DEW (6.21%), indicating that enzymatic hydrolysis enhanced the antioxidant response measured by the DPPH assay. After gastric digestion, all samples showed significant increases in DPPH inhibition (p < 0.05), with DS-DEWH reaching the highest activity (42.67%), which was significantly greater than those of DS-DEW and DEW. Paired comparisons confirmed significant before-and-after increases within each sample, with the largest improvement observed for DS-DEWH.
FRAP results showed a similar overall trend. After digestion, DS-DEWH exhibited the highest ferric-reducing capacity (3.71 ± 0.69 µmol TE/g), however, this change was not statistically significant compared to its pre-digestion value (4.24 ± 0.24 µmol TE/g). In contrast, both DS-DEW showed significant increases in FRAP values following digestion (p < 0.01). Post-digestion FRAP values of DS-DEWH were significantly higher than those of DS-DEW and DEW, indicating a greater reducing capacity measured by the FRAP assay. Similar enhancements in reducing power after enzymatic hydrolysis have been reported for various protein systems [28].
Across samples, higher antioxidant activities were associated with higher total free amino acid (FAA) contents (Figure 3). DS-DEWH, which contained the highest FAA concentration, also exhibited the greatest DPPH and FRAP responses. This finding trend is consistent with previous studies which reported association of higher aromatic and sulfur-containing amino acid contents with stronger antioxidant outcomes in chemical assays [29-31]. Notably, DEW showed relatively higher DPPH activity but lower FRAP values after digestion, indicating assay-dependent differences in antioxidant behavior. Such discrepancies between radical-scavenging and reducing-power assays have been widely reported and reflect differences in the reactive groups involved in each method [9,27].
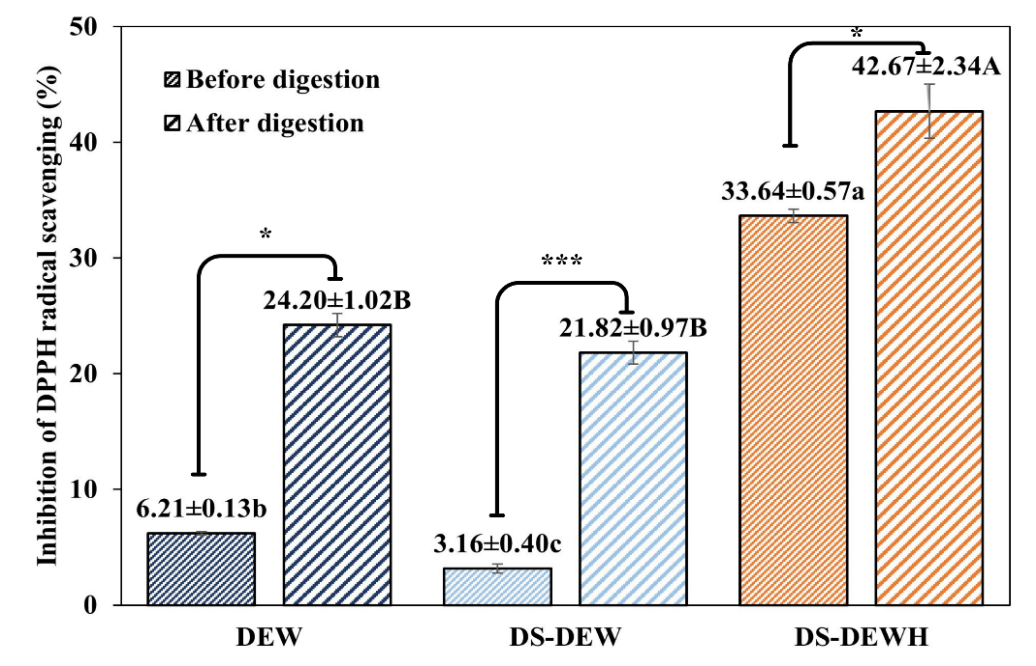
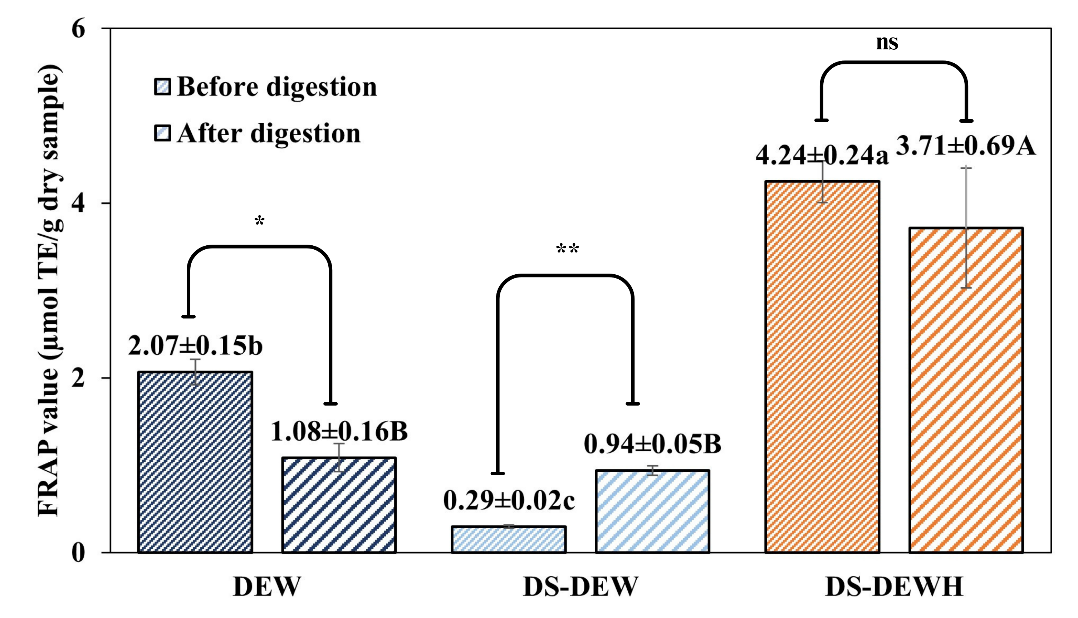
Figure 4 Antioxidant activity of protein samples before and after in vitro gastrointestinal digestion. (Top) DPPH radical scavenging activity and (Bottom) ferric reducing antioxidant power (FRAP). Values are presented as mean ± SD (n = 3). Different letters indicate statistically significant differences among samples (p < 0.05) by DMRT. Lowercase letters (a, b, c) compare values within the “before digestion” group, and uppercase letters (A, B, C) compare values within the “after digestion” group. Asterisks indicate significance levels from paired comparisons before and after digestion of same sample: p < 0.05 (*), p < 0.01 (**), p < 0.001 (***), “ns” = not significant. Duck egg white (DEW), Desalted duck egg white (DS-DEW), Desalted duck egg white hydrolysate (DS-DEWH).
The present findings are consistent with previous reports demonstrating that controlled thermal and enzymatic processing can enhance the antioxidant responses of egg-derived proteins. Wu et al. [32] reported a marked increase in DPPH radical-scavenging activity in preserved egg digests, while Chen et al. [20] showed that sequential thermal and enzymatic treatments of egg white generated peptides with improved antioxidant properties. These studies, together with the present results, suggest that processing-induced structural modification and enzyme specificity play important roles in promoting formation of peptide-rich hydrolysate fractions associated with higher antioxidant responses in chemical assays. In particular, amino acid composition especially the presence of aromatic and sulfur-containing residues - has been widely recognized as a key factor governing antioxidant responses in protein hydrolysates [33].
In this study, enzymatic hydrolysis of desalted duck egg white resulted in increased free amino acid release and enhanced antioxidant responses measured by DPPH and FRAP assays. These observations support the potential of processing-assisted hydrolysis to improve functional properties at the chemical assay level. However, the biological relevance of these antioxidant responses has not been evaluated, and further validation using cell-based or in vivo models is required to confirm physiological significance. It is also recommended that further research on peptide molecular weight distribution (e.g., by size exclusion chromatography-HPLC or gel electrophoresis) or peptide sequencing (LC-MS/MS) should be performed as peptide MW distribution was not measured in this study, and therefore functional implications are inferred based on chemical antioxidant rather than interpretations related to specific peptide structures or structure-function relationships.
This study employed a modified INFOGEST protocol focusing exclusively on the gastric phase to isolate pepsin-driven proteolysis and early-stage peptide stability. Consequently, the digestion behavior and antioxidant responses reported here reflect gastric-phase outcomes only and do not represent complete gastrointestinal digestion. Because peptide generation and functional transformations are substantially influenced by pancreatic enzymes during the intestinal phase, the present findings should be interpreted as early digestion behavior. Future studies incorporating the full gastric-intestinal INFOGEST model are required to evaluate intestinal peptide generation and to better assess physiological relevance.
Conclusions
This study demonstrated that DS-DEW white can be effectively converted into an antioxidant-active protein hydrolysate, based on chemical assays, through dual-enzyme treatment using Protease M “Amano” SD and Thermoase GL30. Enzymatic hydrolysis substantially altered the gastric digestion behavior of DS-DEW, as evidenced by reduced acid consumption during simulated gastric digestion and distinct functional responses at the chemical-assay level. Compared with non-hydrolyzed samples, DS-DEWH exhibited markedly higher free amino acid contents before and after gastric digestion, indicating enhanced availability of low-molecular-weight hydrolysis products and free amino acids under acidic, pepsin-driven conditions. Antioxidant evaluation using DPPH and FRAP assays further showed that DS-DEWH consistently displayed stronger antioxidant responses than DEW and DS-DEW. These improvements were closely associated with increased FAA levels and the presence of amino acids known to contribute to antioxidant activity. Antioxidant comparisons were limited to DEW, DS-DEW, and DS-DEWH due to analytical constraints.
Within the defined experimental scope, the present results demonstrate that the combined desalting and dual-enzyme hydrolysis strategy modified the gastric-phase behavior of duck egg white proteins, resulting in altered pepsin susceptibility, higher free amino acid availability, and enhanced antioxidant responses measured by chemical assays. These findings support the study hypothesis and highlight the potential of DS-DEWH as a value-added protein hydrolysate derived from salted egg by-products. The present work therefore provides an initial functional characterization at the gastric level, while further studies involving full gastrointestinal digestion, detailed peptide characterization, and biological validation are necessary to establish intestinal stability and physiological relevance.
Acknowledgements
This work was supported by Naresuan University (NU), and National Science, Research and Innovation Fund (NSRF), Grant number R2568B049. We thank Amano Enzymes Asia Pacific Co., Ltd., Thailand for providing the enzyme for this study.
Declaration of generative AI in scientific writing
Generative AI tools were used only to improve the readability and language clarity of the manuscript. No content generation or data interpretation was performed by AI. The authors take full responsibility for the content and conclusions of this work.
CRediT Author Statement
Sudarat Jiamyangyuen: Conceptualization, methodology, validation, formal analysis, resources, writing - original draft preparation, visualization, supervision, project administration; Naruemon Tosuk: Conceptualization, methodology, validation, formal analysis, resources, writing - original draft preparation, writing - review and editing; Sonthaya Numthuam: Methodology, validation, formal analysis, resources, writing - original draft preparation, visualization, writing - review and editing; Chayanid Sringarm: Methodology, validation, formal analysis, resources, writing - review and editing, visualization; Thunnalin Winuprasith: Methodology, validation, formal analysis, resources, data curation, writing - original draft preparation, writing - review and editing, supervision; Saowaluk Rungchang: Conceptualization, methodology, validation, formal analysis, resources, data curation, writing - review and editing, supervision, project administration. All authors have read and agreed to the published version of the manuscript.
References
[1] Y Jiang, C Brennan, J Sun, X Zeng, J Chandrapala, B Sun and M Majzoobi. Recent progress of food-derived bioactive peptides: Extraction, purification, function, and encapsulation. Food Frontiers 2024; 5(3), 1240-1264.
[2] ECY Li-Chan. Bioactive peptides and protein hydrolysates: Research trends and challenges for application as nutraceuticals and functional food ingredients. Current Opinion in Food Science 2015; 1(1), 28-37.
[3] Y Sheng, WY Wang, MF Wu, YM Wang, WY Zhu, CF Chi and B Wang. Eighteen novel bioactive peptides from monkfish (Lophius litulon) swim bladders: Production, identification, antioxidant activity, and stability. Marine Drugs 2023; 21(3), 169.
[4] YM Wang, MX Ge, SZ Ran, X Pan, CF Chi and B Wang. Antioxidant peptides from miiuy croaker swim bladders: ameliorating effect and mechanism in NAFLD cell model through regulation of hypolipidemic and antioxidant capacity. Marine Drugs 2025; 23(2), 63.
[5] CC Udenigwe and RE Aluko. Food protein-derived bioactive peptides: Production, processing, and potential health benefits. Journal of Food Science 2012; 77(1), 11-24.
[6] ZF Bhat, S Kumar and HF Bhat. Bioactive peptides of animal origin: A review. Journal of Food Science and Technology 2015; 52(9), 5377-5392.
[7] Y Jiang, J Sun, J Chandrapala, M Majzoobi, C Brennan, XA Zeng and B Sun. Recent progress of food-derived bioactive peptides: Extraction, purification, function, and encapsulation. Food Frontiers 2024; 5(3),1240-1264
[8] M Pan, K Liu, J Yang, S Liu and S Wang. Advances on food-derived peptidic antioxidants - a review. Antioxidants 2020; 9(9), 799.
[9] RJ Elias, SS Kellerby and EA Decker. Antioxidant activity of proteins and peptides. Critical Reviews in Food Science and Nutrition 2008; 48(5), 430-441.
[10] TB Zou, TP He, HB Li, HW Tang and EQ Xia. The structure-activity relationship of the antioxidant peptides from natural proteins. Molecules 2016; 21(1), 21010072.
[11] N Yang and RS Jiang. Recent advances in duck egg research. Asian-Australasian Journal of Animal Sciences 2014; 27(3), 355-362.
[12] C Sun, Z Liu and N Yang. Proteomic comparison of duck and chicken egg whites reveals species-specific protein patterns. Journal of Agricultural and Food Chemistry 2018; 66, 721-731.
[13] ED Abeyrathne, HY Lee and DU Ahn. Egg white proteins and their potential use in food processing or as nutraceutical and pharmaceutical agents - a review. Poultry Science 2014; 92(12), 3292-3299.
[14] R Yu, Y Chi, Y Ma, Y Chi and L Wang. Differences in protein composition and functional properties of egg whites from 4 chicken varieties. Food Bioscience 2022; 46, 101614.
[15] Y Liu, M Huang, X Liu and M Hu. Structural characterization and functional properties of egg white protein treated by electron beam irradiation. Innovative Food Science and Emerging Technologies 2023; 84, 103262.
[16] Y Ren, H Wu, X Li, F Lai and X Xiao. Purification and characterization of high antioxidant peptides from duck egg white protein hydrolysates. Biochemical and Biophysical Research Communications 2014; 452(4), 888-894.
[17] YF Liu, I Oey, P Bremer, A Carne and P Silcock. Bioactive peptides derived from egg proteins: A review. Critical Reviews in Food Science and Nutrition 2018; 58(15), 2508-2530.
[18] AH Manninen. Protein hydrolysates in sports nutrition. Nutrition and Metabolism 2009; 6(1), 38.
[19] Q Liu, B Kong, YL Xiong and X Xia. Antioxidant activity and functional properties of porcine plasma protein hydrolysate as influenced by the degree of hydrolysis. Food Chemistry 2010; 118(2), 403-410.
[20] Y Chen, P Han, B Ma, X Wang, M Ma, N Qiu and X Fu. Effect of thermal treatment on the antioxidant activity of egg white hydrolysate and the preparation of novel antioxidant peptides. International Journal of Food Science and Technology 2022; 57(5), 2590-2599.
[21] Y Mine. Recent advances in the understanding of egg white protein functionality. Trends in Food Science & Technology 1995; 6, 225-232.
[22] GWJ Latimer. Official methods of analysis of AOAC International. Oxford University Press, Oxford, England, 2023.
[23] A Brodkorb, L Egger, M Alminger, P Alvito, R Assunção, S Ballance, T Bohn, C Bourlieu-Lacanal, R Boutrou, F Carrière, A Clemente, M Corredig, D Dupont, C Dufour, C Edwards, M Golding, S Karakaya, B Kirkhus, S Le Feunteun, …, I Recio. INFOGEST static in vitro simulation of gastrointestinal food digestion. Nature Protocols 2019; 14(4), 991-1014.
[24] RA Siddiqi, TP Sinngh, M Rani, DS Sogi and MA Bhat. Diversity in grain, flour, amino acid composition, protein profiling, and proportion of total flour proteins of different wheat cultivars of North India. Frontiers in Nutrition 2020; 7, 00141.
[25] Q Cui, Y Sun, J Cheng and M Guo. Effect of 2-step enzymatic hydrolysis on the antioxidant properties and proteomics of hydrolysates of milk protein concentrate. Food Chemistry 2022; 366, 130711.
[26] Q Shi, M Wei, H Chen, J Gao and P Tong. Desalination of duck egg white by biocoagulation to obtain peptide-ferrous chelate as iron delivery system: Preparation, characterization, and Fe2+ release evaluation in vitro. Journal of Food Science 2021; 86(10), 4678-4690.
[27] A Dávalos, M Miguel, B Bartolomé and R López-Fandiño. Antioxidant activity of peptides derived from egg white proteins by enzymatic hydrolysis. Journal of Food Protection 2004; 67(9), 1939-1944.
[28] ID Nwachukwu and RE Aluko. Structural and functional properties of food protein-derived antioxidant peptides. Journal of Food Biochemistry 2019; 43(1), e12761.
[29] MF Sbroggio, MS Montilha, V Ribeiro, G De Figueiredo, SR Georgetti and LE Kurozawa. Influence of the degree of hydrolysis and type of enzyme on antioxidant activity of okara protein hydrolysates. Food Science and Technology 2016; 36(2), 375-381.
[30] A Dryáková, A Pihlanto, P Marnila, L Čurda and HJ Korhonen. Antioxidant properties of whey protein hydrolysates as measured by 3 methods. European Food Research and Technology 2010; 230(6), 865-874.
[31] H Zhao, W Jiao, K Cui, X Fan, C Shu, W Zhang, J Cao and W Jiang. Near-freezing temperature storage enhances chilling tolerance in nectarine fruit through its regulation of soluble sugars and energy metabolism. Food Chemistry 2019; 289, 426-435.
[32] Y Wu, X Li, M Ma, G Hu, X Fu and J Liu. Characterization of the dynamic gastrointestinal digests of the preserved eggs and their effect and mechanism on HepG2 cells. Foods 2023; 12(4), 800.
[33] F Shahidi and Y Zhong. Measurement of antioxidant activity. Journal of Functional Foods 2015; 18, 757-781.