
Trends
Sci.
2026;
23(7):
12656
Potential of Zingiber officinale var. Rubrum Extract in Inhibiting Helicobacter pylori in Chronic Gastritis Patients: A Pilot Study in Human Focusing on Inflammation, Oxidative Stress, and Quality of Life
Coana Sukmagautama1,2,*, Reviono3, Wachid Putranto4, Eti Poncorini Pamungkasari5,
Hartono5, Risya Cilmiaty5 and Paramasari Dirgahayu6
1Doctoral Program of Medical Sciences, Universitas Sebelas Maret, Surakarta, Indonesia
2Gastroenterology and Hepatology Division, Department of Internal Medicine, Faculty of Medicine,
Universitas Sebelas, Surakarta, Indonesia
3Department of Pulmonology Medicine, Faculty of Medicine, Universitas Sebelas Surakarta, Indonesia
4Department of Internal Medicine, Faculty of Medicine, Universitas Sebelas, Surakarta, Indonesia
5Department of Public Health, Faculty of Medicine, Universitas Sebelas Maret, Surakarta, Indonesia
6Department of Parasitology, Faculty of Medicine, Universitas Sebelas Maret, Surakarta, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 14 November 2025, Revised: 22 December 2025, Accepted: 5 January 2026, Published: 10 March 2026
Abstract
Helicobacter pylori infection is a major global public health problem. Although H. pylori eradication has been shown to reduce the incidence of gastric cancer, increasing resistance to standard antibiotic therapy decreases treatment success rates. Zingiber officinale var. Rubrum contains active compounds with anti-inflammatory and antioxidant thus potential as an adjuvant for inhibiting H. pylori. This study aimed to demonstrate and verify the effect of ZOR extract therapy on the inhibiting of H. pylori in patients with chronic gastritis. Study parameters included TNF-α, IL-6, IL-1β, MDA, and quality of life based on the SF-NDI. A randomized controlled trial with a pre-post test was conducted from July 2024 to June 2025 at Sebelas Maret University Hospital, Sukoharjo. Thirty patients with chronic ulcers due to H. pylori infection were divided equally into 3 groups: A positive control standard eradication therapy, treatment 1 with the addition of 500 mg ZOR capsules, and treatment 2 with the addition of 1 g ZOR capsules. The intervention lasted for 2 weeks. Parameters evaluated before and after the intervention included serum levels of TNF-α, IL-6, IL-1β, MDA, and quality of life using the SF-NDI. Statistical analysis used ANOVA/Kruskal Wallis test with a significance level of p < 0.05. ZOR supplementation at doses of 500 mg and 1 g reduced TNF-α levels. Administration of 1 g ZOR also reduced IL-6 and IL-1β levels between groups. The reduction in MDA was not significant. Quality of life scores were found primarily in the 500 mg and 1 g ZOR groups, with significant results p < 0.05. Zingiber officinale var. Rubrum supplementation can be applied as an adjuvant in H. pylori eradication therapy, potentially providing anti-inflammatory and antioxidant effects thus improving quality of life in patients with chronic gastric ulcers caused by H. pylori infection.
Keywords: Red Ginger, Zingiber officinale, H. pyori, Gastritis, Chronic
Introduction
Helicobacter pylori (H. pylori) infection remains a public health problem worldwide. A global systematic review conducted by Hooi et al. [1] showed that in 2015, approximately 4.4 billion people worldwide were estimated to be positive for H. pylori infection. Although the total prevalence of H. pylori in Indonesia is low, the distribution of H. pylori sufferers is unbalanced between Eastern Indonesia and Western Indonesia 59.4% vs. 40.6% per year 2014 - 2017 with the highest contributor being East Nusa Tenggara 51.4% [2]. The incidence of H. pylori in Indonesia across all age groups was evaluated in 2022, showing that in the 3 - 11 year old age group, H. pylori patients were 16.7%. An increase in the incidence of H. pylori was also reported in the Jakarta area from 2010 to 2021, with the number of H. pylori patients reaching 19.74% [3].
Helicobacter pylori infection is associated with gastritis, peptic ulcers, gastric ulcers, and gastric carcinoma [4]. Changing resistance patterns, with therapeutic implications, and new knowledge regarding indications for pathogen eradication, make medical management of H. pylori a dynamic process that requires regular reassessment [5]. The World Health Organization reported in 2017 that H. pylori is among the 12 most antibiotic-resistant bacterial families. This increase in antibiotic resistance has led to an increased incidence of gastric cancer. Initially, treatment for this bacterial infection showed a success rate of > 90%. However, after 20 years of treatment, antibiotic resistance emerged, including to clarithromycin and metronidazole, the mainstays of H. pylori treatment [6].
Zingiber officinale var. Rubrum (ZOR) is considered to have roles as a gastrointestinal protectant, antimicrobial, antioxidant, anti-inflammatory, anti-cancer, and immunomodulator through active substances including 6-shogaol, 6-gingerol, gingererone, ginger oleoresin, and propanoids [7]. Anti-inflammatory effects through several cellular pathways such as inhibition of 5’-adenosine monophosphate-activated protein kinase, nuclear factor-kappa betha (NF-κB), and activation of the activator protein-1 pathway leading to inhibition of proinflammatory cytokines such as interluekin (IL)-1β, IL-6, IL-8, IL-12, interferon, and tumour necrosis factor-alpha (TNF-α). Furthermore, red ginger is able to inhibit the release of nitric oxide (NO), cyclooxygenase-2 (COX-2) activity, and inducible nitric oxide synthase (iNOS) which is characterized by improving oxidative stress conditions or reducing malondialdehyde MDA [8]. Previous experimental research with a gastric ulcer model in rats showed that Zingiber officinale has an anti-ulcerative effect that improves the clinical condition of patients [9]. Therefore, this study aims to analyze the adjuvant potential of Zingiber officinale var. Rubrum in chronic gastritis patients with H. pylori infection through evaluation parameters of TNF-α, IL-6, IL-1β, MDA, and quality of life based on the Short-Form Nepean Dyspepsia Index (SF-NDI).
Materials and methods
This study was an experimental pre-post test laboratory. Laboratory sampling was carried out at Sebelas Maret University Hospital, Sukorharjo from July to December 2024. This research has obtained ethical approval from the Research Ethics Committee, Faculty of Medicine, Sebelas Maret University (protocol ID 177/01/08/2024, No. 205/UN27.06.11/KEP/EC/2024).
Subject
A total of 30 patients were selected by purposive sampling, aged 18 - 70 years with a diagnosis of mild and moderate chronic gastritis H. pylori +. This study did not involve pregnant/lactating individuals, allergies to standard H. pylori management, and patients who did not have post-treatment control were excluded from the study subjects. Diagnosis of H. pylori is confirmed by histopathology evaluation by pathologist and gastroscopy by internist. The number of samples is determined based on Cohen's f formula for ≥ 3 groups, with the formula λ = f2×N where λ is the non-centrality parameter, N is the total sample, and f is Cohen’s f (effect size). Taking 10 samples per group was carried out with the assumption of a large effect value of f = 0.40 (α = 0.05 and the number of groups was 3) so an N value of 30 was obtained. Thus, a value of λ = 4.8 was obtained so using 30 subjects in total or 10 subjects per group was considered sufficient to detect large differences in this research design pre-post measurement. The low sample size associated with this study was only designed as an initial intervention study to explore biological effects and the results may form the basis for future large-scale trials.
Grouping
Thirty patients were grouped into a positive control group (K+) consisting of 10 patients with standard H. pylori therapy, treatment group 1 (P1) consisting of 10 patients with standard H. pylori therapy and administration of ZOR extract at a dose of 1×500 mg, and treatment group 2 (P2) consisting of 10 patients with standard H. pylori therapy and administration of ZOR extract at a dose of 1×1 g (Figure 1). Subject were randomly allocated into 3 groups using a computer-generated random sequence (Microsoft excel RAND) function prepared by independent assistants. Capsule of ZOR and placebo were coded to conceal treatment identity, and all investigators were blinded to group allocation until data analysis was completed.
Intervention
Standard therapy for H. pylori consists of a double-dose proton pump inhibitor PPI lansoprazole 30 mg twice a day, combined with clarithromycin 500 mg twice a day, and amoxicillin 1 g twice a day for 14 days. Zingiber officinale var. Rubrum extract was administered in capsule form dose 500 mg and obtained from the Herbal Medicine Scientification Clinic of the Tawangmangu Center for Research and Development of Medicinal Plants and Traditional Medicines under the supervision of the Indonesia Ministry of Health (Number: TL.02.04/D.XI.6/24934.1182/2024). The choice of 14 days is in accordance with the national guidelines in force in Indonesia where standard H. pylori eradication therapy can be carried out by administering PPI triple therapy combination therapy (PAC: PPI, Amoxicillin, and Clarithromycin) as the first line for a duration of 14 days. The Indonesian consensus aligns with the American College of Gastroenterology guidelines which provide a duration of triple or quadriple therapy for 14 days [10,11].
The study was conducted by selecting research subjects according to the criteria and then randomizing them into 3 groups. Prior to the intervention, all patients underwent blood sampling to check blood serum parameters and underwent a pre-intervention SF-NDI evaluation. The intervention consisted of standard therapy, either with or without ZOR at a dose of 500 mg or 1 g, for 14 days. After 14 days, repeat blood sampling and SF-NDI testing were performed. The selection of doses of 500 mg/day and 1 g/day in this study was based on previous literature studies. Preclinical studies in experimental animals showed that ZOR doses up to 800 mg/kgBW in musculus showed toxic effects.[12] Human studies have shown the use of ginger powder doses of 3 g per day without any complication [13,14]. In this study, we focused on abdominal effects, where ZOR extract doses of 0.3 - 2 g/day showed mimetic effects by inhibiting HT3. The Food and Drug Administration (FDA) has designated ginger as a dietary supplement, with consumption of no more than 6 g/day causing gastric irritation and loss of protective effects on the gastric mucosa [13-15]. The use of a dose of 500 mg/day is considered to meet the minimum clinical limit, while a dose of 1 g/day is thought to be the optimal dose for abdominal cases, considering that in this study we have tried a dose of 3 g/day and found that patients complained of discomfort, especially excessive heartburn. Doses of 500 mg/day and 1 g/day, we hope to have a good effect on the abdomen but minimal side effects and remain within the safe dose according to the FDA.
We monitor all patients regarding their level of compliance with ZOR supplementation consumption through daily blank tables and we confirm this by telephone every 2 - 3 days so that all patients described in the research results are confirmed to have a 100% level of compliance with ZOR supplement consumption. We also make sure patient did not consume any medication/other supplementation during period of study.
Outcome
Venous blood samples were taken from the study subjects for serological TNF-α, IL-6, IL-1β, and MDA using the ELISA method (https://www.elabscience.com/). Quality of life data collection based on the SF-NDI was carried out directly on the patients. Form were using Bahasa Indonesia and has been validated [16].
Statistical analysis
Researchers conducted statistical analysis using Statistical Package for Social Sciences SPSS version 24 for Windows. Quantitative data on pro TNF-α, IL-6, IL-1β, MDA, and quality of life were conducted normality tests. Statistical tests were conducted using ANOVA test/Kruskal Wallis test followed by Tukey HSD post hoc test/Dunn test. Statistical tests to find the significance of differences in 1 group used paired simple T test/Wiloxon test. Statistical results p < 0.05 were considered significant.
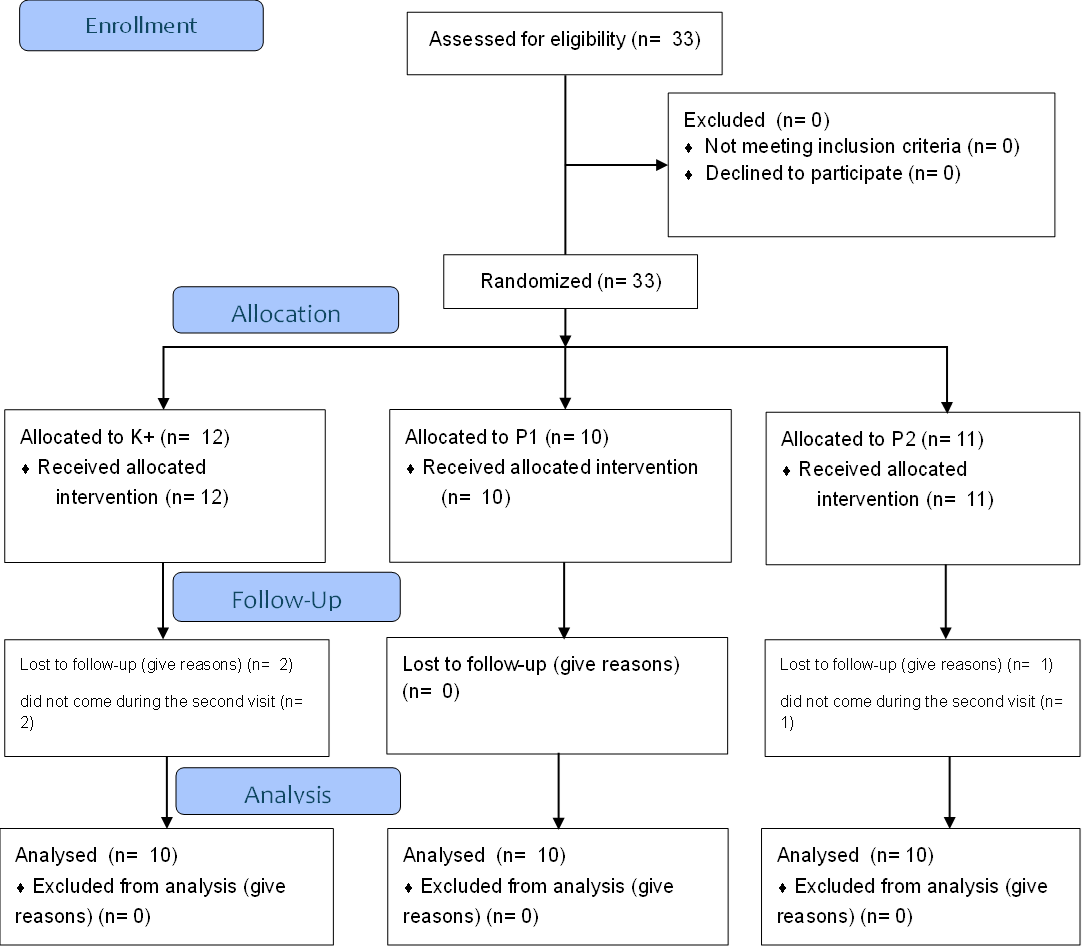
Figure 1 Flowchart consort.
Results and discussion
Analysis of the primary outcomes demonstrates notable trends in the modulation of inflammatory cytokines, oxidative stress markers, and dyspepsia symptom scores across all study groups (Table 1). Baseline values of risk factors for research subjects are presented in Table S1. For the parameters TNF-α, IL-6, IL-1β, the Shapiro-Wilk normality test (p < 0.05) and Levene's homogeneity test (p > 0.05) were obtained in both the pre-intervention and post-intervention groups so that the third parameter was continued with a non-parametric test. For the MDA parameters for groups K+ and P1, the results of the Shapiro-Wilk normality test were obtained (p < 0.05) and Levene’s homogeneity test (p > 0.05), so the test was continued with a non-parametric test, while for the MDA parameters of group P2, the results of the Shapiro-Wilk normality test were obtained (ppre = 0.424; ppost = 0.548) and Levene’s homogeneity test (p > 0.05) so that the test continues with parametric testing. For the SF-NDI parameters, the Shapiro-Wilk normality test results were obtained (p < 0.05) in the pre-intervention K+ and P2 groups and post-intervention P1 with Levene’s homogeneity test results (p > 0.05) (Table S2).
TNF‑α levels exhibited slight reductions in both the 500 mg (P1) and 1 g (P2) ZOR groups after treatment, although these changes were not statistically significant (p > 0.05) (Figure 2). This mild reduction may indicate partial attenuation of systemic inflammation following adjunctive ZOR therapy. In contrast, IL‑6 levels demonstrated a statistically significant increase within the 500 mg ZOR group (p = 0.037), while the 1 g ZOR group also showed a trend toward lower IL‑6 values (p = 0.015 among groups), suggesting a dose‑dependent anti‑inflammatory effect (Figure 3). Similarly, IL‑1β concentrations declined significantly within the 1 g ZOR group (p = 0.005), reinforcing the ability of higher‑dose ZOR supplementation to suppress pro‑inflammatory cytokine activity more effectively (Figure 4).
Regarding oxidative stress, MDA levels decreased notably in the 500 mg group (p = 0.009), indicating reduced lipid peroxidation and improved redox balance, whereas no significant change was observed in the control or 1 g groups (Figure 5). These findings imply that the antioxidant effects of ZOR may be more prominent at moderate doses, potentially reflecting different optimal thresholds for anti‑oxidative and anti‑inflammatory actions. Clinically, the total SF‑NDI scores significantly improved across all groups (Tables 1 and 2; Figure 6), reflecting better quality of life following therapy. For subdomain analysis are present in Table S4. which show each domain significantly improved after intervention all across the group. The most substantial improvements occurred in participants receiving either 500 mg or 1 g ZOR, showing statistically significant pre‑ to post‑intervention reductions (p < 0.05). This improvement corroborates laboratory findings, suggesting that symptom relief aligns with the observed reduction of systemic and gastric inflammation. The p-value between groups showed for TNF-α (p = 0.485), IL-6 (p = 0.100), IL-1β (p = 0.044), MDA (p = 0.315), and SF-NDI (p < 0.001). The IL-1β and SF-NDI parameters were followed up with a post hoc test (Table S3).
Table 1 Primary outcome: TNF-α, IL-6, IL-1β, MDA and SF-NDI.
Parameter |
Pre Intervention |
Post Intervention |
Delta (Δ) |
p within group |
p among group |
|||||
Group |
Mean ± SD |
Min − Max |
Median |
Post Mean ± SD |
Min – Max |
Median |
Mean ± SD |
|||
TNF-α (ng/L) |
K+ |
168.44 ± 125.51 |
87.19 – 482.93 |
116.70 |
170.80 ± 132.29 |
87.71 – 471.13 |
113.88 |
2.36 ± 27.02 |
0.721b |
0.439a |
P1 |
194.90 ± 213.04 |
27.71 – 678.49 |
111.83 |
162.83 ± 188.63 |
24.27 – 592.78 |
77.89 |
32.06 ± 63.73 |
0.203b |
0.235a |
|
P2 |
168.40 ± 80.00 |
87.92 – 357.23 |
138.12 |
166.00 ± 80.68 |
85.52 – 354.83 |
135.72 |
2.40 ± 0.00 |
0.114b |
0.953a |
|
IL-6 (ng/L) |
K+ |
82.59 ± 81.71 |
29.22 – 267.29 |
50.86 |
80.49 ± 70.67 |
34.95 – 218.59 |
48.79 |
2.10 ± 19.00 |
0.646b |
0.088a |
P1 |
105.26 ± 111.44 |
26.57 – 335.09 |
44.18 |
129.35 ± 138.64 |
13.66 – 428.34 |
60.26 |
24.09 ± 31.62 |
0.037b |
0.178a |
|
P2 |
82.51 ± 21.97 |
60.00 – 122.99 |
76.80 |
80.59 ± 25.75 |
57.82 – 121.88 |
69.33 |
1.92 ± 9.85 |
0.093b |
0.015a |
|
IL-1β (pg/mL) |
K+ |
855.16 ± 1018.45 |
280.27 – 2958.31 |
391.22 |
734.32 ± 642.66 |
291.33 – 2,257.22 |
500.22 |
120.83 ± 480.34 |
0.799b |
0.074a |
P1 |
1,014.33 ± 1,154.63 |
222.32 – 3494.74 |
399.80 |
1021.01 ± 1177.31 |
92.57 – 3,723.30 |
478.59 |
6.67 ± 328.66 |
0.646b |
0.081a |
|
P2 |
855.76 ± 325.37 |
634.65 – 1740.50 |
746.15 |
734.19 ± 322.81 |
563.71 – 1,646.41 |
634.80 |
121.57 ± 63.99 |
0.005b |
0.053a |
|
MDA (nmol/mL) |
K+ |
14.64 ± 11.79 |
4.56 – 41.00 |
9.89 |
13.88 ± 11.74 |
4.85 – 36.42 |
8.60 |
0.75 ± 4.78 |
0.575b |
0.388a |
P1 |
23.27 ± 27.80 |
2.87 – 78.13 |
9.36 |
15.64 ± 19.12 |
3.39 – 60.55 |
8.24 |
7.63 ± 19.30 |
0.009b |
0.159a |
|
P2 |
14.60 ± 3.89 |
6.47 – 21.21 |
15.55 |
13.89 ± 2.89 |
10.39 – 19.17 |
13.98 |
0.71 ± 1.93 |
0.271c |
0.572a |
|
SF-NDI (Score Total) |
K+ |
3.65 ± 0.99 |
1.00 – 5.00 |
4.00 |
1.90 ± 0.75 |
1.00 – 4.00 |
2.00) |
1.75 ± 1.24 |
0.005b |
0.005a |
P1 |
3.31 ± 0.92 |
1.00 – 5.00 |
3.00 |
2.05 ± 0.68 |
1.00 – 4.00 |
2.00 |
1.37 ± 0.96 |
< 0.001c |
0.212a |
|
P2 |
3.42 ± 0.63 |
1.00 – 5.00 |
3.00 |
2.05 ± 0.72 |
1.00 – 4.00 |
2.00 |
1.26 ± 1.14 |
0.005b |
0.163a |
|
Note: TNF-α (tumor necrosis factor alfa); IL-6 (interleukin-6); IL-1β (interleukin-1 beta); MDA (malondialdehyde); SF‑NDI (Short‑Form Nepean Dyspepsia Index); K+ (positive control group, treated with H. pylori eradication standard therapy); P1 (treatment group 1, treated with H. pylori eradication standard therapy and ZOR extract dose 1x500 mg); P2 (treatment group 1, treated with H. pylori eradication standard therapy and ZOR extract dose 1x1 g);a = Kruskall‑Wallis Test; b = Wilcoxon Test; c = paired T Test.
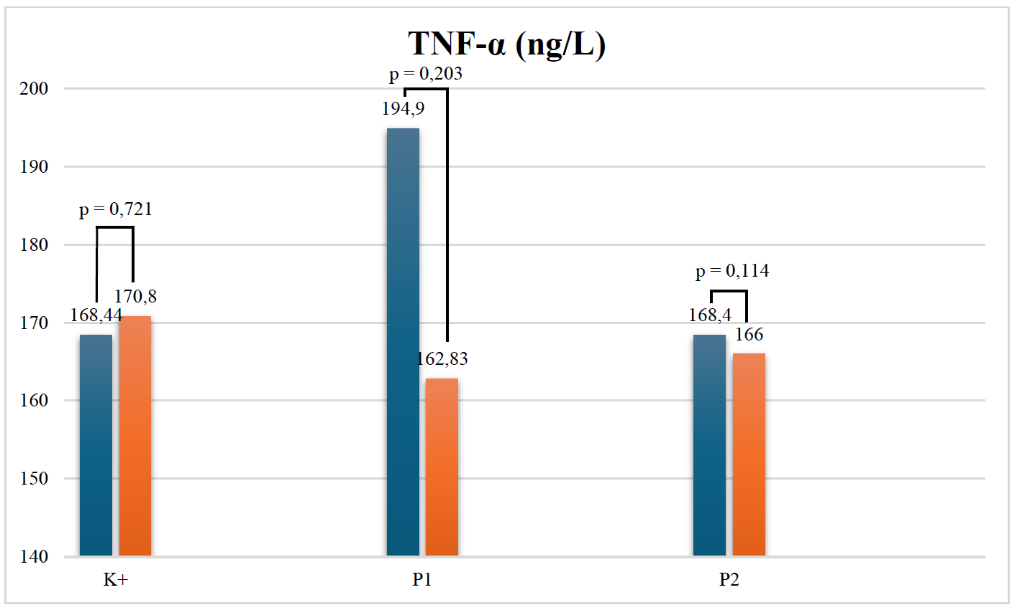
Figure 2 TNF-α level. In ZOR, the 500 mg dose and the 1 g dose showed insignificant suppression of TNF-α values. Note: TNF-α (tumor necrosis factor-alpha); ng/L (nanogram/liter); p (significant value); * (p<0,05); K+ (positive control group, treated with H. pylori eradication standard therapy); P1 (treatment group 1, treated with H. pylori eradication standard therapy and ZOR extract dose 1×500 mg); P2 (treatment group 1, treated with H. pylori eradication standard therapy and ZOR extract dose 1×1 g)
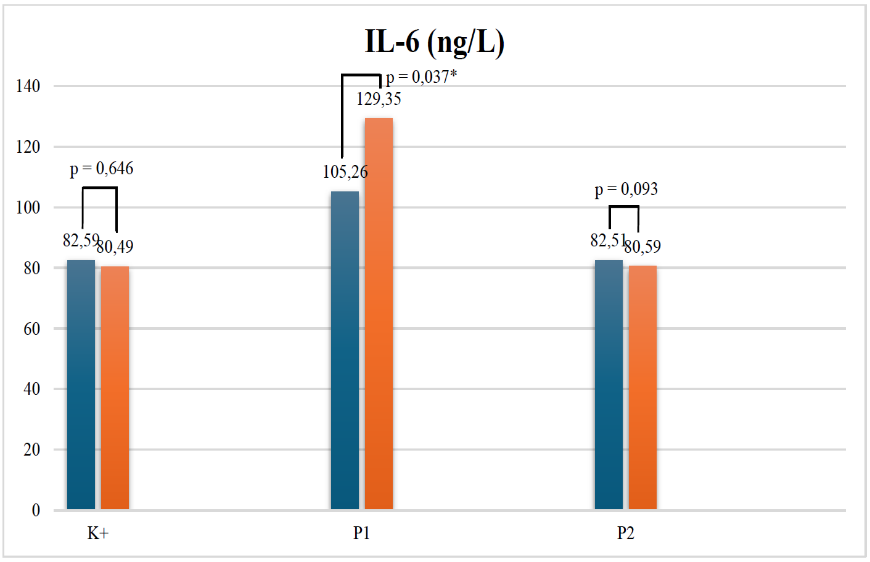
Figure 3 IL-6 level. At a dose of 1 g ZOR, IL-6 suppression was observed. Note: IL-6 (interleukin-six); ng/L (nanogram/liter); p (significant value); * (p<0,05); K+ (positive control group, treated with H. pylori eradication standard therapy); P1 (treatment group 1, treated with H. pylori eradication standard therapy and ZOR extract dose 1×500 mg); P2 (treatment group 1, treated with H. pylori eradication standard therapy and ZOR extract dose 1×1 g)
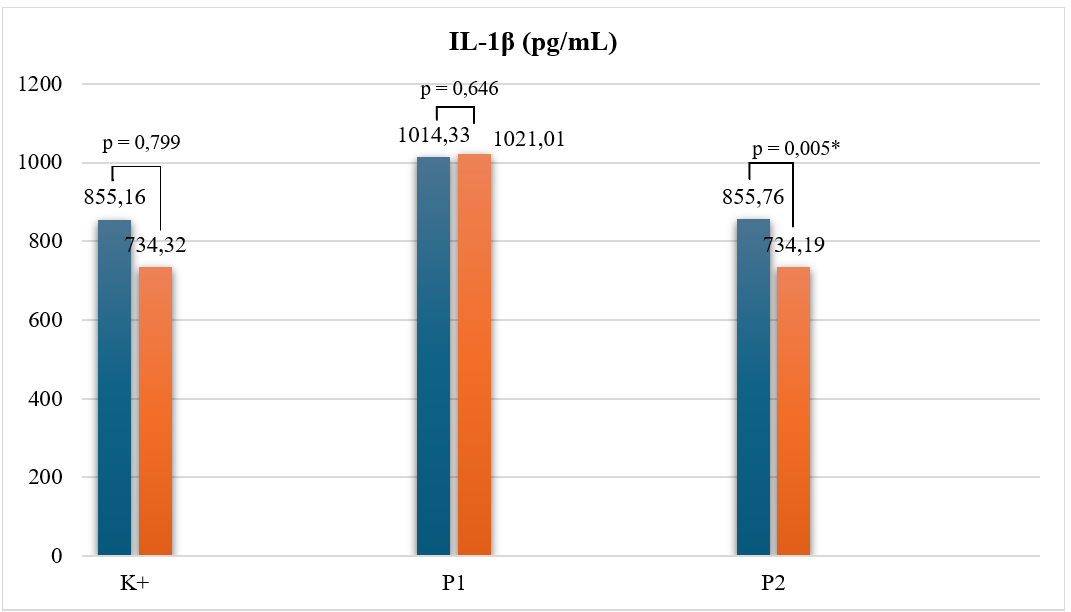
Figure 4 IL-1β level. At a dose of 1 g ZOR, IL-1β suppression was observed. Note: IL-1β (interleukin-one betha); pg/mL (pico/millliter); p (significant value); * (p<0,05); K+ (positive control group, treated with H. pylori eradication standard therapy); P1 (treatment group 1, treated with H. pylori eradication standard therapy and ZOR extract dose 1×500 mg); P2 (treatment group 1, treated with H. pylori eradication standard therapy and ZOR extract dose 1×1 g)
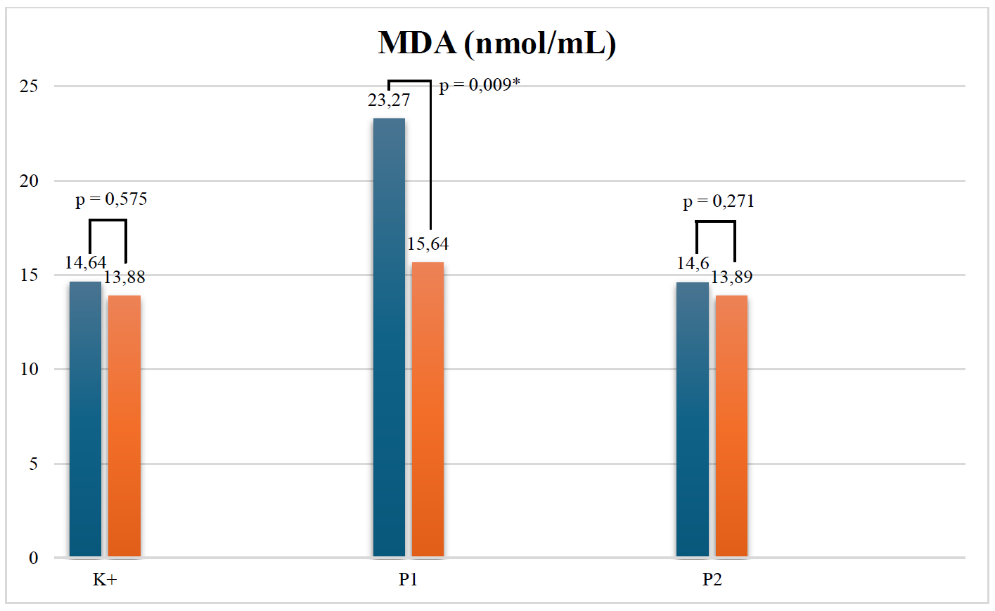
Figure 5 MDA level. In ZOR, the 500 mg dose and the 1 g dose showed suppression of MDA values, significant at lower dose. Note: MDA (malondialdehyde); nmol/mL (nanomole/milliliter); p (significant value); * (p<0,05); K+ (positive control group, treated with H. pylori eradication standard therapy); P1 (treatment group 1, treated with H. pylori eradication standard therapy and ZOR extract dose 1×500 mg); P2 (treatment group 1, treated with H. pylori eradication standard therapy and ZOR extract dose 1×1 g)
Table 2 Short-form Nepean dyspepsia index.
Group |
SF-NDI Score Mean ± SD Per Question (Maximal - Minimal) (Median) |
||||||||
K+ |
P1 |
P2 |
|||||||
Pre |
Post |
∆ |
Pre |
Post |
∆ |
Pre |
Post |
∆ |
|
P1. Emotional disturbance due to stomach discomfort over the past 2 months |
3.80 ± 0.78 (2 - 5) (4) |
2.20 ± 0.42 (2 - 3) (2) |
1.60 ± 0.84 |
3.80 ± 0.42 (3 - 4) (3) |
2.40 ± 0.51 (2 - 3) (2) |
1.40 ± 0.51 |
3.40 ± 1.07 (2 - 5) (4) |
2.30 ± 0.67 (1 - 3) (2) |
1.10 ± 0.87 |
P2. Feeling tense, frustrated, or upset due to stomach symptoms over the past 2 weeks |
3.90 ± 0.87 (2 - 5) (4) |
2.10 ± 0.56 (1 - 3) (2) |
1.80 ± 0.78 |
3.80 ± 0.42 (3 - 4) (3.5) |
2.40 ± 0.51 (2 - 3) (2) |
1.40 ± 0.51 |
3.50 ± 1.08 (2 - 5) (4) |
2.30 ± 0.67 (1 - 3) (2) |
1.20 ± 0.91 |
P3. Interference of stomach discomfort with doing enjoyable activities over the past 2 weeks |
3.90 ± 0.87 (2 - 5) (4) |
2.30 ± 0.82 (1 - 4) (2) |
1.60 ± 0.69 |
3.20 ± 0.42 (3 - 4) (3) |
2.00 ± 0.66 (1 - 3) (2) |
1.20 ± 0.42 |
3.30 ± 0.94 (2 - 5) (3) |
2.00 ± 0.66 (1 - 3) (2) |
1.30 ± 0.67 |
P4. Reduced enjoyment of favorite activities due to stomach symptoms over the past 2 weeks |
3.90 ± 0.87 (2 - 5) (4) |
2.30 ± 0.82 (1 - 4) (2) |
1.60 ± 0.69 |
3.20 ± 0.42 (3 - 4) (3) |
2.00 ± 0.66 (1 - 3) (2) |
1.20 ± 0.42 |
3.30 ± 0.94 (2 - 5) (3) |
2.00 ± 0.66 (1 - 3) (2) |
1.30 ± 0.67 |
P5. Difficulty eating or drinking comfortably because of stomach problems over the past 2 weeks |
3.60 ± 0.84 (2 - 5) (4) |
1.80 ± 0.78 (1 - 3) (2) |
1.80 ± 0.78 |
3.40 ± 0.51 (3 - 4) (3.5) |
1.90 ± 0.99 (1 - 4) (2) |
1.50 ± 0.70 |
3.40 ± 0.69 (2 - 4) (3) |
2.40 ± 0.69 (2 - 4) (2) |
1.00 ± 0.66 |
P6. Reduced enjoyment of eating or drinking because of stomach discomfort over the past 2 weeks |
3.70 ± 0.67 (3 - 5) (4) |
1.90 ± 0.73 (1 - 3) (2) |
1.80 ± 0.78 |
3.40 ± 0.51 (3 - 4) (3.5) |
1.90 ± 0.99 (1 - 4) (2) |
1.50 ± 0.70 |
3.40 ± 0.69 (2 - 4) (3) |
2.40 ± 0.69 (2 - 4) (2) |
1.00 ± 0.21 |
P7. Frequent worry about experiencing stomach problems over the past 2 weeks |
3.50 ± 1.26 (1 - 5) (4) |
1.60 ± 0.96 (1 - 3) (1) |
1.90 ± 1.37 |
3.20 ± 0.63 (2 - 4) (3) |
1.90 ± 0.56 (1 - 3) (1) |
1.30 ± 0.48 |
3.10 ± 1.10 (2 - 5) (3) |
1.40 ± 0.51 (1 - 2) (2) |
1.70 ± 0.94 |
P8. Thinking that stomach symptoms could be caused by a serious illness over the past 2 weeks |
2.80 ± 1.54 (1 - 5) (2) |
1.20 ± 0.63 (1 - 3) (1) |
1.60 ± 1.50 |
2.60 ± 0.69 (1 - 3) (2) |
1.40 ± 0.51 (1 - 2) (1) |
1.20 ± 0.63 |
2.90 ± 1.37 (1 - 5) (3) |
1.30 ± 0.48 (1 - 2) (1) |
1.60 ± 1.26 |
P9. Difficulty performing work or study activities because of stomach discomfort over the past 2 weeks |
3.70 ± 0.94 (2 - 5) (4) |
1.80 ± 0.63 (1 - 3) (2) |
1.90 ± 1.19 |
3.80 ± 0.63 (3 - 5) (3.5) |
2.30 ± 0.67 (2 - 4) (2) |
1.50 ± 0.52 |
3.40 ± 0.69 (2 - 4) (4) |
2.20 ± 0.42 (2 - 3) (2) |
1.20 ± 0.63 |
P10. Decreased enjoyment of work or study because of stomach problems over the past 2 weeks |
3.70 ± 0.94 (2 - 5) (4) |
1.80 ± 0.63 (1 - 3) (2) |
1.90 ± 1.19 |
3.80 ± 0.63 (3 - 5) (3.5) |
2.30 ± 0.67 (2 - 4) (2) |
1.50 ± 0.52 |
3.40 ± 0.69 (2 - 4) (4) |
2.20 ± 0.42 (2 - 3) (2) |
1.20 ± 0.63 |
Note: P1 (treatment group 1, treated with H. pylori eradication standard therapy and ZOR extract dose 1×500 mg); P2 (treatment group 1, treated with H. pylori eradication standard therapy and ZOR extract dose 1×1 g).
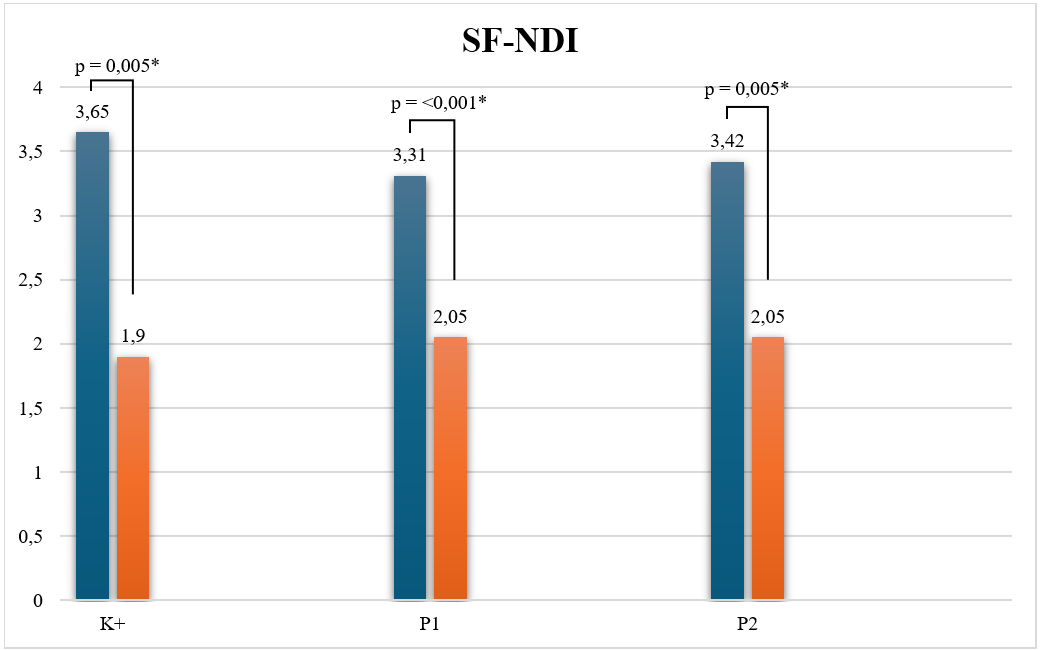
Figure 6 SF-NDI score. In ZOR, the 500 mg dose and the 1 g dose showed significant improvement of SF-NDI. Note: SFNDI (Short-form Nepean dyspepsia index); p (significant value); * (p<0,05); K+ (positive control group, treated with H. pylori eradication standard therapy); P1 (treatment group 1, treated with H. pylori eradication standard therapy and ZOR extract dose 1×500 mg); P2 (treatment group 1, treated with H. pylori eradication standard therapy and ZOR extract dose 1×1 g)
Tumor necrosis factor-α
Elevated pre-intervention TNF-α levels are associated with local and systemic H. pylori infection. H. pylori infection can activate pattern recognition receptors, such as TLRs, on gastric epithelial and immune cells, leading to activation of the NF-κB signaling pathway. Furthermore, heat shock protein (HSP) virulence factors directly induce TNF-α [17-20]. The effectiveness of Zingiber officinale var. Rubrum on TNF-α is attributed to 6-shogaol, 6-gingerol, and Zingerone. Studies have reported that the metabolite 6-shogaol is the most potent TNF-α inhibitor due to its α,β-unsaturated carbonyl group. The metabolite 6-gingerol supports TNF-α suppression through suppression of the JNK pathway [21,22]. Another metabolite is Zingerone which plays a role in modulating Nuclear Factor Erythroid 2–Related Factor 2/Heme Oxygenase-1 (NRF2/HO-1) signals which reduce TNF-α transcription [23]. The mechanism of TNF-α suppression in the NF-κB pathway is caused by the blockade of 6-sgoahol and 6-gingerol on IκB kinase (IKK) causing prevention of IκBα degradation and “trapping” NF-κB in the cytoplasm. This condition prevents TNF-α gene transcription and the secretion of other proteins [23-26]. The decrease in TNF-α transcription is supported by the role of Z. officinale as a PPARγ agonist [22,27]. Zingiber officinale modulation of the MAPK pathway is due to inhibition of ERK, JNK, and p38 phosphorylation by 6-shoagol. Suppression of MAPK signaling leads to decreased stability and translation of TNF-α mRNA [24]. The metabolite 6-shogaol can activate NRF-2, which increases HO-1, thereby suppressing ROS. This reduction in ROS contributes to improved inflammatory conditions, including reduced TNF-α levels [23]. Other findings indicate that 6-shoagol can inhibit the NLRP3 inflammasome, thereby suppressing TNF-α amplification [25].
A study by Amanda et al. [25] on the experimental oral administration of ZOR at doses of 100 mg/kgBW, 200 mg/kgBW, and 400 mg/kgBW for 14 days in a Pfizer-inducing mouse model (0.1 mL) showed a decrease in TNF-α levels (p < 0.05), with the best dose being 100 mg/kgBW [28]. Song et al. [26] reported a decrease in TNF-α mRNA concentration when given GGE03 (steamed ginger extract) at doses of 50, 100 and 200 µg/mL in gastritis caused by H. pylori [29]. Gaus et al. [27] reported that giving H. pylori at a dose of 100 mg/kgBW for 3 weeks before H. pylori induction and 6 weeks after H. pylori induction in mice showed inhibition of COX-2 and NF-ĸβ which suppressed TNF-α production with a 50% inhibitory concentration (IC50) of 8.37 µg/mL [30]. Zhong et al. [28] reported the alignment of TNF-α suppression in a preclinical study in the form of ginger juice administration as a gastroprotectant in a mouse model of gastric ulcer induction. The gastroprotective effect was shown by a decrease in TNF-α (pg/mL) sequentially for the healthy control group, placebo group, 150 g fresh ginger juice group, 150 g fresh ginger boiled juice group, and 50 g dried ginger juice group by 57.70 ± 7.77, 63.90 ± 6.42, 55.75 ± 8.14, 61.35 ± 5.87 and 67.04 ± 6.27 [31]. Another study by Saiah et al. [29] reported the gastroprotective effect of Z. officinale on the administration of active metabolites of Z. officinale, namely butanol fractions at doses of 25, 50 and 100 mg/kgBW which were able to reduce TNF-α concentrations (pg/ml) by 96.78 ± 3.02, 91.06 ± 2.71 and 62.41 ± 3.32, respectively [32]. A systematic review by Morvaridzadeh et al. (2020) reported a significant decrease in TNF-α after Z. officinale administration (SMD: −0.85, 95% CI: −1.48, −0.21, I2 = 89.4%) [33].
Interleukin-6
The pathway of increased proinflammatory cytokine IL-6 in H. pylori infection is analogous to the increase in proinflammatory cytokine TNF-α, namely in the NF-ĸβ pathway. H. pylori infection stimulates gastric epithelial cells and immune cells such as macrophages to produce IL-6 primarily through the activation of several intracellular signaling pathways. Key bacterial virulence factors such as cag pathogenicity island (cagPAI) and outer inflammatory protein A (OipA) strongly induce IL-6 gene expression. These factors activate the MAPK pathway (ERK, JNK, p38), which, together with transcription factors (NF-κB, AP-1, CRE, C/EBP), increases IL-6 transcription. Further studies have shown that H. pylori also triggers STAT3 activation and a positive feedback loop between epithelial cells (GES-1) and monocytes (THP-1), which amplifies IL-6 production in both cell populations. Interleukin-6 plays a role in maintaining local inflammation, immune cell recruitment, and gastric mucosal survival/inflammation during infection [19,34-36]. Helicobacter pylori eradication therapy successfully eliminates bacterial stimuli, halting the activation of MAPK, NF-κB, and STAT3 signaling in gastric epithelial and immune cells. This process leads to a rapid downregulation of IL-6 gene transcription and decreased cytokine secretion. As inflammation and immune cell infiltration subside due to bacterial clearance, local and systemic IL-6 levels decrease significantly, reflecting the attenuation of pro-inflammatory signals and the restoration of gastric mucosal homeostasis [19,34-36].
Yulma et al. [34] showed that administration of ZOR at doses of 100, 200 and 400 mg/kgBW in mice induced with Pfizer showed a decrease in IL-6 concentrations (p < 0.05). A study by Yulma et al. [37] reported the best reduction in IL-6 at a dose of 100 mg/kgBW. Gaus et al. [27] reported that administering Z. officinale to H. pylori-induced mice at a dose of 100 mg/kgBW 3 weeks before and 6 weeks after induction was able to reduce the IC50 of IL-6 by 7.7 µg/mL [30]. Song et al. [26] reported IL-6 inhibition through the NF-κB pathway upon administration of GGE03 at doses of 50, 100 and 200 µg/mL [29]. Hefni et al. [18] reported that administering Z. officinale var. Rubrum extract to mice was able to reduce serum IL-6 concentrations within 14 days of administration through suppression of the NF-κB pathway [21]. Zehsaz et al. [35] reported that in individuals with oxidative stress induction, administration of Z. officinale at a dose of 3×500 mg daily for 6 weeks was able to reduce IL-6 by 17.5% (p < 0.01) [38].
In the 500 mg ZOR dose group, IL-6 levels actually increased after the intervention. Interleukin-6 suppression after Z. officinale supplementation still shows varying results. Morvaridzadeh et al. [30] reported that IL-6 suppression by Z. officinale did not show significant clinical effectiveness (SMD: −0.45, 95% CI: −1.29, 0.38, I2 = 89.2%) [33]. The increase in IL-6 upon administration of ZOR can also be explained by the fact that IL-6 acts as both an anti-inflammatory and pro-inflammatory cytokine. The increase in IL-6 upon ZOR administration can be hypothesized as a mechanism for IL-6 as an anti-inflammatory agent which increases following improvement in inflammatory conditions after ZOR supplementation. As an anti-inflammatory agent, IL-6 works by upregulating anti-inflammatory mediators such as IL-1 receptor antagonist (IL-1ra) and IL-10, both of which suppress the action of major pro-inflammatory cytokines, such as TNF-α and IL-1β. In response to physiological stress or exercise, a transient increase in IL-6 from muscle acts as a signal to trigger systemic anti-inflammatory pathways and increase production of counter-regulators [39,40].
Interleukin-1β
Similar to the induction parameters of the pro-inflammatory cytokines TNF-α and IL-6, the IL-1β induction pathway in H. pylori infection is similar. Helicobacter pylori stimulates pro-IL-1β production, which requires cleavage by the NLRP3 inflammasome. Activation occurs through multiple mechanisms, including the generation of reactive oxygen species, potassium efflux, lysosome destabilization, and the release of extracellular ATP. Furthermore, H. pylori pathogenic factors, particularly CagA and VacA, activate toll-like receptor 4 (TLR4) and NF-κB, which promote IL-1β mRNA transcription. Furthermore, polymorphisms capable of increasing IL-1β production are associated with a higher risk of gastric cancer and more severe gastritis, suggesting IL-1β as a central inflammatory mediator in H. pylori infection [36,41].
The role of Z. officinale in suppressing IL-1β is demonstrated by the roles of 6-gingerol, 6-shogaol, and Zingerone. The 6-gingerol metabolite is able to suppress IL-1β through inhibition of the ERK and p38 MAPK pathways [42]. The 6-shogaol metabolite is able to suppress endothelial inflammation through inhibition of IL-1β by blocking NF-κB translocation by 35% and reducing the expression of adhesion molecules including ICAM-1, VCAM-1, and E-selectin [43]. The role of Zingerone in inhibiting IL-1β is through inhibition of TLR2/4 and activation of MAPK2. Zingerone also works by disrupting the assembly of the Absent in Melanoma 2 (AIM2) inflammasome, reducing the formation of ASC protein specks, and caspse-1 activation [44]. The anti-inflammatory effect synergizes with the antioxidant effect. The metabolite 6-shoagol induces HO-1 via the p38 MAPK pathway, which enhances antioxidant defenses and decreases IL-1β. The metabolite zingerone can activate Nrf2/HO-1, which regulates oxidative stress, triggering the release of IL-1β [23,44-46].
A study by Zehsaz et al. [35] reported in men induced by endurance running training for 6 weeks followed by administration of Z. officinale capsules at a dose of 3x500 mg daily for 6 weeks showed a significant (p < 0.01) decrease in IL-1β in the Z. officinale intervention group by 18.95% [38]. A study by Gaus et al. [27] found that administering Z. officinale at a dose of 100 mg/kgBW 3 weeks before and 6 weeks after induction reduced the IC50 of IL-1β by 3.89 µg/mL [30]. A study in rheumatoid arthritis (RA) patients given 1.5 g of Z. officinale per day showed a decrease in IL-1β mRNA (p = 0.021) and TNF-α mRNA (p = 0.093) [47].
After the ZOR 500 mg intervention, there was an increase in pro-inflammatory cytokines, indicating that ZOR supplementation was influenced by dose. Studies on herbal-based treatments have found that low doses can sometimes act as partial agonists or fail to suppress the signal completely, resulting in uncontrolled or even worsened proinflammatory responses. Natural compounds, including Z. officinale, can exhibit a “J or U shape” hormetic effect, whereby low doses can trigger cell signaling or mild stress, briefly increasing proinflammatory gene expression before suppression occurs at adequate concentrations. Hormetic phenomena (or hormesis) describe a biphasic biological response to a substance or stressor, where low doses are beneficial or stimulatory while higher doses are harmful or inhibitory. Hormesis is a dose–response pattern in which exposure to a low dose of a potentially harmful agent activates adaptive responses that improve function or survival, whereas higher doses cause toxicity or inhibition [39,48-50].
At lower concentrations, Z. officinale compounds may affect immune cell subsets or the microenvironment differently, resulting in variable or even paradoxical cytokine trends, especially if anti-inflammatory pathways such as NRF2/HO-1 activation are not strongly activated. Zingiber officinale's complex phytochemicals act in multiple pathways, such as NF-κB inhibition, requiring higher doses to have an effect, while at low doses, other mechanisms such as MAPK or TLR signaling may temporarily dominate, leading to incomplete or inconsistent cytokine suppression [39,48-50].
The hypothesis reports that herbal supplementation with antioxidant content has pro-oxidant properties at certain doses. Studies report that the content of asobic acid, vitamin E, and polyphenols are pro-oxidant at certain doses, especially if the presence of metal (iron) is known to cause oxidative stress induction [51]. Studies report that grape seed extract as an antioxidant agent is able to suppress increases in ROS and NOX4 at certain doses due to the prooxidant properties of its polyphenol groups [52]. Cellular studies on vitamin E analogues show a transition of antioxidant effects towards prooxidants as indicated by an increase in ROS parameters and apoptosis/necrosis parameters [53,54]. The transition process of antioxidants to prooxidants is based on the Fe3+ to Fe2+ transition mechanism which plays a role in the Fenton reaction which induces ROS and oxidative stress [51,54].
Malondialdehyde
Implementation of H. pylori eradication therapy will essentially eliminate the underlying stimulus for inflammatory cell infiltration and ROS formation. Direct resolution of the infection will suppress proinflammatory cytokines and ROS production. Furthermore, the gastric antioxidant system (e.g., glutathione and ascorbic acid) will recover, further suppressing lipid peroxidation and normalizing MDA levels [55]. Z. officinale secondary metabolites that act as antioxidants are primarily 6-gingerol, 6-shogaol, zingerone, and 6-paradol. Direct ROS elimination is mediated by 6-gingerol and 6-shogaol by neutralizing ROS through a Michael’s addiction reaction that prevents lipid peroxidation [56], [57]. Zingerone, on the other hand, is capable of degrading free radicals through radiolysis and inhibiting the source of ROS, namely xanthine oxidase [58]. The mechanism of endogenous antioxidant enhancement by Z. officinale is mediated by 6-shogaol and Zingerone, which activate Nrf2, which increases HO-1, increasing the synthesis of superoxide dismutase (SOD), catalase (CAT), and glutathione (GSH), which suppresses MDA [58,59].
Endogenous antioxidant enhancement by 6-gingerol occurs through a mitochondrial protection mechanism by maintaining mitochondrial membrane potential and lysosomal integrity. This protective mechanism prevents DNA damage by ROS, as indicated by a decrease in MDA [56,57]. The next pathway is through inhibition of the prooxidant pathway, played by 6-shogaol through LOX-1 blockade and Zingerone through metal chelation [60,61]. Metal chelation prevents the Fenton reaction, which produces hydroxyl radicals. The antioxidant mechanism of Z. officinale in suppressing MDA levels synergizes with anti-inflammatory processes [61]. The metabolite 6-paradol is able to suppress TNF-α/IL-6 levels, which indirectly reduces oxidative stress, indicated by decreased MDA levels. In this study, ZOR at dose 500 mg show better result than 1 gr. It can be hypothesized that the optimal dose of ZOR as an antioxidant is 500 mg. If the dose is increased, it will become a prooxidant, thereby suppressing endogenous antioxidant [62].
Oboh et al. [57] reported that Z. officinale var. Rubrum intervention in rats with brain lipid peroxidation demonstrated an increasing level of MDA inhibition with dose. At a dose of 0.78 mg/mL, the MDA value was 89.29%, while at a dose of 3.13 mg/mL, the MDA value was 16.97% [60]. Saiah et al. [29] in a gastric ulceration model showed an improvement in MDA after administration of the active metabolite of Z. officinale butanol fraction at doses of 25, 50 and 100 mg/kgBW with MDA values of 10.63 ± 0.27, 10.32 ± 0.24 and 4.94 ± 0.07, respectively [32]. Baiubon et al. [60] was similar to the Saiah et al. [29] study model. reported an improvement in MDA levels when given Z. simaoense extract at a dose of 30 mg/kgBW with MDA levels of 0.98 ± 0.07 which was lower than the placebo group (1.42 ± 0.15) and the standard therapy group (1.04 ± 0.10) [63].
Quality of life: SF-NDI
Similar results were found by Mestrovic et al. [61] who demonstrated improvements in quality of life after H. pylori eradication therapy using the Gastrointestinal Symptom Rating Scale (pre vs. post = 23 vs. 19, p < 0.001) in the domains of abdominal pain, reflux symptoms, and maldigestion [64]. The mechanism of action of H. pylori eradication therapy is as previously described. The third regimen works by directly eliminating H. pylori, which is able to block or interrupt pathological processes, including infection, inflammation, and oxidative stress, thus improving clinical manifestations in patients. Amoxicillin is able to lyse the H. pylori cell wall through binding to penicillin-binding proteins. Administration of lansoprazole by increasing gastric pH causes amoxicillin to work more effectively in a low-acid environment. Lansoprazole not only increases the stability and effectiveness of amoxicillin but also allows H. pylori to replicate, making it more susceptible to antibiotic action. Higher pH levels enhance the absorption and bioavailability of antibiotics in gastric fluid, increasing antimicrobial potency, and helping to eradicate biofilm-forming bacteria. Like amoxicillin, clarithromycin works by inhibiting bacterial protein synthesis by binding to the 50S ribosomal subunit in H. pylori, resulting in a reduced bacterial load. H. pylori eradication manifests as mucosal healing, symptom improvement, and reduced inflammation, which collectively restore normal gastrointestinal function [65-67].
Zingiber officinale acts as an antiemetic and antinausea agent by suppressing M1, M3, and M3 cholinergic receptors, as well as 5-HT3/5-HT4 (hydroxytryptamine) receptors [68]. In vitro studies have shown that 6-shogaol, 6-gingerol, and zingerone inhibit vagal afferent neuron signal transmission by suppressing 5-HT receptors, with 6-shogaol being the best suppressor [68,69]. Gingerol is also thought to have effects similar to ondansetron, through suppression of substance P and neurokinin 1 receptors (NK1R) [68,70]. Zingiber officinale plays a role in gastric emptying in dyspepsia patients through its carminative effect, which is a warming sensation on the lower esophageal sphincter to relieve symptoms of postprandial bloating due to belching, flatulence, and decreased contraction rate [71]. This effect is created by Z. officinale by reducing pressure and peptic cramps in the LES. Reduction of nausea and vomiting sensations has also been described in the administration of Z. officinale to patients with liver disorders, through suppression of liver inflammation with a decrease in pro-inflammatory cytokines and ROS markers [68,70,72,73].
The study had several limitations. The primary constraints were the relatively small sample size and the absence of antibiotic resistance testing, which limits the ability to interpret treatment response variability. Additionally, no phenotypic or genotypic analysis of H. pylori strains was performed, the study lacked a negative control group (H. pylori), and absence of chromatographic profiling to confirm the concentration of active metabolites. Future investigations should include a larger number of participants, incorporate antibiotic susceptibility profiling, and apply rigorous control of potential confounding variables. Recommendation to do chromatographic profiling to identify the active metabolites in ZOR. The use of quantitative biomarker analyses such as flow cytometry or qPCR and long-term follow-up assessments is also recommended to better evaluate recurrence rates and the sustained therapeutic effects of the intervention.
Conclusions
ZOR supplementation at doses of 500 mg and 1 g demonstrated notable biological and clinical effects in H. pylori-associated chronic gastritis. More prominently, the 1 g ZOR dose exerted additional benefits, producing measurable decreases in IL‑6 and IL‑1β levels when compared across groups, indicating that higher-dose supplementation may more effectively suppress pro‑inflammatory cytokine production. Although MDA values, a marker of lipid peroxidation, showed a mild reduction following intervention, the difference did not reach statistical significance, implying that antioxidant effects may require longer administration periods or larger sample sizes to be clinically evident. From a clinical standpoint, improvements in quality of life, as assessed by total SF‑NDI scores, were most evident in subjects receiving both the 500 mg and 1 g ZOR formulations. Collectively, these findings suggest that ZOR extract, when integrated with standard H. pylori eradication therapy, has the potential to reduce inflammatory biomarkers and enhance patient‑perceived well-being, with the 1 g dosage offering broader cytokine modulation. The conclusion from this study should corresponded with various limitations of this study, the main ones being the lack of direct quantification of H. pylori by culture or PCR and the absence of antibiotic resistance testing, caution should also be exercised given the small sample size and uncontrolled confounders.
Declaration of generative AI in scientific writing
The authors used Perplexity Pro (perplexity.ai) to im-prove the language and readability of the manuscript. The authors reviewed and edited the content and take full re-sponsibility for its accuracy. Graphical abstract. Other images were not generated or manipulated using AI tools
CRediT author statement
Coana Sukmagautama: Conceptualization; Methodology; Software; Validation; Formal Analysis; Investigation; Resources; Data Curation; Writing – Original Draft; Writing – Review & Editing; Visualization; Supervision; Project Administration. Reviono: Conceptualization; Methodology; Validation; Resources; Data Curation; Writing – Original Draft; Supervision. Wachid Putranto: Conceptualization; Resources; Writing – Original Draft; Writing – Review & Editing; Visualization; Supervision. Eti Poncorini Pamungkasari: Conceptualization; Methodology; Software; Validation; Formal Analysis; Visualization; Supervision. Hartono: Conceptualization; Writing – Original Draft; Writing – Review & Editing; Supervision. Risya Cilmiaty: Conceptualization; Writing – Review & Editing; Supervision. Paramasari Dirgahayu: Conceptualization; Methodology; Supervision; Project Administration.
References
JKY Hooi, WY Lai, WK Ng, MMY Suen, FE Underwood, D Tanyingoh, P Malfertheiner, DY Graham, V Wong, JCY Wu, FKL Chan, JJY Sung, GG Kaplan and SC Ng. Global prevalence of Helicobacter pylori infection: Systematic review and meta-analysis. Gastroenterology 2017; 153, 420-429.
H Maulahela, D Doohan, YAA Rezkhita, AF Syam, LA Waskito, CMA Savitri, M Richardo, A Rahman, YS Namara, H Shatri, A Sanityoso, GA Siregar, E Sudarmo and T Uchida. Helicobacter pylori prevalence in Indonesia: Higher infection risk in Eastern region population. F1000Research 2022; 11, 1581.
M Simadibrata and D Mustikarani. Prevalence and association of Helicobacter pylori infection in bowel disease at Dr. Cipto Mangunkusumo General National Hospital 2010-2021. The Indonesian Journal of Gastroenterology, Hepatology, and Digestive Endoscopy 2024; 24, 222.
SZ Molaoa. Prevalence of Helicobacter pylori infection and the incidence of the associated malignant and peptic ulcer disease at Nelson Mandela Academic Hospital: A retrospective analysis. Journal of Drug Assessment 2021; 10(1), 57-61.
S Diaconu, A Predescu, A Moldoveanu, CS Pop and C Fierbințeanu-Braticevici. Helicobacter pylori infection: Old and new. Journal of Medicine and Life 2017; 10(2), 112-117.
O Nestegard, B Moayeri, FA Halvorsen, T Tønnesen, SW Sørbye, E Paulssen, KM Johnsen, R Goll, JR Florholmen and KK Melby. Helicobacter pylori resistance to antibiotics before and after treatment: Incidence of eradication failure. PLoS One 2022; 17, e0265322.
S Mukjerjee and D Karati. A mechanistic view on phytochemistry, pharmacognostic properties, and pharmacological activities of phytocompounds present in Zingiber officinale: A comprehensive review. Pharmacological Research-Modern Chinese Medicine 2022; 5, 100173.
D Makmun, A Syam, H Maulahela, R Makmun, A Renaldi, S Simadibrata and A Abdullah. Konsensus nasional penatalaksanaan dispepsia dan infeksi Helicobacter pylori di Indonesia (revisi 2021) (in Indonesian). PIPInterna, Jakarta, Indonesia, 2022.
PSD Dewi and MT Ma’ruf. Sub-acute toxicity test of red ginger extract (Zingiber officinale var. rubrum) on mice (Mus musculus). Interdental Jurnal Kedokteran Gigi 2023; 19(1), 1-5.
C Zhang, A Rao, C Chen, Y Li, X Tan, J Long, X Wang, J Cai, J Huang, H Luo, C Li and Y Dang. Pharmacological activity and clinical application analysis of traditional Chinese medicine ginger from the perspective of one source and multiple substances. Chinese Medicine 2024; 19, 97.
S Zhang, X Kou, H Zhao, KK Mak, MK Balijepalli, MR Pichika and A Khan. Zingiber officinale var. rubrum: Red ginger’s medicinal uses. Molecules 2022; 27, 775.
RD Supu, A Diantini and J Levita. Red ginger (Zingiber officinale var. rubrum): Its chemical constituents, pharmacological activities and safety. Fitofarmaka Jurnal Ilmiah Farmasi 2019; 8, 23-29.
IG Arinton, P Samudro and S Soemohardjo. The Nepean dyspepsia index: Translation and validation in Indonesian language. Indonesian Journal of Gastroenterology, Hepatology, and Digestive Endoscopy 2006; 7, 38-41.
F Jaber, S Alsakarneh, A Beran, T Alsharaeh, AJ Salahat, A Abdelshafi, I Mohamed, W Johnson, K Elfert, M Jaber, M Almeqdadi, M Ahmed, H Ghoz, WK Clarkston and JH Helzberg. Impact of Helicobacter pylori eradication on clinical and laboratory parameters in non-alcoholic fatty liver disease patients: A systematic review and meta-analysis of randomized controlled trials. Current Medical Science 2025; 45, 1-10.
AFT Rossi, JC Contiero, F da Silva Manoel-Caetano, FE Severino and AE Silva. Up-regulation of tumor necrosis factor-α pathway survival genes and of the receptor TNFR2 in gastric cancer. World Journal of Gastrointestinal Oncology 2019; 11(4), 281-294.
Y Raza, M Mubarak, MY Memon and MS Alsulaimi. Update on molecular pathogenesis of Helicobacter pylori-induced gastric cancer. World Journal of Gastrointestinal Pathophysiology 2025; 16(2), 107052.
H Sulzbach De Oliveira, V Biolchi, HR Medeiros, DBG Jantsch, Ln De Oliveira Becker Delving, R Reckziegel, MI Goettert, IS Brum and A Pozzobon. Effect of Helicobacter pylori on NFKB1, p38α and TNF-α mRNA expression levels in human gastric mucosa. Experimental and Therapeutic Medicine 2016; 11, 2365-2372.
D Hefni, Y Herdalina and N Suharti. Activity of red ginger extract (Zingiber officinale var. rubrum) against interleukin. International Conference on Contemporary Science and Clinical Pharmacy 2023; 15, 1.
T Sari, S Trisnadi and I Isradji. The effect of red ginger extract on TNF-α and malondialdehyde serum level. International Journal of Multidisciplinary Research and Analysis 2022; 5, 12.
F Ayustaningwarno, G Anjani, AM Ayu and V Fogliano. A critical review of ginger’s (Zingiber officinale) antioxidant, anti-inflammatory, and immunomodulatory activities. Frontiers in Nutrition 2024; 11, 1364836.
HW Jung, CH Yoon, KM Park, HS Han and YK Park. Hexane fraction of Zingiberis rhizoma crudus extract inhibits the production of nitric oxide and proinflammatory cytokines in LPS-stimulated BV2 microglial cells via the NF-kappaB pathway. Food and Chemical Toxicology 2009; 47, 1190-1197.
Yuandani, R Jantan, N Jalil and S Husain. Immunomodulatory effects and mechanisms of the extracts and secondary compounds of Zingiber and Alpinia species: A review. Frontiers in Pharmacology 2023; 14, 1222195.
XH Li, KCY McGrath, S Nammi, AK Heather and BD Roufogalis. Attenuation of liver pro-inflammatory responses by Zingiber officinale via inhibition of NF-kappa B activation in high-fat diet-fed rats. Basic & Clinical Pharmacology & Toxicology 2012; 110, 238-244.
N Aryaeian, M Mahmoudi, F Shahram, S Poursani, F Jamshidi and H Tavakoli. The effect of ginger supplementation on IL2, TNFα, and IL1β cytokines gene expression levels in patients with active rheumatoid arthritis: A randomized controlled trial. Medical Journal of the Islamic Republic of Iran 2019; 33, 154.
D Amanda. 2022, Uji aktivitas imunomodulator ekstrak jahe merah (Zingiber officinale var. rubrum) terhadap tumor nekrosis faktor alfa mencit putih jantan (Mus musculus) dengan metode ELISA (in Indonesian). Bachelor’s Thesis. Universitas Andalas, Indonesia.
MY Song, SH Jeong, HJ Lee, H Kang and JY Kim. Steamed ginger extract exerts anti-inflammatory effects in Helicobacter pylori-infected gastric epithelial cells through inhibition of NF-κB. Journal of Cancer Prevention 2021; 26(4), 289-297
K Gaus, Y Huang, DA Israel, SL Pendland, BA Adeniyi and GB Mahady. Standardized ginger (Zingiber officinale) extract reduces bacterial load and suppresses acute and chronic inflammation in Mongolian gerbils infected with cagA+ Helicobacter pylori. Pharmaceutical Biology 2009; 47, 92-98.
L Zhong, H Tong, J Zhu and M Lv. Pharmacological effects of different ginger juices on the concurrent symptoms in animal models of functional dyspepsia: A comparative study. Food Science & Nutrition 2019; 7(7), 2205-2213.
W Saiah, H Halzoune, R Djaziri, K Tabani, EA Koceir and N Omari. Antioxidant and gastroprotective actions of butanol fraction of Zingiber officinale against diclofenac sodium-induced gastric damage in rats. Journal of Food Biochemistry 2018; 42, e12456.
M Morvaridzadeh, S Fazelian, S Agah, M Khazdouz, M Rahimlou, F Agh, E Potter, S Heshmati and J Heshmati. Effect of ginger (Zingiber officinale) on inflammatory markers: A systematic review and meta-analysis of randomized controlled trials. Cytokine 2020; 135, 155224.
B Yu, L Xiang, MP Peppelenbosch and GM Fuhler. Overlapping cytokines in H. pylori infection and gastric cancer: a tandem meta-analysis. Frontiers in Immunology 2023; 14, 1125658.
XC Yu, QQ Shao, J Ma, M Yu, C Zhang, L Lei, Y Zhou, WC Chen, W Zhang, XH Fang, YZ Zhu, G Wu, XM Wang, SY Han, PC Sun and SZ Ding. Family-based Helicobacter pylori infection status and transmission pattern in central China, and its clinical implications for related disease prevention. World Journal of Gastroenterology 2022; 28(28), 3706-3719.
XY Yuan, Y Zhang, X Zhao, A Chen and P Liu. IL-1β, an important cytokine affecting Helicobacter pylori-mediated gastric carcinogenesis. Microbial Pathogenesis 2023; 174, 105933.
H Yulma. 2022, Uji aktivitas imunomodulator ekstrak jahe merah (Zingiber officinale var. rubrum) terhadap IL-6 pada mencit putih jantan dengan metode ELISA (in Indonesian). Bachelor’s Thesis, Universitas Andalas, Indonesia.
F Zehsaz, N Farhangi and L Mirheidari. The effect of Zingiber officinale R. rhizomes (ginger) on plasma pro-inflammatory cytokine levels in well-trained male endurance runners. Central European Journal of Immunology 2014; 39(2), 174-180.
K Pázmándi, AG Szöllősi and T Fekete. The root causes behind the anti-inflammatory actions of ginger compounds in immune cells. Frontiers in Immunology 2024; 15, 1400956.
M Aliyu, FT Zohora, AU Anka, K Ali, S Maleknia, M Saffarioun and G Azizi. Interleukin-6 cytokine: An overview of the immune regulation, immune dysregulation, and therapeutic approach. International Immunopharmacology 2022; 111, 109130.
RP Semper, R Mejías-Luque, C Groß, F Anderl, A Müller, M Vieth, DH Busch, CP da Costa, J Ruland, O Groß and M Gerhard. Helicobacter pylori-induced IL-1β secretion in innate immune cells is regulated by the NLRP3 inflammasome and requires the Cag pathogenicity island. The Journal of Immunology 2014; 193(7), 3566-3576.
JH Kim, JH Chang, JH Yoon, SH Kwon, JH Bae and KS Kim. Gingerol suppresses interleukin-1β-induced MUC5AC gene expression in human airway epithelial cells. American Journal of Rhinology & Allergy 2009; 23, 385-391.
I Bischoff-Kont, T Primke, LS Niebergall, T Zech and R Fürst. Ginger constituent 6-shogaol inhibits inflammation- and angiogenesis-related cell functions in primary human endothelial cells. Frontiers in Pharmacology 2022; 13, 844767.
Y Song and J Chung. Zingerone-induced autophagy suppresses IL-1β production by increasing the intracellular killing of Aggregatibacter actinomycetemcomitans in THP-1 macrophages. Biomedicines 2023; 11, 2130.
LG Aregawi and C Zoltan. Ginger’s nutritional implication on gastrointestinal health. Clinical Nutrition Open Science 2025; 61, 1-13.
SJ Han, M Kim, VD D’Agati and HT Lee. 6-Shogaol protects against ischemic acute kidney injury by modulating NF-κB and heme oxygenase-1 pathways. American Journal of Physiology-Renal Physiology 2019; 317(3), F743-F756.
N Aryaeian, M Mahmoudi, F Shahram, S Poursani, F Jamshidi and H Tavakoli. The effect of ginger supplementation on IL2, TNFα, and IL1β cytokines gene expression levels in patients with active rheumatoid arthritis: a randomized controlled trial. Medical Journal of the Islamic Republic of Iran 2019; 33, 154.
H Maryam, S Azhar, MN Akhtar, A Asghar, F Saeed, H Ateeq, M Afzaal, N Akram, H Munir, W Anjum and MA Shah. Role of bioactive components of ginger in management of osteoarthritis: A review. International Journal of Food Properties 2023; 26, 1903-1913.
P Ballester, B Cerdá, R Arcusa, J Marhuenda, K Yamedjeu and P Zafrilla. Effect of ginger on inflammatory diseases. Molecules 2022; 27, 7223.
X Zhou, G Münch, H Wohlmuth, S Afzal, MH Kao, A Al-Khazaleh, M Low, D Leach and CG Li. Synergistic inhibition of pro-inflammatory pathways by ginger and turmeric extracts in RAW 264.7 cells. Frontiers in Pharmacology 2022; 13, 818166.
R Sotler, B Poljšak, R Dahmane, T Jukić, D Pavan Jukić, C Rotim, P Trebše and A Starc. Prooxidant activities of antioxidants and their impact on health. Acta Clinica Croatica 2019; 58, 726-736.
VS Chedea, LL Tomoiagǎ, ŞO Macovei, DC Mǎgureanu, ML Iliescu, IC Bocsan, AD Buzoianu, CM Voşloban and RM Pop. Antioxidant/pro-oxidant actions of polyphenols from grapevine and wine by-products - base for complementary therapy in ischemic heart diseases. Frontiers in Cardiovascular Medicine 2021; 8, 750508.
ME Giordano, R Caricato and MG Lionetto. Concentration dependence of the antioxidant and prooxidant activity of Trolox in HeLa cells: involvement in the induction of apoptotic volume decrease. Antioxidants 2020; 9, 1058.
K Jomova, D Valko, M Valko, S Ševčovičová, K Kuča, A Valent and Z Kovalčíková. Reactive oxygen species, toxicity, oxidative stress, and antioxidants: chronic diseases and aging. Archives of Toxicology 2023; 97, 2499-2574.
L Han, X Shu and J Wang. Helicobacter pylori-mediated oxidative stress and gastric diseases: A review. Frontiers in Microbiology 2022; 13, 811258.
G Yang, X Gao, L Jiang, X Sun, X Liu, M Chen, X Yao, Q Sun and S Wang. 6-Gingerol prevents MEHP-induced DNA damage in human umbilical vein endothelial cells. Human & Experimental Toxicology 2017; 36, 1177-1185.
S Wang, G Yang, L Jiang, C Zhang, F Xin, C Geng, X Sun and Y Yang. 6-Gingerol attenuates hydrogen peroxide-induced DNA damage in human umbilical vein endothelial cells. Food Science and Technology Research 2014; 20, 947-954.
B Ahmad, MU Rehman, I Amin, A Arif, S Rasool, SA Bhat, I Afzal, I Hussain, S Bilal and M ur Rahman Mir. A review on pharmacological properties of zingerone (4-(4-hydroxy-3-methoxyphenyl)-2-butanone). The Scientific World Journal 2015; 2015, 816364.
G Figueroa‐González, LI Quintas‐Granados, OD Reyes‐Hernández, IH Caballero‐Florán, SI Peña-Corona, H Cortés, G Leyva-Gómez, S Habtemariam and J Sharifi-Rad. Review of the anticancer properties of 6-shogaol: mechanisms of action in cancer cells and future research opportunities. Food Science & Nutrition 2024; 12(7), 4513-4533.
G Oboh, AJ Akinyemi and AO Ademiluyi. Antioxidant and inhibitory effect of red ginger (Zingiber officinale var. rubra) and white ginger (Zingiber officinale Roscoe) on Fe2+-induced lipid peroxidation in rat brain in vitro. Experimental and Toxicologic Pathology 2012; 64, 31-36.
M Afzal, I Kazmi, AM Quazi, SA Khan, A Zafar, FA Al-Abbasi, F Imam, KS Alharbi, SI Alzarea and N Yadav. 6-Shogaol attenuates traumatic brain injury-induced anxiety/depression-like behavior via inhibition of oxidative stress-influenced expressions of inflammatory mediators TNF-α, IL-1β, and BDNF: Insight into the mechanism. ACS Omega 2022; 7, 140-148.
M Rafeeq, HAS Murad, HM Abdallah and AM El-Halawany. Protective effect of 6-paradol in acetic acid-induced ulcerative colitis in rats. BMC Complementary Medicine and Therapies 2021; 21(1), 28.
P Baiubon, P Kunanusorn, P Khonsung, N Chiranthanut, A Panthong and C Rujjanawate. Gastroprotective activity of the rhizome ethanol extract of Zingiber simaoense YY Qian in rats. Journal of Ethnopharmacology 2016; 194, 571-576.
A Mestrovic, J Bozic, K Vukojevic and A Tonkic. Impact of different Helicobacter pylori eradication therapies on gastrointestinal symptoms. Medicina 2021; 57, 803.
A Shiotani, P Roy, H Lu and DY Graham. Helicobacter pylori diagnosis and therapy in the era of antimicrobial stewardship. Therapeutic Advances in Gastroenterology 2021; 14, 1-19.
H Matsumoto, A Shiotani and DY Graham. Current and future treatment of Helicobacter pylori infections. Helicobacter pylori in Human Diseases: Advances in Microbiology, Infectious Diseases and Public Health 2019; 11, 211-225.
A Gupta, S Shetty, S Mutalik, EM Mathew, A Jha, B Mishra, S Rajpurohit, G Ravi, M Saha and S Moorkoth. Treatment of H. pylori infection and gastric ulcer: Need for novel pharmaceutical formulation. Heliyon 2023; 9, e20406.
MN Bodagh, I Maleki and A Hekmatdoost. Ginger in gastrointestinal disorders: A systematic review of clinical trials. Food Science & Nutrition 2019; 7, 96-108.
Y Huang, MJ Patil, M Yu, P Liptak, BJ Undem, X Dong, G Wang and, S Yu. Effects of ginger constituent 6-shogaol on gastroesophageal vagal afferent C-fibers. Neurogastroenterology & Motility 2019; 31, e13585.
QQ Mao, XY Xu, SY Cao, RY Gan, H Corke, T Beta and HB Li. Bioactive compounds and bioactivities of ginger (Zingiber officinale Roscoe). Foods 2019; 8, 185.
A Kabiraj and R Deshmukh. A review on Chinese herbal medicine used as carminative. Research-Modern Chinese Medicine 2024; 11, 100409.
G Chantharangsikul, W Kitpati, N Soonthornchareonnon, A Sailasuta, A Itharat and W Suvitayavat. Mucus secretion stimulation: a mechanism in gastroprotective effect of Zingiber officinale. Thai Journal of Pharmaceutical Sciences 2016; 40, 1-8.
NS Karampour, A Arzi, A Rezaie, M Pashmforoosh and F Kordi. Gastroprotective effect of zingerone on ethanol-induced gastric ulcers in rats. Medicina 2019; 55(3), 64.
Supplementary Materials
Table S1 Baseline patient risk factor of H. pylori.
Parameter |
Frequency (n) / Percentage (%) |
p |
Alcohol Consumption |
|
0.749a |
None |
26 (86.7%) |
|
Present |
4 (13.3%) |
|
Smoking History |
|
0.427a |
Never |
24 (80.0%) |
|
Active Smoker |
5 (16.7%) |
|
Passive Smoker |
1 (3.3%) |
|
Comorbidities |
|
0.621a |
None |
21 (70.0%) |
|
Present |
9 (30.0%) |
|
Medication Use |
|
0.581a |
None |
17 (56.7%) |
|
Present |
13 (43.3%) |
|
Source of Drinking Water |
|
0.363a |
Bottled / Mineral Water |
10 (33.3%) |
|
Well / Boiled Water |
17 (56.7%) |
|
Mixed |
3 (10.0%) |
|
Note: aChi Square test. For all patients, comorbidities were dominated by hypertension and consumption of hypertension medication which was deemed not to have a good interaction with standard H. pylori therapy and ZOR supplementation.
Table S2 Normality test and homogenity test across all parameter.
Group |
Subjects |
p-value TNF-α |
Pre |
Post |
|
Normality Testa |
Homogenity Testb |
||||
Pre |
Post |
||||
K+ |
10 |
< 0.001 |
< 0.001 |
0.304 |
0.659 |
P1 |
10 |
0.007 |
0.003 |
|
|
P2 |
10 |
0.021 |
< 0.001 |
|
|
|
p-value IL-6 |
|
|||
Normality Testa |
Homogenity Testb |
||||
Pre |
Post |
||||
K+ |
10 |
<0.001 |
<0.001 |
0.341 |
0.178 |
P1 |
10 |
0.001 |
0.005 |
|
|
P2 |
10 |
0.201 |
0.016 |
|
|
|
p-value IL-1β |
|
|||
Normality Testa |
Homogenity Testb |
||||
Pre |
Post |
||||
K+ |
10 |
<0.001 |
<0.001 |
0.419 |
0.201 |
P1 |
10 |
0.001 |
0.003 |
|
|
P2 |
10 |
<0.001 |
<0.001 |
|
|
|
p-value MDA |
|
|||
Normality Testa |
Homogenity Testb |
||||
Pre |
Post |
||||
K+ |
10 |
0.007 |
0.004 |
0.103 |
0.245 |
P1 |
10 |
0.003 |
0.001 |
|
|
P2 |
10 |
0.424 |
0.548 |
|
|
|
p-value SF-NDI |
|
|||
Normality Testa |
Homogenity Testb |
||||
Pre |
Post |
||||
K+ |
10 |
0.002 |
0.364 |
0.587 |
0.306 |
P1 |
10 |
0.055 |
0.135 |
|
|
P2 |
10 |
0.010 |
0.015 |
|
|
Note: aShapiro-Wilk; bLevene’s
Table S3 Post Hoc Dunn test.
Group |
Group |
p-value IL-1β |
p-value SF-NDI |
K+ Pre-intervention |
P1 Pre-intervention |
0.720 |
< 0.001* |
|
P2 Pre-intervention |
0.089 |
0.379 |
|
K+ Post-intervention |
0.609 |
< 0.001* |
|
P1 Post-intervention |
0.609 |
0.170 |
|
P2 Post-intervention |
0.063 |
< 0.001* |
P1 Pre-intervention |
P2 Pre-intervention |
0.017* |
< 0.001* |
|
K+ Post-intervention |
0.878 |
0.542 |
|
P1 Post-intervention |
0.878 |
< 0.001* |
|
P2 Post-intervention |
0.134 |
0.472 |
P2 Pre-intervention |
K+ Post-intervention |
0.025* |
< 0.001* |
|
P1 Post-intervention |
0.025* |
0.621 |
|
P2 Post-intervention |
0.370 |
0.001* |
K+ Post-intervention |
P1 Post-intervention |
1.000 |
0.002* |
|
P2 Post-intervention |
0.179 |
0.913 |
P1 Post-intervention |
P2 Post-intervention |
0.179 |
0.003* |
Note: * (significant, p < 0.05)
Table S4 SF-NDI subdomain evaluation.
SF-NDI Subdomain |
Group |
Time |
Mean ± SD |
Min – Max |
Median |
pa |
Tension/anxiety (P1 - P2) |
K+ |
Pre |
3.85 ± 0.58 |
2 – 5 |
4.0 |
0.001 |
Post |
2.15 ± 0.35 |
1 – 3 |
2.0 |
|||
P1 |
Pre |
3.80 ± 0.30 |
3 – 4 |
3.5 |
0.001 |
|
Post |
2.40 ± 0.36 |
2 – 3 |
2.0 |
|||
P2 |
Pre |
3.45 ± 0.76 |
2 – 5 |
4.0 |
0.001 |
|
Post |
2.30 ± 0.47 |
1 – 3 |
2.0 |
|||
Activities (P3 - P4) |
K+ |
Pre |
3.90 ± 0.62 |
2 – 5 |
4.0 |
0.001 |
Post |
2.30 ± 0.58 |
1 – 4 |
2.0 |
|||
P1 |
Pre |
3.20 ± 0.30 |
3 – 4 |
3.0 |
0.001 |
|
Post |
2.00 ± 0.47 |
1 – 3 |
2.0 |
|||
P2 |
Pre |
3.30 ± 0.66 |
2 – 5 |
3.0 |
< 0.001 |
|
Post |
2.00 ± 0.47 |
1 – 3 |
2.0 |
|||
Eating/drinking (P5 - P6) |
K+ |
Pre |
3.65 ± 0.54 |
2 – 5 |
4.0 |
0.001 |
Post |
1.85 ± 0.53 |
1 – 3 |
2.0 |
|||
P1 |
Pre |
3.40 ± 0.36 |
3 – 4 |
3.5 |
0.001 |
|
Post |
1.90 ± 0.70 |
1 – 4 |
2.0 |
|||
P2 |
Pre |
3.40 ± 0.49 |
2 – 4 |
3.0 |
<0.001 |
|
Post |
2.40 ± 0.49 |
2 – 4 |
2.0 |
|||
Illness concern (P7 - P8) |
K+ |
Pre |
3.15 ± 0.99 |
1 – 5 |
3.0 |
0.001 |
Post |
1.40 ± 0.57 |
1 – 3 |
1.0 |
|||
P1 |
Pre |
2.90 ± 0.47 |
1 – 4 |
2.5 |
0.001 |
|
Post |
1.65 ± 0.38 |
1 – 2 |
1.0 |
|||
P2 |
Pre |
3.00 ± 0.88 |
1 – 5 |
3.0 |
0.001 |
|
Post |
1.35 ± 0.35 |
1 – 2 |
1.0 |
|||
Work/study (P9 - P10) |
K+ |
Pre |
3.70 ± 0.66 |
2 – 5 |
4.0 |
0.001 |
Post |
1.80 ± 0.45 |
1 – 3 |
2.0 |
|||
P1 |
Pre |
3.80 ± 0.45 |
3 – 5 |
3.5 |
0.001 |
|
Post |
2.30 ± 0.47 |
2 – 4 |
2.0 |
|||
P2 |
Pre |
3.40 ± 0.49 |
2 – 4 |
4.0 |
< 0.001 |
|
Post |
2.20 ± 0.30 |
2 – 3 |
2.0 |
Note: aPair T Test