
Trends
Sci.
2026;
23(7):
12681
Development of Microemulsions Containing Ruean-Mhoon-Nok Remedy Extract and Evaluation for Skin Irritation in Healthy Volunteers
Atchanika Taingthum1, Panlop Chakkavittumrong2,
Puritat Kanokkangsadal1 and Intouch Sakpakdeejaroen1,*
1Department of Applied Thai Traditional Medicine, Faculty of Medicine, Thammasat University,
Pathum Thani 12120, Thailand
2Department of Internal Medicine, Faculty of Medicine, Thammasat University, Pathum Thani 12120, Thailand
(*Corresponding author’s e-mail: [email protected])
Received: 17 November 2025, Revised: 18 December 2025, Accepted: 28 December 2025, Published: 5 March 2026
Abstract
Ruean-Mhoon-Nok (RMN), a Thai traditional remedy, has been used topically to treat skin inflammatory disorders, including dermatitis and psoriasis. Previously, the ethanolic extract of RMN and its bioactive compound, hydroxychavicol, demonstrated promising in vitro anti-inflammatory activity by inhibiting NO and IL-6 production. Therefore, this study aimed to develop a stable microemulsion capable of enhancing skin penetration of the RMN extract and to evaluate its potential for skin irritation in healthy volunteers. Two RMN microemulsion formulations were prepared, consisting of an oil phase (Capryol® 90) combined with surfactants (Polyglyceryl-4 caprate or PEG-6 caprylic/capric glycerides) and a co-surfactant (Transcutol® P) at a weight ratio of 1:2. These microemulsions had mean droplet sizes of less than 100 nm and carried a negative surface charge. Furthermore, they remained stable after nine heating-cooling storage cycles. In vitro skin permeation studies showed that the RMN microemulsions were able to penetrate the skin and release hydroxychavicol, achieving a maximum cumulative permeation of 26% over 8 h. In healthy volunteers, the RMN microemulsions were well tolerated, showing no signs of irritation at the maximum dose of 3% RMN extract. In conclusion, the RMN microemulsions are highly promising formulations that warrant further investigation for their efficacy and safety in clinical applications.
Keywords: Ruean-Mhoon-Nok remedy, Thai traditional medicine, Dermatitis, Microemulsion, Skin irritation test
Introduction
Dermatitis is a common skin condition characterized by inflammation, redness, and itching, resulting from dysfunction of the skin barrier. It can affect people of all ages and may be triggered by both endogenous (e.g., genetic and immune system) and exogenous (e.g., environmental, allergenic, and pathogenic) factors [1]. Moreover, this inflammatory condition can cause significant morbidity and impair patients’ quality of life [2]. Treatment options typically include topical corticosteroids, moisturizers, and lifestyle modifications to help manage and prevent recurrent symptoms. However, topical corticosteroids may cause adverse effects, particularly with long-term use, leading to skin atrophy (thinning), fragility, striae, and other complications [3].
In recent years, Asian herbal and traditional medicines have gained considerable attention as alternative options for the treatment of dermatitis due to their potential efficacy and safety [4]. In Thai traditional medicine, Ruean-Mhoon-Nok (RMN) remedy, an herbal medicine listed in the National Thai Traditional Medicine Formulary (2021 Edition), has been utilized as a topical preparation for the management of cutaneous inflammatory disorders, including dermatitis and psoriasis [5]. This remedy consists of nine plant ingredients in equal proportions: the leaves of Casearia grewiifolia Vent., the leaves of Crateva religiosa G. Forst., the leaves of Crateva adansonii DC., the aerial parts of Pouzolzia zeylanica (L.) Benn., the aerial parts of Gonostegia pentandra (Roxb.) Miq., the rhizomes of Alpinia galanga (L.) Willd., the leaves of Piper wallichii (Miq.) Hand.-Mazz., the leaves of Datura metel L., and the leaves of Persicaria chinensis (L.) H. Gross. Several studies have reported that some of these components exhibit anti-inflammatory, anti-allergic, antipsoriatic, and antibacterial activities [6-9]. Furthermore, our recent study revealed promising anti-inflammatory activity of the 95% ethanolic extract of the RMN remedy and its bioactive compound, hydroxychavicol, particularly through inhibition of nitric oxide and interleukin-6 production. This extract also demonstrated in vitro antioxidant activity with high phenolic and flavonoid contents [10], further supporting its use in managing inflammatory skin conditions.
The traditional method of using the RMN remedy involves creating a paste by mixing powdered ingredients with liquor and applying it directly to the affected skin areas. However, the main disadvantages of this conventional method are its impracticality and potential concerns regarding efficacy (e.g., low skin penetration) and safety (e.g., skin irritation), which may hinder the wider use of the RMN remedy. To overcome these limitations, we hypothesize that developing a topical drug delivery system capable of enhancing skin penetration of the RMN extract could improve its efficacy while reducing the risk of skin irritation
Microemulsions (ME) are thermodynamically stable, transparent, isotropic liquid mixtures composed of oil, water, surfactants, and often a co-surfactant. These systems can spontaneously self-assemble upon mixing the components, without the need for high shear conditions [11]. Moreover, the unique properties of microemulsions - such as their ability to incorporate both hydrophilic and lipophilic compounds, low viscosity, and nanometer-sized droplets in the range of 20 - 200 nm - make them a promising and versatile topical drug delivery system for improving skin penetration and reducing irritation [12]. For example, a previous study reported that a stable microemulsion loaded with catechin enhanced skin penetration and deposition of the compound without causing irritation in vivo [13]. Similarly, a microemulsion containing Pouteria macrophylla fruit extract improved skin deposition through pig skin and enhanced the depigmenting effect of the extract [14].
The objectives of this study were therefore to (1) develop and characterize microemulsions containing RMN extract, (2) assess their physical stability and in vitro skin permeation, and (3) evaluate their potential for skin irritation in healthy volunteers.
Materials and methods
Chemicals and reagents
Acetonitrile, methanol, and 95% ethanol were obtained from RCI Labscan (Bangkok, Thailand). Propylene glycol monocaprylate (Capryol® 90) and diethylene glycol monoethyl ether (Transcutol® P) were supplied by Gattefossé (Saint-Priest, France). White petrolatum, isopropyl myristate (IPM), polyglyceryl-4 caprate, PEG-6 caprylic/capric glycerides, Tween 20, propylene glycol, and polyethylene glycol 400 (PEG-400) were obtained from Chanjao Longevity Co., Ltd. (Bangkok, Thailand). Phosphate-buffered saline (PBS), and all other chemicals and reagents that are not specifically mentioned below were purchased from Merck Millipore (Darmstadt, Germany).
Plant materials and preparation of RMN crude extract
The plant ingredients of the RMN remedy were purchased from several herbal shops in Thailand. The rhizomes of A. galanga (TTM 0005447) and the leaves of P. wallichii (TTM 1000663) were obtained from Nakhon Pathom. The leaves of C. grewiifolia (TTM 1000658) were obtained from Chachoengsao, and the leaves of C. adansonii (TTM 1000660) were obtained from Chon Buri. The leaves of D. metel (TTM 0005448) were obtained from Ayutthaya. The leaves of C. religiosa (TTM 1000659), the leaves of P. chinensis (TTM 1000664), the aerial parts of P. zeylanica (TTM 0005449), and the aerial parts of G. pentandra (TTM 1000662) were obtained from Samut Sakhon. All plant specimens were identified by an herbal medicine expert and deposited at the herbarium of Thai Traditional Medicine Research Institute, Department of Thai Traditional and Alternative Medicine, Bangkok, Thailand.
To prepare the RMN crude extract, all plant components were thoroughly cleaned, dried, and pulverized into coarse powder. The RMN remedy was prepared by mixing each plant powder in equal proportions, followed by extraction through maceration with 95% ethanol for 3 days. The extract was filtered through Whatman No. 1 filter paper, and the maceration process was repeated twice more, for a total of 3 times. Finally, the filtrates were combined, concentrated under reduced pressure using a rotary evaporator, and dried in a vacuum dryer. The extract was stored at −20 °C for further experiments.
Determination of hydroxychavicol in RMN extract
The amount of hydroxychavicol, the active compound in the RMN extract, was determined using HPLC (Thermo Fisher Scientific, MA, USA). Briefly, the extract was dissolved in methanol (10 mg/mL) and filtered through a 0.45 μm membrane filter. The sample was then injected into a reversed-phase column, ZORBAX Eclipse XDB-C18 (4.6×250 mm, 5 µm), with a guard column of the same material. The mobile phases consisted of water (A) and acetonitrile (B) with gradient elution as follows: 0 min, 90:10; 10 min, 90:10; 30 min, 70:30; 45 min, 60:40; 53 min, 95:5; 60 min, 95:5; 60.1 min, 90:10; 65 min, 90:10. The flow rate was 1 mL/min, with UV detection at a wavelength of 210 nm. The operating temperature was maintained at room temperature. Additionally, a calibration curve of standard hydroxychavicol was prepared with concentrations ranging from 10 to 400 µg/mL and analyzed in triplicate. The content of hydroxychavicol in the RMN extract was calculated by correlating the peak area with the calibration curve of standard hydroxychavicol.
Screening of microemulsion components
Screening of microemulsion components involves selecting appropriate oils, surfactants, and co-surfactants to achieve high solubility for the RMN extract. Briefly, an excess amount of RMN extract was added to 1 g of selected vehicles. The mixtures were subsequently agitated using a rotator mixer at 30 ± 2 °C for 48 h to achieve drug equilibrium solubility. The supernatants were then collected by centrifugation at 10,000 rpm for 30 min. The amount of hydroxychavicol dissolved in the vehicles was quantified using HPLC. The results were expressed as a percentage of the relative solubility of hydroxychavicol, using 95% ethanol as the comparison standard. Components that delivered high hydroxychavicol solubility were selected to construct pseudo-ternary phase diagrams of the microemulsions.
Construction of pseudo-ternary phase diagrams
Pseudo-ternary phase diagrams were constructed using the aqueous titration method to identify the appropriate components and their ratios, providing a large microemulsion region. Briefly, a mixture of the chosen surfactant and co-surfactant (Smix) was prepared in weight ratios of 1:1 and 1:2. Different mixtures of the oil phase and Smix were then accurately weighed in a glass vial at ratios of 1:9, 2:8, 3:7, 4:6, 5:5, 6:4, 7:3, 8:2, and 9:1. Titration was performed by continuously adding double-distilled water dropwise into the mixtures under magnetic stirring until the transparent and homogeneous microemulsions turned into cloudy emulsions. At this point, the percentages of oil, Smix, and water were calculated to construct pseudo-ternary phase diagrams and identify the microemulsion region using commercial software (CHEMIX School 12.2, Bergen, Norway).
Preparation of microemulsion
The appropriate oil, Smix, and water weight ratios used to prepare oil-in-water (O/W) microemulsions were selected based on the pseudo-ternary phase diagrams. Briefly, the chosen quantities of oil and Smix were mixed in a glass vial. The RMN extract (1% w/w) was dissolved in the mixture under moderate stirring. Subsequently, the aqueous phase was slowly added to the system and continuously stirred overnight at room temperature to obtain a clear, dark green microemulsion. Base (blank) microemulsions were prepared in the same manner, but without the RMN extract. All samples were stored in well-closed containers at 4 °C for further experiments.
Characterization of microemulsion
Microemulsions were visually examined for color, homogeneity, and phase separation. Droplet size, polydispersity index (PDI), and zeta potential were analyzed using dynamic light scattering (DLS) and electrophoretic light scattering (ELS) techniques with a Zetasizer® Pro (Malvern Panalytical, Malvern, UK). In addition, the viscosity of the microemulsions was determined using a DVNext cone/plate rheometer (AMETEK Brookfield, MA, USA) at 60 rpm with a cone spindle (CPA-40Z) at 25 °C, while the pH was measured using an Orion 2-Star™ benchtop pH meter (Thermo Fisher Scientific, MA, USA).
Stability study of microemulsion
The stability of microemulsions was assessed under extreme temperature changes using heating-cooling cycles [15]. Three different batches of microemulsions were placed in glass vials and kept in a refrigerator at 4 °C for 24 h, followed by incubation in a hot-air oven at 45 °C for another 24 h. This step was repeated for a total of nine consecutive cycles. The droplet size, zeta potential, viscosity, and pH of microemulsions were examined at specific cycles (cycles 0, 3, 6, and 9).
In vitro permeation study of RMN microemulsion
To confirm that the RMN microemulsion could penetrate the skin, an in vitro skin permeation study of hydroxychavicol, the active compound in the RMN extract, was performed using a Franz diffusion cell with an effective diffusion area of 1.77 cm2. A synthetic Strat-M® membrane (Merck Millipore, Darmstadt, Germany), designed to mimic human skin, was selected as an alternative non-animal-based model for transdermal diffusion testing [16]. Briefly, the receiver compartment was filled with 12 mL of 40% ethanol in PBS (pH 7.4) and maintained at 32 ± 1 °C under continuous stirring at 600 rpm. The RMN microemulsion was placed in the donor compartment on top of the shiny side of the Strat-M® membrane. At specific time intervals (15, 30 and 45 min, 1, 2, 3, 4, 5, 6, 7 and 8 h), 500 µL of the receptor solution was withdrawn and replaced with an equal volume of fresh medium. The amount of hydroxychavicol in the samples was quantified using HPLC and reported as a percentage of cumulative drug permeation.
Skin irritation and allergic testing of RMN microemulsion in human volunteers
Ethics consideration
The study protocol for the skin irritation and allergic testing of the RMN microemulsion in healthy volunteers (clinical trial phase I) was reviewed and approved by the Human Research Ethics Committee of Thammasat University (Faculty of Medicine), Thailand, prior to commencement of the study (Cert. No. 238/2565; Project No. MTU-EC-TM-1-185/65).
Human volunteers
A total of 36 healthy volunteers aged between 18 and 60 years were examined and recruited by a dermatologist to assess skin irritation [17]. All subjects provided informed consent prior to enrollment. Participants were excluded if they had skin diseases or lesions, tattoos, scars, or burns on the tested area; a history of hyperallergic reactions to the RMN remedy, microemulsion components, or medical adhesive tapes; were pregnant or breastfeeding; or had used antihistamines, corticosteroids, or immunosuppressants within the 2 weeks preceding the study.
Study design
The study was an open-label controlled trial conducted at Thammasat University Hospital, Thammasat University, Pathum Thani, Thailand. The human closed patch test method was used to investigate skin irritation of the RMN extract (0.5%, 1%, and 3% w/w) in either microemulsions or white petrolatum (WP), as well as base (blank) microemulsions and white petrolatum alone (negative control). All tested samples were loaded onto Finn chambers® (8 mm, SmartPractice®, AZ, USA) and applied to the upper back of the volunteers using patch fixation tapes (Tegaderm™ film and Micropore™ tape, 3M Health Care, MN, USA) for 48 h. After removing the patches, skin irritation and allergic reactions were observed and graded twice, at 30 min and 24 h, by a dermatologist using the grading criteria of skin reactions according to the Cosmetic, Toiletry, and Fragrance Association (CTFA) guidelines. The grade was then calculated as a score of skin response using the specified equation below. The scores in each case were compared after converting the values to standardized z-scores (Table 1). Tested samples with z-scores ≤ 1.0 were considered safe for primary skin irritation of the leave-on product [18]. Calculation of score of skin response:
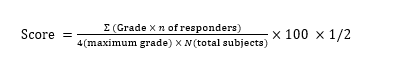
Table 1 Grading criteria for skin reactions by CTFA guideline and human primary irritation index for cosmetic products by human patch test.
Statistical analysis
All experiments were performed in triplicate and expressed as mean ± standard deviation (SD). Statistical analysis was conducted using paired-sample t-test, an independent samples t-test or a 2-way analysis of variance (ANOVA), depending on the experimental design. Differences were considered statistically significant at p < 0.05.
Results and discussion
HPLC analysis of RMN extract
The HPLC chromatogram showed that hydroxychavicol is a major compound in the RMN extract, eluting at a retention time of 33.79 min (Figure 1). The calibration curve of standard hydroxychavicol was linear over the concentration range used, described by the equation y = 48,253x – 364,018, with a coefficient of determination (R²) of 0.9995. The RMN extract contained 23.21 ± 0.21 mg of hydroxychavicol per gram of extract (2.32 ± 0.02% w/w). Therefore, hydroxychavicol was selected as the analytical marker for the RMN extract.
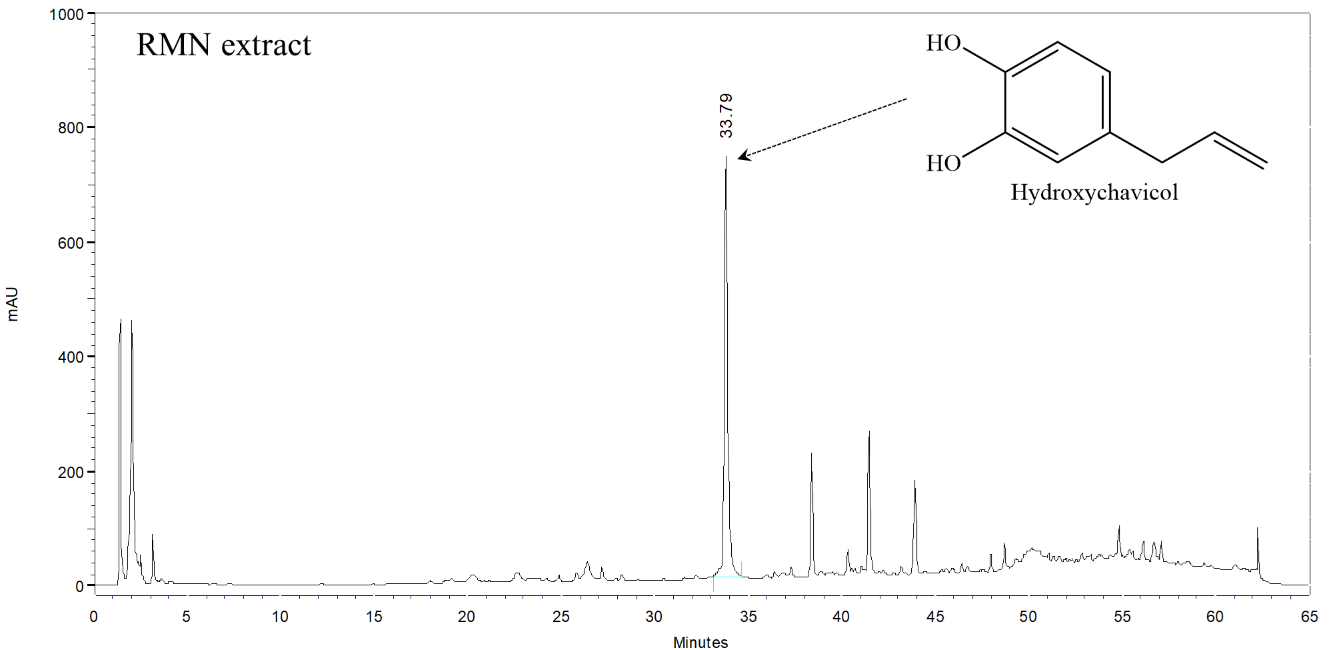
Figure 1 HPLC chromatogram of RMN extract at a wavelength of 210 nm.
Solubility study of RMN extract
The solubility of the RMN extract was assessed in different oils, surfactants, and co-surfactants to identify appropriate components for formulating a stable microemulsion system with a high hydroxychavicol content. The results indicated that IPM exhibited the highest solubility for hydroxychavicol (relative solubility of 99.74 ± 1.16%), followed by Capryol® 90 (relative solubility of 95.75 ± 1.77%), as shown in Table 2. In contrast, liquid paraffin had the lowest solubility among the oils (relative solubility of 28.27 ± 0.36%), likely due to its lower polarity compared with the other oils [19]. Consequently, IPM and Capryol® 90 were selected as the oil phase for the microemulsion formulation because of their high hydroxychavicol solubility, which helps maintain the drug in a solubilized form [20]. In addition, these oils have been used as skin penetration enhancers in topical and transdermal formulations [21,22].
In terms of surfactants, they play a major role in the formation of microemulsions by reducing the interfacial tension between oil and water. Surfactants with the hydrophilic-lipophilic balance (HLB) value between 8 and 16 are ideal for forming stabilized O/W microemulsions [23]. Moreover, nonionic surfactants are widely used in the design of microemulsion formulations due to their low skin irritation and toxicity, as well as their high compatibility with other components [24]. Among the nonionic surfactants tested, polyglyceryl-4 caprate (relative solubility of 98.53 ± 1.92%) and PEG-6 caprylic/capric glycerides (relative solubility of 96.60 ± 1.29%) were the most effective solubilizers for hydroxychavicol, in contrast to Tween 20 (relative solubility of 76.55 ± 1.00%). Hence, polyglyceryl-4 caprate (HLB value of 14-14.5) and PEG-6 caprylic/capric glycerides (HLB value of 12.5 - 14) were selected as the surfactants for the microemulsion formulation.
For co-surfactants, they increase the flexibility of surfactant film and lower the interfacial tension between oil and water phases, promoting the formation and stabilization of microemulsions [25]. In this study, Transcutol® P was selected for the microemulsion formulation because it exhibited the highest solubility of hydroxychavicol, with a relative solubility of 95.39 ± 0.56%. Moreover, Transcutol® P is frequently used in the preparation of transdermal drug delivery systems as it can interact with the intercellular lipid matrix of the stratum corneum, thus allowing drugs to penetrate the skin [26]. Similar findings were observed with PEG-400 (relative solubility of 88.31 ± 1.24%) and propylene glycol (relative solubility of 87.01 ± 1.44%), although both were lower than Transcutol® P.
Table 2 Solubility of hydroxychavicol in various components of microemulsion
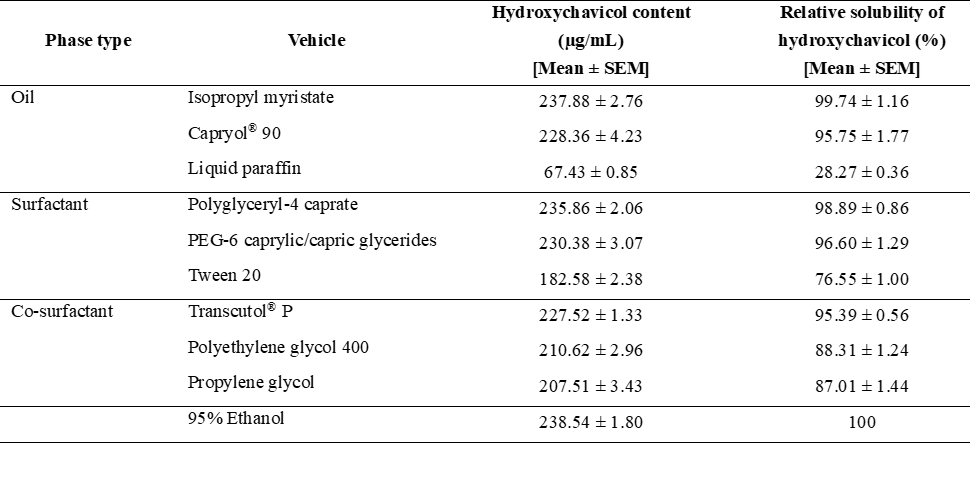
Data represent mean ± SD (n = 3), The hydroxychavicol content of RMN extract in 95% ethanol was used as a reference value for 100% relative solubility of hydroxychavicol.
Pseudo-ternary phase diagrams
Based on the solubility results, the components that yielded a relative solubility of hydroxychavicol greater than 90% were selected to construct the pseudo-ternary phase diagram to identify the microemulsion region and the appropriate ratio of each component. Isopropyl myristate and Capryol® 90 were selected as the oil phase. Polyglyceryl-4 caprate and PEG-6 caprylic/capric glycerides were used as surfactants, combined with Transcutol® P as a co-surfactant. The mixture of surfactant and co-surfactant (Smix) was prepared at weight ratios of 1:1 and 1:2 (Figure 2).
The formation of microemulsion regions is significantly influenced by the type and properties of the oils used [27]. Preliminary studies revealed that the use of Capryol® 90 as the oil phase, combined with different types of Smix at a weight ratio of 1:1 (Figures 2(C) and 2(D)), provided a larger microemulsion region than that observed with IPM (Figures 2(A) and 2(B)). This is because Capryol® 90, a medium-chain fatty acid ester (C8), has amphiphilic properties that enhance microemulsion stability, particularly in systems with high water content (O/W microemulsions). In contrast, IPM, a fatty acid ester (C14), is less polar than Capryol® 90 and often requires higher surfactant concentrations to achieve stable systems, especially when the water phase is dominant [28].
The ratio of surfactant to co-surfactant is also a critical factor influencing the phase behavior and stability of microemulsions. A balanced Smix can lead to the formation of a large and stable microemulsion region [25], as well as clinically proven safe and effective products [29]. Our results showed that all types of Smix at a weight ratio of 1:1 (Figures 2(C) and 2(D)) efficiently formed a large and stable microemulsion region, similar to Smix at a weight ratio of 1:2 (Figures 2(E) and 2(F)). Based on this finding, the optimal ratio of surfactant to co-surfactant of 1:2 was chosen for further studies due to the lower surfactant content, which may reduce skin irritation [30].
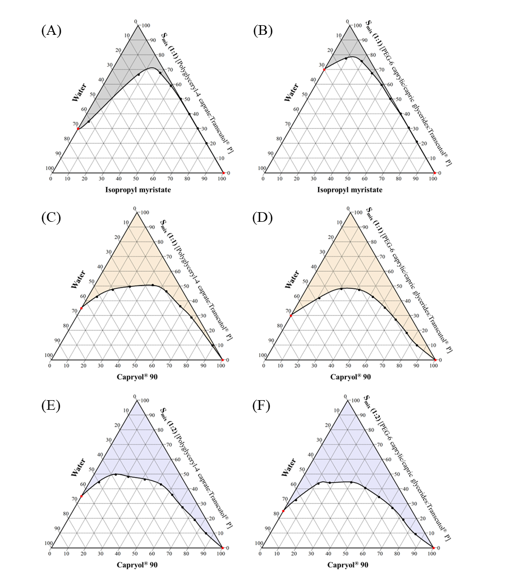
Figure 2 Pseudo-ternary phase diagrams of microemulsions with different oils and Smix ratios.
Preparation and characterization of RMN microemulsions
Based on the microemulsion regions obtained from the pseudo-ternary phase diagrams, 2 optimized O/W microemulsion formulations were successfully prepared using different types of surfactants: Polyglyceryl-4 caprate (ME1) and PEG-6 caprylic/capric glycerides (ME2). These surfactants were mixed with the co-surfactant (Transcutol® P) at a weight ratio of 1:2. The concentrations of oil (Capryol® 90), Smix, and RMN extract were fixed at 5%, 50%, and 1% w/w, respectively. Base (blank) microemulsions were prepared in the same manner, but without the RMN extract. The physicochemical properties of all microemulsion formulations are summarized in Table 3.
The visual appearance of RMN-loaded microemulsions was a dark green, transparent, homogeneous solution, whereas their base formulations were clear and transparent. Droplet size is an important characteristic of microemulsions. In this study, all microemulsion formulations produced droplets below 100 nm with a narrow and uniform size distribution (PDI < 0.2), allowing for enhanced drug penetration through the skin barrier [27]. Specifically, the smallest droplet sizes were found in the ME2 formulations (47.3 ± 0.8 nm for ME2-RMN and 37.9 ± 0.6 for ME2-Base), which used PEG-6 caprylic/capric glycerides (HLB value of 12.5 - 14) as a surfactant. When the surfactant was changed to polyglyceryl-4 caprate (HLB value of 14 - 14.5), the ME1 formulations became larger (60.7 ± 0.6 nm for ME1-RMN and 57.5 ± 0.7 for ME1-Base), although there was only a slight increase in mean droplet size when loading the RMN extract. The concentration of surfactant was still sufficient to maintain the internal phase droplet size [31]. The differences in droplet sizes between ME1 and ME2 formulations may be explained by the effect of different HLB values, surfactant type, and the charge of the interfacial layer surrounding the oil droplets [32].
Regarding zeta potential, the surface charge of microemulsions can significantly affect their stability. Microemulsions with a high positive or negative zeta potential (typically > +30 mV or < –30 mV) are generally considered highly stable colloidal dispersions with low aggregation [33]. The results showed that all microemulsion formulations had a negative surface charge, with zeta potentials ranging from −19.8 ± 0.3 to −48.1 ± 1.2 mV, indicative of colloidal stability. The significant decrease in zeta potential observed in RMN microemulsions may be attributed to negatively charged compounds present in the extract.
For pH, the ME1 formulations exhibited acidic values (5.40 ± 0.02 for ME1-RMN and 5.49 ± 0.02 for ME1-Base), while ME2 formulations were less acidic (5.87 ± 0.02 for ME2-RMN and 6.19 ± 0.07 for ME2-Base). Since ME1 and ME2 formulations used different surfactants, the variation in pH values is most likely influenced by the surfactant itself. Nevertheless, the RMN microemulsions possess pH values within the acceptable range of 4 - 6 for topical products [34].
In terms of viscosity, all microemulsion formulations exhibited low viscosity, ranging from 10.57 ± 0.23 to 15.20 ± 0.17 centipoise (cP). Lower-viscosity microemulsions tend to enhance skin permeation by facilitating drug diffusion through the skin layers. This property may also be beneficial for rapid drug release, which can provide immediate therapeutic effects [35].
Table 3 Physiochemical properties of RMN microemulsions.
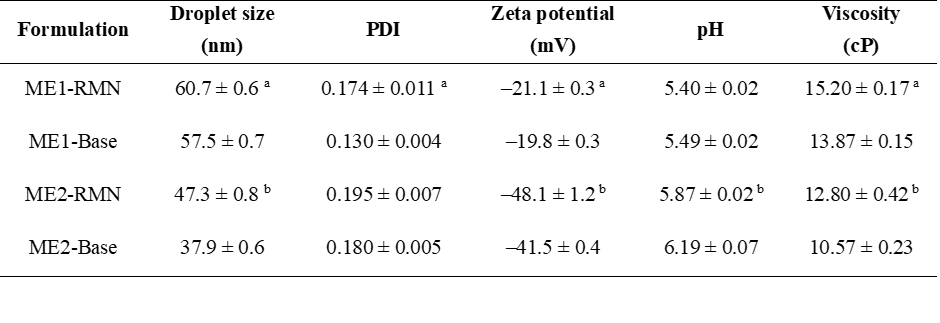
ME1 used polyglyceryl-4 caprate as a surfactant, ME2 used PEG-6 caprylic/capric glycerides as a surfactant. Data represent mean ± SD of each microemulsion formulation prepared from 3 independent batches, Data were analyzed by an independent samples t-test. (a) Significant difference is when p < 0.05 compared with the ME1-Base (n = 3). (b) Significant difference is when p < 0.05 compared with the ME1-Base (n = 3).
Stability of RMN microemulsions
Microemulsions are known for their thermodynamic stability, yet they can also exhibit phase separation under various factors, such as surfactant concentration, salinity, and temperature changes [36]. After 9 heating-cooling cycles, the visual appearance of both RMN and base microemulsion formulations remained homogeneous, without phase separation or drug precipitation. The mean droplet size of all microemulsion formulations also remained stable, with a uniform size distribution (droplet size at 9 cycles ranging from 38.6 ± 0.8 to 60.2 ± 0.8 nm and PDI ranging from 0.139 ± 0.006 to 0.187 ± 0.021) over the duration of the experiment (Figure 3(A)), and still within the microemulsion range (< 200 nm) [37]. This stability may be attributed to the surfactants, which reduce interfacial tension between oil and water, thereby reducing droplet size (increasing surface area) and stabilizing the system [19].
Zeta potential is another crucial parameter for determining the thermodynamic stability of microemulsions. A higher absolute value of zeta potential generally indicates greater stability due to increased electrostatic repulsion between droplets, which prevents coalescence and aggregation [11]. In this study, the zeta potential of ME2 formulations showed a slight decrease after nine cycles (Figure 3(B)), whereas ME1 formulations exhibited a slight increase, but maintained a negative charge within a highly stable range [33].
The pH of all microemulsion formulations decreased slightly due to the impact of temperature changes (pH at 9 cycles ranging from 4.97 ± 0.02 to 5.68 ± 0.03) (Figure 3(C)). Nevertheless, their pH values remained close to skin pH, suitable for topical use [34]. In terms of viscosity, all microemulsion formulations exhibited low viscosity (viscosity at 9 cycles ranging from 11.50 ± 0.53 to 14.97 ± 0.42 cP) without substantial change (Figure 3(D)), facilitating drug diffusion through the vehicle compared with higher-viscosity systems [35].
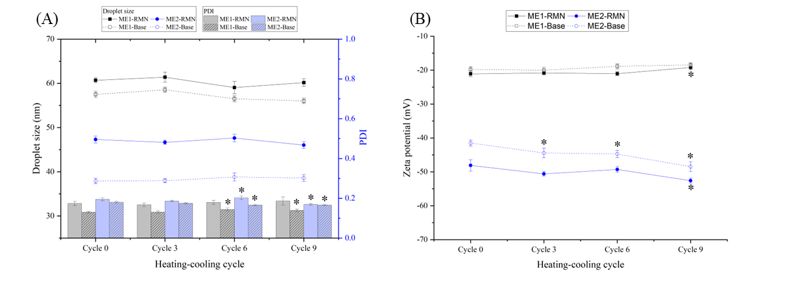
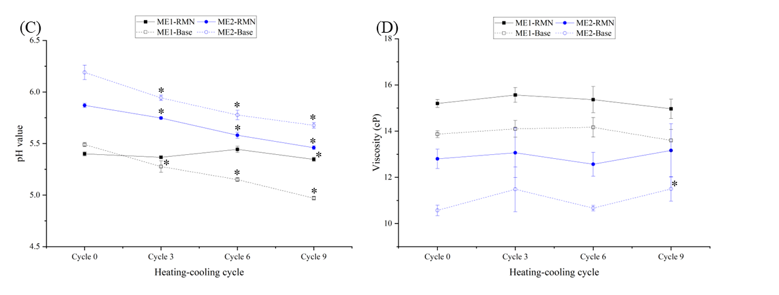
Figure 3 Stability of RMN microemulsions stored under nine heating-cooling cycles. Data represent mean ± SD (n = 3). (*) p < 0.05 when compared with cycle 0 within each sample (paired-samples t-test).
In vitro permeation profile of RMN microemulsion
The permeability of RMN microemulsions was assessed in vitro using Franz diffusion cells. The amount of hydroxychavicol was analyzed by HPLC and reported as percentage cumulative drug permeation (Figure 4). Both ME1-RMN and ME2-RMN formulations showed similar permeation profiles of hydroxychavicol. An initial high permeation of hydroxychavicol (approximately 12% - 15%) was observed within the first 15 min, followed by sustained permeation, reaching a maximum cumulative permeation of 24% - 26% over an 8-hour period. Specifically, the highest permeation of hydroxychavicol was observed with ME2-RMN, with a cumulative permeation of 26.04 ± 1.07%, whereas ME1-RMN achieved 24.07 ± 0.12% through the Strat-M® membrane after 8 h. This may be attributed to the smaller droplet size of ME2-RMN compared to ME1-RMN, allowing easier navigation through the intercellular spaces of the stratum corneum [27]. Similarly, the cumulative permeability of a curcumin nanoemulsion prepared with a surfactant of HLB 11 (droplet size 84 nm) was 4.57-fold higher than that of a formulation using a surfactant with HLB 3 (droplet size 384.7 nm), highlighting the importance of droplet size in enhancing skin delivery [38].
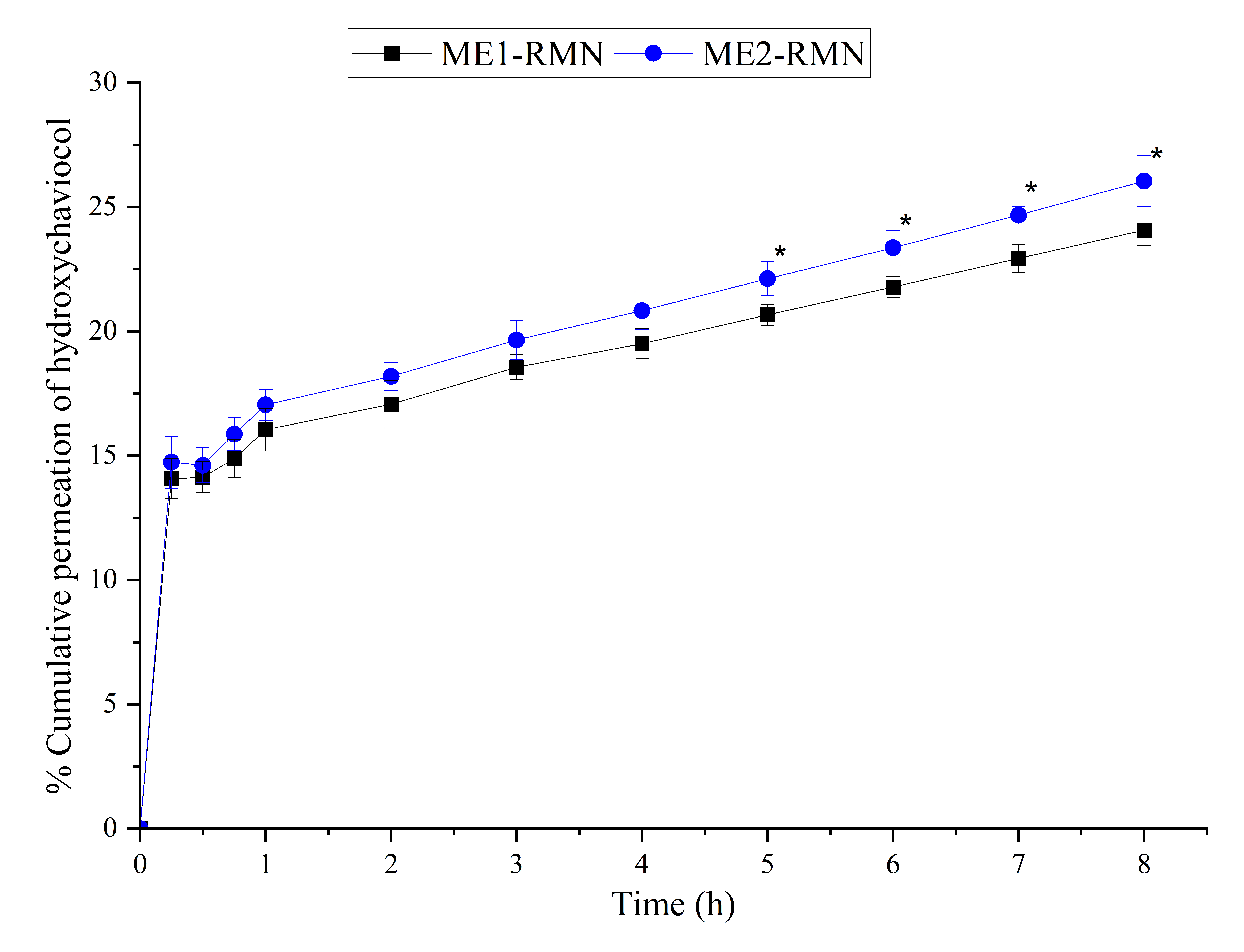
Figure 4 Cumulative permeability profile of hydroxychavicol from RMN-loaded microemulsion formulations: ME1-RMN (black) and ME2-RMN (blue). Data represent mean ± SD (n = 3). (*) p < 0.05 when compared with ME1-RMN at corresponding timepoints (2-way ANOVA; formulation×time interaction).
Skin irritation and allergic testing of RMN microemulsion in healthy volunteers
A human closed patch test was employed to evaluate allergic contact dermatitis and irritant contact dermatitis. Allergic contact dermatitis refers to an immunological response, specifically a type IV or delayed-type hypersensitivity reaction, triggered by exposure to a specific allergen. This condition often manifests as erythema, swelling, and vesiculation at the site of contact. In contrast, irritant contact dermatitis is a non-immunological skin response that results from direct damage to the skin barrier, leading to symptoms such as redness, dryness, and scaling [39]. To characterize the reactions, a positive response at 48 h post-patch application, coupled with a negative response at 72 h, indicates irritation caused by the tested substances. Conversely, a positive reaction observed at 48, 72, or 96 h after patch application indicates an allergic response [17].
In this study, 36 healthy volunteers, consisting of 7 males and 29 females aged between 21 and 41 years (mean age 23.75 ± 4.21 years), were enrolled in the skin patch testing and completed the study. The results of skin assessment are summarized in Table 4. None of the volunteers displayed any skin reactions when exposed to WP (negative control). Similar results were observed when replacing WP with ME1-Base and ME2-Base as vehicle controls, except for volunteers No. 5 and 8, who exhibited faint erythema outside the application site after 48 h (doubtful reaction).
Regarding the RMN extract, WP containing RMN extract at all concentrations (0.5%, 1%, and 3% WP-RMN) did not cause skin reactions in most volunteers. The irritant reactions observed at 48 h in volunteers No. 8 (exposed to 0.5% WP-RMN) and No. 18 (exposed to 0.5% and 1% WP-RMN) were unclear (doubtful reaction). Only one volunteer (No. 18) exhibited slight erythema (CTFA grade 1) to 0.5% WP-RMN at 72 h (score of response = 0.35).
When using microemulsion bases as vehicles, both 0.5% and 1% ME2-RMN did not induce any skin reactions in 35 volunteers, except for volunteer No. 18. In addition, 3 volunteers developed faint erythema at the application site after 48 h when using 0.5% ME1-RMN (volunteers No. 8, 18, and 35), with volunteer No. 18 also displaying faint erythema at 72 h (doubtful reaction). Similarly, faint erythema occurred in 2 volunteers at 48 h after exposure to 1% ME1-RMN (volunteers No. 18 and 25), while volunteer No. 13 developed faint erythema after 72 h (doubtful reaction).
Furthermore, all microemulsion bases containing 3% RMN extract resulted in faint erythema at the application site after 48 and 72 h in a small number of volunteers (up to 6 volunteers in 3% ME1-RMN at 48 h), except for volunteer No. 18 who still exhibited a slight skin reaction (CTFA grade 1) at 72 h (score of response = 0.35 for both 3% ME1-RMN and 3% ME2-RMN).
Based on these findings, one volunteer developed a slight allergic reaction to RMN extract in WP as well as in both microemulsion formulations, suggesting a possible allergy to the RMN extract. In addition, it has been established that a skin response score below 1.40, corresponding to a z-score < 1, is considered within the safe zone for primary skin irritation in cosmetics [18]. Therefore, both RMN microemulsion formulations demonstrated non-irritating effects on the skin and could be safely used without causing irritation in humans. Moreover, the 3% RMN-loaded microemulsion was determined to be the maximum safe concentration for topical application. Nevertheless, it should be avoided in individuals with a history of hypersensitivity to plant-derived ingredients in the RMN remedy.
Table 4 The results of skin patch test from 36 healthy volunteers.
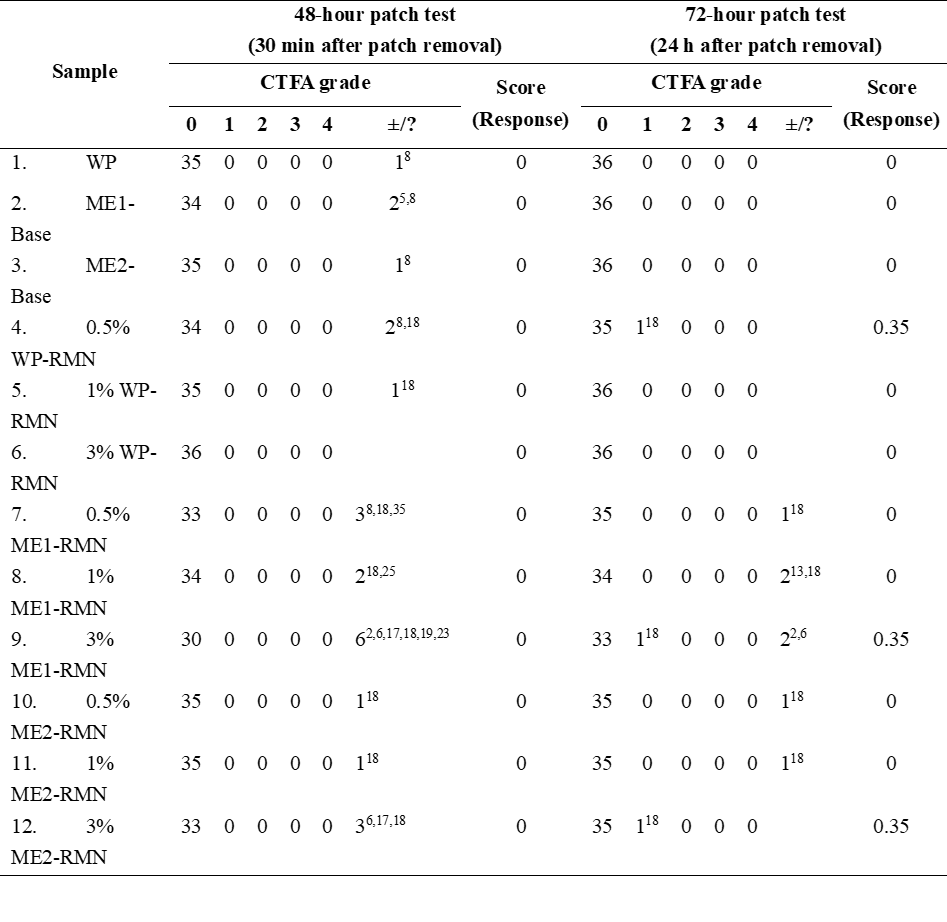
RMN: Ruean-Mhoon-Nok extract, WP: White petrolatum, ME: Microemulsion, ±/?: Doubtful reaction (faint erythema only, no infiltration), X: The total count of people, Superscript: Volunteer’s No., X1-7 refer to male volunteers, X8-36 refer to female volunteers.
Conclusions
In this study, we successfully developed 2 microemulsion formulations containing the RMN remedy extract. Capryol® 90 was identified as the most suitable oil phase, exhibiting a larger microemulsion area compared to IPM. Both polyglyceryl-4 caprate and PEG-6 caprylic/capric glycerides were appropriate surfactants when combined with Transcutol® P as a co-surfactant at a weight ratio of 1:2. The optimized RMN microemulsions had mean droplet sizes below 100 nm and displayed negative charges. They also demonstrated excellent thermodynamic stability during the heating-cooling cycles, as well as effective skin permeation and drug delivery capabilities, particularly in the ME2-RMN formulation.
In the phase I clinical trial, all microemulsion formulations containing 0.5%, 1%, and 3% of RMN extract were non-irritating and could be safely applied to human skin. The 3% RMN microemulsion was the maximum safe dose for topical application. However, further studies on the efficacy and long-term safety of RKN microemulsions are warranted to confirm these findings.
Acknowledgements
This study was supported by Thammasat University Research Unit in Applied Thai Traditional Medicine and Herbal Innovation and a research grant from the Faculty of Medicine, Thammasat University, Thailand Contract No. TP. 2-35/2565. The authors also would like to acknowledge Michael Jan Everts, from the Clinical Research Center, Faculty of Medicine, Thammasat University, for English editorial assistance.
Declaration of Generative AI in Scientific Writing The authors acknowledge the use of generative AI tools (e.g., QuillBot and Grammarly) in the preparation of this manuscript, specifically for language editing and grammar correction. No content generation or data interpretation was performed by AI. The authors take full responsibility for the content and conclusions of this work.
CRediT author statement
Atchanika Taingthum: Conceptualization, Methodology, Data curation, Formal analysis, Investigation, Validation, and Writing – original draft. Panlop Chakkavittumrong: Methodology, Resources, Data curation, Formal analysis, Investigation, Validation, and Visualization. Puritat Kanokkangsadal: Resources, Data curation, Formal analysis, Investigation, Validation, and Visualization. Intouch Sakpakdeejaroen: Conceptualization, Methodology, Data curation, Formal analysis, Investigation, Validation, Project administration, Resources, Supervision, and Writing – review & editing.
References
CE Griffiths, P van de Kerkhof and M Czarnecka-Operacz. Psoriasis and atopic dermatitis. Dermatology and Therapy 2017; 7(S1), 31-41.
A Yakupu, R Aimaier, B Yuan, B Chen, J Cheng, Y Zhao, Y Peng, J Dong and S Lu. The burden of skin and subcutaneous diseases: Findings from the global burden of disease study 2019. Frontiers in Public Health 2023; 11, 1145513.
E Niculet, C Bobeica and AL Tatu. Glucocorticoid-induced skin atrophy: The old and the new. Clinical, Cosmetic and Investigational Dermatology 2020; 13, 1041-1050.
NV Limantara, R Sadono, S Widhiati and R Danarti. Asian herbal medicine for atopic dermatitis: A systematic review. Dermatology Reports 2023; 16(1), 9727.
Ministry of Public Health. National Thai traditional medicine formulary 2021 edition. Samcharoen Panich, Nonthaburi, Thailand, 2021.
H Matsuda, T Morikawa, H Managi and M Yoshikawa. Antiallergic principles from Alpinia galanga: Structural requirements of phenylpropanoids for inhibition of degranulation and release of TNF-alpha and IL-4 in RBL-2H3 cells. Bioorganic & Medicinal Chemistry Letters 2003; 13(19), 3197-3202.
MJ Hossen, SC Kim, YJ Son, KS Baek, E Kim, WS Yang, D Jeong, JG Park, HG Kim, WJ Chung, K Yoon, C Ryou, SY Lee, JH Kim and JY Cho. AP-1-Targeting anti-inflammatory activity of the methanolic extract of Persicaria chinensis. Evidence-Based Complementary and Alternative Medicine 2015; 2015, 608126.
BY Yang, YG Cheng, Y Liu, Y Liu, JY Tan, W Guan, S Gou and HX Kuang. Datura metel L. ameliorates imiquimod-induced psoriasis-like dermatitis and inhibits inflammatory cytokines production through TLR7/8-MyD88-NF-κB-NLRP3 inflammasome pathway. Molecules 2019; 24(11), 2157.
C Nongmai, K Kanokmedhakul, T Promgool, J Paluka, C Suwanphakdee and S Kanokmedhakul. Chemical constituents and antibacterial activity from the stems and leaves of Piper wallichii. Journal of Asian Natural Products Research 2022; 24(4), 344-352.
A Taingthum, I Sakpakdeejaroen, S Panthong and P Kanokkangsadal. Assessment of in vitro antioxidant activities and quantification of total phenolic and flavonoid contents in extracts from the Thai traditional remedy “Ruean-Khi-Nok” and its plant constituents. Asian Medical Journal and Alternative Medicine 2024; 24(1), 9-18.
EB Souto, A Cano, C Martins-Gomes, TE Coutinho, A Zielińska and AM Silva. Microemulsions and nanoemulsions in skin drug delivery. Bioengineering 2022; 9(4), 158
J Leanpolchareanchai and V Teeranachaideekul. Topical microemulsions: Skin irritation potential and anti-inflammatory effects of herbal substances. Pharmaceuticals 2023; 16(7), 999
YH Lin, MJ Tsai, YP Fang, YS Fu, YB Huang and PC Wu. Microemulsion formulation design and evaluation for hydrophobic compound: catechin topical application. Colloids and Surfaces B: Biointerfaces 2018; 161, 121-128.
ACN Brathwaite, T Alencar-Silva, LAC Carvalho, MSF Branquinho, R Ferreira-Nunes, M Cunha-Filho, GM Gelfuso, SS Maria-Engler, JL Carvalho, JKR Silva and T Gratieri. Pouteria macrophylla fruit extract microemulsion for cutaneous depigmentation: Evaluation using a 3D pigmented skin model. Molecules 2022; 27(18), 5982
S Jiamphun and W Chaiyana. Enhancing skin delivery and stability of vanillic and ferulic acids in aqueous enzymatically extracted glutinous rice husk by nanostructured lipid carriers. Pharmaceutics 2023; 15(7), 1961
N Magrode, W Poomanee, K Kiattisin and C Ampasavate. Microemulsions and nanoemulsions for topical delivery of tripeptide-3: from design of experiment to anti-sebum efficacy on facial skin. Pharmaceutics 2024; 16(4), 554.
K Phumlek, A Itharat, P Pongcharoen, P Chakkavittumrong, HY Lee, GS Moon, MH Han, Panthong S, W Ketjinda and NM Davies. Garcinia mangostana hydrogel patch: bactericidal activity and clinical safety for acne vulgaris treatment. Research in Pharmaceutical Sciences 2022; 17(5), 457-467.
SM An, H Ham, EJ Choi, MK Shin, SS An, HO Kim amd JS Koh. Primary irritation index and safety zone of cosmetics: retrospective analysis of skin patch tests in 7440 Korean women during 12 years. International Journal of Cosmetic Science 2014; 36(1), 62-67.
KG Patel, AT Patel, JR Christian, AS Patel, PA Shah and MC Gohel. D-optimal mixture design for optimization of self microemulsifying drug delivery system of simvastatin: Characterization and in vitro evaluation. Bulletin of Pharmaceutical Research 2018; 8(2), 156.
P Chouhan and T Saini. D-optimal design and development of microemulsion based transungual drug delivery formulation of ciclopirox olamine for treatment of onychomycosis. Indian Journal of Pharmaceutical Sciences 2016; 78(4), 498-511
A Eichner, S Stahlberg, S Sonnenberger, S Lange, B Dobner, A Ostermann, TE Schrader, T Hauß, A Schroeter, D Huster and RHH Neubert. Influence of the penetration enhancer isopropyl myristate on stratum corneum lipid model membranes revealed by neutron diffraction and 2H NMR experiments. Biochimica et Biophysica Acta 2017; 1859(5), 745-755.
T Shukla, N Upmanyu, M Agrawal, S Saraf, S Saraf and A Alexander. Biomedical applications of microemulsion through dermal and transdermal route. Biomedicine & Pharmacotherapy 2018; 108, 1477-1494.
K Singpanna, D Charnvanich and V Panapisal. Effect of the HLB values of non-ionic surfactants on size and size distribution and stability of oil/water soybean oil nanoemulsions. Thai Journal of Pharmaceutical Sciences 2021; 45(6), 487-491.
QL Vu, CW Fang, M Suhail and PC Wu. Enhancement of the topical bioavailability and skin whitening effect of genistein by using microemulsions as drug delivery carriers. Pharmaceuticals 2021; 14(12), 1233
L Pavoni, DR Perinelli, G Bonacucina, M Cespi and GF Palmieri. An overview of micro- and nanoemulsions as vehicles for essential oils: Formulation, preparation and stability. Nanomaterials 2020; 10(1), 135.
DW Osborne and J Musakhanian. Skin penetration and permeation properties of Transcutol®-neat or diluted mixtures. AAPS PharmSciTech 2018; 19(8), 3512-3533.
Z Ait-Touchente, N Zine, N Jaffrezic-Renault, A Errachid, N Lebaz, H Fessi and A Elaissari. Exploring the versatility of microemulsions in cutaneous drug delivery: opportunities and challenges. Nanomaterials 2023; 3(10), 1688
A Azeem, M Rizwan, FJ Ahmad, Z Iqbal, RK Khar, M Aqil and S Talegaonkar. Nanoemulsion components screening and selection: A technical note. AAPS PharmSciTech 2009; 10(1), 69-76.
AK Sharma, T Garg, AK Goyal and G Rath. Role of microemulsions in advanced drug delivery. Artificial Cells, Nanomedicine, and Biotechnology 2016; 44(4), 1177-85.
LN Shen, YT Zhang, Q Wang, L Xu and NP Feng. Preparation and evaluation of microemulsion-based transdermal delivery of total flavone of rhizoma arisaematis. International Journal of Nanomedicine 2014; 9, 3453-64.
O Sarheed, M Dibi and K Ramesh. Studies on the effect of oil and surfactant on the formation of alginate-based o/w lidocaine nanocarriers using nanoemulsion template. Pharmaceutics 2020; 12(12), 1223
M Lindner, M Bäumler and A Stäbler. Inter-correlation among the hydrophilic-lipophilic balance, surfactant system, viscosity, particle Size, and stability of candelilla wax-based dispersions. Coatings 2018; 8(12), 469.
MD Arpa, E Çağlar, D Güreşçi, H Sipahi and NÜ Okur. Novel microemulsion containing benzocaine and fusidic acid simultaneously: Formulation, characterization, and in vitro evaluation for wound healing. AAPS PharmSciTech 2024; 25(3), 53.
M Lukić, I Pantelić and SD Savić. Towards optimal pH of the skin and topical formulations: from the current state of the art to tailored products. Cosmetics 2021; 8(3), 69.
L Vlaia, I Olariu, AM Muţ, G Coneac, V Vlaia, DF Anghel, ME Maxim, G Stânga, A Dobrescu, M Suciu, Z Szabadai and D Lupuleasa. New, biocompatible, chitosan-gelled microemulsions based on essential oils and sucrose esters as nanocarriers for topical delivery of fluconazole. Pharmaceutics 2022; 14(1), 75.
Y Liu, S Chen, L Sheng, M Wang and He Liu. The phase behavior and solubilization ability of nonionic surfactant-distillate fraction of crude oil microemulsion system. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2020; 603, 125181.
S Alharthi, ZM Ziora, G Mustafa, P Chaubey, AFE Kirdasy and G Alotaibi. β-caryophyllene-loaded microemulsion-based topical hydrogel: a promising carrier to enhance the analgesic and anti-inflammatory outcomes. Gels 2023; 9(8), 634
CS Chiu, PH Huang, YJ Chan, PH Li and WC Lu. D-limonene nanoemulsion as skin permeation enhancer for curcumin prepared by ultrasonic emulsification. Journal of Agriculture and Food Research 2024; 15, 100932.
G Novak-Bilić, M Vučić, I Japundžić, J Meštrović-Štefekov, S Stanić-Duktaj and L Lugović-Mihić. Irritation and allergic contact dermatitis-skin lesion characteristics. Acta Clinica Croatica 2018; 57(4), 713-720.