Trends
Sci.
2026; 23(7): 12499
In Vivo Analysis of Nuclear Factor Erythroid 2-Related Factor Modulation, Superoxide Dismutase, and Coronary Artery Histopathology by
Allium ascalonicum L Extract in Rat Models of Atherosclerosis
Laily Shofiyah1,5,6, Paramasari Dirgahayu1,2, Eti Poncorini Pamungkasari1,3,
Ida Nurwati1,4, Tonang Dwi Ardyanto1,5,6,*, Risya Cilmiaty1,5,7 and Ratih Puspita Febrinasari1,8
1Doctoral Program of Medical Sciences, Faculty of Medicine, Universitas Sebelas Maret, Surakarta 57126, Indonesia
2Parasitology and Micology Department, Faculty of Medicine, Universitas Sebelas Maret, Surakarta 57126, Indonesia
3Public Health Department, Faculty of Medicine, Universitas Sebelas Maret, Surakarta 57126, Indonesia
4Biochemistry Department, Faculty of Medicine, Universitas Sebelas Maret, Surakarta 57126, Indonesia
5Universitas Sebelas Maret Hospital, Sukoharjo 57126, Indonesia
6Clinical Pathology Department, Faculty of Medicine, Universitas Sebelas Maret, Surakarta 57126, Indonesia
7Oral Disease Department, Faculty of Medicine, Universitas Sebelas Maret, Surakarta 57124, Indonesia
8Pharmacology Department, Faculty of Medicine, Universitas Sebelas Maret, Surakarta 57126, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 2 November 2025, Revised: 28 November 2025, Accepted: 5 December 2025, Published: 1 March 2026
Abstract
Atherosclerosis is a chronic inflammatory disease involving hyperlipidemia, inflammation, and oxidative stress as the main mechanisms. Standard therapies such as statins are not fully effective in preventing the development of atherosclerosis, so additional therapies are still needed. Allium ascalonicum L extract contains quercetin, kaempferol, and allicin, which have antioxidant potential. Objective: To analyze the therapy of Allium ascalonicum L extract variety Bima, sourced from local farmers in Brebes, Central Java, Indonesia combined with atorvastatin, which has not been specifically studied in a rat model of atherosclerosis, the duration of long-term therapy (12 weeks), and to analyze complete and integrated molecular variables. This study simultaneously analyzed oxidative stress and histopathology variables, including serum Nrf2, tissue Nrf2, SOD, and coronary artery histopathology, which are potential therapeutic targets in atherosclerosis, and assessed the correlation between serum Nrf2 and tissue Nrf2, which has not been done in previous studies. Methods: The in vivo experimental study was conducted using rat fed an atherogenic diet to induce atherosclerosis. Treatment groups received Allium ascalonicum L extract and/or atorvastatin for 12 weeks. Results: Allium ascalonicum L extract therapy significantly increased serum Nrf2, tissue Nrf2, and SOD levels (p < 0.05), and histopathology showed a decrease in the degree of atherosclerosis and coronary artery wall thickness. The best effect of Allium ascalonicum L extract was seen in group P3, which was induced with an atherogenic diet and given combination therapy with 500 mg/kgBW of Allium ascalonicum L extract and 0.9 mg/kgBW of atorvastatin. A very strong correlation was found between serum Nrf2 and tissue Nrf2 (r: 0.865). In conclusion, Allium ascalonicum L extract has been proven effective as a preventive therapy in reducing oxidative stress and improving coronary artery histopathology in rat models of atherosclerosis. Serum Nrf2 marker can be proposed as a marker for monitoring atherosclerosis therapy.
Keyword: Atherosclerosis, Allium ascalonicum L, Nrf2, SOD, Coronary histopathology
Introduction
Atherosclerosis is a chronic inflammatory disease involving lipids, thrombosis, vascular walls, and
immune cells. It is the primary cause of hardening and narrowing of the arteries, leading to cardiovascular disease. The global prevalence of atherosclerosis is estimated to be around 1.4% of the adult population, and its prevalence increases with age, accounting for about 31% of deaths. The World Health Organization (WHO) estimates that atherosclerosis by 2030 will be the cause of the most morbidity and mortality, especially in developed countries due to unhealthy lifestyle changes, so atherosclerosis is a major public health problem [1].
The pathogenesis of atherosclerosis involves multiple complex systems, including hyperlipidemia, chronic inflammation, oxidative stress, and endothelial dysfunction [2]. Oxidative stress, which results from an imbalance between free radical production and the antioxidant defense system, plays a key role in cellular and vascular tissue damage [3]. Nuclear factor erythroid 2-related factor 2 (Nrf2) and superoxide dismutase (SOD) are important components in the antioxidant defense system that play a role in protecting cells from oxidative stress. Atherosclerosis causes histopathological changes of the coronary artery, which are key parameters in evaluating the development of atherosclerosis and the effectiveness of therapy [4].
Allium ascalonicum L extract has a high content of flavonoids and phenolics, especially quercetin and kaempferol, also containing organosulfur such as allicin. Feng et al. [5] researched the effect of kaempferol on lipid profile, oxidative stress, and inflammation in atherosclerosis. Research by Liu et al. [6] and Yang et al. [7] examines the effect of allicin on lipid profiles, oxidative stress, apoptosis, and plaque size in atherosclerosis.
Quercetin and kaempferol are natural flavonoid components that have pharmacological mechanisms as strong anti-cholesterol, anti-inflammatory, and powerful antioxidants, so that they can prevent atherosclerosis [8] Quercetin and kaempferol can activate the Nrf2-antioxidant element response (ARE) pathway so that they can activate a series of antioxidant enzymes such as superoxide dismutase (SOD), the most important antioxidant in cardiomyocytes [9,10] Quercetin and kaempferol play a role in inhibiting the activity of NFkB transcription factors so that they can reduce pro-inflammatory cytokines, such as IL-1β, also act as antioxidants, inhibit the COX2 enzyme and regulate the MAPK pathway [11] Allicin is the main organosulfur in Allium ascalonicum L extract which functions as an anti-cholesterol, improves endothelial function, anti-platelet, and reduces oxidative stress by lowering malondialdehyde (MDA) levels, increasing SOD levels, nitric oxide (NO), endothelial nitric oxide synthase (eNOS), Nrf2 and heme oxygenase-1 ( HO-1) [12].
In addition to using standard therapy, additional therapies are also needed to prevent atherosclerosis from using standard therapies that play a role in the various stages of atherosclerosis pathogenesis. The role of Allium ascalonicum L extract in the prevention of atherosclerosis has not been widely researched, and it is still unclear to understand its molecular mechanisms, especially against oxidative stress in coronary arterial atherosclerosis. This study aimed to determine whether Allium ascalonicum L extract plays a role in the pathogenesis of oxidative stress in coronary artery atherosclerosis.
Materials and methods
Research design and research samples
This study was an in vivo experimental study using 36 male Sprague-Dawley rat divided into 6 groups, using a randomization. The rat were induced with an atherogenic diet and simultaneously given therapy for 12 weeks with Allium ascalonicum L extract (500 mg/KgBW), atorvastatin (0.9 mg/KgBW), or a combination of both. Blood samples were taken on day 0 and week 12 of treatment for serum Nrf2 and SOD examinations, and coronary artery tissue samples were taken at week 12 for tissue Nrf2 examination and histopathology. The animal handling procedure is approved by the Research Ethics Committee of the Faculty of Veterinary Medicine, Gadjah Mada University, Indonesia (approval documents number 10/EC-FKH/int/2025).
Plant sources and extract preparations
Raw material Allium ascalonicum L extract variety Bima, sourced from local farmers in Brebes, Central Java, Indonesia. The material was confirmed to be Allium ascalonicum L of the family Amaryllidaceae (document number TL.02.04/D.XI.6/347.015/2025). Fresh Allium ascalonicum L is dried for 48 h at 40 °C, made into a powder, then macerated with a mixture of 70% ethanol and water solvent for 72 h at room temperature. Allium ascalonicum L powder was extracted separately with 70% ethanol using microwave-assisted extraction for 30 min, followed by filtration and evaporation with a rotary vacuum evaporator. The extract was then concentrated using a water bath at 70 °C to obtain the final Allium ascalonicum L extract [13,14].
The dosage and duration of the extract used were based on previous studies and preliminary research. Studies by Muradabadi et al. (2013) and Tran et al. (2020) reported that the effects of Allium ascalonicum L extract were most optimal after administration for 8 - 12 weeks, at doses of 250 and 500 mg/kgBW/day. The researchers conducted preliminary studies to determine the optimal dose of Allium ascalonicum L extract by assessing the effects of the extract at doses of 250, 500, and 750 mg/kgBW in rat models of atherosclerosis induced by an atherogenic diet. The assessment of the optimal dose of Allium ascalonicum L using laboratory and histopathological parameters showed that at a dose of 500 mg/kgBW for 12 weeks, the lipid profile was the best and the SOD level was the highest. Histopathological examination also showed a lower degree of atherosclerosis and coronary artery wall thickness compared to the other dose groups.
Induction of atherogenic diets and research protocols
Male Sprague-Dawley rat were fed an atherogenic diet containing 2% cholesterol, 0.2% cholic acid and 5% lard oil daily for 12 weeks. The rat were divided into 6 groups, namely the normal group (N), the negative control group/KN (atherogenic diet), the positive control group/KP (atherogenic diet and atorvastatin therapy 0.9 mg/kgBW), the P1 group (atherogenic diet and Allium ascalonicum L extract therapy 500 mg/kgBW), the P2 group (atherogenic diet and combination therapy of Allium ascalonicum L extract 250 mg/kgBW and atorvastatin 0.45 mg/kgBW), and the P3 group (atherogenic diet and combination therapy of Allium ascalonicum L extract 500 mg/kgBW and atorvastatin 0.9 mg/kgBW).
Sample collection and biochemical analysis
Nrf2 examination using serum samples and coronary artery tissue samples, using the Sandwich-ELISA method using Rat Nuclear Factor, Erythroid Derived 2, Like 2 (NFE2L2) ELISA Kit Reed Biotech Ltd. SOD examination using the Water-Soluble Tetrazolium Salt-1 (WST-1) method and Biovision Total Superoxide dismutase (T-SOD) Activity Assay Kit reagent. Histopathological examination of Coronary Arteries detects blood vessel damage as a sign of atherosclerosis, then assesses the degree of atherosclerosis using a scoring system.
1) Score 0: Normal, no vascular changes, or diffuse and uniform thickening of the inner intima.
2) Score 1: Fat cells accumulate in the deepest part of the intima, with macrophages and foam cells present.
3) Score 2: Smooth muscle proliferation, medial lipid infiltration, fibrosis/calcification, and increasingly prominent adipocytes.
4) Score 3: Surface damage in the form of fissures, ulcers, hematomas, thrombi, and thick, full adipocytes, which may be accompanied by foam cells.
Statistical analysis
This study used the Shapiro-Wilk normality test and Leveneʼs homogeneity test. Statistical analysis of the difference between the pretest and posttest groups used the Analysis of Variation (ANOVA) test if the data distribution was normal and homogeneous, or the Kruskall Wallis test if the data distribution was not normal and not homogeneous. If the results are significant, a post hoc test is performed. Statistical analysis of differences within the pretest and posttest groups uses the Paired T-Test if the data distribution is normal and homogeneous, or the Wilcoxon test if the data distribution is not normal and not homogeneous. The correlation between serum Nrf2 and tissue Nrf2 is assessed using the Pearson correlation test (if the data distribution is normal) or the Spearmanʼs rank correlation test (if the data distribution is not normal). Data analysis uses the Software Package for Social Science (SPSS) version 26.
Results and discussion
Allium ascalonicum L extract on serum Nrf2 levels
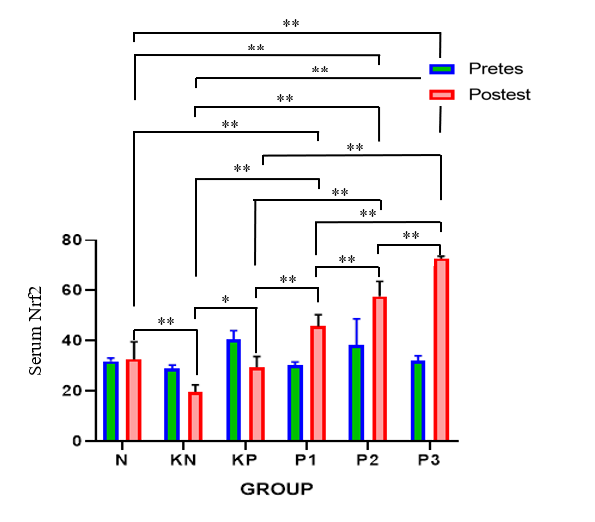
Post-test serum Nrf2 levels were increased in the P1, P2, and P3 groups, with the highest posttest serum Nrf2 levels found in the P3 group (Table 1, Figure 1). Significant pre-post-test differences were found in all groups, except group N. Significant differences were found between the six groups. Post-hoc analysis found significant differences between posttest serum Nrf levels in P1 - P3 and group N with p < 0.001, indicating that the treatment did not achieve the same results as group N.
Table 1 Test of the differences in the effects of Allium ascalonicum L extract on serum Nrf2 levels in rat models of atherosclerosis.
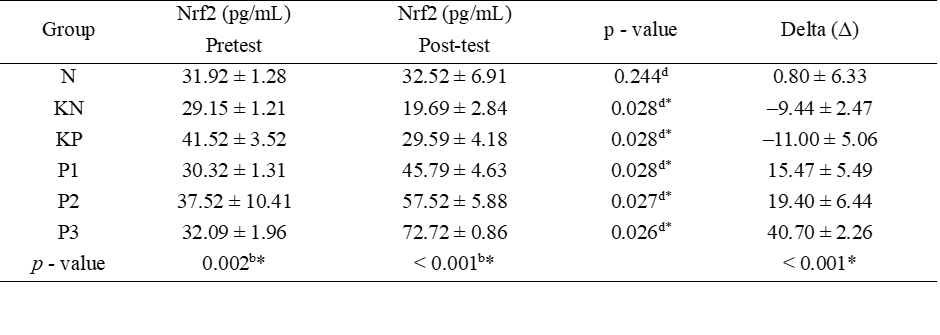
Note: N: Normal; KN: Negative control (atherogenic diet); KP: Positive control (atherogenic diet and administration of atorvastain 0.9 mg/KgBW); P1: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 500 mg/KgBW; P2: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 250 mg/kgBW and atorvastatin 0.45 mg/KgBW; P3: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 500 mg/KgBW and atorvastatin 0.9 mg/KgBW; *: Significant at p < 0.05; b: Kruskal Wallis Test; d: Wilcoxon Test.
![]()
![]()
Figure 1 Bar graphs of mean serum Nrf2 values.
Note: *: Significant at p < 0.05; **: Significant at p < 0.001.
Tabel 2 Post hoc test of posttest serum Nrf2 levels.
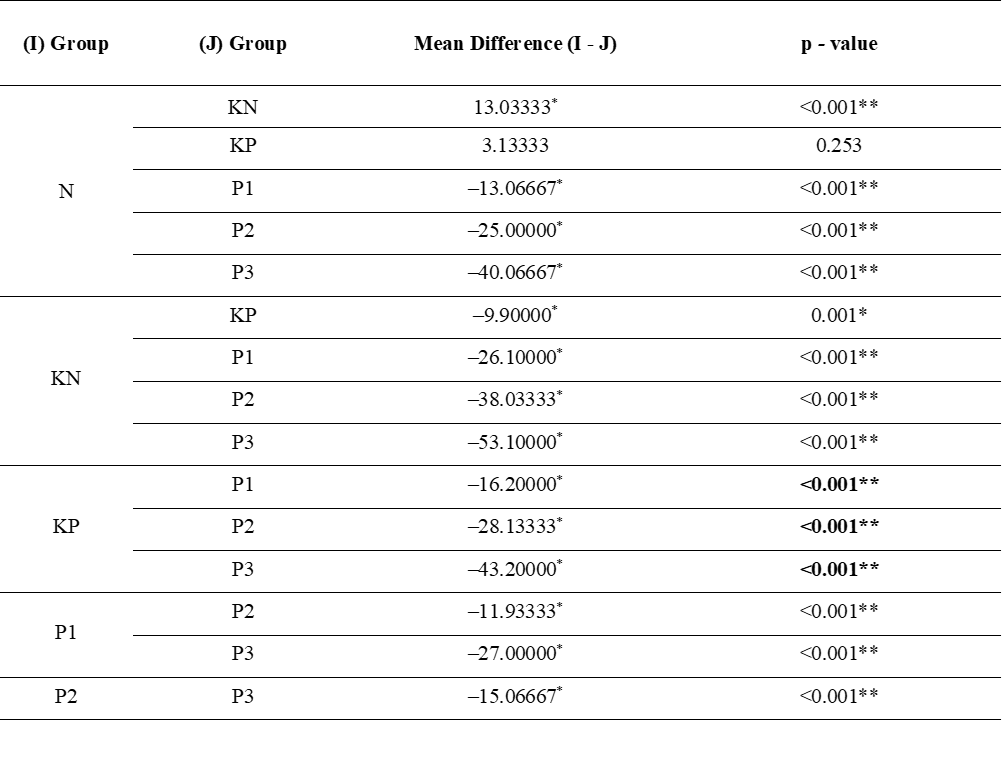
Note: N: normal; KN: Negative control (atherogenic diet); KP: Positive control (atherogenic diet and administration of atorvastain 0.9 mg/KgBW); P1: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 500 mg/KgBW; P2: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 250 mg/KgBW and atorvastatin 0.45 mg/KgBW; P3: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 500 mg/KgBW and atorvastatin 0.9 mg/KgBW; *: Significant at p < 0.05; **: Significant at p < 0.001.
An atherogenic diet can suppress Nrf2 due to chronic oxidative stress. Nuclear factor erythroid 2-related factor 2 is a key transcription factor in the cellular defense against oxidative stress [15,16]. The results of this study showed a significant increase in serum Nrf2 levels after administration of Allium ascalonicum L extract. Nrf2 activation occurs when oxidative stress increases, which causes Nrf2 to move into the cell nucleus and activates the expression of antioxidant genes such as SOD and HO-1 [17]. Allium ascalonicum L extract contains quercetin which is an active ingredient capable of regulating Nrf2 activation through the PI3K/Akt and MAPK pathways [18].
The increase in serum Nrf2 in this study strengthens the evidence that Allium ascalonicum L extract, in addition to having a direct antioxidant effect, can also activate cellular defense mechanisms through Nrf2. The time of atherosclerosis induction greatly affects Nrf2 levels. The longer the induction, the lower the levels. Nuclear factor erythroid 2-related factor 2 is a transcription factor that is predominantly active in the cell nucleus. Therefore, its levels in serum are relatively low and not always consistently detected. Nuclear factor erythroid 2 is related to factor 2, not a primary circulating protein. Therefore, its levels are not as high as in the tissue.
Allium ascalonicum L extract on tissue Nrf2 levels
The highest levels of tissue Nrf2 examination was obtained at P3 group (Table 3, Figure 2). The ANOVA test yielded significant results between groups (p = 0.004). Post hoc test obtained significant differences between the N group and KN, P2, P3; the KP group against P1 - P3; the KN group against P1 - P3; the P1 group against P2 and P3; and the P2 group against P3 (Table 4). In this study, the Spearman Rank correlation test for Nrf2 levels in serum and tissue obtained significant results (p < 0.001) with a strong correlation strength (r = 0.865).
Table 3 Test of the differences in the effects of Allium ascalonicum L extract on tissue Nrf2 levels in rat models of atherosclerosis.
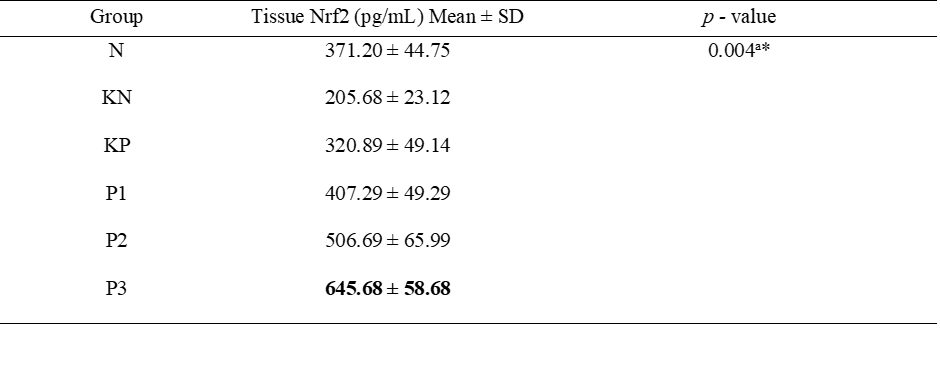
Note: N: Normal; KN: Negative control (atherogenic diet); KP: Positive control (atherogenic diet and administration of atorvastain 0.9 mg/KgBW); P1: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 500 mg/KgBW; P2: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 250 mg/kgBW and atorvastatin 0.45 mg/KgBW; P3: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 500 mg/KgBW and atorvastatin 0.9 mg/KgBW; *: Significant at p < 0.05; a: ANOVA Test.
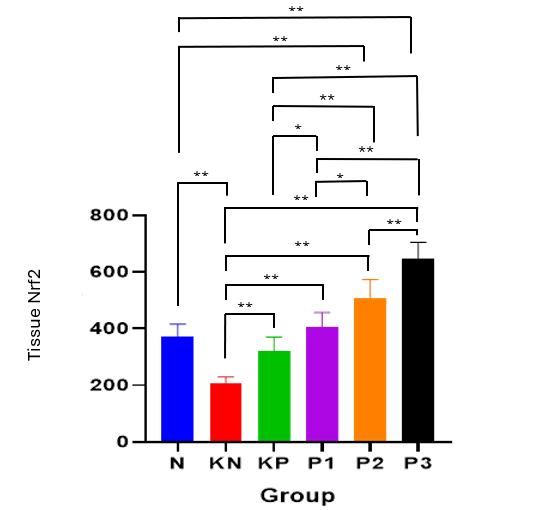
Figure 2 Bar graphs of the mean value of tissue Nrf2.
Note: *: Significant at p < 0.05; **: Significant at p < 0.001.
Table 4 Post hoc test of tissue Nrf2 levels.
(I) Group |
(J) Group |
Mean Difference (I - J) |
p - value |
N |
KN |
165.52333* |
<0.001** |
KP |
50.30833 |
0.093 |
|
P1 |
‒36.08500 |
0.224 |
|
P2 |
‒135.48833* |
<0.001** |
|
P3 |
‒274.47333* |
<0.001** |
|
KN |
KP |
‒115.21500* |
<0.001** |
P1 |
‒201.60833* |
<0.001** |
|
P2 |
‒301.01167* |
<0.001** |
|
P3 |
‒439.99667* |
<0.001** |
|
KP |
P1 |
‒86.39333* |
0.006** |
P2 |
‒185.79667* |
<0.001** |
|
P3 |
‒324.78167* |
<0.001** |
|
P1 |
P2 |
‒99.40333* |
0.0002* |
P3 |
‒238.38833* |
<0.001** |
|
P2 |
P3 |
‒138.98500* |
<0.001** |
Note: N: Normal; KN: Negative control (atherogenic diet); KP: Positive control (atherogenic diet and administration of atorvastain 0.9 mg/KgBW); P1: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 500 mg/KgBW; P2: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 250 mg/KgBW and atorvastatin 0.45 mg/KgBW; P3: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 500 mg/KgBW and atorvastatin 0.9 mg/KgBW; *: Significant at p < 0.05; **: Significant at p < 0.001.
Nrf2 levels in the coronary tissue increased significantly after administration of Allium ascalonicum L extract. These results indicate that the extract not only works systemically but also has a local effect on atherosclerotic target tissues. Allicin is known to be effective in reducing inflammatory markers such as IL-6 and TNF-α, as well as reducing oxidative stress through Nrf2 activation, lowering lipid profiles, and improving blood vessel function. A study by Jiang et al. [19] also showed that allicin from Allium ascalonicum L extract increased the expression of Nrf2 and HO-1 in the heart tissue of ApoE-/- rat, improved coronary artery histopathology, and reduced plaque size. Local activation of Nrf2 plays a major role in preventing endothelial cell apoptosis and improving vascular function [20]. This local effect strengthens the hypothesis that Allium ascalonicum L acts as an anti-atherosclerotic therapeutic agent not only at the systemic level but also directly improves vascular microstructures. Increased serum Nrf2 levels, tissue Nrf2, and SOD activity indicate an increase in endogenous antioxidant capacity, in response to decreased vascular oxidative stress. Nrf2 activation occurs through the release of KEAP1 and activation of the PI3K/AKT and MAPK pathways, according to molecular findings in previous studies.
Correlation of serum Nrf2 and tissue Nrf2 levels
The method to determine the correlation between serum Nrf2 levels and tissue Nrf2 levels is by assessing the correlation using the Spearman Rank correlation test. The results of the correlation test can be seen in Table 5. There was a significant correlation between serum Nrf2 and tissue Nrf2 (p = < 0.001) with a strong correlation rate (0.865). These results suggest that serum samples could represent coronary artery tissue for Nrf2 examination, as Nrf2 tissue sampling is invasive and difficult to apply in clinical studies. This study is in accordance with the research [21], which used blood Nrf2 as a biomarker, and the research [22] reported that serum Nrf2 levels and Nrf2 expression of kidney tissue were both elevated in lupus nephritis.
Table 5 Correlation test of serum Nrf2 levels and tissue Nrf2 levels.
Variable |
Correlation coefficient |
p - value |
Serum Nrf2 levels – Tissue Nrf2 levels |
0.865 |
<0.001e* |
Note: * Significant at p < 0.05; e: Spearman Rank Test.
Local oxidative stress in the coronary arteries can affect biomarkers in circulation. Local oxidative stress conditions in atherosclerosis cause significantly increased production and translocation of Nrf2, and some of the proteins may be released systemically so that it is measurable in the blood. The mechanism of Nrf2/KEAP1 regulation and antioxidant effects can also trigger a systemic response that can increase serum Nrf2 levels [23].
Allium ascolanicum L extract on SOD levels
The SOD level test results showed that the P3 group was able to maintain good SOD levels. (Table 6, Figure 3). ANOVA analysis revealed significant differences between the pretest and posttest groups. Significant intergroup differences in pre-test values were observed, except in the P3 group. Post hoc analysis (Table 7) demonstrated significant differences between post-test groups (p < 0.001), except between P1 and P2.
Table 6 Differential test of the effect of Allium ascalonicum L extract on serum SOD levels in rat models of atherosclerosis.
Group |
SOD (%) Pretest |
SOD (%) Posttest |
p - value |
Delta (Δ) |
N KN KP P1 P2 P3 p - value |
90.48 ± 4.58 85.24 ± 3.69 86.19 ± 4.58 85.10 ± 3.77 85.48 ± 4.08 85.95 ± 5.45 0.985a |
90.22 ± 4.60 37.70 ± 2.58 56.67 ± 5.19 66.19 ± 3.13 67.58 ± 1.57 81.33 ± 3.29 < 0.001a* |
0.028c* 0.027c* 0.028c* 0.028c* 0.028c* 0.173c
|
‒0.26 ± 0.04 ‒47.54 ± 4.88 ‒29.52 ± 5.28 ‒18.91 ± 5.20 ‒17.90 ± 5.39 ‒4.63 ± 6.32 < 0.001* |
Note: N: Normal; KN: Negative control (atherogenic diet); KP: Positive control (atherogenic diet and administration of atorvastain 0.9 mg/KgBW); P1: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 500 mg/KgBW; P2: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 250 mg/kgBW and atorvastatin 0.45 mg/KgBW; P3: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 500 mg/KgBW and atorvastatin 0.9 mg/KgBW; *: Significant at p < 0.05; a: ANOVA Test; c: Paired-t test.

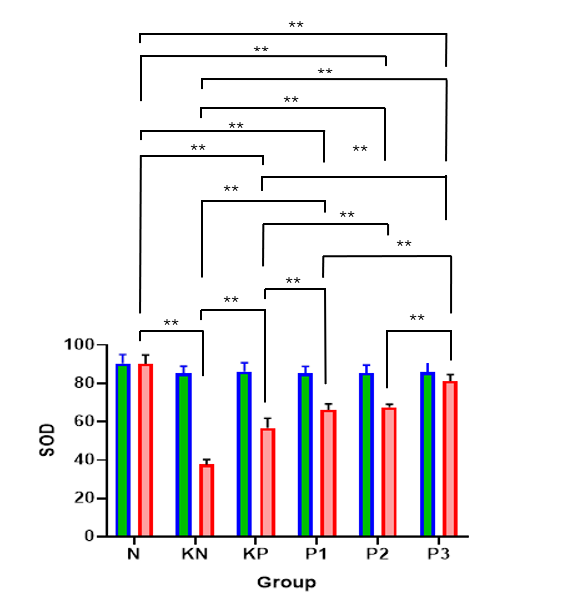
Figure 3 Bar graphs of the mean value of SOD.
Note: *: Significant at p < 0.05; **: Significant at p < 0.001.
Table 7 Post hoc test of post serum SOD levels.
(I) Group |
(J) Group |
Mean Difference (I - J) (%) |
p - value |
N |
KN |
52.52333* |
<0.001** |
KP |
33.55167* |
<0.001** |
|
P1 |
24.03333* |
<0.001** |
|
P2 |
22.64000* |
<0.001** |
|
P3 |
8.89167* |
<0.001** |
|
KN |
KP |
‒18.97167* |
<0.001** |
P1 |
‒28.49000* |
<0.001** |
|
P2 |
‒29.88333* |
<0.001** |
|
P3 |
‒43.63167* |
<0.001** |
|
KP |
P1 |
‒9.51833* |
<0.001** |
P2 |
‒10.91167* |
<0.001** |
|
P3 |
‒24.66000* |
<0.001** |
|
P1 |
P2 |
‒1.39333 |
0.508 |
P3 |
‒15.14167* |
<0.001** |
|
P2 |
P3 |
‒13.74833* |
<0.001** |
Note: N: Normal; KN: Negative control (atherogenic diet); KP: Positive control (atherogenic diet and atorvastatin 0.9 mg/kgBW); P1: Atherogenic diet and Allium ascalonicum L extract 500 mg/kgBW; P2: Atherogenic diet and Allium ascalonicum L extract 250 mg/kgBW and atorvastatin 0.45 mg/kgBW; P3: Atherogenic diet and Allium ascalonicum L extract 500 mg/kgBW and atorvastatin 0.9 mg/kgBW; *: Significant at p < 0.05; **: Highly significant at p < 0.001.
Oxidative stress plays a major role in the pathogenesis of atherosclerosis through the formation of ROS and LDL oxidation. Superoxide dismutase is the main defense enzyme against ROS, while Nrf2 acts as a major transcription factor that regulates the expression of various antioxidant enzymes. Oxidative stress leads to endothelial cell damage, accompanied by inflammatory processes, thereby triggering endothelial dysfunction, which contributes to the initiation and progression of atherosclerosis. A high-fat diet reduces SOD activity due to chronic oxidative stress, with SOD activity in atherosclerosis potentially decreasing by up to 40% - 50% compared to controls [24].
The significant increase in SOD and Nrf2 levels after administration of Allium ascalonicum L extract in this study indicates the activation of endogenous antioxidant pathways. Wang et al. [25] study reported that activation of Nrf2 by flavonoids can increase SOD expression and inhibit atherosclerotic plaque formation in rat. Queretin is also known to activate Nrf2 expression through kinase modules such as PI3K/Akt and MAPK [26]. This study shows that administration of Allium ascalonicum L extract can significantly increase serum SOD levels. Administration of Allium ascalonicum L extract can restore and increase serum SOD activity to near or exceed normal control values, demonstrating its protective role against oxidative stress caused by an atherogenic diet. Superoxide dismutase can be used as a biomarker in oxidative stress because it is the main enzymatic antioxidant that functions to neutralize superoxide anions into hydrogen peroxide [27]. Quercetin and kaempferol in Allium ascalonicum L have been shown in many studies to increase SOD both through direct activation and through Nrf2 induction [26].
Allium ascolanium L extract on coronary artery histopathology
The results showed the most severe degree of atherosclerosis in the KN group and the best results in the N group. Post intervention found the best degree of atherosclerosis in the P3 group. Meanwhile, in the coronary artery thickness examination, the best results were obtained in the N group and heaviest in the KN group. Post intervention found the best results in the P3 group. On the parameters of the degree of atherosclerosis and the thickness of coronary blood vessels, similar results were found (Table 8, Figures 4 and 5). The Kruskall Wallis test found significant results in the parameters of the degree of atherosclerosis and the thickness of coronary blood vessels. The post hoc test (Table 9) was significantly obtained in the N group against KN (p < 0.001), KP (p < 0.001), P1 (p = 0.001), P2 (p = 0.003); KN group against P1 (p = 0.036), P2 (p = 0.010), and P3 (p < 0.001); KP against P2 (p = 0.003); P1 versus P3 (p = 0.010); P2 versus P3 (p = 0.036).
Table 8 Differential test of the effect of Allium ascalonicum L extract on the degree of coronary artery atherosclerosis in rat atherosclerosis model.
Variable / Group |
Median |
Modus |
Mean±SD |
p - value |
N KN KP P1 P2 P3 |
0 2 1 1 1 0 |
0 2 1 1 1 0 |
0.00 ± 0.00 1.83 ± 0.41 1.33 ± 0.52 1.17 ± 0.75 1.00 ± 0.63 0.33 ± 0.52 |
0.002b*
rat
|
Note: N: Normal; KN: Negative control (atherogenic diet); KP: Positive control (atherogenic diet and administration of atorvastain 0.9 mg/KgBW); P1: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 500 mg/KgBW; P2: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 250 mg/kgBW and atorvastatin 0.45 mg/KgBW; P3: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 500 mg/KgBW and atorvastatin 0.9 mg/KgBW; *: Significant at p < 0.05; b: Kruskal Wallis Test.
Figure 4 Bar graphs of the mean value of atherosclerosis degree.
Note: *: Significant at p < 0.05; **: Significant at p < 0.001.
Table 9 Post hoc test of atherosclerosis degree.
(I) Group |
(J) Group |
Mean Difference (I - J) |
p - value |
N |
KN |
‒1.833* |
<0.001** |
KP |
‒1.333* |
<0.001** |
|
P1 |
‒1.167* |
0.001* |
|
P2 |
‒1.000* |
0.003* |
|
P3 |
‒0.333 |
0.282 |
|
KN |
KP |
0.500 |
0.111 |
P1 |
0.667* |
0.036* |
|
P2 |
0.833* |
0.010* |
|
P3 |
1.500* |
<0.001** |
|
KP |
P1 |
0.167 |
0.588 |
P2 |
0.333 |
0.282 |
|
P3 |
1.000* |
0.003* |
|
P1
|
P2 |
0.167 |
0.588 |
P3 |
0.833* |
0.010* |
|
P2 |
P3 |
0.667* |
0.036* |
Note: N: Normal; KN: Negative control (atherogenic diet); KP: Positive control (atherogenic diet and administration of atorvastain 0.9 mg/KgBW); P1: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 500 mg/KgBW; P2: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 250 mg/KgBW and atorvastatin 0.45 mg/KgBW; P3: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 500 mg/KgBW and atorvastatin 0.9 mg/KgBW; *: Significant at p < 0.05; **: Significant at p < 0.001.
Histopathological examination results showed that rat treated with Allium ascalonicum L extract had significantly lower coronary artery wall thickness and atherosclerotic lesion severity compared to the positive control group. These results of this study are in accordance with a study conducted by Yu et al. [28], which reported a decrease in cardiomyocyte hypertrophy, perivascular fibrosis, and interstitial fibrosis after quercetin administration [28]. Atherosclerotic plaques are characterized by thickening of the intima, lipid infiltration, migration of smooth muscle cells, and necrotic core formation. Quercetin can suppress vascular smooth muscle cell migration and prevent proliferation by suppressing ROS and the PI3K/Akt pathway [11,29]. Another study also reported that kaempferol improved intima-media thickness and inhibited arterial inflammation through the PI3K/Akt/Nrf2 pathway, supporting the histopathological findings in this study. Allicin can also increase Nrf2, and increased Nrf2 in tissues will enhance the protective effect against vascular damage [30].
Atherosclerosis causes changes in blood vessel walls, ranging from minor thickening to stenosis, which can lead to hemodynamic disturbances. The results of this study indicate that the greatest decrease in intimal thickness was found in coronary arteries in group P3 (p = 0.003). This is consistent with previous research [31] which states that quercetin administration will significantly reduce the area of atherosclerotic plaque, lipid accumulation and total cholesterol levels, and increase collagen fibers in atherosclerotic plaque. Quercetin is able to alleviate atherosclerotic lesions both in vivo and in vitro through the mechanisms of reducing cellular apoptosis and increasing mitochondrial membrane potential and simultaneously reducing free radicals or ROS. Increasing the dose of quercetin can significantly reduce the thickness of the intima layer of coronary blood vessels [29]. Tissue examination showed a reduction in the degree of atherosclerosis and coronary artery wall thickness especially in the atherogenic diet group and combination therapy of Allium ascalonicum L extract 500 mg/KgBW and atorvastatin 0.9 mg/KgBW.
Table 10 Test of the differences in the effect of Allium ascalonicum L extract on the coronary artery thickness in rat models of atherosclerosis.
Variable/ Group |
Coronary artery thickness (µm) Mean±SD |
p - value |
N KN KP P1 P2 P3 |
134.74 ± 9.56 268.29 ± 57.63 210.58 ± 22.25 164.09 ± 20.10 158.32 ± 9.65 139.68 ± 6.12 |
<0.001b*
|
Note: N: Normal; KN: Negative control (atherogenic diet); KP: Positive control (atherogenic diet and administration of atorvastain 0.9 mg/KgBW); P1: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 500 mg/KgBW; P2: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 250 mg/kgBW and atorvastatin 0.45 mg/KgBW; P3: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 500 mg/KgBW and atorvastatin 0.9 mg/KgBW; *: Significant at p < 0.05; b: Kruskal Wallis Test.
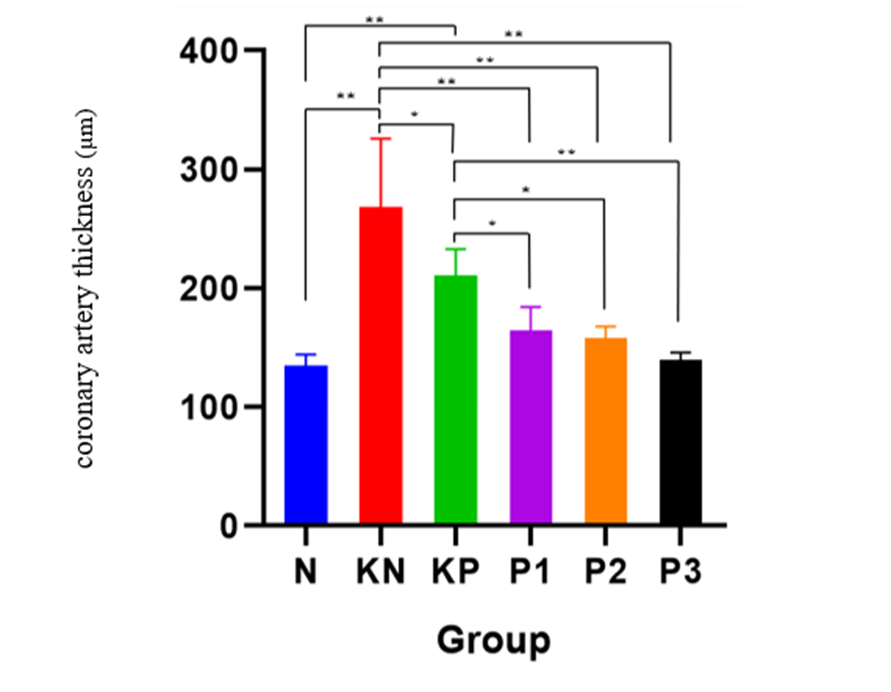
Figure 5 Bar graphs of the mean value of coronary artery thickness.
Note: *: Significant at p < 0.05; **: Significant at p < 0.001.
Table 11 Post Hoc test of coronary artery thickness.
(I) Group |
(J) Group |
Mean Difference (I - J) |
p - value |
N |
KN |
‒133.55167* |
<0.001** |
KP |
‒75.83833* |
<0.001** |
|
P1 |
‒29.34833 |
0.072 |
|
P2 |
‒23.58000 |
0.144 |
|
P3 |
‒4.94167 |
0.755 |
|
KN |
KP |
57.71333* |
0.001* |
P1 |
104.20333* |
<0.001** |
|
P2 |
109.97167* |
<0.001** |
|
P3 |
128.61000* |
<0.001** |
|
KP |
P1 |
46.49000* |
0.006* |
P2 |
52.25833* |
0.002* |
|
P3 |
70.89667* |
<0.001** |
|
P1 |
P2 |
5.76833 |
0.716 |
P3 |
24.40667 |
0.131 |
|
P2 |
P3 |
18.63833 |
0.245 |
Note: N: Normal; KN: Negative control (atherogenic diet); KP: Positive control (atherogenic diet and administration of atorvastain 0.9 mg/KgBW); P1: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 500 mg/KgBW; P2: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 250 mg/KgBW and atorvastatin 0.45 mg/KgBW; P3: Atherogenic diet and administration of Allium ascalonicum L extract at a dose of 500 mg/KgBW and atorvastatin 0.9 mg/KgBW; *: Significant at p < 0.05; **: Significant at p < 0.001.
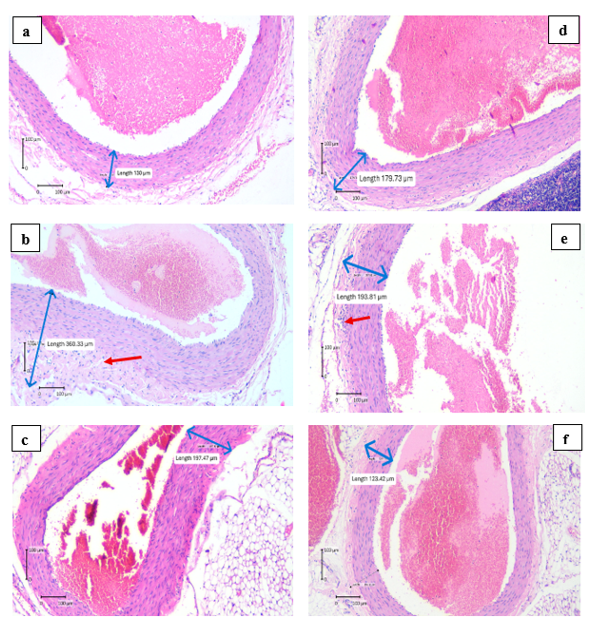
Figure 6 Results of histopathological examination of coronary arteries.
Note: The difference in the coronary arteries thickness was measured by ImageRaster Application with the unit of μm. (a) the coronary arteries thickness of N group, (b) the coronary arteries thickness of KN group, (c) the coronary arteries thickness of KP group, (d) the coronary arteries thickness of P1 group, (e) the coronary arteries thickness of P2 group, (f) the coronary arteries thickness of P3 group.
A limitation of this study is that the administration of Allium ascalonicum L extract cannot yet be directly implemented in clinical practice; further research through human clinical trials is needed. The active compounds, such as quercetin, kaempferol, and allicin in the extract, were not quantitatively analyzed, thus limiting understanding of the effective dose and the specific role of each bioactive ingredient in the observed therapeutic effects. Further molecular evaluation, such as gene or protein expression in the Nrf2/KEAP1 pathway or the NLRP3 inflammasome, was not performed, which could strengthen evidence for the mechanism of action.
Conclusions
Based on this study, it can be concluded that Allium ascalonicum L extract has the potential to be a preventive therapy for atherosclerosis through antioxidant mechanisms. This study strengthens the evidence that Allium ascalonicum L reported to contain active compound, which can decrease oxidative stress, as well as improve the endogenous defense system through Nrf2 activation and SOD levels. Allium ascalonicum L extract can be considered as a combination therapy with statin drugs, as it has a synergistic effect in the prevention of atherosclerosis. The serum Nrf2 marker can be proposed as a marker for monitoring atherosclerosis therapy. Further research in humans is needed to ensure the effectiveness and safety of Allium ascalonicum L extract in preventing atherosclerosis, especially in high-risk populations. Quantitative bioactive content analysis of quercetin, kaempferol and allicin in the extract needs to be performed to ensure the optimal dosage and pharmacological potential of each compound as well as exploration of molecular pathways such as gene expression or Nrf2/KEAP1 proteins will be very useful in strengthening the evidence of its molecular mechanism pathways.
The authors would like to express their sincere gratitude to Universitas Sebelas Maret, Indonesia, for providing access to their laboratory facilities and supporting this research. We are deeply appreciate the support provided by Universitas Sebelas Maret and Univeristas Gadjah Mada, Indonesia, which were essential to the successful completion of this study.
Declaration of Generative AI in Scientific Writing
The authors confirm that generative AI tools (such as ChatGPT by OpenAI) were utilized solely for grammatical improvements during the preparation of this manuscript and graphical abstract. These tools were not used for generating content or interpreting data. The authors take full responsibility for the content and conclusions of this work.
CRediT Author Statement
Laily Shofiyah: Conceptualization, Methodology, Supervision, Validation, Funding acquisition, and Writing –original draft. Paramasari Dirgahayu: Formal analysis, Investigation, Validation, and Visualization. Eti Poncorini Pamungkasari: Data curation, Formal analysis, Investigation and Validation. Ida Nurwati: Formal analysis, Investigation, Validation, and Visualization. Tonang Dwi Ardyanto: Methodology, Project administration, Resources, Supervision and Validation. Risya Cilmiaty: Conceptualization, Resources, Software and Writing –review & editing. Ratih Puspita Febrinasari: Data curation, Investigation, Validation, and Visualization.
References
[1] W Frąk, A Wojtasińska, W Lisińska, E Młynarska, B Franczyk and J Rysz. Pathophysiology of cardiovascular diseases: New insights into molecular mechanisms of atherosclerosis, arterial hypertension, and coronary artery disease. Biomedicines 2022; 10(8), 1938.
[2] A Poznyak, AV Grechko, P Poggio, VA Myasoedova, V Alfieri and AN Orekhov. The diabetes mellitus–atherosclerosis connection: The role of lipid and glucose metabolism and chronic inflammation. International Journal of Molecular Sciences 2020; 21(5), 1835.
[3] TR Kıran, O Otlu and AB Karabulut. Oxidative stress and antioxidants in health and disease. Journal of Laboratory Medicine 2023; 47(1), 1-11.
[4] Ismawati, I Romus and EA Utami. Efek inhibitor proteasom terhadap histopatologi arteri koronaria pada tikus model aterosklerosis. Jurnal Kedokteran dan Kesehatan Indonesia 2020; 16(2), 135-142.
[5] Z Feng, C Wang, Y Jin, Q Meng, J Wu and H Sun. Kaempferol-induced GPER upregulation attenuates atherosclerosis via the PI3K/AKT/Nrf2 pathway. Pharmaceutical Biology 2021; 59(1), 1104-1114.
[6] DS Liu, SL Wang, JM Li, ES Liang, MZ Yan and W Gao. Allicin improves carotid artery intima-media thickness in coronary artery disease patients with hyperhomocysteinemia. Experimental and Therapeutic Medicine 2017; 14(2), 1722-1726.
[7] J Yang, H Si, B Dong and Q Qin. Allicin alleviates coronary atherosclerosis of rat via endothelial nitric oxide synthase(eNOS)/nuclear factor erythroid 2-related factor(Nrf2)/heme oxygenase-1(HO-1) signaling pathway. Applied Biological Chemistry 2023; 66, 28.
[8] MA Mirza, S Mahmood, AR Hilles, A Ali, MZ Khan, SAA Zaidi, Z Iqbal and Y Ge. Quercetin as a therapeutic product: Evaluation of its pharmacological action and clinical applications: A review. Pharmaceuticals 2023; 16(11), 1631.
[9] R Assempoor, MS Daneshvar, A Taghvaei, AS Abroy, A Azimi, JR Nelson and K Hosseini. Atherogenic index of plasma and coronary artery disease: A systematic review. Cardiovascular Diabetology 2025; 24, 35.
[10] Y Jiang, W Wei, J Zhou, S Qiu, Q Yang, JH Huo and W Wang. Decoction derived from Allium ascalonicum L. bulbs and sojae semen praeparatum alleviates wind-cold-type common cold via Nrf2/HO-1 pathway and modulation of Lactobacillus murinus level. Frontiers in Pharmacology 2024; 15, 1364328.
[11] H Yao, J Sun, J Wei, X Zhang, B Chen and Y Lin. Kaempferol protects blood vessels from damage induced by oxidative stress and inflammation in association with the Nrf2 / HO-1 signaling pathway. Frontiers in Pharmacology 2020; 11, 01118.
[12] Y Gao, B Wang, G Qin, S Liang, J Yin, H Jiang, M Liu and X Li. Therapeutic potentials of allicin in cardiovascular disease: Advances and future directions. Chinese Medicine 2024; 19, 93.
[13] RY Elkanawati, SA Sumiwi and J Levita. The effects of the ethanol extract of Allium Ascalonicum L. in High-Fat-High-Fructose-Induced insulin resistance swiss-webster male mice. Journal of Experimental Pharmacology 2025; 17, 207-221.
[14] C Chalif and M Alauhdin. Analysis of the effect of sample mass, solvent volume, and sonication time on the extraction of red onion (Allium ascalonicum L) on quercetin content. Indonesian Journal of Mathematics and Natural Sciences 2024; 47, 37-48.
[15] E Młynarska, J Hajdys, W Czarnik, P Fularski, K Leszto, G Majchrowicz, W Lisińska, J Rysz and B Franczyk. The role of antioxidants in the therapy of cardiovascular diseases: A literature review. Nutrients 2024; 16(16), 2587.
[16] I Tusa, A Menconi, A Tubita and E Rovida. Pathophysiological impact of the MEK5/ERK5 pathway in oxidative stress. Cells 2023; 12(8), 1154.
[17] JA Alonso-Piñeiro, A Gonzalez-Rovira, I Sánchez-Gomar, JA Moreno and MC Durán-Ruiz. Nrf2 and heme Oxygenase‐1 involvement in atherosclerosis related oxidative stress. Antioxidants 2021; 10(9), 1463.
[18] F Li, J Liu, S Tang, J Yan, H Chen, D Li and X Yan. Quercetin regulates inflammation, oxidative stress, apoptosis, and mitochondrial structure and function in H9C2 cells by promoting PVT1 expression. Acta Histochemica 2021; 123(8), 151819.
[19] FY Sun, K Xu, J Zhou, W Zhang, G Duan and M Lei. Allicin protects against LPS-induced cardiomyocyte injury by activating Nrf2-HO-1 and inhibiting NLRP3 pathways. BMC Cardiovascular Disorders 2023; 23(1), 410.
[20] Q Zhang, J Liu, H Duan, R Li, W Peng and C Wu. Activation of Nrf2/HO-1 signaling: An important molecular mechanism of herbal medicine in the treatment of atherosclerosis via the protection of vascular endothelial cells from oxidative stress. Journal of Advanced Research 2021; 34, 43-63.
[21] LE Neilson, JF Quinn and NE Gray. Peripheral blood NRF2 expression as a biomarker in human health and disease. Antioxidants 2021; 10(1), 28.
[22] J Li, Q Guo, X Wei, Y Zhu, M Luo and P Luo. Association of serum Nrf2 protein levels with disease activity and renal impairment in lupus nephritis. Frontiers in Immunology 2024; 15, 1304167.
[23] Z Huang, M Wu, L Zeng and D Wang. The beneficial role of Nrf2 in the endothelial dysfunction of atherosclerosis. Cardiology Research and Practice 2022; 2022, 4287711.
[24] MVDMB Pimentel, A Bertolami, LP Fernandes, LP Barroso and IA Castro. Could a lipid oxidative biomarker be applied to improve risk stratification in the prevention of cardiovascular disease? Biomedicine & Pharmacotherapy 2023; 160, 114345.
[25] CL Wang, XJ Yan, CL Zhang and YW Xu. Elevated serum nuclear factor erythroid 2-related factor 2 levels contribute to a poor prognosis after acute supratentorial intracerebral hemorrhage: A prospective cohort study. Frontiers in Aging Neuroscience 2022; 14, 1014472.
[26] X Luo, X Weng, X Bao, X Bai, Y Lv, S Zhang, Y Chen, C Zhao, M Zeng, J Huang, B Xu, TW Johnson, SJ White, J Li, H Jia and B Yu. A novel anti-atherosclerotic mechanism of quercetin: Competitive binding to KEAP1 via Arg483 to inhibit macrophage pyroptosis. Redox Biology 2022; 57, 102511.
[27] S Liang, J Zhang, R Ning, Z Du, J Liu, JW Batibawa, J Duan and Z Sun. The critical role of endothelial function in fine particulate matter-induced atherosclerosis. Particle and Fibre Toxicology 2020; 17, 61.
[28] S Yu, SR Kim, K Jiang, M Ogrodnik, XY Zhu, CM Ferguson, T Tchkonia, A Lerman, JL Kirkland and LO Lerman. Quercetin reverses cardiac systolic dysfunction in Rat Fed with a High-fat diet: Role of angiogenesis. Oxidative Medicine and Cellular Longevity 2021; 2021, 8875729.
[29] G Luo, L Xiang and L Xiao. Quercetin alleviates atherosclerosis by suppressing oxidized LDL-induced senescence in plaque macrophage via inhibiting the p38MAPK/p16 pathway. The Journal of Nutritional Biochemistry 2023; 116, 109314.
[30] Z Huang, M Wu, L Zeng and D Wang. The beneficial role of Nrf2 in the endothelial dysfunction of atherosclerosis. Cardiology Research and Practice 2022; 2022(1), 4287711.
[31] F Zhang, J Feng, J Zhang, X Kang and D Qian. Quercetin modulates AMPK/SIRT1/NF-κB signaling to inhibit inflammatory/oxidative stress responses in diabetic high fat diet‑induced atherosclerosis in the rat carotid artery. Experimental and Therapeutic Medicine 2020; 20(6), 280.
Supplementary Material