
Trends
Sci.
2026;
23(7):
12682
Green Extraction of Bioactive Compounds from Solanum nigrum L Fruit Using Ultrasound-Assisted Technology: Antioxidant Properties and Proliferative Effects on NIH-3T3 Fibroblasts
In Rahmi Fatria Fajar1,2, Yesi Desmiaty1,*, Kurnia Agustini3,
Nuralih Nuralih3 and Effionora Anwar1
1Faculty of Pharmacy, Pancasila University, South Jakarta 10320, Indonesia
2Faculty of Science and Technology, Al-Kamal Institute of Science and Technology, West Jakarta 11520, Indonesia
3Research Center for Pharmaceutical Ingredients and Traditional Medicine,
National Research and Innovation Agency (BRIN), Bogor 10340, Indonesia
(*Corresponding author’s email: [email protected])
Received: 17 November 2025, Revised: 21 December 2025, Accepted: 28 December 2025, Published: 10 March 2026
Abstract
Skin aging is a process that involves a gradual decline in the function of fibroblasts in the dermis layer. Fibroblasts in the dermis play a role in controlling the structure of the extracellular matrix, supporting wound healing, and maintaining skin tissue stability. When fibroblast function declines, it results in damage to the skin structure, prolonged inflammation, and a reduction in the skin’s ability to regenerate. This study aimed to evaluate the antiaging potential of the extract of Solanum nigrum L fruit from Majenang, Central Java, Indonesia. The effects of solvent type and extraction duration on the bioactive content and biological activities of Solanum nigrum L fruit extracts obtained by Ultrasound-Assisted Extraction (UAE). Proliferation activity was carried out on NIH-3T3 fibroblast cell lines, while antioxidant activity was done with DPPH methods. We also analyzed the content of phenolic and flavonoid compounds. The research method was experimental. Extraction was performed using the Ultrasonic-Assisted Extraction (UAE) method. Phytochemical analysis included the determination of total phenol and total flavonoid content. The activity of the extract was evaluated through antioxidant testing using the DPPH method and testing of the proliferation effect on NIH 3T3 cells. The results indicated that solvent selection and extraction time had a significant influence on both extraction yield and biological activity. Optimal conditions were achieved using 96% ethanol as the solvent and an extraction time of 30 min, yielding a total phenolic content of 61.18 mg GAE/g, flavonoids of 73.99 mg Rutin/g, antioxidant activity of 87.164 ppm, and an EC50 value for NIH-3T3 fibroblast cell proliferation of 12.48 ppm. These proved that Solanum nigrum L fruit has antioxidant activity and can stimulate the proliferation of NIH-3T3 fibroblast cells, making it a promising candidate for further development in the pharmaceutical and cosmetic industries as an active ingredient that supports cell regeneration.
Keywords: Antioxidant, Cosmetic, Fibroblasts, Proliferation, NIH-3T3, Skin aging
Introduction
Fibroblasts are the primary cells in connective tissue that play an essential role in producing and regulating the extracellular matrix, primarily collagen, which provides strength and structural support to tissues, including the skin [1]. They also communicate with stem cells and other cells and tissues to maintain
tissue balance and function [2]. During the skin aging process, fibroblast function declines, resulting in reduced collagen production, which causes the skin to become thin, wrinkled, and saggy. In addition, fibroblasts play a role in wound healing by multiplying and forming new tissue, but this ability also declines with age.
The role of fibroblasts is crucial in the aging process. Recent studies suggest that aging in fibroblasts results in a decline in skin function by permanently stopping cell proliferation and increasing the release of aging-associated secretory phenotypes (SASP) [3]. SASP molecules, which include chemokines and proinflammatory factors, play a role in the development of chronic inflammation, reduce cell proliferation by interfering with the release of essential growth factors, and exacerbate extracellular matrix degradation by activating proteolytic enzymes such as metalloproteinases [1]. Unlike temporary cellular aging during embryogenesis and wound healing, fibroblast aging is permanent and is a significant factor in skin aging, systemic aging, and age-related diseases [4].
Medicinal plants have long been a primary source of bioactive compounds used in the development of drugs, functional foods, and other health products due to their diverse chemical structures and biological activities. Bioactive compounds include alkaloids, phenolics, terpenoids, flavonoids, and various other secondary metabolites. These compounds exhibit a wide range of chemical structures and biological activities, offering significant potential as new molecular candidates for the treatment of multiple diseases [5]. Global demand for products based on natural ingredients continues to rise, driven by growing interest in traditional and alternative medicine, as well as the need for safer and more environmentally friendly active ingredients [5,6].
Extraction is a critical initial step in the analysis and utilization of bioactive compounds from plants. The purpose of extraction is to separate and obtain active components from the plant matrix without damaging their chemical structure, thereby enabling further isolation, identification, and characterization [5,7]. Extraction efficiency is significantly influenced by various factors, including the extraction technique used, the physical-chemical properties of plant components, and the type of solvent selected [4]. Conventional extraction methods such as maceration, decoction, and Soxhlet extraction are still widely used, but they tend to be time-consuming, require large solvent consumption, and may damage thermolabile compounds [8,9]. Innovative extraction methods such as ultrasound-assisted extraction (UAE), microwave-assisted extraction (MAE), and supercritical fluid extraction (SFE) have been developed to enhance extraction yield and quality while reducing energy consumption and environmental impact [10,11]. These innovative technologies offer advantages such as increased extract yields, shorter processing times, lower solvent and energy consumption, and protection against heat-sensitive active compound degradation [8].
One of the prominent techniques is ultrasound-assisted extraction (UAE), which has been proven to significantly accelerate the extraction process while improving the quality and yield of the extract. Unlike conventional methods that require a long time and high solvent consumption, UAE can extract bioactive components completely in just a few minutes, with high reproducibility and reduced solvent use [8].
Sound waves are formed due to variations in pressure, particle movement, and particle velocity in elastic materials. When the frequency of these waves is in the ultrasonic range, they are called ultrasonic waves. Unlike electromagnetic waves, ultrasonic waves are mechanical and can propagate through solid, liquid, or gaseous materials [12]. When cavitation bubbles collapse, the energy released can cause various effects such as extraction, destruction, and emulsification [11]. Ultrasonic cavitation occurs when, in a liquid, small bubbles (cavitation nuclei) vibrate, expand, and absorb energy from acoustic waves due to the influence of ultrasonic waves. Once the accumulated energy reaches a certain threshold, these bubbles collapse suddenly. When cavitation bubbles collapse rapidly, a large amount of energy is released in the form of microjets moving at speeds of approximately 200 to 700 m/s. These microjets generate significant impact pressure. When cavitation bubbles collapse, they produce extremely high temperatures and pressures (around 5,000 K and 2,000 atm), followed by rapid cooling at a rate of up to 109 K/s [13] (Figure 1).
With advancements in technology, optimizing the extraction process not only impacts product efficiency but also the safety and quality of the final product, making it a vital aspect in the pharmaceutical, cosmetic, and plant-based biotechnology industries. The primary advantage of the UAE lies in its ability to produce extracts with high purity while preserving the biological characteristics of active compounds, ensuring that their biological activity and function remain optimal [9]. This is particularly important for applications in pharmaceuticals, food, and cosmetics, where the stability and bioactivity of compounds are critical determinants of final product quality. Additionally, the UAE is considered an economic hub for green technology, as it reduces energy consumption, operational time, solvent costs, and enhances extraction yields.
Skin aging is a complex biological process involving intrinsic factors such as genetics, hormones, and cell metabolism, as well as extrinsic factors such as UV exposure, pollution, and oxidative stress, which cumulatively cause progressive functional decline in the epidermis, dermis, and hypodermis [14]. The epidermis will experience thinning due to delayed keratinocyte cell turnover (decreased by 30% - 50%), reduced natural moisturizing factor (NMF) levels, causing chronic xerosis, and decreased melanocyte density (8% - 20% per decade), resulting in atypical pigmentation and graying hair. Extrinsic photoaging can exacerbate this process through UV-induced ROS, which disrupts DNA repair via nucleotide excision repair (NER) and triggers dysregulated apoptosis, thereby increasing susceptibility to infection and the risk of skin cancers such as melanoma [3]. These conditions are associated with progressive dysfunction of dermal fibroblasts responsible for the synthesis of type I/III collagen, elastin, and proteoglycans, which undergo senescence due to mitochondrial ROS and MMPs as the main triggers, loss of elasticity, and skin xerosis [4].
Nan et al. [3] explain that oxidative stress from excess ROS damages DNA, proteins, and cell lipids, which can trigger telomere shortening, cellular senescence, and activation of the MAPK/AP-1 pathway, which inhibits collagen synthesis by decreasing TGF-β [3]. In the dermis, the accumulation of MMPs degrades collagen/elastin, while AGEs and inflammaging accelerate wrinkling, loss of elasticity, and hyperpigmentation through fibroblast dysfunction and low vascularization [4].
Solanum nigrum L (SN), known as black nightshade, is a plant belonging to the Solanaceae family, widely distributed in tropical and subtropical regions, including Southeast Asia and Indonesia [15]. This plant has been traditionally used in medicine to treat various diseases, including liver disorders, cancer, inflammation, infections, and diabetes [15]. Multiple parts of the SN plant, particularly the fruit and leaves, contain bioactive compounds such as steroids, glycosides, saponins, phenols, and polysaccharides, which exhibit antioxidant activity and play a crucial role in its pharmacological activity [15,16].
Modern research indicates that SN possesses several significant pharmacological effects, including antioxidant, anti-inflammatory, antitumor, antimicrobial, antidiabetic, and hepatoprotective activities [15]. The hepatoprotective effects of SN have been demonstrated through various preclinical studies, where SN extracts have been shown to reduce levels of liver damage markers, including ALT, AST, ALP, and bilirubin, as well as improve liver tissue structure in toxin-induced animal models. This protective mechanism is believed to be related to SNʼs ability to suppress oxidative stress, neutralize free radicals, and enhance cell regeneration. A study by Hong et al. [52] demonstrated that SN extract significantly reduced epidermal and dermal thickness, as well as decreased mast cell and CD8 infiltration in the skin tissue of mice with atopic dermatitis (AD) [12].
SN extract has been proven to possess very high free radical scavenging capacity in various in vitro tests, such as DPPH and FRAP, and is capable of inhibiting the activity of enzymes involved in degenerative and aging processes [15,17]. Quercetin in SN is believed to significantly contribute to the antioxidant mechanism and cellular protective effects against oxidative stress [11]. Additionally, SN antioxidants can improve oxidative status in cellular models, inhibit damage caused by glutamate excitotoxicity, and increase glutathione levels, a crucial endogenous antioxidant in cells [13]. This efficacy supports the potential of SN as a natural ingredient in skin regeneration formulations.
NIH 3T3 cells are a fibroblast cell line derived from mouse embryonic tissue. They are often used as an in vitro model in various biomedical studies, including studies related to cell aging and antiaging effects. Fibroblasts play an essential role in the maintenance and regeneration of skin tissue, which affects skin elasticity and suppleness, two significant factors in the aging process that decline with age [18]. The aging process causes oxidative damage, a decrease in cell proliferation, and changes in collagen production that affect fibroblast function.
Therefore, in this study, we tested compounds on NIH 3T3 cells to provide an overview of the effectiveness of SN in combating aging, increasing cell survival, reducing oxidative damage, and stimulating extracellular matrix synthesis. This study has the potential to support the development of SN for maintaining skin health and appearance.
Materials and methods
Materials and tools
SN fruit was collected from plants growing in Majenang, Central Java, Indonesia. The fruit was identified at the Batu Malang Technical Implementation Unit (UPT) with the number 000.9.3/3392/102.20/2024 and declared as Solanum nigrum L, harvested when the plants reached three months after planting. Na₂CO₃ (Sigma-Aldrich, USA), gallic acid (Sigma-Aldrich, Germany), methanol (Sigma-Aldrich, Germany), Folin-Ciocalteau reagent Sigma-Aldrich, Germany), rutin, AlCl₃, acetic acid, FeCl₃, DPPH (Sigma-Aldrich, Germany), Vitamin C (Himedia, India), Penicillin streptomycin (Sigma-Aldrich USA), Fetal Bovine Serum (FBS) (Sigma-Aldrich USA), Trypsin-EDTA (Gibco, Canada), Sodium Dodecyl Sulfate (SDS) (Sigma-Aldrich, USA), Yellow MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) (Thermo Fisher, USA), NIH3T3 fibroblast cells (Laptiab Serpong, Indonesia), Amphotericin B (Gibco, Israel), Phosphate Buffered Saline (PBS) (Invitrogen)
The tools include Sonicator (Qsonica), Rotary evaporator (Heidolph), spectrophotometer UV-Vis (Biochrom Libra), Elisa Reader (Synergy htx), Laminar Air Flow (Thermo scientific 1300), Centrifuge (Hettic 320 R), incubator (Memmert), Microscope (Axiovert 40 CFL), Shaker (Heidolph), Neraca analitik (Ohaus), ultrasonic bath (Bransonic 8510)
Methods
SN Extraction with UAE
Fresh fruits were washed with running water, dried, and ground using a blender to obtain small particles. The extraction process was carried out using ultrasonic technology with ethanol solvents at concentrations of 96%, 70%, 50% and 30%, with a material-to-solvent ratio of 1:10 and the addition of 1% HCl. Extraction was performed at 40 kHz, with a power of 1,375 Watts at room temperature for 30, 45 and 60 min, and was repeated 3 times. After extraction, the mixture was filtered through filter paper, and the resulting extract was concentrated using a rotary vacuum evaporator until a thick concentrate was obtained [19].
Determination of Total Phenolic Content (TPC)
Total phenolic content was determined using the Folin-Ciocalteu method. The extract, Folin-Ciocalteu reagent, and sodium carbonate were incubated for 2 h at room temperature, and then the absorbance was measured at 730 nm. The results were expressed as milligrams of gallic acid equivalent (GAE) per gram of sample [20].
Determination of Total Flavonoid Content (TFC)
Total flavonoid analysis was performed using the aluminum chloride colorimetric method. A sample of 0.3 g was mixed with 1.5 mL of PA ethanol, followed by the addition of 0.1 mL of sodium acetate solution, 20% aluminum chloride solution, and 2.8 mL of distilled water. The mixture was then incubated at room temperature for 30 min. Absorbance was measured at a wavelength of 730 nm using a UV-Vis spectrophotometer. Total flavonoid content was calculated based on the calibration curve and expressed as mg rutin equivalent per gram of extract, with the test treatment performed in three replicates [19,20].
Antioxidant activity
The antioxidant activity of plant extracts was evaluated using the DPPH (2,2-diphenyl-1-picrylhydrazyl) method, a simple, fast, and economical method for assessing compoundsʼ ability to neutralize free radicals. Changes in color intensity were measured at a wavelength of 517 nm [21]. Plant extract stock solutions were prepared by dissolving 1 mg of extract in 1 mL of dimethyl sulfoxide (DMSO), then serially diluted in methanol at concentrations of 62.5, 125, 250, 500 and 1,000 µg/mL. Ascorbic acid was used as a positive control, and methanol as a negative control. A total of 100 µL of the extract solution or control was added to each well of a 96-well plate, followed by 100 µL of DPPH solution in each well. The mixture was incubated for 30 min in the dark at room temperature to ensure optimal reaction. The test was performed in triplicate [19,22].
Proliferation of NIH-3T3 fibroblasts
Cell culture
NIH-3T3 cells were cultured in high-glucose DMEM medium supplemented with 10% FBS, 1% penicillin, and 0.1% amphotericin B. Culture was performed in a 5% CO₂ incubator at 37 °C with maintained humidity. The medium was changed every 48 to 72 h. Once 80% confluence is reached, cells are harvested and subcultured [23].
MTT Assay
NIH-3T3 cells are seeded in 96-well plates at a density of 10,000 cells/100 µl/well and incubated overnight. After 24 h, the medium is replaced, and the cells are exposed to various concentrations of the test compound (6.25, 12.5, 25, 50, 100, 200 and 400 ppm) for an additional 24 h [18]. After treatment, the medium is removed, and the cells are rinsed with PBS. Next, 100 µl of DMEM medium and 10% MTT solution (Sigma) were added to each well and incubated for 4 h at 37 °C. After incubation, the formed formazan crystals were added to the SDS solution. Observations were made at 24, 48 and 72 h. Absorbance was measured using an ELISA reader at a wavelength of 540 nm. Each treatment was performed in triplicate. Cell viability percentage was calculated by comparing the absorbance of the treatment group with that of the control group.

Data analysis
Research results are presented as the mean ± standard deviation (SD) for each treatment control. Statistical significance of differences between groups was evaluated using GraphPad software (GraphPad Software 10). Comparisons of means between groups were performed using one-way analysis of variance (one-way ANOVA).
Results and discussion
Extraction yield
SN extraction was performed using 100 g of fresh fruit extracted with optimized ethanol solvent at a ratio of 1:10. This extraction process aimed to obtain optimal bioactive compounds. The extraction yield of SN is presented in Table 1.
Table 1 Extraction yield of SN extract.
No |
Sample name |
Extraction yield |
Average |
SD |
||
Test 1 |
Test 2 |
Test 3 |
||||
1 |
SN 9630 |
5.30 |
5.5 |
5.3 |
5.37 |
0.11547 |
2 |
SN 9645 |
4.20 |
4.4 |
4.4 |
4.33 |
0.11547 |
3 |
SN 9660 |
4.70 |
4.6 |
4.65 |
4.65 |
0.05 |
4 |
SN 7030 |
5.80 |
5.5 |
5.7 |
5.67 |
0.152753 |
5 |
SN 7045 |
5.30 |
5 |
5.1 |
5.13 |
0.152753 |
6 |
SN 7060 |
5.10 |
4.9 |
4.9 |
4.97 |
0.11547 |
7 |
SN 5030 |
6.30 |
6.4 |
6 |
6.23 |
0.208167 |
8 |
SN 5045 |
6.10 |
5.9 |
5.9 |
5.97 |
0.11547 |
9 |
SN 5060 |
5.80 |
5.7 |
5.7 |
5.73 |
0.057735 |
10 |
SN 3030 |
5.20 |
5.1 |
5.3 |
5.20 |
0.1 |
11 |
SN 3045 |
5.30 |
5.2 |
5.3 |
5.27 |
0.057735 |
12 |
SN 3060 |
5.30 |
5.1 |
5.4 |
5.27 |
0.152753 |
Notes: SN 9630 = ethanol extract 96% 30 min UAE; SN 9645 = ethanol extract 96% 45 min UAE; SN 9660 = ethanol extract 96% 60 min UAE; SN 7030 = ethanol extract 70% 30 min UAE; SN 7045 = Ethanol extract 70% 45 min UAE; SN 7060 = Ethanol extract 70% 60 min UAE; SN 5030 = Ethanol extract 50% 30 min UAE; SN 5045 = Ethanol extract 50% 45 min UAE; SN 5060 = Ethanol extract 50% 60 min UAE; SN 3030 = Ethanol extract 30% 30 min UAE; SN 3045 = Ethanol extract 30% 45 min UAE; SN 3060 = Ethanol extract 30% 60 min UAE.
Ultrasound-assisted extraction (UAE) is a modern approach widely used to enhance the efficiency of extracting bioactive compounds from natural materials. The primary principle of UAE is the utilization of ultrasonic waves capable of breaking down plant cell walls, thereby accelerating the release of active compounds into the solvent [24]. The advantages of the UAE over conventional methods include shorter extraction times, reduced solvent usage, and a higher yield and quality of the extract [20].
Ultrasonic-assisted extraction (UAE) has been shown to offer significant advantages, including shorter extraction times while maintaining or even enhancing extraction efficiency. This advantage is primarily due to the phenomenon of acoustic cavitation, where ultrasonic waves generate microbubbles that then implode intensely within the extraction medium [8]. These explosions produce physical effects such as turbulence, shear forces, shock waves, and microjets that can break down cell walls, thereby accelerating and enhancing mass transfer rates between the substrate and solvent Figure 1 [11,25,26].
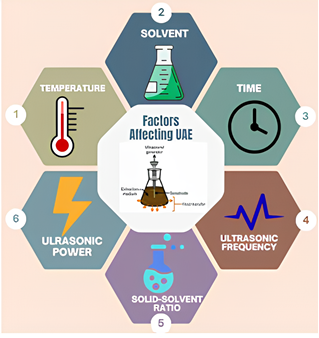
Figure 1 Factors influencing the UAE.
Several studies indicate that optimal extraction in the UAE is generally shorter compared to conventional methods. For example, the highest extraction yield can be achieved in 30 min, where the extraction rate increases sharply in the initial minutes and then slows down after reaching the saturation point [25,27,28]. The study by Papoutsis et al. [27] confirmed that increasing extraction time and ultrasonic power linearly increase total phenolic extraction yield up to an optimal point, after which further increases in time no longer provide significant additional yield [27]. Extending the extraction time beyond the optimal point can reduce yield. This is caused by two main factors: Degradation of active compounds due to prolonged or excessive ultrasonic exposure [25,28] and cavitation saturation effects, where an excessive number of bubbles cause collisions between bubbles, reducing the effectiveness of explosions and even blocking the transmission of ultrasonic energy to the extraction medium [25]. In addition to time, other parameters such as solvent-to-material ratio, temperature, and ultrasonic power also play a crucial role in determining UAE efficiency. Optimization studies using the Response Surface Methodology (RSM) indicate that the appropriate combination of time, temperature, and ultrasonic power can significantly enhance the extraction yield of bioactive compounds such as polyphenols, flavonoids, and antioxidants from various natural materials [27,28,29]. The advantages of the UAE over conventional methods also lie in its more efficient energy use, lower solvent consumption, and lower process temperatures, making it a more environmentally friendly approach and preserving the stability of the extracted bioactive compounds [11,25,26].
Optimization of solvent polarity has been proven to play a crucial role in determining the extraction efficiency and yield of bioactive compounds from SN using ultrasound-assisted extraction (UAE) [30]. The results of the study indicate that the use of 96% ethanol for 30 min yields the highest extraction yield. This aligns with the principle of “like dissolves like”, where polar compounds are more easily extracted using polar or semi-polar solvents [31].
SN contains the following main bioactive compounds: Alkaloids (solasonine, solamargine), steroids, flavonoids (quercetin, rutin), saponins, phenols, and glycosides. These compounds are generally polar to semi-polar in nature, making them easier to extract using polar solvents such as ethanol. Other studies have also reported that ethanol extracts from SN contain high concentrations of flavonoids and phenols, while alkaloid and glycoside content is lower. This indicates that polar solvents are more effective in extracting antioxidant and bioactive compounds from this plant [32].
In addition to the polarity of the solvent, physical properties such as viscosity and volatility also influence the extraction process with UAE. Solvents that are too viscous can reduce the ultrasonic cavitation effect, thereby decreasing extraction efficiency [11,31]. Meanwhile, solvent volatility must be considered to prevent solvent loss during extraction at high temperatures, which can reduce extract yield [11,32].
The optimum UAE conditions depend on the solvent ratio, time, temperature, frequency, and ultrasonic power. Several studies have shown that the optimal UAE time is 15 - 60 min, and beyond that point, the yield/activity begins to decline due to degradation or phenolic oxidation. Extraction times of 45 and 60 min were used to confirm whether the optimal pattern at 30 min still applied to the matrix and experimental conditions used in this study, rather than relying solely on previous reports.
Phytochemical analysis of several SN species, such as Solanum schimperianum, Solanum cordatum, and Solanum nigrum, revealed that phenolic compounds are the primary components contributing to antioxidant activity. Another study on several Solanum species, including Solanum torvum, Solanum aethiopicum, and Solanum macrocarpon, showed varying phenolic content, with 2.03 mg GAE/g in Solanum torvum, 3.89 mg GAE/g in Solanum aethiopicum, and 1.03 mg GAE/g in Solanum macrocarpon.
Total phenolic and total flavonoid content
The determination of total phenolic content using the Folin-Ciocalteu method in this study is expressed as milligrams of gallic acid equivalent per gram of sample. The Folin-Ciocalteu method works based on an oxidation-reduction reaction between phenolic compounds and phosphomolybdic acid-phosphotungstic acid reagents, resulting in a color change from yellow to blue [33]. The intensity of the blue color formed is measured spectrophotometrically and is proportional to the amount of phenolic compounds in the sample [34,35].
The determination of total flavonoid content was performed using the aluminum chloride (AlCl3) colorimetric method, expressed as milligrams of rutin equivalent per gram of sample. The principle of this method involves the formation of a stable complex between (AlCl3) and the hydroxyl and keto groups in the flavonoid structure, resulting in a yellow color. The test results are presented in Figure 2.
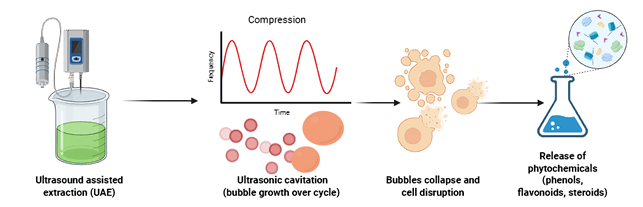
Figure 2 Ultrasound assisted extraction (UAE) process.
Xufei et al. explained that SN contains phenolic compounds and flavonoids such as quercetin, kaempferol, and gallic acid, which have been scientifically proven to neutralize free radicals. Free radicals are the primary contributors to the aging process through oxidative stress mechanisms that damage cells, tissues, and DNA. SN antioxidants can suppress the effects of oxidative stress, thereby slowing cellular damage and restoring redox homeostasis, which helps combat aging and improve skin health [36]. Animal studies have shown that SN leaf extract administration reduces oxidative stress biomarkers and helps maintain endogenous antioxidant enzyme balance after stress exposure, slowing age- and environment-induced damage [37,38].
Antioxidant activity using the DPPH method
SN is recognized for its bioactive compounds, including flavonoids and phenolics, which function as antioxidants. The measurement of antioxidant activity using the DPPH method is based on the principle of measuring the fading of the purple color of DPPH radicals through the reaction of antioxidant compounds that donate hydrogen atoms. DPPH is a molecule containing unstable nitrogen-containing free radicals that can bind hydrogen ions, making it suitable for testing antioxidant activity. The reaction of hydrogen atoms neutralizes the free radicals from DPPH, resulting in the color change of DPPH from purple to yellow [39]. In this study, vitamin C was used as a control. This is because vitamin C has an electron-donating group. This group is located on carbon atoms C2 and C3. The presence of this group allows vitamin C to capture radicals. The calibration curve for vitamin C is shown in Figure 3.
The results showed that the antioxidant activity of SN was influenced by the type of solvent, ethanol concentration, and extraction time. The SN 9630 extract produced an IC₅₀ value of 87.16 ppm, indicating very high antioxidant activity, while the SN 3060 extract had an IC₅₀ of 186.43 ppm when compared to vitamin C as a positive control with an IC₅₀ value of 3.72 ppm. Statistical analysis showed a significant difference with a p - value < 0.0001 below p < 0.05.
It is known that the phenolic and flavonoid content of SN includes quercetin, kaempferol, gallic acid, protocatechuic acid, and rutin. The high levels of phenolic and flavonoid compounds significantly contribute to antioxidant activity. Additionally, steroid saponins and alkaloids can enhance antioxidant effects and provide other pharmacological effects such as anti-inflammatory and hepatoprotective properties [15,16].
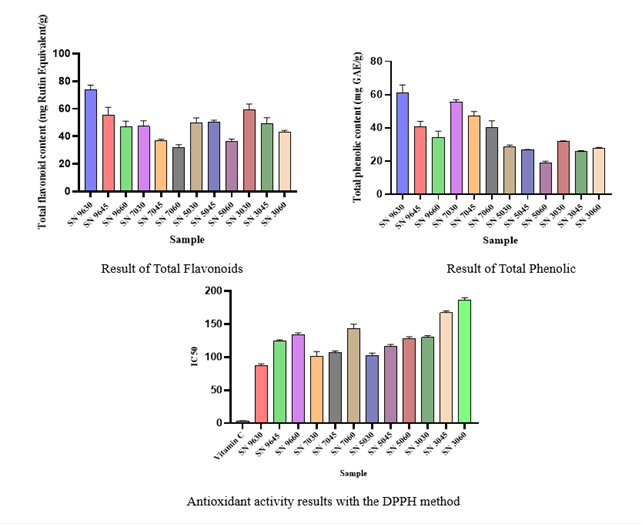
Figure 3 Results of total flavonoids, total phenols, and antioxidants SN 9630 = ethanol extract 96% 30 min UAE; SN 9645 = ethanol extract 96% 45 min UAE; SN 9660 = ethanol extract 96% 60 min UAE; SN 7030 = ethanol extract 70% 30 min UAE; SN 7045 = Ethanol extract 70% 45 min UAE; SN 7060 = Ethanol extract 70% 60 min UAE; SN 5030 = Ethanol extract 50% 30 min UAE; SN 5045 = Ethanol extract 50% 45 min UAE; SN 5060 = Ethanol extract 50% 60 min UAE; SN 3030 = Ethanol extract 30% 30 min UAE; SN 3045 = Ethanol extract 30% 45 min UAE; SN 3060 = Ethanol extract 30% 60 min UAE.
Fibroblast cell proliferation
Cell growth was observed from the initial stages until the cells developed, adhering to the surface of the cover glass and eventually covering the entire surface in a homogeneous and even manner. Cell harvesting was performed when the number of cells significantly increased during subculture, indicating optimal growth.
Fibroblast cell seeding
Primary fibroblast cultures are first maintained until they reach approximately 70% - 80% confluence, which indicates that the cells are in the log phase and actively proliferating, ready for subculture and use in further experiments. If the density is too low or too high, it can alter the growth rate and phenotypic characteristics of the cells. Once confluence is achieved, the medium is discarded, and the cover glass is rinsed with PBS to remove any residual serum that could inactivate trypsin. Next, add 200 μL of trypsin-EDTA solution that has been warmed to 37 °C and incubate the culture for 3 min until the cells appear to detach, change morphology to become round, and detach from the surface [40]. This process is then stopped by adding complete medium containing serum, which inactivates trypsin and allows the cells to be suspended homogeneously.
The suspension of trypsinized cells was then counted using a hemocytometer. A portion of the aliquot was mixed with trypan blue to distinguish live from dead cells, then placed in a hemocytometer chamber and counted under a microscope according to standard procedures. From these calculations, the cell concentration (cells/mL) and viability percentage were obtained, which in this study showed an increase in the number of cells at the 24, 48 and 72-hour observation points, indicating the success of the subculture process [40,41]. Based on these concentration values, the suspension was then diluted to obtain a uniform number of cells per well, for example, 5×10³ cells/well, and seeded into a 96-well plate for treatment mapping and further analysis. Cell growth is visible in Figure 4.

Figure 4 Growth of NIH-3T3 fibroblast cells.
Optimization of fibroblast cell viability time
Morphological observation of NIH-3T3 fibroblast cells after exposure to the extract revealed that the cells maintained a spindle or cylindrical shape, characterized by elongated and tapered ends. To assess cell proliferation, each sample was treated with 100 ppm of the extract, and the viability percentage was calculated. Cell viability was calculated using the MTT assay, where the rate of viability indicates the proportion of live cells after treatment compared to the control. The results showed that the viability of NIH-3T3 fibroblast cells was high at various concentrations of SN extract. These results are consistent with previous studies, which report that SN is non-toxic to normal cells, such as fibroblasts [42].
The MTT assay is one of the most popular methods for assessing cell viability based on cellular metabolic activity. The principle of this assay is the reduction of water-soluble yellow tetrazolium MTT by mitochondrial dehydrogenase enzymes into insoluble purple formazan crystals [43]. The intensity of the formazan color formed is proportional to the number of live cells, making it a reliable indicator of cell viability or proliferation.
The reduction of MTT does not occur exclusively in mitochondria but may involve various other cellular components, including NAD(P)H and biomolecules such as ascorbic acid, glutathione, and others. Additionally, formazan crystals can be found in various cellular organelles, such as the endoplasmic reticulum, plasma membrane, and nucleus, indicating that the MTT assay reflects metabolic activity rather than solely mitochondrial activity [43,44].
One crucial aspect of the MTT assay is the Optimization of conditions such as cell number, MTT concentration, incubation time, and medium composition. Research conducted by Ghasemi et al. [44] confirmed that before applying the MTT assay, strict control and Optimization are necessary to determine the relationship between absorbance values and cell number. Optimal incubation time yields the most representative data for assessing the effect of a treatment or compound toxicity on cells [38]. Optimization of incubation time (24, 48 and 72 h) is necessary to determine the best proliferation, i.e., when cell viability reaches its maximum [45] (Figure 3). Studies indicate that regenerative effects or viability become apparent after 24 - 72 h, while more prolonged exposure may reduce viability due to cumulative toxic effects. Similarly, red okra fruit ethanol extract exhibited the most optimal proliferation inhibition against HeLa cells at a 48-hour exposure, with the lowest IC50 value of [46]. Prolonged exposure time can lead to the accumulation of compounds that cause toxic effects, resulting in apoptosis or cell necrosis [47,48].
Based on the test results, NIH-3T3 fibroblasts in SN extract samples did not exhibit cell death, as indicated by the absence of structural changes in the cells. Cells maintained a spindle or rod-shaped morphology, characterized by elongated and tapered ends. Upon cell death, the cells did not develop further and shrank [49]. The viability percentage was then calculated, and the EC50 was determined for proliferation. The cell viability results are shown in Figure 4.
From the cell proliferation screening results, the sample with the best viability was SN 9630 extract, followed by SN 9645, with a UAE of 45 min. This extract was then subjected to concentration variations ranging from 3.125 to 400 ppm to determine the EC50 value of the extract. Test cells and controls were cultured in 96-well microplates and incubated for 24 h, followed by the addition of the extract and further incubation for 48 h, as determined by the results of the previous screening test. In this test, a blank reading of the absorbance was used, consisting of medium alone (without cells). The cell test results are shown in Figure 4.
The test results showed that not all fibroblast cells underwent cell death, as indicated by the presence of a purple color on the 96-well microplate. However, at a concentration of 400 ppm, a lighter color formed, indicating inhibited cell growth. This suggests that the SN extract can promote the proliferation of fibroblast cells and prevent aging. Fibroblasts play a crucial role in the production of collagen and elastin, proteins that are essential for maintaining skin elasticity and firmness [1]. Therefore, increased skin cell proliferation is directly related to antiaging properties [26]. Viability results are shown in Figure 5.
The results in Figure 5(a) show that administering SN at 100 ppm with incubation times of 24, 48 and 72 h affects cell viability, an indicator of cell proliferation. The highest cell viability was observed at 48 h of incubation, with a value of > 200%, indicating the optimal increase in fibroblast proliferation at that time. At 72 h of incubation, viability decreased, suggesting that prolonged incubation time can reduce fibroblast viability. Thus, 48 h of incubation is considered the optimal time to achieve the highest cell viability. Several studies have shown that incubation at 48 h is optimal [50,51].
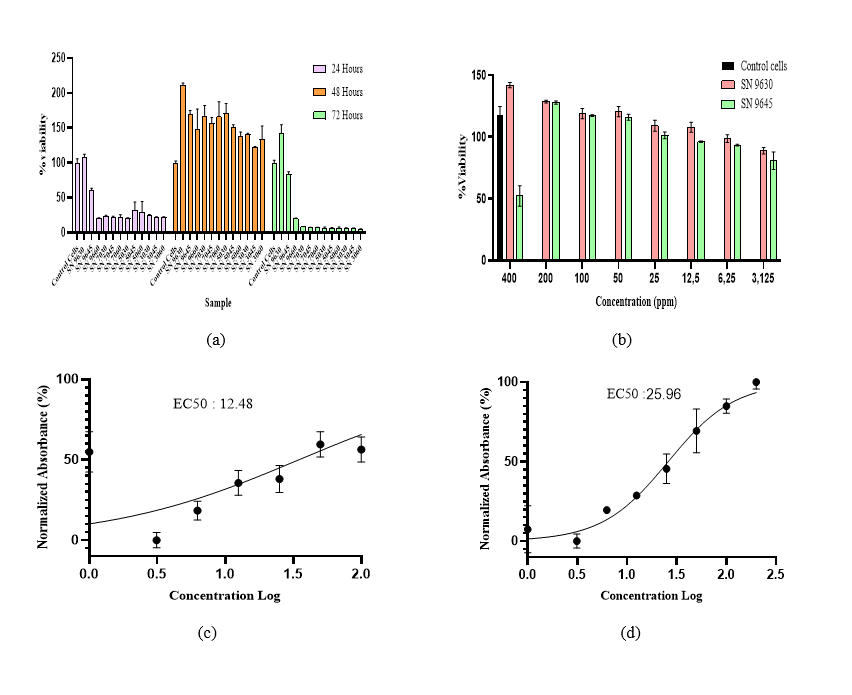
Figure 5 Optimization of cell growth time (a), cell viability concentration series (b), EC50 values of SN 9630(c), SN 9645 (d).
The results of the study indicate that SN 9630 extract at concentrations of 3.125 - 400 ppm was able to induce fibroblast cell proliferation compared to untreated control cells, and SN 9645 at concentrations of 3.125 - 200 ppm was able to induce proliferation. Still, at a concentration of 400 ppm, there was a decrease in cell proliferation. This indicates that the potential effect is dose-dependent. Observations on the samples were continued by calculating the EC50 of the cells, as shown in Figure 5(b). EC50 is used as a parameter to determine the concentration of a substance required to achieve 50% of the maximum effect on fibroblast proliferation [49]. Fibroblasts play a crucial role in skin tissue growth, contributing to the synthesis of proteins such as collagen and elastin, which are primary components of the extracellular matrix (ECM). SN 9630 had an EC50 value of 12.48 ppm. The results of the treatment showed a significant increase in cell viability, as evidenced by a rise from 88% to 141% cell viability. Meanwhile, in SN 9645, the EC50 ppm value was 25.96 ppm, and cell viability ranged from 50% to 127%.
Research by Sooyeon Hong et al. [52] shows that SN significantly reduces the production of proinflammatory cytokines in HaCaT cells stimulated with TNF-α and IFN-γ, inhibits the activation of the p38 MAPK and NF-κB pathways involved in the inflammatory process and skin aging in a mouse model, reduces dermal thickness, lowers serum IgE levels, and reduces mast cell and CD8 T cell infiltration, which are indicators of skin inflammation [52]. Chao-chun et al. research demonstrates that solamargine, derived from Solanum undatum, is effective in treating actinic keratosis (AK) in humans and UVB-induced papillomas in mice. A cream formulation containing solamargin demonstrated improvement in skin aging signs, such as reducing wrinkles, restoring lamin B1, and increasing the production of new elastic fibers [53]. This study shows that SN can inhibit the p38 MAPK and NF κB pathways, and solamargine from Solanum undatum has been proven to reduce inflammation and signs of skin aging, both in mouse models and in humans, by thinning the inflamed dermis, lowering IgE, reducing immune cell infiltration, and improving wrinkles and the elastic fiber structure of the skin.
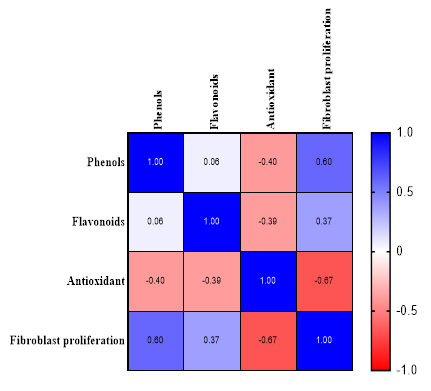
Figure 6 Pearson correlation.
Analysis of cell viability percentage showed that SN extract did not significantly reduce cell viability. This indicates that cell viability testing can serve as an indicator in assessing the safety of natural materials against normal cells. The results of this analysis contribute to the scientific knowledge regarding the safety profile of SN and support previous studies that have stated the low toxicity of the extract on non-cancerous cells.
Pearson’s correlation was used to evaluate the linear relationship between two quantitative variables. In this study, it is crucial to understand the relationship between the content of bioactive compounds (phenols and flavonoids), antioxidant activity, and cellular responses, such as fibroblast proliferation. A positive correlation of r = 0.6 was found between phenols and fibroblasts, indicating that an increase in phenols is associated with an increase in fibroblast cell proliferation. This aligns with previous research suggesting that phenolic compounds can enhance fibroblast proliferation through their antioxidant activity and stimulation of collagen production [54]. Phenolic compounds and flavonoids are the primary contributors to antioxidant activity; however, their effects may vary depending on the specific composition and interactions of compounds within the extract [55].
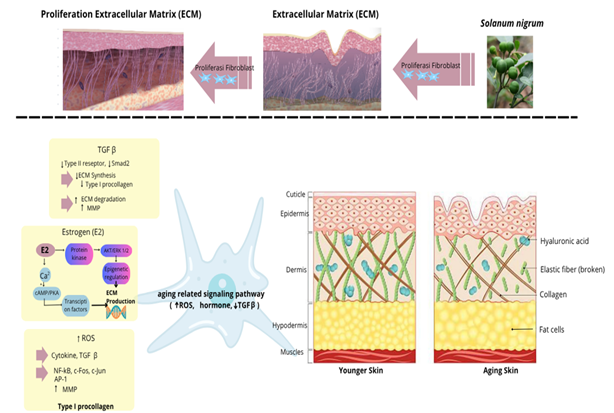
Figure 7 The aging process in fibroblasts.
Fibroblasts are the primary cells in the dermis responsible for producing and maintaining the extracellular matrix (ECM), which includes collagen and elastin. The function and number of fibroblasts play a crucial role in the skin aging process, both intrinsically (due to age and hormonal factors) and extrinsically (due to UV exposure). As age increases, the number of fibroblasts in the skin decreases significantly, resulting in reduced dermal thickness, decreased collagen production, and impaired wound healing [47]. This decline can be caused by senescence, apoptosis, and a decrease in the number of fibroblasts. Fibroblasts undergoing senescence cease dividing and release the senescence-associated secretory phenotype (SASP), which includes the secretion of proinflammatory cytokines, matrix-degrading enzymes (MMPs), and growth factors. SASP leads to collagen degradation, chronic inflammation, and accelerates skin aging [27]. The accumulation of senescent fibroblasts can exacerbate both local and systemic inflammation, thereby accelerating the appearance of aging signs and compromising the skin’s protective function.
Reduced fibroblast activity leads to an imbalance between ECM synthesis and degradation. This results in thinner skin, reduced elasticity, the appearance of wrinkles, and slowed skin regeneration [1,56]. Decreased IGF-1 signaling results in reduced proliferation, collagen synthesis, and tissue repair capacity [47]. Fibroblast proliferation regulation is also influenced by molecular signaling pathways, such as decreased TGF-β, which can promote collagen production, and increased activator protein (AP-1) levels, which inhibit collagen production and stimulate collagen breakdown. Extrinsic aging caused by UV radiation can lead to increased ROS in the dermal layer [57]. These ROS trigger the formation of AP-1, which stimulates the MMP enzyme process involved in collagen degradation. This sequence results in collagen breakdown and reduced collagen production, which constitutes the pathophysiology of skin aging.
Increased fibroblast proliferation under normal conditions is important because fibroblasts are the main cells of the dermis and are responsible for synthesizing and maintaining the extracellular matrix (ECM), particularly collagen, elastin, and proteoglycans. In aging skin, the number and activity of fibroblasts decrease, leading to reduced collagen production, thinning of the dermis, and decreased skin elasticity, which clinically manifests as wrinkles and sagging skin [18]. Thus, agents capable of enhancing the proliferation of normal fibroblasts have the potential to restore fibroblast population density and biosynthetic function, thereby helping to maintain dermal thickness, improve skin firmness, and improve surface texture.
Several studies have shown that stimulation of fibroblast proliferation is accompanied by increased synthesis of collagen and other ECM components, which are directly linked to antiaging effects. Increased fibroblast proliferation and autophagy can improve the structural integrity of the dermis by increasing collagen and elastic fiber production, thereby restoring dermal thickness and density in aging skin [58]. The latest review on collagen also confirms that strategies that stimulate fibroblast growth and activity, for example, through PI3K/Akt or TGF-β activation, increase type I/III collagen expression and can delay skin aging. In vitro studies on collagen peptides and low-molecular-weight recombinant proteins show that treatments that promote fibroblast proliferation also increase the expression of COL1A1, ELN, and other ECM factor genes, as well as strengthen collagen fibers [59]. Thus, increased fibroblast proliferation can be an important indicator of antiaging potential, as these cells become more numerous and more active in rebuilding the extracellular matrix, maintaining tissue homeostasis, and counteracting the thinning and structural damage characteristic of aging skin.
Conclusions
Ultrasound-assisted extraction (UAE) is more advantageous, as it can increase the release of bioactive compounds, shorten extraction time, and reduce solvent use compared to conventional extraction methods. The extraction results yielded an IC₅₀ value for SN extract antioxidant activity of 87.16 - 167.29 ppm, indicating a fairly strong free radical scavenging capacity. SN extract also increased fibroblast cell proliferation, with cell viability > 100% in the MTT assay, and NIH-3T3 fibroblast EC₅₀ values of 12.48 in the SN9630 formula and 25.96 in the SN 9645 formula. Based on these results, SN 9630 was determined to be the best extract, and its high fibroblast cell proliferation activity supports the potential of SN extracts as candidate active ingredients for antiaging cosmetics.
Acknowledgments
This work was funded by Research Grant 2025 from the Health Research Organization, National Research and Innovation Agency (BRIN), Bogor 10340, Indonesia. The author would also like to express gratitude to the Laboratory of Industrial Agro and Biomedical Technology Development, National Research and Innovation Agency (BRIN), Indonesia for providing research facilities
Declaration of Generative AI in Scientific Writing
This manuscript employed a generative artificial intelligence (AI) tool, QuillBot, solely for language and grammar refinement. All scientific content, data analysis, and conclusions presented herein were independently developed by the authors.
CRediT Author Statement
In Rahmi Fatria Fajar: Conceptualization, Methodology, Supervision, Validation, Investigation, Resources, and Writing – original draft. Yesi Desmiaty: Methodology, Data curation, Formal analysis, Investigation, Validation, Resources, Visualization and Writing – review & editing. Kurnia Agustini: Conceptualization, Resources, Methodology, Data curation, Validation, Investigation, Visualization, Project administration, Funding acquisition, and Writing – review & editing. Nuralih nuralih: Data curation, Formal analysis, Investigation, Validation, and Visualization. Effionora Anwar: Data curation, Formal analysis, Investigation, Validation, and Visualization.
References
[1] J Zhang, H Yu, MQ Man and L Hu. Aging in the dermis: Fibroblast senescence and its significance. Aging Cell 2023; 23(2), 14054.
[2] Y Lu, G Pan, Z Wei, Y Li and X Pan. Role of fibroblast autophagy and proliferation in skin anti-aging. Experimental Gerontology 2024; 196, 112559.
[3] L Nan, P Guo, W Hui, F Xia and C Yi. Recent advances in dermal fibroblast senescence and skin aging: Unraveling mechanisms and pioneering therapeutic strategies. Frontiers in Pharmacology 2025; 16, 1592596.
[4] M Wlaschek, P Maity, E Makrantonaki and K Scharffetter-Kochanek. Connective tissue and fibroblast senescence in Skin Aging. Journal of Investigative Dermatology 2021;141(4), 985-992.
[5] X Xiong, W Yang, G Huang and H Huang. Ultrasonic-assisted extraction , characteristics and activity of Ipomoea batatas polysaccharide. Ultrasonics Sonochemistry 2023; 96, 106420.
[6] J Yang, N Li, C Wang, T Chang and H Jiang. Ultrasound-homogenization-assisted extraction of polyphenols from coconut mesocarp: Optimization study. Ultrasonics Sonochemistry 2021; 78, 105739.
[7] Z Xu, Y Cai, Q Ma, Z Zhao, D Yang and X Xu. Optimization of extraction of bioactive compounds from Baphicacanthus cusia leaves by hydrophobic deep eutectic solvents. Molecules 2021; 26(6), 1729.
[8] CP Mungwari, CK King’Ondu, P Sigauke and BA Obadele. Conventional and modern techniques for bioactive compounds recovery from plants: Review. Scientific African 2024; 27, 02509.
[9] PA Uwineza and A Waśkiewicz. Recent advances in supercritical fluid extraction of natural bioactive compounds from natural plant materials. Molecule 2025; 25(17), 3847.
[10] M Saifullah, R McCullum, A McCluskey and Q Vuong. Comparison of conventional extraction technique with ultrasound assisted extraction on recovery of phenolic compounds from lemon scented tea tree (Leptospermum petersonii) leaves. Heliyon 2020; 6(4), 03666.
[11] L Shen, S Pang, M Zhong, Y Sun, A Qayum, Y Liu, A Rashid, B Xu, Q Liang, H Ma and X Ren. A comprehensive review of ultrasonic assisted extraction (UAE) for bioactive components: Principles, advantages, equipment, and combined technologies. Ultrasonics Sonochemistry 2023; 101, 106646.
[12] A Carreira-Casais, P Otero, P Garcia-Perez, P Garcia-Oliveira, AG Pereira, M Carpena, A Soria-Lopez, J Simal-Gandara and MA Prieto. Benefits and drawbacks of ultrasound-assisted extraction for the recovery of bioactive compounds from marine algae. International Journal of Environmental Research and Public Health 2021; 18(17), 9153.
[13] I Călinescu, M Vinatoru, D Ghimpețeanu, V Lavric and TJ Mason. A new reactor for process intensification involving the simultaneous application of adjustable ultrasound and microwave radiation. Ultrasonics Sonochemistry 2021; 77, 105701.
[14] RS Hussein, SBin Dayel, O Abahussein and AA El-Sherbiny. Influences on skin and intrinsic aging: Biological, environmental, and therapeutic insights. Journal of Cosmetic Drmatology 2025; 24(2), 16688.
[15] X Chen, X Dai, Y Liu, Y Yang, L Yuan, X He and G Gong. Solanum nigrum Linn.: An insight into current research on traditional uses, phytochemistry, and pharmacology. Frontiers in Pharmacology 2022; 13, 918071.
[16] A Aouadi, DH Saoud, A Rebiai, MH Ibrahim, M Messaoudi, K Alia, H Zidane, AV Atoki and FMA El-Mordy. Chemical composition’s effect on Solanum nigrum Linn.’s antioxidant capacity and erythrocyte protection: Bioactive components and molecular docking analysis. Open Life Sciences 2024; 19(1), 20220944.
[17] A Campisi, R Acquaviva, G Raciti, A Duro, M Rizzo and NA Santagati. Antioxidant activities of Solanum nigrum L. Leaf extracts determined in in vitro cellular models. Foods 2019; 8(2), 63.
[18] A Zorina, V Zorin, A Isaev, D Kudlay, M Vasileva and P Kopnin. Dermal fibroblasts as the main target for skin anti-age correction using a combination of regenerative medicine methods. Current Issues in Molecular Biology 2023; 45(5), 3829-3847.
[19] J Hao, Z Wang, Y Jia, L Sun, Z Fu, M Zhao, Y Li, N Yuan, B Cong, L Zhao and G Ge. Optimization of ultrasonic-assisted extraction of flavonoids from Lactuca indica L. cv. Mengzao and their antioxidant properties. Frontiers in Nutrition 2023; 10, 1065662.
[20] A El-Baakili, M Fadil and NE Es-Safi. Ultrasonic-assisted extraction for phenolic compounds and antioxidant activity of Moroccan Retama sphaerocarpa L. leaves: Simultaneous optimization by response surface methodology and characterization by HPLC/ESI-MS analysis. Heliyon 2023; 9(6), 17168.
[21] S Baliyan, R Mukherjee, A Priyadarshini, A Vibhuti, A Gupta, RP Pandey and CM Chang. Determination of antioxidants by DPPH radical scavenging activity and quantitative phytochemical analysis of Ficus religiosa. Molecules 2022; 27(4), 1326.
[22] V Buranasudja, D Rani, A Malla, K Kobtrakul and S Vimolmangkang. Insights into antioxidant activities and anti-skin-aging potential of callus extract from Centella asiatica (L.). Scientific Reports 2021; 11, 13459.
[23] AM Cruz, MC Gonçalves, MS Marques, F Veiga, AC Paiva-Santos and PC Pires. In vitro models for antiaging efficacy assessment: A critical update in dermocosmetic research. Cosmetics 2023; 10(2), 66.
[24] A Nurkhasanah, T Fardad, C Carrera, W Setyaningsih and M Palma. Ultrasound-assisted anthocyanins extraction from pigmented corn: Optimization using response surface methodology. Methods Protocols 2023; 6(4), 69.
[25] K Kumar, S Srivastav and VS Sharanagat. Ultrasound assisted extraction (UAE) of bioactive compounds from fruit and vegetable processing by-products: A review. Ultrasonics Sonochemistry 2021; 70, 105325.
[26] G Psakis, F Lia, VP Valdramidis and R Gatt. Exploring hydrodynamic cavitation for citrus waste valorisation in Malta: From beverage enhancement to potato sprouting suppression and water remediation. Frontiers in Chemistry 2024; 12, 1411727.
[27] K Papoutsis, P Pristijono, JB Golding, CE Stathopoulos, MC Bowyer, CJ Scarlett and QV Vuong. Optimizing a sustainable ultrasound-assisted extraction method for the recovery of polyphenols from lemon by-products: Comparison with hot water and organic solvent extractions. European Food Research and Technology 2018; 244, 1353-1365.
[28] NVT Nguyen, CV Nguyen, NT Duong, XTT Dai, KT Nguyen and CTT Le. Optimization of ultrasound-assisted extraction using response surface methodology and quantification of polyphenol compounds in Avicennia officinalis L. from Vietnam. Pharmacia 2024; 71, 115528.
[29] AC Andrade, FT Borsoi, ASMC Saliba, SM de Alencar, GM Pastore and HS Arruda. Optimization of Ultrasonic-assisted extraction of phenolic compounds and antioxidant activity from araticum peel using response surface methodology. Plants 2024; 13(18), 2560.
[30] M Sukma, AWR Akili1, A Hardianto, J Latip and T Herlina. Ultrasound-assisted extraction of flavonoids from Erythrina crista-galli twigs using natural deep eutectic solvents: Process optimization and antioxidant evaluation. Trends in Sciences 2025; 22(12), 11099.
[31] E Herrera-Pool, AL Ramos-Díaz, MA Lizardi-Jiménez, S Pech-Cohuo, T Ayora-Talavera, JC Cuevas-Bernardino, U García-Cruz and N Pacheco. Effect of solvent polarity on the Ultrasound Assisted extraction and antioxidant activity of phenolic compounds from habanero pepper leaves (Capsicum chinense) and its identification by UPLC-PDA-ESI-MS/MS. Ultrasonics Sonochemistry 2021; 76, 105658.
[32] JDC Gomes, FR Procópio, AC Mendonça, LM Vieira, ÍT Perrone, FAR de Barros and PC Stringhet. Optimization of ultrasound-assisted extraction of phenolic compounds from jussara (Euterpe edulis M.) and blueberry (Vaccinium myrtillus) fruits. Food Science and Technology 2018; 38(1), 36316.
[33] M Pérez, I Dominguez-Lopez and RM Lamuela-Raventos. The chemistry behind the folin-ciocalteu method for the estimation of (poly)phenol content in food: Total phenolic intake in a mediterranean dietary pattern. Journal of Agricultural and Food Chem 2023; 71(46), 17543-17553.
[34] M Zugazua-Ganado, A Bordagaray, J Ezenarro, R Garcia-Arrona, M Ostra and M Vidal. Adaptation of the Folin-ciocalteu and fast blue BB spectrophotometric methods to digital image analysis for the determination of total phenolic content: Reduction of reaction time, interferences and sample analysis. LWT 2023; 193, 115756.
[35] H Bouakline, S Bouknana, M Merzouki, I Ziani, A Challioui, M Bnouham, A Tahani and AEL Bachiri. The Phenolic content of pistacia lentiscus leaf extract and its antioxidant and antidiabetic properties. Scientific World Journal 2024; 2024, 1998870.
[36] B Poojar, B Ommurugan, S Adiga, H Thomas, RK Sori, B Poojar, N Hodlur, A Tilak, R Korde and P Gandigawad. Methodology used in the study. Asian Journal Pharmaceutical Clinical Research 2017; 7(10), 4103.
[37] SK Zaidi, MN Hoda, S Tabrez, SA Ansari, MA Jafri, MS Khan, S Hasan, MH Alqahtani, AM Abuzenadah and N Banu. Protective effect of Solanum nigrum leaves extract on immobilization stress induced changes in rat’s brain. Evidence-Based Complementarary and Alternative Medicine 2014; 2014, 912450.
[38] Y Desmiaty, FC Saputri, M Hanafi, R Prastiwi and B Elya. Anti-elastase, anti-tyrosinase, and anti-oxidant of Rubus fraxinifolius stem methanolic extract. Pharmacognosy Journal 2020; 12(2), 271-275.
[39] M Veerapagu, KR Jeya, A Sankaranarayanan and A Rathika. In vitro antioxidant properties of methanolic extract of Solanum nigrum L fruit. The Pharma Innovation Journal 2018; 7(5), 371-374.
[40] H Brem, MS Golinko, O Stojadinovic, A Kodra, RF Diegelmann, S Vukelic, H Entero, DL Coppock and M Tomic-Canic. Primary cultured fibroblasts derived from patients with chronic wounds : A methodology to produce human cell lines and test putative growth factor therapy such as GMCSF. Journal of Translational Medicine 2008; 6, 75.
[41] H Moravvej, H Memariani, M Memariani, M Kabir-salmani, A Shoae-hassani and F Abdollahimajd. Evaluation of fibroblast viability seeded on acellular human amniotic membrane. Biomed Research Internasional 2021; 2021, 5597758.
[42] A Nawaz, A Jamal, A Arif and Z Parveen. In vitro cytotoxic potential of Solanum nigrum against human cancer cell lines. Saudi Journal Biological Sciences 2021; 28(8), 4786-4792.
[43] K Buranaamnuay. The MTT assay application to measure the viability of spermatozoa: A variety of the assay protocols. Open Veterinary Journal 2021; 11(2), 251-269.
[44] M Ghasemi, T Turnbull, S Sebastian and I Kempson. The mtt assay: Utility, limitations, pitfalls, and interpretation in bulk and single-cell analysis. Internasional Journal Molecular Sciences 2021; 22(23), 12827.
[45] RM da Gama, AA Ruggiero, GP Andrade, J Armando-Junior and MAS Pinhal. Optimizing bioactive compounds extraction and cell viability effect of Tithonia diversifolia (Hemsl.) A. Gray dry flower extracts. Journal of Pharmacy and Pharmacognosy Researchs 2025; 13(5), 1313-1326.
[46] AB Achmad. In vitro cytotoxicity test of red okra (abelmoschus esculentus) fruit ethanolic extract on HeLa cells. Journal of Applied Veterinary Science and Technology 2022; 3(1), 22-26.
[47] GA Lozano-Casabianca, SS Arango-Varela, J Aguillón-Osma, MA Llano-Ramírez and ME Maldonado-Celis. Inhibition of cell proliferation and induction of cell cycle arrest in colon cancer cells by lyophilized Mango (Mangifera indica L.) pulp extract. Preventive Nutrition and Food Science 2022; 27(4), 436-447.
[48] R Bar-Shalom, M Bergman, S Grossman, N Azzam, L Sharvit and F Fares. Inula viscosa extract inhibits growth of colorectal cancer cells in vitro and in vivo through induction of apoptosis. Frontiers in Oncology 2019; 9, 227.
[49] S Kumar, VK Maurya, SV Chitti, R Kabir, K Shanker, D Nayak, A Khurana, RK Manchanda, S Gadugu, V Kumar and SK Saxena. Wound healing activity of a novel formulation SKRIN via induction of cell cycle progression and inhibition of PCNA-p21 complex interaction leading to cell survival and proliferation. ACS Pharmacology and Translational Science 2021; 4(1), 352-364
[50] S Manmuan, S Tubtimsri, N Chaothanaphat, N Issaro HM Tantisira and P Manmuan. Determination of the anticancer activity of standardized extract of Centella asiatica (ECa 233) on cell growth and metastatic behavior in oral cancer cells. Research in Pharmaceutical Sciences 2024; 19(2), 121-147.
[51] NM Alfarafisa, AF Khairani, A Achadiyani, Y Chou, F Aulia and MB Firdaus. Assessment of alpha-tocopheryl acetate and metformin hydrochloride as independent agents on human dermal fibroblast viability: Findings from MTT Assay. Althea Medical Journal 2025; 12(2), 125-132.
[52] S Hong, B Lee, JH Kim, EY Kim, M Kim, B Kwon HR Cho, Y Sohn and HS Jung. Solanum nigrum Linne improves DNCB ‑ induced atopic dermatitis ‑ like skin disease in BALB /c mice. Molecular Medicine Reports 2020; 22(4), 2878-2886.
[53] CC Yang, CH Wu, TW Wong, FJ Lai, KC Wei, TK Lin, CS Lin, J Zhao, L Xiang, CH Kao, YJ Wang, CCE Lan and HM Sheu. Effects of Solanum undatum extract (SR-T100) on photocarcinogenesis and photoaging of actinic keratosis. The Journal of Dermatology 2021; 48(3), 344-352.
[54] A Merecz-Sadowska, P Sitarek, E Kucharska, T Kowalczyk, K Zajdel, T Cegliński and R Zajdel. Antioxidant properties of plant-derived phenolic compounds and their effect on skin fibroblast cells. Antioxidants 2021; 10(5), 726.
[55] YM Muflihah, G Gollavelli and YC Ling. Correlation study of antioxidant activity with phenolic and flavonoid compounds in 12 indonesian indigenous herbs. Antioxidants 2021; 10(10), 1530.
[56] TY McCarty and CJ Kearney. Human dermal fibroblast senescence in response to single and recurring oxidative stress. Frontier in Aging 2025; 6, 1504977.
[57] P Singh, SS Bhat, N Singh, BU Venkanna, R Mohamed and RP Rao. Cell-based model systems for validation of various efficacy-based claims for cosmetic ingredients. Cosmetics 2022; 9(5), 107.
[58] F Boraldi, FD Lofaro, S Bonacorsi, A Mazzilli, M Garcia-fernandez and D Quaglino. The role of fibroblasts in skin homeostasis and repair. Biomedicines 2024; 12(7), 1586.
[59] S Dierckx, M Patrizi, M Merino, S González, JL Mullor and R Nergiz-Unal. Collagen peptides affect collagen synthesis and the expression of collagen, elastin, and versican genes in cultured human dermal fibroblasts. Frontiers in Medicine 2024; 11, 1397517.