
Trends
Sci.
2026;
23(7):
12834
Antioxidant Properties of Oryza sativa var. Luem Pua Rice Wine in Alcoholic and Dealcoholized Forms: In HepG2 Cell Studies with Identification of Health-Beneficial Probiotics
Supawadee Srisuwan1, Warachin Gangnonngiw2,3, Benjamart Pratoomthai1,
Direk Aekthammarat1, Surachai Ngamratanapaiboon1 and Sirilug Magerd1,*
1Department of Basic Medical Science, Faculty of Medicine Vajira Hospital, Navamindradhiraj University,
Bangkok 10300, Thailand
2National Center for Genetic Engineering and Biotechnology (BIOTEC),
National Science and Technology Development Agency, Klong Luang, Pathum Thani 12120, Thailand
3Centex Shrimp, Faculty of Science, Mahidol University, Bangkok 10400, Thailand
(*Corresponding author’s e-mail: [email protected])
Received: 28 November 2025, Revised: 16 December 2025, Accepted: 23 December 2025, Published: 10 March 2026
Abstract
Luem Pua black glutinous rice (Oryza sativa L. var. Luem Pua), rich in anthocyanins and phenolics with antioxidant properties. Fermented plant-based beverages contain these compounds and may support probiotic growth. This study investigates Luem Pua rice wine in alcoholic (LPW) and dealcoholized (DLPW) forms over 0-28 days of fermentation, analyzing antioxidants, phenolic/anthocyanin content, cytotoxicity, ROS reduction, and probiotic growth. Phenolic content peaked at 3 days, slightly decreasing later, with DLPW showing higher levels than LPW. DLPW3 and LPW7 had the highest anthocyanins, strong DPPH scavenging, and FRAP values. Cytotoxicity tests showed DLPW3 (20 %v/v) was non-toxic to HepG2 cells, while LPW7 (2.13% ethanol) reduced viability at 48 h. Both, with 0.5 - 1.5 µg/mL anthocyanins, reduced ROS, with higher concentrations more effective. Lactobacillus buchneri, Pediococcus acidilactici, and Lactobacillus fermentum were identified, with higher counts in LPW7. Despite alcohol removal, DLPW3 retained strong bioactivity, highlighting its potential as a nutraceutical, alcohol-free alternative.
Keywords: Luem Pua rice, Wine, Dealcoholized wine, Antioxidant activity, Probiotic microorganisms, Functional beverage, Nutraceutical
Introduction
Luem Pua black glutinous rice (Oryza sativa L. var. Luem Pua), developed as a pure breed by the Phitsanulok Rice Research Center, is a Thai rice variety rich in anthocyanins, which give it its distinctive dark purple or black color. The major anthocyanins include cyanidin-3-glucoside (2,359.93 mg/kg) and peonidin-3-glucoside (324.37 mg/kg), contributing to its potent antioxidant activity. Other antioxidants like catechin, rutin, and tannic acid have also been identified [1]. Previous studies have demonstrated its potential health benefits, such as free radical scavenging in the SK-N-SH neuroblastoma cell line, memory enhancement in
animal models [2], and improved gut transit time with Luem Pua rice tea consumption [3]. Additionally, roasted Luem Pua rice was found to support the growth of the probiotic Lactobacillus amylovorus, beneficial for gut health [4].
Fermented beverages, produced by yeast-driven sugar-to-alcohol conversion, date back to 6,000 BCE in Mesopotamia. Today, many countries leverage local agricultural products for producing these beverages [5]. Moderate consumption of fermented drinks, such as beer and wine, has been associated with reduced risks of cardiovascular disease, cancer prevention, improved bone density, obesity management, and type 2 diabetes [6-9]. These effects are attributed to bioactive compounds like phenolic acids, polyphenols, and probiotics (Lactobacillus, Pediococcus, and Oenococcus oeni) present in fermented beverages [7,8]. Currently, dealcoholized wines, defined by the U.S. Food and Drug Administration (FDA) as non-alcoholic beverages containing less than 0.5% alcohol by volume (ABV) [10], have emerged as a promising alternative for reducing alcohol consumption and serving individuals unable to consume alcohol.
From the aforementioned research report, the Luem Pua black glutinous rice variety exhibits significant bioactive properties influenced by its processing methods. This study aims to investigate the properties of wine produced from this rice, both in alcoholic and dealcoholized forms, by analyzing phenolic compounds, antioxidant activity, and its effect on probiotic growth. These findings could provide a foundation for developing healthier alternatives to traditional beverages while leveraging the nutritional potential of LP rice.
Materials and methods
Preparation of Luem Pua rice Wine (LPW)
The unpolished sticky Thai rice strain of Luem Pua (LP rice) was purchased from a local farm located in Phitsanulok province, Thailand. 6 kg of LP rice was steamed until cooked and left to cool before being mixed well with 85 g of traditional starter culture called “lookpang”, a mixture of starch, yeast (Zygosaccharomyces sp), and mold (Rhizopus oryzae). The streamed rice was divided into 6 equal parts, then put in 6 plastic fermentation tanks, and 500 mL of distilled water was added to each tank. Each tank was fermented for different durations including 1 h (day 0), 3, 7, 14, 21, and 28 days, at room temperature (25 ± 1 °C), and in the absence of light. Samples were collected from each tank at the end of the fermentation period and stored at a temperature of 2 - 4 °C for one week to inhibit activity and precipitate out the yeast. Each of the wine samples was then carefully extracted to avoid disturbing the settled yeast and stored at 2 - 4 °C for further experimentation.
Preparation of Dealcoholized LP rice Wines (DLPW)
50% of each wine sample (except day 0) was subjected to dealcoholization using a rotary evaporator (Laborota 4003, Heidolph, Schwabach, Germany) to obtain a final alcohol content below 0.5% ABV. Each sample was transferred into a 1-L round-bottom flask and heated in a water bath maintained at 45 - 50 °C, under continuous rotation at 130 rpm. Vacuum was applied to achieve a reduced pressure of 60 - 100 mBar. Distillation was carried out for 90 min. Sterile distilled water was added to each sample to restore the original volume. The dealcoholized samples were then cooled to room temperature, and stored at 4 °C until further analysis.
Measurement of ethanol, sugar, and pH of wines
All wine samples, both LPW and DLPW, were analyzed for ethanol content using Gas Chromatography (GC) technique. Injection volume was 1 µL, the injection and detector temperatures were 250 and 280 °C, respectively. Helium was used as carrier gas at a pressure of 150 kPa. The oven temperature was from 50 - 200 °C (10 °C /min). The Sucrose/D-Fructose/D-Glucose test kit (K-SUFRG, Megazyme) was used for the measurement and analysis of sucrose, D-glucose and D-fructose in wines. The sugar content was calculated using the Mega-Calc™ software, supported by the Megazyme company. The monitoring of pH values of wines was performed with pH meter (Mettler Toledo FEP20 FiveEasyTM Plus Laboratory Benchtop pH Meter).
Measurement of total phenolic content and total monomeric anthocyanin content
For measuring total phenolic content, 20 µL of each wine was mixed with 100 µL Folin-Ciocalteu reagent, and 80 µL of 7.5 %(w/v) Na2CO3 was added. The mixture was stirred and allowed to react for 30 min. Absorbance was then measured using a spectrophotometer at a wavelength of 765 nm, and the resulting values were compared the Gallic acid standard [11].
The total monomeric anthocyanin content was determined by using the pH differential technique. This was adapted from the AOAC official method (2005) [12]. 50 µL of wine was diluted with 200 µL of potassium chloride buffer (pH 1.0), and sodium acetate buffer (pH 4.5). The absorbance at wavelengths of 520 and 700 nm was measured to calculate the total monomeric anthocyanin content.
In vitro antioxidant activities of wines
2,2-Diphenyl-1-picrylhydrazyl (DPPH) assay
Each sample was prepared by performing a two-fold serial dilution with 95% ethanol in a 96-well plate. 20 µL of each sample was mixed with 190 µL of 2,2-Diphenyl-1-picrylhydrazyl (DPPH) solution (2 µg/mL) and incubated at room temperature in the dark for 30 min. Ascorbic acid (0 - 1 mg/mL) served as the positive control, while ethanol was used as the blank control. After incubation, absorbance was measured at 517 nm using a spectrophotometer. The percentage of scavenging was calculated and used to generate a four-parameter non-linear regression for IC50 determination. All experiments were conducted in triplicate [13,14].
Ferric Reducing Antioxidant Power (FRAP) assay
The Fe3+ reduction capacity of each sample was evaluated using a modified FRAP assay, with ascorbic acid as an antioxidant control agent. Each extract was prepared in serial dilutions and loaded into a 96-well plate. For each sample, 10 µL of the diluted extract was mixed with 25 µL of phosphate buffer (pH 6.0) and 25 µL of 1% potassium ferricyanide solution. After incubation at 50 °C for 30 min, 10 µL of 10% TCA was added to halt the reaction, followed by centrifugation to remove precipitates. Next, 25 µL of distilled water and 10 µL of 0.1% FeCl3 solution were added to each well. Absorbance was measured at 700 nm using a microplate reader. Ascorbic acid was used as a positive control to gauge antioxidant activity, and IC50 values were calculated from non-linear regression analysis of % scavenging [14].
2,2’-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) assay
To prepare the ABTS radical cation solution, 7 mM ABTS was mixed with 2.45 mM potassium persulfate in deionized water and incubated in the dark at 25 °C for 12 - 16 h. The resulting solution was diluted 1:10 (v/v) with deionized water and then mixed with the sample. Absorbance was measured at 734 nm, and the % scavenging activity was calculated relative to the control and expressed as IC50 values. All experiments were conducted in triplicate [13].
Effect of wines on HepG2 cells viability using 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay
The highest antioxidant properties from both the LPW and DLPW groups, based on in vitro experiments, were selected to determine their cytotoxic effects on HepG2, a hepatocellular carcinoma cell line, as the liver is the primary organ for alcohol metabolism and is highly susceptible to damage caused by chronic alcohol consumption. The cell viability was examined using the MTT assay. HepG2 cells (ATCC HB-8065), were seeded in a 96-well plate (1×104 cells/well) and cultured in 10% of fetal bovine serum (FBS), 1% antibiotic in Dulbecco’s Modified Eagle Medium (DMEM) for 24 h. Then HepG2 cells were treated with LPW or DLPW (0 - 20 %v/v, final conc.), filtered through a 0.45 µm filter paper, compared with ethanol (0 - 10 %v/v, final conc.), as a negative control, for 24 and 48 h. Following treatment, the culture media was removed from all samples and the plate was incubated with MTT solution (0.5 mg/mL in PBS) for 3 h at 37 °C. After discarding the supernatant, the formazan product was dissolved in DMSO, and the color intensity was measured at 570 nm using a microplate reader (TECAN, Männedorf, Switzerland) following a 15 min incubation at 37 °C. The non-cytotoxic concentrations of LPW and DLPW were then selected to evaluate their ability to reduce reactive oxygen species (ROS) generation in HepG2 cells.
Reducing ROS production effect of wines in HepG2 cells
HepG2 cells were seeded in a 96-well black plate (1×104 cells/well) and cultured in 180 µL DMEM and incubated at 37 °C equilibrated with 95% air and 5% CO2 overnight. The cells were incubated with either 20 µL LPW or DLPW at three different anthocyanin concentrations, with final concentration of ethanol which did not induce cell death as determined by the MTT assay, for 24 h. 100 µL DCFHDA (2’,7’-dichlorodihydrofluorescein diacetate) assay working solution (ROS Assay Kit-Highly Sensitive DCFHDA-R252) was added and incubated for 30 min. The media in each well was removed, then 100 µL of DMEM (without serum and antibiotic) was added. The fluorescence intensity of DCF was quantified by iD3 Microplate Reader (SpectraMax®) at 485 nm excitation and 530 nm emission wavelengths.
Measurement of probiotics in wines
This analysis aimed to determine both the total lactic acid bacterial (LAB) count and the probiotic species present in the LPW7 and DLPW3 samples. Total LAB were quantified using the standard method of culturing on MRS agar supplemented with CaCO₃, which allows visualization and quantification of acid-producing colonies. Presumptive LAB isolates were then subjected to phenotypic identification using the API identification system. All procedures were conducted by the Biodiversity Research Centre, Thailand Institute of Scientific and Technological Research (TISTR).
Statistical analysis
All results are presented as mean ± SD. Statistical comparison between control and treatment groups were analyzed with one-way analysis of variance (ANOVA) followed by post-hoc Tukey’s test. All statistical analysis was performed using GraphPad Prism 10 software. Results were considered to be statistically significant at p-value < 0.05.
Results and discussion
Measurement of ethanol, sugar, and pH of wines
The LP rice wine preparations at different fermentation durations resulted in varying ethanol content (Figure 1(A)). The experiment shows that after 1 h (day 0) of fermentation, the ethanol content was 0.01%, as the process of converting starch to sugar precedes the conversion of sugar to ethanol during fermentation by yeast. Therefore, only sugar and no ethanol are detected in the first hour of fermentation. Ethanol levels gradually increase after 3 (9.41 ± 0.25), 7 (10.67 ± 0.15), and 14 (11.69 ± 0.07) days of fermentation, then stabilize by day 21 (11.65 ± 0.16) then slightly decrease by day 28 (11.20 ± 0.13). This correlates with a significant reduction in sugar levels on days 21 and 28 of fermentation (Figure 1(B)).
The wine samples that underwent alcohol removal had an ethanol content of less than 0.025%, which aligns with the U.S. Food and Drug Administration (FDA) (2005) definition of a non-alcoholic beverage, requiring less than 0.5% alcohol by volume (ABV). It was observed that the reduction in ethanol content did not affect sugar levels, as the sugar content in the DLPW group was no different from the LPW group fermented for the same duration (Figure 1(B)). The pH levels of all wines were slightly acidic and showed no other significant differences.
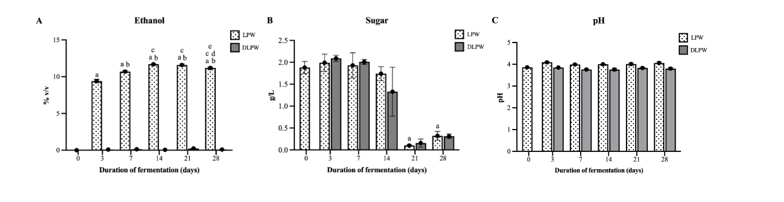
Figure 1 The profiles of Leum Pua rice wines, both alcoholic (LPW) and dealcoholized (DLPW) forms, with different fermentation durations. (A) Ethanol content in wines, (B) Sugar content in wines, and (C) pH levels of wines. Data are expressed as the mean ± SD. The letters above each column indicate the significant difference compared to LPW0 (a); LPW3 (b); LPW7 (c); LPW14 (d); LPW21 (e).
Total phenolic and total monomeric anthocyanin content
The total phenolic and total monomeric anthocyanin contents in rice wine fermented for 3, 7, 14, 21, and 28 days were significantly higher than those in wine fermented for only 1 h (Figure 2). The phenolic content of LPW3 was the highest, gradually decreasing in wines fermented for 7, 14, 21, and 28 days. For DLPW, the total phenolic content was slightly higher than LPW fermented for the same duration but was significantly higher in wines fermented for 3, 21, and 28 days (Figure 2(A)). Regarding total monomeric anthocyanin (Figure 2(B)), the highest levels were observed in LPW7, which decreased gradually in wines fermented for 14, 21, and 28 days. Interestingly, the total monomeric anthocyanin in DLPW3 was higher than LPW3, while dealcoholized wines had lower total monomeric anthocyanin levels compared to alcoholic wines fermented for the same duration. The experimental results were aligned with previous findings in grape wine studies, where phenolic and anthocyanin extraction was highest during early maceration (6 - 10 days), Subsequently, the levels of total phenolic and total anthocyanin gradually decreased [15,16]. Specifically, anthocyanins have been reported to undergo a wide variety of reactions and associations during fermentation due to the presence of ethanol in the wine, which induces monomeric anthocyanins to transform and form various anthocyanin-derived, stable compounds like pyranoanthocyanins and polymeric anthocyanins [17], which could not be investigated by the pH differential technique. In the sample of the wine fermented for 3 days, DLPW3 showed a significantly higher monomeric anthocyanin content compared to LPW3. We hypothesize that during the early stages of wine fermentation, there were still sufficient anthocyanins present on the rice grains which could continuously dissolve into the wine. Furthermore, the ethanol produced at this stage (9.41 %v/v) was not yet at a very high concentration before it was removed, resulting in a limited capacity to facilitate the essential polymerization and condensation reactions required for the conversion of monomeric anthocyanins into polymeric anthocyanins.
However, the observed decrease in total monomeric anthocyanin in DLPW7-28 suggests a different degradation mechanism. While reports exist on the thermal lability of anthocyanins, the dealcoholization process in this study utilized a comparatively mild temperature range of 45 - 50 °C for 90 min. This temperature is significantly below the typical range reported to induce substantial anthocyanin degradation, which is often cited as 80 - 180 °C [18]. Furthermore, this conclusion is supported by the data from the DLPW3 sample, which underwent the identical temperature and time conditions for alcohol removal as DLPW7-28 yet showed no reduction in total monomeric anthocyanin content. Therefore, the observed decrease in total monomeric anthocyanin in DLPW7-28 is highly likely attributed to the loss of ethanol. Specifically, the depletion of the ethanol matrix may reduce the stability of anthocyanins, potentially leading to their increased oxidation or hydrolytic degradation [19]. Additionally, a minor fraction of the anthocyanins may have been physically volatilized and removed alongside the alcohol during the dealcoholization process.
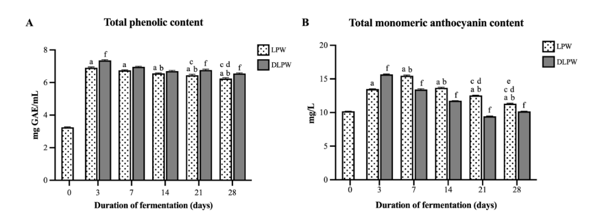 Figure
2
(A) Total phenolic content of wine fermented for different
durations, and (B) Total monomeric anthocyanin content of wine
fermented for different durations. Data is expressed as the mean ±
SD. The letters above each column indicate the significant
difference compared to LPW0 (a); LPW3 (b); LPW7 (c); LPW14 (d);
LPW21 (e); and (f) indicates a significant difference compared to
LPW fermented for the same duration.
Figure
2
(A) Total phenolic content of wine fermented for different
durations, and (B) Total monomeric anthocyanin content of wine
fermented for different durations. Data is expressed as the mean ±
SD. The letters above each column indicate the significant
difference compared to LPW0 (a); LPW3 (b); LPW7 (c); LPW14 (d);
LPW21 (e); and (f) indicates a significant difference compared to
LPW fermented for the same duration.
In vitro antioxidant activities of wines
The antioxidant properties of the wines were analyzed using DPPH, FRAP, and ABTS methods in vitro. It was found that the lowest concentration of wine that could scavenge 50% (IC50) of DPPH radicals was observed in LPW7 (9,087 ± 3,328 ppm) and LPW14 (9,087 ± 3,328 ppm), while in the dealcoholized wine group, the concentrations were DLPW7 (18,943 ± 841 ppm) and DLPW3 (21,644 ± 772 ppm) (Figure 3(A)). However, these concentrations are still considered high compared to the standard, Vitamin C, which scavenged 50% of DPPH radicals at a concentration of 181.10 ± 7.92 ppm (data did not show). Antioxidant activity analyzed using the FRAP method showed that LPW7 had the highest FRAP scavenging capacity with an IC50 value of 51,473 ± 5,628 ppm. In the dealcoholized wine group, DLPW21 and DLPW3 exhibited the best antioxidant activity, as indicated by IC50 values of 60,985 ± 10,117 ppm and 66,958 ± 7,409 ppm, respectively (Figure 3(B)). Testing with the ABTS method revealed that all wine groups exhibited strong antioxidant properties, with significantly lower IC50 values compared to LPW0 (Figure 3(C)). LPW7 and DLPW3 exhibited the highest monomeric anthocyanin content, demonstrating outstanding antioxidant properties based on DPPH and FRAP assays. This aligns with previous studies reporting that monomeric anthocyanins play a major role in the antioxidant activity of various plants [20]. However, other wine conditions, such as LPW14, also demonstrated significant DPPH scavenging and FRAP reducing potential. This may be attributed to polymeric compounds formed during the wine fermentation process, which compensated for the loss of antioxidant capacity due to the degradation of monomeric anthocyanins [21]. Based on the results of total phenolic and total anthocyanin content combined with the antioxidant activities in vitro, LPW7 and DLPW3 were selected for further testing of cytotoxicity and antioxidant effects in HepG2 liver cell cultures.
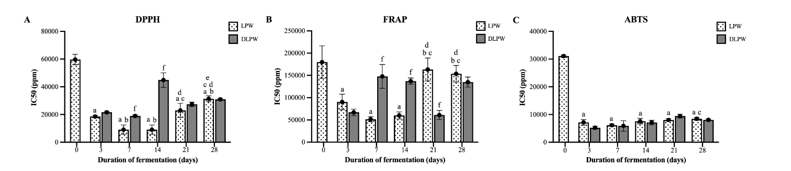 Figure
3
The antioxidant properties of the wines in
vitro
were analyzed using (A) DPPH, (B) FRAP, and (C) ABTS methods. Data
are expressed as the mean ± SD. The letters above each column
indicate the significant difference compared to LPW0 (a); LPW3 (b);
LPW7 (c); LPW14 (d); LPW21 (e); and (f) indicates a significant
difference compared to LPW fermented for the same duration.
Figure
3
The antioxidant properties of the wines in
vitro
were analyzed using (A) DPPH, (B) FRAP, and (C) ABTS methods. Data
are expressed as the mean ± SD. The letters above each column
indicate the significant difference compared to LPW0 (a); LPW3 (b);
LPW7 (c); LPW14 (d); LPW21 (e); and (f) indicates a significant
difference compared to LPW fermented for the same duration.
Effect of wines on HepG2 cells viability using MTT assay
Cytotoxicity testing of LP rice wine on HepG2 cells was performed using the MTT assay technique, compared to ethanol standard concentrations ranging from 0 - 10 %v/v, which was equivalent to 0 - 10 %eth v/v. The MTT assay revealed that ethanol standards at low concentrations 0.14 - 0.25 %v/v induced cell proliferation when treated for 24 and 48 h. However, high ethanol concentrations led to a reduction in cell numbers, that HepG2 cells treated for 48 h with ethanol at concentrations of 5% and 10% showed cell survival rates of only 30.01% and 3.86%, respectively (Figure 4(A)). The effects of DLPW3 and LPW7 on cell viability followed the same trend. HepG2 cells treated with LPW7 for 24 h at low concentrations of 0.4, 4, and 6 %v/v (equivalent to ethanol concentrations of 0.04, 0.43, and 0.64 %eth v/v, respectively) did not result in a reduction in cell number. Instead, there was a significant increase in cell proliferation compared to untreated cells. However, for high concentrations of the LPW7 8, 12, and 20 %v/v (equivalent to ethanol concentrations of 0.85, 1.28, and 2.13 %eth v/v, respectively), there was a tendency to induce a reduction in cell number, but no significant difference was observed compared to untreated cells. In contrast, HepG2 cells treated for 48 h showed cell viability decreasing by up to 50.75% at a concentration of 20 %v/v (2.13 %eth v/v) (Figure 4(B)). It is noteworthy that the LPW7 sample, with an ethanol concentration of 2.13%, induced a greater reduction in cell viability than the 2.5% ethanol standard, as assessed by the MTT assay. This observation suggests that the ethanol concentration in LPW7 was likely too low to be the sole primary cause of the observed cytotoxicity. The results indicate that ethanol in LPW7 is not the only factor responsible for the decreased percentage of HepG2 cell viability; rather, other components such as anthocyanins, phenolic compounds, and other bioactive substances also contribute to this reduction. This conclusion is further supported by comparison of LPW7 with the dealcoholized sample (DLPW3). DLPW3, which contained anthocyanins, phenolic compounds, and other bioactive substances (identical to LPW7 in all respects except for the presence of ethanol), showed no significant reduction in cell viability. Therefore, the marked reduction in cell viability induced by LPW7 is hypothesized to result from a synergistic interaction between the various bioactive components and the ethanol present in the wine. An interesting report suggests that exposure to non-lethal or moderate concentrations of ethanol can alter the structure of the cell membrane. Ethanol is known to disrupt hydrogen bonds that normally maintain the ordered arrangement of fatty acid molecules in the membrane. When these bonds are disrupted, membrane order decreases and permeability increases [22,23]. This increased permeability could potentially facilitate the entry of bioactive compounds, such as anthocyanins and phenolics, into the cell, thereby enhancing their combined effects and leading to a pronounced reduction in HepG2 cell viability
Cytotoxicity testing of DLPW3, for HepG2 cells treated with DLPW3 for 24 h at concentrations of 2 - 12 %v/v (equivalent to ethanol concentrations of 0.002 - 0.012 %eth v/v), showed cell proliferation significantly increasing compared to untreated cells. However, cell numbers decreased at the higher concentration of 20 %v/v (0.02 %eth v/v). HepG2 cells treated with DLPW3 for 48 h did not experience cell death even at the highest concentration of 20 %v/v (0.012 %eth v/v). It has been previously reported that HepG2 cells treated with ethanol at concentrations of 10 - 30 mM (which is comparable to ethanol 0.14 - 0.28%, LPW7 0.4 - 4 %v/v, and all concentrations of DLPW3 used in this study) for 24 h, induced an increase in cell viability by activating the ERK-p90rsk pathway. This leads to an increase in cell cycle progression and a decrease in apoptosis, processes that have been reported to be associated with cancer progression in various types of cells [24-26]. However, it has been reported that the signal strength of the MTT assay is influenced by both cellular viability and metabolic activity of cells. Therefore, the observed increase in MTT activity at low concentrations might primarily be due to enhanced cellular metabolic activity rather than a true increase in cell count, consistent with reports that alcohol concentrations below 1% can increase cellular MTT activity [27]. However, it has also been reported that ethanol concentrations of 10 - 30 mM have no effect on increasing ERK activity in normal liver cells [28].
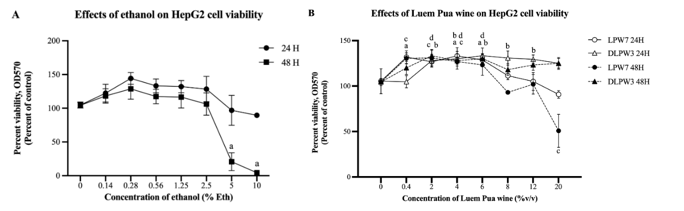 Figure
4
Cytotoxicity testing of LP rice wine on HepG2 cells assessed by the
MTT assay. (A) Effects of ethanol on HepG2 cells. (B) Effects of
LPW7 and DLPW3 on HepG2 cells. Data is expressed as the mean ± SD.
Significant differences compared to the control (0% concentration)
are indicated as follows: (a) LPW7 (24 h); (b) DLPW3 (24 h); (c)
LPW7 (48 h); (d) DLPW3 (48 h).
Figure
4
Cytotoxicity testing of LP rice wine on HepG2 cells assessed by the
MTT assay. (A) Effects of ethanol on HepG2 cells. (B) Effects of
LPW7 and DLPW3 on HepG2 cells. Data is expressed as the mean ± SD.
Significant differences compared to the control (0% concentration)
are indicated as follows: (a) LPW7 (24 h); (b) DLPW3 (24 h); (c)
LPW7 (48 h); (d) DLPW3 (48 h).
Reducing ROS production effect of wines in HepG2 cells
From the cytotoxicity tests on HepG2 cells with LPW7 and DLPW3, it was found that concentrations below 6 %v/v (0.64 %eth v/v of LPW7) did not cause cell death, regardless of whether the treatment duration was 24 or 48 h. The anthocyanin analysis, it was found that LPW7 contained 15.5 µg/mL of anthocyanins, while DLPW3 contained 15.7 µg/mL. Therefore, for testing the ROS-reducing effect in HepG2 cells, wine anthocyanin concentrations of 0.5, 1.0, and 1.5 µg/mL (corresponding to final ethanol concentrations of 2 - 6 %v/v) were selected. The ROS levels were measured using the DCFDA assay, a technique that detects intracellular ROS by measuring the fluorescence emitted after DCFDA is oxidized by ROS into a highly fluorescent compound, dichlorofluorescein (DCF). The experimental results were expressed as percentages relative to the untreated control group (normalized to 100%). The results showed that all concentrations of both types of wine significantly reduced ROS in HepG2 cells compared to the untreated control group, with a trend of greater ROS reduction at higher wine concentrations. Additionally, no significant differences were observed in ROS reduction between LPW7 and DLPW3 at the same concentrations (Figure 5). In this study, we observed that untreated HepG2 cells exhibited very high ROS production. This could be characteristic of cancer cells, as it has been reported that elevated levels of ROS are a hallmark of cancer. Cancer cells inherently have increased ROS levels due to various factors, including genetic aberrations, rapid proliferation and altered cellular metabolism [29-31]. Polyphenols are widely recognized for their ability to protect cells from oxidative stress, attributed to their antioxidant and radical-scavenging properties, and are linked to numerous health benefits. The diverse biological activities of polyphenol-rich wine and wine-derived products have been extensively studied, including their antioxidant, antiproliferative, and anti-inflammatory effects [32-35]. Furthermore, phytochemicals in red wine have been reported to act as protective agents against alcohol-induced damage in HepG2 cells and the human astrocytoma cell line 1321N1, supporting the recommendation of moderate alcohol or red wine consumption [36].
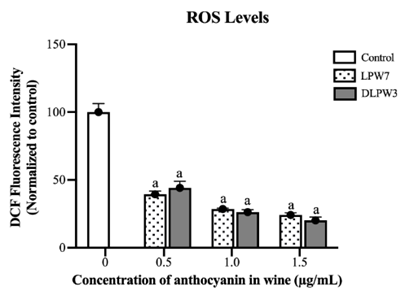
Figure 5 The effect of LPW7 and DLPW3 to reduce ROS in HepG2 cells. Data are expressed as the mean ± SD. a Indicates a significant difference compared to control group.
Probiotics in Luem Pua rice wine
The
results showed the presence of Lactobacillus
buchneri
(84.5% identification) and Pediococcus
acidilactici
(99.1% identification) in LPW7, with a total lactic acid bacteria
(LAB) count of 7.00×10³ CFU/mL. In DLPW3, Lactobacillus
buchneri
(86.8% identification) and Lactobacillus
fermentum
(99.1% identification) were detected, with a total LAB count of 33
CFU/mL. These findings suggest that fermented rice wine may serve as
a promising source of LAB with potential probiotic properties. The
lower count of probiotics in DLPW3 may result from the process of
ethanol removal using a rotary evaporator. This method involves
reducing pressure and applying heat at 40 - 50 °C for 90 min, which
could potentially lead to bacterial death. The detection of lactic
acid bacteria in non-dairy fermented beverages, including wine,
aligns with previous studies that identified various lactic acid
bacteria strains from red wine, confirming their probiotic potential
[8,37]. Furthermore, occasional consumption of fermented beverages
such as wine, beer, cider, and kombucha has been shown to positively
influence gut microbiome diversity [37,38]. The diversity of LAB
identified in Luem Pua rice wine suggests a strong potential for
promoting gut health, comparable to benefits observed in fermented
dairy products and other beverages. This diversity includes three
species with well-established probiotic functions. L.
buchneri
demonstrates high acid and bile tolerance, significant antimicrobial
activity, and notable cholesterol-lowering, neuroprotective, and
antioxidant effects [39,40]. Likewise, P.
acidilactici
is recognized as a promising next-generation probiotic due to its
strong gastrointestinal resilience, antimicrobial properties, and
proven ability to reduce toxic heavy metals in the human
bloodstream. Furthermore, it has been reported to enhance
reproductive performance and offspring quality in male Atlantic
salmon, evidenced by increased gonad weight and sperm concentration
[41-43]. L.
fermentum
is also a beneficial probiotic known for enhancing the immune
response and preventing gastrointestinal and respiratory infections.
It produces potent antimicrobial peptides suitable for use as food
preservatives or antibiotic alternatives, and exhibits key health
functions such as cholesterol-lowering capacity and potential roles
in preventing alcoholic liver disease and colorectal cancer [44].
The consistent presence of L.
buchneri
in both LPW7 and DLPW3 further highlights its potential as a
valuable and reliable probiotic source.
Conclusions
This study examined the antioxidant and probiotic properties of Luem Pua rice wine (LPW) in both its alcoholic and dealcoholized forms (DLPW). It was found that wine fermented for three days (DLPW3) exhibited the highest phenolic content, while wine fermented for seven days (LPW7) had the highest monomeric anthocyanin content. The dealcoholization process effectively preserved bioactive properties. Both LPW and DLPW demonstrated excellent antioxidant activity by reducing ROS in HepG2 liver cells without causing cytotoxicity at moderate concentrations. Additionally, probiotics such as L. buchneri, L. fermentum and P. acidilactici were identified, highlighting the wine’s potential to promote gut health. In conclusion, both alcoholic and dealcoholized forms of Luem Pua rice wine offer substantial health benefits through their antioxidant and probiotic properties. The presence of these beneficial compounds and microorganisms indicates that Luem Pua rice wine possesses strong characteristics for development as a functional beverage. Notably, the DLPW3 variant, containing less than 0.5% alcohol, is particularly suitable for development as a broad-spectrum nutraceutical functional beverage accessible to all consumer groups, including those abstaining from alcohol. The dealcoholized version is particularly suitable for individuals seeking functional beverages without alcohol. Moderate consumption of these wines may help improve health and support gut microbiota balance. Further studies on long-term health effects and practical dietary applications could maximize their potential as health-promoting beverages.
Acknowledgements
This research project was supported by the Navamindradhiraj University Research Fund, Navamindradhiraj University (grant number: วจ.สนธ.86/2564), Thailand. The authors declare no conflicts of interest. We truly appreciate Professor Veerapol Kukongviriyapan from the Department of Pharmacology, Faculty of Medicine, Khon Kaen University, Thailand for his kind support and valuable guidance in the cell culture experiments. And many thanks to Mr. Marcus Fox for his help in reviewing the English grammar of this manuscript.
Declaration of Generative AI in Scientific Writing
The authors used generative AI tools (e.g., Gemini and ChatGPT) to assist in sentence structuring and the initial drafting of the manuscript. Subsequently, the text underwent comprehensive language editing and grammatical verification by a native British speaker, Mr. Marcus Fox. AI was not employed for content generation or data interpretation. The authors maintain full responsibility for the integrity and conclusions of this work.
CRediT Author Statement
Sirilug Magerd and Supawadee Srisuwan: Conceptualization; Methodology; Formal analysis; Investigation; Resources; Data curation; Writing—original draft preparation; Writing—review and editing; Visualization; Supervision; Project administration; Funding acquisition. Warachin Gangnonngiw: Methodology; Formal analysis; Resources. Benjamart Pratoomthai: Methodology; Supervision. Direk Aekthammarat: Methodology; Funding acquisition. Surachai Ngamratanapaiboon: Methodology.
References
[1] P Suwannalert and S Rattanachitthawat. High levels of phytophenolics and antioxidant activities in Oryza Sativa - unpolished Thai rice strain of Leum Phua. Tropical Journal of Pharmaceutical Research 2011. https://doi.org/10.4314/tjpr.v10i4.8
[2] S Srisuwan, T Arkaravichien, S Mahatheeranont, P Puangsombat, P Seekhaw, ANL Noenplab and J Sattayasai. Effects of aqueous extract of unpolished dark purple glutinous rice, var Luem Pua, on ROS in SK-N-SH cells and scopolamine-induced memory deficit in mice. Tropical Journal of Pharmaceutical Research 2015. Tropical Journal of Pharmaceutical Research 2015; 14(9), 1635-1641.
[3] K Thipart, K Phunikhom, ANL Noenplab and J Sattayasai. Dark Purple Luem Pua rice extract reduces disease severity in rat models of inflammatory bowel disease. Tropical Journal of Pharmaceutical Research 2020; 19(3), 629-636.
[4] T Chumphon, P Sriprasertsak and S Promsai. Development of rice as potential carriers for probiotic Lactobacillus amylovorus. International Journal of Food Science and Technology 2016; 51(5), 1260-1267.
[5] VB Shet, SD Sagar, MN Bollamma, TJ Mary, RC Vaman, A Aparna and S Yumnam. Production of fermented fruit juice and value addition by blending medicinal plants. Journal of Bacteriology & Mycology: Open Access 2017; 5(6), 395-397.
[6] JB German and RL Walzem. The health benefits of wine. Annual Review of Nutrition 2000; 20, 561-593.
[7] S Arranz, G Chiva-Blanch, P Valderas-Martínez, A Medina-Remón, RM Lamuela-Raventós and R Estruch. Wine, beer, alcohol and polyphenols on cardiovascular disease and cancer. Nutrient 2012; 4(7), 759-781.
[8] A García-Ruiz, DGD Llano, A Esteban-Fernández, T Requena, B Bartolomé and MV Moreno-Arribas. Assessment of probiotic properties in lactic acid bacteria isolated from wine. Food Microbiololy 2014. https://doi.org/10.1016/j.fm.2014.06.015
[9] K Kondo. Beer and health: Preventive effects of beer components on lifestyle-related diseases. BioFactors 2004; 22(1-4), 303-310.
[10] U.S. Food and Drug Administration. CPG Sec 510.400 - Dealcoholized wine and malt beverages-Labeling, Available at: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/cpg-sec-510400-dealcoholized-wine-and-malt-beverages-labeling, accessed March 2025.
[11] VL Singleton and JA Rossi. Colorimetry of total phenolics with photomolybdic-phosphotungstic acid reagents. American Journal of Enology and Viticulture 1965; 16, 144-158.
[12] J Lee, RW Durst and RE Wrolstad. Determination of total monomeric anthocyanin pigment content of fruit juices, beverages, natural colorants, and wines by the pH differential method: Collaborative study. Journal of AOAC International 2005; 88(5), 1269-1278.
[13] K Theeraraksakul, K Jaengwang, K Choowongkomon and L Tabtimmai. Exploring the biological functions and anti-melanogenesis of Phallus indusiatus for mushroom-based cosmetic applications. Cosmetics 2023; 10(5), 121.
[14] C Sonklin, AM Alashi, N Laohakunjit and RE Aluko. Functional characterization of mung bean meal protein-derived antioxidant peptides. Molecules 2021; 26(6), 1515.
[15] R Gil-Muñoz, E Gómez-Plaza, A Martínez and JM López-Roca. Evolution of the CIELAB and other spectrophotometric parameters during wine fermentation. Influence of some pre and postfermentative factors. Food Research International 1997; 30(9), 699-705.
[16] V Ivanova, B Vojnoski and M Stefova. Effect of winemaking treatment and wine aging on phenolic content in Vranec wines. Journal of Food Science and Technology 2012; 49, 161-172.
[17] F He, NN Liang, L Mu, QH Pan, J Wang, MJ Reeves and CQ Duan. Anthocyanins and their variation in red wines II. Anthocyanin derived pigments and their color evolution. Molecules 2012; 17(2), 1483-1519.
[18] MS Ursu, I Aprodu, SA Milea, E Enachi, G Râpeanu, GE Bahrim and N Stǎnciuc. Thermal degradation kinetics of anthocyanins extracted from purple maize flour extract and the effect of heating on selected biological functionality. Foods 2020; 9(11), 1593.
[19] P Silva. Low-alcohol and nonalcoholic wines: From production to cardiovascular health, along with their economic effects. Beverages 2024; 10(3), 49.
[20] AMS Azima, A Noriham and N Manshoor. Anthocyanin content in relation to the antioxidant activity and colour properties of Garcinia mangostana peel, Syzigium cumini and Clitoria ternatea extracts. International Food Research Journal 2014; 21(6), 2369-2375.
[21] C Brownmiller, LR Howard and RL Prior. Processing and storage effects on monomeric anthocyanins, percent polymeric color, and antioxidant capacity of processed blueberry products. Journal of Food Science 2008; 73(5), H72-H79.
[22] DB Goldstein. Effect of alcohol on cellular membranes. Annals of Emergency Medicine 1986; 15(9), 1013-1018.
[23] M Patra, E Salonen, E Terama, I Vattulainen, R Faller, BW Lee, J Holopainen and M Karttunen. Under the influence of alcohol: The effect of ethanol and methanol on lipid bilayers. Biophysical Journal 2006; 90(4), 1121-1135.
[24] HS Kim, SJ Kim, J Bae, Y Wang, SY Park, YS Min, HDJ and UD Sohn. The p90rsk-mediated signaling of ethanol-induced cell proliferation in HepG2 cell line. Korean Journal of Physiology & Pharmacology 2016; 20(6), 595-603.
[25] A Zoubeidi, A Zardan, RM Wiedmann, J Locke, E Beraldi, L Fazli and ME Gleave. Hsp27 promotes insulin-like growth factor-I survival signaling in prostate cancer via p90Rsk-dependent phosphorylation and inactivation of BAD. Cancer Research 2010; 70(6), 2307-2317.
[26] LA Kubasiak, OM Hernandez, NH Bishopric and KA Webster. Hypoxia and acidosis activate cardiac myocyte death through the Bcl-2 family protein BNIP3. The Proceedings of the National Academy of Sciences 2002; 99(20), 12825-12830.
[27] N Kar, D Gupta and J Bellare. Ethanol affects fibroblast behavior differentially at low and high doses: A comprehensive, dose-response evaluation. Toxicology Reports 2021; 8, 1054-1066.
[28] M Hennig, MT Yip-Schneider, P Klein, S Wentz, JM Matos, C Doyle, J Choi, H Wu, A O’Mara, A Menze, S Noble, IH McKillop and CM Schmidt. Ethanol-TGFalpha-MEK signaling promotes growth of human hepatocellular carcinoma. Journal of Surgical Research 2009; 154(2), 187-195.
[29] MA Shah and HA Rogoff. Implications of reactive oxygen species on cancer formation and its treatment. Seminars in Oncology 2021; 48(3), 238-245.
[30] S Kumari, AK Badana, MG Murali, G Shailender and R Malla. Reactive oxygen species: A key constituent in cancer survival. Biomarker Insights 2018; 13, 1177271918755391.
[31] M Schieber and NS Chandel. ROS function in redox signaling and oxidative stress. Current Biology 2014; 24(10), R453-R462.
[32] C Fuchs, T Bakuradze, R Steinke, R Grewal, GP Eckert and E Richling. Polyphenolic composition of extracts from winery by-products and effects on cellular cytotoxicity and mitochondrial functions in HepG2 cells. Journal of Functional Foods 2020; 70, 103988.
[33] S Hogan, C Canning, S Sun, X Sun and K Zhou. Effects of grape pomace antioxidant extract on oxidative stress and inflammation in diet induced obese mice. Journal of Agricultural and Food Chemistry 2010; 58(21), 11250-11256.
[34] MJ Jara-Palacios, D Hernanz, T Cifuentes-Gomez, ML Escudero-Gilete, FJ Heredia and JPE Spencer. Assessment of white grape pomace from winemaking as source of bioactive compounds, and its antiproliferative activity. Food Chemistry 2015; 183, 78-82.
[35] B Rodríguez-Morgado, M Candiracci, C Santa-María, E Revilla, B Gordillo, J Parrado and A Castaño. Obtaining from grape pomace an enzymatic extract with anti-inflammatory properties. Plant Foods for Human Nutrition 2015; 70, 42-49.
[36] A Gupta, ME Ellis and KA Oduse. The roles of phytochemicals in red wine as a protective agent against alcohol damage. International Food Research Journal 2013; 20(3), 1191-1197.
[37] A Vilela, F Cosme and A Inês. Wine and non-dairy fermented beverages: A novel source of pro- and prebiotics. Fermentation 2020; 6(4), 113.
[38] CIL Roy, PM Wells, J Si, J Raes, JT Bell and TD Spector. Red wine consumption associated with increased gut microbiota α-diversity in 3 independent cohorts. Gastroenterology 2020; 158(1), 270-272.
[39] XQ Zeng, DD Pan and YX Guo. The probiotic properties of Lactobacillus buchneri P2. Journal of Applied Microbiology 2010; 108(6), 2059-2066.
[40] MJ Cheon, SM Lim, NK Lee and HD Paik. Probiotic properties and neuroprotective effects of Lactobacillus buchneri KU200793 isolated from Korean fermented foods. International Journal of Molecular Sciences 2020; 21(4), 1227.
[41] SB Peter, Z Qiao, HN Godspower, SB Ajeje, M Xu, X Zhang, T Yang and Z Rao. Biotechnological innovations and therapeutic application of Pediococcus and Lactic Acid Bacteria: The next-generation microorganism. Frontiers in Bioengineering and Biotechnology 2022; 9, 802031.
[42] P Feng, J Yang, S Zhao, Z Ling, R Han, Y Wu, ES Salama, A Kakade, A Khan, W Jin, W Zhang, BH Jeon, J Fan, M Liu, T Mamtimin, P Liu and X Li. Human supplementation with Pediococcus acidilactici GR-1 decreases heavy metals levels through modifying the gut microbiota and metabolome. npj Biofilms and Microbiomes 2022; 8, 63.
[43] R Díaz, D Carrasco, J Quiñones, A Martínez, G Sepúlveda, I Pérez-Núñez, R Huaiquipán, D Cancino-Baier, JF Beltrán, JG Farías, EA Paz and N Sepúlveda. The probiotic Pediococcus acidilactici in the feed of Salmonids: A strategy to improve reproductive parameters. Animals 2025; 15(11), 1659.
[44] K Naghmouchi, Y Belguesmia, F Bendali, G Spano, BS Seal and D Drider. Lactobacillus fermentum: A bacterial species with potential for food preservation and biomedical applications. Critical Reviews in Food Science and Nutrition 2020; 60(20), 3387-3399.