
Trends
Sci.
2026;
23(7):
12903
Design and Characterization of Microcapsule Loading Vernonia amygdalina Leaf Extract: A Phytochemical and Formulation-Based Approach to Antihypertensive Therapy Candidate
Lusi
Agus Setiani1,*,
Syamsu Nur2,
![]() ,
Septia Andini1,
,
Septia Andini1,
![]() ,
Nabila Yuliana Putri1,
,
Nabila Yuliana Putri1,
Putri Navy Irawan1, Risca Melia Widyanti1 and Ike Rohana1
1Pharmacy Study Program, Pakuan University, West Java 16143, Indonesia
2Department of Pharmaceutical Analysis and Medicinal Chemistry, Universitas Almarisah Madani,
Makassar City, South Sulawesi 90242, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 5 December 2025, Revised: 30 December 2025, Accepted: 10 January 2026, Published: 15 March 2026
Abstract
Vernonia amygdalina has historically been utilized in traditional medicine and is recognized for its bioactive constituents, including alkaloids, flavonoids, tannins, steroids, and saponins, which may have antihypertensive properties. This work sought to develop and assess a microencapsulated formulation of V. amygdalina leaf extract to enhance stability, bioavailability, and therapeutic efficacy. The extract was procured using ethanol maceration, standardized through organoleptic, physicochemical, and phytochemical evaluations, and subsequently characterized by LC-ESI-MS, which verified flavonoids (luteolin, apigenin, quercetin) and phenolic acids as the predominant components. Microencapsulation was executed using spray drying utilizing gum arabic and maltodextrin as encapsulating agents. Four formulations were evaluated, exhibiting entrapment efficiencies from 73.35% to 98.45%, water contents between 3.53% and 5.12%, and yields ranging from 72.40% to 84.35%. Formula 1 (gum arabic:maltodextrin ratio 7:1) exhibited the most advantageous properties, comprising the minimal water content (3.53%), maximal yield (84.35%), and nearly total entrapment efficiency (98.45%). The average particle size was 987.7 nm, with a zeta potential of –54.0 mV, signifying robust colloidal stability. SEM examination verified spherical shape with consistent distribution. The quantitative results indicate that microencapsulation enhanced extract stability and facilitated the regulated release of bioactive components, hence improving delivery performance. In conclusion, the refined formulation of V. amygdalina extract establishes a robust foundation for its advancement as a phytopharmaceutical or nutraceutical candidate for supplementary hypertension treatment, necessitating additional in vivo and clinical assessment.
Keywords: African leaf, LC-ESI-MS characterization, Hypertension, Maltodextrin, Microencapsulation, Vernonia amygdalina
Introduction
Hypertension is a leading global health problem and a major risk factor for cardiovascular disease, contributing to millions of deaths annually [1-3]. In Indonesia, prevalence continues to rise, with more than 63 million cases reported in 2018 [2,4]. Despite the availability of conventional antihypertensive drugs, long-term use is often associated with adverse effects and poor patient compliance, underscoring the need for safer and more acceptable adjunctive therapies [5-7].
Natural products and bioactive compounds from plants have emerged as promising candidates due to their multi-target mechanisms of action, low toxicity, and potential synergy with conventional drugs. One prominent plant is Vernonia amygdalina, known as African leaf or bitter leaf. This plant is traditionally used in Africa to treat hypertension, malaria, and diabetes, and has been empirically utilized in Indonesia as a herbal remedy [8-10]. Phytochemical analysis indicates that V. amigdalina leaf extract contains flavonoids, alkaloids, tannins, and saponins, which exhibit antioxidant, anti-inflammatory, and antihypertensive activities [11,12].
Experimental research on male Sprague-Dawley rats induced by prednisone-NaCl hypertension demonstrated that V. amigdalina leaf extract significantly reduced systolic and diastolic blood pressure. The most effective dose was 200 mg/kg body weight, which reduced blood pressure by 22.83% in 14 days, comparable to the effect of captopril, a standard ACE inhibitor [13]. This antihypertensive activity is primarily attributed to flavonoids, particularly flavonols and flavones, which act as natural ACE inhibitors, preventing the conversion of angiotensin I to angiotensin II and reducing vascular resistance. These findings provide strong scientific evidence that V. amigdalina leaf extract has clinical potential as an adjuvant therapy [14,15].
However, despite promising pharmacological evidence, crude V. amygdalina extract faces critical limitations: low bioavailability, poor stability, and a strong bitter taste that reduces patient acceptability. These challenges represent a clear research gap, as they hinder the translation of traditional use into standardized formulations. Microencapsulation technology offers a potential solution by protecting bioactive compounds from degradation, improving stability, masking bitterness, and enabling controlled release [16,17].
Therefore, this study focused on the development and evaluation of a microencapsulated V. amigdalina leaf extract formula as an antihypertensive adjuvant. Using an in vitro model, the formula's Effectiveness in inhibiting ACE enzyme activity, a key target for hypertension therapy, was tested. The study's results are expected to provide robust scientific evidence regarding the potential of microencapsulated V. amigdalina leaf extract as a safe and effective adjunctive therapy, while also opening up opportunities for the future development of nutraceutical products for cardiovascular health.
Materials and methods
Sample preparation
Vernonia amygdalina leaf samples were collected from Kayu Manis Village, Tanah Sareal District, Bogor City, West Java. Fresh leaves (35 kg) were washed thoroughly with running water and drained to facilitate drying. They were then subjected to wet sorting to remove unwanted parts. The leaves were sun-dried, followed by dry sorting to eliminate residual impurities such as sand. Once completely dried, the leaves were ground using a grinder and passed through a 60‑mesh sieve, yielding 4.9 kg of powdered herbal material
Extraction
The extraction of Vernonia amygdalina powdered leaves was performed using the maceration method with 70% ethanol as the solvent. A plant-to-solvent ratio of 1:10 was applied, corresponding to 4.9 kg of powdered leaves and 49 L of 70% ethanol. Maceration was carried out for 3 days at room temperature and protected from light with stirring every 5 h. The mixture was then filtered, and the residue was re-macerated for an additional 48 h. This process yielded 40 L of liquid extract. The filtrate was subsequently concentrated using a rotary evaporator, followed by a water bath, to remove residual solvent. The procedure produced 1,048 g of thick extract. The extraction method was adapted from Setiani et al. [13].
Standardization of raw materials of dried leaves and extracts
The extract of Vernonia amygdalina was subjected to phytochemical screening through standard qualitative tests. Alkaloids were identified using precipitation reactions involving Mayer’s, Dragendorff’s, and Bouchardat’s reagents. The presence of tannins was verified through the ferric chloride reaction, resulting in a distinctive blue-black or green-brown coloration. Flavonoids were identified through a magnesium–HCl reaction, as evidenced by color changes from red to purple. Saponins were identified through the generation of stable foam following heating and agitation in water. The Liebermann-Burchard reaction was employed to differentiate between steroids and triterpenoids, resulting in a green to bluish-green coloration for steroids and a red to purple coloration for triterpenoids. The standardized procedures validated the presence of essential secondary metabolites, thereby ensuring the authenticity and quality of the raw materials and extracts for subsequent formulation studies [18].
Non-specific paramaters
Loss on drying of simplicia
One g of powdered Myrmecodia (Sarang Semut) tubers was weighed and placed in a covered porcelain crucible. The crucible was heated at 105 °C for 30 min. The sample was then spread evenly in the crucible and dried in an oven at 105 °C for another 30 min. The lid was removed, and heating was continued at 105 °C until a constant weight was obtained [19].
Moisture content of extract
One g of extract was accurately weighed into a calibrated porcelain crucible and dried at 105 °C for 5 h. The sample was then weighed, and drying was continued with reweighing at 1‑hour intervals until a constant weight was achieved. The difference between 2 consecutive weighings did not exceed 0.25% [19].
Ash content of extract
Two g of extract were weighed into a porcelain crucible that had been previously ignited and tared. The sample was leveled and incinerated until a constant weight was obtained. The ash content was calculated relative to the dried material [19].
Ash content of simplicia (crude drug)
Two g of powdered crude drug were weighed into a porcelain crucible that had been previously ignited and tared. The sample was leveled and incinerated until a constant weight was obtained. The ash content was calculated relative to the dried material [19].
Total flavonoid content
A total of 0.2 g of V. amygdalina leaf extract was dissolved in 10 mL of ethanol p.a. and homogenized to obtain a sample solution with a concentration of 20,000 ppm. Subsequently, 1 mL of the 20,000 ppm extract solution was mixed with 3 mL of ethanol p.a., 0.2 mL of 10% AlCl₃ solution, 0.2 mL of 1 M sodium acetate solution, and 5.6 mL of distilled water. The resulting mixture was homogenized and incubated at room temperature for the designated duration. Absorbance was measured using UV-Vis spectrophotometry at the maximum wavelength and recorded in triplicate [20]. The flavonoid content was expressed as mg QE/g dry weight, calculated using the following formula:
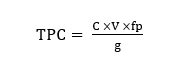
Notation description: TPC = Total Phenolic Content; C = Phenolic concentration (mg/L); V = Sample volume (L); fp = Dilution factor; g = Sample weight (g)
LC-ESI-MS analysis of V. amygdalina extract
The LC-MS QTOF analysis method, according to the 18(LCMS/MS) QTOF-16-2/MU /MU/MU/SMM-SIG standard from SIG Laboratory, was carried out systematically to ensure the accuracy and validity of the results. The extract sample was first dissolved in an LC-MS-grade solvent, then filtered through a 0.22 µm membrane to remove particles. Liquid chromatography analysis was carried out using a C18 column with a mobile phase of water and acetonitrile containing 0.1% formic acid, run in a gradient to separate the active compounds. The QTOF instrument was operated in positive and negative ionization modes with an m/z range of 50 - 1,500, equipped with a lock-mass system to maintain mass accuracy. MS/MS data were acquired with graded collision energies to produce diagnostic fragments. The analysis results were then processed by assessing 4 main criteria: mass error ≤ 5 ppm; isotope match with RMS value ≤ 6 ppm and percentage ≤ 10%; signal intensity or response ≥ 300; and the presence of a fragment match for at least 1 mass ion.
Microencapsulation formulation
Optimization of the microencapsulation formula for Vernonia amygdalina (African leaf) extract was carried out using fourth formulations, varying in coating concentration, stirring speed, and stirring duration. The coating concentration variations were adapted from [21].
Microencapsulation emulsion preparation
The ingredients were weighed according to the formulation (Table 1). Maltodextrin and gum arabic were dissolved and hydrated in distilled water, then homogenized using a Silent Crusher at 5,000 rpm for 15 min. After homogenization, the V. amygdalina leaf extract-previously dissolved in distilled water for 10 min - was gradually added to the mixture. The final emulsion was dried using a spray dryer with an inlet temperature of 125 °C, outlet temperature of 60 °C, and a flow rate of 10 mL [21,22].
Table 1 Coating Variations of microencapsulation emulsion.
V. amygdalina Leaf Extract (g) |
Gum Arabic (g) |
Maltodextrin (g) |
Distilled Water (mL) |
75 |
7 |
1 |
300 |
75 |
6 |
2 |
300 |
75 |
5 |
3 |
300 |
75 |
4 |
4 |
300 |
Stiring speed variation
The ingredients were weighed according to the formulation (Table 2). Maltodextrin and gum arabic were dissolved and hydrated in distilled water, then homogenized using a Silent Crusher at varying speeds of 5,000, 10,000, and 15,000 rpm for 15 min. After homogenization, the V. amygdalina leaf extract - previously dissolved in distilled water (LC-MS grade, Merck) for 10 min - was gradually added to the mixture. The resulting mixture was then dried using a laboratory-scale spray dryer (Büchi Mini Spray Dryer B-290, Switzerland) operated at an inlet temperature of 125 °C, outlet temperature of 60 °C, and a feed flow rate of 10 mL/min [22,23].
Table 2 Variation of stiring speed and stiring time.
V. amygdalina Leaf Extract (g) |
Gum Arabic (g) |
Maltodextrin (g) |
Distilled Water (mL) |
Stirring Speed (rpm) |
75 |
7 |
1 |
300 |
5,000 |
75 |
7 |
1 |
300 |
10,000 |
75 |
7 |
1 |
300 |
15,000 |
Stiring time variation
The ingredients were weighed according to the formulation (Table 3). Maltodextrin and gum arabic were dissolved and hydrated in distilled water, then homogenized using a Silent Crusher (Heidolph, Germany) at 5,000 rpm for 15 min. After homogenization, the Vernonia amygdalina leaf extract - previously dissolved in distilled water (LC-MS grade, Merck) - was gradually added and stirred for 5, 10, and 15 min, respectively. The resulting mixture was then dried using a laboratory-scale spray dryer (Büchi Mini Spray Dryer B-290, Switzerland) operated at an inlet temperature of 125 °C, outlet temperature of 60 °C, and a feed flow rate of 10 mL/min [22,23].
Table 3 Variation of stiring time.
V. amygdalina Leaf Extract (g) |
Gum Arabic (g) |
Maltodextrin (g) |
Distilled Water (mL) |
Stirring Time (min) |
75 |
7 |
1 |
300 |
5 |
75 |
7 |
1 |
300 |
10 |
75 |
7 |
1 |
300 |
15 |
Evaluation of microencapsulation characteristics
The microencapsulation characteristics were evaluated using several key parameters, including moisture content, recovery yield, particle size, zeta potential, polydispersity index, and scanning electron microscopy (SEM) analysis [24,25].
Entrapment efficiency
Encapsulation efficiency was determined by measuring total flavonoid content using a quercetin standard curve. A blank solution was prepared by mixing ethanol p.a., sodium acetate (1 M), and distilled water. Standard solutions of quercetin (30 - 70 ppm) were reacted with ethanol p.a., 10% AlCl₃, sodium acetate, and distilled water, then incubated for 30 min at room temperature and measured at 438 nm in triplicate. Extract (500 mg) was dissolved in ethanol p.a., filtered, and diluted to 10 mL. Each 1 mL sample was mixed with ethanol p.a., AlCl₃, sodium acetate, and distilled water to a final volume of 10 mL, vortexed for 10 min, and incubated for 30 min. Absorbance was measured at the maximum wavelength, and total flavonoid content was calculated using the standard curve [24]. Encapsulation efficiency was then calculated using the formula:
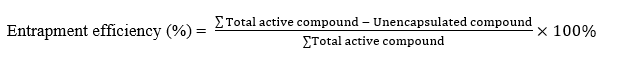
Results and discussion.
Characteristics of Vernonia amygdalina simplicia
The organoleptic evaluation of Vernonia amygdalina simplicia showed that the dried material was in the form of a fine powder, green in color, with a characteristic odor and a distinctly bitter taste (Table 4).
Table 4 Characteristic Result of Vernonia amygdalina simplicial.
Parameter |
Result |
Organoleptic Shape Odor Color Taste |
Powder Characteristic Green Bitter |
Drying Loss |
0.37 ± 0.00 |
Ash Content |
9.49 ± 0.15 |
Acid-insoluble ash content |
0.38 ± 0.00 |
Ethanol-soluble extractive value |
9.15 ± 0.54 |
Water-soluble extractive value |
13.37 ± 1.74 |
These findings align with the unique characteristics of Vernonia amygdalina, which contains sesquiterpene lactones and flavonoids that contribute to its bitter taste. The green color indicates the presence of chlorophyll and other plant pigments that remain stable after drying, while the distinctive aroma suggests volatile compounds. Bitter taste is a crucial marker of quality because it is directly related to the bioactive compounds that contribute to the pharmacological activity of plants, particularly alkaloids. These results confirm that herbal medicines meet the expected sensory profile, a crucial aspect for ensuring authenticity and quality before further extraction and formulation [26,27]. This organoleptic parameter also serves as an initial indicator for quality control, complementing physicochemical and phytochemical analyses in the standardization process of herbal raw materials [28].
In addition to organoleptic properties, other characteristics analyzed in the herbal medicine included drying loss and total ash content (Table 4). Drying loss was recorded at 0.37 ± 0.00%, well below the generally recommended maximum limit for herbal raw materials (≤ 10%). This low water content indicates good stability and minimal risk of microbial growth, thus ensuring the quality and shelf life of the herbal medicine [29,30]. The total ash content of 9.49 ± 0.15% reflects the presence of inorganic compounds such as minerals and other non-volatile substances. While ash content indicates mineral residues, excessively high values may indicate contamination or adulteration [19,31]. The results obtained are still within the acceptable range for herbal medicine, thus supporting the purity and authenticity of the material. Overall, the organoleptic profile, low drying loss, and standard ash content confirm that the V. amygdalina herbal medicine meets the initial quality standards and is suitable for further extraction and formulation processes.
Characteristic of extract of Vernonia amygdalina
The extract exhibited a thick texture, a distinctive aroma, a brown color, and a pronounced bitter taste (Table 5). These sensory characteristics align with the known profile of Vernonia amygdalina, a plant widely used in ethnopharmacological practices. The bitter taste primarily indicates the presence of alkaloids and saponins, compounds often associated with therapeutic properties. The ash content of 6.24 ± 0.47% reflects total inorganic residues and indicates a moderate mineral content (Table 5). This value is within acceptable limits for herbal extracts, indicating minimal contamination and appropriate processing. Meanwhile, the moisture content of 8.68 ± 0.53% is relatively low, conducive to extract stability, reduced risk of microbial growth, and extended shelf life.
Overall, the sensory profile, ash content, and moisture content obtained confirm that the V. amygdalina extract meets initial quality standards and is suitable for further development.
Table 5 Characteristic result of Vernonia amygdalina extract.
Parameter |
Result |
Organoleptic: Shape Odor Color Taste |
Viscous Characteristic Brown Bitter |
Ash Content |
6.24 ± 0.47 |
Water content |
8.68 ± 0.53 |
Acid-insoluble ash content |
0.47 ± 0.03 |
Ethanol-soluble extractive value |
|
Water-soluble extractive value |
|
Phytocompounds
|
+
+ + + + |
Note: + indicates the result show positive contained the compounds.
Qualitative phytochemical screening of V. amygdalina extract revealed the presence of several bioactive secondary metabolites, including alkaloids, flavonoids, tannins, steroids, and saponins. These compounds were positively identified using specific reagents, indicating a rich phytochemical profile with diverse pharmacological potential.
Alkaloids were detected using the Mayer, Bouchardat, and Dragendorff tests, confirming their presence in the extract [32]. Alkaloids are known to possess various pharmacological activities, such as anti-inflammatory, antimicrobial, and cytotoxic properties. Some alkaloids have also been reported to exert vasodilatory effects and regulate vascular tone, thereby contributing to blood pressure regulation. Their presence suggests a possible antihypertensive mechanism, either through interaction with adrenergic receptors or by inhibiting the ACE enzyme [33-35].
Flavonoids, known for their antioxidant properties, play a crucial role in neutralizing reactive oxygen species (ROS) and maintaining endothelial function. Since oxidative stress is a significant factor in hypertension, flavonoids may help by increasing nitric oxide (NO) availability and improving blood vessel relaxation. The flavonoid content of this extract supports its potential to reduce vascular resistance and maintain cardiovascular health [36,37].
Tannins have astringent and antimicrobial properties and may contribute to antihypertensive effects through antioxidant activity and lipid profile regulation. Several studies have shown that tannins can inhibit ACE activity and improve endothelial function, thereby helping to lower blood pressure.
Steroids identified in the extract may have anti-inflammatory and membrane-stabilizing effects. Although their direct role in hypertension is not fully understood, some plant steroids are known to influence fluid balance and vascular reactivity, indirectly supporting blood pressure regulation [38,39].
Saponins, known for their surfactant and emulsification properties, may increase the bioavailability of other active compounds. Furthermore, saponins exhibit cholesterol-lowering effects and vasorelaxant activity, both of which are beneficial in the management of hypertension. Their immunomodulatory properties also support vascular health by suppressing systemic inflammation [40,41].
Overall, the presence of these phytoconstituents confirms the therapeutic potential of V. amygdalina, not only for cosmetic and dermatological applications, but also for cardiovascular health. The composition of this extract is consistent with its traditional use in treating hypertension and provides a strong scientific basis for further pharmacological research and the development of antihypertensive formulations.
Analysis of compounds by LC-ESI-MS
The LC‑ESI‑MS chromatogram of Vernonia amygdalina ethanol extract revealed a diverse profile of secondary metabolites, dominated by flavonoids and phenolic acids. Strong signals were observed at retention times of 6.54, 8.29, 13.38, and 16.99 min, corresponding to compounds with hydroxylated aromatic rings, conjugated systems, and glycosidic bonds (Figure 1).
LC-ESI-MS analysis of the ethanol extract of Vernonia amygdalina leaves revealed a diverse secondary metabolite profile, dominated by flavonoid and phenolic compounds. The base peak chromatogram showed clear ion intensities over the 0 - 20-minute retention time range, with strong signals at 6.54, 8.29, 13.38 and 16.99 min (Figure 1). Each peak was evaluated using 4 analytical criteria - mass error (≤ 5 ppm), isotope match (≤ 6 ppm RMS and ≤ 10% deviation), signal intensity (≥ 300 counts), and fragment ion match. Peaks fulfilling all criteria were classified as positive, confirming major constituents. Minor peaks with lower intensity (< 300 counts) were categorized as low abundance, indicating trace metabolites that may contribute synergistically. These peaks correspond to compounds with distinctive structural features, such as hydroxylated aromatic rings, conjugated systems, and glycosidic bonds, as shown in the molecular structure (Figure 2). The identified compounds included flavonols, flavones, flavonoid glycosides, and phenolic acids, all of which are known to modulate cardiovascular function.
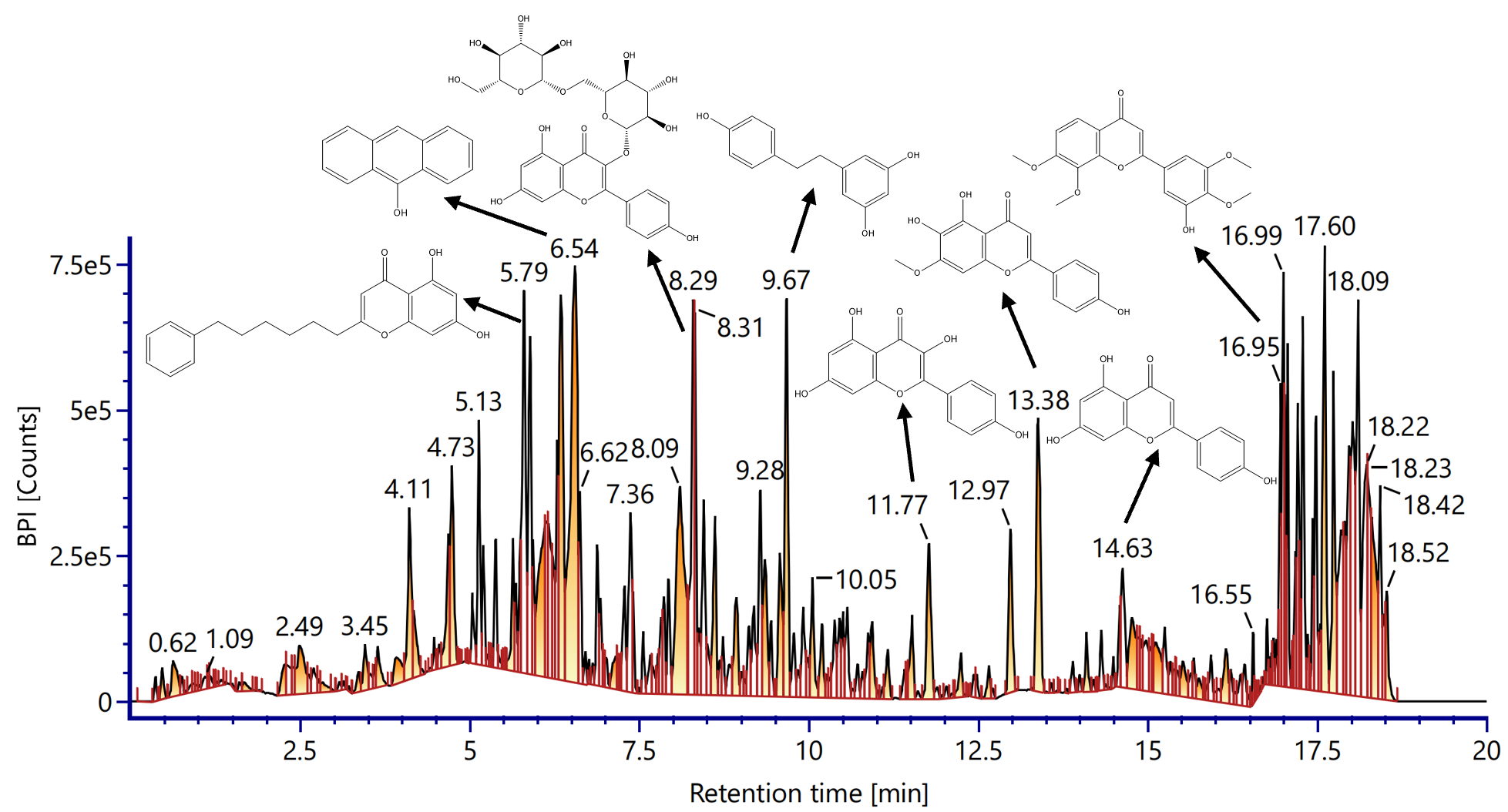
Figure 1 LC-ESI-MS chromatogram (positive ion mode) of ethanol extract of Vernonia amygdalina leaves showing base peak intensity (BPI) profile against retention time (0 - 20 min).
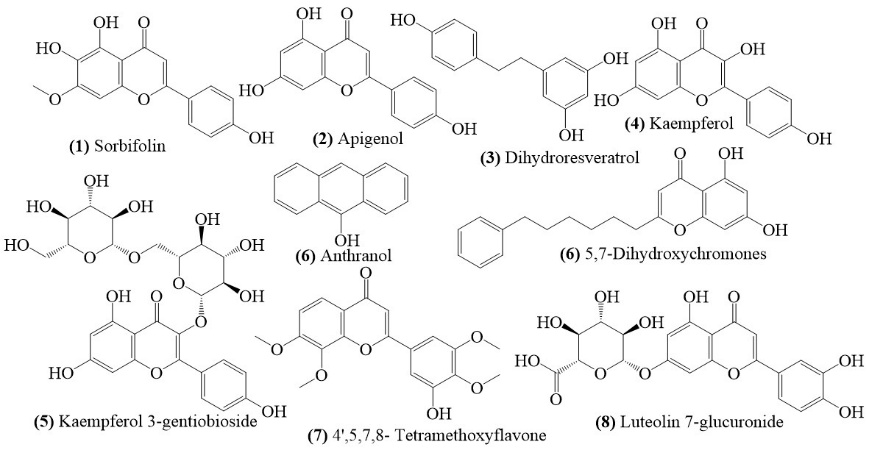
Figure 2 Chemical structure of secondary metabolite compounds identified from ethanol extract of Vernonia amygdalina leaves based on LC-ESI-MS analysis.
From a pharmacodynamic perspective, several detected flavonoids - such as luteolin, apigenin, and quercetin - are known to inhibit angiotensin-converting enzyme (ACE), a key regulator of the renin-angiotensin system that plays a role in vasoconstriction and elevated blood pressure. By suppressing ACE activity, these compounds contribute to systemic vasodilation and reduced vascular resistance [42,43]. Furthermore, phenolic acids such as ferulic acid and caffeic acid possess potent antioxidant properties that can reduce oxidative stress while maintaining endothelial function. Some flavonoids also increase nitric oxide (NO) availability, thereby supporting blood vessel relaxation via an endothelium-dependent mechanism. Furthermore, some compounds act as calcium channel modulators, restricting the influx of calcium ions into vascular smooth muscle cells and thereby reducing vasoconstriction [44].
Overall, the LC-ESI-MS findings provide strong scientific evidence for the ethnopharmacological use of Vernonia amygdalina as a natural antihypertensive agent. The presence of structurally diverse and pharmacologically active flavonoids and phenolics confirms the therapeutic potential of this plant extract in the management of hypertension. This insight not only strengthens traditional claims but also opens the door to the development of standardized phytopharmaceuticals based on bioactive compounds from V. amygdalina.
Total flavonoid content
The LC-ESI-MS characterization results indicate that most of the compounds identified in the V. amigdalina leaf extract are flavonoid derivatives with significant pharmacological potential. Therefore, determining the total flavonoid content expressed as quercetin equivalents is an essential step in providing supporting quantitative data. This parameter not only validates the bioextract’s activity but also provides a scientific basis for establishing dosage regimens for the development of natural product formulations [24].
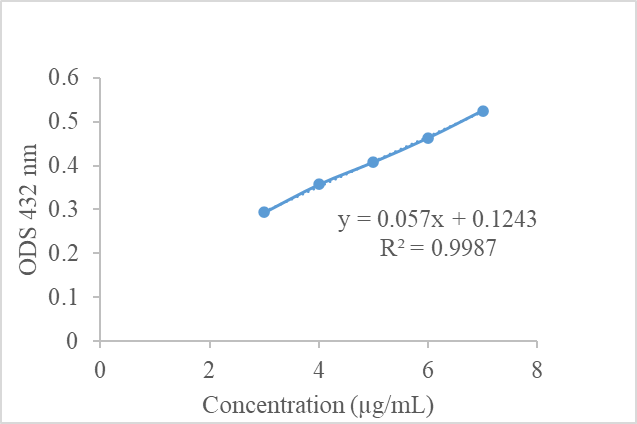
Figure 3 Scatter plot showing the relationship between concentration (µg/mL) and optical density (OD) measured at a wavelength of 432 nm. The linear regression equation obtained is y = 0.057x + 0.1243, with a coefficient of determination R² = 0.9987, indicating a very strong correlation and high validity as a standard curve for quantitative analysis. The standard curve data were analysis triplicate (n = 3).
The figure shows a graph of the relationship between solution concentration (µg/mL) on the x-axis and optical density (OD) at 432 nm on the y-axis. The data points exhibit a consistent linear pattern, with a fitted regression line to illustrate the trend. The resulting regression equation is y = 0.057x + 0.1243, with a coefficient of determination (R²) of 0.9987, indicating a robust linear correlation between concentration and absorbance. This graph serves as a standard calibration curve for quantitative analysis, including the determination of flavonoid levels in V. amigdalina leaf extract. The R² value, which is close to 1, indicates that the regression model has high accuracy in predicting OD from concentration.
The results of the total flavonoid determination showed that the ethanol extract of V. amigdalina leaf contains 2.99 ± 0.0019 mg/g extract, expressed as quercetin equivalents. This quantitative value reflects the abundance of flavonoid compounds in the extract, consistent with the LC-ESI-MS findings that identified various flavonoid derivatives. The total flavonoid content depends on the intrinsic composition of the extract, and accurate measurement provides an essential benchmark for correlating phytochemical richness with biological activity. Determining flavonoid levels as quercetin equivalents also provides a standardized reference for designing dosage regimens for natural product-based therapies [24,45]. These data not only strengthen the scientific basis of V. amigdalina leaf extract as an antihypertensive candidate but also ensure reproducibility and reliability in future nutraceutical development.
The microencapsulation formulation containing V. amygdalina leaf extract was optimized using various parameters, including combinations of gum arabic and maltodextrin coatings, stirring speed, and stirring time. These test parameters were carried out to ensure that the resulting microencapsulation had physically and chemically stable characteristics. The preliminary trials indicated that stirring speeds of 500 - 1,200 rpm and times of 10 - 30 min were chosen to optimize droplet dispersion and wall material integrity. Speeds below 500 rpm caused phase separation, while those above 1,200 rpm led to shear degradation. Similarly, mixing times under 10 min gave incomplete homogenization, whereas longer than 30 min offered no further benefit. Entrapment efficiency (EE, %) was used as the main optimization indicator, with results shown in Figure 4 to identify the most suitable formulation conditions.
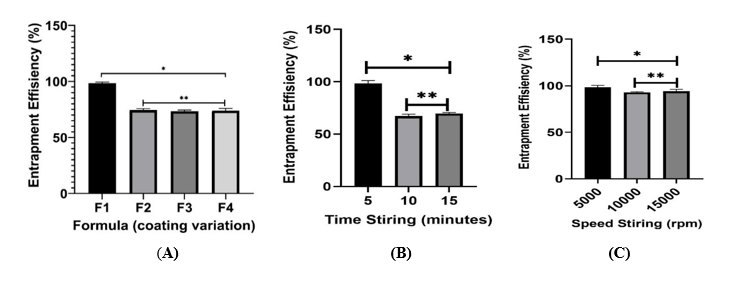
Figure 4 Optimization of microcapsulation formula for loading V. amygdalina extract, which is varied by the ratio of gum arabic layer: Maltodextrin (A) for F1 (7:1), F2 (6:2), F3 (5:3), and F4 (4:4), stirring time (B), and stirring speed (C). Note: *indicates a significant difference in characteristics between the formulas (p-value < 0.05, Tukey HSD, n = 3), whereas **indicates no significant difference in characteristics (p-value > 0.05, Tukey HSD, n = 3).
The optimization of coating materials indicates that the ratio of gum arabic to maltodextrin significantly influences microencapsulation performance. Among the 4 formulations tested, F1 (7:1 ratio) demonstrated the highest entrapment efficiency at 98.45 ± 0.82%, surpassing F2 (74.69 ± 0.35%), F3 (73.34 ± 0.01%), and F4 (73.98 ± 0.06%). The enhanced efficiency is due to the elevated concentration of gum arabic, recognized for creating a dense, protective matrix surrounding the core material. The emulsifying and film-forming properties of gum arabic improve droplet stability, which reduces the leakage of bioactive compounds during the spray drying process [46]. The high efficiency noted in F1 suggests that almost all flavonoids were effectively retained within the microcapsules, thereby ensuring optimal preservation of antihypertensive activity.
All formulations achieved the acceptable entrapment efficiency threshold (60% - 100%). However, the significant difference between F1 and the other formulas underscores the importance of coating composition. Maltodextrin, while effective in decreasing stickiness and enhancing powder flow, offers diminished encapsulation at elevated concentrations. The synergistic balance between gum arabic and maltodextrin is essential, with gum arabic serving as the primary stabilizer [21,23,46]. In addition to coating composition, process parameters like stirring time and speed significantly affect encapsulation results. Optimization indicated that a stirring duration of 5 min at a speed of 5,000 rpm achieved maximum efficiency, ensuring droplet integrity and promoting uniform coating. Excessive stirring compromised droplet structure due to extended shear forces, whereas inadequate stirring did not create a stable protective layer [47,48]. Higher stirring speeds similarly induced turbulence and mechanical stresses, leading to the fragmentation of droplets into unstable forms, which accounts for the reduced efficiency noted in formulations like F4.
The microencapsulation performance of 4 formulations was assessed based on water content, recovery yield, entrapment efficiency, particle size, and zeta potential. The results are described in Table 6.
Table 6 The microencapsulation performance results of formulations.
Formula (Gum arab:maltodextrin) |
Water Content (%) |
Recovery (%) |
Entrapment Efficiency (%) |
Particle Size (nm) |
Zeta Potential (mV) |
Formula 1 (7:1) |
3.53 ± 0.15* |
84.17 ± 0.16* |
98.45 ± 0.82* |
987.7 ± 83.3* |
−54.0 ± 0.00* |
Formula 2 (6:2) |
4.83 ± 0.31*d |
81.19 ± 0.08* |
74.45 ± 0.35cd |
1026.5 ± 103.2cd |
−46.7 ± 0.00*d |
Formula 3 (5:3) |
5.03 ± 0.06* |
77.41 ± 0.13* |
73.35 ± 0.02bd |
1147 ± 282.1bd |
−39.2 ± 0.00* |
Formula 4 (4:4) |
4.60 ± 0.17*b |
70.54 ± 0.37* |
73.98 ± 0.06bc |
1123.3 ± 90.08bc |
−46.1 ± 0.00*b |
Note: *indicates a significant difference in characteristics between the formulas (p-value < 0.05, Tukey HSD, n = 3), whereas abcdidentical alphabets indicate no significant difference in characteristics (p-value > 0.05, Tukey HSD, n = 3).
Formula 1 exhibited the lowest water content (3.53 ± 0.15%), suggesting superior drying efficiency and improved stability. Lower moisture levels are desirable for prolonging shelf life and minimizing microbial growth. Formulas 2 - 4 showed slightly higher water content, which may affect long-term stability and require optimization of drying conditions. Formula 1 demonstrated the highest recovery (84.17 ± 0.16%) and entrapment efficiency (98.45 ± 0.82%), indicating effective encapsulation of the active compounds. High entrapment efficiency is critical for preserving bioactivity and ensuring controlled release [46,48]. The decline in both parameters across Formulas 2 - 4 suggests reduced encapsulation performance, possibly due to formulation composition or processing variables.
All formulations yielded microcapsules within the submicron to low-micron range, hence validating their appropriateness for microencapsulation-based delivery systems. Formula 1 exhibited the lowest particle size (987.7 ± 83.3 nm), which is beneficial for ensuring uniform dispersion and enhancing rehydration qualities during application. In contrast, Formulas 2 to 4 yielded bigger particles (> 1,100 nm), potentially impairing dispersibility and diminishing the efficacy of bioactive release. Research indicates that microencapsulation systems with particle sizes under 5 µm (5,000 nm) have enhanced stability and controlled release relative to bigger particles. Jafari et al. [49] indicated that spray-dried microcapsules with reduced particle sizes exhibited enhanced solubility and encapsulation efficiency. Thus, the particle size results in this study reinforce the superior characteristics of Formula 1, which not only meets the threshold for effective microencapsulation systems but also demonstrates enhanced stability and delivery potential for Vernonia amygdalina bioactives.
In zeta potential values in Figure 6, indicating a stable nano-system. Zeta potential is an essential metric for assessing the stability of colloidal and nanoencapsulation systems. Values exceeding ± 30 mV are deemed adequate to provide electrostatic repulsion among particles, thus inhibiting aggregation and maintaining dispersion stability [50,51]. Increased negative readings, especially below –40 mV, signify intensified repulsive forces and improved colloidal stability. This analysis revealed that the zeta potential values of the formulations varied from –39.2 to –54.0 mV, indicating that all systems fell under the stability threshold for dispersions. Formula 1 demonstrated the lowest zeta potential (–54.0 mV), indicating enhanced electrostatic stability and a less propensity for particle aggregation. In contrast, Formula 3 exhibited the least negative value (–39.2 mV), which, while remaining within the permissible stability range, indicates a comparatively elevated risk of particle contact and a probable decrease in shelf life. The report has continuously highlighted the significance of zeta potential in forecasting colloidal stability. Tamboli et al. [51] assert that nano-formulations exhibiting zeta potential values exceeding ± 40 mV exhibit prolonged stability owing to robust repulsive forces among particles. Research conducted by Akhavan Mahdavi et al. [21]; Sakulnarmrat and Konczak [52] indicates that encapsulation systems utilizing gum arabic and maltodextrin attain improved stability when zeta potential values are sustained below –40 mV [21,50,52]. These findings corroborate the results of Formula 1 in this study, underscoring its exceptional efficacy in preserving particle integrity and guaranteeing product quality. Overall, the ANOVA results confirmed that variations in the composition of gum arabic and maltodextrin had a significant effect on the performance of microencapsulation, with Formula 1 demonstrating the most optimal outcome.
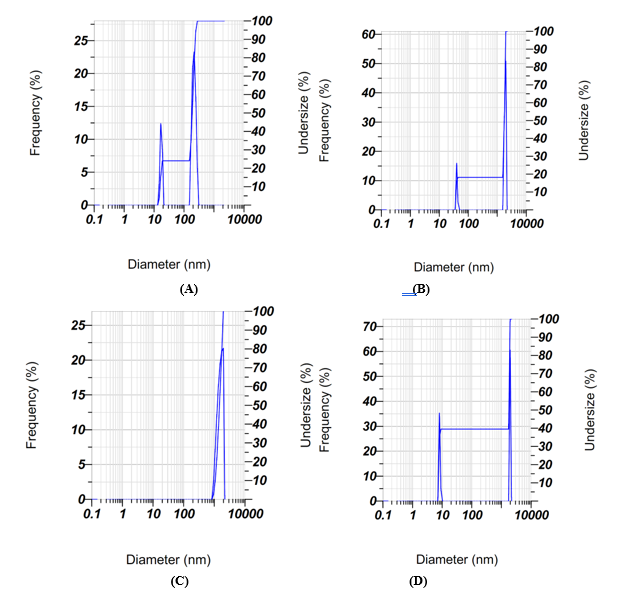
Figure 5 Particle size distribution of microencapsulation formulations, showing diameter versus frequency (%). Graphs A–D represent the particle size profiles of Formula 1 (7:1), Formula 2 (6:2), Formula 3 (5:3), and Formula 4 (4:4), respectively. The plots show diameter (nm) versus frequency (%), highlighting differences in particle uniformity and dispersion among formulations. Formula 1 exhibits a narrower distribution with smaller average particle size, indicating more homogeneous microcapsules, while Formulas 2 - 4 display broader distributions with larger diameters, reflecting reduced uniformity and stability.
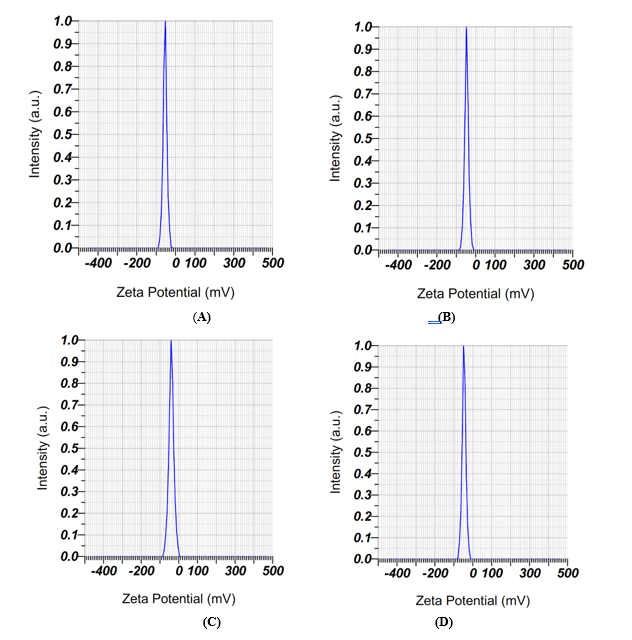
Figure 6 Zeta potential analysis of microencapsulation formulations, showing zeta potential (mV) versus intensity (a.u). Graphs A - D represent the zeta potential profiles of Formula 1 (7:1), Formula 2 (6:2), Formula 3 (5:3), and Formula 4 (4:4), respectively. Each plot shows zeta potential (mV) versus intensity (a.u.), highlighting differences in electrostatic stability among formulations. Formula 1 exhibited the most negative peak (–54.0 mV), indicating superior colloidal stability, while Formulas 2 - 4 showed less negative values (–39.2 to –46.7 mV), reflecting comparatively lower but still acceptable stability.
The SEM analysis yielded significant insights into the morphology of the microcapsules (Figure 7). Formula 1 primarily displayed spherical and homogeneous particles, thereby validating the effective creation of a protective coating layer. Irregular morphologies were observed, including collapsed, wrinkled, and fragmented particles. Irregularities frequently observed in spray-dried microencapsulation systems can be attributed to several factors such as incomplete coating of the core material, rapid moisture evaporation during drying leading to surface shrinkage and collapse, and mechanical stress during stirring or atomization that disrupts particle uniformity [53-55].
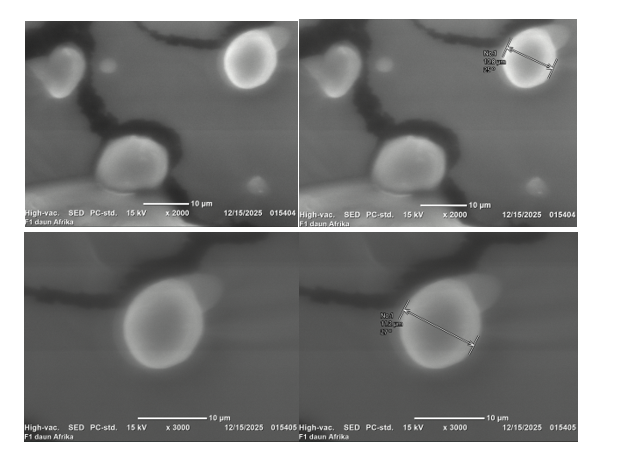
Figure 7 SEM images of African leaf extract microcapsules coated with gum arabic:maltodextrin (7:1), showing predominantly spherical and homogeneous particles with minor irregularities, confirming successful encapsulation and structural integrity (magnification of 2,000× and 3,000×).
Irregular structures, despite their smaller proportions, can affect powder flowability, dispersibility, and rehydration properties. Research suggests that irregular or fractured particles can diminish encapsulation efficiency by exposing core materials to environmental stress, which may hasten the degradation of bioactives [49]. In contrast, wrinkled surfaces have been linked to enhanced rehydration capacity owing to their increased surface area, although they may negatively affect long-term stability
This study indicates that the prevalence of spherical particles in Formula 1 demonstrates the effectiveness of the optimized gum arabic:maltodextrin ratio (7:1) and controlled stirring conditions (5,000 rpm for 5 min) in reducing morphological irregularities. The observed irregular particles likely indicate natural variability in the spray-drying process, rather than systemic instability. The SEM findings indicate the superior performance of Formula 1 and underscore the necessity for additional process refinement to minimize morphological heterogeneity and maintain consistent product quality.
Overall, Formula 1 integrates optimal coating composition and controlled process parameters to deliver superior microencapsulation performance. Its characteristics - low moisture, high recovery, excellent entrapment efficiency, favorable particle size, and strong zeta potential - support its application as a phytopharmaceutical platform for antihypertensive therapy. The system ensures bioactive preservation, sustained release, and improved absorption, aligning with the therapeutic goals of natural product-based formulations [55]. Future studies should investigate long-term stability, in vivo bioavailability, and clinical efficacy to confirm the therapeutic potential of this optimized formulation.
Conclusions
The research indicated that the microencapsulation of Vernonia amygdalina extract with gum arabic and maltodextrin significantly improved stability and regulated the release of bioactive chemicals. The 7:1 coating ratio (F1) yielded excellent performance, underpinned by advantageous physicochemical characteristics and uniform microcapsule morphology. These findings provide a robust basis for the development of V. amygdalina-derived phytopharmaceutical or nutraceutical products as supplementary alternatives for hypertension, and underscore the necessity for additional in vivo and clinical assessments.
Acknowledgements
The author would like to thank the Ministry of Education, Culture, Research, and Technology of the Republic of Indonesia for the research grant funding that has been given to Dr. Apt. Lusi Agus Setian, M.Sc., through a fundamental research grant with contract No: 7928/LL4/PG/2025.
Declaration of generative AI in scientific writing
We, as the authors of this manuscript, acknowledge the use of basic generative AI tools (e.g., the Copilot Rewriter web tool and Grammarly) in the preparation of this manuscript, particularly for language editing and grammar correction. We did not use AI for any content generation or data interpretation. Each author conceived the ideas and the interpretation of this work.
CRediT author statement
Lusi Agus Setiani: Conceptualization; Methodology; validation; Funding acquisition. Syamsu Nur: Data curation; validation; supervision; Reviewing and Editing. Septia Andini: Visualization; Investigation; supervision; Visualization. Nabila Yuliana Putri: Writing - Original draft preparation. Putri Navy Irawan: Software; Validation; investigation. Risca Melia Widyanti: Writing - Investigation. Ike Rohana: Software; investigation.
References
[1] S Goorani, S Zangene and JD Imig. Hypertension: A continuing public healthcare issue. International Journal of Molecule Sciences 2025; 26(1), 0123.
[2] KT Mills, A Stefanescu and J He. The global epidemiology of hypertension. Nature Reviews Nephrology 2020; 16(4), 223-237.
[3] AE Moran, R Gupta, A Pathni, K Osi, G Dessie, X Zhang, D Cazabon, B Banigbe, M Farrell and O Erojikwe. Implementation of global hearts hypertension control programs in 32 low- and middle-income countries: JACC international. Journal of the American College of Cardiology 2023; 82(17), 1868-1884.
[4] K Kario, A Okura, S Hoshide and M Mogi. The WHO Global report 2023 on hypertension warning the emerging hypertension burden in globe and its treatment strategy. Hypertension Research 2024; 47(10), 1099-1102.
[5] B Heidari, E Avenatti and K Nasir. Pharmacotherapy for essential hypertension: A brief review. Methodist DeBakey Cardiovascular Journal 2022; 18(1), 5-16.
[6] E Grossman, P Verdecchia, A Shamiss, F Angeli and G Reboldi. Diuretic treatment of hypertension. Diabetes Care 2011; 34(S2), S246.
[7] S Laurent. Antihypertensive drugs. Pharmacology Research 2017; 124, 116-125.
[8] N Nasim, IS Sandeep and S Mohanty. Plant-derived natural products for drug discovery: Current approaches and prospects. Nucleus 2022; 65(5), 399-411.
[9] SL Ooi and SC Pak. Natural products with pharmaceutical activities. Molecules 2025; 30(23), 4557.
[10] CB Aware, DN Patil, SS Suryawanshi, PR Mali, MR Rane, RG Gurav and JP Jadhav. Natural bioactive products as promising therapeutics: A review of natural product-based drug development. South African Journal of Botany 2022; 151, 512-528.
[11] AM Tura, M Anbessa, ED Tulu and BZ Tilinti. Exploring Vernonia amygdalina’s leaf extracts for phytochemical screening and its anti-bacterial activities. International Journal of Food Properties 2024; 27(6), 960-974.
[12] GI Edo, PO Samuel, AN Jikah, FO Onoharigho, LI Idu, P Obasohan, AR Opiti, J Electric, VO Ikpekoro, CF Otunuya, E Ugbuwe, J Ongulu, M Ijide, ID Nwaose, SR Ajakaye and JE Owigho. Biological and bioactive components of bitter leaf (Vernonia amygdalina leaf): Insight on health and nutritional benefits. A review. Food Chemistry Advances 2023; 3, 100488.
[13] LA Setiani, N Herlina, V Oktaviani and O Cahyani. The potential of African leaf extract (Gymnanthemum amygdalinum Del.) as antihypertensive in male white rats. Proceedings of the International Conference on Natural Sciences, Mathematics, Application, and Technology (ICON-SMART) 2022; 2, 10-17.
[14] D Maaliki, AA Shaito, G Pintus, A El-Yazbi and AH Eid. Flavonoids in hypertension: A brief review of the underlying mechanisms. Current Opinion in Pharmacology 2019; 45, 57-65.
[15] AP Haynes, S Desta, T Ahmad, K Neikirk, A Hinton, N Bloodworth and A Kirabo. The antioxidative effects of flavones in hypertensive disease. Biomedicines 2023; 11(11), 2877.
[16] A Szpicer, W Bińkowska, A Stelmasiak, I Wojtasik-Kalinowska, A Czajkowska, S Mierzejewska, Z Domiszewski, T Rydzkowski, J Piepiórka-Stepuk and A Półtorak. Innovative microencapsulation techniques of bioactive compounds: Impact on physicochemical and sensory properties of food products and industrial applications. Applied Sciences 2025; 15(22), 11908.
[17] D Das Emon, MS Islam, MAR Mazumder, MG Aziz and MS Rahman. Recent applications of microencapsulation techniques for delivery of functional ingredient in food products: A comprehensive review. Food Chemistry Advances 2025; 6, 100923.
[18] JB Harborne. Phytochemical methods: A guide to modern techniques of plant analysis. 3rd ed. Chapman & Hall, London, 1998.
[19] S Nur, A Aisyah, N Nursamsiar, F Sami, A Fadri, N Khairi and A Sapra. Standardization and GC-MS analysis of Kersen (Muntingia calabura L.) fruit ethanol extract as an herbal raw material. Bulletin of Pharmaceutical Sciences Assiut 2023; 46(2), 185037.
[20] S Nur, FJ Sami, M Marwati, N Nursamsiar, A Fadri and K Khairuddin. Phenolic and flavonoid content of black mulberry (Morus nigra L.) stem and their evaluation antioxidant and cytotoxic profile. Borneo Journal of Pharmacy 2022; 5(4), 384-395.
[21] SA Mahdavi, SM Jafari, E Assadpoor and D Dehnad. Microencapsulation optimization of natural anthocyanins with maltodextrin, gum Arabic and gelatin. International Journal of Biological Macromolecules 2016; 85, 379-385.
[22] L Sadiah, R Indiarto and Y Cahyana. Karakteristik dan senyawa fenolik mikrokapsul ekstrak daun kelor (Moringa oleifera) dengan kombinasi maltodekstrin dan whey protein iso (in Indonesian). Jurnal Teknologi Industri Pertanian 2022; 32(3), 273-282.
[23] J de Cássia Gomes da Rocha, TCB Rigolon, LLR Borges, ALAA Nascimento, NA Neves, ÍT Perrone, R Stephani and PC Stringheta. Anthocyanin stability in a mix of phenolic extracts microencapsulated by maltodextrine, whey protein and gum arabic. Journal of Food and Nutrition Research 2023; 11(1), 1-12.
[24] S Nur, MFN Alwani, FJ Sami, A Sapra, AN Aisyah, M Indrisari, Nursamsiar, NA Yusuf and Aristianti. Transfersome-based delivery of Muntingia calabura fruit extract for anti-aging applications. Biocatalysis and Agricultural Biotechnology 2025; 66, 103604.
[25] S Nur, FJ Sami, A Sapra, AN Aisyah, A Burhan, Megawati, Marwati, NA Yusuf, Mislia and Nursamsiar. Application Box-Behnken design for the optimum cream formula of Muntingia calabura fruit extract: In vitro and In vivo studies as an antiaging product. Research Journal of Pharmacy and Technology 2024; 17(12), 5325-5335.
[26] K Qiao, M Zhao, Y Huang, L Liang and Y Zhang. Bitter perception and effects of foods rich in bitter compounds on human health: A comprehensive review. Foods 2024; 13(23), 3747.
[27] X Chu, W Zhu, X Li, E Su and J Wang. Bitter flavors and bitter compounds in foods: identification, perception, and reduction techniques. Food Research International 2024; 183, 114234.
[28] F Husain, Y Ysrafil, RPA Daud, AN Aisyah, A Fadri and S Nur. Authentication of medicinal chemicals in traditional herbal products (Jamu) by UV-Vis spectrophotometry. Hacettepe University Journal of the Faculty of Pharmacy 2023; 43(2), 1090673.
[29] CDA Irudaya, U Venkatesan, R Srinivasan, S Moorthy and K Raja. Evaluation of stability and determination of shelf life of an herbal food supplement: Naturovita. Journal of Pharmacognosy and Phytochemistry 2023; 12(1), 219-225.
[30] M Vera Zambrano, B Dutta, DG Mercer, HL MacLean and MF Touchie. Assessment of moisture content measurement methods of dried food products in small-scale operations in developing countries: A review. Trends in Food Science and Technology 2019; 88, 484-496.
[31] A Monti, N Di Virgilio and G Venturi. Mineral composition and ash content of six major energy crops. Biomass and Bioenergy 2008; 32(3), 216-223.
[32] P Dey, A Kundu, A Kumar, M Gupta, BM Lee, T Bhakta, S Dash and HS Kim. Analysis of alkaloids (indole alkaloids, isoquinoline alkaloids, tropane alkaloids). In: AS Silva, SF Nabavi, M Saeedi and SM Nabavi (Eds.). Recent advances in natural products analysis. Elsevier, Amsterdam, Netherland, 2020, p. 505-567.
[33] YS Ch’ng, YC Loh, CS Tan, M Ahmad, MZ Asmawi, WMW Omar and MF Yam. Vasorelaxant properties of Vernonia amygdalina ethanol extract and its possible mechanism. Pharmacognosy Biology 2017; 55(12), 2083-2094.
[34] EC Pham, VV Doan, TVL Thi, CV Ngo and LV Van. In vivo and in silico antihypertensive, anti-inflammatory, and analgesic activities of Vernonia amygdalina Del. leaf extracts. Heliyon 2024; 10(5), e38634.
[35] Z Zhou, J Chen, Y Cui, R Zhao, H Wang, R Yu, T Jin, J Guo and Y Cong. Antihypertensive activity of different components of Veratrum alkaloids through metabonomic data analysis. Phytomedicine 2023; 120, 155033.
[36] R Caminiti, C Carresi, R Mollace, R Macrì, F Scarano, F Oppedisano, J Maiuolo, M Serra, S Ruga, S Nucera, A Tavernese, M Gliozzi, V Musolino, E Palma, C Muscoli, S Rubattu, M Volterrani, M Federici, M Volpe and V Mollace. The potential effect of natural antioxidants on endothelial dysfunction associated with arterial hypertension. Frontiers in Cardiovascular Medicine 2024; 11, 1345218.
[37] H Li and Q Zhang. Research progress of flavonoids regulating endothelial function. Pharmaceuticals 2023; 16(9), 1201.
[38] MA Asif, SR Lisa and N Qais. Exploring the anti-hypertensive properties of medicinal plants and their bioactive metabolites: An extensive review. American Journal of Plant Sciences 2021; 12(11), 1705-1740.
[39] TAFT Anuar and A Ismail. Southeast Asian medicinal plants with angiotensin converting enzyme (ACE) inhibition properties. Pharmacognosy Journal 2020; 12(6), 1429-1439.
[40] H Luo, J Chen, C Su and L Zha. Advances in the Bioactivities of Phytochemical Saponins in the Prevention and Treatment of Atherosclerosis. Nutrients 2022; 14(23), 4998.
[41] X Ma, J Lu, XR Gu, Y Jia, B Shen, Y Weiming, GH Du and CB Zheng. Cardioprotective effects and mechanisms of saponins on cardiovascular disease. Natural Product Communications 2022; 17(12), 1-8.
[42] D Zielińska, M Starowicz, M Wronkowska and H Zieliński. Angiotensin-converting enzyme inhibitory activity of selected phenolic acids, flavonoids, their o-glucosides, and low-molecular-weight phenolic metabolites in relation to their oxidation potentials. Metabolites 2025; 15(7), 443.
[43] L Guerrero, J Castillo, M Quiñones, S Garcia-Vallvé, L Arola, G Pujadas and B Muguerza. Inhibition of Angiotensin-Converting Enzyme Activity by Flavonoids: Structure-Activity Relationship Studies. PLoS One 2012; 7(12), e49493.
[44] H Silva and NMF Lopes. Cardiovascular effects of caffeic acid and its derivatives: A comprehensive review. Frontiers in Physiology 2020; 11, 595516.
[45] OR Alara, NH Abdurahman, CI Ukaegbu and NA Kabbashi. Extraction and characterization of bioactive compounds in Vernonia amygdalina leaf ethanolic extract comparing Soxhlet and microwave-assisted extraction techniques. Journal of Taibah University for Science 2019; 13(1), 414-422.
[46] D Hudiyanti, MF Al Khafiz, K Anam, P Siahaan, L Suyati, S Sunarsih and SM Christa. Prospect of Gum Arabic–Cocoliposome Matrix to Encapsulate Curcumin for Oral Administration. Polymers 2024; 16(7), 944.
[47] MV Loureiro, A Aguiar, RG dos Santos, JC Bordado, I Pinho and AC Marques. Design of Experiment for Optimizing Microencapsulation by the Solvent Evaporation Technique. Polymers 2024; 16(1), 111.
[48] EF Apriani, S Septina and A Ahmadi. Effect of stirring speed and stirring time on characterization of clindamycin HCL ethosomal and ICH Q1A (R2) stability test. Farmacia 2023; 71(5), 956-965.
[49] SM Jafari, E Assadpoor, Y He and B Bhandari. Encapsulation efficiency of food flavours and oils during spray drying. Drying Technology 2008; 26(7), 816-835.
[50] D Story, A Aminoroaya, Z Skelton, M Kumari, Y Zhang and BR Smith. Nanoparticle-based therapies in hypertension. Hypertension 2023; 80(11), 2506-2514.
[51] AMM Tamboli, J Munaf Tade and Zeta Potential. A comprehensive review. International Research Journal of Pharmacy and Medical Sciences 2025; 8, 115-124.
[52] K Sakulnarmrat and I Konczak. Encapsulation of Melodorum fruticosum Lour. Anthocyanin-rich extract and its incorporation into model food. LWT 2022; 153, 112546.
[53] A Al-Hamayda, B Abu-Jdayil, M Ayyash and J Tannous. Advances in microencapsulation techniques using Arabic gum: A comprehensive review. Industrial Crops and Products 2023; 205, 117556.
[54] W Yang, Y Gong, Y Wang, C Wu, X Zhang, J Li and D Wu. Design of gum Arabic/gelatin composite microcapsules and their cosmetic applications in encapsulating tea tree essential oil. RSC Advances 2024; 14(9), 4880-4889.
[55] N Nadali, A Pahlevanlo, M Sarabi-Jamab and A Balandari. Effect of maltodextrin with different dextrose equivalents on the physicochemical properties of spray-dried barberry juice (Berberis vulgaris L.). Journal of Food Science and Technology 2022; 59(7), 2855-2866.