
Trends
Sci.
2026;
23(7):
12840
The Active Fraction of Physalis angulata Linn. Increases PPARγ and GLUT4 mRNA Expression in an In Vitro Insulin-Resistant C2C12 Cells
Jessica Gita Batoteng1,2, Mae Sri Hartati Wahyuningsih3,4,* and Fara Silvia Yuliani3,4
1Biomedical Science Master Study Program, Faculty of Medicine, Public Health, and Nursing, Universitas Gadjah Mada, Yogyakarta 55281, Indonesia
2Faculty of Medicine, Universitas Atma Jaya Yogyakarta, Yogyakarta 55281, Indonesia
3Department of Pharmacology, Faculty of Medicine, Public Health, and Nursing, Universitas Gadjah Mada,
Yogyakarta 55281, Indonesia
4Center for Herbal Medicine, Faculty of Medicine, Public Health, and Nursing, Universitas Gadjah Mada,
Yogyakarta 55281, Indonesia
(*Corresponding author’s e-mail: [email protected])
Received: 28 November 2025, Revised: 30 December 2025, Accepted: 6 January 2026, Published: 10 March 2026
Abstract
Insulin resistance, a key feature of type 2 diabetes, is characterized by impaired glucose uptake associated with dysregulation of PPARγ and GLUT4. This study investigated the effects of the active fraction of Physalis angulata Linn. (P. angulata), commonly known as ciplukan, on PPARγ and GLUT4 mRNA expression in an in vitro insulin-resistant C2C12 cell model. Insulin resistance was induced using palmitate, followed by treatment with the active fraction of P. angulata (100 μg/mL) for 4, 12, and 24 h. PPARγ and GLUT4 mRNA expression levels were quantified using reverse transcription quantitative real-time polymerase chain reaction (RT-qPCR). The active fraction of P. angulata significantly increased PPARγ mRNA expression at 4, 12, and 24 h and GLUT4 mRNA expression at 12 h compared with the control group (p < 0.05). These results demonstrate a time-dependent upregulation of genes involved in lipid metabolism and glucose transport in insulin-resistant C2C12 cells. The findings suggest that the active fraction of P. angulata may influence the expression of PPARγ and GLUT4 mRNA in insulin-resistant C2C12 cells, warranting further investigation.
Keywords: P. angulata active fraction, Insulin resistance, GLUT4, PPAR, Good health and well-being, C2C12 myotubes, Palmitate-induced insulin resistance
Introduction
Type 2 diabetes mellitus is a metabolic disorder characterized by impaired insulin action resulting from insulin resistance and/or insufficient insulin secretion. This condition accounts for more than 90% of all diabetes cases and predominantly affects adults, representing a substantial global health burden. The increasing prevalence of type 2 diabetes mellitus, including in Indonesia, underscores the need for effective therapeutic strategies to improve glucose homeostasis and insulin sensitivity [1,2].
At the molecular level, insulin resistance in type 2 diabetes mellitus is closely associated with dysregulation of peroxisome proliferator-activated receptor gamma (PPARγ) and glucose transporter type 4 (GLUT4). GLUT4 is the primary insulin-responsive glucose transporter in skeletal muscle and is responsible for approximately 80% of postprandial glucose uptake. Reduced expression of GLUT4 or impaired translocation of this transporter to the plasma membrane leads to decreased glucose uptake and subsequent hyperglycemia. Peroxisome proliferator-activated receptor gamma plays a critical regulatory role in glucose metabolism by regulating the transcription of genes involved in insulin sensitivity, including GLUT4. Decreased expression of PPARγ further exacerbates insulin resistance, creating a vicious cycle of impaired insulin signaling and glucose utilization. Several studies have demonstrated that reduced transcription of PPARγ and GLUT4 mRNA results in diminished protein expression, impaired GLUT4 translocation, and decreased glucose uptake in skeletal muscle cells [3-5].
Given the limitations of current pharmacological therapies, increasing attention has been directed toward herbal-based interventions as complementary strategies for the management of type 2 diabetes mellitus. Physalis angulata L. (Ciplukan) is a medicinal plant containing various bioactive compounds, including flavonoids, alkaloids, steroids, and phytosterols, which have been reported to exhibit antihyperglycemic activity. Experimental studies have shown that active fractions of Physalis angulata enhance glucose uptake in skeletal muscle cells. Wahyuningsih et al. [6] reported that Fraction I of Physalis angulata increased glucose consumption in C2C12 myotubes by 26.47%. Consistently, the active fraction of Physalis angulata has been shown to enhance the phosphorylation of insulin receptor substrate-1 at Tyr-612 and Akt/protein kinase B at Ser-473, which are key components of the insulin signaling pathway [7]. Furthermore, Rakhmawati et al. [8] identified campesterol and stigmasterol as key constituents that facilitate GLUT4 translocation through AS160-related signaling mechanisms [8]. These findings suggest that Physalis angulata may improve insulin sensitivity through modulation of insulin signaling pathways in skeletal muscle cells.
Despite growing evidence of its antihyperglycemic effects, the molecular mechanisms underlying the action of Physalis angulata, particularly its effects on GLUT4 and PPARγ mRNA expression, remain insufficiently explored. Therefore, this study aims to evaluate the effects of active fractions of Physalis angulata L. on GLUT4 and PPARγ mRNA expression in insulin-resistant C2C12 cells as an in vitro skeletal muscle model. Elucidating these molecular effects is expected to provide mechanistic insight into the antidiabetic potential of Physalis angulata and to support its development as a complementary therapeutic option for type 2 diabetes mellitus.
Materials and methods
Materials
DMEM was sourced from Gibco (Lot: 2512444), FBS (Fetal Bovine Serum) from Sigma-Aldrich (Lot: 001671877), HS (Horse Serum) from Sigma-Aldrich (Lot: 22B441). Antibiotics were from Elabscience (Lot: GY1142RN8913), and BSA was from Elabscience (Lot: DI02260R2362). The GOD PAP kit was from DSI (Lot: 60159945), The RNA extraction kit used was obtained from Favorgen, while the cDNA synthesis kit was sourced from Smobio, and the qPCR kit was SensiFAST SYBR Lo-ROX. Primers for GLUT4, PPARγ, and housekeeping gene β-actin were obtained from Macrogen. The active fraction of P. angulata was provided by the Department of Pharmacology and Therapy, Faculty of Medicine, Public Health, and Nursing, Universitas Gadjah Mada (UGM). This study has obtained approval from the Ethics Committee of the Faculty of Medicine, Public Health, and Nursing, Universitas Gadjah Mada, Yogyakarta, Indonesia (Approval Number: KE/FK/0379/EC/2024).
Cell culture and differentiation
C2C12 cells obtained from Department of Pharmacology and Therapy, Faculty of Medicine, Public Health, and Nursing, UGM. The cells were at passage 50. C2C12 myoblasts were maintained in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% FBS at 37 °C in a 5% CO2. Differentiation of C2C12 cells was induced by replacing the medium with DMEM containing 2% Horse Serum (HS). Experiments were conducted after 6 to 8 days of differentiation.
Palmitate-induced insulin resistance in C2C12 myotubes
Insulin resistance was induced in differentiated C2C12 myotubes using palmitate. A palmitate stock solution (500 mM) was prepared by dissolving palmitic acid in absolute ethanol with heating at 70 °C. A 10% bovine serum albumin (BSA) solution was prepared in basal DMEM and incubated at 37 °C until fully dissolved. Palmitate was conjugated to BSA to obtain a 5 mM palmitate-BSA stock solution, which was aliquoted and stored at ‒20 °C. Working concentrations of palmitate-BSA were prepared by dilution in DMEM free-serum (DMEM-FS). Vehicle control cells were treated with BSA containing an equivalent concentration of ethanol without palmitate. To induce insulin resistance, differentiated C2C12 myotubes were exposed to 0.75 mM palmitate-BSA for 16 h. After induction, cells were washed once with phosphate-buffered saline (PBS) to remove residual palmitate prior to subsequent treatments.
Glucose consumption assay
Glucose consumption was assessed by measuring residual glucose concentration in the culture medium using the glucose oxidase-phenol 4-aminoantipyrine peroxidase (GOD-PAP) method according to the manufacturer’s instructions. C2C12 cells were seeded at a density of 3×10³ cells per well in 96-well plates and differentiated. Fully differentiated cells were divided into 6 groups: Media control (KM, culture medium without cells), cell control (KS, untreated cells) and four palmitate-treated groups (0.25, 0.5, 0.75, and 1 mM). Cells were treated with palmitate as described above and incubated for 16 h. Following incubation, culture supernatants were collected and incubated with the GOD-PAP reagent. Absorbance was measured at 490 nm using an ELISA reader, and glucose concentrations (mg/dL) were calculated using a standard calibration curve according to the following formula:
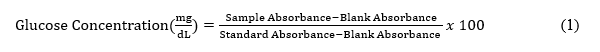
Fraction Physalis angulata L. preparation
The active fraction of Physalis angulata L. was prepared using a bioassay-guided fractionation method as previously described by Wahyuningsih et al. [6]. The active fraction used in this study corresponds to Fraction I, which exhibited the highest glucose uptake-enhancing activity (26.47%) among Fractions I - IV in C2C12 myoblast cells. Fraction I represents a mixture of compounds derived from the methanol-insoluble partition of the chloroform extract and separated by vacuum liquid chromatography using a gradient polarity mobile phase, with thin-layer chromatography employed to monitor the fractionation process. Based on prior characterization, this fraction comprises multiple classes of compounds, predominantly flavonoids, and also contains phytosterols, with campesterol and stigmasterol identified as representative phytosterol constituents [6,8].
The manufacturing process consisted of three main stages: Extraction, partitioning, and fractionation. A total of 500 g of dried P. angulata herb powder was extracted using the maceration method. Maceration was carried out with continuous stirring for 24 h at room temperature using methanol and chloroform solvents. The success of the extraction process was monitored using thin-layer chromatography (TLC) and a glucose consumption assay [6].
Based on previous in vitro studies using C2C12 myotubes, the active fraction was applied at a concentration of 100 µg/mL, which has been reported to be effective and non-cytotoxic under insulin-resistant conditions [9]. The experimental grouping, including palmitate exposure, active fraction treatment, insulin stimulation prior to harvesting, and sampling time points before RT-qPCR analysis, is summarized in Table 1.
Table 1 Experimental grouping.
Group |
Palmitate |
Insulin |
Active fraction of P. Angulata L. 100 μg/mL |
Solvent of active fraction of physalis angulata L. (DMSO + DMEM free serum) |
Sampling time |
|
KS |
‒ |
+ |
‒ |
+ |
Hour 0 |
|
F1 |
+ |
+ |
+ |
‒ |
Hour 4 |
|
F2 |
+ |
+ |
+ |
‒ |
Hour 12 |
|
F3 |
+ |
+ |
+ |
‒ |
Hour 24 |
|
Experimental Grouping. KS: Cell control; F1: Fraction 1; F2: Fraction 2; F3: Fraction 3.
Treatment with P. angulata active fraction
Differentiated C2C12 myotubes were seeded in 6-well plates at a density of 2.5×10⁵ cells per well in a total volume of 2 mL medium. Insulin resistance was induced by exposure to palmitate (0.75 mM) for 16 h. After induction, cells were washed once with phosphate-buffered saline (PBS) to remove residual palmitate prior to fraction treatment. The active fraction of Physalis angulata L. was dissolved in DMSO and diluted with DMEM free-serum (DMEM-FS) to obtain a final concentration of 100 µg/mL, based on previous studies. The final DMSO concentration in all treatment and control conditions was maintained at 0.1 %(v/v). Cells were treated with the active fraction at 100 µg/mL for 4, 12, or 24 h. Vehicle control cells received an equivalent concentration of DMSO (0.1 %(v/v) without the active fraction. Following fraction exposure, all groups were stimulated with insulin (100 nM) for 30 min prior to sample harvesting. Cells were then washed with cold PBS, collected, and stored at −80 °C for subsequent RT-qPCR analysis. Each experimental condition was performed using technical replicates.
Reverse transcription-quantitative polymerase chain reaction (RT-qPCR)
RNA extraction was performed using the FavorPrep™ Tissue Total RNA Mini Kit, following the manufacturerʼs instructions. Insulin stimulation was applied only immediately prior to cell harvesting to activate the insulin signaling pathway and was not included during the treatment period. All experimental groups received the same insulin stimulation prior to RT-qPCR analysis. cDNA synthesis was conducted using the Smobio ExcelRT™ Reverse Transcription Kit II, and quantitative PCR (qPCR) was carried out with the SensiFAST™ SYBR® Lo-ROX Kit (Cat. No: BIO-94005) in a total reaction volume of 20 µL. qPCR was performed using the 7,500 Fast Real-Time PCR System (Applied Biosystems, Singapore). The reaction followed a two-step PCR protocol: An initial denaturation at 95 C for 2 min, followed by 40 cycles of 95 C for 5 s, annealing at 62 C for 30 s, and extension at 72 C for 20 s. The mRNA expression levels of PPARγ and GLUT4 were normalized to the housekeeping gene β-actin. The relative expression of the target genes was determined using the 2^-ΔΔCT method. The primer sequences were as follows: For GLUT4, the forward primer was 5’-TTGCACACGGCTTCCGAACG-3’, and the reverse primer: 5’-GATCT CTGGAAACCCGACGG-3’ [5]. For PPARγ, the forward primer was 5’-GCCAAGGTGCTCCAGAAGAT-3’, and the reverse primer: 5’-AGGAACACGTTGTCAGCGG-3’ [10]. For the housekeeping gene β-actin, the forward primer was 5’- AGCCCAGAACATCATCCCTG -3’, and the reverse primer: 5’-CACCACCTTCTTGATGTCATC-3’ [11].
Data and statistical analysis
Experiments were performed using two independent technical replicates, with cells cultured in separate wells. Data from insulin resistance validation and mRNA expression of GLUT4 and PPARγ were analyzed using GraphPad Prism (version 10.3.1) for Windows. The normality of the data was evaluated using the Shapiro-Wilk test. Data that were normally distributed (p > 0.05) were analyzed with One-Way ANOVA, followed by Tukeyʼs multiple comparisons test to determine significant differences between groups. Statistical significance was defined as p < 0.05.
Results and discussion
Differentiation of c2c12 cells
Figure 1 shows the differentiation of C2C12 myoblasts into multinucleated myotubes over an 8-day period. At day 0, cells displayed a round and dispersed morphology characteristic of the proliferative myoblast stage. By days 2 and 4, cells exhibited elongation and alignment, indicating the onset of cell fusion. More prominent tubular structures were observed at day 6, and by day 8, most cells had differentiated into multinucleated myotubes with muscle fiber like morphology.
These morphological changes are consistent with previous reports describing the differentiation of C2C12 cells from mononucleated myoblasts into elongated, multinucleated myotubes, which represent a well-established in vitro skeletal muscle model. Differentiated C2C12 myotubes have been reported to exhibit metabolic characteristics relevant to insulin signaling studies, including insulin-responsive glucose uptake and high GLUT4 expression [12].

Figure 1 Differentiation of C2C12 cells from myoblasts to myotubes. Differentiation at day 0 (a), day 2 (b), day 4 (c), day 6 (d), and day 8 (e). Images of the cells were taken at 10x magnification using an inverted Nikon light microscope.
Validation of insulin resistance in C2C12 cells via glucose consumption assay
Glucose consumption in C2C12 cells was evaluated using the glucose oxidase-phenol 4-aminoantipyrine peroxidase (GOD-PAP) method following exposure to different concentrations of palmitate. As shown in Figure 2, treatment with 0.75 mM palmitate resulted in a significantly higher glucose concentration in the culture medium compared with the cell control group (p < 0.05), indicating reduced glucose consumption and successful induction of insulin resistance. In contrast, palmitate concentrations of 0.25, 0.5, and 1 mM did not produce statistically significant differences in glucose levels compared with controls. Based on these results, 0.75 mM palmitate was selected for subsequent experiments involving the active fraction of Physalis angulata L.
Palmitate is widely used to induce insulin resistance in skeletal muscle cells due to its lipotoxic effects, which involve mitochondrial dysfunction, oxidative stress, and activation of inflammatory pathways [13,14]. Previous studies have shown that palmitate promotes the accumulation of lipid intermediates such as diacylglycerol and ceramide, leading to impaired insulin signaling through inhibition of IRS-1 and Akt phosphorylation [15,16]. In addition, palmitate-induced oxidative stress and activation of inflammatory mediators, including NF-κB, contribute to disrupted glucose metabolism and reduced insulin responsiveness in C2C12 myotubes [17]. These mechanisms provide a biological explanation for the reduced glucose consumption observed following exposure to 0.75 mM palmitate in the present study and support its use as an effective concentration for modeling insulin resistance in vitro.
Nevertheless, it should be acknowledged that cell viability was not directly assessed in the present study; therefore, although insulin resistance was established based on glucose consumption data and prior validation studies, the observed molecular changes should be interpreted with consideration of this limitation.
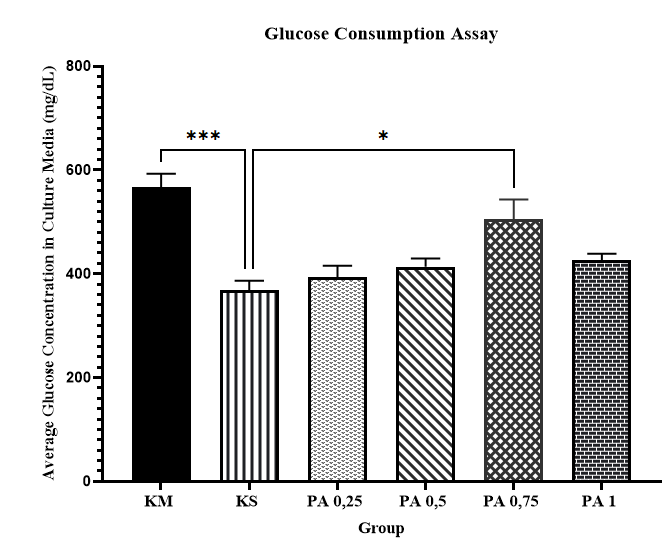
Figure 2 Glucose consumption assay in insulin-resistant C2C12 cells after treatment with varying concentrations of palmitate. The data are expressed as mean ± SEM and were analyzed using One-Way ANOVA, followed by Tukeyʼs post hoc multiple comparisons test. KM = Media Control, KS = Cell Control, PA 0.25 = 0.25 mM palmitate, PA 0.5 = 0.5 mM palmitate, PA 0.75 = 0.75 mM palmitate, PA 1 = 1 mM palmitate. **p < 0.05 KS vs. PA 0.75 (n = 3), **p < 0.001 KS vs. KM (n = 3).
Effects of P. angulata active fractions on mRNA expression of PPARγ and mRNA expression GLUT4 in Insulin-resistant C2C12 cells
Treatment with the active fraction of Physalis angulata significantly altered PPARγ and GLUT4 mRNA expression in insulin-resistant C2C12 cells in a time-dependent manner. As shown in Figure 3, PPARγ mRNA expression was significantly increased at 4 h (p < 0.05), reached the highest level at 12 h (p < 0.01), and remained significantly elevated at 24 h (p < 0.05) compared with the control group.
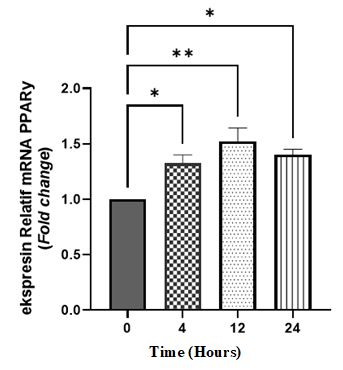
Figure 3 Relative mRNA expression of PPARγ in insulin-resistant C2C12 cells after treatment with P. angulata active fractions. The results are shown as mean ± SEM and were statistically analyzed using One-Way ANOVA, followed by Tukey’s multiple comparisons test. Groups: 0 = control, 4 = 4-hour incubation, 12 = 12-hour incubation, 24 = 24-hour incubation. *p < 0.05 (4 h vs. 0 h and 24 h vs. 0 h) (n = 2)., *p < 0.05 (12 h vs. 0 h) (n= 2).
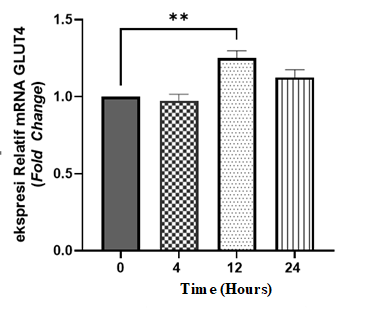
Figure 4 Relative mRNA expression of GLUT4 in insulin-resistant C2C12 cells after treatment with P. angulata active fractions. Data are presented as mean ± SEM and analyzed using One-Way ANOVA followed by Tukeyʼs multiple comparisons test. Groups: 0 = control, 4 = 4-hour incubation, 12 = 12-hour incubation, 24 = 24-hour incubation. *p < 0.01 (12 h vs. 0 h), (n = 2).
As shown in Figure 4, GLUT4 mRNA expression exhibited a distinct temporal pattern. A significant increase in GLUT4 mRNA expression was observed only at 12 h (p < 0.01), whereas no significant changes were detected at 4 or 24 h. These results indicate a transient induction of GLUT4 mRNA expression following treatment with the active fraction. Detailed quantitative RT-qPCR data, including mean ± SEM and fold-change values for PPARγ and GLUT4 at each time-point relative to control, are provided in Supplementary Table S1.
PPARγ is a key transcriptional regulator involved in lipid metabolism and insulin sensitivity in skeletal muscle, whereas GLUT4 mediates insulin-stimulated glucose uptake. Disruption of these pathways is a hallmark of palmitate-induced insulin resistance, characterized by reduced GLUT4 expression and impaired glucose handling [16,18]. The earlier and sustained increase in PPARγ mRNA expression compared with the delayed and transient induction of GLUT4 suggests a sequential transcriptional response rather than simultaneous regulation.
Previous studies have demonstrated that increased PPARγ expression can enhance GLUT4 gene transcription and improve glucose uptake in skeletal muscle cells [5,19]. The temporal pattern observed in the present study, with peak GLUT4 mRNA expression occurring after maximal PPARγ induction, is consistent with this regulatory relationship.
The decline in both transcripts at 24 h, although PPARγ expression remained above control levels, may reflect a transient transcriptional response to the active fraction. The biological activity of this fraction is supported by previous studies identifying Fraction I of P. angulata as effective in improving glucose metabolism [6]. This activity has been attributed to bioactive constituents such as flavonoids and phytosterols, including stigmasterol and campesterol, which have been reported to modulate glucose metabolism through PPAR-dependent pathways [8,20].
Although the present study focused on transcriptional changes at the mRNA level, other signaling pathways involved in glucose metabolism may also contribute to the observed effects and warrant further investigation. Overall, these findings suggest that the active fraction of P. angulata modulates PPARγ and GLUT4 expression at the mRNA level in insulin-resistant C2C12 cells.
While these findings provide valuable insight, several limitations should be acknowledged. In addition to the absence of direct cell viability assessment and the limited sample size, C2C12 cells were used at relatively high passage numbers without formal validation of differentiation capacity or insulin responsiveness. Although efforts were made to limit variability by using closely related passage ranges, complete uniformity across experimental groups could not be ensured. Accordingly, the findings reflect the experimental model and study design employed.
Conclusions
This study shows that treatment with the active fraction of Physalis angulata Linn. (100 μg/mL) is associated with increased mRNA expression of PPARγ and GLUT4 in insulin-resistant C2C12 cells in a time-dependent manner. PPARγ mRNA expression was significantly elevated at 4, 12, and 24 h, while a significant increase in GLUT4 mRNA expression was observed at 12 h. These findings indicate that the active fraction of P. angulata is linked to changes in the expression of PPARγ and GLUT4 at the mRNA level under insulin-resistant conditions. Further studies are required to elucidate the underlying molecular mechanisms and to determine the functional and translational relevance of these observations.
Acknowledgements
The authors would like to express their gratitude to the Laboratory of Pharmacology and Therapy, Parasitology Laboratory, and LRT of the Faculty of Medicine, Public Health, and Nursing, UGM, for providing the laboratory facilities and equipment essential for this research. The authors also gratefully the support and resources provided by Universitas Atma Jaya Yogyakarta 55281, Indonesia.
Declaration of Generative AI in Scientific Writing
The authors acknowledge the use of generative AI tools (e.g., ChatGPT by OpenAI) in the preparation of this manuscript, specifically for language editing and grammar refinement. No content generation or data interpretation was performed by AI, and the authors take full responsibility for all content and conclusions presented in this work.
CRediT Author Statement
Jessica Gita Batoteng: Conceptualization; Data Curation; Writing - Original Draft; Visualization; Formal analysis. Mae Sri Hartati Wahyuningsih: Conceptualization; Methodology; Writing - Review & Editing; Supervision. Fara Silvia Yuliani: Writing - Review & Editing; Validation; Data Curation; Formal analysis.
References
[1] D Lindarto, E Decroli, H Permana, KW Sucipto, Y Kusnadi, Budiman, MR Ikhsan, L Sasiarini and H Sanusi. Pedoman Pengelolaan dan Pencegahan Diabetes Melitus Tipe 2 di Indonesia 2021. Perkumpulan Endokrinologi Indonesia, Indonesia, 2021.
[2] WHO, Available at: https://www.who.int/health-topics/diabetes#tab=tab_1, accessed July 2023.
[3] U Galicia-Garcia, A Benito-Vicente, S Jebari, A Larrea-Sebal, H Siddiqi, KB Uribe, H Ostolaza and C Martín. Pathophysiology of type 2 diabetes mellitus. International Journal of Molecular Sciences 2020; 21(17), 6275.
[4] K Ahmad, S Shaikh, JH Lim, SS Ahmad, HJ Chun, EJ Lee and I Choi. Therapeutic application of natural compounds for skeletal muscle-associated metabolic disorders: A review on diabetes perspective. Biomedicine & Pharmacotherapy 2023; 168, 115642.
[5] S Estrada-Soto, K Ornelas-Mendoza, G Navarrete-Vázquez, F Chávez-Silva, JC Almanza-Pérez, R Villalobos-Molina, E Ortiz-Barragán, H Loza-Rodríguez, JC Rivera-Leyva, A Flores-Flores, I Perea-Arango, JG Rodríguez-Carpena and G Ávila-Villarreal. Insulin sensitization by PPARγ and GLUT-4 overexpression/translocation mediates the antidiabetic effect of Plantago australis. Pharmaceuticals 2023; 16(4), 535.
[6] MSH Wahyuningsih, KSS Wiwekananda, APR Putri, DAA Nugrahaningsih and MM Yuniyanti. Bioassay guided fractionation of Ciplukan (Physalis angulata L.) monitored by glucose consumption assay and thin layer chromatography on myoblast cells. Traditional Medicine Journal 2023; 28(1), 22-30.
[7] FB Papilaya, MSH Wahyuningsih and RA Syarif. Active fraction of Ciplukan (Physalis angulate L.) increases phosphorylation on IRS-1 Tyr-612 and Akt Ser-473 in 24-hour incubation time. Journal of Applied Pharmaceutical Science 2025; 15(5), 121-128.
[8] R Rakhmawati, ST Wahyudi, MSH Wahyuningsih, Mustofa and AH Sadewa. GC-MS and in silico analyses revealed the potential inhibitory activity of compounds isolated from Ciplukan herb (Physalis angulata L.) targeting GLUT-4 receptors. Journal of Applied Pharmaceutical Science 2024; 14(8), 142-149.
[9] YC Prasetyo. Efek fraksi aktif herba Ciplukan (Physalis Angulata Linn) terhadap konsumsi glukosa sel C2C12 Myotube: Kajian pada model resistensi insulin secara in vitro. Universitas Gadjah Mada, Indonesia, 2023.
[10] Ensembl, Transcript: Exons - Mus musculus (ENSMUST00000000450). Available at: https://asia.ensembl.org/Mus_musculus/Transcript/Exons?db=core;g=ENSMUSG00000000440;r=6:115337912-115467360;t=ENSMUST00000000450, accessed April 2024.
[11] Y Xing, J Zhang, H Wei, H Zhang, Y Guan, X Wang and X Tong. Reduction of the PI3K/Akt related signaling activities in skeletal muscle tissues involves insulin resistance in intrauterine growth restriction rats with catch-up growth. PLoS One 2019; 14(5), 0216665.
[12] CY Wong, H Al-Salami and CR Dass. C2C12 cell model: Its role in understanding of insulin resistance at the molecular level and pharmaceutical development at the preclinical stage. Journal of Pharmacy and Pharmacology 2020; 72(12), 1667-1693.
[13] MH Abu Bakar, KA Shariff, JS Tan and LK Lee. Celastrol attenuates inflammatory responses in adipose tissues and improves skeletal muscle mitochondrial functions in high fat diet-induced obese rats via upregulation of AMPK/SIRT1 signaling pathways. European Journal of Pharmacology 2020; 883, 173371.
[14] KC Anderson, J Liu and Z Liu. Interplay of fatty acids, insulin and exercise in vascular health. Lipids in Health and Disease 2025; 24(4), 1-13.
[15] Y Geng, KN Faber, VE de Meijer, H Blokzijl and H Moshage. How does hepatic lipid accumulation lead to lipotoxicity in non ‑ alcoholic fatty liver disease? Hepatology International 2021; 15(1), 31-35.
[16] WT Chuang, CC Yen, CS Huang, HW Chen and CK Lii. Benzyl isothiocyanate ameliorates high-fat diet-induced hyperglycemia by enhancing Nrf2-dependent antioxidant defense-mediated IRS-1/AKT/TBC1D1 signaling and GLUT4 expression in Skeletal Muscle. Journal of Agricultural and Food Chemistry 2020; 68(51), 15228-15238.
[17] G Taheripak, N Sabeti, N Najar, Z Razavi, S Saharkhiz and I Alipourfard. SIRT1 activation attenuates palmitate induced apoptosis in C2C12 muscle cells. Molecular Biology Reports 2024; 51(1), 354.
[18] VL Tokarz, S Mylvaganam and A Klip. Palmitate-induced insulin resistance causes actin filament stiffness and GLUT4 mis-sorting without altered Akt signalling. Journal of Cell Science 2023; 136(21), 261300.
[19] S Dewanjee, P Paul, TK Dua, S Bhowmick and A Saha. Chapter 38 - Big leaf mahogany seeds: Swietenia macrophylla seeds offer possible phytotherapeutic intervention against diabetic pathophysiology. Academic Press, Massachusetts, 2020, p. 543-565.
[20] B Salehi, C Quispe, J Sharifi-Rad, N Cruz-Martins, M Nigam, AP Mishra, DA Konovalov, V Orobinskaya, IM Abu-Reidah, W Zam, F Sharopov, T Venneri, R Capasso, W Kukula-Koch, A Wawruszak and W Koch. Phytosterols: From preclinical evidence to potential clinical applications. Frontiers in Pharmacology 2021; 11, 599959.
Supplementary Material
Table S1 Relative mRNA expression (2-ΔΔCT) of PPARγ and GLUT4 in C2C12 cells at different time-points.
Gene |
Time-point |
Fold-change vs control (mean ± SEM) |
p-value vs control |
PPARγ |
0 h |
1.00 ± 0.00 |
- |
PPARγ |
4 h |
1.32 ± 0.07 |
0.0430* |
PPARγ |
12 h |
1.52 ± 0.11 |
0.0017* |
PPARγ |
24 h |
1.40 ± 0.04 |
0.0117* |
GLUT4 |
0 h |
1.00 ± 0.00 |
- |
GLUT4 |
4 h |
0.97 ± 0.04 |
>0.05 |
GLUT4 |
12 h |
1.24 ± 0.04 |
0.0048* |
GLUT4 |
24 h |
1.12 ± 0.05 |
>0.05 |