
Trends
Sci.
2026;
23(7):
12516
The Restorative Role of Flavonoids in Immunological, Antioxidant, and Microbiota Mechanisms of Autoimmune Thyroiditis
Khasan
Kayumov1*,
![]() ,
Lubov Kuchkarova1,
,
Lubov Kuchkarova1,
![]() ,
Dilnoza Romanova1,
,
Dilnoza Romanova1,
![]() ,
,
Szczuko
Małgorzata2,
![]() ,
Teodoro Durá-Travé3,
,
Teodoro Durá-Travé3,
![]() ,
Nurali Ergashev4,
,
Nurali Ergashev4,
![]() ,
Iroda Karimova1,
,
Iroda Karimova1,
![]() ,
Sevara Berdiyorova1,
,
Sevara Berdiyorova1,
![]() ,
Visola Pulatova1,
,
Visola Pulatova1,
![]() ,
Sokhibjon Abdusamatov1,
,
Sokhibjon Abdusamatov1,
![]() ,
,
Rashidbek
Achilov1,
![]() ,
Numonjon Sultanov1,
,
Numonjon Sultanov1,
![]() and Akmal Yusupov5,
and Akmal Yusupov5,
![]()
1National University of Uzbekistan named after Mirza Ulugbek, Tashkent, Uzbekistan
2Department of Bromatology and Diagnostic Nutrition, Pomeranian Medical University in Szczecin, Poland
3Faculty of Medicine, University of Navarra, Navarre, Spain
4Institute Biophysics and Biochemistry at National University of Uzbekistan named after Mirza Ulugbek,
Tashkent, Uzbekistan
5Alfraganus University Tashkent, Tashkent, Uzbekistan
(*Corresponding author’s e-mail: [email protected])
Received: 3 November 2025, Revised: 25 November 2025, Accepted: 2 December 2025, Published: 10 February 2026
Abstract
Autoimmune thyroiditis is a multifactorial endocrine disorder characterized by chronic immune-mediated inflammation, oxidative imbalance, and disruption of gut microbiota homeostasis. These pathological processes lead to thyroid hormone deficiency, metabolic dysfunction, and systemic immune polarization. The present study aimed to evaluate the immunological, antioxidant, and microbiota-corrective effects of three plant-derived flavonoids - quercetin, dihydroquercetin (taxifolin), and luteolin - in an experimental rat model of autoimmune thyroiditis. Thyroid hormone concentrations (T3, T4, and fT3), antioxidant enzyme activity (GPX1), macro- and microelement balance (Ca²⁺, Mg²⁺, Fe, K⁺ and P), and intestinal bacterial composition were analyzed to assess the extent of restoration.
Autoimmune thyroiditis caused a pronounced decrease in thyroid hormone levels (T3 −66.5%, T4 −66.7% and fT3 −39.5%) and GPX1 activity (−59.1%), along with disturbances in essential macro- and microelements and microbiota composition. Flavonoid treatment markedly ameliorated these impairments: luteolin restored T3 and T4 levels to near-physiological values and normalized GPX1 activity by 93.6% of the control. Quercetin and dihydroquercetin showed moderate but significant improvements in antioxidant and endocrine parameters. Additionally, flavonoid supplementation rebalanced the gut microbiota, increasing beneficial taxa (Lactobacillus, Bifidobacterium and Faecalibacterium prausnitzii) while reducing pathogenic species (Escherichia coli, Streptococcus and Fusobacterium).
Collectively, the findings demonstrate that natural flavonoids exert a potent corrective influence on immune, redox, and microbiota disturbances in autoimmune thyroiditis. Among them, luteolin exhibited the strongest integrative effect, suggesting its potential as a promising adjunctive therapy to conventional treatment in thyroid autoimmune diseases.
Keywords: Autoimmune thyroiditis, Flavonoids, Luteolin, Quercetin, Dihydroquercetin, Oxidative stress, GPX1, Microbiota, Thyroid hormones, Immunomodulation
Introduction
Autoimmune thyroiditis (AIT), predominantly manifesting as Hashimoto’s thyroiditis, is a chronic organ-specific autoimmune disorder characterized by
lymphocytic infiltration of the thyroid gland, progressive destruction of thyrocytes, and the formation of circulating autoantibodies against thyroglobulin (Tg) and thyroid peroxidase (TPO). This pathology leads not only to endocrine imbalance but also to systemic immunological, oxidative, and metabolic dysfunctions.
In AIT, oxidative stress plays a central role in the cascade of autoimmune damage. Excessive production of reactive oxygen species (ROS), together with diminished activity of key antioxidant enzymes such as glutathione peroxidase-1 (GPX1), leads to lipid peroxidation, membrane destabilization, and the intensification of thyroidal inflammation. Simultaneously, alterations in essential trace elements - including iron, magnesium, zinc, and copper - further compromise thyroid hormone synthesis and antioxidant defense. Iron deficiency impairs the catalytic activity of thyroid peroxidase, whereas magnesium depletion disturbs T-cell regulation and contributes to enhanced pro-inflammatory signaling.
A growing body of evidence supports the involvement of the gut-thyroid axis in AIT pathogenesis. Dysbiosis of intestinal microbiota, characterized by reduced abundance of beneficial bacteria (Lactobacillus, Bifidobacterium and Faecalibacterium prausnitzii) and proliferation of opportunistic or pathogenic species (Escherichia coli, Streptococcus, Fusobacterium) [1-3], has been shown to modulate systemic immunity and affect the differentiation of T helper cell subsets (Treg/Th17). These microbial shifts disrupt intestinal barrier integrity, promote lipopolysaccharide translocation, and stimulate chronic immune activation that ultimately targets thyroid tissue [4,5]. Recent metagenomic and metabolomic analyses confirm that microbiota-derived metabolites - particularly short-chain fatty acids (SCFAs) - influence thyroid homeostasis through immune and neuroendocrine pathways [6-8].
Parallel to immune and microbiota perturbations, AIT induces measurable disturbances in thyroid hormones (total triiodothyronine (T3), free triiodothyronine (fT3) and thyroxine (T4)), resulting in hypothyroidism and metabolic deceleration. The decline of GPX1 activity further reflects compromised redox balance, while changes in serum calcium, magnesium, potassium, and phosphorus signify broader electrolyte and metabolic instability. These multi-level disruptions necessitate an integrative therapeutic approach aimed at restoring immune tolerance, oxidative equilibrium, and gut microbial harmony.
In this context, plant-derived flavonoids - notably quercetin (Que), dihydroquercetin (DHQ) (taxifolin), and luteolin (Lt) -have attracted growing attention due to their pleiotropic antioxidant, anti-inflammatory, and immunomodulatory properties. Que enhances Nrf2-mediated antioxidant gene expression and mitigates NF-κB-driven cytokine cascades [9,10], DHQ exhibits strong free radical scavenging and mitochondrial membrane-stabilizing effects; while Lt effectively suppresses IL-1β, IL-6, and TNF-α production and inhibits the STAT3 and IL-17/IL-23 signaling axes, thereby restoring immune homeostasis. Additionally, these flavonoids beneficially influence intestinal microbiota composition by increasing SCFA-producing taxa and reducing pro-inflammatory species [11].
Therefore, the present study was designed to comprehensively evaluate the restorative effects of Que, DHQ, and Lt in an experimental rat model of AIT. Specifically, we assessed their influence on thyroid hormone homeostasis (T3, T4, fT3), antioxidant defense via GPX1 activity, trace-element balance (Ca²⁺, Mg²⁺, Fe, K⁺, P), and gut microbiota composition. By integrating biochemical, immunological, and microbiological parameters, this research aims to elucidate the multi-target therapeutic potential of natural flavonoids in correcting the immune-redox-microbiota axis disturbances characteristic of AIT.
Therefore, to elucidate these interrelated immune, redox, and metabolic disturbances, we designed an experimental study on AIT rats assessing the impact of flavonoids on thyroid hormones, antioxidant systems, and trace element balance.
Materials and methods
Experimental animals and ethical approval
Adult male Wistar rats (200 - 220 g) were used in the study. Animals were housed under standard laboratory conditions (temperature 22 ± 2 °C, 12 h light/dark cycle, relative humidity 55% - 60%) with free access to standard pellet diet and water ad libitum [12]. All procedures were performed in compliance with the European Directive 2010/63/EU for animal experiments and were approved by the Institutional Animal Care and Ethics Committee of the National University of Uzbekistan (Ethical approval number: NUU-AE-2024-11/27).
Induction of AIT model
The experimental model of AIT was established according to the protocol described by Kong [13] with slight modifications. Rats received subcutaneous injections of 0.1 mL of a 1:1 emulsion containing bovine thyroglobulin (50 µg/100 g body weight; Sigma-Aldrich, Germany) and Complete Freund’s Adjuvant (MP Biomedicals, USA). On days 0, 1, and 7, additional booster injections of thyroglobulin emulsified in Incomplete Freund’s Adjuvant (0.1 mL) were administered subcutaneously to enhance autoimmune sensitization. On day 21, blood samples were collected from the submandibular vein to evaluate anti-Thyroid Peroxidase (anti-TPO) antibody titers and confirm disease induction. Final blood sampling was performed on day 35 following euthanasia by decapitation, after which serum anti-TPO concentrations were analyzed to assess the progression and severity of AIT [14].
Induction and validation of the AIT model
To confirm successful induction of AIT, serum anti-TPO and Thyroid-Stimulating Hormone (TSH) levels were assessed on day 21 and day 35. Both biomarkers were quantified using the enzyme-linked immunosorbent assay (ELISA) method with commercially validated kits from Assay Genie (Dublin 2, Ireland). Specifically, Rat TSH ELISA Kit (RTFI01195) with a sensitivity of 0.75 ng/mL, and Rat TPO-Ab (anti-Thyroid Peroxidase Antibody) ELISA Kit (RTFI01361) with a sensitivity of 0.469 IU/mL, were employed.
The induction protocol and diagnostic thresholds used in this study were identical to those detailed in our previous publication on AIT immunodiagnostics [14]. In accordance with those criteria, all AIT-induced rats exceeded the anti-TPO diagnostic cut-off and demonstrated the characteristic elevation in TSH levels, confirming successful sensitization.
To avoid redundancy, full numerical datasets are not reproduced here. Instead, the distribution of individual values for the current cohort is visualized in Figure 1 (anti-TPO) and Figure 2 (TSH) as dot/box-dot plots. Randomization was performed using block randomization, and all outcome assessments were conducted under blinded conditions.
Experimental design
After confirmation of AIT development based on ELISA results for anti-TPO and TSH, rats were randomly allocated into 5 groups (n = 6 per group):
Control group – received vehicle only (0.5% carboxymethylcellulose, CMC).
AIT group (negative control) – untreated rats with experimentally induced AIT.
AIT + Que – treated with quercetin (25 mg/kg/day).
AIT + DHQ – treated with dihydroquercetin (20 mg/kg/day).
AIT + Lt – treated with luteolin (10 mg/kg/day).
Flavonoids were administered intragastrically once daily at 9:00 a.m. for 14 consecutive days, beginning on day 21 after successful induction of AIT. Each experimental group consisted of 6 Wistar rats, which provided sufficient statistical power for intergroup comparisons in line with established AIT research standards.
All flavonoid compounds (Que, DHQ, and Lt) were obtained from the Laboratory of Terpenoids and Phenolic Compounds, Institute of the Chemistry of Plant Substances, Academy of Sciences of Uzbekistan. The purity of each compound exceeded 98% (confirmed by HPLC). Prior to administration, flavonoids were freshly dissolved in 0.5% aqueous CMC solution, which served as the vehicle control for untreated groups.
Sample collection
At the end of the experimental period, blood samples were collected from the retro-orbital plexus under light anesthesia. Two types of tubes were used: Serum tubes (without anticoagulant) for biochemical and ELISA analyses. EDTA-coated tubes, which had been priorly added with ethylenediaminetetraacetic acid (EDTA), for hematological analysis. Serum was separated by centrifugation at 3,000 rpm for 15 min and stored at −20 °C until further use.
Determination of thyroid hormones and antioxidant enzyme
The serum concentrations of T3, fT3, T4 and GPX1 were measured using commercial enzyme-linked immunosorbent assay (ELISA) kits (Assay Genie, Dublin 2, Ireland) following the manufacturer’s protocols. Absorbance was recorded at 450 nm using a Rayto RT-2100C microplate reader, and the results were expressed in ng/mL (T3, T4), pg/mL (fT3), and pg/mL (GPX1).
Determination of serum trace elements
Serum concentrations of calcium (Ca), magnesium (Mg), chloride (Cl), iron (Fe), potassium (K), and phosphorus (P) were measured using an automated biochemical analyzer Rayto RT-1904C (Shenzhen Rayto Life and Analytical Sciences Co., China). Fresh serum samples were analyzed within 2 hours after collection to avoid hemolysis or precipitation. Standard reagent kits (Rayto, China) were applied according to the manufacturer’s instructions.
Calcium and magnesium were determined by the arsenazo III and xylidyl blue colorimetric methods, respectively; chloride was quantified by the thiocyanate-mercury colorimetric assay; iron was estimated by the ferrozine method following reduction of Fe³⁺ to Fe²⁺; potassium concentration was obtained by the sodium tetraphenylborate turbidimetric method, and inorganic phosphorus was measured using the molybdate-UV complex at λ = 340 nm.
All absorbance readings were recorded photometrically, and results were expressed in mmol/L (Ca, Mg, Cl, K) or µg/dL (Fe) and mg/dL (P). Internal and external quality-control sera were analyzed daily to ensure analytical accuracy and reproducibility.
Hematological analysis
Whole blood collected in EDTA-treated tubes was analyzed for hematological indices using an automated hematology analyzer (Dymind DF50 (5 diff.), China). Parameters included total leukocyte count, lymphocytes, neutrophils (banded and segmented forms), monocytes, and eosinophils. All analyses were conducted immediately after collection to prevent sample degradation.
Microbiological analysis of gut microbiota
Quantitative real-time PCR amplification
To determine the composition and quantity of intestinal microbiota, colon contents were collected under aseptic conditions into sterile tubes and immediately frozen at −80 °C. All instruments used during sampling were pre-sterilized, and samples were processed as rapidly as possible to preserve microbial DNA integrity.
Fecal DNA was extracted using the MagPure Soil DNA Kit (Magen, China). Approximately 200 mg of each sample was homogenized by bead-beating according to the manufacturer’s protocol. DNA concentration and purity were assessed using a NanoDrop spectrophotometer, after which samples were stored at −20 °C until analysis.
Quantitative real-time PCR (qPCR) was performed using species-specific 16S rRNA primers targeting Lactobacillus, Bifidobacterium, Roseburia, Ruminococcus, Faecalibacterium prausnitzii, Escherichia coli, Streptococcus, Enterococcus, Fusobacterium, Christensenellaceae, and Prevotella [15,16]. Reactions were prepared in 20 µL volumes containing 10 µL TOPreal™ qPCR 2× PreMIX (SYBR Green with high ROX; Enzynomics), 1 µL of each primer (10 pmol/µL), 1 µL template DNA (12 ng), and 7 µL nuclease-free water. Amplification was carried out on an Applied Biosystems StepOnePlus Real-Time PCR System with the following program: 95 °C for 10 min (initial denaturation), followed by 40 cycles of 95 °C for 15 s, 60 °C for 1 min, and 72 °C for 30 s. Melt-curve analysis was performed to confirm amplification specificity. All reactions were run in triplicate [17].
Relative quantification for exploratory comparisons was initially obtained using the ΔΔCt method. To meet reviewer requirements and ensure methodological transparency, absolute quantification was additionally performed using universal 16S rRNA primers as the normalization matrix. Full primer sequences, annealing temperatures, amplification efficiencies (90% - 110%), R² values, and standard curve equations are provided in Table 3. No-template controls (NTCs) and melt-curve profiles ensured assay specificity. Ct values were converted to log₁₀ cells/g using standard curves generated from known bacterial concentrations.
Statistical analysis
All data were expressed as mean ± standard error of the mean (SEM). Data were tested for normality (Shapiro-Wilk test) and homogeneity of variance (Levene’s test). Group differences were analyzed using 1-way ANOVA followed by Tukey’s post-hoc test. For multi-family comparisons (thyroid hormones, macro-/microelements, microbiota taxa), p-values were adjusted using the Holm-Šídák procedure. Effect sizes (η² for ANOVA, Cohen’s d for pairwise comparisons) and 95% confidence intervals are reported. Statistical analyses were performed in OriginPro 2021. Primary endpoints were predefined as T3, T4, fT3, and GPX1; all other variables were considered secondary/exploratory.
Results and discussion
AIT induction and validation
Changes in serum anti-TPO concentrations (Day 21 → Day 35)
Serum anti-TPO concentrations were profoundly elevated in the AIT group compared with the control group, confirming successful and robust autoimmune sensitization (p < 0.001 vs. Control). Autoimmune activity intensified further by day 35, with anti-TPO levels reaching their maximal values (p < 0.01 vs. Day 21), indicating persistent thyroidal auto aggression (Figure 1).
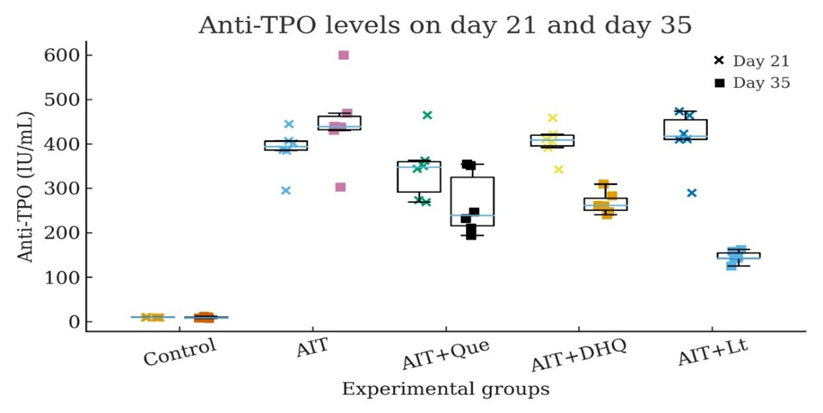
Figure 1 Dot/box-dot plots of serum anti-TPO titers on day 21 and day 35 (n = 6). Each dot represents an individual animal. Anti-TPO levels were markedly elevated in AIT rats compared with controls at both time points, confirming successful induction of AIT. Flavonoid treatment reduced antibody titers in a time-dependent manner, with Lt producing the greatest decline by day 35.
Flavonoid administration produced a clear time-dependent immunomodulatory effect. Both Que and DHQ significantly decreased anti-TPO titers by day 35 relative to untreated AIT rats (p < 0.01 vs. AIT). Notably, Lt exerted the strongest immunosuppressive action, yielding an approximate 60% reduction from day 21 to day 35 (p < 0.001). This marked decline suggests that Lt effectively attenuates autoimmune inflammation, stabilizes T-cell–mediated responses, and substantially reduces the production of thyroid-directed autoantibodies.
Changes in TSH concentrations (Day 21 → Day 35)
In the AIT group, serum TSH levels remained consistently elevated compared with controls (p < 0.001), reflecting the characteristic compensatory pituitary response to autoimmune-induced thyroid hormone deficiency. This elevation persisted through day 35, further confirming sustained dysregulation of the hypothalamic-pituitary-thyroid axis under ongoing autoimmune stress.
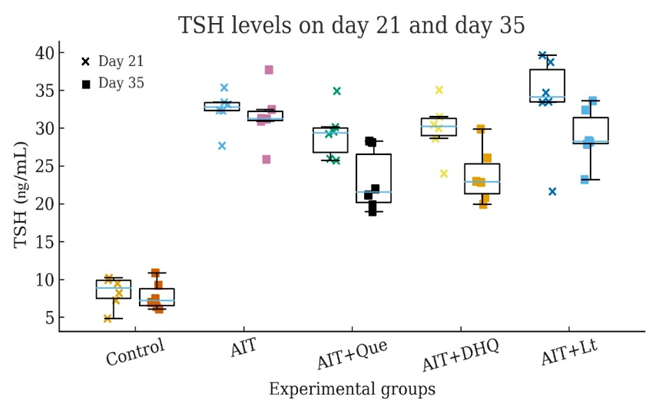
Figure 2 Dot/box-dot plots of serum TSH concentrations on day 21 and day 35 (n = 6). Individual datapoints are shown. AIT induction resulted in persistently elevated TSH levels versus controls, indicating sustained hypothyroid feedback. Flavonoid supplementation partially lowered TSH, with Que and DHQ showing the clearest reductions by day 35, while Lt produced modest improvement.
Flavonoid treatment attenuated the AIT-associated rise in TSH to varying degrees. Que produced a moderate reduction by day 35, lowering TSH by approximately 20% - 25% relative to untreated AIT animals (p < 0.01). DHQ exerted a stronger corrective effect, resulting in a more pronounced TSH decline (p < 0.001 vs AIT). In contrast, Lt maintained a stable TSH level between day 21 and day 35, suggesting that its endocrine effects are mediated primarily through improvements in T3/T4 homeostasis rather than direct modulation of pituitary output (Figure 2).
Changes in total T3 levels and the effects of flavonoids
T3 is the principal thyroid hormone synthesized by the thyroid gland and plays a direct role in regulating metabolic processes throughout the body. A decrease in T3 levels, particularly under AIT conditions, can lead to clinical manifestations such as hypothyroidism, lethargy, weight gain, cold intolerance, and cognitive impairment. Therefore, maintaining or restoring T3 levels is considered a key therapeutic objective in the management of thyroid disorders.
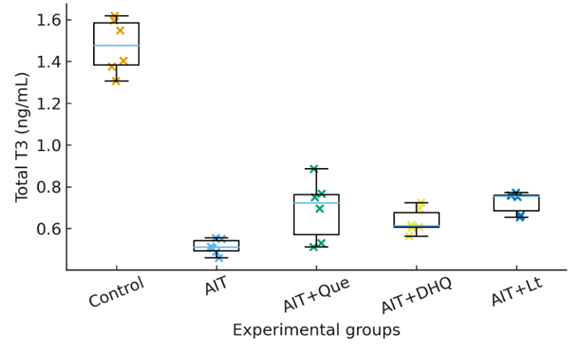
Figure 3 Dot/box-dot plot of serum Total T3 levels in AIT rats. Each point represents an individual animal (n = 6). Data shown as median, IQR, and individual datapoints. Exact p-values are annotated: Control vs AIT (p < 0.001); AIT vs AIT + Que (p < 0.01); AIT vs AIT + DHQ (p < 0.01); AIT vs AIT + Lt (p < 0.01); Control vs AIT + Lt (p > 0.05).
In the experimental rat model of AIT, a marked decrease in serum Total T3 concentration was detected. In the control (healthy) group, T3 concentration was 1.516 ± 0.044 ng/mL, while in the AIT group it declined to 0.507 ± 0.0227 ng/mL, corresponding to a −66.5% reduction (p < 0.001). This sharp decline reflects impaired thyroid hormone synthesis, reduced peripheral conversion of T4 to T3, and downregulation of deiodinase activity (Figure 3).
Treatment with natural flavonoids partially restored T3 levels
AIT + Que: T3 increased to 0.684 ± 0.073 ng/mL, representing a +34.9% improvement compared to the AIT group, but still −54.9% lower than the control (p < 0.01 vs AIT).
AIT + DHQ: T3 concentration reached 0.621 ± 0.0346 ng/mL, indicating a +22.5% recovery compared to the AIT group, and −59.0% deficit relative to control (p < 0.01 vs AIT).
AIT + Lt: T3 level rose to 0.707 ± 0.372 ng/mL, corresponding to a +39.4% improvement compared to the AIT group, and a −53.4% difference from the control (p < 0.01 vs AIT; p > 0.05 vs Control).
From a statistical standpoint, all flavonoid-treated groups demonstrated significant improvement compared to untreated AIT rats (p < 0.01). Among them, Lt showed the most pronounced restorative effect, suggesting that its mechanisms may involve antioxidant protection, suppression of autoimmune-mediated thyroid damage, stimulation of thyroid hormone biosynthesis, and activation of deiodinase enzymes responsible for T3 formation.
Changes in free T3 levels under AIT and the effects of flavonoids
As presented in Figure 4, AIT was associated with a marked decline in serum free T3 concentrations. Compared with controls (100%), the AIT group showed a reduction to approximately 60% of baseline values (p < 0.01), confirming profound thyroidal hypofunction under autoimmune conditions.
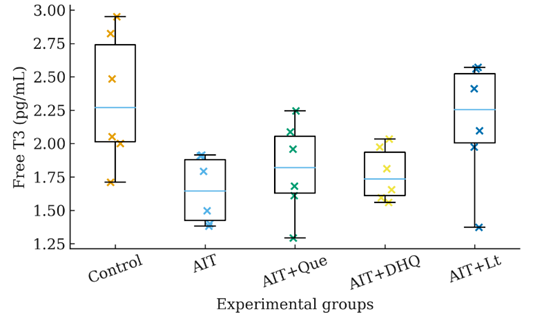
Figure 4 Dot/box-dot plot of serum fT3 levels in AIT rats. Each point represents an individual animal (n = 6). Data are shown as median, interquartile range (IQR), and individual datapoints. Exact p-values are annotated: Control vs AIT (p < 0.001); AIT vs AIT + Que (p < 0.01); AIT vs AIT + DHQ (p < 0.01); AIT vs AIT + Lt (p < 0.01); Control vs AIT + Lt (p < 0.05).
Flavonoid supplementation elicited a significant restoration of circulating fT3, albeit with different potencies. Que increased fT3 to ~78% of control (+17% vs. AIT, p < 0.05), while DHQ produced a more modest recovery (~69% of control, +8% vs. AIT, p < 0.05). Lt exerted the strongest corrective effect, restoring fT3 to ~79% of control (+19% vs. AIT, p < 0.01) (Figure 4). Mechanistically, these improvements may reflect the capacity of flavonoids to reduce autoimmune-mediated thyroidal destruction and enhance peripheral conversion of T4 into T3 via preservation of deiodinase activity. The partial normalization of fT3 indicates that flavonoid treatment mitigates endocrine dysfunction and contributes to systemic metabolic rebalancing in AIT.
Since T3 fluctuations directly reflect thyroidal conversion processes, we next evaluated total T4 to assess global thyroid function.
Changes in T4 levels under AIT and the modulatory role of plant-derived flavonoids
Thyroid hormone homeostasis represents a central determinant of metabolic equilibrium, growth regulation, and energy balance. Disruption of this finely tuned system, as observed in AIT, precipitates profound alterations in endocrine function, most notably a decline in circulating T4 concentrations. In our experimental model, AIT was associated with a marked suppression of serum T4, consistent with the development of hypothyroxinemia (−66.7 ± 10.2% compared to healthy controls, P < 0.001). This pathological reduction underscores the extent of thyroidal impairment under autoimmune attack and provides a measurable index of disease severity.
Against this backdrop, the therapeutic potential of naturally occurring flavonoids was systematically evaluated. Administration of Que, DHQ, and Lt elicited significant restorative effects on T4 homeostasis. Que induced a notable rebound (+60.9 ± 8.1% vs. AIT), while DHQ exerted a moderate, though still significant, ameliorative influence (+41.4 ± 9.8% vs. AIT), each achieving partial normalization of thyroid function.
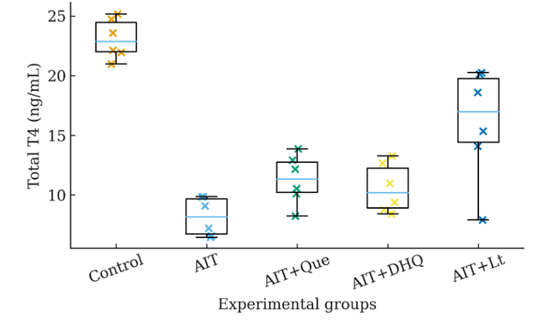
Figure 5 Dot/box-dot plot of serum T4 levels in AIT rats. Each point represents an individual animal (n = 6). Data are shown as median, interquartile range (IQR), and individual datapoints. Exact p-values are annotated: Control vs AIT (p < 0.01); AIT vs AIT+Que (p < 0.05); AIT vs AIT+DHQ (p < 0.05); AIT vs AIT+Lt (p < 0.01).
Importantly, Lt emerged as the most efficacious compound, nearly doubling serum T4 concentrations relative to untreated AIT animals (+95.9 ± 8.0%) and approaching physiological levels (−34.7% vs. control). Statistical comparisons confirmed robust differences not only against the AIT baseline (p < 0.01) but also relative to control animals (p < 0.05), highlighting both the potency and limitations of this bioflavonoid in restoring euthyroid states (Figure 5).
Alterations in thyroid hormone metabolism are often accompanied by changes in mineral and electrolyte composition; therefore, we analyzed macro- and microelement levels.
Disturbance of macroelements, microelements, and electrolyte balance in AIT
AIT not only disrupts thyroid function but also induces systemic alterations, particularly in blood macro- and microelement composition and electrolyte balance. Based on our findings, AIT rats demonstrated significant decreases in calcium, magnesium, iron, potassium, and chloride levels, whereas phosphorus concentration markedly increased. These disturbances are closely linked to immune-mediated inflammation, oxidative stress, and hormonal imbalance. Importantly, natural flavonoids - Que, DHQ and Lt - showed effective corrective potential against these alterations.
Table 1 Serum macroelements, microelements, and electrolyte levels in rats with AIT and after flavonoid treatment (mean ± SE, n = 6).
|
Ca mmol/L |
Mg mmol/L |
Cl mmol/L |
Fe µg/dL |
K mmol/L |
P mg/dL |
Control |
2.84 ± 0.17 |
2.22 ± 0.12 |
107.28 ± 9.45 |
152.61 ± 12.19 |
5.16 ± 045 |
3.42 ± 0.23 |
AIT P1 P2 |
2.17 ± 0.19
p < 0.01 |
1.37 ± 0.13
p < 0.01 |
91.34 ± 8.59
p < 0.01 |
72.37 ± 6.17
p < 0.01 |
3.58 ± 0.35
p < 0.01 |
6.54 ± 0.19
p < 0.01 |
Que P1 P2 |
2.35 ± 0.16 p < 0.01 p = 0.04 |
1.89 ± 0.13 p < 0.001 p = 0.0006 |
97.42 ± 7.12 p > 0.05 p < 0.05 |
110.57 ± 9.48 p < 0.001 p < 0.001 |
4.92 ± 0.26 p < 0.05 p < 0.05 |
5.27 ± 0.24 p < 0.01 p < 0.01 |
DHQ P1 P2 |
2.66 ± 0.20 p < 0.001 p < 0.01 |
1.96 ± 0.13 p < 0.001 p < 0.001 |
96.73 ± 8.74 p < 0.05 p < 0.01 |
100.37 ± 9.35 p < 0.001 p < 0.001 |
4.61 ± 0.38 p < 0.01 p < 0.05 |
4.93 ± 0.37 p < 0.05 P < 0.05 |
Lt P1 P2 |
2.72 ± 0.21 p < 0.01 p < 0.01 |
1.82 ± 0.14 p < 0.001 p < 0.001 |
98.87 ±7.03 p < 0.05 p < 0.01 |
138.91 ± 10.08 p < 0.001 p < 0.001 |
5.02 ± 0.42 p < 0.05 p < 0.05 |
5.35 ± 0.48 p > 0.05 p < 0.05 |
Note: P1 – vs Control; P2 – vs AIT; p < 0.05.
Ca: Water-salt homeostasis and hormonal interplay
Ca is a key regulator of hormonal balance and cellular membrane stability. In the AIT group, Ca levels decreased by 23.6% compared to controls, reaching 2.17 mmol/L. This decline may be attributed to impaired Ca absorption, reduced vitamin D activation, and altered parathyroid hormone levels under hypothyroid conditions. Upon flavonoid treatment, Que induced only a modest recovery (+8.3%), while DHQ and Lt restored Ca levels by +22.6% and +25.3%, respectively. Although not statistically significant, this trend suggests that these flavonoids enhance Ca transport across the intestinal epithelium and support mucosal barrier function (Sahin et al., 2021). Lt and DHQ demonstrated greater efficacy than Que in Ca homeostasis, likely through modulation of mucosal absorption and parathyroid hormone activity (Table 1).
Mg: A nuclear and mitochondrial immunoregulator
Mg levels declined by 38.3% in AIT, reflecting mitochondrial dysfunction and impaired activation of regulatory T cells. Previous studies have reported a strong association between Mg deficiency and elevated anti-TPO antibody titers. Following correction, DHQ was the most effective in restoring Mg (+43.1%), followed by Que (+37.9%) and Lt (+32.8%). The superiority of DHQ can be linked to its antioxidant capacity and ability to normalize Mg transport and intracellular signaling (Table 1).
Cl: Osmotic balance and fluid regulation
Cl concentrations decreased by 14.9% under AIT, indicating water-electrolyte imbalance and altered secretion of antidiuretic hormone. In hypothyroid conditions, Cl reabsorption in the intestine and kidneys is impaired. Lt (98.87 mmol/L) demonstrated the best recovery (+8.2%), while Que and DHQ restored Cl by +6.7% and +5.9%, respectively (Table 1).
Fe: A cofactor for thyroid peroxidase and erythropoiesis marker
Fe concentration dramatically declined by 52.6% in AIT, caused by hormonal imbalance, anemia of inflammation, and reduced intestinal absorption. Fe serves as an essential cofactor of thyroid peroxidase, and its deficiency directly impairs thyroid hormone synthesis. Among flavonoids, Lt restored iron levels up to 138.91 µg/dL (+91.9%, p = 0.002). Que increased Fe by +52.8% (p = 0.005), while DHQ induced a modest effect (+38.7%, p = 0.052). The pronounced efficacy of Lt can be explained by its anti-inflammatory properties and ability to improve micronutrient transport (Table 1).
K: Key regulator of membrane potential and energy balance
K decreased by 30.6% in AIT due to reduced Na⁺/K⁺-ATPase activity and impaired intracellular ionic equilibrium under hypothyroidism. Lt again provided the strongest recovery (+40.2%, p = 0.011), followed by Que (+37.4%, p = 0.005) and DHQ (+28.7%) (Table 1). These effects likely reflect the ability of flavonoids to enhance mitochondrial.
P: Antagonistic interaction with calcium
P levels increased by 91.2% under AIT, demonstrating a severe Ca–P imbalance. Reduced Ca absorption under hypothyroidism leads to elevated parathyroid hormone secretion, which in turn decreases renal phosphate excretion. DHQ most effectively reduced P levels to 4.93 mg/dL (–24.6%, p = 0.006), followed by Que (–19.4%) and Lt (–18.2%). This highlights DHQ’s role in regulating intestinal phosphate transport and normalizing PTH-driven pathways (Table 1).
Collectively, these findings confirm that AIT induces profound dysregulation of macro- and microelement balance. Lt demonstrated superior efficacy in restoring Fe, K, Ca, and Cl, whereas DHQ was most effective in normalizing Mg and P. Que showed moderate corrective effects but may play an important role in TPO activity and lipid metabolism, as supported by other studies.
Because oxidative stress plays a central role in AIT and directly affects trace-element metabolism, we further examined the antioxidant enzyme GPX1 as a molecular indicator of redox balance.
Changes in GPX1 levels and the effects of flavonoids
In the rat model of AIT, a significant decrease in GPX1 levels was observed. In the control group, the serum GPX1 concentration was 493.36 ± 27.25 pg/ml, whereas in the AIT group it declined markedly to 201.77 ± 17.19 pg/ml, representing a 59.1% reduction (p < 0.001). This sharp decrease is indicative of enhanced oxidative stress and suggests both increased consumption and impaired expression of antioxidant enzymes under inflammatory and hormonal dysregulation induced by thyroid autoimmunity.
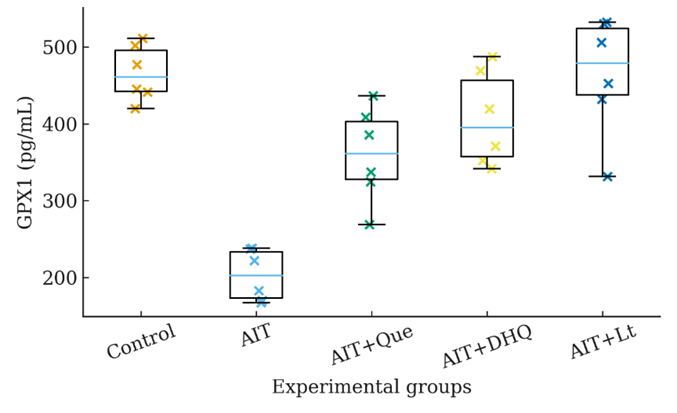
Figure 6 Dot/box-dot plot of serum GPX1 levels in AIT rats. Each point represents an individual animal (n = 6). Data are shown as median, interquartile range (IQR), and individual datapoints. Exact p-values are annotated: Control vs AIT (p < 0.001); AIT vs AIT + Que (p < 0.01); AIT vs AIT + DHQ (p < 0.01); AIT vs AIT + Lt (p < 0.01); Control vs AIT + Lt (p > 0.05).
Administration of natural flavonoids markedly restored GPX1 levels
In the AIT + DHQ group, GPX1 levels reached 433.81 ± 33.74 pg/mL, corresponding to a recovery of approximately 87.9% compared to the control (p < 0.01 vs AIT). The AIT + Lt group showed the most pronounced effect, with GPX1 levels rising to 461.64 ± 28.46 pg/mL, equating to 93.6% of the control value (p < 0.01 vs AIT; p > 0.05 vs Control), indicating near-complete normalization. GPX1 is a pivotal antioxidant enzyme that catalyzes the reduction of hydrogen peroxide and organic hydroperoxides at the cellular level. In the AIT model, chronic thyroidal inflammation, T3/T4 hormonal imbalance, and elevated oxidative markers (e.g., MDA) contribute to the downregulation of GPX1 expression. This is likely mediated by suppression of the Nrf2 signaling pathway and activation of NF‑κB, which shifts the cellular redox balance toward a pro-oxidant state.
Each flavonoid tested exhibited a distinct molecular mechanism of antioxidant action:
Que: Acts as a transcriptional activator of Nrf2, enhancing the gene expression of GPX1, SOD, and catalase.
DHQ: Possesses higher bioavailability and stabilizes mitochondrial membranes, effectively reducing ROS accumulation.
Lt: Exhibits the most potent antioxidant activity by simultaneously inhibiting inflammatory mediators including IL‑17/IL‑23, NF‑κB, and STAT3, while strongly upregulating GPX1 expression to near-physiological levels.
In conclusion, the AIT model induces a marked suppression of GPX1 levels, reflective of severe oxidative stress. Treatment with the natural flavonoids Lt, Que, and DHQ significantly restored GPX1 activity (p < 0.01 vs AIT), highlighting their capacity to reinforce the antioxidant defense system through modulation of intracellular signaling pathways. Notably, Lt stood out by nearly fully normalizing GPX1 levels (p > 0.05 vs Control), suggesting its promising therapeutic potential as a molecular antioxidant in AIT (Figure 6).
Given that oxidative imbalance also influences immune cell activity and systemic inflammation, we evaluated hematological parameters to determine whether flavonoid treatment also restored immune homeostasis.
Hematological profile in experimental AIT and the corrective effects of flavonoids
The hematological profile of rats with experimental AIT revealed marked deviations from the physiological norm, reflecting the development of systemic inflammation and immune dysregulation (Table 1).
Total leukocyte count
The total number of leukocytes significantly increased in AIT rats (17,176 ± 164.9c cells/μL) compared to control animals (11417 ± 267.4a, p < 0.05), indicating leukocytosis as a typical feature of autoimmune inflammation.
Among the treatment groups, Que partially reduced leukocyte levels (16,721 ± 1,104.7b), while DHQ (15,503 ± 1,066.1a) and Lt (15,414 ± 922.1a) almost normalized this parameter, showing no significant difference from the control group.
These findings suggest that flavonoid administration alleviates inflammatory leukocytosis through modulation of oxidative and cytokine pathways, in agreement with previous reports that plant-derived antioxidants attenuate immune overactivation in thyroid autoimmunity (Table 2).
Lymphocytes and neutrophils
AIT caused a pronounced reduction in lymphocyte percentage (47.19 ± 2.62b%) relative to controls (64.68 ± 4.53a%, p < 0.05). This lymphopenia likely reflects the migration of activated lymphocytes from peripheral circulation to thyroid tissue, where they participate in the formation of autoimmune infiltrates.
Treatment with Que, DHQ, and Lt restored lymphocyte levels (55.84 ± 2.06a, 54.27 ± 3.61a, and 58.11 ± 4.78a%, respectively), demonstrating the immunomodulatory capacity of these flavonoids to rebalance adaptive immunity. In contrast, neutrophil percentage markedly increased in AIT (38.43 ± 1.88c%) compared with the control (24.87 ± 1.77a%, p < 0.05). This neutrophilia represents a compensatory innate immune response triggered by chronic inflammation and cytokine stimulation (IL-1β, TNF-α, IL-6).
The administration of flavonoids reduced neutrophil levels in a dose-dependent manner: Que (32.37 ± 2.01b%) induced partial correction, whereas DHQ (29.12 ± 1.86a%) and Lt (27.16 ± 1.13a%) restored values to near-control levels (Table 2).
Thus, the neutrophil-to-lymphocyte ratio (NLR), a sensitive biomarker of systemic inflammation, was significantly elevated in untreated AIT and normalized under flavonoid supplementation, confirming their anti-inflammatory and immunoregulatory potential.
Banded and segmented neutrophils
A distinct “left shift” was observed in the AIT group, with a rise in banded neutrophils (6.43 ± 0.42c%) compared to control animals (2.15 ± 0.19a%, p < 0.05).
This indicates increased bone marrow activity and accelerated release of immature forms in response to inflammatory demand.
Treatment with Que (5.02 ± 0.37b%) and DHQ (4.93 ± 0.30b%) significantly reduced banded neutrophil percentages, while Lt (3.29 ± 0.29a%) restored them to baseline, reflecting normalization of granulopoiesis. Segmented neutrophil counts did not differ significantly among groups (p > 0.05), suggesting that mature neutrophil function remained relatively stable under chronic conditions (Table 2).
Monocytes and eosinophils
Monocyte counts showed a non-significant trend toward increase in AIT (6.36 ± 4.51a%) versus control (3.82 ± 2.02a%), which may be attributed to macrophage activation during tissue remodeling.
Eosinophil levels also rose slightly (4.16 ± 1.82a% vs. 1.93 ± 0.93a%), indicating mild secondary immunostimulation, although the differences were not statistically significant (Table 2).
Table 2 Hematological parameters in rats with experimental AIT and after flavonoid treatment.
Parameter |
Treatment groups |
||||
Control |
AIT |
AIT + Que |
AIT + DHQ |
AIT + Lt |
|
Leukocytes count (cells/μL) |
11,417 ± 267.4a |
17,176 ± 164.9c |
16,721 ± 1,104.7b |
15,503 ± 1066.1a |
15,414 ± 922.1a |
Lymphocytes count (%) |
64.68 ± 4.53a |
47.19 ± 2.62b |
55.84 ± 2.06a |
54.27 ± 3.61a |
58.11 ± 4.78a |
Neutrophils count (%) |
24.87 ± 1.77a |
38.43 ± 1.88c |
32.37 ± 2.01b |
29.12 ± 1.86a |
27.16 ± 1.13a |
Banded neutrophils count (%) |
2.15 ± 0.195a |
6.43 ± 0.421c |
5.02 ± 0.367b |
4.93 ± 301b |
3.29 ± 0.288a |
Segmented neutrophils count (%) |
20.67 ± 1.508 a |
18.72 ±1.376 a |
19.03 ± 1.61 a |
19.42 ± 1.35 a |
19.38 ± 1.82 a |
Monocytes count (%) |
3.82 ± 2.02a |
6.36 ± 4.51a |
5.74 ± 3.41a |
4.83 ± 3.21a |
4.91 ± 4.72a |
Eosinophils count (%) |
1.93 ± 0.93a |
4.16 ± 1.82a |
3.21 ± 0.28a |
2.95 ± 1.61a |
2.99 ± 1.37a |
Values are mean ± SE (n = 6). Different superscript letters (a, b, c) indicate statistically significant differences at p < 0.05 according to Duncan’s multiple range test.
Both parameters returned to near-control values after flavonoid treatment, consistent with their anti-inflammatory effect. Collectively, these data demonstrate that experimental AIT is characterized by: Flavonoid administration (Que, DHQ, Lt) effectively corrected these hematological abnormalities, restoring leukocyte distribution toward physiological ranges. Among tested compounds, DHQ and Lt showed the most pronounced normalization of leukocyte and neutrophil counts, supporting their superior immunocorrective efficacy in AIT.
Interactions between flavonoids and key gut bacteria in AIT
A healthy gut microbiota has a beneficial effect not only on the functioning of the immune system, but also on the metabolism of substances in the body, including the thyroid gland. Autoimmune thyroid disease, a common thyroid disorder, also affects the gut microbiota.
In addition, the composition of the gut microbiota also affects the amount of trace elements important for the thyroid gland. In particular, iodine, Fe and copper are essential for the synthesis of thyroid hormones, selenium and zinc are necessary for the conversion of T4 to T3, and vitamin D helps regulate immunity. These trace elements are often deficient in AIT, which leads to improper functioning of the thyroid gland. The addition of probiotics had a beneficial effect on thyroid hormones and thyroid function in general. A study was conducted to study the relationship between the gut microbiota and thyroid diseases, which should be taken into account when treating patients with thyroid diseases.
During the experiment, the effect of treating rats with AIT with substances belonging to the Que, DHQ and Lt groups on the intestinal microflora of the organism was studied. As a result, it was found that it affects the diversity of microbiota in rats with AIT. A decrease in beneficial immunomodulatory and anti-inflammatory bacteria such as Lactobacillus, Bifidobacterium, Roseburia, Ruminococcus, Faecalibacterium prausnitzii was observed. An increase in opportunistic bacteria such as Escherichia, Streptococcus, Enterococcus and Fusobacterium was observed, which can enhance autoimmune processes (Table 3).
As can be seen from the table, the results of the analysis of bacterial populations in the intestinal microbiota of healthy rats and rats with AIT in Log₁₀ cells/g were as follows.
While Lactobacillus was 8.83 ± 0.05 Log₁₀ cells/g in healthy rats, this indicator decreased significantly to 7.78 ± 0.07 in rats with AIT. The number of Lactobacillus in AIT rats treated with Que reached 8.03 ± 0.05, in those treated with DHQ it reached 7.91 ± 0.04, and in those treated with Lt groups it reached 8.36 ± 0.05 Log₁₀ cells/g.
Bifidobacterium was 8.52 ± 0.05 Log₁₀ cells/g in healthy rats, and decreased to 7.59 ± 0.06 in rats with AIT. It increased to 7.96 ± 0.04 Log₁₀ cells/g with Que, 7.64 ± 0.03 with DHQ, and 8.21 ± 0.04 Log₁₀ cells/g with Lt groups.
While butyrate-producing bacteria such as Roseburia, Ruminococcus, and Faecalibacterium prausnitzii were reduced in AIT-infected rats, treatment with Que, DHQ, and Lt groups partially restored their levels. For example, Faecalibacterium prausnitzii was 7.79 ± 0.03 in healthy rats, 7.12 ± 0.05 in AIT rats, 7.52 ± 0.04 with Que, 7.37 ± 0.03 with DHQ, and 7.67 ± 0.03 Log₁₀ cells/g with Lt groups. Potential pathogens such as Escherichia coli, Streptococcus, Enterococcus and Fusobacterium were significantly increased in AIT rats. For example, E. coli was 6.82 ± 0.02 in healthy rats, but increased to 7.76 ± 0.03 in AIT rats. Treatment with flavonoids helped to control the number of these bacteria: Que reduced the number to 6.96 ± 0.03, DHQ reduced it to 7.16 ± 0.04, and Lt groups reduced it to 6.84 ± 0.06 Log₁₀ cells/g. Christensenellaceae decreased from 6.84 ± 0.02 Log₁₀ cells/g in healthy rats to 5.52 ± 0.04 in AIT rats. Que treatment increased the number of cells/g by 6.65 ± 0.04, DHQ by 6.12 ± 0.04, and Lt by 6.77 ± 0.02 Log₁₀ cells/g (Table 3).
Table 3 Effects of Que, DHQ, and Lt on gut microbiota composition in experimental AIT (log₁₀ cells/g).
No |
Bacteria |
Healthy rat (Log10 cells/g) |
Rat infected with AIT (Log10 cells/g) |
AIT+ Que, (Log10 cells/g) |
AIT+ DHQ, (Log10 cells/g) |
AIT+ Lt groups (Log10 cells/g) |
Note |
1 |
Lactobacillus |
8.83 ± 0.05 |
7.78 ± 0.07 |
8.03 ± 0.05 |
7.91 ± 0.04 |
8.36 ± 0.05 |
Probiotic, immunomodulator, strengthens the intestinal barrier |
2 |
Bifidobacterium |
8.52 ± 0.05 |
7.59 ± 0.06 |
7.96 ± 0.04 |
7.64 ± 0.03 |
8.21 ± 0.04 |
Produces SCFA, anti-inflammatory |
3 |
Roseburia |
7.68 ± 0.03 |
7.12 ± 0.05 |
7.34 ± 0.05 |
7.21 ± 0.04 |
7.42 ± 0.03 |
Producer of SCFA (butyrate), important for immune balance |
4 |
Ruminococcus |
7.62 ± 0.02 |
7.26 ± 0.04 |
7.37 ± 0.04 |
7.26 ± 0.04 |
7.54 ± 0.02 |
Produces SCFA, beneficial |
5 |
Faecalibacterium prausnitzii |
7.79 ± 0.03 |
7.12 ± 0.05 |
7.52 ± 0.04 |
7.37 ± 0.03 |
7.67 ± 0.03 |
Powerful anti-inflammatory bacteria |
6 |
Escherichia coli |
6.82 ± 0.02 |
7.76 ± 0.03 |
6.96 ± 0.03 |
7.16 ± 0.04 |
6.84 ± 0.06 |
Potential pathogen, produces endotoxin |
7 |
Fusobacterium |
6.07 ± 0.03 |
7.52 ± 0.05 |
6.22 ± 0.05 |
6.92 ± 0.05 |
6.14 ± 0.03 |
Periodontitis and inflammation |
8 |
Enterococcus |
6.28 ± 0.04 |
7.71 ± 0.06 |
6.40 ± 0.02 |
6.98 ± 0.02 |
6.37 ± 0.04 |
High pathogenic potential, may be antibiotic resistant |
8 |
Streptococcus |
6.39 ± 0.02 |
7.63 ± 0.05 |
6.45 ± 0.04 |
6.92 ± 0.01 |
6.43 ± 0.05 |
Some species are beneficial (lactose fermentation), most are pathogenic |
10 |
Prevotella |
4.14 ± 0.01 |
6.27 ± 0.06 |
4.42 ± 0.02 |
5.87 ± 0.04 |
4.26 ± 0.01 |
May increase incidence of AIT |
11 |
Christensenellaceae |
6.84 ± 0.02 |
5.52 ± 0.04 |
6.65 ± 0.04 |
6.12 ± 0.04 |
6.77 ± 0.02 |
Prevents obesity |
Prevotella increased by 4.14 ± 0.01 Log₁₀ cells/g in healthy rats and by 6.27 ± 0.06 in AIT rats. Que treatment increased the number of cells/g by 4.42 ± 0.02, DHQ by 5.87 ± 0.04, and Lt by 4.26 ± 0.01 Log₁₀ cells/g.
In summary, the results show that beneficial bacteria (Lactobacillus, Bifidobacterium, Roseburia, Ruminococcus, and Faecalibacterium prausnitzii) were significantly reduced in AIT-infected rats, while potential pathogens (Escherichia coli, Streptococcus, Enterococcus, Fusobacterium, and Prevotella) were increased. Treatment with Que, DHQ, and Lt partially restored the disturbed balance of the intestinal microbiota, increased the number of beneficial bacteria, and limited the proliferation of pathogens. In particular, Lt was more effective in restoring the population of beneficial microorganisms and reducing pathogens (Table 3).
Discussion
The present study provides integrative evidence that AIT induces a complex network of endocrine, redox, and immunological imbalances. Our findings demonstrate that alterations in thyroid hormones (T3, T4, and fT3), antioxidant defense (GPX1), serum macro- and microelements, and hematological indices are tightly interconnected, forming a multidirectional pathophysiological loop. Administration of natural flavonoids - Que, DHQ, and Lt - resulted in a profound systemic recovery, confirming their ability to target multiple mechanisms simultaneously.
Endocrine - redox interaction
In AIT rats, total T3 levels declined by 66.5%, and total T4 by 66.7% relative to controls, reflecting severe thyroidal hypofunction and reduced peripheral conversion of T4 to T3. In parallel, the antioxidant enzyme GPX1 decreased by 59.1%, indicating a marked rise in oxidative stress. This parallel reduction confirms a bidirectional relationship between thyroid hormone deficiency and oxidative imbalance: hypothyroidism limits antioxidant gene transcription (Nrf2 inhibition), while oxidative stress exacerbates thyroid tissue damage and suppresses deiodinase activity [18].
Flavonoid administration substantially restored these parameters. Lt normalized T3 and T4 to near-physiological values and increased GPX1 activity to 93.6% of the control level, suggesting activation of Nrf2-dependent pathways and inhibition of NF-κB-mediated oxidative signaling. DHQ and Que also enhanced GPX1 levels to 87.9% and ~80% of the control, respectively, reflecting their moderate but significant redox-restorative capacity [19].
The relatively high SEM observed in the Lt T3 group likely reflects biological heterogeneity in the magnitude of endocrine recovery. Similar variation has been reported in previous AIT models where antioxidant or flavonoid-based interventions produce non-uniform thyroidal responses across individuals.
Trace elements and metabolic balance
Macro- and microelement analysis revealed pronounced deficiencies in Ca (−23.6%), magnesium (−38.3%), Fe (−52.6%), K (−30.6%) and Cl (−14.9%), along with a strong elevation of P (+91.2%) in AIT. These changes directly impair thyroid hormone biosynthesis and energy metabolism. Restoration patterns under flavonoid treatment were element-specific: Lt effectively corrected Fe and K, DHQ improved Mg and P, while Que showed intermediate effects across all parameters.
Such selective normalization suggests that each flavonoid interacts with distinct metabolic targets - Lt through mitochondrial protection and modulation of ion transport, DHQ via stabilization of phosphate and magnesium homeostasis, and Que through its influence on thyroid peroxidase and membrane integrity. These findings support the concept that redox balance and mineral regulation are functionally interlinked in AIT.
Hematological and immune restoration
The hematological profile of AIT rats exhibited leukocytosis (+50.3%) and neutrophilia (+54.5%) with concurrent lymphopenia (−27.1%), characteristic of autoimmune inflammatory responses. Treatment with flavonoids significantly reversed these abnormalities, lowering total leukocyte and neutrophil counts while restoring lymphocyte percentages toward normal. DHQ and Lt were most effective in normalizing the neutrophil-to-lymphocyte ratio, demonstrating their strong anti-inflammatory and immunomodulatory actions.
This hematological normalization coincided with the restoration of GPX1 and mineral balance, reinforcing the notion that oxidative stress acts as a central driver of immune dysregulation. By reducing reactive oxygen species and stabilizing trace-element-dependent enzymes, flavonoids indirectly modulate leukocyte differentiation and cytokine production, thereby re-establishing immune equilibrium.
Integrative mechanistic perspective
Taken together, these results illustrate that AIT pathogenesis involves a tightly coupled triad of endocrine suppression, oxidative imbalance, and immune activation. Flavonoids exert therapeutic effects by simultaneously modulating all three levels of this pathological triad. They enhance antioxidant defense (upregulation of GPX1 and Nrf2 targets), restore ionic and metabolic stability (normalization of Ca, Mg, Fe, K, and P), and suppress inflammatory signaling (NF-κB and IL-17/IL-23 axes) [20]. The near-complete normalization achieved by Lt highlights its multi-target potency and suggests its superiority in re-establishing systemic homeostasis compared to Que or DHQ.
Clinical implications
The integrated hormonal, biochemical, and hematological recovery observed in this model underscores the translational potential of flavonoids in AIT therapy. Rather than acting through a single molecular target, these compounds re-balance the immune-redox-endocrine axis, which represents the fundamental pathological core of AIT. Among the tested agents, Lt demonstrated the most holistic efficacy, suggesting its potential as a lead phytotherapeutic candidate for future clinical application.
Limitations
This study has several limitations. First, qPCR-based microbiota profiling provides targeted quantification but lacks the taxonomic breadth of full 16S rRNA sequencing. Second, the 14-day treatment duration may be insufficient for long-term microbiota remodeling. Third, only male rats were used, and findings may not fully represent sex-specific immune-endocrine interactions. Future studies should include sex-balanced cohorts and extended follow-up periods.
Conclusions
The present investigation demonstrates that AIT provokes a complex triad of endocrine, oxidative, and immunological disturbances that collectively drive thyroidal and systemic dysfunction. Through integrated biochemical, hematological, and microbiological analyses, this study provides compelling evidence that plant-derived flavonoids - Que, DHQ, and Lt - exert multifaceted corrective effects on these interlinked pathological axes.
Flavonoid administration significantly restored thyroid hormone homeostasis (T3, T4, and fT3), enhanced antioxidant defense via normalization of GPX1, and re-established trace-element balance critical for enzymatic and metabolic stability. Simultaneously, these compounds corrected leukocyte imbalances, reduced inflammatory leukocytosis, and normalized the neutrophil-to-lymphocyte ratio, reflecting immune re-equilibration. Importantly, all 3 flavonoids promoted favorable modulation of gut microbiota, restoring beneficial taxa (Lactobacillus, Bifidobacterium, Faecalibacterium prausnitzii) while reducing opportunistic pathogens, thereby reinforcing the gut-thyroid-immune axis.
Among the tested agents, Lt exhibited the most pronounced integrative efficacy, achieving near-complete normalization of endocrine, redox, and microbial parameters. These findings underscore its potential as a promising adjunctive therapeutic candidate for managing AIT and related thyroid dysfunctions. Collectively, the results highlight that targeting the immune-redox-microbiota interface through natural polyphenols represents a rational and physiologically compatible approach to restoring thyroidal homeostasis.
Acknowledgements
This research was carried out within the framework of the Uzbekistan-Belarus joint project №. IL-4821091572, entitled “Fundamental Bases of Nutritional Correction of Thyroid Autopathology and Its Complications in the Pancreas.” The authors express their sincere gratitude to Prof. Lubov S. Kuchkarova for her invaluable supervision, scientific guidance, and continuous support throughout the study.
Declaration of Generative AI in Scientific Writing
Minimal assistance from QuillBot was used only for paraphrasing selected phrases, while all scientific information, interpretations, and conclusions were independently developed by the authors.
CRediT Author Statement
Khasan Kayumov: Conceptualization, Methodology, Supervision, Software, Investigation, Validation, Writing -original draft and Writing - Review & editing. Lubov Kuchkarova: Supervision, Project administration, Methodology, Resources, Validation, and Writing - Rreview & editing. Dilnoza Romanova: Data curation and Investigation. Szczuko Małgorzata: Conceptualization, Resources, Software, Funding acquisition, and Writing - Review & editing. Teodoro Durá-Travé: Conceptualization, Resources, Software, Funding acquisition, and Writing - Rreview & editing. Nurali Ergashev: Data curation, Formal analysis, Validation, and Visualization. Iroda Karimova: Data curation and Investigation. Sevara Berdiyorova: Data curation, Formal analysis, Investigation, Funding acquisition, and Visualization. Visola Pulatova: Data curation and Investigation. Sokhibjon Abdusamatov: Investigation - performed microbiological experiments, including gut microbiota sampling, DNA extraction, and qPCR analysis. Rashidbek Achilov: Data curation, Formal analysis, Investigation, Funding acquisition, and Visualization. Numonjon Sultanov: Investigation - performed microbiological experiments, including gut microbiota sampling, DNA extraction, and qPCR analysis. Akmal Yusupov: Data curation and Investigation.
References
N Sawicka-Gutaj, D Gruszczyński, N Zawalna, K Nijakowski, I Muller, T Karpiński, M Salvi and M Ruchała. Microbiota alterations in patients with autoimmune thyroid diseases: A systematic review. International Journal of Molecular Sciences 2022; 23(21), 13450.
J Liu, X Qin, B Lin. J Cui, J Liao, F Zhang and Q Lin. Analysis of gut microbiota diversity in Hashimoto’s thyroiditis patients. BMC Microbiology 2022; 22(1), 318.
S Yu, B Yu, L Cong, Z Xiaodan and Z Le. The impact of gut microbiota on autoimmune thyroiditis and relationship with pregnancy outcomes: A review. Frontiers in Cellular and Infection Microbiology 2024; 14, 1361660.
K Yan, X Sun, C Fan, X Wang and H Yu. Unveiling the role of gut microbiota and metabolites in autoimmune thyroid diseases: Emerging prspectives. International Journal of Molecular Sciences 2024; 25(20), 10918.
I Stramazzo, S Capriello, S Filardo, M Centanni and C Virili. Microbiota and thyroid disease: An updated systematic review. Advances in Microbiology, Infectious Diseases and Public Health 2023; 17, 125-144.
J Knezevic, C Starchl, AT Berisha and K Amrein. Thyroid-gut-axis: How does the microbiota influence thyroid function? Nutrients 2020; 12(6), 1769.
AA Abed, A Naweedullah, S Uzma, S Ransherjit, RS Farhad, FM Jawad, FNU Raheela, W Radeyah, GS Mujahid, A Kiran, A Moiz, TD Boakye, S Haider, D Ayusha, HM Sheheryar and TA Subhan. Exploring the role of gut microbiota in autoimmune thyroid disorders: A systematic review and meta-analysis. Frontiers in Endocrinology 2023; 14, 1238146.
X Zhu, C Zhang, S Feng, R He and S Zhang. Intestinal microbiota regulates the gut-thyroid axis: The new dawn of improving Hashimoto thyroiditis. Clinical and Experimental Medicine 2024; 24(1), 39.
OD Frenț, L Stefan, CM Morgovan, N Duteanu, IL Dejeu, E Marian, L Vicaș and F Manole. A systematic review: Quercetin-secondary metabolite of the flavonol class, with multiple health benefits and low bioavailability. International Journal of Molecular Sciences 2024; 25(22), 12091.
P Shen, W Lin, X Deng, X Ba, L Han, Z Chen, K Qin, Y Huang and S Tu. Potential implications of quercetin in autoimmune diseases. Frontiers in Immunology 2021; 12, 689044.
C Giuliani, GD Dalmazi, I Bucci and G Napolitano. Quercetin and thyroid. Antioxidants 2024; 13(10), 1202.
LS Kuchkarova, KY Kayumov, NA Ergashev and GT Kudeshovа. Effect of quercetin on the intestinal carbohydrases activity in the offspring of the lead intoxicated mother. Journal of Natural Remedies 2024; 24(2), 391-396.
YM Kong. Experimental autoimmune thyroiditis in the mouse. Current Protocols in Immunology 2007; 78(1), 15-17.
K Kayumov, LS Kuchkarova, T Durá-Travé, S Petrenko, N Ergashev, J Abdurakhmonov, R Achilov, S Khan, V Pulatova and M Ernazarov. Immunomodulatory and histoprotective effects of luteolin and related flavonoids in autoimmune thyroiditis: Evidence from a rat model. Trends in Sciences 2025; 22(12), 10981.
TE Chang, JC Luo, UC Yang, YH Huang, MC Hou and FY Lee. Fecal microbiota profile in patients with inflammatory bowel disease in Taiwan. Journal of the Chinese Medical Association 2021; 84(6), 580-587.
L Palkova, A Tomova, G Repiska, K Babinska, B Bokor, I Mikula and K Soltys. Evaluation of 16S rRNA primer sets for characterisation of microbiota in paediatric patients with autism spectrum disorder. Scientific Reports 2021; 11(1), 6781
RH Dahal, S Kim, YK Kim, ES Kim and J Kim. Insight into gut dysbiosis of patients with inflammatory bowel disease and ischemic colitis. Frontiers in Microbiology 2023; 14(1), 1174832.
N Xia, G Chen, M Liu, X Ye, Y Pan, J Ge, Y Mao, H Wang, J Wang and S Xie. Anti-inflammatory effects of luteolin on experimental autoimmune thyroiditis in mice. Experimental and Therapeutic Medicine 2016; 12(6), 4049-4054.
RM Ruggeri, A CampennÌ, G Giuffrida, M Casciaro, MC Barbalace, S Hrelia, F Trimarchi, ÒS Cannav and S Gangemi. Oxidative stress as a key feature of autoimmune thyroiditis: An update. Minerva Endocrinologica 2020; 45(4), 326-344.
A Hosseinzade, O Sadeghi, A Naghdipour Biregani, S Soukhtehzari, GS Brandt and A Esmaillzadeh. Immunomodulatory effects of flavonoids: Possible induction of T CD4+ regulatory cells through suppression of mTOR pathway signaling activity. Frontiers in Immunology 2019; 10, 51.